Exploring the Photothermo-Catalytic Performance of Brookite TiO2-CeO2 Composites
Abstract
1. Introduction
2. Results and Discussion
2.1. Brookite TiO2-CeO2 Composites Characterization
2.2. Surface Properties of Brookite TiO2-CeO2 Composites
2.3. (Photo)Catalytic Oxidation of 2-Propanol
2.4. (Photo)Catalytic Oxidation of Ethanol
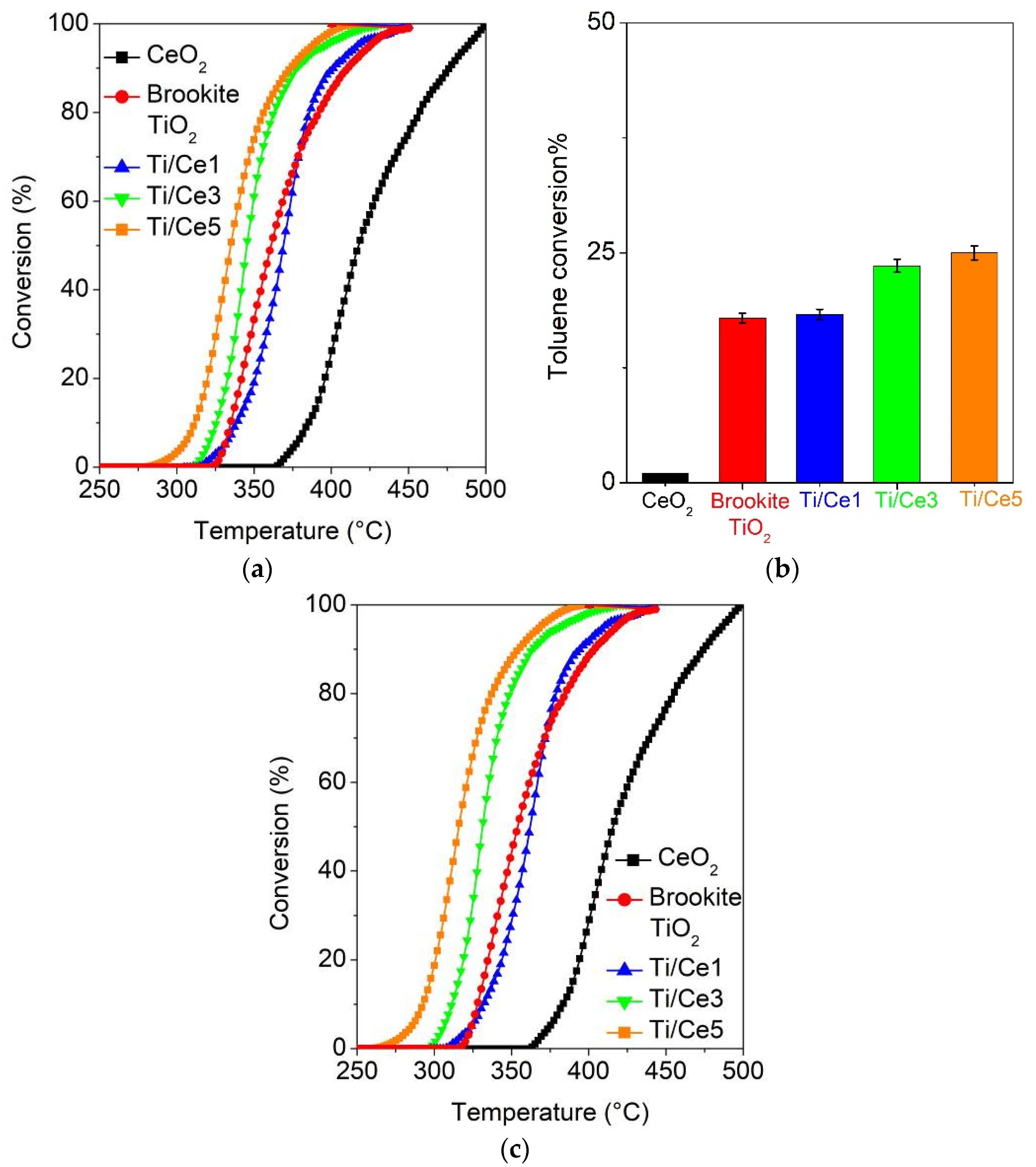
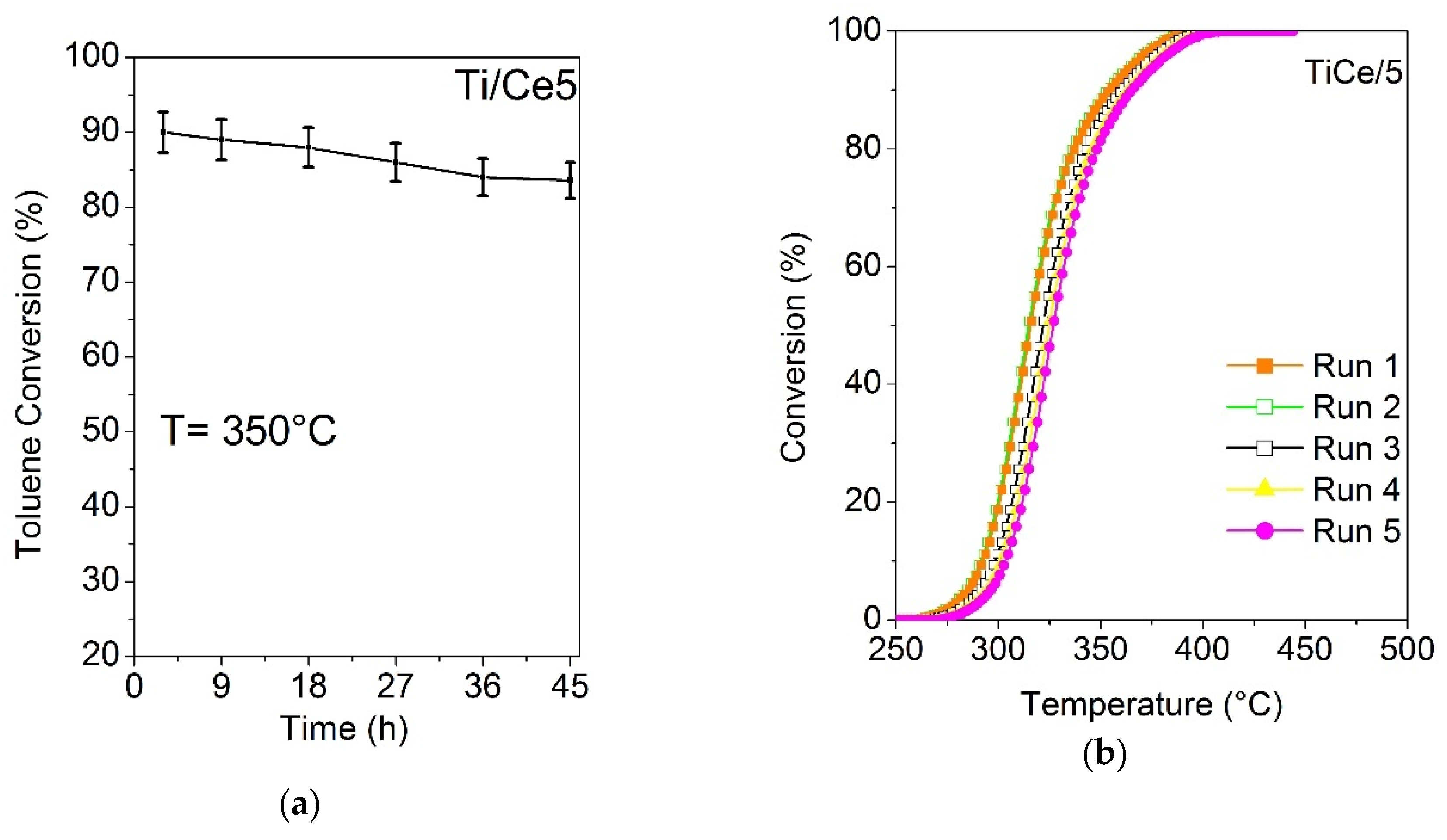
2.5. (Photo)Catalytic Oxidation of Toluene
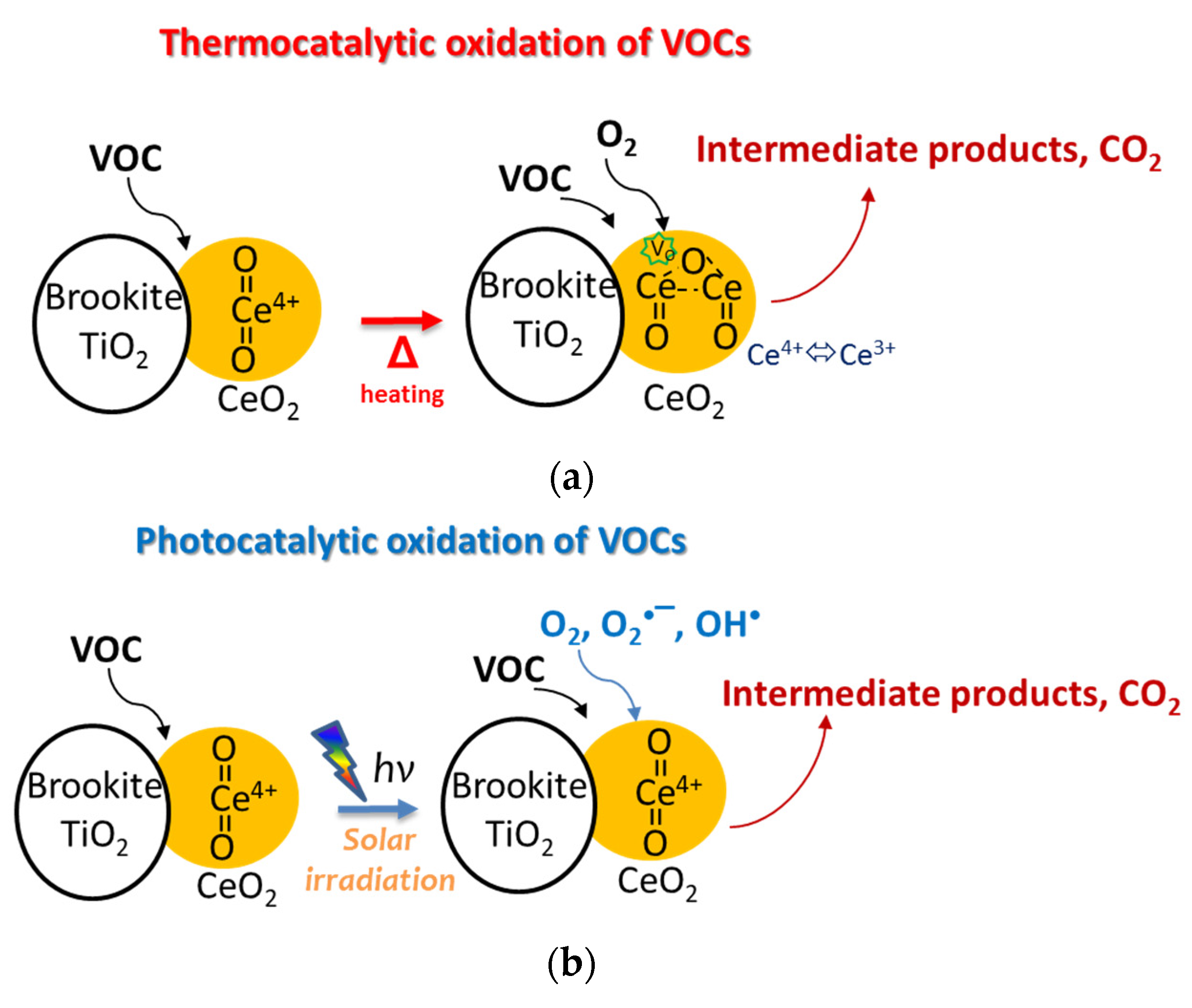
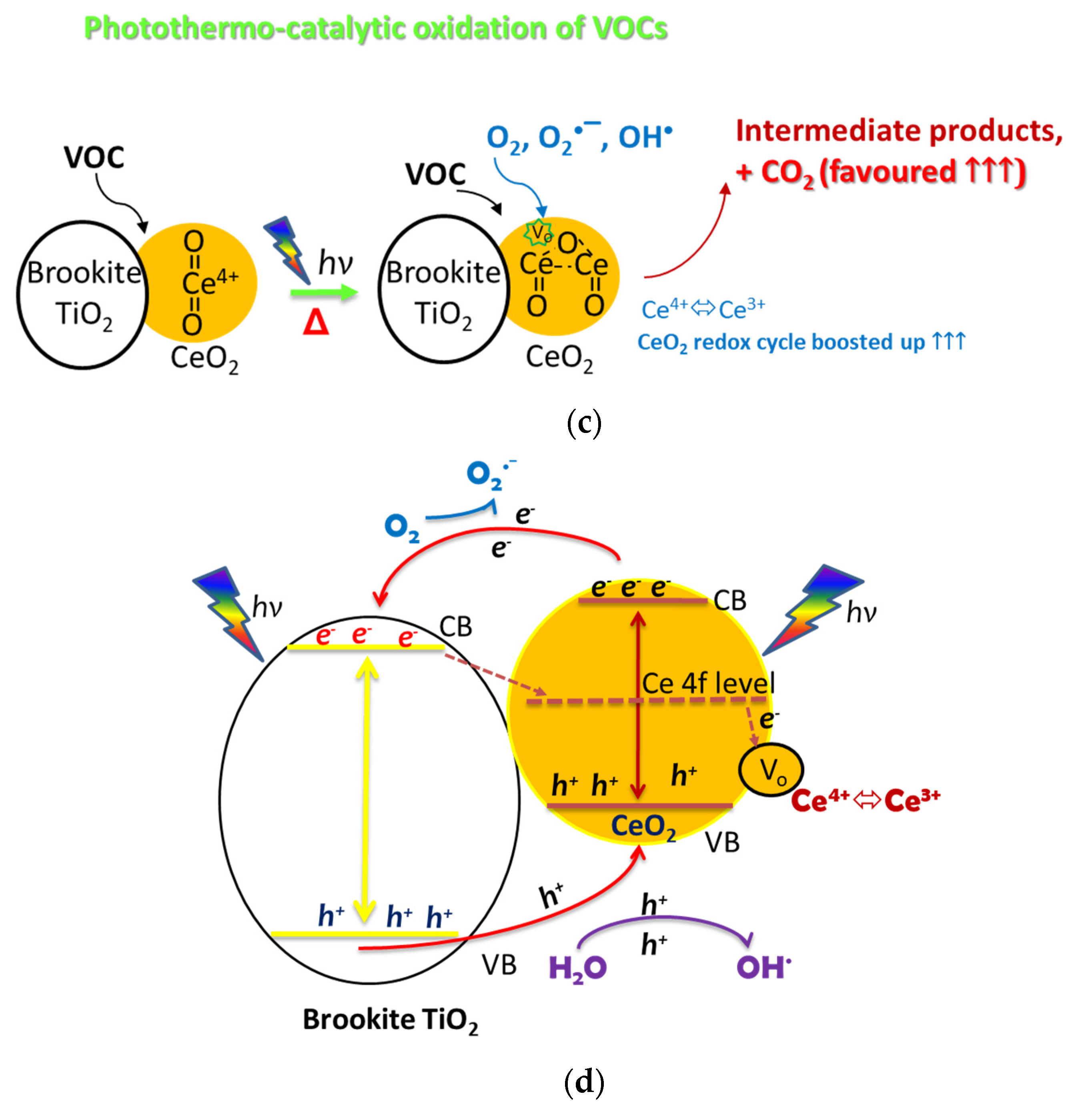
2.6. Photothermo-Catalytic Mechanisms
3. Materials and Methods
3.1. Catalysts Synthesis
3.2. Characterization Measurements
3.3. (Photo)Catalytic Measurements
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Shah, K.W.; Li, W. A review on catalytic nanomaterials for volatile organic compounds VOC removal and their applications for healthy buildings. Nanomaterials 2019, 9, 910. [Google Scholar] [CrossRef]
- Bautista-Gutierrez, K.P.; Herrera-May, A.L.; Santamaría-López, J.M.; Honorato-Moreno, A.; Zamora-Castro, S.A. Recent progress in nanomaterials for modern concrete infrastructure: Advantages and challenges. Materials (Basel) 2019, 12, 3458. [Google Scholar] [CrossRef]
- Truppi, A.; Petronella, F.; Placido, T.; Striccoli, M.; Agostiano, A.; Curri, M.L.; Comparelli, R. Visible-light-active TiO2-based hybrid nanocatalysts for environmental applications. Catalysts 2017, 7, 100. [Google Scholar] [CrossRef]
- Varma, A.; James, A.R.; Daniel, S.A. A Review on Nano TiO2—A Repellent in Paint. In Lecture Notes in Civil Engineering; Springer Nature Switzerland AG: Charm, Switzerland, 2020; Volume 46, pp. 908–919. [Google Scholar]
- Hamidi, F.; Aslani, F. TiO2-based photocatalytic cementitious composites: Materials, properties, influential parameters, and assessment techniques. Nanomaterials 2019, 9, 1444. [Google Scholar] [CrossRef]
- Enea, D.; Bellardita, M.; Scalisi, P.; Alaimo, G.; Palmisano, L. Effects of weathering on the performance of self-cleaning photocatalytic paints. Cem. Concr. Compos. 2019, 96, 77–86. [Google Scholar] [CrossRef]
- Katal, R.; Masudy-Panah, S.; Tanhaei, M.; Farahani, M.H.D.A.; Jiangyong, H. A review on the synthesis of the various types of anatase TiO2 facets and their applications for photocatalysis. Chem. Eng. J. 2020, 384, 123384. [Google Scholar] [CrossRef]
- Al Jitan, S.; Palmisano, G.; Garlisi, C. Synthesis and surface modification of TiO2-based photocatalysts for the conversion of CO2. Catalysts 2020, 10, 227. [Google Scholar] [CrossRef]
- Karthikeyan, C.; Arunachalam, P.; Ramachandran, K.; Al-Mayouf, A.M.; Karuppuchamy, S. Recent advances in semiconductor metal oxides with enhanced methods for solar photocatalytic applications. J. Alloys Compd. 2020, 828, 154281. [Google Scholar] [CrossRef]
- Fiorenza, R.; Sciré, S.; D’Urso, L.; Compagnini, G.; Bellardita, M.; Palmisano, L. Efficient H2 production by photocatalytic water splitting under UV or solar light over variously modified TiO2-based catalysts. Int. J. Hydrogen Energy 2019, 44, 14796–14807. [Google Scholar] [CrossRef]
