Statistical Model-Based Classification to Detect Patient-Specific Spike-and-Wave in EEG Signals
Abstract
1. Introduction
2. Materials and Methods
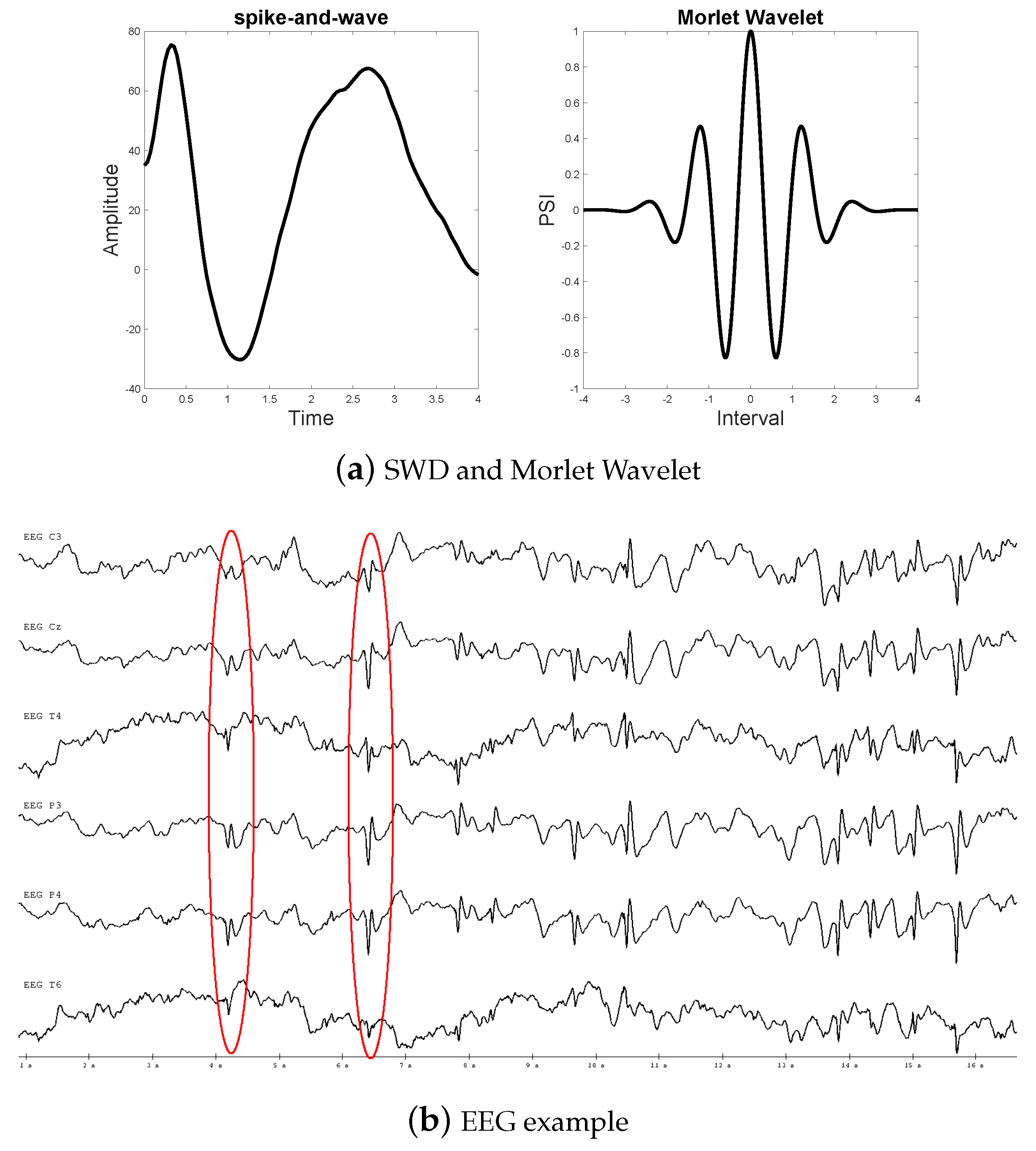
2.1. Database
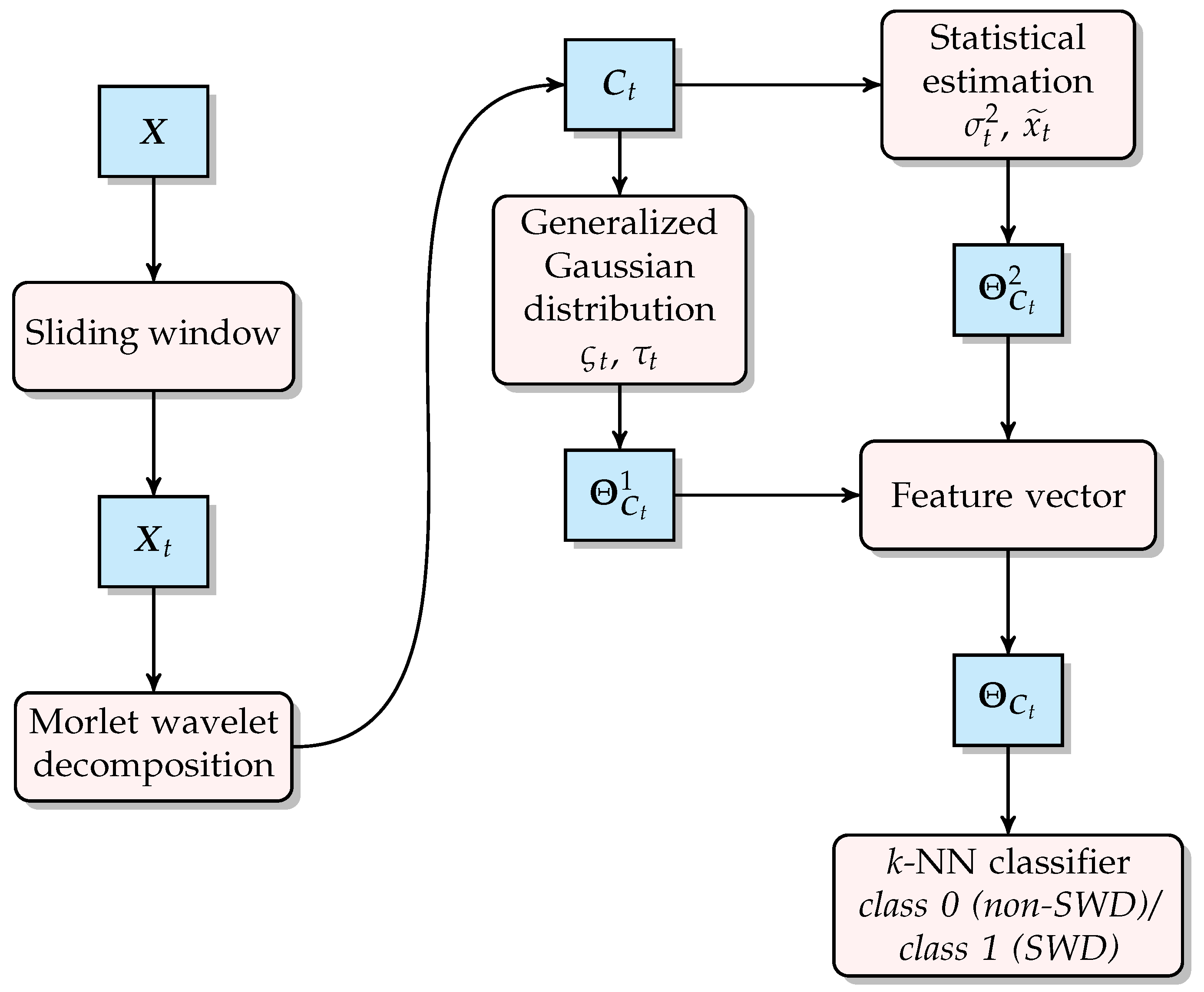
