Palladium Catalysts Based on Porous Aromatic Frameworks, Modified with Ethanolamino-Groups, for Hydrogenation of Alkynes, Alkenes and Dienes
Abstract
1. Introduction
2. Results and Discussion
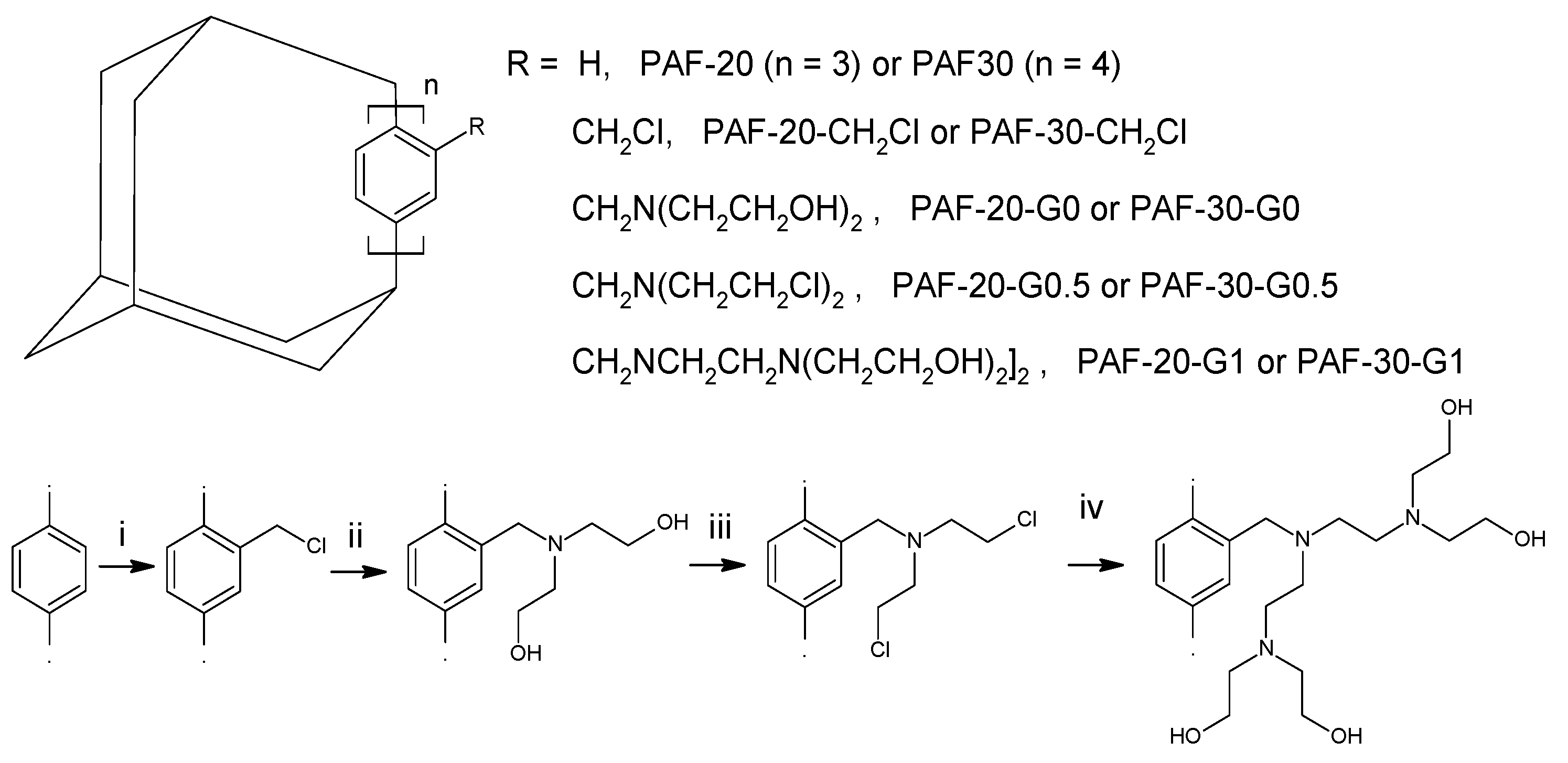
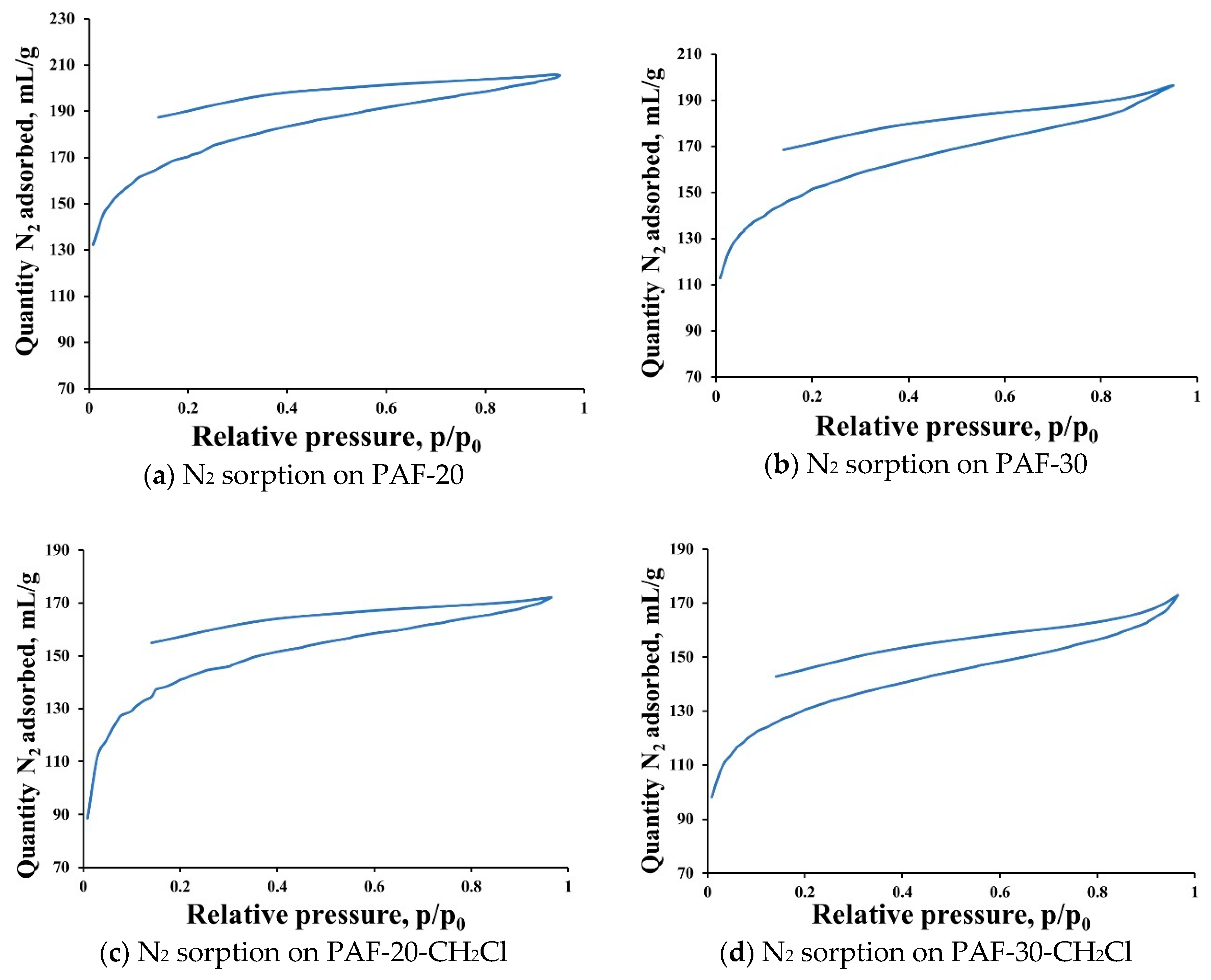
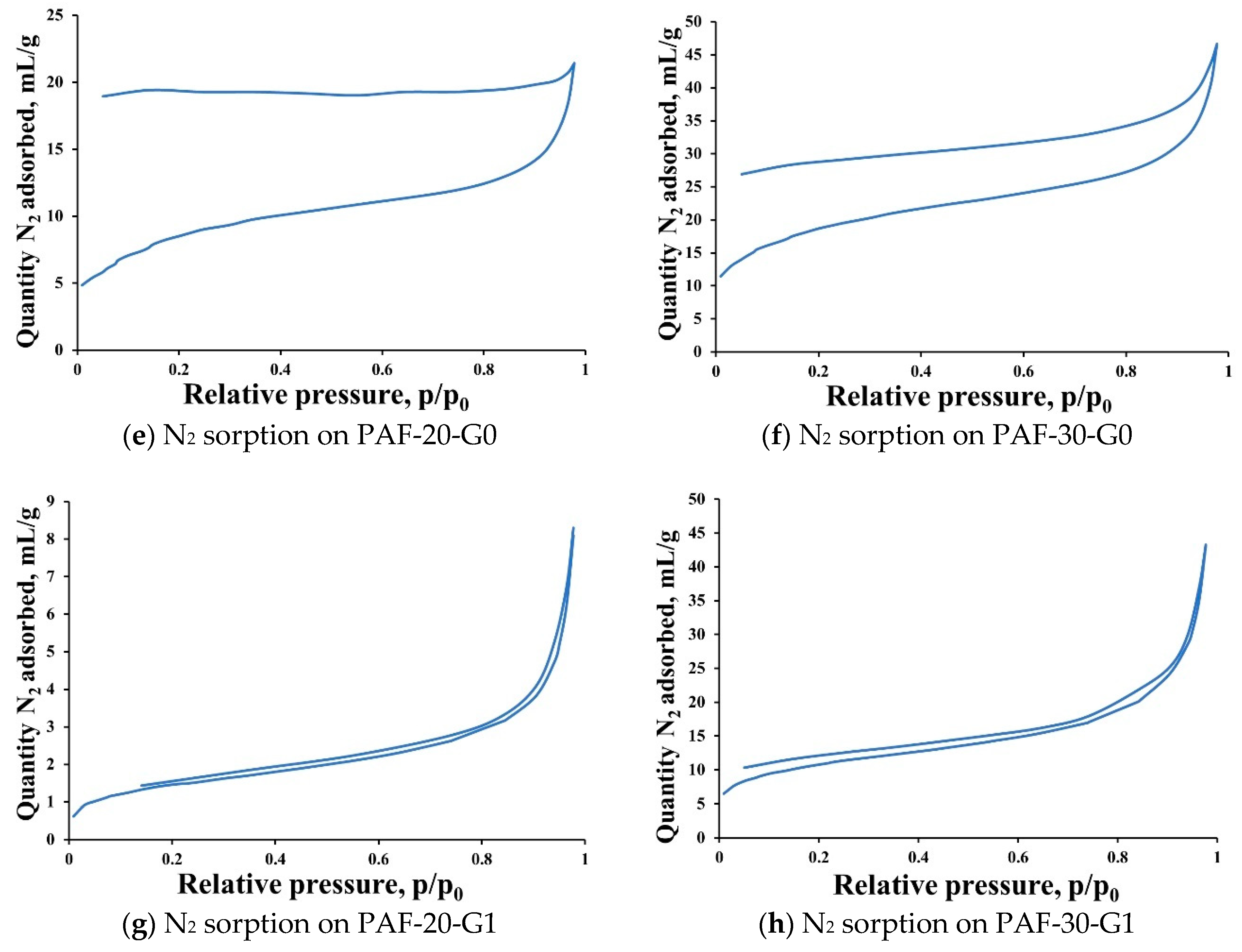
2.1. Synthesis and Characterization of Supports
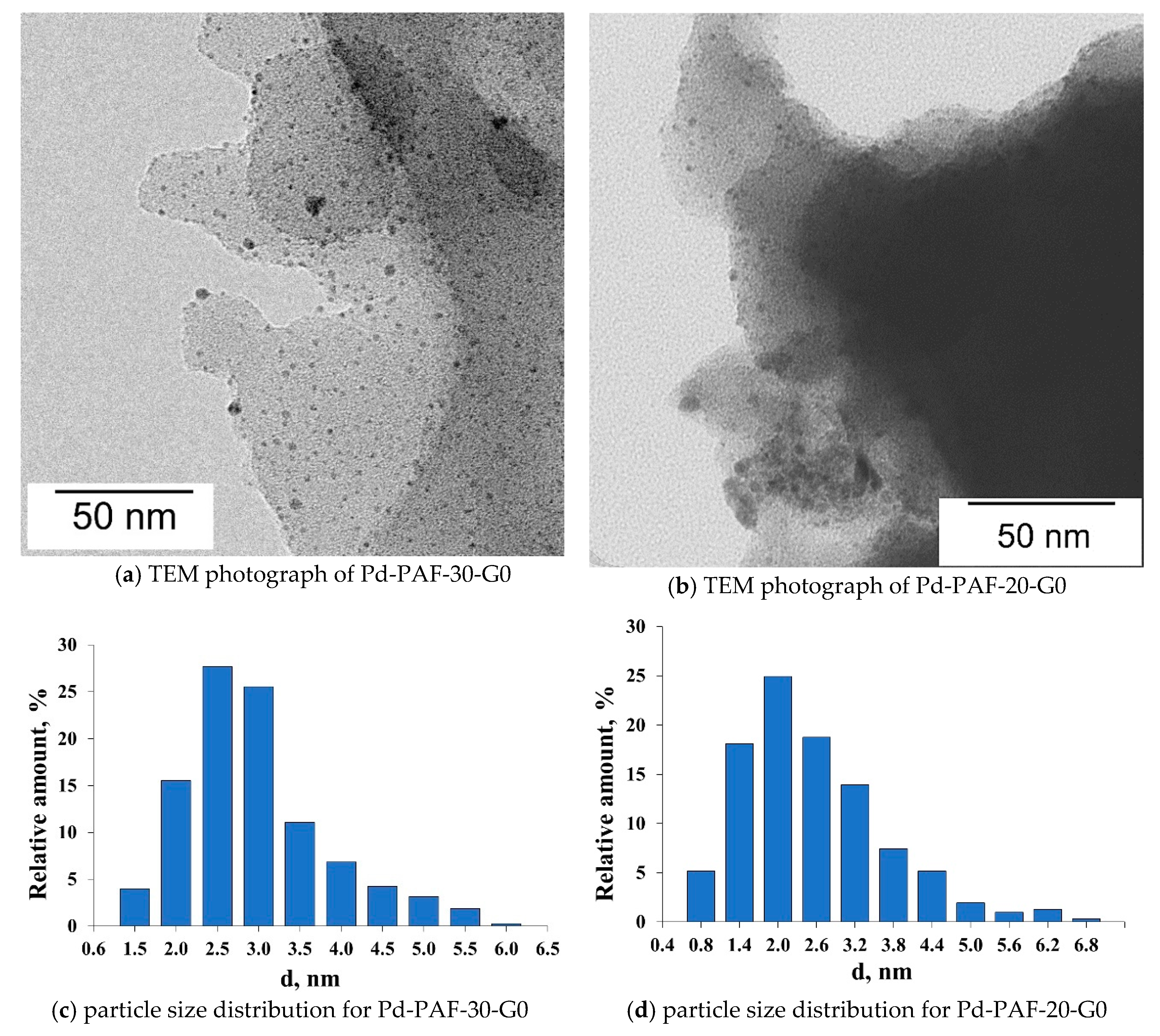
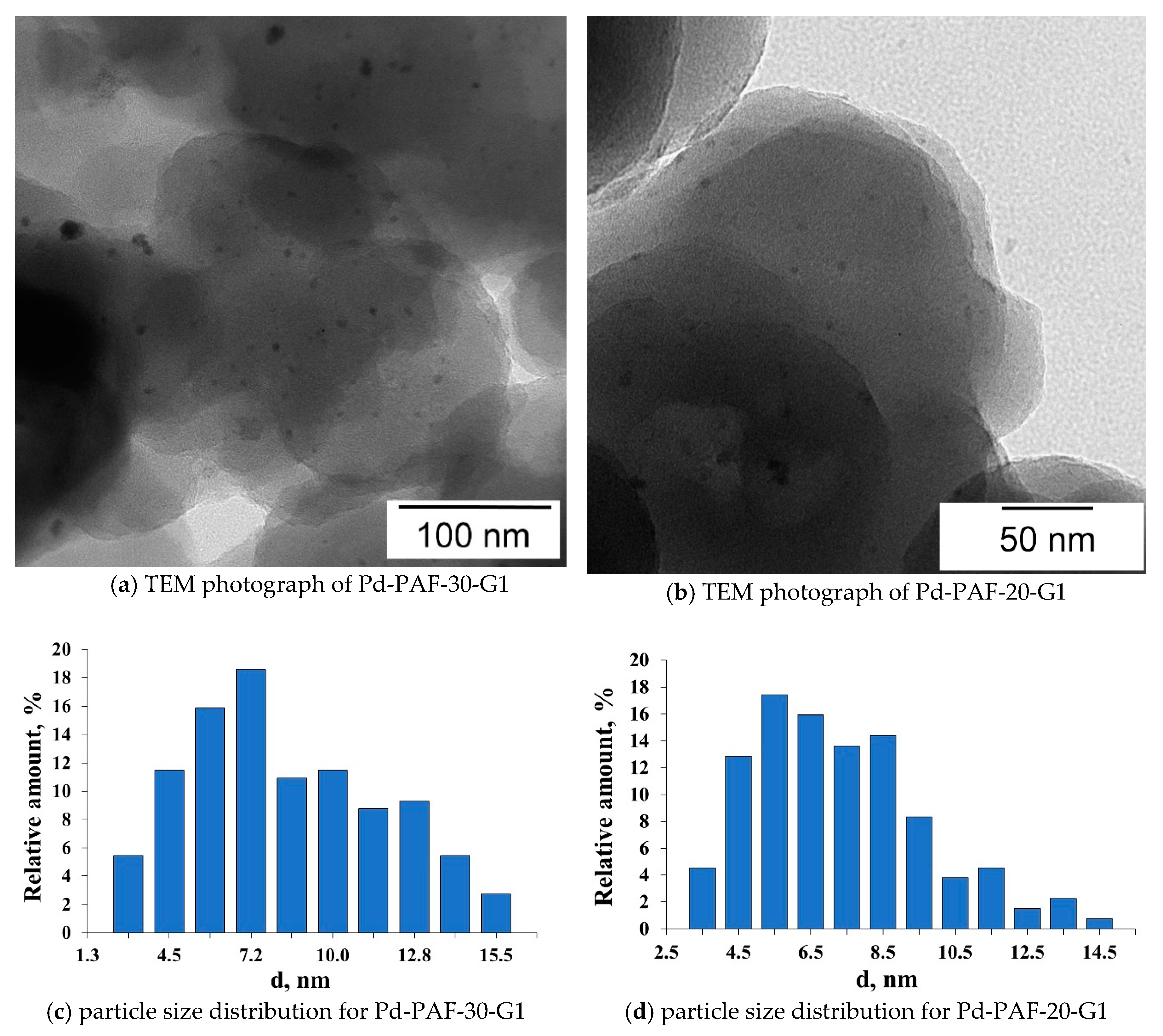
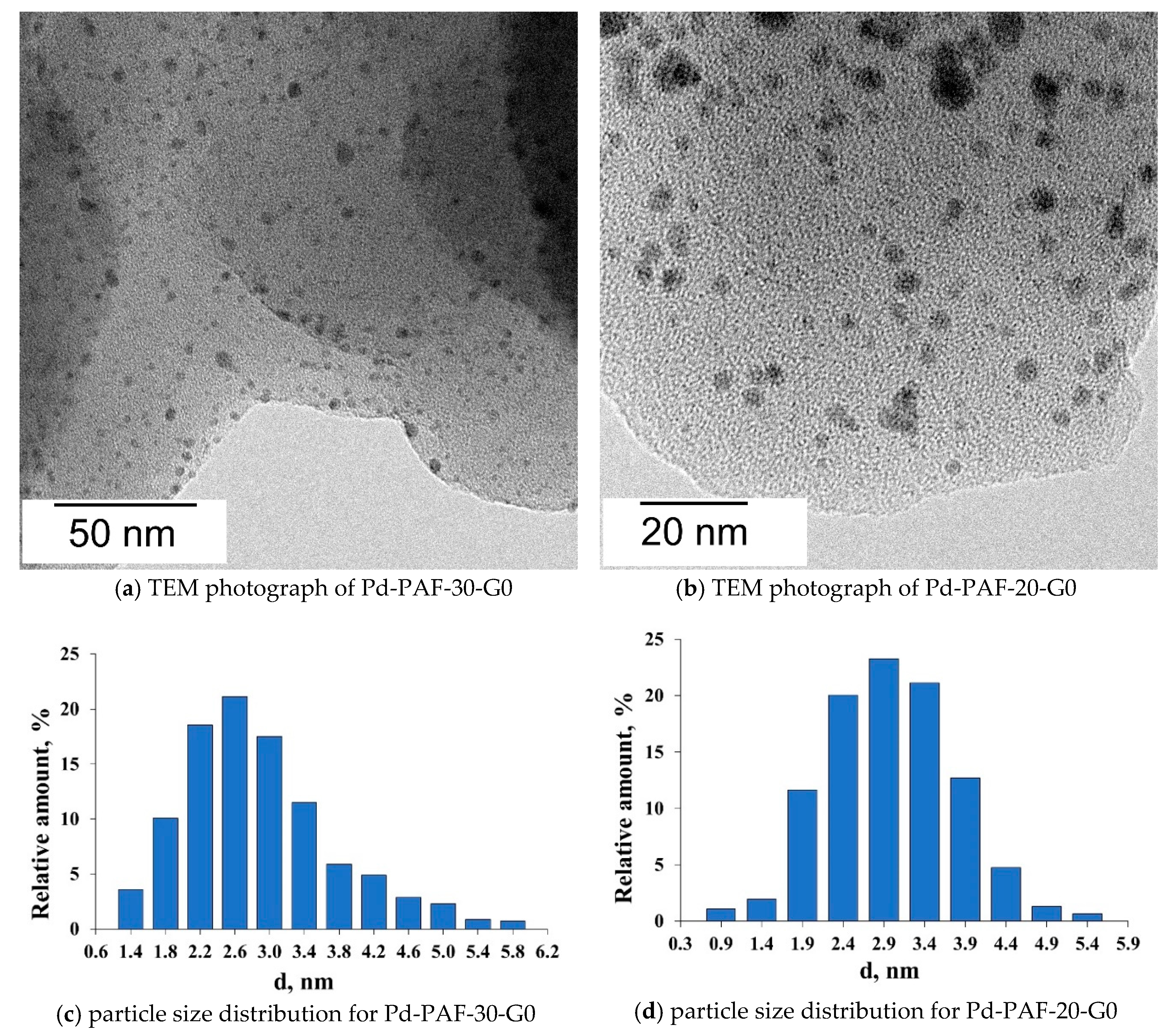
2.2. Characterization of Palladium Catalysts
2.3. Catalytic Activity
3. Materials and Methods
3.1. Used Reagents
3.2. Synthesis of PAF-20-CH2Cl
3.3. Synthesis of PAF-20-G0
3.4. Synthesis of PAF-20-G1
3.5. Synthesis of Catalyst Pd-PAF-G0 and Pd-PAF-G1
3.6. Catalytic Experiments
3.7. Characterization
3.7.1. Low Temperature Nitrogen Adsorption
3.7.2. Transmission Electron Microscopy (TEM)
3.7.3. X-ray Photoelectron Spectroscopy (XPS)
3.7.4. Gas-Liquid Chromatography
3.7.5. Atomic Absorption Spectroscopy
4. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Li, L.; Zhou, C.; Zhao, H.; Wang, R. Spatial control of palladium nanoparticles in flexible click-based porous organic polymers for hydrogenation of olefins and nitrobenzene. Nano Res. 2015, 8, 709–721. [Google Scholar] [CrossRef]
- Li, L.; Zhao, H.; Wang, R. Tailorable synthesis of porous organic polymers decorating ultrafine palladium nanoparticles for hydrogenation of olefins. ACS Catal. 2015, 5, 948–955. [Google Scholar] [CrossRef]
- Garg, G.; Foltran, S.; Favier, I.; Pla, D.; Medina-González, Y.; Gómez, M. Palladium nanoparticles stabilized by novel choline-based ionic liquids in glycerol applied in hydrogenation reactions. Catal. Today 2020, 346, 69–75. [Google Scholar] [CrossRef]
- Chung, J.; Kim, C.; Jeong, H.; Yu, T.; Binh, D.H.; Jang, J.; Lee, J.; Kim, B.M.; Lim, B. Selective semihydrogenation of alkynes on shape-controlled palladium nanocrystals. Chem. Asian J. 2013, 8, 919–925. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Y.; Riduan, S.N. Functional porous organic polymers for heterogeneous catalysis. Chem. Soc. Rev. 2012, 41, 2083–2094. [Google Scholar] [CrossRef]
- Zhang, Q.; Yang, Y.; Zhang, S. Novel functionalized microporous organic networks based on triphenylphosphine. Chem. Eur. J. 2013, 19, 10024–10029. [Google Scholar] [CrossRef]
- Hausoul, P.J.C.; Eggenhuisen, T.M.; Nand, D.; Baldus, M.; Weckhuysen, B.M.; Klein Gebbink, R.J.M.; Bruijnincx, P.C.A. Development of a 4,4′-biphenyl/phosphine-based COF for the heterogeneous Pd-catalysed telomerisation of 1,3-butadiene. Catal. Sci. Technol. 2013, 3, 2571–2579. [Google Scholar] [CrossRef][Green Version]