- Fujishima, A.; Rao, T.N.; Tryk, D.A. Titanium dioxide photocatalysis. J. Photochem. Photobiol. C Photochem. Rev. 2000, 1, 1–21. [Google Scholar] [CrossRef]
- Di Paola, A.; Bellardita, M.; Palmisano, L. Brookite, the Least Known TiO2 Photocatalyst. Catalysts 2013, 3, 36–73. [Google Scholar] [CrossRef]
- Liu, L.; Zhao, H.; Andino, J.M.; Li, Y. Photocatalytic CO2 Reduction with H2O on TiO2 Nanocrystals: Comparison of Anatase, Rutile, and Brookite Polymorphs and Exploration of Surface Chemistry. ACS Catal. 2012, 2, 1817–1828. [Google Scholar] [CrossRef]
- Ramos-Delgado, N.A.; Hinojosa-Reyes, L.; Guzman-Mar, I.L.; Gracia-Pinilla, M.A.; Hernández-Ramírez, A. Synthesis by sol–gel of WO3/TiO2 for solar photocatalytic degradation of malathion pesticide. Catal. Today 2013, 209, 35–40. [Google Scholar] [CrossRef]
- Kum, J.M.; Yoo, S.H.; Ali, G.; Cho, S.O. Photocatalytic hydrogen production over CuO and TiO2 nanoparticles mixture. Int. J. Hydrogen Energy 2013, 38, 13541–13546. [Google Scholar] [CrossRef]
- Sood, S.; Mehta, S.K.; Sinha, A.S.K.; Kansal, S.K. Bi2O3/TiO2 heterostructures: Synthesis, characterization and their application in solar light mediated photocatalyzed degradation of an antibiotic, ofloxacin. Chem. Eng. J. 2016, 290, 45–52. [Google Scholar] [CrossRef]
- Bellardita, M.; Fiorenza, R.; Palmisano, L.; Scirè, S. Photocatalytic and photothermocatalytic applications of cerium oxide-based materials. In Cerium Oxide (CeO₂): Synthesis, Properties and Applications; Elsevier: Amsterdam, The Netherlands, 2020; pp. 109–167. [Google Scholar]
- Fiorenza, R.; Bellardita, M.; D’Urso, L.; Compagnini, G.; Palmisano, L.; Scirè, S. Au/TiO2-CeO2 catalysts for photocatalytic water splitting and VOCs oxidation reactions. Catalysts 2016, 6, 121. [Google Scholar] [CrossRef]
- Sciré, S.; Palmisano, L. Cerium Oxide (CeO₂): Synthesis, Properties and Applications; Elsevier: Amsterdam, The Netherlands, 2020; ISBN 9780128156612. [Google Scholar]
- Fiorenza, R.; Balsamo, S.A.; D’Urso, L.; Sciré, S.; Brundo, M.V.; Pecoraro, R.; Scalisi, E.M.; Privitera, V.; Impellizzeri, G. CeO2 for Water Remediation: Comparison of Various Advanced Oxidation Processes. Catalysts 2020, 10, 446. [Google Scholar] [CrossRef]
- Liu, B.; Zhao, X.; Zhang, N.; Zhao, Q.; He, X.; Feng, J. Photocatalytic mechanism of TiO2–CeO2 films prepared by magnetron sputtering under UV and visible light. Surf. Sci. 2005, 595, 203–211. [Google Scholar] [CrossRef]
- Galindo, F.; Gómez, R.; Aguilar, M. Photodegradation of the herbicide 2,4-dichlorophenoxyacetic acid on nanocrystalline TiO2–CeO2 sol–gel catalysts. J. Mol. Catal. A Chem. 2008, 281, 119–125. [Google Scholar] [CrossRef]
- Fiorenza, R.; Bellardita, M.; Palmisano, L.; Scirè, S. A comparison between photocatalytic and catalytic oxidation of 2-Propanol over Au/TiO2-CeO2 catalysts. J. Mol. Catal. A Chem. 2016, 415, 56–64. [Google Scholar] [CrossRef]
- Montecchio, F.; Persson, H.; Engvall, K.; Delin, J.; Lanza, R. Development of a stagnation point flow system to screen and test TiO 2 -based photocatalysts in air purification applications. Chem. Eng. J. 2016, 306, 734–744. [Google Scholar] [CrossRef]
