Mucins as Potential Biomarkers for Early Detection of Cancer
Abstract
Simple Summary
Abstract
1. Introduction
2. Mucin Deregulation in Cancer
3. Mucins as Cancer Biomarkers
4. Lung Cancer Diagnosis and Mucin Biomarkers
5. Breast Cancer Early Detection and Mucin Biomarkers
6. Role of Mucins in the Early Detection of Ovarian Cancer
7. Colorectal Cancer Screening and Mucin Biomarkers
8. Mucins in Early Detection of Pancreatic Ductal Adenocarcinoma
9. Mucin Biomarkers in Prostate Cancer Diagnosis
10. Mucin Biomarkers in Liver Cancer
11. Mucin Autoantibodies for Early Cancer Detection
12. Circulating Exosomes as a Cancer Biomarker
13. Conclusions and Future Perspective
Author Contributions
Funding
Conflicts of Interest
Abbreviations
References
- Crosby, D.; Bhatia, S.; Brindle, K.M.; Coussens, L.M.; Dive, C.; Emberton, M.; Esener, S.; Fitzgerald, R.C.; Gambhir, S.S.; Kuhn, P.; et al. Early detection of cancer. Science 2022, 375, eaay9040. [Google Scholar] [CrossRef]
- Sung, H.; Ferlay, J.; Siegel, R.L.; Laversanne, M.; Soerjomataram, I.; Jemal, A.; Bray, F. Global Cancer Statistics 2020: GLOBOCAN Estimates of Incidence and Mortality Worldwide for 36 Cancers in 185 Countries. CA A Cancer J. Clin. 2021, 71, 209–249. [Google Scholar] [CrossRef] [PubMed]
- Siegel, R.L.; Miller, K.D.; Wagle, N.S.; Jemal, A. Cancer statistics, 2023. CA A Cancer J. Clin. 2023, 73, 17–48. [Google Scholar] [CrossRef] [PubMed]
- Pashayan, N.; Pharoah, P.D.P. The challenge of early detection in cancer. Science 2020, 368, 589–590. [Google Scholar] [CrossRef] [PubMed]
- Zou, J.; Wang, E. Cancer Biomarker Discovery for Precision Medicine: New Progress. Curr. Med. Chem. 2019, 26, 7655–7671. [Google Scholar] [CrossRef]
- Mäbert, K.; Cojoc, M.; Peitzsch, C.; Kurth, I.; Souchelnytskyi, S.; Dubrovska, A. Cancer biomarker discovery: Current status and future perspectives. Int. J. Radiat. Biol. 2014, 90, 659–677. [Google Scholar] [CrossRef]
- Ishige, F.; Hoshino, I.; Iwatate, Y.; Chiba, S.; Arimitsu, H.; Yanagibashi, H.; Nagase, H.; Takayama, W. MIR1246 in body fluids as a biomarker for pancreatic cancer. Sci. Rep. 2020, 10, 8723. [Google Scholar] [CrossRef]
- Kaur, J.; Jacobs, R.; Huang, Y.; Salvo, N.; Politis, C. Salivary biomarkers for oral cancer and pre-cancer screening: A review. Clin. Oral. Investig. 2018, 22, 633–640. [Google Scholar] [CrossRef] [PubMed]
- Lam, W.K.J.; Chan, K.C.A. Plasma DNA for early cancer detection—Opportunities and challenges. Expert Rev. Mol. Diagn. 2019, 19, 5–7. [Google Scholar] [CrossRef]
- Macdonald, I.K.; Parsy-Kowalska, C.B.; Chapman, C.J. Autoantibodies: Opportunities for Early Cancer Detection. Trends Cancer 2017, 3, 198–213. [Google Scholar] [CrossRef]
- Shimomura, A.; Shiino, S.; Kawauchi, J.; Takizawa, S.; Sakamoto, H.; Matsuzaki, J.; Ono, M.; Takeshita, F.; Niida, S.; Shimizu, C.; et al. Novel combination of serum microRNA for detecting breast cancer in the early stage. Cancer Sci 2016, 107, 326–334. [Google Scholar] [CrossRef]
- Tang, Q.; Cheng, J.; Cao, X.; Surowy, H.; Burwinkel, B. Blood-based DNA methylation as biomarker for breast cancer: A systematic review. Clin. Epigenet. 2016, 8, 115. [Google Scholar] [CrossRef] [PubMed]
- Rachagani, S.; Torres, M.P.; Moniaux, N.; Batra, S.K. Current status of mucins in the diagnosis and therapy of cancer. Biofactors 2009, 35, 509–527. [Google Scholar] [CrossRef]
- Filippou, P.S.; Ren, A.H.; Korbakis, D.; Dimitrakopoulos, L.; Soosaipillai, A.; Barak, V.; Frenkel, S.; Pe’er, J.; Lotem, M.; Merims, S.; et al. Exploring the potential of mucin 13 (MUC13) as a biomarker for carcinomas and other diseases. Clin. Chem. Lab. Med. 2018, 56, 1945–1953. [Google Scholar] [CrossRef]
- Attallah, A.M.; El-Far, M.; Ibrahim, A.R.; El-desouky, M.A.; Omran, M.M.; Elbendary, M.S.; Attallah, K.A.; Qura, E.R.; Abdallah, S.O. Clinical value of a diagnostic score for colon cancer based on serum CEA, CA19-9, cytokeratin-1 and mucin-1. Br. J. Biomed. Sci. 2018, 75, 122–127. [Google Scholar] [CrossRef] [PubMed]
- Wang, S.; You, L.; Dai, M.; Zhao, Y. Mucins in pancreatic cancer: A well-established but promising family for diagnosis, prognosis and therapy. J. Cell. Mol. Med. 2020, 24, 10279–10289. [Google Scholar] [CrossRef] [PubMed]
- Björkman, K.; Mustonen, H.; Kaprio, T.; Haglund, C.; Böckelman, C. Mucin 16 and kallikrein 13 as potential prognostic factors in colon cancer: Results of an oncological 92-multiplex immunoassay. Tumor Biol. 2019, 41, 1010428319860728. [Google Scholar] [CrossRef]
- Bugge, A.S.; Kongerud, J.S.; Valberg, M.; Solberg, S.K.; Brustugun, O.T.; Lund, M.B. Long-term survival after surgical resection for non-small cell lung cancer. Eur. Respir. J. 2017, 50, PA4292. [Google Scholar] [CrossRef]
- Kim, B.H. Surgical resection versus ablation for early hepatocellular carcinoma: The debate is still open. Clin. Mol. Hepatol. 2022, 28, 174–176. [Google Scholar] [CrossRef]
- Oettle, H.; Neuhaus, P.; Hochhaus, A.; Hartmann, J.T.; Gellert, K.; Ridwelski, K.; Niedergethmann, M.; Zülke, C.; Fahlke, J.; Arning, M.B.; et al. Adjuvant Chemotherapy With Gemcitabine and Long-term Outcomes Among Patients With Resected Pancreatic Cancer: The CONKO-001 Randomized Trial. JAMA 2013, 310, 1473–1481. [Google Scholar] [CrossRef]
- Redaniel, M.T.; Martin, R.M.; Gillatt, D.; Wade, J.; Jeffreys, M. Time from diagnosis to surgery and prostate cancer survival: A retrospective cohort study. BMC Cancer 2013, 13, 559. [Google Scholar] [CrossRef]
- Tewari, K.S.; Java, J.J.; Eskander, R.N.; Monk, B.J.; Burger, R.A. Early initiation of chemotherapy following complete resection of advanced ovarian cancer associated with improved survival: NRG Oncology/Gynecologic Oncology Group study. Ann. Oncol. 2016, 27, 114–121. [Google Scholar] [CrossRef]
- van den Berg, I.; Coebergh van den Braak, R.R.J.; van Vugt, J.L.A.; Ijzermans, J.N.M.; Buettner, S. Actual survival after resection of primary colorectal cancer: Results from a prospective multicenter study. World J. Surg. Oncol. 2021, 19, 96. [Google Scholar] [CrossRef]
- Magnus, N.; Garnier, D.; Meehan, B.; McGraw, S.; Lee, T.H.; Caron, M.; Bourque, G.; Milsom, C.; Jabado, N.; Trasler, J.; et al. Tissue factor expression provokes escape from tumor dormancy and leads to genomic alterations. Proc. Natl. Acad. Sci. USA 2014, 111, 3544–3549. [Google Scholar] [CrossRef] [PubMed]
- Hisada, Y.; Mackman, N. Tissue Factor and Cancer: Regulation, Tumor Growth, and Metastasis. Semin. Thromb. Hemost. 2019, 45, 385–395. [Google Scholar] [CrossRef]
- Schneider, G.; Schmidt-Supprian, M.; Rad, R.; Saur, D. Tissue-specific tumorigenesis: Context matters. Nat. Rev. Cancer 2017, 17, 239–253. [Google Scholar] [CrossRef] [PubMed]
- Gum, J.R., Jr. Mucin genes and the proteins they encode: Structure, diversity, and regulation. Am. J. Respir. Cell. Mol. Biol. 1992, 7, 557–564. [Google Scholar] [CrossRef] [PubMed]
- Ho, S.B.; Niehans, G.A.; Lyftogt, C.; Yan, P.S.; Cherwitz, D.L.; Gum, E.T.; Dahiya, R.; Kim, Y.S. Heterogeneity of mucin gene expression in normal and neoplastic tissues. Cancer Res. 1993, 53, 641–651. [Google Scholar]
- Jonckheere, N.; Skrypek, N.; Frénois, F.; Van Seuningen, I. Membrane-bound mucin modular domains: From structure to function. Biochimie 2013, 95, 1077–1086. [Google Scholar] [CrossRef]
- Andrianifahanana, M.; Moniaux, N.; Batra, S.K. Regulation of mucin expression: Mechanistic aspects and implications for cancer and inflammatory diseases. Biochim. Biophys. Acta 2006, 1765, 189–222. [Google Scholar] [CrossRef]
- Jonckheere, N.; Vincent, A.; Neve, B.; Van Seuningen, I. Mucin expression, epigenetic regulation and patient survival: A toolkit of prognostic biomarkers in epithelial cancers. Biochim. Biophys. Acta Rev. Cancer 2021, 1876, 188538. [Google Scholar] [CrossRef] [PubMed]
- Kim, Y.S.; Gum, J., Jr.; Brockhausen, I. Mucin glycoproteins in neoplasia. Glycoconj. J. 1996, 13, 693–707. [Google Scholar] [CrossRef]
- Desseyn, J.L.; Tetaert, D.; Gouyer, V. Architecture of the large membrane-bound mucins. Gene 2008, 410, 215–222. [Google Scholar] [CrossRef]
- Spencer, D.I.R.; Price, M.R.; Tendler, S.J.B.; De Matteis, C.I.; Stadie, T.; Hanisch, F.-G. Effect of glycosylation of a synthetic MUC1 mucin-core-related peptide on recognition by anti-mucin antibodies. Cancer Lett. 1996, 100, 11–15. [Google Scholar] [CrossRef] [PubMed]
- Burchell, J.M.; Beatson, R.; Graham, R.; Taylor-Papadimitriou, J.; Tajadura-Ortega, V. O-linked mucin-type glycosylation in breast cancer. Biochem. Soc. Trans. 2018, 46, 779–788. [Google Scholar] [CrossRef]
- Nagata, K.; Horinouchi, M.; Saitou, M.; Higashi, M.; Nomoto, M.; Goto, M.; Yonezawa, S. Mucin expression profile in pancreatic cancer and the precursor lesions. J. Hepatobiliary Pancreat. Surg. 2007, 14, 243–254. [Google Scholar] [CrossRef] [PubMed]
- Lakshmanan, I.; Ponnusamy, M.P.; Macha, M.A.; Haridas, D.; Majhi, P.D.; Kaur, S.; Jain, M.; Batra, S.K.; Ganti, A.K. Mucins in lung cancer: Diagnostic, prognostic, and therapeutic implications. J. Thorac. Oncol. 2015, 10, 19–27. [Google Scholar] [CrossRef]
- Giuntoli, R.L., 2nd; Rodriguez, G.C.; Whitaker, R.S.; Dodge, R.; Voynow, J.A. Mucin gene expression in ovarian cancers. Cancer Res 1998, 58, 5546–5550. [Google Scholar]
- Kaur, S.; Kumar, S.; Momi, N.; Sasson, A.R.; Batra, S.K. Mucins in pancreatic cancer and its microenvironment. Nat. Rev. Gastroenterol. Hepatol. 2013, 10, 607–620. [Google Scholar] [CrossRef]
- Yan, P.S.; Ho, S.B.; Itzkowitz, S.H.; Byrd, J.C.; Siddiqui, B.; Kim, Y.S. Expression of native and deglycosylated colon cancer mucin antigens in normal and malignant epithelial tissues. Lab. Investig. 1990, 63, 698–706. [Google Scholar] [PubMed]
- Krishn, S.R.; Kaur, S.; Smith, L.M.; Johansson, S.L.; Jain, M.; Patel, A.; Gautam, S.K.; Hollingsworth, M.A.; Mandel, U.; Clausen, H.; et al. Mucins and associated glycan signatures in colon adenoma-carcinoma sequence: Prospective pathological implication(s) for early diagnosis of colon cancer. Cancer Lett. 2016, 374, 304–314. [Google Scholar] [CrossRef] [PubMed]
- Byrd, J.C.; Bresalier, R.S. Mucins and mucin binding proteins in colorectal cancer. Cancer Metastasis Rev. 2004, 23, 77–99. [Google Scholar] [CrossRef] [PubMed]
- Yonezawa, S.; Sato, E. Expression of mucin antigens in human cancers and its relationship with malignancy potential. Pathol. Int. 1997, 47, 813–830. [Google Scholar] [CrossRef]
- Diaz, L.K.; Wiley, E.L.; Morrow, M. Expression of epithelial mucins Muc1, Muc2, and Muc3 in ductal carcinoma in situ of the breast. Breast J. 2001, 7, 40–45. [Google Scholar] [CrossRef]
- Walsh, M.D.; Clendenning, M.; Williamson, E.; Pearson, S.-A.; Walters, R.J.; Nagler, B.; Packenas, D.; Win, A.K.; Hopper, J.L.; Jenkins, M.A.; et al. Expression of MUC2, MUC5AC, MUC5B, and MUC6 mucins in colorectal cancers and their association with the CpG island methylator phenotype. Mod. Pathol. 2013, 26, 1642–1656. [Google Scholar] [CrossRef]
- Bauer, A.K.; Umer, M.; Richardson, V.L.; Cumpian, A.M.; Harder, A.Q.; Khosravi, N.; Azzegagh, Z.; Hara, N.M.; Ehre, C.; Mohebnasab, M.; et al. Requirement for MUC5AC in KRAS-dependent lung carcinogenesis. JCI Insight 2018, 3, e120941. [Google Scholar] [CrossRef]
- Duruisseaux, M.; Antoine, M.; Rabbe, N.; Rodenas, A.; Mc Leer-Florin, A.; Lacave, R.; Poulot, V.; Duchêne, B.; Van Seuningen, I.; Cadranel, J.; et al. Lepidic predominant adenocarcinoma and invasive mucinous adenocarcinoma of the lung exhibit specific mucin expression in relation with oncogenic drivers. Lung Cancer 2017, 109, 92–100. [Google Scholar] [CrossRef]
- Vasseur, R.; Skrypek, N.; Duchêne, B.; Renaud, F.; Martínez-Maqueda, D.; Vincent, A.; Porchet, N.; Van Seuningen, I.; Jonckheere, N. The mucin MUC4 is a transcriptional and post-transcriptional target of K-ras oncogene in pancreatic cancer. Implication of MAPK/AP-1, NF-κB and RalB signaling pathways. Biochim. Biophys. Acta (BBA)—Gene Regul. Mech. 2015, 1849, 1375–1384. [Google Scholar] [CrossRef]
- Yamada, N.; Kitamoto, S.; Yokoyama, S.; Hamada, T.; Goto, M.; Tsutsumida, H.; Higashi, M.; Yonezawa, S. Epigenetic regulation of mucin genes in human cancers. Clin. Epigenet. 2011, 2, 85–96. [Google Scholar] [CrossRef]
