A Short Review on Ni Based Catalysts and Related Engineering Issues for Methane Steam Reforming
Abstract
1. Introduction
2. Ni Based Powder Catalysts
2.1. Monometallic Catalysts
2.2. Polymetallic Catalysts, Promoters
| Catalysts (Particle Size) | Preparation Procedure | Operative Conditions MSR | Selected Catalyst CH4 conv./vol % Versus Temperature/Pressure (Time on Stream, Deactivation) | Ref. |
|---|---|---|---|---|
| Ni/SiO2Al2O3 (5–8 ± 1.2 nm) | 10 wt % SiO2Al2O3, NiCl2*6H2O, NaOH, ethylene glycol, N2H4*H2O. T = 90 °C. Tcalcination = 900 °C. | WHSV = 1700 mL/g*h, CH4/H2O = 1, T = 650–900 °C. | XCH4 ≈ 100%, T = 750 °C, P = 1-bar (50 h, 0.41%) | [16] |
| Ni/SiO2Al2O3 (18–20 ± 3.1 nm) | 10 wt % SiO2Al2O3, Ni(NO3)2*6H2O, H2O. Tcalcination = 900 °C. | WHSV = 1700 mL/g*h; CH4/H2O = 1; T = 650–900 °C. | XCH4 ≈ 75%, T = 750 °C, P = 1-bar (50 h, 9.2%) XCH4 ≈ 100%, T = 900 °C, P = 1-bar | [16] |
| Ni/MgAl2O4 (0.25–0.30 mm) | MgAl2O4, Ni(NO3)2*6H2O, H2O. Tcalcination = 350 °C, 500 °C, 650 °C, 850 °C, 1000 °C. | WHSV = 0.34 gcath/mol; CH4/H2O/N2 = 1/5/1; T = 600 °C; P = 1–10-bar | XCH4 = 40–50%, T = 600 °C, P = 1-bar XCH4 = 35–45%, T = 600 °C, P = 5-bar XCH4 = 35–45%, T = 600 °C, P = 10-bar | [17] |
| NiAl4O7 (8−10 nm), NiAl2O4 (6−7 nm), Ni2Al2O5 (3−4 nm) | Ni(NO3)2*6H2O, Al(NO3)3*9H2O, ethylene glycol, citric acid, H2O. Tcalcination = 1000 °C. | GHSV = 65,500 h−1; H2O/CH4N2 = 2.4/1/3.4; T = 700 °C; P = 1-bar | NiAl4O7 not active NiAl2O4 XCH4 = 78%, T = 700 °C; P = 1 bar (12 h, 2%) Ni2Al2O5 XCH4 = 82 %, T = 700 °C, P = 1 bar (12 h, 7%) | [18] |
| Ni/ZnLaAlO4 (50–60 nm) Pt/ZnLaAlO4 (50−60 nm) Ru/ZnLaAlO4 (≈40 nm) Ni/ɣ-Al2O3 (50−60 nm) | Ni(NO3)2*6H2O or H2PtCl6*6H2O or Ru(N=O)(NO3)3, ZnLaAlO4 or ɣ-Al2O3, H2O. Tcalcination = 700 °C. | GHSV = 3500,7000 and 10,500 h−1; H2O/CH4/Ar = 3/1/3; T = 600−800 °C; P = 1 bar | Ni/ZnLaAlO4 XCH4 ≈ 72%, T = 700 °C, P = 1 bar (10,500 h−1, 30 h, ≈8%) Pt/ZnLaAlO4, XCH4 ≈ 88% XCH4 ≈ 72%, T = 700 °C, P = 1 bar (10,500 h−1, 30 h, ≈1%) Ru/ZnLaAlO4 XCH4 ≈ 98%, T = 700 °C, P = 1 bar (10,500 h−1, 30 h, ≈0%) | [20] |
| Ni/Y2Zr2O7_x x = HT, CP, GNC | Ni(NO3)2*6H2O, Y2Zr2O7_x, H2O. Tcalcination = 800 °C. x = HT, CP, GNC | WHSV = 72,000 mL/h gcat; H2O/CH4 = 2/1; T = 550–800 °C; P = 1, 20 atm | Ni/Y2Zr2O7_HT XCH4 ≈ 83%, T = 800 °C, P = 1 atm (100 h, 11%) Ni/Y2Zr2O7_CP XCH4 ≈ 60%, T = 800 °C, P = 1 atm (100 h, 15%) Ni/Y2Zr2O7_GNC XCH4 ≈ 98%, T = 800 °C, P = 1 atm (100 h, 0%) | [21] |
| Ni–La0.6Sr0.4FeO3-δ (50 nm) Ni–SrTi0.7Fe0.3O3-δ (25 nm) | Ni(acac)2, acetone, La0.6Sr0.4FeO3-δ or SrTi0.7Fe0.3O3-δ Tcalcination = 600 °C. | H2O/CH4 = 1/1; T = 550–600 °C; P = 1 bar | Ni–SrTi0.7Fe0.3O3-δ XCH4 ≈ 90%, T = 600 °C, P = 1 bar | [22] |
| Ni/γ-Al2O3 (<10 nm) | γ-Al2O3, H2O, Ni(NO3)2*6H2O, butyric acid. Ni/butyric acid 0-1 Tcalcination = 700 °C. | Flow = Natural gas (92 vol % methane and 8 vol % ethane) 5 mL/min., steam (11 mL/min.), and nitrogen (18 mL/min.). | 10Ni/γ-Al2O3 Ni/butyric acid = 0.25 Xnatual gas ≈ 64%, T = 550 °C (1000 min, 0 %) | [23] |
| Ni/Ce0.95M0.05O2-d (M = Zr, Pr, La) (17−35 nm) | Ce0.95M0.05O2-d (M = Zr, Pr, La), H2O, Ni(NO3)2*6H2O. Tcalcination = 600, 750, 900 °C. | Weight/Flow = 1 g min/Nml), CH4 (12.5 %), H2O/CH4 (0.5, 1, 1.5 or 3). | 5% Ni/CeZr5 XCH4 = 72%, T = 600 °C, R = 3. (4 h, ≈12%) | [24] |
| Ni/γ-Al2O3 (<50 nm) | γ-Al2O3, H2O, Ni(NO3)2*6H2O, glycine. Tcalcination = 400 °C | WHSV = 45,000−360,000 scm3g−1h−1, H2O/CH4 = 2/1, T = 650 °C. | CH4 ≈ 10% vol, H2 ≈ 50% vol T = 650 °C (50 h, 0%) | [25] |
| Ni/Ni0.4Mg0.6O (18−28 nm) | Reduction of Ni0.4Mg0.6O with hydrogen. | WHSV = 15,800 cm3g−1h−1, H2O/CH4 = 0.5. | XCH4 ≈ 99%, T = 800 °C. (100 h, 0%) | [26] |
| Ni/Y2B2O7 (B = Ti, Sn, Zr, or Ce) | Ni(NO3)2*6H2O), H2O, Y2B2O7 (B = Ti, Sn, Zr, or Ce) Tcalcination = 800 °C. | WHSV = 36,000 mLh−1gcat−1, H2O/CH4 = 2:1. | Ni/Y2Ti2O4 XCH4 ≈ 85%, T = 750 °C (50 h, ≈ 5%) Ni/Y2Sn2O4 XCH4 ≈ 9%, T = 750 °C (50 h, ≈2%) Ni/Y2Zr2O4 XCH4 ≈ 43%, T = 750 °C (50 h, ≈35%) Ni/Y2Ce2O4 XCH4 ≈ 65%, T = 750 °C (50 h, ≈5%) | [27] |
| mNi/xLa-Si (m = 7.5,12.5,15,17.5,22.5) (x = 0,1.0,2.0,3.0,5.0,7.0) (14–20 nm) | Ni(NO3)2*6H2O, La(NO3)3*6H2O, PEG, H2O, ethylene glycol, nitric acid, TEOS. Tcalcination = 800 °C. | GHSV = 1.584 × 105 mL/gcath, CH4/CO2/H2O = 1/0.4/0.8, T = 730–830 °C, P = 1 atm | 17.5Ni/3.0La-Si XCH4 ≈ 85%, T = 800 °C (60 h, ≈5%) | [28] |