- Boyjoo, Y.; Sun, H.; Liu, J.; Pareek, V.K.; Wang, S. A review on photocatalysis for air treatment: From catalyst development to reactor design. Chem. Eng. J. 2017, 310, 537–559. [Google Scholar] [CrossRef]
- Vikrant, K.; Park, C.M.; Kim, K.H.; Kumar, S.; Jeon, E.C. Recent advancements in photocatalyst-based platforms for the destruction of gaseous benzene: Performance evaluation of different modes of photocatalytic operations and against adsorption techniques. J. Photochem. Photobiol. C Photochem. Rev. 2019, 41, 100316. [Google Scholar] [CrossRef]
- Fiorenza, R.; Condorelli, M.; D’Urso, L.; Compagnini, G.; Bellardita, M.; Palmisano, L.; Scirè, S. Catalytic and photothermo-catalytic applications of TiO2-CoOx composites. J. Photocatal. 2020, 1, 1–13. [Google Scholar] [CrossRef]
- Di Paola, A.; Cufalo, G.; Addamo, M.; Bellardita, M.; Campostrini, R.; Ischia, M.; Ceccato, R.; Palmisano, L. Photocatalytic activity of nanocrystalline TiO2 (brookite, rutile and brookite-based) powders prepared by thermohydrolysis of TiCl4 in aqueous chloride solutions. Colloids Surfaces A Physicochem. Eng. Asp. 2008, 317, 366–376. [Google Scholar] [CrossRef]
- Bellardita, M.; Di Paola, A.; Megna, B.; Palmisano, L. Absolute crystallinity and photocatalytic activity of brookite TiO2 samples. Appl. Catal. B Environ. 2017, 201, 150–158. [Google Scholar] [CrossRef]
- Tompsett, G.A.; Bowmaker, G.A.; Cooney, R.P.; Metson, J.B.; Rodgers, K.A.; Seakins, J.M. The Raman spectrum of brookite, TiO2 (Pbca, Z = 8). J. Raman Spectrosc. 1995, 26, 57–62. [Google Scholar] [CrossRef]
- Monai, M.; Montini, T.; Fornasiero, P. Brookite: Nothing new under the sun? Catalysts 2017, 7, 304. [Google Scholar] [CrossRef]
- Fiorenza, R.; Bellardita, M.; Barakat, T.; Scirè, S.; Palmisano, L. Visible light photocatalytic activity of macro-mesoporous TiO2-CeO2 inverse opals. J. Photochem. Photobiol. A Chem. 2018, 352, 25–34. [Google Scholar] [CrossRef]
- Wang, Y.; Zou, Y.; Shang, Q.; Tan, X.; Yu, T.; Huang, X.; Shi, W.; Xie, Y.; Yan, G.; Wang, X. Quasi-Spherical Brookite TiO2 Nanostructures Synthesized Using Solvothermal Method in the Presence of Oxalic Acid. Trans. Tianjin Univ. 2018, 24, 326–339. [Google Scholar] [CrossRef]
- Su, W.; Zhang, J.; Feng, Z.; Chen, T.; Ying, P.; Li, C. Surface Phases of TiO 2 Nanoparticles Studied by UV Raman Spectroscopy and FT-IR Spectroscopy. J. Phys. Chem. C 2008, 112, 7710–7716. [Google Scholar] [CrossRef]
- Choudhury, B.; Borah, B.; Choudhury, A. Ce–Nd codoping effect on the structural and optical properties of TiO2 nanoparticles. Mater. Sci. Eng. B 2013, 178, 239–247. [Google Scholar] [CrossRef]
- Zeng, M.; Li, Y.; Mao, M.; Bai, J.; Ren, L.; Zhao, X. Synergetic Effect between Photocatalysis on TiO2 and Thermocatalysis on CeO2 for Gas-Phase Oxidation of Benzene on TiO2/CeO2 Nanocomposites. ACS Catal. 2015, 5, 3278–3286. [Google Scholar] [CrossRef]
- Pigeot-Rémy, S.; Gregori, D.; Hazime, R.; Hérissan, A.; Guillard, C.; Ferronato, C.; Cassaignon, S.; Colbeau-Justin, C.; Durupthy, O. Size and shape effect on the photocatalytic efficiency of TiO2 brookite. J. Mater. Sci. 2019, 54, 1213–1225. [Google Scholar] [CrossRef]
