Results of a 20 Year Retrospective Analysis of Early-Stage Cervical Cancer: Should 3 cm Be Considered the New Ariadne’s Thread in Early Cervical Cancer Treatment?
Abstract
Simple Summary
Abstract
1. Introduction
2. Materials and Methods
2.1. Patients Characteristics
2.2. Patient Management
2.3. Follow-Up
2.4. Statistical Analysis
3. Results
4. Discussion
4.1. Conization
4.2. Mini-Invasive Surgery versus Open Surgery and Tumor Size
4.3. Strengths and Limitations
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Sung, H.; Ferlay, J.; Siegel, R.L.; Laversanne, M.; Soerjomataram, I.; Jemal, A.; Bray, F. Global Cancer Statistics 2020: GLOBOCAN Estimates of Incidence and Mortality Worldwide for 36 Cancers in 185 Countries. CA Cancer J. Clin. 2021, 71, 209–249. [Google Scholar] [CrossRef]
- Bhatla, N.; Berek, J.S.; Cuello Fredes, M.; Denny, L.A.; Grenman, S.; Karunaratne, K.; Kehoe, S.; Konishi, I.; Olawaiye, A.; Prat, J.; et al. Revised FIGO staging for carcinoma of the cervix uteri. Int. J. Gynecol. Obstet. 2019, 145, 129–135. [Google Scholar] [CrossRef] [PubMed]
- Cibula, D.; Pötter, R.; Planchamp, F.; Avall-Lundqvist, E.; Fischerova, D.; Haie Meder, C.; Köhler, C.; Landoni, F.; Lax, S.; Lindegaard, J.C.; et al. The European Society of Gynaecological Oncology/European Society for Radiotherapy and Oncology/European Society of Pathology guidelines for the management of patients with cervical cancer. Radiother. Oncol. 2018, 127, 404–416. [Google Scholar] [CrossRef]
- National Comprehensive Cancer Network. NCCN Clinical Practice Guidelines in Oncology: Cervical Cancer. (Version I.2022) NCCN. 2021. Available online: https://www.nccn.org/professionals/physician_gls/pdf/cervical.pdf (accessed on 1 September 2022).
- Querleu, D.; Cibula, D.; Abu-Rustum, N.R. 2017 Update on the Querleu–Morrow Classification of Radical Hysterectomy. Ann. Surg Oncol. 2017, 24, 3406–3412. [Google Scholar] [CrossRef] [PubMed]
- Zhao, Y.; Hang, B.; Xiong, G.W.; Zhang, X.W. Laparoscopic Radical Hysterectomy in Early Stage Cervical Cancer: A Systematic Review and Meta-Analysis. J. Laparoendosc. Adv. Surg. Tech. 2017, 27, 1132–1144. [Google Scholar] [CrossRef]
- Garabedian, C.; Merlot, B.; Bresson, L.; Tresch, E.; Narducci, F.; Leblanc, E. Minimally invasive surgical management of early-stage cervical cancer: An analysis of the risk factors of surgical complications and of oncologic outcomes. Int. J. Gynecol. Cancer 2015, 25, 714–721. [Google Scholar] [CrossRef]
- Ramirez, P.T.; Frumovitz, M.; Pareja, R.; Lopez, A.; Vieira, M.; Ribeiro, M.; Buda, A.; Yan, X.; Shuzhong, Y.; Chetty, N.; et al. Minimally Invasive versus Abdominal Radical Hysterectomy for Cervical Cancer. N. Engl. J. Med. 2018, 379, 1895–1904. [Google Scholar] [CrossRef]
