MOF Encapsulation of Ru Olefin Metathesis Catalysts to Block Catalyst Decomposition
Abstract
1. Introduction
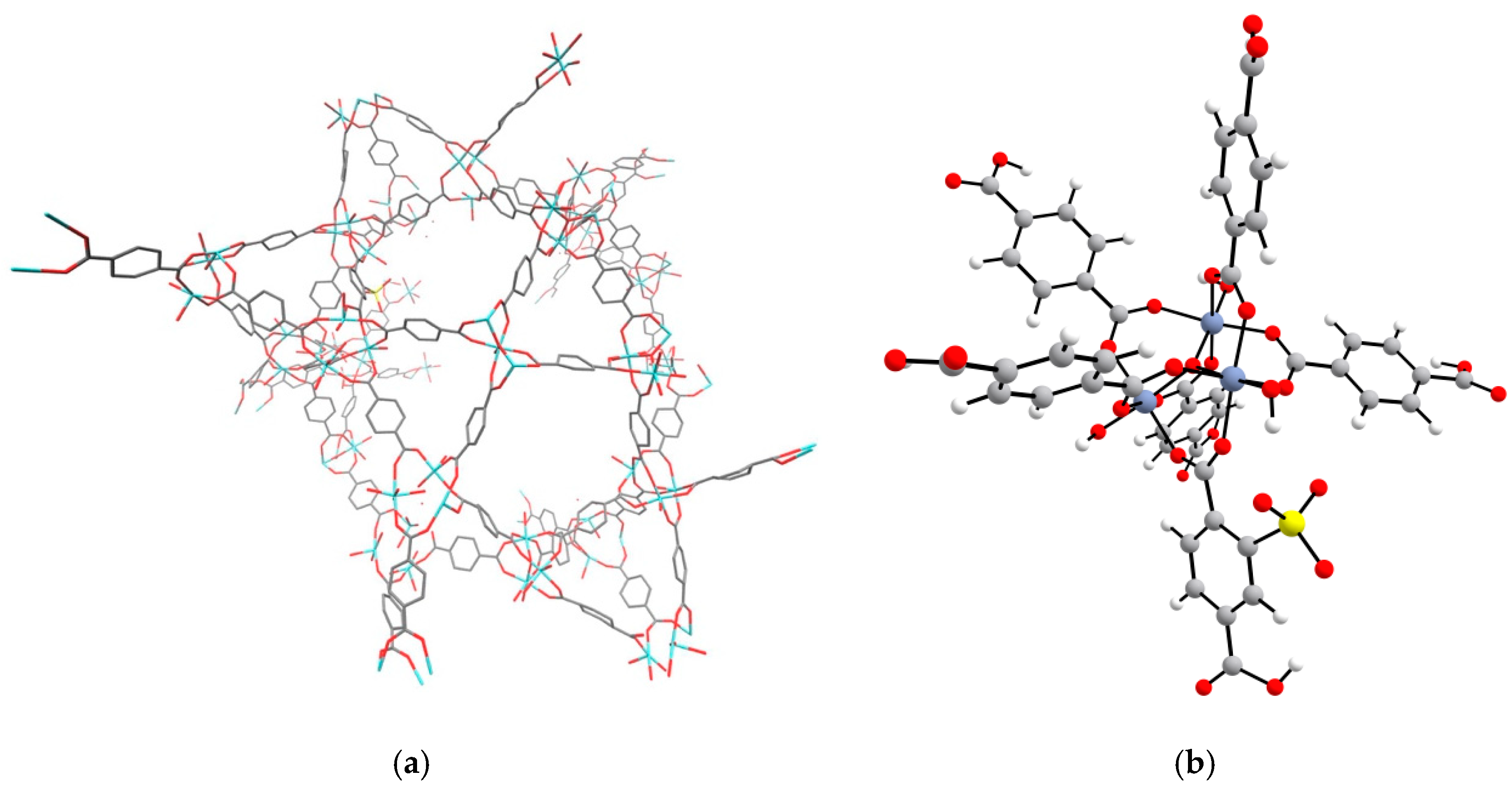
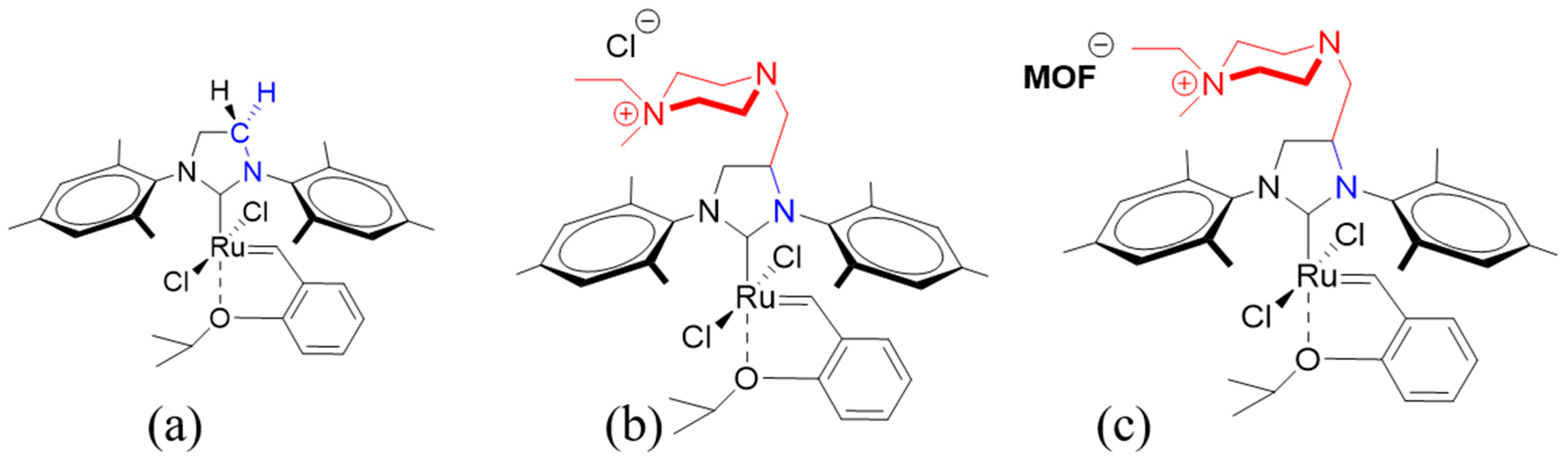
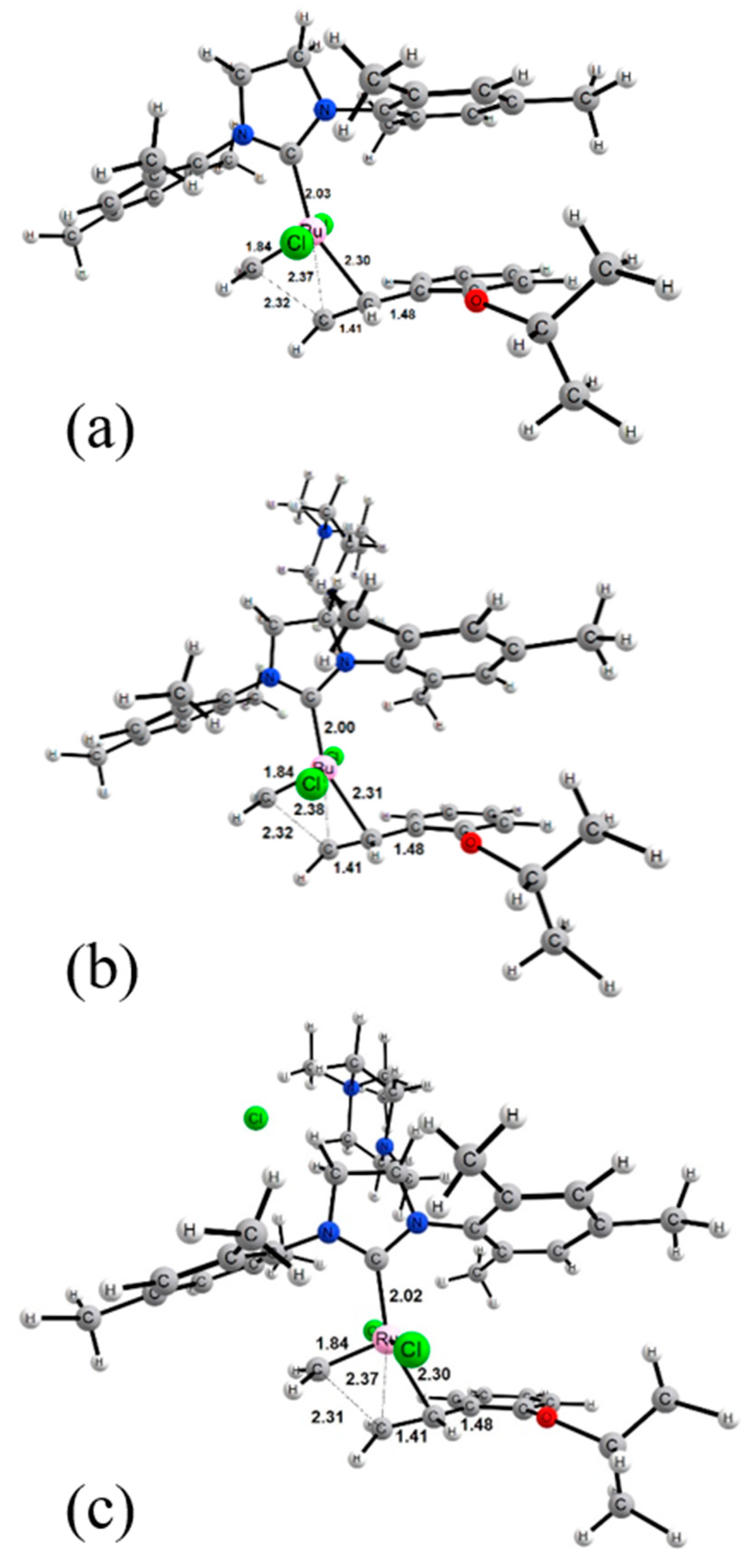
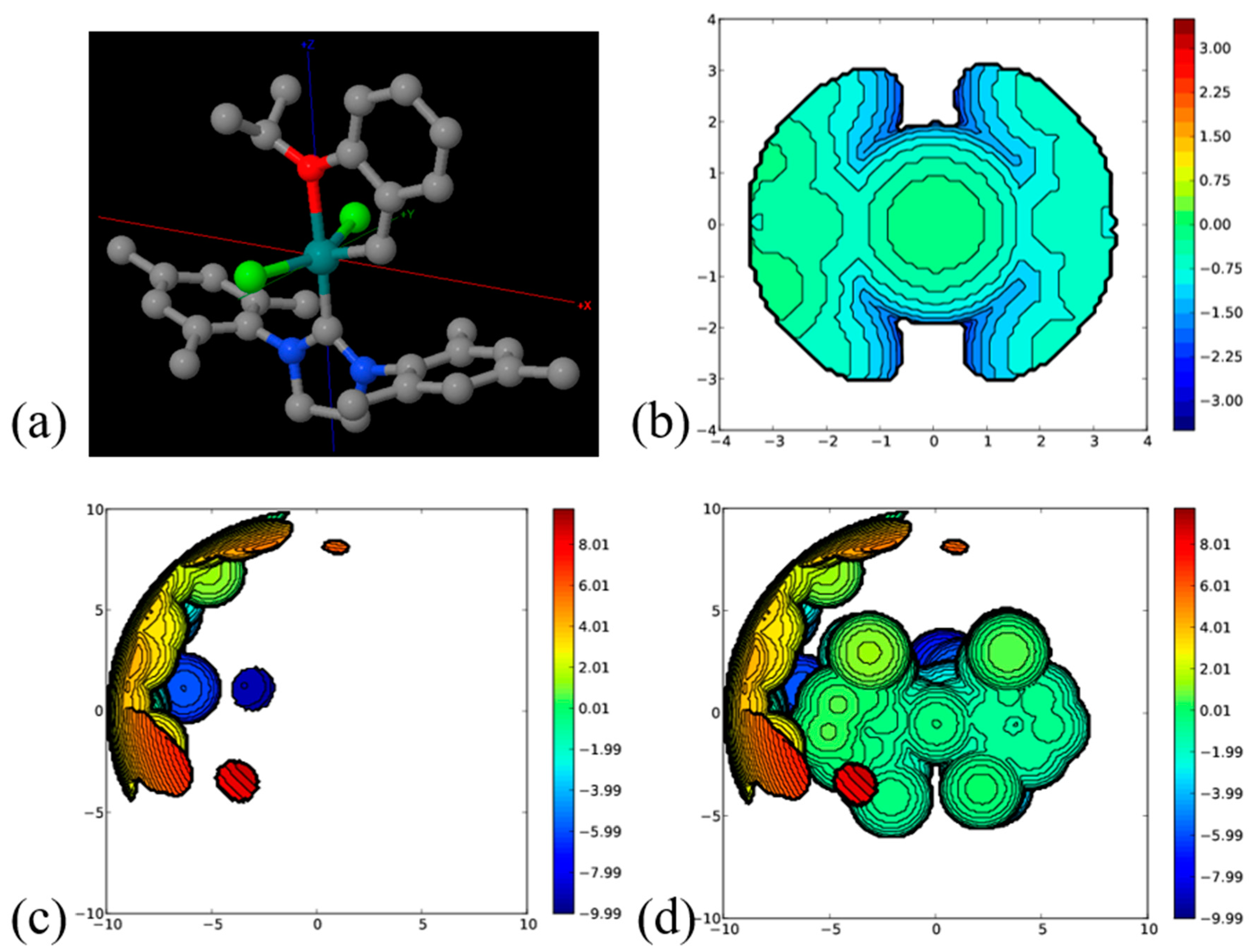
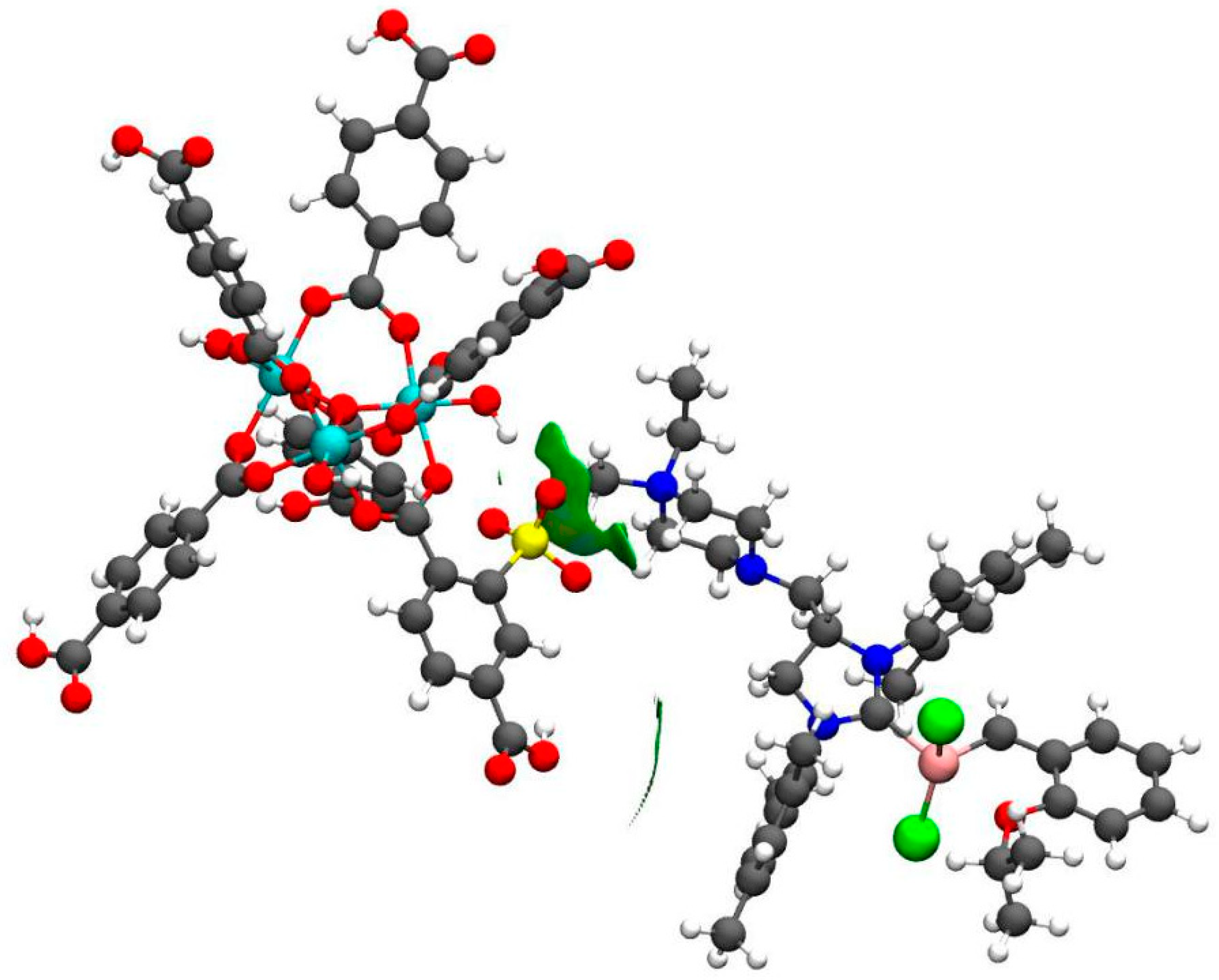
2. Results and Discussion
3. Conclusions
4. Computational Details
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Sytniczuk, A.; Forcher, G.; Grotjahn, D.B.; Grela, K. Sequential alkene isomerization and ring-closing metathesis in production of Macrocyclic Musks from biomass. Chem. Eur. J. 2018, 24, 10403–10408. [Google Scholar] [CrossRef] [PubMed]
- Rouen, M.; Queval, P.; Borré, E.; Falivene, L.; Poater, A.; Berthod, M.; Hugues, F.; Cavallo, L.; Baslé, O.; Olivier-Bourbigou, H.; et al. Selective metathesis of α-olefins from bio-sourced Fischer–Tropsch feeds. ACS Catal. 2016, 6, 7970–7976. [Google Scholar] [CrossRef]
- Sytniczuk, A.; Dąbrowski, M.; Banach, L.; Urban, M.; Czarnocka-Śniadała, S.; Milewski, M.; Kajetanowicz, A.; Grela, K. At long last: Olefin metathesis macrocyclization at high concentration. J. Am. Chem. Soc. 2018, 140, 8895–8901. [Google Scholar] [CrossRef] [PubMed]
- Grubbs, R.H. Handbook of Olefin Metathesis; Wiley-VCH: Weinheim, Germany, 2003. [Google Scholar]
- Samojłowicz, C.; Bieniek, M.; Grela, K. Ruthenium-based olefin metathesis catalysts bearing N-Heterocyclic Carbene Ligands. Chem. Rev. 2009, 109, 3708–3742. [Google Scholar] [CrossRef] [PubMed]
- Vougioukalakis, G.C.; Grubbs, R.H. Ruthenium-based heterocyclic carbene-coordinated olefin metathesis catalysts. Chem. Rev. 2010, 110, 1746–1787. [Google Scholar] [CrossRef]
- Ogba, O.M.; Warner, N.C.; O’Leary, D.J.; Grubbs, R.H. Recent advances in ruthenium-based olefin metathesis. Chem. Soc. Rev. 2019, 47, 4510–4544. [Google Scholar] [CrossRef]
- Poater, A.; Cavallo, L. A Comprehensive study of olefin metathesis catalyzed by Ru-based catalysts. Beilstein J. Org. Chem. 2015, 11, 1767–1780. [Google Scholar] [CrossRef]
- Grela, K.; Kajetanowicz, A. Progress in metathesis chemistry. Beilstein J. Org. Chem. 2019, 15, 2765–2766. [Google Scholar] [CrossRef]
- Bailey, G.A.; Foscato, M.; Higman, C.S.; Day, C.; Jensen, V.R.; Fogg, D.E. Bimolecular coupling as a vector for decomposition of fast-initiating olefin metathesis catalysts. J. Am. Chem. Soc. 2018, 140, 6931–6944. [Google Scholar] [CrossRef]
- Van Rensburg, W.J.; Steynberg, P.J.; Meyer, W.H.; Kirk, M.M.; Forman, G.S. DFT Prediction and experimental observation of substrate-induced catalyst decomposition in ruthenium-catalyzed olefin metathesis. J. Am. Chem. Soc. 2004, 126, 14332–14333. [Google Scholar] [CrossRef]
- Poater, A.; Bahri-Laleh, N.; Cavallo, L. Rationalizing current strategies to protect n-heterocyclic Carbene-based ruthenium catalysts active in olefin metathesis from C–H (De)Activation. Chem. Commun. 2011, 47, 6674–6676. [Google Scholar] [CrossRef] [PubMed]
- Manzini, S.; Poater, A.; Nelson, D.J.; Cavallo, L.; Slawin, A.M.Z.; Nolan, S.P. Insights into the decomposition of olefin metathesis pre-catalysts. Angew. Chem. Int. Ed. 2014, 53, 8995–8999. [Google Scholar] [CrossRef] [PubMed]
- Pump, E.; Poater, A.; Bahri-Laleh, N.; Credendino, R.; Serra, L.; Scarano, V.; Cavallo, L. Regio, Stereo and chemoselectivity of 2nd generation grubbs ruthenium-catalyzed olefin metathesis. Catal. Today 2020, in press. [Google Scholar] [CrossRef]
- Ahmed, T.S.; Montgomery, T.P.; Grubbs, R.H. Using stereoretention for the synthesis of E-Macrocycles with ruthenium-based olefin metathesis catalysts. Chem. Sci. 2018, 9, 3580–3583. [Google Scholar] [CrossRef] [PubMed]