- Karakhanov, E.A.; Maksimov, A.L.; Zolotukhina, A.V.; Kardasheva, Y.S. Hydrogenation catalysts based on metal nanoparticles stabilized by organic ligands. Russ. Chem. Bull. 2013, 62, 1465–1492. [Google Scholar]
- Karakhanov, E.; Maximov, A.; Kardasheva, Y.; Semernina, V.; Zolotukhina, A.; Ivanov, A.; Abbott, G.; Rosenberg, E.; Vinokurov, V. Pd nanoparticles in dendrimers immobilized on silica-polyamine composites as catalysts for selective hydrogenation. ACS Appl. Mater. Interfaces 2014, 6, 8807–8816. [Google Scholar] [CrossRef]
- Karakhanov, E.A.; Maximov, A.L.; Zolotukhina, A.V. Selective semi-hydrogenation of phenyl acetylene by Pd nanocatalysts encapsulated into dendrimer networks. Mol. Catal. 2019, 469, 98–110. [Google Scholar] [CrossRef]
- Chen, H.; He, Y.; Pfefferle, L.D.; Pu, W.; Wu, Y.; Qi, S. Phenol Catalytic Hydrogenation over Palladium Nanoparticles Supported on Metal-Organic Frameworks in the Aqueous Phase. ChemCatChem 2018, 10, 2558–2570. [Google Scholar] [CrossRef]
- Jansen, J.F.G.A.; De Brabander-Van Den Berg, E.M.M.; Meijer, E.W. Encapsulation of guest molecules into a dendritic box. Science 1994, 266, 1226–1229. [Google Scholar] [CrossRef] [PubMed]
- Niu, Y.; Crooks, R.M. Dendrimer-encapsulated metal nanoparticles and their applications to catalysis. C. R. Chim. 2003, 6, 1049–1059. [Google Scholar] [CrossRef]
- Yates, C.R.; Hayes, W. Synthesis and applications of hyperbranched polymers. Eur. Polym. J. 2004, 40, 1257–1281. [Google Scholar] [CrossRef]
- Karakhanov, E.A.; Maximov, A.L.; Skorkin, V.A.; Zolotukhina, A.V.; Smerdov, A.S.; Tereshchenko, A.Y. Nanocatalysts based on dendrimers. Pure Appl. Chem. 2009, 81, 2013–2023. [Google Scholar] [CrossRef]
- Karakhanov, E.A.; Maximov, A.L.; Zakharyan, E.M.; Zolotukhina, A.V.; Ivanov, A.O. Palladium nanoparticles on dendrimer-containing supports as catalysts for hydrogenation of unsaturated hydrocarbons. Mol. Catal. 2017, 440, 107–119. [Google Scholar] [CrossRef]
- Karakanov, E.A.; Zolotukhina, A.V.; Ivanov, A.O.; Maximov, A.L. Dendrimer-Encapsulated Pd Nanoparticles, Immobilized in Silica Pores, as Catalysts for Selective Hydrogenation of Unsaturated Compounds. Chem. Open 2019, 8, 358–381. [Google Scholar] [CrossRef]
- Krishnan, G.R.; Sreekumar, K. Synthesis and Characterization of Polystyrene Supported Catalytically Active Poly(amidoamine) Dendrimer-Palladium Nanoparticle Conjugates. Soft Mater. 2010, 8, 114–129. [Google Scholar] [CrossRef]