- Tsutsumida, H.; Goto, M.; Kitajima, S.; Kubota, I.; Hirotsu, Y.; Wakimoto, J.; Batra, S.K.; Imai, K.; Yonezawa, S. MUC4 expression correlates with poor prognosis in small-sized lung adenocarcinoma. Lung Cancer 2007, 55, 195–203. [Google Scholar] [CrossRef] [PubMed]
- Saitou, M.; Goto, M.; Horinouchi, M.; Tamada, S.; Nagata, K.; Hamada, T.; Osako, M.; Takao, S.; Batra, S.K.; Aikou, T.; et al. MUC4 expression is a novel prognostic factor in patients with invasive ductal carcinoma of the pancreas. J. Clin. Pathol. 2005, 58, 845–852. [Google Scholar] [CrossRef] [PubMed]
- Lindemann, K.; Kristensen, G.; Mirza, M.R.; Davies, L.; Hilpert, F.; Romero, I.; Ayhan, A.; Burges, A.; Rubio, M.J.; Raspagliesi, F.; et al. Poor concordance between CA-125 and RECIST at the time of disease progression in patients with platinum-resistant ovarian cancer: Analysis of the AURELIA trial. Ann. Oncol. 2016, 27, 1505–1510. [Google Scholar] [CrossRef] [PubMed]
- Streppel, M.M.; Vincent, A.; Mukherjee, R.; Campbell, N.R.; Chen, S.H.; Konstantopoulos, K.; Goggins, M.G.; Van Seuningen, I.; Maitra, A.; Montgomery, E.A. Mucin 16 (cancer antigen 125) expression in human tissues and cell lines and correlation with clinical outcome in adenocarcinomas of the pancreas, esophagus, stomach, and colon. Hum. Pathol. 2012, 43, 1755–1763. [Google Scholar] [CrossRef]
- Manne, A.; Esnakula, A.; Abushahin, L.; Tsung, A. Understanding the Clinical Impact of MUC5AC Expression on Pancreatic Ductal Adenocarcinoma. Cancers 2021, 13, 3059. [Google Scholar] [CrossRef]
- Javanbakht, M.; Akhavanmoghadam, J.; Talaei, A.J.; Aghyani, M.; Mozafari, M.; Khedmat, L.; Mohebbi, M. Differential expression of two genes Oct-4 and MUC5AC associates with poor outcome in patients with gastric cancer. Clin. Exp. Pharm. Physiol 2017, 44, 1099–1105. [Google Scholar] [CrossRef]
- Dong, Y.; Zhou, L.; Zhao, D.; Li, K.; Liu, Z.; Che, N.; Liu, H. MUC5AC enhances tumor heterogeneity in lung adenocarcinoma with mucin production and is associated with poor prognosis. Jpn. J. Clin. Oncol. 2020, 50, 701–711. [Google Scholar] [CrossRef]
- Liu, D.; Chang, C.H.; Gold, D.V.; Goldenberg, D.M. Identification of PAM4 (clivatuzumab)-reactive epitope on MUC5AC: A promising biomarker and therapeutic target for pancreatic cancer. Oncotarget 2015, 6, 4274–4285. [Google Scholar] [CrossRef]
- Rico, S.D.; Höflmayer, D.; Büscheck, F.; Dum, D.; Luebke, A.M.; Kluth, M.; Hube-Magg, C.; Hinsch, A.; Möller-Koop, C.; Perez, D.; et al. Elevated MUC5AC expression is associated with mismatch repair deficiency and proximal tumor location but not with cancer progression in colon cancer. Med. Mol. Morphol. 2021, 54, 156–165. [Google Scholar] [CrossRef] [PubMed]
- Hollingsworth, M.A.; Swanson, B.J. Mucins in cancer: Protection and control of the cell surface. Nat. Rev. Cancer 2004, 4, 45–60. [Google Scholar] [CrossRef]
- Jing, X.; Liang, H.; Hao, C.; Yang, X.; Cui, X. Overexpression of MUC1 predicts poor prognosis in patients with breast cancer. Oncol. Rep. 2019, 41, 801–810. [Google Scholar] [CrossRef]
- Duncan, T.J.; Watson, N.F.; Al-Attar, A.H.; Scholefield, J.H.; Durrant, L.G. The role of MUC1 and MUC3 in the biology and prognosis of colorectal cancer. World J. Surg. Oncol. 2007, 5, 31. [Google Scholar] [CrossRef]
- Kufe, D.W. Mucins in cancer: Function, prognosis and therapy. Nat. Rev. Cancer 2009, 9, 874–885. [Google Scholar] [CrossRef]
- Hawkes, N. Cancer survival data emphasise importance of early diagnosis. BMJ 2019, 364, l408. [Google Scholar] [CrossRef] [PubMed]
- Trinidad, C.V.; Tetlow, A.L.; Bantis, L.E.; Godwin, A.K. Reducing Ovarian Cancer Mortality Through Early Detection: Approaches Using Circulating Biomarkers. Cancer Prev. Res. 2020, 13, 241. [Google Scholar] [CrossRef] [PubMed]
- Rodriguez, A. Contribution to the Screening and Early Detection in Breast Cancer. J. Glob. Oncol. 2018, 4, 130s. [Google Scholar] [CrossRef]
- Young, M.R.; Wagner, P.D.; Ghosh, S.; Rinaudo, J.A.; Baker, S.G.; Zaret, K.S.; Goggins, M.; Srivastava, S. Validation of Biomarkers for Early Detection of Pancreatic Cancer: Summary of The Alliance of Pancreatic Cancer Consortia for Biomarkers for Early Detection Workshop. Pancreas 2018, 47, 135–141. [Google Scholar] [CrossRef] [PubMed]
- Cancer Stat Facts: Lung and Bronchus Cancer. National Cancer Institute. Available online: https://seer.cancer.gov/statfacts/html/lungb.html (accessed on 26 December 2022).
- Wang, S.; Du, M.; Zhang, J.; Xu, W.; Yuan, Q.; Li, M.; Wang, J.; Zhu, H.; Wang, Y.; Wang, C.; et al. Tumor evolutionary trajectories during the acquisition of invasiveness in early stage lung adenocarcinoma. Nat. Commun. 2020, 11, 6083. [Google Scholar] [CrossRef]
- Khan, P.; Siddiqui, J.A.; Maurya, S.K.; Lakshmanan, I.; Jain, M.; Ganti, A.K.; Salgia, R.; Batra, S.K.; Nasser, M.W. Epigenetic landscape of small cell lung cancer: Small image of a giant recalcitrant disease. Semin. Cancer Biol. 2020, 83, 57–76. [Google Scholar] [CrossRef]
- Khan, P.; Siddiqui, J.A.; Lakshmanan, I.; Ganti, A.K.; Salgia, R.; Jain, M.; Batra, S.K.; Nasser, M.W. RNA-based therapies: A cog in the wheel of lung cancer defense. Mol. Cancer 2021, 20, 54. [Google Scholar] [CrossRef]
- Zhang, C.; Zhang, J.; Xu, F.P.; Wang, Y.G.; Xie, Z.; Su, J.; Dong, S.; Nie, Q.; Shao, Y.; Zhou, Q.; et al. Genomic Landscape and Immune Microenvironment Features of Preinvasive and Early Invasive Lung Adenocarcinoma. J. Thorac. Oncol. 2019, 14, 1912–1923. [Google Scholar] [CrossRef] [PubMed]
- Force, U.S.P.S.T.; Krist, A.H.; Davidson, K.W.; Mangione, C.M.; Barry, M.J.; Cabana, M.; Caughey, A.B.; Davis, E.M.; Donahue, K.E.; Doubeni, C.A.; et al. Screening for Lung Cancer: US Preventive Services Task Force Recommendation Statement. JAMA 2021, 325, 962–970. [Google Scholar] [CrossRef]
- Key Statistics for Lung Cancer. American Cancer Society. Available online: https://www.cancer.org/cancer/lung-cancer/about/key-statistics.html (accessed on 29 August 2021).
- de Groot, P.M.; Wu, C.C.; Carter, B.W.; Munden, R.F. The epidemiology of lung cancer. Transl. Lung Cancer Res. 2018, 7, 220–233. [Google Scholar] [CrossRef] [PubMed]
- Herbst, R.S.; Morgensztern, D.; Boshoff, C. The biology and management of non-small cell lung cancer. Nature 2018, 553, 446–454. [Google Scholar] [CrossRef]
- National Lung Screening Trial Research, T.; Aberle, D.R.; Adams, A.M.; Berg, C.D.; Black, W.C.; Clapp, J.D.; Fagerstrom, R.M.; Gareen, I.F.; Gatsonis, C.; Marcus, P.M.; et al. Reduced lung-cancer mortality with low-dose computed tomographic screening. N. Engl. J. Med. 2011, 365, 395–409. [Google Scholar] [CrossRef]
- de Koning, H.J.; van der Aalst, C.M.; de Jong, P.A.; Scholten, E.T.; Nackaerts, K.; Heuvelmans, M.A.; Lammers, J.J.; Weenink, C.; Yousaf-Khan, U.; Horeweg, N.; et al. Reduced Lung-Cancer Mortality with Volume CT Screening in a Randomized Trial. N. Engl. J. Med. 2020, 382, 503–513. [Google Scholar] [CrossRef]
- Mazzone, P.J.; Silvestri, G.A.; Patel, S.; Kanne, J.P.; Kinsinger, L.S.; Wiener, R.S.; Soo Hoo, G.; Detterbeck, F.C. Screening for Lung Cancer: CHEST Guideline and Expert Panel Report. Chest 2018, 153, 954–985. [Google Scholar] [CrossRef]
- Smith, R.A.; Andrews, K.S.; Brooks, D.; Fedewa, S.A.; Manassaram-Baptiste, D.; Saslow, D.; Brawley, O.W.; Wender, R.C. Cancer screening in the United States, 2018: A review of current American Cancer Society guidelines and current issues in cancer screening. CA Cancer J. Clin. 2018, 68, 297–316. [Google Scholar] [CrossRef] [PubMed]
- Liu, C.; Xiang, X.; Han, S.; Lim, H.Y.; Li, L.; Zhang, X.; Ma, Z.; Yang, L.; Guo, S.; Soo, R.; et al. Blood-based liquid biopsy: Insights into early detection and clinical management of lung cancer. Cancer Lett. 2022, 524, 91–102. [Google Scholar] [CrossRef]
- Sun, Y.; Huo, C.; Qiao, Z.; Shang, Z.; Uzzaman, A.; Liu, S.; Jiang, X.; Fan, L.Y.; Ji, L.; Guan, X.; et al. Comparative Proteomic Analysis of Exosomes and Microvesicles in Human Saliva for Lung Cancer. J. Proteome Res. 2018, 17, 1101–1107. [Google Scholar] [CrossRef]
- Rao, T.D.; Tian, H.; Ma, X.; Yan, X.; Thapi, S.; Schultz, N.; Rosales, N.; Monette, S.; Wang, A.; Hyman, D.M.; et al. Expression of the Carboxy-Terminal Portion of MUC16/CA125 Induces Transformation and Tumor Invasion. PLoS ONE 2015, 10, e0126633. [Google Scholar] [CrossRef]
- Xu, M.; Wang, D.C.; Wang, X.; Zhang, Y. Correlation between mucin biology and tumor heterogeneity in lung cancer. Semin. Cell Dev. Biol. 2017, 64, 73–78. [Google Scholar] [CrossRef] [PubMed]
- Rose, M.C. Mucins: Structure, function, and role in pulmonary diseases. Am. J. Physiol. 1992, 263, L413–L429. [Google Scholar] [CrossRef] [PubMed]
- Meldrum, O.W.; Chotirmall, S.H. Mucus, Microbiomes and Pulmonary Disease. Biomedicines 2021, 9, 675. [Google Scholar] [CrossRef]
- Smet, A.; Breugelmans, T.; Michiels, J.; Lamote, K.; Arras, W.; De Man, J.G.; Heyndrickx, L.; Hauner, A.; Huizing, M.; Malhotra-Kumar, S.; et al. A dynamic mucin mRNA signature associates with COVID-19 disease presentation and severity. JCI Insight 2021, 6, e151777. [Google Scholar] [CrossRef] [PubMed]
- Buisine, M.P.; Devisme, L.; Copin, M.C.; Durand-Reville, M.; Gosselin, B.; Aubert, J.P.; Porchet, N. Developmental mucin gene expression in the human respiratory tract. Am. J. Respir. Cell Mol. Biol. 1999, 20, 209–218. [Google Scholar] [CrossRef]
- Copin, M.C.; Buisine, M.P.; Devisme, L.; Leroy, X.; Escande, F.; Gosselin, B.; Aubert, J.P.; Porchet, N. Normal respiratory mucosa, precursor lesions and lung carcinomas: Differential expression of human mucin genes. Front. Biosci. 2001, 6, D1264–D1275. [Google Scholar] [CrossRef]
- Copin, M.C.; Devisme, L.; Buisine, M.P.; Marquette, C.H.; Wurtz, A.; Aubert, J.P.; Gosselin, B.; Porchet, N. From normal respiratory mucosa to epidermoid carcinoma: Expression of human mucin genes. Int. J. Cancer 2000, 86, 162–168. [Google Scholar] [CrossRef]
- Perrais, M.; Pigny, P.; Copin, M.C.; Aubert, J.P.; Van Seuningen, I. Induction of MUC2 and MUC5AC mucins by factors of the epidermal growth factor (EGF) family is mediated by EGF receptor/Ras/Raf/extracellular signal-regulated kinase cascade and Sp1. J. Biol. Chem. 2002, 277, 32258–32267. [Google Scholar] [CrossRef]
- Okuda, K.; Chen, G.; Subramani, D.B.; Wolf, M.; Gilmore, R.C.; Kato, T.; Radicioni, G.; Kesimer, M.; Chua, M.; Dang, H.; et al. Localization of Secretory Mucins MUC5AC and MUC5B in Normal/Healthy Human Airways. Am. J. Respir. Crit. Care Med. 2019, 199, 715–727. [Google Scholar] [CrossRef]
- Williams, S.J.; Wreschner, D.H.; Tran, M.; Eyre, H.J.; Sutherland, G.R.; McGuckin, M.A. Muc13, a novel human cell surface mucin expressed by epithelial and hemopoietic cells. J. Biol. Chem. 2001, 276, 18327–18336. [Google Scholar] [CrossRef] [PubMed]
- Davies, J.R.; Kirkham, S.; Svitacheva, N.; Thornton, D.J.; Carlstedt, I. MUC16 is produced in tracheal surface epithelium and submucosal glands and is present in secretions from normal human airway and cultured bronchial epithelial cells. Int. J. Biochem. Cell Biol. 2007, 39, 1943–1954. [Google Scholar] [CrossRef]
- Yu, C.J.; Yang, P.C.; Shew, J.Y.; Hong, T.M.; Yang, S.C.; Lee, Y.C.; Lee, L.N.; Luh, K.T.; Wu, C.W. Mucin mRNA expression in lung adenocarcinoma cell lines and tissues. Oncology 1996, 53, 118–126. [Google Scholar] [CrossRef] [PubMed]
- Pan, D.; Chen, J.; Feng, C.; Wu, W.; Wang, Y.; Tong, J.; Zhou, D. Preferential Localization of MUC1 Glycoprotein in Exosomes Secreted by Non-Small Cell Lung Carcinoma Cells. Int. J. Mol. Sci. 2019, 20, 323. [Google Scholar] [CrossRef] [PubMed]
- Awaya, H.; Takeshima, Y.; Yamasaki, M.; Inai, K. Expression of MUC1, MUC2, MUC5AC, and MUC6 in atypical adenomatous hyperplasia, bronchioloalveolar carcinoma, adenocarcinoma with mixed subtypes, and mucinous bronchioloalveolar carcinoma of the lung. Am. J. Clin. Pathol. 2004, 121, 644–653. [Google Scholar] [CrossRef]
- Sun, Y.; Sun, X.; You, C.; Ma, S.; Luo, Y.; Peng, S.; Tang, F.; Tian, X.; Wang, F.; Huang, Z.; et al. MUC3A promotes non-small cell lung cancer progression via activating the NFkappaB pathway and attenuates radiosensitivity. Int. J. Biol. Sci. 2021, 17, 2523–2536. [Google Scholar] [CrossRef]
