Establishment, Maintenance, and Performance of the Cooperative Osteosarcoma Study Group (COSS)
Abstract
Simple Summary
Abstract
1. Origins and Early COSS History
“If we operate they die, if we don’t operate they die. This meeting should be concluded with prayers.”(Sir Stanford Cade, 1955)
2. Structure of the COSS Group
2.1. Multi-Disciplinarity
2.2. Multi-Centricity
2.3. Multi-Nationality
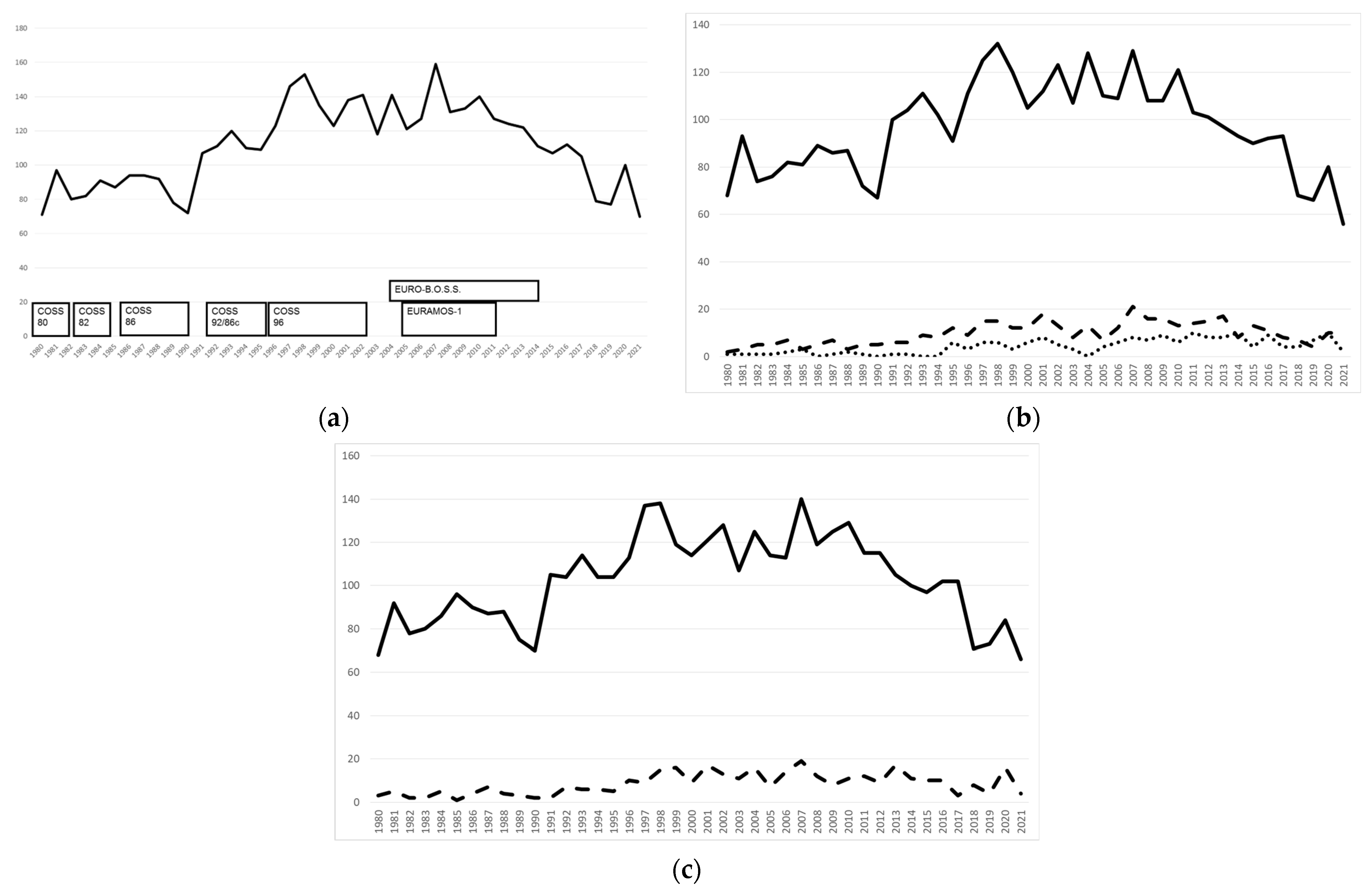
2.4. Patient Recruitment
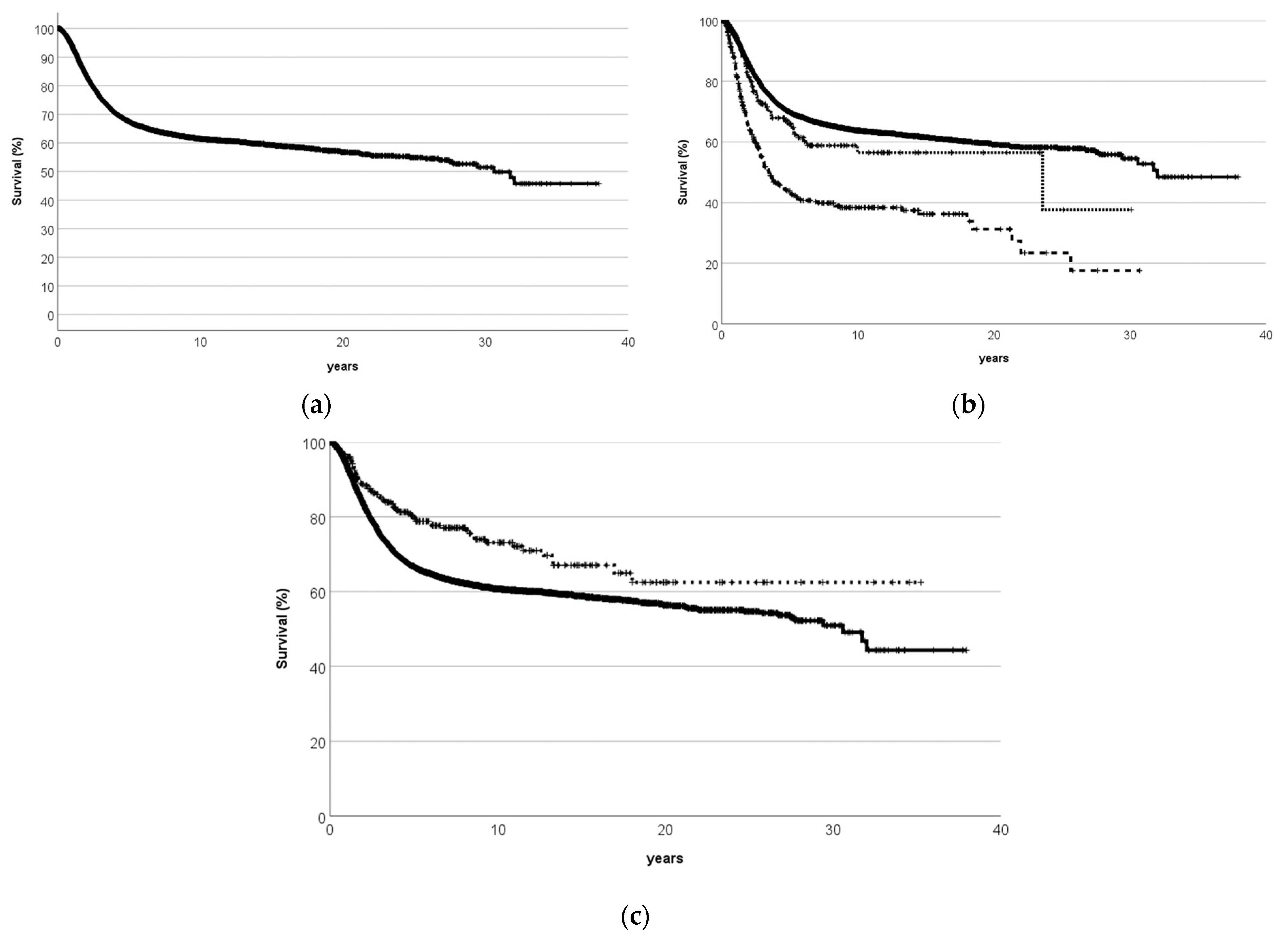
2.5. Long-Term Follow-Up and Cancer Survivorship
3. Aims of the COSS Group
4. Prospective COSS Trials
4.1. Groupwise Trials
4.2. Intergroup Trials
5. COSS Registry
5.1. Rationale
5.2. Recruitment
5.3. Published Groupwise Analyses
6. Adolescent and Young Adult Oncology
7. Intergroup Collaboration Using Anonymized Data
8. Bringing European Researchers Together
9. Past, Current, and Future Challenges to Collaboration
9.1. Maintenance of Multi-Disciplinary Collaboration
9.2. Ever Increasing Regulatory Demands
9.3. Financial Sustainability
10. Current and Future COSS Projects
11. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Jaffe, N.; Frei, E., 3rd; Traggis, D.; Bishop, Y. Adjuvant methotrexate and citrovorum-factor treatment of osteogenic sarcoma. N. Engl. J. Med. 1974, 291, 994–997. [Google Scholar] [CrossRef] [PubMed]
- Cortes, E.P.; Holland, J.F.; Wang, J.J.; Sinks, L.F.; Blom, J.; Senn, H.; Bank, A.; Glidewell, O. Amputation and adriamycin in primary osteosarcoma. N. Engl. J. Med. 1974, 291, 998–1000. [Google Scholar] [CrossRef] [PubMed]
- Rosen, G.; Suwansirikul, S.; Kwon, C.; Tan, C.; Wu, S.J.; Beattie, E.J., Jr.; Murphy, M.L. High-dose methotrexate with citrovorum factor rescue and adriamycin in childhood osteogenic sarcoma. Cancer 1974, 33, 1151–1163. [Google Scholar] [CrossRef]
- Bacci, G.; Pagani, P.A. Supplementary antiblastic polychemotherapy in localized malignant bone tumors. I. Ewing’s sarcoma and osteosarcoma. Chir. Organi Mov. 1975, 62, 491–506. [Google Scholar]
- Rosen, G.; Marcove, R.C.; Caparros, B.; Nirenberg, A.; Kosloff, C.; Huvos, A.G. Primary osteogenic sarcoma: The rationale for preoperative chemotherapy and delayed surgery. Cancer 1979, 43, 2163–2177. [Google Scholar] [CrossRef]
- Winkler, K.; Gaedicke, G.; Grosch-Wörner, I.; Marsmann, G.; Delling, G.; Landbeck, G. Chemotherapy of osteosarcoma (author’s transl). Dtsch. Med. Wochenschr. 1977, 102, 1831–1835. [Google Scholar] [CrossRef]
- Kotz, R.; Leber, H.; Ramach, W.; Arbes, H.; Wolf, A. Clinical observations on the use of high-dose methotrexate treatment in osteogenic sarcoma (author’s transl). Wien. Klin. Wochenschr. 1977, 89, 474–479. [Google Scholar]
- Winkler, K.; Beron, G.; Schellong, G.; Stollmann, B.; Prindull, G.; Lasson, U.; Brandeis, W.; Henze, G.; Ritter, J.; Russe, W.; et al. Cooperative osteosarcoma study COSS-77: Results after 4 years. Klin. Padiatr. 1982, 194, 251–256. [Google Scholar] [CrossRef]
- Purfürst, C.; Beron, G.; Torggler, S.; Kotz, R.; Salzer-Kuntschik, M.; Winkler, K. Results of the COSS-77 and COSS-80 studies on adjuvant chemotherapy in osteosarcoma of the extremities. Klin. Padiatr. 1985, 197, 233–238. [Google Scholar] [CrossRef]
- Enneking, W.F.; Spanier, S.S.; Goodman, M.A. A system for the surgical staging of musculoskeletal sarcoma. Clin. Orthop. Relat. Res. 1980, 153, 106–120. [Google Scholar] [CrossRef]
- Smeland, S.; Bielack, S.S.; Whelan, J.; Bernstein, M.; Hogendoorn, P.; Krailo, M.D.; Gorlick, R.; Janeway, K.A.; Ingleby, F.C.; Anninga, J.; et al. Survival and prognosis with osteosarcoma: Outcomes in more than 2000 patients in the EURAMOS-1 (European and American Osteosarcoma Study) cohort. Eur. J. Cancer. 2019, 109, 36–50. [Google Scholar] [CrossRef] [PubMed]
- Blaauwbroek, R.; Tuinier, W.; Meyboom-de Jong, B.; Kamps, W.A.; Postma, A. Shared care by paediatric oncologists and family doctors for long-term follow-up of adult childhood cancer survivors: A pilot study. Lancet Oncol. 2008, 9, 232–238. [Google Scholar] [CrossRef]
- McLoone, J.K.; Chen, W.; Wakefield, C.E.; Johnston, K.; Bell, R.; Thornton-Benko, E.; Cohn, R.J.; Signorelli, C. Childhood cancer survivorship care: A qualitative study of healthcare providers’ professional preferences. Front. Oncol. 2022, 12, 945911. [Google Scholar] [CrossRef] [PubMed]
- Blaauwbroek, R.; Zwart, N.; Bouma, M.; Meyboom-de Jong, B.; Kamps, W.A.; Postma, A. The willingness of general practitioners to be involved in the follow-up of adult survivors of childhood cancer. J. Cancer Surviv. 2007, 1, 292–297. [Google Scholar] [CrossRef] [PubMed]
- Taggart, J.; Chin, M.; Liauw, W.; Goldstein, D.; Dolezal, A.; Plahn, J.; Harris, M.F. Challenges and solutions to sharing a cancer follow-up e-care plan between a cancer service and general practice. Public Health Res. Pract. 2021, 31, 31122108. [Google Scholar] [CrossRef] [PubMed]
- Winkler, K.; Beron, G.; Kotz, R.; Salzer-Kuntschik, M.; Beck, J.; Beck, W.; Brandeis, W.; Ebell, W.; Erttmann, R.; Havers, W.; et al. Adjuvant chemotherapy in osteosarcoma—Effects of cisplatinum, BCD, and fibroblast interferon in sequential combination with HD-MTX and adriamycin. Preliminary results of the COSS 80 study. J. Cancer Res. Clin. Oncol. 1983, 106, 1–7. [Google Scholar] [CrossRef]
