Exosome-Enhanced Sonodynamic Therapy in Cancer: Emerging Synergies and Modulation of the Tumor Microenvironment
Simple Summary
Abstract
1. Introduction
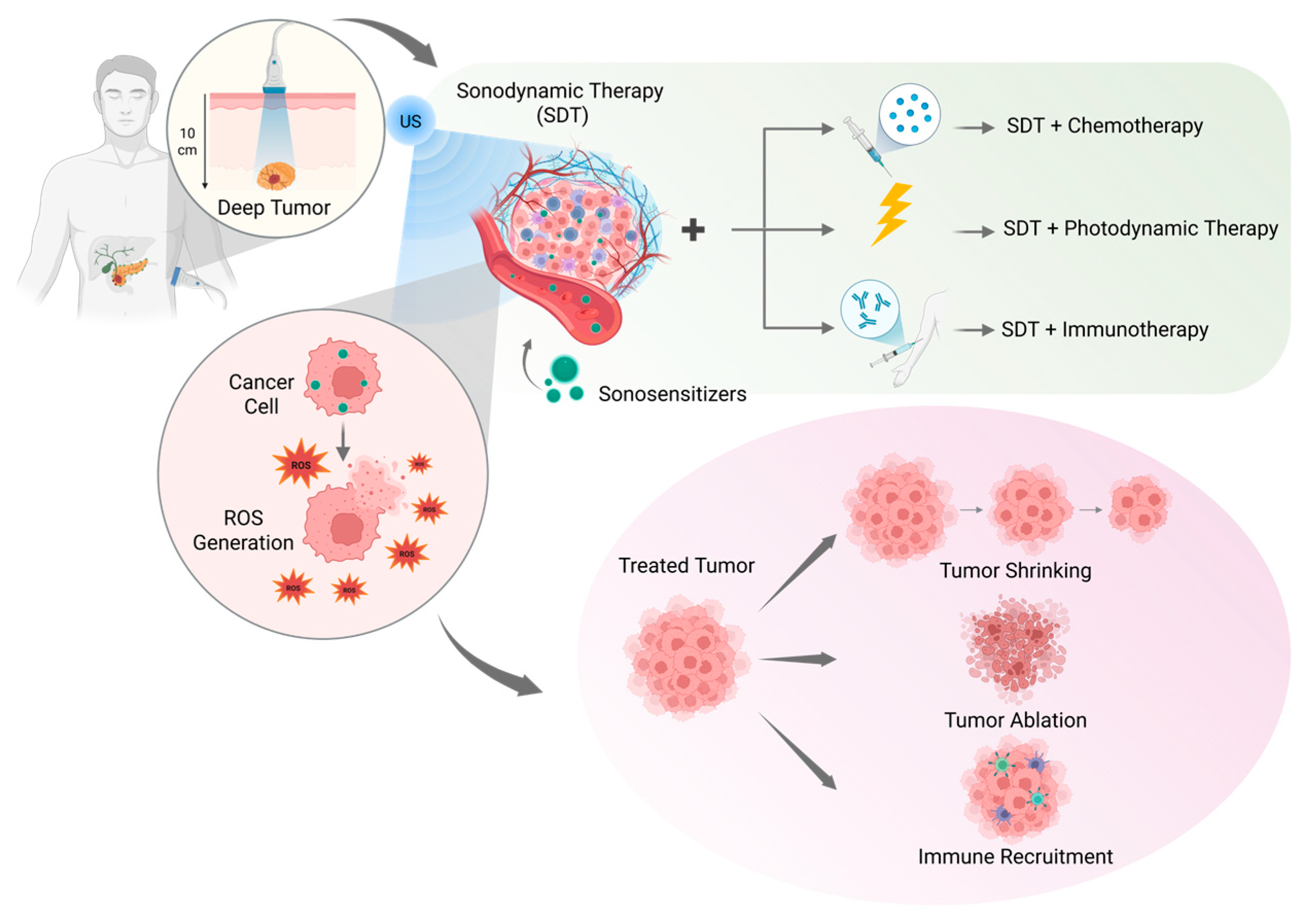
2. Mechanisms of SDT
3. SDT and Cancer Applications
4. EVs in Nanomedicine and Cancer
4.1. Overview of Exosomes and EVs
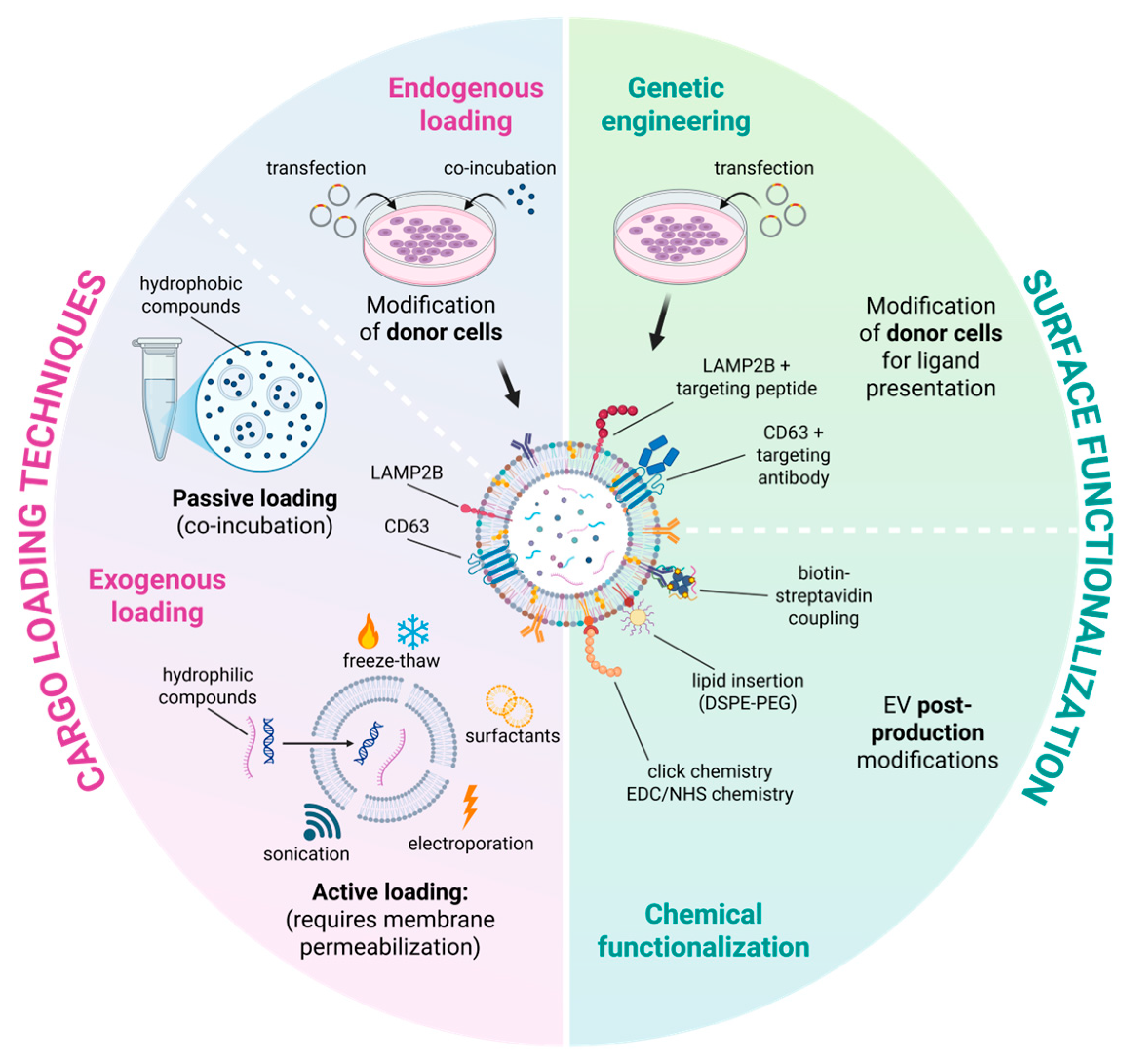
4.2. Exosome Engineering Strategies for Therapeutic Applications
4.2.1. Cargo Loading Techniques
4.2.2. Surface Functionalization
4.2.3. EV Biomimetics and Artificial EVs
4.3. Exosomes in the TME
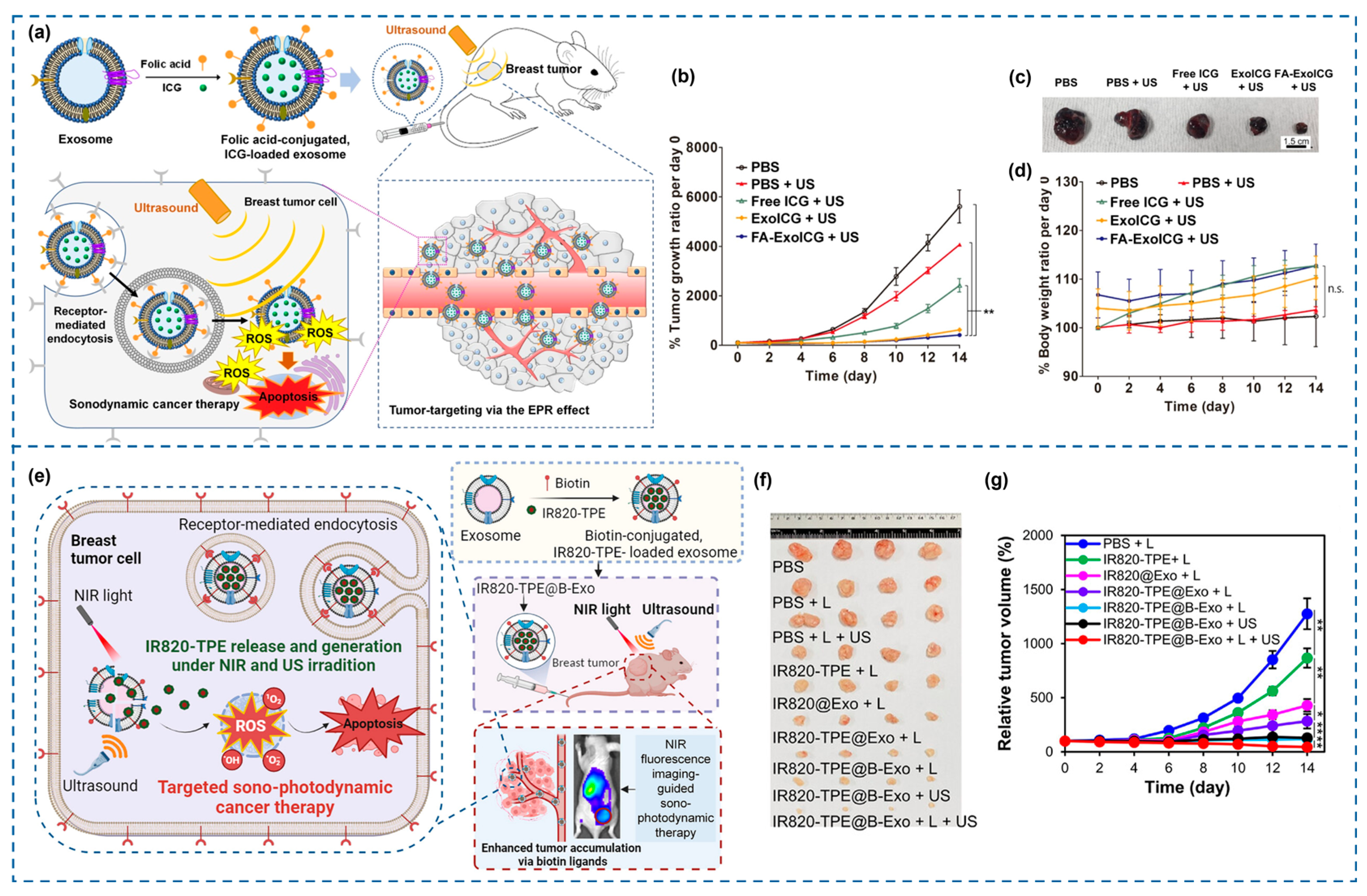
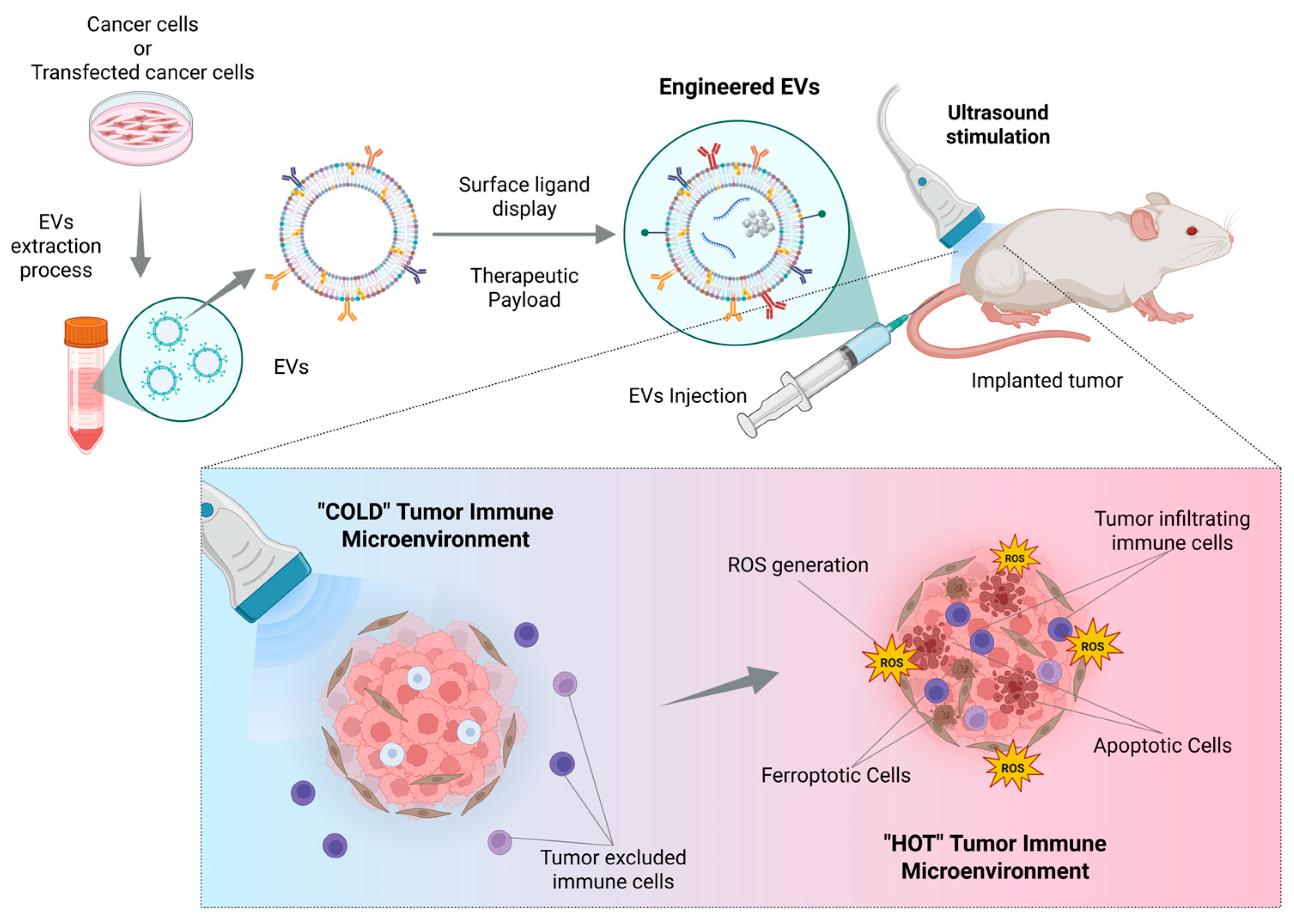
5. Integration of EVs in SDT
6. Modulating the TME via EVs-SDT
7. Challenges and Clinical Translation
8. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
Abbreviations
| SDT | Sonodynamic Therapy |
| EVs | Extracellular Vesicles |
| ROS | Reactive Oxygen Species |
| US | Ultrasound |
| NP | Nanoparticle |
| TME | Tumor Microenvironment |
| EV-SDT | Extracellular Vesicle-Enhanced Sonodynamic Therapy |
| PDT | Photodynamic Therapy |
| PTT | Photothermal Therapy |
| DAMPs | Damage-Associated Molecular Patterns |
| ICD | Immunogenic Cell Death |
| miRNA | microRNA |
| VEGF | Vascular Endothelial Growth Factor |
| FGF | Fibroblast Growth Factor |
| PTX | Paclitaxel |
| GBM | Glioblastoma |
| EMT | Epithelial-Mesenchymal Transition |
| ICG | Indocyanine Green |
| CAT | Catalase |
| Ce6 | Chlorin e6 |
| GMP | Good Manufacturing Practice |
| 5-ALA | 5-Aminolevulinic acid |
References
- Bray, F.; Laversanne, M.; Sung, H.; Ferlay, J.; Siegel, R.L.; Soerjomataram, I.; Jemal, A. Global Cancer Statistics 2022: GLOBOCAN Estimates of Incidence and Mortality Worldwide for 36 Cancers in 185 Countries. CA Cancer J. Clin. 2024, 74, 229–263. [Google Scholar] [CrossRef]
- Deivayanai, V.C.; Thamarai, P.; Karishma, S.; Saravanan, A.; Yaashikaa, P.R.; Vickram, A.S.; Hemavathy, R.V.; Kumar, R.R.; Rishikesavan, S.; Shruthi, S. A Comprehensive Review on Advances in Nanoparticle-Mediated Cancer Therapeutics: Current Research and Future Perspectives. Cancer Pathog. Ther. 2024, 3, 293–308. [Google Scholar] [CrossRef]
- Xu, M.; Han, X.; Xiong, H.; Gao, Y.; Xu, B.; Zhu, G.; Li, J. Cancer Nanomedicine: Emerging Strategies and Therapeutic Potentials. Molecules 2023, 28, 5145. [Google Scholar] [CrossRef]
- Yang, N.; Li, J.; Yu, S.; Xia, G.; Li, D.; Yuan, L.; Wang, Q.; Ding, L.; Fan, Z.; Li, J. Application of Nanomaterial-Based Sonodynamic Therapy in Tumor Therapy. Pharmaceutics 2024, 16, 603. [Google Scholar] [CrossRef]
- Zhou, H.; Chen, Y.; Li, P.; He, X.; Zhong, J.; Hu, Z.; Liu, L.; Chen, Y.; Cui, G.; Sun, D.; et al. Sonodynamic Therapy for Breast Cancer: A Literature Review. Open Chem. 2022, 20, 1045–1056. [Google Scholar] [CrossRef]
- Yang, M.; Wang, X.; Peng, M.; Wang, F.; Hou, S.; Xing, R.; Chen, A. Nanomaterials Enhanced Sonodynamic Therapy for Multiple Tumor Treatment. Nano-Micro Lett. 2025, 17, 157. [Google Scholar] [CrossRef] [PubMed]
- Yu, J.; Hu, J.-R.; Tian, Y.; Lei, Y.-M.; Hu, H.-M.; Lei, B.-S.; Zhang, G.; Sun, Y.; Ye, H.-R. Nanosensitizer-Assisted Sonodynamic Therapy for Breast Cancer. J. Nanobiotechnol. 2025, 23, 281. [Google Scholar] [CrossRef] [PubMed]
- Chen, H.; Zhou, X.; Gao, Y.; Zheng, B.; Tang, F.; Huang, J. Recent Progress in Development of New Sonosensitizers for Sonodynamic Cancer Therapy. Drug Discov. Today 2014, 19, 502–509. [Google Scholar] [CrossRef] [PubMed]
- Wang, L.; Yu, X.; Zhou, J.; Su, C. Extracellular Vesicles for Drug Delivery in Cancer Treatment. Biol. Proced. Online 2023, 25, 28. [Google Scholar] [CrossRef]
- Nguyen, V.N.; Dao, T.N.T.; Cho, M.; Jeong, H.; Nguyen-Le, M.T.; Shin, Y.; Yoon, J. Recent Advances in Extracellular Vesicle-Based Organic Nanotherapeutic Drugs for Precision Cancer Therapy. Coord. Chem. Rev. 2023, 479, 215006. [Google Scholar] [CrossRef]
- Tang, Y.; Liu, X.; Sun, M.; Xiong, S.; Xiao, N.; Li, J.; He, X.; Xie, J. Recent Progress in Extracellular Vesicle-Based Carriers for Targeted Drug Delivery in Cancer Therapy. Pharmaceutics 2023, 15, 1902. [Google Scholar] [CrossRef]
- Zhang, A.; Zheng, X.; Yan, G.; Liu, X.; Xie, D.; Xu, X.; Sun, M.; Liu, Z. Sonodynamic Biomimetic-Nanomedicine Fight Cancers. J. Nanobiotechnol. 2025, 23, 548. [Google Scholar] [CrossRef] [PubMed]
- Sattiraju, A.; Kang, S.; Giotti, B.; Chen, Z.; Marallano, V.J.; Brusco, C.; Ramakrishnan, A.; Shen, L.; Tsankov, A.M.; Hambardzumyan, D.; et al. Hypoxic Niches Attract and Sequester Tumor-Associated Macrophages and Cytotoxic T Cells and Reprogram Them for Immunosuppression. Immunity 2023, 56, 1825–1843.e6. [Google Scholar] [CrossRef]
- Zhu, Y.; Yu, X.; Thamphiwatana, S.D.; Zheng, Y.; Pang, Z. Nanomedicines Modulating Tumor Immunosuppressive Cells to Enhance Cancer Immunotherapy. Acta Pharm. Sin. B 2020, 10, 2054–2074. [Google Scholar] [CrossRef]
- Canavese, G.; Ancona, A.; Racca, L.; Canta, M.; Dumontel, B.; Barbaresco, F.; Limongi, T.; Cauda, V. Nanoparticle-Assisted Ultrasound: A Special Focus on Sonodynamic Therapy against Cancer. Chem. Eng. J. 2018, 340, 155–172. [Google Scholar] [CrossRef] [PubMed]
- Gong, F.; Cheng, L.; Yang, N.; Gong, Y.; Ni, Y.; Bai, S.; Wang, X.; Chen, M.; Chen, Q.; Liu, Z. Preparation of TiH1.924 Nanodots by Liquid-Phase Exfoliation for Enhanced Sonodynamic Cancer Therapy. Nat. Commun. 2020, 11, 3712. [Google Scholar] [CrossRef]
- Han, Y.; Yu, X.; Marfavi, Z.; Chen, Y.; Zhang, L.; Chu, J.; Sun, K.; Li, M.; Tao, K. A Perspective on Ultrasound-Triggered Production of Reactive Oxygen Species by Inorganic Nano/Microparticles. Adv. NanoBiomed Res. 2024, 4, 2400060. [Google Scholar] [CrossRef]
- Vighetto, V.; Pascucci, E.; Percivalle, N.M.; Troia, A.; Meiburger, K.M.; Broek, M.R.P.; van den; Segers, T.; Cauda, V. Functional Nanocrystal as Effective Contrast Agents for Dual-Mode Imaging: Live-Cell Sonoluminescence and Contrast-Enhanced Echography. Ultrason. Sonochem. 2025, 113, 107242. [Google Scholar] [CrossRef] [PubMed]
- Wang, W.; Tasset, A.; Pyatnitskiy, I.; Mohamed, H.G.; Taniguchi, R.; Zhou, R.; Rana, M.; Lin, P.; Capocyan, S.L.C.; Bellamkonda, A.; et al. Ultrasound Triggered Organic Mechanoluminescence Materials. Adv. Drug Deliv. Rev. 2022, 186, 114343. [Google Scholar] [CrossRef]
