Enhanced UV Light Emission by Core-Shell Upconverting Particles Powering up TiO2 Photocatalysis in Near-Infrared Light
Abstract
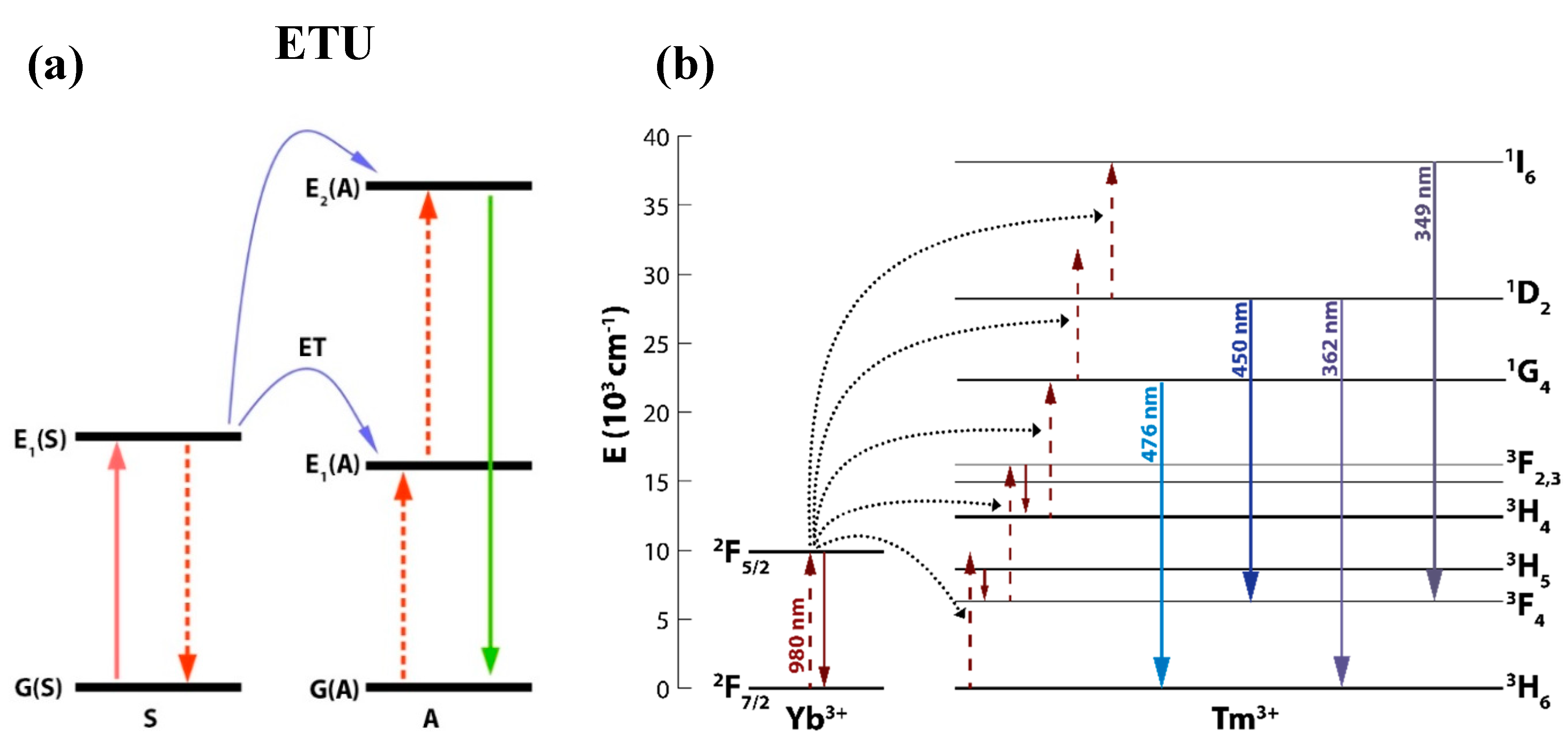
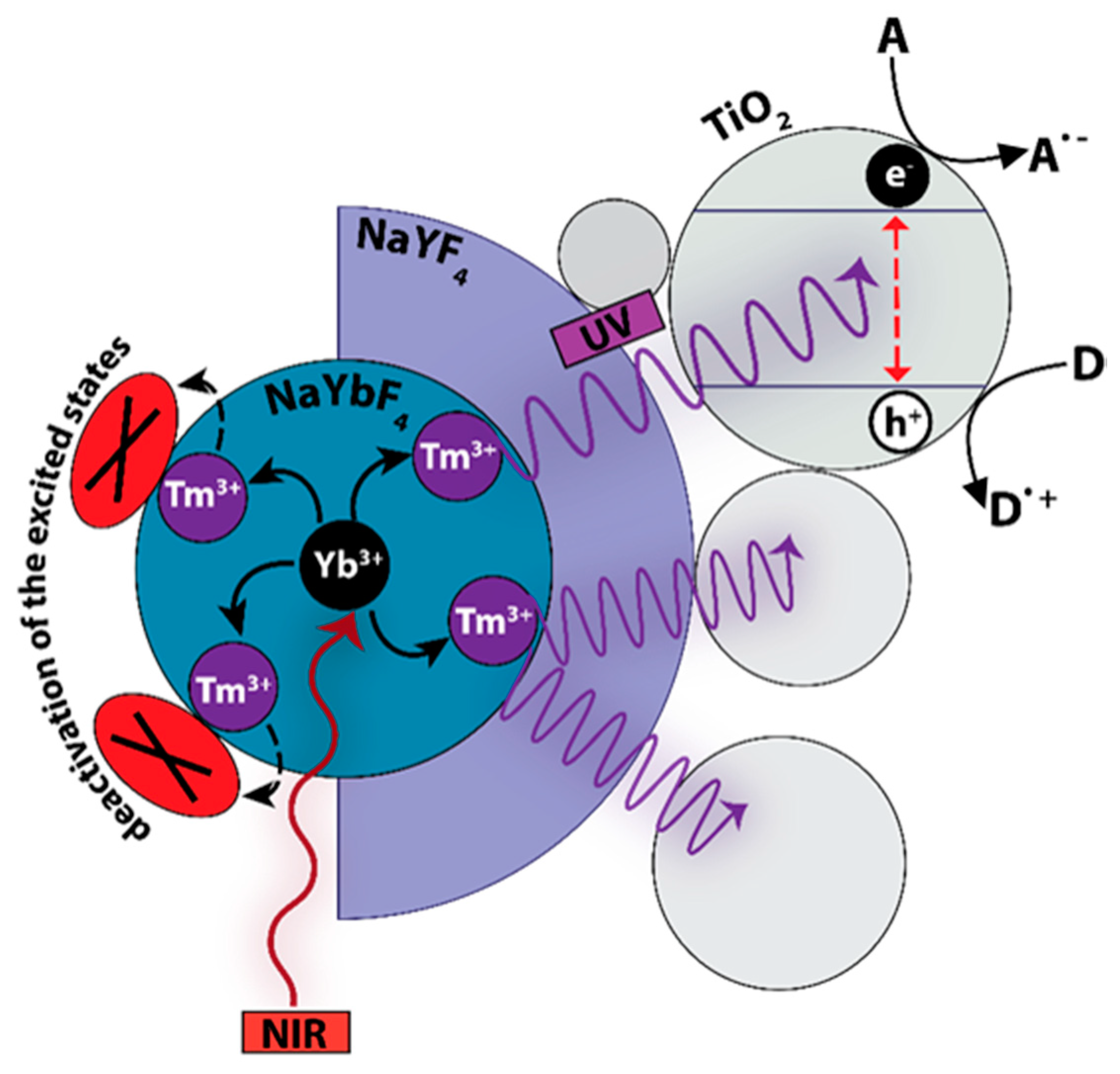
1. Introduction
2. Results and Discussion
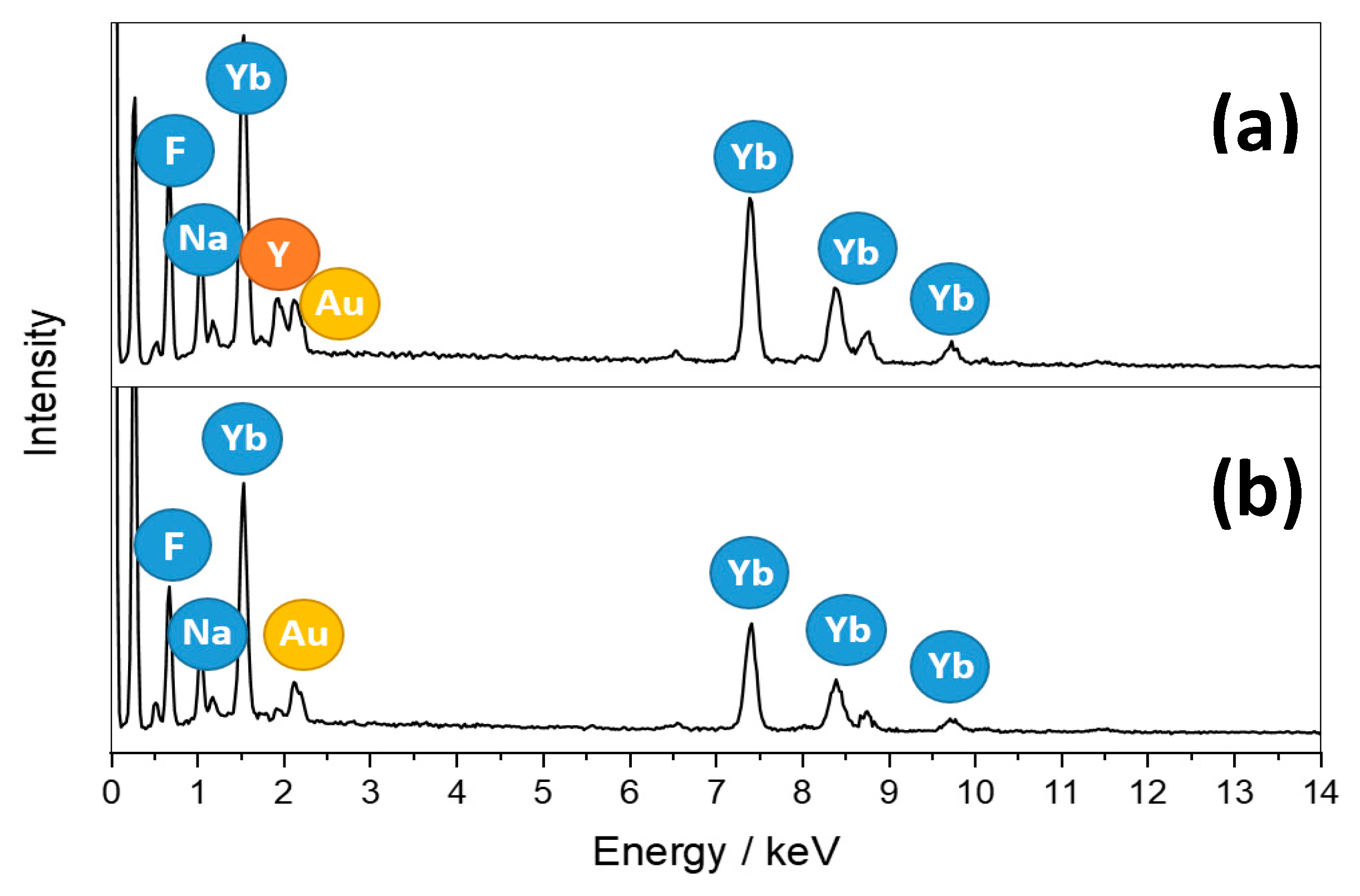
2.1. Morphology and Elemental Analysis
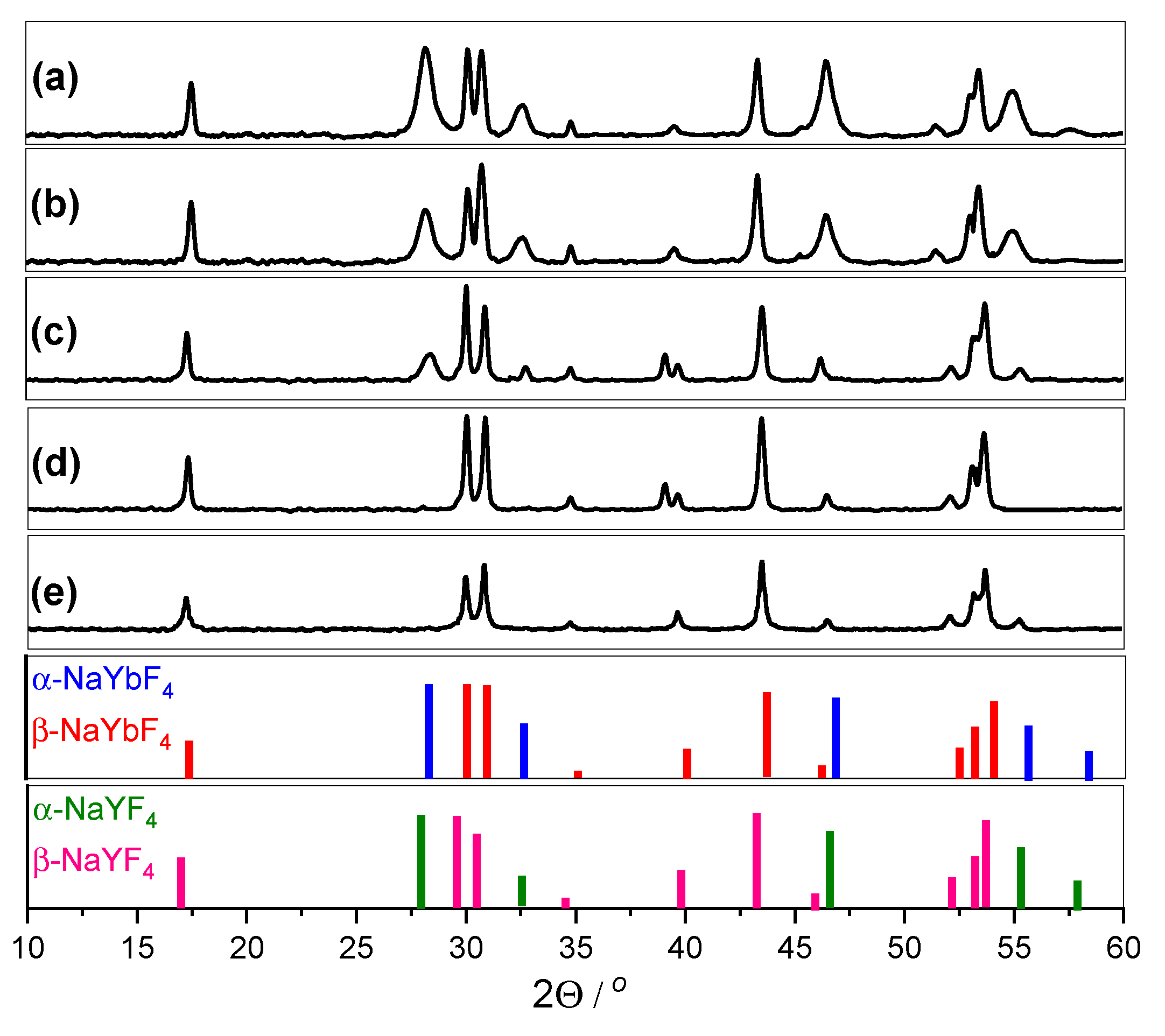
2.2. Structural Analysis—XRD
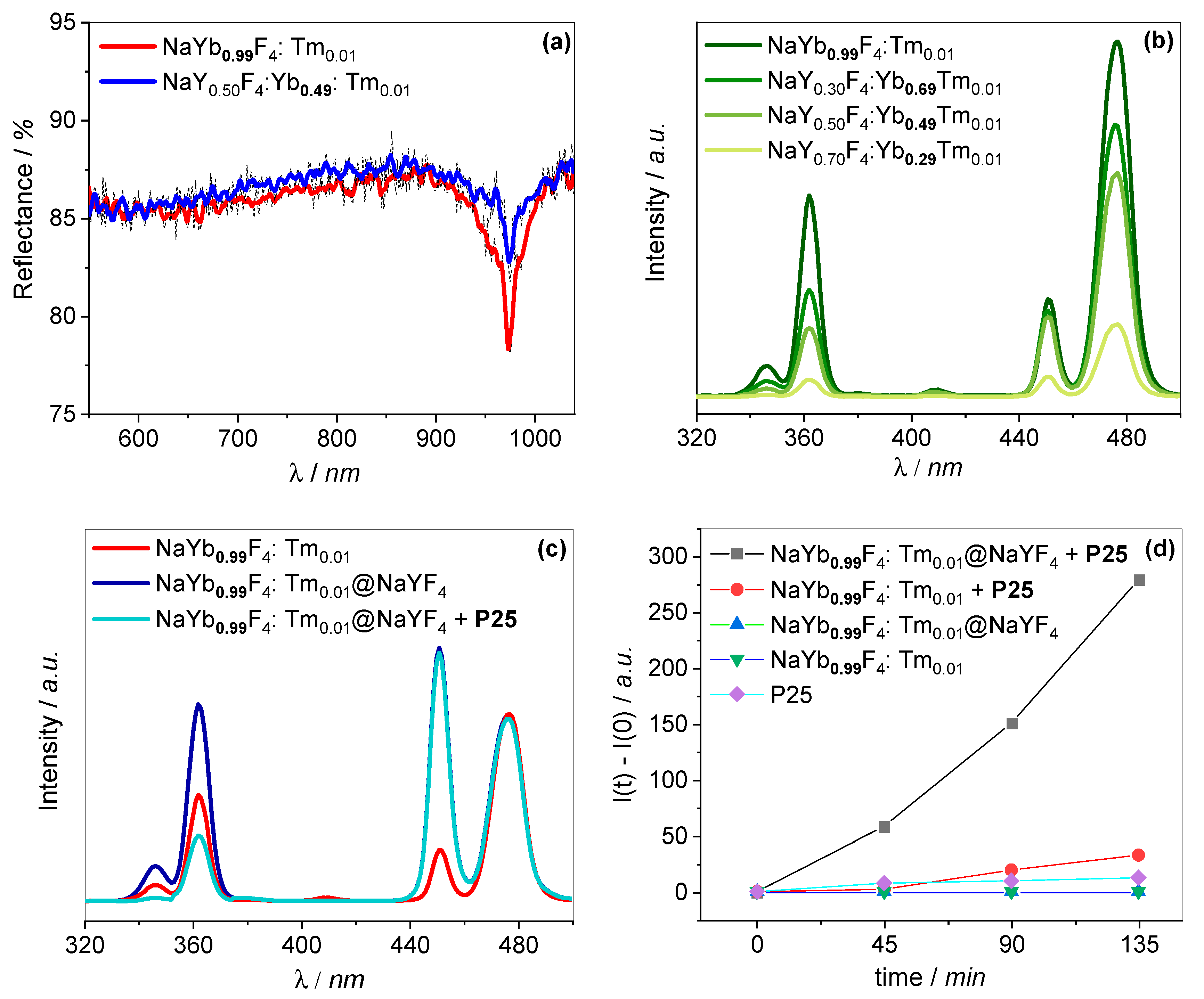
2.3. Spectroscopic Analysis
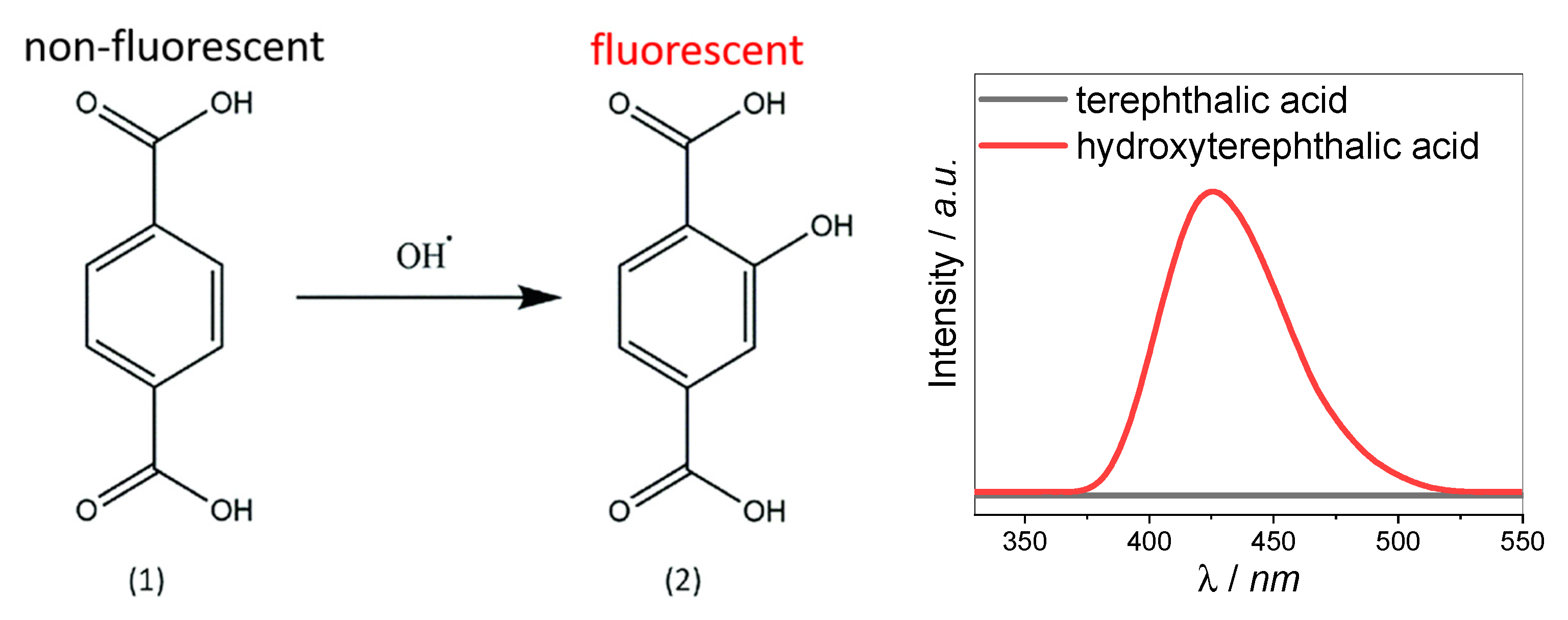
2.4. Photocatalytic Activity
3. Materials and Methods
3.1. Materials
3.2. Synthesis of NaYxF4:Yb0.99−xTm0.01 Microcrystals
3.3. Synthesis of the Core-Shell NaYb0.99F4:Tm0.01@NaYF4 Crystals
3.4. Characterization
3.5. Photocatalytic Measurements
