1. Introduction
Butyrylcholinesterase (EC 3.1.1.8) is a hydrolase enzyme catalyzing the reaction of butyrylthiocholine hydrolysis to release thiocholine and butyric acid. Butyrylcholinesterase (BChE), as well as acetylcholinesterase (AChE), has been commonly used to monitor inhibitors of cholinesterases in the air, water, and biological fluids [
1,
2]. These enzymes catalyze similar reactions, differing only in substrate, but, in contrast to BChE, AChE is inhibited by excess of substrate [
1].
Numerous studies show high sensitivity of BChE to organophosphorus and carbamate pesticides [
3,
4,
5]. The specific effect of these pesticides on BChE activity provided the basis for developing very effective biosensors to determine them in environmental samples or food [
2,
3]. Moreover, this enzyme can be used to detect nerve agents, and thus security agencies and the military take a close interest in it [
6,
7,
8,
9,
10]. The sensitivity of BChE for detecting organophosphorus pesticides such as paraoxon and chlorophos is 6 and 10 times, respectively, higher than the sensitivity of AChE [
3].
Analytes in environmental samples and biological fluids are usually determined using electrochemical (amperometric and potentiometric) and optical BChE-based biosensors [
2,
11,
12,
13]. There are studies reporting the use of fluorescence methods involving enzyme conjugates [
14].
Despite the high sensitivity of enzyme-based analytical methods, maintaining the high activity and stability of the enzymes during storage and use, including their use as the recognition element in biosensors, still remains a challenge. Enzyme preparations can be stabilized by adding polyelectrolytes, amino acids, sugars, or nanoparticles [
14,
15,
16,
17]. To enhance the stability of enzymes further, the resulting solutions are dehydrated by evaporation or freeze drying on supports. In a study by Gibson et al., to increase the stability of glucose oxidase preparations, they were first supplemented with sucrose and then dehydrated [
18]. The resulting enzyme preparations retained their catalytic activity for one month when stored at a temperature of 37 °C.
Development of BChE preparations stable in storage and use is a problem that has been addressed in numerous studies [
10,
19,
20,
21]. A study by Lonshakova-Mukina et al. [
22] reported that starch and gelatin gels produced a substantial stabilizing effect on BChE activity during long-term storage. Immobilization of the enzyme preparations into those polymers increased their storage time at a temperature of 4 °C to more than two years. At the same time, there are very few available data on thermodynamic properties of the BChE immobilized in natural polymer carriers.
Most researchers analyze kinetics of thermal inactivation of proteins in aqueous solutions [
15,
23,
24,
25,
26] rather than in viscous media created by high-molecular-weight materials such as starch and gelatin. Gelatin and starch gels were found to enhance temperature and pH stability of proteins such as enzymes of luminous bacteria [
27,
28,
29]. In the presence of gelatin gel, the activity of those enzymes at different temperatures increased by a factor of two, and their optimum pH range became wider. Hence, high-molecular-weight polymers can effectively stabilize protein molecules. Previous studies of BChE stability were mainly conducted with low-molecular-weight substances such as glycerol or sucrose [
16]. The novelty of the present research is the use of starch and gelatin gels to study thermal inactivation of BChE. We found that the thermal stability of BChE was enhanced in the presence of these natural polymers.
2. Results
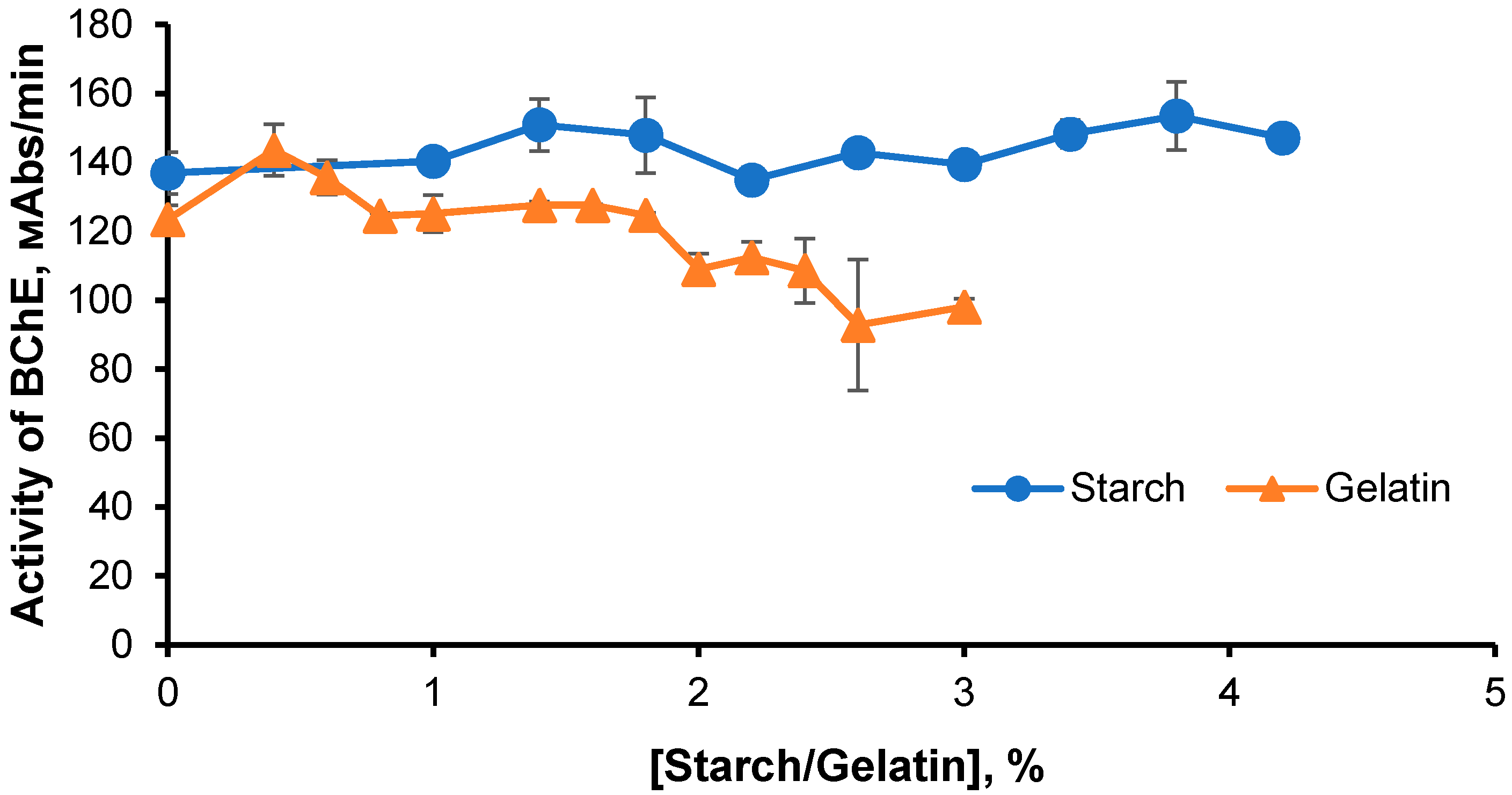
The effects of starch and gelatin on BChE activity were determined by studying the rate of enzymatic hydrolysis as dependent on concentrations of stabilizers in the solution at a temperature of 25 °C (
Figure 1). Starch concentrations were varied between 1% and 4.2% and gelatin concentrations between 0.4% and 3%.