- Liu, R.G.; Zhang, Q.Y.; Lang, Y.H.; Peng, Z.Z.; Li, L.B. Sonodynamic Therapy, a Treatment Developing from Photodynamic Therapy. Photodiagno. Photodyn. Ther. 2017, 19, 159–166. [Google Scholar] [CrossRef]
- Choi, V.; Rajora, M.A.; Zheng, G. Activating Drugs with Sound: Mechanisms Behind Sonodynamic Therapy and the Role of Nanomedicine. Bioconjug. Chem. 2020, 31, 967–989. [Google Scholar] [CrossRef]
- Gong, Z.; Dai, Z. Design and Challenges of Sonodynamic Therapy System for Cancer Theranostics: From Equipment to Sensitizers. Adv. Sci. 2021, 8, 2002178. [Google Scholar] [CrossRef] [PubMed]
- McHale, A.P.; Callan, J.F.; Nomikou, N.; Fowley, C.; Callan, B. Sonodynamic Therapy: Concept, Mechanism and Application to Cancer Treatment. Adv. Exp. Med. Biol. 2016, 880, 429–450. [Google Scholar] [CrossRef]
- Wood, A.K.W.; Sehgal, C.M. A Review of Low-Intensity Ultrasound for Cancer Therapy. Ultrasound Med. Biol. 2015, 41, 905–928. [Google Scholar] [CrossRef]
- Nowak, K.M.; Schwartz, M.R.; Breza, V.R.; Price, R.J. Sonodynamic Therapy: Rapid Progress and New Opportunities for Non-Invasive Tumor Cell Killing with Sound. Cancer Lett. 2022, 532, 215592. [Google Scholar] [CrossRef] [PubMed]
- Yuan, H.; Ma, J.; Huang, W.; Gong, P.; Shi, F.; Xu, X.; Fu, C.; Wang, X.; Wong, Y.K.; Long, Y.; et al. Antitumor Effects of a Distinct Sonodynamic Nanosystem through Enhanced Induction of Immunogenic Cell Death and Ferroptosis with Modulation of Tumor Microenvironment. JACS Au 2023, 3, 1507–1520. [Google Scholar] [CrossRef]
- Wang, M.-F.; Guo, J.; Yuan, S.-J.; Li, K.; Zhang, Q.; Lei, H.-M.; Wu, J.-L.; Zhao, L.; Xu, Y.-H.; Chen, X. Targeted Sonodynamic Therapy Induces Tumor Cell Quasi-Immunogenic Ferroptosis and Macrophage Immunostimulatory Autophagy in Glioblastoma. Biomaterials 2025, 315, 122913. [Google Scholar] [CrossRef]
- Zhang, Y.; Zhao, Y.; Zhang, Y.; Liu, Q.; Zhang, M.; Tu, K. The Crosstalk between Sonodynamic Therapy and Autophagy in Cancer. Front. Pharmacol. 2022, 13, 961725. [Google Scholar] [CrossRef] [PubMed]
- Li, X.; Zhang, X.; Zheng, L.; Kou, J.; Zhong, Z.; Jiang, Y.; Wang, W.; Dong, Z.; Liu, Z.; Han, X.; et al. Hypericin-Mediated Sonodynamic Therapy Induces Autophagy and Decreases Lipids in THP-1 Macrophage by Promoting ROS-Dependent Nuclear Translocation of TFEB. Cell Death Dis. 2016, 7, e2527. [Google Scholar] [CrossRef]
- Wu, M.; Zhang, Z.; Li, D.; Ruan, X.; Yang, J.; Chen, S.; Li, X.; Ling, W. Integrating Oxygen-Boosted Sonodynamic Therapy and Ferroptosis via Engineered Exosomes for Effective Cancer Treatment. Theranostics 2025, 15, 68–85. [Google Scholar] [CrossRef]
- Ma, J.; Yuan, H.; Zhang, J.; Sun, X.; Yi, L.; Li, W.; Li, Z.; Fu, C.; Zheng, L.; Xu, X.; et al. An Ultrasound-Activated Nanoplatform Remodels Tumor Microenvironment through Diverse Cell Death Induction for Improved Immunotherapy. J. Control. Release 2024, 370, 501–515. [Google Scholar] [CrossRef] [PubMed]
- Chen, J.; Zhan, Q.; Li, L.; Xi, S.; Cai, L.; Liu, R.; Chen, L. Cell-Membrane Targeting Sonodynamic Therapy Combination with FSP1 Inhibition for Ferroptosis-Boosted Immunotherapy. Mater. Today Bio 2024, 30, 101407. [Google Scholar] [CrossRef]
- Yamaguchi, T.; Kitahara, S.; Kusuda, K.; Okamoto, J.; Horise, Y.; Masamune, K.; Muragaki, Y. Current Landscape of Sonodynamic Therapy for Treating Cancer. Cancers 2021, 13, 6184. [Google Scholar] [CrossRef]
- Jeong, Y.-G.; Park, J.-H.; Khang, D. Sonodynamic and Acoustically Responsive Nanodrug Delivery System: Cancer Application. Int. J. Nanomed. 2024, 19, 11767–11788. [Google Scholar] [CrossRef]
- Radivoievych, A.; Prylutska, S.; Zolk, O.; Ritter, U.; Frohme, M.; Grebinyk, A. Comparison of Sonodynamic Treatment Set-Ups for Cancer Cells with Organic Sonosensitizers and Nanosonosensitizers. Pharmaceutics 2023, 15, 2616. [Google Scholar] [CrossRef]
- Li, D.; Yang, Y.; Li, D.; Pan, J.; Chu, C.; Liu, G. Organic Sonosensitizers for Sonodynamic Therapy: From Small Molecules and Nanoparticles toward Clinical Development. Small 2021, 17, 2101976. [Google Scholar] [CrossRef]
- Yu, Z.; Cao, W.; Han, C.; Wang, Z.; Qiu, Y.; Wang, J.; Wei, M.; Wang, J.; Zhang, S.; Liu, S.; et al. Biomimetic Metal-Organic Framework Nanoparticles for Synergistic Combining of SDT-Chemotherapy Induce Pyroptosis in Gastric Cancer. Front. Bioeng. Biotechnol. 2022, 10, 796820. [Google Scholar] [CrossRef]
- Li, Y.; Zhou, Q.; Deng, Z.; Pan, M.; Liu, X.; Wu, J.; Yan, F.; Zheng, H. IR-780 Dye as a Sonosensitizer for Sonodynamic Therapy of Breast Tumor. Sci. Rep. 2016, 6, 25968. [Google Scholar] [CrossRef]
- Umemura, S.; Yumita, N.; Umemura, K.; Nishigaki, R. Sonodynamically Induced Effect of Rose Bengal on Isolated Sarcoma 180 Cells. Cancer Chemother. Pharmacol. 1999, 43, 389–393. [Google Scholar] [CrossRef] [PubMed]
- He, L.-L.; Wu, X.-X.; Wang, Y.-X.; Liu, X.-P.; Song, Y.-L.; Yang, Y.-J.; Gao, Y.-P.; Wang, X.; Liu, B.; Wang, X. Spectroscopic Investigation on the Sonodynamic Damage to Protein in the Presence of Eosine B. Ultrason. Sonochem. 2015, 26, 93–98. [Google Scholar] [CrossRef] [PubMed]
- Zhu, J.-X.; Zhu, W.-T.; Hu, J.-H.; Yang, W.; Liu, P.; Liu, Q.-H.; Bai, Y.-X.; Xie, R. Curcumin-Loaded Poly(L-Lactide-Co-Glycolide) Microbubble-Mediated Sono-Photodynamic Therapy in Liver Cancer Cells. Ultrasound Med. Biol. 2020, 46, 2030–2043. [Google Scholar] [CrossRef]
- Ninomiya, K.; Noda, K.; Ogino, C.; Kuroda, S.; Shimizu, N. Enhanced OH Radical Generation by Dual-Frequency Ultrasound with TiO2 Nanoparticles: Its Application to Targeted Sonodynamic Therapy. Ultrason. Sonochem 2014, 21, 289–294. [Google Scholar] [CrossRef] [PubMed]
- Yang, G.; Ji, J.; Liu, Z. Multifunctional MnO2 Nanoparticles for Tumor Microenvironment Modulation and Cancer Therapy. Wiley Interdiscip. Rev. Nanomed. Nanobiotechnol. 2021, 13, e1720. [Google Scholar] [CrossRef]
- Conte, M.; Carofiglio, M.; Vander Pol, R.S.; Wood, A.; Hernandez, N.; Joubert, A.; Caffey, C.; Chua, C.Y.X.; Grattoni, A.; Cauda, V. Acoustically Driven Hybrid Nanocrystals for In Vivo Pancreatic Cancer Treatment. ACS Appl. Mater. Interfaces 2025, 17, 11873–11887. [Google Scholar] [CrossRef]