- Alvarez, J.; Sun, L.; Crooks, R.M. Electroactive composite dendrimer films containing thiophene-terminated poly(amidoamine) dendrimers cross-linked by poly(3-methylthiophene). Chem. Mater. 2002, 14, 3995–4001. [Google Scholar] [CrossRef]
- Murugan, E.; Rangasamy, R. Synthesis, characterization, and heterogeneous catalysis of polymer-supported poly(propyleneimine) dendrimer stabilized gold nanoparticle catalyst. J. Polym. Sci. Part A Polym. Chem. 2010, 48, 2525–2532. [Google Scholar] [CrossRef]
- Li, L.; Zhao, H.; Wang, J.; Wang, R. Facile fabrication of ultrafine palladium nanoparticles with size- and location-control in click-based porous organic polymers. ACS Nano 2014, 8, 5352–5364. [Google Scholar]
- Karakhanov, E.; Kardasheva, Y.; Kulikov, L.; Maximov, A.; Zolotukhina, A.; Vinnikova, M.; Ivanov, A. Sulfide catalysts supported on porous aromatic frameworks for naphthalene hydroprocessing. Catalysts 2016, 6, 1–11. [Google Scholar]
- Yuan, Y.; Zhu, G. Porous Aromatic Frameworks as a Platform for Multifunctional Applications. ACS Cent. Sci. 2019, 5, 409–418. [Google Scholar] [PubMed]
- Maximov, A.; Zolotukhina, A.; Kulikov, L.; Kardasheva, Y.; Karakhanov, E. Ruthenium catalysts based on mesoporous aromatic frameworks for the hydrogenation of arenes. React. Kinet. Mech. Catal. 2016, 117, 729–743. [Google Scholar]
- Wang, F.; Mielby, J.; Richter, F.H.; Wang, G.; Prieto, G.; Kasama, T.; Weidenthaler, C.; Bongard, H.J.; Kegnæs, S.; Fürstner, A.; et al. A polyphenylene support for pd catalysts with exceptional catalytic activity. Angew. Chem. Int. Ed. 2014, 53, 8645–8648. [Google Scholar]
- Kulikov, L.A.; Terenina, M.V.; Kryazheva, I.Y.; Karakhanov, E.A. Unsaturated-compound hydrogenation nanocatalysts based on palladium and platinum particles immobilized in pores of mesoporous aromatic frameworks. Pet. Chem. 2017, 57, 222–229. [Google Scholar]
- Su, J.; Chen, J.S. Synthetic porous materials applied in hydrogenation reactions. Microporous Mesoporous Mater. 2017, 237, 246–259. [Google Scholar]
- Ben, T.; Ren, H.; Shengqian, M.; Cao, D.; Lan, J.; Jing, X.; Wang, W.; Xu, J.; Deng, F.; Simmons, J.M.; et al. Targeted synthesis of a porous aromatic framework with high stability and exceptionally high surface area. Angew. Chem. Int. Ed. 2009, 48, 9457–9460. [Google Scholar]
- Garibay, S.J.; Weston, M.H.; Mondloch, J.E.; Colón, Y.J.; Farha, O.K.; Hupp, J.T.; Nguyen, S.T. Accessing functionalized porous aromatic frameworks (PAFs) through a de novo approach. CrystEngComm 2013, 15, 1515–1519. [Google Scholar]