The highest BChE activity was observed at a gelatin concentration of 0.5% (
Figure 1), but this concentration was not high enough for gel formation (at this concentration, gelatin solutions were sols). At gelatin concentrations between 1% and 2%, BChE activity did not change, and the addition of the enzyme to solutions with higher gelatin concentrations decreased its activity. At starch concentrations above 1%, the activity of the enzyme was higher than in the gelatin medium.
Mechanisms of thermal inactivation of BChE were investigated in the presence of 1.4% gelatin and 3% starch gels. We chose these concentrations because they are suitable for preparation of the immobilizing reagents for analyzing anticholinesterase agents [
29]. At lower concentrations, a gel network is not formed, and immobilizing reagents based on higher concentrations are inconvenient to use because of undesirable kinetic characteristics (the time to reach the maximum of BChE activity is too long). Moreover, it is difficult to measure out very viscous solutions when preparing doses of immobilized reagents intended for conducting one analysis.
To estimate the stabilizing effect of the starch and gelatin gels on BChE, the enzyme was exposed to temperatures between 25 and 65 °C for 2 to 30 min in the presence of the gels, and changes in the enzyme activity were analyzed. A significant decrease in BChE activity was observed at a temperature of 50 °C or higher. In the buffer solution, the residual BChE activity remained unchanged even after a 30 min exposure to 40 °C, but activity dropped to a near-zero level after less than 5 min exposure to 65 °C.
Graphs showing the dependence of the natural logarithm of BChE residual activity on the time of exposure to elevated temperatures are not linear. Hence, BChE thermal inactivation kinetics does not correspond to simple first-order kinetics. Overall kinetics at temperatures between 55 and 64 °C can be described using the two-exponential term model [