- Supruniuk, K.; Radziejewska, I. MUC1 is an oncoprotein with a significant role in apoptosis (Review). Int. J. Oncol. 2021, 59. [Google Scholar] [CrossRef] [PubMed]
- Kishikawa, S.; Hayashi, T.; Saito, T.; Takamochi, K.; Kohsaka, S.; Sano, K.; Sasahara, N.; Sasa, K.; Kurihara, T.; Hara, K.; et al. Diffuse expression of MUC6 defines a distinct clinicopathological subset of pulmonary invasive mucinous adenocarcinoma. Mod. Pathol. 2021, 34, 786–797. [Google Scholar] [CrossRef] [PubMed]
- Kim, M.; Hwang, J.; Kim, K.A.; Hwang, S.; Lee, H.J.; Jung, J.Y.; Lee, J.G.; Cha, Y.J.; Shim, H.S. Genomic characteristics of invasive mucinous adenocarcinoma of the lung with multiple pulmonary sites of involvement. Mod. Pathol. 2021, 35, 202–209. [Google Scholar] [CrossRef]
- Lei, Y.; Zang, R.; Lu, Z.; Zhang, G.; Huang, J.; Liu, C.; Wang, Z.; Mao, S.; Che, Y.; Wang, X.; et al. ERO1L promotes IL6/sIL6R signaling and regulates MUC16 expression to promote CA125 secretion and the metastasis of lung cancer cells. Cell Death Dis. 2020, 11, 853. [Google Scholar] [CrossRef]
- Kim, D.M.; Noh, H.B.; Park, D.S.; Ryu, S.H.; Koo, J.S.; Shim, Y.B. Immunosensors for detection of Annexin II and MUC5AC for early diagnosis of lung cancer. Biosens. Bioelectron. 2009, 25, 456–462. [Google Scholar] [CrossRef]
- Fushimi, A.; Morimoto, Y.; Ishikawa, S.; Yamashita, N.; Bhattacharya, A.; Daimon, T.; Rajabi, H.; Jin, C.; Hagiwara, M.; Yasumizu, Y.; et al. Dependence on the MUC1-C Oncoprotein in Classic, Variant, and Non-neuroendocrine Small Cell Lung Cancer. Mol. Cancer Res. 2022, 20, 1379–1390. [Google Scholar] [CrossRef] [PubMed]
- Savarese-Brenner, B.; Heugl, M.; Rath, B.; Schweizer, C.; Obermayr, E.; Stickler, S.; Hamilton, G. MUC1 and CD147 Are Promising Markers for the Detection of Circulating Tumor Cells in Small Cell Lung Cancer. Anticancer Res. 2022, 42, 429–439. [Google Scholar] [CrossRef]
- Molina, R.; Auge, J.M.; Escudero, J.M.; Marrades, R.; Vinolas, N.; Carcereny, E.; Ramirez, J.; Filella, X. Mucins CA 125, CA 19.9, CA 15.3 and TAG-72.3 as tumor markers in patients with lung cancer: Comparison with CYFRA 21-1, CEA, SCC and NSE. Tumour Biol. 2008, 29, 371–380. [Google Scholar] [CrossRef] [PubMed]
- Yang, Q.; Zhang, P.; Wu, R.; Lu, K.; Zhou, H. Identifying the Best Marker Combination in CEA, CA125, CY211, NSE, and SCC for Lung Cancer Screening by Combining ROC Curve and Logistic Regression Analyses: Is It Feasible? Dis. Markers 2018, 2018, 2082840. [Google Scholar] [CrossRef] [PubMed]
- Korbakis, D.; Dimitromanolakis, A.; Prassas, I.; Davis, G.J.; Barber, E.; Reckamp, K.L.; Blasutig, I.; Diamandis, E.P. Serum LAMC2 enhances the prognostic value of a multi-parametric panel in non-small cell lung cancer. Br. J. Cancer 2015, 113, 484–491. [Google Scholar] [CrossRef]
- Horimasu, Y.; Ishikawa, N.; Tanaka, S.; Hirano, C.; Iwamoto, H.; Ohshimo, S.; Fujitaka, K.; Hamada, H.; Hattori, N.; Kohno, N. MUC1 in lung adenocarcinoma: Cross-sectional genetic and serological study. BMC Cancer 2017, 17, 263. [Google Scholar] [CrossRef]
- Song, Y.; Zhu, X.; Lin, Z.; Luo, L.; Wen, D. The potential value of serum chemerin in patients with breast cancer. Sci. Rep. 2021, 11, 6564. [Google Scholar] [CrossRef]
- Chen, C.; Chen, Q.; Dong, Y.; Liu, X. The Clinical Significance of sICAM-1 in Differentiating Benign Breast Lesions from Breast Cancer. Ann. Clin. Lab. Sci. 2020, 50, 650–656. [Google Scholar]
- Roy, L.D.; Dillon, L.M.; Zhou, R.; Moore, L.J.; Livasy, C.; El-Khoury, J.M.; Puri, R.; Mukherjee, P. A tumor specific antibody to aid breast cancer screening in women with dense breast tissue. Genes Cancer 2017, 8, 536–549. [Google Scholar] [CrossRef]
- Woolas, R.P.; Xu, F.-J.; Jacabs, I.J.; Yu, Y.-H.; Daly, L.; Brechuck, A.; Soper, J.T.; Clarke-pearson, D.L.; Oram, D.H.; Bast, R.C., Jr. Elevation of Multiple Serum Markers in Patients With Stage I Ovarian Cancer. JNCI J. Natl. Cancer Inst. 1993, 85, 1748–1751. [Google Scholar] [CrossRef]
- Guo, J.; Yang, W.L.; Pak, D.; Celestino, J.; Lu, K.H.; Ning, J.; Lokshin, A.E.; Cheng, Z.; Lu, Z.; Bast, R.C., Jr. Osteopontin, Macrophage Migration Inhibitory Factor and Anti-Interleukin-8 Autoantibodies Complement CA125 for Detection of Early Stage Ovarian Cancer. Cancers 2019, 11, 596. [Google Scholar] [CrossRef]
- Simmons, A.R.; Fourkala, E.O.; Gentry-Maharaj, A.; Ryan, A.; Sutton, M.N.; Baggerly, K.; Zheng, H.; Lu, K.H.; Jacobs, I.; Skates, S.; et al. Complementary Longitudinal Serum Biomarkers to CA125 for Early Detection of Ovarian Cancer. Cancer Prev. Res. 2019, 12, 391–400. [Google Scholar] [CrossRef]
- Engelstaedter, V.; Heublein, S.; Schumacher, A.L.; Lenhard, M.; Engelstaedter, H.; Andergassen, U.; Guenthner-Biller, M.; Kuhn, C.; Rack, B.; Kupka, M.; et al. Mucin-1 and its relation to grade, stage and survival in ovarian carcinoma patients. BMC Cancer 2012, 12, 600. [Google Scholar] [CrossRef] [PubMed]
- Jeschke, U.; Wiest, I.; Schumacher, A.L.; Kupka, M.; Rack, B.; Stahn, R.; Karsten, U.; Mayr, D.; Friese, K.; Dian, D. Determination of MUC1 in sera of ovarian cancer patients and in sera of patients with benign changes of the ovaries with CA15-3, CA27.29, and PankoMab. Anticancer Res. 2012, 32, 2185–2189. [Google Scholar] [PubMed]
- Gao, Y.; Wang, J.; Zhou, Y.; Sheng, S.; Qian, S.Y.; Huo, X. Evaluation of Serum CEA, CA19-9, CA72-4, CA125 and Ferritin as Diagnostic Markers and Factors of Clinical Parameters for Colorectal Cancer. Sci. Rep. 2018, 8, 2732. [Google Scholar] [CrossRef] [PubMed]
- Al-Khayal, K.; Abdulla, M.; Al-Obaid, O.; Zubaidi, A.; Vaali-Mohammed, M.A.; Alsheikh, A.; Ahmad, R. Differential expression of mucins in Middle Eastern patients with colorectal cancer. Oncol. Lett. 2016, 12, 393–400. [Google Scholar] [CrossRef] [PubMed]
- Zhong, W.; Yu, Z.; Zhan, J.; Yu, T.; Lin, Y.; Xia, Z.S.; Yuan, Y.H.; Chen, Q.K. Association of serum levels of CEA, CA199, CA125, CYFRA21-1 and CA72-4 and disease characteristics in colorectal cancer. Pathol. Oncol. Res. 2015, 21, 83–95. [Google Scholar] [CrossRef]
- Björkman, K.; Mustonen, H.; Kaprio, T.; Kekki, H.; Pettersson, K.; Haglund, C.; Böckelman, C. CA125: A superior prognostic biomarker for colorectal cancer compared to CEA, CA19-9 or CA242. Tumour Biol. 2021, 43, 57–70. [Google Scholar] [CrossRef]
- Luka, J.; Arlen, P.M.; Bristol, A. Development of a serum biomarker assay that differentiates tumor-associated MUC5AC (NPC-1C ANTIGEN) from normal MUC5AC. J. Biomed. Biotechnol. 2011, 2011, 934757. [Google Scholar] [CrossRef]
- Kaur, S.; Smith, L.M.; Patel, A.; Menning, M.; Watley, D.C.; Malik, S.S.; Krishn, S.R.; Mallya, K.; Aithal, A.; Sasson, A.R.; et al. A Combination of MUC5AC and CA19-9 Improves the Diagnosis of Pancreatic Cancer: A Multicenter Study. Am. J. Gastroenterol. 2017, 112, 172–183. [Google Scholar] [CrossRef]
- Zhang, J.; Wang, Y.; Zhao, T.; Li, Y.; Tian, L.; Zhao, J.; Zhang, J. Evaluation of serum MUC5AC in combination with CA19-9 for the diagnosis of pancreatic cancer. World J. Surg. Oncol. 2020, 18, 31. [Google Scholar] [CrossRef]
- Gu, Y.L.; Lan, C.; Pei, H.; Yang, S.N.; Liu, Y.F.; Xiao, L.L. Applicative Value of Serum CA19-9, CEA, CA125 and CA242 in Diagnosis and Prognosis for Patients with Pancreatic Cancer Treated by Concurrent Chemoradiotherapy. Asian Pac. J. Cancer Prev. 2015, 16, 6569–6573. [Google Scholar] [CrossRef]
- Yokoyama, S.; Kitamoto, S.; Higashi, M.; Goto, Y.; Hara, T.; Ikebe, D.; Yamaguchi, T.; Arisaka, Y.; Niihara, T.; Nishimata, H.; et al. Diagnosis of pancreatic neoplasms using a novel method of DNA methylation analysis of mucin expression in pancreatic juice. PLoS ONE 2014, 9, e93760. [Google Scholar] [CrossRef]
- Chen, Y.; Gao, S.G.; Chen, J.M.; Wang, G.P.; Wang, Z.F.; Zhou, B.; Jin, C.H.; Yang, Y.T.; Feng, X.S. Serum CA242, CA199, CA125, CEA, and TSGF are Biomarkers for the Efficacy and Prognosis of Cryoablation in Pancreatic Cancer Patients. Cell Biochem. Biophys. 2015, 71, 1287–1291. [Google Scholar] [CrossRef]
- Yang, C.; Cheng, H.; Luo, G.; Lu, Y.; Guo, M.; Jin, K.; Wang, Z.; Yu, X.; Liu, C. The metastasis status and tumor burden-associated CA125 level combined with the CD4/CD8 ratio predicts the prognosis of patients with advanced pancreatic cancer: A new scoring system. Eur. J. Surg. Oncol. 2017, 43, 2112–2118. [Google Scholar] [CrossRef] [PubMed]
- Arai, T.; Fujita, K.; Fujime, M.; Irimura, T. Expression of sialylated MUC1 in prostate cancer: Relationship to clinical stage and prognosis. Int. J. Urol. 2005, 12, 654–661. [Google Scholar] [CrossRef]
- Bilen, M.A.; Reyes, A.; Bhowmick, D.; Maa, A.; Bast, R., Jr.; Pisters, L.L.; Lin, S.H.; Logothetis, C.J.; Tu, S.M. Variant prostate carcinoma and elevated serum CA-125. Can. J. Urol. 2014, 21, 7442–7448. [Google Scholar]
- Devine, P.L.; Walsh, M.D.; McGuckin, M.A.; Quin, R.J.; Hohn, B.G.; Clague, A.; Samaratunga, H. Prostate-specific antigen (PSA) and cancer-associated serum antigen (CASA) in distinguishing benign and malignant prostate disease. Int. J. Biol. Markers 1995, 10, 221–225. [Google Scholar] [CrossRef]
- Somovilla, V.J.; Bermejo, I.A.; Albuquerque, I.S.; Martinez-Saez, N.; Castro-Lopez, J.; Garcia-Martin, F.; Companon, I.; Hinou, H.; Nishimura, S.I.; Jimenez-Barbero, J.; et al. The Use of Fluoroproline in MUC1 Antigen Enables Efficient Detection of Antibodies in Patients with Prostate Cancer. J. Am. Chem. Soc. 2017, 139, 18255–18261. [Google Scholar] [CrossRef] [PubMed]
- Elias, J.; Kew, M.C. Evaluation of CA 125 as a serum marker of hepatocellular carcinoma. Int. J. Cancer 1990, 46, 805–807. [Google Scholar] [CrossRef] [PubMed]
- Lopez, J.B.; Balasegaram, M.; Thambyrajah, V. Serum CA 125 as a marker of hepatocellular carcinoma. Int. J. Biol. Markers 1996, 11, 178–182. [Google Scholar] [CrossRef]
- Huang, Y.; Zeng, J.; Liu, T.; Lin, X.; Guo, P.; Zeng, J.; Zhou, W.; Liu, J. Prognostic Significance of Elevated Preoperative Serum CA125 Levels After Curative Hepatectomy for Hepatocellular Carcinoma. OncoTargets Ther. 2020, 13, 4559–4567. [Google Scholar] [CrossRef]
- Qin, C.; Gao, Y.; Li, J.; Huang, C.; He, S. Predictive effects of preoperative serum CA125 and AFP levels on post-hepatectomy survival in patients with hepatitis B-related hepatocellular carcinoma. Oncol. Lett. 2021, 21, 487. [Google Scholar] [CrossRef]
- Xu, H.; Inagaki, Y.; Tang, W.; Guo, Q.; Wang, F.; Seyama, Y.; Midorikawa, Y.; Gai, R.; Kokudo, N.; Sugawara, Y.; et al. Elevation of serum KL-6 mucin levels in patients with cholangiocarcinoma. Hepato-Gastroenterology 2008, 55, 2000–2004. [Google Scholar]
- Tamaki, N.; Kuno, A.; Matsuda, A.; Tsujikawa, H.; Yamazaki, K.; Yasui, Y.; Tsuchiya, K.; Nakanishi, H.; Itakura, J.; Korenaga, M.; et al. Serum Wisteria Floribunda Agglutinin-Positive Sialylated Mucin 1 as a Marker of Progenitor/Biliary Features in Hepatocellular Carcinoma. Sci. Rep. 2017, 7, 244. [Google Scholar] [CrossRef]
- Zhou, S.; Wang, Z.; Li, M.; Wu, L. Elevated Preoperative Serum CA125 Predicts Larger Tumor Diameter in Patients with Hepatocellular Carcinoma and Low AFP Levels. BioMed Res. Int. 2019, 2019, 6959637. [Google Scholar] [CrossRef] [PubMed]
- Marmot, M.G.; Altman, D.G.; Cameron, D.A.; Dewar, J.A.; Thompson, S.G.; Wilcox, M. The benefits and harms of breast cancer screening: An independent review. Br. J. Cancer 2013, 108, 2205–2240. [Google Scholar] [CrossRef] [PubMed]
- McKinney, S.M.; Sieniek, M.; Godbole, V.; Godwin, J.; Antropova, N.; Ashrafian, H.; Back, T.; Chesus, M.; Corrado, G.S.; Darzi, A.; et al. International evaluation of an AI system for breast cancer screening. Nature 2020, 577, 89–94. [Google Scholar] [CrossRef]
- Lei, S.; Zheng, R.; Zhang, S.; Wang, S.; Chen, R.; Sun, K.; Zeng, H.; Zhou, J.; Wei, W. Global patterns of breast cancer incidence and mortality: A population-based cancer registry data analysis from 2000 to 2020. Cancer. Commun. 2021, 41, 1183–1194. [Google Scholar] [CrossRef]
- Cancer Stat Facts: Female Breast Cancer. National Cancer Institute. Available online: https://seer.cancer.gov/statfacts/html/breast.html (accessed on 26 December 2022).
