Models of Hepatocellular Carcinoma and Biomarker Strategy
Abstract
:1. Introduction
2. Discussion
2.1. In Vitro Studies
HCC Cell Lines and in Vitro Efficacy
2.2. In Vivo Studies
2.2.1. Animal Models of HCC—General Remarks
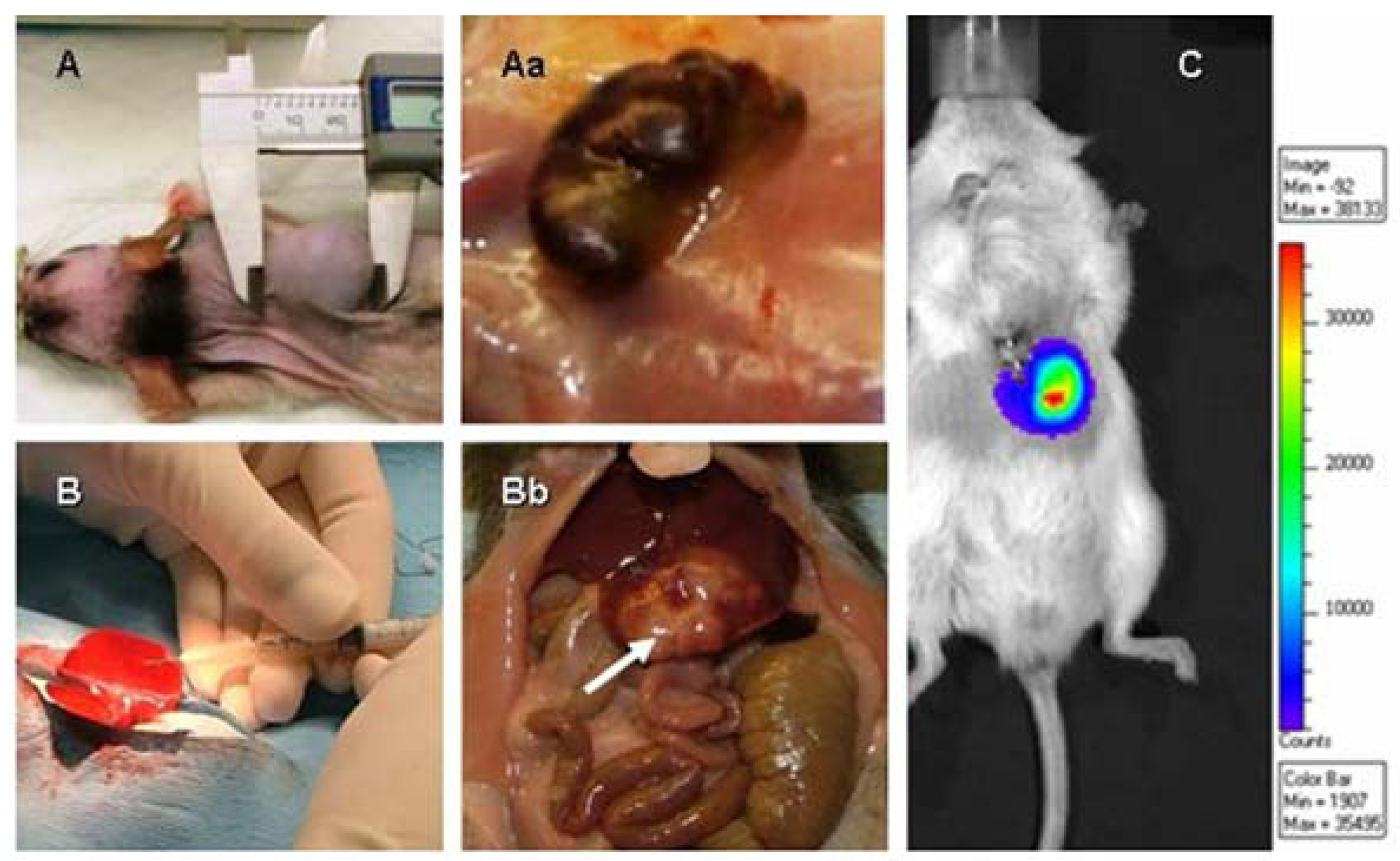
2.2.2. Use of Cell Cocktails to Better Predict Clinical Outcome
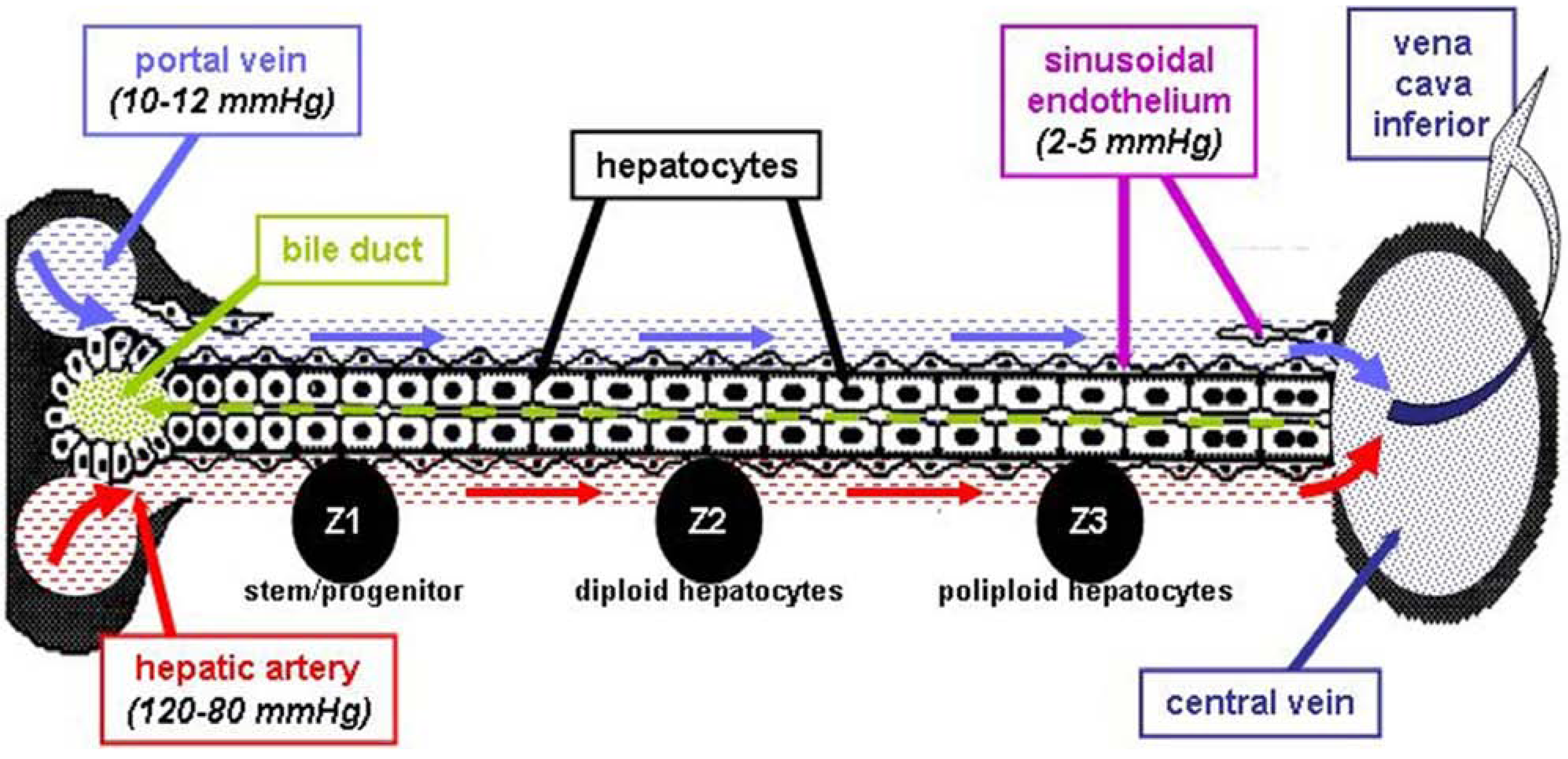
2.2.3. Liver Vasculature—Relevance for Models of HCC
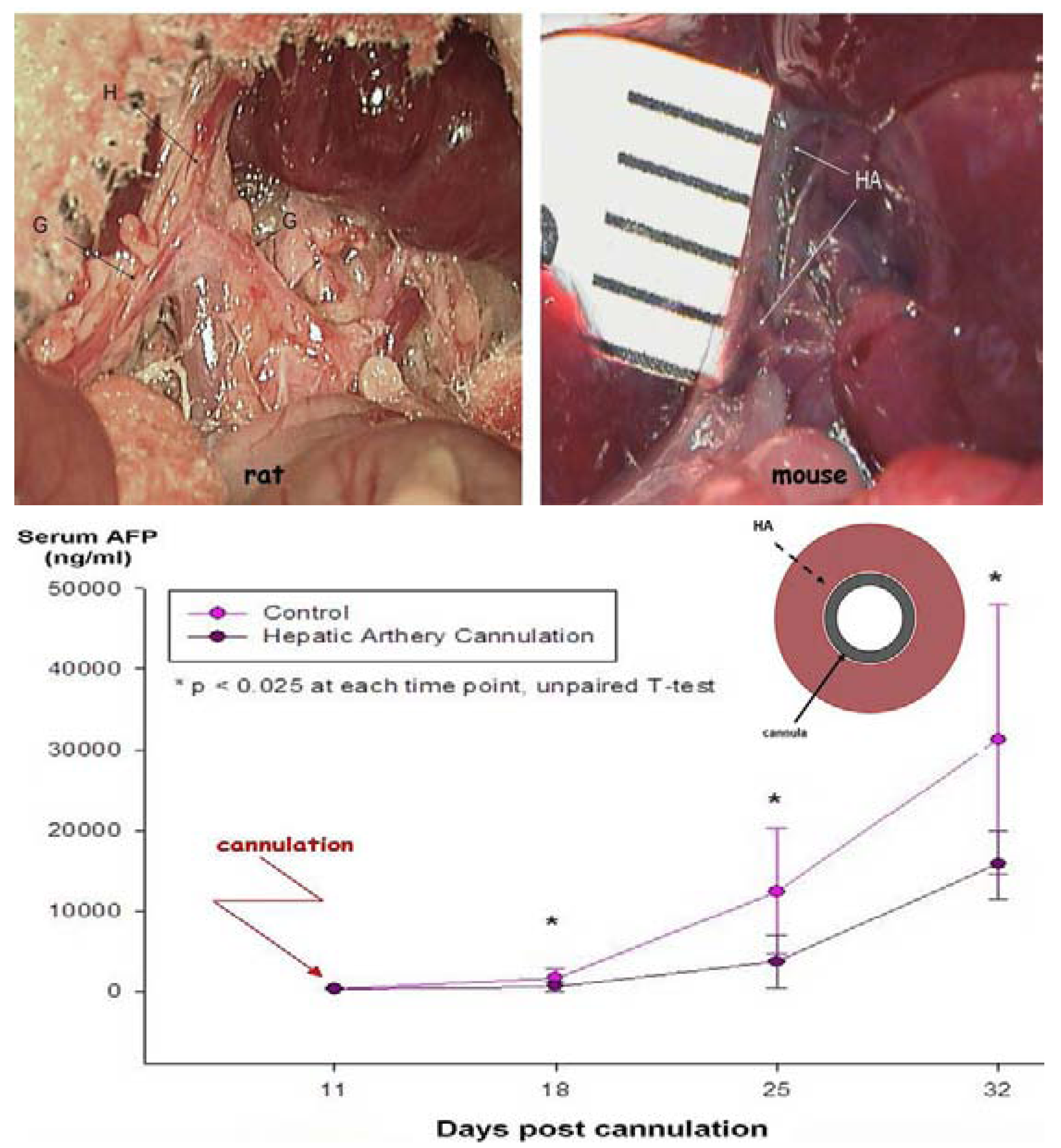
2.2.4. Drug Delivery—Clinical and Preclinical Relevance
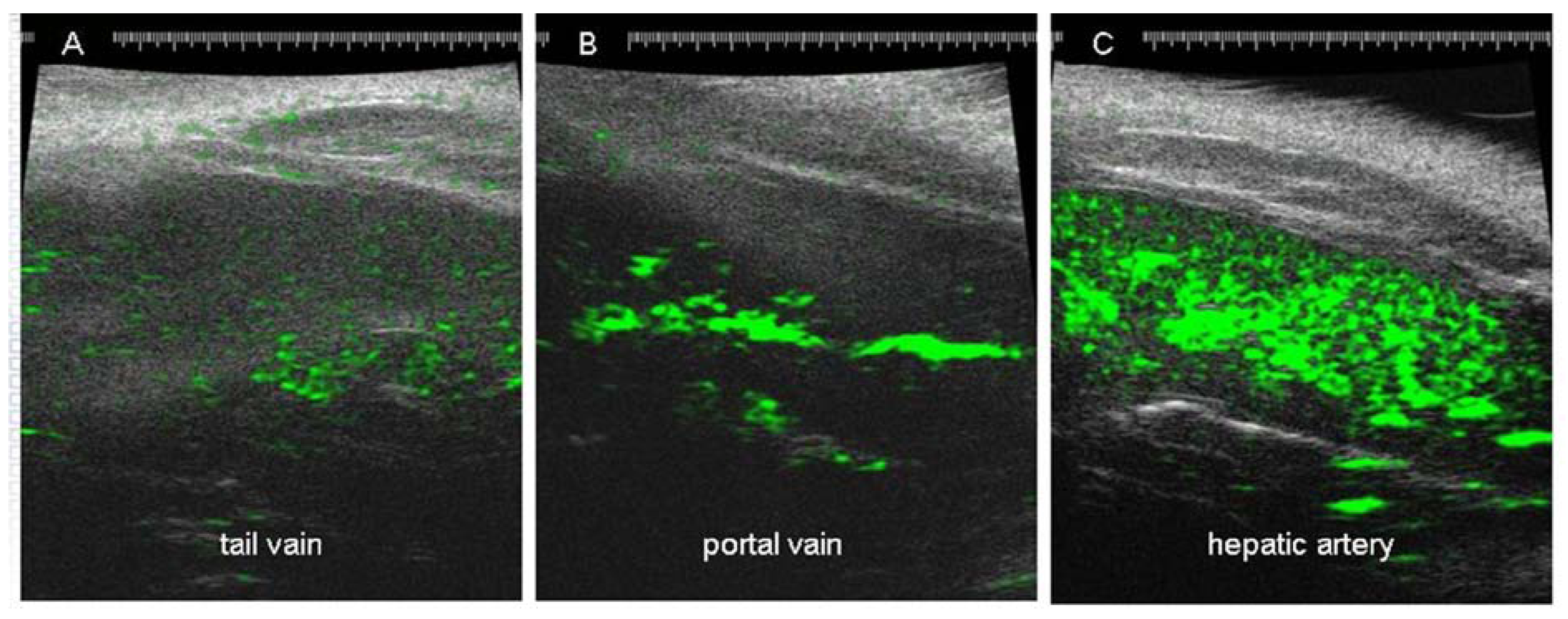
2.2.5. Use of Biomarkers in Preclinical Models of HCC
3. Conclusions
Declare
References
- Parkin, D.M.; Bray, F.; Ferlay, J.; Pisani, P. Estimating the world cancer burden: Globcon 2000. Int. J. Cancer 2001, 94, 153–156. [Google Scholar] [CrossRef]