30]:
where
ai is the residual activity at time t,
A is an undefined constant term, and
k1 and
k2 are apparent first-order kinetic constants of inactivation.
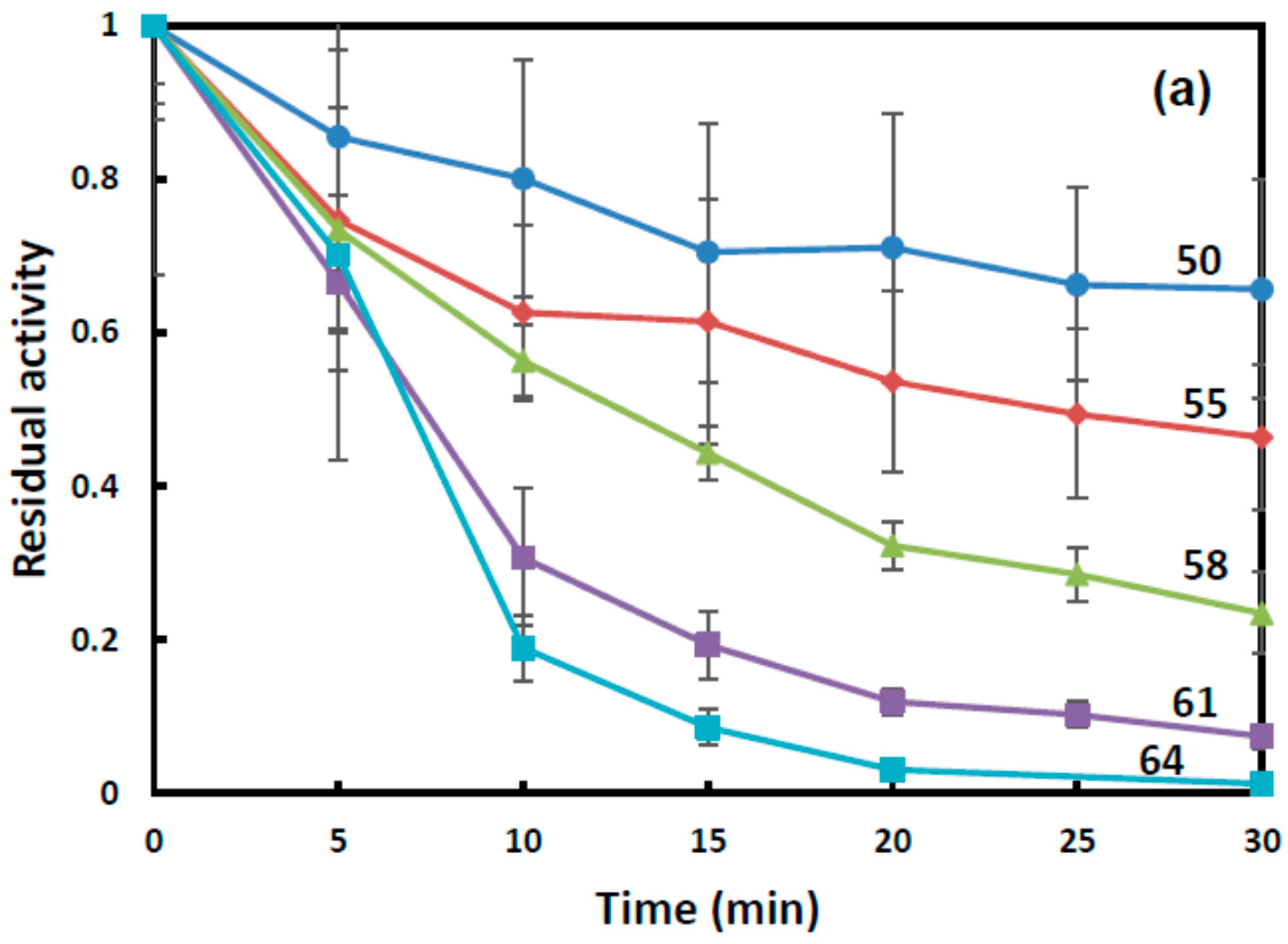
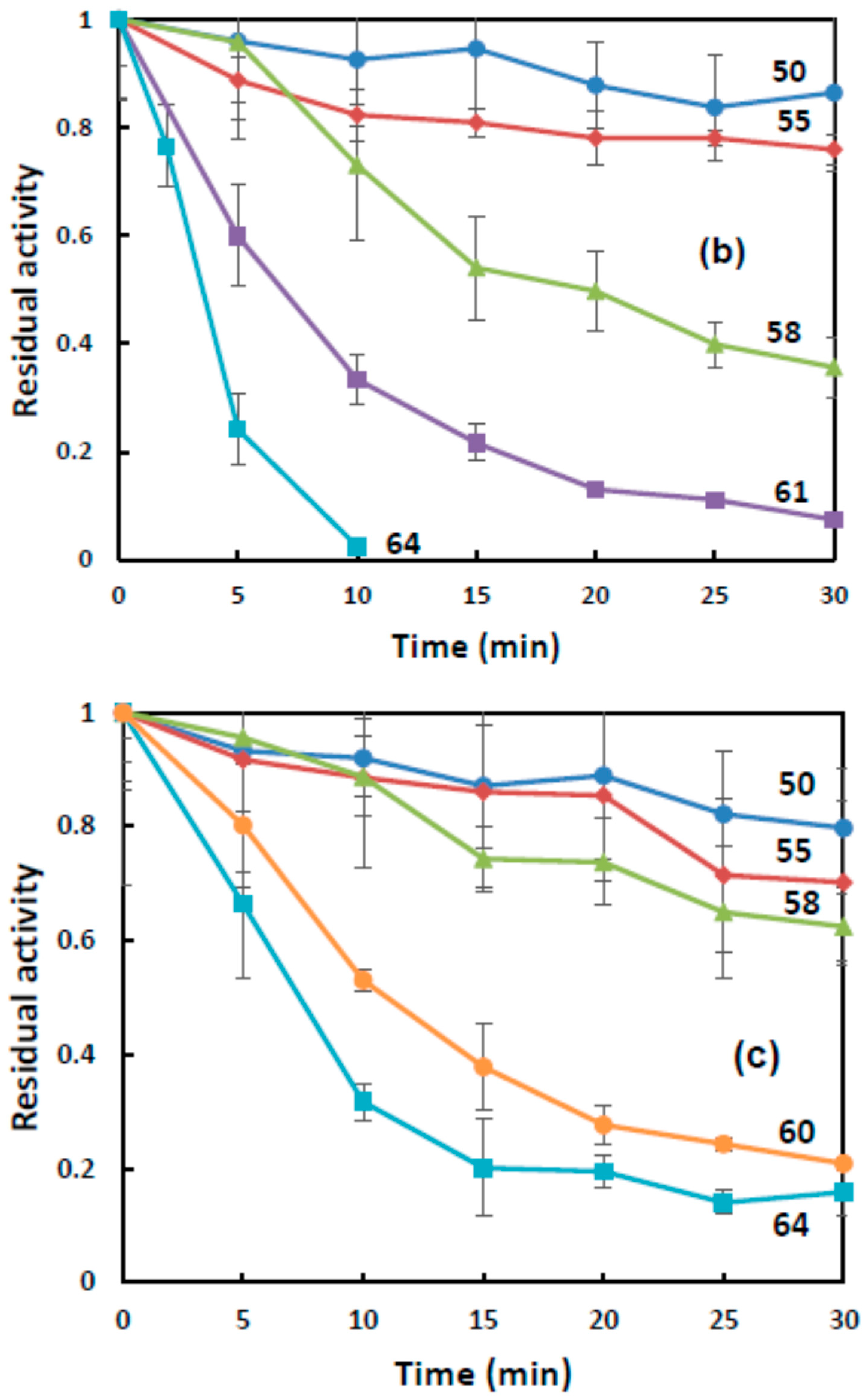
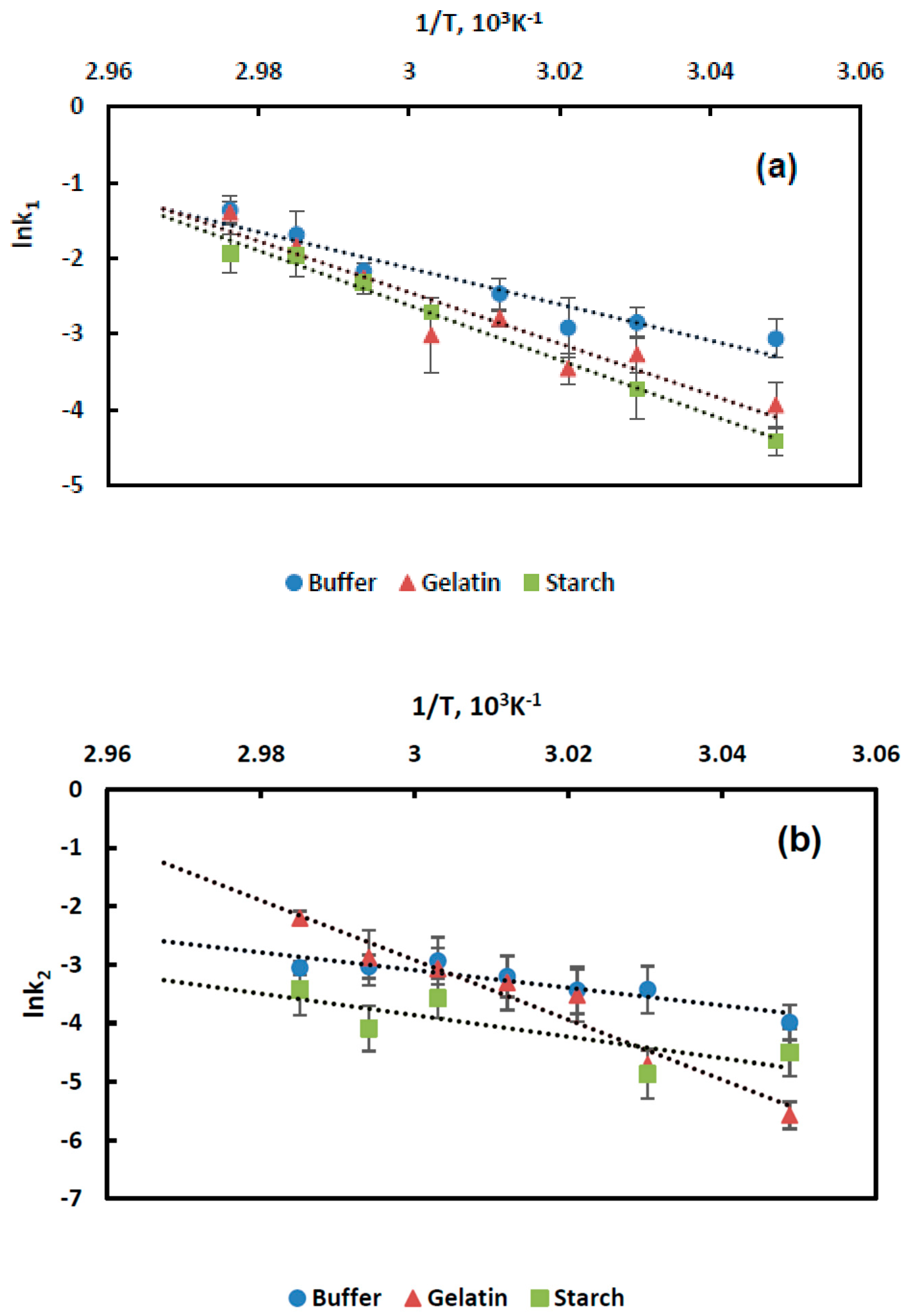
Figure 2 is time dependence of the residual BChE activity at temperatures between 50 and 64 °C. In the starch gel, the residual BChE activity decreased at a slower rate with an increase in the duration of exposure to high temperatures than in the gelatin gel and buffer. For instance, at a temperature of 64 °C, the residual BChE activity was retained for 30 min in the starch gel and for 10 min in the gelatin gel.
Then, semi-log plots were made to show kinetic dependences of BChE thermal inactivation.
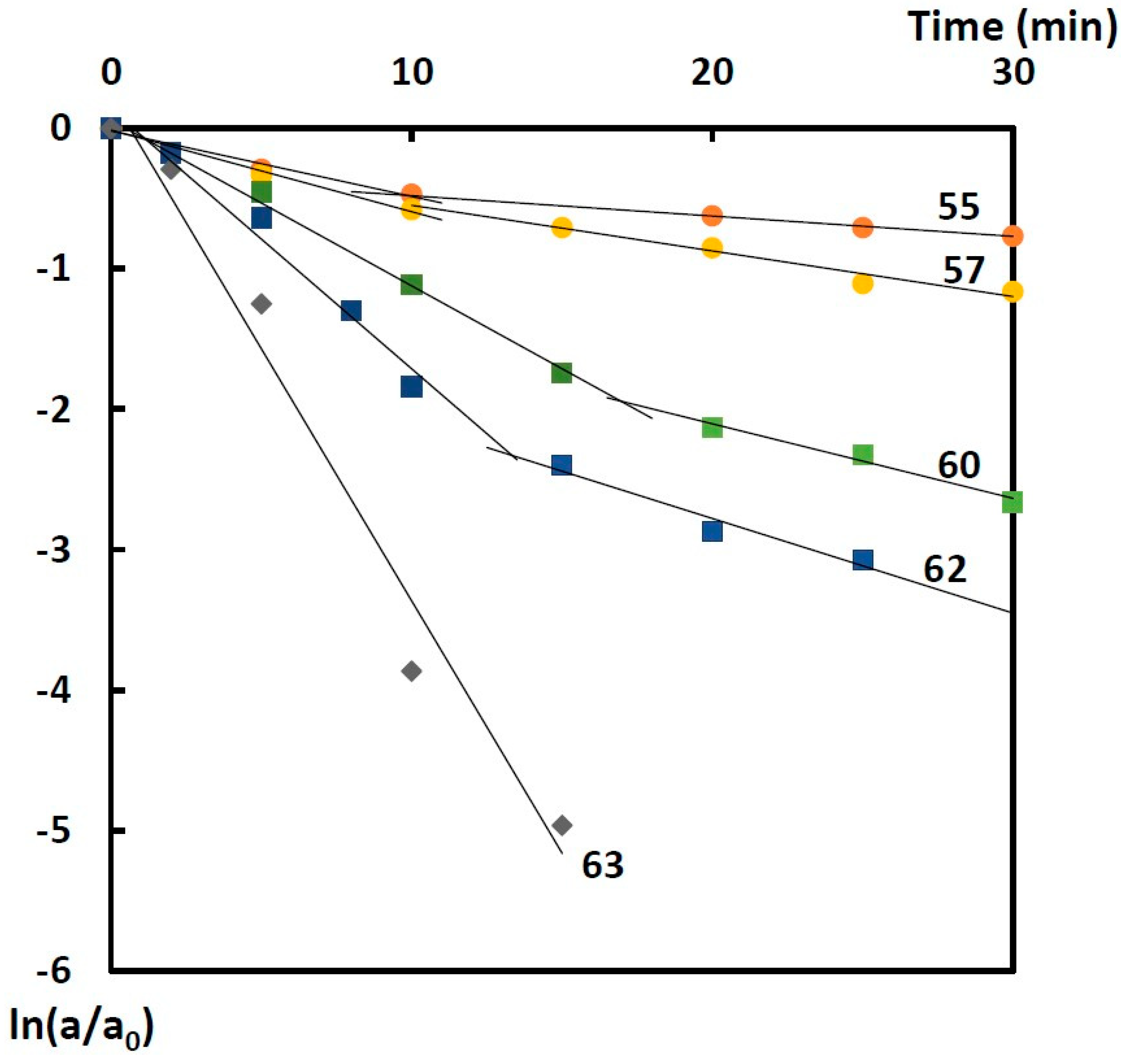
Figure 3 is an example of kinetic dependence of BChE thermal inactivation in the buffer solution. Similar dependences of BChE thermal inactivation were obtained in the presence of the starch and gelatin gels. All dependences in the graph of the first-order equation are represented by broken lines, which is characteristic of the dissociative thermal inactivation of oligomeric enzymes [
31]. The starting sections of the kinetic curves correspond to enzyme dissociation into subunits (fast inactivation) and the second linear sections to denaturation (slow inactivation). The exceptions were temperature dependences of BChE thermal inactivation in the buffer solution at 63 °C and in the gelatin gel at 64 °C, for which quasi-first-order irreversible inactivation was observed.
Constants of thermal inactivation rates,
k1 and
k2, for fast and slow inactivation were determined as the slope of the curve of BChE thermal inactivation in the absence and presence of stabilizing additives for each temperature dependence.
