WT1 Expression Is Associated with Poor Overall Survival after Azacytidine and DLI in a Cohort of Adult AML and MDS Patients
Abstract
Simple Summary
Abstract
1. Introduction
2. Patients and Treatment
3. Statistical Analysis
4. Results
4.1. Patient and Transplant Characteristics
4.2. Relapse and Treatment Characteristics
4.3. Safety and Toxicity
4.4. GVHD
4.5. Treatment Response and Survival
4.6. WT1
5. Discussion
6. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Schmid, C.; de Wreede, L.C.; van Biezen, A.; Finke, J.; Ehninger, G.; Ganser, A.; Volin, L.; Niederwieser, D.; Beelen, D.; Alessandrino, P.; et al. Outcome after relapse of myelodysplastic syndrome and secondary acute myeloid leukemia following allogeneic stem cell transplantation: A retrospective registry analysis on 698 patients by the Chronic Malignancies Working Party of the European Society of Blood and Marrow Transplantation. Haematologica 2018, 103, 237–245. [Google Scholar]
- Schmid, C.; Labopin, M.; Nagler, A.; Bornhäuser, M.; Finke, J.; Fassas, A.; Volin, L.; Gürman, G.; Maertens, J.; Bordigoni, P.; et al. EBMT Acute Leukemia Working Party. Donor Lymphocyte Infusion in the Treatment of First Hematological Relapse After Allogeneic Stem-Cell Transplantation in Adults With Acute Myeloid Leukemia: A Retrospective Risk Factors Analysis and Comparison With Other Strategies by the EBMT Acute Leukemia Working Party. J. Clin. Oncol. 2007, 25, 4938–4945. [Google Scholar] [PubMed]
- Craddock, C.; Labopin, M.; Robin, M.; Finke, J.; Chevallier, P.; Yakoub-Agha, I.; Bourhis, J.H.; Sengelov, H.; Blaise, D.; Luft, T.; et al. Clinical activity of azacitidine in patients who relapse after allogeneic stem cell transplantation for acute myeloid leukemia. Haematologica 2016, 101, 879–883. [Google Scholar] [CrossRef] [PubMed]
- Platzbecker, U.; Wermke, M.; Radke, J.; Oelschlaegel, U.; Seltmann, F.; Kiani, A.; Klut, I.M.; Knoth, H.; Röllig, C.; Schetelig, J.; et al. Azacitidine for treatment of imminent relapse in MDS or AML patients after allogeneic HSCT: Results of the RELAZA trial. Leukemia 2012, 26, 381–389. [Google Scholar] [CrossRef]
- Czibere, A.; Bruns, I.; Kröger, N.; Platzbecker, U.; Lind, J.; Zohren, F.; Fenk, R.; Germing, U.; Schröder, T.; Gräf, T.; et al. 5-Azacytidine for the treatment of patients with acute myeloid leukemia or myelodysplastic syndrome who relapse after allo-SCT: A retrospective analysis. Bone Marrow Transplant. 2010, 45, 872–876. [Google Scholar] [CrossRef] [PubMed]