2.2. Methodology
2.2.1. Morlet Wavelet
2.2.2. Generalized Gaussian Distribution
2.2.3. Feature Parameters
2.2.4. k-Nearest Neighbors Classification
3. Results
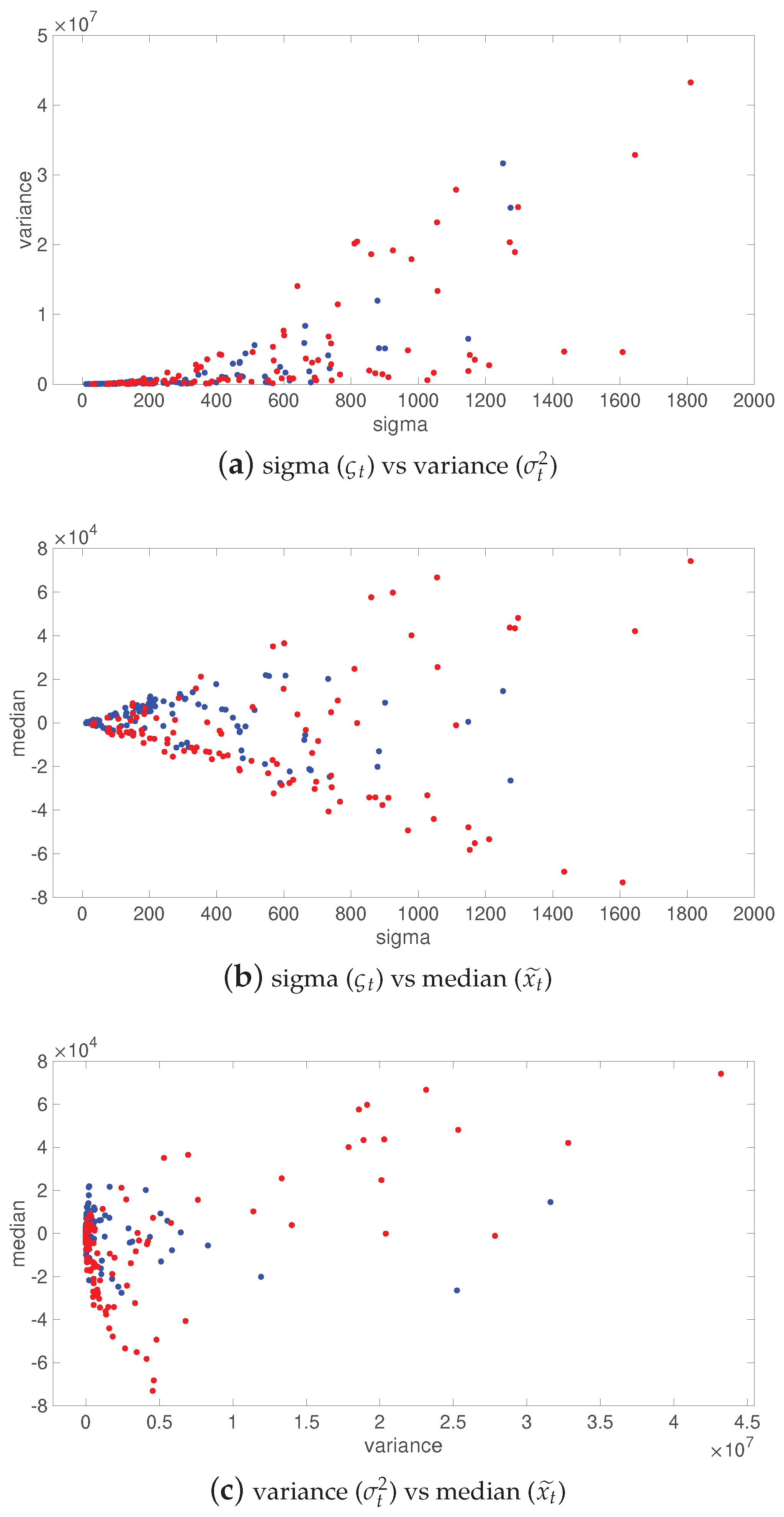
- Scale parameter vs. variance : for class 1 (SWD), one observes a direct relationship between the variance and sigma, where both parameters grow proportionally. For class 0 (non-SWD), both sigma and variance remain in a limited range of values.