Figure 4a,b is temperature dependence of the constants of fast and slow inactivation rates represented as Arrhenius plot. The Arrhenius activation energy of temperature inactivation in the buffer solution was 47.3 kcal/mol and 29.9 kcal/mol for the fast and slow inactivation, respectively. In the presence of the starch gel, the Arrhenius activation energy was 71.7 kcal/mol and 36.6 kcal/mol, and in the presence of the gelatin gel, it was 67.2 kcal/mol and 101.3 kcal/mol for the fast and slow inactivation, respectively. The higher activation energies of the BChE temperature inactivation in the presence of the starch and gelatin gels suggested enhancement of enzyme stability by the starch and gelatin gels.
The classical thermodynamic Eyring equation was used to determine activation parameters Δ
H#, Δ
S#, and Δ
G#, which characterized thermal inactivation of BChE (
Table 1). In the gelatin solution, thermal inactivation enthalpy and entropy increased for both the first and the second phases of thermal inactivation. In the starch gel, thermal inactivation enthalpy and entropy increased for the fast phase but decreased for the slow phase of thermal inactivation.
The calculated values of the change in the free energy in both phases of BChE thermal inactivation varied between 20 and 23 kcal∙mol−1. These values are consistent with the values of this parameter obtained for most proteins.
3. Discussion
Addition of various stabilizing substances considerably enhances enzyme stability, including thermal stability. Stabilization of enzymes is mainly caused by a decrease in the molecular and submolecular motions or molecular vibrations [
19]. Low-molecular-weight stabilizers are known to increase BChE stability, e.g., activation parameters of temperature inactivation of the enzyme are decreased in the presence of deuterium oxide [
15]. At the same time, the oligomeric protein is still inactivated in a two-phase process.
