Core Patient-Centered Outcomes for Adolescents and Young Adults with Cancer: A Comprehensive Review of the Literature from the STRONG-AYA Project
Simple Summary
Abstract
1. Introduction
2. Materials and Methods
2.1. Search Strategy
2.2. Study Selection
2.3. Data Extraction
2.4. Quality Assurance
3. Results
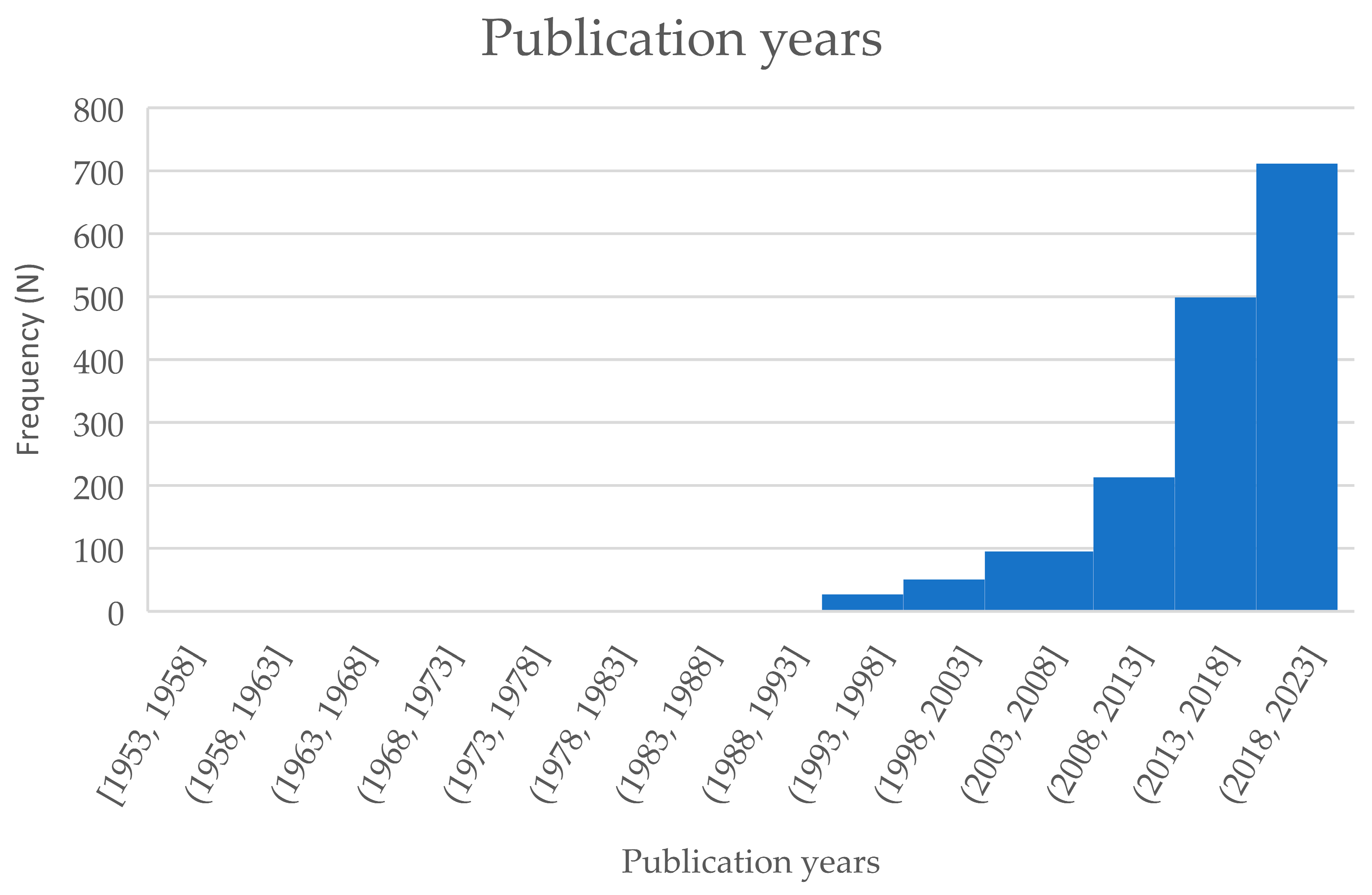
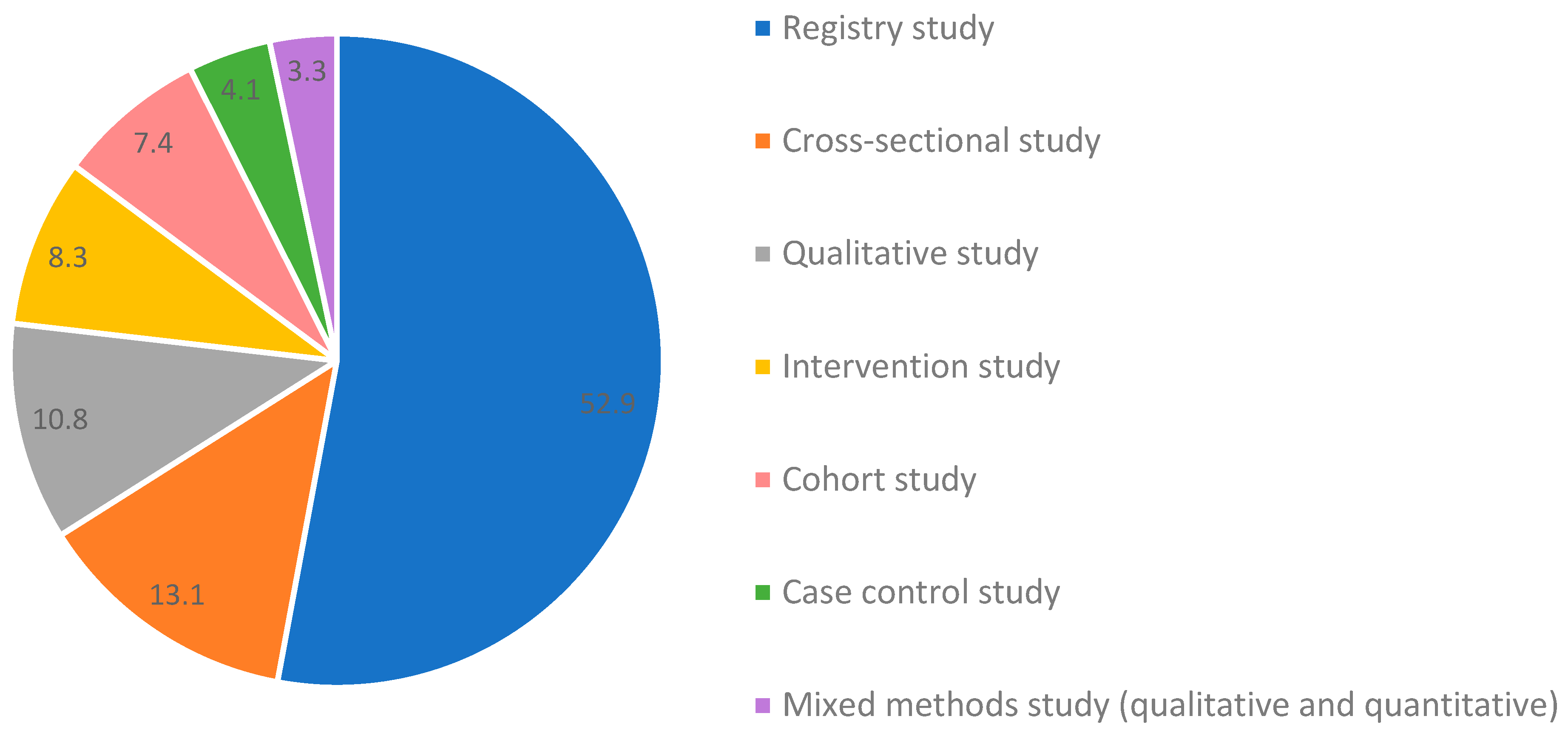
3.1. Study Characteristics
3.2. Outcomes, Measurement Tools, and Case-Mix Factors
3.2.1. Outcomes
3.2.2. Measurement Tools
3.2.3. Case-Mix Factor(s)
4. Discussion
4.1. Future Perspectives
4.2. Strengths and Limitations
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
Abbreviations
| AYA | Adolescent and young adult |
| COS | Core outcome set |
| COMET | Core Outcome Measures in Effectiveness Trials |
| ICHOM | International Consortium for Health Outcomes Measurement |
| PRISMA | Preferred Reporting Items for Systematic Reviews and Meta-Analyses |
| MeSH | Medical Subject Headings |
| Nr | Number |
| SES | Social economic status |
| GvHD | Graft versus host disease |
| HIV | Human immunodeficiency virus |
| HPIs | Helicobacter pylori infections |
| EBV | Epstein–Barr virus |
| HPV | Human papilloma virus |
| COPD | Chronic obstructive pulmonary disease |
| SEER | Surveillance, Epidemiology, and End Results |
| PROMs | Patient-reported outcome measures |
| HADS | Hospital Anxiety and Depression Scale |
| PedsQL | Pediatric Quality of Life Inventory |
| EORTC QLQ-C30 | European Organization for Research and Treatment of Cancer Quality of Life Group C30 |
| BSI | Brief Symptom Inventory |
| SF | Short Form Survey |
| COVID-19 | Corona virus disease |
Appendix A. Search String for the Literature Review per Database
Appendix B. In- and Exclusion Criteria
- Population: AYAs (13 up until (and including) 40 years old at initial cancer diagnosis) or a subset of this age range (e.g., adolescents OR young adults only). The AYA age range will be flexibly applied, because lower and upper age limits for AYAs differ per country or per study;
- Mixed samples will be included if age-stratified outcomes are available for the target population (i.e., AYAs; adolescents; young adults);
