Agro-Waste Derived Biomass Impregnated with TiO2 as a Potential Adsorbent for Removal of As(III) from Water
Abstract
1. Introduction
2. Results and Discussion
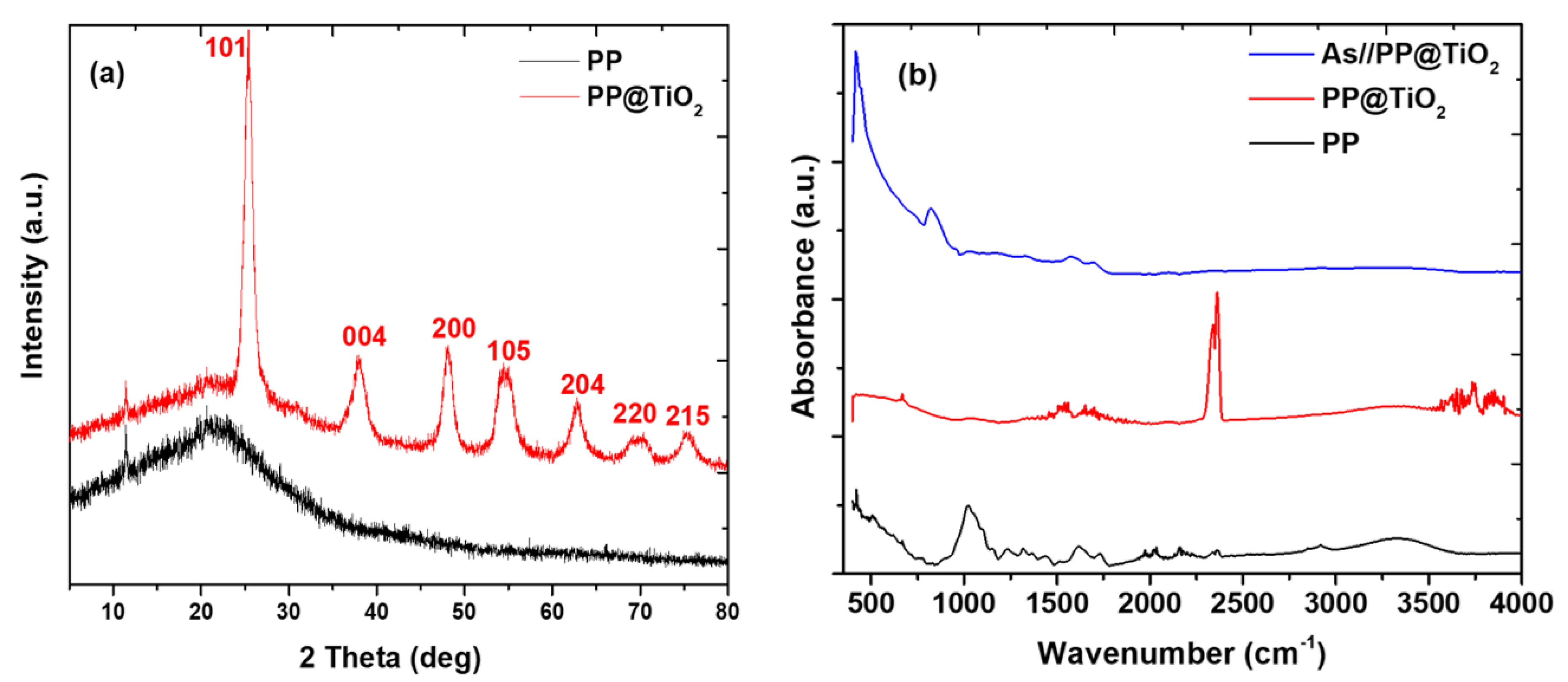
2.1. Adsorbent Characterization
2.2. Adsorption Isotherm
2.3. Effect of pH and As(III) Adsorption Mechanism
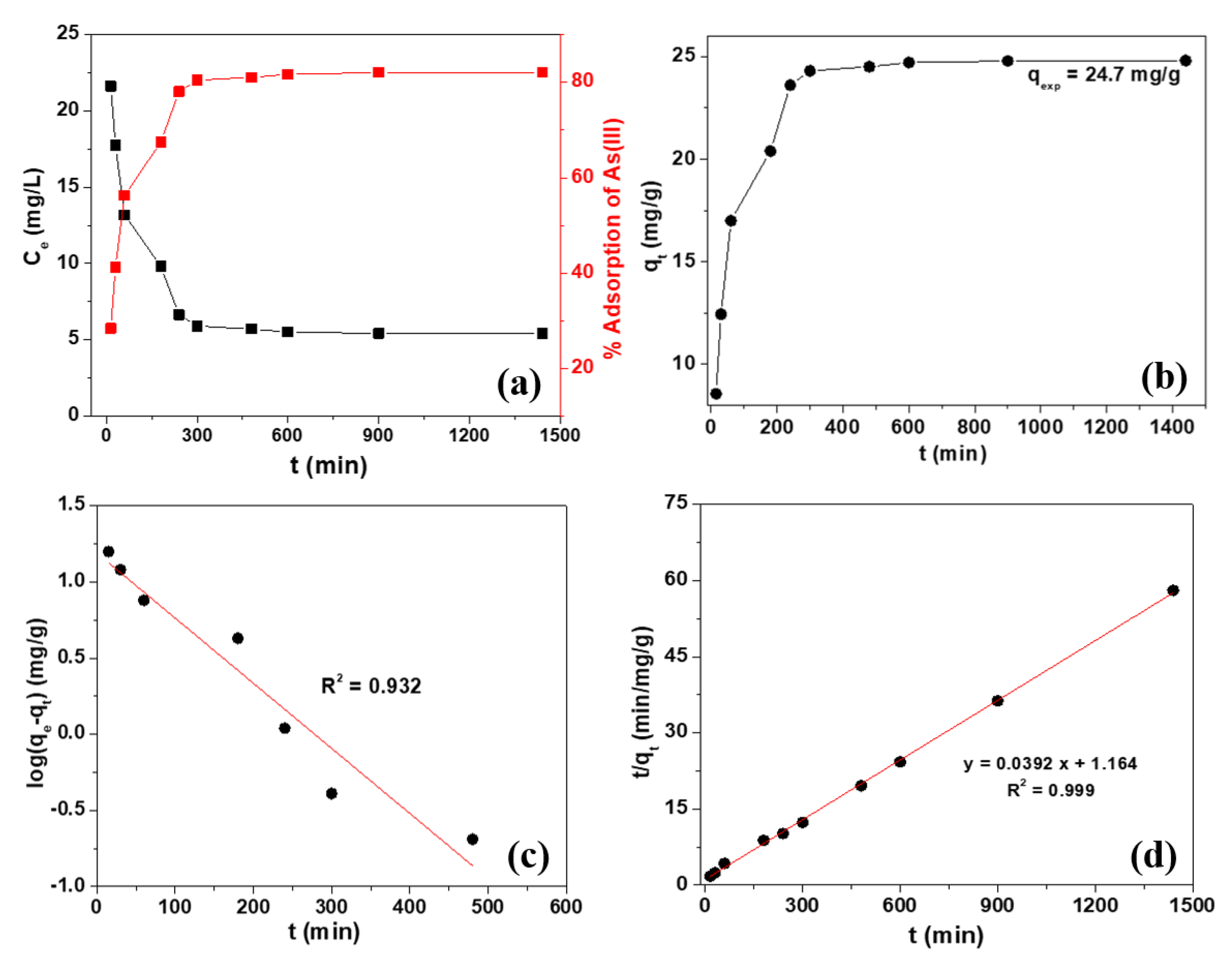
2.4. Adsorption Kinetics
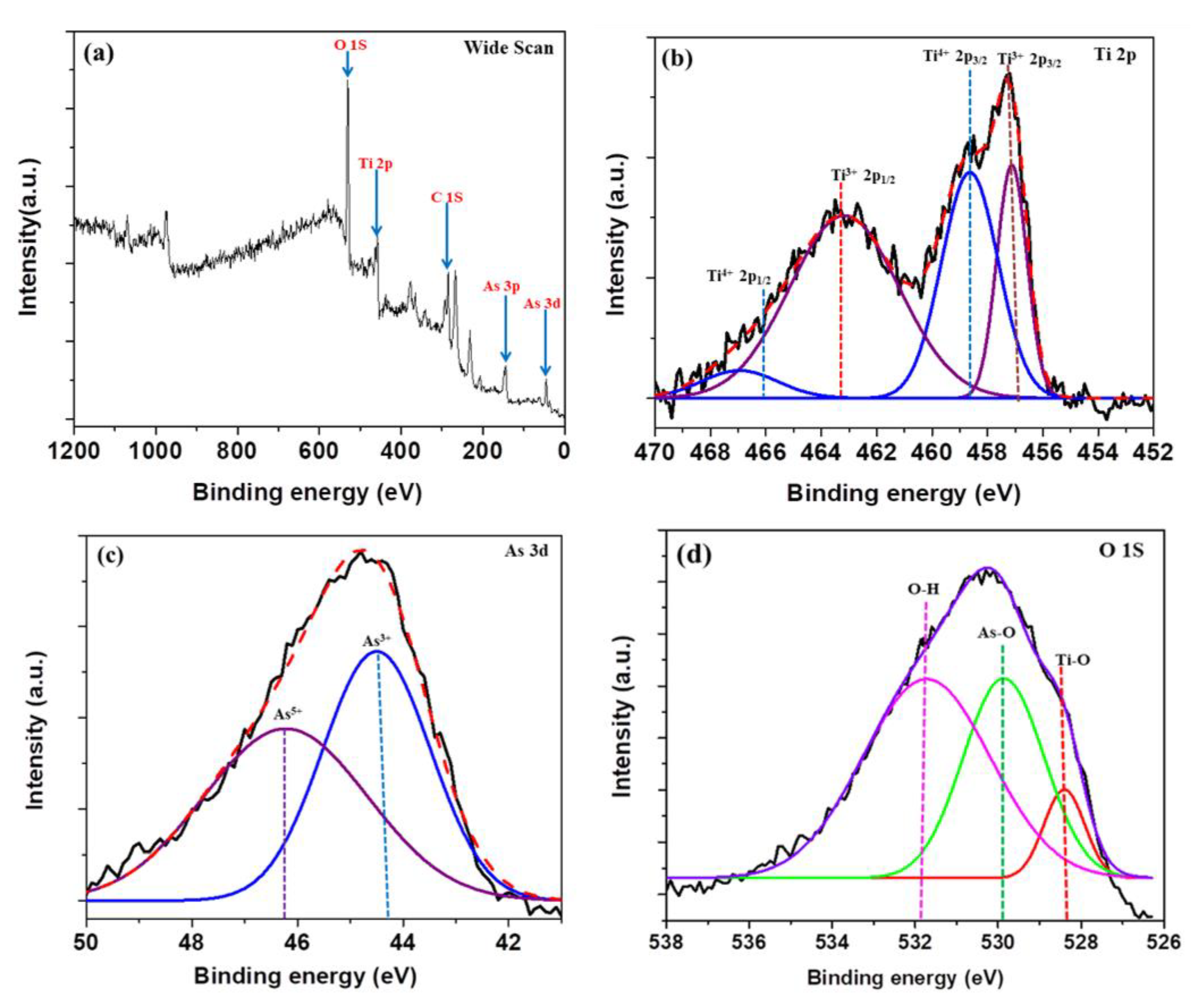
2.5. X-ray Photoelectron Spectroscopy (XPS) Studies
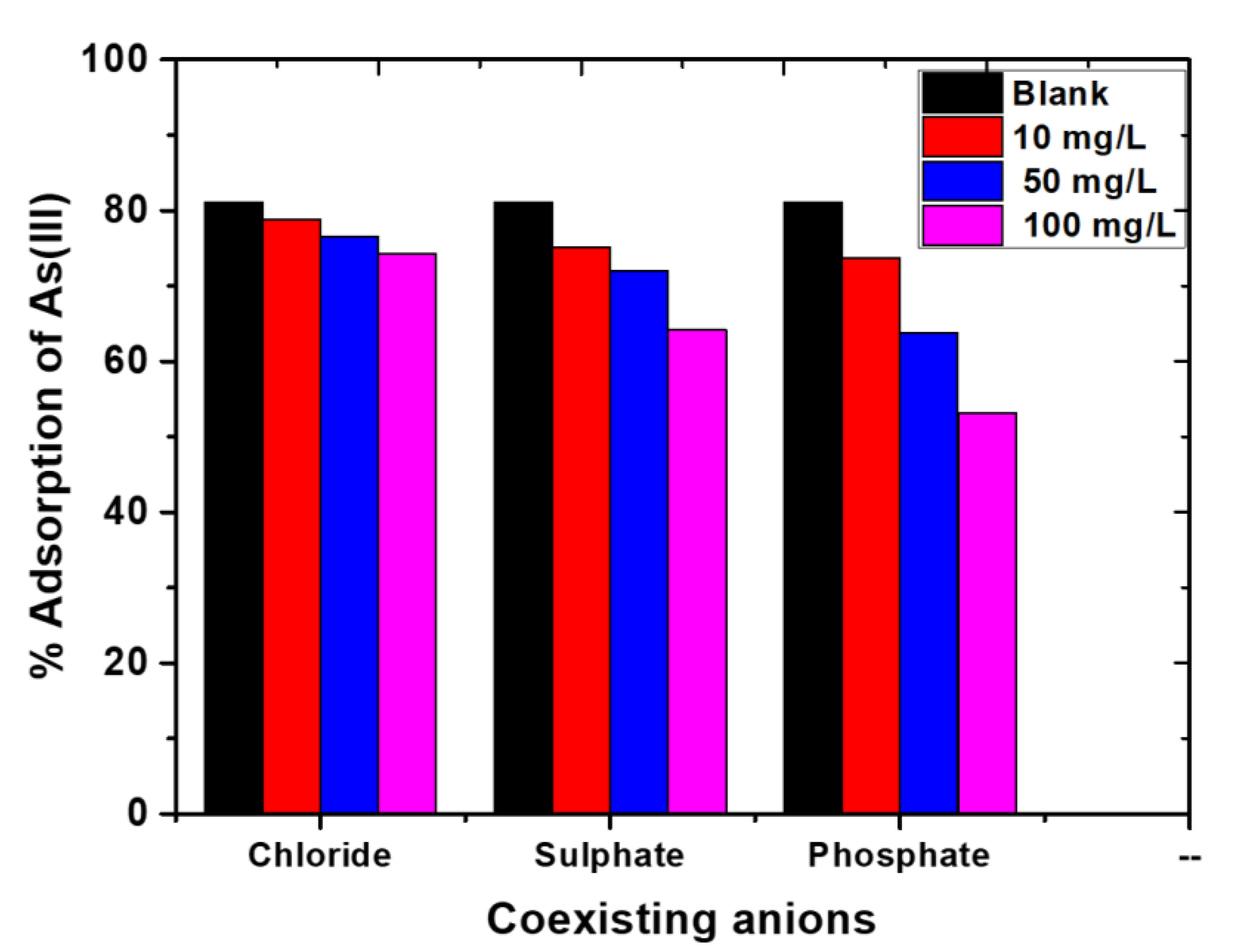
2.6. Effect of Common Coexisting Anions
2.7. Desorption Study and Reusability of PP@TiO2
3. Materials and Methods
3.1. Materials
3.2. Synthesis of TiO2 Impregnated Pomegranate Peels (PP@TiO2)
3.3. Characterizations
3.4. Batch Adsorption Studies
3.5. X-ray Photoelectron Spectroscopy (XPS) Studies
3.6. Desorption Study and Reusability of PP@TiO2
3.7. Analysis of Arsenic Concentration
4. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Shannon, M.A.; Bohn, P.W.; Elimelech, M.; Georgiadis, J.G.; Marinas, B.J.; Mayes, A.M. Science and technology for water purification in the coming decades. Nature 2008, 452, 301–310. [Google Scholar] [CrossRef] [PubMed]