- Carofiglio, M.; Mesiano, G.; Rosso, G.; Conte, M.; Zuccheri, M.; Pignochino, Y.; Cauda, V. Targeted Lipid-Coated ZnO Nanoparticles Coupled with Ultrasound: A Sonodynamic Approach for the Treatment of Osteosarcoma as 3D Spheroid Models. Mater. Today Commun. 2024, 40, 109826. [Google Scholar] [CrossRef]
- Osminkina, L.A.; Sivakov, V.A.; Mysov, G.A.; Georgobiani, V.A.; Natashina, U.A.; Talkenberg, F.; Solovyev, V.V.; Kudryavtsev, A.A.; Timoshenko, V.Y. Nanoparticles Prepared from Porous Silicon Nanowires for Bio-Imaging and Sonodynamic Therapy. Nanoscale Res. Lett. 2014, 9, 463. [Google Scholar] [CrossRef]
- Yumita, N.; Iwase, Y.; Imaizumi, T.; Sakurazawa, A.; Kaya, Y.; Nishi, K.; Ikeda, T.; Umemura, S.-I.; Chen, F.-S.; Momose, Y. Sonodynamically-Induced Anticancer Effects by Functionalized Fullerenes. Anticancer Res. 2013, 33, 3145–3151. [Google Scholar]
- Chen, P.; Zhang, P.; Shah, N.H.; Cui, Y.; Wang, Y. A Comprehensive Review of Inorganic Sonosensitizers for Sonodynamic Therapy. Int. J. Mol. Sci. 2023, 24, 12001. [Google Scholar] [CrossRef] [PubMed]
- Wang, T.; Du, M.; Chen, Z. Sonosensitizers for Sonodynamic Therapy: Current Progress and Future Perspectives. Ultrasound Med. Biol. 2025, 51, 727–734. [Google Scholar] [CrossRef]
- Barati, A.H.; Mokhtari-Dizaji, M. Ultrasound Dose Fractionation in Sonodynamic Therapy. Ultrasound Med. Biol. 2010, 36, 880–887. [Google Scholar] [CrossRef]
- Sofuni, A.; Itoi, T. Current Status and Future Perspective of Sonodynamic Therapy for Cancer. J. Med. Ultrason 2022. [Google Scholar] [CrossRef] [PubMed]
- Wu, P.; Dong, W.; Guo, X.; Qiao, X.; Guo, S.; Zhang, L.; Wan, M.; Zong, Y. ROS-Responsive Blended Nanoparticles: Cascade-Amplifying Synergistic Effects of Sonochemotherapy with On-Demand Boosted Drug Release During SDT Process. Adv. Healthc. Mater. 2019, 8, 1900720. [Google Scholar] [CrossRef] [PubMed]
- An, J.; Hu, Y.-G.; Cheng, K.; Li, C.; Hou, X.-L.; Wang, G.-L.; Zhang, X.-S.; Liu, B.; Zhao, Y.-D.; Zhang, M.-Z. ROS-Augmented and Tumor-Microenvironment Responsive Biodegradable Nanoplatform for Enhancing Chemo-Sonodynamic Therapy. Biomaterials 2020, 234, 119761. [Google Scholar] [CrossRef]
- Yan, P.; Liu, L.-H.; Wang, P. Sonodynamic Therapy (SDT) for Cancer Treatment: Advanced Sensitizers by Ultrasound Activation to Injury Tumor. ACS Appl. Bio Mater. 2020, 3, 3456–3475. [Google Scholar] [CrossRef]
- Xu, M.; Zhou, L.; Zheng, L.; Zhou, Q.; Liu, K.; Mao, Y.; Song, S. Sonodynamic Therapy-Derived Multimodal Synergistic Cancer Therapy. Cancer Lett. 2021, 497, 229–242. [Google Scholar] [CrossRef]
- Wang, X.; Zhong, X.; Gong, F.; Chao, Y.; Cheng, L. Newly Developed Strategies for Improving Sonodynamic Therapy. Mater. Horiz. 2020, 7, 2028–2046. [Google Scholar] [CrossRef]
- Muz, B.; de la Puente, P.; Azab, F.; Azab, A.K. The Role of Hypoxia in Cancer Progression, Angiogenesis, Metastasis, and Resistance to Therapy. Hypoxia 2015, 3, 83–92. [Google Scholar] [CrossRef]
- Chen, Z.; Han, F.; Du, Y.; Shi, H.; Zhou, W. Hypoxic Microenvironment in Cancer: Molecular Mechanisms and Therapeutic Interventions. Signal Transduct. Target. Ther. 2023, 8, 70. [Google Scholar] [CrossRef]
- Huang, B.; Chen, S.; Pei, W.; Xu, Y.; Jiang, Z.; Niu, C.; Wang, L. Oxygen-Sufficient Nanoplatform for Chemo-Sonodynamic Therapy of Hypoxic Tumors. Front. Chem. 2020, 8, 358. [Google Scholar] [CrossRef]
- Li, J.; Yue, Z.; Tang, M.; Wang, W.; Sun, Y.; Sun, T.; Chen, C. Strategies to Reverse Hypoxic Tumor Microenvironment for Enhanced Sonodynamic Therapy. Adv. Healthc. Mater. 2024, 13, e2302028. [Google Scholar] [CrossRef] [PubMed]
- Yang, K.; Yue, L.; Yu, G.; Rao, L.; Tian, R.; Wei, J.; Yang, Z.; Sun, C.; Zhang, X.; Xu, M.; et al. A Hypoxia Responsive Nanoassembly for Tumor Specific Oxygenation and Enhanced Sonodynamic Therapy. Biomaterials 2021, 275, 120822. [Google Scholar] [CrossRef]
- Fu, J.; Li, T.; Zhu, Y.; Hao, Y. Ultrasound-Activated Oxygen and ROS Generation Nanosystem Systematically Modulates Tumor Microenvironment and Sensitizes Sonodynamic Therapy for Hypoxic Solid Tumors. Adv. Funct. Mater. 2019, 29, 1906195. [Google Scholar] [CrossRef]
- Gao, F.; He, G.; Yin, H.; Chen, J.; Liu, Y.; Lan, C.; Zhang, S.; Yang, B. Titania-Coated 2D Gold Nanoplates as Nanoagents for Synergistic Photothermal/Sonodynamic Therapy in the Second near-Infrared Window. Nanoscale 2019, 11, 2374–2384. [Google Scholar] [CrossRef]
- Liang, S.; Deng, X.; Chang, Y.; Sun, C.; Shao, S.; Xie, Z.; Xiao, X.; Ma, P.; Zhang, H.; Cheng, Z.; et al. Intelligent Hollow Pt-CuS Janus Architecture for Synergistic Catalysis-Enhanced Sonodynamic and Photothermal Cancer Therapy. Nano Lett. 2019, 19, 4134–4145. [Google Scholar] [CrossRef]
- Liu, Y.; Zhen, W.; Jin, L.; Zhang, S.; Sun, G.; Zhang, T.; Xu, X.; Song, S.; Wang, Y.; Liu, J.; et al. All-in-One Theranostic Nanoagent with Enhanced Reactive Oxygen Species Generation and Modulating Tumor Microenvironment Ability for Effective Tumor Eradication. ACS Nano 2018, 12, 4886–4893. [Google Scholar] [CrossRef]
- Li, X.; Sun, Y.; Wang, Y.; Zhou, Y.; Bao, Y.; Zhang, Z.; Liu, S.; Yang, H.; Zhang, R.; Xia, P.; et al. Amplifying Radiotherapy by Evoking Mitochondrial Oxidative Stress Using a High-Performance Aggregation-Induced Emission Sonosensitizer. Curr. Med. Chem. 2025, 32, 380–395. [Google Scholar] [CrossRef]
- Liu, J.; Shi, M.; Zhao, H.; Bai, X.; Lin, Q.; Guan, X.; Wu, B.; E., M. Ultrasound-Activated Nano-Oxygen Sensitizer for Sonodynamic–Radiotherapy of Esophageal Cancer. Nanoscale Adv. 2025, 7, 2209–2221. [Google Scholar] [CrossRef]
- Zha, B.; Yang, J.; Dang, Q.; Li, P.; Shi, S.; Wu, J.; Cui, H.; Huangfu, L.; Li, Y.; Yang, D.; et al. A Phase I Clinical Trial of Sonodynamic Therapy Combined with Temozolomide in the Treatment of Recurrent Glioblastoma. J. Neurooncol 2023, 162, 317–326. [Google Scholar] [CrossRef]
- Huangfu, L.; Zha, B.; Li, P.; Wang, L.; Liu, X.; Cui, H.; Li, Y.; Wu, J.; Shi, S.; Yang, Y.; et al. A Phase I Clinical Trial of Sonodynamic Therapy Combined with Radiotherapy for Brainstem Gliomas. Int. J. Cancer 2025, 156, 1005–1014. [Google Scholar] [CrossRef]