- Nechmad, N.B.; Phatake, R.; Ivry, E.; Poater, A.; Lemcoff, N. Unprecedented selectivity of ruthenium iodide Benzylidenes in olefin metathesis reactions. Angew. Chem. Int. Ed. 2020, 59, 3539–3543. [Google Scholar] [CrossRef] [PubMed]
- Jana, A.; Grela, K. Forged and fashioned for faithfulness—Ruthenium olefin metathesis catalysts bearing ammonium tags. Chem. Commun. 2018, 54, 122–139. [Google Scholar] [CrossRef]
- Mauduit, M.; Schmid, T.E.; Dumas, A.; Colombel-Rouen, S.; Crévisy, C.; Baslé, O. From environmentally friendly reusable ionic-tagged ruthenium-based complexes to industrially relevant homogeneous catalysts: Toward a sustainable olefin metathesis. Synlett 2017, 28, 773–798. [Google Scholar] [CrossRef]
- Patrzałek, M.; Piątkowski, J.; Kajetanowicz, A.; Grela, K. Anion metathesis in facile preparation of olefin metathesis catalysts bearing a quaternary ammonium chloride tag. Synlett 2019, 30, 1981–1987. [Google Scholar] [CrossRef]
- Kluciar, M.; Grela, K.; Mauduit, M. Ruthenium-based complexes containing a benzimidazolium tag covalently connected to n-heterocyclic Carbene ligands: Environmentally friendly catalysts for olefin metathesis transformations. Dalton Trans. 2013, 42, 7354–7358. [Google Scholar] [CrossRef]
- Szczepaniak, G.; Kosiński, K.; Grela, K. Towards “cleaner” olefin metathesis: Tailoring the NHC ligand of second generation ruthenium catalysts to afford auxiliary traits. Green Chem. 2014, 16, 4474–4492. [Google Scholar] [CrossRef]
- Skowerski, K.; Białecki, J.; Czarnocki, S.J.; Żukowska, K.; Grela, K. Effective immobilisation of a metathesis catalyst bearing an ammonium-tagged NHC ligand on various solid supports. Beilstein J. Org. Chem. 2016, 12, 5–15. [Google Scholar] [CrossRef] [PubMed]
- Skowerski, K.; Szczepaniak, G.; Wierzbicka, C.; Gułajski, L.; Bieniek, M.; Grela, K. Highly active catalysts for olefin metathesis in water. Catal. Sci. Technol. 2012, 2, 2424–2427. [Google Scholar] [CrossRef]
- Chołuj, A.; Zieliński, A.; Grela, K.; Chmielewski, M.J. Metathesis@MOF: Simple and robust immobilization of olefin metathesis catalysts inside (Al)MIL-101-NH2. ACS Catal. 2016, 6, 6343–6349. [Google Scholar] [CrossRef]
- Espallargas, G.M.; Coronado, E. Magnetic functionalities in MOFs: From the framework to the pore. Chem. Soc. Rev. 2018, 47, 533–557. [Google Scholar] [CrossRef] [PubMed]
- Chołuj, A.; Karczykowski, R.; Chmielewski, M.J. Simple and robust immobilization of a ruthenium olefin metathesis catalyst inside MOFs by acid–base reaction. Organometallics 2019, 38, 3392–3396. [Google Scholar] [CrossRef]
- Chołuj, A.; Krzesiński, P.; Ruszczyńska, A.; Bulska, E.; Kajetanowicz, A.; Grela, K. Noncovalent immobilization of cationic ruthenium complex in a metal–Organic framework by ion exchange leading to a heterogeneous olefin metathesis catalyst for use in green solvents. Organometallics 2019, 38, 3397–3405. [Google Scholar] [CrossRef]
- Chołuj, A.; Nogaś, W.; Patrzałek, M.; Krzesiński, P.; Chmielewski, M.J.; Kajetanowicz, A.; Grela, K. Preparation of ruthenium olefin metathesis catalysts immobilized on MOF, SBA-15, and 13X for probing heterogeneous boomerang effect. Catalysts 2020, 10, 438. [Google Scholar] [CrossRef]
- Slugovc, C.; Perner, B.; Stelzer, F.; Mereiter, K. “Second generation” ruthenium carbene complexes with a cis-Dichloro arrangement. Organometallics 2004, 23, 3622–3626. [Google Scholar] [CrossRef]
- Ung, T.; Hejl, A.; Grubbs, R.H.; Schrodi, Y. Latent ruthenium olefin metathesis catalysts that contain an N-Heterocyclic carbene ligand. Organometallics 2004, 23, 5399–5401. [Google Scholar] [CrossRef]