- Tian, Y.; Song, J.; Zhu, Y.; Zhao, H.; Muhammad, F.; Ma, T.; Chen, M.; Zhu, G. Understanding the desulphurization process in an ionic porous aromatic framework. Chem. Sci. 2019, 10, 606–613. [Google Scholar] [PubMed]
- Vilian, A.T.E.; Puthiaraj, P.; Kwak, C.H.; Hwang, S.K.; Huh, Y.S.; Ahn, W.S.; Han, Y.K. Fabrication of Palladium Nanoparticles on Porous Aromatic Frameworks as a Sensing Platform to Detect Vanillin. ACS Appl. Mater. Interfaces 2016, 8, 12740–12747. [Google Scholar] [CrossRef] [PubMed]
- Yang, Y.; Wang, T.; Jing, X.; Zhu, G. Phosphine-based porous aromatic frameworks for gold nanoparticle immobilization with superior catalytic activities. J. Mater. Chem. A 2019, 7, 10004–10009. [Google Scholar] [CrossRef]
- Nikolaev, S.A.; Zanaveskin, L.N.; Smirnov, V.V.; Averyanov, V.A.; Zanaveskin, K.L. Catalytic hydrogenation of alkyne and alkadiene impurities from alkenes. Practical and theoretical aspects. Russ. Chem. Rev. 2009, 78, 231–247. [Google Scholar] [CrossRef]
- Mallat, T.; Baiker, A. Selectivity enhancement in heterogeneous catalysis induced by reaction modifiers. Appl. Catal. A Gen. 2000, 200, 3–22. [Google Scholar] [CrossRef]
- Xing, R.; Liu, Y.; Wu, H.; Li, X.; He, M.; Wu, P. Preparation of active and robust palladium nanoparticle catalysts stabilized by diamine-functionalized mesoporous polymers. Chem. Commun. 2008, 47, 6297–6299. [Google Scholar] [CrossRef]
- Karakhanov, E.; Maximov, A.; Terenina, M.; Vinokurov, V.; Kulikov, L.; Makeeva, D.; Glotov, A. Selective hydrogenation of terminal alkynes over palladium nanoparticles within the pores of amino-modified porous aromatic frameworks. Catal. Today 2019. [Google Scholar] [CrossRef]
- Verde-Sesto, E.; Merino, E.; Rangel-Rangel, E.; Corma, A.; Iglesias, M.; Sánchez, F. Postfunctionalized Porous Polymeric Aromatic Frameworks with an Organocatalyst and a Transition Metal Catalyst for Tandem Condensation-Hydrogenation Reactions. ACS Sustain. Chem. Eng. 2016, 4, 1078–1084. [Google Scholar] [CrossRef]
- Li, L.; Chen, Z.; Zhong, H.; Wang, R. Urea-based porous organic frameworks: Effective supports for catalysis in neat water. Chem. Eur. J. 2014, 20, 3050–3060. [Google Scholar] [CrossRef]
- Zhong, H.; Gong, Y.; Zhang, F.; Li, L.; Wang, R. Click-based porous organic framework containing chelating terdentate units and its application in hydrogenation of olefins. J. Mater. Chem. A 2014, 2, 7502–7508. [Google Scholar] [CrossRef]
