Peripheral Blood TCRβ Repertoire, IL15, IL2 and Soluble Ligands for NKG2D Activating Receptor Predict Efficacy of Immune Checkpoint Inhibitors in Lung Cancer
Abstract
Simple Summary
Abstract
1. Introduction
2. Materials and Methods
2.1. Population
2.2. Sample Type and Processing
2.2.1. Sample Processing
2.2.2. Sequencing and Analysis of the TCRβ Repertoire
2.2.3. Determination of Cytokines in Peripheral Blood
2.3. Statistical Analysis
3. Results
3.1. Descriptive Analysis
Patient and Tumor Disease Characteristics
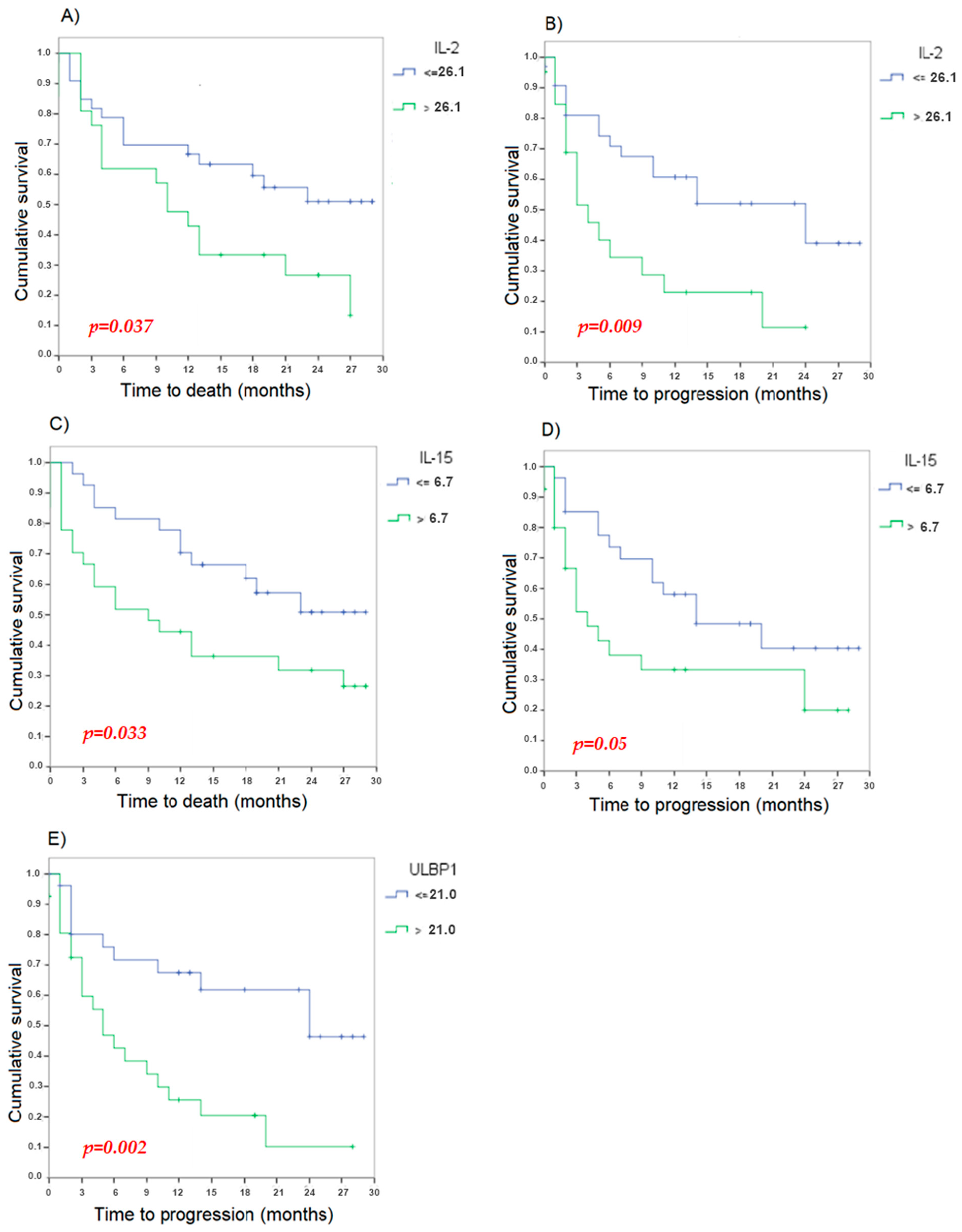
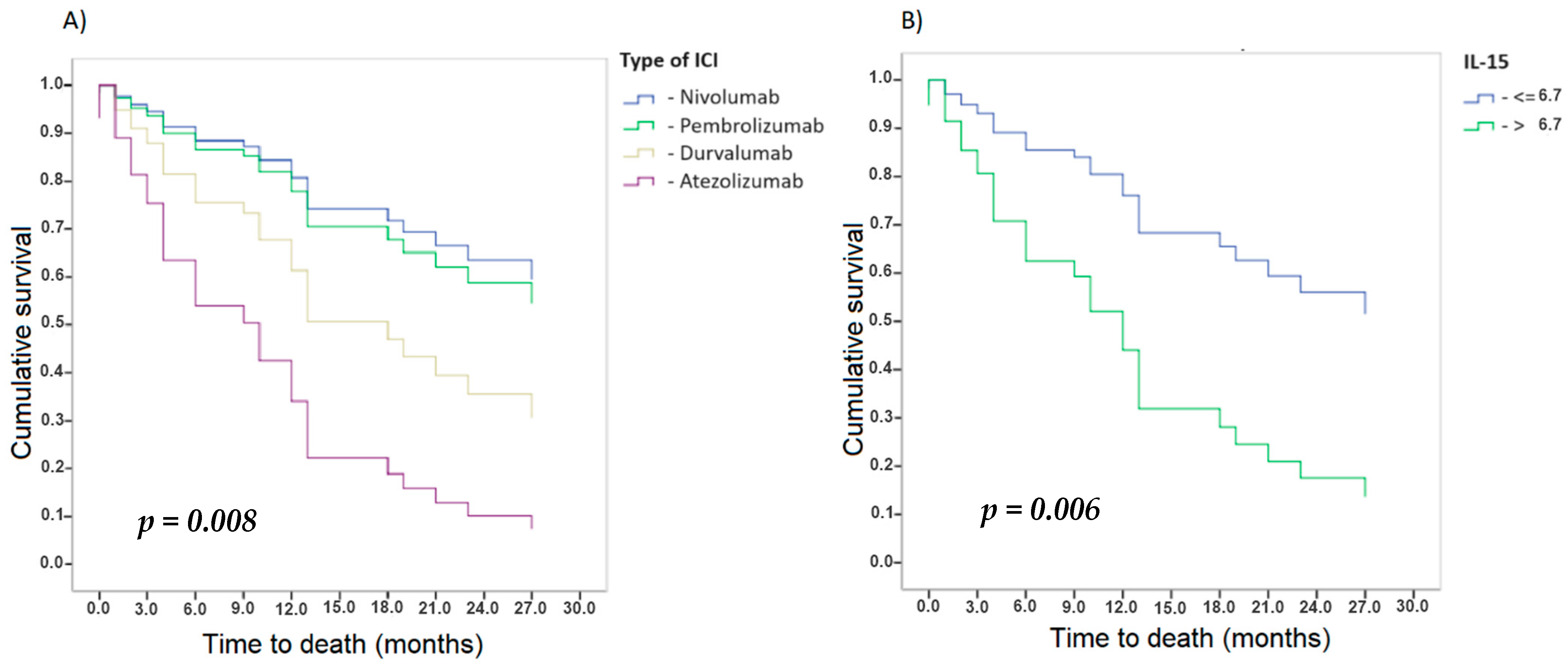
3.2. Survival Analysis
3.2.1. Clinical Pathological Features
3.2.2. TCRβ Repertoire
TCRβ Sequencing Data Analysis
3.2.3. Cytokines and Other Soluble Factors
Analysis of the Determination of Cytokines
3.3. Multivariate Analysis
4. Discussion
5. Limitations
6. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Antón, A. (Ed.) Inmunología Tumoral e Inmunoterapia del Cáncer; Amazing Books: Zaragoza, Spain, 2018. [Google Scholar]
- Lim, S.W.; Ahn, M.J. Current status of immune checkpoint inhibitors in treatment of non-small cell lung cancer. Korean J. Intern. Med. 2019, 34, 50–59. [Google Scholar] [CrossRef]
- Sánchez de Cos Escuín, J. Nueva inmunoterapia y cáncer de pulmón. Arch. Bronconeumol. 2017, 53, 682–687. [Google Scholar] [CrossRef] [PubMed]
- Chen, D.S.; Mellman, I. Elements of cancer immunity and the cancer–immune set point. Nature 2017, 541, 321–330. [Google Scholar] [CrossRef]
- Bai, J.; Gao, Z.; Li, X.; Dong, L.; Han, W.; Nie, J. Regulation of PD-1/PD-L1 pathway and resistance to PD-1/PD-L1 blockade. Oncotarget 2017, 8, 110693. [Google Scholar] [CrossRef]
- Sharma, P.; Hu-Lieskovan, S.; Wargo, J.A.; Ribas, A. Primary, Adaptive, and Acquired Resistance to Cancer Immunotherapy. Cell 2017, 168, 707–723. [Google Scholar] [CrossRef] [PubMed]
- Lesterhuis, W.J.; Bosco, A.; Millward, M.J.; Small, M.; Nowak, A.K.; Lake, R.A. Dynamic versus static biomarkers in cancer immune checkpoint blockade: Unravelling complexity. Nat. Rev. Drug Discov. 2017, 16, 264–272. [Google Scholar] [CrossRef]
- Aversa, I.; Malanga, D.; Fiume, G.; Palmieri, C. Molecular T-cell repertoire analysis as source of prognostic and predictive biomarkers for checkpoint blockade immunotherapy. Int. J. Mol. Sci. 2020, 21, 2378. [Google Scholar] [CrossRef]
- McNeel, D.G. TCR diversity—A universal cancer immunotherapy biomarker? J. Immunother. Cancer 2016, 4, 69. [Google Scholar] [CrossRef] [PubMed]
- Cha, E.; Klinger, M.; Hou, Y.; Cummings, C.; Ribas, A.; Faham, M.; Fong, L. Improved survival with T cell clonotype stability after anti-CTLA-4 treatment in cancer patients. Sci. Transl. Med. 2014, 6, 238ra70. [Google Scholar] [CrossRef]
