Catalytic Performance of a Recombinant Organophosphate-Hydrolyzing Phosphotriesterase from Brevundimonas diminuta in the Presence of Surfactants
Abstract
1. Introduction
2. Results and Discussion
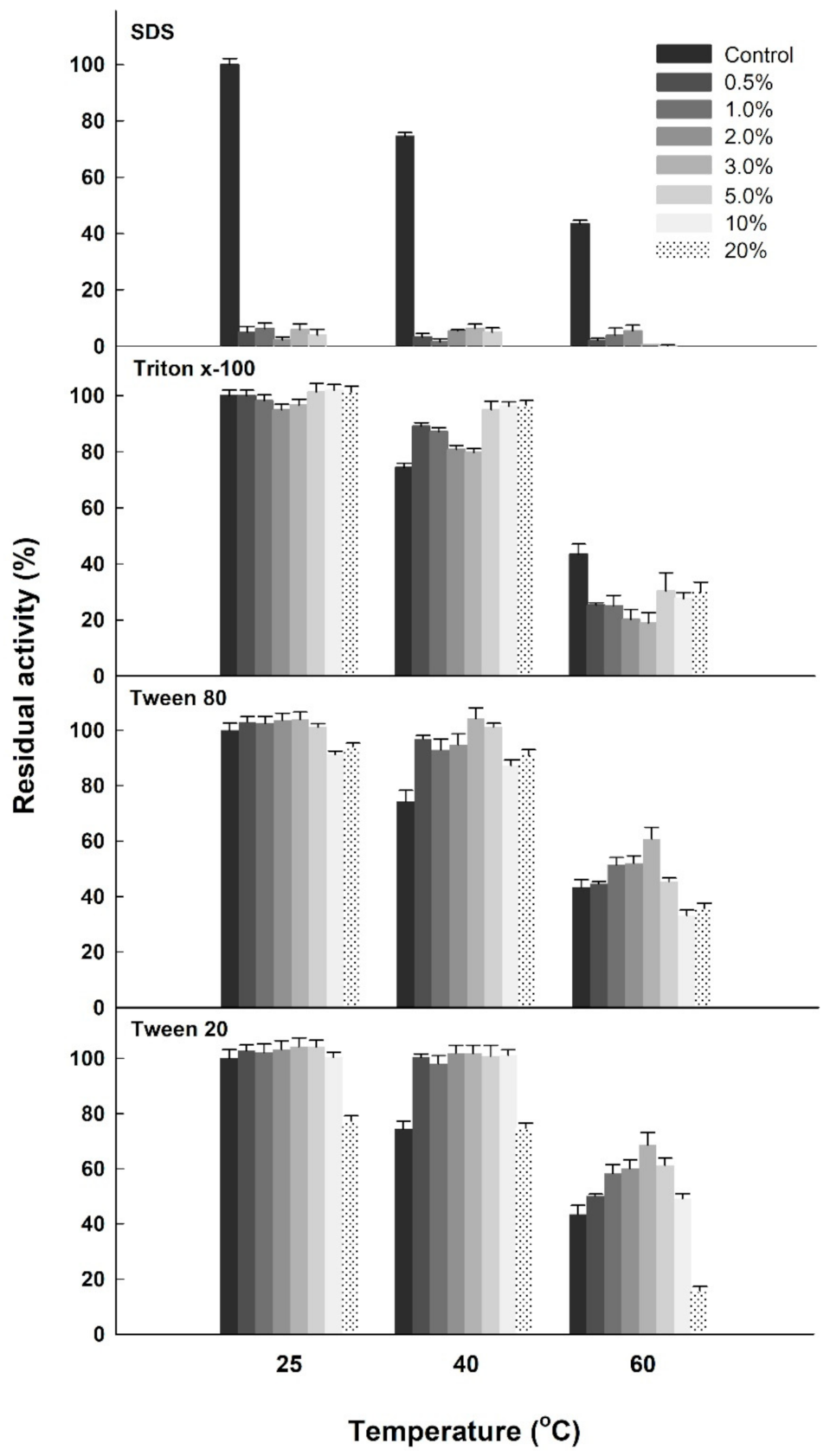
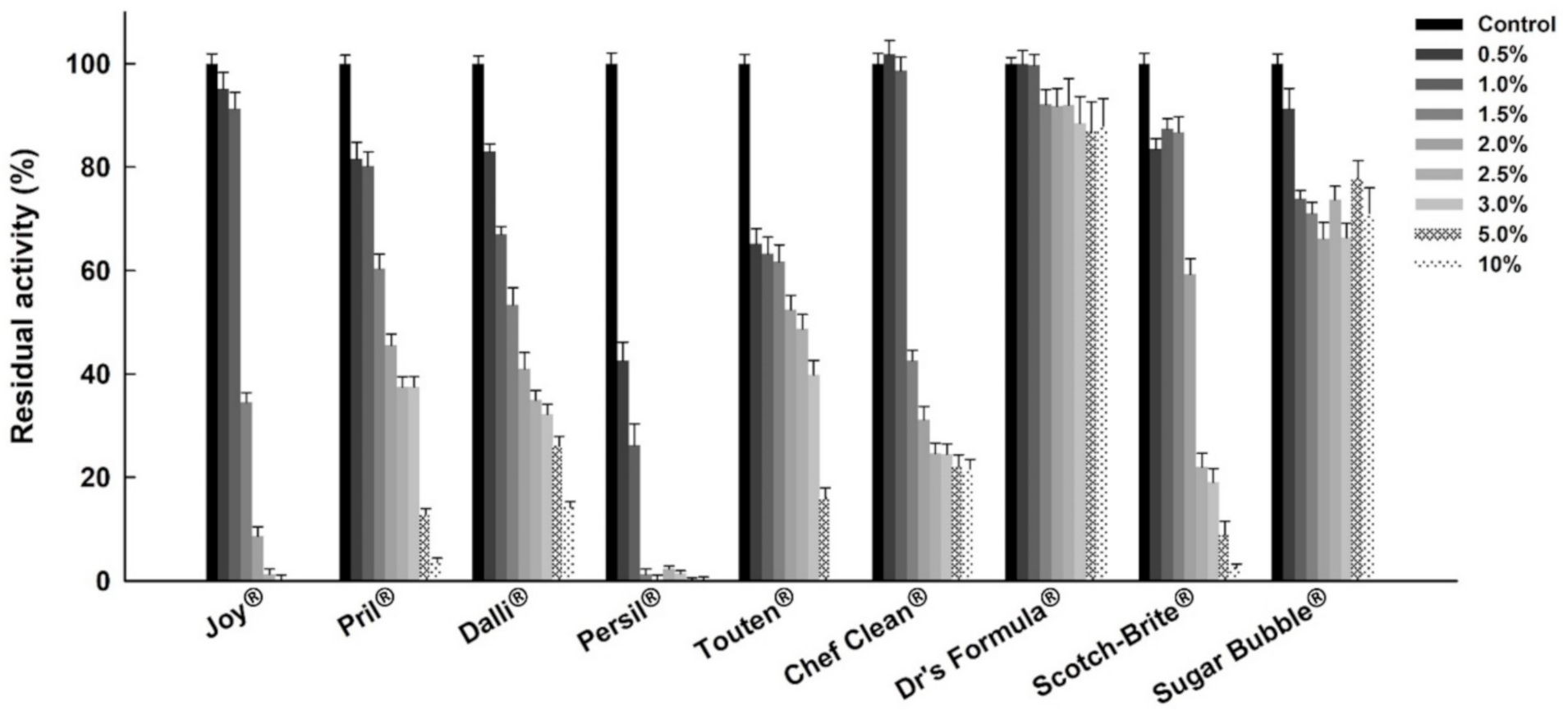
2.1. Impacts of Surfactants and Commercial Detergents on the Esterase Activity
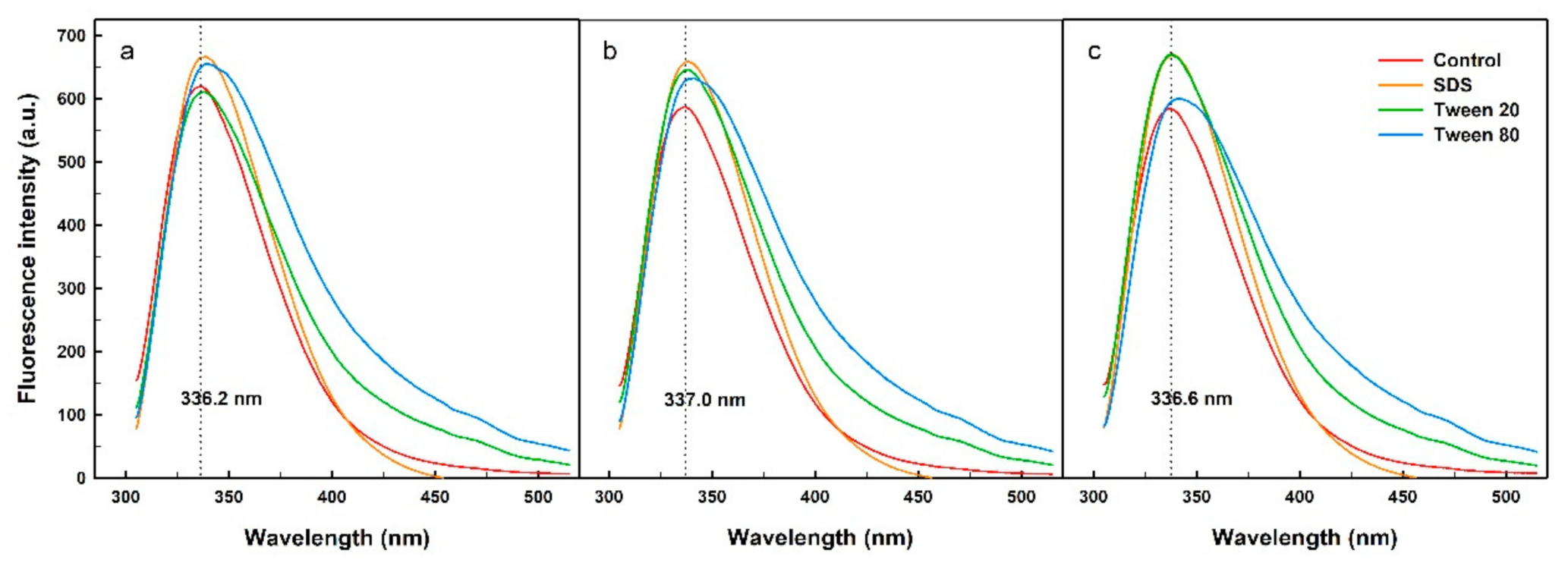
2.2. Fluorescence Spectra of the His-Tagged Enzyme in the Presence of Surfactants
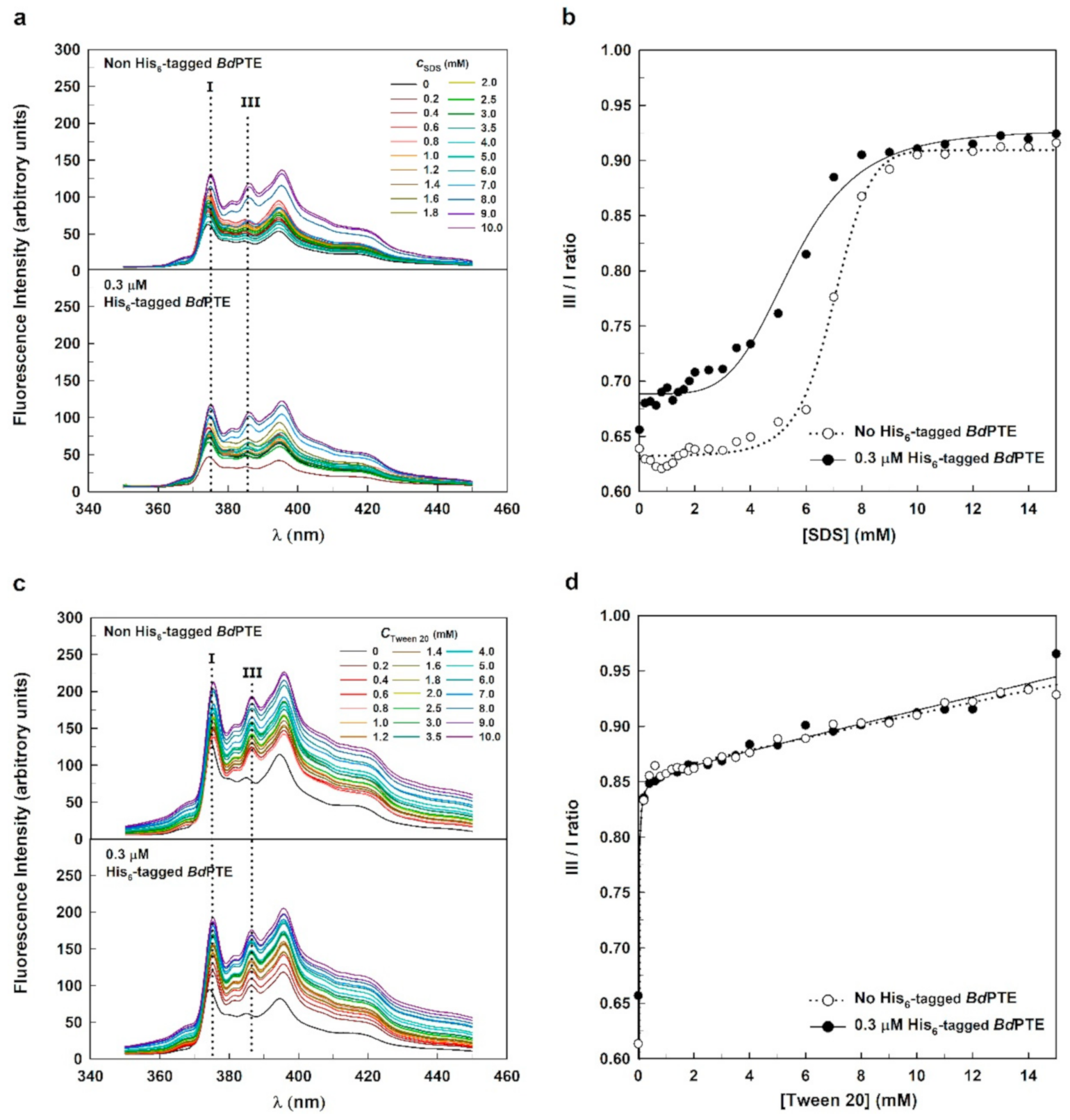
2.3. Pyrene Fluorescence Spectra of SDS and Tween 20 in the Absence and Presence of the His-Tagged Enzyme
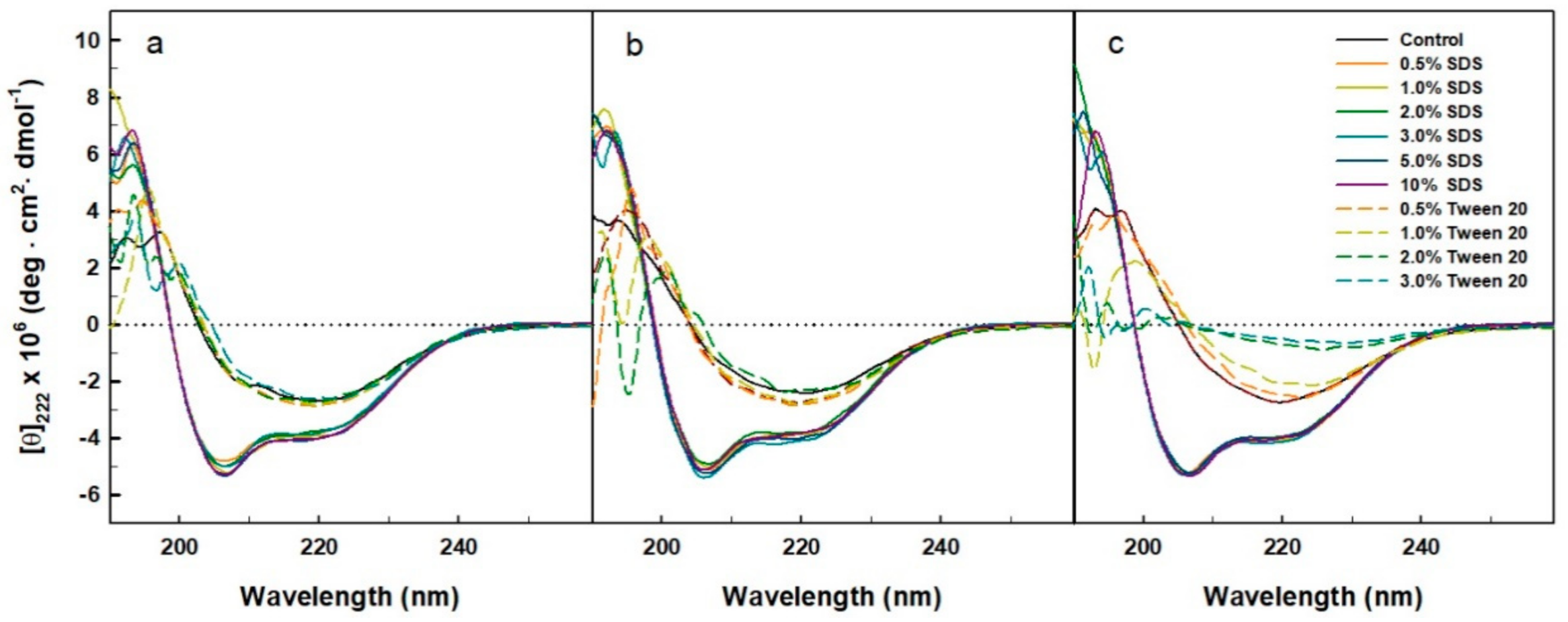
2.4. Far-UV CD Spectra of the His-Tagged Enzyme in the Presence of Surfactants
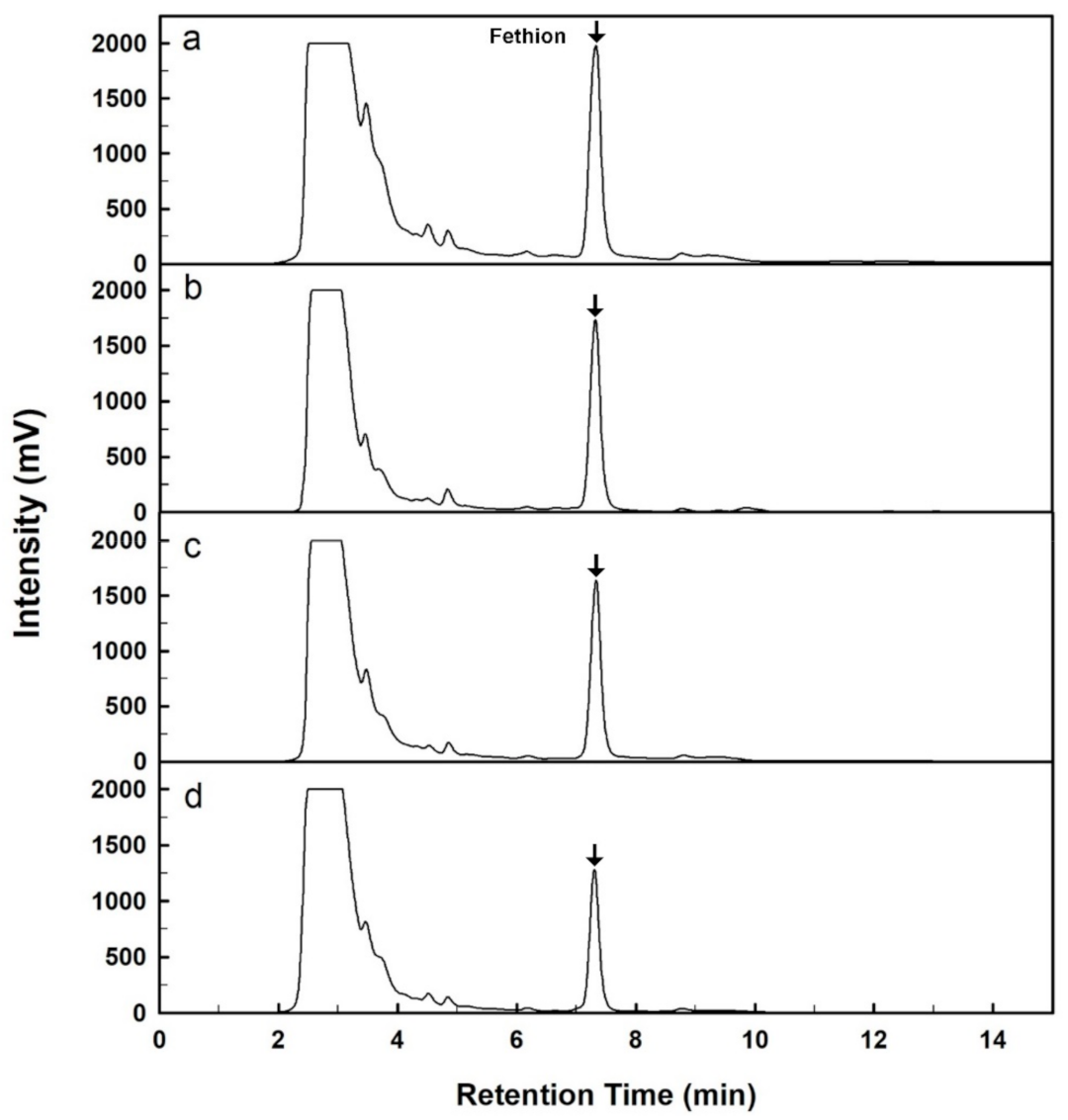
2.5. Removal of Fenthion from the Artificially Contaminated Sample
3. Materials and Methods
3.1. Chemicals and Reagents
3.2. Enzyme Preparation and Esterase Activity Assay
3.3. Effects of Surfactants and Commercial Detergents on the Esterase Activity
3.4. Fluorescence Studies
3.5. Circular Dichorism (CD) Studies
3.6. Enzyme Treatment of the Fenthion-Contaminated Chinese Cabbage Leaves
4. Conclusions
Author Contributions
Funding
Data Availability Statement
Conflicts of Interest
References
- Singh, R.S.; Singh, T.; Pandey, A. Microbial enzymes- an overview. In Biomass, Biofuels, Biochemicals; Singh, R.S., Singhania, R.R., Pandey, A., Larroche, E.T., Eds.; Elsevier: Amsterdam, The Netherlands, 2019; pp. 1–40. [Google Scholar]
- Kumar, A.; Singh, S. Directed evolution: Tailoring biocatalysts for industrial applications. Crit. Rev. Biotechnol. 2012, 33, 365–378. [Google Scholar] [CrossRef]
- Gürkök, S. Microbial enzymes in detergents: A review. Int. J. Sci. Eng. Res. 2019, 10, 75–81. [Google Scholar]
- Kumar, C.G.; Malik, R.K.; Tiwari, M.P. Novel enzyme-based detergents: An Indian perspective. Curr. Sci. 1998, 75, 1312–1318. [Google Scholar]
- Olsen, H.S.; Falholt, P. The role of enzymes in modern detergency. J. Surfactants Deterg. 1998, 1, 555–567. [Google Scholar] [CrossRef]
- Zhang, J.; Zhang, Y.; Li, W.; Li, X.; Lian, X. Optimizing Detergent Formulation with Enzymes. J. Surfactants Deterg. 2014, 17, 1059–1067. [Google Scholar] [CrossRef]