- Vehlow, K.; Gessler, S.; Blechert, S. Deactivation of ruthenium olefin metathesis catalysts through intramolecular Carbene–Arene bond formation. Angew. Chem. Int. Ed. 2007, 46, 8082–8085. [Google Scholar] [CrossRef]
- Romero, P.E.; Piers, W.; McDonald, R. Rapidly initiating ruthenium olefin-metathesis catalysts. Angew. Chem. Int. Ed. 2004, 43, 6161–6165. [Google Scholar] [CrossRef] [PubMed]
- Romero, P.E.; Piers, W. Direct observation of a 14-Electron ruthenacyclobutane relevant to olefin metathesis. J. Am. Chem. Soc. 2005, 127, 5032–5033. [Google Scholar] [CrossRef] [PubMed]
- Wenzel, A.G.; Grubbs, R.H. Ruthenium metallacycles derived from 14-Electron complexes. New insights into olefin metathesis intermediates. J. Am. Chem. Soc. 2006, 128, 16048–16049. [Google Scholar] [CrossRef] [PubMed]
- van der Eide, E.F.; Romero, P.E.; Piers, W. Generation and spectroscopic characterization of ruthenacyclobutane and ruthenium olefin carbene intermediates relevant to ring closing metathesis catalysis. J. Am. Chem. Soc. 2008, 130, 4485–4491. [Google Scholar] [CrossRef]
- Vorfalt, T.; Leuthäußer, S.; Plenio, H. An [(NHC)(NHCEWG)RuCl2(CHPh)] complex for the efficient formation of sterically hindered olefins by ring-closing metathesis. Angew. Chem. Int. Ed. 2009, 48, 5191–5194. [Google Scholar] [CrossRef]
- Huang, J.; Stevens, E.D.; Nolan, S.P.; Petersen, J.L. Olefin metathesis-active ruthenium complexes bearing a nucleophilic Carbene Ligand. J. Am. Chem. Soc. 1999, 121, 2674–2678. [Google Scholar] [CrossRef]
- Weskamp, T.; Kohl, F.J.; Hieringer, W.; Gleich, D.; Herrmann, W.A. Highly active ruthenium catalysts for olefin metathesis: The synergy of N-Heterocyclic Carbenes and Coordinatively labile ligands. Angew. Chem. Int. Ed. 1999, 38, 2416–2419. [Google Scholar] [CrossRef]
- Grubbs, R.H.; Burk, P.L.; Carr, D.D. Mechanism of the olefin metathesis reaction. J. Am. Chem. Soc. 1975, 97, 3265–3267. [Google Scholar] [CrossRef]
- Sanford, M.S.; Love, J.A.; Grubbs, R.H. Mechanism and activity of ruthenium olefin metathesis catalysts. J. Am. Chem. Soc. 2001, 123, 6543–6554. [Google Scholar] [CrossRef]
- Conrad, J.C.; Parnas, H.H.; Snelgrove, J.L.; Fogg, D.E. Highly efficient Ru−Pseudohalide catalysts for olefin metathesis. J. Am. Chem. Soc. 2005, 127, 11882–11883. [Google Scholar] [CrossRef]
- Manzini, S.; Urbina-Blanco, C.A.; Nelson, D.; Poater, A.; Lebl, T.; Meiries, S.; Slawin, A.M.Z.; Falivene, L.; Cavallo, L.; Nolan, S.P. Evaluation of an olefin metathesis pre-catalyst with a bulky and electron-rich N-heterocyclic carbene. J. Organomet. Chem. 2015, 780, 43–48. [Google Scholar] [CrossRef]
- Małecki, P.; Gajda, K.; Gajda, R.; Woźniak, K.; Trzaskowski, B.; Kajetanowicz, A.; Grela, K. Specialized ruthenium olefin metathesis catalysts bearing bulky unsymmetrical NHC ligands: Computations, synthesis, and application. ACS Catal. 2018, 9, 587–598. [Google Scholar] [CrossRef]
- Vyboishchikov, S.; Bühl, M.; Thiel, W. Mechanism of olefin metathesis with catalysis by ruthenium carbene complexes: Density functional studies on model systems. Chem. Eur. J. 2002, 8, 3962–3975. [Google Scholar] [CrossRef]
- Occhipinti, G.; Bjørsvik, H.-R.; Jensen, V.R. Quantitative structure−activity relationships of ruthenium catalysts for olefin metathesis. J. Am. Chem. Soc. 2006, 128, 6952–6964. [Google Scholar] [CrossRef]
- Ben-Asuly, A.; Aharoni, A.; Diesendruck, C.E.; Vidavsky, Y.; Goldberg, I.; Straub, B.F.; Lemcoff, N. Photoactivation of ruthenium olefin metathesis initiators. Organometallics 2009, 28, 4652–4655. [Google Scholar] [CrossRef]