- Scale parameter vs. median : as sigma grows, median increases then decreases for both SWD and non-SWD, but is larger for SWD. A cone-shaped pattern can be observed.
- Variance vs. median : as the variance grows, the median increases then decreases for SWD, while it remains in a small range (cluster) for non-SWD.
4. Discussion
5. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
Abbreviations
| EEG | Electroencephalography |
| FLENI | Fight against Pediatric Neurological Disease |
| GGD | Generalized Gaussian distribution |
| k-NN | k-nearest neighbors |
| SWD | Spike-and-wave discharge |
References
- Schomer, D.L.; da Silva, F.H.L. Niedermeyer’s Electroencephalography Basic Principles, Clinical Applications, and Related Fields; LWW: Philadelphia, PA, USA, 2010. [Google Scholar]
- Bergstrom, R.; Choi, J.; Manduca, A.; Shin, H.; Worrell, G.; Howe, C. Automated identification of multiple seizure-related and interictal epileptiform event types in the EEG of mice. Sci. Rep. 2013, 3, 1483. [Google Scholar] [CrossRef] [PubMed]
- Bhuyan, R.; Jahan, W.; Upadhyaya, N. Interictal wave pattern study in EEG of epilepsy patients. Int. J. Res. Med. Sci. 2013, 5, 3378–3384. [Google Scholar] [CrossRef][Green Version]
- Quintero-Rincón, A.; Pereyra, M.; D’Giano, C.; Batatia, H.; Risk, M. A new algorithm for epilepsy seizure onset detection and spread estimation from EEG signals. J. Phys. Conf. Ser. 2016, 705, 12032. [Google Scholar] [CrossRef]
- Gajic, D.; Djurovic, Z.; Gennaro, S.D.; Gustafsson, F. Classification of EEG signals for detection of epileptic seizures based on wavelets and statistical pattern recognition. Biomed. Eng. Appl. Basis Commun. 2014, 26, 1450021. [Google Scholar] [CrossRef]
- Navakatikyan, M.; Colditz, P.; Burke, C.; Inder, T.; Richmond, J.; Williams, C. Seizure detection algorithm for neonates based on wave-sequence analysis. Clin. Neurophysiol. 2006, 117, 1190–1203. [Google Scholar] [CrossRef]
- Siuly, S.; Kabir, E.; Wang, H.; Zhang, Y. Exploring sampling in the detection of multicategory EEG signals. Comput. Math. Methods Med. 2015, 2015, 576437. [Google Scholar] [CrossRef]
- Subasi, A.; Ercelebi, E. Classification of EEG signals using neural network and logistic regression. Comput. Methods Programs Biomed. 2005, 78, 87–99. [Google Scholar] [CrossRef]
- Quintero-Rincón, A.; Alanis, M.; Muro, V.; D’Giano, C. Spike-and-Wave detection in epileptic signals using cross-correlation and decision trees. Rev. Argent. BioingenierÍa Bioeng. Argent. Soc. 2018, 21, 1–4. [Google Scholar]
- Donos, C.; Dumpelmann, M.; Schulze-Bonhage, A. Early Seizure Detection Algorithm Based on Intracranial EEG and Random Forest Classification. Int. J. Neural Syst. 2015, 5, 1550023. [Google Scholar] [CrossRef]
- Fu, R.; Tian, Y.; Shi, P.; Bao, T. Automatic Detection of Epileptic Seizures in EEG Using Sparse CSP and Fisher Linear Discrimination Analysis Algorithm. J. Med. Syst. 2020, 4, 1–13. [Google Scholar] [CrossRef]
