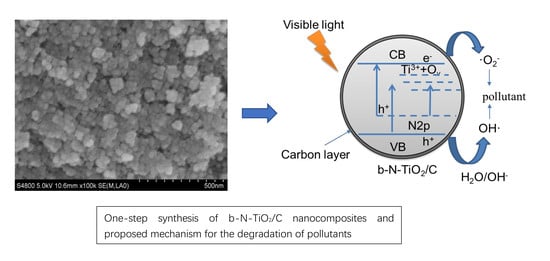
One-Step Synthesis of b-N-TiO2/C Nanocomposites with High Visible Light Photocatalytic Activity to Degrade Microcystis aeruginosa
Abstract
1. Introduction
2. Results and Discussion
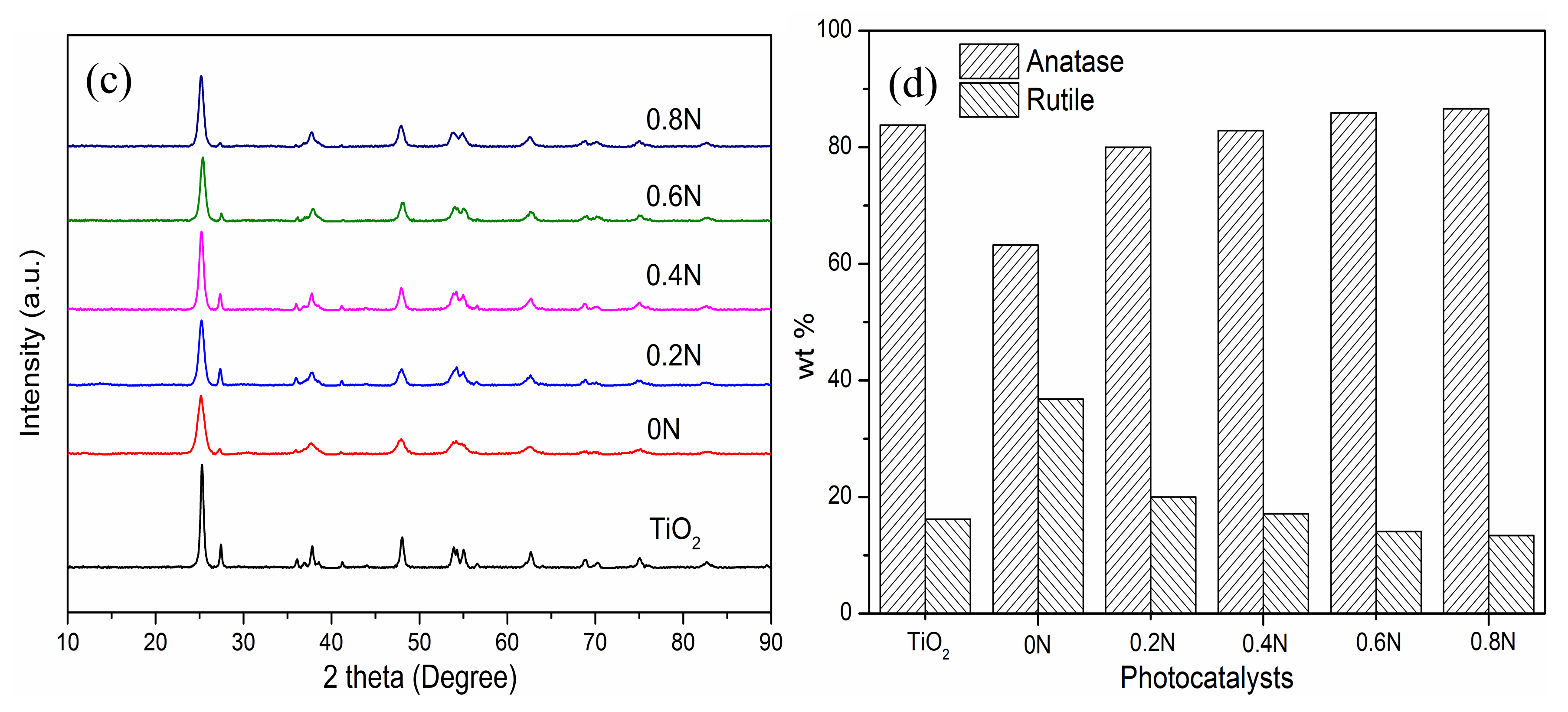
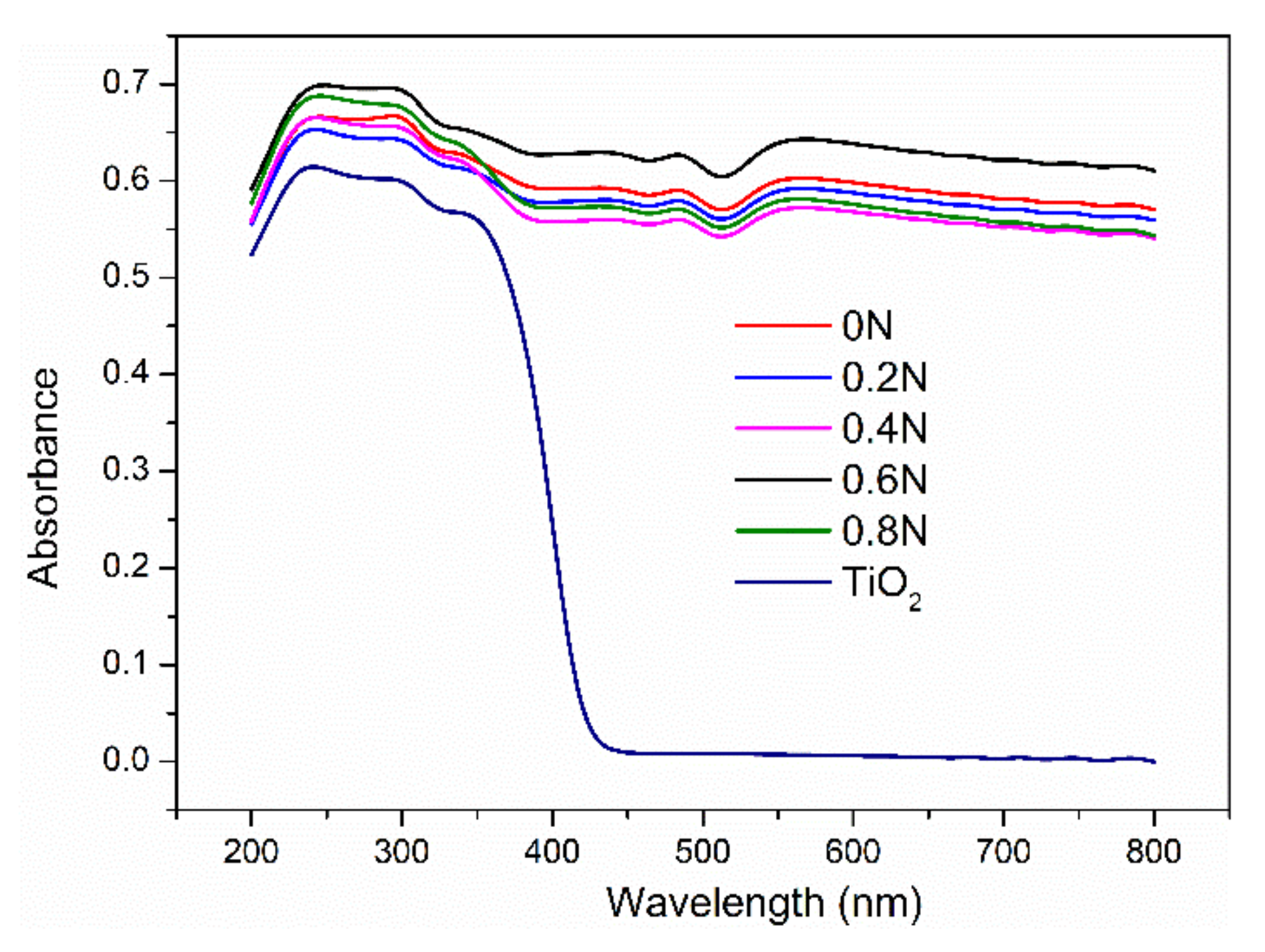
2.1. Characterization of Photocatalysts
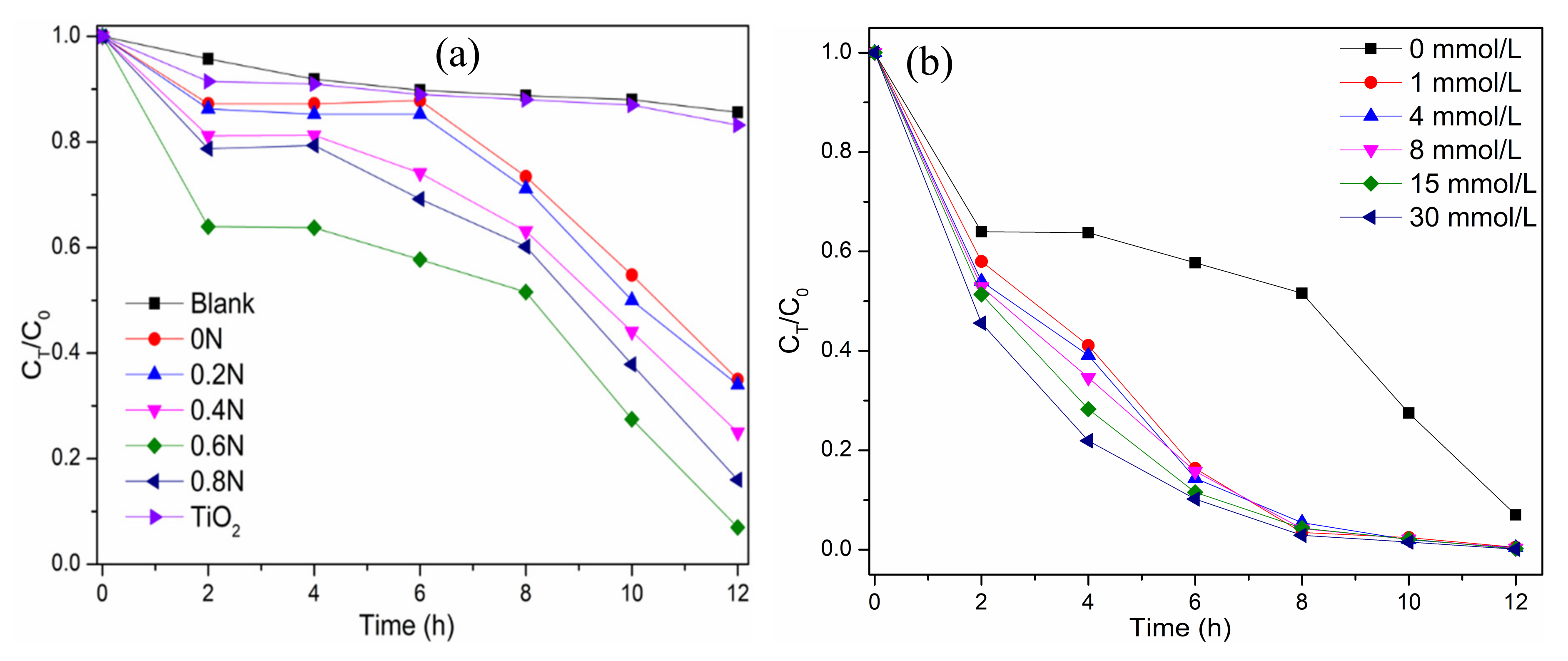
2.2. Photocatalytic Performance
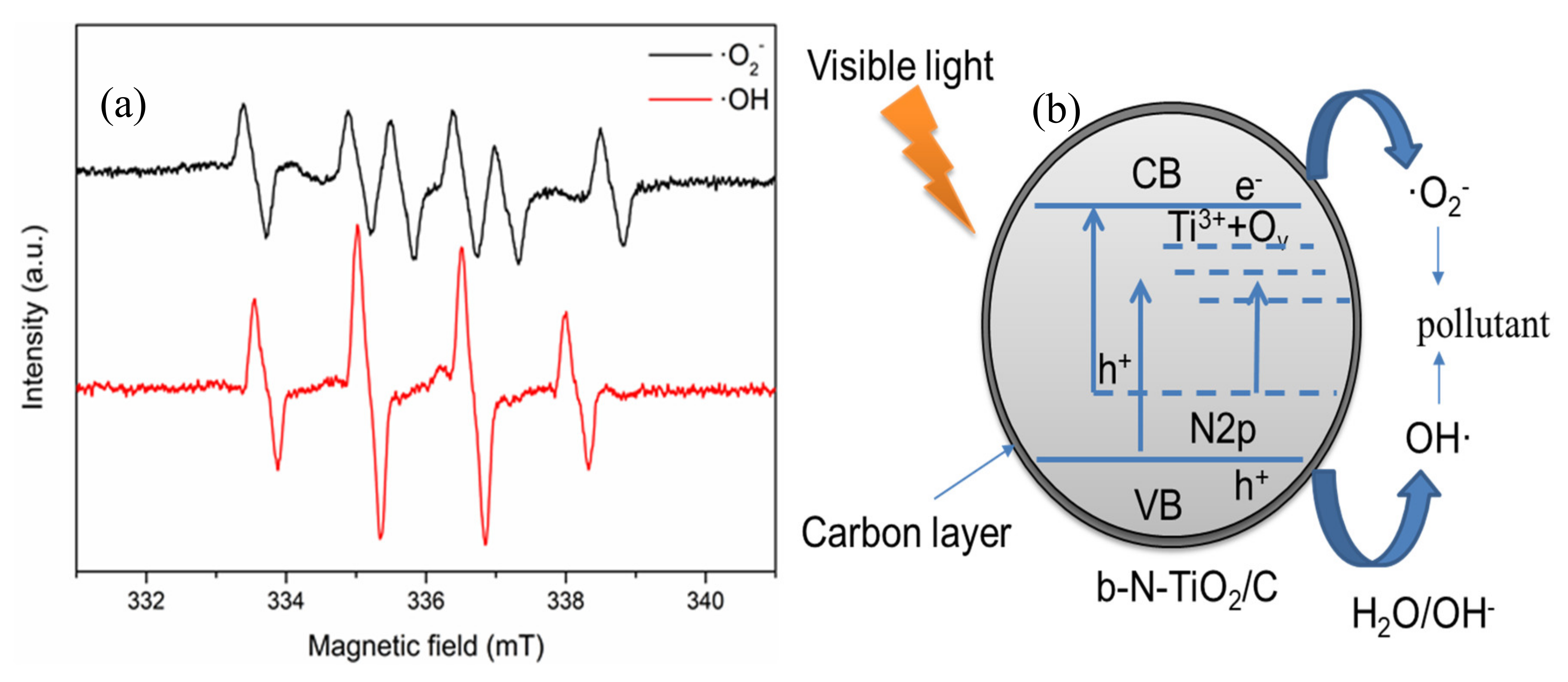
2.3. Photocatalytic Mechanism
3. Materials and Methods
3.1. Chemicals and Materials
3.2. Preparation of Photocatalysts
3.3. Photocatalytic Degradation
3.4. Analytical Methods
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Conflicts of Interest
References
- Paerl, H.W.; Gardner, W.S.; Havens, K.E.; Joyner, A.R.; McCarthy, M.J.; Newell, S.E.; Qin, B.; Scott, J.T. Mitigating cyanobacterial harmful algal blooms in aquatic ecosystems impacted by climate change and anthropogenic nutrients. Harmful Algae 2016, 54, 213–222. [Google Scholar] [CrossRef] [PubMed]
- Wang, X.; Wang, X.; Zhao, J.; Song, J.; Su, C.; Wang, Z. Surface modified TiO2 floating photocatalyst with PDDA for efficient adsorption and photocatalytic inactivation of Microcystis aeruginosa. Water Res. 2018, 131, 320–333. [Google Scholar] [CrossRef] [PubMed]
- Song, J.; Wang, X.; Ma, J.; Wang, X.; Wang, J.; Zhao, J. Visible-light-driven in situ inactivation of Microcystis aeruginosa with the use of floating g-C3N4 heterojunction photocatalyst: Performance, mechanisms and implications. Appl. Catal. B Environ. 2018, 226, 83–92. [Google Scholar] [CrossRef]
