Influence of Cathode Catalyst Layer with SiO2-Coated Pt/Ketjen Black Catalysts on Performance for Polymer Electrolyte Fuel Cells
Abstract
:1. Introduction
2. Results and Discussion
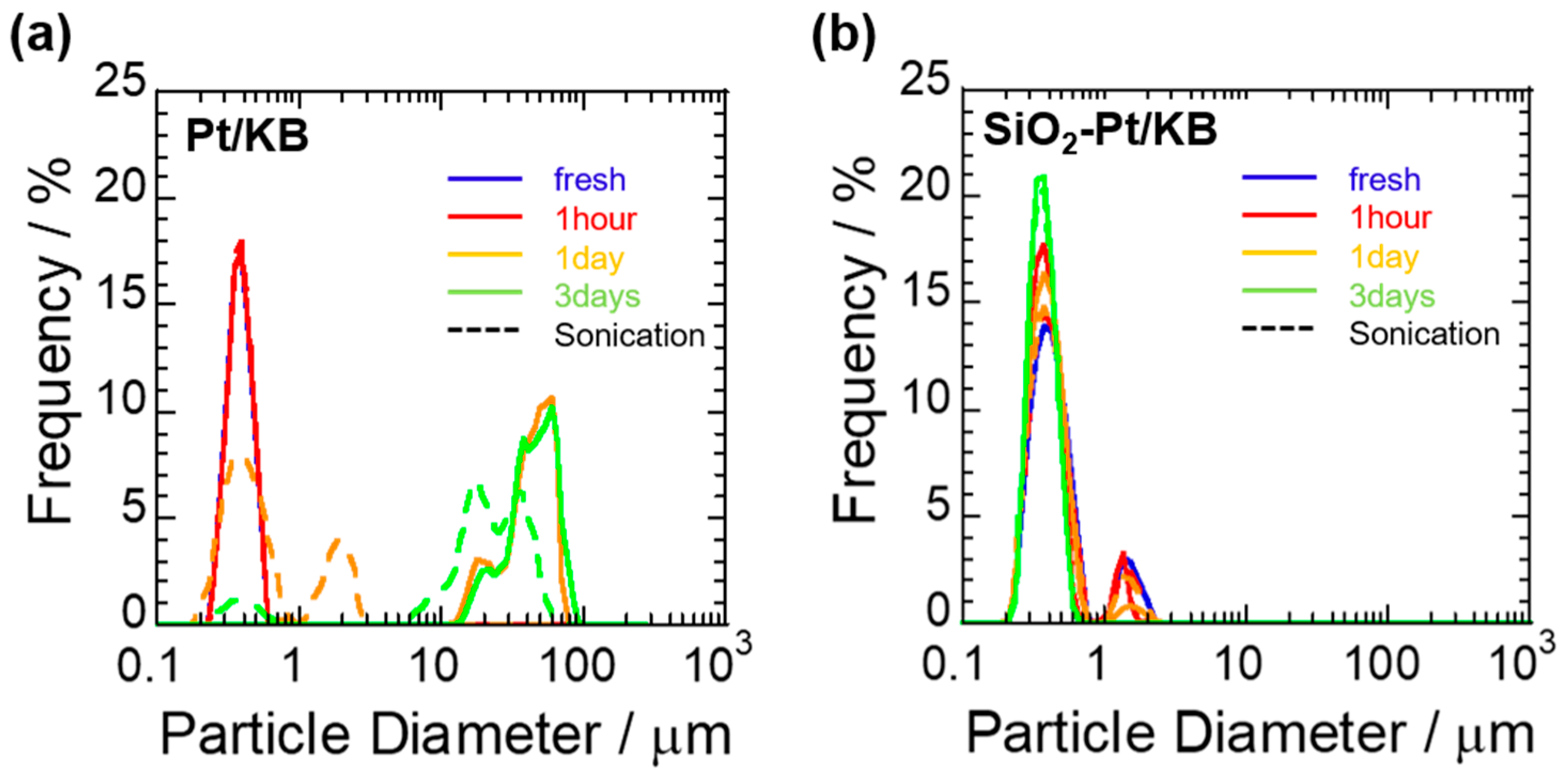
2.1. Particle Size Distribution of the Catalyst Ink
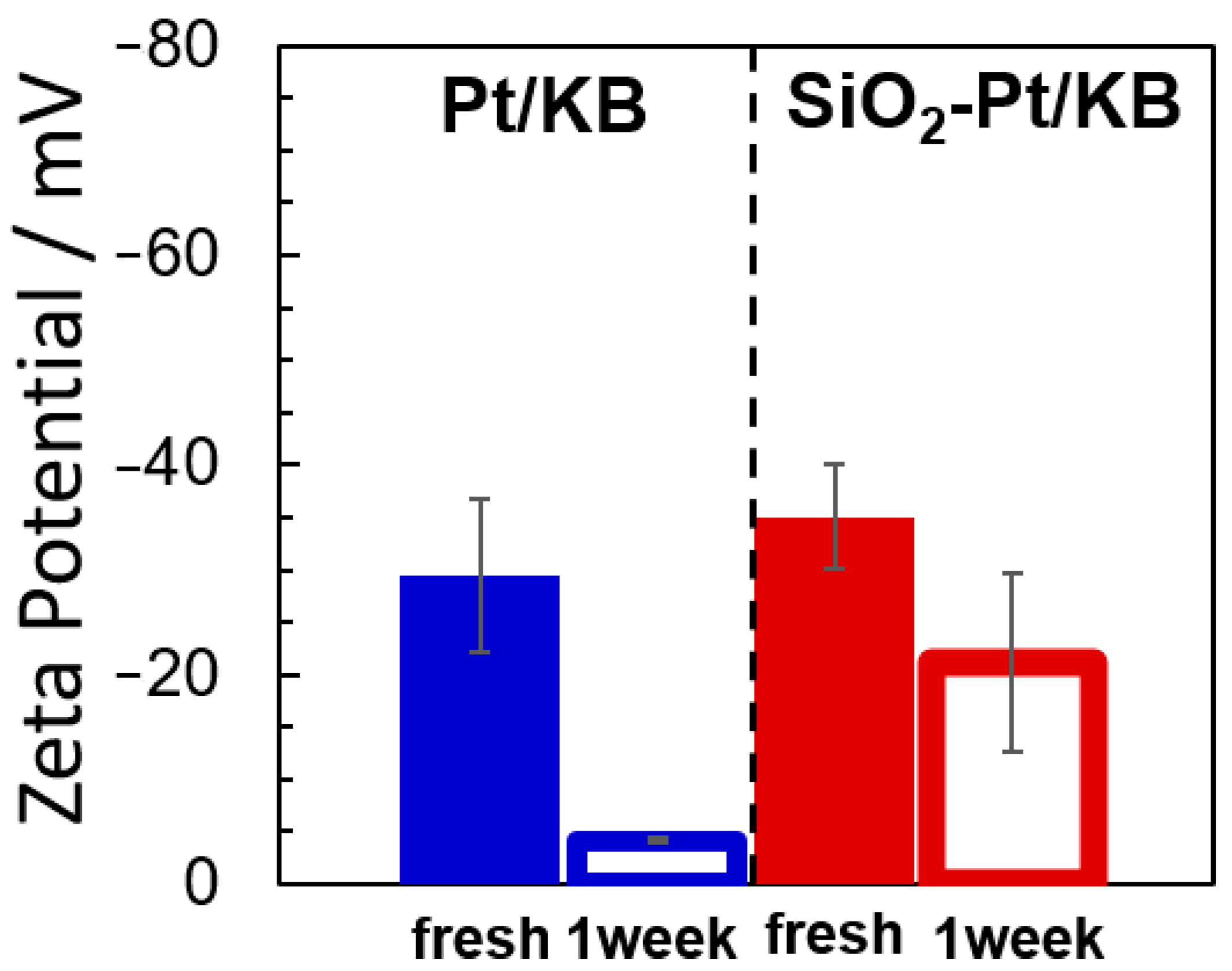
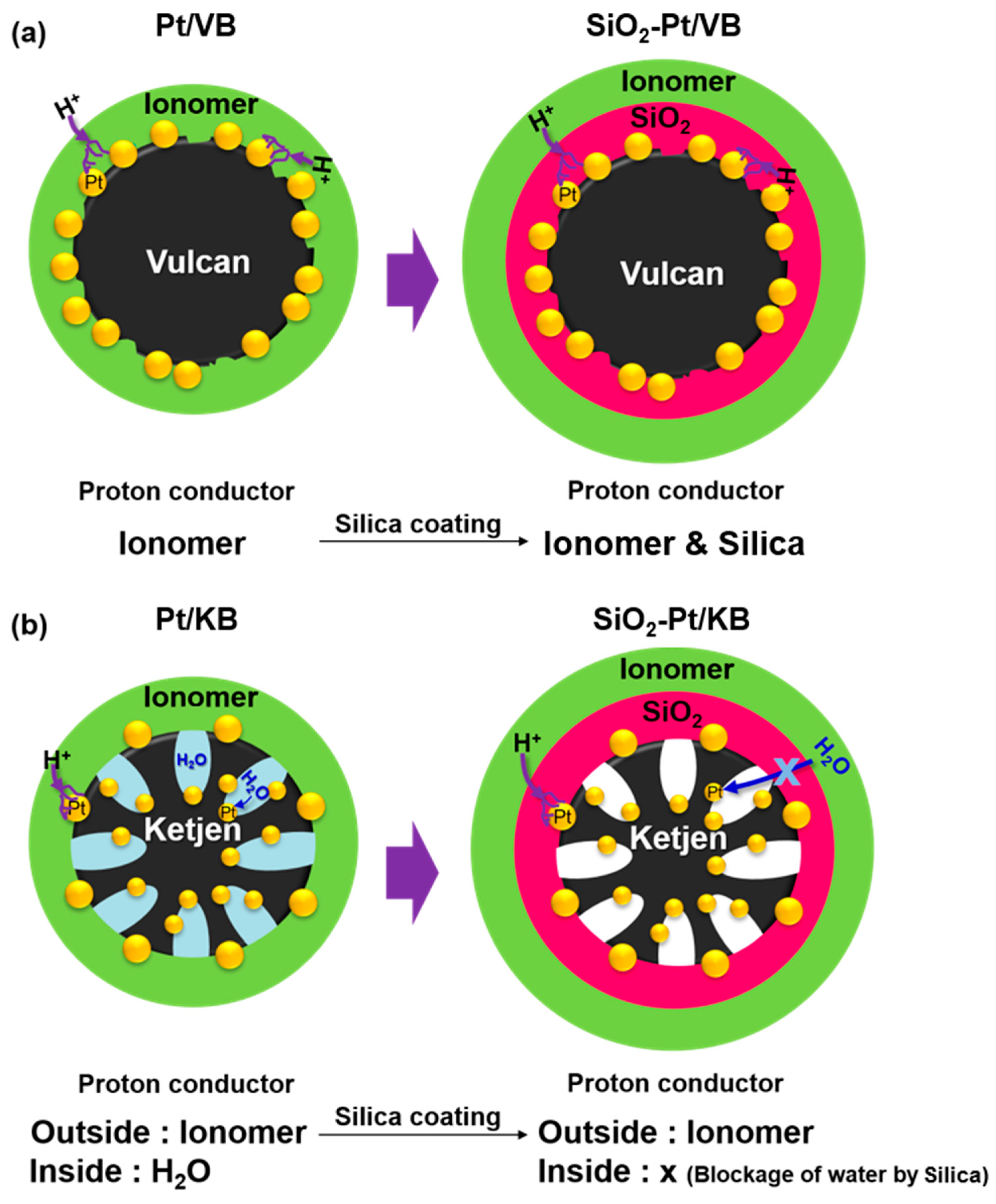
2.2. Morphology of Cathode CLs
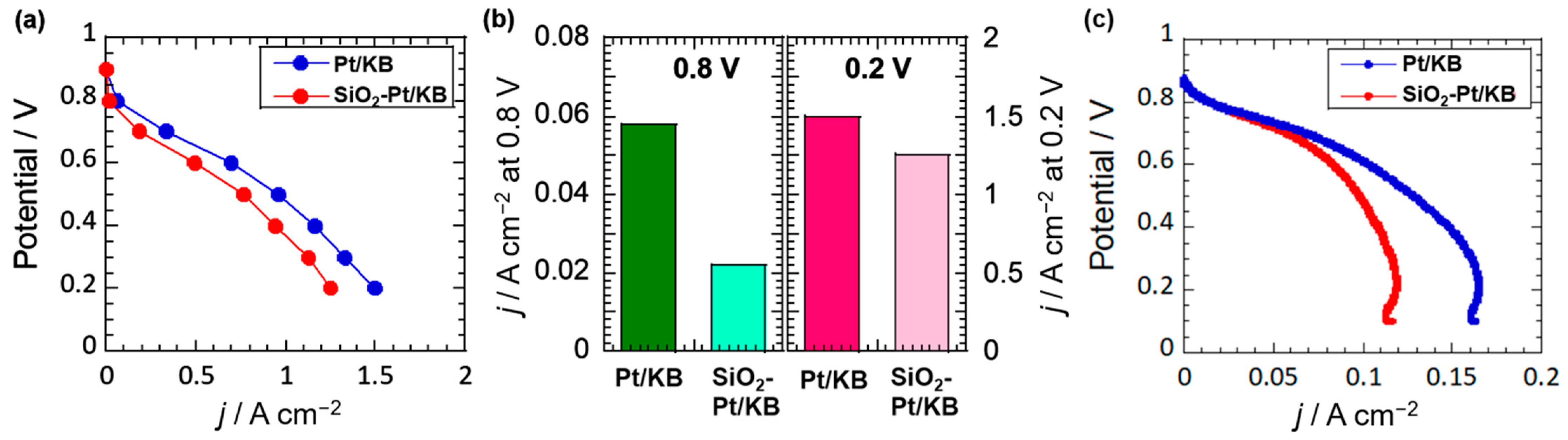
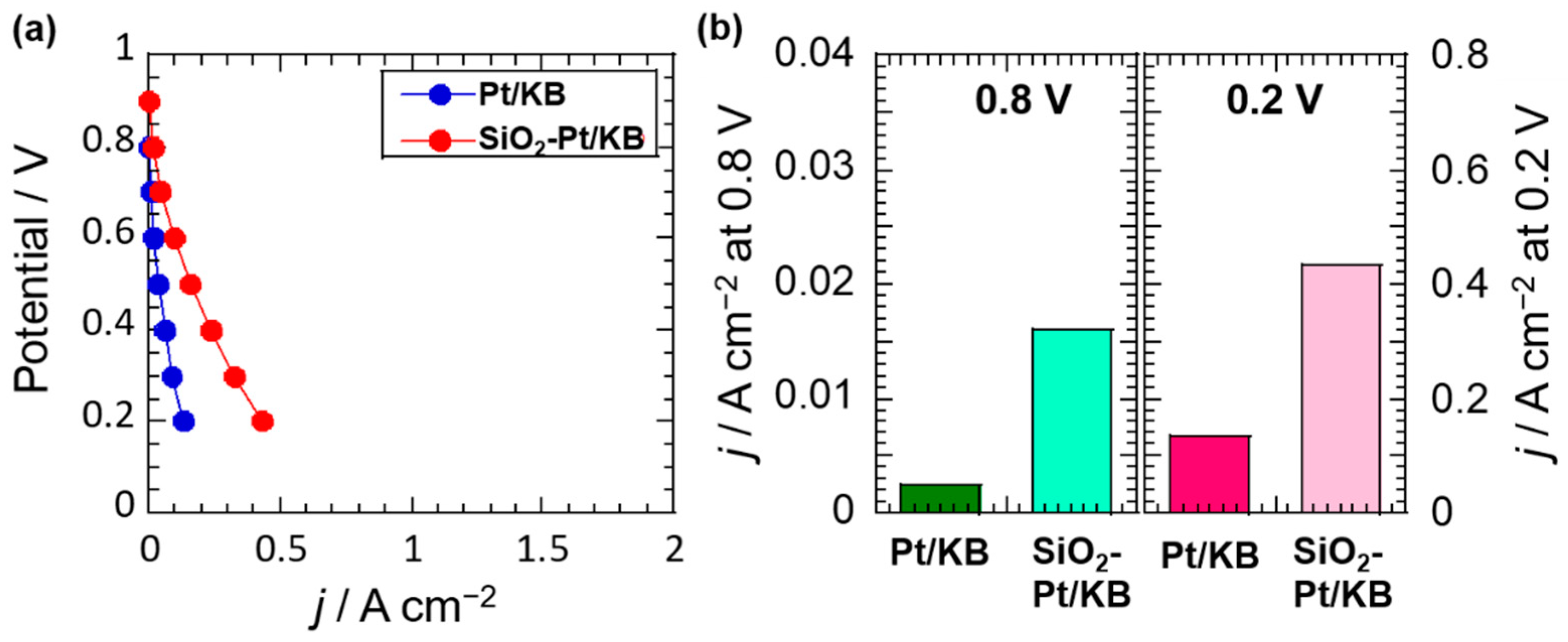
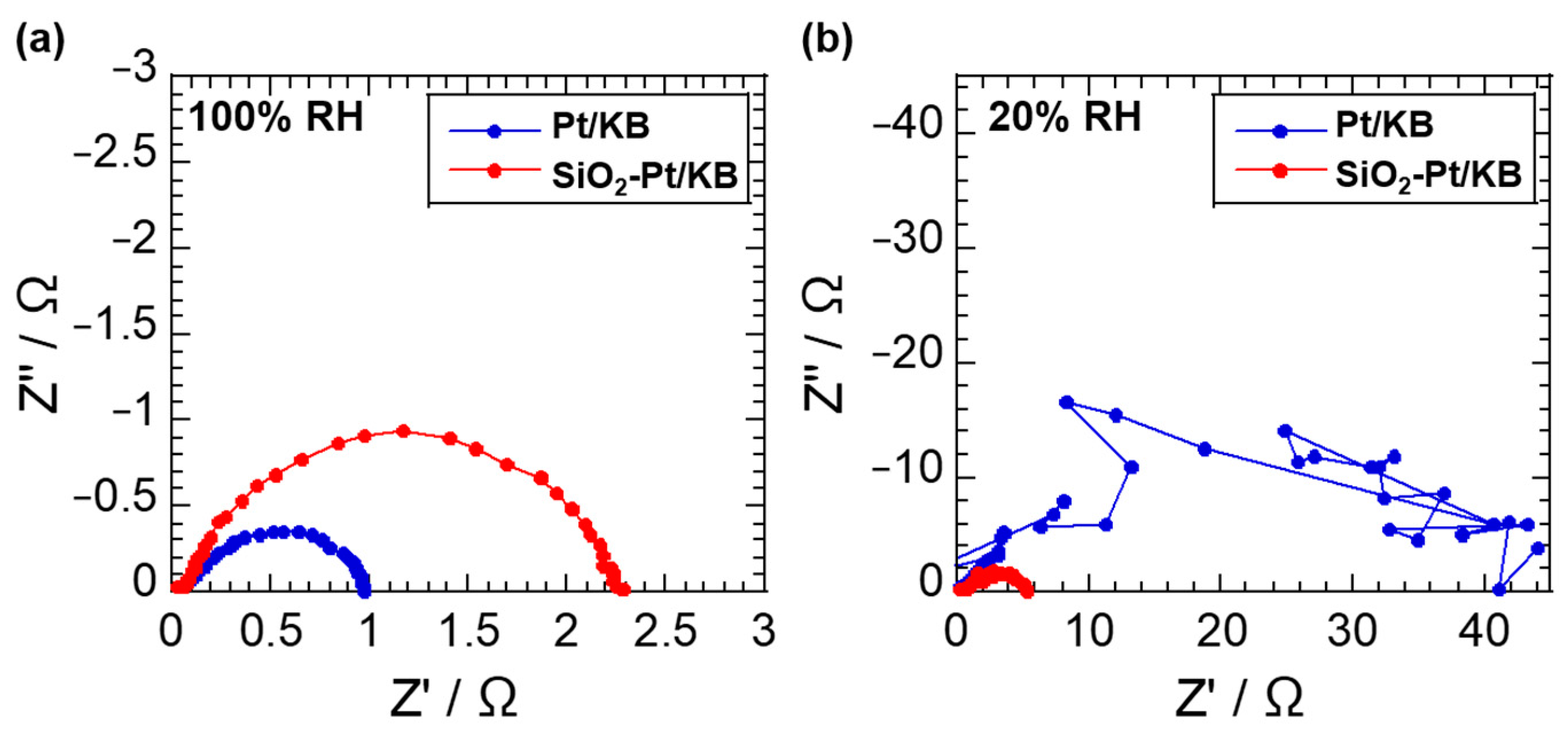
2.3. Electrochemical Analysis of Pt Catalysts
3. Materials and Methods
3.1. Preparation of SiO2-Coated Pt Catalyst
3.2. Fabrication of the MEA
3.3. Characterization of Catalysts, Ink, CLs
3.4. Electrochemical Measurements
4. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Debe, M.K. Electrocatalyst approaches and challenges for automotive fuel cells. Nature 2012, 486, 43–51. [Google Scholar] [CrossRef]
- Jung, N.; Chung, D.Y.; Ryu, J.; Yoo, S.J.; Sung, Y.E. Pt-based nanoarchitecture and catalyst design for fuel cell applications. Nano Today 2014, 9, 433–456. [Google Scholar] [CrossRef]