- Schroeder, T.; Czibere, A.; Platzbecker, U.; Bug, G.; Uharek, L.; Luft, T.; Giagounidis, A.; Zohren, F.; Bruns, I.; Wolschke, C.; et al. Azacitidine and donor lymphocyte infusions as first salvage therapy for relapse of AML or MDS after allogeneic stem cell transplantation. Leukemia 2013, 27, 1229–1235. [Google Scholar] [CrossRef]
- Atanackovic, D.; Luetkens, T.; Kloth, B.; Fuchs, G.; Cao, Y.; Hildebrandt, Y.; Meyer, S.; Bartels, K.; Reinhard, H.; Lajmi, N.; et al. Cancer-testis antigen expression and its epigenetic modulation in acute myeloid leukemia. Am. J. Hematol. 2011, 86, 918–922. [Google Scholar] [CrossRef]
- Pinto, A.; Maio, M.; Attadia, V.; Zappacosta, S.; Cimino, R. Modulation of HLA-DR antigens expression in human myeloid leukaemia cells by cytarabine and 5-aza-2’-deoxycytidine. Lancet 1984, 324, 867–868. [Google Scholar] [CrossRef]
- Goodyear, O.; Agathanggelou, A.; Novitzky-Basso, I.; Siddique, S.; McSkeane, T.; Ryan, G.; Vyas, P.; Cavenagh, J.; Stankovic, T.; Moss, P.; et al. Induction of a CD8+ T-cell response to the MAGE cancer testis antigen by combined treatment with azacitidine and sodium valproate in patients with acute myeloid leukemia and myelodysplasia. Blood 2010, 116, 1908–1918. [Google Scholar] [CrossRef]
- Sánchez-Abarca, L.I.; Gutierrez-Cosio, S.; Santamaría, C.; Caballero-Velazquez, T.; Blanco, B.; Herrero-Sánchez, C.; García, J.L.; Carrancio, S.; Hernández-Campo, P.; González, F.J.; et al. Imunomodulatory effect of 5-azacytidine (5-azaC): Potential role in the transplantation setting. Blood 2010, 115, 107–121. [Google Scholar] [CrossRef]
- Choi, J.; Ritchey, J.; Prior, J.L.; Holt, M.; Shannon, W.D.; Deych, E.; Piwnica-Worms, D.R.; DiPersio, J.F. In vivo administration of hypomethylating agents mitigate graft-versus-host disease without sacrificing graft-versus-leukemia. Blood 2010, 116, 129–139. [Google Scholar] [CrossRef]
- Cooper, M.L.; Choi, J.; Karpova, D.; Vij, K.; Ritchey, J.; Schroeder, M.A.; DiPersio, J.F. Azacitidine Mitigates Graft-versus-Host Disease via Differential Effects on the Proliferation of T Effectors and Natural Regulatory T Cells In Vivo. J. Immunol. 2017, 198, 3746–3754. [Google Scholar] [CrossRef]