- Yue, W.; Chen, L.; Yu, L.; Zhou, B.; Yin, H.; Ren, W.; Liu, C.; Guo, L.; Zhang, Y.; Sun, L.; et al. Checkpoint Blockade and Nanosonosensitizer-Augmented Noninvasive Sonodynamic Therapy Combination Reduces Tumour Growth and Metastases in Mice. Nat. Commun. 2019, 10, 2025. [Google Scholar] [CrossRef]
- Bulner, S.; Prodeus, A.; Gariepy, J.; Hynynen, K.; Goertz, D.E. Enhancing Checkpoint Inhibitor Therapy with Ultrasound Stimulated Microbubbles. Ultrasound Med. Biol. 2019, 45, 500–512. [Google Scholar] [CrossRef]
- Lin, H.; Xiong, W.; Fu, L.; Yi, J.; Yang, J. Damage-Associated Molecular Patterns (DAMPs) in Diseases: Implications for Therapy. Mol. Biomed. 2025, 6, 60. [Google Scholar] [CrossRef]
- Wang, T.; Peng, W.; Du, M.; Chen, Z. Immunogenic Sonodynamic Therapy for Inducing Immunogenic Cell Death and Activating Antitumor Immunity. Front. Oncol. 2023, 13, 1167105. [Google Scholar] [CrossRef]
- Duan, X.; Chan, C.; Lin, W. Nanoparticle-Mediated Immunogenic Cell Death Enables and Potentiates Cancer Immunotherapy. Angew. Chem. Int. Ed. Engl. 2019, 58, 670–680. [Google Scholar] [CrossRef]
- Liao, H.; Chen, M.; Liao, Z.; Luo, Y.; Chen, S.; Tan, W.; Wang, L.; Niu, C. Sonodynamic Therapy-Boosted Biomimetic Nanoplatform Targets Ferroptosis and CD47 as Vulnerabilities for Cancer Immunotherapy. J. Nanobiotechnol. 2025, 23, 432. [Google Scholar] [CrossRef]
- Wang, X.; Wang, C.; Tian, H.; Wu, B.; Cheng, W. Tumor Microenvironment-Related Sonodynamic Therapy Mediates Tumor Therapy. Adv. Ther. 2023, 6, 2300265. [Google Scholar] [CrossRef]
- Chen, Q.; Sun, T.; Jiang, C. Recent Advancements in Nanomedicine for ‘Cold’ Tumor Immunotherapy. Nano-Micro Lett. 2021, 13, 92. [Google Scholar] [CrossRef]
- Collins, V.G.; Hutton, D.; Hossain-Ibrahim, K.; Joseph, J.; Banerjee, S. The Abscopal Effects of Sonodynamic Therapy in Cancer. Br. J. Cancer 2025, 132, 409–420. [Google Scholar] [CrossRef]
- Huang, J.; Xiao, Z.; An, Y.; Han, S.; Wu, W.; Wang, Y.; Guo, Y.; Shuai, X. Nanodrug with Dual-Sensitivity to Tumor Microenvironment for Immuno-Sonodynamic Anti-Cancer Therapy. Biomaterials 2021, 269, 120636. [Google Scholar] [CrossRef]
- Nesbitt, H.; Logan, K.; Thomas, K.; Callan, B.; Gao, J.; McKaig, T.; Taylor, M.; Love, M.; Stride, E.; McHale, A.P.; et al. Sonodynamic Therapy Complements PD-L1 Immune Checkpoint Inhibition in a Murine Model of Pancreatic Cancer. Cancer Lett. 2021, 517, 88–95. [Google Scholar] [CrossRef]
- Yang, Y.; Huang, J.; Liu, M.; Qiu, Y.; Chen, Q.; Zhao, T.; Xiao, Z.; Yang, Y.; Jiang, Y.; Huang, Q.; et al. Emerging Sonodynamic Therapy-Based Nanomedicines for Cancer Immunotherapy. Adv. Sci. 2023, 10, 2204365. [Google Scholar] [CrossRef]
- Son, S.; Kim, J.H.; Wang, X.; Zhang, C.; Yoon, S.A.; Shin, J.; Sharma, A.; Lee, M.H.; Cheng, L.; Wu, J.; et al. Multifunctional Sonosensitizers in Sonodynamic Cancer Therapy. Chem. Soc. Rev. 2020, 49, 3244–3261. [Google Scholar] [CrossRef]
- Kalluri, R.; LeBleu, V.S. The Biology, Function, and Biomedical Applications of Exosomes. Science 2020, 367, eaau6977. [Google Scholar] [CrossRef]
- Kumar, M.A.; Baba, S.K.; Sadida, H.Q.; Marzooqi, S.A.; Jerobin, J.; Altemani, F.H.; Algehainy, N.; Alanazi, M.A.; Abou-Samra, A.-B.; Kumar, R.; et al. Extracellular Vesicles as Tools and Targets in Therapy for Diseases. Signal Transduct. Target. Ther. 2024, 9, 27. [Google Scholar] [CrossRef]
- Chiabotto, G.; Gai, C.; Deregibus, M.C.; Camussi, G. Salivary Extracellular Vesicle-Associated exRNA as Cancer Biomarker. Cancers 2019, 11, 891. [Google Scholar] [CrossRef]
- De Giorgis, V.; Barberis, E.; Manfredi, M. Extracellular Vesicles Proteins for Early Cancer Diagnosis: From Omics to Biomarkers. Semin. Cancer Biol. 2024, 104–105, 18–31. [Google Scholar] [CrossRef]
- Pegtel, D.M.; Gould, S.J. Exosomes. Annu. Rev. Biochem. 2019, 88, 487–514. [Google Scholar] [CrossRef]
- Berumen Sánchez, G.; Bunn, K.E.; Pua, H.H.; Rafat, M. Extracellular Vesicles: Mediators of Intercellular Communication in Tissue Injury and Disease. Cell Commun. Signal. 2021, 19, 104. [Google Scholar] [CrossRef]
- Wandrey, M.; Jablonska, J.; Stauber, R.H.; Gül, D. Exosomes in Cancer Progression and Therapy Resistance: Molecular Insights and Therapeutic Opportunities. Life 2023, 13, 2033. [Google Scholar] [CrossRef]
- Ten, A.; Kumeiko, V.; Farniev, V.; Gao, H.; Shevtsov, M. Tumor Microenvironment Modulation by Cancer-Derived Extracellular Vesicles. Cells 2024, 13, 682. [Google Scholar] [CrossRef]
- Yang, X.; Zhang, Y.; Zhang, Y.; Zhang, S.; Qiu, L.; Zhuang, Z.; Wei, M.; Deng, X.; Wang, Z.; Han, J. The Key Role of Exosomes on the Pre-Metastatic Niche Formation in Tumors. Front. Mol. Biosci. 2021, 8, 703640. [Google Scholar] [CrossRef] [PubMed]
- Chen, J.; Hu, S.; Liu, J.; Jiang, H.; Wang, S.; Yang, Z. Exosomes: A Double-edged Sword in Cancer Immunotherapy. MedComm 2025, 6, e70095. [Google Scholar] [CrossRef]
- Elsharkasy, O.M.; Nordin, J.Z.; Hagey, D.W.; de Jong, O.G.; Schiffelers, R.M.; Andaloussi, S.E.; Vader, P. Extracellular Vesicles as Drug Delivery Systems: Why and How? Adv. Drug Deliv. Rev. 2020, 159, 332–343. [Google Scholar] [CrossRef]
- Ha, D.; Yang, N.; Nadithe, V. Exosomes as Therapeutic Drug Carriers and Delivery Vehicles across Biological Membranes: Current Perspectives and Future Challenges. Acta Pharm. Sin. B 2016, 6, 287–296. [Google Scholar] [CrossRef]
- Luan, X.; Sansanaphongpricha, K.; Myers, I.; Chen, H.; Yuan, H.; Sun, D. Engineering Exosomes as Refined Biological Nanoplatforms for Drug Delivery. Acta Pharmacol. Sin. 2017, 38, 754–763. [Google Scholar] [CrossRef]
- Vader, P.; Mol, E.A.; Pasterkamp, G.; Schiffelers, R.M. Extracellular Vesicles for Drug Delivery. Adv. Drug Deliv. Rev. 2016, 106, 148–156. [Google Scholar] [CrossRef] [PubMed]
- Gilligan, K.E.; Dwyer, R.M. Engineering Exosomes for Cancer Therapy. Int. J. Mol. Sci. 2017, 18, 1122. [Google Scholar] [CrossRef]
- Kim, M.S.; Haney, M.J.; Zhao, Y.; Mahajan, V.; Deygen, I.; Klyachko, N.L.; Inskoe, E.; Piroyan, A.; Sokolsky, M.; Okolie, O.; et al. Development of Exosome-Encapsulated Paclitaxel to Overcome MDR in Cancer Cells. Nanomedicine 2016, 12, 655–664. [Google Scholar] [CrossRef]
