UV-Catalyzed Persulfate Oxidation of an Anthraquinone Based Dye
Abstract
1. Introduction
2. Results and Discussion
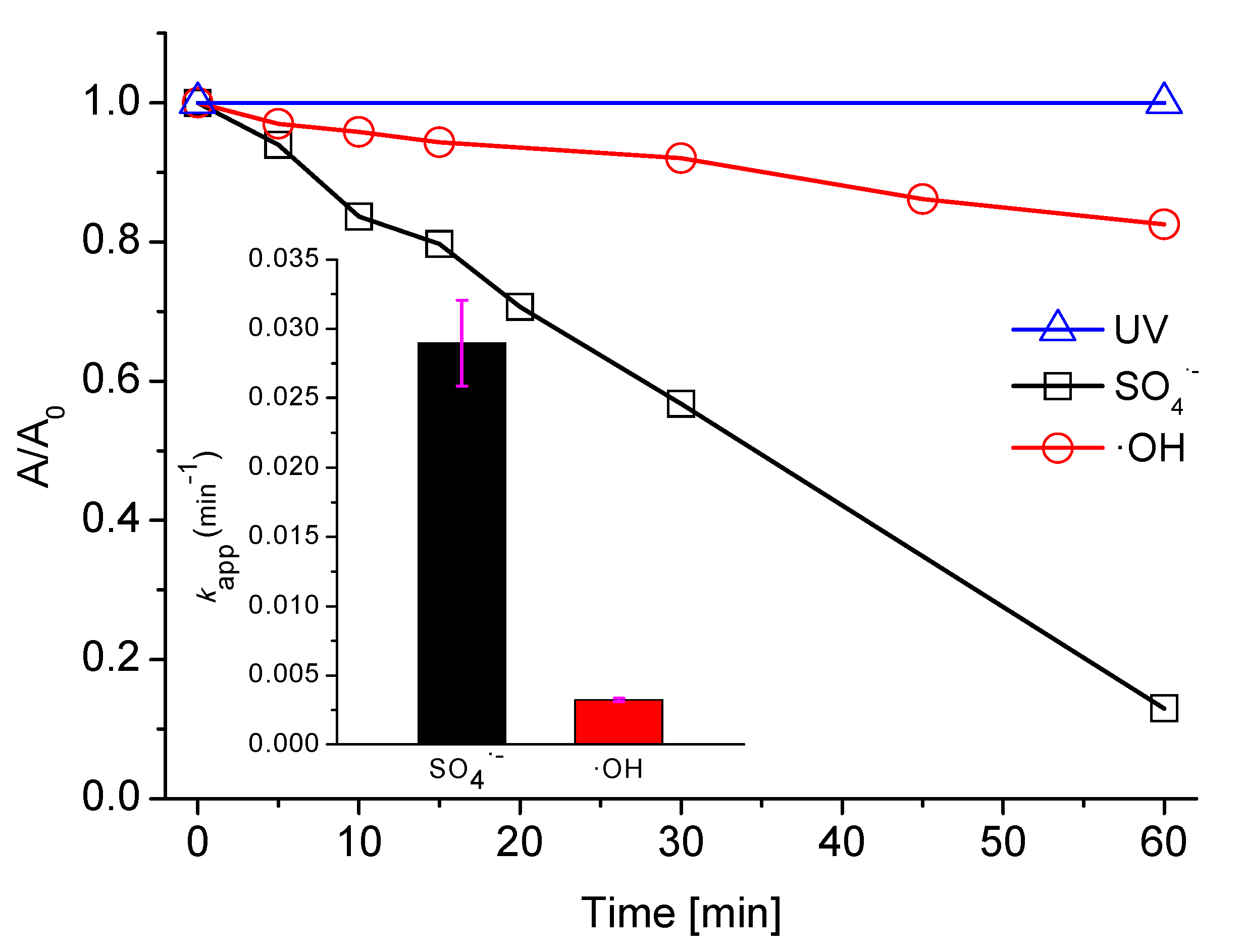
2.1. Influence of •OH and SO4•− on AB129 Decolorization
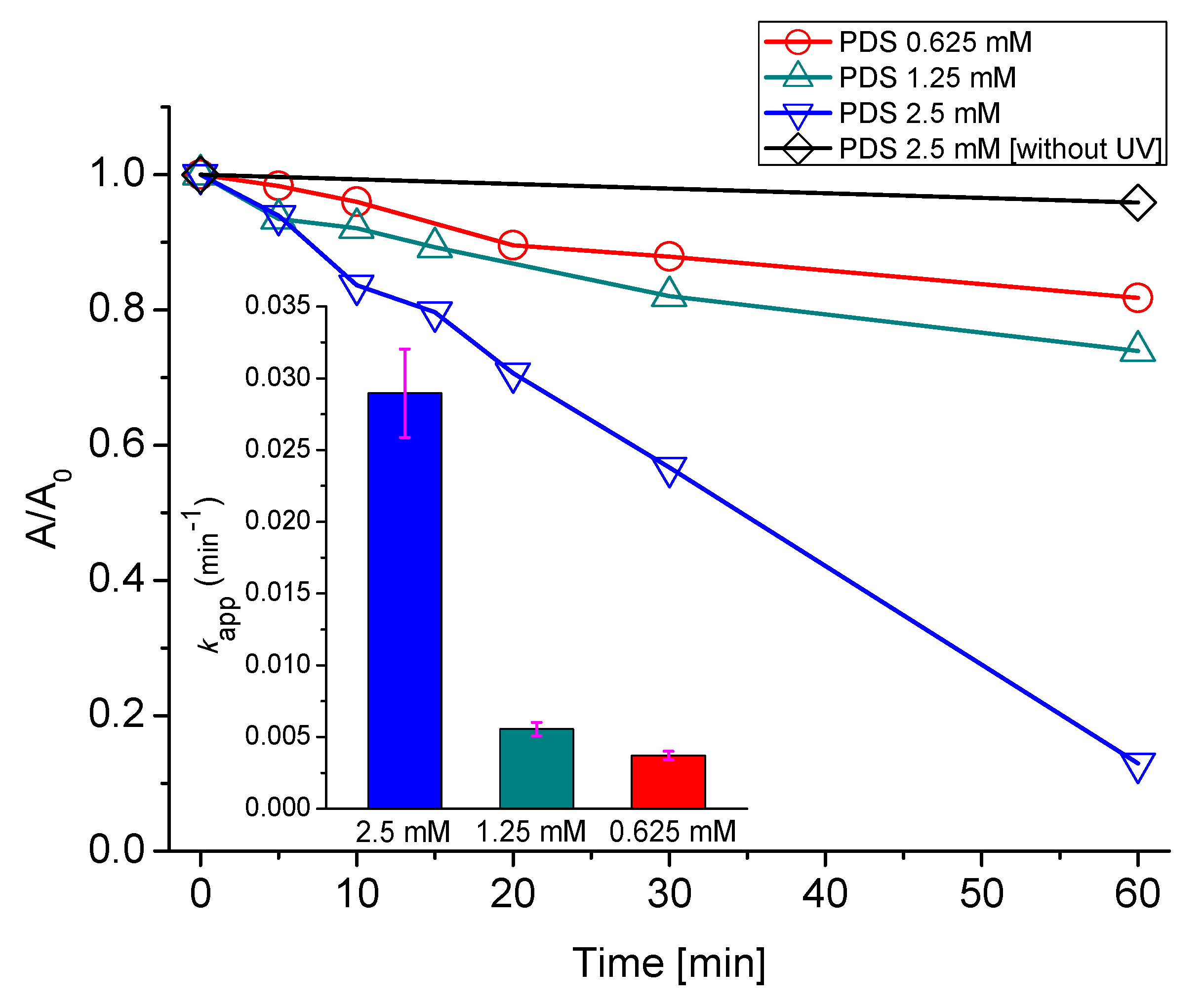
2.2. Effect of PDS Concentration
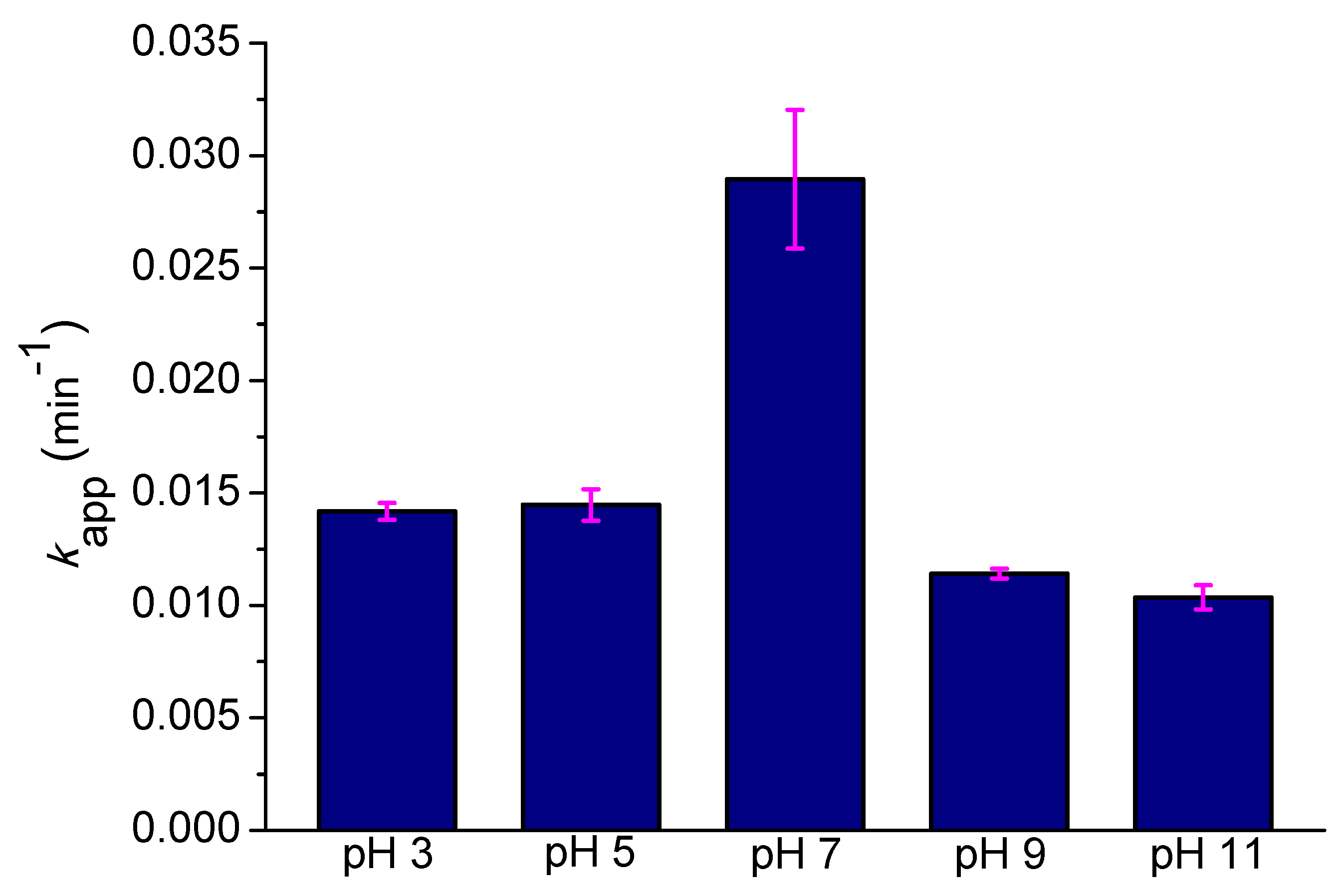
2.3. Effect of the Initial pH
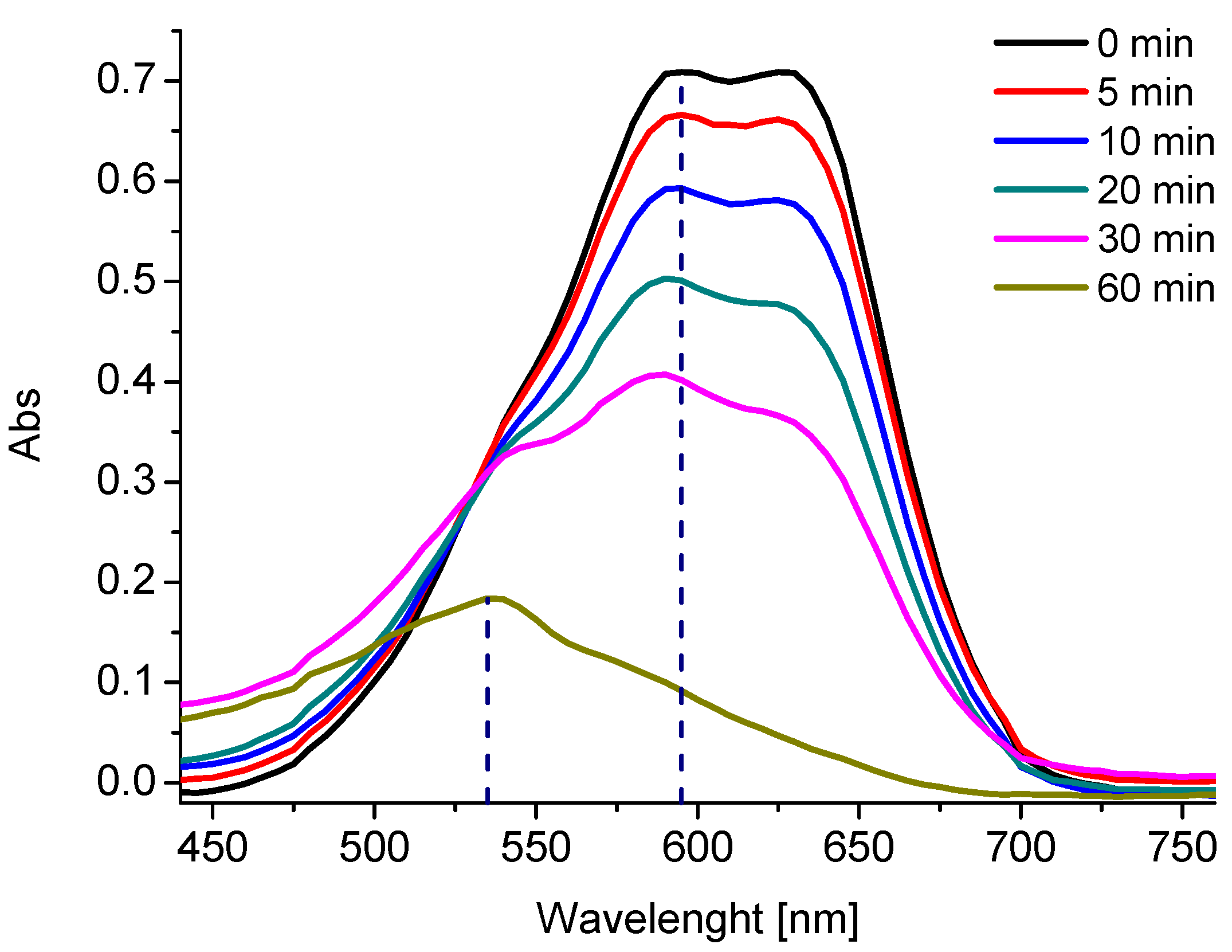
2.4. Formation of by-Products
2.5. Ecotoxicity
3. Materials and Methods
3.1. Chemicals
3.2. Analytical
3.3. Decolorization Test
3.4. Kinetic Test and AB129 Structure Modelling
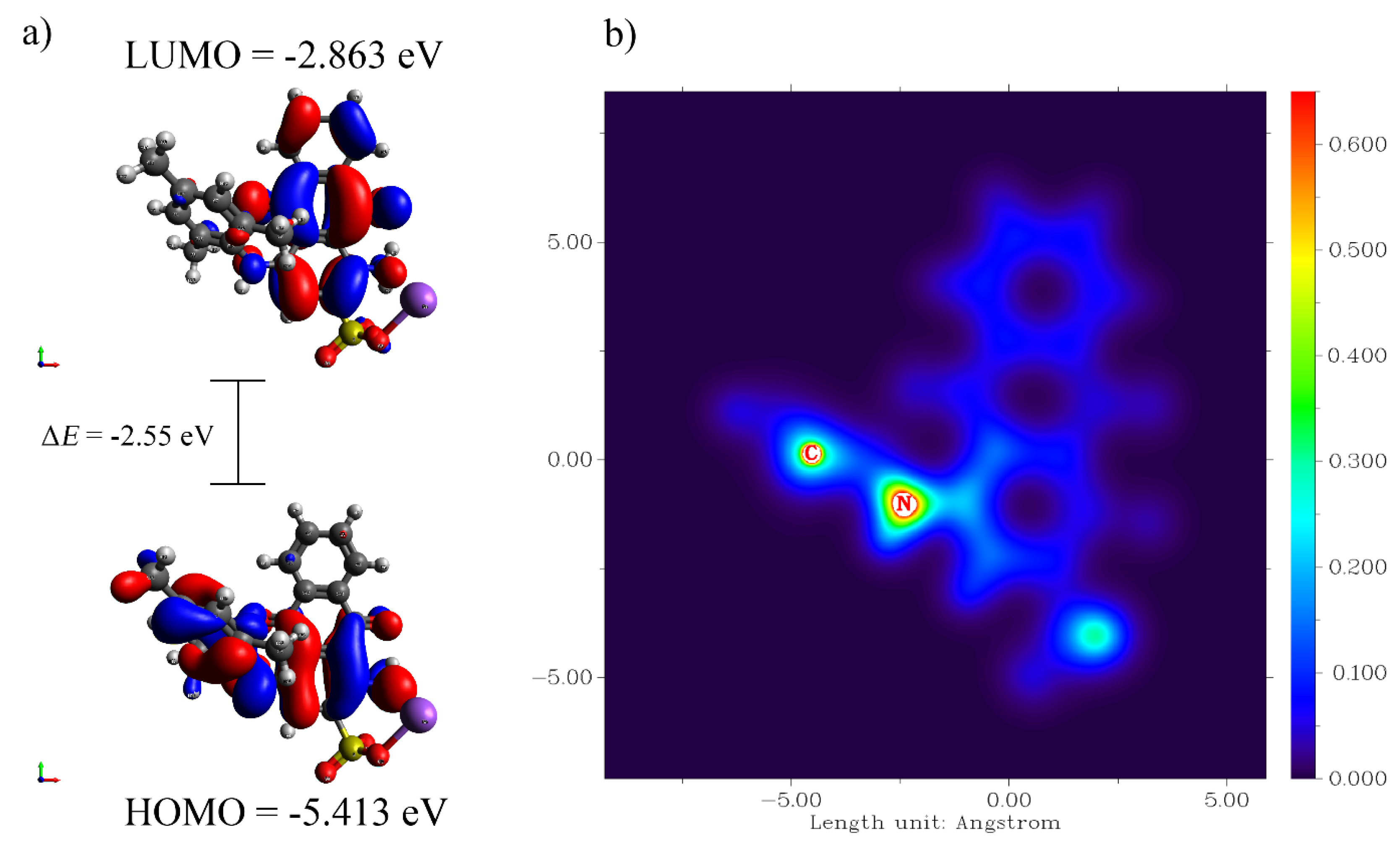
3.5. Quantum Chemical Analysis
3.6. Ecotoxicity Test
4. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Pal, A.; He, Y.; Jekel, M.; Reinhard, M.; Gin, K.Y.H. Emerging contaminants of public health significance as water quality indicator compounds in the urban water cycle. Environ. Int. 2014, 71, 46–62. [Google Scholar] [CrossRef]