- Winkler, K.; Beron, G.; Kotz, R.; Salzer-Kuntschik, M.; Beck, J.; Beck, W.; Brandeis, W.; Ebell, W.; Erttmann, R.; Göbel, U. Neoadjuvant chemotherapy for osteogenic sarcoma: Results of a Cooperative German/Austrian study. J. Clin. Oncol. 1984, 2, 617–624. [Google Scholar] [CrossRef]
- Bielack, S.; Beck, J.; Delling, G.; Gerein, V.; Grümayer, R.; Hiddemann, W.; Jobke, A.; Jürgens, H.; Kornhuber, G.; Kotz, R.; et al. Neoadjuvant chemotherapy of osteosarcoma. Results of the cooperative studies COSS-80 and COSS-82 after 7 and 5 years. Klin. Padiatr. 1989, 201, 275–284. [Google Scholar] [CrossRef] [PubMed]
- Winkler, K.; Beron, G.; Delling, G.; Heise, U.; Kabisch, H.; Purfürst, C.; Berger, J.; Ritter, J.; Jürgens, H.; Gerein, V. Neoadjuvant chemotherapy of osteosarcoma: Results of a randomized cooperative trial (COSS-82) with salvage chemotherapy based on histological tumor response. J. Clin. Oncol. 1988, 6, 329–337. [Google Scholar] [CrossRef] [PubMed]
- Bielack, S.S.; Erttmann, R.; Looft, G.; Purfürst, C.; Delling, G.; Winkler, K.; Landbeck, G. Platinum disposition after intraarterial and intravenous infusion of cisplatin for osteosarcoma. Cooperative Osteosarcoma Study Group COSS. Cancer Chemother. Pharmacol. 1989, 24, 376–380. [Google Scholar] [CrossRef]
- Winkler, K.; Bielack, S.; Delling, G.; Salzer-Kuntschik, M.; Purfürst, C.; Heise, U.; Jürgens, H.; Kotz, R.; Gadner, H.; Ritter, J.; et al. Intensified chemotherapy with ifosfamide (IFO) and influence of intraarterial (i.a.) versus intravenous (i.v.) infusion of cisplatinum (DDP). Preliminary results. Chir. Organi Mov. 1990, 75 (Suppl. S1), 54–56. [Google Scholar]
- Winkler, K.; Bielack, S.; Delling, G.; Salzer-Kuntschik, M.; Kotz, R.; Greenshaw, C.; Jürgens, H.; Ritter, J.; Kusnierz-Glaz, C.; Erttmann, R.; et al. Effect of intraarterial versus intravenous cisplatin in addition to systemic doxorubicin, high-dose methotrexate, and ifosfamide on histologic tumor response in osteosarcoma (study COSS-86). Cancer 1990, 66, 1703–1710. [Google Scholar] [CrossRef]
- Bieling, P.; Bielack, S.; Delling, G.; Jürgens, H.; Kotz, R.; Dose, C.; Astheimer, H.; Exner, G.; Gadner, H.; Graf, N.; et al. Neoadjuvant chemotherapy of osteosarcoma. Preliminary results of the cooperative COSS-86 osteosarcoma study. Klin. Padiatr. 1991, 203, 220–230. [Google Scholar] [CrossRef]
- Fuchs, N.; Bielack, S.S.; Epler, D.; Bieling, P.; Delling, G.; Körholz, D.; Graf, N.; Heise, U.; Jürgens, H.; Kotz, R.; et al. Long-term results of the co-operative German-Austrian-Swiss osteosarcoma study group’s protocol COSS-86 of intensive multidrug chemotherapy and surgery for osteosarcoma of the limbs. Ann. Oncol. 1998, 9, 893–899. [Google Scholar] [CrossRef]
- Bielack, S.S.; Bieling, P.; Erttmann, R.; Winkler, K. Intraarterial chemotherapy for osteosarcoma: Does the result really justify the effort? Cancer Treat. Res. 1993, 62, 85–92. [Google Scholar] [CrossRef]
- Bielack, S.S.; Erttmann, R.; Kempf-Bielack, B.; Winkler, K. Impact of scheduling on toxicity and clinical efficacy of doxorubicin: What do we know in the mid-nineties? Eur. J. Cancer 1996, 32, 1652–1660. [Google Scholar] [CrossRef]
- Marina, N.; Bielack, S.; Whelan, J.; Smeland, S.; Krailo, M.; Sydes, M.R.; Butterfass-Bahloul, T.; Calaminus, G.; Bernstein, M. International collaboration is feasible in trials for rare conditions: The EURAMOS experience. Cancer Treat. Res. 2009, 152, 339–353. [Google Scholar] [CrossRef]
- Whelan, J.S.; Bielack, S.S.; Marina, N.; Smeland, S.; Jovic, G.; Hook, J.M.; Krailo, M.; Anninga, J.; Butterfass-Bahloul, T.; Böhling, T.; et al. EURAMOS collaborators. EURAMOS-1, an international randomised study for osteosarcoma: Results from pre-randomisation treatment. Ann. Oncol. 2015, 26, 407–414. [Google Scholar] [CrossRef]
- Marina, N.M.; Smeland, S.; Bielack, S.S.; Bernstein, M.; Jovic, G.; Krailo, M.D.; Hook, J.M.; Arndt, C.; van den Berg, H.; Brennan, B.; et al. Comparison of MAPIE versus MAP in patients with a poor response to preoperative chemotherapy for newly diagnosed high-grade osteosarcoma (EURAMOS-1): An open-label, international, randomised controlled trial. Lancet Oncol. 2016, 17, 1396–1408. [Google Scholar] [CrossRef]
- Bielack, S.S.; Smeland, S.; Whelan, J.S.; Marina, N.; Jovic, G.; Hook, J.M.; Krailo, M.D.; Gebhardt, M.; Pápai, Z.; Meyer, J.; et al. EURAMOS-1 investigators. Methotrexate, Doxorubicin, and Cisplatin (MAP) Plus Maintenance Pegylated Interferon Alfa-2b Versus MAP Alone in Patients with Resectable High-Grade Osteosarcoma and Good Histologic Response to Preoperative MAP: First Results of the EURAMOS-1 Good Response Randomized Controlled Trial. J. Clin. Oncol. 2015, 33, 2279–2287. [Google Scholar] [CrossRef]
- Calaminus, G.; Jenney, M.; Hjorth, L.; Baust, K.; Bernstein, M.; Bielack, S.; Vos, P.; Hogendoorn, P.C.W.; Jovic, G.; Krailo, M.; et al. Quality of Life of Patients With Osteosarcoma in the European American Osteosarcoma Study-1 (EURAMOS-1): Development and Implementation of a Questionnaire Substudy. affected patients. JMIR Res. Protoc. 2019, 8, e14406. [Google Scholar] [CrossRef]