- Wang, X.; Wang, X.; Zhao, J.; Song, J.; Zhou, L.; Ma, R.; Wang, J.; Tong, X.; Chen, Y. Efficient visible light-driven in situ photocatalytic destruction of harmful alga by worm-like N,P co-doped TiO2/expanded graphite carbon layer (NPT-EGC) floating composites. Catal. Sci. Technol. 2017, 7, 2335–2346. [Google Scholar] [CrossRef]
- Wang, X.; Song, J.; Zhao, J.; Wang, Z.; Wang, X. In-situ active formation of carbides coated with NP-TiO2 nanoparticles for efficient adsorption-photocatalytic inactivation of harmful algae in eutrophic water. Chemosphere 2019, 228, 351–359. [Google Scholar] [CrossRef]
- Jin, Y.; Zhang, S.; Xu, H.; Ma, C.; Sun, J.; Li, H.; Pei, H. Application of N-TiO2 for visible-light photocatalytic degradation of Cylindrospermopsis raciborskii—More difficult than that for photodegradation of Microcystis aeruginosa? Environ. Pollut. 2019, 245, 642–650. [Google Scholar] [CrossRef]
- Lu, Z.; Zhang, K.; Liu, X.; Shi, Y. High efficiency inactivation of microalgae in ballast water by a new proposed dual-wave UV-photocatalysis system (UVA/UVC-TiO2). Environ. Sci. Pollut. R. 2019, 26, 7785–7792. [Google Scholar] [CrossRef]
- Chen, X.; Burda, C. The Electronic Origin of the Visible-Light Absorption Properties of C-, N- and S-Doped TiO2 Nanomaterials. J. Am. Chem. Soc. 2008, 130, 5018–5019. [Google Scholar] [CrossRef]