- Studies conducted in other study populations, such as healthcare providers, friends, parents, or carers of AYAs, will be included only if the participants provide information on the outcomes of AYAs;
- Study population at study participation: on and/or off treatment, including at diagnosis, during treatment, or following treatment (patients and/or survivors). There is no limit for the time since diagnosis for AYA cancer survivors. Patients on maintenance treatment will be included;
- Written in English language;
- Any type of malignant tumor;
- Study designs: prospective intervention studies, randomized controlled trials (RCTs), observational cohort studies, case–control studies, cross-sectional studies, qualitative studies, registry studies, mixed-methods studies (qualitative and quantitative methods);
- Studies focusing on all types of biological, physical, psychological, or social outcomes.
- Non-English articles;
- Full text is unavailable;
- Conference abstracts or posters;
- Article focusing on non-malignant tumor type(s);
- Non-human study;
- Study population consisting exclusively of adolescent and young adult participants without a cancer diagnosis;
- Articles describing study protocols, case reports/series, reviews/meta-analyses, expert opinions, theoretical papers, policy documents/guidelines, consensus letters, editorials;
- Study population consisting exclusively of childhood cancer patients aged under 13 years at initial cancer diagnosis and/or adult cancer patients aged over 40 years at initial cancer diagnosis;
- Study population consisting of childhood and AYA cancer patients OR AYA and adult cancer patients with NO age-stratified outcomes available (i.e., AYA-specific outcomes cannot be identified);
- Articles focusing on outcomes not of interest (such as solely focusing on the outcome incidence).
References
- Janssen, S.H.; van der Graaf, W.T.; van der Meer, D.J.; Manten-Horst, E.; Husson, O. Adolescent and young adult (AYA) cancer survivorship practices: An overview. Cancers 2021, 13, 4847. [Google Scholar] [CrossRef] [PubMed]
- Darlington, A.-S.E.; Wakefield, C.E.; van Erp, L.M.; van der Graaf, W.T.; Cohn, R.J.; Grootenhuis, M.A. Psychosocial consequences of surviving cancer diagnosed and treated in childhood versus in adolescence/young adulthood: A call for clearer delineation between groups. Cancer 2022, 128, 2690–2694. [Google Scholar] [CrossRef]