- Melamed, A.; Margul, D.J.; Chen, L.; Keating, N.L.; del Carmen, M.G.; Yang, J.; Seagle, B.-L.L.; Alexander, A.; Barber, E.L.; Rice, L.W.; et al. Survival after Minimally Invasive Radical Hysterectomy for Early-Stage Cervical Cancer. N. Engl. J. Med. 2018, 379, 1905–1914. [Google Scholar] [CrossRef] [PubMed]
- Nitecki, R.; Ramirez, P.T.; Frumovitz, M.; Krause, K.J.; Tergas, A.I.; Wright, J.D.; Alejandro Rauh-Hain, J.; Melamed, A. Survival After Minimally Invasive vs Open Radical Hysterectomy for Early-Stage Cervical Cancer: A Systematic Review and Meta-analysis. JAMA Oncol. 2020, 6, 1019. [Google Scholar] [CrossRef]
- Cusimano, M.C.; Baxter, N.N.; Gien, L.T.; Moineddin, R.; Liu, N.; Dossa, F.; Willows, K.; Ferguson, S.E. Impact of surgical approach on oncologic outcomes in women undergoing radical hysterectomy for cervical cancer. Am. J. Obstet. Gynecol. 2019, 221, 619.e1–619.e24. [Google Scholar] [CrossRef]
- Obermair, A.; Asher, R.; Pareja, R.; Frumovitz, M.; Lopez, A.; Moretti-Marques, R.; Rendon, G.; Ribeiro, R.; Tsunoda, A.; Behan, V.; et al. Incidence of adverse events in minimally invasive vs open radical hysterectomy in early cervical cancer: Results of a randomized controlled trial. Am. J. Obstet. Gynecol. 2020, 222, 249.e1–249.e10. [Google Scholar] [CrossRef] [PubMed]
- Pomel, C.; Lecuru, F.; Leblanc, E.; Mathevet, P.; Colombo, P.E.; Guyon, F.; Collinet, P.; Nadeau, C.; Huchon, C.; Frederic, S.; et al. Cancer du col utérin opérable de stade précoce par chirurgie minimale invasive. Recommandations des chirurgiens du groupe GINECO. Bull. Du Cancer 2019, 106, 939–940. [Google Scholar] [CrossRef] [PubMed]
- Mo, L. Effects of a Simulated CO2 Pneumoperitoneum Environment on the Proliferation, Apoptosis, and Metastasis of Cervical Cancer Cells In Vitro. Med. Sci. Monit. 2014, 20, 2497–2503. [Google Scholar] [CrossRef] [PubMed]
- Volz, J.; Köster, S.; Spacek, Z.; Paweletz, N. The influence of pneumoperitoneum used in laparoscopic surgery on an intraabdominal tumor growth. Cancer 1999, 86, 770–774. [Google Scholar] [CrossRef]
- Malzoni, M.; Tinelli, R.; Cosentino, F.; Perone, C.; Iuzzolino, D.; Rasile, M.; Tinelli, A. Laparoscopic radical hysterectomy with lymphadenectomy in patients with early cervical cancer: Our instruments and technique. Surg. Oncol. 2009, 18, 289–297. [Google Scholar] [CrossRef]
- Chiva, L.; Zanagnolo, V.; Querleu, D.; Martin-Calvo, N.; Arévalo-Serrano, J.; Căpîlna, M.E.; Fagotti, A.; Kucukmetin, A.; Mom, C.; Chakalova, G.; et al. SUCCOR study: An international European cohort observational study comparing minimally invasive surgery versus open abdominal radical hysterectomy in patients with stage IB1 cervical cancer. Int. J. Gynecol. Cancer 2020, 30, 1269–1277. [Google Scholar] [CrossRef]