- El Serag, H.B.; Mason, A.C. Rising incidence of hepatocellular carcinoma in the United States. N. Eng. J. Med. 1999, 340, 745–750. [Google Scholar] [CrossRef]
- Dalton, W.S; Friend, S.H. Cancer biomarkers—An invitation to the table. Science 2006, 312, 1165–1168. [Google Scholar] [CrossRef]
- Nerenstone, S.R.; Ihde, D.C.; Friedeman, M.A. Clinical trials in primary hepatocellular carcinoma: Current status and future directions. Cancer Treat. Rev. 1988, 15, 1–31. [Google Scholar] [CrossRef]
- Lai, C.-L.; Wu, P.-C.; Chan, G.C.B.; Lok, A.S.F.; Lin, H.-J. Doxorubicin versus no antitumor therapy in inoperable hepatocellular carcinoma. Cancer 1988, 62, 479–483. [Google Scholar] [CrossRef]
- Strumberg, D.; Richly, H.; Hilger, R.A.; Schleucher, N.; Kofee, S.; Tewes, M.; Faghih, M.; Brendel, E.; Voliotis, D.; Haase, C.G.; et al. Clinical and pharmacokinetic study of the novel Raf kinase and vascular endothelial growth factor inhibitor BAY 43–9006 in patients with advanced refractory solid tumors. J. Clin. Oncol. 2005, 23, 965–972. [Google Scholar]
- Llovet, J.; Ricci, S.; Mazzaferro, V.; Hilgard, P.; Raoul, J.; Zeuzem, S.; Paulin-Costello, M.; Moscovici, M.; Voliotis, D.; Bruix, J. For the SHARP Investigators Study Group. Sorafenib improves survival in advanced hepatocellular carcinoma (HCC). Results of phase III randomized placebo-controlled trial (SHARP trial). J. Clin. Oncol. 2007, 25. Abstr. LBA1. [Google Scholar]