| Ni/Ln2Ti2O7 (Ln = La, Pr, Sm, and Y) (16.6–17.5 nm) | Ni(NO3)2*6H2O, H2O, Ln2Ti2O7 (Ln = La, Pr, Sm, and Y). Tcalcination = 800 °C. | WHSV = 72,000 mL/gh, H2O/CH4 = 2/1, T = 600–800 °C, P = 1 atm. | Ni/Y2Ti2O7 XCH4 ≈ 85%, T = 800 °C (50 h, ≈5%) | [29] |
| yNi/Ce1−XZrXO2-δ (X = 0, 0.15, y = 2,5,10). | Ni(NO3)2*6H2O, H2O, Ce1-XZrXO2-δ (X = 0, 0.15). Tcalcination = 600 °C. | τ = 1 mg min/Nml H2O/CH4 = 1/1, T = 600 °C. | 5Ni/Ce0.85Zr0.15O2-δ XCH4 ≈ 70%, T = 600 °C (4 h) | [30] |
| Ni/γ-Al2O3 (11.4–14.2 nm) | γ-Al2O3, Ni(NO3)2*6H2O, H2O, amine. Ni/Amine = 1/6, Amine = ammonia, ethylamine, diethylamine, triethylamine. | GHSV = 24 × 103 mL/gcat h, H2O/CH4 = 3/1, T = 500–800 °C, | 5Ni/γ-Al2O3 Amine = diethylamine XCH4 ≈ 90%, T = 700 °C (500 min, ≈0%) | [32] |
| Ni/Al2O3 (6–12 nm) | Al2O3, Ni(NO3)2*6H2O, H2O. Tcalcination = 550 °C. | GHSV = 100 mL/min., CH4/CO2 = 1/0.48, H2O/CH4 = 1.2/0.48, 3.5/0.48, 6.1/0.48. T = 600–700 °C. | 10 wt % Ni/Al2O3, XCH4 ≈ 99%, T = 700 °C (20 h, ≈0%) | [33] |
| Catalyst (Particle Size) | Preparation Procedure | Operative Conditions | Selected Catalyst CH4 conv./CH4 conv. Rate Temperature/Pressure (Time on Stream, Deactivation) | Ref |
|---|---|---|---|---|
| Ni-X/α-Al2O3 X = Ru, Rh, Ir (7–30 nm) | α-Al2O3, Ni(NO3)2*6H2O, H2O, Rh(NO3)2*xH2O or IrCl3*6H2O or Ru(NO3)3(NO). Tcalcination = 450 °C. | GHSV = 11,195 h−1; CH4/H2O/H2 = 2/8/0.8; T = 500 °C; P = 30 bar | Ni-Ir/α-Al2O3 CH4 conv. Rate ≈ 0.17 mol/gcat*h Ni-Ru/α-Al2O3 CH4 conv. rate ≈ 0.22 mol/gcat*h Ni-Rh/α-Al2O3 CH4 conv. rate ≈ 0.22 mol/gcat*h | [40] |
| Ni/CaZrO3/α-Al2O3 X = 5,10,15 wt % | α-Al2O3, H2O, Ca(NO3)2*4H2O, ZrO(NO3)2*H2O. Tcalcination = 800 °C. Ni(NO3)2*6H2O, H2O. Tcalcination = 700 °C. | WHSV = 60,000 mL/h g; H2O/CH4/Ar = 1/1/3, 1/3/6 and 3/1/6. | 15Ni/15CaZrO3/α-Al2O3 XCH4 ≈ 67%, T = 700 °C S/C = 1 (10 h, ≈0%) | [41] |
| xPt/15 wt %Ni/MgAl2O4 x = 0–1 wt % (7.6–14.9 nm) | 15 wt %Ni/MgAl2O4, H2PtCl6*6H2O, H2O. Tcalcination = 600 °C. | WHSV = 0.34 gcat*h/mol; CH4/H2O/N2 = 1/5/1 T = 600 °C; P = 1–10 bar | T = 600 °C, P = 1 atm x = 0.00 wt % XCH4 ≈ 48% (8 h, ≈1%) x = 0.01 wt % XCH4 ≈ 50% (8 h, ≈2%) x = 0.05 wt % XCH4 ≈ 59% (8 h, ≈1%) x = 0.1 wt % XCH4 ≈ 62% (8 h, ≈1%) x = 0.3 wt % XCH4 ≈ 63% (8 h, ≈3%) x = 1.0 wt % XCH4 ≈ 19% (8 h, ≈2%) | [42] |
| Ni/Mg/γ-Al2O3 (21.9 nm) | 1.Mg(NO3)2*6H2O, γ-Al2O3 H2O. 2.Ni(NO3)2*6H2O., Mg/ γ-Al2O3 H2O. Tcalcination = 550 °C. | GHSV = 60 L/gh, H2O/CH4 = 1.24, T = 750–850 °C. | XCH4 ≈ 60%, T = 850 °C | [43] |
| Ni/MgAl2O4 (14.9 nm) | 1.Mg(NO3)2*6H2O, Al(NO3)3*9H2O, NaOH, Na2CO3, H2O. 2.Ni(NO3)2*6H2O, MgAl2O4, H2O. Tcalcination = 900 °C. | GHSV = 60 L/gh, H2O/CH4 = 1.24, T = 750–850 °C. | XCH4 ≈ 74%, T = 850 °C | [43] |
| NiMgAlOX (42.1 nm) | Mg(NO3)2*6H2O, Al(NO3)3*9H2O, Ni(NO3)2*6H2O, NaOH, Na2CO3, H2O. Tcalcination= 550 °C. | GHSV = 60 L/gh, H2O/CH4 = 1.24, T = 750–850 °C. | XCH4 ≈ 35%, T = 850 °C | [43] |
| xNi-yLa/α-Al2O3 (x/y = 7/3, 8/2, 9/1) | α-Al2O3, Ni(NO3)2*6H2O, La(NO3)3*6H2O, H2O. Tcalcination = 700 °C. | GHSV = 32 × 103 mL/gcath, CH4/H2O = 1/3, 10% CH4/Ar, T = 500–800 °C. | 7Ni-3La/α-Al2O3 XCH 4 ≈ 87%, T = 600 °C (12 h, ≈0%) | [44] |
| xNi-yCu-tZn/γ-Al2O3 (x = 5,10,15; y = 1,3,5; t = 1,3,5) | γ-Al2O3, Ni(NO3)2*6H2O, Zn (NO3)*6H2O, Cu (NO3)*6H2O, H2O. Tcalcination = 900 °C. | Methane molar rate (mol/min.) = 0.0002719 H2O/CH4 ratio = 4, T = 700°C. | 15Ni-1Cu-5Zn/γ-Al2O3 XCH4 ≈ 95%, T = 700 °C (10 h, ≈3%) | [45] |
2.3. Deactivation Studies
2.4. Kinetics and Simulations
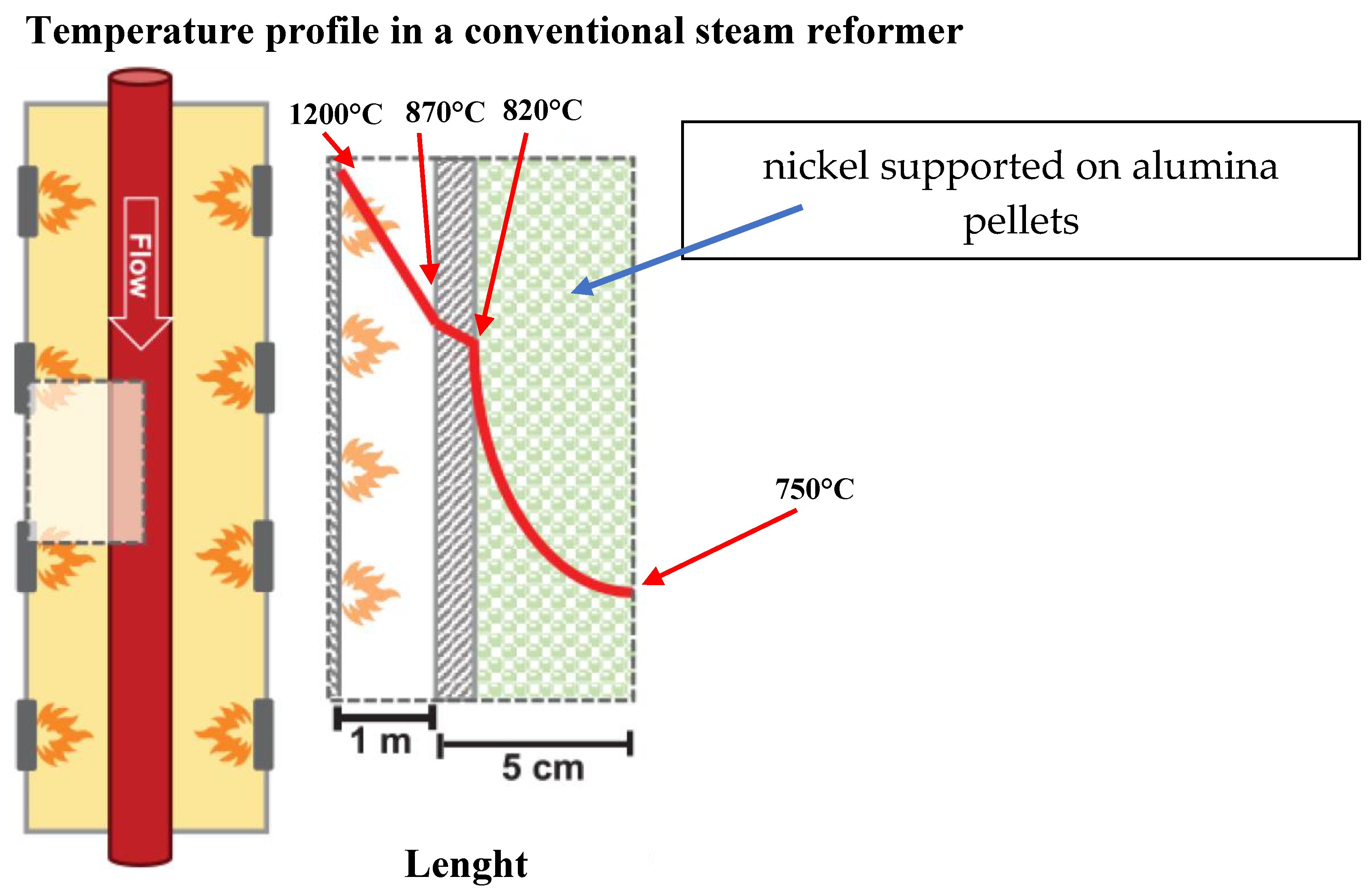
3. Ni Based Structured Catalysts
3.1. Microstructured Catalytic Reactors
3.2. Structured Catalytic Reactors
3.2.1. Metallic Carriers
3.2.2. Ceramic Carriers
4. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Angeli, S.D.; Monteleone, G.; Giaconia, A.; Lemonidou, A.A. State-of-the-art catalysts for CH4 steam reforming at low temperature—Review. Int. J. Hydrogen Energy 2014, 39, 1979–1997. [Google Scholar] [CrossRef]
- Sikander, U.; Sufian, S.; Salam, M.A. A review of hydrotalcite based catalysts for hydrogen production systems. Int. J. Hydrogen Energy 2017, 42, 19851–19868. [Google Scholar] [CrossRef]
- Ma, Y.; Ma, Y.; Long, G.; Li, J.; Hu, X.; Ye, Z.; Wang, Z.; Buckley, C.E.; Dong, D. Synergistic promotion effect of MgO and CeO2 on nanofibrous Ni/Al2O3 catalysts for methane partial oxidation. Fuel 2019, 258, 116103. [Google Scholar] [CrossRef]