- Leblanc, E. Comment je fais… la préparation vaginale à une hystérectomie/colpectomie radicale laparoscopique ou procédé de «Schautheim». Gynécologie Obs. Fertilité. 2007, 35, 263–264. [Google Scholar] [CrossRef]
- Köhler, C.; Hertel, H.; Herrmann, J.; Marnitz, S.; Mallmann, P.; Favero, G.; Plaikner, A.; Martus, P.; Gajda, M.; Schneider, A.; et al. Laparoscopic radical hysterectomy with transvaginal closure of vaginal cuff—A multicenter analysis. Int. J. Gynecol. Cancer 2019, 29, 845–850. [Google Scholar] [CrossRef]
- Martin-Hirsch, P.; Wood, N.; Whitham, N.L.; Macdonald, R.; Kirwan, J.; Anagnostopoulos, A.; Hutson, R.; Theophilou, G.; Otify, M.; Smith, M.; et al. Survival of women with early-stage cervical cancer in the UK treated with minimal access and open surgery. BJOG Int. J. Obstet. Gy. 2019, 126, 956–959. [Google Scholar] [CrossRef]
- Alfonzo, E.; Wallin, E.; Ekdahl, L.; Staf, C.; Rådestad, A.F.; Reynisson, P.; Stålberg, K.; Falconer, H.; Persson, J.; Dahm-Kähler, P. No survival difference between robotic and open radical hysterectomy for women with early-stage cervical cancer: Results from a nationwide population-based cohort study. Eur. J. Cancer 2019, 116, 169–177. [Google Scholar] [CrossRef]
- Kanno, K.; Andou, M.; Yanai, S.; Toeda, M.; Nimura, R.; Ichikawa, F.; Teishikata, Y.; Shirane, T.; Sakate, S.; Kihira, T.; et al. Long-term oncological outcomes of minimally invasive radical hysterectomy for early-stage cervical cancer: A retrospective, single-institutional study in the wake of the LACC trial. J. Obstet. Gynaecol. Res. 2019, 45, 2425–2434. [Google Scholar] [CrossRef]
- Wang, S.Y.; Yin, L.; Guan, X.M.; Xiao, B.B.; Zhang, Y.; Delgado, A. Single Port Transumbilical Laparoscopic Surgery versus Conventional Laparoscopic Surgery for Benign Adnexal Masses: A Retrospective Study of Feasibility and Safety. Chin. Med. J. 2016, 129, 1305–1310. [Google Scholar] [CrossRef] [PubMed]
- Querleu, D.; Morrow, C.P. Classification of radical hysterectomy. Lancet Oncol. 2008, 9, 297–303. [Google Scholar] [CrossRef] [PubMed]
- Matsuo, K.; Shimada, M.; Yamaguchi, S.; Matoda, M.; Nakanishi, T.; Kikkawa, F.; Ohmichi, M.; Okamoto, A.; Sugiyama, T.; Mikami, M. Association of Radical Hysterectomy Surgical Volume and Survival for Early-Stage Cervical Cancer. Obstet. Gynecol. 2019, 133, 1086–1098. [Google Scholar] [CrossRef] [PubMed]
- Casarin, J.; Bogani, G.; Papadia, A.; Ditto, A.; Pinelli, C.; Garzon, S.; Donadello, N.; Laganà, A.S.; Cromi, A.; Mueller, M.; et al. Preoperative Conization and Risk of Recurrence in Patients Undergoing Laparoscopic Radical Hysterectomy for Early Stage Cervical Cancer: A Multicenter Study. J. Minim. Invasive Gynecol. 2021, 28, 117–123. [Google Scholar] [CrossRef] [PubMed]
