Prognostic Impact of the AML60+ Score for Elderly Patients with Acute Myeloid Leukemia Treated with Hypomethylating Agents: A Retrospective Multicentric Analysis
Simple Summary
Abstract
1. Introduction
2. Materials and Methods
2.1. Study Cohort
2.2. Molecular Profiling
2.3. Prognostic Scoring Systems
2.4. Statistical Analysis
3. Results
3.1. Patient Population
3.2. Distribution of Risk Groups According to the Different Scores
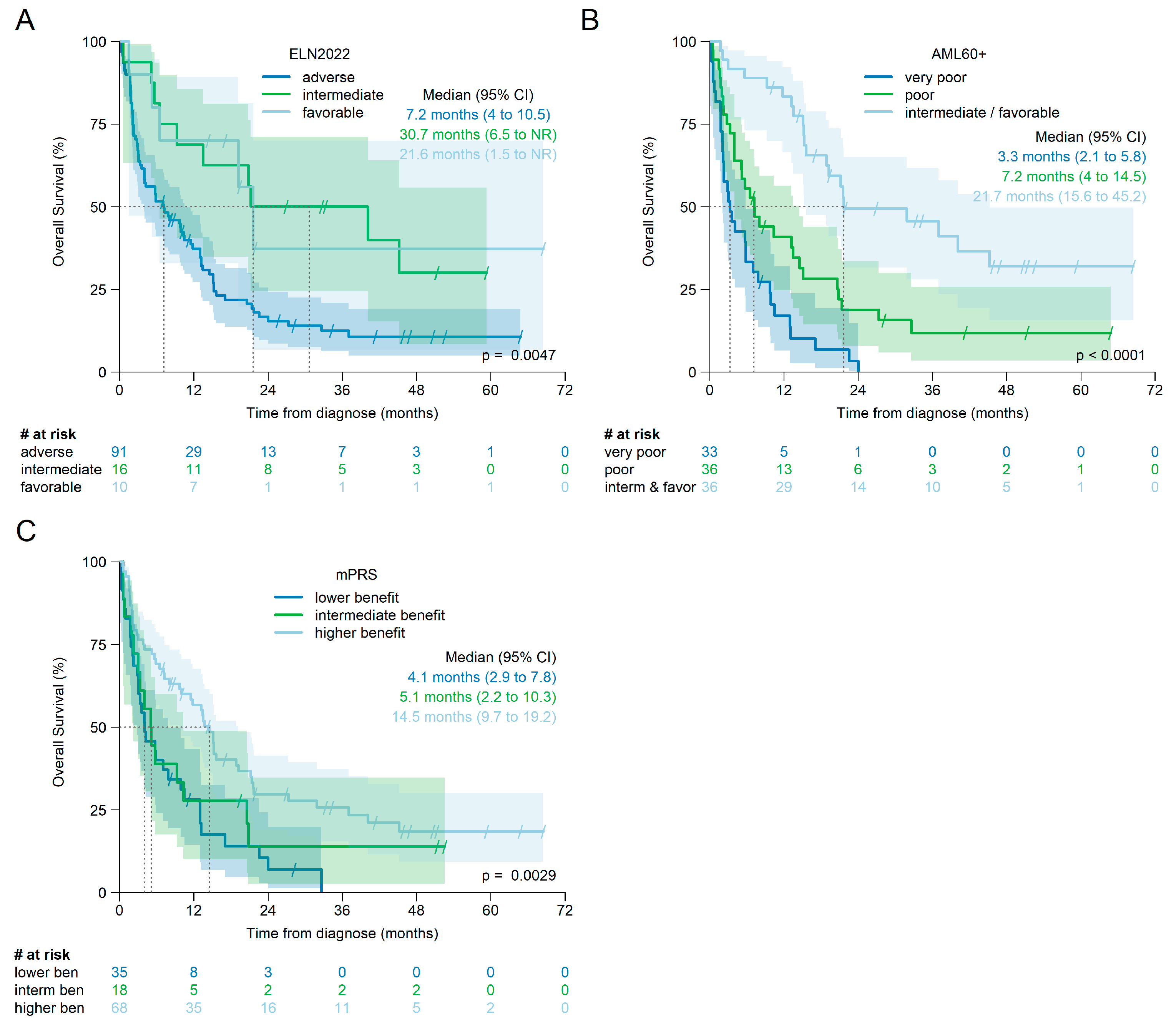
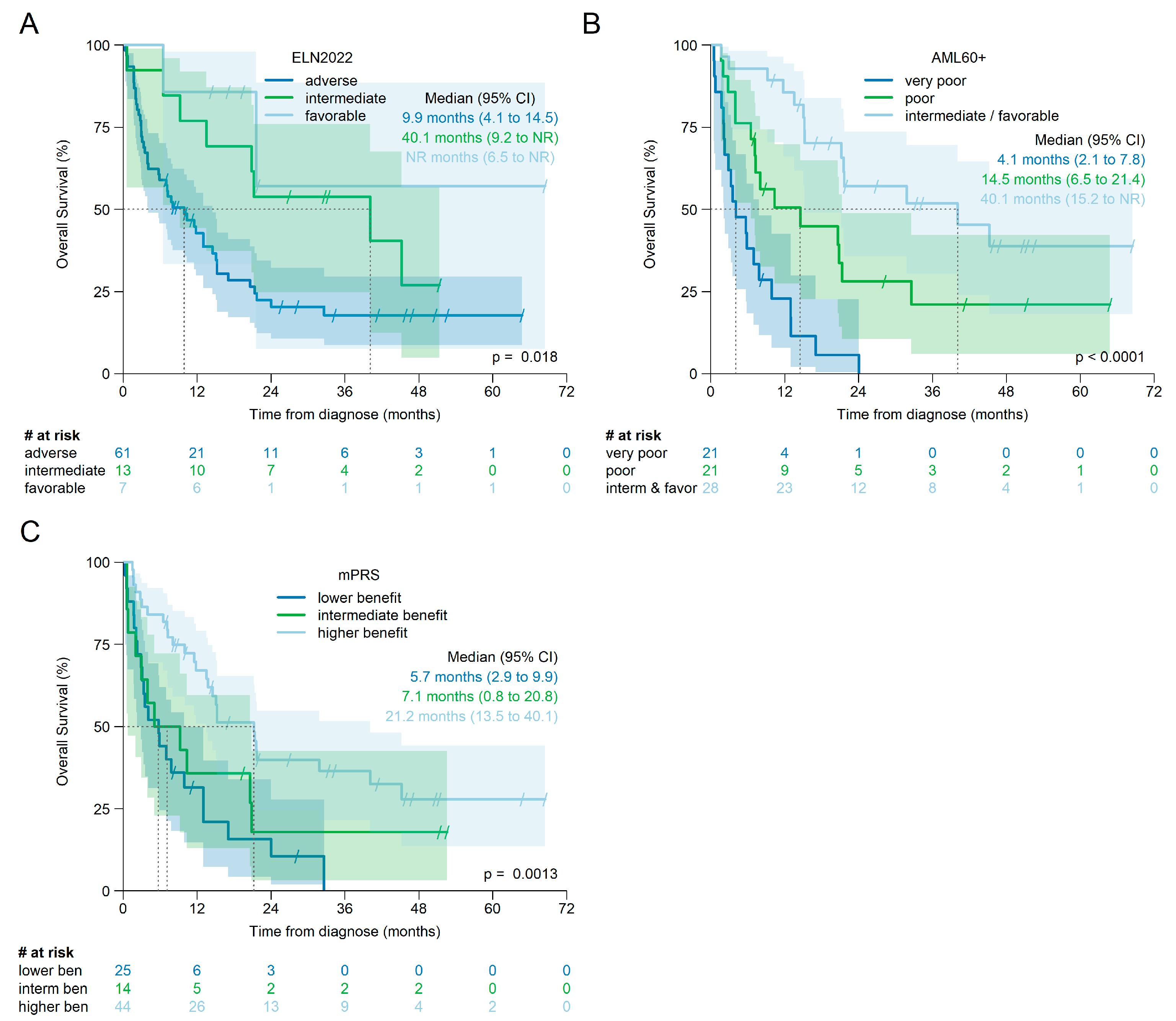
3.3. Risk Stratification of Patients Stratified According ELN2022 by AML60+
3.4. Risk Stratification of Patients Stratified According ELN2022 by mPRS
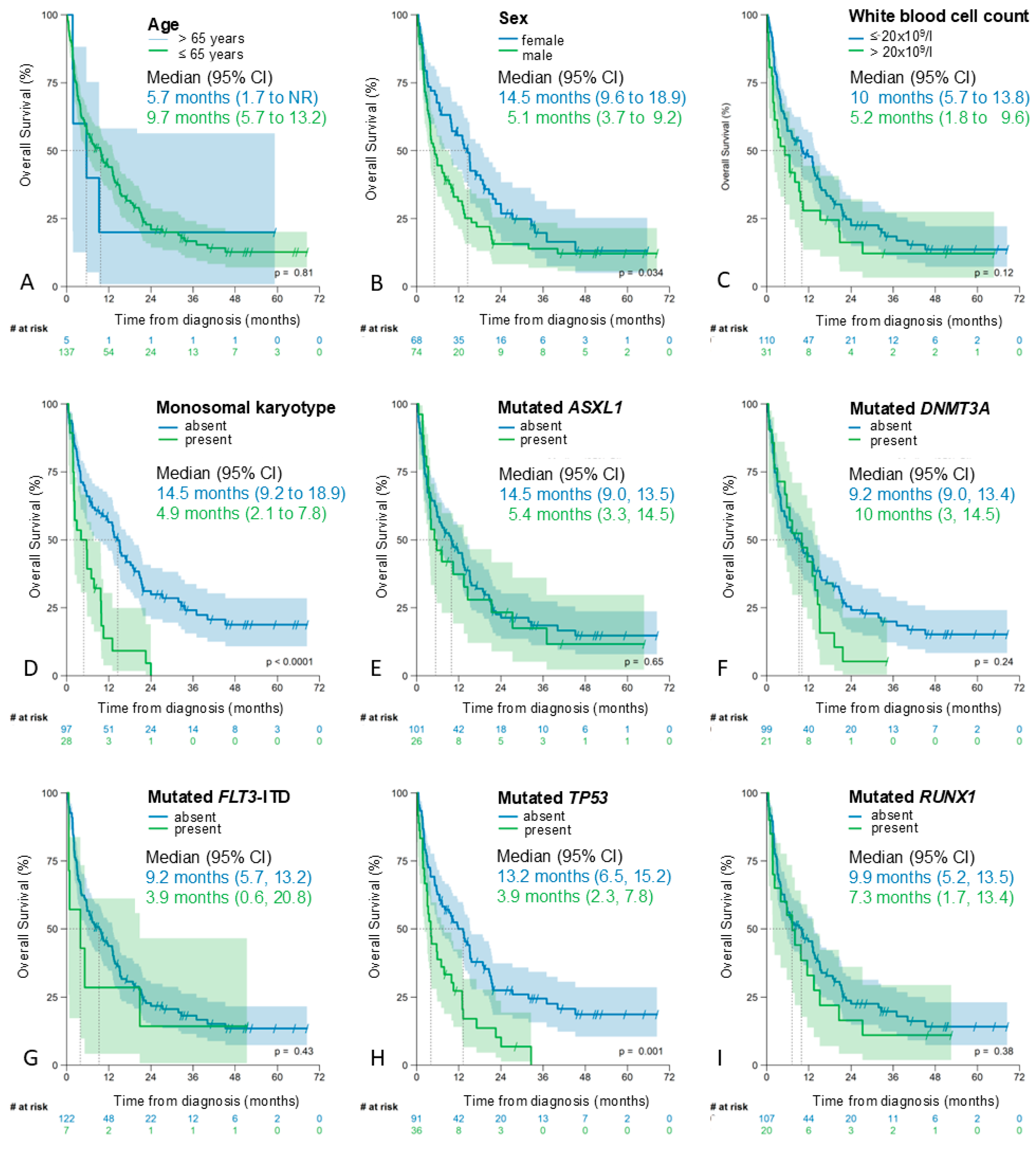
3.5. Prognostic Impact of the Single Risk Factors According to the AML60+ in Univariate Analysis
3.6. Frequency and Prognostic Impact of NPM1 and IDH2 Mutations in the Risk Groups According to AML60+
3.7. Prognosis According to ELN2022, mPRS, and AML60+
3.8. Comparison of the C-Indices
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
Abbreviations
| AML | Acute myeloid leukemia |
| ASXL | Additional Sex Combs-Like 1 |
| CI | Confidence interval |
| DNMT3A | DNA-Metyhltransferase 3A |
| Dec | Decitabine |
| ELN | European Leukemia Net |
| FLT3 | FMS-like Tyrosinkinase 3 |
| ICC | International Consensus Classification |
| HR | Hazard ratio |
| HMA | Hypomethylating agent |
| IDH | Isocitrate Dehydrogenase |
| ITD | Internal Tandem |
| IQR | Inter Quartile range |
| mPRS | Molecular Prognostic Score |
| NHS | National Health Service |
| NPM | Nucleophosmin 1 |
| OS | Overall survival |
| RAS | Rat Sarcoma |
| TP53 | Tumor Protein p53 |
| RUNX1 | Runt-related transcription factor 1 |
| Ven | Venetoclax |
| WBC | White blood cell count |
| WHO | World Health Organization |
References
- DiNardo, C.D.; Erba, H.P.; Freeman, S.D.; Wei, A.H. Acute Myeloid Leukaemia. Lancet 2023, 401, 2073–2086. [Google Scholar] [CrossRef] [PubMed]
- Roman, E.; Smith, A.; Appleton, S.; Crouch, S.; Kelly, R.; Kinsey, S.; Cargo, C.; Patmore, R. Myeloid Malignancies in the Real-World: Occurrence, Progression and Survival in the UK’s Population-Based Haematological Malignancy Research Network 2004–15. Cancer Epidemiol. 2016, 42, 186–198. [Google Scholar] [CrossRef]
- Döhner, H.; Wei, A.H.; Appelbaum, F.R.; Craddock, C.; DiNardo, C.D.; Dombret, H.; Ebert, B.L.; Fenaux, P.; Godley, L.A.; Hasserjian, R.P.; et al. Diagnosis and Management of AML in Adults: 2022 Recommendations from an International Expert Panel on Behalf of the ELN. Blood 2022, 140, 1345–1377. [Google Scholar] [CrossRef] [PubMed]
- Shimony, S.; Stahl, M.; Stone, R.M. Acute Myeloid Leukemia: 2025 Update on Diagnosis, Risk-Stratification, and Management. Am. J. Hematol. 2025, 100, 860–891. [Google Scholar] [CrossRef]