- Miao, X.; Yue, X.; Ji, Z.; Shen, X.; Zhou, H.; Liu, M.; Xu, K.; Zhu, J.; Zhu, G.; Kong, L.; et al. Nitrogen-doped carbon dots decorated on g-C3N4/Ag3PO4 photocatalyst with improved visible light photocatalytic activity and mechanism insight. Appl. Catal. B Environ. 2018, 227, 459–469. [Google Scholar] [CrossRef]
- Asahi, R.; Morikawa, T.; Ohwaki, T.; Aoki, K.; Taga, Y. Visible-light photocatalysis in nitrogen-doped titanium oxides. Science 2001, 293, 269–271. [Google Scholar] [CrossRef]
- Wang, W.; Tade, M.O.; Shao, Z. Nitrogen-doped simple and complex oxides for photocatalysis: A review. Prog. Mater. Sci. 2018, 92, 33–63. [Google Scholar] [CrossRef]
- Cao, Y.; Xing, Z.; Shen, Y.; Li, Z.; Wu, X.; Yan, X.; Zou, J.; Yang, S.; Zhou, W. Mesoporous black Ti3+/N-TiO2 spheres for efficient visible-light-driven photocatalytic performance. Chem. Eng. J. 2017, 325, 199–207. [Google Scholar] [CrossRef]
- Zhang, K.; Park, J.H. Surface Localization of Defects in Black TiO2: Enhancing Photoactivity or Reactivity. J. Phys. Chem. Lett. 2017, 8, 199–207. [Google Scholar] [CrossRef] [PubMed]
- Hirakawa, H.; Hashimoto, M.; Shiraishi, Y.; Hirai, T. Photocatalytic Conversion of Nitrogen to Ammonia with Water on Surface Oxygen Vacancies of Titanium Dioxide. J. Am. Chem. Soc. 2017, 139, 10929–10936. [Google Scholar] [CrossRef]
- Gómez-Polo, C.; Larumbe, S.; Monge, M. Room temperature ferromagnetism and absorption red-shift in nitrogen-doped TiO2 nanoparticles. J. Alloys Compd. 2014, 612, 450–455. [Google Scholar] [CrossRef]
- Zhao, W.; Liu, S.; Zhang, S.; Wang, R.; Wang, K. Preparation and visible-light photocatalytic activity of N-doped TiO2 by plasma-assisted sol-gel method. Catal. Today 2019, 337, 37–43. [Google Scholar] [CrossRef]