- Lichtman, A. Inmunología Celular y Molecular, 6th ed.; Journal of Chemical Information and Modeling; Elsevier Saunders: Amsterdam, The Netherlands, 2015; Volume 53, pp. 1–532. [Google Scholar]
- Quagliata, L.; Looney, T.; Storkus, W.; Taylor, J.; Topacio-Hall, D.; Lowman, G. T cell repertoire sequencing reveals dynamics of response to dendritic cell vaccine plus dasatinib for checkpoint blockade resistant metastatic melanoma. Ann. Oncol. 2019, 30, v496–v497. [Google Scholar] [CrossRef]
- Dong, N.; Moreno-Manuel, A.; Calabuig-Fariñas, S.; Gallach, S.; Zhang, F.; Blasco, A.; Aparisi, F.; Meri-Abad, M.; Guijarro, R.; Sirera, R.; et al. Characterization of circulating t cell receptor repertoire provides information about clinical outcome after pd-1 blockade in advanced non-small cell lung cancer patients. Cancers 2021, 13, 2950. [Google Scholar] [CrossRef]
- Robert, L.; Tsoi, J.; Wang, X.; Emerson, R.; Homet, B.; Chodon, T.; Mok, S.; Huang, R.R.; Cochran, A.J.; Comin-Anduix, B.; et al. CTLA4 blockade broadens the peripheral T-cell receptor repertoire. Clin. Cancer Res. 2014, 20, 2424–2432. [Google Scholar] [CrossRef]
- Sesma, A.; Pardo, J.; Cruellas, M.; Gálvez, E.M.; Gascón, M.; Isla, D.; Martínez-Lostao, L.; Ocáriz, M.; Paño, J.R.; Quílez, E.; et al. From tumor mutational burden to blood T cell receptor: Looking for the best predictive biomarker in lung cancer treated with immunotherapy. Cancers 2020, 12, 2974. [Google Scholar] [CrossRef]
- Looney, T.J.; Topacio-Hall, D.; Lowman, G.; Conroy, J.; Morrison, C.; Oh, D.; Fong, L.; Zhang, L. TCR Convergence in Individuals Treated with Immune Checkpoint Inhibition for Cancer. Front. Immunol. 2020, 10, 2985. [Google Scholar] [CrossRef]
- Zhang, L.; Looney, T.; Lowman, G.; Oh, D.; Fong, L. Peripheral blood TCRB repertoire convergence and clonal expansion predict response to anti-CTLA-4 monotherapy for cancer. Res. Sq. 2024, in press.
- Gascón-Ruiz, M.; Ramírez-Labrada, A.; Lastra, R.; Martínez-Lostao, L.; Paño-Pardo, J.R.; Sesma, A.; Zapata-García, M.; Moratiel, A.; Quílez, E.; Torres-Ramón, I.; et al. A Subset of PD-1-Expressing CD56bright NK Cells Identifies Patients with Good Response to Immune Checkpoint Inhibitors in Lung Cancer. Cancers 2023, 15, 329. [Google Scholar] [CrossRef]
- Han, J.; Duan, J.; Bai, H.; Wang, Y.; Wan, R.; Wang, X.; Chen, S.; Tian, Y.; Wang, D.; Fei, K.; et al. TCR repertoire diversity of peripheral PD-1þCD8þ T cells predicts clinical outcomes after immunotherapy in patients with non–small cell lung cancer. Cancer Immunol. Res. 2020, 8, 146–154. [Google Scholar] [CrossRef]
- Anagnostou, V.; Forde, P.M.; White, J.R.; Niknafs, N.; Hruban, C.; Naidoo, J.; Marrone, K.; Sivakumar, I.K.A.; Bruhm, D.C.; Rosner, S.; et al. Dynamics of Tumor and Immune Responses during Immune Checkpoint Blockade in Non-Small Cell Lung Cancer. HHS Public Access 2019, 79, 1214–1225. [Google Scholar] [CrossRef]