- Benitez, D.; Tkatchouk, E.; Goddard, W.A., III. Relevance of cis- and trans-dichloride Ru intermediates in grubbs-ii olefin metathesis catalysis (H2IMesCl2Ru=CHR). Chem. Commun. 2008, 6194–6196. [Google Scholar] [CrossRef]
- Correa, A.; Cavallo, L. The elusive mechanism of olefin metathesis promoted by (NHC)Ru-Based Catalysts: A trade between steric, electronic, and solvent effects. J. Am. Chem. Soc. 2006, 128, 13352–13353. [Google Scholar] [CrossRef]
- Barbasiewicz, M.; Szadkowska, A.; Bujok, R.; Grela, K. Structure and activity peculiarities of ruthenium quinoline and quinoxaline complexes: Novel metathesis catalysts. Organometallics 2006, 25, 3599–3604. [Google Scholar] [CrossRef]
- Benítez, D.; Goddard, W.A. The isomerization equilibrium between Cis and trans chloride ruthenium olefin metathesis catalysts from quantum mechanics calculations. J. Am. Chem. Soc. 2005, 127, 12218–12219. [Google Scholar] [CrossRef]
- Cavallo, L. Mechanism of ruthenium-catalyzed olefin metathesis reactions from a theoretical perspective. J. Am. Chem. Soc. 2002, 124, 8965–8973. [Google Scholar] [CrossRef]
- Adlhart, C.; Chen, P. Ligand rotation distinguishes first- and second-generation ruthenium metathesis catalysts. Angew. Chem. Int. Ed. 2002, 41, 4484–4487. [Google Scholar] [CrossRef]
- Adlhart, C.; Chen, P. Mechanism and activity of ruthenium olefin metathesis catalysts: The role of ligands and substrates from a theoretical perspective. J. Am. Chem. Soc. 2004, 126, 3496–3510. [Google Scholar] [CrossRef] [PubMed]
- Stewart, I.C.; Benítez, D.; O’Leary, D.J.; Tkatchouk, E.; Day, M.W.; Goddard, W.A.; Grubbs, R.H. Conformations of N-Heterocyclic carbene ligands in ruthenium complexes relevant to olefin metathesis. J. Am. Chem. Soc. 2009, 131, 1931–1938. [Google Scholar] [CrossRef] [PubMed]
- Voccia, M.; Nolan, S.P.; Cavallo, L.; Poater, A. The activity of indenylidene derivatives in olefin metathesis catalysts. Beilstein J. Org. Chem. 2018, 14, 2956–2963. [Google Scholar] [CrossRef]
- Yang, H.-C.; Huang, Y.-C.; Lan, Y.; Luh, T.-Y.; Zhao, Y.; Truhlar, D.G. Carbene rotamer switching explains the reverse trans effect in forming the grubbs second-generation olefin metathesis catalyst. Organometallics 2011, 30, 4196–4200. [Google Scholar] [CrossRef]
- Zhao, Y.; Truhlar, D.G. Attractive noncovalent interactions in the mechanism of grubbs second-generation Ru catalysts for olefin metathesis. Org. Lett. 2007, 9, 1967–1970. [Google Scholar] [CrossRef]
- Poater, A.; Ragone, F.; Correa, A.; Cavallo, L. Comparison of different ruthenium–alkylidene bonds in the activation step with N-Heterocyclic carbene Ru-Catalysts for olefins metathesis. Dalton Trans. 2011, 40, 11066–11069. [Google Scholar] [CrossRef]
- Mathew, J.; Koga, N.; Suresh, C.H. C−H Bond activation through σ-Bond metathesis and agostic interactions: Deactivation pathway of a grubbs second-generation catalyst. Organometallics 2008, 27, 4666–4670. [Google Scholar] [CrossRef]
- Fogg, D.E. Introduction to the virtual issue on olefin metathesis—Fundamentals and frontiers. Organometallics 2017, 36, 1881–1883. [Google Scholar] [CrossRef]
- Manzini, S.; Nelson, D.J.; Lebl, T.; Poater, A.; Cavallo, L.; Slawin, A.M.Z.; Nolan, S.P. From ruthenium olefin metathesis catalyst to (eta(5)-3-phenylindenyl)hydrido complex via alcoholysis. Chem. Commun. 2014, 50, 2205–2207. [Google Scholar] [CrossRef]
- Hong, S.H.; Chlenov, A.; Day, M.W.; Grubbs, R.H. Double C—H Activation of an N-Heterocyclic carbene ligand in a ruthenium olefin metathesis catalyst. Angew. Chem. Int. Ed. 2007, 46, 5148–5151. [Google Scholar] [CrossRef]