- Wong, W.Y.; Daud, W.R.W.; Mohamad, A.B.; Kadhum, A.A.H.; Loh, K.S.; Majlan, E.H. Recent progress in nitrogen-doped carbon and its composites as electrocatalysts for fuel cell applications. Int. J. Hydrogen Energy 2013, 38, 9370–9386. [Google Scholar] [CrossRef]
- Samad, S.; Loh, K.S.; Wong, W.Y.; Lee, T.K.; Sunarso, J.; Chong, S.T.; Wan Daud, W.R. Carbon and non-carbon support materials for platinum-based catalysts in fuel cells. Int. J. Hydrogen Energy 2018, 43, 7823–7854. [Google Scholar] [CrossRef]
- Inoue, G.; Ohnishi, T.; So, M.; Park, K.; Ono, M.; Tsuge, Y. Simulation of carbon black aggregate and evaluation of ionomer structure on carbon in catalyst layer of polymer electrolyte fuel cell. J. Power Sources 2019, 439, 227060. [Google Scholar] [CrossRef]
- Lee, D.; Hwang, S. Effect of loading and distributions of Nafion ionomer in the catalyst layer for PEMFCs. Int. J. Hydrogen Energy 2008, 33, 2790–2794. [Google Scholar] [CrossRef]
- Suzuki, A.; Sen, U.; Hattori, T.; Miura, R.; Nagumo, R.; Tsuboi, H.; Hatakeyama, N.; Endou, A.; Takaba, H.; Williams, M.C.; et al. Ionomer content in the catalyst layer of polymer electrolyte membrane fuel cell (PEMFC): Effects on diffusion and performance. Int. J. Hydrogen Energy 2011, 36, 2221–2229. [Google Scholar] [CrossRef]
- Kulikovsky, A. The effect of Nafion film on the cathode catalyst layer performance in a low–Pt PEM fuel cell. Electrochem. Commun. 2019, 103, 61–65. [Google Scholar] [CrossRef]
- Park, K.; Ohnishi, T.; Goto, M.; So, M.; Takenaka, S.; Tsuge, Y.; Inoue, G. Improvement of cell performance in catalyst layers with silica-coated Pt/carbon catalysts for polymer electrolyte fuel cells. Int. J. Hydrogen Energy 2019, 45, 1867–1877. [Google Scholar] [CrossRef]
- Takenaka, S.; Matsumori, H.; Matsune, H.; Kishida, M. Highly durable Pt cathode catalysts for polymer electrolyte fuel cells; coverage of carbon black-supported Pt catalysts with silica layers. Appl. Catal. A Gen. 2011, 409–410, 248–256. [Google Scholar] [CrossRef]