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Conflicts of Interest
References
- Nakata, K.; Fujishima, A. TiO2 photocatalysis: Design and applications. J. Photochem. Photobiol. C 2012, 13, 169–189. [Google Scholar] [CrossRef]
- Hoffmann, M.R.; Martin, S.T.; Choi, W.; Bahnemann, D.W. Environmental applications of semiconductor photocatalysis. Chem. Rev. 1995, 95, 69–96. [Google Scholar] [CrossRef]
- Mills, A.; Davies, R.H.; Worsley, D. Water purification by semiconductor photocatalysis. Chem. Soc. Rev. 1993, 22, 417–425. [Google Scholar] [CrossRef]
- Sang, Y.; Liu, H.; Umar, A. Photocatalysis from UV/Vis to Near-Infrared Light: Towards Full Solar-Light Spectrum Activity. Chem. Cat. Chem. 2015, 7, 559–573. [Google Scholar]
- Macyk, W.; Szaciłowski, K.; Stochel, G.; Buchalska, M.; Kuncewicz, J.; Łabuz, P. Titanium (IV) complexes as direct TiO2 photosensitizers. Coord. Chem. Rev. 2010, 254, 2687–2701. [Google Scholar] [CrossRef]
- Zhang, Q.; Yang, F.; Xu, Z.; Chaker, M.; Ma, D. Are lanthanide-doped upconversion materials good candidates for photocatalysis? Nanoscale Horiz. 2019, 4, 579–591. [Google Scholar] [CrossRef]
- Yoon, T.P.; Ischay, M.A.; Du, J. Visible light photocatalysis as a greener approach to photochemical synthesis. Nature Chem. 2010, 2, 527. [Google Scholar] [CrossRef]
- Kisch, H.; Macyk, W. Visible-light photocatalysis by modified titania. Chem. Phys. Chem. 2002, 3, 399–400. [Google Scholar] [CrossRef]
