Insights into the Kinetics Degradation of Bisphenol A by Catalytic Wet Air Oxidation with Metals Supported onto Carbon Nanospheres
Abstract
:1. Introduction
2. Results and Discussion
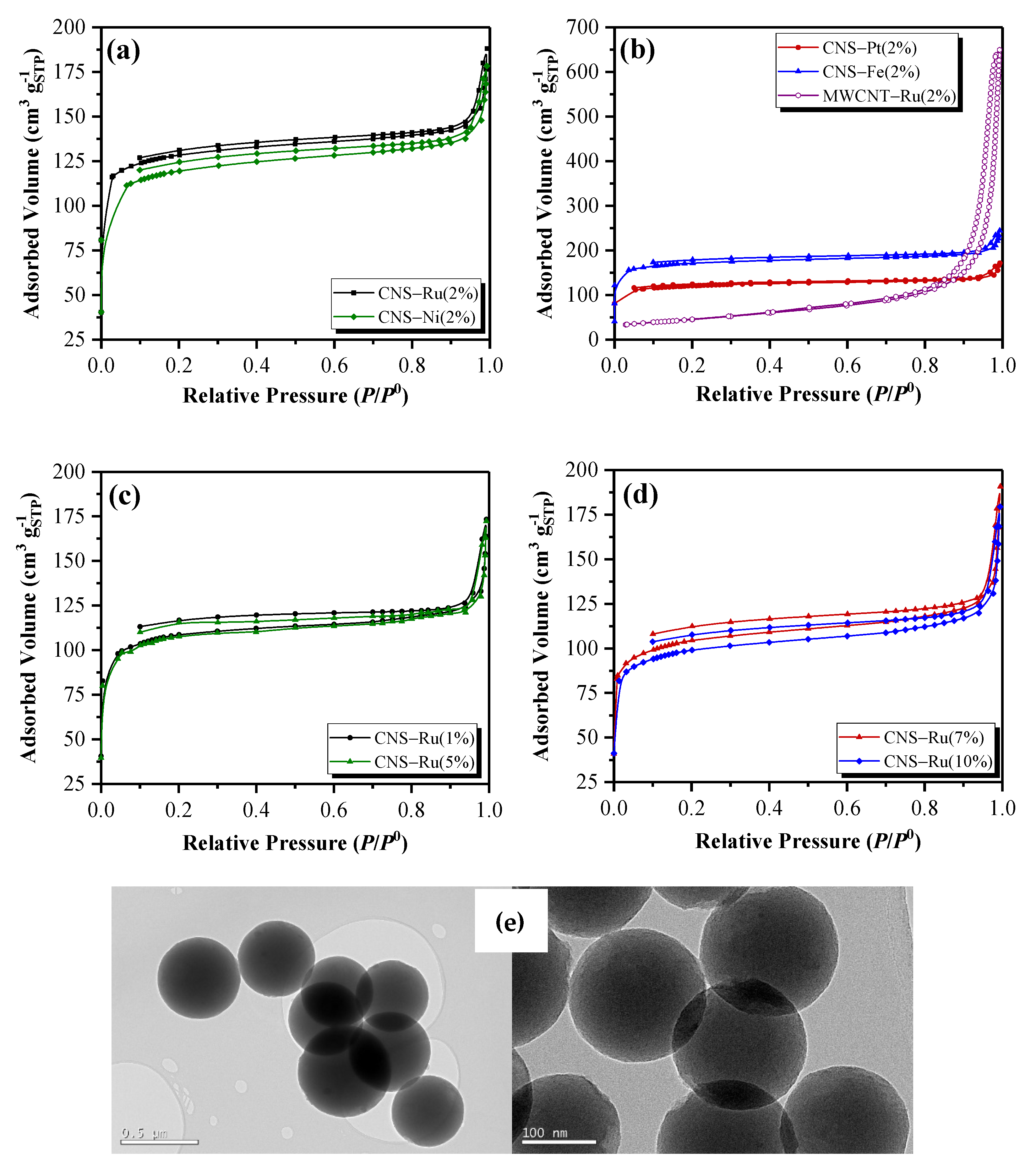
2.1. Catalyst Characterization
2.2. Influence of the Ruthenium Load in the CNS-Ru Catalyst
2.3. Influence of the Metal in the Catalyst for the BPA Degradation
2.4. Reusability and Stability Tests with CNS-Ru and CNS-Pt
2.5. Kinetics Models Determination
2.5.1. Simple Potential Model
2.5.2. Complex Potential Model
2.6. Proof of Concept: Treatment of a Real Hospital Wastewater Effluent with CNS-Ru and CNS-Pt
3. Materials and Methods
3.1. Materials
3.2. Synthesis of the Catalyst
3.3. Catalyst Characterization
3.4. Characterization of the Wastewater Matrix
3.5. WAO and CWAO Experiments
3.6. Sample Analysis
4. Conclusions
- The first minimum value to achieve a conversion above 97% in 90 min of reaction time was a 2 wt. % of Ru in the CNS-Ru catalyst. Leaching increased when metal content in the catalyst increased.