- Ucciero, A.; Pagnoni, F.; Scotti, L.; Pisterna, A.; Barone-Adesi, F.; Gaidano, G.; Patriarca, A.; Lunghi, M. Venetoclax with Hypomethylating Agents in Newly Diagnosed Acute Myeloid Leukemia: A Systematic Review and Meta-Analysis of Survival Data from Real-World Studies. Cancers 2023, 15, 4618. [Google Scholar] [CrossRef]
- Wang, Y.; Chen, Y.; Ji, D.; Ge, L.; Zhang, Y.; Liu, L.; Jiang, L.; Jin, F.; Xia, L. Meta-Analysis on the Effectiveness and Safety of Venetoclax-Based Combination Therapy with Hypomethylation in Acute Myeloid Leukemia. Eur. J. Med. Res. 2025, 30, 330. [Google Scholar] [CrossRef] [PubMed]
- Madarang, E.; Lykon, J.; Zhao, W.; Sekeres, M.A.; Bradley, T.; Chandhok, N.S.; Taylor, J.; Venugopal, S.; Koru-Sengul, T.; Iyer, S.G.; et al. Venetoclax and Hypomethylating Agents in Octogenarians and Nonagenarians with Acute Myeloid Leukemia. Blood Neoplasia 2024, 1, 100016. [Google Scholar] [CrossRef]
- Wei, A.H.; Loo, S.; Daver, N. How I Treat Patients with AML Using Azacitidine and Venetoclax. Blood 2025, 145, 1237–1250. [Google Scholar] [CrossRef]
- Pratz, K.W.; Jonas, B.A.; Pullarkat, V.; Thirman, M.J.; Garcia, J.S.; Döhner, H.; Récher, C.; Fiedler, W.; Yamamoto, K.; Wang, J.; et al. Long-Term Follow-up of VIALE-A: Venetoclax and Azacitidine in Chemotherapy-Ineligible Untreated Acute Myeloid Leukemia. Am. J. Hematol. 2024, 99, 615–624. [Google Scholar] [CrossRef]
- Niscola, P.; Gianfelici, V.; Giovannini, M.; Piccioni, D.; Mazzone, C.; de Fabritiis, P. Very Long-Term Efficacy of Venetoclax Combined with Hypomethylating Agents in Two AML Elderly: Is It the Time for Treatment Discontinuation Strategies? Ann. Hematol. 2024, 103, 1787–1788. [Google Scholar] [CrossRef]
- Chua, C.C.; Hammond, D.; Kent, A.; Tiong, I.S.; Konopleva, M.Y.; Pollyea, D.A.; DiNardo, C.D.; Wei, A.H. Treatment-Free Remission after Ceasing Venetoclax-Based Therapy in Patients with Acute Myeloid Leukemia. Blood Adv. 2022, 6, 3879–3883. [Google Scholar] [CrossRef]
- Garciaz, S.; Dumas, P.-Y.; Bertoli, S.; Sallman, D.A.; Decroocq, J.; Belhabri, A.; Orvain, C.; Aspas Requena, G.; Simand, C.; Laribi, K.; et al. Outcomes of Acute Myeloid Leukemia Patients Who Responded to Venetoclax and Azacitidine and Stopped Treatment. Am. J. Hematol. 2024, 99, 1870–1876. [Google Scholar] [CrossRef]
- Döhner, H.; Pratz, K.W.; DiNardo, C.D.; Wei, A.H.; Jonas, B.A.; Pullarkat, V.A.; Thirman, M.J.; Récher, C.; Schuh, A.C.; Babu, S.; et al. Genetic Risk Stratification and Outcomes among Treatment-Naive Patients with AML Treated with Venetoclax and Azacitidine. Blood 2024, 144, 2211–2222. [Google Scholar] [CrossRef]