- Asahi, R.Y.O.J.I.; Morikawa, T.A.K.E.S.H.I.; Ohwaki, T.; Aoki, K.; Taga, Y. Visible-light photocatalysis in nitrogen-doped titanium oxides. Science 2001, 293, 269–271. [Google Scholar] [CrossRef] [PubMed]
- Auzel, F. Upconversion and anti-stokes processes with f and d ions in solids. Chem. Rev. 2004, 104, 139–174. [Google Scholar] [CrossRef] [PubMed]
- Yang, W.; Li, X.; Chi, D.; Zhang, H.; Liu, X. Lanthanide-doped upconversion materials: Emerging applications for photovoltaics and photocatalysis. Nanotechnology 2014, 25, 482001. [Google Scholar] [CrossRef] [PubMed]
- Altavilla, C. Upconverting Nanomaterials: Perspectives, Synthesis, and Applications; CRC Press: Boca Raton, FL, USA, 2016. [Google Scholar]
- Liu, X.; Yan, C.-H.; Capobianco, J.A. Photon upconversion nanomaterials. Chem. Soc. Rev. 2015, 44, 1299–1301. [Google Scholar] [CrossRef] [PubMed]
- Haase, M.; Schafer, H. Upconverting nanoparticles. Angew. Chem. Int. Ed. 2011, 50, 5808–5829. [Google Scholar] [CrossRef] [PubMed]
- Mai, H.X.; Zhang, Y.W.; Sun, L.D.; Yan, C.H. Highly efficient multicolor up-conversion emissions and their mechanisms of monodisperse NaYF4: Yb, Er core and core/shell-structured nanocrystals. J. Phys. Chem. C 2007, 20, 13721–13729. [Google Scholar] [CrossRef]
- Charbouillot, T.; Brigante, M.; Mailhot, G.; Maddigapu, P.R.; Minero, C.; Vione, D. Performance and selectivity of the terephthalic acid probe for •OH as a function of temperature, pH and composition of atmospherically relevant aqueous media. J. Photochem. Photobiol. A 2011, 222, 70–76. [Google Scholar] [CrossRef]