- Boada, M.; Echarte, L.; Guillermo, C.; Cristina Tourino, L.D.; Grille, S. 5-Azacytidine restores interleukin 6-increased production in mesenchymal stromal cells from myelodysplastic patients. Hematol. Transfus. Cell Ther. 2021, 43, 35–42. [Google Scholar] [CrossRef] [PubMed]
- Sugiyama, H. WT1 (Wilms’ tumor gene 1): Biology and cancer immunotherapy. Jpn. J. Clin. Oncol. 2010, 40, 377–387. [Google Scholar] [CrossRef] [PubMed]
- Rampal, R.; Figueroa, M.E. Wilms tumor 1 mutations in the pathogenesis of acute myeloid leukemia. Haematologica 2016, 101, 672–679. [Google Scholar] [CrossRef] [PubMed]
- Ito, K.; Oji, Y.; Tatsumi, N.; Shimizu, S.; Kanai, Y.; Nakazawa, T.; Asada, M.; Jomgeow, T.; Aoyagi, S.; Nakano, Y.; et al. Antiapoptotic function of 17AA(+)WT1 (Wilms’ tumor gene) isoforms on the intrinsic apoptosis pathway. Oncogene 2006, 25, 4217–4229. [Google Scholar] [CrossRef]
- Cilloni, D.; Renneville, A.; Hermitte, F.; Hills, R.K.; Daly, S.; Jovanovic, J.V.; Gottardi, E.; Fava, M.; Schnittger, S.; Weiss, T.; et al. Real-time quantitative polymerase chain reaction detection of minimal residual disease by standardized WT1 assay to enhance risk stratification in acute myeloid leukemia: A European LeukemiaNet study. J. Clin. Oncol. 2009, 27, 5195–5201. [Google Scholar] [CrossRef]
- Lange, T.; Hubmann, M.; Burkhardt, R.; Franke, G.N.; Cross, M.; Scholz, M.; Leiblein, S.; Al-Ali, H.K.; Edelmann, J.; Thiery, J.; et al. Monitoring of WT1 expression in PB and CD34(+) donor chimerism of BM predicts early relapse in AML and MDS patients after hematopoietic cell transplantation with reduced-intensity conditioning. Leukemia 2011, 25, 498–505. [Google Scholar] [CrossRef]
- Cancer Genome Atlas Research Network; Ley, T.J.; Miller, C.; Ding, L.; Raphael, B.J.; Mungall, A.J.; Robertson, A.; Hoadley, K.; Triche, T.J., Jr.; Laird, P.W.; et al. Genomic and epigenomic landscapes of adult de novo acute myeloid leukemia. N. Engl. J. Med. 2013, 368, 2059–2074. [Google Scholar]
- Döhner, H.; Estey, E.; Grimwade, D.; Amadori, S.; Appelbaum, F.R.; Büchner, T.; Dombret, H.; Ebert, B.L.; Fenaux, P.; Larson, R.A.; et al. Diagnosis and management of AML in adults: 2017 ELN recommendations from an international expert panel. Blood 2017, 129, 424–447. [Google Scholar] [CrossRef]