- Otzen, D. Protein–surfactant interactions: A tale of many states. Biochim. Biophys. Acta Proteins Proteom. 2011, 1814, 562–591. [Google Scholar] [CrossRef]
- Karmande, G.M.; Baah, J.; Cheng, K.-J.; McAllister, T.A.; Shelford, J.A. Effects of tTween 60 and tTween 80 on protease activity, thiol group reactivity, protein adsorption, and cellulose degradation by rumen microbial enzymes. J. Dairy Sci. 2000, 83, 536–542. [Google Scholar] [CrossRef]
- Shome, A.; Roy, S.; Das, P.K. Nonionic Surfactants: A Key to Enhance the Enzyme Activity at Cationic Reverse Micellar Interface. Langmuir 2007, 23, 4130–4136. [Google Scholar] [CrossRef]
- Antony, N.; Balachandran, S.; Mohanan, P.V. Effect of Surfactants on Catalytic Activity of Diastase α-amylase. J. Surfactants Deterg. 2013, 17, 703–708. [Google Scholar] [CrossRef]
- Kristensen, J.B.; Börjesson, J.; Bruun, M.H.; Tjerneld, F.; Jørgensen, H. Use of surface active additives in enzymatic hydrolysis of wheat straw lignocellulose. Enzym. Microb. Technol. 2007, 40, 888–895. [Google Scholar] [CrossRef]
- Duan, X.; Zou, C.; Wu, J. Triton X-100 enhances the solubility and secretion ration of aggregation-prone pullulanase produced in Escherichia coli. Bioresour. Technol. 2015, 194, 137–143. [Google Scholar] [CrossRef] [PubMed]
- Delorme, V.; Dhouib, R.; Canaan, S.; Fotiadu, F.; Carrière, F.; Cavalier, J.-F. Effects of Surfactants on Lipase Structure, Activity, and Inhibition. Pharm. Res. 2011, 28, 1831–1842. [Google Scholar] [CrossRef] [PubMed]
- Otzen, D.E. Amyloid formation in surfactants and alcohols: Membrane mimetics or structural switchers? Curr. Prot. Pept. Sci. 2010, 11, 355–371. [Google Scholar] [CrossRef] [PubMed]
- Otzen, D.E. Proteins in a brave new surfactant world. Curr. Opin. Colloid Interface Sci. 2015, 20, 161–169. [Google Scholar] [CrossRef]
- Otzen, D.E.; Christiansen, L.; Schulein, M. A comparative study of the unfolding of the endoglucanase Cel45 from Humicola insolens in denaturant and surfactant. Protein Sci. 1999, 8, 1878–1887. [Google Scholar] [CrossRef]