- Park, K.; Matsune, H.; Kishida, M.; Takenaka, S. Carbon-supported Pd-Ag catalysts with silica-coating layers as active and durable cathode catalysts for polymer electrolyte fuel cells. Int. J. Hydrogen Energy 2017, 42, 18951–18958. [Google Scholar] [CrossRef]
- Takenaka, S.; Miyamoto, H.; Utsunomiya, Y.; Matsune, H.; Kishida, M. Catalytic Activity of Highly Durable Pt/CNT Catalysts Covered with Hydrophobic Silica Layers for the Oxygen Reduction Reaction in PEFCs. J. Phys. Chem. C 2014, 118, 774–783. [Google Scholar] [CrossRef]
- Soboleva, T.; Zhao, X.; Malek, K.; Xie, Z.; Navessin, T.; Holdcroft, S. On the micro-, meso-, and macroporous structures of polymer electrolyte membrane fuel cell catalyst layers. ACS Appl. Mater. Interfaces 2010, 2, 375–384. [Google Scholar] [CrossRef] [Green Version]
- Ramaswamy, N.; Gu, W.; Ziegelbauer, J.M.; Kumaraguru, S. Carbon Support Microstructure Impact on High Current Density Transport Resistances in PEMFC Cathode. J. Electrochem. Soc. 2020, 167, 064515. [Google Scholar] [CrossRef]
- Mohanta, P.K.; Ripa, M.S.; Regnet, F.; Jörissen, L. Effects of supports BET surface areas on membrane electrode assembly performance at high current loads. Catalysts 2021, 11, 195. [Google Scholar] [CrossRef]