- Abou Alfa, G.K.; Johnson, P.; Knox, J.; Lacava, J.; Leung, T.; Mori, A.; Leberre, M.A.; Voliotis, D.; Saltz, L. Preliminary results from a phase II, rendomized, double-blind study of sorafenib plus doxorubicin versus placebo plus doxorubicin in patients with advanced hepatocellular carcinoma. Eur. J. Cancer 2007, 5, 259. [Google Scholar]
- Zhu, A.X.; Sahani, D.V.; de Tomaso, E.; Duda, D.; Sindhwani, V.; Yoon, S.S.; Blaszkowsky, L.S.; Clark, J.W.; Ryan, D.P.; Jain, R.K. A phase II study of sunitinib in patients with advanced hepatocellular carcinoma. J. Clin. Oncol. 2007, 25. Abstr. 4637. [Google Scholar]
- Faivre, S.J.; Raymond, E.; Douillard, J.; Boucher, E.; Lim, H.Y.; Kim, J.S.; Lanzalone, S.; Lechuga, M.J.; Sherman, L.; Cheng, A. Assessment of safety and drug-induced tumor necrosis with sunitanib in patients with unresectable hepatocellular carcinoma (HCC). J. Clin. Oncol. 2007, 25. Abstr. 3546. [Google Scholar]
- Yang, R.; Rescoria, F.J.; Reilly, C.R.; Faught, P.R.; Sanghvi, N.T.; Lumeng, L.; Franklin, T.D; Grosfeld, J.L. A reproducible rat liver cancer model for experimental therapy: introducing a technique of intrahepatic tumor implantation. J. Surg. Res. 1992, 52, 193–198. [Google Scholar] [CrossRef]
- Bagi, C.M. Challenges in Developing Preclinical Models of Hepatocellular Carcinoma. In 7th Annual Cancer Drugs. Research and Development Conference, San Diego, CA, USA, 2010; pp. 163–170.