- Tang, D.; Sun, X.; Zhao, D.; Zhu, J.; Zhang, W.; Xu, X.; Zhao, Z. Nitrogen-Doped Carbon Xerogels Supporting Palladium Nanoparticles for Selective Hydrogenation Reactions: The Role of Pyridine Nitrogen Species. ChemCatChem 2018, 10, 1291–1299. [Google Scholar] [CrossRef]
- Méry, D.; Astruc, D. Dendritic catalysis: Major concepts and recent progress. Coord. Chem. Rev. 2006, 250, 1965–1979. [Google Scholar] [CrossRef]
- Lu, S.; Hu, Y.; Wan, S.; McCaffrey, R.; Jin, Y.; Gu, H.; Zhang, W. Synthesis of Ultrafine and Highly Dispersed Metal Nanoparticles Confined in a Thioether-Containing Covalent Organic Framework and Their Catalytic Applications. J. Am. Chem. Soc. 2017, 139, 17082–17088. [Google Scholar] [CrossRef] [PubMed]
- Wang, Q.; Tsumori, N.; Kitta, M.; Xu, Q. Fast Dehydrogenation of Formic Acid over Palladium Nanoparticles Immobilized in Nitrogen-Doped Hierarchically Porous Carbon. ACS Catal. 2018, 8, 12041–12045. [Google Scholar] [CrossRef]
- Neeli, C.K.P.; Puthiaraj, P.; Lee, Y.R.; Chung, Y.M.; Baeck, S.H.; Ahn, W.S. Transfer hydrogenation of nitrobenzene to aniline in water using Pd nanoparticles immobilized on amine-functionalized UiO-66. Catal. Today 2018, 303, 227–234. [Google Scholar] [CrossRef]
- Sadjadi, S.; Koohestani, F. Pd immobilized on polymeric network containing imidazolium salt, cyclodextrin and carbon nanotubes: Efficient and recyclable catalyst for the hydrogenation of nitroarenes in aqueous media. J. Mol. Liq. 2020, 301, 112414. [Google Scholar] [CrossRef]
- Zhou, S.; Shang, L.; Zhao, Y.; Shi, R.; Waterhouse, G.I.N.; Huang, Y.; Zheng, L.; Zhang, T. Pd Single-Atom Catalysts on Nitrogen-Doped Graphene for the Highly Selective Photothermal Hydrogenation of Acetylene to Ethylene. Adv. Mater. 2019, 31, 1900509. [Google Scholar] [CrossRef]
- Crooks, R.M.; Zhao, M.; Sun, L.; Chechik, V.; Yeung, L.K. Dendrimer-encapsulated metal nanoparticles: Synthesis, characterization, and applications to catalysis. Acc. Chem. Res. 2001, 34, 181–190. [Google Scholar] [CrossRef]
- Boronoev, M.P.; Zolotukhina, A.V.; Ignatyeva, V.I.; Terenina, M.V.; Maximov, A.L.; Karakhanov, E.A. Palladium Catalysts Based on Mesoporous Organic Materials in Semihydrogenation of Alkynes. Macromol. Symp. 2016, 363, 57–63. [Google Scholar] [CrossRef]
- King, A.S.H.; Twyman, L.J. Heterogeneous and solid supported dendrimer catalysts. J. Chem. Soc. Perkin 2002, 2, 2209–2218. [Google Scholar] [CrossRef]