- Takenaka, S.; Iga, A.; Park, K.; Tanabe, E.; Matsune, H.; Kishida, M. Carbon nanofiber supports for the preparation of Pt-based metal nanoparticles with high tolerance to sintering. Appl. Catal. A Gen. 2013, 450, 211–221. [Google Scholar] [CrossRef]
- Park, Y.C.; Tokiwa, H.; Kakinuma, K.; Watanabe, M.; Uchida, M. Effects of carbon supports on Pt distribution, ionomer coverage and cathode performance for polymer electrolyte fuel cells. J. Power Sources 2016, 315, 179–191. [Google Scholar] [CrossRef] [Green Version]
- Takenaka, S.; Goto, M.; Masuda, Y.; Emura, S.; Kishida, M. Improvement in the durability of carbon black-supported Pt cathode catalysts by silica-coating for use in PEFCs. Int. J. Hydrogen Energy 2018, 43, 7473–7482. [Google Scholar] [CrossRef]
- Soboleva, T.; Malek, K.; Xie, Z.; Navessin, T.; Holdcroft, S. PEMFC catalyst layers: The role of micropores and mesopores on water sorption and fuel cell activity. ACS Appl. Mater. Interfaces 2011, 3, 1827–1837. [Google Scholar] [CrossRef] [PubMed]