- Tsiampalis, A.; Frontistis, Z.; Binas, V.; Kiriakidis, G.; Mantzavinos, D. Degradation of sulfamethoxazole using iron-doped titania and simulated solar radiation. Catalysts 2019, 9, 612. [Google Scholar] [CrossRef]
- Şen, S.; Demirer, G.N. Anaerobic treatment of real textile wastewater with a fluidized bed reactor. Water Res. 2003, 37, 1868–1878. [Google Scholar] [CrossRef]
- Ozturk, E.; Yetis, U.; Dilek, F.B.; Demirer, G.N. A chemical substitution study for a wet processing textile mill in Turkey. J. Clean. Prod. 2009, 17, 239–247. [Google Scholar] [CrossRef]
- Inoue, M.; Okada, F.; Sakurai, A.; Sakakibara, M. A new development of dyestuffs degradation system using ultrasound. Ultrason. Sonochem. 2006, 13, 313–320. [Google Scholar] [CrossRef]
- Eren, Z. Ultrasound as a basic and auxiliary process for dye remediation: A review. J. Environ. Manag. 2012, 104, 127–141. [Google Scholar] [CrossRef]
- Julkapli, N.M.; Bagheri, S.; Hamid, S.B.A. Recent advances in heterogeneous photocatalytic decolorization of synthetic dyes. Sci. World J. 2014, 2014, 692307. [Google Scholar] [CrossRef]