- Matthews, R.W. The radiation chemistry of the terephthalate dosimeter. Radiat. Res. 1980, 83, 27–41. [Google Scholar] [CrossRef] [PubMed]
- Bogdan, N.; Vetrone, F.; Ozin, G.A.; Capobianco, J.A. Synthesis of ligand-free colloidally stable water dispersible brightly luminescent lanthanide-doped upconverting nanoparticles. Nano Lett. 2011, 11, 835–840. [Google Scholar] [CrossRef] [PubMed]

© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Jarosz-Duda, A.; O’Callaghan, P.; Kuncewicz, J.; Łabuz, P.; Macyk, W. Enhanced UV Light Emission by Core-Shell Upconverting Particles Powering up TiO2 Photocatalysis in Near-Infrared Light. Catalysts 2020, 10, 232. https://doi.org/10.3390/catal10020232
Jarosz-Duda A, O’Callaghan P, Kuncewicz J, Łabuz P, Macyk W. Enhanced UV Light Emission by Core-Shell Upconverting Particles Powering up TiO2 Photocatalysis in Near-Infrared Light. Catalysts. 2020; 10(2):232. https://doi.org/10.3390/catal10020232
Chicago/Turabian StyleJarosz-Duda, Agnieszka, Paulina O’Callaghan, Joanna Kuncewicz, Przemysław Łabuz, and Wojciech Macyk. 2020. "Enhanced UV Light Emission by Core-Shell Upconverting Particles Powering up TiO2 Photocatalysis in Near-Infrared Light" Catalysts 10, no. 2: 232. https://doi.org/10.3390/catal10020232
APA StyleJarosz-Duda, A., O’Callaghan, P., Kuncewicz, J., Łabuz, P., & Macyk, W. (2020). Enhanced UV Light Emission by Core-Shell Upconverting Particles Powering up TiO2 Photocatalysis in Near-Infrared Light. Catalysts, 10(2), 232. https://doi.org/10.3390/catal10020232