- Keegan, T.H.; Ries, L.A.; Barr, R.D.; Geiger, A.M.; Dahlke, D.V.; Pollock, B.H.; Bleyer, W.A.; National Cancer Institute Next Steps for Adolescent and Young Adult Oncology Epidemiology Working Group. Comparison of cancer survival trends in the United States of adolescents and young adults with those in children and older adults. Cancer 2016, 122, 1009–1016. [Google Scholar] [CrossRef] [PubMed]
- Zebrack, B.J. Psychological, social, and behavioral issues for young adults with cancer. Cancer 2011, 117, 2289–2294. [Google Scholar] [CrossRef]
- Sodergren, S.C.; Husson, O.; Rohde, G.E.; Tomaszewska, I.M.; Vivat, B.; Yarom, N.; Griffiths, H.; Darlington, A.-S.; On Behalf of the European Organization for Research and Treatment of Cancer Quality of Life Group. A life put on pause: An exploration of the health-related quality of life issues relevant to adolescents and young adults with cancer. J. Adolesc. Young Adult Oncol. 2018, 7, 453–464. [Google Scholar] [CrossRef] [PubMed]
- Hammond, C. Against a singular message of distinctness: Challenging dominant representations of adolescents and young adults in oncology. J. Adolesc. Young Adult Oncol. 2017, 6, 45–49. [Google Scholar] [CrossRef] [PubMed]
- Husson, O.; Zebrack, B.J.; Block, R.; Embry, L.; Aguilar, C.; Hayes-Lattin, B.; Cole, S. Health-related quality of life in adolescent and young adult patients with cancer: A longitudinal study. J. Clin. Oncol. 2017, 35, 652–659. [Google Scholar] [CrossRef] [PubMed]
- Abdelhadi, O.A.; Joseph, J.; Pollock, B.H.; Keegan, T.H. Additional medical costs of chronic conditions among adolescent and young adult cancer survivors. J. Cancer Surviv. 2021, 16, 487–496. [Google Scholar] [CrossRef]
- Mellblom, A.; Kiserud, C.E.; Rueegg, C.S.; Ruud, E.; Loge, J.H.; Fosså, S.D.; Lie, H.C. Self-reported late effects and long-term follow-up care among 1889 long-term Norwegian Childhood, Adolescent, and Young Adult Cancer Survivors (the NOR-CAYACS study). Support. Care Cancer 2021, 29, 2947–2957. [Google Scholar] [CrossRef]