- Ghaly, A.E.; Ananthashankar, R.; Alhattab, M.; Ramakrishnan, V.V. Production, Characterization and Treatment of Textile Effluents: A Critical Review. J. Chem. Eng. Process. Technol. 2014, 5, 1000182. [Google Scholar]
- Chequer, F.D.; de Oliveira, G.A.R.; Ferraz, E.R.A.; Carvalho, J.; Zanoni, M.B.; de Oliveir, D.P. Textile Dyes: Dyeing Process and Environmental Impact. Eco-Friendly Text. Dye. Finish. 2013, 6, 151–176. [Google Scholar]
- Wang, J.; Guo, B.; Zhang, X.; Zhang, Z.; Han, J.; Wu, J. Sonocatalytic degradation of methyl orange in the presence of TiO2 catalysts and catalytic activity comparison of rutile and anatase. Ultrason. Sonochem. 2005, 12, 331–337. [Google Scholar] [CrossRef]
- Wijetunga, S.; Li, X.F.; Jian, C. Effect of organic load on decolourization of textile wastewater containing acid dyes in upflow anaerobic sludge blanket reactor. J. Hazard. Mater. 2010, 177, 792–798. [Google Scholar] [CrossRef]
- Entezari, M.H.; Al-Hoseini, Z.S.; Ashraf, N. Fast and efficient removal of Reactive Black 5 from aqueous solution by a combined method of ultrasound and sorption process. Ultrason. Sonochem. 2008, 15, 433–437. [Google Scholar] [CrossRef] [PubMed]
- Crini, G.; Badot, P.-M. Sorption Processes and Pollution: Conventional and Non-Conventional Sorbents for Pollutant Removal from Wastewaters; Presses universitaires de Franche-Comté: Besançon, France, 2010; ISBN 2848673044. [Google Scholar]
- Puvaneswari, N.; Muthukrishnan, J.; Gunasekaran, P. Toxicity assessment and microbial degradation of azo dyes. Indian J. Exp. Biol. 2006, 44, 618–626. [Google Scholar]
- Oh, S.W.; Kang, M.N.; Cho, C.W.; Lee, M.W. Detection of carcinogenic amines from dyestuffs or dyed substrates. Dye. Pigment. 1997, 33, 119–135. [Google Scholar] [CrossRef]
- Fat’hi, M.R.; Asfaram, A.; Hadipour, A.; Roosta, M. Kinetics and thermodynamic studies for removal of Acid Blue 129 from aqueous solution by almond shell. J. Environ. Health Sci. Eng. 2014, 12, 26. [Google Scholar] [CrossRef] [PubMed]
- Acid Blue 129. Available online: http://datasheets.scbt.com/sc-214468.pdf (accessed on 22 April 2020).