- Binnewies, M.; Roberts, E.W.; Kersten, K.; Chan, V.; Fearon, D.F.; Merad, M.; Coussens, L.M.; Gabrilovich, D.I.; Ostrand-Rosenberg, S.; Hedrick, C.C.; et al. Understanding the tumor immune microenvironment (TIME) for effective therapy. Nat. Med. 2018, 24, 541–550. [Google Scholar] [CrossRef]
- Zhang, J.; Ji, Z.; Caushi, J.X.; El Asmar, M.; Anagnostou, V.; Cottrell, T.R.; Chan, H.Y.; Suri, P.; Guo, H.; Merghoub, T.; et al. Compartmental analysis of T-Cell clonal dynamics as a function of pathologic response to neoadjuvant PD-1 blockade in resectable non-small cell lung cancer. Clin. Cancer Res. 2020, 26, 1327–1337. [Google Scholar] [CrossRef]
- Huang, A.C.; Postow, M.A.; Orlowski, R.J.; Mick, R.; Bengsch, B.; Manne, S.; Xu, W.; Harmon, S.; Giles, J.R.; Wenz, B.; et al. T-cell invigoration to tumour burden ratio associated with anti-PD-1 response. Nature 2017, 545, 60–65. [Google Scholar] [CrossRef]
- Puig-Saus, C.; Sennino, B.; Peng, S.; Wang, C.L.; Pan, Z.; Yuen, B.; Purandare, B.; An, D.; Quach, B.B.; Nguyen, D.; et al. Neoantigen-targeted CD8(+) T cell responses with PD-1 blockade therapy. Nature 2023, 615, 697–704. [Google Scholar] [CrossRef]
- Kuehm, L.M.; Wolf, K.; Zahour, J.; DiPaolo, R.J.; Teague, R.M. Checkpoint blockade immunotherapy enhances the frequency and effector function of murine tumor-infiltrating T cells but does not alter TCRβ diversity. Cancer Immunol. Immunother. 2019, 68, 1095–1106. [Google Scholar] [CrossRef]
- Lozano, A.X.; Chaudhuri, A.A.; Nene, A.; Bacchiocchi, A.; Earland, N.; Vesely, M.D.; Usmani, A.; Turner, B.E.; Steen, C.B.; Luca, B.A.; et al. T cell characteristics associated with toxicity to immune checkpoint blockade in patients with melanoma. Nat. Med. 2022, 28, 353–362. [Google Scholar] [CrossRef]
- Balbach, M.L.; Axelrod, M.L.; Balko, J.M.; Bankhead, A.; Shaffer, T.; Lim, L.; Guo, J.; Hernandez, J.; Li, M.; Iams, W.T. Peripheral T-cell receptor repertoire dynamics in small cell lung cancer. Transl. Lung Cancer Res. 2023, 12, 257–265. [Google Scholar] [CrossRef]
- Laza-Briviesca, R.; Cruz-Bermúdez, A.; Nadal, E.; Insa, A.; García-Campelo, M.d.R.; Huidobro, G.; Dómine, M.; Majem, M.; Rodríguez-Abreu, D.; Martínez-Martí, A.; et al. Blood biomarkers associated to complete pathological response on NSCLC patients treated with neoadjuvant chemoimmunotherapy included in NADIM clinical trial. Clin. Transl. Med. 2021, 11, e491. [Google Scholar] [CrossRef]
- Badoual, C.; Bouchaud, G.; Agueznay, N.E.H.; Mortier, E.; Hans, S.; Gey, A.; Fernani, F.; Peyrard, S.; -Puig, P.L.; Bruneval, P.; et al. The soluble alpha chain of interleukin-15 receptor: A proinflammatory molecule associated with tumor progression in head and neck cancer. Cancer Res. 2008, 68, 3907–3914. [Google Scholar] [CrossRef]