- Sing, K.S.W. Reporting physisorption data for gas/solid systems with special reference to the determination of surface area and porosity (Recommendations 1984). Pure Appl. Chem. 1985, 57, 603–619. [Google Scholar] [CrossRef]
- Shen, X.; Tian, B.; Zhang, J. Tailored preparation of titania with controllable phases of anatase and brookite by an alkalescent hydrothermal route. Catal. Today 2013, 201, 151–158. [Google Scholar] [CrossRef]
- Wei, X.; Lü, X.; Wu, Z.; Ding, J.; Xie, J.; Wei, W.; Mao, D. Reactivity and mechanistic insight into visible-light-induced anerobic selective reaction by Ag/brookite titania. J. Nanosci. Nanotechnol. 2012, 12, 8017–8022. [Google Scholar] [CrossRef]
- Asadollahi, E.; Youzbashi, A.A.; Keyanpour-Rad, M. Synthesis and investigation of crystal structure and optical properties of brookite TiO2 nanoparticles capped with (2-chloroquinoline-3-yl) methanol. J. Mol. Struct. 2017, 1128, 612–618. [Google Scholar] [CrossRef]
- Yamashita, H.; Harada, M.; Misaka, J.; Takeuchi, M.; Neppolian, B.; Anpo, M. Photocatalytic degradation of organic compounds diluted in water using visible light-responsive metal ion-implanted TiO2 catalysts: Fe ion-implanted TiO2. Catal. Today 2003, 84, 191–196. [Google Scholar] [CrossRef]
- Yu, J.; Yue, L.; Liu, S.; Huang, B.; Zhang, X. Hydrothermal preparation and photocatalytic activity of mesoporous Au–TiO2 nanocomposite microspheres. J. Colloid Interface Sci. 2009, 334, 58–64. [Google Scholar] [CrossRef]
- Liqiang, J.; Yichun, Q.; Baiqi, W.; Shudan, L.; Baojiang, J.; Libin, Y.; Wei, F.; Honggang, F.; Jiazhong, S. Review of photoluminescence performance of nano-sized semiconductor materials and its relationships with photocatalytic activity. Sol. Energy Mater. Sol. Cells 2006, 90, 1773–1787. [Google Scholar] [CrossRef]
- Xie, J.; Lü, X.; Liu, J.; Shu, H. Brookite titania photocatalytic nanomaterials: Synthesis, properties, and applications. Pure Appl. Chem. 2009, 81, 2407–2415. [Google Scholar] [CrossRef]
- Lei, Y.; Zhang, L.D.; Meng, G.W.; Li, G.H.; Zhang, X.Y.; Liang, C.H.; Chen, W.; Wang, S.X. Preparation and photoluminescence of highly ordered TiO2 nanowire arrays. Appl. Phys. Lett. 2001, 78, 1125–1127. [Google Scholar] [CrossRef]
- Gao, X.; Jiang, Y.; Zhong, Y.; Luo, Z.; Cen, K. The activity and characterization of CeO2-TiO2 catalysts prepared by the sol–gel method for selective catalytic reduction of NO with NH3. J. Hazard. Mater. 2010, 174, 734–739. [Google Scholar] [CrossRef] [PubMed]
- Fang, J.; Bi, X.; Si, D.; Jiang, Z.; Huang, W. Spectroscopic studies of interfacial structures of CeO2–TiO2 mixed oxides. Appl. Surf. Sci. 2007, 253, 8952–8961. [Google Scholar] [CrossRef]
- Gulino, A.; Taverner, A.E.; Warren, S.; Harris, P.; Egdell, R.G. A photoemission study of Sb-doped TiO2. Surf. Sci. 1994, 315, 351–361. [Google Scholar] [CrossRef]
- Qiu, L.; Liu, F.; Zhao, L.; Ma, Y.; Yao, J. Comparative XPS study of surface reduction for nanocrystalline and microcrystalline ceria powder. Appl. Surf. Sci. 2006, 252, 4931–4935. [Google Scholar] [CrossRef]