- Palma, V.; Miccio, M.; Ricca, A.; Meloni, E.; Ciambelli, P. Monolithic catalysts for methane steam reforming intensification: Experimental and numerical investigations. Fuel 2014, 138, 80–90. [Google Scholar] [CrossRef]
- Alvarez-Galvan, C.; Melian, M.; Ruiz-Matas, L.; Eslava, J.L.; Navarro, R.M.; Ahmadi, M.; Roldan Cuenya, B.; Fierro, J.L.G. Partial Oxidation of Methane to Syngas Over Nickel-Based Catalysts: Influence of Support Type, Addition of Rhodium, and Preparation Method. Front. Chem. 2019, 7, 1–16. [Google Scholar] [CrossRef]
- Pinheiro, A.L.; Pinheiro, A.N.; Valentini, A.; Filho, J.M.; de Sousa, F.F.; de Sousa, J.R.; Rocha, M.; da Graça, C.; Bargiela, P.; Oliveira, A.C. Analysis of coke deposition and study of the structural features of MAl2O4 catalysts for the dry reforming of methane. Catal. Commun. 2009, 11, 11–14. [Google Scholar] [CrossRef]
- Hu, D.; Liu, C.; Li, L.; Lv, K.-L.; Zhang, Y.-H.; Li, J.-L. Carbon dioxide reforming of methane over nickel catalysts supported on TiO2(001) nanosheets. Int. J. Hydrogen Energy 2018, 43, 21345–21354. [Google Scholar] [CrossRef]
- Lercher, J.A.; Bitter, J.H.; Hally, W.; Niessen, W.; Seshan, K. Design of stable catalysts for methane-carbon dioxide reforming. Stud. Surf. Sci. Catal. 1996, 101, 463–472. [Google Scholar] [CrossRef]
- Pinheiro, A.N.; Valentini, A.; Sasaki, J.M.; Oliveira, A.C. Highly stable dealuminated zeolite support for the production of hydrogen by dry reforming of methane. Appl. Catal. A Gen. 2009, 355, 156–168. [Google Scholar] [CrossRef]
- Ramezani, Y.; Meshkani, F.; Rezaei, M. Promotional effect of Mg in trimetallic nickelmanganese-magnesium nanocrystalline catalysts in CO2 reforming of methane. Int. J. Hydrogen Energy 2018, 43, 22347–22356. [Google Scholar] [CrossRef]
- Alrafei, B.; Polaert, I.; Ledoux, A.; Azzolina-Jury, F. Remarkably stable and efficient Ni and Ni-Co catalysts for CO2 methanation. Catal. Today 2019. [Google Scholar] [CrossRef]
- Oton, L.F.; Coelho, D.C.; Oliveira Alcineia, C.; de Araujo, J.C.S.; Lang, R.; Rodríguez-Castellón, E.; Rodríguez-Aguado, E.; Lucredio, A.F.; Assaf, E.M.; Reyna-Alvarado, J.; et al. Structural transformation of vanadate nanotubes into vanadate oxides nanostructures during the dry reforming of methane. Mol. Catal. 2020, 480, 110641. [Google Scholar] [CrossRef]
- Rezaei, R.; Moradi, G. Study of the performance of dry methane reforming in a microchannel reactor using sputtered Ni/Al2O3 coating on stainless steel. Int. J. Hydrogen Energy 2018, 43, 21374–21385. [Google Scholar] [CrossRef]
- Chotirach, M.; Tungasmita, S.; Tungasmita, D.N.; Tantayanon, S. Titanium nitride promoted Ni-based SBA-15 catalyst for dry reforming of methane. Int. J. Hydrogen Energy 2018, 43, 21322–21332. [Google Scholar] [CrossRef]
- Sepehri, S.; Rezaei, M.; Wang, Y.; Younesi, A.; Arandiyan, H. The evaluation of autothermal methane reforming for hydrogen production over Ni/CeO2 catalysts. Int. J. Hydrogen Energy 2018, 43, 22340–22346. [Google Scholar] [CrossRef]
- Ali, S.; Al-Marri, M.J.; Abdelmoneim, A.J.; Kumar, A.; Khader, M.M. Catalytic evaluation of nickel nanoparticles in methane steam reforming. Int. J. Hydrogen Energy 2016, 41, 22876–22885. [Google Scholar] [CrossRef]
- Katheria, S.; Gupta, A.; Deo, G.; Kunzru, D. Effect of calcination temperature on stability and activity of Ni/MgAl2O4 catalyst for steam reforming of methane at high pressure condition. Int. J. Hydrogen Energy 2016, 41, 14123–14132. [Google Scholar] [CrossRef]
- Rogers, J.L.; Mangarella, M.C.; D’Amico, A.D.; Gallagher, J.R.; Dutzer, M.R.; Stavitski, E.; Miller, J.T.; Sievers, C. Differences in the Nature of Active Sites for Methane Dry Reforming and Methane Steam Reforming over Nickel Aluminate Catalysts. ACS Catal. 2016, 6, 5873–5886. [Google Scholar] [CrossRef]
- Pechini, M.P. Method of Preparing Lead and Alkaline Earth Titanates and Niobates and Coating Method Using the Same to Form a Capacitor. U.S. Patent 3,3306,97A, 11 July 1967. [Google Scholar]
- Khani, Y.; Shariatinia, Z.; Bahadoran, F. High catalytic activity and stability of ZnLaAlO4 supported Ni, Pt and Ru nanocatalysts applied in the dry, steam and combined dry-steam reforming of methane. Chem. Eng. J. 2016, 299, 353–366. [Google Scholar] [CrossRef]