- Bataller, A.; Bazinet, A.; DiNardo, C.D.; Maiti, A.; Borthakur, G.; Daver, N.G.; Short, N.J.; Jabbour, E.J.; Issa, G.C.; Pemmaraju, N.; et al. Prognostic Risk Signature in Patients with Acute Myeloid Leukemia Treated with Hypomethylating Agents and Venetoclax. Blood Adv. 2024, 8, 927–935. [Google Scholar] [CrossRef]
- Döhner, H.; DiNardo, C.D.; Appelbaum, F.R.; Craddock, C.; Dombret, H.; Ebert, B.L.; Fenaux, P.; Godley, L.A.; Hasserjian, R.P.; Larson, R.A.; et al. Genetic Risk Classification for Adults with AML Receiving Less-Intensive Therapies: The 2024 ELN Recommendations. Blood 2024, 144, 2169–2173. [Google Scholar] [CrossRef]
- Hoff, F.W.; Blum, W.G.; Huang, Y.; Welkie, R.L.; Swords, R.T.; Traer, E.; Stein, E.M.; Lin, T.L.; Archer, K.J.; Patel, P.A.; et al. Beat-AML 2024 ELN–Refined Risk Stratification for Older Adults with Newly Diagnosed AML given Lower-Intensity Therapy. Blood Adv. 2024, 8, 5297–5305. [Google Scholar] [CrossRef] [PubMed]
- Gangat, N.; Elbeih, A.; Ghosoun, N.; McCullough, K.; Aperna, F.; Johnson, I.M.; Abdelmagid, M.; Al-Kali, A.; Alkhateeb, H.B.; Begna, K.H.; et al. Mayo Genetic Risk Models for Newly Diagnosed Acute Myeloid Leukemia Treated with Venetoclax + Hypomethylating Agent. Am. J. Hematol. 2025, 100, 260–271. [Google Scholar] [CrossRef] [PubMed]
- Versluis, J.; Metzner, M.; Wang, A.; Gradowska, P.; Thomas, A.; Jakobsen, N.A.; Kennedy, A.; Moore, R.; Boertjes, E.; Vonk, C.M.; et al. Risk Stratification in Older Intensively Treated Patients With AML. J. Clin. Oncol. 2024, 42, 4084–4094. [Google Scholar] [CrossRef] [PubMed]
- Arber, D.A.; Orazi, A.; Hasserjian, R.P.; Borowitz, M.J.; Calvo, K.R.; Kvasnicka, H.-M.; Wang, S.A.; Bagg, A.; Barbui, T.; Branford, S.; et al. International Consensus Classification of Myeloid Neoplasms and Acute Leukemias: Integrating Morphologic, Clinical, and Genomic Data. Blood 2022, 140, 1200–1228. [Google Scholar] [CrossRef] [PubMed]
- Khoury, J.D.; Solary, E.; Abla, O.; Akkari, Y.; Alaggio, R.; Apperley, J.F.; Bejar, R.; Berti, E.; Busque, L.; Chan, J.K.C.; et al. The 5th Edition of the World Health Organization Classification of Haematolymphoid Tumours: Myeloid and Histiocytic/Dendritic Neoplasms. Leukemia 2022, 36, 1703–1719. [Google Scholar] [CrossRef]
- Huls, G.; Chitu, D.A.; Pabst, T.; Klein, S.K.; Stussi, G.; Griskevicius, L.; Valk, P.J.M.; Cloos, J.; van de Loosdrecht, A.A.; Breems, D.; et al. Ibrutinib Added to 10-Day Decitabine for Older Patients with AML and Higher Risk MDS. Blood Adv. 2020, 4, 4267–4277. [Google Scholar] [CrossRef]
- Griffioen, M.S.; de Leeuw, D.C.; Janssen, J.J.W.M.; Smit, L. Targeting Acute Myeloid Leukemia with Venetoclax; Biomarkers for Sensitivity and Rationale for Venetoclax-Based Combination Therapies. Cancers 2022, 14, 3456. [Google Scholar] [CrossRef]