- Casarin, J.; Buda, A.; Bogani, G.; Fanfani, F.; Papadia, A.; Ceccaroni, M.; Malzoni, M.; Pellegrino, A.; Ferrari, F.; Greggi, S.; et al. Predictors of recurrence following laparoscopic radical hysterectomy for early-stage cervical cancer: A multi-institutional study. Gynecol. Oncol. 2020, 159, 164–170. [Google Scholar] [CrossRef]
- Gennari, P.; Gerken, M.; Mészáros, J.; Klinkhammer-Schalke, M.; Ortmann, O.; Eggemann, H.; Ignatov, A. Minimal-invasive or open approach for surgery of early cervical cancer: The treatment center matters. Arch Gynecol. Obs. 2021, 304, 503–510. [Google Scholar] [CrossRef]
- Landoni, F.; Maneo, A.; Colombo, A.; Placa, F.; Milani, R.; Perego, P.; Favini, G.; Ferri, L.; Mangioni, C. Randomised study of radical surgery versus radiotherapy for stage Ib-IIa cervical cancer. Lancet 1997, 350, 535–540. [Google Scholar] [CrossRef]
- Bataille, B.; Escande, A.; Le Tinier, F.; Parent, A.; Bogart, E.; Collinet, P.; Bresson, L.; Narducci, F.; Leblanc, E.; Lemaire, A.-S.; et al. Outcomes of Pre-Operative Brachytherapy Followed by Hysterectomy for Early Cervical Cancer. Int. J. Gynecol. Cancer 2019, 30, 181–186. [Google Scholar] [CrossRef]
- Escande, A.; Gouy, S.; Mazeron, R.; Bentivegna, E.; Bacorro, W.; Maroun, P.; Schernberg, A.; Oberlander, A.-S.; Dumas, I.; Genestie, C.; et al. Outcome of early stage cervical cancer patients treated according to a radiosurgical approach: Clinical results and prognostic factors. Gynecol. Oncol. 2017, 144, 541–546. [Google Scholar] [CrossRef]
- Uzan, C.; Merlot, B.; Gouy, S.; Belghiti, J.; Haie-Meder, C.; Nickers, P.; Fabrice, N.; Morice, P.; Leblanc, E. Laparoscopic radical hysterectomy after preoperative brachytherapy for stage IB1 cervical cancer: Feasibility, results, and surgical implications in a large bicentric study of 162 consecutive cases. Ann. Surg Oncol. 2013, 20, 872–880. [Google Scholar] [CrossRef] [PubMed]
- Lamblin, G.; Rouffiac, M.; Mathevet, P.; Martin, E.; Peignaux-Casasnovas, K.; Chabert, P.; Lebail-Carval, K.; Chene, G. Surgery alone or in association with preoperative uterovaginal brachytherapy for stage IB1 cervical cancer: Toxicities profiles. Gynecol. Obstet. Fertil. 2015, 43, 485–490. [Google Scholar] [CrossRef] [PubMed]
- Atlan, D.; Touboul, E.; Deniaud-Alexandre, E.; Lefranc, J.P.; Antoine, J.M.; Jannet, D.; Lhuillier, P.; Uzan, M.; Huart, J.; Genestie, C.; et al. Operable Stages IB and II cervical carcinomas: A retrospective study comparing preoperative uterovaginal brachytherapy and postoperative radiotherapy. Int. J. Radiat. Oncol. Biol. Phys. 2002, 54, 780–793. [Google Scholar] [CrossRef] [PubMed]