- Hughes, T.; Harper, A.; Gupta, S.; Frazier, A.L.; van der Graaf, W.T.; Moreno, F.; Joseph, A.; Fidler-Benaoudia, M.M. The current and future global burden of cancer among adolescents and young adults: A population-based study. Lancet Oncol. 2024, 25, 1614–1624. [Google Scholar] [CrossRef] [PubMed]
- Williamson, P.R.; Altman, D.G.; Blazeby, J.M.; Clarke, M.; Devane, D.; Gargon, E.; Tugwell, P. Developing core outcome sets for clinical trials: Issues to consider. Trials 2012, 13, 1–8. [Google Scholar] [CrossRef] [PubMed]
- Husson, O.; Reeve, B.B.; Darlington, A.-S.; Cheung, C.K.; Sodergren, S.; Van Der Graaf, W.T.; Salsman, J.M. Next step for global adolescent and young adult oncology: A core patient-centered outcome set. JNCI J. Natl. Cancer Inst. 2022, 114, 496–502. [Google Scholar] [CrossRef]
- Ramsey, I.; Eckert, M.; Hutchinson, A.D.; Marker, J.; Corsini, N. Core outcome sets in cancer and their approaches to identifying and selecting patient-reported outcome measures: A systematic review. J. Patient-Rep. Outcomes 2020, 4, 1–12. [Google Scholar] [CrossRef]
- STRONGAYA. Available online: https://strongaya.eu/ (accessed on 11 January 2024).
- Williamson, P.R.; Altman, D.G.; Bagley, H.; Barnes, K.L.; Blazeby, J.M.; Brookes, S.T.; Clarke, M.; Gargon, E.; Gorst, S.; Harman, N. The COMET handbook: Version 1.0. Trials 2017, 18, 1–50. [Google Scholar] [CrossRef] [PubMed]
- ICHOM. Patient-Centered Outcome Measures. 2023. Available online: https://www.ichom.org/patient-centered-outcome-measures/ (accessed on 19 December 2023).
- Husson, O.; Janssen, S.H.; Reeve, B.B.; Sodergren, S.C.; Cheung, C.K.; McCabe, M.G.; Salsman, J.M.; van der Graaf, W.T.; Darlington, A.-S. Protocol for the development of a Core Outcome Set (COS) for Adolescents and Young Adults (AYAs) with cancer. BMC Cancer 2024, 24, 126. [Google Scholar] [CrossRef] [PubMed]
- Moher, D.; Shamseer, L.; Clarke, M.; Ghersi, D.; Liberati, A.; Petticrew, M.; Shekelle, P.; Stewart, L.A.; Group, P.-P. Preferred reporting items for systematic review and meta-analysis protocols (PRISMA-P) 2015 statement. Syst. Rev. 2015, 4, 1–9. [Google Scholar] [CrossRef] [PubMed]
- COMET. Core Outcome Set (COS) for Adolescents and Young Adults (AYAs) with Cancer. Available online: https://www.comet-initiative.org/Studies/Details/2663 (accessed on 19 December 2023).