- Breast Cancer Early Detection and Diagnosis. American Cancer Society. Available online: https://www.cancer.org/cancer/breast-cancer/screening-tests-and-early-detection/mammograms.html (accessed on 29 August 2021).
- Lauby-Secretan, B.; Scoccianti, C.; Loomis, D.; Benbrahim-Tallaa, L.; Bouvard, V.; Bianchini, F.; Straif, K.; International Agency for Research on Cancer Handbook Working Group. Breast-cancer screening--viewpoint of the IARC Working Group. N. Engl. J. Med. 2015, 372, 2353–2358. [Google Scholar] [CrossRef]
- Oeffinger, K.C.; Fontham, E.T.; Etzioni, R.; Herzig, A.; Michaelson, J.S.; Shih, Y.-C.T.; Walter, L.C.; Church, T.R.; Flowers, C.R.; LaMonte, S.J. Breast cancer screening for women at average risk: 2015 guideline update from the American Cancer Society. JAMA 2015, 314, 1599–1614. [Google Scholar] [CrossRef] [PubMed]
- Siu, A.L.; Force, U.S.P.S.T. Screening for Breast Cancer: U.S. Preventive Services Task Force Recommendation Statement. Ann. Intern. Med. 2016, 164, 279–296. [Google Scholar] [CrossRef]
- Lehman, C.D.; Wellman, R.D.; Buist, D.S.; Kerlikowske, K.; Tosteson, A.N.; Miglioretti, D.L.; Breast Cancer Surveillance, C. Diagnostic Accuracy of Digital Screening Mammography With and Without Computer-Aided Detection. JAMA Intern. Med. 2015, 175, 1828–1837. [Google Scholar] [CrossRef]
- Wise, J. Computer aided mammography yields no clear benefit, research finds. BMJ 2015, 351, h5164. [Google Scholar] [CrossRef]
- Elmore, J.G.; Jackson, S.L.; Abraham, L.; Miglioretti, D.L.; Carney, P.A.; Geller, B.M.; Yankaskas, B.C.; Kerlikowske, K.; Onega, T.; Rosenberg, R.D.; et al. Variability in interpretive performance at screening mammography and radiologists’ characteristics associated with accuracy. Radiology 2009, 253, 641–651. [Google Scholar] [CrossRef]
- Liu, A.; Yin, L.; Ma, Y.; Han, P.; Wu, Y.; Wu, Y.; Ye, Z. Quantitative breast density measurement based on three-dimensional images: A study on cone-beam breast computed tomography. Acta Radiol. 2021, 63, 1023–1031. [Google Scholar] [CrossRef] [PubMed]
- Gao, Y.; Moy, L.; Heller, S.L. Digital Breast Tomosynthesis: Update on Technology, Evidence, and Clinical Practice. Radiographics 2021, 41, 321–337. [Google Scholar] [CrossRef] [PubMed]
- Mukhopadhyay, P.; Chakraborty, S.; Ponnusamy, M.P.; Lakshmanan, I.; Jain, M.; Batra, S.K. Mucins in the pathogenesis of breast cancer: Implications in diagnosis, prognosis and therapy. Biochim. Biophys. Acta 2011, 1815, 224–240. [Google Scholar] [CrossRef]
- Chen, W.; Zhang, Z.; Zhang, S.; Zhu, P.; Ko, J.K.; Yung, K.K. MUC1: Structure, Function, and Clinic Application in Epithelial Cancers. Int. J. Mol. Sci. 2021, 22, 6567. [Google Scholar] [CrossRef]
- Jin, D.; Jiao, Y.; Ji, J.; Jiang, W.; Ni, W.; Wu, Y.; Ni, R.; Lu, C.; Qu, L.; Ni, H.; et al. Identification of prognostic risk factors for pancreatic cancer using bioinformatics analysis. PeerJ 2020, 8, e9301. [Google Scholar] [CrossRef]
- Chu, J.-S.; Chang, K.-J. Mucin expression in mucinous carcinoma and other invasive carcinomas of the breast. Cancer Lett. 1999, 142, 121–127. [Google Scholar] [CrossRef] [PubMed]
- Bieglmayer, C.; Szepesi, T.; Neunteufel, W. Follow-up of metastatic breast cancer patients with a mucin-like carcinoma-associated antigen: Comparison to CA 15.3 and carcinoembryonic antigen. Cancer Lett. 1988, 42, 199–206. [Google Scholar] [CrossRef]
- Kufe, D.; Inghirami, G.; Abe, M.; Hayes, D.; Justi-Wheeler, H.; Schlom, J. Differential reactivity of a novel monoclonal antibody (DF3) with human malignant versus benign breast tumors. Hybridoma 1984, 3, 223–232. [Google Scholar] [CrossRef] [PubMed]
- Kufe, D.W. MUC1-C oncoprotein as a target in breast cancer: Activation of signaling pathways and therapeutic approaches. Oncogene 2013, 32, 1073–1081. [Google Scholar] [CrossRef]
- Hayes, D.F.; Sekine, H.; Ohno, T.; Abe, M.; Keefe, K.; Kufe, D.W. Use of a murine monoclonal antibody for detection of circulating plasma DF3 antigen levels in breast cancer patients. J. Clin. Investig. 1985, 75, 1671–1678. [Google Scholar] [CrossRef] [PubMed]
- Abe, M.; Kufe, D. Structural analysis of the DF3 human breast carcinoma-associated protein. Cancer Res. 1989, 49, 2834–2839. [Google Scholar] [PubMed]
- Perey, L.; Hayes, D.F.; Maimonis, P.; Abe, M.; O’Hara, C.; Kufe, D.W. Tumor selective reactivity of a monoclonal antibody prepared against a recombinant peptide derived from the DF3 human breast carcinoma-associated antigen. Cancer Res. 1992, 52, 2563–2568. [Google Scholar]
- Siroy, A.; Abdul-Karim, F.W.; Miedler, J.; Fong, N.; Fu, P.; Gilmore, H.; Baar, J. MUC1 is expressed at high frequency in early-stage basal-like triple-negative breast cancer. Hum. Pathol. 2013, 44, 2159–2166. [Google Scholar] [CrossRef]
- Kelly, V.J.; Wu, S.T.; Gottumukkala, V.; Coelho, R.; Palmer, K.; Nair, S.; Erick, T.; Puri, R.; Ilovich, O.; Mukherjee, P. Preclinical evaluation of an (111)In/(225)Ac theranostic targeting transformed MUC1 for triple negative breast cancer. Theranostics 2020, 10, 6946–6958. [Google Scholar] [CrossRef] [PubMed]
- Yousefi, M.; Dehghani, S.; Nosrati, R.; Zare, H.; Evazalipour, M.; Mosafer, J.; Tehrani, B.S.; Pasdar, A.; Mokhtarzadeh, A.; Ramezani, M. Aptasensors as a new sensing technology developed for the detection of MUC1 mucin: A review. Biosens. Bioelectron. 2019, 130, 1–19. [Google Scholar] [CrossRef]
- Ahirwar, R.; Khan, N.; Kumar, S. Aptamer-based sensing of breast cancer biomarkers: A comprehensive review of analytical figures of merit. Expert Rev. Mol. Diagn. 2021, 21, 703–721. [Google Scholar] [CrossRef]
- Rakha, E.A.; Boyce, R.W.; Abd El-Rehim, D.; Kurien, T.; Green, A.R.; Paish, E.C.; Robertson, J.F.; Ellis, I.O. Expression of mucins (MUC1, MUC2, MUC3, MUC4, MUC5AC and MUC6) and their prognostic significance in human breast cancer. Mod. Pathol. 2005, 18, 1295–1304. [Google Scholar] [CrossRef] [PubMed]
- Pereira, M.; Dias, A.; Reis, C.; Schmitt, F. Immunohistochemical study of the expression of MUC5AC and MUC6 in breast carcinomas and adjacent breast tissues. J. Clin. Pathol. 2001, 54, 210–213. [Google Scholar] [CrossRef]
- Walsh, M.; McGuckin, M.; Devine, P.; Hohn, B.; Wright, R. Expression of MUC2 epithelial mucin in breast carcinoma. J. Clin. Pathol. 1993, 46, 922–925. [Google Scholar] [CrossRef] [PubMed]
- Astashchanka, A.; Shroka, T.M.; Jacobsen, B.M. Mucin 2 (MUC2) modulates the aggressiveness of breast cancer. Breast Cancer Res. Treat. 2019, 173, 289–299. [Google Scholar] [CrossRef] [PubMed]
- Valque, H.; Gouyer, V.; Gottrand, F.; Desseyn, J.L. MUC5B leads to aggressive behavior of breast cancer MCF7 cells. PLoS ONE 2012, 7, e46699. [Google Scholar] [CrossRef]
- Budzik, M.P.; Fudalej, M.M.; Badowska-Kozakiewicz, A.M. Histopathological analysis of mucinous breast cancer subtypes and comparison with invasive carcinoma of no special type. Sci. Rep. 2021, 11, 5770. [Google Scholar] [CrossRef]
- Aoki, K.; Kametaka, H.; Makino, H.; Fukada, T.; Akiyama, T.; Hyakutake, Y.; Yoshida, H.; Mitomi, H.; Seike, K. [A Case of Primary Breast Cancer and Gastric Cancer Metastasis to the Skin-Usefulness of Immunohistochemistry in Differentiating Primary Breast Cancer from Metastatic Breast Cancer]. Gan Kagaku Ryoho 2020, 47, 2352–2354. [Google Scholar]
- Oral, O.; Unverdi, H.; Kumcu, E.; Turkbey, D.; Dogan, S.; Hucumenoglu, S. Associations between the expression of mucins (MUC1, MUC2, MUC5AC and MUC6) and clinicopathologic parameters of human breast carcinomas. Indian J. Pathol. Microbiol. 2020, 63, 551–558. [Google Scholar] [CrossRef]
- Matulonis, U.A.; Sood, A.K.; Fallowfield, L.; Howitt, B.E.; Sehouli, J.; Karlan, B.Y. Ovarian cancer. Nat. Rev. Dis. Prim. 2016, 2, 16061. [Google Scholar] [CrossRef]
- Lheureux, S.; Gourley, C.; Vergote, I.; Oza, A.M. Epithelial ovarian cancer. Lancet 2019, 393, 1240–1253. [Google Scholar] [CrossRef]
- Franier, B.; Thompson, M. Early stage detection and screening of ovarian cancer: A research opportunity and significant challenge for biosensor technology. Biosens. Bioelectron. 2019, 135, 71–81. [Google Scholar] [CrossRef] [PubMed]
- Bonifácio, V.D.B. Ovarian Cancer Biomarkers: Moving Forward in Early Detection. Adv. Exp. Med. Biol. 2020, 1219, 355–363. [Google Scholar] [CrossRef] [PubMed]
- Blyuss, O.; Burnell, M.; Ryan, A.; Gentry-Maharaj, A.; Mariño, I.P.; Kalsi, J.; Manchanda, R.; Timms, J.F.; Parmar, M.; Skates, S.J.; et al. Comparison of Longitudinal CA125 Algorithms as a First-Line Screen for Ovarian Cancer in the General Population. Clin. Cancer Res. 2018, 24, 4726–4733. [Google Scholar] [CrossRef]
- Zhang, M.; Cheng, S.; Jin, Y.; Zhao, Y.; Wang, Y. Roles of CA125 in diagnosis, prediction, and oncogenesis of ovarian cancer. Biochim. Biophys. Acta Rev. Cancer 2021, 1875, 188503. [Google Scholar] [CrossRef]
- Dikmen, Z.G.; Colak, A.; Dogan, P.; Tuncer, S.; Akbiyik, F. Diagnostic performances of CA125, HE4, and ROMA index in ovarian cancer. Eur. J. Gynaecol. Oncol. 2015, 36, 457–462. [Google Scholar] [PubMed]
- Ferraro, S.; Braga, F.; Lanzoni, M.; Boracchi, P.; Biganzoli, E.M.; Panteghini, M. Serum human epididymis protein 4 vs carbohydrate antigen 125 for ovarian cancer diagnosis: A systematic review. J. Clin. Pathol. 2013, 66, 273–281. [Google Scholar] [CrossRef] [PubMed]
- Bagnoli, M.; Canevari, S.; Califano, D.; Losito, S.; Maio, M.D.; Raspagliesi, F.; Carcangiu, M.L.; Toffoli, G.; Cecchin, E.; Sorio, R.; et al. Development and validation of a microRNA-based signature (MiROvaR) to predict early relapse or progression of epithelial ovarian cancer: A cohort study. Lancet Oncol. 2016, 17, 1137–1146. [Google Scholar] [CrossRef] [PubMed]
- Ferraro, S.; Robbiano, C.; Tosca, N.; Panzeri, A.; Paganoni, A.M.; Panteghini, M. Serum human epididymis protein 4 vs. carbohydrate antigen 125 in ovarian cancer follow-up. Clin. Biochem. 2018, 60, 84–90. [Google Scholar] [CrossRef]
- Singha, B.; Harper, S.L.; Goldman, A.R.; Bitler, B.G.; Aird, K.M.; Borowsky, M.E.; Cadungog, M.G.; Liu, Q.; Zhang, R.; Jean, S.; et al. CLIC1 and CLIC4 complement CA125 as a diagnostic biomarker panel for all subtypes of epithelial ovarian cancer. Sci. Rep. 2018, 8, 14725. [Google Scholar] [CrossRef] [PubMed]
- Zheng, X.; Chen, S.; Li, L.; Liu, X.; Liu, X.; Dai, S.; Zhang, P.; Lu, H.; Lin, Z.; Yu, Y.; et al. Evaluation of HE4 and TTR for diagnosis of ovarian cancer: Comparison with CA-125. J. Gynecol. Obs. Hum. Reprod. 2018, 47, 227–230. [Google Scholar] [CrossRef]
- Dochez, V.; Caillon, H.; Vaucel, E.; Dimet, J.; Winer, N.; Ducarme, G. Biomarkers and algorithms for diagnosis of ovarian cancer: CA125, HE4, RMI and ROMA, a review. J. Ovarian Res. 2019, 12, 28. [Google Scholar] [CrossRef]
- Wang, Q.; Wu, Y.; Zhang, H.; Yang, K.; Tong, Y.; Chen, L.; Zhou, Q.; Guan, S. Clinical Value of Serum HE4, CA125, CA72-4, and ROMA Index for Diagnosis of Ovarian Cancer and Prediction of Postoperative Recurrence. Clin. Lab. 2019, 65. [Google Scholar] [CrossRef]
- El Bairi, K.; Afqir, S.; Amrani, M. Is HE4 Superior over CA-125 in the Follow-up of Patients with Epithelial Ovarian Cancer? Curr. Drug Targets 2020, 21, 1026–1033. [Google Scholar] [CrossRef] [PubMed]
- Sun, J.; Cui, X.W.; Li, Y.S.; Wang, S.Y.; Yin, Q.; Wang, X.N.; Gu, L. The value of 18F-FDG PET/CT imaging combined with detection of CA125 and HE4 in the diagnosis of recurrence and metastasis of ovarian cancer. Eur. Rev. Med. Pharm. Sci. 2020, 24, 7276–7283. [Google Scholar] [CrossRef]