- Gauci, P.A.; Kee, D.L.C.; Thamphya, B.; Schiappa, R.; Delotte, J.; Chand-Fouche, M.E.; Hannoun-Levi, J.M. Preoperative high-dose-rate brachytherapy for high-risk early-stage cervical cancer: Long-term clinical outcome analysis. Brachytherapy 2022, 21, 273–282. [Google Scholar] [CrossRef]
- Varela Cagetti, L.; Gonzague-Casabianca, L.; Zemmour, C.; Lambaudie, E.; Houvenaeghel, G.; Provansal, M.; Sabatier, R.; Sabiani, L.; Blache, G.; Jauffret, C.; et al. The impact of modern preoperative high-dose-rate brachytherapy in early-stage cervical cancer. Gynecol. Oncol. 2021, 161, 166–172. [Google Scholar] [CrossRef]
- Hannoun-Levi, J.M.; Chand-Fouche, M.E.; Gautier, M.; Dejean, C.; Marcy, M.; Fouche, Y. Interstitial preoperative high-dose-rate brachytherapy for early stage cervical cancer: Dose–volume histogram parameters, pathologic response and early clinical outcome. Brachytherapy 2013, 12, 148–155. [Google Scholar] [CrossRef]
- Chacon, E.; Manzour, N.; Zanagnolo, V.; Querleu, D.; Núñez-Córdoba, J.M.; Martin-Calvo, N.; Căpîlna, M.E.; Fagotti, A.; Kucukmetin, A.; Mom, C.; et al. SUCCOR cone study: Conization before radical hysterectomy. Int. J. Gynecol. Cancer 2022, 32, 117–124. [Google Scholar] [CrossRef]
- Bizzarri, N.; Anchora, L.P.; Kucukmetin, A.; Ratnavelu, N.; Korompelis, P.; Carbone, V.; Fedele, C.; Bruno, M.; Vizzielli, G.; Gallotta, V.; et al. Protective Role of Conization Before Radical Hysterectomy in Early-Stage Cervical Cancer: A Propensity-Score Matching Study. Ann Surg Oncol. 2021, 28, 3585–3594. [Google Scholar] [CrossRef]
- Landoni, F.; Maneo, A.; Zapardiel, I.; Zanagnolo, V.; Mangioni, C. Class I versus class III radical hysterectomy in stage IB1-IIA cervical cancer. A prospective randomized study. Eur. J. Surg. Oncol. (EJSO) 2012, 38, 203–209. [Google Scholar] [CrossRef]
- Corrado, G.; Vizza, E.; Legge, F.; Anchora, L.P.; Sperduti, I.; Fagotti, A.; Mancini, E.; Gallotta, V.; Zampa, A.; Chiofalo, B.; et al. Comparison of Different Surgical Approaches for Stage IB1 Cervical Cancer Patients: A Multi-institution Study and a Review of the Literature. Int. J. Gynecol. Cancer 2018, 28, 1020–1028. [Google Scholar] [CrossRef]
- Piedimonte, S.; Pond, G.R.; Plante, M.; Nelson, G.; Kwon, J.; Altman, A.; Feigenberg, T.; Elit, L.; Lau, S.; Sabourin, J.; et al. Comparison of outcomes between abdominal, minimally invasive and combined vaginal-laparoscopic hysterectomy in patients with stage IAI/IA2 cervical cancer: 4C (Canadian Cervical Cancer Collaborative) study. Gynecol. Oncol. 2022, 166, 230–235. [Google Scholar] [CrossRef] [PubMed]




| Age | ||
| Median-(Range) | 46 | (25; 80) |
| Mean-SD | 47.6 | 11.6 |
| BMI in kg/m² | ||