- Gitlin, I.; Gudiksen, K.L.; Whitesides, G.M. Peracetylated Bovine Carbonic Anhydrase (BCA-Ac18) Is Kinetically More Stable than Native BCA to Sodium Dodecyl Sulfate. J. Phys. Chem. B 2006, 110, 2372–2377. [Google Scholar] [CrossRef] [PubMed]
- Tanford, C. The hydrophobic effect and the organization of living matter. Science 1978, 200, 1012–1018. [Google Scholar] [CrossRef]
- Srivastava, R.; Alam, M.S. Effect of pH and surfactant on the protein: A perspective from theory and experiments. Int. J. Biol. Macromol. 2018, 107, 1519–1527. [Google Scholar] [CrossRef]
- Timasheff, S.N. Protein Hydration, Thermodynamic Binding, and Preferential Hydration. Biochemistry 2002, 41, 13473–13482. [Google Scholar] [CrossRef]
- Vlasova, I.; Zhuravleva, V.; Saletsky, A. Denaturation of bovine serum albumin initiated by sodium dodecyl sulfate as moni-tored via the intrinsic fluorescence of the protein. Russ. J. Phys. Chem. B 2014, 8, 385–390. [Google Scholar] [CrossRef]
- Saha, D.; Ray, D.; Kohlbrecher, J.; Aswal, V.K. Unfolding and Refolding of Protein by a Combination of Ionic and Nonionic Surfactants. ACS Omega 2018, 3, 8260–8270. [Google Scholar] [CrossRef] [PubMed]
- Kumari, S.; Halder, S.; Aggrawal, R.; Aswal, V.K.; Sundar, G.; Saha, S.K. Refolding of protein unfolded by gemini surfactants using β-cyclodextrin and sodium dodecyl sulfate in aqueous medium: Study on role of spacer chain of surfactants. J. Mol. Liq. 2020, 300, 112238. [Google Scholar] [CrossRef]
- Pedersen, J.N.; Lyngsø, J.; Zinn, T.; Otzen, D.E.; Pedersen, J.S. A complete picture of protein unfolding and refolding in sur-factants. Chem. Sci. 2020, 11, 699–712. [Google Scholar] [CrossRef]
- Das, S.; Singh, D.K. Purification and characterization of phosphotriesterases from Pseudomonas aeruginosa F10B and Calvibacter michiganese subsp. insidiosum SBL11. Can. J. Microbiol. 2006, 52, 157–168. [Google Scholar] [CrossRef]
- Hong, S.-B.; Raushel, F.M. Metal−Substrate Interactions Facilitate the Catalytic Activity of the Bacterial Phosphotriesterase. Biochemistry 1996, 35, 10904–10912. [Google Scholar] [CrossRef]
- Raushel, F.M.; Holden, H.M. Phosphotriesterase: An Enzyme in Search of Its Natural Substrate. Adv. Enzymol. Relat. Areas Mol. Biol. 2000, 74, 51–93. [Google Scholar] [CrossRef]