In our study we used physical gel because of the formation of collagen type triple helices when cooling the solutions. Gelatin gel networks are formed at temperatures below 40 °C and at gelatin concentrations above the critical gelation concentration. Depending on the temperature, gelatin can have different dispersion structures: if the temperature is lower than that of gelation (T < Tg), gelatin is gel-like; if the temperature is higher than that of gelation (T > Tg), gelatin is a colloid. When the temperature is equal to that of gelation (T = Tg), gelatin is in a sol-to-gel transition state [
32,
33]. In our study, we dealt with low-concentration water/gelatin solutions, which are capable of gelation at temperatures below 27 °C. Thus, the 1.4% gelatin gel used in the present work had low thermal stability. Hence, at high temperatures, collagen type triple helices “melt”, and gelatin is converted from gel to sol. Nevertheless, the activation energies of thermal inactivation calculated in this study suggest the stabilizing effect of the gelatin medium on BChE. It seems likely that within the temperature range studied here, this stabilizer prevents unfolding processes, as the overall viscosity of the gelatin solution was certainly reduced with the temperature rise [
34].
The potato starch used in the present study has high solubility (82% at 95 °C) and contains an appreciable amount of covalently bonded phosphate monoester groups (0.06–0.10%) linked exclusively to the potato amylopectin molecule. The repulsion of like charges in aqueous solutions helps to untangle the individual polymer molecules; thus, the potato amylopectin increases the viscosity and the “thickening power” of potato starch pastes [
35]. Moreover, potato starch has a low critical concentration. Even 0.1 g of dry starch per 100 mL of water produces a paste at 95 °C, in which the swollen granules occupy virtually the entire volume. As the temperature range of gelation of potato starch is between 59 and 68 °C, we are certain that the gel structure of starch was not destroyed at the temperatures of thermal inactivation of BChE used in this study.