- So, M.; Ohnishi, T.; Park, K.; Ono, M.; Tsuge, Y.; Inoue, G. The effect of solvent and ionomer on agglomeration in fuel cell catalyst inks: Simulation by the Discrete Element Method. Int. J. Hydrogen Energy 2019, 44, 28984–28995. [Google Scholar] [CrossRef]
- Inoue, G.; Kawase, M. Effect of porous structure of catalyst layer on effective oxygen diffusion coefficient in polymer electrolyte fuel cell. J. Power Sources 2016, 327, 1–10. [Google Scholar] [CrossRef]
- Ishikawa, H.; Sugawara, Y.; Inoue, G.; Kawase, M. Effects of Pt and ionomer ratios on the structure of catalyst layer: A theoretical model for polymer electrolyte fuel cells. J. Power Sources 2018, 374, 196–204. [Google Scholar] [CrossRef]
- So, M.; Park, K.; Tsuge, Y.; Inoue, G. A Particle Based Ionomer Attachment Model for a Fuel Cell Catalyst Layer. J. Electrochem. Soc. 2020, 167, 013544. [Google Scholar] [CrossRef] [Green Version]
- Terao, T.; Inoue, G.; Kawase, M.; Kubo, N.; Yamaguchi, M.; Yokoyama, K.; Tokunaga, T.; Shinohara, K.; Hara, Y.; Hara, T. Development of novel three-dimensional reconstruction method for porous