- Pala, A.; Tokat, E. Color removal from cotton textile industry wastewater in an activated sludge system with various additives. Water Res. 2002, 36, 2920–2925. [Google Scholar] [CrossRef]
- Harrelkas, F.; Azizi, A.; Yaacoubi, A.; Benhammou, A.; Pons, M.N. Treatment of textile dye effluents using coagulation-flocculation coupled with membrane processes or adsorption on powdered activated carbon. Desalination 2009, 235, 330–339. [Google Scholar] [CrossRef]
- Cooper, P. Removing colour from dyehouse waste waters—A critical review of technology available. J. Soc. Dye. Colour. 1993, 109, 97–100. [Google Scholar] [CrossRef]
- Fane, A.G.; Fell, C.J.D. A review of fouling and fouling control in ultrafiltration. Desalination 1987, 62, 117–136. [Google Scholar] [CrossRef]
- Georgiou, D.; Melidis, P.; Aivasidis, A. Use of a microbial sensor: Inhibition effect of azo-reactive dyes on activated sludge. Bioprocess Biosyst. Eng. 2002, 25, 79–83. [Google Scholar]
- Katheresan, V.; Kansedo, J.; Lau, S.Y. Efficiency of various recent wastewater dye removal methods: A review. J. Environ. Chem. Eng. 2018, 6, 4676–4697. [Google Scholar] [CrossRef]
- Li, Q.; Wang, L.; Fang, X.; Zhang, L.; Li, J.; Xie, H. Synergistic effect of photocatalytic degradation of hexabromocyclododecane in water by UV/TiO2/persulfate. Catalysts 2019, 9, 189. [Google Scholar] [CrossRef]
- Tehrani, A.R.; Mahmood, N.M.; Arami, M. Study of the efficiency of effective parameters on decolorization of CI. Reactive black 5 wastewater by ozonation. J. Color Sci. Technol. 2008, 2, 67–75. [Google Scholar]
- Muruganandham, M.; Swaminathan, M. Photochemical oxidation of reactive azo dye with UV-H2O2 process. Dye. Pigment. 2004, 62, 269–275. [Google Scholar] [CrossRef]
- Mohey El-Dein, A.; Libra, J.A.; Wiesmann, U. Mechanism and kinetic model for the decolorization of the azo dye Reactive Black 5 by hydrogen peroxide and UV radiation. Chemosphere 2003, 52, 1069–1077. [Google Scholar] [CrossRef]
- Lovato, M.E.; Gilliard, M.B.; Cassano, A.E.; Martín, A.M. Kinetics of the degradation of n-butyl benzyl phthalate using O3/UV, direct photolysis, direct ozonation and UV effects. Environ. Sci. Pollut. Res. 2015, 22, 909–917. [Google Scholar] [CrossRef]
- Tehrani-Bagha, A.R.; Amini, F.L. Decolorization of a Reactive Dye by UV-Enhanced Ozonation. Prog. Col. Color. Coat. 2010, 3, 1–8. [Google Scholar]
- Ghernaout, D. Advanced oxidation phenomena in electrocoagulation process: A myth or a reality? Desalin. Water Treat. 2013, 51, 7536–7554. [Google Scholar] [CrossRef]
- Argun, M.E.; Karatas, M. Application of Fenton process for decolorization of reactive black 5 from synthetic wastewater: Kinetics and thermodynamics. Environ. Prog. Sustain. Energy 2011, 30, 540–548. [Google Scholar] [CrossRef]
- Meriç, S.; Kaptan, D.; Ölmez, T. Color and COD removal from wastewater containing Reactive Black 5 using Fenton’s oxidation process. Chemosphere 2004, 54, 435–441. [Google Scholar] [CrossRef]
- Li, J.; Li, R.; Zou, L.; Liu, X. Efficient degradation of norfloxacin and simultaneous electricity generation in a persulfate-photocatalytic fuel cell system. Catalysts 2019, 9, 835. [Google Scholar] [CrossRef]
- Rickman, K.A.; Mezyk, S.P. Kinetics and mechanisms of sulfate radical oxidation of β-lactam antibiotics in water. Chemosphere 2010, 81, 359–365. [Google Scholar] [CrossRef] [PubMed]
- Olmez-Hanci, T.; Arslan-Alaton, I. Comparison of sulfate and hydroxyl radical based advanced oxidation of phenol. Chem. Eng. J. 2013, 224, 10–16. [Google Scholar] [CrossRef]
- Zheng, J.; Li, J.; Bai, J.; Xiaohantan, X.; Zeng, Q.; Li, L.; Zhou, B. Efficient degradation of refractory organics using sulfate radicals generated directly from WO3 photoelectrode and the catalytic reaction of sulfate. Catalysts 2017, 7, 346. [Google Scholar] [CrossRef]
- Neta, P.; Madhavan, V.; Zemel, H.; Fessenden, R.W. Rate Constants and Mechanism of Reaction of SO4 with Aromatic Compounds. J. Am. Chem. Soc. 1977, 99, 163–164. [Google Scholar] [CrossRef]
- Buxton, G.V.; Greenstock, C.L.; Helman, W.P.; Ross, A.B. Critical Review of rate constants for reactions of hydrated electrons, hydrogen atoms and hydroxyl radicals (⋅OH/⋅O− in Aqueous Solution. J. Phys. Chem. Ref. Data 1988, 17, 513. [Google Scholar] [CrossRef]