- Murphy, D.E.; de Jong, O.G.; Brouwer, M.; Wood, M.J.; Lavieu, G.; Schiffelers, R.M.; Vader, P. Extracellular Vesicle-Based Therapeutics: Natural versus Engineered Targeting and Trafficking. Exp. Mol. Med. 2019, 51, 1–12. [Google Scholar] [CrossRef]
- Wiklander, O.P.B.; Brennan, M.Á.; Lötvall, J.; Breakefield, X.O.; El Andaloussi, S. Advances in Therapeutic Applications of Extracellular Vesicles. Sci. Transl. Med. 2019, 11, eaav8521. [Google Scholar] [CrossRef] [PubMed]
- Palakurthi, S.S.; Shah, B.; Kapre, S.; Charbe, N.; Immanuel, S.; Pasham, S.; Thalla, M.; Jain, A.; Palakurthi, S. A Comprehensive Review of Challenges and Advances in Exosome-Based Drug Delivery Systems. Nanoscale Adv. 2024, 6, 5803–5826. [Google Scholar] [CrossRef]
- Sadeghi, S.; Tehrani, F.R.; Tahmasebi, S.; Shafiee, A.; Hashemi, S.M. Exosome Engineering in Cell Therapy and Drug Delivery. Inflammopharmacology 2023, 31, 145–169. [Google Scholar] [CrossRef]
- Armstrong, J.P.K.; Holme, M.N.; Stevens, M.M. Re-Engineering Extracellular Vesicles as Smart Nanoscale Therapeutics. ACS Nano 2017, 11, 69–83. [Google Scholar] [CrossRef]
- Serrano, D.R.; Juste, F.; Anaya, B.J.; Ramirez, B.I.; Sánchez-Guirales, S.A.; Quispillo, J.M.; Hernandez, E.M.; Simon, J.A.; Trallero, J.M.; Serrano, C.; et al. Exosome-Based Drug Delivery: A Next-Generation Platform for Cancer, Infection, Neurological and Immunological Diseases, Gene Therapy and Regenerative Medicine. Pharmaceutics 2025, 17, 1336. [Google Scholar] [CrossRef] [PubMed]
- Carobolante, G.; Mantaj, J.; Ferrari, E.; Vllasaliu, D. Cow Milk and Intestinal Epithelial Cell-Derived Extracellular Vesicles as Systems for Enhancing Oral Drug Delivery. Pharmaceutics 2020, 12, 226. [Google Scholar] [CrossRef] [PubMed]
- Aqil, F.; Munagala, R.; Jeyabalan, J.; Agrawal, A.K.; Gupta, R. Exosomes for the Enhanced Tissue Bioavailability and Efficacy of Curcumin. AAPS J. 2017, 19, 1691–1702. [Google Scholar] [CrossRef] [PubMed]
- Li, J.; Wang, J.; Chen, Z. Emerging Role of Exosomes in Cancer Therapy: Progress and Challenges. Mol. Cancer 2025, 24, 13. [Google Scholar] [CrossRef]
- Zhou, Y.; Zhou, W.; Chen, X.; Wang, Q.; Li, C.; Chen, Q.; Zhang, Y.; Lu, Y.; Ding, X.; Jiang, C. Bone Marrow Mesenchymal Stem Cells-Derived Exosomes for Penetrating and Targeted Chemotherapy of Pancreatic Cancer. Acta Pharm. Sin. B 2020, 10, 1563–1575. [Google Scholar] [CrossRef]
- Wang, P.; Wang, H.; Huang, Q.; Peng, C.; Yao, L.; Chen, H.; Qiu, Z.; Wu, Y.; Wang, L.; Chen, W. Exosomes from M1-Polarized Macrophages Enhance Paclitaxel Antitumor Activity by Activating Macrophages-Mediated Inflammation. Theranostics 2019, 9, 1714–1727. [Google Scholar] [CrossRef]
- Cao, X.-H.; Liang, M.-X.; Wu, Y.; Yang, K.; Tang, J.-H.; Zhang, W. Extracellular Vesicles as Drug Vectors for Precise Cancer Treatment. Nanomedicine 2021, 16, 1519–1537. [Google Scholar] [CrossRef]
- Koh, H.B.; Kim, H.J.; Kang, S.-W.; Yoo, T.-H. Exosome-Based Drug Delivery: Translation from Bench to Clinic. Pharmaceutics 2023, 15, 2042. [Google Scholar] [CrossRef]
- Huang, J.; Chen, H.; Li, N.; Liu, P.; Yang, J.; Zhao, Y. Emerging Technologies towards Extracellular Vesicles Large-Scale Production. Bioact. Mater. 2025, 52, 338–365. [Google Scholar] [CrossRef]
- Yang, Z.; Shi, J.; Xie, J.; Wang, Y.; Sun, J.; Liu, T.; Zhao, Y.; Zhao, X.; Wang, X.; Ma, Y.; et al. Large-Scale Generation of Functional mRNA-Encapsulating Exosomes via Cellular Nanoporation. Nat. Biomed. Eng. 2020, 4, 69–83. [Google Scholar] [CrossRef]
- Yang, X.; Pan, Z.; Choudhury, M.R.; Yuan, Z.; Anifowose, A.; Yu, B.; Wang, W.; Wang, B. Making Smart Drugs Smarter: The Importance of Linker Chemistry in Targeted Drug Delivery. Med. Res. Rev. 2020, 40, 2682–2713. [Google Scholar] [CrossRef] [PubMed]
- Gabaran, S.G.; Ghasemzadeh, N.; Rahnama, M.; Karatas, E.; Akbari, A.; Rezaie, J. Functionalized Exosomes for Targeted Therapy in Cancer and Regenerative Medicine: Genetic, Chemical, and Physical Modifications. Cell Commun. Signal. 2025, 23, 265. [Google Scholar] [CrossRef]
- Liu, Q.; Li, D.; Pan, X.; Liang, Y. Targeted Therapy Using Engineered Extracellular Vesicles: Principles and Strategies for Membrane Modification. J. Nanobiotechnol. 2023, 21, 334. [Google Scholar] [CrossRef] [PubMed]
- Chao, T.; Zhao, J.; Gao, R.; Wang, H.; Guo, J.; Gao, Z.; Wang, Y. Exosome Surface Modification and Functionalization: A Narrative Review of Emerging Technologies and Their Application Potential in Precision Medicine. Adv. Technol. Neurosci. 2025, 2, 27. [Google Scholar] [CrossRef]
- Limongi, T.; Susa, F.; Dumontel, B.; Racca, L.; Perrone Donnorso, M.; Debellis, D.; Cauda, V. Extracellular Vesicles Tropism: A Comparative Study between Passive Innate Tropism and the Active Engineered Targeting Capability of Lymphocyte-Derived EVs. Membranes 2021, 11, 886. [Google Scholar] [CrossRef]
- Hong, L.; Li, W.; Li, Y.; Yin, S. Nanoparticle-Based Drug Delivery Systems Targeting Cancer Cell Surfaces. RSC Adv. 2023, 13, 21365–21382. [Google Scholar] [CrossRef]
- Lorenzoni, S.; Rodríguez-Nogales, C.; Blanco-Prieto, M.J. Targeting Tumor Microenvironment with RGD-Functionalized Nanoparticles for Precision Cancer Therapy. Cancer Lett. 2025, 614, 217536. [Google Scholar] [CrossRef]
- Qiu, M.; Zou, J.; Yang, Z.; Yang, D.; Wang, R.; Guo, H. Strategies for Targeting Peptide-Modified Exosomes and Their Applications in the Lungs. Int. J. Nanomed. 2024, 19, 8175–8188. [Google Scholar] [CrossRef] [PubMed]
- Smyth, T.; Petrova, K.; Payton, N.M.; Persaud, I.; Redzic, J.S.; Graner, M.W.; Smith-Jones, P.; Anchordoquy, T.J. Surface Functionalization of Exosomes Using Click Chemistry. Bioconjug. Chem. 2014, 25, 1777–1784. [Google Scholar] [CrossRef]
- Xu, L.; Chopdat, R.; Li, D.; Al-Jamal, K.T. Development of a Simple, Sensitive and Selective Colorimetric Aptasensor for the Detection of Cancer-Derived Exosomes. Biosens. Bioelectron. 2020, 169, 112576. [Google Scholar] [CrossRef] [PubMed]
- Kim, M.S.; Haney, M.J.; Zhao, Y.; Yuan, D.; Deygen, I.; Klyachko, N.L.; Kabanov, A.V.; Batrakova, E.V. Engineering Macrophage-Derived Exosomes for Targeted Paclitaxel Delivery to Pulmonary Metastases: In Vitro and in Vivo Evaluations. Nanomed. Nanotechnol. Biol. Med. 2018, 14, 195–204. [Google Scholar] [CrossRef]