- Lachowiez, C.A.; DiNardo, C.D. Mutation- and MRD-Informed Treatments for Transplant-Ineligible Patients. Hematol. Am. Soc. Hematol. Educ. Program. 2024, 2024, 168–177. [Google Scholar] [CrossRef]
- DeZern, A.E.; Zeidan, A.M.; Barnard, J.; Hand, W.; Al Ali, N.; Brown, F.; Zimmerman, C.; Roboz, G.J.; Garcia-Manero, G.; Steensma, D.P.; et al. Differential Response to Hypomethylating Agents Based on Sex: A Report on Behalf of the MDS Clinical Research Consortium (MDS CRC). Leuk. Lymphoma 2017, 58, 1325–1331. [Google Scholar] [CrossRef]
- Maggioni, G.; Bersanelli, M.; Travaglino, E.; Piérola, A.A.; Kasprzak, A.; Montserrat, A.S.; Sauta, E.; Sala, C.; Matteuzzi, T.; Meggendorfer, M.; et al. A Sex-Informed Approach to Improve the Personalised Decision Making Process in Myelodysplastic Syndromes: A Multicentre, Observational Cohort Study. Lancet Haematol. 2023, 10, e117–e128. [Google Scholar] [CrossRef] [PubMed]
- Hilberink, J.R.; van Zeventer, I.A.; Chitu, D.A.; Pabst, T.; Klein, S.K.; Stussi, G.; Griskevicius, L.; Valk, P.J.M.; Cloos, J.; van de Loosdrecht, A.A.; et al. Age and Sex Associate with Outcome in Older AML and High Risk MDS Patients Treated with 10-Day Decitabine. Blood Cancer J. 2023, 13, 93. [Google Scholar] [CrossRef] [PubMed]
- Ozga, M.; Nicolet, D.; Mrózek, K.; Yilmaz, A.S.; Kohlschmidt, J.; Larkin, K.T.; Blachly, J.S.; Oakes, C.C.; Buss, J.; Walker, C.J.; et al. Sex-Associated Differences in Frequencies and Prognostic Impact of Recurrent Genetic Alterations in Adult Acute Myeloid Leukemia (Alliance, AMLCG). Leukemia 2024, 38, 45–57. [Google Scholar] [CrossRef] [PubMed]
- Othman, J.; Lam, H.P.J.; Leong, S.; Basheer, F.; Abdallah, I.; Fleming, K.; Mehta, P.; Yassin, H.; Laurie, J.; Austin, M.; et al. Real-World Outcomes of Newly Diagnosed AML Treated with Venetoclax and Azacitidine or Low-Dose Cytarabine in the UK NHS. Blood Neoplasia 2024, 1, 100017. [Google Scholar] [CrossRef]
- Tettero, J.M.; Cloos, J.; Bullinger, L. Acute Myeloid Leukemia: Does Sex Matter? Leukemia 2024, 38, 2329–2331. [Google Scholar] [CrossRef]
- Shimony, S.; Bewersdorf, J.P.; Shallis, R.M.; Liu, Y.; Schaefer, E.J.; Zeidan, A.M.; Goldberg, A.D.; Stein, E.M.; Marcucci, G.; Lindsley, R.C.; et al. Hypomethylating Agents plus Venetoclax Compared with Intensive Induction Chemotherapy Regimens in Molecularly Defined Secondary AML. Leukemia 2024, 38, 762–768. [Google Scholar] [CrossRef]
- Ikoma, Y.; Nakamura, N.; Kaneda, Y.; Takamori, H.; Seki, T.; Hiramoto, N.; Kitagawa, J.; Kanda, J.; Fujita, K.; Morishita, T.; et al. Impact of Myelodysplasia-Related Gene Mutations and Residual Mutations at Remission in Venetoclax/Azacitidine for AML. Leukemia 2025, 39, 1–6. [Google Scholar] [CrossRef]