- Fang, X.; Zhang, X.; Guo, Y.; Chen, M.; Liu, W.; Xu, X.; Peng, H.; Gao, Z.; Wang, X.; Li, C. Highly active and stable Ni/Y2Zr2O7 catalysts for methane steam reforming: On the nature and effective preparation method of the pyrochlore support. Int. J. Hydrogen Energy 2016, 41, 11141–11153. [Google Scholar] [CrossRef]
- Thalinger, R.; Gocyla, M.; Heggen, M.; Dunin-Borkowski, R.; Grünbacher, M.; Stöger-Pollach, M.; Schmidmair, D.; Klötzer, B.; Penner, S. Ni–perovskite interaction and its structural and catalytic consequences in methane steam reforming and methanation reactions. J. Catal. 2016, 337, 26–35. [Google Scholar] [CrossRef]
- Yoo, J.; Park, S.; Hwan Song, J.; Yoo, S.; Kyu Song, I. Hydrogen production by steam reforming of natural gas over butyric acid-assisted nickel/alumina catalyst. Int. J. Hydrogen Energy 2017, 42, 28377–28385. [Google Scholar] [CrossRef]
- Iglesias, I.; Baronetti, G.; Marino, F. Ni/Ce0.95M0.05O2-d (M = Zr, Pr, La) for methane steam reforming at mild conditions. Int. J. Hydrogen Energy 2017, 42, 29735–29744. [Google Scholar] [CrossRef]
- Aghayan, M.; Potemkin, D.I.; Rubio-Marcos, F.; Uskov, S.I.; Snytnikov, P.V.; Hussainova, I. Template-Assisted Wet-Combustion Synthesis of Fibrous Nickel Based Catalyst for Carbon Dioxide Methanation and Methane Steam Reforming. ACS Appl. Mater. Interfaces 2017, 9, 43553–43562. [Google Scholar] [CrossRef]
- Park, Y.S.; Kang, M.; Byeon, P.; Chung, S.Y.; Nakayama, T.; Ko, T.; Hwang, H. Fabrication of a regenerable Ni supported NiO-MgO catalyst for methane steam reforming by exsolution. J. Power Sources 2018, 397, 318–324. [Google Scholar] [CrossRef]
- Zhang, X.; Peng, L.; Fang, X.; Cheng, Q.; Liu, W.; Peng, H.; Gao, Z.; Zhou, W.; Wang, X. Ni/Y2B2O7 (B=Ti, Sn, Zr and Ce) catalysts for methane steam reforming: On the effects of B site replacement. Int. J. Hydrogen Energy 2018, 43, 8298–8312. [Google Scholar] [CrossRef]
- Chen, C.; Wang, X.; Chen, X.; Liang, X.; Zou, X.; Lu, X. Combined steam and CO2 reforming of methane over one-pot prepared Ni/La-Si catalysts. Int. J. Hydrogen Energy 2019, 44, 4780–4793. [Google Scholar] [CrossRef]
- Fang, X.; Xu, L.; Zhang, X.; Zhang, K.; Dai, H.; Liu, W.; Xu, X.; Wang, X.; Zhou, W. Effect of rare earth element (Ln = La, Pr, Sm, and Y) on physicochemical properties of the Ni/Ln2Ti2O7 catalysts for the steam reforming of methane. Mol. Catal. 2019, 468, 130–138. [Google Scholar] [CrossRef]
- Iglesias, I.; Baronetti, G.; Alemany, L.; Mariño, F. Insight into Ni/Ce1-XZrXO2-δ support interplay for enhanced methane steam reforming. Int. J. Hydrogen Energy 2019, 44, 3668–3680. [Google Scholar] [CrossRef]
- Iglesias, I.; Forti, M.; Baronetti, G.; Mariño, F. Zr-enhanced stability of ceria based supports for methane steam reforming at severe reaction conditions. Int. J. Hydrogen Energy 2019, 44, 8121–8132. [Google Scholar] [CrossRef]
- Sebai, I.; Boulahaouache, A.; Trari, M.; Salhi, N. Preparation and characterization of 5%Ni/γ-Al2O3 catalysts by complexation with NH3 derivatives active in methane steam reforming. Int. J. Hydrogen Energy 2019, 44, 9949–9958. [Google Scholar] [CrossRef]
- Dan, M.; Mihet, M.; Lazar, M.D. Hydrogen and/or syngas production by combined steam and dry reforming of methane on nickel catalysts. Int. J. Hydrogen Energy 2020. [Google Scholar] [CrossRef]
- Hu, Y.H.; Ruckenstein, E. Catalytic Conversion of Methane to Synthesis Gas by Partial Oxidation and CO2 Reforming. Adv. Catal. 2004, 48, 297–345. [Google Scholar] [CrossRef]
- Rostrup-Nielsen, J.R. New aspects of syngas production and use. Catal. Today 2000, 63, 159–164. [Google Scholar] [CrossRef]
- Liu, C.-J.; Ye, J.; Jiang, J.; Pan, Y. Progresses in the Preparation of Coke Resistant Ni-based Catalyst for Steam and CO2 Reforming of Methane. ChemCatChem 2011, 3, 529–541. [Google Scholar] [CrossRef]
- Ligthart, D.A.J.M.; Pieterse, J.A.Z.; Hensen, E.J.M. The role of promoters for Ni catalysts in low temperature (membrane) steam methane reforming. Appl. Cat. A Gen. 2011, 405, 108–119. [Google Scholar] [CrossRef]
- Weststrate, C.J.; Saib, A.M.; Niemantsverdriet, J.W. Promoter segregation in Pt and Ru promoted cobalt model catalysts during oxidation–reduction treatments. Catal. Today 2013, 215, 2–7. [Google Scholar] [CrossRef]
- Available online: https://www.britannica.com/science/promoter-catalysis (accessed on 14 March 2020).