- van Haaften-Day, C.; Shen, Y.; Xu, F.; Yu, Y.; Berchuck, A.; Havrilesky, L.J.; de Bruijn, H.W.; van der Zee, A.G.; Bast, R.C., Jr.; Hacker, N.F. OVX1, macrophage-colony stimulating factor, and CA-125-II as tumor markers for epithelial ovarian carcinoma: A critical appraisal. Cancer 2001, 92, 2837–2844. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Z.; Yu, Y.; Xu, F.; Berchuck, A.; van Haaften-Day, C.; Havrilesky, L.J.; de Bruijn, H.W.; van der Zee, A.G.; Woolas, R.P.; Jacobs, I.J.; et al. Combining multiple serum tumor markers improves detection of stage I epithelial ovarian cancer. Gynecol. Oncol. 2007, 107, 526–531. [Google Scholar] [CrossRef] [PubMed]
- Granato, T.; Midulla, C.; Longo, F.; Colaprisca, B.; Frati, L.; Anastasi, E. Role of HE4, CA72.4, and CA125 in monitoring ovarian cancer. Tumour Biol. 2012, 33, 1335–1339. [Google Scholar] [CrossRef]
- Muinao, T.; Deka Boruah, H.P.; Pal, M. Multi-biomarker panel signature as the key to diagnosis of ovarian cancer. Heliyon 2019, 5, e02826. [Google Scholar] [CrossRef] [PubMed]
- Barr, C.E.; Funston, G.; Jeevan, D.; Sundar, S.; Mounce, L.T.A.; Crosbie, E.J. The Performance of HE4 Alone and in Combination with CA125 for the Detection of Ovarian Cancer in an Enriched Primary Care Population. Cancers 2022, 14, 2124. [Google Scholar] [CrossRef]
- Kim, B.; Park, Y.; Kim, B.; Ahn, H.J.; Lee, K.A.; Chung, J.E.; Han, S.W. Diagnostic performance of CA 125, HE4, and risk of Ovarian Malignancy Algorithm for ovarian cancer. J. Clin. Lab. Anal. 2019, 33, e22624. [Google Scholar] [CrossRef]
- Singh, A.P.; Senapati, S.; Ponnusamy, M.P.; Jain, M.; Lele, S.M.; Davis, J.S.; Remmenga, S.; Batra, S.K. Clinical potential of mucins in diagnosis, prognosis, and therapy of ovarian cancer. Lancet Oncol. 2008, 9, 1076–1085. [Google Scholar] [CrossRef]
- Freeman, H.J. Early stage colon cancer. World J. Gastroenterol. 2013, 19, 8468–8473. [Google Scholar] [CrossRef] [PubMed]
- Ladabaum, U.; Dominitz, J.A.; Kahi, C.; Schoen, R.E. Strategies for Colorectal Cancer Screening. Gastroenterology 2020, 158, 418–432. [Google Scholar] [CrossRef]
- Wolf, A.M.D.; Fontham, E.T.H.; Church, T.R.; Flowers, C.R.; Guerra, C.E.; LaMonte, S.J.; Etzioni, R.; McKenna, M.T.; Oeffinger, K.C.; Shih, Y.T.; et al. Colorectal cancer screening for average-risk adults: 2018 guideline update from the American Cancer Society. CA Cancer J. Clin. 2018, 68, 250–281. [Google Scholar] [CrossRef] [PubMed]
- Imperiale, T.F.; Kahi, C.J. Cost-effectiveness of Future Biomarkers for Colorectal Cancer Screening: Quantified Futility or Call for Innovation? Clin. Gastroenterol. Hepatol. 2018, 16, 483–485. [Google Scholar] [CrossRef] [PubMed]
- Sawbridge, D.; Probert, C. Population-based screening in colorectal cancer—Current practice and future developments: Faecal biomarkers review. J. Gastrointestin. Liver Dis. 2014, 23, 195–202. [Google Scholar] [CrossRef]
- Bosch, L.J.W.; de Wit, M.; Pham, T.V.; Coupé, V.M.H.; Hiemstra, A.C.; Piersma, S.R.; Oudgenoeg, G.; Scheffer, G.L.; Mongera, S.; Sive Droste, J.T.; et al. Novel Stool-Based Protein Biomarkers for Improved Colorectal Cancer Screening: A Case-Control Study. Ann. Intern. Med. 2017, 167, 855–866. [Google Scholar] [CrossRef]
- Carethers, J.M. Fecal DNA Testing for Colorectal Cancer Screening. Annu. Rev. Med. 2020, 71, 59–69. [Google Scholar] [CrossRef]
- Dong, L.; Ren, H. Blood-based DNA Methylation Biomarkers for Early Detection of Colorectal Cancer. J. Proteom. Bioinform. 2018, 11, 120–126. [Google Scholar] [CrossRef]
- Duran-Sanchon, S.; Moreno, L.; Augé, J.M.; Serra-Burriel, M.; Cuatrecasas, M.; Moreira, L.; Martín, A.; Serradesanferm, A.; Pozo, À.; Costa, R.; et al. Identification and Validation of MicroRNA Profiles in Fecal Samples for Detection of Colorectal Cancer. Gastroenterology 2020, 158, 947–957.e4. [Google Scholar] [CrossRef]
- Niedermaier, T.; Balavarca, Y.; Brenner, H. Stage-Specific Sensitivity of Fecal Immunochemical Tests for Detecting Colorectal Cancer: Systematic Review and Meta-Analysis. Am. J. Gastroenterol. 2020, 115, 56–69. [Google Scholar] [CrossRef] [PubMed]
- Slaby, O. Non-coding RNAs as Biomarkers for Colorectal Cancer Screening and Early Detection. Adv. Exp. Med. Biol. 2016, 937, 153–170. [Google Scholar] [CrossRef] [PubMed]
- Yörüker, E.E.; Holdenrieder, S.; Gezer, U. Blood-based biomarkers for diagnosis, prognosis and treatment of colorectal cancer. Clin. Chim. Acta 2016, 455, 26–32. [Google Scholar] [CrossRef] [PubMed]
- Thomas, D.S.; Fourkala, E.O.; Apostolidou, S.; Gunu, R.; Ryan, A.; Jacobs, I.; Menon, U.; Alderton, W.; Gentry-Maharaj, A.; Timms, J.F. Evaluation of serum CEA, CYFRA21-1 and CA125 for the early detection of colorectal cancer using longitudinal preclinical samples. Br. J. Cancer 2015, 113, 268–274. [Google Scholar] [CrossRef] [PubMed]
- Pothuraju, R.; Krishn, S.R.; Gautam, S.K.; Pai, P.; Ganguly, K.; Chaudhary, S.; Rachagani, S.; Kaur, S.; Batra, S.K. Mechanistic and Functional Shades of Mucins and Associated Glycans in Colon Cancer. Cancers 2020, 12, 649. [Google Scholar] [CrossRef]
- Krishn, S.R.; Kaur, S.; Sheinin, Y.M.; Smith, L.M.; Gautam, S.K.; Patel, A.; Jain, M.; Juvvigunta, V.; Pai, P.; Lazenby, A.J.; et al. Mucins and associated O-glycans based immunoprofile for stratification of colorectal polyps: Clinical implication for improved colon surveillance. Oncotarget 2017, 8, 7025–7038. [Google Scholar] [CrossRef]
- Wang, H.; Jin, S.; Lu, H.; Mi, S.; Shao, W.; Zuo, X.; Yin, H.; Zeng, S.; Shimamoto, F.; Qi, G. Expression of survivin, MUC2 and MUC5 in colorectal cancer and their association with clinicopathological characteristics. Oncol. Lett. 2017, 14, 1011–1016. [Google Scholar] [CrossRef]
- Bonde, A.; Smith, D.A.; Kikano, E.; Yoest, J.M.; Tirumani, S.H.; Ramaiya, N.H. Overview of serum and tissue markers in colorectal cancer: A primer for radiologists. Abdom. Radiol. 2021, 46, 5521–5535. [Google Scholar] [CrossRef]
- Liu, Z.; Gu, Y.; Li, X.; Zhou, L.; Cheng, X.; Jiang, H.; Huang, Y.; Zhang, Y.; Xu, T.; Yang, W.; et al. Mucin 16 Promotes Colorectal Cancer Development and Progression Through Activation of Janus Kinase 2. Dig. Dis. Sci. 2021, 67, 2195–2208. [Google Scholar] [CrossRef]
- Singhi, A.D.; Koay, E.J.; Chari, S.T.; Maitra, A. Early Detection of Pancreatic Cancer: Opportunities and Challenges. Gastroenterology 2019, 156, 2024–2040. [Google Scholar] [CrossRef] [PubMed]
- Zhang, L.; Sanagapalli, S.; Stoita, A. Challenges in diagnosis of pancreatic cancer. World J. Gastroenterol. 2018, 24, 2047–2060. [Google Scholar] [CrossRef]
- Yao, W.; Maitra, A.; Ying, H. Recent insights into the biology of pancreatic cancer. EBioMedicine 2020, 53, 102655. [Google Scholar] [CrossRef] [PubMed]
- Kleeff, J.; Korc, M.; Apte, M.; La Vecchia, C.; Johnson, C.D.; Biankin, A.V.; Neale, R.E.; Tempero, M.; Tuveson, D.A.; Hruban, R.H.; et al. Pancreatic cancer. Nat. Rev. Dis. Prim. 2016, 2, 16022. [Google Scholar] [CrossRef] [PubMed]
- Kaur, S.; Baine, M.J.; Jain, M.; Sasson, A.R.; Batra, S.K. Early diagnosis of pancreatic cancer: Challenges and new developments. Biomark. Med. 2012, 6, 597–612. [Google Scholar] [CrossRef]
- McGuigan, A.; Kelly, P.; Turkington, R.C.; Jones, C.; Coleman, H.G.; McCain, R.S. Pancreatic cancer: A review of clinical diagnosis, epidemiology, treatment and outcomes. World J. Gastroenterol. 2018, 24, 4846–4861. [Google Scholar] [CrossRef] [PubMed]
- Moutinho-Ribeiro, P.; Iglesias-Garcia, J.; Gaspar, R.; Macedo, G. Early pancreatic cancer—The role of endoscopic ultrasound with or without tissue acquisition in diagnosis and staging. Dig. Liver. Dis. 2019, 51, 4–9. [Google Scholar] [CrossRef]
- Rachagani, S.; Torres, M.P.; Kumar, S.; Haridas, D.; Baine, M.; Macha, M.A.; Kaur, S.; Ponnusamy, M.P.; Dey, P.; Seshacharyulu, P.; et al. Mucin (Muc) expression during pancreatic cancer progression in spontaneous mouse model: Potential implications for diagnosis and therapy. J. Hematol. Oncol. 2012, 5, 68. [Google Scholar] [CrossRef] [PubMed]
- Balagué, C.; Audié, J.-P.; Porchet, N.; Real, F.X. In situ hybridization shows distinct patterns of mucin gene expression in normal, benign, and malignant pancreas tissues. Gastroenterology 1995, 109, 953–964. [Google Scholar] [CrossRef]
- Park, H.-U.; Kim, J.-W.; Kim, G.E.; Bae, H.-I.; Crawley, S.C.; Yang, S.C.; Gum, J.R., Jr.; Batra, S.K.; Rousseau, K.; Swallow, D.M.; et al. Aberrant Expression of MUC3 and MUC4 Membrane-Associated Mucins and Sialyl Lex Antigen in Pancreatic Intraepithelial Neoplasia. Pancreas 2003, 26, e48–e54. [Google Scholar] [CrossRef]
- Adsay, N.V.; Merati, K.; Andea, A.; Sarkar, F.; Hruban, R.H.; Wilentz, R.E.; Goggins, M.; Iocobuzio-Donahue, C.; Longnecker, D.S.; Klimstra, D.S. The Dichotomy in the Preinvasive Neoplasia to Invasive Carcinoma Sequence in the Pancreas: Differential Expression of MUC1 and MUC2 Supports the Existence of Two Separate Pathways of Carcinogenesis. Mod. Pathol. 2002, 15, 1087–1095. [Google Scholar] [CrossRef]
- Stiles, Z.E.; Khan, S.; Patton, K.T.; Jaggi, M.; Behrman, S.W.; Chauhan, S.C. Transmembrane mucin MUC13 distinguishes intraductal papillary mucinous neoplasms from non-mucinous cysts and is associated with high-risk lesions. HPB 2019, 21, 87–95. [Google Scholar] [CrossRef]
- Xu, W.; Zhang, M.; Liu, L.; Yin, M.; Xu, C.; Weng, Z. Association of mucin family members with prognostic significance in pancreatic cancer patients: A meta-analysis. PLoS ONE 2022, 17, e0269612. [Google Scholar] [CrossRef]
- Wang, S.; You, L.; Dai, M.; Zhao, Y. Quantitative assessment of the diagnostic role of mucin family members in pancreatic cancer: A meta-analysis. Ann. Transl. Med. 2021, 9, 192. [Google Scholar] [CrossRef] [PubMed]
- Striefler, J.K.; Riess, H.; Lohneis, P.; Bischoff, S.; Kurreck, A.; Modest, D.P.; Bahra, M.; Oettle, H.; Sinn, M.; Bläker, H.; et al. Mucin-1 Protein Is a Prognostic Marker for Pancreatic Ductal Adenocarcinoma: Results From the CONKO-001 Study. Front. Oncol. 2021, 11, 670396. [Google Scholar] [CrossRef] [PubMed]
- Dolscheid-Pommerich, R.C.; Manekeller, S.; Walgenbach-Brünagel, G.; Kalff, J.C.; Hartmann, G.; Wagner, B.S.; Holdenrieder, S. Clinical Performance of CEA, CA19-9, CA15-3, CA125 and AFP in Gastrointestinal Cancer Using LOCI™-based Assays. Anticancer Res. 2017, 37, 353–359. [Google Scholar] [CrossRef]
- Zhang, M.; Zhang, Y.; Fu, J.; Zhang, L. Serum CA125 levels are decreased in rectal cancer but increased in fibrosis-associated diseases and in most types of cancers. Prog. Mol. Biol. Transl. Sci. 2019, 162, 241–252. [Google Scholar] [CrossRef]
- Xu, H.X.; Li, S.; Wu, C.T.; Qi, Z.H.; Wang, W.Q.; Jin, W.; Gao, H.L.; Zhang, S.R.; Xu, J.Z.; Liu, C.; et al. Postoperative serum CA19-9, CEA and CA125 predicts the response to adjuvant chemoradiotherapy following radical resection in pancreatic adenocarcinoma. Pancreatology 2018, 18, 671–677. [Google Scholar] [CrossRef]
- Song, J.; Sokoll, L.J.; Chan, D.W.; Zhang, Z. Validation of Serum Biomarkers That Complement CA19-9 in Detecting Early Pancreatic Cancer Using Electrochemiluminescent-Based Multiplex Immunoassays. Biomedicines 2021, 9, 1897. [Google Scholar] [CrossRef]
- Li, N.S.; Lin, W.L.; Hsu, Y.P.; Chen, Y.T.; Shiue, Y.L.; Yang, H.W. Combined Detection of CA19-9 and MUC1 Using a Colorimetric Immunosensor Based on Magnetic Gold Nanorods for Ultrasensitive Risk Assessment of Pancreatic Cancer. ACS Appl. Bio. Mater. 2019, 2, 4847–4855. [Google Scholar] [CrossRef] [PubMed]