- Wei, S.; Wu, R.; Xu, X.; Jian, J.; Wang, H.; Sun, Y. One-step synthetic approach for core-shelled black anatase titania with high visible light photocatalytic performance. Chem. Eng. J. 2016, 299, 120–125. [Google Scholar] [CrossRef]
- Jia, F.; Yao, Z.; Jiang, Z.; Li, C. Preparation of carbon coated TiO2 nanotubes film and its catalytic application for H2 generation. Catal. Commun. 2011, 12, 497–501. [Google Scholar] [CrossRef]
- Wang, D.-H.; Jia, L.; Wu, X.-L.; Lu, L.-Q.; Xu, A.-W. One-step hydrothermal synthesis of N-doped TiO2/C nanocomposites with high visible light photocatalytic activity. Nanoscale 2012, 4, 576–584. [Google Scholar] [CrossRef]
- Liu, X.; Xing, Z.; Zhang, Y.; Li, Z.; Wu, X.; Tan, S.; Yu, X.; Zhu, Q.; Zhou, W. Fabrication of 3D flower-like black N-TiO2-x@MoS2 for unprecedented-high visible-light-driven photocatalytic performance. Appl. Catal. B Environ. 2017, 201, 119–127. [Google Scholar] [CrossRef]
- Yan, X.; Xing, Z.; Cao, Y.; Hu, M.; Li, Z.; Wu, X.; Zhu, Q.; Yang, S.; Zhou, W. In-Situ C-N-S-tridoped single crystal black TiO2 nanosheets with exposed {001} facets as efficient visible-light-driven photocatalysts. Appl. Catal. B Environ. 2017, 219, 572–579. [Google Scholar] [CrossRef]
- Hu, M.; Xing, Z.; Cao, Y.; Li, Z.; Yan, X.; Xiu, Z.; Zhao, T.; Yang, S.; Zhou, W. Ti3+ self-doped mesoporous black TiO2/SiO2/g-C3N4 sheets heterojunctions as remarkable visible-lightdriven photocatalysts. Appl. Catal. B Environ. 2018, 226, 499–508. [Google Scholar] [CrossRef]