- Morales-Cano, F.; Lundegaard, L.F.; Tiruvalam, R.R.; Falsig, H.; Skjøth-Rasmussen, M.S. Improving the sintering resistance of Ni/Al2O3 steam-reforming catalysts by promotion with noble metals. Appl. Catal. A Gen. 2015, 498, 117–125. [Google Scholar] [CrossRef]
- Lertwittayanon, K.; Youravong, W.; Jye Lau, W. Enhanced catalytic performance of Ni/α-Al2O3 catalyst modified with CaZrO3 nanoparticles in steam-methane reforming. Int. J. Hydrogen Energy 2017, 42, 28254–28265. [Google Scholar] [CrossRef]
- Jaiswar, V.K.; Katheria, S.; Deo, G.; Kunzru, D. Effect of Pt doping on activity and stability of Ni/MgAl2O4 catalyst for steam reforming of methane at ambient and high pressure condition. Int. J. Hydrogen Energy 2017, 42, 18968–18976. [Google Scholar] [CrossRef]
- Azancot, L.; Bobadilla, L.F.; Santos, J.L.; Cordoba, J.M.; Centeno, M.A.; Odriozola, J.A. Influence of the preparation method in the metal-support interaction and reducibility of Ni-Mg-Al based catalysts for methane steam reforming. Int. J. Hydrogen Energy 2019, 44, 19827–19840. [Google Scholar] [CrossRef]
- Boudjeloud, M.; Boulahouache, A.; Rabia, C.; Salhi, N. La-doped supported Ni catalysts for steam reforming of methane. Int. J. Hydrogen Energy 2019, 44, 9906–9913. [Google Scholar] [CrossRef]
- Nazari, M.; Mehdi Alavi, S. An investigation of the simultaneous presence of Cu and Zn in different Ni/Al2O3 catalyst loads using Taguchi design of experiment in steam reforming of methane. Int. J. Hydrogen Energy 2020, 45, 691–702. [Google Scholar] [CrossRef]
- Phanawadee, P.; Laipraseard, K.; Yablonsky, G.S.; Constales, D.; Jamroonrote, W.; Jaipet, P. Estimation of the remaining lifetime of deactivated catalyst via the spatial average catalyst activity illustrated by the water–gas shift and steam methane reforming processes. Reac. Kinet. Mech. Cat. 2017, 121, 371–385. [Google Scholar] [CrossRef]
- Argyle, M.D.; Bartholomew, C.H. Heterogeneous Catalyst Deactivation and Regeneration: A Review. Catalysts 2015, 5, 145–269. [Google Scholar] [CrossRef]
- Ochoa, A.; Bilbao, J.; Gayubo, A.G.; Castano, P. Coke formation and deactivation during catalytic reforming of biomass and waste pyrolysis products: A review. Renew. Sustain. Energy Rev. 2020, 119, 109600. [Google Scholar] [CrossRef]
- Hashemnejad, S.M.; Parvari, M. Deactivation and Regeneration of Nickel-Based Catalysts for Steam-Methane Reforming. Chin. J. Catal. 2011, 32, 273–279. [Google Scholar] [CrossRef]
- Jablonski, W.S.; Villano, S.M.; Dean, A.M. A comparison of H2S, SO2, and COS poisoning on Ni/YSZ and Ni/K2O-CaAl2O4 during methane steam and dry reforming. Appl. Cat. A General 2015, 502, 399–409. [Google Scholar] [CrossRef]
- Yang, X. An experimental investigation on the deactivation and regeneration of a steam reforming catalyst. Renew. Energy 2017, 112, 17–24. [Google Scholar] [CrossRef]
- Laprune, D.; Theodoridi, C.; Tuel, A.; Farrusseng, D.; Meunier, F.C. Effect of polyaromatic tars on the activity for methane steam reforming of nickel particles embedded in silicalite-1. Appl. Catal. B Environ. 2017, 204, 515–524. [Google Scholar] [CrossRef]
- Li, S.; Burel, L.; Aquino, C.; Tuel, A.; Morfin, F.; Rousset, J.-L.; Farrusseng, D. Ultimate size control of encapsulated gold nanoparticles. Chem. Commun. 2013, 49, 8507–8509. [Google Scholar] [CrossRef] [PubMed]
- Haynes, D.J.; Shekhawat, D.; Berry, D.; Roy, A.; Spivey, J.J. Effect of calcination temperature on steam reforming activity of Ni-based pyrochlore catalysts. J. Rare Earths 2019. [Google Scholar] [CrossRef]
- Hernandez, A.D.; Kaisalo, N.; Simell, P.; Scarsella, M. Effect of H2S and thiophene on the steam reforming activity of nickel and rhodium catalysts in a simulated coke oven gas stream. Appl. Cat. B Environ. 2019, 258, 117977. [Google Scholar] [CrossRef]
- Turchetti, L.; Murmura, M.A.; Monteleone, G.; Giaconia, A.; Lemonidou, A.A.; Angeli, S.D.; Palma, V.; Ruocco, C.; Annesini, M.C. Kinetic assessment of Ni-based catalysts in low-temperature methane/biogas steam reforming. Int. J. Hydrogen Energy 2016, 41, 16865–16877. [Google Scholar] [CrossRef]
- Kechagiopoulos, P.N.; Angeli, S.D.; Lemonidouba, A.A. Low temperature steam reforming of methane: A combined isotopic and microkinetic study. Appl. Cat. B Environ. 2017, 205, 238–253. [Google Scholar] [CrossRef]
- Mundhwa, M.; Thurgood, C.P. Methane steam reforming at low steam to carbon ratios over alumina and yttria-stabilized-zirconia supported nickel-spinel catalyst: Experimental study and optimization of microkinetic model. Fuel Process. Technol. 2017, 168, 27–39. [Google Scholar] [CrossRef]
- German, E.D.; Sheintuch, M. Methane steam reforming rates over Pt, Rh and Ni(111) accounting for H tunneling and for metal lattice vibrations. Surf. Sci. 2017, 656, 126–139. [Google Scholar] [CrossRef]
- Thallam Thattai, A.; van Biert, L.; Aravind, P.V. On direct internal methane steam reforming kinetics in operating solid oxide fuel cells with nickel-ceria anodes. J. Power Sources 2017, 370, 71–86. [Google Scholar] [CrossRef]
- Che, F.; Gray, J.T.; Ha, S.; McEwen, J.-S. Reducing Reaction Temperature, Steam Requirements, and Coke Formation During Methane Steam Reforming Using Electric Fields: A Microkinetic Modeling and Experimental Study. ACS Catal. 2017, 7, 6957–6968. [Google Scholar] [CrossRef]
- Khzouz, M.; Gkanas, E.I. Experimental and Numerical Study of Low Temperature Methane Steam Reforming for Hydrogen Production. Catalysts 2018, 8, 5. [Google Scholar] [CrossRef]
- Ji, G.; Zhao, M.; Wang, G. Computational fluid dynamic simulation of a sorption-enhanced palladium membrane reactor for enhancing hydrogen production from methane steam reforming. Energy 2018, 147, 884–895. [Google Scholar] [CrossRef]