| Median-(Range) | 23.9 | (15.2; 46.4) |
| Mean-SD | 25.6 | 5.8 |
| History of conization | ||
| No | 154 | 64.4% |
| Yes | 85 | 35.6% |
| Presence of LVSI | ||
| No | 190 | 79.5% |
| Yes | 49 | 20.5% |
| Histological type | ||
| Epidermoid | 172 | 72.0% |
| Adenocarcinoma | 60 | 25.1% |
| Epidermoid and Adenocarcinoma | 5 | 2.1% |
| Glassy cell | 2 | 0.8% |
| Grade of Differentiation | ||
| Low | 78 | 32.6% |
| Medium | 63 | 26.4% |
| Well Differentiated | 98 | 41% |
| Largest diameter in cm | ||
| ≤2 | 80 | 33.5% |
| 2 to 3 | 101 | 42.3% |
| >3 | 58 | 24.3% |
| FIGO Stage | ||
| IA2 | 10 | 4.2% |
| IB1 | 70 | 29.3% |
| IB2 | 141 | 59.0% |
| IIA1 | 18 | 7.5% |
| Association between Prognostic Factors and Recurrence Free Survival | Number of Events/N | Univariate Analysis | Multivariate Analysis | ||||
|---|---|---|---|---|---|---|---|
| HR | 95% CI | p-Value | HR | 95% CI | p-Value | ||
| Age at treatment | 0.04 | NS (2) | |||||
| HR/10 years | - | 1.37 | 1.02–1.84 | NS | |||
| Body Mass Index | 0.28 | - | |||||
| HR/1 kg/m² | - | 1.03 | 0.97–1.09 | - | |||
| History of Conization | 0.009 | 0.01 | |||||
| No | 26/154 | 1 | 1 | ||||
| Yes | 3/85 | 0.20 | 0.06–0.67 | 0.21 | 0.06–0.70 | ||
| Lymphovascular space invasion | 0.99 | - | |||||
| No | 23/190 | 1 | - | ||||
| Yes | 6/49 | 1.00 | 0.41–2.46 | - | |||
| Histological type | 0.49 | - | |||||
| Squamous cell | 23/172 | 1 | - | ||||
| Adenocarcinoma | 5/60 | 0.56 | 0.21–1.47 | - | |||
| Other (1) | 1/7 | 1.03 | 0.14–7.63 | - | |||
| Grade of Differentiation | 0.29 | - | |||||
| Low | 10/78 | 1 | - | ||||
| Middle | 10/63 | 1.33 | 0.55–3.18 | - | |||
| High | 8/97 | 0.63 | 0.25–1.59 | - | |||
| Stage | 0.03 | NS (2) | |||||
| I | 24/221 | 1 | NS | ||||
| II | 5/18 | 2.87 | 1.09–7.52 | NS | |||
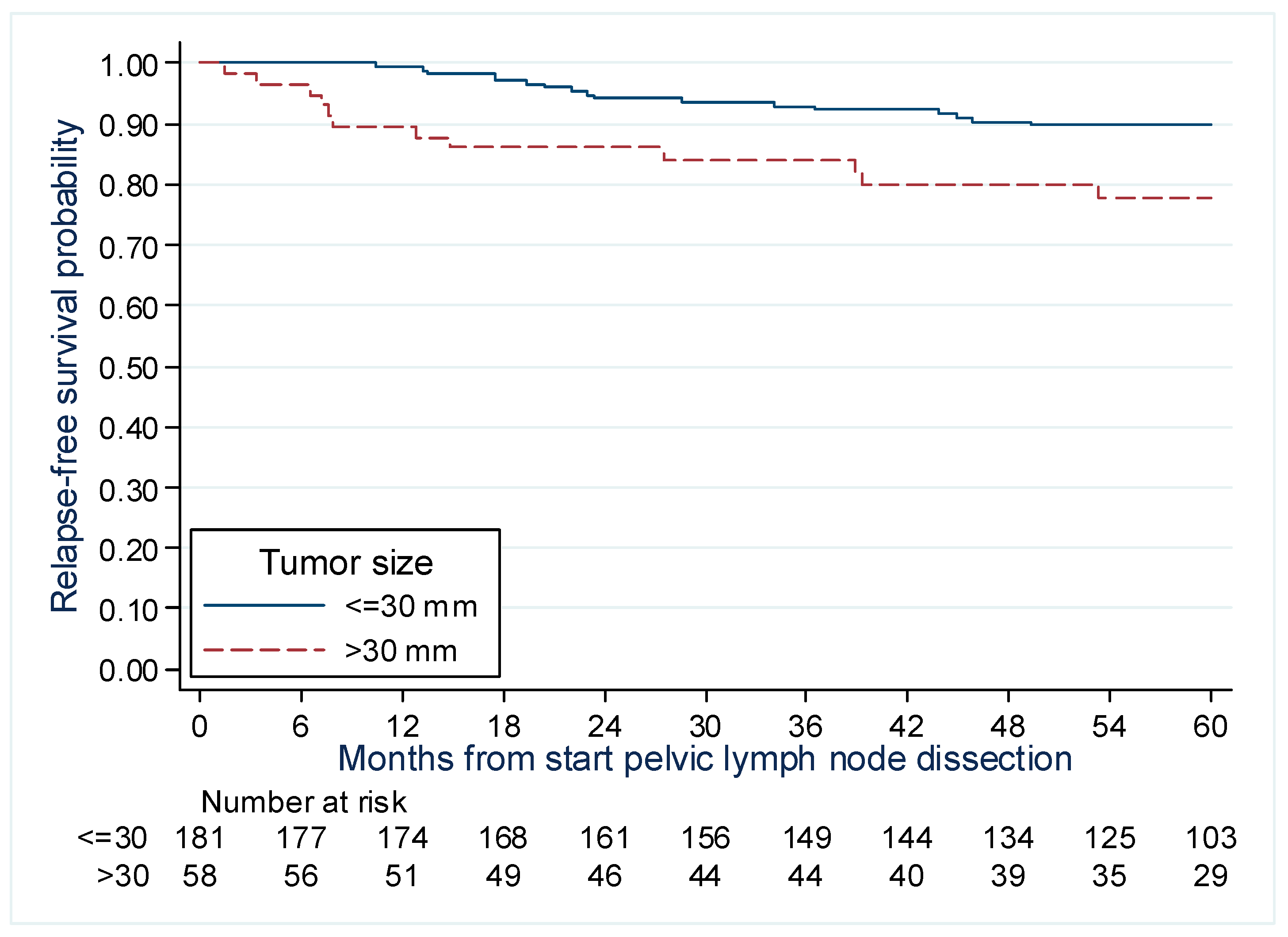
| Size of the Lesion | 0.021 | - (3) | |||||
| HR/10 mm | - | 1.65 | 1.08–2.52 | - | |||
| Size of the Lesion | 0.06 | - | |||||
| ≤20 mm | 7/80 | 1 | - | ||||
| (20–30] mm | 10/101 | 1.13 | 0.43–2.96 | - | |||
| >30 mm | 12/58 | 1.60 | 1.03–6.62 | - | |||
| Size of the Lesion | 0.018 | 0.031 | |||||
| ≤30 mm | 17/181 | 1 | 1 | ||||
| >30 mm | 12/58 | 2.43 | 1.16–5.09 | 2.26 | 1.08–4.73 | ||
| Brachytherapy | 0.066 | NS (2) | |||||
| No | 9/114 | 1 | NS | ||||
| Yes | 20/125 | 2.09 | 0.95–4.59 | NS | |||
| Size of the lesion and Brachytherapy | 0.16 (4) | ||||||
| size ≤ 20 mm without brachytherapy | 7/75 | - | - | ||||
| size ≤ 20 mm with brachytherapy | 0/5 | - | - | ||||
| size >20 mm without brachytherapy | 2/39 | - | - | ||||
| size > 20 mm with brachytherapy | 20/120 | - | - | ||||