- Ossadtchi, A.; Greenblatt, R.E.; Towle, V.L.; Kohrman, M.H.; Kamada, K. Inferring spatiotemporal network patterns from intracranial EEG data. Clin. Neurophysiol. 2010, 121, 823–835. [Google Scholar] [CrossRef]
- Wilson, S.B.; Emerson, R. Spike detection: A review and comparison of algorithms. Clin. Neurophysiol. 2002, 113, 1873–1881. [Google Scholar] [CrossRef]
- Hese, P.V.; Martens, J.; Waterschoot, L.; Boon, P.; Lemahieu, I. Automatic detection of spike and wave discharges in the EEG of genetic absence epilepsy rats from Strasbourg. IEEE Trans. Biomed. Eng. 2009, 56, 706–717. [Google Scholar] [CrossRef] [PubMed]
- Pearce, P.; Friedman, D.; Lafrancois, J.; Iyengar, S.; Fenton, A.; Maclusky, N.; Scharfman, H. Spike wave discharges in adult Sprague Dawley rats and their implications for animal models of temporal lobe epilepsy. Epilepsy Behav. 2014, 32, 121–131. [Google Scholar] [CrossRef] [PubMed]
- M Le Van Quyen, J.F.; Lachaux, J.; Rodriguez, E.; Lutz, A.; Martinerie, J.; Varela, F.J. Comparison of Hilbert transform and wavelet methods for the analysis of neuronal synchrony. J. Neurosci. Methods 2001, 111, 83–98. [Google Scholar] [CrossRef]
- Puspita, J.W.; Gunadharma, A.I.J.S. Classification of epileptiform and wicket spike of EEG pattern using backpropagation neural network. AIP Conf. Proc. 2017, 1825, 020018. [Google Scholar]
- Gupta, A.; Singh, P.; Karlekar, M. A Novel Signal Modeling Approach for Classification of Seizure and Seizure-Free EEG Signals. IEEE Trans. Neural Syst. Rehabil. Eng. 2018, 5, 925–935. [Google Scholar] [CrossRef]
- Quintero-Rincón, A.; D’Giano, C.; Batatia, H. A quadratic linear-parabolic model-based EEG classification to detect epileptic seizures. J. Biomed. Res. 2020, 3, 203–210. [Google Scholar] [CrossRef]
- Quintero-Rincón, A.; Prendes, J.; Muro, V.; D’Giano, C. Study on Spike-and-wave detection in epileptic signals using t-location-scale distribution and the k-nearest neighbors classifier. IEEE URUCON Congr. Electron. Electr. Eng. Comput. 2017, 2017, 1–4. [Google Scholar]
- Li, P.; Karmakar, C.; Yearwood, J.; Venkatesh, S.; Palaniswami, M.; Liu, C. Detection of epileptic seizure based on entropy analysis of short-term EEG. PLoS ONE 2018, 3, e0193691. [Google Scholar] [CrossRef]
- Jirka, J.; Prauzek, M.; Krejcar, O.; Kuca, K. Automatic epilepsy detection using fractal dimensions segmentation and GP-SVM classification. Neuropsychiatr. Dis. Treat. 2018, 14, 2439–2449. [Google Scholar] [CrossRef]
- Paul, Y. Various epileptic seizure detection techniques using biomedical signals: A review. Brain Inform. 2018, 5, 1–19. [Google Scholar] [CrossRef] [PubMed]
- Subasi, A.; Alkana, A.; Koklukayab, E.; Kiymik, M.K. Analysis of Epileptic Seizure. Detection Methods Based on Parameter Estimation, Power Spectrum Density And Morlet Wavelet Transform. Neural Netw. 2005, 18, 985–997. [Google Scholar] [CrossRef]
- Xanthopoulos, P.; Liu, C.C.; Zhang, J.; Miller, E.R.; Nair, S.P.; Uthman, B.M.; Kelly, K.; Pardalos, P.M. A robust spike and wave algorithm for detecting seizures in a genetic absence seizure model. In Proceedings of the Annual International Conference of the IEEE Engineering in Medicine and Biology Society, Minneapolis, MN, USA, 3–6 September 2009; pp. 2184–2187. [Google Scholar]