- De Oliveira Rocha, K.; Marques, C.M.P.; Correa Bueno, J.M. Effect of Au doping of Ni/Al2O3 catalysts used in steam reforming of methane: Mechanism, apparent activation energy, and compensation effect. Chem. Eng. Sci. 2019, 207, 844–852. [Google Scholar] [CrossRef]
- Pashchenko, D. Combined methane reforming with a mixture of methane combustion products and steam over a Ni-based catalyst: An experimental and thermodynamic study. Energy 2019, 185, 573–584. [Google Scholar] [CrossRef]
- Pashchenko, D. Numerical study of steam methane reforming over a pre-heated Ni-based catalyst with detailed fluid dynamics. Fuel 2019, 236, 686–694. [Google Scholar] [CrossRef]
- Unruean, P.; Plianwong, T.; Pruksawan, S.; Kitiyanan, B.; Ziff, R.M. Kinetic Monte-Carlo Simulation of Methane Steam Reforming over a Nickel Surface. Catalysts 2019, 9, 946. [Google Scholar] [CrossRef]
- Liu, L.; Hong, D.; Guo, X. Insight into CaO addition on coking resistance of Ni surface for sorption enhanced methane steam reforming: A density functional study. Appl. Surf. Sci. 2019, 475, 887–895. [Google Scholar] [CrossRef]
- Vogt, C.; Kranenborg, J.; Monai, M.; Weckhuysen, B.M. Structure Sensitivity in Steam and Dry Methane Reforming over Nickel: Activity and Carbon Formation. ACS Catal. 2020, 10, 1428–1438. [Google Scholar] [CrossRef]
- Chen, K.; Zhao, Y.; Zhang, W.; Feng, D.; Sun, S. The intrinsic kinetics of methane steam reforming over a nickel-based catalyst in a micro fluidized bed reaction system. Int. J. Hydrogen Energy 2020, 45, 1615–1628. [Google Scholar] [CrossRef]
- Karthik, G.M.; Buwa, V. Particle-Resolved Simulations of Methane Steam Reforming in Multilayered Packed Beds. AIChE J. 2018, 64, 4162–4176. [Google Scholar] [CrossRef]
- Pashchenko, D. Experimental investigation of reforming and flow characteristics of a steam methane reformer filled with nickel catalyst of various shapes. Energy Convers. Manag. 2019, 185, 465–472. [Google Scholar] [CrossRef]
- Park, H.-G.; Han, S.-Y.; Jun, K.-W.; Woo, Y.; Park, M.-J.; Kim, S.K. Bench-Scale Steam Reforming of Methane for Hydrogen Production. Catalysts 2019, 9, 615. [Google Scholar] [CrossRef]
- Kim, Y.; Cho, E.; Ko, C.H. Preparation of Ni-based egg-shell-type catalyst on cylinder-shaped alumina pellets and its application for hydrogen production via steam methane reforming. Int. J. Hydrogen Energy 2019, 44, 5314–5323. [Google Scholar] [CrossRef]
- Fukuhara, C.; Yamamoto, K.; Makiyama, Y.; Kawasaki, W.; Watanabe, R. A metal-honeycomb-type structured catalyst for steam reforming of methane: Effect of preparation condition change on reforming performance. Appl. Catal. A Gen. 2015, 492, 190–200. [Google Scholar] [CrossRef]
- Gouveia Gil, A.; Wu, Z.; Chadwick, D.; Li, K. Microstructured Catalytic Hollow Fiber Reactor for Methane Steam Reforming. Ind. Eng. Chem. Res. 2015, 54, 5563–5571. [Google Scholar] [CrossRef]
- Zhang, N.; Chen, X.; Chu, B.; Cao, C.; Jin, Y.; Cheng, Y. Catalytic performance of Ni catalyst for steam methane reforming in a micro-channel reactor at high pressure. Chem. Eng. Process. Process. Intensif. 2017, 118, 19–25. [Google Scholar] [CrossRef]
- Calisan, A.; Ogulgonen, C.G.; Yilmaz, A.; Uner, D.; Kincal, S. Steam methane reforming over structured reactors under concentrated solar irradiation. Int. J. Hydrogen Energy 2019, 44, 18682–18693. [Google Scholar] [CrossRef]
- Wismann, S.T.; Engbæk, J.S.; Vendelbo, S.B.; Bendixen, F.B.; Eriksen, W.L.; Aasberg-Petersen, K.; Frandsen, C.; Chorkendor, I.; Mortensen, P.M. Electrified methane reforming: A compact approach to greener industrial hydrogen production. Science 2019, 364, 756–759. [Google Scholar] [CrossRef]
- Kumar, A.; Baldea, M.; Edgar, T.F. A physics-based model for industrial steam-methane reformer optimization with non-uniform temperature field. Comput. Chem. Eng. 2017, 105, 224–236. [Google Scholar] [CrossRef]
- Govender, S.; Friedrich, H.B. Monoliths: A Review of the Basics, Preparation Methods and Their Relevance to Oxidation (review). Catalysts 2017, 7, 62. [Google Scholar] [CrossRef]
- Xu, Y.; Ma, Y.; Demura, M.; Hirano, T. Enhanced catalytic activity of Ni3Al foils towards methane steam reforming by water vapor and hydrogen pretreatments. Int. J. Hydrogen Energy 2016, 41, 7352–7362. [Google Scholar] [CrossRef]
- Mundhwa, M.; Parmar, R.D.; Thurgood, C.P. A comparative parametric study of a catalytic plate methane reformer coated with segmented and continuous layers of combustion catalyst for hydrogen production. J. Power Sources 2017, 344, 85–102. [Google Scholar] [CrossRef]
- Mundhwa, M.; Thurgood, C.P. Numerical study of methane steam reforming and methane combustion over the segmented and continuously coated layers of catalysts in a plate reactor. Fuel Process. Technol. 2017, 158, 57–72. [Google Scholar] [CrossRef]
- Hirano, T.; Xu, Y. Catalytic properties of a pure Ni coil catalyst for methane steam reforming. Int. J. Hydrogen Energy 2017, 42, 30621–30629. [Google Scholar] [CrossRef]
- Xu, Y.; Harimoto, T.; Hirano, T.; Ohata, H.; Kunieda, H.; Hara, Y.; Miyata, Y. Catalytic performance of a high-cell-density Ni honeycomb catalyst for methane steam reforming. Int. J. Hydrogen Energy 2018, 43, 15975–15984. [Google Scholar] [CrossRef]
- Xu, Y.; Harimoto, T.; Wang, L.; Hirano, T.; Kunieda, H.; Hara, Y.; Miyata, Y. Effect of steam and hydrogen treatments on the catalytic activity of pure Ni honeycomb for methane steam reforming. Chem. Eng. Process. Process. Intensif. 2018, 129, 63–70. [Google Scholar] [CrossRef]
- Settar, A.; Abboudi, S.; Lebaal, N. Effect of inert metal foam matrices on hydrogen production intensification of methane steam reforming process in wall-coated reformer. Int. J. Hydrogen Energy 2018, 43, 12386–12397. [Google Scholar] [CrossRef]