- The better metal of those tested was Ru against Fe and Ni, which suffered more leaching. MWCNT as support adsorbed a significant amount of BPA.
- Reusability of CNS-Ru was possible, while the activity of the CNS-Pt in the reuse reactions decreased.
- Experimental data were adjusted to a potential model to reproduce the kinetics behavior of the reactions. Another more complex potential model was employed for CWAO reactions, considering the variety of different parameters, such as temperature, pollutant concentration, total pressure, and catalyst concentration. This model was successfully proposed to imitate the experimental data of CWAO process.
- Achieved BPA degradation in the hospital wastewater was higher with CNS-Pt (85%), followed by CNS-Ru (78%) in 180 min of reaction time.
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- The European Parliament and the Council of the European Union Directive 2013/39/EU of the European Parliament and of the Council of 12 August 2013 amending Directives 2000/60/EC and 2008/105/EC as regards priority substances in the field of water policy. Off. J. Eur. Union. L Ser. 2013, 226, 1–17.
- Rathi, B.S.; Kumar, P.S. Application of adsorption process for effective removal of emerging contaminants from water and wastewater. Environ. Pollut. 2021, 280, 116995. [Google Scholar] [CrossRef] [PubMed]
- Ahamad, A.; Madhav, S.; Singh, A.K.; Kumar, A.; Singh, P. Types of Water Pollutants: Conventional and Emerging. In Sensors in Water Pollutants Monitoring: Role of Material; Pooja, D., Kumar, P., Singh, P., Patil, S., Eds.; Springer: Singapore, 2020; pp. 21–41. ISBN 978-981-15-0671-0. [Google Scholar]
- Sèdes, L.; Desdoits-Lethimonier, C.; Rouaisnel, B.; Holota, H.; Thirouard, L.; Lesne, L.; Damon-Soubeyrand, C.; Martinot, E.; Saru, J.P.; Mazaud-Guittot, S.; et al. Crosstalk between BPA and FXRα signaling pathways lead to alterations of undifferentiated germ cell homeostasis and male fertility disorders. Stem Cell Rep. 2018, 11, 944–958. [Google Scholar] [CrossRef] [Green Version]
- Rubin, B.S. Bisphenol A: An endocrine disruptor with widespread exposure and multiple effects. J. Steroid Biochem. Mol. Biol. 2011, 127, 27–34. [Google Scholar] [CrossRef] [PubMed]
- Helmestam, M.; Davey, E.; Stavreus-Evers, A.; Olovsson, M. Bisphenol A affects human endometrial endothelial cell angiogenic activity in vitro. Reprod. Toxicol. 2014, 46, 69–76. [Google Scholar] [CrossRef]
- Norwegian Environment Agency List of Priority Substances; Norwegian Environment Agency: Oslo, Norway, 2017; pp. 1–16.