- Sun, F.X.; Tang, Z.Y.; Liu, K.D.; Ye, S.L.; Xue, Q.; Gao, D.M.; Ma, Z.C. Establishment of a metastatic model of human hepatocellular carcinoma in nude mice via orthotopic implantation of histologically intact tissues. Int. J. Cancer 1996, 66, 239–243. [Google Scholar] [CrossRef]
- Hoffman, R.M. Orthotopic metastatic mouse models for anticancer drug discovery and evaluation: a bridge to the clinic. Invest. New Drugs 1999, 17, 343–359. [Google Scholar] [CrossRef]
- Gutmann, D.H.; Hunter-Schaedle, K.; Shannon, K.M. Harnessing preclinical mouse models to inform human cancer trials. J. Clin. Invest. 2006, 116, 847–852. [Google Scholar] [CrossRef]
- Kramer, M.G.; Hernandez-Alcoceba, R.; Qian, C.; Prieto, J. Evaluation of hepatocellular carcinoma models for preclinical research. Drug Discovery Today: Disease models. Drug Disc. Today 2005, 2, 41–49. [Google Scholar]
- Daniel, V.C.; Marchionni, L.; Hierman, J.S.; Rhodes, J.T.; Devereux, W.L.; Rudin, C.M.; Young, R.; Parmigiani, G.; Dorsch, M.; Peacock, C.D.; Watkins, D.N. A primary xenograft model of small-cell lung cancer reveals irreversible changes in gene expression imposed by culture in vitro. Cancer Res. 2009, 69, 3364–3373. [Google Scholar] [CrossRef]
- Marx, J. Medicine. Building better mouse models for studying cancer. Science 2003, 299, 1972–1975. [Google Scholar] [CrossRef]
- Simonetti, R.G.; Cottone, M.; Craxi, A.; Pagliaro, L.; Rapicetta, M.; Chionne, P.; Costantino, A. Prevalence of antibodies to hepatitis C virus in hepatocellular carcinoma. Lancet 1989, 2, 1338. [Google Scholar]
- Patt, Y.Z.; Charnsangavej, B.; Yoffe, R.; Smith, R.; Lawrence, D.; Chuang, V.; Carrasco, H.; Roh, M.; Chase, J.; Fisher, H. Hepatic arterial infusion of floxouridine, leucovorin, doxorubicin, and cisplatin for hepatocellular carcinoma: Effects of hepatitis B and C viral infection on drug toxicity and patient survival. J. Clin. Oncol. 1994, 12, 1204–1211. [Google Scholar]
- Buendia, M.A.; Hepatitis, B. Viruses and hepatocellular carcinoma. Adv. Cancer Res. 1992, 59, 167–226. [Google Scholar] [CrossRef]
- Okubo, H.; Takei, Y.; Serizawa, N.; Enomoto, N.; Ikejima, K.; Sato, N. Orthotopic hepatocellular carcinoma model with a controlled and reproducible tumorogenicity. Hepatology 2007, 22, 423–428. [Google Scholar]
- Killion, J.J.; Radinsky, R.; Fidler, I.J. Orthotopic models are necessary to predict therapy of transplantable tumors in mice. Cancer Metastasis Rev. 1998, 17, 279–284. [Google Scholar] [CrossRef]
- Frijhoff, A.F.; Conti, C.J.; Sanderowicz, A.M. Advances in molecular carcinogenesis: Current and future use of mouse models to screen and validate molecularly targeted anticancer drugs. Mol. Carcinog. 2004, 39, 183–194. [Google Scholar] [CrossRef]
- Barajas, M.; Mazzolini, G.; Genove, G.; Bilbao, R.; Narvaiza, I.; Schmitz, V.; Sangro, B.; Melero, I.; Qian, C.; Prieto, J. Gene therapy of orthotopic hepatocellular carcinoma in rats using adenovirus coding for interleukin 12. Hepatology 2001, 33, 52–61. [Google Scholar] [CrossRef]
- Wilmanns, C.; Fan, D.; O’Brian, C.A.; Bucana, C.D.; Fidler, I.J. Orthotopic and ectopic organ environments differentially influence the sensitivity of murine colon carcinoma cells to doxorubicin and 5-fluorouracil. Int. J. Cancer 1992, 52, 98–104. [Google Scholar] [CrossRef]