| Age | Size (mm) | Figo Stage | Histology | Grade | Preoperatory Conization | Invasion of the Paracervix or Vagina | Lympho-Vascular Space Invasion | Status of Pelvic Nodes | Preoperatory Brachytherapy | Adjuvant Treatment | Time to Recurrence (Months) | Site of Recurrence | Treatment of Disease Recurrence | Death |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| 50 | 20 | IB2 | AC | G1 | No | No | Non | Negative | No | No | 35.9 | Aortic node | CT + RT | No |
| 55 | 20 | IB2 | ASC | G1 | No | No | Yes | Negative | No | Yes | 22.6 | Centro-pelvis | CT | Yes |
| 74 | 19 | IIA1 | SCC | G2 | No | Yes | Yes | Negative | No | No | 50.2 | Vagina | RTCT | Yes |
| 62 | 20 | IIA1 | SCC | G1 | No | Yes | Non | Negative | No | Yes | 23.7 | Vagina and lung | BT + CT | Yes |
| 59 | 14 | IIA1 | SCC | G2 | No | Yes | Non | Negative | No | No | 17.8 | Vagina | S + RT | No |
| 41 | 12 | IIA1 | SCC | G1 | No | Yes | Non | Negative | No | No | 23.3 | Vagina | S + RT | No |
| 57 | 28 | IB2 | SCC | G1 | No | No | No | Negative | Yes | No | 18 | Lung | S + CT + RT | Yes |
| 52 | 24 | IB2 | SCC | G3 | No | No | No | Negative | Yes | No | 15 | Para-rectal and lung | S + CT | Yes |
| 64 | 30 | IB2 | SCC | G2 | No | No | No | Negative | Yes | No | 11.5 | Iliac node | RTCT | Yes |
| 67 | 30 | IB2 | SCC | G1 | No | No | No | Negative | Yes | No | 105 | Lung | S | No |
| 44 | 25 | IB2 | SCC | G1 | No | No | Yes | Negative | No | Yes | 115.9 | Node, lung, and bone | CT + RT | Yes |
| 63 | 30 | IB2 | SCC | G2 | No | No | No | Negative | Yes | No | 34.6 | Iliac node and lung | S + CT | Yes |
| 38 | 21 | IB2 | AC | G3 | No | No | Yes | Negative | No | Yes | 96.6 | Peritoneal carcinosis | S + CT + RT | No |
| 44 | 23 | IB2 | SCC | G3 | No | No | Yes | Negative | Yes | No | 10.5 | Sacrum node | RTCT | Yes |
| 38 | 25 | IB2 | SCC | G3 | Yes | No | Yes | Negative | Yes | No | 33 | Iliac node | RTCT | No |
| 55 | 25 | IB2 | AC | G2 | Yes | Yes | No | Negative | Yes | No | 42.1 | Node, lung, and bone | CT | Yes |
| 46 | 24 | IB2 | AC | G3 | No | No | Yes | Negative | Yes | No | 7.7 | Vagina and node, lung | S + RTCT | No |
| 63 | 30 | IB2 | SCC | G3 | No | No | No | Negative | Yes | No | 26.8 | Lung | CT | Yes |
| 53 | 28 | IB2 | SCC | G3 | No | No | No | Negative | Yes | No | 10.3 | Lung, bone, and cerebral | CT | Yes |
| 64 | 39 | IB2 | AC | G1 | No | No | No | Negative | Yes | No | 121.2 | Iliac node and rectum | S | No |
| 39 | 38 | IB2 | SCC | G2 | No | No | No | Negative | Yes | No | 25.8 | Iliac node and sigmoid | S + RTCT | Yes |
| 53 | 35 | IB2 | SCC | G2 | No | No | No | Negative | Yes | No | 4.7 | Iliac node | S + RTCT | Yes |
| 54 | 32 | IB2 | SCC | G1 | No | No | No | Negative | Yes | No | 93.6 | Lung and bone | CT | Yes |