- Bryant, E.; Martin, P.J. Documentation of engraftment and characterization of chimerism following hematopoietic cell transplantation. In Thomas’ Hematopoietic Cell Transplantation; Blume, K.G., Forman, S.J., Appelbaum, F.R., Eds.; Blackwell Publishing Ltd.: Oxford, UK, 2004; pp. 234–243. [Google Scholar]
- Armand, P.; Kim, H.T.; Logan, B.R.; Wang, Z.; Alyea, E.P.; Kalaycio, M.E.; Maziarz, R.T.; Antin, J.H.; Soiffer, R.J.; Weisdorf, D.J.; et al. Validation and refinement of the Disease Risk Index for allogeneic stem cell transplantation. Blood 2014, 123, 3664–3671. [Google Scholar] [CrossRef]
- Frairia, C.; Aydin, S.; Audisio, E.; Riera, L.; Aliberti, S.; Allione, B.; Busca, A.; D’Ardia, S.; Dellacasa, C.M.; Demurtas, A.; et al. Post-remissional and pre-transplant role of minimal residual disease detected by WT1 in acute myeloid leukemia: A retrospective cohort study. Leuk. Res. 2017, 61, 10–17. [Google Scholar] [CrossRef]
- Therneau, T.M.; Grambsch, P.M.; Fleming, T.R. Martingale-based residuals for survival models. Biometrika 1990, 77, 147–160. [Google Scholar] [CrossRef]
- Klein, J.P.; Moeschberger, S. Survival Analysis: Techniques for Censored and Truncated Data; Springer: Berlin, Germany, 1997. [Google Scholar]
- Patel, C.; Stenke, L.; Varma, S.; Lindberg, M.L.; Björkholm, M.; Sjöberg, J.; Viktorsson, K.; Lewensohn, R.; Landgren, O.; Gottesman, M.M.; et al. Multidrug resistance in relapsed acute myeloid leukemia: Evidence of biological heterogeneity. Cancer 2013, 119, 3076–3083. [Google Scholar] [CrossRef]
- Kroeger, H.; Jelinek, J.; Estécio, M.R.; He, R.; Kondo, K.; Chung, W.; Zhang, L.; Shen, L.; Kantarjian, H.M.; Bueso-Ramos, C.E.; et al. Aberrant CpG island methylation in acute myeloid leukemia is accentuated at relapse. Blood 2008, 112, 1366–1373. [Google Scholar] [CrossRef] [PubMed]
- Hackl, H.; Astanina, K.; Wieser, R. Molecular and genetic alterations associated with therapy resistance and relapse of acute myeloid leukemia. J. Hematol. Oncol. 2017, 10, 51. [Google Scholar] [CrossRef] [PubMed]
- Schroeder, T.; Rachlis, E.; Bug, G.; Stelljes, M.; Klein, S.; Steckel, N.K.; Wolf, D.; Ringhoffer, M.; Czibere, A.; Nachtkamp, K.; et al. Treatment of acute myeloid leukemia or myelodysplastic syndrome relapse after allogeneic stem cell transplantation with azacitidine and donor lymphocyte infusions: A retrospective multicenter analysis from the German Cooperative Transplant Study Group. Biol. Blood Marrow Transplant. 2015, 21, 653–660. [Google Scholar] [CrossRef]