- Labonte, P.; Kadhim, S.; Bowlin, T.; Mounir, S. Inhibitionof tumor growth with doxorubicin in a new orthotopically implanted human hepatocellular carcinoma model. Hepatol. Res. 2000, 18, 72–85. [Google Scholar] [CrossRef]
- Davis, H.C.; Morse, I.S. Segmental liver revascularization. Arch. Surg. 1957, 74, 525–527. [Google Scholar] [CrossRef]
- McCuskey, R.S. The hepatic microvascular system. In The Liver: Biology Pathology; Arias, I.M., Boyer, J.L., Fausto, N., Jacoby, B.W., Schachter, D.A., Shafritz, D.A., Eds.; Raven Press: New York, NY, USA, 1994; pp. 1089–1106. [Google Scholar]
- Rubaltelly, L.; Maschio, A.; Candiani, F.; Miotto, D. The role of vascularization in the formation of echographic patterns of hepatic metastases: microangiographic and echographic study. Br. J. Radiol. 1980, 53, 1166–1168. [Google Scholar] [CrossRef]
- Archer, S.G.; Gray, B.N. Vascularization of small liver metastases. Br. J. Surg. 1989, 76, 545–548. [Google Scholar] [CrossRef]
- Nakashima, Y.; Nakashima, O.; Hsia, C.C.; Kojiro, M.; Tabor, E. Vascularization of small hepatocellular carcinomas: correlation with differentiation. Liver 1999, 19, 12–18. [Google Scholar] [CrossRef]
- Matsui, O.; Kadoya, M.; Kameyama, T.; Yoshikawa, J.; Takashima, T.; Nakanuma, Y.; Unoura, M.; Kobayashi, K.; Izumi, R.; Ida, M. Benign and malignant nodulus in cirrhotic liver: distinction based on blood supply. Radiology 1991, 178, 493–497. [Google Scholar]
- Ackerman, N.B. The blood supply of experimental liver metastasis: IV. Changes in vascularity with increasing tumor growth. Surgery 1974, 75, 589–596. [Google Scholar]
- Ishikawa, T.; Imai, M.; Kamimura, H.; Tsuchiya, A.; Togashi, T.; Watanabe, K.; Seki, K.-I.; Ohta, H.; Yoshida, T.; Kemimura, T. Improved survival for hepatocellular carcinoma with portal vein tumor thrombosis treated by intra-arterial chemotherapy combining etoposide, carboplatin, epirubicin and pharmacokinetic modulating chemotherapy by 5-FU and enteric-coated tagafur/uracil: A pilot study. World J. Gastroenterol. 2007, 13, 5465–5470. [Google Scholar]
- Jarnagin, W.R.; Schwartz, L.H.; Gultekin, D.H.; Gönen, M.; Haviland, D.; Shia, J.; D’Angelica, M.; Fong, Y.; DeMatteo, R.; Tse, A.; Blumgart, L.H.; Kemeny, N. Regional chemotherapy for unresectable primary liver cancer: results of phase II clinical trial and assessment of DCE-MRI as biomarker of survival. Annal. Oncol 2009, 20, 1589–1595. [Google Scholar] [CrossRef]
- Fracanzani, A.L.; Burdick, L.; Borzio, M.; Roncalli, M.; Bonelli, N.; Borzio, F.; Maraschi, A.; Fiorelli, G.; Fargion, S. Contrast enhanced doppler ultrasonography in the diagnosis of hepatocellular carcinoma and premalignant lesions in patients with cirrhosis. Hepatology 2001, 34, 1109–1112. [Google Scholar] [CrossRef]
- Delbeke, D.; Martin, W.H.; Sandler, M.P.; Chapman, W.C.; Wright, J.K.; Pinson, C.W. Evaluation of benign vs. Malignant hepatic lesions with positron emission tomography. Arch. Surg. 1998, 133, 510–515. [Google Scholar] [CrossRef]
- Ho, C-L.; Yu, S.C.H.; Yeung, A.W.C. 11C-Acetate PET imaging in hepatocellular carcinoma and other liver masses. J. Nucl. Med. 2003, 44, 213–221. [Google Scholar]