- Soğukömeroğulları, H.G.; Karataş, Y.; Celebi, M.; Gülcan, M.; Sönmez, M.; Zahmakiran, M. Palladium nanoparticles decorated on amine functionalized graphene nanosheets as excellent nanocatalyst for the hydrogenation of nitrophenols to aminophenol counterparts. J. Hazard. Mater. 2019, 369, 96–107. [Google Scholar] [CrossRef]
- Yang, J.; Yuan, M.; Xu, D.; Zhao, H.; Zhu, Y.; Fan, M.; Zhang, F.; Dong, Z. Highly dispersed ultrafine palladium nanoparticles encapsulated in a triazinyl functionalized porous organic polymer as a highly efficient catalyst for transfer hydrogenation of aldehydes. J. Mater. Chem. A 2018, 6, 18242–18251. [Google Scholar] [CrossRef]
- Xu, D.; Wang, F.; Yu, G.; Zhao, H.; Yang, J.; Yuan, M.; Zhang, X.; Dong, Z. Aminal-based Hypercrosslinked Polymer Modified with Small Palladium Nanoparticles for Efficiently Catalytic Reduction of Nitroarenes. ChemCatChem 2018, 10, 4569–4577. [Google Scholar] [CrossRef]
- Yuan, Y.; Sun, F.; Ren, H.; Jing, X.; Wang, W.; Ma, H.; Zhao, H.; Zhu, G. Targeted synthesis of a porous aromatic framework with a high adsorption capacity for organic molecules. J. Mater. Chem. 2011, 21, 13498–13502. [Google Scholar] [CrossRef]
- Gangadharan, D.; Dhandhala, N.; Dixit, D.; Thakur, R.S.; Popat, K.M.; Anand, P.S. Investigation of solid supported dendrimers for water disinfection. J. Appl. Polym. Sci. 2012, 124, 1384–1391. [Google Scholar] [CrossRef]
- Lu, W.; Sculley, J.P.; Yuan, D.; Krishna, R.; Wei, Z.; Zhou, H.C. Polyamine-tethered porous polymer networks for carbon dioxide capture from flue gas. Angew. Chem. Int. Ed. 2012, 51, 7480–7484. [Google Scholar] [CrossRef]
- Law, R.V.; Sherrington, D.C.; Snape, C.E.; Ando, I.; Korosu, H. Solid State 13C MAS NMR Studies of Anion Exchange Resins and Their Precursors. Ind. Eng. Chem. Res. 1995, 34, 2740–2749. [Google Scholar] [CrossRef]
- Rangel-Rangel, E.; Verde-Sesto, E.; Rasero-Almansa, A.M.; Iglesias, M.; Sánchez, F. Porous aromatic frameworks (PAFs) as efficient supports for N-heterocyclic carbene catalysts. Catal. Sci. Technol. 2016, 6, 6037–6045. [Google Scholar] [CrossRef]
- Thommes, M.; Kaneko, K.; Neimark, A.V.; Olivier, J.P.; Rodriguez-Reinoso, F.; Rouquerol, J.; Sing, K.S.W. Physisorption of gases, with special reference to the evaluation of surface area and pore size distribution (IUPAC Technical Report). Pure Appl. Chem. 2015, 87, 1051–1069. [Google Scholar] [CrossRef]
- Jeromenok, J.; Weber, J. Restricted access: On the nature of adsorption/desorption hysteresis in amorphous, microporous polymeric materials. Langmuir 2013, 29, 12982–12989. [Google Scholar] [CrossRef]


| Samples | Materials Based on PAF-20 | Materials Based on PAF-30 | ||