- Alejo-González, K.; Hanson-Viana, E.; Vazquez-Duhalt, R. Enzymatic detoxification of organophosphorus pesticides and related toxicants. J. Pestic. Sci. 2018, 43, 1–9. [Google Scholar] [CrossRef]
- Manco, G.; Porzio, E.; Zuzumoto, Y. Enzymatic detoxification: A sustainable means of degrading toxic organophosphate pes-ticides and chemical warfare nerve agents. J. Chem. Technol. Biotechnol. 2018, 93, 2064–2082. [Google Scholar] [CrossRef]
- Benning, M.M.; Kuo, J.M.; Raushel, F.M.; Holden, H.M. Three-Dimensional Structure of Phosphotriesterase: An Enzyme Capable of Detoxifying Organophosphate Nerve Agents. Biochemistry 1994, 33, 15001–15007. [Google Scholar] [CrossRef] [PubMed]
- Benning, M.M.; Kuo, J.M.; Raushel, F.M.; Holden, H.M. Three-dimensional structure of the binuclear metal center of phos-photriesterase. Biochemistry 1995, 34, 7973–7978. [Google Scholar] [CrossRef]
- Bigley, A.N.; Raushel, F.M. Catalytic mechanisms for phosphotriesterases. Biochim. Biophys. Acta Proteins Proteom. 2013, 1834, 443–453. [Google Scholar] [CrossRef] [PubMed]
- Chi, M.C.; Liao, T.Y.; Lin, M.G.; Lin, L.L.; Wang, T.F. Expression and physiochemical characterization of an N-terminal pol-yhistidine-tagged phosphotriesterase from the soil bacterium Brevundimonas diminuta. Biocatal. Agric. Biotechnol. 2020, 29, 101811. [Google Scholar] [CrossRef]
- Shi, Y.; Luo, W.; Tian, W.-X.; Zhang, T.; Zhou, H.-M. Inactivation and conformational changes of fatty acid synthase from chicken liver during unfolding by sodium dodecyl sulfate. Int. J. Biochem. Cell Biol. 1998, 30, 1319–1330. [Google Scholar] [CrossRef]
- Wu, X.-Q.; Xu, H.; Yue, H.; Liu, K.-Q.; Wang, X.-Y. Inhibition Kinetics and the Aggregation of α-Glucosidase by Different Denaturants. Protein J. 2009, 28, 448–456. [Google Scholar] [CrossRef] [PubMed]
- He, B.; Zhang, T.; Zhou, H.M. Comparison of inactivation and conformational changes of aminoacylase during denaturation in lithium dodecylsulfate solution. Int. J. Biol. Macromol. 1997, 20, 53–62. [Google Scholar] [CrossRef]
- Zheng, Y.-B.; Meng, F.-G.; Chen, B.-Y.; Wang, X.-C. Inactivation and conformational changes of lactate dehydrogenase from porcine heart in sodium dodecyl sulfate solutions. Int. J. Biol. Macromol. 2002, 31, 97–102. [Google Scholar] [CrossRef]