- Jang, S.C.; Kim, O.Y.; Yoon, C.M.; Choi, D.-S.; Roh, T.-Y.; Park, J.; Nilsson, J.; Lötvall, J.; Kim, Y.-K.; Gho, Y.S. Bioinspired Exosome-Mimetic Nanovesicles for Targeted Delivery of Chemotherapeutics to Malignant Tumors. ACS Nano 2013, 7, 7698–7710. [Google Scholar] [CrossRef]
- Kooijmans, S.A.A.; de Jong, O.G.; Schiffelers, R.M. Exploring Interactions between Extracellular Vesicles and Cells for Innovative Drug Delivery System Design. Adv. Drug Deliv. Rev. 2021, 173, 252–278. [Google Scholar] [CrossRef]
- Rosso, G.; Cauda, V. Biomimicking Extracellular Vesicles with Fully Artificial Ones: A Rational Design of EV-BIOMIMETICS toward Effective Theranostic Tools in Nanomedicine. ACS Biomater. Sci. Eng. 2023, 9, 5924–5932. [Google Scholar] [CrossRef]
- Yoon, J.; Jo, W.; Jeong, D.; Kim, J.; Jeong, H.; Park, J. Generation of Nanovesicles with Sliced Cellular Membrane Fragments for Exogenous Material Delivery. Biomaterials 2015, 59, 12–20. [Google Scholar] [CrossRef]
- Chen, Y.; Douanne, N.; Wu, T.; Kaur, I.; Tsering, T.; Erzingatzian, A.; Nadeau, A.; Juncker, D.; Nerguizian, V.; Burnier, J.V. Leveraging Nature’s Nanocarriers: Translating Insights from Extracellular Vesicles to Biomimetic Synthetic Vesicles for Biomedical Applications. Sci. Adv. 2025, 11, eads5249. [Google Scholar] [CrossRef] [PubMed]
- Sundaram, V.; Aryal, S. Emerging Biomimetic Drug Delivery Nanoparticles Inspired by Extracellular Vesicles. Wiley Interdiscip. Rev. Nanomed. Nanobiotechnol. 2025, 17, e70025. [Google Scholar] [CrossRef]
- Rosso, G.; Van Veen, S.M.A.; Sancho-Albero, M.; Tamboia, G.; Empereur-Mot, C.; Perego, C.; Kuipers, M.E.; Dumontel, B.; Ajó, A.; Nolte-’t Hoen, E.N.; et al. Rational Design of EV-Mimicking Nanoparticles with Polarity-Based Recognition Potential for Advanced Nanocarrier Development. ACS Appl. Nano Mater. 2025, 8, 13257–13273. [Google Scholar] [CrossRef] [PubMed]
- Parveen, S.; Gupta, P.; Kumar, S.; Banerjee, M. Lipid Polymer Hybrid Nanoparticles as Potent Vehicles for Drug Delivery in Cancer Therapeutics. Med. Drug Discov. 2023, 20, 100165. [Google Scholar] [CrossRef]
- Liu, X.; Xiao, C.; Xiao, K. Engineered Extracellular Vesicles-like Biomimetic Nanoparticles as an Emerging Platform for Targeted Cancer Therapy. J. Nanobiotechnol. 2023, 21, 287. [Google Scholar] [CrossRef] [PubMed]
- Li, L.; He, D.; Guo, Q.; Zhang, Z.; Ru, D.; Wang, L.; Gong, K.; Liu, F.; Duan, Y.; Li, H. Exosome-Liposome Hybrid Nanoparticle Codelivery of TP and miR497 Conspicuously Overcomes Chemoresistant Ovarian Cancer. J. Nanobiotechnol. 2022, 20, 50. [Google Scholar] [CrossRef]
- Soltanmohammadi, F.; Gharehbaba, A.M.; Zangi, A.R.; Adibkia, K.; Javadzadeh, Y. Current Knowledge of Hybrid Nanoplatforms Composed of Exosomes and Organic/Inorganic Nanoparticles for Disease Treatment and Cell/Tissue Imaging. Biomed. Pharmacother. 2024, 178, 117248. [Google Scholar] [CrossRef]
- Tang, L.; Zhang, W.; Qi, T.; Jiang, Z.; Tang, D. Exosomes Play a Crucial Role in Remodeling the Tumor Microenvironment and in the Treatment of Gastric Cancer. Cell Commun. Signal. 2025, 23, 82. [Google Scholar] [CrossRef]
- Zhou, K.; Guo, S.; Li, F.; Sun, Q.; Liang, G. Exosomal PD-L1: New Insights into Tumor Immune Escape Mechanisms and Therapeutic Strategies. Front. Cell Dev. Biol. 2020, 8, 569219. [Google Scholar] [CrossRef] [PubMed]
- Xie, Q.-H.; Zheng, J.-Q.; Ding, J.-Y.; Wu, Y.-F.; Liu, L.; Yu, Z.-L.; Chen, G. Exosome-Mediated Immunosuppression in Tumor Microenvironments. Cells 2022, 11, 1946. [Google Scholar] [CrossRef]
- Kamerkar, S.; Leng, C.; Burenkova, O.; Jang, S.C.; McCoy, C.; Zhang, K.; Dooley, K.; Kasera, S.; Zi, T.; Sisó, S.; et al. Exosome-Mediated Genetic Reprogramming of Tumor-Associated Macrophages by exoASO-STAT6 Leads to Potent Monotherapy Antitumor Activity. Sci. Adv. 2022, 8, eabj7002. [Google Scholar] [CrossRef]
- Escudier, B.; Dorval, T.; Chaput, N.; André, F.; Caby, M.-P.; Novault, S.; Flament, C.; Leboulaire, C.; Borg, C.; Amigorena, S.; et al. Vaccination of Metastatic Melanoma Patients with Autologous Dendritic Cell (DC) Derived-Exosomes: Results of Thefirst Phase I Clinical Trial. J. Transl. Med. 2005, 3, 10. [Google Scholar] [CrossRef]
- Fridman, E.S.; Ginini, L.; Gil, Z. The Role of Extracellular Vesicles in Metabolic Reprogramming of the Tumor Microenvironment. Cells 2022, 11, 1433. [Google Scholar] [CrossRef]
- Jung, K.O.; Youn, H.; Lee, C.-H.; Kang, K.W.; Chung, J.-K. Visualization of Exosome-Mediated miR-210 Transfer from Hypoxic Tumor Cells. Oncotarget 2016, 8, 9899–9910. [Google Scholar] [CrossRef]
- He, Q.; Ye, A.; Ye, W.; Liao, X.; Qin, G.; Xu, Y.; Yin, Y.; Luo, H.; Yi, M.; Xian, L.; et al. Cancer-Secreted Exosomal miR-21-5p Induces Angiogenesis and Vascular Permeability by Targeting KRIT1. Cell Death Dis. 2021, 12, 576. [Google Scholar] [CrossRef]
- Kosaka, N.; Iguchi, H.; Hagiwara, K.; Yoshioka, Y.; Takeshita, F.; Ochiya, T. Neutral Sphingomyelinase 2 (nSMase2)-Dependent Exosomal Transfer of Angiogenic microRNAs Regulate Cancer Cell Metastasis. J. Biol. Chem. 2013, 288, 10849–10859. [Google Scholar] [CrossRef]
- Wang, L.; Yang, L.; Zhuang, T.; Shi, X. Tumor-Derived Exosomal miR-29b Reduces Angiogenesis in Pancreatic Cancer by Silencing ROBO1 and SRGAP2. J. Immunol. Res. 2022, 2022, 4769385. [Google Scholar] [CrossRef]
- Lu, J.; Liu, Q.-H.; Wang, F.; Tan, J.-J.; Deng, Y.-Q.; Peng, X.-H.; Liu, X.; Zhang, B.; Xu, X.; Li, X.-P. Exosomal miR-9 Inhibits Angiogenesis by Targeting MDK and Regulating PDK/AKT Pathway in Nasopharyngeal Carcinoma. J. Exp. Clin. Cancer Res. 2018, 37, 147. [Google Scholar] [CrossRef]
- Rahman, M.A.; Barger, J.F.; Lovat, F.; Gao, M.; Otterson, G.A.; Nana-Sinkam, P. Lung Cancer Exosomes as Drivers of Epithelial Mesenchymal Transition. Oncotarget 2016, 7, 54852–54866. [Google Scholar] [CrossRef] [PubMed]
- Guo, W.; Wang, T.; Huang, C.; Ning, S.; Guo, Q.; Zhang, W.; Yang, H.; Zhu, D.; Huang, Q.; Qian, H.; et al. Platelet Membrane-Coated C-TiO2 Hollow Nanospheres for Combined Sonodynamic and Alkyl-Radical Cancer Therapy. Nano Res. 2023, 16, 782–791. [Google Scholar] [CrossRef]