- Lancheros, J.C.; Madera-Parra, C.A.; Caselles-Osorio, A.; Torres-López, W.A.; Vargas-Ramírez, X.M. Ibuprofen and Naproxen removal from domestic wastewater using a horizontal subsurface flow constructed wetland coupled to ozonation. Ecol. Eng. 2019, 135, 89–97. [Google Scholar] [CrossRef]
- Tran, N.H.; Reinhard, M.; Gin, K.Y. Occurrence and fate of emerging contaminants in municipal wastewater treatment plants from different geographical regions—A review. Water Res. 2018, 133, 182–207. [Google Scholar] [CrossRef] [PubMed]
- Česen, M.; Ahel, M.; Terzić, S.; Heath, D.J.; Heath, E. The occurrence of contaminants of emerging concern in Slovenian and Croatian wastewaters and receiving Sava river. Sci. Total Environ. 2019, 650, 2446–2453. [Google Scholar] [CrossRef] [PubMed]
- Peña-Álvarez, A.; Castillo-Alanís, A. Identificación y cuantificación de contaminantes emergentes en aguas residuales por microextracción en fase sólida-cromatografía de gases-espectrometría de masas (MEFS-CG-EM). TIP Rev. Espec. Cienc. Químico-Biológicas 2015, 18, 29–42. [Google Scholar] [CrossRef] [Green Version]
- Corrales, J.; Kristofco, L.A.; Baylor Steele, W.; Yates, B.S.; Breed, C.S.; Spencer Williams, E.; Brooks, B.W. Global assessment of bisphenol a in the environment: Review and analysis of its occurrence and bioaccumulation. Dose-Response An Int. J. 2015, 13, 1559325815598308. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Martín, J.; Camacho-Muñoz, D.; Santos, J.L.; Aparicio, I.; Alonso, E. Determination of emerging and priority industrial pollutants in surface water and wastewater by liquid chromatography-negative electrospray ionization tandem mass spectrometry. Anal. Bioanal. Chem. 2014, 406, 3709–3716. [Google Scholar] [CrossRef] [PubMed]
- Oluwole, A.O.; Omotola, E.O.; Olatunji, O.S. Pharmaceuticals and personal care products in water and wastewater: A review of treatment processes and use of photocatalyst immobilized on functionalized carbon in AOP degradation. BMC Chem. 2020, 14, 62. [Google Scholar] [CrossRef] [PubMed]
- Oliveira, A.S.; Baeza, J.A.; Saenz de Miera, B.; Calvo, L.; Rodriguez, J.J.; Gilarranz, M.A. Aqueous phase reforming coupled to catalytic wet air oxidation for the removal and valorisation of phenolic compounds in wastewater. J. Environ. Manag. 2020, 274, 111199. [Google Scholar] [CrossRef] [PubMed]
- Liu, N.; Lu, N.; Yu, H.; Chen, S.; Quan, X. Degradation of aqueous bisphenol A in the CoCN/Vis/PMS system: Catalyst design, reaction kinetic and mechanism analysis. Chem. Eng. J. 2021, 407, 127228. [Google Scholar] [CrossRef]
- Chu, J.H.; Kang, J.K.; Park, S.J.; Lee, C.G. Bisphenol A degradation using waste antivirus copper film with enhanced sono-Fenton-like catalytic oxidation. Chemosphere 2021, 276, 130218. [Google Scholar] [CrossRef]
- Li, S.; Wu, Y.; Zheng, Y.; Jing, T.; Tian, J.; Zheng, H.; Wang, N.; Nan, J.; Ma, J. Free-radical and surface electron transfer dominated bisphenol A degradation in system of ozone and peroxydisulfate co-activated by CoFe2O4-biochar. Appl. Surf. Sci. 2021, 541, 147887. [Google Scholar] [CrossRef]
- Rathnayake, B.; Heponiemi, A.; Huovinen, M.; Ojala, S.; Pirilä, M.; Loikkanen, J.; Azalim, S.; Saouabe, M.; Brahmi, R.; Vähäkangas, K.; et al. Photocatalysis and catalytic wet air oxidation: Degradation and toxicity of bisphenol A containing wastewaters. Environ. Technol. 2020, 41, 3272–3283. [Google Scholar] [CrossRef]