- Budde, A.; Baust, K.; Weinhold, L.; Bernstein, M.; Bielack, S.; Dhooge, C.; Hjorth, L.; Janeway, K.A.; Jenney, M.; Krailo, M.D.; et al. Linking EORTC QLQ-C-30 and PedsQL/PEDQOL physical functioning scores in patients with osteosarcoma. Eur. J. Cancer 2022, 170, 209–235. [Google Scholar] [CrossRef]
- Ferrari, S.; Bielack, S.S.; Smeland, S.; Longhi, A.; Egerer, G.; Hall, K.S.; Donati, D.; Kevric, M.; Brosjö, O.; Comandone, A.; et al. EURO-B.O.S.S.: A European study on chemotherapy in bone-sarcoma patients aged over 40: Outcome in primary high-grade osteosarcoma. Tumori J. 2018, 104, 30–36. [Google Scholar] [CrossRef]
- Bielack, S.S.; Kempf-Bielack, B.; Delling, G.; Exner, G.U.; Flege, S.; Helmke, K.; Kotz, R.; Salzer-Kuntschik, M.; Werner, M.; Winkelmann, W.; et al. Prognostic factors in high-grade osteosarcoma of the extremities or trunk: An analysis of 1702 patients treated on neoadjuvant cooperative osteosarcoma study group protocols. J. Clin. Oncol. 2002, 20, 776–790. [Google Scholar] [CrossRef]
- Rehan, N.; Bieling, P.; Winkler, P.; Helmke, K.; Maas, R.; Baldini, N.; Heise, U.; Fuchs, N.; Winkler, K. The prognostic significance of tumor volume in osteosarcoma with neoadjuvant chemotherapy. Klin. Padiatr. 1993, 205, 200–209. [Google Scholar] [CrossRef]
- Bieling, P.; Rehan, N.; Winkler, P.; Helmke, K.; Maas, R.; Fuchs, N.; Bielack, S.; Heise, U.; Jurgens, H.; Treuner, J.; et al. Tumor size and prognosis in aggressively treated osteosarcoma. J. Clin. Oncol. 1996, 14, 848–858. [Google Scholar] [CrossRef]
- Graf, N.; Winkler, K.; Betlemovic, M.; Fuchs, N.; Bode, U. Methotrexate pharmacokinetics and prognosis in osteosarcoma. J. Clin. Oncol. 1994, 12, 1443–1451. [Google Scholar] [CrossRef]
- Eselgrim, M.; Grunert, H.; Kühne, T.; Zoubek, A.; Kevric, M.; Bürger, H.; Jürgens, H.; Mayer-Steinacker, R.; Gosheger, G.; Bielack, S.S. Dose intensity of chemotherapy for osteosarcoma and outcome in the Cooperative Osteosarcoma Study Group (COSS) trials. Pediatr. Blood Cancer 2006, 47, 42–50. [Google Scholar] [CrossRef]
- Sauerbrey, A.; Bielack, S.; Kempf-Bielack, B.; Zoubek, A.; Paulussen, M.; Zintl, F. High-dose chemotherapy (HDC) and autologous hematopoietic stem cell transplantation (ASCT) as salvage therapy for relapsed osteosarcoma. Bone Marrow Transplant. 2001, 27, 933–937. [Google Scholar] [CrossRef]
- Kelley, L.M.; Schlegel, M.; Hecker-Nolting, S.; Kevric, M.; Haller, B.; Rössig, C.; Reichardt, P.; Kager, L.; Kühne, T.; Gosheger, G.; et al. Pathological Fracture and Prognosis of High-Grade Osteosarcoma of the Extremities: An Analysis of 2847 Consecutive Cooperative Osteosarcoma Study Group (COSS) Patients. J. Clin. Oncol. 2020, 38, 823–833. [Google Scholar] [CrossRef]
- Winkler, K.; Torggler, S.; Beron, G.; Bode, U.; Gerein, V.; Jürgens, H.; Kusnierz-Glaz, C.; Kotz, R.; Salzer-Kuntschik, M.; Schmoll, H.J.; et al. Results of treatment in primary disseminated osteosarcoma. Analysis of the follow-up of patients in the cooperative osteosarcoma studies COSS-80 and COSS-82. Onkologie 1989, 12, 92–96. [Google Scholar] [CrossRef]
- Kager, L.; Zoubek, A.; Pötschger, U.; Kastner, U.; Flege, S.; Kempf-Bielack, B.; Branscheid, D.; Kotz, R.; Salzer-Kuntschik, M.; Winkelmann, W.; et al. Cooperative German-Austrian-Swiss Osteosarcoma Study Group. Primary metastatic osteosarcoma: Presentation and outcome of patients treated on neoadjuvant Cooperative Osteosarcoma Study Group protocols. J. Clin. Oncol. 2003, 21, 2011–2018. [Google Scholar] [CrossRef] [PubMed]
- Kager, L.; Zoubek, A.; Kastner, U.; Kempf-Bielack, B.; Potratz, J.; Kotz, R.; Exner, G.U.; Franzius, C.; Lang, S.; Maas, R.; et al. Cooperative Osteosarcoma Study Group. Skip metastases in osteosarcoma: Experience of the Cooperative Osteosarcoma Study Group. J. Clin. Oncol. 2006, 24, 1535–1541. [Google Scholar] [CrossRef]
- Schuster, A.J.; Kager, L.; Reichardt, P.; Baumhoer, D.; Csóka, M.; Hecker-Nolting, S.; Lang, S.; Lorenzen, S.; Mayer-Steinacker, R.; von Kalle, T.; et al. High-Grade Osteosarcoma of the Foot: Presentation, Treatment, Prognostic Factors, and Outcome of 23 Cooperative Osteosarcoma Study Group COSS Patients. Sarcoma 2018, 2018, 1632978. [Google Scholar] [CrossRef]
- Daecke, W.; Bielack, S.; Martini, A.K.; Ewerbeck, V.; Jürgens, H.; Kotz, R.; Winkelmann, W.; Kabisch, H.; Kevric, M.; Bernd, L. Osteosarcoma of the hand and forearm: Experience of the Cooperative Osteosarcoma Study Group. Ann. Surg. Oncol. 2005, 12, 322–331. [Google Scholar] [CrossRef]
- Bielack, S.S.; Wulff, B.; Delling, G.; Göbel, U.; Kotz, R.; Ritter, J.; Winkler, K. Osteosarcoma of the trunk treated by multimodal therapy: Experience of the Cooperative Osteosarcoma study group (COSS). Med. Pediatr. Oncol. 1995, 24, 6–12. [Google Scholar] [CrossRef]
- Ozaki, T.; Flege, S.; Liljenqvist, U.; Hillmann, A.; Delling, G.; Salzer-Kuntschik, M.; Jürgens, H.; Kotz, R.; Winkelmann, W.; Bielack, S.S. Osteosarcoma of the spine: Experience of the Cooperative Osteosarcoma Study Group. Cancer 2002, 94, 1069–1077. [Google Scholar] [CrossRef] [PubMed]