- Manzini, S.; Urbina-Blanco, C.A.; Poater, A.; Slawin, A.M.Z.; Cavallo, L.; Nolan, S.P. From olefin metathesis catalyst to alcohol racemization catalyst in one step. Angew. Chem. Int. Ed. 2011, 51, 1042–1045. [Google Scholar] [CrossRef] [PubMed]
- Poater, A.; Cavallo, L. Mechanistic insights into the double C–H (de)activation route of a Ru-based olefin metathesis catalyst☆. J. Mol. Catal. A Chem. 2010, 324, 75–79. [Google Scholar] [CrossRef]
- Jawiczuk, M.; Młodzikowska-Pieńsko, K.; Osella, S.; Trzaskowski, B. Molecular modeling of mechanisms of decomposition of ruthenium metathesis catalysts by acrylonitrile. Organometallics 2020, 39, 239–246. [Google Scholar] [CrossRef]
- Webster, C.E. Computational insights into degenerate ethylene exchange with a grubbs-type catalyst. J. Am. Chem. Soc. 2007, 129, 7490–7491. [Google Scholar] [CrossRef] [PubMed]
- Stewart, I.C.; Keitz, B.K.; Kuhn, K.M.; Thomas, R.M.; Grubbs, R.H. Nonproductive events in ring-closing metathesis using ruthenium catalysts. J. Am. Chem. Soc. 2010, 132, 8534–8535. [Google Scholar] [CrossRef]
- Poater, A.; Vummaleti, S.V.C.; Pump, E.; Cavallo, L. Comparing Ru and Fe-catalyzed olefin metathesis. Dalton Trans. 2014, 43, 11216–11220. [Google Scholar] [CrossRef]
- Bantreil, X.; Poater, A.; Urbina-Blanco, C.A.; Bidal, Y.D.; Falivene, L.; Randall, R.A.M.; Cavallo, L.; Slawin, A.M.Z.; Cazin, C.S.J. Synthesis and reactivity of ruthenium phosphite indenylidene complexes. Organometallics 2012, 31, 7415–7426. [Google Scholar] [CrossRef]
- Hong, S.H.; Wenzel, A.G.; Salguero, T.T.; Day, M.W.; Grubbs, R.H. Decomposition of ruthenium olefin metathesis catalysts. J. Am. Chem. Soc. 2007, 129, 7961–7968. [Google Scholar] [CrossRef]
- Urbina-Blanco, C.A.; Poater, A.; Lebl, T.; Manzini, S.; Slawin, A.M.Z.; Cavallo, L.; Nolan, S.P. The activation mechanism of Ru–Indenylidene complexes in olefin metathesis. J. Am. Chem. Soc. 2013, 135, 7073–7079. [Google Scholar] [CrossRef]
- Martin, R.L.; Hay, P.J.; Pratt, L. Hydrolysis of ferric ion in water and conformational equilibrium. J. Phys. Chem. A 1998, 102, 3565–3573. [Google Scholar] [CrossRef]
- Poater, A.; Pump, E.; Vummaleti, S.V.C.; Cavallo, L. The right computational recipe for olefin metathesis with ru-based catalysts: The whole mechanism of ring-closing olefin metathesis. J. Chem. Theory Comput. 2014, 10, 4442–4448. [Google Scholar] [CrossRef] [PubMed]
- Nuñez-Zarur, F.; Solans-Monfort, X.; Pleixats, R.; Rodríguez-Santiago, L.; Sodupe, M. DFT study on the recovery of hoveyda-grubbs-type catalyst precursors in enyne and diene ring-closing metathesis. Chem. Eur. J. 2013, 19, 14553–14565. [Google Scholar] [CrossRef]
- Tracz, A.; Gawin, A.; Bieniek, M.; Olszewski, T.K.; Skowerski, K. Ammonium NHC-Tagged olefin metathesis catalysts—Influence of counter-ions on catalytic activity. New J. Chem. 2018, 42, 8609–8614. [Google Scholar] [CrossRef]
- Parr, R.G.; Szentpály, L.V.; Liu, S. Electrophilicity index. J. Am. Chem. Soc. 1999, 121, 1922–1924. [Google Scholar] [CrossRef]
- Geerlings, P.; De Proft, F.; Langenaeker, W. Conceptual density functional theory. Chem. Rev. 2003, 103, 1793–1873. [Google Scholar] [CrossRef]
- Koopmans, T. Über die Zuordnung von wellenfunktionen und eigenwerten zu den einzelnen elektronen eines atoms. Physica 1934, 1, 104–113. [Google Scholar] [CrossRef]
- Poater, A.; Solans-Monfort, X.; Clot, E.; Copéret, C.; Eisenstein, O. DFT Calculations of d0 M(NR)(CHtBu)(X)(Y) (M = Mo, W.; R = CPh3, 2,6-iPr–C6H3; X and Y = CH2tBu, OtBu, OSi(OtBu)3) olefin metathesis catalysts: Structural, spectroscopic and electronic properties. Dalton Trans. 2006, 2006, 3077–3087. [Google Scholar] [CrossRef]