- Sitnikova, E.; Hramov, A.E.; Koronovsky, A.A.; van Luijtelaar, G. Sleep spindles and spike-wave discharges in EEG: Their generic features, similarities and distinctions disclosed with Fourier transform and continuous wavelet analysis. J. Neurosci. Methods 2009, 180, 304–316. [Google Scholar] [CrossRef]
- Richard, C.D.; Tanenbaum, A.; Audit, B.; Arneodo, A.; Khalil, A.; Frankel, W. SWDreader A Wavelet-Based Algorithm Using Spectral Phase to Characterize Spike-Wave Morphological Variation in Genetic Models of Absence Epilepsy. J. Neurosci. Methods 2014, 242, 127–140. [Google Scholar] [CrossRef]
- Pollen, D.A. Intracellular studies of cortical neurons during thalamic induced wave and spike. Electroencephalogr. Clin. Neurophysiol. 1964, 17, 398–404. [Google Scholar] [CrossRef]
- Ovchinnikov, A.; Luttjohann, A.; Hramov, A.; van Luijtelaar, G. An algorithm for real-time detection of spike-wave discharges in rodents. J. Neurosci. Methods 2010, 94, 172–178. [Google Scholar] [CrossRef] [PubMed]
- Rodgers, K.; Dudek, F.; Barth, D. 2. Progressive, Seizure-Like, Spike-Wave Discharges Are Common in Both Injured and Uninjured Sprague-Dawley Rats: Implications for the Fluid Percussion Injury Model of Post-Traumatic Epilepsy. J. Neurosci. 2015, 35, 9194–9204. [Google Scholar] [CrossRef]
- Blumenfeld, H. Cellular and Network Mechanisms of Spike-Wave Seizures. Epilepsia 2005, 46, 21–33. [Google Scholar] [CrossRef] [PubMed]
- Avoli, M. A brief history on the oscillating roles of thalamus and cortex in absence seizures. Epilepsia 2012, 53, 779–789. [Google Scholar] [CrossRef]
- Puspita, J.W.; Gunadharma, S.; Indratno, S.W.; Soewono, E. Bayesian approach to identify spike and sharp waves in EEG data of epilepsy patients. Biomed. Signal Process. Control 2017, 35, 63–69. [Google Scholar] [CrossRef]
- Zhu, J.D.; Lin, C.F.; Chang, S.H.; Wang, J.H.; Peng, T.I.; Chien, Y.Y. Analysis of spike waves in epilepsy using Hilbert-Huang transform. J. Med. Syst. 2014, 39, 1–13. [Google Scholar] [CrossRef] [PubMed]
- Medvedeva, T.M.; Sysoeva, M.V.; Luijtelaa, G.; Sysoev, I.V. Modeling spike-wave discharges by a complex network of neuronal oscillators. Arch. Psychiatry Clin. Neurosci. 2018, 98, 271–282. [Google Scholar] [CrossRef]
- Olejarczyk, E.; Rudner, R.; Marciniak, R.; Wartak, M.; Stasiowski, M.; Jalowiecki, P.; Sobieszek, A. Detection of the EEG spike-wave patterns evoked by volatile anaesthetics. IFMBE Proc. 2009, 25, 407–409. [Google Scholar]
- Zibrandtsen, I.C.; Nielsen, J.M.; Kjaer, T.W. Quantitative characteristics of spike-wave paroxysms in genetic generalized epilepsy. Clin. Neurophysiol. 2020, 131, 1230–1240. [Google Scholar] [CrossRef]
- Haghighi, H.S.; Markazi, A.H. Dynamic origin of spike and wave discharges in the brain. NeuroImage 2019, 197, 69–79. [Google Scholar] [CrossRef]