- Seike, M.; Yanaihara, N.; Bowman, E.D.; Zanetti, K.A.; Budhu, A.; Kumamoto, K.; Mechanic, L.E.; Matsumoto, S.; Yokota, J.; Shibata, T.; et al. Use of a cytokine gene expression signature in lung adenocarcinoma and the surrounding tissue as a prognostic classifier. J. Natl. Cancer Inst. 2007, 99, 1257–1269. [Google Scholar] [CrossRef]
- Fiore, P.F.; Di Matteo, S.; Tumino, N.; Mariotti, F.R.; Pietra, G.; Ottonello, S.; Negrini, S.; Bottazzi, B.; Moretta, L.; Mortier, E.; et al. Interleukin-15 and cancer: Some solved and many unsolved questions. J. Immunother. Cancer 2020, 8, e001428. [Google Scholar] [CrossRef]
- Mlecnik, B.; Bindea, G.; Angell, H.K.; Sasso, M.S.; Obenauf, A.C.; Fredriksen, T.; Lafontaine, L.; Bilocq, A.M.; Kirilovsky, A.; Tosolini, M.; et al. Functional network pipeline reveals genetic determinants associated with in situ lymphocyte proliferation and survival of cancer patients. Sci. Transl. Med. 2014, 6, 228ra37. [Google Scholar] [CrossRef]
- Mao, X.-C.; Yang, C.-C.; Yang, Y.-F.; Yan, L.-J.; Ding, Z.-N.; Liu, H.; Yan, Y.-C.; Dong, Z.-R.; Wang, D.-X.; Li, T. Peripheral cytokine levels as novel predictors of survival in cancer patients treated with immune checkpoint inhibitors: A systematic review and meta-analysis. Front. Immunol. 2022, 13, 884592. [Google Scholar] [CrossRef]
- Ni, G.; Zhang, L.; Yang, X.; Li, H.; Ma, B.; Walton, S.; Wu, X.; Yuan, J.; Wang, T.; Liu, X. Targeting interleukin-10 signalling for cancer immunotherapy, a promising and complicated task. Hum. Vaccin. Immunother. 2020, 16, 2328–2332. Available online: https://pubmed.ncbi.nlm.nih.gov/32159421 (accessed on 11 March 2020). [CrossRef]
- Brooks, D.G.; Ha, S.-J.; Elsaesser, H.; Sharpe, A.H.; Freeman, G.J.; Oldstone, M.B.A. IL-10 and PD-L1 operate through distinct pathways to suppress T-cell activity during persistent viral infection. Proc. Natl. Acad. Sci. USA 2008, 105, 20428–20433. [Google Scholar] [CrossRef]
- Lamichhane, P.; Karyampudi, L.; Shreeder, B.; Krempski, J.; Bahr, D.; Daum, J.; Kalli, K.R.; Goode, E.L.; Block, M.S.; Cannon, M.J.; et al. IL10 Release upon PD-1 Blockade Sustains Immunosuppression in Ovarian Cancer. Cancer Res. 2017, 77, 6667–6678. [Google Scholar] [CrossRef]
- Sun, Z.; Fourcade, J.; Pagliano, O.; Chauvin, J.-M.; Sander, C.; Kirkwood, J.M.; Zarour, H.M. IL10 and PD-1 Cooperate to Limit the Activity of Tumor-Specific CD8+ T Cells. Cancer Res. 2015, 75, 1635–1644. [Google Scholar] [CrossRef]
- Naing, A.; Papadopoulos, K.P.; Autio, K.A.; Ott, P.A.; Patel, M.R.; Wong, D.J.; Falchook, C.S.; Pant, S.; Whiteside, M.; Rasco, D.R.; et al. Safety, Antitumor Activity, and Immune Activation of Pegylated Recombinant Human Interleukin-10 (AM0010) in Patients with Advanced Solid Tumors. J. Clin. Oncol. 2016, 34, 3562–3569. [Google Scholar] [CrossRef]