- Rathore, E.; Biswas, K. Selective and ppb level removal of Hg(II) from water: Synergistic role of graphene oxide and SnS2. J. Mater. Chem. A 2018, 6, 13142–13152. [Google Scholar] [CrossRef]
- Raj, K.R.; Kardam, A.; Srivastava, S. Development of polyethyleneimine modified Zea mays as a high capacity biosorbent for the removal of As (III) and As (V) from aqueous system. Int. J. Miner. Process. 2013, 122, 66–70. [Google Scholar] [CrossRef]
- Ratnaike, R.N. Acute and chronic arsenic toxicity. Postgrad. Med. J. 2003, 79, 391–396. [Google Scholar] [CrossRef] [PubMed]
- Mohan, D.; Pittman, C.U., Jr. Arsenic removal from water/wastewater using adsorbents—A critical review. J. Hazard. Mater. 2007, 142, 1–53. [Google Scholar] [CrossRef]
- Shankar, S.; Shanker, U. Arsenic contamination of groundwater: A review of sources, prevalence, health risks, and strategies for mitigation. Sci. World J. 2014, 2014, 1–18. [Google Scholar] [CrossRef] [PubMed]
- Wang, S.; Gao, B.; Zimmerman, A.R.; Li, Y.; Ma, L.; Harris, W.G.; Migliaccio, K.W. Removal of arsenic by magnetic biochar prepared from pinewood and natural hematite. Bioresour. Technol. 2015, 175, 391–395. [Google Scholar] [CrossRef]