- Rashid, T.; Iqbal, D.; Hazafa, A.; Hussain, S.; Sher, F.; Sher, F. Formulation of zeolite supported nano-metallic catalyst and applications in textile effluent treatment. J. Environ. Chem. Eng. 2020, 8, 104023. [Google Scholar] [CrossRef]
- Baloyi, J.; Ntho, T.; Moma, J. Synthesis of highly active and stable Al/Zr pillared clay as catalyst for catalytic wet oxidation of phenol. J. Porous Mater. 2019, 26, 583–597. [Google Scholar] [CrossRef]
- Sun, W.; Wei, H.; yang An, L.; Jin, C.; Wu, H.; Xiong, Z.; Pu, C.; Sun, C. Oxygen vacancy mediated La1-xCexFeO3-δ perovskite oxides as efficient catalysts for CWAO of acrylic acid by A-site Ce doping. Appl. Catal. B Environ. 2019, 245, 20–28. [Google Scholar] [CrossRef]
- Soares, O.S.; Rocha, R.P.; Órfão, J.J.; Pereira, M.F.; Figueiredo, J.L. Mechanothermal Approach for N-, S-, P-, and B-Doping of Carbon Nanotubes: Methodology and Catalytic Performance in Wet Air Oxidation. C 2019, 5, 30. [Google Scholar] [CrossRef] [Green Version]
- Nieto-Márquez, A.; Romero, R.; Romero, A.; Valverde, J.L. Carbon nanospheres: Synthesis, physicochemical properties and applications. J. Mater. Chem. 2011, 21, 1664–1672. [Google Scholar] [CrossRef]
- Brunauer, S.; Deming, L.S.; Deming, W.E.; Teller, E. On a Theory of the van der Waals Adsorption of Gases. J. Am. Chem. Soc. 1940, 62, 1723–1732. [Google Scholar] [CrossRef]
- Thommes, M.; Kaneko, K.; Neimark, A.V.; Olivier, J.P.; Rodriguez-Reinoso, F.; Rouquerol, J.; Sing, K.S. Physisorption of gases, with special reference to the evaluation of surface area and pore size distribution (IUPAC Technical Report). Pure Appl. Chem. 2015, 87, 1051–1069. [Google Scholar] [CrossRef] [Green Version]
- Martín Martínez, J.M. Adsorción Física de Gases y Vapores por Carbones; Secretariado de Publicaciones de la Universidad de Alicante: Alicante, Spain, 1990; ISBN 8486809339. [Google Scholar]
- Serra-Pérez, E.; Álvarez-Torrellas, S.; Águeda, V.I.; Delgado, J.A.; Ovejero, G.; García, J. Insights into the removal of Bisphenol A by catalytic wet air oxidation upon carbon nanospheres-based catalysts: Key operating parameters, degradation intermediates and reaction pathway. Appl. Surf. Sci. 2019, 473, 726–737. [Google Scholar] [CrossRef]
- Levenspiel, O. Chemical Reaction Engineering, 3rd ed.; John Wiley & Sons: New York, NY, USA, 1999; ISBN 047125424X. [Google Scholar]
- Eftaxias, A.; Font, J.; Fortunyb, A.; Giralt, J.; Fabregat, A.; Stüber, F. Kinetic modelling of catalytic wet air oxidation of phenol by simulated annealing. Appl. Catal. B Environ. 2001, 33, 175–190. [Google Scholar] [CrossRef] [Green Version]
- Ovejero, G.; Rodríguez, A.; Vallet, A.; García, J. Catalytic wet air oxidation of a non-azo dye with Ni/MgAlO catalyst. Chem. Eng. J. 2013, 215–216, 168–173. [Google Scholar] [CrossRef]
- Gomes, H.T.; Figueiredo, J.L.; Faria, J.L.; Serp, P.; Kalck, P. Carbon-supported iridium catalysts in the catalytic wet air oxidation of carboxylic acids: Kinetics and mechanistic interpretation. J. Mol. Catal. A Chem. 2002, 182–183, 47–60. [Google Scholar] [CrossRef]
- Filip, J.; Cajthaml, T.; Najmanová, P.; Černík, M.; Zbořil, R. (Eds.) Advanced Nano-Bio Technologies for Water and Soil Treatment; Springer: New York, NY, USA, 2020; ISBN 978-3-030-29839-5. [Google Scholar]
- Rice, E.W.; Baird, R.B.; Eaton, A.D. (Eds.) Standard Methods for the Examination of Water and Wastewater, 23rd ed.; American Public Health Association, American Water Works Association, Water Environment Federation: Washington, DC, USA, 2017; ISBN 9780875532875. [Google Scholar]