Thus, the increase in activation energy suggested an enhancement of thermal stability of the enzyme in the presence of the solutions of starch and gelatin. This effect is not obvious, as it is not observed for all enzymes. In [
27,
36], the authors showed that the viscous environments of gelatin and starch did not affect thermal inactivation of the enzymes of luminous bacteria.
The present study showed that the use of natural polymers enhanced thermal stability of BChE. The general reaction path can be schematized as follows: E4 ↔ E1 → Ed, where E4 is the tetrameric enzyme in the native state, E1—monomers, and Ed—irreversibly denatured monomers. A similar mechanism of thermal inactivation is observed for both oligomeric cholinesterases [
15] and other oligomeric enzymes [
37]. Nonlinear temperature dependence can be attributed to the occurrence of at least two successive processes: reversible dissociation of the oligomeric enzyme into monomers and subsequent irreversible denaturation of these monomers. The higher values of residual activity after long-term exposure of BChE to elevated temperatures in the presence of the starch gel compared with the corresponding parameters in the buffer solution and gelatin gel are indicative of the stabilizing effect of the starch gel on the enzyme. A possible reason for this is that starch retains its gel structure at elevated temperatures, whereas the temperature of gelatin gel formation is considerably below 50 °C [
38]. Hence, within the temperature range studied here, this stabilizer prevents unfolding processes as the overall viscosity of the solution increases. These data are consistent with the activation energies of thermal inactivation calculated in this study. The increase in activation energy suggested an enhancement of thermal stability of the enzyme in the presence of the viscous solutions of starch and gelatin.
Protein denaturation is accompanied by a significant increase in entropy and enthalpy, resulting in changes in activation entropy ΔS# and activation enthalpy ΔH#. Results of this study demonstrate that in the presence of the gelatin gel, the activation entropy of the dissociation process is lower than the activation entropy of the thermal inactivation process. This fact suggests that the activated complex of the tetrameric enzyme is structured to a higher degree than the activated complex of the enzyme in the second phase of thermal inactivation. An opposite effect is observed in the starch gel. In this medium, entropy of activation of the slow process is negative. In the starch medium, unfolding processes may be suppressed because the gel structure of starch is retained at elevated temperatures. The positive value of the change of free energy for both processes suggests that thermal inactivation in both the buffer solution and the gels is not a spontaneous process.
In a previous study, starch and gelatin gels were used as stabilizing additives to produce stable enzyme preparations [
22]. In that study, however, BChE preparations based on starch or gelatin gel were solidified; they were first measured out and then dried at 8 °C. Dehydration appears to enhance enzyme stability further, for long-term storage [
21].
4. Materials and Methods
Freeze-dried BChE from equine serum 900 U/mg (Sigma, USA) was used in the study. Other reagents used were butyrylthiocholine iodide (≥99.0%) (Fluka, Sigma-Aldrich, Switzerland), 5-5′-dithiobis (2-nitrobenzoic acid) (≥98%) and starch from potato (Sigma-Aldrich, Germany), gelatin from porcine skin with medium gel strength (Fluka, Sigma, Germany). Solutions of butyrylcholinesterase, starch, and gelatin were prepared using a 0.05 M potassium-phosphate buffer, pH 8, at 25 °C.
Gelatin gel was prepared as follows. Buffer solution was added to gelatin powder, and it was left to swell for 30 min. Then, it was heated to a temperature of 80 °C while being stirred thoroughly. To prepare starch gel, the necessary amount of dry starch was added to the buffer solution; it was brought to a boil and then cooled to a temperature of 25 °C.
BChE activity was determined in the presence of different gelatin (0.4% to 3%) and starch (1% to 4.2%) concentrations and with no stabilizers (control) at a temperature of 25 °C. Thermal inactivation of BChE was studied by incubating the enzymes for 2–30 min at temperatures ranging from 50 to 64 °C in a 1.4% gelatin solution, 3% starch solution, and with no stabilizers (control). A portion of 0.05 M phosphate buffer pH 8 was used as a control solution.
BChE activity was analyzed using Ellman’s method [
39]. To perform measurements, 1.8 mL of starch/gelatin suspension or buffer solution with 0.11 U BChE, 100 µL 0.02 mM 5-5′-dithiobis (2-nitrobenzoic acid), 100 µL 2 mM butyrylthiocholine iodide were sequentially added to the spectrophotometer cuvette. BChE activity was determined by measuring the absorbance of the reaction solution at a 412 nm absorption wavelength. BChE residual activity,
a/
a0, was determined after exposure of the enzyme to various temperatures;
a0 is the starting activity of BChE, and
a is the activity of BChE after heating time. Measurements were done using a Shimadzu UV-2700 spectrophotometer (Japan). Each data point was determined in at least 3–5 parallel measurements.
Activation energies
Ea were determined using the Arrhenius equation:
where
Ai is the Arrhenius pre-exponential factor,
Ea is the Arrhenius activation energies,
R is the universal gas constant,
T is temperature in Kelvin.
The activation enthalpy Δ
H#, the activation entropy Δ
S#, and the free energy of inactivation Δ
G were calculated according to the Eyring equations:
where
kB is Boltzmann’s constant,
h is Plank’s constant.
The activation entropy ΔS# was obtained by plotting T × ln(k/T) versus T and from the temperature dependence of ΔG (3).
5. Conclusions
Using starch and gelatin gels can maintain catalytic activity of BChE exposed to elevated temperatures over long periods. Thus, there is positive evidence in favor of using these polymer gels to produce enzyme preparations that are stable in storage and use. Starch gel is, however, the better choice, as the use of this stabilizer not only increases the Arrhenius energy but also decreases entropy of activation of BChE thermal inactivation, which indicates that the rate of this process slows down.