- Mu, H.; Zhou, S.M.; Xia, Y.; Zou, H.; Meng, F.; Yan, Y.B. Inactivation and unfolding of the hyperthermophilic inorganic py-rophosphatase from Thermus thermophiles by sodium dodecyl sulfate. Int. J. Mol. Sci. 2009, 10, 2849–2859. [Google Scholar] [CrossRef]
- Hou, H.; He, H.; Wang, Y. Effects of SDS on the activity and conformation of protein tyrosine phosphatase from Thermus thermophiles HB27. Sci. Rep. 2020, 10, 3195. [Google Scholar] [CrossRef]
- Merone, L.; Mandrich, L.; Rossi, M.; Manco, G. A thermostable phosphotriesterase from the archaeon Sulfolobus solfataricus: Cloning, overexpression and properties. Extremophiles 2005, 9, 297–305. [Google Scholar] [CrossRef]
- Hiblot, J.; Gotthard, G.; Chabriere, E.; Elias, M. Characterisation of the organophosphate hydrolase catalytic activity of SsoPox. Sci. Rep. 2012, 2, 779. [Google Scholar] [CrossRef]
- del Giudice, I.; Coppolecchia, R.; Merone, L.; Porzio, E.; Carusone, T.M.; Mandrich, L.; Worek, F.; Manco, G. An efficient thermostable organophosphate hydrolase and its application in pesticide decontamination. Biotechnol. Bioeng. 2015, 113, 724–734. [Google Scholar] [CrossRef] [PubMed]
- Bai, Y.-P.; Luo, X.-J.; Zhao, Y.-L.; Li, C.-X.; Xu, D.-S.; Xu, J.-H. Efficient Degradation of Malathion in the Presence of Detergents Using an Engineered Organophosphorus Hydrolase Highly Expressed by Pichia pastoris without Methanol Induction. J. Agric. Food Chem. 2017, 65, 9094–9100. [Google Scholar] [CrossRef] [PubMed]
- Sode, K.; Nakamura, H. Compatibility of phosphotriesterase from Flavobacterium sp. with detergents. Biotechnol. Lett. 1997, 19, 1239–1242. [Google Scholar] [CrossRef]
- Eriksson, T.; Börjesson, J.; Tjerneld, F. Mechanism of surfactant effect in enzymatic hydrolysis of lignocellulose. Enzym. Microb. Technol. 2002, 31, 353–364. [Google Scholar] [CrossRef]
- Goswami, D. Lipase Catalysis in Presence of Nonionic Surfactants. Appl. Biochem. Biotechnol. 2019, 191, 744–762. [Google Scholar] [CrossRef]
- Lou, H.; Zeng, M.; Hu, Q.; Cai, C.; Lin, X.; Qiu, X.; Yang, D.; Pang, Y. Nonionic surfactants enhanced enzymatic activity hydrolysis of cellulose by reducing cellulose deactivation caused by shear force and air-liquid interface. Bioresour. Technol. 2018, 249, 1–8. [Google Scholar] [CrossRef]