- Ortiz de García, S.A.; Pinto Pinto, G.; García-Encina, P.A.; Irusta-Mata, R. Ecotoxicity and environmental risk assessment of pharmaceuticals and personal care products in aquatic environments and wastewater treatment plants. Ecotoxicology 2014, 23, 1517–1533. [Google Scholar] [CrossRef]
- Abbas, M.; Adil, M.; Ehtisham-ul-Haque, S.; Munir, B.; Yameen, M.; Ghaffar, A.; Shar, G.A.; Asif Tahir, M.; Iqbal, M. Vibrio fischeri bioluminescence inhibition assay for ecotoxicity assessment: A review. Sci. Total Environ. 2018, 626, 1295–1309. [Google Scholar] [CrossRef]
- Donner, E.; Kosjek, T.; Qualmann, S.; Kusk, K.O.; Heath, E.; Revitt, D.M.; Ledin, A.; Andersen, H.R. Ecotoxicity of carbamazepine and its UV photolysis transformation products. Sci. Total Environ. 2013, 443, 870–876. [Google Scholar] [CrossRef] [PubMed] [Green Version]









| Catalyst | %C | %H | %N |
|---|---|---|---|
| CNS-Ru(2%) | 69.50 | 2.96 | 3.51 |
| CNS-Pt(2%) | 68.13 | 3.10 | 3.34 |
| CNS-Fe(2%) | 63.03 | 3.10 | 3.43 |
| CNS-Ni(2%) | 65.98 | 3.39 | 3.16 |
| Catalyst | Metal—Active Phase | Weight Content (%) |
|---|---|---|
| CNS-Ru(2%) | Ru | 1.95 |
| CNS-Pt(2%) | Pt | 2.05 |
| CNS-Fe(2%) | Fe | 2.07 |
| CNS-Ni(2%) | Ni | 2.03 |
| CNS-Ru(1%) | Ru | 0.97 |
| CNS-Ru(5%) | Ru | 4.86 |
| CNS-Ru(7%) | Ru | 6.93 |
| CNS-Ru(10%) | Ru | 10.2 |
| Catalyst | Metal | wt. (%) | ABET (m2 g−1) | VP (cm3 g−1) | Vm (cm3 g−1) | Am (m2 g−1) | Aext (m2 g−1) |
|---|---|---|---|---|---|---|---|
| CNS | Ru | 2 | 340 ± 6 | 0.326 | 0.102 | 335 | 119 |
| Pt | 2 | 330 ± 6 | 0.331 | 0.150 | 248 | 82 | |
| Fe | 2 | 586 ± 9 | 0.361 | 0.199 | 433 | 153 | |
| Ni | 2 | 406 ± 6 | 0.260 | 0.134 | 290 | 116 | |
| MWCNT | Ru | 2 | 160 ± 0.3 | 0.916 | 0.009 | 21 | 138 |
| CNS | Ru | 1 | 372 ± 5 | 0.238 | 0.119 | 258 | 114 |
| Ru | 5 | 367 ± 5 | 0.280 | 0.113 | 245 | 118 | |
| Ru | 7 | 358 ± 5 | 0.277 | 0.109 | 238 | 120 | |
| Ru | 10 | 340 ± 5 | 0.245 | 0.104 | 227 | 113 |
| Parameter | Value |
|---|---|
| k0 (mmol1−a·La·gRu−1·min−1) | 8606 |
| Ea (kJ·mol−1) | 31.03 ± 2.55 |
| a | 0.97 ± 0.05 |
| R2 | 0.988 |
| Parameter | Value |
|---|---|
| k0 (mmol1−a·La+c·gRu−1−c·min−1·bar−b) | 1014 |
| Ea (kJ·mol−1) | 31.60 ± 2.85 |
| a | 0.99 ± 0.05 |
| b | 0.55 ± 0.06 |
| c | 0.78 ± 0.03 |
| R2 | 0.988 |
| Experiment | T (°C) | P (bar) | [CNS-Ru(2%)] (gRu L−1) | CBPA 0 (mg L−1) |
|---|---|---|---|---|
| 1 | 110 | 20 | 0.04 | 20 |
| 2 | 130 | 20 | 0.04 | 20 |
| 3 | 140 | 20 | 0.04 | 20 |
| 4 | 150 | 20 | 0.04 | 20 |
| 5 | 130 | 30 | 0.04 | 20 |
| 6 | 130 | 40 | 0.04 | 20 |
| 7 | 130 | 50 | 0.04 | 20 |
| 8 | 130 | 20 | 0.01 | 20 |
| 9 | 130 | 20 | 0.02 | 20 |
| 10 | 130 | 20 | 0.06 | 20 |
| 11 | 130 | 20 | 0.04 | 5 |
| 12 | 130 | 20 | 0.04 | 10 |
| 13 | 130 | 20 | 0.04 | 30 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Serra-Pérez, E.; Rodríguez, J.G. Insights into the Kinetics Degradation of Bisphenol A by Catalytic Wet Air Oxidation with Metals Supported onto Carbon Nanospheres. Catalysts 2021, 11, 1293. https://doi.org/10.3390/catal11111293
Serra-Pérez E, Rodríguez JG. Insights into the Kinetics Degradation of Bisphenol A by Catalytic Wet Air Oxidation with Metals Supported onto Carbon Nanospheres. Catalysts. 2021; 11(11):1293. https://doi.org/10.3390/catal11111293
Chicago/Turabian StyleSerra-Pérez, Estrella, and Juan García Rodríguez. 2021. "Insights into the Kinetics Degradation of Bisphenol A by Catalytic Wet Air Oxidation with Metals Supported onto Carbon Nanospheres" Catalysts 11, no. 11: 1293. https://doi.org/10.3390/catal11111293