- Válková, V.; Polák, J.; Marková, M.; Vítek, A.; Hájková, H.; Sálek, C.; Procházka, B.; Cetkovský, P.; Trněný, M. Minimal residual disease detectable by quantitative assessment of WT1 gene before allogeneic stem cell transplantation in patients in first remission of acute myeloid leukemia has an impact on their future prognosis. Clin. Transplant. 2013, 27, E21–E29. [Google Scholar] [CrossRef]
- Buckley, S.A.; Wood, B.L.; Othus, M.; Hourigan, C.S.; Ustun, C.; Linden, M.A.; DeFor, T.E.; Malagola, M.; Anthias, C.; Valkova, V.; et al. Minimal residual disease prior to allogeneic hematopoietic cell transplantation in acute myeloid leukemia: A meta-analysis. Haematologica 2017, 102, 865–873. [Google Scholar] [CrossRef]
- Šálek, C.; Vydra, J.; Cerovská, E.; Šestáková, Š.; Ransdorfová, Š.; Válková, V.; Cetkovský, P.; Remešová, H. WT1 Expression in Peripheral Blood at Diagnosis and During the Course of Early Consolidation Treatment Correlates with Survival in Patients with Intermediate and Poor-Risk Acute Myeloid Leukemia. Clin. Lymphoma Myeloma Leuk. 2020, 20, e998–e1009. [Google Scholar] [CrossRef]
- Valkova, V.; Vydra, J.; Markova, M.; Cerovska, E.; Vrana, M.; Marinov, I.; Cechova, H.; Cetkovsky, P.; Vitek, A.; Salek, C. WT1 Gene Expression in Peripheral Blood Before and After Allogeneic Stem Cell Transplantation is a Clinically Relevant Prognostic Marker in AML—A Single-center 14-year Experience. Clin. Lymphoma Myeloma Leuk. 2021, 21, e145–e151. [Google Scholar] [CrossRef] [PubMed]
- Rautenberg, C.; Lauseker, M.; Kaivers, J.; Jäger, P.; Fischermanns, C.; Pechtel, S.; Haas, R.; Kobbe, G.; Germing, U.; Schroeder, T. Prognostic impact of pretransplant measurable residual disease assessed by peripheral blood WT1-mRNA expression in patients with AML and MDS. Eur. J. Haematol. 2021, 107, 283–292. [Google Scholar] [CrossRef]
- Chapuis, A.G.; Egan, D.N.; Bar, M.; Schmitt, T.M.; McAfee, M.S.; Paulson, K.G.; Voillet, V.; Gottardo, R.; Ragnarsson, G.B.; Bleakley, M.; et al. T cell receptor gene therapy targeting WT1 prevents acute myeloid leukemia relapse post-transplant. Nat. Med. 2019, 25, 1064–1072. [Google Scholar] [CrossRef] [PubMed]