- Falivene, L.; Cao, Z.; Petta, A.; Serra, L.; Poater, A.; Oliva, R.; Scarano, V.; Cavallo, L. Towards the online computer-aided design of catalytic pockets. Nat. Chem. 2019, 11, 872–879. [Google Scholar] [CrossRef]
- Poater, A.; Cavallo, L. Comparing families of olefin polymerization precatalysts using the percentage of buried volume. Dalton Trans. 2009, 8875–8883. [Google Scholar] [CrossRef]
- Mariz, R.; Poater, A.; Gatti, M.; Drinkel, E.; Bürgi, J.J.; Luan, X.; Blumentritt, S.; Linden, A.; Cavallo, L.; Dorta, R. C2-symmetric chiral disulfoxide ligands in rhodium-catalyzed 1,4-addition: From ligand synthesis to the enantioselection pathway. Chem. Eur. J. 2010, 16, 14335–14347. [Google Scholar] [CrossRef] [PubMed]
- Jacobsen, H.; Correa, A.; Poater, A.; Costabile, C.; Cavallo, L. Understanding the M(NHC) (NHC=N-Heterocyclic Carbene) bond. Coord. Chem. Rev. 2009, 253, 687–703. [Google Scholar] [CrossRef]
- Falivene, L.; Credendino, R.; Poater, A.; Petta, A.; Serra, L.; Oliva, R.; Scarano, V.; Cavallo, L. SambVca 2. A web tool for analyzing catalytic pockets with topographic steric maps. Organometallics 2016, 35, 2286–2293. [Google Scholar] [CrossRef]
- Poater, A.; Cosenza, B.; Correa, A.; Giudice, S.; Ragone, F.; Scarano, V.; Cavallo, L. SambVca: A web application for the calculation of the buried volume of N-heterocyclic carbene ligands. Eur. J. Inorg. Chem. 2009, 2009, 1759–1766. [Google Scholar] [CrossRef]
- Turias, F.; Poater, J.; Chauvin, R.; Poater, A. How carbo-benzenes fit molecules in their inner core as do biologic ion carriers? Struct. Chem. 2016, 27, 240–259. [Google Scholar] [CrossRef]
- Poater, A.; Falivene, L.; Urbina-Blanco, C.A.; Manzini, S.; Nolan, S.P.; Cavallo, L. How does the addition of steric hindrance to a typical n-heterocyclic carbene ligand affect catalytic activity in olefin metathesis? Dalton Trans. 2013, 42, 7433–7439. [Google Scholar] [CrossRef]
- Johnson, E.R.; Keinan, S.; Mori-Sánchez, P.; Contreras-Garcia, J.; Cohen, A.; Yang, W. Revealing noncovalent interactions. J. Am. Chem. Soc. 2010, 132, 6498–6506. [Google Scholar] [CrossRef]
- Contreras-Garcia, J.; Johnson, E.R.; Keinan, S.; Chaudret, R.; Piquemal, J.-P.; Beratan, D.N.; Yang, W. NCIPLOT: A program for plotting noncovalent interaction regions. J. Chem. Theory Comput. 2011, 7, 625–632. [Google Scholar] [CrossRef]
- Poater, J.; Gimferrer, M.; Poater, A. Covalent and ionic capacity of MOFs to sorb small gas molecules. Inorg. Chem. 2018, 57, 6981–6990. [Google Scholar] [CrossRef]
- Masdemont, J.; Luque-Urrutia, J.A.; Gimferrer, M.; Milstein, D.; Poater, A. Mechanism of coupling of alcohols and amines to generate aldimines and H2 by a pincer manganese catalyst. ACS Catal. 2019, 9, 1662–1669. [Google Scholar] [CrossRef]
- Jans, A.C.H.; Gómez-Suárez, A.; Nolan, S.P.; Reek, J.N.H. A switchable gold catalyst by encapsulation in a self-assembled cage. Chem. Eur. J. 2016, 22, 14836–14839. [Google Scholar] [CrossRef] [PubMed]
- Frisch, M.J.; Trucks, G.W.; Schlegel, H.B.; Scuseria, G.E.; Robb, M.A.; Cheeseman, J.R.; Scalmani, G.; Barone, V.; Mennucci, B.; Petersson, G.A.; et al. Gaussian 09, Revision E.01; Gaussian, Inc.: Wallingford, CT, USA, 2009. [Google Scholar]
- Becke, A.D. Density-functional exchange-energy approximation with correct asymptotic behavior. Phys. Rev. A 1988, 38, 3098–3100. [Google Scholar] [CrossRef] [PubMed]