- Ozaki, T.; Flege, S.; Kevric, M.; Lindner, N.; Maas, R.; Delling, G.; Schwarz, R.; von Hochstetter, A.R.; Salzer-Kuntschik, M.; Berdel, W.E.; et al. Osteosarcoma of the pelvis: Experience of the Cooperative Osteosarcoma Study Group. J. Clin. Oncol. 2003, 21, 334–341. [Google Scholar] [CrossRef]
- Zils, K.; Bielack, S.; Wilhelm, M.; Werner, M.; Schwarz, R.; Windhager, R.; Hofmann-Wackersreuther, G.; Andus, T.; Kager, L.; Kuehne, T.; et al. Osteosarcoma of the mobile spine. Ann. Oncol. 2013, 24, 2190–2195. [Google Scholar] [CrossRef]
- Jasnau, S.; Meyer, U.; Potratz, J.; Jundt, G.; Kevric, M.; Joos, U.K.; Jürgens, H.; Bielack, S.S. Cooperative Osteosarcoma Study Group COSS. Craniofacial osteosarcoma Experience of the cooperative German-Austrian-Swiss osteosarcoma study group. Oral Oncol. 2008, 44, 286–294. [Google Scholar] [CrossRef]
- Bielack, S.S.; Kempf-Bielack, B.; Heise, U.; Schwenzer, D.; Winkler, K. Combined modality treatment for osteosarcoma occurring as a second malignant disease. Cooperative German-Austrian-Swiss Osteosarcoma Study Group. J. Clin. Oncol. 1999, 17, 1164. [Google Scholar] [CrossRef]
- Bielack, S.S.; Rerin, J.S.; Dickerhoff, R.; Dilloo, D.; Kremens, B.; von Stackelberg, A.; Vormoor, J.; Jürgens, H. Cooperative German-Austrian-Swiss Osteosarcoma Study Group (COSS). Osteosarcoma after allogeneic bone marrow transplantation. A report of four cases from the Cooperative Osteosarcoma Study Group (COSS). Bone Marrow Transplant. 2003, 31, 353–359. [Google Scholar] [CrossRef]
- Zils, K.; Bielack, S. Osteosarcoma after bone marrow transplantation: Still a challenge. J. Pediatr. Hematol. Oncol. 2015, 37, 74. [Google Scholar] [CrossRef]
- Bielack, S.S.; Hecker-Nolting, S.; Kevric, M.; Juergens, H. More on osteosarcoma and phylloides tumor. J. Pediatr. Hematol. Oncol. 2015, 37, 158–159. [Google Scholar] [CrossRef] [PubMed]
- Zils, K.; Klingebiel, T.; Behnisch, W.; Mueller, H.L.; Schlegel, P.G.; Fruehwald, M.; Suttorp, M.; Simon, T.; Werner, M.; Bielack, S. Osteosarcoma in patients with Rothmund-Thomson syndrome. Pediatr. Hematol. Oncol. 2015, 32, 32–40. [Google Scholar] [CrossRef] [PubMed]
- Goldstein-Jackson, S.Y.; Gosheger, G.; Delling, G.; Berdel, W.E.; Exner, G.U.; Jundt, G.; Machatschek, J.N.; Zoubek, A.; Jürgens, H.; Bielack, S.S. Cooperative Osteosarcoma Study Group COSS. Extraskeletal osteosarcoma has a favourable prognosis when treated like conventional osteosarcoma. J. Cancer Res. Clin. Oncol. 2005, 131, 520–526. [Google Scholar] [CrossRef]
- Kager, L.; Zoubek, A.; Dominkus, M.; Lang, S.; Bodmer, N.; Jundt, G.; Klingebiel, T.; Jürgens, H.; Gadner, H.; Bielack, S. COSS Study Group. Osteosarcoma in very young children: Experience of the Cooperative Osteosarcoma Study Group. Cancer 2010, 116, 5316–5324. [Google Scholar] [CrossRef]
- Hecker-Nolting, S.; Baumhoer, D.; Blattmann, C.; Kager, L.; Kühne, T.; Kevric, M.; Lang, S.; Mettmann, V.; Sorg, B.; Werner, M.; et al. Osteosarcoma pre-diagnosed as another tumor: A report from the Cooperative Osteosarcoma Study Group (COSS). J. Cancer Res. Clin. Oncol. 2022. [Google Scholar] [CrossRef]
- Kempf-Bielack, B.; Bielack, S.S.; Jürgens, H.; Branscheid, D.; Berdel, W.E.; Exner, G.U.; Göbel, U.; Helmke, K.; Jundt, G.; Kabisch, H.; et al. Osteosarcoma relapse after combined modality therapy: An analysis of unselected patients in the Cooperative Osteosarcoma Study Group (COSS). J. Clin. Oncol. 2005, 23, 559–568. [Google Scholar] [CrossRef]
- Bielack, S.S.; Kempf-Bielack, B.; Branscheid, D.; Carrle, D.; Friedel, G.; Helmke, K.; Kevric, M.; Jundt, G.; Kühne, T.; Maas, R.; et al. Second and subsequent recurrences of osteosarcoma: Presentation, treatment, and outcomes of 249 consecutive cooperative osteosarcoma study group patients. J. Clin. Oncol. 2009, 27, 557–565. [Google Scholar] [CrossRef] [PubMed]
- Franke, M.; Hardes, J.; Helmke, K.; Jundt, G.; Jürgens, H.; Kempf-Bielack, B.; Kevric, M.; Tunn, P.U.; Werner, M.; Bielack, S. Solitary skeletal osteosarcoma recurrence. Findings from the Cooperative Osteosarcoma Study Group. Pediatr. Blood Cancer 2011, 56, 771–776. [Google Scholar] [CrossRef]
- Andreou, D.; Bielack, S.S.; Carrle, D.; Kevric, M.; Kotz, R.; Winkelmann, W.; Jundt, G.; Werner, M.; Fehlberg, S.; Kager, L.; et al. The influence of tumor- and treatment-related factors on the development of local recurrence in osteosarcoma after adequate surgery. An analysis of 1355 patients treated on neoadjuvant Cooperative Osteosarcoma Study Group protocols. Ann. Oncol. 2011, 22, 1228–1235. [Google Scholar] [CrossRef]
- Gotta, J.; Bielack, S.; Hecker-Nolting, S.; Sorg, B.; Kevric, M.; Salzmann-Manrique, E.; Klingebiel, T. When Your Ankle Becomes a Knee—Long-Term Functional Outcome and Quality of Life with a Rotationplasty after Resection of Malignant Limb Tumors. Klin. Padiatr. 2022, 234, 154–162. [Google Scholar] [CrossRef] [PubMed]
- Bielack, S.S.; Schroeders, A.; Fuchs, N.; Bacci, G.; Bauer, H.C.; Mapeli, S.; Tomeno, B.; Winkler, K. Malignant fibrous histiocytoma of bone: A retrospective EMSOS study of 125 cases. European Musculo-Skeletal Oncology Society. Acta Orthop. Scand. 1999, 70, 353–360. [Google Scholar] [CrossRef]