- Castro, F.; Cardoso, A.P.; Gonçalves, R.M.; Serre, K.; Oliveira, M.J. Interferon-gamma at the crossroads of tumor immune surveillance or evasion. Front. Immunol. 2018, 9, 847. [Google Scholar] [CrossRef]
- Chen, H.; Liakou, C.I.; Kamat, A.; Pettaway, C.; Ward, J.F.; Tang, D.N.; Sun, J.; Jungbluth, A.A.; Troncoso, P.; Logothetis, C.; et al. Anti-CTLA-4 therapy results in higher CD4+ICOShi T cell frequency and IFN-gamma levels in both nonmalignant and malignant prostate tissues. Proc. Natl. Acad. Sci. USA 2009, 106, 2729–2734. [Google Scholar] [CrossRef]
- Peng, W.; Liu, C.; Xu, C.; Lou, Y.; Chen, J.; Yang, Y.; Yagita, H.; Overwijk, W.W.; Lizée, G.; Radvanyi, L.; et al. PD-1 blockade enhances T-cell migration to tumors by elevating IFN-γ inducible chemokines. Cancer Res. 2012, 72, 5209–5218. [Google Scholar] [CrossRef]
- Higgs, B.W.; Morehouse, C.A.; Streicher, K.; Brohawn, P.Z.; Pilataxi, F.; Gupta, A.; Ranade, K. Interferon Gamma Messenger RNA Signature in Tumor Biopsies Predicts Outcomes in Patients with Non-Small Cell Lung Carcinoma or Urothelial Cancer Treated with Durvalumab. Clin. Cancer Res. 2018, 24, 3857–3866. [Google Scholar] [CrossRef]
- Karachaliou, N.; Gonzalez-Cao, M.; Crespo, G.; Drozdowskyj, A.; Aldeguer, E.; Gimenez-Capitan, A.; Teixido, C.; Molina-Vila, M.A.; Viteri, S.; de los Llanos Gil, M.; et al. Interferon gamma, an important marker of response to immune checkpoint blockade in non-small cell lung cancer and melanoma patients. Ther. Adv. Med. Oncol. 2018, 10, 1758834017749748. [Google Scholar] [CrossRef] [PubMed]
- Toiyama, Y.; Fujikawa, H.; Kawamura, M.; Matsushita, K.; Saigusa, S.; Tanaka, K.; Inoue, Y.; Uchida, K.; Mohri, Y.; Kusunoki, M. Evaluation of CXCL10 as a novel serum marker for predicting liver metastasis and prognosis in colorectal cancer. Int. J. Oncol. 2012, 40, 560–566. [Google Scholar] [CrossRef]
- Hilpert, J.; Grosse-Hovest, L.; Grünebach, F.; Buechele, C.; Nuebling, T.; Raum, T.; Steinle, A.; Salih, H.R. Comprehensive analysis of NKG2D ligand expression and release in leukemia: Implications for NKG2D-mediated NK cell responses. J. Immunol. 2012, 189, 1360–1371. [Google Scholar] [CrossRef]
- Salih, H.R.; Goehlsdorf, D.; Steinle, A. Release of MICB Molecules by Tumor Cells: Mechanism and Soluble MICB in Sera of Cancer Patients. Hum. Immunol. 2006, 67, 188–195. Available online: https://www.sciencedirect.com/science/article/pii/S0198885906000292 (accessed on 26 June 2024). [CrossRef]
- Waldhauer, I.; Steinle, A. Proteolytic release of soluble UL16-binding protein 2 from tumor cells. Cancer Res. 2006, 66, 2520–2526. [Google Scholar] [CrossRef]
- Madjd, Z.; Spendlove, I.; Moss, R.; Bevin, S.; E Pinder, S.; Watson, N.F.S.; Ellis, I.; Durrant, L.G. Upregulation of MICA on high-grade invasive operable breast carcinoma. Cancer Immun. 2007, 7, 17. [Google Scholar]