- Tang, W.Z. Huren an Photocatalytic degradation kinetics and mechanism of acid blue 40 by TiO2/UV in aqueous solution. Chemosphere 1995, 31, 4171–4183. [Google Scholar] [CrossRef]
- Li, X.; Tang, S.; Yuan, D.; Tang, J.; Zhang, C.; Li, N.; Rao, Y. Improved degradation of anthraquinone dye by electrochemical activation of PDS. Ecotoxicol. Environ. Saf. 2019, 177, 77–85. [Google Scholar] [CrossRef]
- Wacławek, S.; Lutze, H.V.; Grübel, K.; Padil, V.V.T.; Černík, M.; Dionysiou, D.D. Chemistry of persulfates in water and wastewater treatment: A review. Chem. Eng. J. 2017, 330, 44–62. [Google Scholar] [CrossRef]
- Yang, J.; Zeng, Z.; Huang, Z.; Cui, Y. Acceleration of persulfate activation by MIL-101(Fe) with vacuum thermal activation: Effect of FeII/FeIII mixed-valence center. Catalysts 2019, 9, 906. [Google Scholar] [CrossRef]
- Duan, X.; Sun, H.; Kang, J.; Wang, Y.; Indrawirawan, S.; Wang, S. Insights into heterogeneous catalysis of persulfate activation on dimensional-structured nanocarbons. ACS Catal. 2015, 5, 4629–4636. [Google Scholar] [CrossRef]
- Ji, Y.; Shi, Y.; Yang, Y.; Yang, P.; Wang, L.; Lu, J.; Li, J.; Zhou, L.; Ferronato, C.; Chovelon, J.M. Rethinking sulfate radical-based oxidation of nitrophenols: Formation of toxic polynitrophenols, nitrated biphenyls and diphenyl ethers. J. Hazard. Mater. 2019, 361, 152–161. [Google Scholar] [CrossRef] [PubMed]
- Gözmen, B.; Kayan, B.; Gizir, A.M.; Hesenov, A. Oxidative degradations of reactive blue 4 dye by different advanced oxidation methods. J. Hazard. Mater. 2009, 168, 129–136. [Google Scholar] [CrossRef] [PubMed]
- Lizama, C.; Freer, J.; Baeza, J.; Mansilla, H.D. Optimized photodegradation of reactive blue 19 on TiO2 and ZnO suspensions. Catal. Today 2002, 76, 235–246. [Google Scholar] [CrossRef]
- Tehrani-Bagha, A.R.; Mahmoodi, N.M.; Menger, F.M. Degradation of a persistent organic dye from colored textile wastewater by ozonation. Desalination 2010, 260, 34–38. [Google Scholar] [CrossRef]
- Bilal, M.; Rasheed, T.; Iqbal, H.M.N.; Li, C.; Wang, H.; Hu, H.; Wang, W.; Zhang, X. Photocatalytic degradation, toxicological assessment and degradation pathway of C.I. Reactive Blue 19 dye. Chem. Eng. Res. Des. 2018, 129, 384–390. [Google Scholar] [CrossRef]
- Becelic-Tomin, M.; Dalmacija, B.; Rajic, L.; Tomasevic, D.; Kerkez, D.; Watson, M.; Prica, M. Degradation of anthraquinone dye reactive blue 4 in pyrite ash catalyzed fenton reaction. Sci. World J. 2014, 2014, 234654. [Google Scholar] [CrossRef]
- Lovato, M.E.; Fiasconaro, M.L.; Martín, C.A. Degradation and toxicity depletion of RB19 anthraquinone dye in water by ozone-based technologies. Water Sci. Technol. 2017, 75, 813–822. [Google Scholar] [CrossRef]
- Radovic, M.; Mitrovic, J.; Kostic, M.; Bojic, D.; Petrovic, M.; Najdanovic, S.; Bojic, A. Comparison of ultraviolet radiation/hydrogen peroxide, Fenton and photo-Fenton processes for the decolorization of reactive dyes. Hem. Ind. 2015, 69, 657–665. [Google Scholar] [CrossRef]
- Sharma, S.; Patel, S.; Ruparelia, J. Feasibility study on degradation of RR120 dye from water by O3, O3/UV and O3/UV/Persulfate. In In Multi-disciplinary Sustainable Engineering: Current and Future Trends, Proceedings of the 5th Nirma University International Conference on Engineering, Ahmedabad, India, 26–28 November 2015; CRC Press: Boca Raton, FL, USA, 2016; p. 233. [Google Scholar]
- Herrmann, H.; Reese, A.; Zellner, R. Time-resolved UV/VIS diode array absorption spectroscopy of SOx− (x=3, 4, 5) radical anions in aqueous solution. J. Mol. Struct. 1995, 348, 183–186. [Google Scholar] [CrossRef]
- Tan, C.; Gao, N.; Deng, Y.; Zhang, Y.; Sui, M.; Deng, J.; Zhou, S. Degradation of antipyrine by UV, UV/H2O2 and UV/PS. J. Hazard. Mater. 2013, 260, 1008–1016. [Google Scholar] [CrossRef] [PubMed]
- Shah, N.S.; He, X.; Khan, H.M.; Khan, J.A.; O’Shea, K.E.; Boccelli, D.L.; Dionysiou, D.D. Efficient removal of endosulfan from aqueous solution by UV-C/peroxides: A comparative study. J. Hazard. Mater. 2013, 263, 584–592. [Google Scholar] [CrossRef] [PubMed]