- Hompland, I.; Ferrari, S.; Bielack, S.; Palmerini, E.; Hall, K.S.; Picci, P.; Hecker-Nolting, S.; Donati, D.M.; Blattmann, C.; Bjerkehagen, B.; et al. Outcome in dedifferentiated chondrosarcoma for patients treated with multimodal therapy: Results from the EUROpean Bone over 40 Sarcoma Study. Eur. J. Cancer 2021, 151, 150–158. [Google Scholar] [CrossRef]
- Jürgens, H.; Beron, G.; Winkler, K. Toxicity associated with combination chemotherapy for osteosarcoma: A report of the cooperative osteosarcoma study (COSS 80). J. Cancer Res. Clin. Oncol. 1983, 106, 14–18. [Google Scholar] [CrossRef]
- Langer, T.; Stöhr, W.; Bielack, S.; Paulussen, M.; Treuner, J.; Beck, J.D. German Late Effects Working Group in the German Society of Pediatric Oncology and Hematology. Late effects surveillance system for sarcoma patients. Pediatr. Blood Cancer 2004, 42, 373–379. [Google Scholar] [CrossRef] [PubMed]
- Paulides, M.; Kremers, A.; Stöhr, W.; Bielack, S.; Jürgens, H.; Treuner, J.; Beck, J.D.; Langer, T. German Late Effects Working Group in the Society of Pediatric Oncology and Haematology (GPOH). Prospective longitudinal evaluation of doxorubicin-induced cardiomyopathy in sarcoma patients: A report of the late effects surveillance system (LESS). Pediatr. Blood Cancer 2006, 46, 489–495. [Google Scholar] [CrossRef] [PubMed]
- Stöhr, W.; Langer, T.; Kremers, A.; Bielack, S.; Lamprecht-Dinnesen, A.; Frey, E.; Beck, J.D. German Late Effects Working Group in the German Society of Pediatric Oncology and Hematology. Cisplatin-induced ototoxicity in osteosarcoma patients: A report from the late effects surveillance system. Cancer Investig. 2005, 23, 201–207. [Google Scholar] [CrossRef]
- Nitz, A.; Kontopantelis, E.; Bielack, S.; Koscielniak, E.; Klingebiel, T.; Langer, T.; Paulides, M. Prospective evaluation of cisplatin- and carboplatin-mediated ototoxicity in paediatric and adult soft tissue and osteosarcoma patients. Oncol. Lett. 2013, 5, 311–315. [Google Scholar] [CrossRef]
- Stöhr, W.; Paulides, M.; Bielack, S.; Jürgens, H.; Koscielniak, E.; Rossi, R.; Langer, T.; Beck, J.D. Nephrotoxicity of cisplatin and carboplatin in sarcoma patients: A report from the late effects surveillance system. Pediatr. Blood Cancer 2007, 48, 140–147. [Google Scholar] [CrossRef]
- Stöhr, W.; Paulides, M.; Bielack, S.; Jürgens, H.; Treuner, J.; Rossi, R.; Langer, T.; Beck, J.D. Ifosfamide-induced nephrotoxicity in 593 sarcoma patients: A report from the Late Effects Surveillance System. Pediatr. Blood Cancer 2007, 48, 447–452. [Google Scholar] [CrossRef] [PubMed]
- Paulides, M.; Dörr, H.G.; Stöhr, W.; Bielack, S.; Koscielniak, E.; Klingebiel, T.; Jürgens, H.; Bölling, T.; Willich, N.; Sauer, R.; et al. Late Effects Surveillance System. Thyroid function in paediatric and young adult patients after sarcoma therapy: A report from the Late Effects Surveillance System. Clin. Endocrinol. 2007, 66, 727–731. [Google Scholar] [CrossRef]
- Paulides, M.; Stöhr, W.; Laws, H.J.; Graf, N.; Lakomek, M.; Berthold, F.; Schmitt, K.; Niggli, F.; Jürgens, H.; Bielack, S.; et al. Immunity against tetanus and diphtheria after childhood sarcoma treatment. Klin. Padiatr. 2010, 222, 196. [Google Scholar] [CrossRef]
- Paulides, M.; Stöhr, W.; Laws, H.J.; Graf, N.; Lakomek, M.; Berthold, F.; Schmitt, K.; Niggli, F.; Jürgens, H.; Bielack, S.; et al. Antibody levels against tetanus and diphtheria after polychemotherapy for childhood sarcoma: A report from the Late Effects Surveillance System. Vaccine 2011, 29, 1565–1568. [Google Scholar] [CrossRef]
- Langer, T.; Clemens, E.; Broer, L.; Maier, L.; Uitterlinden, A.G.; de Vries, A.C.H.; van Grotel, M.; Pluijm, S.F.M.; Binder, H.; Mayer, B.; et al. PanCareLIFE consortium. Association of candidate pharmacogenetic markers with platinum-induced ototoxicity: PanCareLIFE dataset. Data Brief 2020, 32, 106227. [Google Scholar] [CrossRef] [PubMed]
- Langer, T.; Clemens, E.; Broer, L.; Maier, L.; Uitterlinden, A.G.; de Vries, A.C.H.; van Grotel, M.; Pluijm, S.F.M.; Binder, H.; Mayer, B.; et al. PanCareLIFE consortium. Usefulness of current candidate genetic markers to identify childhood cancer patients at risk for platinum-induced ototoxicity: Results of the European PanCareLIFE cohort study. Eur. J. Cancer 2020, 138, 212–224. [Google Scholar] [CrossRef]
- Escherich, G.; Bielack, S.; Maier, S.; Braungart, R.; Brümmendorf, T.H.; Freund, M.; Grosse, R.; Hoferer, A.; Kampschulte, R.; Koch, B.; et al. Building a National Framework for Adolescent and Young Adult Hematology and Oncology and Transition from Pediatric to Adult Care: Report of the Inaugural Meeting of the “AjET” Working Group of the German Society for Pediatric Oncology and Hematology. J. Adolesc. Young Adult Oncol. 2017, 6, 194–199. [Google Scholar] [CrossRef]
- Saloustros, E.; Stark, D.P.; Michailidou, K.; Mountzios, G.; Brugieres, L.; Peccatori, F.A.; Jezdic, S.; Essiaf, S.; Douillard, J.Y.; Bielack, S. The care of adolescents and young adults with cancer: Results of the ESMO/SIOPE survey. ESMO Open 2017, 2, e000252. [Google Scholar] [CrossRef]
- Wilhelm, M.; Dirksen, U.; Bielack, S.S.; Whelan, J.S.; Lewis, I.J.; Jürgens, H.; Ferrari, S.; Sundby Hall, K.; Cleton-Jansen, A.M.; Stark, D. ENCCA (European Network for Cancer research in Children and Adolescents). ENCCA WP17-WP7 consensus paper on teenagers and young adults (TYA) with bone sarcomas. Ann Oncol. 2014, 25, 1500–1505. [Google Scholar] [CrossRef]