- Hogendorf, P.; Durczyński, A.; Skulimowski, A.; Kumor, A.; Poznańska, G.; Strzelczyk, J. Growth differentiation factor (GDF-15) concentration combined with Ca125 levels in serum is superior to commonly used cancer biomarkers in differentiation of pancreatic mass. Cancer Biomark. 2018, 21, 505–511. [Google Scholar] [CrossRef] [PubMed]
- Wang, G.; Lipert, R.J.; Jain, M.; Kaur, S.; Chakraboty, S.; Torres, M.P.; Batra, S.K.; Brand, R.E.; Porter, M.D. Detection of the potential pancreatic cancer marker MUC4 in serum using surface-enhanced Raman scattering. Anal. Chem. 2011, 83, 2554–2561. [Google Scholar] [CrossRef]
- Banaei, N.; Foley, A.; Houghton, J.M.; Sun, Y.; Kim, B. Multiplex detection of pancreatic cancer biomarkers using a SERS-based immunoassay. Nanotechnology 2017, 28, 455101. [Google Scholar] [CrossRef]
- Welch, H.G.; Albertsen, P.C. Prostate cancer diagnosis and treatment after the introduction of prostate-specific antigen screening: 1986-2005. J. Natl. Cancer Inst. 2009, 101, 1325–1329. [Google Scholar] [CrossRef]
- Naji, L.; Randhawa, H.; Sohani, Z.; Dennis, B.; Lautenbach, D.; Kavanagh, O.; Bawor, M.; Banfield, L.; Profetto, J. Digital Rectal Examination for Prostate Cancer Screening in Primary Care: A Systematic Review and Meta-Analysis. Ann. Fam. Med. 2018, 16, 149–154. [Google Scholar] [CrossRef]
- Karunasinghe, N.; Minas, T.Z.; Bao, B.-Y.; Lee, A.; Wang, A.; Zhu, S.; Masters, J.; Goudie, M.; Huang, S.-P.; Jenkins, F.J.; et al. Assessment of factors associated with PSA level in prostate cancer cases and controls from three geographical regions. Sci. Rep. 2022, 12, 55. [Google Scholar] [CrossRef]
- Tarantino, G.; Crocetto, F.; Vito, C.D.; Martino, R.; Pandolfo, S.D.; Creta, M.; Aveta, A.; Buonerba, C.; Imbimbo, C. Clinical factors affecting prostate-specific antigen levels in prostate cancer patients undergoing radical prostatectomy: A retrospective study. Future Sci. OA 2021, 7, Fso643. [Google Scholar] [CrossRef] [PubMed]
- Etzioni, R.; Penson, D.F.; Legler, J.M.; di Tommaso, D.; Boer, R.; Gann, P.H.; Feuer, E.J. Overdiagnosis due to prostate-specific antigen screening: Lessons from U.S. prostate cancer incidence trends. J. Natl. Cancer Inst. 2002, 94, 981–990. [Google Scholar] [CrossRef]
- Joshi, M.D.; Ahmad, R.; Yin, L.; Raina, D.; Rajabi, H.; Bubley, G.; Kharbanda, S.; Kufe, D. MUC1 oncoprotein is a druggable target in human prostate cancer cells. Mol. Cancer 2009, 8, 3056–3065. [Google Scholar] [CrossRef] [PubMed]
- Singh, A.P.; Chauhan, S.C.; Bafna, S.; Johansson, S.L.; Smith, L.M.; Moniaux, N.; Lin, M.F.; Batra, S.K. Aberrant expression of transmembrane mucins, MUC1 and MUC4, in human prostate carcinomas. Prostate 2006, 66, 421–429. [Google Scholar] [CrossRef]
- Franks, L.M. Latent carcinoma of the prostate. J. Pathol. Bacteriol. 1954, 68, 603–616. [Google Scholar] [CrossRef]
- Franks, L.M. Estrogen-treated prostatic cancer: The variation in responsiveness of tumor ells. Cancer 1960, 13, 490–501. [Google Scholar] [CrossRef]
- Franks, L.M.; O’Shea, J.D.; Thomson, A.E. Mucin in the Prostate: A Histochemical Study in Normal Glands, Latent, Clinical, and Colloid Cancers. Cancer 1964, 17, 983–991. [Google Scholar] [CrossRef] [PubMed]
- Middleton, E.; Williams, P.C. Electrolyte and fluid appetite and balance in male guinea-pigs: Effects of stilboestrol treatment and renal function tests. J. Endocrinol. 1974, 61, 381–399. [Google Scholar] [CrossRef]
- Wong, N.; Major, P.; Kapoor, A.; Wei, F.; Yan, J.; Aziz, T.; Zheng, M.; Jayasekera, D.; Cutz, J.C.; Chow, M.J.; et al. Amplification of MUC1 in prostate cancer metastasis and CRPC development. Oncotarget 2016, 7, 83115–83133. [Google Scholar] [CrossRef]
- Yasumizu, Y.; Rajabi, H.; Jin, C.; Hata, T.; Pitroda, S.; Long, M.D.; Hagiwara, M.; Li, W.; Hu, Q.; Liu, S.; et al. MUC1-C regulates lineage plasticity driving progression to neuroendocrine prostate cancer. Nat. Commun. 2020, 11, 338. [Google Scholar] [CrossRef] [PubMed]
- Hagiwara, M.; Yasumizu, Y.; Yamashita, N.; Rajabi, H.; Fushimi, A.; Long, M.D.; Li, W.; Bhattacharya, A.; Ahmad, R.; Oya, M.; et al. MUC1-C Activates the BAF (mSWI/SNF) Complex in Prostate Cancer Stem Cells. Cancer Res. 2021, 81, 1111–1122. [Google Scholar] [CrossRef]
- Rico, S.D.; Mahnken, M.; Buscheck, F.; Dum, D.; Luebke, A.M.; Kluth, M.; Hube-Magg, C.; Hinsch, A.; Hoflmayer, D.; Moller-Koop, C.; et al. MUC5AC Expression in Various Tumor Types and Nonneoplastic Tissue: A Tissue Microarray Study on 10,399 Tissue Samples. Technol. Cancer Res. Treat. 2021, 20, 15330338211043328. [Google Scholar] [CrossRef]
- Santos, N.J.; Ximenes, P.P.; Constantino, F.B.; Carvalho, H.F.; Felisbino, S.L. Mucinous metaplasia in Pten conditional knockout mice and mucin family genes as prognostic markers for prostate cancer. Life Sci. 2022, 293, 120264. [Google Scholar] [CrossRef] [PubMed]
- Cao, H.; Fang, X.; Li, H.; Li, H.; Kong, J. Ultrasensitive detection of mucin 1 biomarker by immuno-loop-mediated isothermal amplification. Talanta 2017, 164, 588–592. [Google Scholar] [CrossRef] [PubMed]
- Collaboration, G.B.o.D.L.C. The Burden of Primary Liver Cancer and Underlying Etiologies From 1990 to 2015 at the Global, Regional, and National Level: Results From the Global Burden of Disease Study 2015. JAMA Oncol. 2017, 3, 1683–1691. [Google Scholar] [CrossRef]
- Lin, L.; Yan, L.; Liu, Y.; Qu, C.; Ni, J.; Li, H. The Burden and Trends of Primary Liver Cancer Caused by Specific Etiologies from 1990 to 2017 at the Global, Regional, National, Age, and Sex Level Results from the Global Burden of Disease Study 2017. Liver Cancer 2020, 9, 563–582. [Google Scholar] [CrossRef]
- Hennedige, T.; Venkatesh, S.K. Advances in computed tomography and magnetic resonance imaging of hepatocellular carcinoma. World J. Gastroenterol. 2016, 22, 205–220. [Google Scholar] [CrossRef]
- Jiang, H.Y.; Chen, J.; Xia, C.C.; Cao, L.K.; Duan, T.; Song, B. Noninvasive imaging of hepatocellular carcinoma: From diagnosis to prognosis. World J. Gastroenterol. 2018, 24, 2348–2362. [Google Scholar] [CrossRef] [PubMed]
- Zhou, F.; Shang, W.; Yu, X.; Tian, J. Glypican-3: A promising biomarker for hepatocellular carcinoma diagnosis and treatment. Med. Res. Rev. 2018, 38, 741–767. [Google Scholar] [CrossRef] [PubMed]
- Gao, Y.X.; Yang, T.W.; Yin, J.M.; Yang, P.X.; Kou, B.X.; Chai, M.Y.; Liu, X.N.; Chen, D.X. Progress and prospects of biomarkers in primary liver cancer (Review). Int. J. Oncol. 2020, 57, 54–66. [Google Scholar] [CrossRef] [PubMed]
- Huang, Z.; Zhou, J.K.; Peng, Y.; He, W.; Huang, C. The role of long noncoding RNAs in hepatocellular carcinoma. Mol. Cancer 2020, 19, 77. [Google Scholar] [CrossRef]
- Singal, A.G.; Hoshida, Y.; Pinato, D.J.; Marrero, J.; Nault, J.-C.; Paradis, V.; Tayob, N.; Sherman, M.; Lim, Y.S.; Feng, Z.; et al. International Liver Cancer Association (ILCA) White Paper on Biomarker Development for Hepatocellular Carcinoma. Gastroenterology 2021, 160, 2572–2584. [Google Scholar] [CrossRef]
- Wu, X.; Wang, Q.; Lu, Y.; Zhang, J.; Yin, H.; Yi, Y. Clinical application of thioredoxin reductase as a novel biomarker in liver cancer. Sci. Rep. 2021, 11, 6069. [Google Scholar] [CrossRef]
- Yuan, H.; Wang, J.; Wang, F.; Zhang, N.; Li, Q.; Xie, F.; Chen, T.; Zhai, R.; Wang, F.; Guo, Y.; et al. Mucin 1 gene silencing inhibits the growth of SMMC-7721 human hepatoma cells through Bax-mediated mitochondrial and caspase-8-mediated death receptor apoptotic pathways. Mol. Med. Rep. 2015, 12, 6782–6788. [Google Scholar] [CrossRef]
- Li, Q.; Liu, G.; Shao, D.; Wang, J.; Yuan, H.; Chen, T.; Zhai, R.; Ni, W.; Tai, G. Mucin1 mediates autocrine transforming growth factor beta signaling through activating the c-Jun N-terminal kinase/activator protein 1 pathway in human hepatocellular carcinoma cells. Int. J. Biochem. Cell Biol. 2015, 59, 116–125. [Google Scholar] [CrossRef]
- Huang, Y.; Huang, X.; Zeng, J.; Lin, J. Knockdown of MUC16 (CA125) Enhances the Migration and Invasion of Hepatocellular Carcinoma Cells. Front. Oncol. 2021, 11, 667669. [Google Scholar] [CrossRef] [PubMed]
- Dai, Y.; Liu, L.; Zeng, T.; Liang, J.Z.; Song, Y.; Chen, K.; Li, Y.; Chen, L.; Zhu, Y.H.; Li, J.; et al. Overexpression of MUC13, a Poor Prognostic Predictor, Promotes Cell Growth by Activating Wnt Signaling in Hepatocellular Carcinoma. Am. J. Pathol. 2018, 188, 378–391. [Google Scholar] [CrossRef]
- Cao, Y.; Karsten, U.; Otto, G.; Bannasch, P. Expression of MUC1, Thomsen-Friedenreich antigen, Tn, sialosyl-Tn, and alpha2,6-linked sialic acid in hepatocellular carcinomas and preneoplastic hepatocellular lesions. Virchows Arch. 1999, 434, 503–509. [Google Scholar] [CrossRef]
- Wang, J.; Liu, G.; Li, Q.; Wang, F.; Xie, F.; Zhai, R.; Guo, Y.; Chen, T.; Zhang, N.; Ni, W.; et al. Mucin1 promotes the migration and invasion of hepatocellular carcinoma cells via JNK-mediated phosphorylation of Smad2 at the C-terminal and linker regions. Oncotarget 2015, 6, 19264–19278. [Google Scholar] [CrossRef] [PubMed]
- Li, Q.; Wang, F.; Liu, G.; Yuan, H.; Chen, T.; Wang, J.; Xie, F.; Zhai, R.; Wang, F.; Guo, Y.; et al. Impact of Mucin1 knockdown on the phenotypic characteristics of the human hepatocellular carcinoma cell line SMMC-7721. Oncol. Rep. 2014, 31, 2811–2819. [Google Scholar] [CrossRef] [PubMed]
- Bozkaya, G.; Korhan, P.; Cokaklı, M.; Erdal, E.; Sağol, O.; Karademir, S.; Korch, C.; Atabey, N. Cooperative interaction of MUC1 with the HGF/c-Met pathway during hepatocarcinogenesis. Mol. Cancer 2012, 11, 64. [Google Scholar] [CrossRef]
- Sasaki, M.; Nakanuma, Y. Expression of mucin core protein of mammary type in primary liver cancer. Hepatology 1994, 20, 1192–1197. [Google Scholar] [CrossRef] [PubMed]
- Zimmerman, R.L.; Burke, M.; Young, N.A.; Solomides, C.C.; Bibbo, M. Diagnostic utility of Glut-1 and CA 15-3 in discriminating adenocarcinoma from hepatocellular carcinoma in liver tumors biopsied by fine-needle aspiration. Cancer 2002, 96, 53–57. [Google Scholar] [CrossRef]
- Yuan, S.F.; Li, K.Z.; Wang, L.; Dou, K.F.; Yan, Z.; Han, W.; Zhang, Y.Q. Expression of MUC1 and its significance in hepatocellular and cholangiocarcinoma tissue. World J. Gastroenterol. 2005, 11, 4661–4666. [Google Scholar] [CrossRef]
- Pedersen, J.W.; Wandall, H.H. Autoantibodies as Biomarkers in Cancer. Lab. Med. 2011, 42, 623–628. [Google Scholar] [CrossRef]
- Jiang, D.; Zhang, X.; Liu, M.; Wang, Y.; Wang, T.; Pei, L.; Wang, P.; Ye, H.; Shi, J.; Song, C.; et al. Discovering Panel of Autoantibodies for Early Detection of Lung Cancer Based on Focused Protein Array. Front. Immunol. 2021, 12, 8922. [Google Scholar] [CrossRef]
- Yang, B.; Li, X.; Ren, T.; Yin, Y. Autoantibodies as diagnostic biomarkers for lung cancer: A systematic review. Cell Death Discov. 2019, 5, 126. [Google Scholar] [CrossRef]
- Kunihiro, A.G.; Sarrett, S.M.; Lastwika, K.J.; Solan, J.L.; Pisarenko, T.; Keinänen, O.; Rodriguez, C.; Taverne, L.R.; Fitzpatrick, A.L.; Li, C.I.; et al. CD133 as a Biomarker for an Autoantibody-to-ImmunoPET Paradigm for the Early Detection of Small Cell Lung Cancer. J. Nucl. Med. 2022, 63, 1701–1707. [Google Scholar] [CrossRef]
- Kaaks, R.; Fortner, R.T.; Hüsing, A.; Barrdahl, M.; Hopper, M.; Johnson, T.; Tjønneland, A.; Hansen, L.; Overvad, K.; Fournier, A.; et al. Tumor-associated autoantibodies as early detection markers for ovarian cancer? A prospective evaluation. Int. J. Cancer 2018, 143, 515–526. [Google Scholar] [CrossRef] [PubMed]