- Nolan, N.T.; Synnott, D.W.; Seery, M.K.; Hinder, S.J.; Van Wassenhoven, A.; Pillai, S.C. Effect of N-doping on the photocatalytic activity of sol-gel TiO2. J. Hazard. Mater. 2012, 211, 88–94. [Google Scholar] [CrossRef]
- Kumar, R.; Govindarajan, S.; Siri Kiran Janardhana, R.K.; Rao, T.N.; Joshi, S.V.; Anandan, S. Facile One-Step Route for the Development of in Situ Cocatalyst-Modified Ti3+ Self-Doped TiO2 for Improved Visible-Light Photocatalytic Activity. ACS Appl. Mater. Inter. 2016, 8, 27642–27653. [Google Scholar] [CrossRef] [PubMed]
- Naldoni, A.; Allieta, M.; Santangelo, S.; Marelli, M.; Fabbri, F.; Cappelli, S.; Bianchi, C.L.; Psaro, R.; Dal Santo, V. Effect of Nature and Location of Defects on Bandgap Narrowing in Black TiO2 Nanoparticles. J. Am. Chem. Soc. 2012, 134, 7600–7603. [Google Scholar] [CrossRef] [PubMed]
- Asahi, R.; Morikawa, T. Nitrogen complex species and its chemical nature in TiO2 for visible-light sensitized photocatalysis. Chem. Phys. 2007, 339, 57–63. [Google Scholar] [CrossRef]
- Zhao, L.; Chen, X.; Wang, X.; Zhang, Y.; Wei, W.; Sun, Y.; Antonietti, M.; Titirici, M.-M. One-Step Solvothermal Synthesis of a Carbon@TiO2 Dyade Structure Effectively Promoting Visible-Light Photocatalysis. Adv. Mater. 2010, 22, 3317–3321. [Google Scholar] [CrossRef]
- Zhou, W.; Sun, F.; Pan, K.; Tian, G.; Jiang, B.; Ren, Z.; Tian, C.; Fu, H. Well-Ordered Large-Pore Mesoporous Anatase TiO2 with Remarkably High Thermal Stability and Improved Crystallinity: Preparation, Characterization, and Photocatalytic Performance. Adv. Funct. Mater. 2011, 21, 1922–1930. [Google Scholar] [CrossRef]