- Ogata, F.; Nagai, N.; Toda, M.; Otani, M.; Saenjum, C.; Nakamura, T.; Kawasaki, N. Removal of arsenic(III) ion from aqueous media using complex nickel-aluminum and nickel-aluminum-zirconium hydroxides. Water 2020, 12, 1697. [Google Scholar] [CrossRef]
- Shehzad, K.; Ahmad, M.; He, J.; Liu, T.; Xu, W.; Liu, J. Synthesis of ultra-large ZrO2 nanosheets as novel adsorbents for fast and efficient removal of As (III) from aqueous solutions. J. Colloid Interface Sci. 2019, 533, 588–597. [Google Scholar] [CrossRef]
- Aryal, M.; Ziagova, M.; Liakopoulou-Kyriakides, M. Study on arsenic biosorption using Fe (III)–treated biomass of Staphylococcus xylosus. Chem. Eng. J. 2010, 162, 178–185. [Google Scholar] [CrossRef]
- WHO. Arsenic in Drinking-water. Background document for development of WHO Guidelines for Drinking-water Quality. In Guidelines for Drinking-water Quality; (No. WHO/SDE/WSH/03.04/75/Rev/1); World Health Organization: Geneva, Switzerland, 2011; Available online: http://www.who.int/water_sanitation_health/dwq/chemicals/arsenic.pdf (accessed on 16 April 2020).