- Quintero-Rincón, A.; Carenzo, C.; Ems, J.; Hirschson, L.; Muro, V.; D’Giano, C. Spike-and-wave epileptiform discharge pattern detection based on Kendall’s Tau-b Coefficient. Appl. Med. Inform. 2019, 1, 1–8. [Google Scholar]
- Tenneti, S.V.; Vaidyanathan, P.P. Absence Seizure Detection Using Ramanujan Filter Banks. In Proceedings of the 52nd Asilomar Conference on Signals, Systems, and Computers, Pacific Grove, CA, USA, 28–31 October 2018; pp. 1913–1917. [Google Scholar]
- Polivannyi, F.; Igasaki, T.; Neshiger, N.M.R. Wavelet Transform-Based Algorithm for Single Spike-and-Wave Discharges Detection in Epileptic Patients’ Electroencephalogram. In Proceedings of the 8th International Conference on BioMedical Engineering and Informatics, Shenyang, China, 14–16 October 2015; pp. 255–259. [Google Scholar]
- Quintero-Rincón, A.; Prendes, J.; Pereyra, M.; Batatia, H.; Risk, M. Multivariate Bayesian Classification of Epilepsy EEG Signals. In Proceedings of the IEEE 12th Image, Video, and Multidimensional Signal Processing Workshop (IVMSP), Bordeaux, France, 11–12 July 2016; pp. 1–5. [Google Scholar]
- Quintero-Rincón, A.; Pereyra, M.; D’Giano, C.; Batatia, H.; Risk, M. A visual EEG epilepsy detection method based on a wavelet statistical representation and the Kullback-Leibler divergence. In IFMBE Proceedings; Springer: Singapore, 2017; Volume 60, pp. 13–16. [Google Scholar]
- Quintero-Rincón, A.; Pereyra, M.; D’Giano, C.; Risk, M.; Batatia, H. Fast statistical model-based classification of epileptic EEG signals. Biocybern. Biomed. Eng. 2018, 38, 877–889. [Google Scholar] [CrossRef]
- Ahuja, N.; Lertrattanapanich, S.; Bose, N. Properties determining choice of mother wavelet. IEE Proc. Vis. Image Signal Process. 2005, 152, 659–664. [Google Scholar] [CrossRef]
- Abry, P. Ondelettes et Turbulence. Multirésolutions, Algorithmes de Décomposition, Invariance D’échelles; Diderot Editeur: Paris, France, 1997. [Google Scholar]
- Do, M.N.; Vetterli, M. Wavelet-Based Texture Retrieval Using Generalized Gaussian Density and Kullback-Leibler Distance. IEEE Trans. Image Process. 2002, 11, 146–158. [Google Scholar] [CrossRef]
- Quintero-Rincón, A.; D’Giano, C.; Risk, M. Epileptic seizure prediction using Pearson’s product-moment correlation coefficient of a linear classifier from generalized Gaussian modeling. Neurol. Argent. 2018, 10, 201–217. [Google Scholar]
- Bishop, C.M. Pattern Recognition and Machine Learning (Information Science and Statistics); Springer: New York, NY, USA, 2006. [Google Scholar]
- Barber, D. Bayesian Reasoning and Machine Learning; Cambridge University Press: Cambridge, UK, 2012. [Google Scholar]
- van der Maaten, L.; Hinton, H. Visualizing Data using t-SNE. J. Mach. Learn. Res. 2008, 9, 2579–2605. [Google Scholar]
- Zemouri, R.; Lévesque, M.; Amyot, N.; Hudon, C.; Kokoko, O. Deep Variational Autoencoder: An efficient tool for PHM frameworks. In Proceedings of the Prognostics and Health Management Conference (PHM-Besancon), Besancon, France, 4–7 May 2020; pp. 235–240. [Google Scholar]