- Li, K.; Mandai, M.; Hamanishi, J.; Matsumura, N.; Suzuki, A.; Yagi, H.; Yamaguchi, K.; Baba, T.; Fujii, S.; Konishi, I. Clinical significance of the NKG2D ligands, MICA/B and ULBP2 in ovarian cancer: High expression of ULBP2 is an indicator of poor prognosis. Cancer Immunol. Immunother. 2009, 58, 641–652. [Google Scholar] [CrossRef] [PubMed]
- Wensveen, F.M.; Jelenčić, V.; Polić, B. NKG2D: A master regulator of immune cell responsiveness. Front. Immunol. 2018, 9, 441. [Google Scholar] [CrossRef] [PubMed]
- Le Bert, N.; Gasser, S. Advances in NKG2D ligand recognition and responses by NK cells. Immunol. Cell Biol. 2014, 92, 230–236. [Google Scholar] [CrossRef]
- Van Tong, H.; Song, L.H.; Hoan, N.X.; Cuong, B.K.; Sy, B.T.; Son, H.A.; Quyet, D.; Binh, V.Q.; Kremsner, P.G.; Bock, C.T.; et al. Soluble MICB protein levels and platelet counts during hepatitis B virus infection and response to hepatocellular carcinoma treatment. BMC Infect. Dis. 2015, 15, 25. [Google Scholar] [CrossRef]
- Diestelhorst, J.; Junge, N.; Jonigk, D.; Schlue, J.; Falk, C.S.; Manns, M.P.; Baumann, U.; Jaeckel, E.; Taubert, R. Baseline IL-2 and the AIH score can predict the response to standard therapy in paediatric autoimmune hepatitis. Sci. Rep. 2018, 8, 419. [Google Scholar] [CrossRef] [PubMed]
- Boyman, O.; Sprent, J. The role of interleukin-2 during homeostasis and activation of the immune system. Nat. Rev. Immunol. 2012, 12, 180–190. [Google Scholar] [CrossRef] [PubMed]
- Waldmann, T.A. The biology of interleukin-2 and interleukin-15: Implications for cancer therapy and vaccine design. Nat. Rev. Immunol. 2006, 6, 595–601. [Google Scholar] [CrossRef] [PubMed]

| Variable | Total: 55 Patients (N) | (%) |
|---|---|---|
| Sex | ||
| Males | 39 | 70.9 |
| Female | 16 | 29.1 |
| Age | ||
| Mean age: 65.02 | ||
| <5 | 47 | 85 |
| ≥75 | 8 | 15 |
| ECOG | ||
| ECOG 0 | 36 | 65.5 |
| ECOG 1 | 19 | 34.5 |
| IMC | ||
| <30 kg/m2 | 46 | 83.6 |
| ≥30 kg/m2 | 9 | 16.4 |
| Race | ||
| Caucasian | 54 | 98.2 |
| Others | 1 | 1.8 |
| Smoking Habit | ||
| Never smoker | 2 | 3.60 |
| Former smoker/Current smoker | 53 | 96.40 |
| PD-L1 | ||
| <1% | 10 | 18.20 |
| 1–49% | 21 | 38.20 |
| ≥50% | 16 | 29.10 |
| Unknown | 8 | 14.50 |
| LDH | ||
| Normal (≤214 U/L) | 35 | 63.60 |
| High (>214 U/L) | 20 | 36.40 |
| LIPI Score | ||
| Poor | 27 | 49.10 |
| Intermediate | 27 | 49.10 |
| Good | 1 | 1.80 |
| Histology | ||
| Adenocarcinoma | 22 | 40 |
| Squamous | 33 | 60 |
| Tumor Stage | ||
| III | 16 | 29.1 |
| IV | 39 | 70.9 |
| Treatment Indication | ||
| Locally advanced | 14 | 25.50 |
| First line | 18 | 32.70 |
| Second line or more | 23 | 41.80 |
| ICI | ||
| Durvalumab | 14 | 25.50 |
| Pembrolizumab | 21 | 38.20 |
| Atezolizumab | 18 | 32.70 |
| Nivolumab | 2 | 3.60 |
| ICI Response | ||
| Complete response (CR) | 10 | 18.80 |
| Partial response (PR) | 13 | 23.60 |
| Stable disease (SD) | 12 | 21.80 |