- Chen, X.; Liu, L.; Yu, P.Y.; Mao, S.S. Increasing solar absorption for photocatalysis with black hydrogenated titanium dioxide nanocrystals. Science 2011, 331, 746–750. [Google Scholar] [CrossRef]
- Zhang, S.; Li, W.; Jin, Z.; Yang, J.; Zhang, J.; Du, Z.; Zhang, Z. Study on ESR and inter-related properties of vacuum-dehydrated nanotubed titanic acid. J. Solid State Chem. 2004, 177, 1365–1371. [Google Scholar] [CrossRef]
- Lin, Z.A.; Lu, W.C.; Wu, C.Y.; Chang, K.S. Facile fabrication and tuning of TiO2 nanoarchitectured morphology using magnetron sputtering and its applications to photocatalysis. Ceram. Int. 2014, 40, 15523–15529. [Google Scholar] [CrossRef]
- Di Valentin, C.; Pacchioni, G.; Selloni, A. Origin of the different photoactivity of N-doped anatase and rutile TiO2. Phys. Rev. B 2004, 70, 085116. [Google Scholar] [CrossRef]
- Li, D.; Ohashi, N.; Hishita, S.; Kolodiazhnyi, T.; Haneda, H. Origin of visible-light-driven photocatalysis: A comparative study on N/F-doped and N-F-codoped TiO2 powders by means of experimental characterizations and theoretical calculations. J. Solid State Chem. 2005, 178, 3293–3302. [Google Scholar] [CrossRef]
- Zuo, F.; Wang, L.; Wu, T.; Zhang, Z.; Borchardt, D.; Feng, P. Self-Doped Ti3+ Enhanced Photocatalyst for Hydrogen Production under Visible Light. J. Am. Chem. Soc. 2010, 132, 11856–11857. [Google Scholar] [CrossRef] [PubMed]
- Chen, C.; Cai, W.; Long, M.; Zhou, B.; Wu, Y.; Wu, D.; Feng, Y. Synthesis of Visible-Light Responsive Graphene Oxide/TiO2 Composites with p/n Heterojunction. ACS Nano 2010, 4, 6425–6432. [Google Scholar] [CrossRef]
- Liu, Y.; Zhu, Y.; Xu, J.; Bai, X.; Zong, R.; Zhu, Y. Degradation and mineralization mechanism of phenol by BiPO4 photocatalysis assisted with H2O2. Appl. Catal. B Environ. 2013, 142–143, 561–567. [Google Scholar] [CrossRef]
- Zhang, C.; Yu, K.; Feng, Y.; Chang, Y.; Yang, T.; Xuan, Y.; Lei, D.; Lou, L.-L.; Liu, S. Novel 3DOM-SrTiO3/Ag/Ag3PO4 ternary Z-scheme photocatalysts with remarkably improved activity and durability for contaminant degradation. Appl. Catal. B Environ. 2017, 210, 77–87. [Google Scholar] [CrossRef]
- Hoang, S.; Berglund, S.P.; Hahn, N.T.; Bard, A.J.; Mullins, C.B. Enhancing Visible Light Photo-oxidation of Water with TiO2 Nanowire Arrays via Cotreatment with H2 and NH3: Synergistic Effects between Ti3+ and N. J. Am. Chem. Soc. 2012, 134, 3659–3662. [Google Scholar] [CrossRef]
- Lei, Y.; Zhang, L.D.; Meng, G.W.; Li, G.H.; Zhang, X.Y.; Liang, C.H.; Chen, W.; Wang, S.X. Preparation and photoluminescence of highly ordered TiO2 nanowire arrays. Appl. Phys. Lett. 2001, 78, 1125–1127. [Google Scholar] [CrossRef]
- Jing, L.; Qu, Y.; Wang, B.; Li, S.; Jiang, B.; Yang, L.; Fu, W.; Fu, H.; Sun, J. Review of photoluminescence performance of nano-sized semiconductor materials and its relationships with photocatalytic activity. Sol. Energy Mater. Sol. Cells 2006, 90, 1773–1787. [Google Scholar] [CrossRef]
- Llansola-Portoles, M.J.; Bergkamp, J.J.; Finkelstein-Shapiro, D.; Sherman, B.D.; Kodis, G.; Dimitrijevic, N.M.; Gust, D.; Moore, T.A.; Moore, A.L. Controlling Surface Defects and Photophysics in TiO2 Nanoparticles. J. Phys. Chem. A 2014, 118, 10631–10638. [Google Scholar] [CrossRef] [PubMed]
- Tafen, D.N.; Wang, J.; Wu, N.; Lewis, J.P. Visible light photocatalytic activity in nitrogen-doped TiO2 nanobelts. Appl. Phys. Lett. 2009, 94, 093101. [Google Scholar] [CrossRef]



© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Zhang, X.; Cai, M.; Cui, N.; Chen, G.; Zou, G.; Zhou, L. One-Step Synthesis of b-N-TiO2/C Nanocomposites with High Visible Light Photocatalytic Activity to Degrade Microcystis aeruginosa. Catalysts 2020, 10, 579. https://doi.org/10.3390/catal10050579
Zhang X, Cai M, Cui N, Chen G, Zou G, Zhou L. One-Step Synthesis of b-N-TiO2/C Nanocomposites with High Visible Light Photocatalytic Activity to Degrade Microcystis aeruginosa. Catalysts. 2020; 10(5):579. https://doi.org/10.3390/catal10050579
Chicago/Turabian StyleZhang, Xu, Min Cai, Naxin Cui, Guifa Chen, Guoyan Zou, and Li Zhou. 2020. "One-Step Synthesis of b-N-TiO2/C Nanocomposites with High Visible Light Photocatalytic Activity to Degrade Microcystis aeruginosa" Catalysts 10, no. 5: 579. https://doi.org/10.3390/catal10050579
APA StyleZhang, X., Cai, M., Cui, N., Chen, G., Zou, G., & Zhou, L. (2020). One-Step Synthesis of b-N-TiO2/C Nanocomposites with High Visible Light Photocatalytic Activity to Degrade Microcystis aeruginosa. Catalysts, 10(5), 579. https://doi.org/10.3390/catal10050579