| Method | Features | Classifier | Accuracy in % | Ref. |
|---|---|---|---|---|
| Generalized Gaussian distribution (GGD) | GGD parameters, variance and median from time–frequency Morlet decomposition | 10-NN | 92 | our |
| Kendall’s Tau-b Coefficient | Kendall’s Tau-b coefficient significance | SpPIn | 94 | [39] |
| Ramanujan Filter Bank (RFB) | Spectrum from RFB | Empirical | >80 | [40] |
| t-location-scale distribution (TLS) | TLS parameters | 1-NN | 100 | [20] |
| Cross-correlation | Correlation coefficient | Decision trees | 97 | [9] |
| Walsh transformation (WT) | First and second orden from WT | Bayesian | >70 | [33] |
| Hilbert–Huang transform | Intrinsic mode functions energy | WA | - | [34] |
| Cross-correlation | Wavelet spectrum correlation | AET | 100 | [41] |
| Mean | std | Variance | Bounds | |
|---|---|---|---|---|
| Class 0 | 293 | 267.8017 | 71,718 | [12, 1275] |
| Class 1 | 542 | 406.2597 | 165,047 | [31, 1811] |
| Mean | std | Variance | Bounds | |
|---|---|---|---|---|
| Class 0 | 1.446 × 10 | 4.235 × 10 | 1.794 × 10 | [9.46 × 10, 3.162 × 10] |
| Class 1 | 4.32 × 10 | 7.892 × 10 | 6.228 × 10 | [2.715 × 10, 4.321 × 10] |
| Mean | std | Variance | Bounds | |
|---|---|---|---|---|
| Class 0 | 1.089 × 10 | 1.002 × 10 | 1.004 × 10 | [−2.769 × 10, 2.179 × 10] |
| Class 1 | −6.125 × 10 | 2.672 × 10 | 7.140 × 10 | [−7.325 × 10, 7.406 × 10] |
| Method | Features | Classifier | TPR | TNR | ACC | Training | Testing | Ref. |
|---|---|---|---|---|---|---|---|---|
| GGD | GGD parameters, variance and median from time-frequency Morlet wavelet decomposition | 10-NN | 95 | 87 | 92 | 212 | 96 | Actual |
| Kendall’s Tau-b Coefficient | Kendall’s Tau-b coefficient significance in time domain | SpPIn | - | 94 | 94 | 300 | 300 | [39] |
| TLS | TLS parameters in time domain | 1-NN | 100 | 100 | 100 | 192 | 46 | [20] |
| Cross-correlation | Correlation coefficient in time domain | Decision trees | 86 | 98 | 97 | 96 | 46 | [9] |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Quintero-Rincón, A.; Muro, V.; D’Giano, C.; Prendes, J.; Batatia, H. Statistical Model-Based Classification to Detect Patient-Specific Spike-and-Wave in EEG Signals. Computers 2020, 9, 85. https://doi.org/10.3390/computers9040085
Quintero-Rincón A, Muro V, D’Giano C, Prendes J, Batatia H. Statistical Model-Based Classification to Detect Patient-Specific Spike-and-Wave in EEG Signals. Computers. 2020; 9(4):85. https://doi.org/10.3390/computers9040085
Chicago/Turabian StyleQuintero-Rincón, Antonio, Valeria Muro, Carlos D’Giano, Jorge Prendes, and Hadj Batatia. 2020. "Statistical Model-Based Classification to Detect Patient-Specific Spike-and-Wave in EEG Signals" Computers 9, no. 4: 85. https://doi.org/10.3390/computers9040085
APA StyleQuintero-Rincón, A., Muro, V., D’Giano, C., Prendes, J., & Batatia, H. (2020). Statistical Model-Based Classification to Detect Patient-Specific Spike-and-Wave in EEG Signals. Computers, 9(4), 85. https://doi.org/10.3390/computers9040085