- Dumontel, B.; Susa, F.; Limongi, T.; Vighetto, V.; Debellis, D.; Canta, M.; Cauda, V. Nanotechnological Engineering of Extracellular Vesicles for the Development of Actively Targeted Hybrid Nanodevices. Cell Biosci. 2022, 12, 61. [Google Scholar] [CrossRef] [PubMed]
- Nguyen Cao, T.G.; Kang, J.H.; You, J.Y.; Kang, H.C.; Rhee, W.J.; Ko, Y.T.; Shim, M.S. Safe and Targeted Sonodynamic Cancer Therapy Using Biocompatible Exosome-Based Nanosonosensitizers. ACS Appl. Mater. Interfaces 2021, 13, 25575–25588. [Google Scholar] [CrossRef]
- Nguyen Cao, T.G.; Kang, J.H.; Kim, W.; Lim, J.; Kang, S.J.; You, J.Y.; Truong Hoang, Q.; Kim, W.J.; Rhee, W.J.; Kim, C.; et al. Engineered Extracellular Vesicle-Based Sonotheranostics for Dual Stimuli-Sensitive Drug Release and Photoacoustic Imaging-Guided Chemo-Sonodynamic Cancer Therapy. Theranostics 2022, 12, 1247–1266. [Google Scholar] [CrossRef]
- Wu, T.; Liu, Y.; Cao, Y.; Liu, Z. Engineering Macrophage Exosome Disguised Biodegradable Nanoplatform for Enhanced Sonodynamic Therapy of Glioblastoma. Adv. Mater. 2022, 34, e2110364. [Google Scholar] [CrossRef] [PubMed]
- Li, Y.; Huang, C.; Xu, Y. Colon Cancer Exosome-Derived Biomimetic Nanoplatform for Curcumin-Mediated Sonodynamic Therapy and Calcium Overload. Front. Bioeng. Biotechnol. 2022, 10, 1069676. [Google Scholar] [CrossRef]
- Nguyen, V.-N.; Nguyen Cao, T.G.; Jeong, H.; Truong Hoang, Q.; Pham, B.T.T.; Bang, J.; Koh, C.W.; Kang, J.H.; Lee, J.H.; Wu, X.; et al. Tumor-Targeted Exosome-Based Heavy Atom-Free Nanosensitizers with Long-Lived Excited States for Safe and Effective Sono-Photodynamic Therapy of Solid Tumors. Adv. Healthc. Mater. 2025, 14, 2500927. [Google Scholar] [CrossRef]
- Nguyen Cao, T.G.; Truong Hoang, Q.; Hong, E.J.; Kang, S.J.; Kang, J.H.; Ravichandran, V.; Kang, H.C.; Ko, Y.T.; Rhee, W.J.; Shim, M.S. Mitochondria-Targeting Sonosensitizer-Loaded Extracellular Vesicles for Chemo-Sonodynamic Therapy. J. Control. Release 2023, 354, 651–663. [Google Scholar] [CrossRef]
- Nguyen Cao, T.G.; Truong Hoang, Q.; Kang, J.H.; Kang, S.J.; Ravichandran, V.; Rhee, W.J.; Lee, M.; Ko, Y.T.; Shim, M.S. Bioreducible Exosomes Encapsulating Glycolysis Inhibitors Potentiate Mitochondria-Targeted Sonodynamic Cancer Therapy via Cancer-Targeted Drug Release and Cellular Energy Depletion. Biomaterials 2023, 301, 122242. [Google Scholar] [CrossRef]
- Wang, D.; Wan, Z.; Yang, Q.; Chen, J.; Liu, Y.; Lu, F.; Tang, J. Sonodynamical Reversion of Immunosuppressive Microenvironment in Prostate Cancer via Engineered Exosomes. Drug Deliv. 2022, 29, 702–713. [Google Scholar] [CrossRef]
- Wu, J.; Huang, J.; Yu, J.; Xu, M.; Liu, J.; Pu, K. Exosome-Inhibiting Polymeric Sonosensitizer for Tumor-Specific Sonodynamic Immunotherapy. Adv. Mater. 2024, 36, e2400762. [Google Scholar] [CrossRef] [PubMed]
- Qu, X.; Fan, Q.; Liu, Y.; Zhang, J.; Yuan, B.; Cai, X.; Ji, L.; Zhuang, R.; Dong, Z. Synergizing Sono-Piezo with Exosome Suppression Using Doping-Engineered Hydroxyapatite for Potentiated Tumor Treatment through Immunoactivation. J. Nanobiotechnol. 2025, 23, 495. [Google Scholar] [CrossRef]
- Lener, T.; Gimona, M.; Aigner, L.; Börger, V.; Buzas, E.; Camussi, G.; Chaput, N.; Chatterjee, D.; Court, F.A.; Del Portillo, H.A.; et al. Applying Extracellular Vesicles Based Therapeutics in Clinical Trials—An ISEV Position Paper. J. Extracell. Vesicles 2015, 4, 30087. [Google Scholar] [CrossRef] [PubMed]
- Reiner, A.T.; Witwer, K.W.; van Balkom, B.W.M.; de Beer, J.; Brodie, C.; Corteling, R.L.; Gabrielsson, S.; Gimona, M.; Ibrahim, A.G.; de Kleijn, D.; et al. Concise Review: Developing Best-Practice Models for the Therapeutic Use of Extracellular Vesicles. Stem Cells Transl. Med. 2017, 6, 1730–1739. [Google Scholar] [CrossRef]
- Villata, S.; Canta, M.; Cauda, V. EVs and Bioengineering: From Cellular Products to Engineered Nanomachines. Int. J. Mol. Sci. 2020, 21, 6048. [Google Scholar] [CrossRef]
- Buschmann, D.; Mussack, V.; Byrd, J.B. Separation, Characterization, and Standardization of Extracellular Vesicles for Drug Delivery Applications. Adv. Drug Deliv. Rev. 2021, 174, 348–368. [Google Scholar] [CrossRef]
- Smyth, T.; Kullberg, M.; Malik, N.; Smith-Jones, P.; Graner, M.W.; Anchordoquy, T.J. Biodistribution and Delivery Efficiency of Unmodified Tumor-Derived Exosomes. J. Control. Release 2015, 199, 145–155. [Google Scholar] [CrossRef] [PubMed]
- Wiklander, O.P.B.; Nordin, J.Z.; O’Loughlin, A.; Gustafsson, Y.; Corso, G.; Mäger, I.; Vader, P.; Lee, Y.; Sork, H.; Seow, Y.; et al. Extracellular Vesicle in Vivo Biodistribution Is Determined by Cell Source, Route of Administration and Targeting. J. Extracell. Vesicles 2015, 4, 26316. [Google Scholar] [CrossRef] [PubMed]
- Pedrioli, G.; Piovesana, E.; Vacchi, E.; Balbi, C. Extracellular Vesicles as Promising Carriers in Drug Delivery: Considerations from a Cell Biologist’s Perspective. Biology 2021, 10, 376. [Google Scholar] [CrossRef] [PubMed]
- Hu, C.; Hou, B.; Xie, S. Application of Nanosonosensitizer Materials in Cancer Sono-Dynamic Therapy. RSC Adv. 2022, 12, 22722–22747. [Google Scholar] [CrossRef]
- Moosa, S. Pilot Study of Sonodynamic Therapy with 5-ALA for the Treatment of Recurrent Glioblastoma Using Neuronavigation-Guided Low-Intensity Focused Ultrasound. 2024. Available online: https://clinicaltrials.gov/study/NCT06039709 (accessed on 15 October 2025).
- SonALAsense, Inc. A Phase 2 Dose-Escalation Study Examining the Safety, Pharmacokinetics, and Preliminary Efficacy of Ascending Drug and Energy Dose Combinations for Sonodynamic Therapy Using SONALA-001 in Combination with Exablate 4000 Type 2.0 MR-Guided Focused Ultrasound in Subjects with Diffuse Intrinsic Pontine Glioma (DIPG) and Diffuse Midline Glioma (DMG). 2024. Available online: https://www.clinicaltrials.gov/study/NCT05123534 (accessed on 15 October 2025).
- Zheng, Y. Clinical Investigation of Sonodynamic Therapy in Conjunction with Chemoradiotherapy for Glioblastoma Management. 2025. Available online: https://clinicaltrials.gov/study/NCT07130149 (accessed on 15 October 2025).