- Augsberger, C.; Hänel, G.; Xu, W.; Pulko, V.; Hanisch, L.J.; Augustin, A.; Challier, J.; Hunt, K.; Vick, B.; Rovatti, P.E.; et al. Targeting intracellular WT1 in AML with a novel RMF-peptide-MHC-specific T-cell bispecific antibody. Blood 2021, 138, 2655–2669. [Google Scholar] [CrossRef]
- Claiborne, J.; Bandyopathyay, D.; Roberts, C.; Hawks, K.; Aziz, M.; Simmons, G.; Wiedl, C.; Chung, H.; Clark, W.; McCarty, J.; et al. Managing post allograft relapse of myeloid neoplasms: Azacitidine and donor lymphocyte infusions as salvage therapy. Leuk. Lymphoma 2019, 60, 2733–2743. [Google Scholar] [CrossRef]
- de Bueger, M.; Bakker, A.; Van Rood, J.J.; Van der Woude, F.; Goulmy, E. Tissue distribution of human minor histocompatibility antigens. Ubiquitous versus restricted tissue distribution indicates heterogeneity among human cytotoxic T lymphocyte-defined non-MHC antigens. J. Immunol. 1992, 149, 1788–1794. [Google Scholar] [CrossRef]
- Bacchetta, R.; Lucarelli, B.; Sartirana, C.; Gregori, S.; Lupo Stanghellini, M.T.; Miqueu, P.; Tomiuk, S.; Hernandez-Fuentes, M.; Gianolini, M.E.; Greco, R.; et al. Immunological Outcome in Haploidentical-HSC Transplanted Patients Treated with IL-10-Anergized Donor T Cells. Front. Immunol. 2014, 5, 16. [Google Scholar] [CrossRef]
- Orti, G.; Barba, P.; Fox, L.; Salamero, O.; Bosch, F.; Valcarcel, D. Donor lymphocyte infusions in AML and MDS: Enhancing the graft-versus-leukemia effect. Exp. Hematol. 2017, 48, 1–11. [Google Scholar] [CrossRef] [PubMed]
- Rautenberg, C.; Bergmann, A.; Pechtel, S.; Fischermanns, C.; Haas, R.; Germing, U.; Kobbe, G.; Schroeder, T. Wilm’s Tumor 1-guided preemptive treatment with hypomethylating agents for molecular relapse of AML and MDS after allogeneic transplantation. Bone Marrow Transplant. 2021, 56, 442–450. [Google Scholar] [CrossRef]
- Guillaume, T.; Thépot, S.; Peterlin, P.; Ceballos, P.; Bourgeois, A.L.; Garnier, A.; Orvain, C.; Giltat, A.; François, S.; Bris, Y.L.; et al. Prophylactic or Preemptive Low-Dose Azacitidine and Donor Lymphocyte Infusion to Prevent Disease Relapse following Allogeneic Transplantation in Patients with High-Risk Acute Myelogenous Leukemia or Myelodysplastic Syndrome. Transplant. Cell. Ther. 2021, 27, 839.e1–839.e6. [Google Scholar] [CrossRef]