- Sasaki, K.; Ravandi, F.; Kadia, T.M.; Borthakur, G.; Short, N.J.; Jain, N.; Daver, N.G.; Jabbour, E.J.; Garcia-Manero, G.; Loghavi, S.; et al. Prediction of Survival with Lower Intensity Therapy among Older Patients with Acute Myeloid Leukemia. Cancer 2023, 129, 1017–1029. [Google Scholar] [CrossRef]
- Bewersdorf, J.P.; Zeidan, A.M. Hyperleukocytosis and Leukostasis in Acute Myeloid Leukemia: Can a Better Understanding of the Underlying Molecular Pathophysiology Lead to Novel Treatments? Cells 2020, 9, 2310. [Google Scholar] [CrossRef]
- Suárez, E.U.; Boluda, B.; Lavilla, E.; Tormo, M.; Botella, C.; Gil, C.; Vives, S.; Rodríguez, C.; Serrano, J.; Sayas, M.J.; et al. Do NPM1 and FLT3-ITD Mutations Modify Prognosis in Patients Treated with Non-Intensive Regimens? Ann. Hematol. 2024, 103, 2845–2851. [Google Scholar] [CrossRef] [PubMed]
- Chen, Y.; Cao, J.; Ye, Y.; Luo, L.; Zheng, X.; Yang, X.; Zheng, Z.; Zheng, J.; Yang, T.; Hu, J. Hypomethylating Agents Combined with Low-Dose Chemotherapy for Elderly Patients with Acute Myeloid Leukaemia Unfit for Intensive Chemotherapy: A Real-World Clinical Experience. J. Chemother. 2023, 35, 322–329. [Google Scholar] [CrossRef] [PubMed]
- Kiyoi, H.; Naoe, T.; Nakano, Y.; Yokota, S.; Minami, S.; Miyawaki, S.; Asou, N.; Kuriyama, K.; Jinnai, I.; Shimazaki, C.; et al. Prognostic Implication of FLT3 and N-RAS Gene Mutations in Acute Myeloid Leukemia. Blood 1999, 93, 3074–3080. [Google Scholar]
- Kottaridis, P.D.; Gale, R.E.; Frew, M.E.; Harrison, G.; Langabeer, S.E.; Belton, A.A.; Walker, H.; Wheatley, K.; Bowen, D.T.; Burnett, A.K.; et al. The Presence of a FLT3 Internal Tandem Duplication in Patients with Acute Myeloid Leukemia (AML) Adds Important Prognostic Information to Cytogenetic Risk Group and Response to the First Cycle of Chemotherapy: Analysis of 854 Patients from the United Kingdom Medical Research Council AML 10 and 12 Trials. Blood 2001, 98, 1752–1759. [Google Scholar] [CrossRef]
- Shahzad, M.; Amin, M.K.; Daver, N.G.; Shah, M.V.; Hiwase, D.; Arber, D.A.; Kharfan-Dabaja, M.A.; Badar, T. What Have We Learned about TP53-Mutated Acute Myeloid Leukemia? Blood Cancer J. 2024, 14, 202. [Google Scholar] [CrossRef]
- Senapati, J.; Loghavi, S.; Garcia-Manero, G.; Tang, G.; Kadia, T.; Short, N.J.; Abbas, H.A.; Arani, N.; DiNardo, C.D.; Borthakur, G.; et al. Clinical Interrogation of TP53 Aberrations and Its Impact on Survival in Patients with Myeloid Neoplasms. Haematologica 2025, 110, 1304–1315. [Google Scholar] [CrossRef]