- Universität Münster. A Phase 1 Single Center Clinical Trial Evaluating Safety of 5-Aminolevulinic Acid (5-ALA) Combined with CV01 Delivery of Ultrasound for Sonodynamic Therapy (SDT) in Patients with Newly Diagnosed High-Grade Glioma (HGG) Prior to Resection and Standard Adjuvant Therapy. 2025. Available online: https://clinicaltrials.gov/study/NCT06665724 (accessed on 15 October 2025).
| EVs Origin | Engineering Strategy | Target Tumor Model | Sonodynamic Treatment | Therapeutic Outcome | Ref. |
|---|---|---|---|---|---|
| Lymphocyte cell line (IST-EBV-TW6B) | Sensitizer: ZnO NCs; Ligand: anti-CD20; Method: Freeze-thaw loading and surface modification. | Burkitt’s lymphoma Daudi cells (CCL213) | Shock waves: 250 shots of 12.5 MPa, 4 shots/s, 3 times/day, every 3 h (in vitro) | Effect: Selective CD20+ cytotoxicity; Outcome: Significant on-demand cancer cell death. | [149] |
| Human embryonic kidney HEK-293T cells | Sensitizer: ICG; Ligand: Folic acid; Method: Surface functionalization and cargo loading. | Breast cancer cells (MCF-7) and human dermal fibroblasts (h-DFB) in vitro; MCF-7 xenografts in vivo | US: 0.3 W/cm2, 1 MHz, 60/70 s; 0.5 W/cm2, 1 MHz, 3 min | Effect: Selective tumor accumulation; Outcome: Effective suppression of tumor growth. | [150] |
| Human embryonic kidney HEK-293T cells | Sensitizer: ICG; Cargo: PTX + sodium bicarbonate; Method: Combined nanocarrier encapsulation. | Breast cancer cells (MCF-7) in vitro, MCF-7 xenografts in vivo | US: 0.3 W/cm2, 1 MHz, 1 min (in vitro) 0.5 W/cm2, 1 MHz, 3 min | Effect: Facilitated cytoplasmic drug release; Outcome: Significant inhibition of proliferation. | [151] |
| Human reticulum cell sarcoma cells (J774A.1) | Cargo: CAT-Silica NPs; Ligand: AS1411 aptamer; Method: Aptamer-decorated exosome embedding. | Human keratinocyte (HaCat), endothelial (bEnd 3) and GBM (U87) cells (in vitro); luciferase-expressing U87 tumors (in vivo) | US: 1.5 W/cm2, 1 MHz, 40% duty cycle (in vivo) | Effect: Growth inhibition; Outcome: Reduced metastasis and primary tumor volume. | [152] |
| Murine colon adenocarcinoma cells (CT26) | Sensitizer: Curcumin; Cargo: Calcium carbonate NPs; Method: Exosomal co-encapsulation. | Colon adenocarcinoma cells (CT26) in vitro; CT26 subcutaneous tumor model (in vivo) | US: 1.5 W/cm2, 1 MHz, 30% duty cycle, 10 min (in vivo) | Effect: pH-responsive/SDT synergy; Outcome: Robust tumor suppressor effect | [153] |
| Human embryonic kidney HEK-293T cells | Sensitizer: 4-(1,2,2-triphenylvinyl)-1,1′-biphenyl (TPE)-IR820; Ligand: Biotin; Method: Loading into biotin-conjugated exosomes | Human breast cancer cells (MCF-7) in vitro; MCF-7 xenografts in vivo | US: 0.5 W/cm2, 1 MHz, 2 min (in vitro and in vivo) | Effect: Sono-PDT synergy; Outcome: Maximized tumor targeting and growth inhibition. | [154] |
| EVs Origin | Engineering Strategy | Target Tumor Model | Sonodynamic Treatment | TME Modulation | Ref |
|---|---|---|---|---|---|
| Human embryonic kidney HEK-293T cells | Sensitizer: Ce6-TPP (mitochondria-targeted); Cargo: Piperlongumine; Method: Co-encapsulation. | Human breast cancer cells (MCF-7) and human dermal fibroblasts (h-DFB) in vitro; MCF-7 xenografts in vivo | US: 0.3 W/cm2, 1 MHz, 1 min (in vitro) 0.3 W/cm2, 1 MHz, 3 min (in vivo) | Mitochondrial oxidative stress: selective and maximal cytotoxic destruction of cancer cells | [155] |
| Human embryonic kidney HEK-293T cells | Sensitizer: Ce6-TPP; Cargo: Glycolysis inhibitors; Lipids: GSH-responsive diselenide-bearing | Human breast cancer cells (MCF-7) in vitro; MCF-7 xenografts in vivo | US: 0.5 W/cm2, 1 MHz, 2 min (in vitro) 0.5 W/cm2, 1 MHz, 3 min (in vivo) | Glycolysis inhibition: energy depletion and oxidative stress-induced tumor death | [156] |
| Human embryonic kidney HEK-293T cells | Sensitizer: Ce6; Adjuvant: R848 (TLR7/8 agonist); Method: Cargo loading. | Murine dendritic cells (DC2.4), murine macrophage cells (RAW 264.7) in vitro; murine embryonic prostate cells (RM-1) tumors in vivo | US: 0.1/0.5/1 W/cm2, 1 MHz, 20% duty cycle, 5 min (in vitro) 2 W/cm2, 1 MHz, 20% duty cycle, 5 min (in vivo) | Immune remodeling (Cold-to-Hot): macrophage repolarization, DC maturation, and T-cell activation. | [157] |
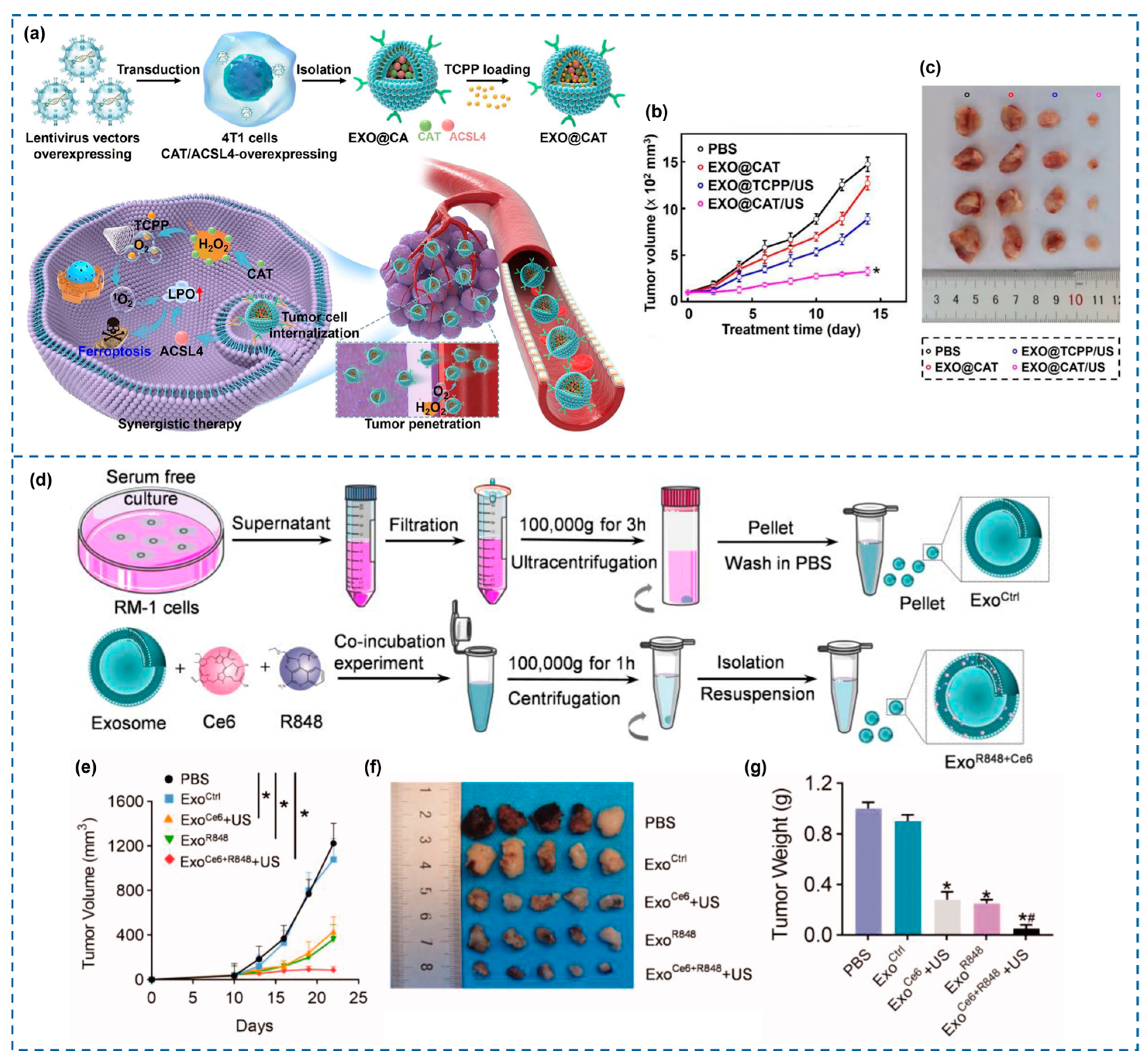
| Transfected murine breast cancer cells (4T1) | Sensitizer: TCPP (tetrakis (4-carboxyphenyl) porphyrin); EV Modification: CAT/Acyl-CoA enrichment via transfection. | Murine breast cancer cells (4T1) in vitro; | US: 1.5 W/cm2, 1 MHz, 3 min (in vitro and in vivo) | Hypoxia alleviation: O2 production-driven nanoconstruct motion; metastatic modulation | [30] |
| ID Trial | Tumor Type | Drug Used with SDT | Enrolled Patients | Ref. |
|---|---|---|---|---|
| ChiCTR2200065992 | Brainstem gliomas | Hematoporphyrin (Xipofen) | 11 | [69] |
| NCT06039709 | GBM | (5-ALA) | 11 (est.) | [168] |
| NCT05123534 | Diffuse Intrinsic Pontine Glioma and Diffuse Midline Glioma | Proprietary intravenous formulation of 5-ALA (SONALA-001) | 15 | [169] |
| NCT07130149 | GBM | Hiporfirin® | 230 (est.) | [170] |
| NCT06665724 | Recurrent High Grade Glioma | 5-aminolevulinic acid hydrochloride (5-ALA HCl, Gliolan®) | 14 (est.) | [171] |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license.
Share and Cite
Chiabotto, G.; Conte, M.; Cauda, V. Exosome-Enhanced Sonodynamic Therapy in Cancer: Emerging Synergies and Modulation of the Tumor Microenvironment. Cancers 2026, 18, 118. https://doi.org/10.3390/cancers18010118
Chiabotto G, Conte M, Cauda V. Exosome-Enhanced Sonodynamic Therapy in Cancer: Emerging Synergies and Modulation of the Tumor Microenvironment. Cancers. 2026; 18(1):118. https://doi.org/10.3390/cancers18010118
Chicago/Turabian StyleChiabotto, Giulia, Marzia Conte, and Valentina Cauda. 2026. "Exosome-Enhanced Sonodynamic Therapy in Cancer: Emerging Synergies and Modulation of the Tumor Microenvironment" Cancers 18, no. 1: 118. https://doi.org/10.3390/cancers18010118
APA StyleChiabotto, G., Conte, M., & Cauda, V. (2026). Exosome-Enhanced Sonodynamic Therapy in Cancer: Emerging Synergies and Modulation of the Tumor Microenvironment. Cancers, 18(1), 118. https://doi.org/10.3390/cancers18010118