- USEPA. Drinking Water Requirements for States and Public Water System. 2017. Available online: https://www.epa.gov/dwreginfo/chemical–contaminant–rules (accessed on 16 April 2020).
- Bureau of Indian Standard (BIS). Indian Standards, Drinking Water Specification, 2nd ed.; Bureau of Indian Standards: New Delhi, India, 2012.
- National Drinking Water Quality Standards (NDWQS), Nepal. Nepal Gazette (B.S. 2063–03–12); Government of Nepal/Ministry of Physical Planning: Kathmandu, Nepal, 2006.
- Sanjoy, K.M.; Anjali, P.; Trasankar, P. Arsenic removal from real-life groundwater by adsorption on laterite soil. J. Hazard. Mater. 2008, 153, 811–820. [Google Scholar] [CrossRef]
- Joshi, S.; Sharma, M.; Kumari, A.; Shrestha, S.; Shrestha, B. Arsenic removal from water by adsorption onto iron oxide/nano–porous carbon magnetic composite. Appl. Sci. 2019, 9, 3732. [Google Scholar] [CrossRef]
- Chowdhury, T.; Zhang, L.; Zhang, J.; Aggarwal, S. Removal of arsenic (III) from aqueous solution using metal-organic framework-graphene oxide nanocomposite. Nanomaterials 2018, 8, 1062. [Google Scholar] [CrossRef] [PubMed]
- Thapa, S.; Pokhrel, M.R. Removal of As(III) from aqueous solution using Fe(III) loaded pomegranate waste. J. Nepal Chem. Soc. 2012, 30, 29–36. [Google Scholar] [CrossRef]
- Tamayo, R.; Espinoza-González, R.; Gracia, F.; Rodrigues-Filho, U.P.; Flores, M.; Sacari, E. As(III) removal from aqueous solution by calcium titanate nanoparticles prepared by the sol-gel method. Nanomaterials 2019, 9, 733. [Google Scholar] [CrossRef]
- Biswas, B.K.; Inoue, J.I.; Inoue, K.; Ghimire, K.N.; Harada, H.; Ohto, K.; Kawakita, H. Adsorptive removal of As (V) and As (III) from water by a Zr (IV)-loaded orange waste gel. J. Hazard. Mater. 2008, 154, 1066–1074. [Google Scholar] [CrossRef]
- Ghimire, K.N.; Inoue, K.; Makino, K.; Miyajima, T. Adsorptive removal of arsenic using orange juice residue. Sep. Sci. Technol. 2002, 37, 2785–2799. [Google Scholar] [CrossRef]
- Yu, L.; Peng, X.; Ni, F.; Li, J.; Wang, D.; Luan, Z. Arsenite removal from aqueous solutions by γ-Fe2O3-TiO2 magnetic nanoparticles through simultaneous photocatalytic oxidation and adsorption. J. Hazard. Mater. 2013, 246, 10–17. [Google Scholar] [CrossRef]
- Ortega, A.; Oliva, I.; Contreras, K.E.; González, I.; Cruz-Díaz, M.R.; Rivero, E.P. Arsenic removal from water by hybrid electro–regenerated anion exchange resin/electrodialysis process. Sep. Purif. Technol. 2017, 184, 319–326. [Google Scholar] [CrossRef]
- Wickramasinghe, S.R.; Han, B.; Zimbron, J.; Shen, Z.; Karim, M.N. Arsenic removal by coagulation and filtration: Comparison of groundwaters from the United States and Bangladesh. Desalination 2004, 169, 231–244. [Google Scholar] [CrossRef]
- Litter, M.I.; Morgada, M.E.; Bundschuh, J. Possible treatments for arsenic removal in Latin American waters for human consumption. Environ. Pollut. 2010, 158, 1105–1118. [Google Scholar] [CrossRef] [PubMed]
- Sharma, V.K.; Sohn, M. Aquatic arsenic: Toxicity, speciation, transformations, and remediation. Environ. Int. 2009, 35, 743–759. [Google Scholar] [CrossRef] [PubMed]
- Pokhrel, M.R.; Poudel, B.R.; Aryal, R.L.; Paudyal, H.; Ghimire, K.N. Removal and recovery of phosphate from water and wastewater using metal–loaded agricultural waste-based adsorbents: A review. JIST 2019, 24, 77–89. [Google Scholar] [CrossRef]
- Amin, M.N.; Kaneco, S.; Kitagawa, T.; Begum, A.; Katsumata, H.; Suzuki, T.; Ohta, K. Removal of arsenic in aqueous solutions by adsorption onto waste rice husk. Ind. Eng. Chem. Res. 2006, 45, 8105–8110. [Google Scholar] [CrossRef]