- Ashraf, M.A.; Sanz, O.; Montes, M.; Specchia, S. Insights into the effect of catalyst loading on methane steam reforming and controlling regime for metallic catalytic monoliths. Int. J. Hydrogen Energy 2018, 43, 11778–11792. [Google Scholar] [CrossRef]
- Shigarov, A.B.; Кirillov, V.A.; Аmosov, Y.I.; Brayko, A.S.; Avakov, V.B.; Landgraf, I.К.; Urusov, A.R.; Jivulko, S.A.; Izmaylovich, V.V. Membrane reformer module with Ni-foam catalyst for pure hydrogen production from methane: Experimental demonstration and modelling. Int. J. Hydrogen Energy 2017, 42, 6713–6728. [Google Scholar] [CrossRef]
- Pajak, M.; Mozdzierz, M.; Chalusiak, M.; Kimijima, S.; Szmyd, J.S.; Brus, G. A numerical analysis of heat and mass transfer processes in a macro-patterned methane/steam reforming reactor. Int. J. Hydrogen Energy 2018, 43, 20474–20487. [Google Scholar] [CrossRef]
- Katheria, S.; Deo, G.; Kunzru, D. Rh-Ni/MgAl2O4 catalyst for steam reforming of methane: Effect of Rh doping, calcination temperature and its application on metal monoliths. Appl. Catal. A 2019, 570, 308–318. [Google Scholar] [CrossRef]
- Balzarotti, R.; Ambrosetti, M.; Beretta, A.; Groppi, G.; Tronconi, E. Investigation of packed conductive foams as a novel reactor configuration for methane steam reforming. Chem. Eng. J. 2019. [Google Scholar] [CrossRef]
- Palma, V.; Ricca, A.; Meloni, E.; Martino, M.; Miccio, M.; Ciambelli, P. Experimental and numerical investigations on structured catalysts for methane steam reforming intensification. J. Clean. Prod. 2016, 111, 217–230. [Google Scholar] [CrossRef]
- Ricca, A.; Palma, V.; Martino, M.; Meloni, E. Innovative catalyst design for methane steam reforming intensification. Fuel 2017, 198, 175–182. [Google Scholar] [CrossRef]
- Palma, V.; Ricca, A.; Martino, M.; Meloni, E. Innovative structured catalytic systems for methane steam reforming intensification. Chem. Eng. Process. Process. Intensif. 2017, 120, 207–215. [Google Scholar] [CrossRef]
- Palma, V.; Martino, M.; Meloni, E.; Ricca, A. Novel structured catalysts configuration for intensification of steam reforming of methane. Int. J. Hydrogen Energy 2017, 42, 1629–1638. [Google Scholar] [CrossRef]
- Noh, Y.S.; Lee, K.-Y.; Moon, D.J. Hydrogen production by steam reforming of methane over nickel based structured catalysts supported on calcium aluminate modified SiC. Int. J. Hydrogen Energy 2019, 44, 21010–21019. [Google Scholar] [CrossRef]
- Ashraf, M.A.; Sanz, O.; Italiano, C.; Vita, A.; Montes, M.; Specchia, S. Analysis of Ru/La-Al2O3 catalyst loading on alumina monoliths and controlling regimes in methane steam reforming. Chem. Eng. J. 2018, 334, 1792–1807. [Google Scholar] [CrossRef]
- Inbamrung, P.; Sornchamni, T.; Prapainainar, C.; Tungkamani, S.; Narataruksa, P.; Jovanovic, G.N. Modeling of a square channel monolith reactor for methane steam reforming. Energy 2018, 152, 383–400. [Google Scholar] [CrossRef]
- Jeong, A.; Shin, D.; Baek, S.M.; Nam, J.H. Effectiveness factor correlations from simulations of washcoat nickel catalyst layers for small-scale steam methane reforming applications. Int. J. Hydrogen Energy 2018, 43, 15398–15411. [Google Scholar] [CrossRef]
- Leonzio, G. ANOVA analysis of an integrated membrane reactor for hydrogen production by methane steam reforming. Int. J. Hydrogen Energy 2019, 44, 11535–11545. [Google Scholar] [CrossRef]
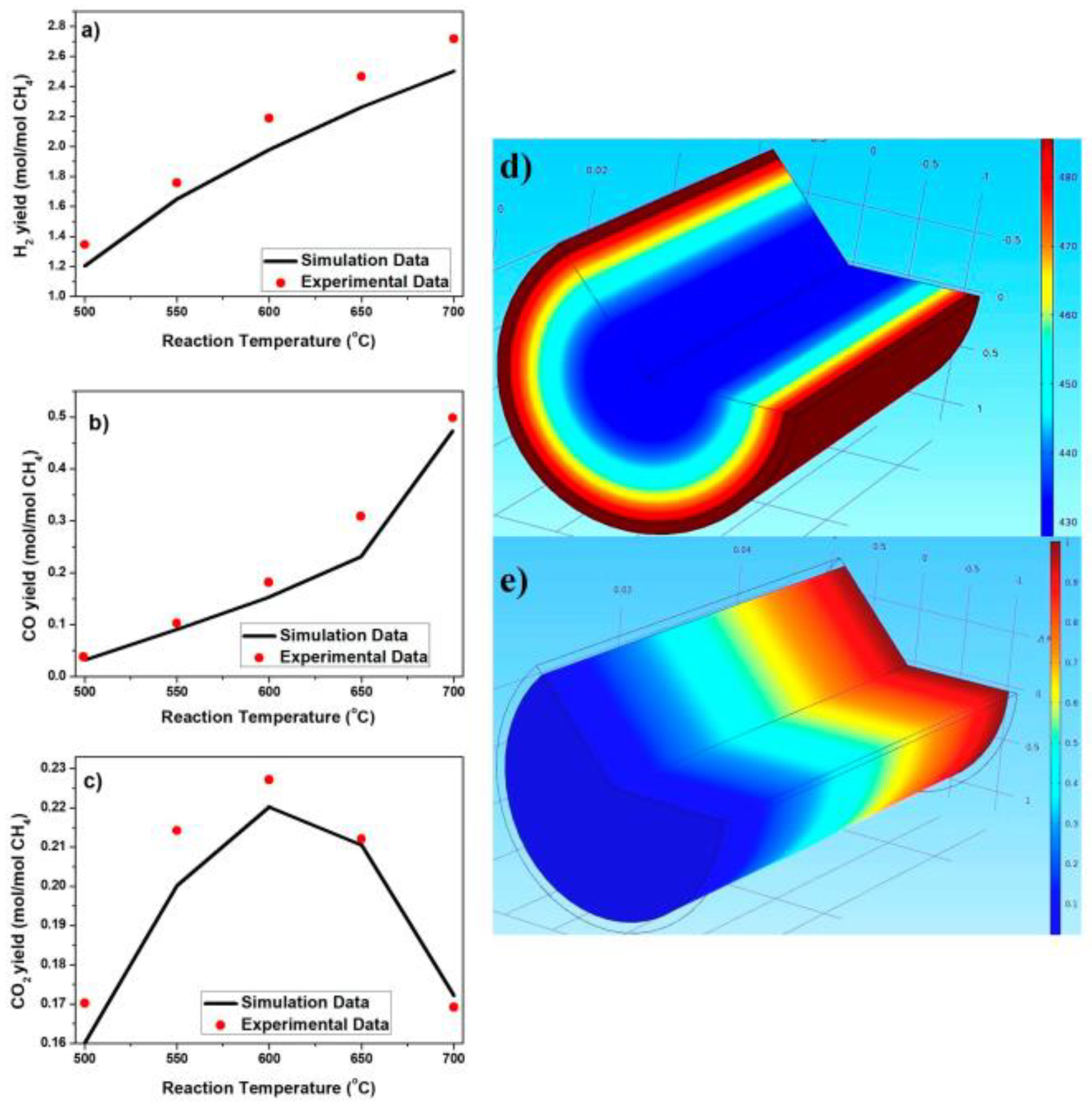



| Structured Carrier Shape | Catalyst | Operating Conditions | Main Characteristics | Main Performance | Ref. | |||||
|---|---|---|---|---|---|---|---|---|---|---|
| Ceramic Packed bed | Ni/αAl2O3 | Initial mass fractions CH4 = 0.1966 H2 = 0.0005 CO = 0.0007 CO2 = 0.1753 H2O = 0.6269 Ret = 50,000 | Surface area, mm2 | DP, Pa | Ts, °C * | Qsink, W * | XCH4, % * | η1 * | η3 * | |
| Cylinder | 1885 | 1109 | 723.87 | 1159 | 1.984 | 0.269 | 0.452 | [71] | ||
| Trilobe | 1637 | 165 | 727.96 | 787 | 1.373 | 0.393 | 0.595 | |||
| Daisy | 1955 | 164 | 732.33 | 868 | 1.475 | 0.438 | 0.639 | |||
| Hollow | 2428 | 197 | 742.47 | 1062 | 1.831 | 0.551 | 0.766 | |||
| Cylcut | 3484 | 208 | 750.66 | 1243 | 2.136 | 0.689 | 0.857 | |||
| 7-hole | 3737 | 218 | 752.06 | 1283 | 2.187 | 0.690 | 0.865 | |||
| NiO = 14.5%; SiO2 = 0.2%; support CaO-MgO-La2O3-αAl2O3 | T = 500–700 °C P = 3-bar S/C = 1–4 Residence time = 1–8 kgcat ·s/mol | surface area = 4.5 m2/gcat bulk density = 680 kg/m3 average porosity = 41% | @ S/C = 1, flow rate = 15 g/s, p = 3-bar, T = 700 °C | [72] | ||||||
| Cylinder | XCH4 = 62% | |||||||||
| Raschig ring | XCH4 = 70% | |||||||||
| 7-hole cylinder | XCH4 = 75% | |||||||||
| 7-hole sphere | XCH4 = 80% | |||||||||
| 1-hole cylinder | Ni = 20 wt %, CaO-Al2O3 = 80 wt % | T = 500–850 °C P = 0.2–1 MPa S/C = 2–3.3 GHSV = 2–40 l CH4/(h.gcat) | surface area = 21.26 m2/gcat density = 1.8 g/cm3 pore size = 111 Å | Equilibrium CH4 conversion reached @ GHSV = 2 l CH4/(h.gcat), T > 800 °C, P = 1 MPa, S/C = 3 | [73] | |||||
| cylinder-shaped alumina pellets | E-5Ni ** | T = 700–800 °C S/C = 3 WHSV = 3000 mL/(gcat h) | Ni = 5.6 wt % Surface area = 204 m2/g | XCH4 = 96% @ 700 °C XCH4 = 99% @ 800 °C | [74] | |||||
| E-3.5Ni ** | Ni = 3.5 wt % Surface area = 167 m2/g | XCH4 = 96% @ 700 °C XCH4 = 97% @ 800 °C | ||||||||
| E-3.5Ni-5Mg ** | Ni = 3.1 wt % Mg = 4.5 wt % Surface area = 120 m2/g | XCH4 = 95% @ 700 °C XCH4 = 98% @ 800 °C | ||||||||
| Micro-channel reactor Metal- ceramic plate | Ni = 15.87 wt % | T = 800–900 °C S/C = 3–5 P = 0.5–2 MPa GHSV = 40,000 to 240,000 h−1 | Surface area = 108.75 m2/g Catalyst layer thickness = 70 mm | Equilibrium CH4 conversion value and H2 productivity = 0.1 m3/h @ T = 900 °C, GHSV = 240,000 h−1, P = 2 MPa, S/C = 3 | [77] | |||||
| Structured Carrier Shape | Catalyst | Operating Conditions | Main Characteristics | Main Performance | Ref. |
|---|---|---|---|---|---|