| Progressive disease (PD) | 15 | 27.30 |
| Not evaluable (NE) | 5 | |
| Death | ||
| Yes | 32 | 58.20 |
| No | 23 | 41.80 |
| Immune-Mediated Toxicity | ||
| Yes | 25 | 45.50 |
| No | 30 | 54.50 |
| Immune-Mediated Toxicity | ||
| Skin | 6 | 24 |
| Pneumonitis | 6 | 24 |
| Endocrine | 5 | 20 |
| Musculoskeletal | 5 | 20 |
| Renal | 4 | 16 |
| Liver | 3 | 12 |
| Colitis | 2 | 8 |
| Overall Survival | Progression-Free Survival | |
|---|---|---|
| IC (11.13–26.87) | IC (2.81–17.19) | |
| p value | p value | |
| Sex | ||
| Males | 0.396 | 0.646 |
| Female | ||
| Age | ||
| Mean age: 65.02 | 0.065 | |
| <75 | 0.170 | |
| ≥75 | ||
| ECOG | ||
| ECOG 0 | 0.000 | 0.643 |
| ECOG 1 | ||
| IMC | ||
| <30 kg/m2 | 0.695 | 0.889 |
| ≥30 kg/m2 | ||
| Race | ||
| Caucasian | 0.196 | 0.113 |
| Others | ||
| Smoking Habit | ||
| Never smoker | 0.165 | 0.528 |
| Former smoker/Current smoker | ||
| PD-L1 | ||
| <1% | 0.194 | 0.389 |
| 1–49% | ||
| ≥50% | ||
| Unknown | ||
| LDH | ||
| Normal (≤214 U/L) | 0.017 | 0.086 |
| High (>214 U/L) | ||
| LIPI Score | ||
| Poor | 0.000 | 0.005 |
| Intermediate | ||
| Good | ||
| Histology | ||
| Adenocarcinoma | 0.487 | 0.713 |
| Squamous | ||
| Tumor Stage | ||
| III | 0.000 | 0.034 |
| IV | ||
| Treatment Indication | ||
| Locally advanced | 0.000 | 0.076 |
| First line | ||
| Second line or more | ||
| ICI | ||
| Durvalumab | 0.000 | 0.354 |
| Pembrolizumab | ||
| Atezolizumab | ||
| Nivolumab | ||
| ICI Response | ||
| Complete response (CR) | 0.000 | 0.000 |
| Partial response (PR) | ||
| Stable disease (SD) | ||
| Progressive disease (PD) | ||
| Not evaluable (NE) | ||
| Immune-Mediated Toxicity | ||
| Yes | 0.051 | 0.030 |
| No | ||
| Immune-Mediated Toxicity | ||
| Skin | ||
| Pneumonitis | 0.588 | 0.697 |
| Endocrine | ||
| Musculoskeletal | ||
| Renal | ||
| Liver | ||
| Colitis |
| Cytokine | Median | Mean | Standard Deviation | Interquartile Range |
|---|---|---|---|---|
| MICA | 119.5 | 128.0 | 49.5 | 44.9 |
| MICB | 117.4 | 126.7 | 42.4 | 49.6 |
| ULBP1 | 21.0 | 42.7 | 96.7 | 16.1 |
| ULBP2 | 157.5 | 159.5 | 22.9 | 27.9 |
| CXCL10 | 16.4 | 19.0 | 11.0 | 11.1 |
| IL10 | 2.8 | 6.7 | 9.2 | 9.9 |
| ULBP4 | 0.0 | 27.6 | 81.4 | 20.6 |
| IFNγ | 25.5 | 26.5 | 8.0 | 5.1 |
| IL4 | 0.0 | 18.0 | 23.8 | 37.3 |
| IL2 | 26.1 | 26.7 | 3.5 | 2.9 |
| IL15 | 6.7 | 7.0 | 3.8 | 2.8 |
| IL12 | 0.0 | 137.7 | 412.2 | 153.9 |
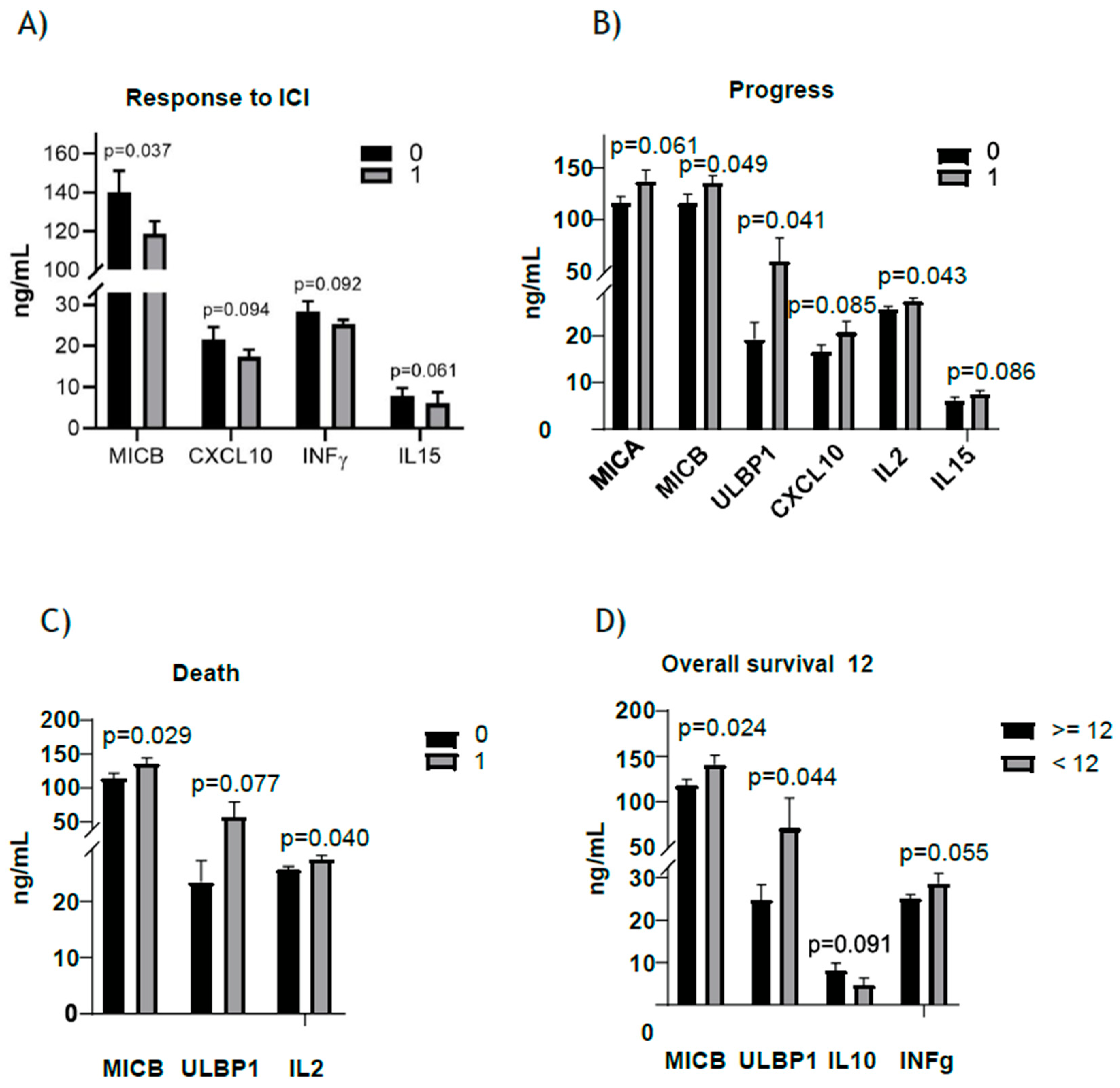
| Cytokines | Response to ICI | N | Mean | p Value |
|---|---|---|---|---|
| MICA | No | 20 | 133.7 | 0.516 |
| Yes | 34 | 124.6 | ||
| MICB | No | 20 | 140.2 | 0.037 |
| Yes | 34 | 118.8 | ||
| CXCL10 | No | 20 | 21.6 | 0.094 |
| Yes | 34 | 17.5 | ||
| IFNγ | No | 20 | 28.4 | 0.092 |
| Yes | 34 | 25.4 | ||
| ULBP1 | No | 20 | 52.1 | 0.589 |
| Yes | 34 | 37.2 | ||
| ULBP2 | No | 20 | 162.8 | 0.206 |