- Yao, S.; Liu, Z.; Shi, Z. Arsenic removal from aqueous solutions by adsorption onto iron oxide/activated carbon magnetic composite. J. Environ. Health Sci. Eng. 2014, 12, 58. [Google Scholar] [CrossRef]
- Bissen, M.; Vieillard-Baron, M.M.; Schindelin, A.J.; Frimmel, F.H. TiO2-catalyzed photooxidation of arsenite to arsenate in aqueous samples. Chemosphere 2001, 44, 751–757. [Google Scholar] [CrossRef]
- Guan, X.H.; Du, J.S.; Meng, X.G.; Sun, Y.K.; Sun, B.; Hu, Q.H. Application of titanium dioxide in arsenic removal from water: A review. J. Hazard. Mater. 2012, 215, 1–16. [Google Scholar] [CrossRef]
- Yao, S.; Jia, Y.; Shi, Z.; Zhao, S. Photocatalytic oxidation of arsenite by a composite of titanium dioxide and activated carbon fiber. Photochem. Photobiol. 2010, 86, 1215–1221. [Google Scholar] [CrossRef]
- Lee, H.; Choi, W. Photocatalytic oxidation of arsenite in TiO2 suspension: Kinetics and mechanisms. Environ. Sci. Technol. 2002, 36, 3872–3878. [Google Scholar] [CrossRef]
- Ferguson, M.A.; Hoffmann, M.R.; Hering, J.G. TiO2-photocatalyzed As (III) oxidation in aqueous suspensions: Reaction kinetics and effects of adsorption. Environ. Sci. Technol. 2005, 39, 1880–1886. [Google Scholar] [CrossRef]
- Dutta, P.K.; Pehkonen, S.O.; Sharma, V.K.; Ray, A.K. Photocatalytic oxidation of arsenic (III): Evidence of hydroxyl radicals. Environ. Sci. Technol. 2005, 39, 1827–1834. [Google Scholar] [CrossRef] [PubMed]
- Zhang, F.S.; Itoh, H. Photocatalytic oxidation and removal of arsenite from water using slag–iron oxide-TiO2 adsorbent. Chemosphere 2006, 65, 125–131. [Google Scholar] [CrossRef] [PubMed]
- Bang, S.; Patel, M.; Lippincott, L.; Meng, X. Removal of arsenic from groundwater by granular titanium dioxide adsorbent. Chemosphere 2005, 60, 389–397. [Google Scholar] [CrossRef] [PubMed]
- López Paraguay, M.Z.; Cortes, J.A.; Pérez-Robles, J.F.; Alarcón-Herrera, M.T. Adsorption of arsenite from groundwater using titanium dioxide. Clean (Weinh.) 2014, 42, 713–721. [Google Scholar] [CrossRef]
- Pena, M.E.; Korfiatis, G.P.; Patel, M.; Lippincott, L.; Meng, X.G. Adsorption of As(V) and As(III) by nanocrystalline titanium dioxide. Water Res. 2006, 39, 2327–2337. [Google Scholar] [CrossRef]
- Pirila, M.; Martikainen, M.; Ainassaari, K.; Kuokkanen, T.; Keiski, R.L. Removal of aqueous As(III) and As(V) by hydrous titanium dioxide. J. Colloid Interface Sci. 2011, 353, 257–262. [Google Scholar] [CrossRef]
- Wei, Z.G.; Liang, K.; Wu, Y.; Zou, Y.D.; Zuo, J.H.; Arriagada, D.C.; Pan, Z.C.; Hu, G.H. The effect of pH on the adsorption of arsenic(III) and arsenic(V) at the TiO2 anatase [1 0 1] surface. J. Colloid Interface Sci. 2016, 562, 252–259. [Google Scholar] [CrossRef]
- Shakoor, M.B.; Niazi, N.K.; Bibi, I.; Shahid, M.; Sharif, F.; Bashir, S.; Shaheen, M.S.; Wang, H.; Ok, Y.S.; Rinklebe, J. Arsenic removal by natural and chemically modified watermelon rind in aqueous solutions and groundwater. Sci. Total Environ. 2018, 645, 1444–1455. [Google Scholar] [CrossRef]
- Pehlivan, E.; Tran, H.T.; Ouédraogo, W.K.I.; Schmidt, C.; Zachmann, D.; Bahadir, M. Sugarcane bagasse treated with hydrous ferric oxide as a potential adsorbent for the removal of As (V) from aqueous solutions. Food Chem. 2013, 138, 133–138. [Google Scholar] [CrossRef]
- Gupta, A.; Vidyarthi, S.R.; Sankararamakrishnan, N. Concurrent removal of As (III) and As (V) using green low cost functionalized biosorbent-Saccharum officinarum bagasse. J. Environ. Chem. Eng. 2015, 3, 113–121. [Google Scholar] [CrossRef]
- Wiącek, A.E.; Gozdecka, A.; Jurak, M. Physicochemical characteristics of chitosan-TiO2 biomaterial. 1. Stability and swelling properties. Ind. Eng. Chem. Res. 2018, 57, 1859–1870. [Google Scholar] [CrossRef]