| 52 | 33 | IB2 | SCC | G3 | No | No | No | Negative | Yes | No | 4.2 | Peritoneal carcinosis | CT | Yes |
| 48 | 39 | IB2 | SCC | G2 | No | No | No | Negative | Yes | No | 10.7 | Iliac node and bone and lung | CT | Yes |
| 48 | 39 | IB2 | AC | G1 | No | No | No | Negative | Yes | No | 36.4 | Lung | CT | Yes |
| 40 | 39 | IIA1 | SCC | G2 | No | Yes | Yes | Negative | No | No | 2.5 | Left side pelvis | S + CT | No |
| 62 | 37 | IB2 | SCC | G2 | No | No | No | Negative | Yes | No | 6.2 | Iliac node and lung | CT + RT | Yes |
| 38 | 34 | IB2 | SCC | G1 | No | No | No | Negative | No | No | 3.4 | Iliac node and p carcinosis | CT + RT | Yes |
| 54 | 35 | IB2 | SCC | G3 | No | No | No | Negative | Yes | No | 39.4 | Node, bone, and lung | CT | Yes |
| 38 | 34 | IB2 | SCC | G3 | No | No | No | Negative | Yes | No | 4.1 | Peritoneal, bone, and lung | CT | Yes |
| 70 | 36 | IB2 | SCC | G3 | No | No | No | Negative | Yes | No | 52 | Lung, hepatic, and bone | CT | Yes |
| 49 | 36 | IB2 | SCC | G3 | Yes | No | No | Negative | Yes | No | 12.4 | Iliac node and lung | CT | Yes |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Serouart, B.; Cordoba, A.; Martinez-Gomez, C.; Bogart, E.; Le Deley, M.C.; Leblanc, É.; Hudry, D.; Escande, A.; Le Tinier, F.; Pasquesoone, C.; et al. Results of a 20 Year Retrospective Analysis of Early-Stage Cervical Cancer: Should 3 cm Be Considered the New Ariadne’s Thread in Early Cervical Cancer Treatment? Cancers 2023, 15, 1570. https://doi.org/10.3390/cancers15051570
Serouart B, Cordoba A, Martinez-Gomez C, Bogart E, Le Deley MC, Leblanc É, Hudry D, Escande A, Le Tinier F, Pasquesoone C, et al. Results of a 20 Year Retrospective Analysis of Early-Stage Cervical Cancer: Should 3 cm Be Considered the New Ariadne’s Thread in Early Cervical Cancer Treatment? Cancers. 2023; 15(5):1570. https://doi.org/10.3390/cancers15051570
Chicago/Turabian StyleSerouart, Benjamin, Abel Cordoba, Carlos Martinez-Gomez, Emilie Bogart, Marie Cecile Le Deley, Éric Leblanc, Delphine Hudry, Alexandre Escande, Florence Le Tinier, Camille Pasquesoone, and et al. 2023. "Results of a 20 Year Retrospective Analysis of Early-Stage Cervical Cancer: Should 3 cm Be Considered the New Ariadne’s Thread in Early Cervical Cancer Treatment?" Cancers 15, no. 5: 1570. https://doi.org/10.3390/cancers15051570
APA StyleSerouart, B., Cordoba, A., Martinez-Gomez, C., Bogart, E., Le Deley, M. C., Leblanc, É., Hudry, D., Escande, A., Le Tinier, F., Pasquesoone, C., Taieb, S., El Hajj, H., & Narducci, F. (2023). Results of a 20 Year Retrospective Analysis of Early-Stage Cervical Cancer: Should 3 cm Be Considered the New Ariadne’s Thread in Early Cervical Cancer Treatment? Cancers, 15(5), 1570. https://doi.org/10.3390/cancers15051570