- Bramer, W.M.; De Jonge, G.B.; Rethlefsen, M.L.; Mast, F.; Kleijnen, J. A systematic approach to searching: An efficient and complete method to develop literature searches. J. Med. Libr. Assoc. JMLA 2018, 106, 531. [Google Scholar] [CrossRef] [PubMed]
- Bramer, W.M.; Giustini, D.; de Jonge, G.B.; Holland, L.; Bekhuis, T. De-duplication of database search results for systematic reviews in EndNote. J. Med. Libr. Assoc. JMLA 2016, 104, 240. [Google Scholar] [CrossRef]
- Ouzzani, M.; Hammady, H.; Fedorowicz, Z.; Elmagarmid, A. Rayyan—A web and mobile app for systematic reviews. Syst. Rev. 2016, 5, 1–10. [Google Scholar] [CrossRef] [PubMed]
- Dodd, S.; Clarke, M.; Becker, L.; Mavergames, C.; Fish, R.; Williamson, P.R. A taxonomy has been developed for outcomes in medical research to help improve knowledge discovery. J. Clin. Epidemiol. 2018, 96, 84–92. [Google Scholar] [CrossRef] [PubMed]
- van der Meer, D.J.; Karim-Kos, H.E.; van der Mark, M.; Aben, K.K.; Bijlsma, R.M.; Rijneveld, A.W.; van der Graaf, W.T.; Husson, O. Incidence, survival, and mortality trends of cancers diagnosed in adolescents and young adults (15–39 years): A population-based study in The Netherlands 1990–2016. Cancers 2020, 12, 3421. [Google Scholar] [CrossRef]
- Page, M.J.; McKenzie, J.E.; Bossuyt, P.M.; Boutron, I.; Hoffmann, T.C.; Mulrow, C.D.; Shamseer, L.; Tetzlaff, J.M.; Akl, E.A.; Brennan, S.E. The PRISMA 2020 statement: An updated guideline for reporting systematic reviews. Int. J. Surg. 2021, 88, 105906. [Google Scholar] [CrossRef] [PubMed]
- Sodergren, S.C.; Husson, O.; Rohde, G.E.; Tomaszewska, I.M.; Griffiths, H.; Pessing, A.; Yarom, N.; Hooker, L.; Din, A.; Darlington, A.S. Does age matter? A comparison of health-related quality of life issues of adolescents and young adults with cancer. Eur. J. Cancer Care 2018, 27, e12980. [Google Scholar] [CrossRef] [PubMed]
- Salsman, J.M.; Danhauer, S.C.; Moore, J.B.; Canzona, M.R.; Victorson, D.E.; Zebrack, B.J.; Reeve, B.B. Optimizing the measurement of health-related quality of life in adolescents and young adults with cancer. Cancer 2020, 126, 4818–4824. [Google Scholar] [CrossRef]
- Husson, O.; Sodergren, S.C.; Darlington, A.S. The importance of a collaborative health-related quality of life measurement strategy for adolescents and young adults with cancer. Cancer 2021, 127, 1712–1713. [Google Scholar] [CrossRef]
- Rothmund, M.; Pilz, M.J.; Schlosser, L.; Arraras, J.I.; Groenvold, M.; Holzner, B.; van Leeuwen, M.; Petersen, M.A.; Schmidt, H.; Young, T. Equipercentile equating of scores from common patient-reported outcome measures of physical function in patients with cancer. J. Clin. Epidemiol. 2024, 165, 111203. [Google Scholar] [CrossRef]

| Core Area | % * | Outcome Domain | % * | Outcome Categories (Descending Prevalence) |
|---|---|---|---|---|
| Epidemiology | 44.9 | Mortality/survival | 43.1 | Overall survival |
| Survival (specified and non-specified) | ||||
| Event-free survival | ||||
| Other: Mortality (specified and non-specified); Disease-free survival; Progression-free survival; Cancer-specific survival; Relative survival; Relapse-free survival; Recurrence-free survival; Cancer-specific mortality; Disease-specific death; Disease-specific survival; Metastasis-free survival; Standard mortality ratio; All-cause mortality; Treatment-related mortality; Excess mortality/conditional survival; Death hazard ratio; Overall mortality; Miscellaneous | ||||
| Incidence 1 | 8.2 | Cancer incidence | ||
| Prevalence | ||||
| Miscellaneous | ||||
| Physiological/clinical | 42.4 | Blood and lymphatic system outcomes | 1.8 | Blood markers |
| Hematologic diseases (specified and non-specified) | ||||
| Thrombosis | ||||
| Other: Lymphatic outcomes; Miscellaneous | ||||
| Cardiac outcomes | 1.0 | Cardiac diseases (specified and non-specified) | ||
| Incidence of cardiac diseases | ||||
| Miscellaneous | ||||
| Congenital, familial, and genetic outcomes | 5.3 | Gene expression/mutations | ||
| Family history | ||||