- Cheng, H.; Zhao, Y.-L.; Luo, X.-J.; Xu, D.-S.; Cao, X.; Xu, J.-H.; Dai, Q.; Zhang, X.-Y.; Ge, J.; Bai, Y.-P. Cross-linked enzyme-polymer conjugates with excellent stability and detergent-enhanced activity for efficient organophosphate degradation. Bioresour. Bioprocess. 2018, 5, 49. [Google Scholar] [CrossRef]
- Helle, S.S.; Duff, S.J.B.; Cooper, D.G. Effect of surfactants on cellulose hydrolysis. Biotechnol. Bioeng. 1993, 42, 611–617. [Google Scholar] [CrossRef]
- Seo, D.J.; Fujita, H.; Sakoda, A. Structural changes of lignocellulose by a nonionic surfactant, Tween 20, and their effects on cellulose adsorption and saccharification. Bioresour. Technol. 2011, 102, 9605–9612. [Google Scholar] [CrossRef]
- Yang, M.; Zhang, A.; Liu, B.; Li, W.; Xing, J. Improvement of cellulose conversion caused by the protection of Tween-80 on the adsorbed cellulase. Biochem. Eng. J. 2011, 56, 125–129. [Google Scholar] [CrossRef]
- Sipos, B.; Szilágyi, M.; Sebestyén, Z.; Perazzini, R.; Dienes, D.; Jakab, E.; Crestini, C.; Réczey, K. Mechanism of the positive effect of poly(ethylene glycol) addition in enzymatic hydrolysis of steam pretreated lignocelluloses. Comptes Rendus Biol. 2011, 334, 812–823. [Google Scholar] [CrossRef]
- Kirk, O.; Borchert, T.V.; Fuglsang, C.C. Industrial enzyme applications. Curr. Opin. Biotechnol. 2002, 13, 345–351. [Google Scholar] [CrossRef]
- Bjerre, J.; Simonsen, O.; Vind, J. Detergent enzymes- from discovery to product. HPC Today 2013, 8, 37–41. [Google Scholar]
- Krouwer, A.J.J.; Adams, C.D. Engineering an improved cellulase for fabric care in liquid detergents. SOFW J. 2016, 142, 64–67. [Google Scholar]
- Lund, H.; Kaasgaard, S.G.; Skagerlind, P.; Jorgensen, L.; Jørgensen, C.I.; van de Weert, M. Protease and Amylase Stability in the Presence of Chelators Used in Laundry Detergent Applications: Correlation Between Chelator Properties and Enzyme Stability in Liquid Detergents. J. Surfactants Deterg. 2011, 15, 265–276. [Google Scholar] [CrossRef]