- Perdew, J.P. Density-functional approximation for the correlation energy of the inhomogeneous electron gas. Phys. Rev. B 1986, 33, 8822–8824. [Google Scholar] [CrossRef] [PubMed]
- Schäfer, A.; Horn, H.; Ahlrichs, R. Fully optimized contracted gaussian basis sets for atoms Li to Kr. J. Chem. Phys. 1992, 97, 2571–2577. [Google Scholar] [CrossRef]
- Küchle, W.; Dolg, M.; Stoll, H.; Preuss, H. Energy-adjusted pseudopotentials for the actinides. Parameter sets and test calculations for thorium and thorium monoxide. J. Chem. Phys. 1994, 100, 7535–7542. [Google Scholar] [CrossRef]
- Barone, V.; Cossi, M. Quantum calculation of molecular energies and energy gradients in solution by a conductor solvent model. J. Phys. Chem. A 1998, 102, 1995–2001. [Google Scholar] [CrossRef]
- Tomasi, J.; Persico, M. Molecular interactions in solution: An overview of methods based on continuous distributions of the solvent. Chem. Rev. 1994, 94, 2027–2094. [Google Scholar] [CrossRef]
- Perdew, J.P.; Burke, K.; Ernzerhof, M. Generalized gradient approximation made simple. Phys. Rev. Lett. 1996, 77, 3865–3868. [Google Scholar] [CrossRef] [PubMed]
- Kendall, R.A.; Dunning, T.H., Jr.; Harrison, R.J. Electron affinities of the first-row atoms revisited. Systematic basis sets and wave functions. J. Chem. Phys. 1992, 96, 6796–6806. [Google Scholar] [CrossRef]
- Grimme, S.; Antony, J.; Ehrlich, S.; Krieg, H. A aonsistent and accurate Ab Initio parametrization of density functional dispersion correction (DFT-D) for the 94 elements H-Pu. J. Chem. Phys. 2010, 132, 154104. [Google Scholar] [CrossRef] [PubMed]
- Hutter, J.; Iannuzzi, M.; Schiffmann, F.; Vandevondele, J. cp2k: Atomistic simulations of condensed matter systems. Wiley Interdiscip. Rev. Comput. Mol. Sci. 2014, 4, 15–25. [Google Scholar] [CrossRef]
- Perdew, J.P.; Ruzsinszky, A.; Csonka, G.I.; Vydrov, O.A.; Scuseria, G.E.; Constantin, L.A.; Zhou, X.; Burke, K. Restoring the density-gradient expansion for exchange in solids and surfaces. Phys. Rev. Lett. 2007, 100, 136406. [Google Scholar] [CrossRef] [PubMed]
- Vandevondele, J.; Hutter, J. Gaussian basis sets for accurate calculations on molecular systems in gas and condensed phases. J. Chem. Phys. 2007, 127, 114105. [Google Scholar] [CrossRef] [PubMed]
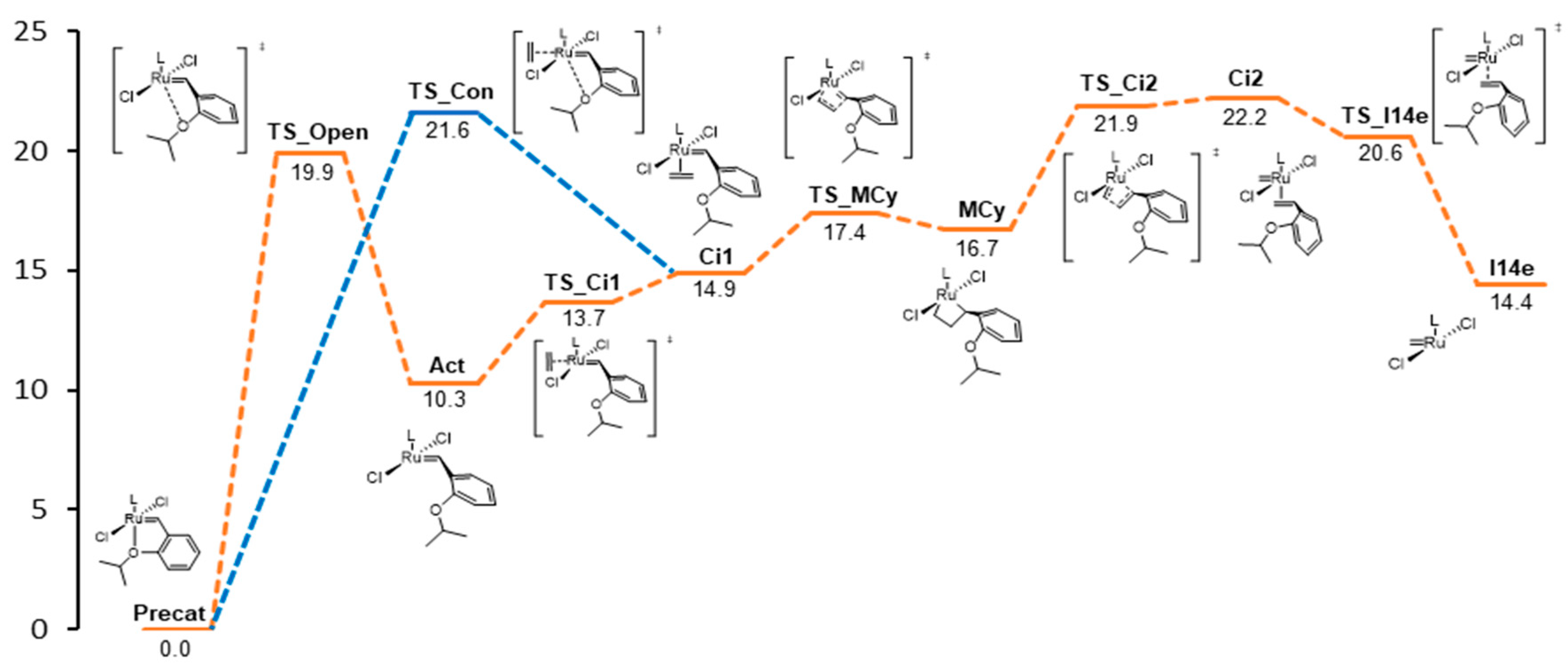

| Catalyst | Precat | TS_open | Act | TS_Ci1 | Ci1 | TS_MCy | MCy | TS_Ci2 | Ci2 | TS_I14e | I14e |
|---|---|---|---|---|---|---|---|---|---|---|---|
| HOV | 0.0 | 19.9 | 10.3 | 13.7 | 14.9 | 17.4 | 16.7 | 21.9 | 22.2 | 20.6 | 14.4 |
| AquaMetTM+ | 0.0 | 15.8 | 9.0 | 12.5 | 15.0 | 16.6 | 16.1 | 19.9 | 19.0 | 19.4 | 13.4 |
| AquaMetTM | 0.0 | 15.4 | 10.9 | 13.7 | 14.2 | 17.8 | 16.4 | 20.4 | 21.7 | 19.9 | 14.0 |
| Catalyst | Bond | Precat | Act | Ci1 | MCy | Ci2 | I14e |
|---|---|---|---|---|---|---|---|
| HOV | Ru=Cylidene(1) | 1.848 | 1.846 | 1.873 | 2.035 | 2.241 | - |
| Ru=Cylidene(2) | - | - | 2.276 | 1.980 | 1.814 | 1.809 | |
| Ru–CNHC | 1.965 | 1.927 | 2.033 | 2.016 | 2.076 | 1.933 | |
| Ru–O | 2.304 | - | - | - | - | - | |
| AquaMetTM+ | Ru=Cylidene(1) | 1.850 | 1.851 | 1.877 | 2.042 | 2.283 | - |
| Ru=Cylidene(2) | - | - | 2.291 | 1.981 | 1.824 | 1.809 | |
| Ru–CNHC | 1.953 | 1.916 | 2.014 | 2.007 | 1.981 | 1.917 | |
| Ru–O | 2.296 | - | - | - | - | - | |
| AquaMetTM | Ru=Cylidene(1) | 1.848 | 1.845 | 1.871 | 2.033 | 2.240 | - |
| Ru=Cylidene(2) | - | - | 2.275 | 1.979 | 1.814 | 1.809 | |
| Ru–CNHC | 1.966 | 1.935 | 2.034 | 2.015 | 2.079 | 1.939 | |
| Ru–O | 2.311 | - | - | - | - | - |
| Catalyst | Precat | Act | I14e | |||||||
|---|---|---|---|---|---|---|---|---|---|---|
| μ | η | ε | μ | η | ε | μ | η | ε | ||