| Genetic testing | ||||
| Other: Miscellaneous | ||||
| Endocrine outcomes | 1.5 | Endocrine diseases (specified and non-specified) | ||
| Thyroid diseases | ||||
| Menopausal outcomes | ||||
| Other: Miscellaneous | ||||
| Ear and labyrinth outcomes | 0.2 | Hearing loss | ||
| Ototoxicity | ||||
| Eye outcomes | 0.2 | Diseases relating to vision (specified and non-specified) | ||
| Posterior scleral bowing | ||||
| Cataracts | ||||
| Gastrointestinal outcomes | 0.9 | Appetite | ||
| Constipation | ||||
| Mouth issues | ||||
| Other: Vomiting; Diarrhea; Swallowing; Dental problems; Gastrointestinal diseases (specified and non-specified); Miscellaneous | ||||
| General outcomes | 6.4 | Fatigue | ||
| Symptoms (specified and non-specified) | ||||
| Pain | ||||
| Other: Sleep; Nausea; Anthropometric outcomes; General health; Dyspnea; Dizziness; Miscellaneous | ||||
| Hepatobiliary outcomes | 0.4 | Cirrhosis | ||
| Chronic liver diseases | ||||
| Liver transplantation | ||||
| Other: Liver diseases (specified and non-specified); Miscellaneous | ||||
| Immune system outcomes | 1.2 | Graft versus host disease (GvHD) outcomes | ||
| Immune system diseases (specified and non-specified) | ||||
| Human immunodeficiency virus (HIV) | ||||
| Other: Miscellaneous | ||||
| Infection and infestation outcomes | 1.2 | Infections (specified and non-specified) | ||
| Pancreatitis | ||||
| Helicobacter pylori infections (HPIs) | ||||
| Other: Epstein–Barr virus (EBV); Human papilloma virus (HPV); Miscellaneous | ||||
| Injury and poisoning outcomes | 0.0 | |||
| Metabolism and nutrition outcomes | 0.9 | Diabetes mellitus | ||
| Metabolic outcomes (specified and non-specified) | ||||
| Dietary outcomes | ||||
| Other: Miscellaneous | ||||
| Musculoskeletal and connective tissue outcomes | 0.7 | Musculoskeletal outcomes (specified and non-specified) | ||
| Osteonecrosis | ||||
| Strength and flexibility outcomes | ||||
| Other: Miscellaneous | ||||
| Outcomes relating to neoplasms: benign, malignant, and unspecified (including cysts and polyps) | 22.7 | Stage | ||
| Tumor characteristics (specified and non-specified) | ||||
| Treatment characteristics (specified and non-specified) | ||||
| Other: Tumor type; Metastases; Disease status; Relapses; Recurrences; Location; Second malignancies; Time intervals; Symptoms (specified and non-specified); Subsequent malignant neoplasms; Miscellaneous | ||||
| Nervous system outcomes | 0.9 | Nervous system outcomes (specified and non-specified) | ||
| Stroke | ||||
| Seizure | ||||
| Other: Neurotoxicity; Miscellaneous | ||||
| Pregnancy, puerperium, and perinatal outcomes | 2.0 | Childbirth outcomes | ||
| Pregnancy outcomes (specified and non-specified) | ||||
| Baby/offspring characteristics (specified and non-specified) | ||||
| Other: Characteristics of the delivery (specified and non-specified); Maternal outcomes; Desire for children; Miscellaneous | ||||
| Renal and urinary outcomes | 0.9 | Renal diseases (specified and non-specified) | ||
| Treatment characteristics | ||||
| Urinary outcomes (specified and non-specified) | ||||
| Other: Miscellaneous | ||||
| Reproductive system and breast outcomes | 6.7 | Fertility (specified and non-specified) | ||
| Preservation | ||||
| Emotions | ||||
| Other: Information provision/counseling; Sexual health; Knowledge; Menstrual outcomes; Contraceptive use; Breast outcomes; Miscellaneous | ||||
| Psychiatric outcomes | 0.6 | Suicide | ||
| Psychiatric outcomes (specified and non-specified) | ||||
| Miscellaneous | ||||
| Respiratory, thoracic, and mediastinal outcomes | 1.2 | Respiratory diseases (specified and non-specified) | ||
| Shortness of breath | ||||
| Chronic obstructive pulmonary disease (COPD) | ||||
| Other: Asthma; Pulmonary fibrosis; Miscellaneous | ||||
| Skin and subcutaneous tissue outcomes | 0.1 | Skin changes | ||
| Vascular outcomes | 0.8 | Vascular outcomes (specified and non-specified) | ||
| Blood pressure | ||||
| Miscellaneous | ||||