- Badar, T.; Nanaa, A.; Atallah, E.; Shallis, R.M.; Guilherme, S.d.C.C.; Goldberg, A.D.; Saliba, A.N.; Patel, A.; Bewersdorf, J.P.; DuVall, A.S.; et al. Comparing Venetoclax in Combination with Hypomethylating Agents to Hypomethylating Agent-Based Therapies for Treatment Naive TP53-Mutated Acute Myeloid Leukemia: Results from the Consortium on Myeloid Malignancies and Neoplastic Diseases (COMMAND). Blood Cancer J. 2024, 14, 32. [Google Scholar] [CrossRef]
- Rücker, F.G.; Schlenk, R.F.; Bullinger, L.; Kayser, S.; Teleanu, V.; Kett, H.; Habdank, M.; Kugler, C.-M.; Holzmann, K.; Gaidzik, V.I.; et al. TP53 Alterations in Acute Myeloid Leukemia with Complex Karyotype Correlate with Specific Copy Number Alterations, Monosomal Karyotype, and Dismal Outcome. Blood 2012, 119, 2114–2121. [Google Scholar] [CrossRef]
- Kadia, T.M.; Jain, P.; Ravandi, F.; Garcia-Manero, G.; Andreef, M.; Takahashi, K.; Borthakur, G.; Jabbour, E.; Konopleva, M.; Daver, N.G.; et al. TP53 Mutations in Newly Diagnosed Acute Myeloid Leukemia: Clinicomolecular Characteristics, Response to Therapy, and Outcomes. Cancer 2016, 122, 3484–3491. [Google Scholar] [CrossRef] [PubMed]
- Pollyea, D.A.; Pratz, K.W.; Wei, A.H.; Pullarkat, V.; Jonas, B.A.; Recher, C.; Babu, S.; Schuh, A.C.; Dail, M.; Sun, Y.; et al. Outcomes in Patients with Poor-Risk Cytogenetics with or without TP53 Mutations Treated with Venetoclax and Azacitidine. Clin. Cancer Res. 2022, 28, 5272–5279. [Google Scholar] [CrossRef] [PubMed]
- Venugopal, S.; Maiti, A.; DiNardo, C.D.; Qiao, W.; Ning, J.; Loghavi, S.; Daver, N.G.; Kadia, T.M.; Rausch, C.R.; Alvarado, Y.; et al. Prognostic Impact of Conventional Cytogenetics in Acute Myeloid Leukemia Treated with Venetoclax and Decitabine. Leuk. Lymphoma 2021, 62, 3501–3505. [Google Scholar] [CrossRef]
- Gangat, N.; Johnson, I.; McCullough, K.; Farrukh, F.; Al-Kali, A.; Alkhateeb, H.; Begna, K.; Mangaonkar, A.; Litzow, M.; Hogan, W.; et al. Molecular Predictors of Response to Venetoclax plus Hypomethylating Agent in Treatment-Naïve Acute Myeloid Leukemia. Haematologica 2022, 107, 2501–2505. [Google Scholar] [CrossRef]
- Gangat, N.; Ilyas, R.; Johnson, I.M.; McCullough, K.; Al-Kali, A.; Alkhateeb, H.B.; Begna, K.H.; Mangaonkar, A.; Litzow, M.R.; Hogan, W.; et al. Outcome of Patients with Acute Myeloid Leukemia Following Failure of Frontline Venetoclax plus Hypomethylating Agent Therapy. Haematologica 2023, 108, 3170–3174. [Google Scholar] [CrossRef]
- Lin, J.; Liu, X.; Ying, S.; Zhu, Y.; Huang, W.; Zheng, W.; Ye, X.; Shi, J.; Luo, Y.; He, J.; et al. Selective Efficacy of Venetoclax in Unfit Patients with Acute Myeloid Leukemia with Myelodysplasia-Related Gene Mutations under Low-Intensity Therapy. Ann. Hematol. 2025, 104, 2717–2729. [Google Scholar] [CrossRef]