- Stark, D.; Bielack, S.; Brugieres, L.; Dirksen, U.; Duarte, X.; Dunn, S.; Erdelyi, D.J.; Grew, T.; Hjorth, L.; Jazbec, J.; et al. Teenagers and young adults with cancer in Europe: From national programmes to a European integrated coordinated project. Eur. J. Cancer Care 2016, 25, 419–427. [Google Scholar] [CrossRef]
- Ferrari, A.; Stark, D.; Peccatori, F.A.; Fern, L.; Laurence, V.; Gaspar, N.; Bozovic-Spasojevic, I.; Smith, O.; De Munter, J.; Derwich, K.; et al. Adolescents and young adults (AYA) with cancer: A position paper from the AYA Working Group of the European Society for Medical Oncology (ESMO) and the European Society for Paediatric Oncology (SIOPE). ESMO Open 2021, 6, 100096. [Google Scholar] [CrossRef]
- Collins, M.; Wilhelm, M.; Conyers, R.; Herschtal, A.; Whelan, J.; Bielack, S.; Kager, L.; Kühne, T.; Sydes, M.; Gelderblom, H.; et al. Benefits and adverse events in younger versus older patients receiving neoadjuvant chemotherapy for osteosarcoma: Findings from a meta-analysis. J. Clin. Oncol. 2013, 31, 2303–2312. [Google Scholar] [CrossRef]
- Grimer, R.J.; Cannon, S.R.; Taminiau, A.M.; Bielack, S.; Kempf-Bielack, B.; Windhager, R.; Dominkus, M.; Saeter, G.; Bauer, H.; Meller, I.; et al. Osteosarcoma over the age of forty. Eur. J. Cancer 2003, 39, 157–163. [Google Scholar] [CrossRef]
- Grimer, R.J.; Bielack, S.; Flege, S.; Cannon, S.R.; Foleras, G.; Andreeff, I.; Sokolov, T.; Taminiau, A.; Dominkus, M.; San-Julian, M.; et al. European Musculo Skeletal Oncology Society. Periosteal osteosarcoma—A European review of outcome. Eur. J. Cancer 2005, 41, 2806–2811. [Google Scholar] [CrossRef]
- Longhi, A.; Bielack, S.S.; Grimer, R.; Whelan, J.; Windhager, R.; Leithner, A.; Gronchi, A.; Biau, D.; Jutte, P.; Krieg, A.H.; et al. Extraskeletal osteosarcoma: A European Musculoskeletal Oncology Society study on 266 patients. Eur. J. Cancer 2017, 74, 9–16. [Google Scholar] [CrossRef]
- Hauben, E.I.; Bielack, S.; Grimer, R.; Jundt, G.; Reichardt, P.; Sydes, M.; Taminiau, A.H.; Hogendoorn, P.C. Clinico-histologic parameters of osteosarcoma patients with late relapse. Eur. J. Cancer 2006, 42, 460–466. [Google Scholar] [CrossRef]
- Widemann, B.C.; Balis, F.M.; Kempf-Bielack, B.; Bielack, S.; Pratt, C.B.; Ferrari, S.; Bacci, G.; Craft, A.W.; Adamson, P.C. High-dose methotrexate-induced nephrotoxicity in patients with osteosarcoma. Cancer 2004, 100, 2222–2232. [Google Scholar] [CrossRef]
- Frezza, A.M.; Cesari, M.; Baumhoer, D.; Biau, D.; Bielack, S.; Campanacci, D.A.; Casanova, J.; Esler, C.; Ferrari, S.; Funovics, P.T.; et al. Mesenchymal chondrosarcoma: Prognostic factors and outcome in 113 patients. A European Musculoskeletal Oncology Society study. Eur. J. Cancer 2015, 51, 374–381. [Google Scholar] [CrossRef]
- Kube, S.J.; Blattmann, C.; Bielack, S.S.; Kager, L.; Kaatsch, P.; Kühne, T.; Sorg, B.; Kevric, M.; Jabar, S.; Hallmen, E.; et al. Secondary malignant neoplasms after bone and soft tissue sarcomas in children, adolescents, and young adults. Cancer 2022, 128, 1787–1800. [Google Scholar] [CrossRef]
- Brecht, I.B.; Graf, N.; Schweinitz, D.V.; Frühwald, M.C.; Bielack, S.S.; Schneider, D.T. Networking for children and adolescents with very rare tumors: Foundation of the GPOH Pediatric Rare Tumor Group. Klin. Padiatr. 2009, 221, 181–185. [Google Scholar] [CrossRef] [PubMed]
- Brecht, I.B.; Bremensdorfer, C.; Schneider, D.T.; Frühwald, M.C.; Offenmüller, S.; Mertens, R.; Vorwerk, P.; Koscielniak, E.; Bielack, S.S.; Benesch, M.; et al. Rare malignant pediatric tumors registered in the German Childhood Cancer Registry 2001–2010. Pediatr. Blood Cancer 2014, 61, 1202–1209. [Google Scholar] [CrossRef] [PubMed]
- Thomas, D.M.; Wilhelm, M.; Cleton-Jansen, A.M.; Dirksen, U.; Entz-Werlé, N.; Gelderblom, H.; Hassan, B.; Jürgens, H.; Koster, J.; Kovar, H.; et al. Workshop Report on the European Bone Sarcoma Networking Meeting: Integration of Clinical Trials with Tumor Biology. J. Adolesc. Young Adult Oncol. 2011, 1, 118–123. [Google Scholar] [CrossRef]
- Anninga, J.; Cleton-Jansen, A.M.; Hassan, B.; Amary, M.A.; Baumhoer, D.; Blay, J.Y.; Brugieres, L.; Ferrari, S.; Jürgens, J.; Kempf-Bielack, B.; et al. Workshop report on the 2nd Joint ENCCA/EuroSARC European bone sarcoma network meeting: Integration of clinical trials with tumour biology. Clin. Sarcoma Res. 2014, 4, 4. [Google Scholar] [CrossRef]
- Kager, L.; Whelan, J.; Dirksen, U.; Hassan, B.; Anninga, J.; Bennister, L.; Bovée, J.V.M.G.; Brennan, B.; Broto, J.M.; Brugières, L.; et al. The ENCCA-WP7/EuroSarc/EEC/PROVABES/EURAMOS 3rd European Bone Sarcoma Networking Meeting/Joint Workshop of EU Bone Sarcoma Translational Research Networks: Vienna, Austria, September 24–25, 2015. Workshop Report. Clin. Sarcoma Res. 2016, 6, 3. [Google Scholar] [CrossRef] [PubMed]
- Arbeitsgemeinschaft der wissenschaftlich-Medizinischen Fachgesellschaften. Osteosarkome—Deutsche Leitlinie. AWMF-Register-Nr. 025/005 Osteosarkome Version 06/2021. AWMF-online, 2021. Available online: https://register.awmf.org/de/leitlinien/detail/025-005 (accessed on 25 February 2023).