- Hong, C.-Q.; Weng, X.-F.; Huang, X.-C.; Chu, L.-Y.; Wei, L.-F.; Lin, Y.-W.; Chen, L.-Y.; Liu, C.-T.; Xu, Y.-W.; Peng, Y.-H. A Panel of Tumor-associated Autoantibodies for the Detection of Early-stage Breast Cancer. J. Cancer 2021, 12, 2747–2755. [Google Scholar] [CrossRef]
- Kobayashi, M.; Katayama, H.; Fahrmann, J.F.; Hanash, S.M. Development of autoantibody signatures for common cancers. Semin. Immunol. 2020, 47, 101388. [Google Scholar] [CrossRef]
- Zhuang, L.; Huang, C.; Ning, Z.; Yang, L.; Zou, W.; Wang, P.; Cheng, C.-S.; Meng, Z. Circulating tumor-associated autoantibodies as novel diagnostic biomarkers in pancreatic adenocarcinoma. Int. J. Cancer 2023, 152, 1013–1024. [Google Scholar] [CrossRef]
- Wang, H.; Li, X.; Zhou, D.; Huang, J. Autoantibodies as biomarkers for colorectal cancer: A systematic review, meta-analysis, and bioinformatics analysis. Int. J. Biol. Markers 2019, 34, 334–347. [Google Scholar] [CrossRef] [PubMed]
- Pedersen, J.W.; Gentry-Maharaj, A.; Nøstdal, A.; Fourkala, E.-O.; Dawnay, A.; Burnell, M.; Zaikin, A.; Burchell, J.; Papadimitriou, J.T.; Clausen, H.; et al. Cancer-associated autoantibodies to MUC1 and MUC4—A blinded case–control study of colorectal cancer in UK collaborative trial of ovarian cancer screening. Int. J. Cancer 2014, 134, 2180–2188. [Google Scholar] [CrossRef]
- Saif, M.W.; Zalonis, A.; Syrigos, K. The clinical significance of autoantibodies in gastrointestinal malignancies: An overview. Expert Opin. Biol. Ther. 2007, 7, 493–507. [Google Scholar] [CrossRef]
- Frietze, K.M.; Roden, R.B.; Lee, J.-H.; Shi, Y.; Peabody, D.S.; Chackerian, B. Identification of Anti-CA125 Antibody Responses in Ovarian Cancer Patients by a Novel Deep Sequence–Coupled Biopanning Platform. Cancer Immunol. Res. 2016, 4, 157–164. [Google Scholar] [CrossRef]
- Cramer, D.W.; Williams, K.; Vitonis, A.F.; Yamamoto, H.S.; Stuebe, A.; Welch, W.R.; Titus, L.; Fichorova, R.N. Puerperal mastitis: A reproductive event of importance affecting anti-mucin antibody levels and ovarian cancer risk. Cancer Causes Control. 2013, 24, 1911–1923. [Google Scholar] [CrossRef]
- Chapman, C.; Murray, A.; Chakrabarti, J.; Thorpe, A.; Woolston, C.; Sahin, U.; Barnes, A.; Robertson, J. Autoantibodies in breast cancer: Their use as an aid to early diagnosis. Ann. Oncol. 2007, 18, 868–873. [Google Scholar] [CrossRef] [PubMed]
- Blixt, O.; Bueti, D.; Burford, B.; Allen, D.; Julien, S.; Hollingsworth, M.; Gammerman, A.; Fentiman, I.; Taylor-Papadimitriou, J.; Burchell, J.M. Autoantibodies to aberrantly glycosylated MUC1 in early stage breast cancer are associated with a better prognosis. Breast Cancer Res. 2011, 13, R25. [Google Scholar] [CrossRef]
- Tang, Y.; Wang, L.; Zhang, P.; Wei, H.; Gao, R.; Liu, X.; Yu, Y.; Wang, L. Detection of circulating anti-mucin 1 (MUC1) antibodies in breast tumor patients by indirect enzyme-linked immunosorbent assay using a recombinant MUC1 protein containing six tandem repeats and expressed in Escherichia coli. Clin. Vaccine Immunol. 2010, 17, 1903–1908. [Google Scholar] [CrossRef]
- Guillen-Poza, P.A.; Sanchez-Fernandez, E.M.; Artigas, G.; Garcia Fernandez, J.M.; Hinou, H.; Ortiz Mellet, C.; Nishimura, S.I.; Garcia-Martin, F. Amplified Detection of Breast Cancer Autoantibodies Using MUC1-Based Tn Antigen Mimics. J. Med. Chem. 2020, 63, 8524–8533. [Google Scholar] [CrossRef] [PubMed]
- Xia, J.; Shi, J.; Wang, P.; Song, C.; Wang, K.; Zhang, J.; Ye, H. Tumour-Associated Autoantibodies as Diagnostic Biomarkers for Breast Cancer: A Systematic Review and Meta-Analysis. Scand. J. Immunol. 2016, 83, 393–408. [Google Scholar] [CrossRef] [PubMed]
- Laidi, F.; Bouziane, A.; Errachid, A.; Zaoui, F. Usefulness of Salivary and Serum Auto-antibodies Against Tumor Biomarkers HER2 and MUC1 in Breast Cancer Screening. Asian Pac. J. Cancer Prev. 2016, 17, 335–339. [Google Scholar] [CrossRef]
- Hirasawa, Y.; Kohno, N.; Yokoyama, A.; Kondo, K.; Hiwada, K.; Miyake, M. Natural autoantibody to MUC1 is a prognostic indicator for non-small cell lung cancer. Am. J. Respir. Crit. Care Med. 2000, 161, 589–594. [Google Scholar] [CrossRef]
- Pedersen, J.W.; Blixt, O.; Bennett, E.P.; Tarp, M.A.; Dar, I.; Mandel, U.; Poulsen, S.S.; Pedersen, A.E.; Rasmussen, S.; Jess, P.; et al. Seromic profiling of colorectal cancer patients with novel glycopeptide microarray. Int. J. Cancer 2011, 128, 1860–1871. [Google Scholar] [CrossRef] [PubMed]
- Burford, B.; Gentry-Maharaj, A.; Graham, R.; Allen, D.; Pedersen, J.W.; Nudelman, A.S.; Blixt, O.; Fourkala, E.O.; Bueti, D.; Dawnay, A.; et al. Autoantibodies to MUC1 glycopeptides cannot be used as a screening assay for early detection of breast, ovarian, lung or pancreatic cancer. Br. J. Cancer 2013, 108, 2045–2055. [Google Scholar] [CrossRef] [PubMed]
- Kim, H.; Kim, D.W.; Cho, J.-Y. Exploring the key communicator role of exosomes in cancer microenvironment through proteomics. Proteom. Sci. 2019, 17, 5. [Google Scholar] [CrossRef]
- Tamura, T.; Yoshioka, Y.; Sakamoto, S.; Ichikawa, T.; Ochiya, T. Extracellular vesicles as a promising biomarker resource in liquid biopsy for cancer. Extracell. Vesicles Circ. Nucleic Acids 2021, 2, 148–174. [Google Scholar] [CrossRef]
- Hon, K.W.; Abu, N.; Ab Mutalib, N.-S.; Jamal, R. Exosomes As Potential Biomarkers and Targeted Therapy in Colorectal Cancer: A Mini-Review. Front. Pharmacol. 2017, 8, 583. [Google Scholar] [CrossRef]
- Shen, M.; Di, K.; He, H.; Xia, Y.; Xie, H.; Huang, R.; Liu, C.; Yang, M.; Zheng, S.; He, N.; et al. Progress in exosome associated tumor markers and their detection methods. Mol. Biomed. 2020, 1, 3. [Google Scholar] [CrossRef]
- Sandfeld-Paulsen, B.; Jakobsen, K.R.; Bæk, R.; Folkersen, B.H.; Rasmussen, T.R.; Meldgaard, P.; Varming, K.; Jørgensen, M.M.; Sorensen, B.S. Exosomal Proteins as Diagnostic Biomarkers in Lung Cancer. J. Thorac. Oncol. 2016, 11, 1701–1710. [Google Scholar] [CrossRef]
- Osteikoetxea, X.; Benke, M.; Rodriguez, M.; Pálóczi, K.; Sódar, B.W.; Szvicsek, Z.; Szabó-Taylor, K.; Vukman, K.V.; Kittel, Á.; Wiener, Z.; et al. Detection and proteomic characterization of extracellular vesicles in human pancreatic juice. Biochem. Biophys. Res. Commun. 2018, 499, 37–43. [Google Scholar] [CrossRef]
- Yang, K.S.; Ciprani, D.; O’Shea, A.; Liss, A.S.; Yang, R.; Fletcher-Mercaldo, S.; Mino-Kenudson, M.; Fernández-Del Castillo, C.; Weissleder, R. Extracellular Vesicle Analysis Allows for Identification of Invasive IPMN. Gastroenterology 2021, 160, 1345–1358. [Google Scholar] [CrossRef]
- Atlihan-Gundogdu, E.; Ilem-Ozdemir, D.; Ekinci, M.; Ozgenc, E.; Demir, E.S.; Sánchez-Dengra, B.; González-Alvárez, I. Recent developments in cancer therapy and diagnosis. J. Pharm. Investig. 2020, 50, 349–361. [Google Scholar] [CrossRef]
- Echeverría, C.; Nualart, F.; Ferrada, L.; Smith, G.J.; Godoy, A.S. Hexose Transporters in Cancer: From Multifunctionality to Diagnosis and Therapy. Trends Endocrinol. Metab. 2021, 32, 198–211. [Google Scholar] [CrossRef] [PubMed]
- Chan, C.Y.; Tan, K.V.; Cornelissen, B. PARP Inhibitors in Cancer Diagnosis and Therapy. Clin. Cancer Res. 2021, 27, 1585. [Google Scholar] [CrossRef]
- Lee, Y.T.; Tan, Y.J.; Oon, C.E. Molecular targeted therapy: Treating cancer with specificity. Eur. J. Pharmacol. 2018, 834, 188–196. [Google Scholar] [CrossRef]
- McCormack, V.; Aggarwal, A. Early cancer diagnosis: Reaching targets across whole populations amidst setbacks. Br. J. Cancer 2021, 124, 1181–1182. [Google Scholar] [CrossRef]
- Whitaker, K. Earlier diagnosis: The importance of cancer symptoms. Lancet Oncol. 2020, 21, 6–8. [Google Scholar] [CrossRef]
- Hwang, T.J.; Carpenter, D.; Lauffenburger, J.C.; Wang, B.; Franklin, J.M.; Kesselheim, A.S. Failure of Investigational Drugs in Late-Stage Clinical Development and Publication of Trial Results. JAMA Intern. Med. 2016, 176, 1826–1833. [Google Scholar] [CrossRef] [PubMed]
- Shiri, P.; Ramezanpour, S.; Amani, A.M.; Dehaen, W. A patent review on efficient strategies for the total synthesis of pazopanib, regorafenib and lenvatinib as novel anti-angiogenesis receptor tyrosine kinase inhibitors for cancer therapy. Mol. Divers. 2022, 26, 2981–3002. [Google Scholar] [CrossRef]
- Funston, G.; Hamilton, W.; Abel, G.; Crosbie, E.J.; Rous, B.; Walter, F.M. The diagnostic performance of CA125 for the detection of ovarian and non-ovarian cancer in primary care: A population-based cohort study. PLoS Med. 2020, 17, e1003295. [Google Scholar] [CrossRef]
- Liu, L.; Xiang, J.; Chen, R.; Fu, D.; Hong, D.; Hao, J.; Li, Y.; Li, J.; Li, S.; Mou, Y.; et al. The clinical utility of CA125/MUC16 in pancreatic cancer: A consensus of diagnostic, prognostic and predictive updates by the Chinese Study Group for Pancreatic Cancer (CSPAC). Int. J. Oncol. 2016, 48, 900–907. [Google Scholar] [CrossRef] [PubMed]
- Bottoni, P.; Scatena, R. The Role of CA 125 as Tumor Marker: Biochemical and Clinical Aspects. Adv. Exp. Med. Biol. 2015, 867, 229–244. [Google Scholar] [CrossRef] [PubMed]
- Heitzer, E.; Perakis, S.; Geigl, J.B.; Speicher, M.R. The potential of liquid biopsies for the early detection of cancer. NPJ Precis. Oncol. 2017, 1, 36. [Google Scholar] [CrossRef] [PubMed]
- Rebbeck, T.R.; Burns-White, K.; Chan, A.T.; Emmons, K.; Freedman, M.; Hunter, D.J.; Kraft, P.; Laden, F.; Mucci, L.; Parmigiani, G.; et al. Precision Prevention and Early Detection of Cancer: Fundamental Principles. Cancer Discov. 2018, 8, 803. [Google Scholar] [CrossRef]
- Walter, F.M.; Thompson, M.J.; Wellwood, I.; Abel, G.A.; Hamilton, W.; Johnson, M.; Lyratzopoulos, G.; Messenger, M.P.; Neal, R.D.; Rubin, G.; et al. Evaluating diagnostic strategies for early detection of cancer: The CanTest framework. BMC Cancer 2019, 19, 586. [Google Scholar] [CrossRef] [PubMed]
- Thomas, D.; Rathinavel, A.K.; Radhakrishnan, P. Altered glycosylation in cancer: A promising target for biomarkers and therapeutics. Biochim. Et Biophys. Acta (BBA)—Rev. Cancer 2021, 1875, 188464. [Google Scholar] [CrossRef] [PubMed]
- Locke, W.J.; Guanzon, D.; Ma, C.; Liew, Y.J.; Duesing, K.R.; Fung, K.Y.C.; Ross, J.P. DNA Methylation Cancer Biomarkers: Translation to the Clinic. Front. Genet. 2019, 10, 1150. [Google Scholar] [CrossRef] [PubMed]
- Pinho, S.S.; Reis, C.A. Glycosylation in cancer: Mechanisms and clinical implications. Nat. Rev. Cancer 2015, 15, 540–555. [Google Scholar] [CrossRef]
- Wei, Z.-W.; Wu, J.; Huang, W.-B.; Li, J.; Lu, X.-F.; Yuan, Y.-J.; Xiong, W.-J.; Zhang, X.-H.; Wang, W.; He, Y.-L.; et al. Immune-infiltration based signature as a novel prognostic biomarker in gastrointestinal stromal tumour. eBioMedicine 2020, 57, 102850. [Google Scholar] [CrossRef]
- Kresovich, J.K.; O’Brien, K.M.; Xu, Z.; Weinberg, C.R.; Sandler, D.P.; Taylor, J.A. Prediagnostic Immune Cell Profiles and Breast Cancer. JAMA Netw. Open 2020, 3, e1919536. [Google Scholar] [CrossRef]
- Gao, X.-P.; Dong, J.-J.; Xie, T.; Guan, X. Integrative Analysis of MUC4 to Prognosis and Immune Infiltration in Pan-Cancer: Friend or Foe? Front. Cell Dev. Biol. 2021, 9, 695544. [Google Scholar] [CrossRef]
- Fan, H.Y.; Duan, D.M.; Liu, Y.F. The diagnostic value of determination of serum GOLPH3 associated with CA125, CA19.9 in patients with ovarian cancer. Eur. Rev. Med. Pharm. Sci. 2017, 21, 4039–4044. [Google Scholar]
- Yang, Z.J.; Yang, G.; Jiang, Y.M.; Ran, Y.L.; Yang, Z.H.; Zhang, W.; Zhang, J.Q.; Pan, Z.M.; Li, L. [Clinical study of autoantibody spectrum against ovarian cancer associated antigens combined with CA(125) in detecting and monitoring ovarian cancer]. Zhonghua Fu Chan Ke Za Zhi 2011, 46, 113–118. [Google Scholar] [PubMed]