media for polymer electrolyte fuel cells using focused ion beam-scanning electron microscope tomography. J. Power Sources 2017, 347, 108–113. [Google Scholar] [CrossRef]
- Liu, H.; Epting, W.K.; Litster, S. Gas Transport Resistance in Polymer Electrolyte Thin Films on Oxygen Reduction Reaction Catalysts. Langmuir 2015, 31, 9853–9858. [Google Scholar] [CrossRef] [PubMed]
- Mukherjee, P.P.; Wang, C.-Y. Stochastic Microstructure Reconstruction and Direct Numerical Simulation of the PEFC Catalyst Layer. J. Electrochem. Soc. 2006, 153, A840. [Google Scholar] [CrossRef]
- Inoue, G.; Yokoyama, K.; Ooyama, J.; Terao, T.; Tokunaga, T.; Kubo, N.; Kawase, M. Theoretical examination of effective oxygen diffusion coefficient and electrical conductivity of polymer electrolyte fuel cell porous components. J. Power Sources 2016, 327, 610–621. [Google Scholar] [CrossRef]

| Catalysts | Composition (Unit: Mass%) | ||
|---|---|---|---|
| SiO2 | Pt | C | |
| Pt/KB | - | 46.5 | 53.5 |
| SiO2-Pt/KB | 14.4 | 39.8 | 45.8 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Park, K.; Goto, M.; So, M.; Takenaka, S.; Tsuge, Y.; Inoue, G. Influence of Cathode Catalyst Layer with SiO2-Coated Pt/Ketjen Black Catalysts on Performance for Polymer Electrolyte Fuel Cells. Catalysts 2021, 11, 1517. https://doi.org/10.3390/catal11121517
Park K, Goto M, So M, Takenaka S, Tsuge Y, Inoue G. Influence of Cathode Catalyst Layer with SiO2-Coated Pt/Ketjen Black Catalysts on Performance for Polymer Electrolyte Fuel Cells. Catalysts. 2021; 11(12):1517. https://doi.org/10.3390/catal11121517
Chicago/Turabian StylePark, Kayoung, Masaki Goto, Magnus So, Sakae Takenaka, Yoshifumi Tsuge, and Gen Inoue. 2021. "Influence of Cathode Catalyst Layer with SiO2-Coated Pt/Ketjen Black Catalysts on Performance for Polymer Electrolyte Fuel Cells" Catalysts 11, no. 12: 1517. https://doi.org/10.3390/catal11121517
APA StylePark, K., Goto, M., So, M., Takenaka, S., Tsuge, Y., & Inoue, G. (2021). Influence of Cathode Catalyst Layer with SiO2-Coated Pt/Ketjen Black Catalysts on Performance for Polymer Electrolyte Fuel Cells. Catalysts, 11(12), 1517. https://doi.org/10.3390/catal11121517