- Li, Q.; Su, H.; Tan, T. Synthesis of ion-imprinted chitosan–TiO2 adsorbent and its multi-functional performances. Biochem. Eng. J. 2008, 38, 212–218. [Google Scholar] [CrossRef]
- Wu, S.; Kan, J.; Dai, X.; Shen, X.; Zhang, K.; Zhu, M. Ternary carboxymethyl chitosan-hemicellulose-nanosized TiO2 composite as effective adsorbent for removal of heavy metal contaminants from water. Fibers Polym. 2017, 18, 22–32. [Google Scholar] [CrossRef]
- Tao, Y.; Ye, L.; Pan, J.; Wang, Y.; Tang, B. Removal of Pb (II) from aqueous solution on chitosan/TiO2 hybrid film. J. Hazard. Mater. 2009, 161, 718–722. [Google Scholar] [CrossRef] [PubMed]
- Carvalho, J.; Araújo, J.; Castro, F. Alternative low-cost adsorbent for water and wastewater decontamination derived from eggshell waste: An overview. Waste Biomass Valor. 2011, 2, 157–167. [Google Scholar] [CrossRef]
- Özsin, G.; Kılıç, M.; Apaydın-Varol, E.; Pütün, A.E. Chemically activated carbon production from agricultural waste of chickpea and its application for heavy metal adsorption: Equilibrium, kinetic, and thermodynamic studies. Appl. Water Sci. 2019, 9, 56. [Google Scholar] [CrossRef]
- Yu, J.; Kang, Y.; Yin, W.; Fan, J.; Guo, Z. Removal of antibiotics from aqueous solutions by a carbon adsorbent derived from protein-waste-doped biomass. ACS Omega 2020. [Google Scholar] [CrossRef]
- Cruz, G.J.F.; Mondal, D.; Rimaycuna, J.; Soukup, K.; Gomez, M.M.; Solis, J.L.; Lang, J. Agrowaste derived biochars impregnated with ZnO for removal of arsenic and lead in water. J. Environ. Chem. Eng. 2020, 8, 103800. [Google Scholar] [CrossRef]
- Pincus, L.N.; Melnikov, F.; Yamani, J.S.; Zimmerman, J.B. Multifunctional photoactive and selective adsorbent for arsenite and arsenate: Evaluation of nano titanium dioxide–enabled chitosan cross-linked with copper. J. Hazard. Mater. 2018, 358, 145–154. [Google Scholar] [CrossRef]
- Fausey, C.L.; Zucker, I.; Shaulsky, E.; Zimmerman, J.B.; Elimelech, M. Removal of arsenic with reduced graphene oxide-TiO2-enabled nanofibrous mats. Chem. Eng. J. 2019, 375, 122040. [Google Scholar] [CrossRef]
- Liu, H.; Zuo, K.; Vecitis, C.D. Titanium dioxide-coated carbon nanotube network filter for rapid and effective arsenic sorption. Environ. Sci. Technol. 2014, 48, 13871–13879. [Google Scholar] [CrossRef]
- Pant, B.; Pant, H.R.; Park, M. Fe1−xS modified TiO2 NPs embedded carbon nanofiber composite via electrospinning: A potential electrode material for supercapacitors. Molecules 2020, 25, 1075. [Google Scholar] [CrossRef] [PubMed]
- Pant, B.; Pant, H.R.; Park, M.; Liu, Y.; Choi, J.W.; Barakat, N.A.M.; Kim, H.Y. Electrospun CdS-TiO2 doped carbon nanofibers for visible-light-induced photocatalytic hydrolysis of ammonia borane. Cata. Commun. 2014, 50, 63–68. [Google Scholar] [CrossRef]
- Choi, C.; Hwang, K.J.; Kim, Y.J.; Kim, G.; Park, J.Y.; Jin, S. Rice-straw-derived hybrid TiO2–SiO2 structures with enhanced photocatalytic properties for removal of hazardous dye in aqueous solutions. Nano Energy 2016, 20, 76–83. [Google Scholar] [CrossRef]
- Gallios, G.P.; Tolkou, A.K.; Katsoyiannis, I.A.; Stefusova, K.; Vaclavikova, M.; Deliyanni, E.A. Adsorption of arsenate by nanoscaled activated carbon modified by iron and manganese oxides. Sustainability 2017, 9, 1684. [Google Scholar] [CrossRef]
- Barkat, M.; Nibou, D.; Chegrouche, S.; Mellah, A. Kinetics and thermodynamics studies of chromium(VI) ions adsorption onto activated carbon from aqueous solutions. Chem. Eng. Process. Process Intensif. 2009, 48, 38–47. [Google Scholar] [CrossRef]