|---|---|---|---|---|
| SBET, m2/g | Total Pore Volume (BJH), cm3/g | SBET, m2/g | Total Pore Volume (BJH), cm3/g | |
| PAF | 579 | 0.316 | 506 | 0.311 |
| PAF-CH2Cl | 472 | 0.264 | 436 | 0.262 |
| PAF-G0 | 29 | 0.026 | 61 | 0.054 |
| PAF-G1 | 5 | 0.001 | 38 | 0.007 |
| Material | Element Content, Mass. % | |
|---|---|---|
| Cl | N | |
| PAF-20-CH2Cl | 3.08% | - |
| PAF-30-CH2Cl | 3.00% | - |
| PAF-20-G0 | 0.91% | 1.84% |
| PAF-30-G0 | 1.19% | 1.68% |
| PAF-20-G1 | 2.43% | 1.88% |
| PAF-30-G1 | 3.30% | 2.16% |
| Material | Pd-PAF-20-G0 | Pd-PAF-30-G0 | Pd-PAF-20-G1 | Pd-PAF-30-G1 |
|---|---|---|---|---|
| Pd, mass % | 2.4 | 1.0 | 0.6 | 1.8 |
| Catalysts | C | O | N | Pd | Cl |
|---|---|---|---|---|---|
| Pd-PAF-20-G0 | 82.2 at.% | 13.3 at.% | 1.5 at.% | 2.8 at.% | 0.1 at.% |
| Pd-PAF-30-G0 | 85.6 at.% | 11.2 at.% | 1.7 at.% | 1.3 at.% | 0.2 at.% |
| Pd-PAF-20-G1 | 87.9 at.% | 6.8 at.% | 3.4 at.% | 0.3 at.% | 0.6 at.% |
| Pd-PAF-30-G1 | 87.0 at.% | 9.2 at.% | 3.0 at.% | 0.3 at.% | 0.5 at.% |
| Catalyst | Parameter | Pd0 | PdOx |
|---|---|---|---|
| Pd-PAF-20-G0 | Binding energy, eV | Pd 3d5/2 334.85 eV | Pd 3d5/2 336.58 eV |
| Pd 3d3/2 340.10 eV | Pd 3d3/2 341.82 eV | ||
| Content, % | 67 | 33 | |
| Pd-PAF-30-G0 | Binding energy, eV | Pd 3d5/2 334.65 eV | Pd 3d5/2 336.75 eV |
| Pd 3d3/2 339.96 eV | Pd 3d3/2 341.91 eV | ||
| Content, % | 56 | 44 | |
| Pd-PAF-20-G1 | Binding energy, eV | Pd 3d5/2 335.11 eV | Pd 3d5/2 336.8 eV |
| Pd 3d3/2 340.47 eV | Pd 3d3/2 342.43 eV | ||
| Content, % | 48 | 51 | |
| Pd-PAF-30-G1 | Binding energy, eV | Pd 3d5/2 335.45 eV | Pd 3d5/2 337.07 eV |
| Pd 3d3/2 340.89 eV | Pd 3d3/2 342.56 eV | ||
| Content, % | 56 | 44 |
| Substrate | Reaction Products | Product Yield, % | |||
|---|---|---|---|---|---|
| Pd-PAF-20-G0 | Pd-PAF-30-G0 | Pd-PAF-20-G1 | Pd-PAF-30-G1 | ||
| Hexyne-1 | Hexene-1 | 85 | 94 | 9 | 35 |
| Hexane | 4 | 6 | - | - | |
| Hexene-1 | Hexane | 34 | 100 | <1 | <1 |
| Cyclohexene | Cyclohexane | 11 | 12 | - | - |
| 1,3-cyclohexadiene | Cyclohexadiene | 7 | 9 | - | - |
| Octyne-1 | Octene-1 | 6 | 99 | - | - |
| Octyne-4 | Octene-4 | 3 | 4 | - | - |
| Octene-1 | Octane | 7 | 99 | <1 | 1 |
| Isomerization products | 85 | <1 | 5 | 5 | |
| 2,5-dimethyl-2,4-hexadiene | 2,5-dimethyl-3-hexene | 8 | 5 | <1 | <1 |
| 2,5-dimethylhexane | <1 | 5 | <1 | 1 | |
| 2,5-dimethyl-2-hexene | 18 | 82 | - | - | |
| Phenylacetylene | Styrene | 21 | 37 | - | - |
| Styrene | Ethylbenzene | 10 | 26 | - | - |
| 4-methoxystyrene | 4-methoxyethylbenzene | 3 | 4 | - | - |
| Substrate | Pd-PAF-20-G0 | Pd-PAF-30-G0 | Pd-PAF-20-G1 | Pd-PAF-30-G1 |
|---|---|---|---|---|
| Hexyne-1 | 94,600 | 323,400 | 100,700 | 189,800 |
| Hexene-1 | 34,600 | 305,100 | - | - |
| Cyclohexene | 11,200 | 36,600 | - | - |
| 1,3-cyclohexadiene | 7100 | 27,400 | ||
| Octyne-1 | 7100 | 302,000 | - | - |
| Octyne-4 | 5600 | 12,200 | ||
| Octene-1 | 7100 | 305,100 | - | - |
| 2,5-dimethyl-2,4-hexadiene | 27,500 | 294,900 | - | - |
| Phenylacetylene | 22,400 | 115,900 | - | - |
| Styrene | 10,200 | 79,300 | - | - |
| 4-methoxystyrene | 5600 | 12,200 |
| Catalyst | Product Yield, % | ||||
|---|---|---|---|---|---|
| Cycle 1 | Cycle 2 | Cycle 3 | Cycle 4 | Cycle 5 | |
| Pd-PAF-20-G0 | 34 | 28 | 16 | 16 | 17 |
| Pd-PAF-30-G0 | 99 | 98 | 86 | 84 | 83 |
| Sample | C | O | N | Cl | Pd | |
|---|---|---|---|---|---|---|
| Pd-PAF-20-G0 | Before reaction | 82.2 at.% | 13.3 at.% | 1.5 at.% | 0.1 at.% | 2.8 at.% |
| After 5 runs | 79.4 at.% | 16.1 at.% | 2.1 at.% | 0.2 at.% | 2.1 at.% | |
| Pd-PAF-30-G0 | Before reaction | 85.6 at.% | 11.2 at.% | 1.7 at.% | 0.2 at.% | 1.3 at.% |
| After 5 runs | 85.6 at.% | 12.3 at.% | 0.9 at.% | 0.2 at.% | 1.0 at.% | |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Kulikov, L.; Kalinina, M.; Makeeva, D.; Maximov, A.; Kardasheva, Y.; Terenina, M.; Karakhanov, E. Palladium Catalysts Based on Porous Aromatic Frameworks, Modified with Ethanolamino-Groups, for Hydrogenation of Alkynes, Alkenes and Dienes. Catalysts 2020, 10, 1106. https://doi.org/10.3390/catal10101106
Kulikov L, Kalinina M, Makeeva D, Maximov A, Kardasheva Y, Terenina M, Karakhanov E. Palladium Catalysts Based on Porous Aromatic Frameworks, Modified with Ethanolamino-Groups, for Hydrogenation of Alkynes, Alkenes and Dienes. Catalysts. 2020; 10(10):1106. https://doi.org/10.3390/catal10101106
Chicago/Turabian StyleKulikov, Leonid, Maria Kalinina, Daria Makeeva, Anton Maximov, Yulia Kardasheva, Maria Terenina, and Eduard Karakhanov. 2020. "Palladium Catalysts Based on Porous Aromatic Frameworks, Modified with Ethanolamino-Groups, for Hydrogenation of Alkynes, Alkenes and Dienes" Catalysts 10, no. 10: 1106. https://doi.org/10.3390/catal10101106
APA StyleKulikov, L., Kalinina, M., Makeeva, D., Maximov, A., Kardasheva, Y., Terenina, M., & Karakhanov, E. (2020). Palladium Catalysts Based on Porous Aromatic Frameworks, Modified with Ethanolamino-Groups, for Hydrogenation of Alkynes, Alkenes and Dienes. Catalysts, 10(10), 1106. https://doi.org/10.3390/catal10101106