| HOV | Gas | −0.125 | 0.059 | 0.134 | −0.129 | 0.069 | 0.120 | −0.133 | 0.079 | 0.111 |
| Solvent | −0.139 | 0.117 | 0.082 | −0.141 | 0.124 | 0.080 | −0.142 | 0.142 | 0.071 | |
| AquaMetTM+ | Gas | −0.184 | 0.064 | 0.265 | −0.187 | 0.072 | 0.243 | −0.197 | 0.084 | 0.232 |
| Solvent | −0.157 | 0.119 | 0.103 | −0.158 | 0.124 | 0.101 | −0.161 | 0.145 | 0.090 | |
| AquaMetTM | Gas | −0.115 | 0.057 | 0.116 | −0.115 | 0.066 | 0.099 | −0.118 | 0.076 | 0.091 |
| Solvent | −0.135 | 0.116 | 0.078 | −0.134 | 0.123 | 0.073 | −0.136 | 0.141 | 0.065 | |
| Catalyst | Atom | Precat | Act | Ci1 | MCy | Ci2 | I14e |
|---|---|---|---|---|---|---|---|
| HOV | Ru | −0.253 | −0.157 | −0.337 | −0.269 | −0.283 | −0.178 |
| Ru=Cylidene(1) | −0.001 | −0.024 | 0.051 | −0.164 | −0.443 | - | |
| Ru=Cylidene(2) | - | - | −0.426 | −0.333 | −0.147 | −0.205 | |
| CNHC | 0.487 | 0.505 | 0.458 | 0.481 | 0.462 | - | |
| O | −0.475 | −0.531 | −0.529 | −0.541 | −0.544 | - | |
| Cl1 | −0.268 | −0.253 | −0.296 | −0.308 | −0.303 | - | |
| Cl2 | −0.269 | −0.264 | −0.288 | −0.306 | −0.298 | - | |
| AquaMetTM+ | Ru | −0.253 | −0.156 | −0.342 | −0.274 | −0.342 | −0.174 |
| Ru=Cylidene(1) | 0.002 | −0.020 | 0.050 | −0.161 | −0.465 | - | |
| Ru=Cylidene(2) | - | - | −0.424 | −0.328 | −0.133 | −0.197 | |
| CNHC | 0.485 | 0.502 | 0.464 | 0.482 | 0.473 | - | |
| O | −0.476 | −0.533 | −0.531 | −0.545 | −0.537 | - | |
| Cl1 | −0.253 | −0.240 | −0.291 | −0.308 | −0.294 | - | |
| Cl2 | −0.264 | −0.263 | −0.286 | −0.295 | −0.327 | - | |
| AquaMetTM | Ru | −0.250 | −0.157 | −0.336 | −0.263 | −0.282 | −0.179 |
| Ru=Cylidene(1) | −0.009 | −0.034 | 0.046 | −0.171 | −0.445 | - | |
| Ru=Cylidene(2) | - | - | −0.427 | −0.333 | −0.148 | −0.210 | |
| CNHC | 0.489 | 0.508 | 0.463 | 0.486 | 0.464 | - | |
| O | −0.475 | −0.531 | −0.530 | −0.546 | −0.546 | - | |
| Cl1 | −0.261 | −0.245 | −0.294 | −0.305 | −0.294 | - | |
| Cl2 | −0.273 | −0.269 | −0.291 | −0.312 | −0.309 | - |
| Model Catalyst | Precat | Act | Ci1 | MCy | Ci2 | I14e |
|---|---|---|---|---|---|---|
| AquaMetTM+ (a) | 0.0 | 13.2 | 6.3 | 0.2 | 12.9 | 18.0 |
| AquaMetTM (a) | 0.0 | 11.8 | 5.2 | −2.4 | 13.1 | 13.2 |
| MOF-AquaMetTM (a) | 0.0 | 2.1 | −3.1 | 3.1 | 3.9 | 8.2 |
| AquaMetTM+ (b) | 0.0 | 13.6 | 6.0 | 1.0 | 7.0 | 14.4 |
| AquaMetTM (b) | 0.0 | 14.7 | 5.8 | −0.1 | 9.9 | 10.8 |
| HOV(b) | 0.0 | 12.9 | 5.6 | 0.5 | 10.4 | 13.0 |
| HOV(b,c) | 0.0 | 10.5 | 15.0 (18.7) | 11.6 (15.4) | 20.0 (23.8) | 9.8 (9.0) |
| Model Catalyst | Radius | 3.5 | 5.0 | 8.0 | 10.0 | 12.0 | 15.0 |
|---|---|---|---|---|---|---|
| HOV | 33.1 | 35.4 | 16.6 | 8.7 | 5.0 | 0.0 |
| AquaMetTM | 33.3 | 35.6 | 18.7 | 11.0 | 7.1 | 3.7 |
| MOF | 0.0 | 0.0 | 0.9 | 5.0 | 10.2 | 16.9 |
| MOF-AquaMetTM | 32.9 | 39.6 | 24.8 | 18.4 | 18.6 | 21.3 |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Pareras, G.; Tiana, D.; Poater, A. MOF Encapsulation of Ru Olefin Metathesis Catalysts to Block Catalyst Decomposition. Catalysts 2020, 10, 687. https://doi.org/10.3390/catal10060687
Pareras G, Tiana D, Poater A. MOF Encapsulation of Ru Olefin Metathesis Catalysts to Block Catalyst Decomposition. Catalysts. 2020; 10(6):687. https://doi.org/10.3390/catal10060687
Chicago/Turabian StylePareras, Gerard, Davide Tiana, and Albert Poater. 2020. "MOF Encapsulation of Ru Olefin Metathesis Catalysts to Block Catalyst Decomposition" Catalysts 10, no. 6: 687. https://doi.org/10.3390/catal10060687
APA StylePareras, G., Tiana, D., & Poater, A. (2020). MOF Encapsulation of Ru Olefin Metathesis Catalysts to Block Catalyst Decomposition. Catalysts, 10(6), 687. https://doi.org/10.3390/catal10060687