- Neta, P.; Huie, R.E.; Ross, A.B. Rate Constants for Reactions of Inorganic Radicals in Aqueous Solution. J. Phys. Chem. Ref. Data 1988, 17, 1027–1284. [Google Scholar] [CrossRef]
- De Laat, J.; Le, T.G. Kinetics and modeling of the Fe(III)/H2O2 system in the presence of sulfate in acidic aqueous solutions. Environ. Sci. Technol. 2005, 39, 1811–1818. [Google Scholar] [CrossRef]
- Yang, Z.; Su, R.; Luo, S.; Spinney, R.; Cai, M.; Xiao, R.; Wei, Z. Comparison of the reactivity of ibuprofen with sulfate and hydroxyl radicals: An experimental and theoretical study. Sci. Total Environ. 2017, 590, 751–760. [Google Scholar] [CrossRef]
- Ghauch, A.; Baalbaki, A.; Amasha, M.; El Asmar, R.; Tantawi, O. Contribution of persulfate in UV-254 nm activated systems for complete degradation of chloramphenicol antibiotic in water. Chem. Eng. J. 2017, 317, 1012–1025. [Google Scholar] [CrossRef]
- Silveira, J.E.; Garcia-Costa, A.L.; Cardoso, T.O.; Zazo, J.A.; Casas, J.A. Indirect decolorization of azo dye Disperse Blue 3 by electro-activated persulfate. Electrochim. Acta 2017, 258, 927–932. [Google Scholar] [CrossRef]
- Lin, H.; Zhang, H.; Hou, L. Degradation of C. I. Acid Orange 7 in aqueous solution by a novel electro/Fe3O4/PDS process. J. Hazard. Mater. 2014, 276, 182–191. [Google Scholar] [CrossRef]
- Liang, C.; Wang, Z.S.; Bruell, C.J. Influence of pH on persulfate oxidation of TCE at ambient temperatures. Chemosphere 2007, 66, 106–113. [Google Scholar] [CrossRef]
- Asghar, A.; Bello, M.M.; Raman, A.A.A.; Daud, W.M.A.W.; Ramalingam, A.; Zain, S.B.M. Predicting the degradation potential of Acid blue 113 by different oxidants using quantum chemical analysis. Heliyon 2019, 5, e02396. [Google Scholar] [CrossRef]
- Lang, A.R. Dyes and Pigments: New Research; Nova Science Publishers: Hauppauge, NY, USA, 2009; ISBN 978-1-60876-195-1. [Google Scholar]
- Cinar, Z. The Role of Molecular Modeling in TiO2 Photocatalysis. Molecules 2017, 22, 556. [Google Scholar] [CrossRef]
- Liu, S.; Rong, C.; Lu, T. Information conservation principle determines electrophilicity, nucleophilicity, and regioselectivity. J. Phys. Chem. A 2014, 118, 3698–3704. [Google Scholar] [CrossRef] [PubMed]
- Xiao, R.; Ye, T.; Wei, Z.; Luo, S.; Yang, Z.; Spinney, R. Quantitative Structure–Activity Relationship (QSAR) for the Oxidation of Trace Organic Contaminants by Sulfate Radical. Environ. Sci. Technol. 2015, 49, 13394–13402. [Google Scholar] [CrossRef] [PubMed]
- Ziegler, P.; Sree, K.S.; Appenroth, K.J. Duckweeds for water remediation and toxicity testing. Toxicol. Environ. Chem. 2016, 98, 1127–1154. [Google Scholar] [CrossRef]
- Žaltauskaitė, J.; Sujetovienė, G.; Čypaitė, A.; Aužbikavičiūtė, A. Lemna Minor as a Tool for Wastewater Toxicity Assessment and Pollutants Removal Agent. In Proceedings of the 9th International Conference on Environmental Engineering, Vilnius, Lithuania, 22–23 May 2014. [Google Scholar]
- Mkandawire, M.; Teixeira Da Silva, J.A.; Dudel, E.G. The lemna bioassay: Contemporary issues as the most standardized plant bioassay for aquatic ecotoxicology. Crit. Rev. Environ. Sci. Technol. 2014, 44, 154–197. [Google Scholar] [CrossRef]
- Sackey, L.N.A.; Kočí, V.; van Gestel, C.A.M. Ecotoxicological effects on Lemna minor and Daphnia magna of leachates from differently aged landfills of Ghana. Sci. Total Environ. 2020, 698, 134295. [Google Scholar] [CrossRef]
- Castro, A.M.; Nogueira, V.; Lopes, I.; Rocha-Santos, T.; Pereira, R. Evaluation of the potential toxicity of effluents from the textile industry before and after treatment. Appl. Sci. 2019, 9, 3804. [Google Scholar] [CrossRef]
- Kudlek, E. Identification of Degradation By-Products of Selected Pesticides during Oxidation and Chlorination Processes. Ecol. Chem. Eng. S 2019, 26, 571–581. [Google Scholar] [CrossRef]
- Neamtu, M.; Siminiceanu, I.; Yediler, A.; Kettrup, A. Kinetics of decolorization and mineralization of reactive azo dyes in aqueous solution by the UV/H2O2 oxidation. Dye. Pigment. 2002, 53, 93–99. [Google Scholar] [CrossRef]
- Palencia, M.; Martínez, J.M.; Arrieta, Á. Removal of Acid Blue 129 dye by Polymer-Enhanced Ultrafiltration (PEUF). J. Sci. Technol. Appl. 2017, 2, 65–74. [Google Scholar] [CrossRef]
- Stumm, W.; Morgan, J.J. Aquatic Chemistry: Chemical Equilibria and Rates in Natural Waters, 3rd ed.; Wiley: Hoboken, NJ, USA, 2012; ISBN 1118591488. [Google Scholar]
- Silvestri, D.; Wacławek, S.; Venkateshaiah, A.; Krawczyk, K.; Sobel, B.; Padil, V.V.T.; Černík, M.; Varma, R.S. Synthesis of Ag nanoparticles by a chitosan-poly(3-hydroxybutyrate) polymer conjugate and their superb catalytic activity. Carbohydr. Polym. 2020, 232, 115806. [Google Scholar] [CrossRef]
- Hanwell, M.D.; Curtis, D.E.; Lonie, D.C.; Vandermeersch, T.; Zurek, E.; Hutchison, G.R. Avogadro: An advanced semantic chemical editor, visualization, and analysis platform. J. Cheminform. 2012, 4, 17. [Google Scholar] [CrossRef]
- Neese, F. The ORCA program system. WIRES Comput. Mol. Sci. 2012, 2, 73–78. [Google Scholar] [CrossRef]
- Lu, T.; Chen, F. Multiwfn: A multifunctional wavefunction analyzer. J. Comput. Chem. 2012, 33, 580–592. [Google Scholar] [CrossRef]
- Fu, R.; Lu, T.; Chen, F.W. Comparing methods for predicting the reactive site of electrophilic substitution. Acta Phys.-Chim. Sin. 2014, 30, 628–639. [Google Scholar]
- Santana, C.M.; Ferrera, Z.S.; Padrón, M.E.T.; Rodríguez, J.J.S. Methodologies for the extraction of phenolic compounds from environmental samples: New approaches. Molecules 2009, 14, 298–320. [Google Scholar] [CrossRef] [PubMed]

| Chemical Formula: C23H19N2NaO5S |  Acid blue 129 structure |
| Name: Sodium-1-amino-4-(2, 4, 6-trimethylanilino) anthraquinone-2-sulfonate | |
| Molecular weight: 458.46 | |
| Melting point: >300 °C |
| Method Used | Dye | Decolorization [%]/Time [h] | Reference |
|---|---|---|---|
| AOP (wet air, wet peroxide, Fenton, photocatalytic,) | Reactive Blue 4 | 100%, 100%, 99%/1 and 100%/0.75 | [45] |
| AOP (TiO2 and ZnO nanoparticles + photodegradation) | Reactive Blue 19 | >95%/0.5 | [46] |
| AOP (ozonation) | Reactive Blue 19 | ~100%/0.3 | [47] |
| AOP (TiO2 + photodegradation) | Reactive Blue 19 | ~75%/3 | [48] |
| AOP (Fenton reaction with pyrite ash) | Reactive Blue 4 | 100%/0.5 | [49] |
| UV radiation and ozonation | Reactive Blue 19 | 100%/0.1 | [50] |
| AOP (Fenton, photo-Fenton), UV radiation | Reactive Blue 19 | 81%, 98%, 42%/0.3 | [51] |
| AOP (Sulfate radical + UV) | Acid Blue 129 | 87%/1 | This work |
| Daphnia Magna | ||||||
|---|---|---|---|---|---|---|
| PDS/AB129 [mM] | Time [min] | |||||
| 0 * | 5 | 10 | 20 | 30 | 45 | |
| Toxicity Effect [%] | ||||||
| 0.2/0.2 | 25 | 40 | 35 | 30 | 25 | 20 |
| 0.5/0.5 | 30 | 40 | 45 | 40 | 30 | 20 |
| 1/1 | 40 | 55 | 65 | 50 | 35 | 25 |
| 2/2 | 50 | 60 | 65 | 55 | 40 | 30 |
| Lemna Minor | ||||||
|---|---|---|---|---|---|---|
| PDS/AB129 [mM] | Time [min] | |||||
| 0 * | 5 | 10 | 20 | 30 | 45 | |
| Toxicity Effect [%] | ||||||
| 0.2/0.2 | 8 | 17 | 8 | 0 | 0 | 0 |
| 0.5/0.5 | 25 | 25 | 25 | 8 | 0 | 0 |
| 1/1 | 33 | 25 | 33 | 17 | 8 | 8 |
| 2/2 | 33 | 33 | 25 | 17 | 17 | 17 |
| Toxicity Effect E% | Toxicity Classification |
|---|---|
| <25 | non-toxic |
| 25–50 | low toxic |
| 50.01–75 | toxic |
| >75 | highly toxic |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Krawczyk, K.; Wacławek, S.; Kudlek, E.; Silvestri, D.; Kukulski, T.; Grübel, K.; Padil, V.V.T.; Černík, M. UV-Catalyzed Persulfate Oxidation of an Anthraquinone Based Dye. Catalysts 2020, 10, 456. https://doi.org/10.3390/catal10040456
Krawczyk K, Wacławek S, Kudlek E, Silvestri D, Kukulski T, Grübel K, Padil VVT, Černík M. UV-Catalyzed Persulfate Oxidation of an Anthraquinone Based Dye. Catalysts. 2020; 10(4):456. https://doi.org/10.3390/catal10040456
Chicago/Turabian StyleKrawczyk, Kamil, Stanisław Wacławek, Edyta Kudlek, Daniele Silvestri, Tomasz Kukulski, Klaudiusz Grübel, Vinod V. T. Padil, and Miroslav Černík. 2020. "UV-Catalyzed Persulfate Oxidation of an Anthraquinone Based Dye" Catalysts 10, no. 4: 456. https://doi.org/10.3390/catal10040456
APA StyleKrawczyk, K., Wacławek, S., Kudlek, E., Silvestri, D., Kukulski, T., Grübel, K., Padil, V. V. T., & Černík, M. (2020). UV-Catalyzed Persulfate Oxidation of an Anthraquinone Based Dye. Catalysts, 10(4), 456. https://doi.org/10.3390/catal10040456