| Functioning | 47.0 | Physical functioning | 9.7 | Physical health |
| Physical functioning | ||||
| Physical activity | ||||
| Other: Health behavior (specified and non-specified); Smoking; Sexual functioning; Alcohol; Drugs; Dietary behavior; Performance status; Appearance; Miscellaneous | ||||
| Social functioning | 9.7 | Social functioning | ||
| Social support | ||||
| Psychosocial health | ||||
| Other: Online support; Social involvement in care-related decisions; Social needs; Isolation; Disclosure; Miscellaneous | ||||
| Role functioning | 6.3 | Employment | ||
| School | ||||
| Role functioning | ||||
| Other: Partner outcomes; Functional well-being; Intimacy; Miscellaneous | ||||
| Emotional functioning/well-being | 19.2 | Distress | ||
| Mental health | ||||
| Depression | ||||
| Other: Anxiety; Emotional functioning; Concerns; Coping; Worry; Body image; Fear of cancer recurrence; Self-efficacy; (Post-traumatic) Growth; Post-traumatic stress; Hope; Needs; Stress; Mental support; Fear; Self-esteem; Satisfaction; Cancer-related meaning; Uncertainty; Loneliness; Identity; Resilience; Regret; Psychosocial experiences; Identity; Body image; Miscellaneous | ||||
| Cognitive functioning | 5.8 | Cognitive functioning | ||
| Spiritual | ||||
| Knowledge | ||||
| Other: Religious outcomes; Beliefs; Attitudes; Meaning of life; Cognitive health; Literacy; Goal setting; Concentration; Memory; Miscellaneous | ||||
| Global quality of life | 5.6 | Quality of life | ||
| Perceived health status | 3.1 | Cancer experience | ||
| Impact of cancer | ||||
| Health status | ||||
| Other: Future; Participation in life; Miscellaneous | ||||
| Delivery of care | 23.4 | Healthcare use | ||
| Needs | ||||
| Fertility-related care | ||||
| Other: Trial participation/availability; Care experiences; Care preferences; Time intervals; Acceptability; Characteristics of care center; Perceptions; Adherence; Palliative care; Communication outcomes; Satisfaction with care; Pediatric and/or/vs. adult care; Source of information; Attendance to care; Documentation of care; Compliance; Miscellaneous | ||||
| Personal circumstances | 4.5 | Financial burden | ||
| Insurance | ||||
| Expenditures | ||||
| Other: Financial distress; Benefits; Practical challenges; Living situation; Miscellaneous | ||||
| Resource use | 6.1 | Economic | 0.8 | Costs |
| Cost-driven behavior | ||||
| Miscellaneous | ||||
| Hospital | 2.3 | Hospitalizations | ||
| Length of stay | ||||
| Emergency department care | ||||
| Other: Finances; Miscellaneous | ||||
| Need for further intervention | 3.1 | Feasibility | ||
| Efficacy | ||||
| Validation | ||||
| Other: Safety; Accuracy; Evaluation; Reliability; Miscellaneous | ||||
| Societal/carer burden | 0.0 | |||
| Adverse events | 4.5 | Adverse events | 4.5 | Toxicity |
| Complications (specified and non-specified) | ||||
| Adverse events (specified and non-specified) | ||||
| Other: Morbidity; Miscellaneous |
| Nr. | Case-Mix Factors (Examples) | N | % * |
|---|---|---|---|
| 1 | Age (age at diagnosis; age at study participation) | 897 | 74.7 |
| 2 | Sex (sex; gender) | 711 | 59.2 |
| 3 | Malignant (sub)types (tumor types; histological subtypes; date of diagnosis) | 667 | 55.5 |
| 4 | Treatment (type of treatment; number of treatments; treatment dosage; date of treatment) | 488 | 40.6 |
| 5 | Stage (stage; TNM classification; tumor size; metastasis; prognosis) | 473 | 39.4 |
| 6 | Ethnicity (race; migration background; ethnicity) | 314 | 26.1 |
| 7 | Socioeconomic status (SES) (socioeconomic position; median income) | 190 | 15.8 |
| 8 | Education (educational level; current or highest grade in school; change in school life) | 146 | 12.2 |
| 9 | Partner (partner status; marital status; sexual orientation) | 140 | 11.7 |
| 10 | Delivery of care (type of treatment center; location of center; patient provider communication; healthcare access; unmet service needs) | 87 | 7.2 |
| 11 | Insurance (insurance status; insured by Medicaid; insurance provider) | 85 | 7.1 |