| Yes | 34 | 157.5 | ||
| IL10 | No | 20 | 4.0 | 0.162 |
| Yes | 34 | 5.6 | ||
| ULBP4 | No | 20 | 9.2 | 0.302 |
| Yes | 34 | 7.1 | ||
| IL4 | No | 20 | 19.6 | 0.356 |
| Yes | 34 | 17.0 | ||
| IL2 | No | 20 | 26.3 | 0.319 |
| Yes | 34 | 26.0 | ||
| IL15 | No | 20 | 7.4 | 0.061 |
| Yes | 34 | 6.2 | ||
| IL12 | No | 20 | 204.6 | 0.183 |
| Yes | 34 | 98.4 |
| Covariable | Coefficient Estimate (Bi) | SD Estimation | Sig. | Exp (B) (HR.) | IC (HR) 95% |
|---|---|---|---|---|---|
| TI Locally advanced | - | - | 0.006 | - | - |
| TI First line | 2.363 | 0.901 | 0.009 | 10.627 | 1.816–62.181 |
| TI Second line or more | 2.708 | 0.852 | 0.001 | 14.998 | 2.823–79.679 |
| ECOG [1] | 0.915 | 0.393 | 0.020 | 2.496 | 1.155–5.391 |
| Staging (IV) | 2.295 | 1.081 | 0.034 | 9.929 | 1.193–82.625 |
| IL-15 (>6.7) | 1.098 | 0.401 | 0.006 | 3.000 | 1.368–6.578 |
| Atezolizumab | - | - | 0.008 | - | - |
| Nivolumab | −1.615 | 1.082 | 0.135 | 0.199 | 0.024–1.657 |
| Pembrolizumab | −1.457 | 0.444 | 0.001 | 0.233 | 0.098–0.557 |
| Durvalumab | −0.790 | 1.277 | 0.536 | 0.454 | 0.037–5.540 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Sesma, A.; Pardo, J.; Isla, D.; M. Gálvez, E.; Gascón-Ruiz, M.; Martínez-Lostao, L.; Moratiel, A.; Paño-Pardo, J.R.; Quílez, E.; Torres-Ramón, I.; et al. Peripheral Blood TCRβ Repertoire, IL15, IL2 and Soluble Ligands for NKG2D Activating Receptor Predict Efficacy of Immune Checkpoint Inhibitors in Lung Cancer. Cancers 2024, 16, 2798. https://doi.org/10.3390/cancers16162798
Sesma A, Pardo J, Isla D, M. Gálvez E, Gascón-Ruiz M, Martínez-Lostao L, Moratiel A, Paño-Pardo JR, Quílez E, Torres-Ramón I, et al. Peripheral Blood TCRβ Repertoire, IL15, IL2 and Soluble Ligands for NKG2D Activating Receptor Predict Efficacy of Immune Checkpoint Inhibitors in Lung Cancer. Cancers. 2024; 16(16):2798. https://doi.org/10.3390/cancers16162798
Chicago/Turabian StyleSesma, Andrea, Julian Pardo, Dolores Isla, Eva M. Gálvez, Marta Gascón-Ruiz, Luis Martínez-Lostao, Alba Moratiel, J. Ramón Paño-Pardo, Elisa Quílez, Irene Torres-Ramón, and et al. 2024. "Peripheral Blood TCRβ Repertoire, IL15, IL2 and Soluble Ligands for NKG2D Activating Receptor Predict Efficacy of Immune Checkpoint Inhibitors in Lung Cancer" Cancers 16, no. 16: 2798. https://doi.org/10.3390/cancers16162798
APA StyleSesma, A., Pardo, J., Isla, D., M. Gálvez, E., Gascón-Ruiz, M., Martínez-Lostao, L., Moratiel, A., Paño-Pardo, J. R., Quílez, E., Torres-Ramón, I., Yubero, A., Zapata-García, M., Domingo, M. P., Esteban, P., Sanz Pamplona, R., Lastra, R., & Ramírez-Labrada, A. (2024). Peripheral Blood TCRβ Repertoire, IL15, IL2 and Soluble Ligands for NKG2D Activating Receptor Predict Efficacy of Immune Checkpoint Inhibitors in Lung Cancer. Cancers, 16(16), 2798. https://doi.org/10.3390/cancers16162798