6. Patents
Patent RF 2546245. Enzyme preparation based on immobilized butyrylcholinesterase and method of its preparation; Esimbekova E.N., Lonshakova-Mukina V.I., Kratasyuk V.A.
Patent RF 2704264. Express method for determination of butyrylcholinesterase inhibitors in water and aqueous extracts; Esimbekova E.N., Lonshakova-Mukina V.I., Kratasyuk V.A.
Author Contributions
Conceptualization, E.N.E. and V.A.K.; methodology, E.N.E.; validation, E.N.E. and V.I.L.-M.; investigation, V.I.L.-M.; resources, V.A.K.; data curation, V.I.L.-M.; writing—original draft preparation, V.I.L.-M.; writing—review and editing, E.N.E.; supervision, E.N.E.; project administration, V.A.K.; funding acquisition, E.N.E. All authors have read and agreed to the published version of the manuscript.
Funding
The research was funded by the Government of Krasnoyarsk Territory, Krasnoyarsk Regional Fund of Science, and the Russian Foundation for Basic Research [project No 20-44-242001].
Conflicts of Interest
The authors declare no conflict of interest. The funders had no role in the design of the study; in the collection, analyses, or interpretation of data; in the writing of the manuscript; or in the decision to publish the results.
References
- Andreescu, S.; Marty, J.-L. Twenty years research in cholinesterase biosensors: From basic research to practical applications. Biomol. Eng. 2006, 23, 1–15. [Google Scholar] [CrossRef] [PubMed]
- Van Dyk, J.S.; Pletschke, B. Review on the use of enzymes for the detection of organochlorine, organophosphate and carbamate pesticides in the environment. Chemosphere 2011, 82, 291–307. [Google Scholar] [CrossRef] [PubMed]
- Arduini, F.; Ricci, F.; Tuta, C.S.; Moscone, D.; Amine, A.; Palleschi, G. Detection of carbamic and organophosphorous pesticides in water samples using a cholinesterase biosensor based on Prussian Blue-modified screen printed electrode. Anal. Chim. Acta 2006, 580, 155–162. [Google Scholar] [CrossRef] [PubMed]
- Kumaran, S.; Morita, M. Application of a cholinesterase biosensor to screen for organophosphorus pesticides extracted from soil. Talanta 1995, 42, 649–655. [Google Scholar] [CrossRef]
- Gogol, E.V.; Evtugyn, G.A.; Marty, J.-L.; Budnikov, H.C.; Winter, V.G. Amperometric biosensors based on nafion coated screen-printed electrodesfor the determination of cholinesterase inhibitors. Talanta 2000, 53, 379–389. [Google Scholar] [CrossRef]
- Amine, A.; Arduini, F.; Moscone, D.; Palleschi, G. Recent advances in biosensors based on enzyme inhibition. Biosens. Bioelectron. 2016, 76, 180–194. [Google Scholar] [CrossRef] [PubMed]
- Benschop, H.P.; De Jong, L.P.A. Nerve agent stereoisomers: Analysis, isolation and toxicology. Acc. Chem. Res. 1988, 21, 368–374. [Google Scholar] [CrossRef]
- Arduini, F.; Amine, A.; Moscone, D.; Palleschi, G. Biosensors based on cholinesterase inhibition for insecticides, nerve agents and aflatoxin B1 detection (review). Microchim. Acta 2010, 170, 193–214. [Google Scholar] [CrossRef]
- Arduini, F.; Palleschi, G. Disposable Electrochemical Biosensor Based on Cholinesterase Inhibition with Improved Shelf-Life and Working Stability for Nerve Agent Detection; Nikolelis, D., Ed.; Springer: Dordrecht, The Netherlands, 2012; pp. 261–278. [Google Scholar] [CrossRef]
- Zeman, J.; Vetchý, D.; Pavloková, S.; Franc, A.; Pitschmann, V.; Dominik, M.; Urbanová, M.; Šeděnková, I. Tubes for detection of cholinesterase inhibitors—Unique effects of Neusilin on the stability of butyrylcholinesterase-impregnated carriers. Enzym. Microb. Technol. 2019, 128, 26–33. [Google Scholar] [CrossRef] [PubMed]
- Viveros, L.; Paliwal, S.; McCrae, D.; Wild, J.; Simonian, A. A fluorescence-based biosensor for the detection of organophosphate pesticides and chemical warfare agents. Sens. Actuators B 2006, 115, 150–157. [Google Scholar] [CrossRef]
- Liu, G.; Lin, Y. Biosensor based on self-assembling acetylcholinesterase on carbon nanotubes for flflow injection/amperometric detection of organophosphate pesticides and nerve agents. Anal. Chem. 2006, 78, 835–843. [Google Scholar] [CrossRef] [PubMed]
- Arduin, F.; Amine, A.; Moscone, D.; Ricci, F.; Palleschi, G. Fast, sensitive and cost-effective detection of nerve agents in the gas phase using a portable instrument and an electrochemical biosensor. Anal. Bioanal. Chem. 2007, 388, 1049–1057. [Google Scholar] [CrossRef] [PubMed]
- Zhang, W.; Asiri, A.M.; Liu, D.; Du, D.; Lin, Y. Nanomaterial-based biosensors for environmental and biological monitoring of organophosphorus pesticides and nerve agents. Trends Anal. Chem. 2014, 54, 1–10. [Google Scholar] [CrossRef]
- Masson, P.; Laurentie, M. Stability of butyrylcholinesterase: Thermal inactivation in water and deuterium oxide. Biochim. Biophys. Acta 1988, 957, 111–121. [Google Scholar] [CrossRef]