- Kübelbeck, S.; Mikhael, J.; Keller, H.; Konradi, R.; Andrieu-Brunsen, A.; Baier, G. Enzyme-Polymer Conjugates to Enhance Enzyme Shelf Life in a Liquid Detergent Formulation. Macromol. Biosci. 2018, 18, e1800095. [Google Scholar] [CrossRef] [PubMed]
- Al-Ghanayem, A.A.; Joseph, B. Current prospective in using cold-active enzymes as eco-friendly detergent additive. Appl. Microbiol. Biotechnol. 2020, 104, 2871–2882. [Google Scholar] [CrossRef] [PubMed]
- Ghisaidoobe, A.B.T.; Chung, S.J. Intrinsic Tryptophan Fluorescence in the Detection and Analysis of Proteins: A Focus on Förster Resonance Energy Transfer Techniques. Int. J. Mol. Sci. 2014, 15, 22518–22538. [Google Scholar] [CrossRef]
- Kalyanasundaram, K.; Thomas, J.K. Environmental effects on vibronic band intensities in pyrene monomer florescence and their application in studies of micellar systems. J. Am. Chem. Soc. 1976, 99, 2039–2044. [Google Scholar] [CrossRef]
- Gorla, P.; Pandey, J.P.; Parthasarathy, S.; Merrick, M.; Siddavattam, D. Organophosphate hydrolase in Brevundimonas diminuta is targeted to the periplasmic face of the inner membrane by the twin arginine translocation (Tat) pathway. J. Bacteriol. 2009, 191, 6292–6299. [Google Scholar] [CrossRef]
- Wallace, T.C.; Bailey, R.L.; Blumberg, J.B.; Burton-Freeman, B.; Chen, C.-Y.O.; Crowe-White, K.M.; Drewnowski, A.; Hooshmand, S.; Johnson, E.; Lewis, R.; et al. Fruits, vegetables, and health: A comprehensive narrative, umbrella review of the science and recommendations for enhanced public policy to improve intake. Crit. Rev. Food Sci. Nutr. 2020, 60, 2174–2211. [Google Scholar] [CrossRef] [PubMed]
- Knežević, Z.; Serdar, M. Screening of fresh fruit and vegetables for pesticide residues on Croatian market. Food Control. 2009, 20, 419–422. [Google Scholar] [CrossRef]
- Popp, J.; Pető, K.; Nagy, J. Pesticide productivity and food security. A review. Agron. Sustain. Dev. 2013, 33, 243–255. [Google Scholar] [CrossRef]
- Costa, L.G. Organophosphorus Compounds at 80: Some Old and New Issues. Toxicol. Sci. 2018, 162, 24–35. [Google Scholar] [CrossRef] [PubMed]
- Peter, J.V.; Sudarsan, T.I.; Moran, J.L. Clinical features of organophosphate poisoning: A review of different classification systems and approaches. Indian J. Crit. Care Med. 2014, 18, 735–745. [Google Scholar] [CrossRef]