- Rashidi, A.; Huselton, E.J.; Stefanski, H.E.; DeFor, T.E.; Shanley, R.; Choi, J.; DiPersio, J.F.; Juckett, M.; Miller, J.S.; Weisdorf, D.J.; et al. A Multicenter Phase 2 Clinical Trial of 10-Day Decitabine, Dose-Escalated Donor Lymphocyte Infusion, and Ruxolitinib for Relapsed Acute Myeloid Leukemia and Myelodysplastic Syndromes after Allogeneic Hematopoietic Cell Transplantation. Transplant. Cell. Ther. 2023, 29, 328.e1–328.e6. [Google Scholar] [CrossRef] [PubMed]
- Schroeder, T.; Stelljes, M.; Christopeit, M.; Esseling, E.; Scheid, C.; Mikesch, J.H.; Rautenberg, C.; Jäger, P.; Cadeddu, R.P.; Drusenheimer, N.; et al. Azacitidine, lenalidomide and donor lymphocyte infusions for relapse of myelodysplastic syndrome, acute myeloid leukemia and chronic myelomonocytic leukemia after allogeneic transplant: The Azalena-Trial. Haematologica 2023, 108, 3001–3010. [Google Scholar] [CrossRef] [PubMed]
| Variable | n= | %/IQR |
|---|---|---|
| Patients | 29 | |
| Median age at HSCT, years | 52.3 | 25–68 |
| Diagnosis before HSCT | ||
| AML | 20 | 69 |
| MDS | 9 | 31 |
| Cytogenetic/molecular risk stratification | ||
| Adverse | 17 | 58.6 |
| Intermediate | 9 | 31 |
| Missing | 3 | 10.3 |
| Disease Risk Index stratification | ||
| Intermediate | 12 | 41.4 |
| High | 11 | 37.9 |
| Very high | 6 | 20.7 |
| Reason for high-risk stratification | ||
| Primary refractory | 9 | 31 |
| Relapse | 7 | 24.1 |
| t-AML or post-MDS/MPS | 4 | 13.8 |
| Extramedullary disease | 1 | 3.4 |
| Disease status at HSCT | ||
| CR1/>CR1 | 19 | 65.5 |
| SD | 3 | 10.3 |
| Relapse/progressive | 7 | 24.1 |
| Conditioning regimen | ||
| MAC (sequential) | 25 (5) | 86.2 (17.2) |
| RIC | 4 | 13.8 |
| Donor type | ||
| MSD | 7 | 24.1 |
| MUD | 16 | 55.2 |
| Haploidentical | 6 | 20.7 |
| Graft source | ||
| PBSC | 21 | 72.4 |
| BM | 8 | 27.6 |
| WT1 at transplant, range copies | 184 | 15–7335 |
| Variable | n= | %/IQR |
|---|---|---|
| Median time to relapse, months | 8.3 | 5.7–16.1 |
| Diagnosis at relapse | ||
| AML | 24 | 82.8 |
| MDS | 5 | 17.2 |
| BM chimerism at relapse, median | 85.5 | 0–99% |
| WT1 at relapse, median copies | 1570 | 89–10665 |
| aGVHD before relapse | ||
| Grade I | 6 | 20.7 |
| Grade II | 9 | 31.0 |
| Grade III | 1 | 3.4 |
| cGVHD before relapse | ||
| Mild | 4 | 13.8 |
| Moderate | 6 | 20.7 |
| Severe | 1 | 3.4 |
| Immunosuppression at relapse | 18 | 62.1 |
| Taper/stop before AZA start | 17 | 58.6 |