- Yantasee, W.; Warner, C.L.; Sangvanich, T.; Addleman, R.S.; Carter, T.G.; Wiacek, R.J.; Fryxell, G.E.; Timchalk, C.; Warner, M.G. Removal of heavy metals from aqueous systems with thiol functionalized superparamagnetic nanoparticles. Environ. Sci. Technol. 2007, 41, 5114–5119. [Google Scholar] [CrossRef]
- Langmuir, I. The constitution and fundamental properties of solids and liquids. JACS 1916, 38, 2221–2295. [Google Scholar] [CrossRef]
- Freundlich, H. Uber die adsorption in losungen. J. Phys. Chem. 1916, 57, 385–470. [Google Scholar] [CrossRef]
- Chandra, V.; Park, J.; Chun, Y.; Lee, J.W.; Hwang, I.C.; Kim, K.S. Water-dispersible magnetite-reduced graphene oxide composites for arsenic removal. ACS Nano 2010, 4, 3979–3986. [Google Scholar] [CrossRef]
- Feng, L.; Cao, M.; Ma, X.; Zhu, Y.; Hu, C. Superparamagnetic high–surface–area Fe3O4 nanoparticles as adsorbents for arsenic removal. J. Hazard Mater. 2012, 217–218, 439–446. [Google Scholar] [CrossRef] [PubMed]
- Chen, W.; Parette, R.; Zou, J.; Cannon, F.S.; Dempsey, B.A. Arsenic removal by iron modified activated carbon. Water Res. 2007, 41, 1851–1858. [Google Scholar] [CrossRef] [PubMed]
- Lenoble, V.; Bouras, O.; Deluchat, V.; Serpaud, B.; Bollinger, J.C. Arsenic adsorption onto pillared clays and iron oxides. J. Colloid Interface Sci. 2002, 255, 52–58. [Google Scholar] [CrossRef] [PubMed]
- Liu, X.; Ao, H.; Xiong, X.; Xiao, J.; Liu, J. Arsenic removal from water by iron–modified bamboo charcoal. Water Air Soil Pollut. 2012, 223, 1033–1044. [Google Scholar] [CrossRef]
- Setyono, D.; Valiyaveettil, S. Chemically modified sawdust as renewable adsorbent for arsenic removal from water. ACS Sustain. Chem. Eng. 2014, 2, 2722–2729. [Google Scholar] [CrossRef]
- Manju, G.N.; Raji, C.; Anirudhan, T.S. Evaluation of coconut husk carbon for the removal of arsenic from water. Water Res. 1998, 32, 3062–3070. [Google Scholar] [CrossRef]
- Xia, C.L.; Jing, Y.; Jia, Y.Z.; Yue, D.Y.; Ma, J.; Yin, X.J. Adsorption properties of congo red from aqueous solution on modified hectorite: Kinetic and thermodynamic studies. Desalination 2011, 265, 81–87. [Google Scholar] [CrossRef]
- Ghaedi, M.; Hajati, S.; Karimi, F.; Barazesh, B.; Ghezelbash, G. Equilibrium, kinetic and isotherm of some metal ion biosorption. J. Ind. Eng. Chem. 2013, 19, 987–992. [Google Scholar] [CrossRef]
- Cai, X.; Li, J.; Liu, Y.; Yan, Z.; Tan, X.; Liu, S.; Zeng, G.; Gu, Y.; Hu, X.; Jiang, L. Titanium dioxide-coated biochar composites as adsorptive and photocatalytic degradation materials for the removal of aqueous organic pollutants. J. Chem. Technol. Biotechnol. 2018, 93, 783–791. [Google Scholar] [CrossRef]
- Ajith, N.; Bhattacharyya, K.; Ipte, P.R.; Satpati, A.K.; Tripathi, A.K.; Verma, R.; Swain, K.K. Interaction of arsenic (III) and arsenic (V) on manganese dioxide: XPS and electrochemical investigations. J. Environ. Sci. Health A 2019, 54, 277–285. [Google Scholar] [CrossRef]
- Chong, M.N.; Jin, B.; Chow, C.W.; Saint, C. Recent developments in photocatalytic water treatment technology: A review. Water Res. 2010, 44, 2997–3027. [Google Scholar] [CrossRef] [PubMed]
- Su, H.; Lv, X.; Zhang, Z.; Yu, J.; Wang, T. Arsenic removal from water by photocatalytic functional Fe2O3–TiO2 porous ceramic. J. Porous Mater. 2017, 24, 1227–1235. [Google Scholar] [CrossRef]
- Paudyal, H.; Ohto, K.; Kawakita, H.; Inoue, K. Recovery of fluoride from water through adsorption using orange–waste gel, followed by desorption using saturated lime water. J. Matter. Cycles Waste Manag. 2020, 22, 1484–1491. [Google Scholar] [CrossRef]