- Mahajan, R.; Chatterjee, S. A simple HPLC–DAD method for simultaneous detection of two organophosphates, profenofos and fenthion, and validation by soil microcosm experiment. Environ. Monit. Assess. 2018, 190, 327. [Google Scholar] [CrossRef]
- Rochu, D.; Beaufet, N.; Renault, F.; Viguié, N.; Masson, P. The wild type bacterial Co2+/Co2+-phosphotriesterase shows a mid-dle-range thermostability. Biochim. Biophys. Acta 2002, 1594, 207–218. [Google Scholar] [CrossRef]
- Li, H.; Hu, D.; Liang, F.; Huang, X.; Zhu, Q. Influence factors on the critical micelle concentration determination using pyrene as a probe and a simple method of preparing samples. R. Soc. Open Sci. 2020, 7, 192092. [Google Scholar] [CrossRef]
- Chaudhuri, T.K.; Arai, M.; Terada, T.P.; Ikura, T.; Kuwajima, K. Equilibrium and Kinetic Studies on Folding of the Authentic and Recombinant Forms of Human α-Lactalbumin by Circular Dichroism Spectroscopy. Biochemistry 2000, 39, 15643–15651. [Google Scholar] [CrossRef]
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Chi, M.-C.; Liao, T.-Y.; Lin, M.-G.; Lin, L.-L.; Wang, T.-F. Catalytic Performance of a Recombinant Organophosphate-Hydrolyzing Phosphotriesterase from Brevundimonas diminuta in the Presence of Surfactants. Catalysts 2021, 11, 597. https://doi.org/10.3390/catal11050597
Chi M-C, Liao T-Y, Lin M-G, Lin L-L, Wang T-F. Catalytic Performance of a Recombinant Organophosphate-Hydrolyzing Phosphotriesterase from Brevundimonas diminuta in the Presence of Surfactants. Catalysts. 2021; 11(5):597. https://doi.org/10.3390/catal11050597
Chicago/Turabian StyleChi, Meng-Chun, Ting-Yu Liao, Min-Guan Lin, Long-Liu Lin, and Tzu-Fan Wang. 2021. "Catalytic Performance of a Recombinant Organophosphate-Hydrolyzing Phosphotriesterase from Brevundimonas diminuta in the Presence of Surfactants" Catalysts 11, no. 5: 597. https://doi.org/10.3390/catal11050597
APA StyleChi, M.-C., Liao, T.-Y., Lin, M.-G., Lin, L.-L., & Wang, T.-F. (2021). Catalytic Performance of a Recombinant Organophosphate-Hydrolyzing Phosphotriesterase from Brevundimonas diminuta in the Presence of Surfactants. Catalysts, 11(5), 597. https://doi.org/10.3390/catal11050597