- Lai, A.; Liu, W.; Zhou, C.; Li, Y.; Wei, S.; Liu, K.; Gong, B.; Gong, X.; Liu, Y.; Zhang, G.; et al. Prognostic Impact of Co-Mutations in Adults with IDH1/2-Mutated Acute Myeloid Leukemia. Blood Sci. 2025, 7, e00231. [Google Scholar] [CrossRef]
- Hoff, F.W.; Huang, Y.; Welkie, R.L.; Swords, R.T.; Traer, E.; Stein, E.M.; Lin, T.L.; Patel, P.A.; Robert H Collins, J.; Baer, M.R.; et al. Molecular Characterization of Newly Diagnosed Acute Myeloid Leukemia Patients Aged 60 Years or Older: A Report from the Beat AML Clinical Trial. Blood Cancer J. 2025, 15, 55. [Google Scholar] [CrossRef]
- Yao, Y.; Zhou, Y.; Zhuo, N.; Xie, W.; Meng, H.; Lou, Y.; Mao, L.; Tong, H.; Qian, J.; Yang, M.; et al. Co-mutation landscape and its prognostic impact on newly diagnosed adult patients with NPM1-mutated de novo acute myeloid leukemia. Blood Cancer J. 2024, 14, 118. [Google Scholar] [CrossRef]
- DiNardo, C.D.; Tiong, I.S.; Quaglieri, A.; MacRaild, S.; Loghavi, S.; Brown, F.C.; Thijssen, R.; Pomilio, G.; Ivey, A.; Salmon, J.M.; et al. Molecular Patterns of Response and Treatment Failure after Frontline Venetoclax Combinations in Older Patients with AML. Blood 2020, 135, 791–803. [Google Scholar] [CrossRef]
- Venugopal, S.; DiNardo, C.D.; Loghavi, S.; Qiao, W.; Ravandi, F.; Konopleva, M.; Kadia, T.; Bhalla, K.; Jabbour, E.; Issa, G.C.; et al. Differential Prognostic Impact of RUNX1 Mutations According to Frontline Therapy in Patients with Acute Myeloid Leukemia. Am. J. Hematol. 2022, 97, 1560–1567. [Google Scholar] [CrossRef]


| Characteristic | Whole Population |
|---|---|
| n | 142 |
| Age [years], median, (IQR) | 77 (74–81) |
| Female n, (%) | 68 (48) |
| Male n, (%) | 74 (52) |
| AML according to ICC2022 n, (%) | 122 * (86) |
| AML with recurrent genetic abnormality n, (%) | 21 * (14) |
| AML with Mutated NPM1 | 17 (11.5) |
| AML with KMT2A Rearrangement | 3 (2) |
| AML with inv(3)(q21.3q26.2) | 1 (0.5) |
| AML with mutated TP53 n, (%) | 26 (18) |
| AML with myelodysplasia-related gene mutation n, (%) | 45 (32) |
| AML with myelodysplasia-related cytogenetic abnormality n, (%) | 4 (2.8) |
| AML not otherwise specified n, (%) | 13 (9) |
| Subtype unknown due to insufficient work-up n, (%) | 13 (9) |
| MDS/AML according to ICC2022 n, (%) | 20 (14) |
| MDS/AML with mutated TP53 n, (%) | 8 (5.6) |
| MDS/AML with myelodysplasia-related gene mutation n, (%) | 9 (6.3) |
| MDS/AML not otherwise specified n, (%) | 1 (0.7) |
| Subtype unknown due to insufficient work-up n, (%) | 2 (1.4) |
| Hemoglobin | |
| available | 141/142 |
| [g/L], median (IQR) | 88 (76–102) |
| Leukocytes | |
| available | 141/142 |
| (×109/L), median (IQR) | 3.3 (1.8–15) |
| Platelet count | |
| available | 141/142 |
| (×109/L), median (IQR) | 66 (36–111) |
| Neutrophils | |
| available | 139/142 |
| (×109/L), median (IQR) | 0.93 (0.36–3.1) |