- LeBleu, V.S.; Kalluri, R. Exosomes as a Multicomponent Biomarker Platform in Cancer. Trends Cancer 2020, 6, 767–774. [Google Scholar] [CrossRef] [PubMed]
- Wang, L.F.; Lee, C.H.; Liang, S.S.; Hung, C.C.; Wu, Y.R.; Chien, C.Y.; Lee, C.H.; Chen, J.Y. Mucin 5AC is significantly upregulated in exosomes from the nasal lavage fluid and may promote the expression of COX-2, VEGF and MMP-9: An implication in nasal polyp pathogenesis. Rhinology 2021, 59, 328–336. [Google Scholar] [CrossRef] [PubMed]
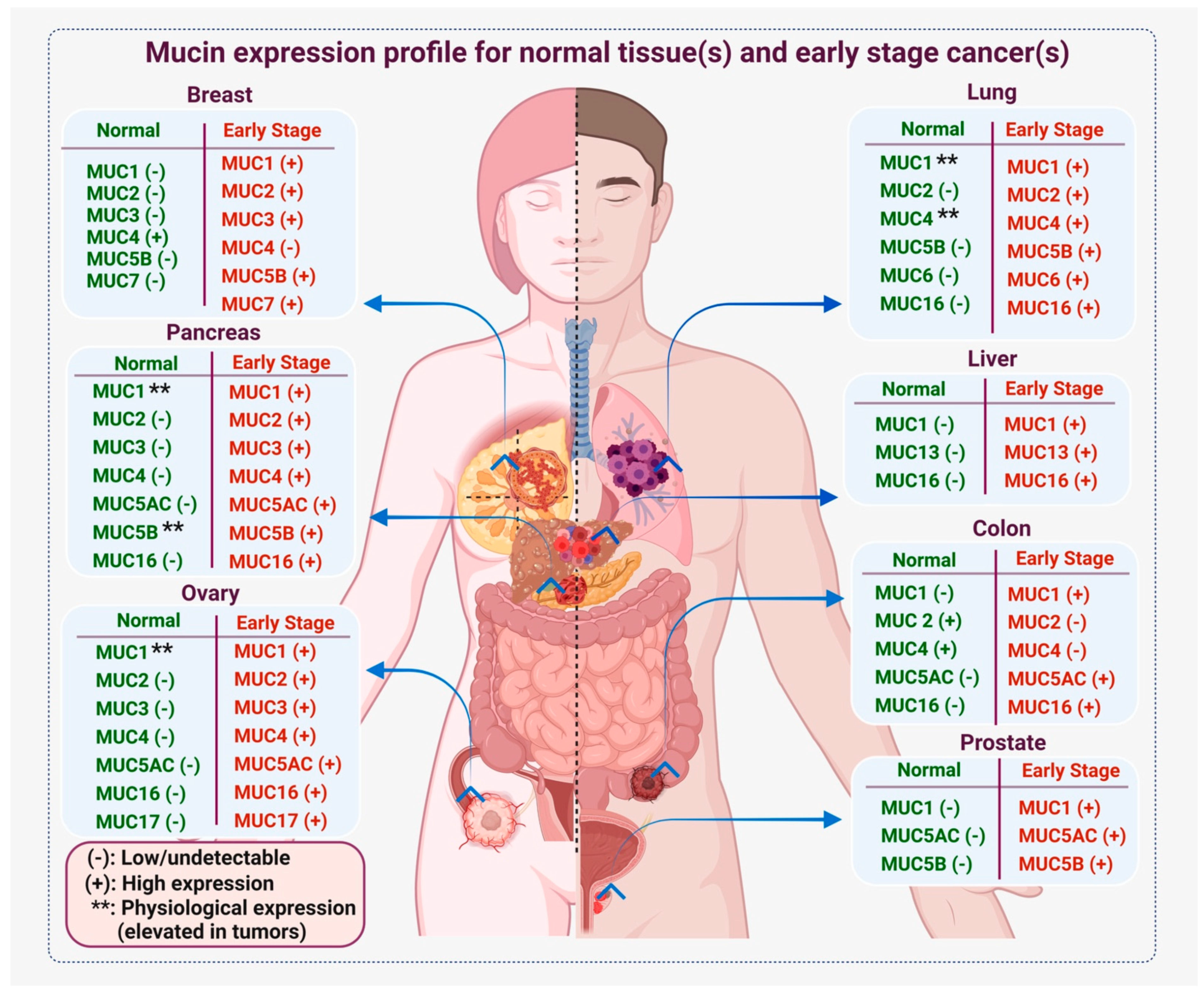
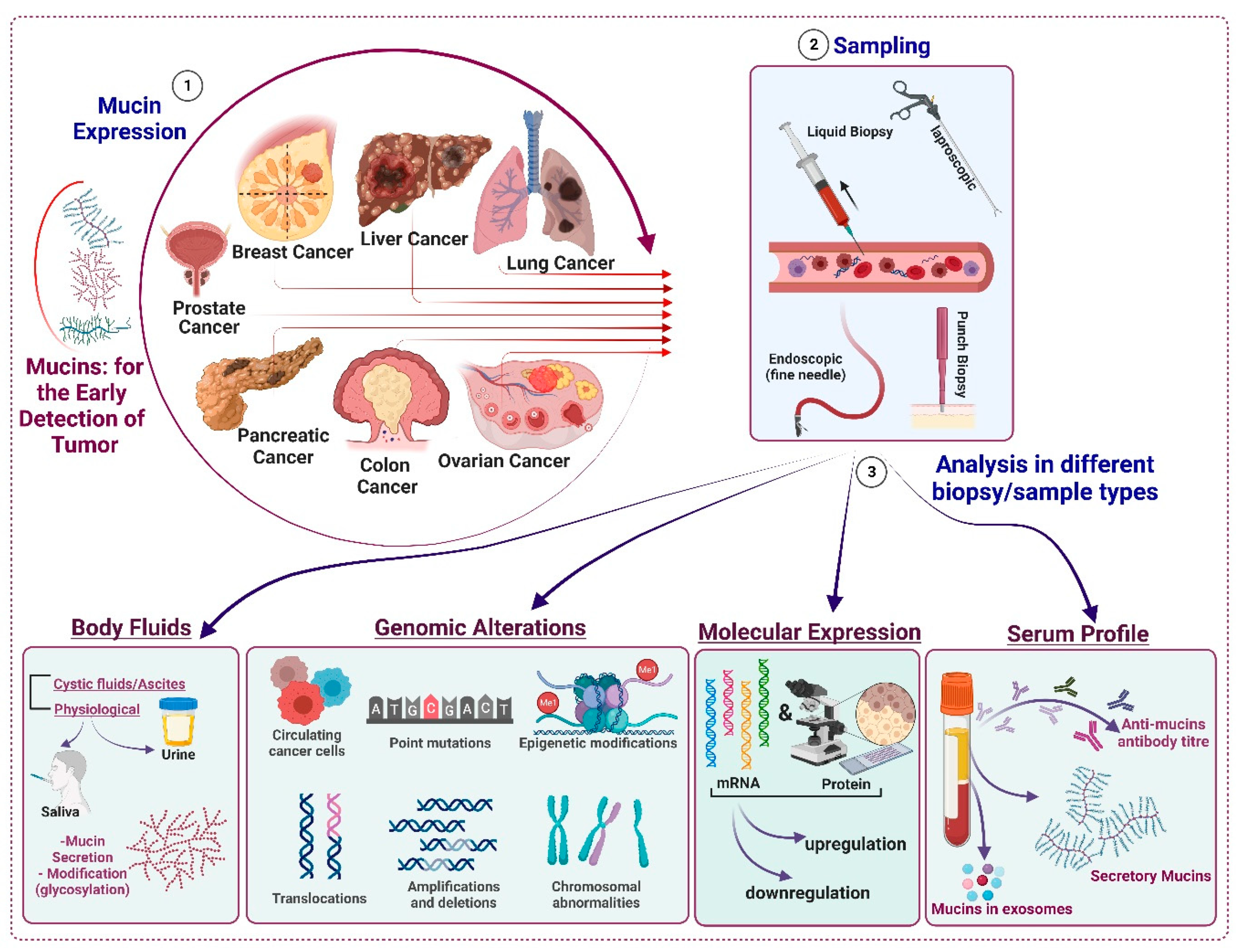
| S. No. | Sample(s) | Mucin(s)/Combination | Stage of Detection | Readout | Ref. |
|---|---|---|---|---|---|
| 1-A: Lung cancer | |||||
| 1 | Serum samples/ tumor tissues (IHC/ELISA), n = 80 samples | MUC16, IL6 | NSCLC | High MUC16 and IL6 can be used to detect LC from liquid biopsies as they are positively associated with distant organ metastasis. | [101] |
| 2 | Exosomes from cell lines and patient plasma samples (n = 27 samples) | MUC1 | NSCLC | MUC1 is enriched in the exosomes of cancer cell lines and patients (plasma) and can be used to detect NSCLC at an early stage. | [95] |
| 3 | Serum samples (n = 633) | CEA + CA125(MUC16) or CY211/NSE/SCC | NSCLC | The combination marker for NSCLC screening is CEA + CA125 (with positive cut-off range of 0.577CEA + 0.035CA125 ng/mL). | [106] |
| 4 | Serum samples (n = 289 suspected/unconfirmed, and n = 417 NSCLC, n = 96 SCLC) | CA125, CA19.9, CA15.3, TAG72-3, CYFRA21-1, CEA, SCC, NSE | NSCLC and SCLC | High CA125, CA15-3, CEA, CYFRA21-1, SCC, and TAG72-3 are early markers for NSCLC. High serum expression of CEA, TAG72-3, CA15-3, and CA125 denotes adenocarcinoma. High SCC, CEA, and CYFRA21-1 indicate squamous carcinoma. | [105] |
| 1-B: Breast cancer | |||||
| 1 | Serum sample (n = 248 samples) | CA15-3(MUC1) and chemerin | Breast cancer and benign breast tumor patients | High chemerin with CA15-3 in the serum samples provided better diagnostic performance and could be used to characterize histologic grades. | [109] |
| 2 | Plasma samples (n = 200 BC patient, n = 47 benign breast lesions samples) | CEA and CA15-3 | Breast cancer and benign breast lesions | High CEA and CA15-3 are seen in early-stage and cancer patients with node or distant organ metastasis. | [110] |
| 3 | Serum and tumor tissues (n = 433 samples) | MUC1 | Primary and metastatic breast cancer tissues | High MUC1 in circulation/serum at the early stage can be detected in advance (two years before compared to other detection methods). Inclusion with multi-model screening strategies may diagnose tumors in women missed by mammography and irrespective of breast tissue density. | [111] |
| 1-C: Ovarian cancer | |||||
| 1 | Serum samples (n = 46 stage 1 OC, n = 237 benign pelvic masses, n = 204 healthy controls) | CA125, M-CSF, and OVX1 | Stage1 ovarian cancer, benign pelvic masses, and healthy women | A panel of CA125, M-CSF, and OVX1 tumor markers can identify early-stage ovarian cancer with extremely high sensitivity and moderate specificity. | [112] |
| 2 | Serum sample (n = 71 early-stage and n = 45 late-stage OC patients, n = 131 healthy controls) | OPN, MIF, IL8 AAb, and CA125 | Early- and late-stage ovarian cancer patients | Combining OPN, MIF, IL8, and CA125 enhances the sensitivity of detection of OC patients compared to healthy controls. | [113] |
| 3 | Serum specimens (n = 75 invasive epithelial ovarian cancer and n = 547 healthy controls) | HE4 + CA72-4 and CA125 | Pre-clinical invasive epithelial ovarian cancer and healthy controls | Combining HE4 + CA72-4 complements CA125 as a biomarker panel for longitudinal screening by multiplex assay. | [114] |
| 4 | Serum samples (n = 118 patients with ovarian tumors) | CA 15–3 and CA 27.29 | Malignant and benign disease. | The serum concentration of CA 15–3 and CA 27.29 increased in malignant than in patients with benign disease. | [115] |
| 5 | Serum sample; n = 123 patients, either benign (n = 83 patients) or malignant (n = 40 patients) | CA15-3, CA27.29, and Panko Mab | Benign patients and malignant disease of the ovaries | PankoMab (anti-MUC1 antibody) has strong diagnostic potential in discriminating sera from patients with benign ovarian diseases vs. normal sera. | [116] |
| 1-D: Colorectal cancer | |||||
| 1 | Serum panel (CEA, CA19-9, CK1, and MUC1) (n = 150 colon cancer, n = 50 benign, and n = 35 healthy controls) | CEA, CA19-9, CK1, and MUC1 | Early-stage, benign, and healthy controls | Serum levels of CEA, CA19-9, CK1, and MUC1 gradually increase in benign disease to colon cancer compared to healthy controls. | [15] |
| 2 | Serum sample (n = 279 colorectal cancer patients vs. healthy controls) | CEA + CA19-9 + CA72-4 + CA125 + ferritin | Diagnostic potential and tumor status in CRC | Combining CEA + CA19-9 + CA72-4 + CA125 + ferritin has the diagnostic potential and evaluates the tumor status in colorectal cancer. | [117] |
| 3 | Serum and tissue sample (n = 22 CRC patients vs. healthy controls) | MUC1, MUC2 | Early- and late-stage CRC patients | Increased MUC1 protein was observed in serum of late-stage CRC patients compared to control, whereas MUC2 was downregulated in CRC patients, as analyzed in tissue samples. | [118] |
| 4 | Patients (n = 373) with CRC evaluated pre- and post-surgery | CEA, CA19-9, CA125, CYFRA21-1, and CA72-4 | Colorectal cancer patients | Combination of CEA, CA19-9, CA125, CYFRA21-1, and CA72-4 correlates with poor tumor differentiation and metastasis | [119] |
| 5 | Serum sample (n = 322 CRC patients vs. healthy controls) | CA125 and CEA | Colorectal cancer patients | CA125 turns out to be an independent prognostic factor in CRC with greater reliability than CEA. | [120] |
| 6 | Serum sample (n = 28 normal, n = 41 CRC/PC sera at 1 month, n = 33 CRC/PC sera at 2 months, and n = 25 CRC/PC sera at 3 months) | MUC5AC (NPC-1C Ab) | Colorectal/pancreatic cancer | MUC5AC(NPC-1C) antibody can discriminate the serum of cancer patients from normal donors in colorectal and pancreatic cancer. | [121] |
| 1-E: Pancreatic cancer | |||||
| 1 | Serum and tissue samples (n = 346 samples) | MUC5AC, CA19-9 | CP, early-stage resectable vs. late-stage non-resectable PC | Differentiates between early- and late-stage PC, and PC from CP. | [122] |
| 2 | Serum sample; early-stage PC (n = 30), late-stage PC (n = 31), 29 benign controls, 25 CPs, and 34 healthy controls | MUC5AC, CA19-9 | CP vs. early PC And early vs. late PC, compared to healthy controls | Serum MUC5AC in patients with PC (210.1 (100.5–423.8) ng/mL) and combined biomarker panel (MUC5AC and CA19-9) showed a better performance. | [123] |
| 3 | Serum sample (n = 92 samples) | CA19.9, CA125, CEA, and CA242 | Normal, benign, and PC | The combination panel enhanced the diagnostic efficiency. | [124] |
| 4 | Pancreatic juice (n = 191 samples) | MUC1, MUC2, and MUC4 | IPMNs and PC | DNA methylation status differentiates between intestinal-type and gastric-type IPMNs. | [125] |
| 5 | Serum sample (n = 31 samples) | CA19.9, CA125, CEA, and CA242 | PDAC patients undergoing cryoablation therapy | CA19.9, CEA, and TSGF for treatment assessment; CA242 for tumor staging, LN, and liver metastasis; TSGF for tumor differentiation. | [126] |
| 6 | Blood sample (n = 369 samples) | CA125 and CD4/CD8 ratio | Advanced-stage PDAC patients | Better prognosis with combined CA125 and CD4/CD8 ratio. | [127] |
| 1-F: Prostate cancer | |||||
| 1 | Serum samples and tissues biopsy (n = 57 samples) | Sialylated MUC1 and PSA | Clinical stages and prognosis | Sialylated MUC1 increases with disease progression. | [128] |
| 2 | Serum samples (n = 11 patients) | Serum MUC16/CA-125 | Tumor type or metastasis | PCa patient with elevated serum MUC16 (CA-125) had a high chance of persistent urinary symptoms and visceral metastasis. | [129] |
| 3 | Serum samples (n = 303 patients with benign and malignant disease) | Comparison of serum MUC1 and PSA | Benign vs. malignant disease | Elevated MUC1 (91% specificity) in benign disease, also high MUC1 in PSA-negative samples. | [130] |
| 4 | Serum samples | Serum anti-MUC1 antibodies and natural antigen for prostate | Early detection | High level of MUC1 antibodies in the early stages of PCa. | [131] |
| 1-G: Liver cancer | |||||
| 1 | Serum samples of n = 115 HCC patients with a history of amebic hepatic abscess (62 patients), chronic hepatitis (40 patients), and acute viral hepatitis (41 patients) | Comparison of MUC16 and AFP serum level | HCC vs. benign hepatic disease | CA125 is a highly sensitive marker for HCC but lacks specificity. | [132] |
| 2 | Serum samples from HCC patients | MUC16 and AFP analysis | HCC vs. normal controls | MUC16 could complement AFP in diagnosing HCC. MUC16 is more sensitive than AFP (92% vs. 58.8%). | [133] |
| 3 | Serum samples of n = 3440 HCC patients underwent curative hepatectomy | MUC16 and AFP | Retrospective, HCC vs. normal individuals | Elevated preoperative MUC16 in 409 patients, correlated with younger age, females, and higher AFP level. CA125 served as an independent prognostic factor of OS and RFS. | [134] |
| 4 | Serum samples of n = 306 hepatitis-B-virus-related HCC patients | MUC16 and AFP | Preoperative HCC patients | A high MUC16 level was found to be risk factor for OS and DFS and correlated with the worst prognosis. | [135] |
| 5 | Serum sample from HCC patients (n = 27), CC patients (n = 8), metastatic liver cancer patients (30), healthy controls (n = 19) | MUC1 (KL-6) | Established HCC and CC | Significant differences in MUC16 levels of CC and HCC patients were seen compared to controls. All CC patients and 18.5% of HSS patients showed positivity above the cut-off (248 U/mL). | [136] |
| 6 | Serum sample of HCC patients (n = 144) who underwent complete radiofrequency ablation of primary HCC. | Wisteria Floribunda agglutinin (WFA)-positive sialylated MUC1 | HCC patients after radiofrequency ablation | WFA-positive MUC1 correlated with HCC recurrence and was found to be associated with histological features of HCC. | [137] |
| 7 | Serum samples of n = 427 HCC patients with serum AFP level ≤200 ng/mL | MUC16 level analysis with cut off 30 U/mL | Preoperative serum analysis | CA125 levels were associated with maximum tumor diameter (>5 cm) and CA125 was found to be an independent risk factor of DFS and OS. | [138] |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Gautam, S.K.; Khan, P.; Natarajan, G.; Atri, P.; Aithal, A.; Ganti, A.K.; Batra, S.K.; Nasser, M.W.; Jain, M. Mucins as Potential Biomarkers for Early Detection of Cancer. Cancers 2023, 15, 1640. https://doi.org/10.3390/cancers15061640
Gautam SK, Khan P, Natarajan G, Atri P, Aithal A, Ganti AK, Batra SK, Nasser MW, Jain M. Mucins as Potential Biomarkers for Early Detection of Cancer. Cancers. 2023; 15(6):1640. https://doi.org/10.3390/cancers15061640
Chicago/Turabian StyleGautam, Shailendra K., Parvez Khan, Gopalakrishnan Natarajan, Pranita Atri, Abhijit Aithal, Apar K. Ganti, Surinder K. Batra, Mohd W. Nasser, and Maneesh Jain. 2023. "Mucins as Potential Biomarkers for Early Detection of Cancer" Cancers 15, no. 6: 1640. https://doi.org/10.3390/cancers15061640
APA StyleGautam, S. K., Khan, P., Natarajan, G., Atri, P., Aithal, A., Ganti, A. K., Batra, S. K., Nasser, M. W., & Jain, M. (2023). Mucins as Potential Biomarkers for Early Detection of Cancer. Cancers, 15(6), 1640. https://doi.org/10.3390/cancers15061640