- Paudyal, H.; Pangeni, B.; Inoue, K.; Harada, H.; Kawakita, H.; Ohto, K.; Alam, S. Adsorptive removal of trace concentration of fluoride using orange waste treated using concentrated sulfuric acid. IJMSA 2017, 6, 212. [Google Scholar] [CrossRef]
- Zhang, S.; Lu, X. Treatment of wastewater containing reactive brilliant blue KN-R using TiO2/BC composite as heterogeneous photocatalyst and adsorbent. Chemosphere 2018, 206, 777–783. [Google Scholar] [CrossRef]
- Zhang, H.; Wang, Z.; Li, R.; Guo, J.; Li, Y.; Zhu, J.; Xie, X. TiO2 supported on reed straw biochar as an adsorptive and photocatalytic composite for the efficient degradation of sulfamethoxazole in aqueous matrices. Chemosphere 2017, 185, 351–360. [Google Scholar] [CrossRef]
- Pant, B.; Park, M.; Park, S.J. Recent advances in TiO2 films prepared by sol-gel methods for photocatalytic degradation of organic pollutants and antibacterial activities. Coatings 2019, 9, 613. [Google Scholar] [CrossRef]

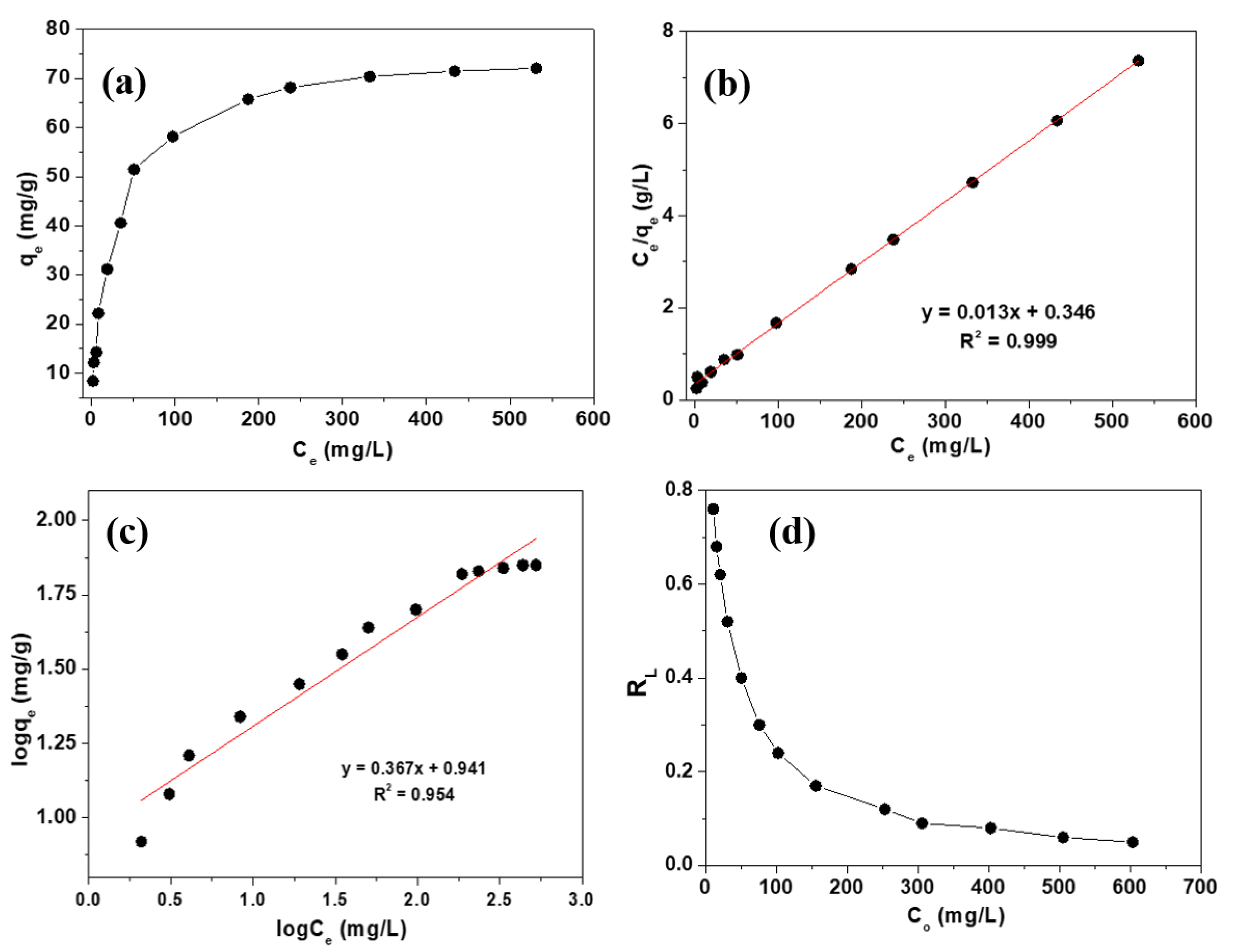


| Adsorbate | Adsorbent | Langmuir Model | Freundlich Model | ||||
|---|---|---|---|---|---|---|---|
| qm (mg/g) | b (L/mg) | R2 | KF (mg/g) | n | R2 | ||
| As(III) | PP@TiO2 | 76.92 | 0.03 | 0.999 | 8.72 | 2.72 | 0.954 |
| Adsorbent | Optimum pH | qm (mg/g) | Reference |
|---|---|---|---|
| Orange juice residue | 10.0 | 68.16 | [21] |
| Watermelon rind | 8.2 | 3.40 | [42] |
| Thiol functionalized sugarcane bagasse | 7 | 28.57 | [44] |
| Granular titanium dioxide | 7 | 32.4 | [37] |
| Fe3O4 nanoparticles | 7 | 46.06 | [65] |
| Iron–modified activated carbon | 7.6–8.0 | 38.8 | [66] |
| Amorphous iron hydroxide | 6–8 | 28.0 | [67] |
| Fe3O4/sugarcane bagasse activated carbon composite | 8 | 6.69 | [16] |
| ZrO2 nanosheets | 6 | 74.9 | [9] |
| Iron modified bamboo charcoal | 4–5 | 7.23 | [68] |
| Fe(III) loaded pomegranate waste | 9 | 50.0 | [18] |
| Al-based MOF graphene–oxide nanocomposite | 6.1 | 65.0 | [17] |
| ZrO2–sawdust | 7 | 29.0 | [69] |
| Copper–impregnated coconut husk carbon | 6.5 | 20.35 | [70] |
| TiO2 impregnated pomegranate peels (PP@TiO2) | 7 | 76.92 | This study |
| Order | Adsorbate | R2 | qe (exp) (mg/g) | qe (cal) (mg/g) | k1 (min–1) | k2 (mg/g/min) |
|---|---|---|---|---|---|---|
| Pseudo–2nd | As(III) | 0.999 | 24.7 | 25.51 | – | 1.32 × 10−3 |
| Pseudo–1st | As(III) | 0.932 | 24.7 | 15.45 | 9.85 × 10−3 | – |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Poudel, B.R.; Aryal, R.L.; Bhattarai, S.; Koirala, A.R.; Gautam, S.K.; Ghimire, K.N.; Pant, B.; Park, M.; Paudyal, H.; Pokhrel, M.R. Agro-Waste Derived Biomass Impregnated with TiO2 as a Potential Adsorbent for Removal of As(III) from Water. Catalysts 2020, 10, 1125. https://doi.org/10.3390/catal10101125
Poudel BR, Aryal RL, Bhattarai S, Koirala AR, Gautam SK, Ghimire KN, Pant B, Park M, Paudyal H, Pokhrel MR. Agro-Waste Derived Biomass Impregnated with TiO2 as a Potential Adsorbent for Removal of As(III) from Water. Catalysts. 2020; 10(10):1125. https://doi.org/10.3390/catal10101125
Chicago/Turabian StylePoudel, Bhoj Raj, Ram Lochan Aryal, Sitaram Bhattarai, Agni Raj Koirala, Surendra Kumar Gautam, Kedar Nath Ghimire, Bishweshwar Pant, Mira Park, Hari Paudyal, and Megh Raj Pokhrel. 2020. "Agro-Waste Derived Biomass Impregnated with TiO2 as a Potential Adsorbent for Removal of As(III) from Water" Catalysts 10, no. 10: 1125. https://doi.org/10.3390/catal10101125
APA StylePoudel, B. R., Aryal, R. L., Bhattarai, S., Koirala, A. R., Gautam, S. K., Ghimire, K. N., Pant, B., Park, M., Paudyal, H., & Pokhrel, M. R. (2020). Agro-Waste Derived Biomass Impregnated with TiO2 as a Potential Adsorbent for Removal of As(III) from Water. Catalysts, 10(10), 1125. https://doi.org/10.3390/catal10101125