| Blasts peripheral blood | |
| available | 140/142 |
| (%), median, IQR | 8.3 (0.5–23) |
| Blasts bone marrow | |
| available | 141/142 |
| (%) median, IQR | 35 (25–63) |
| HR | 95% CI | p | |
|---|---|---|---|
| ELN2022 | |||
| Adverse * | -- | -- | |
| Intermediate | 0.41 | 0.21, 0.79 | 0.008 |
| Favorable | 0.40 | 0.16, 0.99 | 0.047 |
| mPRS | |||
| Lower benefit * | -- | -- | |
| Intermediate benefit | 0.73 | 0.39, 1.36 | 0.3 |
| Higher benefit | 0.46 | 0.29, 0.73 | <0.001 |
| AML60+ | |||
| Very poor * | -- | -- | -- |
| Poor | 0.47 | 0.28, 0.78 | 0.004 |
| Intermediate/favorable | 0.17 | 0.10, 0.31 | <0.001 |
| HR | 95% CI | p | |
|---|---|---|---|
| ELN2022 | |||
| Adverse * | -- | -- | |
| Intermediate | 0.47 | 0.22, 1.00 | 0.049 |
| Favorable | 0.24 | 0.06, 0.97 | 0.045 |
| mPRS | |||
| Lower benefit * | -- | -- | |
| Intermediate benefit | 0.67 | 0.32, 1.40 | 0.3 |
| Higher benefit | 0.36 | 0.20, 0.64 | <0.001 |
| AML60+ | |||
| Very poor * | -- | -- | -- |
| Poor | 0.32 | 0.16, 0.65 | 0.002 |
| Intermediate/favorable | 0.14 | 0.07, 0.30 | <0.001 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Petermichl, V.; Fuchs, S.; Weber, M.; Gobat, K.; Micheloud, C.; Graf, L.; Gerth, Y.; Goede, J.S.; Lehmann, T.; Driessen, C.; et al. Prognostic Impact of the AML60+ Score for Elderly Patients with Acute Myeloid Leukemia Treated with Hypomethylating Agents: A Retrospective Multicentric Analysis. Cancers 2025, 17, 2658. https://doi.org/10.3390/cancers17162658
Petermichl V, Fuchs S, Weber M, Gobat K, Micheloud C, Graf L, Gerth Y, Goede JS, Lehmann T, Driessen C, et al. Prognostic Impact of the AML60+ Score for Elderly Patients with Acute Myeloid Leukemia Treated with Hypomethylating Agents: A Retrospective Multicentric Analysis. Cancers. 2025; 17(16):2658. https://doi.org/10.3390/cancers17162658
Chicago/Turabian StylePetermichl, Verena, Stefan Fuchs, Matthias Weber, Katrin Gobat, Charlotte Micheloud, Lukas Graf, Yannick Gerth, Jeroen S. Goede, Thomas Lehmann, Christoph Driessen, and et al. 2025. "Prognostic Impact of the AML60+ Score for Elderly Patients with Acute Myeloid Leukemia Treated with Hypomethylating Agents: A Retrospective Multicentric Analysis" Cancers 17, no. 16: 2658. https://doi.org/10.3390/cancers17162658
APA StylePetermichl, V., Fuchs, S., Weber, M., Gobat, K., Micheloud, C., Graf, L., Gerth, Y., Goede, J. S., Lehmann, T., Driessen, C., Mey, U. J. M., Cathomas, R., Cogliatti, S., & Silzle, T. (2025). Prognostic Impact of the AML60+ Score for Elderly Patients with Acute Myeloid Leukemia Treated with Hypomethylating Agents: A Retrospective Multicentric Analysis. Cancers, 17(16), 2658. https://doi.org/10.3390/cancers17162658