- Masson, P.; Lushchekina, S.; Schopfer, L.M.; Lockridge, O. Effects of viscosity and osmotic stress on the reaction of human butyrylcholinesterase with cresyl saligenin phosphate, a toxicant related to aerotoxic syndrome: Kinetic and molecular dynamics studies. Biochem. J. 2013, 454, 387–399. [Google Scholar] [CrossRef]
- Arduini, F.; Forchielli, M.; Amine, A.; Neagu, D.; Cacciotti, I.; Nanni, F.; Moscone, D.; Palleschi, G. Screen-printed biosensor modified with carbon black nanoparticles for the determination of paraoxon based on the inhibition of butyrylcholinesterase. Microchim. Acta 2015, 182, 643–651. [Google Scholar] [CrossRef]
- Gibson, T.D.; Pierce, B.L.J.; Hulbert, J.N.; Gillespie, S. Improvements in the stability characteristics of biosensors using protein-polyelectrolyte complexes. Sens. Actuators B Chem. 1996, 33, 13–18. [Google Scholar] [CrossRef]
- Bromberg, A.; Marix, S.; Frishman, G. Kinetic study of the thermal inactivation of cholinesterase enzymes immobilized in solid matrices. Biochim. Biophys. Acta 2008, 1784, 961–966. [Google Scholar] [CrossRef] [PubMed]
- Luckarifi, H.R.; Johnson, G.R.; Spain, J.C. Silica-immobilized enzyme reactor; application to cholinesterase-inhibition studies. J. Chromatogr. B 2006, 843, 310–316. [Google Scholar] [CrossRef] [PubMed]
- Esimbekova, E.N.; Govorun, A.E.; Lonshakova-Mukina, V.I.; Kratasyuk, V.A. Gelatin and Starch: What Better Stabilizes the Enzyme Activity? Dokl. Biol. Sci. 2020, 491, 43–46. [Google Scholar] [CrossRef] [PubMed]
- Lonshakova-Mukina, V.I.; Esimbekova, E.N.; Kratasyuk, V.A. Stabilization of Butyrylcholinesterase by the Entrapment into the Natural Polymer-Based Gels. Dokl. Biochem. Biophys. 2018, 479, 98–100. [Google Scholar] [CrossRef] [PubMed]
- Tanford, C. Protein denaturation. Adv. Protein Chem. 1968, 23, 121–282. [Google Scholar] [CrossRef] [PubMed]
- Privalov, P.L. Stability of proteins, small globular proteins. Adv. Protein Chem. 1979, 33, 167–241. [Google Scholar] [CrossRef] [PubMed]
- O’Fagian, C. Understanding and increasing protein stability. Biochim. Biophys. Acta 1995, 1252, 1–14. [Google Scholar] [CrossRef]
- O’Fagian, C. Enzyme stabilization—recent experimental progress. Enz. Microb. Technol. 2003, 33, 137–149. [Google Scholar] [CrossRef]
- Bezrukikh, A.; Esimbekova, E.; Nemtseva, E.; Kratasyuk, V.; Shimomura, O. Gelatin and starch as stabilizers for the coupled enzyme system of luminous bacteria NADH:FMN-oxidoreductase-luciferase. Anal. Bioanal. Chem. 2014, 406, 5743–5747. [Google Scholar] [CrossRef] [PubMed]
- Lonshakova-Mukina, V.; Esimbekova, E.; Kratasyuk, V. Impact of enzyme stabilizers on the characteristics of biomodules for bioluminescent biosensors. Sens. Actuator B Chem. 2015, 213, 244–247. [Google Scholar] [CrossRef]
- Esimbekova, E.N.; Lonshakova-Mukina, V.I.; Bezrukikh, A.E.; Kratasyuk, V.A. Design of Multicomponent Reagents for Enzymatic Assays. Dokl. Biochem. Biophys. 2015, 461, 102–105. [Google Scholar] [CrossRef] [PubMed]
- Henley, J.; Sadana, A. Deactivation theory. Biotechnol. Bioeng. 1986, 28, 1277–1285. [Google Scholar] [CrossRef] [PubMed]
- Poltorak, O.M.; Chukhray, E.S.; Torshin, I.Y. Dissociative thermal inactivation, stability, and activity of oligomeric enzymes. Biochem. Biokhimia 1998, 63, 303–311. [Google Scholar]
- de Carvalho, W.; Djabourov, M. Physical gelation under shear for gelatin gels. Rheol. Acta 1997, 36, 591–609. [Google Scholar] [CrossRef]
- Papon, P.; Leblond, J.; Meijer, P.H.E. Gelation and Transitions in Biopolymers. In The Physics of Phase Transitions. Advanced Texts in Physics; Springer: Berlin/Heidelberg, Germany, 2002. [Google Scholar]
- Gulnov, D.; Nemtseva, E.; Kratasyuk, V. Contrasting relationship between macroand microviscosity of the gelatin- and starch-based suspensions and gels. Polym. Bull. 2016, 73, 3421–3435. [Google Scholar] [CrossRef]
- Appelqvist, I.; Debet, M. Starch-biopolymer interactions—A review. Food Rev. Int. 1997, 13, 163–224. [Google Scholar] [CrossRef]
- Govorun, A.E.; Esimbekova, E.N.; Kratasyuk, V.A. NAD(P)H:FMN-oxidoreductase functioning under macromolecular crowding: In vitro modeling. Dokl. Biochem. Biophys. 2019, 486, 213–215. [Google Scholar] [CrossRef] [PubMed]
- Atyaksheva, L.F.; Pilipenko, O.S.; Chukhrai, E.S.; Poltorak, O.M. Similarity of and differences between the mechanisms of thermal inactivation of β-galactosidases of different origins. Russian J. Phys. Chem. A 2008, 82, 864–869. [Google Scholar] [CrossRef]
- Ross-Murphy, S.B. Structure and rheology of gelatin gels: Recent progress. Polymer 1992, 33, 2622–2627. [Google Scholar] [CrossRef]
- Ellman, G.L.; Courtne, K.D.; Andres, V.; Featherstone, R.M. A new and rapid colorimetric determination of acetylcholinesterase activity. Biochem. Pharmacol. 1961, 7, 88–95. [Google Scholar] [CrossRef]
| Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).