| Number of AZA cycles, median | 2 | 1–24 |
| 1–3 cycle | 13 | 44.8 |
| 4–6 cycles | 8 | 27.6 |
| >6 cycles | 8 | 27.6 |
| Number of DLI, median | 1 | 0–6 |
| no DLI | 11 | 37.9 |
| 1–3 DLI | 14 | 48.3 |
| 4–6 DLI | 4 | 13.7 |
| GVHD post AZA/DLI | 3 | 10.3 |
| Response to AZA/DLI | ||
| CR | 7 | 24.1 |
| SD | 5 | 17.2 |
| PD | 17 | 58.6 |
| Duration of response, months | ||
| CR | 19.5 | 11.5–29.7 |
| SD | 11.9 | 1.6–17.4 |
| Survival Analysis | Univariate Model | ||||
|---|---|---|---|---|---|
| Median (IQR) | p * | HR (95% CI) | p ** | ||
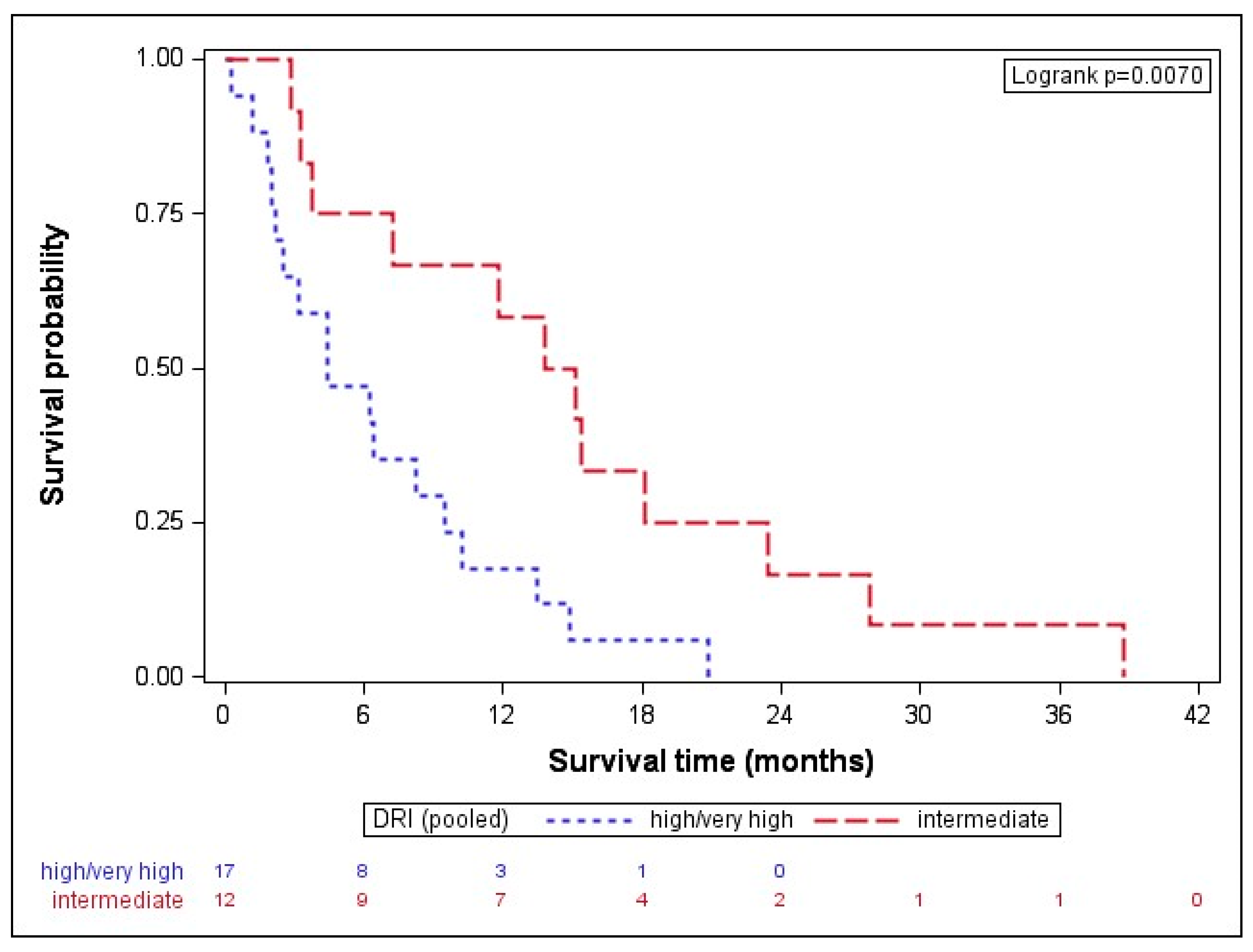
| DRI high/very high intermediate | 4.44 (2.20–9.47) 14.47 (5.51–20.78) | 0.0070 | high/very high vs. intermediate | 3.05 (1.31; 7.11) | 0.0096 |
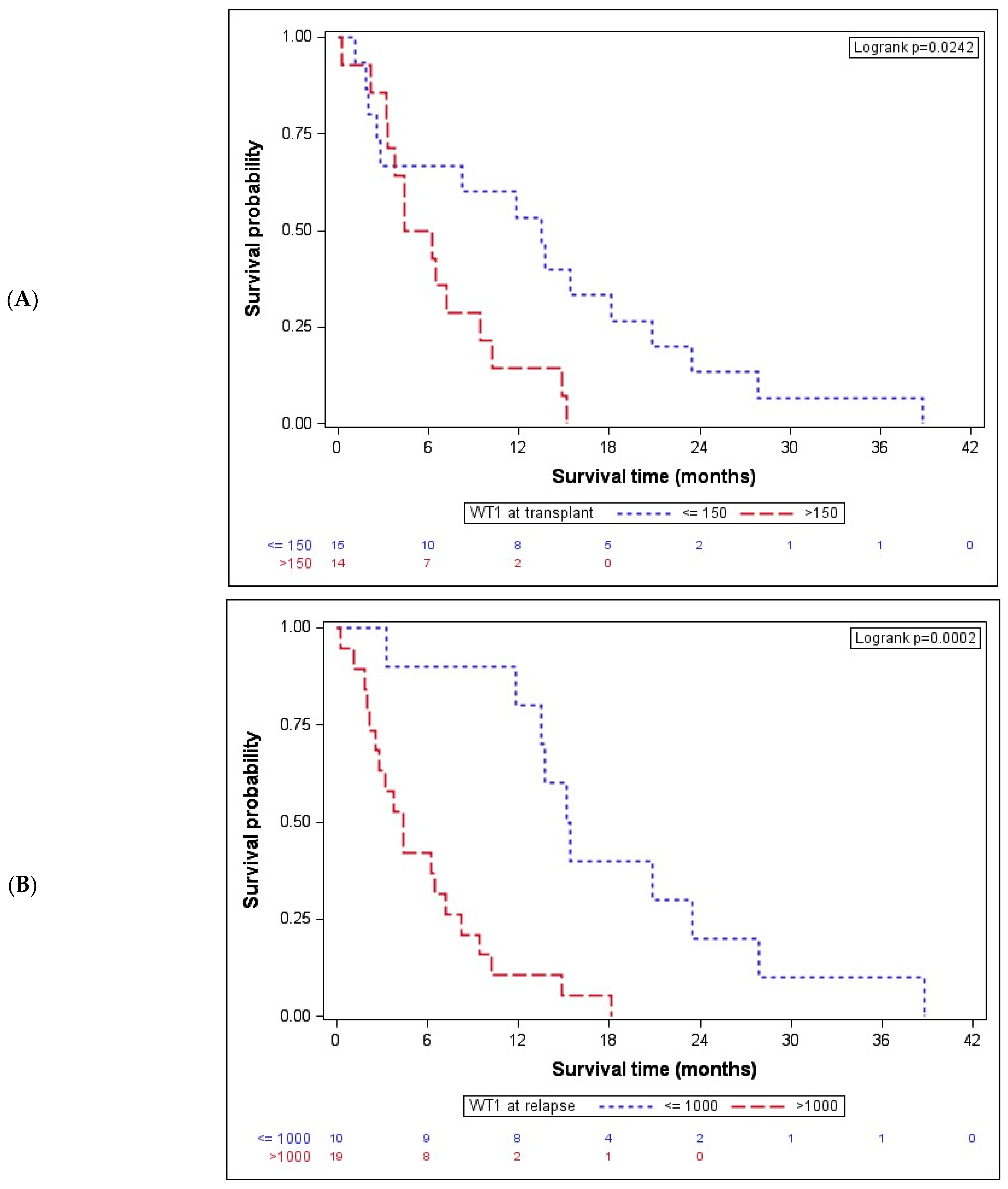
| WT1 at transplant ≤150 >150 | 13.48 (2.53–20.81) 5.33 (3.29–9.47) | 0.0242 | >150 vs. ≤150 | 2.65 (1.11; 6.34) | 0.0289 |
| WT1 at relapse ≤1000 >1000 | 15.27 (13.48–23.44) 4.41 (2.20–8.25) | 0.0002 | >1000 vs. ≤1000 | 5.39 (2.04; 14.24) | 0.0007 |
| LDH at relapse ≤390 >390 | 11.84 (4.44–15.16) 3.79 (2.01–8.35) | 0.0286 | >390 vs. ≤390 | 2.37 (1.07; 5.25) | 0.0335 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Aydin, S.; Schmitz, J.; Dellacasa, C.M.; Dogliotti, I.; Giaccone, L.; Busca, A. WT1 Expression Is Associated with Poor Overall Survival after Azacytidine and DLI in a Cohort of Adult AML and MDS Patients. Cancers 2024, 16, 3070. https://doi.org/10.3390/cancers16173070
Aydin S, Schmitz J, Dellacasa CM, Dogliotti I, Giaccone L, Busca A. WT1 Expression Is Associated with Poor Overall Survival after Azacytidine and DLI in a Cohort of Adult AML and MDS Patients. Cancers. 2024; 16(17):3070. https://doi.org/10.3390/cancers16173070
Chicago/Turabian StyleAydin, Semra, Jennifer Schmitz, Chiara M. Dellacasa, Irene Dogliotti, Luisa Giaccone, and Alessandro Busca. 2024. "WT1 Expression Is Associated with Poor Overall Survival after Azacytidine and DLI in a Cohort of Adult AML and MDS Patients" Cancers 16, no. 17: 3070. https://doi.org/10.3390/cancers16173070
APA StyleAydin, S., Schmitz, J., Dellacasa, C. M., Dogliotti, I., Giaccone, L., & Busca, A. (2024). WT1 Expression Is Associated with Poor Overall Survival after Azacytidine and DLI in a Cohort of Adult AML and MDS Patients. Cancers, 16(17), 3070. https://doi.org/10.3390/cancers16173070