- Strauss, S.J.; Frezza, A.M.; Abecassis, N.; Bajpai, J.; Bauer, S.; Biagini, R.; Bielack, S.; Blay, J.Y.; Bolle, S.; Bonvalot, S.; et al. Bone sarcomas: ESMO–EURACAN–GENTURIS–ERN PaedCan Clinical Practice Guideline for diagnosis, treatment and follow-up. Ann. Oncol. 2021, 32, 1520–1536. [Google Scholar] [CrossRef]
- Andritsch, E.; Beishon, M.; Bielack, S.; Bonvalot, S.; Casali, P.; Crul, M.; Delgado Bolton, R.; Donati, D.M.; Douis, H.; Haas, R.; et al. ECCO Essential Requirements for Quality Cancer Care: Soft Tissue Sarcoma in Adults and Bone Sarcoma. A critical review. Crit. Rev. Oncol. Hematol. 2017, 110, 94–105. [Google Scholar] [CrossRef] [PubMed]
- Bielack, S.; Jürgens, H.; Jundt, G.; Kevric, M.; Kühne, T.; Reichardt, P.; Zoubek, A.; Werner, M.; Winkelmann, W.; Kotz, R. Osteosarcoma: The COSS experience. Cancer Treat. Res. 2009, 152, 289–308. [Google Scholar] [CrossRef] [PubMed]
- Isakoff, M.S.; Bielack, S.S.; Meltzer, P.; Gorlick, R. Osteosarcoma: Current Treatment and a Collaborative Pathway to Success. J. Clin. Oncol. 2015, 33, 3029–3035. [Google Scholar] [CrossRef] [PubMed]
- Beird, H.C.; Bielack, S.S.; Flanagan, A.M.; Gill, J.; Heymann, D.; Janeway, K.A.; Livingston, J.A.; Roberts, R.D.; Strauss, S.J.; Gorlick, R. Osteosarcoma. Nat. Rev. Dis. Prim. 2022, 8, 77. [Google Scholar] [CrossRef]
- Plana, A.; Furner, B.; Palese, M.; Dussault, N.; Birz, S.; Graglia, L.; Kush, M.; Nicholson, J.; Hecker-Nolting, S.; Gaspar, N.; et al. Pediatric Cancer Data Commons: Federating and Democratizing Data for Childhood Cancer Research. JCO Clin. Cancer Inform. 2021, 5, 1034–1043. [Google Scholar] [CrossRef] [PubMed]
| Publication | Ref. | Doi | Topic |
|---|---|---|---|
| Groupwise trials | |||
| Winkler1982 | [8] | 10.1055/s-0028-1105579 | COSS-77: first adjuvant trial results |
| Winkler 1983 | [12] | 10.1007/BF00625042 | COSS-80: preliminary trial results |
| Winkler 1984 | [13] | 10.1200/JCO.1984.2.6.617 | COSS-80: final trial results |
| Purfürst 1985 | [9] | 10.1055/s-2008-1033974 | COSS-77 and -80: updated trial results |
| Winkler 1988 | [15] | 10.1200/JCO.1988.6.2.329 | COSS-82: final trial results |
| Bielack 1989 | [14] | 10.1055/s-2008-1026715 | COSS-80 and COSS-82: updated trial results |
| Winkler 1990 | [17] | - | COSS-86: preliminary trial results |
| Winkler 1990 | [18] | 10.1002/1097-0142(19901015)66:8<1703::aid-cncr2820660809>3.0.co;2-v | COSS-86: final trial results |
| Bieling 1996 | [19] | 10.1055/s-2007-1025433 | COSS-86: preliminary trial results |
| Fuchs 1998 | [20] | 10.1023/a:1008391103132 | COSS-86: updated trial results |
| Bielack 2009 | [30] | 10.1007/978-1-4419-0284-9_15 | COSS: pooled results |
| Intergroup trials | |||
| Marina 2009 | [23] | 10.1007/978-1-4419-0284-9_18 | EURAMOS-1: design |
| Whelan 2015 | [24] | 10.1093/annonc/mdu526 | EURAMOS-1: pre-randomization results |
| Bielack 2015 | [26] | 10.1200/JCO.2014.60.0734 | EURAMOS-1: poor responder results |
| Marina 2016 | [25] | 10.1016/S1470-2045(16)30214-5 | EURAMOS-1: good responder results |
| Ferrari 2018 | [29] | 10.5301/tj.5000696 | EURO-B.O.S.S S.: osteosarcoma results (>40 years) |
| Smeland 2019 | [11] | 10.1016/j.ejca.2018.11.027 | EURAMOS-1: updated trial results |
| Calaminus 2019 | [27] | 10.1016/j.ejca.2022.03.018 | EURAMOS-1: quality of life methodology |
| Hompland 2021 | [61] | 10.1016/j.ejca.2021.04.017 | EURO-B.O.S.S: dedifferentiated chondrosarcomas in patients 41–65 |
| Budde 2022 | [28] | 10.1016/j.ejca.2022.03.018 | EURAMOS-1: quality of life results |
| Patient-related variables and outcomes | |||
| Grimer 2003 | [80] | 10.1016/s0959-8049(02)00478-1 | EMSOS: osteosarcoma over the age of forty |
| Bielack 2003 | [48] | 10.1038/sj.bmt.1703864 | Osteosarcoma after bone marrow transplantation |
| Kager 2010 | [53] | 10.1002/cncr.25287 | Osteosarcoma in very young children |
| Collins 2013 | [79] | 10.1200/JCO.2012.43.8598 | Intergroup meta-analysis: younger vs. older patients with osteosarcoma |
| Bielack 2015 | [50] | 10.1097/MPH.0000000000000197 | Osteosarcoma and phyllodes tumor |
| Zils 2015 | [49] | 10.1097/MPH.0b013e3182a2719c | Osteosarcoma after bone marrow transplantation |
| Zils 2015 | [51] | 10.3109/08880018.2014.987939 | Osteosarcoma in Rothmund–Thomson syndrome |
| Gotta 2022 | [59] | 10.1055/a-1681-1916 | Questionnaire: long-term function and quality of life with a rotationplasty |
| Tumor-related variables and outcomes | |||
| Bieling 1996 | [33] | 10.1200/JCO.1996.14.3.848 | Initial tumor size and prognosis |
| Rehan 1993 | [32] | 10.1055/s-2007-1025228 | Initial tumor size and prognosis |
| Bielack 1995 | [42] | 10.1002/mpo.2950240103 | Osteosarcoma of the trunk |
| Bielack 1999 | [47] | 10.1200/JCO.1999.17.4.1164 | Osteosarcoma as secondary malignancy |
| Bielack 2002 | [31] | 10.1200/JCO.2002.20.3.776 | Prognostic factors in osteosarcoma |
| Ozaki 2002 | [43] | - | Osteosarcoma of the spine |
| Ozaki 2003 | [44] | 10.1200/JCO.2003.01.142 | Osteosarcoma of the pelvis |
| Daecke 2005 | [41] | 10.1245/ASO.2005.06.002 | Osteosarcoma of the hand and forearm |
| Jasnau 2008 | [46] | 10.1016/j.oraloncology.2007.03.001 | Craniofacial osteosarcoma |
| Zils 2013 | [45] | 10.1093/annonc/mdt154 | Osteosarcoma of the mobile spine |
| Schuster 2018 | [40] | 10.1155/2018/1632978 | High-grade osteosarcomas of the foot |