- Bêche, E.; Charvin, P.; Perarnau, D.; Abanades, S.; Flamant, G. Ce 3d XPS investigation of cerium oxides and mixed cerium oxide (CexTiyOz). Surf. Interface Anal. 2008, 40, 264–267. [Google Scholar] [CrossRef]
- Ji, P.; Zhang, J.; Chen, F.; Anpo, M. Ordered Mesoporous CeO2 Synthesized by Nanocasting from Cubic Ia3d Mesoporous MCM-48 Silica: Formation, Characterization and Photocatalytic Activity. J. Phys. Chem. C 2008, 112, 17809–17813. [Google Scholar] [CrossRef]
- Watanabe, S.; Ma, X.; Song, C. Characterization of Structural and Surface Properties of Nanocrystalline TiO2−CeO 2 Mixed Oxides by XRD, XPS, TPR, and TPD. J. Phys. Chem. C 2009, 113, 14249–14257. [Google Scholar] [CrossRef]
- Eriksen, S.; Egdell, R.G. Electronic excitations at oxygen deficient TiO2(110) surfaces: A study by EELS. Surf. Sci. 1987, 180, 263–278. [Google Scholar] [CrossRef]
- Bharti, B.; Kumar, S.; Lee, H.-N.; Kumar, R. Formation of oxygen vacancies and Ti3+ state in TiO2 thin film and enhanced optical properties by air plasma treatment. Sci. Rep. 2016, 6, 32355. [Google Scholar] [CrossRef] [PubMed]
- Cafun, J.-D.; Kvashnina, K.O.; Casals, E.; Puntes, V.F.; Glatzel, P. Absence of Ce 3+ Sites in Chemically Active Colloidal Ceria Nanoparticles. ACS Nano 2013, 7, 10726–10732. [Google Scholar] [CrossRef]
- Tanner, P.A.; Mak, C.S.K.; Edelstein, N.M.; Murdoch, K.M.; Liu, G.; Huang, J.; Seijo, L.; Barandiarán, Z. Absorption and Emission Spectra of Ce3+ in Elpasolite Lattices. J. Am. Chem. Soc. 2003, 125, 13225–13233. [Google Scholar] [CrossRef] [PubMed]
- Holz, M.C.; Kähler, K.; Tölle, K.; van Veen, A.C.; Muhler, M. Gas-phase oxidation of 2-propanol over Au/TiO2 catalysts to probe metal-support interactions. Phys. Status Solidi 2013, 250, 1094–1106. [Google Scholar] [CrossRef]
- Sboui, M.; Bouattour, S.; Liotta, L.F.; La Parola, V.; Gruttadauria, M.; Marcì, G.; Boufi, S. Paper-TiO2 composite: An effective photocatalyst for 2-propanol degradation in gas phase. J. Photochem. Photobiol. A Chem. 2018, 350, 142–151. [Google Scholar] [CrossRef]
- García-López, E.I.; Marcì, G.; Dozzi, M.V.; Palmisano, L.; Selli, E. Photoactivity of shape-controlled TiO2 in gas-solid regime under solar irradiation. Catal. Today 2019, 328, 118–124. [Google Scholar] [CrossRef]
- Godoi, D.R.M.; Villullas, H.M.; Zhu, F.-C.; Jiang, Y.-X.; Sun, S.-G.; Guo, J.; Sun, L.; Chen, R. A comparative investigation of metal-support interactions on the catalytic activity of Pt nanoparticles for ethanol oxidation in alkaline medium. J. Power Sources 2016, 311, 81–90. [Google Scholar] [CrossRef]
- Liu, S.Y.; Yang, S.M. Complete oxidation of 2-propanol over gold-based catalysts supported on metal oxides. Appl. Catal. A Gen. 2008, 334, 92–99. [Google Scholar] [CrossRef]
- Haffad, D.; Chambellan, A.; Lavalley, J.C. Propan-2-ol transformation on simple metal oxides TiO2, ZrO2 and CeO2. J. Mol. Catal. A Chem. 2001, 168, 153–164. [Google Scholar] [CrossRef]