| honeycomb | Ni/Ce | T = 450–650 °C S/C = 2 P = 1 atm | Surface area = 152 m2/g | XCH4 = 79.1% SCO = 59.4% SCO2 = 40.6% @ 650 °C | [75] |
| Rolled foils | Ni3Al | T = 600–800 °C S/C = 1 GHSV = 0.091 m3/(hm2) | total geometrical surface = 0.0066 m2 | XCH4 = 90% SCO = 99% SCO2 = 2% @ 650 °C | [82] |
| Rolled foils | Ni | GHSV = 455–2880 h−1 S/C = 0.62–2.48 T= 700–800 °C | Geometric surface area = 66.4 cm2 Pore density = 700 cpsi specific surface area = 88.1 cm2/cm3 | XCH4 = 94.0% SCO = 91.1% SCO2 = 8.9% H2 yield = 77.6% H2 production rate = 8.7 mL/min. H2 production rate/Volume = 11.6 mL/min./cm3 @ T = 800 °C, GHSV = 535 h−1, S/C = 1.24 | [85] |
| Rolled foils | Ni | GHSV = 6400 h−1 S/C = 1.362 T = 600–900 °C | Geometric surface area = 43.72 cm2 Pore density = 2300 cpsi specific surface area = 87.2 cm2/cm3 | XCH4 = 55.7% @ T = 800 °C | [86] |
| Rolled foils | Ni | GHSV = 6400 h−1 S/C = 1.362 T = 600–900 °C | Geometric surface area = 43.72 cm2 Pore density = 2300 cpsi specific surface area = 87.2 cm2/cm3 | XCH4 = 90% @ T = 900 °C, Ni honeycomb catalysts steam treated at 900 °C for 1 h followed by H2 reduction at 900 °C for 1 h | [87] |
| foam | Ni | w0CH4 = 21.28 w0H2O = 71.45 w0H2 = 2.60 w0CO = 1.19 w0N2 = 3.48 T0 = 550 °C P = 1-bar | ppi = 10 Kp = 1.2 m2 *** Cf = 0.097 *** ρs = 8900 Kg/m3 *** λs = 71 W/m K *** | XCH4 = 29.491% H2 production rate = 47.645 | [88] |
| Ni-Cr | w0CH4 = 21.28 w0H2O = 71.45 w0H2 = 2.60 w0CO = 1.19 w0N2 = 3.48 T0 = 550 °C P = 1-bar | ppi = 20 Kp = 1.185 m2 *** Cf = 0.1 *** ρs = 8420 Kg/m3 *** λs = 22.5 W/m K *** | XCH4 = 22.534% H2 production rate = 41.015 | ||
| Ni-Fe-Cr | w0CH4 = 21.28 w0H2O = 71.45 w0H2 = 2.60 w0CO = 1.19 w0N2 = 3.48 T0 = 550 °C P = 1-bar | ppi = 10 Kp = 1.49 m2 *** Cf = 0.099 *** ρs = 6285 Kg/m3 *** λs = 134.87 W/m K *** | XCH4 = 37.400% H2 production rate = 53.577 | ||
| Fecralloy® monoliths | 1.5 wt %Ru/3 wt %La-Al2O3 | T = 600–900 °C WHSV = 55 Nl/(h gcat) S/C = 3 | cpsi = 461 catalysts loading = 109 mg | Equilibrium CH4 conversion values @ 800 °C XCH4 = 52 % @ 600 °C | [89] |
| T = 600–900 °C WHSV = 55 Nl/(h gcat) S/C = 3 | cpsi = 461 catalyst loading = 171.8 mg | Equilibrium CH4 conversion values @ 800 °C XCH4 = 61% @ 600 °C | |||
| T = 600–900 °C WHSV = 55 Nl/(h gcat) S/C = 3 | cpsi = 461 catalyst loading = 216.9 mg | Equilibrium CH4 conversion values @ 800 °C XCH4 = 62 % @ 600 °C | |||
| Membrane reformer with Ni foam | 10 %Ni/10 %MgO/Ni-foam | P = 1-bar T = 400–600 °C S/C = 2 | 4 mm thick foam discs, 60 ppi | Equilibrium CH4 conversion values @ 600 °C for the catalytic discs | [90] |
| FeCralloy monolith | 0.5 wt %Rh-15 wt %Ni/MgAl2O4 | Twall = 600 °C, P = 1-bar, S/C=5, GHSV = 6000 h−1 and W/Fao = 0.34 gcat-h/gmol | XCH4 = 50% H2/CO = 37 H2/CO2 = 4.6 SCO = 11.0 × 10−2 | [92] | |
| Catalytic alumina egg-shell particles with diameter of 600 μm, filling the voids of FeCrAlY open cell foams of 12 PPI and copper open cell foams of 10 and 40 PPI | Rh/Al2O3 | GHSV = 5000 and 10,000 h−1 T = 600–800 °C S/C = 3.5 | λs = 16 W/(m K) – FeCrAlY foam λs = 380 W/(m K) – Cu foam | WT* – CT * = 40 for Cu foam WT* – CT * = 50 for FeCrAlY foam XCH4 = 86.4% for Cu foam, XCH4 = 75% for FeCrAlY foam @ Toven = 700 °C | [93] |
| SiC monolith | 30 wt %Ni | GHSV = 5000 to 25,000 h−1 T = 600–800 °C P = 1-bar S/C = 3 | Direct impregnation λs = 350 W/(m K) – SiC monolith Flow through configuration | Equilibrium CH4 conversion values for T > 800 °C | [94] |
| 20 wt %Ni/20 wt %CeO2 | GHSV = 1250 to 5000 h−1 T = 550–800 °C P = 1-bar S/C = 3 | Washcoating + Ni direct impregnation λs = 350 W/(m K) – SiC monolith Flow through and Wall Flow configuration | Equilibrium CH4 conversion values for T > 800 °C - Flow Through Equilibrium CH4 conversion values for T > 750 °C – wall Flow | ||
| SiC monolith | 5 wt %Ni/20 wt %CeO2 | GHSV = 100,000 h−1 Toven = 500–950 °C P = 1-bar S/C = 3 | λs = 350 W/(m K) Flow through and Wall Flow configuration | Equilibrium CH4 conversion values at Tgas = 800 °C (Toven = 900 °C) for both configurations | [95] |
| Cordierite monolith | 5 wt %Ni/20 wt %CeO2 | GHSV = 100,000 h−1 Toven = 500–950 °C P = 1-bar S/C = 3 | λs = 3 W/(m K) Flow through configuration | XCH4 = 80% at Toven = 900 °C | |
| SiC monolith | 5 wt %Ni/20 wt %CeO2/Al2O3 | GHSV = 100,000 h−1 Toven = 500–950 °C P = 1-bar S/C = 3 | λs = 350 W/(m K) Flow through and Wall Flow configuration | Equilibrium CH4 conversion values at Tgas = 750 °C (Toven = 850 °C) for the wall flow configuration | [96,97] |
| calcium aluminate modified SiC | 10%Ni | GHSV = 30,000 h−1 T = 650–850 °C P = 1-bar S/C = 1 to 3 | 1 mm granules λs = 1.16 W/(m K) | XCH4 = 97.9 % at T = 650 °C and S/C = 3 XCH4 = 81.3 % at T = 850 °C and S/C = 1 | [98] |
| αAlumina monolith | 1.5 wt %Ru/ 3 wt %La-Al2O3 | T = 600–900 °C S/C = 3 WHSV = 27 to 368 Nl h−1 gcat −1 | Catalyst loading = 100 to 250 mg | Equilibrium CH4 conversion values at T > 800 °C for 100 and 150 mg catalyst loadings | [99] |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Meloni, E.; Martino, M.; Palma, V. A Short Review on Ni Based Catalysts and Related Engineering Issues for Methane Steam Reforming. Catalysts 2020, 10, 352. https://doi.org/10.3390/catal10030352
Meloni E, Martino M, Palma V. A Short Review on Ni Based Catalysts and Related Engineering Issues for Methane Steam Reforming. Catalysts. 2020; 10(3):352. https://doi.org/10.3390/catal10030352
Chicago/Turabian StyleMeloni, Eugenio, Marco Martino, and Vincenzo Palma. 2020. "A Short Review on Ni Based Catalysts and Related Engineering Issues for Methane Steam Reforming" Catalysts 10, no. 3: 352. https://doi.org/10.3390/catal10030352
APA StyleMeloni, E., Martino, M., & Palma, V. (2020). A Short Review on Ni Based Catalysts and Related Engineering Issues for Methane Steam Reforming. Catalysts, 10(3), 352. https://doi.org/10.3390/catal10030352