| Kelley 2020 | [36] | 10.1200/JCO.19.00827 | Pathological fracture and prognosis |
| Hecker-Nolting 2022 | [54] | 10.1007/s00432-022-04156-1 | Osteosarcoma pre-diagnosed as another tumor |
| Primary and secondary metastatic disease | |||
| Winkler 1989 | [37] | 10.1159/000216608 | Primary metastatic osteosarcoma |
| Kager 2003 | [38] | 10.1200/JCO.2003.08.132 | Primary metastatic osteosarcoma |
| Kempf-Bielack 2005 | [55] | 10.1200/JCO.2005.04.063 | Osteosarcoma relapse after combined modality therapy |
| Kager 2006 | [39] | 10.1200/JCO.2005.04.2978 | Primary skip metastases |
| Hauben 2006 | [83] | 10.1016/j.ejca.2005.09.032 | Intergroup analysis: late osteosarcoma relapses |
| Bielack 2009 | [56] | 10.1200/JCO.2008.16.2305 | Second and subsequent osteosarcoma recurrences |
| Franke 2011 | [57] | 10.1002/pbc.22864 | Solitary skeletal osteosarcoma recurrences |
| Andreou 2011 | [58] | 10.1093/annonc/mdq589 | Local osteosarcoma recurrences |
| Osteosarcoma variants and non-osteosarcomas | |||
| Bielack 1999 | [60] | 10.3109/17453679908997824 | EMSOS: undifferentiated pleomorphic sarcoma |
| Grimer 2005 | [81] | 10.1016/j.ejca.2005.04.052 | EMSOS: periosteal osteosarcoma |
| Goldstein-Jackson 2005 | [52] | 10.1007/s00432-005-0687-7 | Eextraskeletal osteosarcoma |
| Brecht 2014 | [88] | 10.1002/pbc.24997 | STEP: rare malignant pediatric tumors |
| Frezza 2015 | [85] | 10.1016/j.ejca.2014.11.007 | EMSOS: mesenchymal chondrosarcoma |
| Longhi 2017 | [82] | 10.1016/j.ejca.2016.12.016 | EMSOS: extraskeletal osteosarcoma |
| Anti-tumor drugs | |||
| Bielack 1989 | [16] | 10.1007/BF00257446 | Tumor tissue cisplatin levels after i.a. vs. i.v. infusion |
| Graf 1994 | [99] | 10.1200/JCO.1994.12.7.1443 | Methotrexate pharmacokinetics and prognosis |
| Sauerbrey 2003 | [35] | 10.1038/sj.bmt.1703023 | High-dose chemotherapy in relapsed osteosarcoma |
| Widemann 2004 | [84] | 10.1002/cncr.20255 | Meta-analysis: high-dose methotrexate-induced nephrotoxicity |
| Eselgrim 2006 | [34] | 10.1002/pbc.20608 | Dose intensity of chemotherapy and outcomes |
| Side effects of therapy | |||
| Jürgens 1983 | [62] | 10.1007/BF00625045 | Toxicity of osteosarcoma chemotherapy |
| Langer 2004 | [63] | 10.1002/pbc.10325 | LESS: overview of late toxicity in sarcoma patients |
| Stöhr 2005 | [65] | 10.1081/cnv-200055951 | LESS: cisplatin-induced ototoxicity in osteosarcoma |
| Paulides 2006 | [64] | 10.1002/pbc.20492 | LESS: doxorubicin-induced cardiomyopathy in sarcoma |
| Stöhr 2007 | [67] | 10.1002/pbc.20812 | LESS: nephrotoxicity of cisplatin and carboplatin in sarcoma |
| Stöhr 2007 | [68] | 10.1002/pbc.208 | LESS: ifosfamide nephrotoxicity in sarcoma |
| Paulides 2007 | [69] | 10.1111/j.1365-2265.2007.02813.x | LESS: thyroid function in pediatric and young-adult sarcoma |
| Paulides 2010 | [70] | 10.1055/s-0030-1249609 | LESS: Immunity against tetanus and diphtheria after childhood sarcoma |
| Paulides 2011 | [71] | 10.1016/j.vaccine.2010.12.084 | LESS: antibodies against tetanus and diphtheria after childhood sarcoma |
| Nitz 2013 | [66] | 10.3892/ol.2012.997 | LESS: cisplatin- and carboplatin-mediated ototoxicity in sarcoma |
| Langer 2020 | [72] | 10.1016/j.dib.2020.106227 | PanCareLIFE: association of pharmacogenetic markers and platinum ototoxicity |
| Langer 2020 | [73] | 10.1016/j.ejca.2020.07.019 | PanCareLIFE: genetic markers and platinum-induced ototoxicity |
| Kube 2022 | [86] | 10.1002/cncr.34110 | COSS, CESS, and CWS: secondary malignancies after sarcomas |
| Guidelines/guidance/consensus papers | |||
| Wilhelm 2014 | [76] | 10.1093/annonc/mdu153 | ENCCA WP17-WP7: consensus on teenagers/young adults with bone sarcoma |
| Andritsch 2017 | [94] | 10.1016/j.critrevonc.2016.12.002 | ECCO: essential requirements for quality sarcoma care |
| AWMF 2021 | [92] | - | Expert consensus: German osteosarcoma guidelines |
| Strauss 2021 | [93] | 10.1016/j.annonc.2021.08.1995 | ESMO-EURACAN-GENTURIS-ERN PaedCan: European sarcoma guidelines |
| International reviews | |||
| Isakoff 2015 | [95] | 10.1200/JCO.2014.59.4895 | Osteosarcoma treatment and a collaborative pathway to success |
| Beird 2022 | [96] | 10.1038/s41572-022-00409-y | Osteosarcoma |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Bielack, S.S.; Kager, L.; Kühne, T.; Langer, T.; Reichardt, P.; Blattmann, C.; Kevric, M.; Mettmann, V.; Sorg, B.; Hecker-Nolting, S. Establishment, Maintenance, and Performance of the Cooperative Osteosarcoma Study Group (COSS). Cancers 2023, 15, 1520. https://doi.org/10.3390/cancers15051520
Bielack SS, Kager L, Kühne T, Langer T, Reichardt P, Blattmann C, Kevric M, Mettmann V, Sorg B, Hecker-Nolting S. Establishment, Maintenance, and Performance of the Cooperative Osteosarcoma Study Group (COSS). Cancers. 2023; 15(5):1520. https://doi.org/10.3390/cancers15051520
Chicago/Turabian StyleBielack, Stefan S., Leo Kager, Thomas Kühne, Thorsten Langer, Peter Reichardt, Claudia Blattmann, Matthias Kevric, Vanessa Mettmann, Benjamin Sorg, and Stefanie Hecker-Nolting. 2023. "Establishment, Maintenance, and Performance of the Cooperative Osteosarcoma Study Group (COSS)" Cancers 15, no. 5: 1520. https://doi.org/10.3390/cancers15051520
APA StyleBielack, S. S., Kager, L., Kühne, T., Langer, T., Reichardt, P., Blattmann, C., Kevric, M., