Comparative Study of ZnO Thin Films Doped with Transition Metals (Cu and Co) for Methylene Blue Photodegradation under Visible Irradiation
Abstract
1. Introduction
2. Results and Discussion
2.1. Structural Study
2.2. Raman Study
2.3. Morphological Study
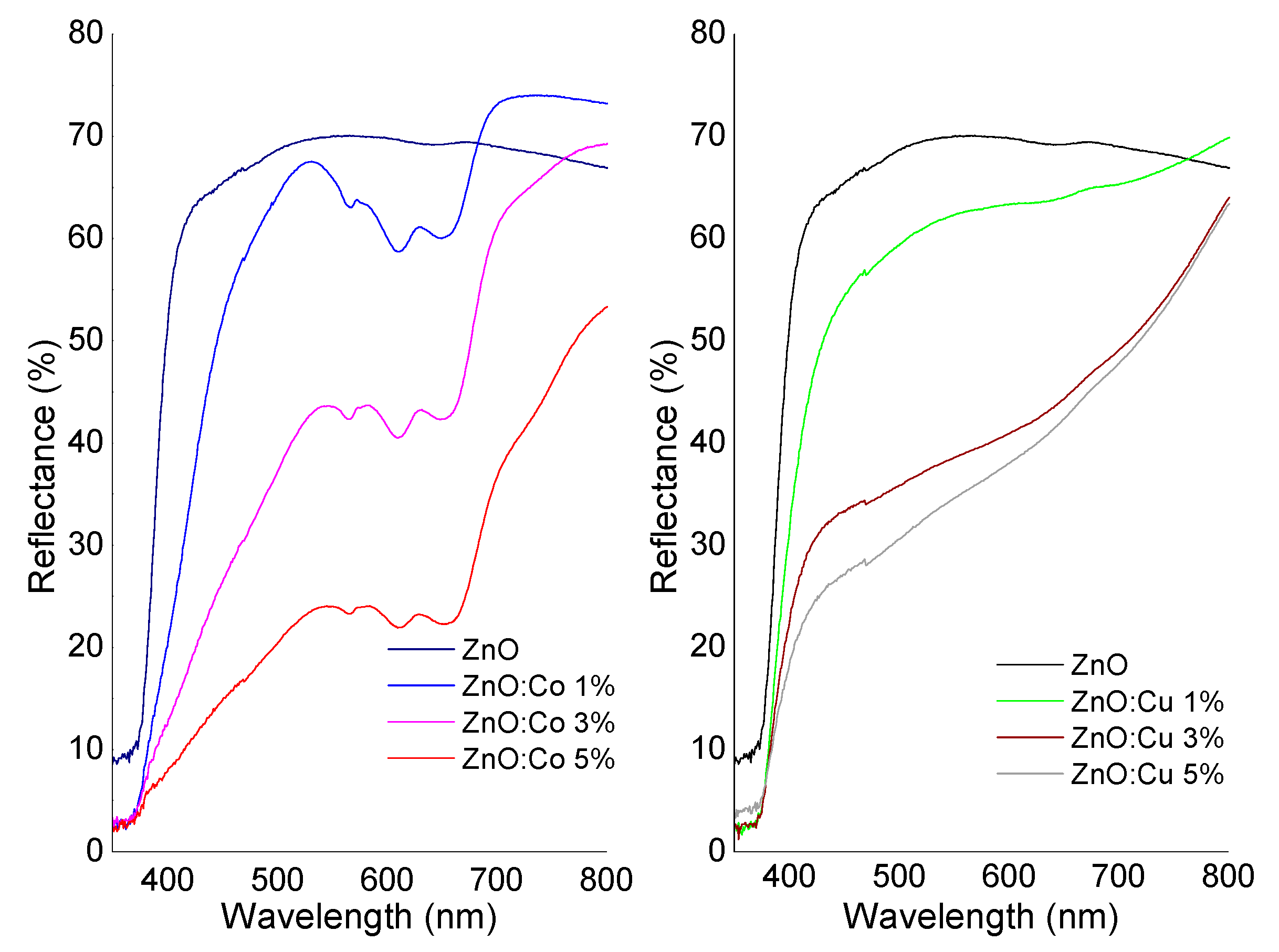
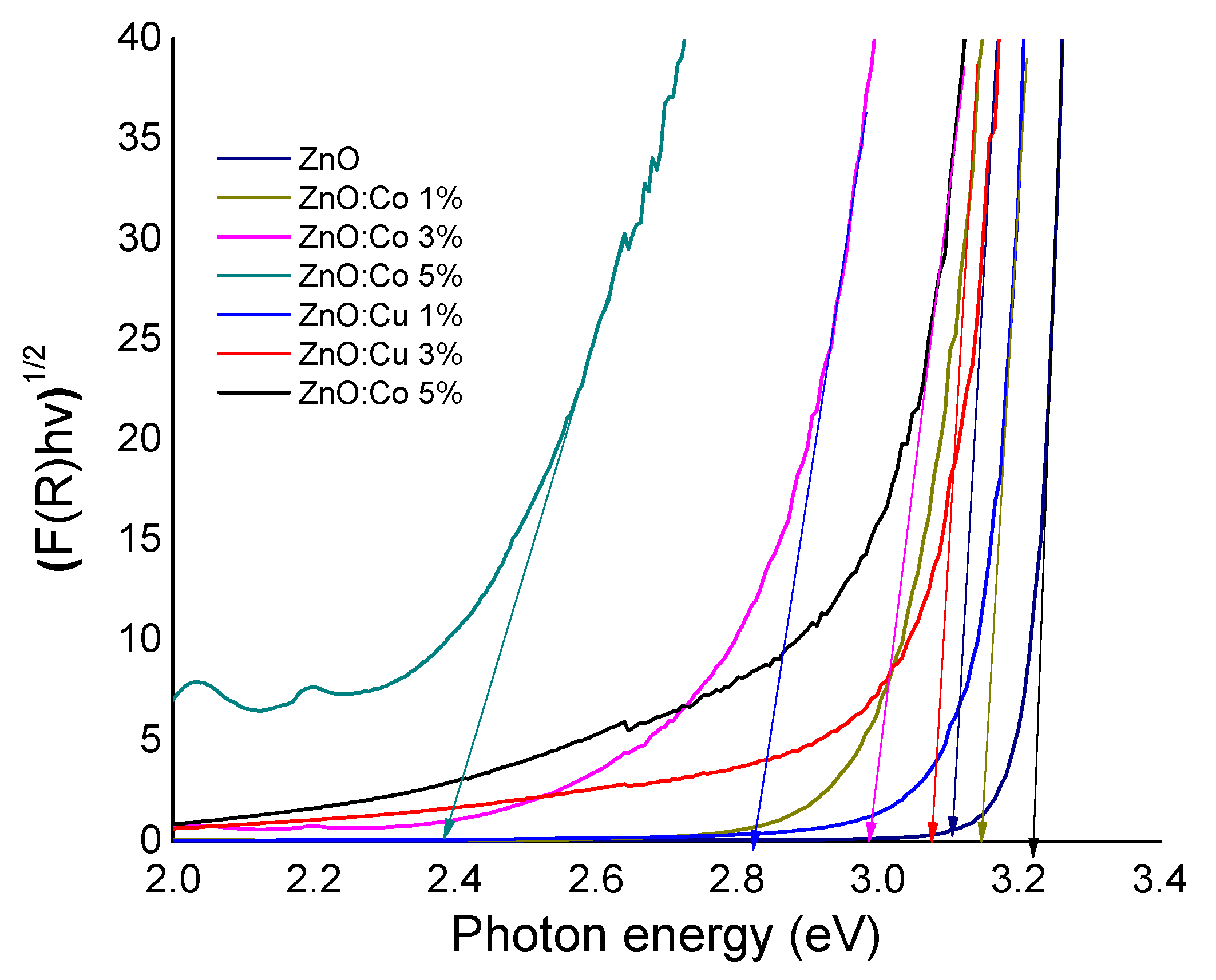
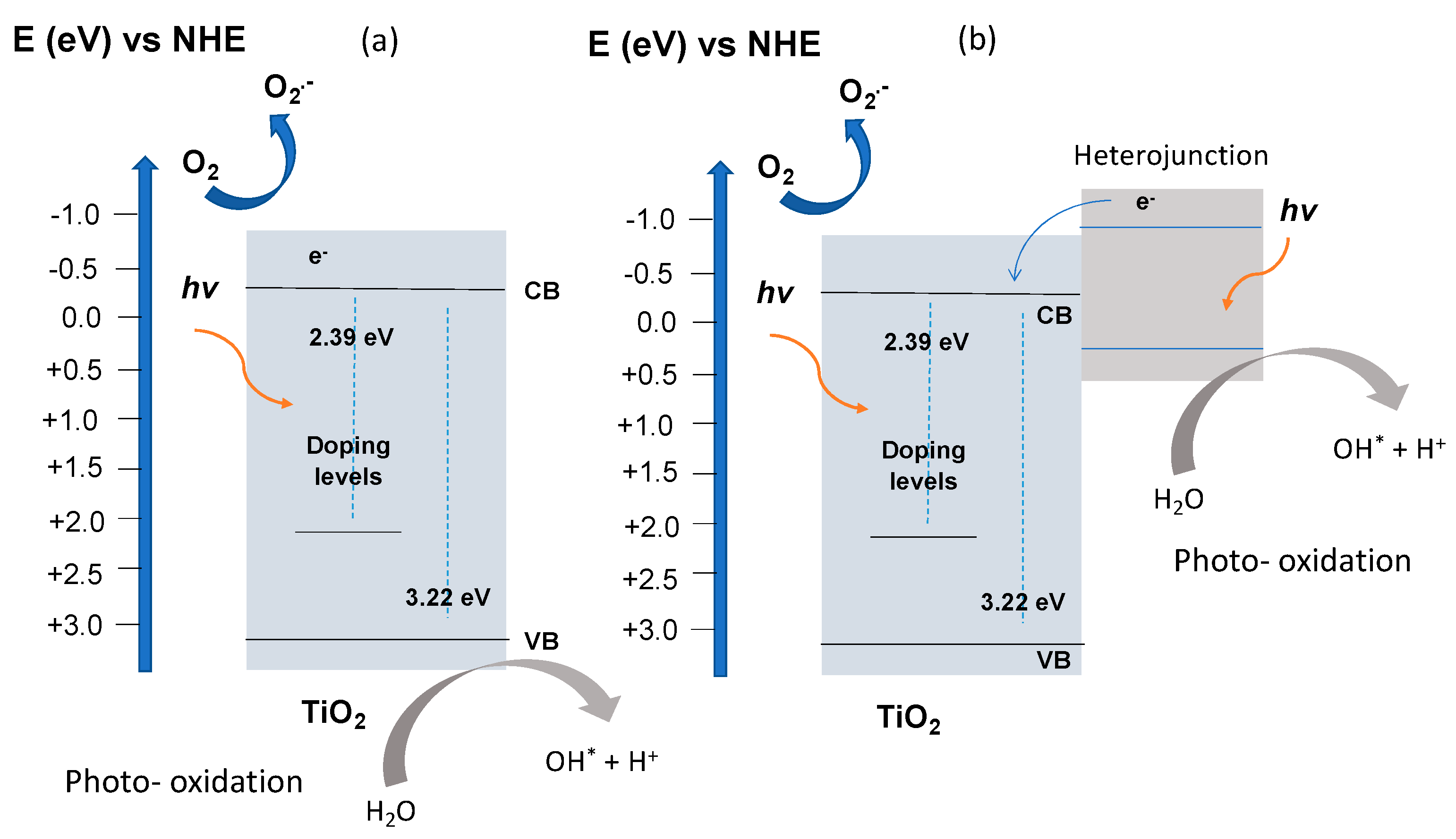
2.4. Optical Study
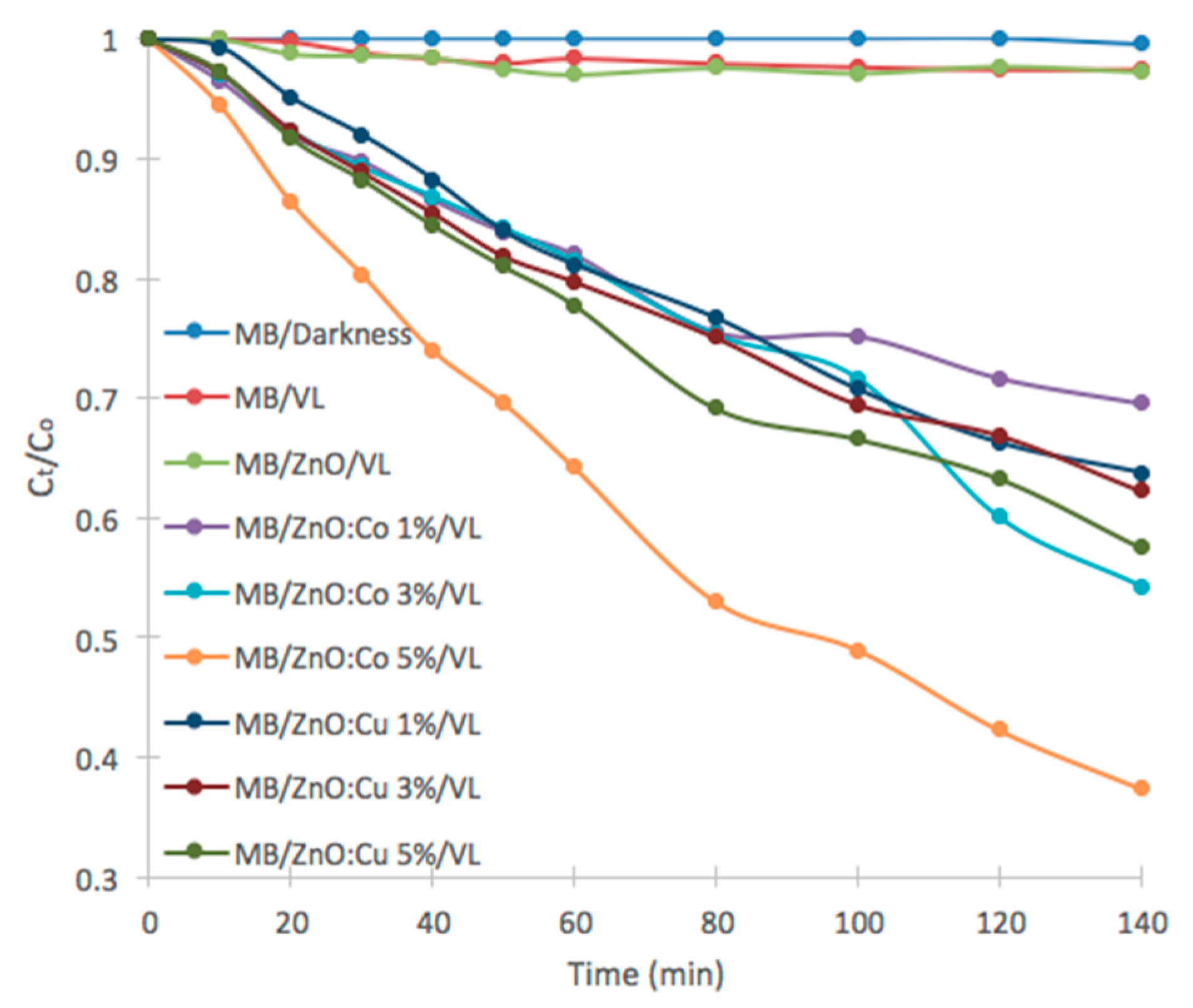
2.5. Photocatalytic Study
3. Materials and Methods
3.1. Synthesis and Characterization
3.2. Photocatalytic Test
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Lellis, B.; Fávaro-Polonio, C.Z.; Pamphile, J.A.; Polonio, J.C. Effects of textile dyes on health and the environment and bioremediation potential of living organisms. Biotechnol. Res. Innov. 2019, 3, 275–290. [Google Scholar] [CrossRef]
- Hassan, M.M.; Carr, C.M. A critical review on recent advancements of the removal of reactive dyes from dyehouse effluent by ion-exchange adsorbents. Chemosphere 2018, 209, 201–219. [Google Scholar] [CrossRef]
- Fabbri, D.; López-Muñoz, M.J.; Daniele, A.; Medana, C.; Calza, P. Photocatalytic abatement of emerging pollutants in pure water and wastewater effluent by TiO 2 and Ce-ZnO: Degradation kinetics and assessment of transformation products. Photochem. Photobiol. Sci. 2019, 18, 845–852. [Google Scholar] [CrossRef]
- Zelinski, D.W.; dos Santos, T.P.M.; Takashina, T.A.; Leifeld, V.; Igarashi-Mafra, L. Photocatalytic Degradation of Emerging Contaminants: Artificial Sweeteners. Water. Air. Soil Pollut. 2018, 229, 1–12. [Google Scholar] [CrossRef]
- Regulska, E.; Rivera-Nazario, D.; Karpinska, J.; Plonska-Brzezinska, M.; Echegoyen, L. Zinc Porphyrin-Functionalized Fullerenes for the Sensitization of Titania as a Visible-Light Active Photocatalyst: River Waters and Wastewaters Remediation. Molecules 2019, 24, 1118. [Google Scholar] [CrossRef] [PubMed]
- Ansari, S.A.; Ansari, S.G.; Foaud, H.; Cho, M.H. Facile and sustainable synthesis of carbon-doped ZnO nanostructures towards the superior visible light photocatalytic performance. New J. Chem. 2017, 41, 9314–9320. [Google Scholar] [CrossRef]
- Naldoni, A.; Riboni, F.; Guler, U.; Boltasseva, A.; Shalaev, V.M.; Kildishev, A.V. Solar-Powered Plasmon-Enhanced Heterogeneous Catalysis. Nanophotonics 2016, 5, 112–133. [Google Scholar] [CrossRef]
- Sedghi, R.; Heidari, F. A novel & effective visible light-driven TiO2/magnetic porous graphene oxide nanocomposite for the degradation of dye pollutants. RSC Adv. 2016, 6, 49459–49468. [Google Scholar]
- Balu, S.; Uma, K.; Pan, G.T.; Yang, T.C.K.; Ramaraj, S.K. Degradation of methylene blue dye in the presence of visible light using SiO2@α-Fe2O3 nanocomposites deposited on SnS2 flowers. Materials 2018, 11, 1030. [Google Scholar] [CrossRef]
- Loh, K.; Gaylarde, C.C.; Shirakawa, M.A. Photocatalytic activity of ZnO and TiO2 ‘nanoparticles’ for use in cement mixes. Constr. Build. Mater. 2018, 167, 853–859. [Google Scholar] [CrossRef]
- Saravanakkumar, D.; Oualid, H.A.; Brahmi, Y.; Ayeshamariam, A.; Karunanaithy, M.; Saleem, A.M.; Kaviyarasu, K.; Sivaranjani, S.; Jayachandran, M. Synthesis and characterization of CuO/ZnO/CNTs thin films on copper substrate and its photocatalytic applications. OpenNano 2019, 4, 100025. [Google Scholar] [CrossRef]
- Zyoud, A.; Zaatar, N.; Saadeddin, I.; Helal, M.H.; Campet, G.; Hakim, M.; Park, D.; Hilal, H.S. Alternative natural dyes in water purification: Anthocyanin as TiO2-sensitizer in methyl orange photo-degradation. Solid State Sci. 2011, 13, 1268–1275. [Google Scholar] [CrossRef]
- Elango, G.; Roopan, S.M. Efficacy of SnO2 nanoparticles toward photocatalytic degradation of methylene blue dye. J. Photochem. Photobiol. B Biol. 2016, 155, 34–38. [Google Scholar] [CrossRef]
- Das, A.; Malakar, P.; Nair, R.G. Engineering of ZnO nanostructures for efficient solar photocatalysis. Mater. Lett. 2018, 219, 76–80. [Google Scholar] [CrossRef]
- Schumann, J.; Eichelbaum, M.; Lunkenbein, T.; Thomas, N.; Álvarez Galván, M.C.; Schlögl, R.; Behrens, M. Promoting Strong Metal Support Interaction: Doping ZnO for Enhanced Activity of Cu/ZnO:M (M = Al, Ga, Mg) Catalysts. ACS Catal. 2015, 5, 3260–3270. [Google Scholar] [CrossRef]
- Kumari, V.; Mittal, A.; Jindal, J.; Yadav, S.; Kumar, N. S-, N- and C-doped ZnO as semiconductor photocatalysts: A review. Front. Mater. Sci. 2019, 13, 1–22. [Google Scholar] [CrossRef]
- Bharat, T.C.; Mondal, S.; Gupta, H.S.; Singh, P.K.; Das, A.K. Synthesis of Doped Zinc Oxide Nanoparticles: A Review. Mater. Today Proc. 2019, 11, 767–775. [Google Scholar] [CrossRef]
- Poornaprakash, U.; Chalapathi, K.; Subramanyam, S.V.; Prabhakar Vattikuti, Y.; Shun, S.P. Effects of Ce incorporation on the structural, morphological, optical, magnetic, and photocatalytic characteristics of ZnO nanoparticles. Mater. Res. Express 2019, 6, 105356. [Google Scholar] [CrossRef]
- Poornaprakash, B.; Subramanyam, K.; Vattikuti, S.V.P.; Pratap Reddy, M.S. Achieving enhanced ferromagnetism in ZnTbO nanoparticles through Cu co-doping. Ceram. Int. 2019, 45, 16347–16352. [Google Scholar] [CrossRef]
- Poornaprakash, B.; Chalapathi, U.; Poojitha, P.T.; Vattikuti, S.V.P.; Reddy, M.S.P. (Al, Cu) Co-doped ZnS nanoparticles: Structural, chemical, optical, and photocatalytic properties. J. Mater. Sci. Mater. Electron. 2019, 30, 9897–9902. [Google Scholar] [CrossRef]
- Kaur, M.; Umar, A.; Mehta, S.K.; Singh, S.; Kansal, S.K.; Fouad, H.; Alothman, O.Y. Rapid Solar-Light Driven Superior Photocatalytic Degradation of Methylene Blue Using MoS₂-ZnO Heterostructure Nanorods Photocatalyst. Material 2018, 11, 2254. [Google Scholar]
- Vallejo, W.; Díaz-Uribe, C.; Rios, K. Methylene Blue Photocatalytic Degradation under Visible Irradiation on In2S3 Synthesized by Chemical Bath Deposition. Adv. Phys. Chem. 2017, 2017, 1–5. [Google Scholar] [CrossRef]
- Subash, B.; Krishnakumar, B.; Swaminathan, M.; Shanthi, M. Highly Efficient, Solar Active, and Reusable Photocatalyst: Zr-Loaded Ag–ZnO for Reactive Red 120 Dye Degradation with Synergistic Effect and Dye-Sensitized Mechanism. Langmuir 2013, 29, 939–949. [Google Scholar] [CrossRef] [PubMed]
- Aby, H.; Kshirsagar, A.; Pk, K. Plasmon Mediated Photocatalysis by Solar Active Ag/ZnO Nanostructures: Degradation of Organic Pollutants in Aqueous Conditions. J. Mater Sci Nanotechnol 2016, 4. [Google Scholar] [CrossRef]
- Díaz-Uribe, C.; Viloria, J.; Cervantes, L.; Vallejo, W.; Navarro, K.; Romero, E.; Quiñones, C. Photocatalytic Activity of Ag-TiO2 Composites Deposited by Photoreduction under UV Irradiation. Int. J. Photoenergy 2018, 2018, 1–8. [Google Scholar] [CrossRef]
- Chen, L.; Tran, T.T.; Huang, C.; Li, J.; Yuan, L.; Cai, Q. Synthesis and photocatalytic application of Au/Ag nanoparticle-sensitized ZnO films. Appl. Surf. Sci. 2013, 273, 82–88. [Google Scholar] [CrossRef]
- Poornaprakash, B.; Chalapathi, U.; Poojitha, P.T.; Vattikuti, S.V.P.; Park, S.H. Co-Doped ZnS Quantum Dots: Structural, Optical, Photoluminescence, Magnetic, and Photocatalytic Properties. J. Supercond. Nov. Magn. 2020, 33, 539–544. [Google Scholar] [CrossRef]
- Youssef, Z.; Colombeau, L.; Yesmurzayeva, N.; Baros, F.; Vanderesse, R.; Hamieh, T.; Toufaily, J.; Frochot, C.; Roques-Carmes, T.; Acherar, S. Dye-sensitized nanoparticles for heterogeneous photocatalysis: Cases studies with TiO2, ZnO, fullerene and graphene for water purification. Dye. Pigment. 2018, 159, 49–71. [Google Scholar] [CrossRef]
- Vallejo, W.; Diaz-Uribe, C.; Cantillo, Á. Methylene blue photocatalytic degradation under visible irradiation on TiO2 thin films sensitized with Cu and Zn tetracarboxy-phthalocyanines. J. Photochem. Photobiol. A Chem. 2015, 299, 80–86. [Google Scholar] [CrossRef]
- Vallejo, W.; Rueda, A.; Díaz-Uribe, C.; Grande, C.; Quintana, P. Photocatalytic activity of graphene oxide–TiO2 thin films sensitized by natural dyes extracted from Bactris guineensis. R. Soc. Open Sci. 2019, 6, 181824. [Google Scholar] [CrossRef]
- Diaz-Uribe, C.; Vallejo, W.; Camargo, G.; Muñoz-Acevedo, A.; Quiñones, C.; Schott, E.; Zarate, X. Potential use of an anthocyanin-rich extract from berries of Vaccinium meridionale Swartz as sensitizer for TiO2 thin films—An experimental and theoretical study. J. Photochem. Photobiol. A Chem. 2019, 384, 112050. [Google Scholar] [CrossRef]
- Hamid, S.B.A.; Teh, S.J.; Lai, C.W. Photocatalytic Water Oxidation on ZnO: A Review. Catalysts 2017, 7, 93. [Google Scholar] [CrossRef]
- Türkyılmaz, Ş.Ş.; Güy, N.; Özacar, M. Photocatalytic efficiencies of Ni, Mn, Fe and Ag doped ZnO nanostructures synthesized by hydrothermal method: The synergistic/antagonistic effect between ZnO and metals. J. Photochem. Photobiol. A Chem. 2017, 341, 39–50. [Google Scholar] [CrossRef]
- Bouzid, H.; Faisal, M.; Harraz, F.A.; Al-Sayari, S.A.; Ismail, A.A. Synthesis of mesoporous Ag/ZnO nanocrystals with enhanced photocatalytic activity. Catal. Today 2015, 252, 20–26. [Google Scholar] [CrossRef]
- Altintas Yildirim, O.; Arslan, H.; Sönmezoğlu, S. Facile synthesis of cobalt-doped zinc oxide thin films for highly efficient visible light photocatalysts. Appl. Surf. Sci. 2016, 390, 111–121. [Google Scholar] [CrossRef]
- Ahmad, M.; Ahmed, E.; Ahmed, W.; Elhissi, A.; Hong, Z.L.; Khalid, N.R. Enhancing visible light responsive photocatalytic activity by decorating Mn-doped ZnO nanoparticles on graphene. Ceram. Int. 2014, 40, 10085–10097. [Google Scholar] [CrossRef]
- Polat, İ.; Yılmaz, S.; Altın, İ.; Bacaksız, E.; Sökmen, M. The influence of Cu-doping on structural, optical and photocatalytic properties of ZnO nanorods. Mater. Chem. Phys. 2014, 148, 528–532. [Google Scholar] [CrossRef]
- Mittal, M.; Sharma, M.; Pandey, O.P. UV–Visible light induced photocatalytic studies of Cu doped ZnO nanoparticles prepared by co-precipitation method. Sol. Energy 2014, 110, 386–397. [Google Scholar] [CrossRef]
- Kuriakose, S.; Satpati, B.; Mohapatra, S. Highly efficient photocatalytic degradation of organic dyes by Cu doped ZnO nanostructures. Phys. Chem. Chem. Phys. 2015, 17, 25172–25181. [Google Scholar] [CrossRef]
- Thennarasu, G.; Sivasamy, A. Metal ion doped semiconductor metal oxide nanosphere particles prepared by soft chemical method and its visible light photocatalytic activity in degradation of phenol. Powder Technol. 2013, 250, 1–12. [Google Scholar] [CrossRef]
- Lu, Y.; Lin, Y.; Wang, D.; Wang, L.; Xie, T.; Jiang, T. A high performance cobalt-doped ZnO visible light photocatalyst and its photogenerated charge transfer properties. Nano Res. 2011, 4, 1144–1152. [Google Scholar] [CrossRef]
- Kuriakose, S.; Satpati, B.; Mohapatra, S. Enhanced photocatalytic activity of Co doped ZnO nanodisks and nanorods prepared by a facile wet chemical method. Phys. Chem. Chem. Phys. 2014, 16, 12741. [Google Scholar] [CrossRef]
- Poornaprakash, B.; Chalapathi, U.; Subramanyam, K.; Vattikuti, S.V.P.; Park, S.H. Wurtzite phase Co-doped ZnO nanorods: Morphological, structural, optical, magnetic, and enhanced photocatalytic characteristics. Ceram. Int. 2020, 46, 2931–2939. [Google Scholar] [CrossRef]
- Rajbongshi, B.M.; Samdarshi, S.K. Cobalt-doped zincblende–wurtzite mixed-phase ZnO photocatalyst nanoparticles with high activity in visible spectrum. Appl. Catal. B Environ. 2014, 144, 435–441. [Google Scholar] [CrossRef]
- Rajbongshi, B.M.; Samdarshi, S.K. ZnO and Co-ZnO nanorods—Complementary role of oxygen vacancy in photocatalytic activity of under UV and visible radiation flux. Mater. Sci. Eng. B 2014, 182, 21–28. [Google Scholar] [CrossRef]
- Muchuweni, E.; Sathiaraj, T.S.; Nyakotyo, H. Synthesis and characterization of zinc oxide thin films for optoelectronic applications. Heliyon 2017, 3, e00285. [Google Scholar] [CrossRef]
- Yuhas, B.D.; Zitoun, D.O.; Pauzauskie, P.J.; He, R.; Yang, P. Transition-Metal Doped Zinc Oxide Nanowires. Angew. Chemie 2006, 118, 434–437. [Google Scholar] [CrossRef]
- Wang, X.; Sø, L.; Su, R.; Wendt, S.; Hald, P.; Mamakhel, A.; Yang, C.; Huang, Y.; Iversen, B.B.; Besenbacher, F. The influence of crystallite size and crystallinity of anatase nanoparticles on the photo-degradation of phenol. J. Catal. 2014, 310, 100–108. [Google Scholar] [CrossRef]
- Lima, M.K.; Fernandes, D.M.; Silva, M.F.; Baesso, M.L.; Neto, A.M.; de Morais, G.R.; Nakamura, C.V.; de Oliveira Caleare, A.; Hechenleitner, A.A.W.; Pineda, E.A.G. Co-doped ZnO nanoparticles synthesized by an adapted sol–gel method: Effects on the structural, optical, photocatalytic and antibacterial properties. J. Sol-Gel Sci. Technol. 2014, 72, 301–309. [Google Scholar] [CrossRef]
- Calleja, J.M.; Cardona, M. Resonant Raman scattering in ZnO. Phys. Rev. B 1977, 16, 3753–3761. [Google Scholar] [CrossRef]
- Cuscó, R.; Alarcón-Lladó, E.; Ibáñez, J.; Artús, L.; Jiménez, J.; Wang, B.; Callahan, M.J. Temperature dependence of Raman scattering in ZnO. Phys. Rev. B 2007, 75, 165202. [Google Scholar] [CrossRef]
- Wang, W.; Zhou, Q.; Fei, X.; He, Y.; Zhang, P.; Zhang, G.; Peng, L.; Xie, W. Synthesis of CuO nano- and micro-structures and their Raman spectroscopic studies. CrystEngComm 2010, 12, 2232. [Google Scholar] [CrossRef]
- Winiarski, J.; Tylus, W.; Szczygieł, B. EIS and XPS investigations on the corrosion mechanism of ternary Zn–Co–Mo alloy coatings in NaCl solution. Appl. Surf. Sci. 2016, 364, 455–466. [Google Scholar] [CrossRef]
- Xuan, H.; Yao, C.; Hao, X.; Liu, C.; Ren, J.; Zhu, Y.; Xu, C.; Ge, L. Fluorescence enhancement with one-dimensional photonic crystals/nanoscaled ZnO composite thin films. Colloids Surfaces A Physicochem. Eng. Asp. 2016, 497, 251–256. [Google Scholar] [CrossRef]
- Hasnidawani, J.N.; Azlina, H.N.; Norita, H.; Bonnia, N.N.; Ratim, S.; Ali, E.S. Synthesis of ZnO Nanostructures Using Sol-Gel Method. Procedia Chem. 2016, 19, 211–216. [Google Scholar] [CrossRef]
- Pourrahimi, A.M.; Liu, D.; Pallon, L.K.H.; Andersson, R.L.; Martínez Abad, A.; Lagarón, J.-M.; Hedenqvist, M.S.; Ström, V.; Gedde, U.W.; Olsson, R.T. Water-based synthesis and cleaning methods for high purity ZnO nanoparticles–comparing acetate, chloride, sulphate and nitrate zinc salt precursors. RSC Adv. 2014, 4, 35568–35577. [Google Scholar] [CrossRef]
- Simmons, E.L. Relation of the Diffuse Reflectance Remission Function to the Fundamental Optical Parameters. Opt. Acta Int. J. Opt. 1972, 19, 845–851. [Google Scholar] [CrossRef]
- Pal, M.; Pal, U.; Jiménez, J.M.G.Y.; Pérez-Rodríguez, F. Effects of crystallization and dopant concentration on the emission behavior of TiO2:Eu nanophosphors. Nanoscale Res. Lett. 2012, 7, 1. [Google Scholar] [CrossRef]
- Viezbicke, B.D.; Patel, S.; Davis, B.E.; Birnie, D.P. Evaluation of the Tauc method for optical absorption edge determination: ZnO thin films as a model system. Phys. Status Solidi 2015, 252, 1700–1710. [Google Scholar] [CrossRef]
- Srikant, V.; Clarke, D.R. On the optical band gap of zinc oxide. J. Appl. Phys. 1998, 83, 5447–5451. [Google Scholar] [CrossRef]
- El-Atab, N.; Chowdhury, F.; Ulusoy, T.G.; Ghobadi, A.; Nazirzadeh, A.; Okyay, A.K.; Nayfeh, A. ~3-nm ZnO Nanoislands Deposition and Application in Charge Trapping Memory Grown by Single ALD Step. Sci. Rep. 2016, 6, 38712. [Google Scholar] [CrossRef] [PubMed]
- Liu, X.-C.; Shi, E.-W.; Chen, Z.-Z.; Zhang, H.-W.; Song, L.-X.; Wang, H.; Yao, S.-D. Structural, optical and magnetic properties of Co-doped ZnO films. J. Cryst. Growth 2006, 296, 135–140. [Google Scholar] [CrossRef]
- Qiu, X.; Li, G.; Sun, X.; Li, L.; Fu, X. Doping effects of Co 2+ ions on ZnO nanorods and their photocatalytic properties. Nanotechnology 2008, 19, 215703. [Google Scholar] [CrossRef] [PubMed]
- Ramya, E.; Rao, M.V.; Jyothi, L.; Rao, D.N. Photoluminescence and Nonlinear Optical Properties of Transition Metal (Ag, Ni, Mn) Doped ZnO Nanoparticles. J. Nanosci. Nanotechnol. 2018, 18, 7072–7077. [Google Scholar] [CrossRef] [PubMed]
- Xu, H.; Shi, M.; Liang, C.; Wang, S.; Xia, C.; Xue, C.; Hai, Z.; Zhuiykov, S. Effect of Zinc Acetate Concentration on Optimization of Photocatalytic Activity of p-Co3O4/n-ZnO Heterostructures. Nanoscale Res. Lett. 2018, 13, 195. [Google Scholar] [CrossRef]
- Konstantinou, I.K.; Albanis, T.A. TiO2—Assisted photocatalytic degradation of azo dyes in aqueous solution: Kinetic and mechanistic investigations A review. Appl. Catal. B Environ. 2004, 49, 1–14. [Google Scholar] [CrossRef]
- Jayswal, S.; Moirangthem, R.S. Construction of a solar spectrum active SnS/ZnO p–n heterojunction as a highly efficient photocatalyst: The effect of the sensitization process on its performance. New J. Chem. 2018, 42, 13689–13701. [Google Scholar] [CrossRef]
- Jun Park, S.; Sankar Das, G.; Schütt, F.; Adelung, R.; Kumar Mishra, Y.; Malika Tripathi, K.; Kim, T. Visible-light photocatalysis by carbon-nano-onion-functionalized ZnO tetrapods: Degradation of 2,4-dinitrophenol and a plant-model-based ecological assessment. Asia Mater. 2019, 11, 1–13. [Google Scholar]
- Vallejo, W.; Cantillo, A.; Dias-Uribe, C. Methylene Blue Photodegradation under Visible Irradiation on Ag-Doped ZnO Thin Films. Int. J. Photoenergy 2020, 2020, 112. [Google Scholar] [CrossRef]
- Prasad, C.; Tang, H.; Liu, Q.Q.; Zulfiqar, S.; Shah, S.; Bahadur, I. An overview of semiconductors/layered double hydroxides composites: Properties, synthesis, photocatalytic and photoelectrochemical applications. J. Mol. Liq. 2019, 289, 111114. [Google Scholar] [CrossRef]
- Hernández-Alonso, M.D.; Fresno, F.; Suárez, S.; Coronado, J.M. Development of alternative photocatalysts to TiO2: Challenges and opportunities. Energy Environ. Sci. 2009, 2, 1231–1257. [Google Scholar] [CrossRef]
- Pérez, J.A.; Gallego, J.L.; Wilson Stiven Roman, H.R.L. Zinc Oxide Nanostructured Thin Films. Sci. Tech. 2008, 39, 416–421. [Google Scholar]
- Ramírez Vinasco, D.; Vera, L.; Patricia, L.; Riascos Landázuri, H. Zn1-xMnxO Thin Films. Sci. Tech. 2009, 41, 273–278. [Google Scholar]
- Quiñones, C.; Ayala, J.; Vallejo, W. Methylene blue photoelectrodegradation under UV irradiation on Au/Pd-modified TiO2 films. Appl. Surf. Sci. 2010, 257, 367–371. [Google Scholar] [CrossRef]
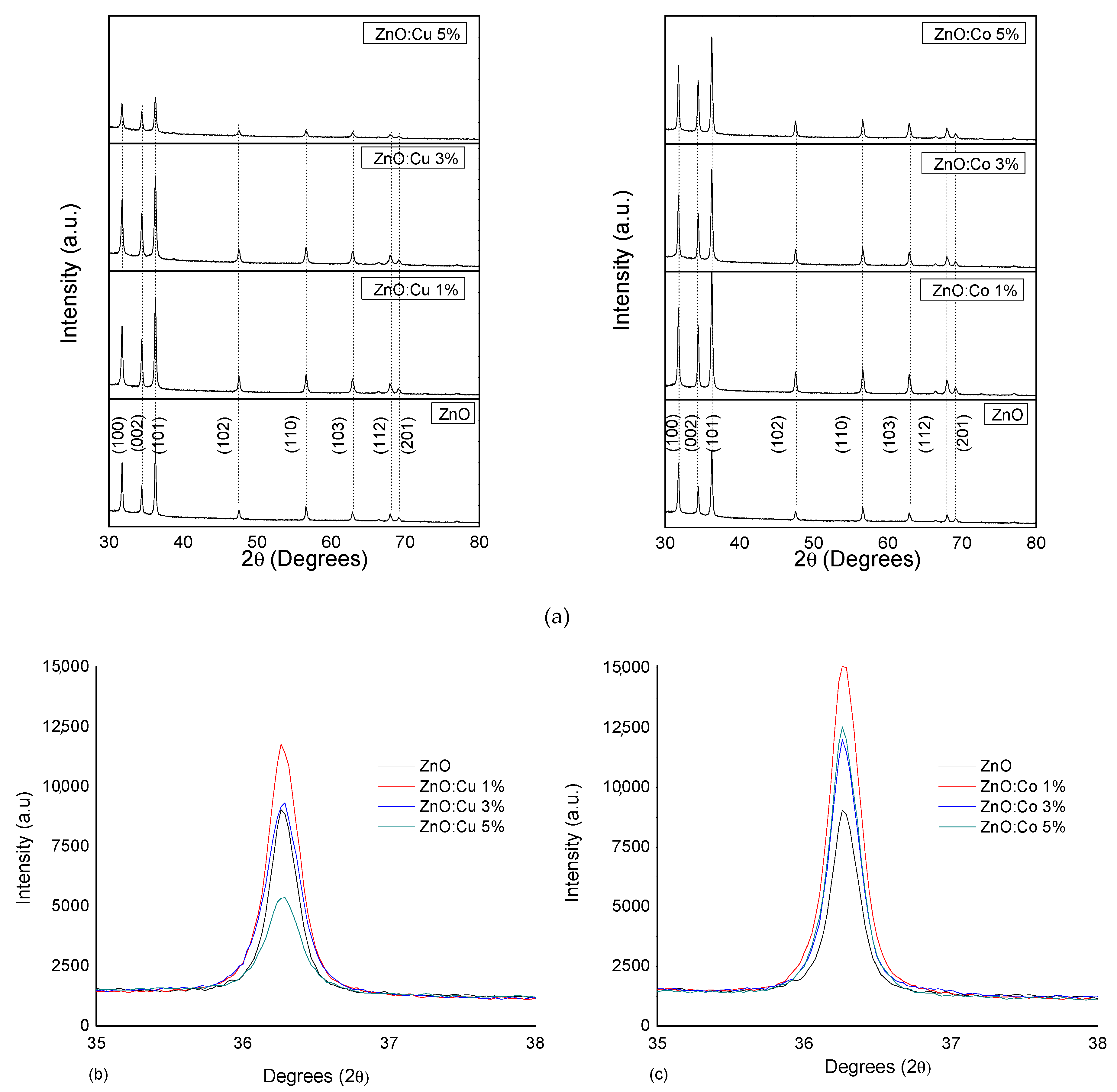


| Thin Film | FWHM * (101) | Intensity (101) Peak | Grain Size (nm) |
|---|---|---|---|
| ZnO | 0.2396 | 8949 | 34.9 |
| ZnO:Co 1% | 0.2615 | 14,997 | 32.0 |
| ZnO:Co 3% | 0.2552 | 11,836 | 32.3 |
| ZnO:Co 5% | 0.2583 | 12,495 | 32.4 |
| ZnO:Cu 1% | 0.2601 | 11,649 | 32.1 |
| ZnO:Cu 3% | 0.2803 | 9202 | 29.8 |
| ZnO:Cu 5% | 0.3075 | 5300 | 27.2 |
| Catalyst | kap × 10−3 (min−1) | Degradation (%) | Band Gap (eV) |
|---|---|---|---|
| ZnO | 0.2 | 2.7 | 3.22 |
| ZnO:Co 1% | 2.6 | 30.4 | 3.17 |
| ZnO:Co 3% | 4.2 | 45.7 | 2.83 |
| ZnO:Co 5% | 7.2 | 62.6 | 2.39 |
| ZnO:Cu 1% | 3.4 | 36.2 | 3.12 |
| ZnO:Cu 3% | 3.4 | 37.7 | 3.07 |
| ZnO:Cu 5% | 4.0 | 42.5 | 3.01 |
| Catalysts/Reference | Pollutant/Molar Concentration | Degradation (%)/Time Test | kap × 10−3 (min−1) |
|---|---|---|---|
| SnS/ZnO [67] | Rhodamine B/5 ppm Methyl Orange/5 ppm | 99%/175 min 82%/125 min | 21.2 13.9 |
| Carbon-ZnO [68] | 2,4-dinitrophenol/25 ppm | 92%/140 min | 18.3 |
| ZnO:Co [41] | Alizarin Red/20 ppm | 93%/60 min | — |
| ZnO:Cu [39] | Methyl Orange/5 ppm | 80%/30 min | 23 |
| ZnO:Ag [69] | Methylene Blue/10 ppm | 65%/140 min | 4.1 |
| ZnO:Co/this work | Methylene Blue/10 ppm | 63%/140 min | 7.2 |
| ZnO:Cu/this work | Methylene Blue/10 ppm | 43%/140 min | 4.0 |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Vallejo, W.; Cantillo, A.; Salazar, B.; Diaz-Uribe, C.; Ramos, W.; Romero, E.; Hurtado, M. Comparative Study of ZnO Thin Films Doped with Transition Metals (Cu and Co) for Methylene Blue Photodegradation under Visible Irradiation. Catalysts 2020, 10, 528. https://doi.org/10.3390/catal10050528
Vallejo W, Cantillo A, Salazar B, Diaz-Uribe C, Ramos W, Romero E, Hurtado M. Comparative Study of ZnO Thin Films Doped with Transition Metals (Cu and Co) for Methylene Blue Photodegradation under Visible Irradiation. Catalysts. 2020; 10(5):528. https://doi.org/10.3390/catal10050528
Chicago/Turabian StyleVallejo, William, Alvaro Cantillo, Briggitte Salazar, Carlos Diaz-Uribe, Wilkendry Ramos, Eduard Romero, and Mikel Hurtado. 2020. "Comparative Study of ZnO Thin Films Doped with Transition Metals (Cu and Co) for Methylene Blue Photodegradation under Visible Irradiation" Catalysts 10, no. 5: 528. https://doi.org/10.3390/catal10050528
APA StyleVallejo, W., Cantillo, A., Salazar, B., Diaz-Uribe, C., Ramos, W., Romero, E., & Hurtado, M. (2020). Comparative Study of ZnO Thin Films Doped with Transition Metals (Cu and Co) for Methylene Blue Photodegradation under Visible Irradiation. Catalysts, 10(5), 528. https://doi.org/10.3390/catal10050528