- Olivia, M.R.; Saini, S. Liver cancer imaging: role of CT, MRI, US and PET. Cancer Imaging 2004, 4, S42–S46. [Google Scholar] [CrossRef]
- Saini, S.; Nelson, R.C. Technique for MR imaging of the liver. Radiology 1995, 197, 575–577. [Google Scholar]
- Weinmann, H.J.; Ebert, W.; Misselwitz, B.; Schmitt-Willich, H. Tissue-specific MR contrast agents. Eur. J. Radiol. 2003, 46, 33–44. [Google Scholar] [CrossRef]
- Soling, A.; Rainov, N.G. Bioluminescence imaging in vivo – application to cancer research. Expert. Opin. Biol. Ther. 2003, 3, 1163–1172. [Google Scholar]
- Sarraf-Yazdi, S.; Mi, J.; Dewhirst, M.W.; Clary, B.M. Use of in vivo bioluminescence imaging to predict hepatic tumor burden in mice. J. Surg. Res. 2004, 120, 249–255. [Google Scholar] [CrossRef]
- Ives, A.A.; Pathan, A.A.; Navarrete, C.V.; Williams, R.; Bertoletti, A.; Behboudi, S. Analysis of CD4+ T-cell responses to a novel alpha-fetoprotein-derived epitope in hepatocellular carcinoma patients. Clin. Cancer Res. 2005, 11, 6686–6694. [Google Scholar] [CrossRef]
- McIntire, K.R.; Vogel, C.L.; Primack, A.; Waldmann, T.A.; Kyalwazi, S.K. Effect of surgical and chemotherapeutic treatment on alpha-fetoprotein levels in patients with hepatocellular carcinoma. Cancer 2006, 37, 677–683. [Google Scholar]
- Bagi, C.M.; Christensen, J.; Cohen, D.P.; Roberts, G.W.; Wilkie, D.; Swanson, T.; Tuthill, T.; Andresen, J.C. Sunitinib and PF-562,271 (FAK/Pyk2 inhibitor) effectively block growth and recovery of human hepatocellular carcinoma in a rat xenograft model. Cancer Biol. Ther. 2009, 8, 1–10. [Google Scholar] [CrossRef]
- Boukaz, A.; de Jong, N. WFUMB safety symposium on echo-contrast agents: nature and types of ultrasound contrast agents. Ultrasound Med. Biol. 2007, 33, 187–196. [Google Scholar] [CrossRef]
- Bekeredjian, R.; Kroll, R.D.; Fein, E.; Tinkov, S.; Coester, C.; Winter, G.; Katus, H.A.; Kulaksiz, H. Ultrasound targeted microbubble destruction increases capillary permeability in hepatomas. Ultrasound Med. Biol. 2007, 33, 1592–1598. [Google Scholar] [CrossRef]
- Lassau, N.; Chebil, M.; Chami, L.; Roche, A. A new functional imaging technique for the early functional evaluation of antiangiogenic treatment: dynamic contrast-enhanced ultrasonography (DCE-US). Targ. Oncol. 2008, 3, 111–117. [Google Scholar] [CrossRef]
- Kang, J.; Wu, X.; Wang, Z.; Ran, H.; Xu, C.; Wu, J.; Wang, Z.; Zhang, Y. Antitumor effect of docetaxel-loaded lipid microbubbles combined with ultrasound-targeted microbubble activation on VX2 rabbit liver tumors. J. Ultrasound Med. 2010, 29, 61–70. [Google Scholar]
© 2010 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Bagi, C.M.; Andresen, C.J. Models of Hepatocellular Carcinoma and Biomarker Strategy. Cancers 2010, 2, 1441-1452. https://doi.org/10.3390/cancers2031441
Bagi CM, Andresen CJ. Models of Hepatocellular Carcinoma and Biomarker Strategy. Cancers. 2010; 2(3):1441-1452. https://doi.org/10.3390/cancers2031441
Chicago/Turabian StyleBagi, Cedo M., and Catharine J. Andresen. 2010. "Models of Hepatocellular Carcinoma and Biomarker Strategy" Cancers 2, no. 3: 1441-1452. https://doi.org/10.3390/cancers2031441
APA StyleBagi, C. M., & Andresen, C. J. (2010). Models of Hepatocellular Carcinoma and Biomarker Strategy. Cancers, 2(3), 1441-1452. https://doi.org/10.3390/cancers2031441