- Fiorenza, R.; Bellardita, M.; Scirè, S.; Palmisano, L. Effect of the addition of different doping agents on visible light activity of porous TiO2 photocatalysts. Mol. Catal. 2018, 455, 108–120. [Google Scholar] [CrossRef]
- O’Malley, A.; Hodnett, B.K. The influence of volatile organic compound structure on conditions required for total oxidation. Catal. Today 1999, 54, 31–38. [Google Scholar] [CrossRef]
- Baldi, M.; Finocchio, E.; Milella, F.; Busca, G. Catalytic combustion of C3 hydrocarbons and oxygenates over Mn3O4. Appl. Catal. B Environ. 1998, 16, 43–51. [Google Scholar] [CrossRef]
- Scirè, S.; Minicò, S.; Crisafulli, C.; Satriano, C.; Pistone, A. Catalytic combustion of volatile organic compounds on gold/cerium oxide catalysts. Appl. Catal. B Environ. 2003, 40, 43–49. [Google Scholar] [CrossRef]
- Maira, A.J.; Yeung, K.L.; Soria, J.; Coronado, J.M.; Belver, C.; Lee, C.Y.; Augugliaro, V. Gas-phase photo-oxidation of toluene using nanometer-size TiO2 catalysts. Appl. Catal. B Environ. 2001, 29, 327–336. [Google Scholar] [CrossRef]
- Augugliaro, V.; Coluccia, S.; Loddo, V.; Marchese, L.; Martra, G.; Palmisano, L.; Schiavello, M. Photocatalytic oxidation of gaseous toluene on anatase TiO2 catalyst: Mechanistic aspects and FT-IR investigation. Appl. Catal. B Environ. 1999, 20, 15–27. [Google Scholar] [CrossRef]
- Zhang, L.; Menendez-Flores, V.M.; Murakami, N.; Ohno, T. Improvement of photocatalytic activity of brookite titanium dioxide nanorods by surface modification using chemical etching. Appl. Surf. Sci. 2012, 258, 5803–5809. [Google Scholar] [CrossRef]
- Li, J.-J.; Yu, E.-Q.; Cai, S.-C.; Chen, X.; Chen, J.; Jia, H.-P.; Xu, Y.-J. Noble metal free, CeO2/LaMnO3 hybrid achieving efficient photo-thermal catalytic decomposition of volatile organic compounds under IR light. Appl. Catal. B Environ. 2019, 240, 141–152. [Google Scholar] [CrossRef]
- Kong, J.; Xiang, Z.; Li, G.; An, T. Introduce oxygen vacancies into CeO2 catalyst for enhanced coke resistance during photothermocatalytic oxidation of typical VOCs. Appl. Catal. B Environ. 2020, 269, 118755. [Google Scholar] [CrossRef]
- Shayegan, Z.; Lee, C.-S.; Haghighat, F. TiO2 photocatalyst for removal of volatile organic compounds in gas phase—A review. Chem. Eng. J. 2018, 334, 2408–2439. [Google Scholar] [CrossRef]
- Huang, Y.; Ho, S.; Lu, Y.; Niu, R.; Xu, L.; Cao, J.; Lee, S. Removal of Indoor Volatile Organic Compounds via Photocatalytic Oxidation: A Short Review and Prospect. Molecules 2016, 21, 56. [Google Scholar] [CrossRef] [PubMed]
- Koli, V.B.; Kim, J.-S. Photocatalytic oxidation for removal of gases toluene by TiO2-CeO2 nanocomposites under UV light irradiation. Mater. Sci. Semicond. Process. 2019, 94, 70–79. [Google Scholar] [CrossRef]
- Muñoz-Batista, M.J.; Gómez-Cerezo, M.N.; Kubacka, A.; Tudela, D.; Fernández-García, M. Role of interface contact in CeO2-TiO2 photocatalytic composite materials. ACS Catal. 2014, 4, 63–72. [Google Scholar] [CrossRef]
- Yamakata, A.; Vequizo, J.J.M. Curious behaviors of photogenerated electrons and holes at the defects on anatase, rutile, and brookite TiO2 powders: A review. J. Photochem. Photobiol. C Photochem. Rev. 2019, 40, 234–243. [Google Scholar] [CrossRef]
- Scirè, S.; Riccobene, P.M.; Crisafulli, C. Ceria supported group IB metal catalysts for the combustion of volatile organic compounds and the preferential oxidation of CO. Appl. Catal. B Environ. 2010, 101, 109–117. [Google Scholar] [CrossRef]
- Scirè, S.; Fiorenza, R.; Gulino, A.; Cristaldi, A.; Riccobene, P.M. Selective oxidation of CO in H2-rich stream over ZSM5 zeolites supported Ru catalysts: An investigation on the role of the support and the Ru particle size. Appl. Catal. A Gen. 2016, 520, 82–91. [Google Scholar] [CrossRef]
- Fiorenza, R.; Spitaleri, L.; Gulino, A.; Scirè, S. Ru–Pd bimetallic catalysts supported on CeO2-MnOx oxides as efficient systems for H2 purification through CO preferential Oxidation. Catalysts 2018, 8, 203. [Google Scholar] [CrossRef]
- Gulino, A. Structural and electronic characterization of self-assembled molecular nanoarchitectures by X-ray photoelectron spectroscopy. Anal. Bioanal. Chem. 2013, 405, 1479–1495. [Google Scholar] [CrossRef]
- Cantarella, M.; Di Mauro, A.; Gulino, A.; Spitaleri, L.; Nicotra, G.; Privitera, V.; Impellizzeri, G. Selective photodegradation of paracetamol by molecularly imprinted ZnO nanonuts. Appl. Catal. B Environ. 2018, 238, 509–517. [Google Scholar] [CrossRef]


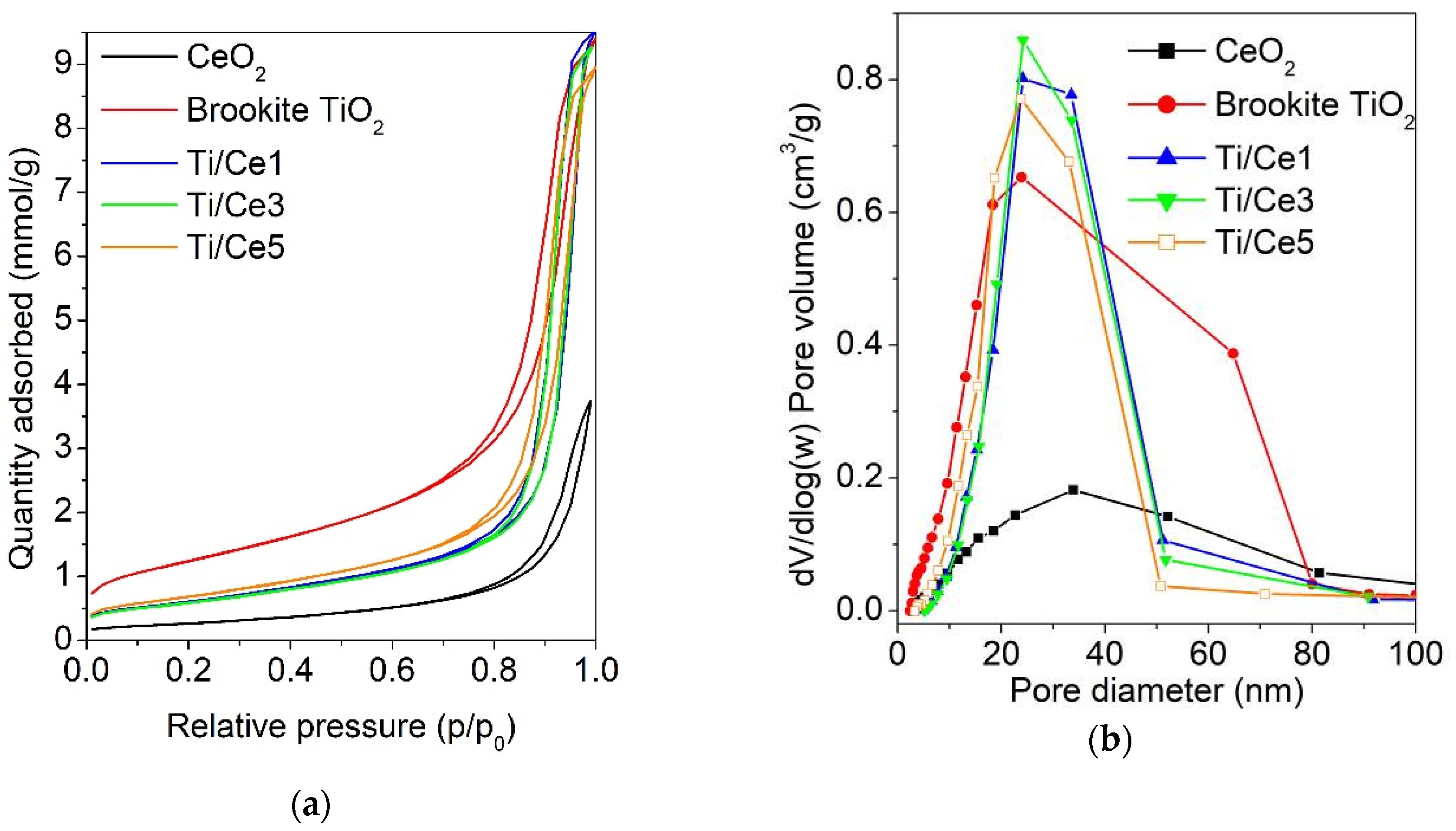
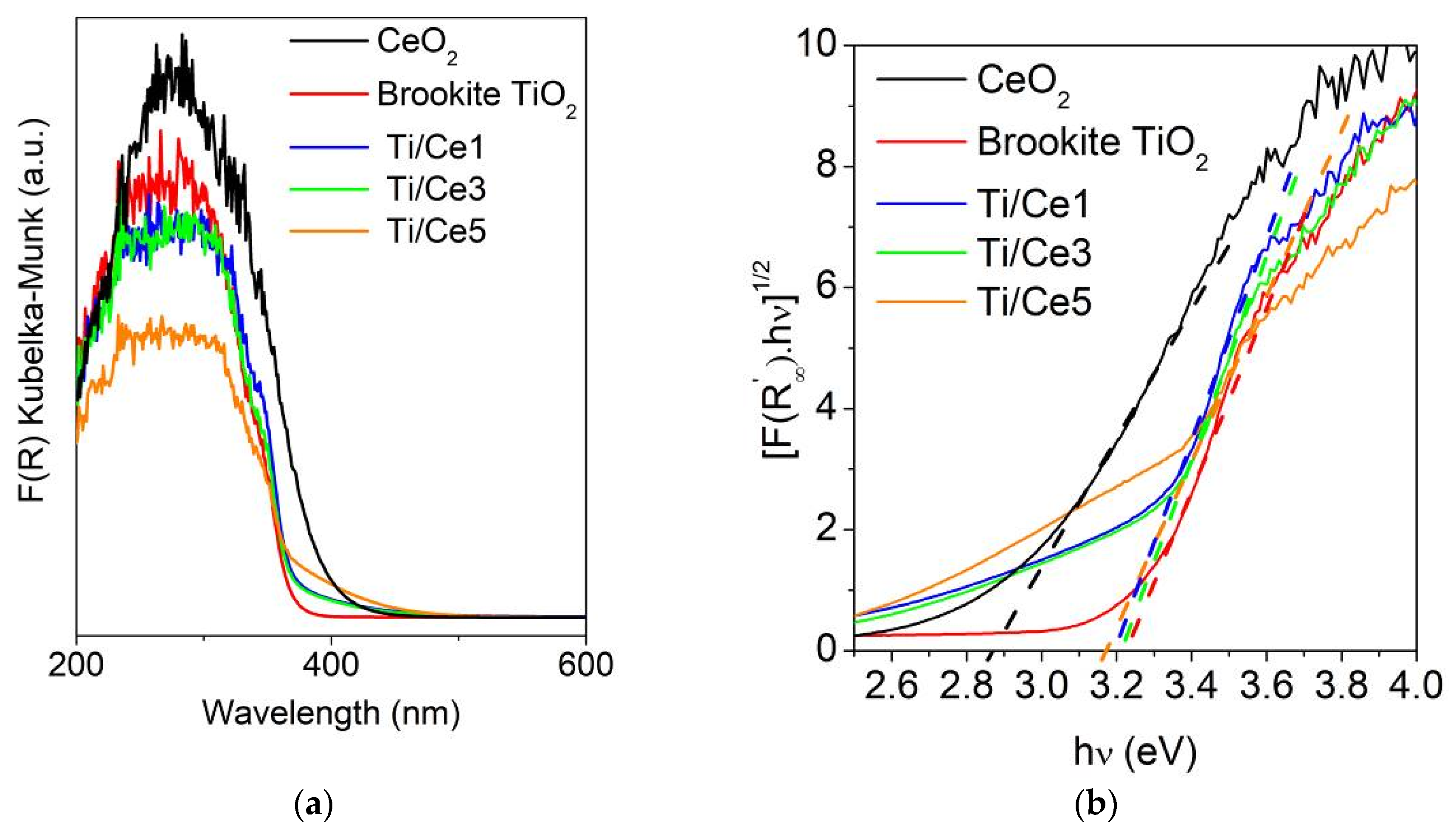




| Samples | SA (m2/g) | dp (nm) | vp (cm3/g) | Eg (eV) |
|---|---|---|---|---|
| CeO2 | 60 | 32.1 | 0.14 | 2.87 |
| Brookite TiO2 | 100 | 27.3 | 0.33 | 3.22 |
| Ti/Ce1 | 69 | 24.9 | 0.35 | 3.20 |
| Ti/Ce3 | 68 | 25.2 | 0.34 | 3.21 |
| Ti/Ce5 | 66 | 24.7 | 0.33 | 3.19 |
| Samples | O 1s | Ti 2p3/2,1/2 | Ce 3d5/2,3/2 |
|---|---|---|---|
| CeO2 | 529.4, 531.6 | / | 882.9–901.5, 889.2–907.8, 898.8–917.3 |
| Brookite TiO2 | 530.8 | 459.3–465.1 | / |
| Ti/Ce1 | 530.5 | 459.0–464.8 | 883.2–901.8, 886.5–905.1, 917.5 |
| Ti/Ce3 | 530.5 | 459.0–464.8 | 883.2–901.8, 886.5–905.1, 917.5 |
| Ti/Ce5 | 530.5 | 459.0–464.8 | 883.2–901.8, 886.5–905.1, 917.5 |
| Samples | 2-Propanol Conversion | Conversion to CO2 | Maximum Conversion to Acetone |
|---|---|---|---|
| CeO2 | T10 = 293 °C | T10 = 336 °C | 39% (365 °C) |
| T50 = 342 °C | T50 = 386 °C | ||
| T90 = 417 °C | T90 = 434 °C | ||
| Brookite TiO2 | T10 = 209 °C | T10 = 271 °C | 93% (261 °C) |
| T50 = 226 °C | T50 = 294 °C | ||
| T90 = 258 °C | T90 = 323 °C | ||
| Ti/Ce1 | T10 = 195 °C | T10 = 253 °C | 79% (248 °C) |
| T50 = 222 °C | T50 = 277 °C | ||
| T90 = 256 °C | T90 = 306 °C | ||
| Ti/Ce3 | T10 = 194 °C | T10 = 212 °C | 47% (248 °C) |
| T50 = 213 °C | T50 = 260 °C | ||
| T90 = 253 °C | T90 = 294 °C | ||
| Ti/Ce5 | T10 = 176 °C | T10 = 199 °C | 41% (221 °C) |
| T50 = 208 °C | T50 = 238 °C | ||
| T90 = 257 °C | T90 = 281 °C |
| Samples | 2-Propanol Conversion | Conversion to CO2 | Maximum Conversion to Acetone |
|---|---|---|---|
| CeO2 | T10 = 281 °C | T10 = 374 °C | 52% (367 °C) |
| T50 = 348 °C | T50 = 403 °C | ||
| T90 = 407 °C | T90 = 416 °C | ||
| Brookite TiO2 | T10 = 170 °C | T10 = 260 °C | 96% (235 °C) |
| T50 = 192 °C | T50 = 282 °C | ||
| T90 = 218 °C | T90 = 303 °C | ||
| Ti/Ce1 | T10 = 162 °C | T10 = 240 °C | 97% (225 °C) |
| T50 = 178 °C | T50 = 265 °C | ||
| T90 = 208 °C | T90 = 287 °C | ||
| Ti/Ce3 | T10 = 130 °C | T10 = 172 °C | 61% (180 °C) |
| T50 = 152 °C | T50 = 200 °C | ||
| T90 = 196 °C | T90 = 224 °C | ||
| Ti/Ce5 | T10 = 114 °C | T10 = 166 °C | 57% (165 °C) |
| T50 = 151 °C | T50 = 187 °C | ||
| T90 = 199 °C | T90 = 214 °C |
| Samples | Ethanol Conversion | Conversion to CO2 | Maximum Conversion to Acetaldehyde |
|---|---|---|---|
| CeO2 | T10 = 299 °C | T10 = 342 °C | 38% (375 °C) |
| T50 = 347 °C | T50 = 393 °C | ||
| T90 = 426 °C | T90 = 440 °C | ||
| Brookite TiO2 | T10 = 244 °C | T10 = 279 °C | 69% (271 °C) |
| T50 = 258 °C | T50 = 303 °C | ||
| T90 = 290 °C | T90 = 330 °C | ||
| Ti/Ce1 | T10 = 222 °C | T10 = 273 °C | 71% (268 °C) |
| T50 = 248 °C | T50 = 298 °C | ||
| T90 = 284 °C | T90 = 327 °C | ||
| Ti/Ce3 | T10 = 218 °C | T10 = 232 °C | 44% (271 °C) |
| T50 = 237 °C | T50 = 282 °C | ||
| T90 = 274 °C | T90 = 315 °C | ||
| Ti/Ce5 | T10 = 187 °C | T10 = 208 °C | 40% (232 °C) |
| T50 = 216 °C | T50 = 249 °C | ||
| T90 = 270 °C | T90 = 292 °C |
| Samples | Ethanol Conversion | Conversion to CO2 | Maximum Conversion to Acetaldehyde |
|---|---|---|---|
| CeO2 | T10 = 294 °C | T10 = 337 °C | 40% (360 °C) |
| T50 = 343 °C | T50 = 386 °C | ||
| T90 = 418 °C | T90 = 435 °C | ||
| Brookite TiO2 | T10 = 221 °C | T10 = 256 °C | 68% (253 °C) |
| T50 = 236 °C | T50 = 279 °C | ||
| T90 = 268 °C | T90 = 307 °C | ||
| Ti/Ce1 | T10 = 198 °C | T10 = 249 °C | 72% (244 °C) |
| T50 = 224 °C | T50 = 273 °C | ||
| T90 = 259 °C | T90 = 302 °C | ||
| Ti/Ce3 | T10 = 178 °C | T10 = 192 °C | 43% (228 °C) |
| T50 = 197 °C | T50 = 242 °C | ||
| T90 = 236 °C | T90 = 275 °C | ||
| Ti/Ce5 | T10 = 145 °C | T10 = 167 °C | 39% (190 °C) |
| T50 = 174 °C | T50 = 207 °C | ||
| T90 = 226 °C | T90 = 250 °C |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Bellardita, M.; Fiorenza, R.; D'Urso, L.; Spitaleri, L.; Gulino, A.; Compagnini, G.; Scirè, S.; Palmisano, L. Exploring the Photothermo-Catalytic Performance of Brookite TiO2-CeO2 Composites. Catalysts 2020, 10, 765. https://doi.org/10.3390/catal10070765
Bellardita M, Fiorenza R, D'Urso L, Spitaleri L, Gulino A, Compagnini G, Scirè S, Palmisano L. Exploring the Photothermo-Catalytic Performance of Brookite TiO2-CeO2 Composites. Catalysts. 2020; 10(7):765. https://doi.org/10.3390/catal10070765
Chicago/Turabian StyleBellardita, Marianna, Roberto Fiorenza, Luisa D'Urso, Luca Spitaleri, Antonino Gulino, Giuseppe Compagnini, Salvatore Scirè, and Leonardo Palmisano. 2020. "Exploring the Photothermo-Catalytic Performance of Brookite TiO2-CeO2 Composites" Catalysts 10, no. 7: 765. https://doi.org/10.3390/catal10070765
APA StyleBellardita, M., Fiorenza, R., D'Urso, L., Spitaleri, L., Gulino, A., Compagnini, G., Scirè, S., & Palmisano, L. (2020). Exploring the Photothermo-Catalytic Performance of Brookite TiO2-CeO2 Composites. Catalysts, 10(7), 765. https://doi.org/10.3390/catal10070765