| 12 | Comorbidities (pre-existing and not effects of treatment) (comorbidities; pre-treatment mental health diagnosis) | 80 | 6.7 |
| 13 | Geographical area/residence (urban/rural residence; (place of) residence; distance to care center) | 81 | 6.7 |
| 14 | Symptoms (symptoms, complaints; duration of symptoms; complications; late effects) | 74 | 6.2 |
| 15 | Genetics (family history of cancer; cytogenetic/molecular genetic characteristics; genetic mutations) | 66 | 5.5 |
| 16 | Tumor markers (white blood cell count; serum levels; expression of tumor markers) | 61 | 5.1 |
| 17 | Children (having children; caregiving for children at diagnosis) | 56 | 4.7 |
| 18 | Location (of tumor) (primary tumor site; tumor location) | 54 | 4.5 |
| 19 | Employment (being employed, type of work; change in work life) | 52 | 4.3 |
| 20 | Lifestyle (physical activity; sleep; alcohol intake; smoking status) | 50 | 4.2 |
| 21 | Health status (health status; performance status; functional status) | 39 | 3.2 |
| 22 | Maternity/fertility characteristics (ever been pregnant; maternal age; infertility; menarche status) | 35 | 2.9 |
| 23 | Anthropometric measures (obesity status; body mass index) | 32 | 2.7 |
| 24 | Psychological mechanisms (anxiety, depression; psychological outcomes; coping) | 33 | 2.7 |
| 25 | Secondary malignancies (second malignancies; history of cancer; stage of second malignancy) | 30 | 2.5 |
| 26 | Sociodemographic characteristics (living status; language; religion; personality; health literacy; family structure) | 26 | 2.2 |
| 27 | Recurrence/relapse (recurrence; relapse; site of recurrence; time since relapse) | 25 | 2.1 |
| 28 | Social support (social support; social factors; social desirability) | 17 | 1.4 |
| 29 | Registry characteristics (type of registry or cohort; survey year) | 11 | 0.9 |
| 30 | Virus/bacteria (HIV status; HPV status) | 10 | 0.8 |
| 31 | Trial participation (clinical/study trial participation; clinical trial enrollment) | 6 | 0.5 |
| 32 | COVID-19 characteristics (pre/post COVID-19 pandemic; adherence to COVID-19 precautions; COVID impact) | 4 | 0.3 |
| 33 | Miscellaneous (communication; stressful life event(s)) | 25 | 2.1 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Janssen, S.H.M.; van der Graaf, W.T.A.; Hurley-Wallace, A.; Vlooswijk, C.; Padilla, C.S.; Cairns, C.; Tyler, C.J.; Holthuis, E.I.; Rohde, G.E.; Hunt, K.J.; et al. Core Patient-Centered Outcomes for Adolescents and Young Adults with Cancer: A Comprehensive Review of the Literature from the STRONG-AYA Project. Cancers 2025, 17, 454. https://doi.org/10.3390/cancers17030454
Janssen SHM, van der Graaf WTA, Hurley-Wallace A, Vlooswijk C, Padilla CS, Cairns C, Tyler CJ, Holthuis EI, Rohde GE, Hunt KJ, et al. Core Patient-Centered Outcomes for Adolescents and Young Adults with Cancer: A Comprehensive Review of the Literature from the STRONG-AYA Project. Cancers. 2025; 17(3):454. https://doi.org/10.3390/cancers17030454
Chicago/Turabian StyleJanssen, Silvie H. M., Winette T. A. van der Graaf, Anna Hurley-Wallace, Carla Vlooswijk, Catarina S. Padilla, Charlotte Cairns, Connor J. Tyler, Emily I. Holthuis, Gudrun E. Rohde, Katherine J. Hunt, and et al. 2025. "Core Patient-Centered Outcomes for Adolescents and Young Adults with Cancer: A Comprehensive Review of the Literature from the STRONG-AYA Project" Cancers 17, no. 3: 454. https://doi.org/10.3390/cancers17030454
APA StyleJanssen, S. H. M., van der Graaf, W. T. A., Hurley-Wallace, A., Vlooswijk, C., Padilla, C. S., Cairns, C., Tyler, C. J., Holthuis, E. I., Rohde, G. E., Hunt, K. J., Way, K., Angoumis, K., Lankhorst, L. H., Engel, M. F. M., Rothmund, M.-S., Reuvers, M. J. P., Collaço, N., Wagland, R., Sodergren, S. C., ... Husson, O. (2025). Core Patient-Centered Outcomes for Adolescents and Young Adults with Cancer: A Comprehensive Review of the Literature from the STRONG-AYA Project. Cancers, 17(3), 454. https://doi.org/10.3390/cancers17030454







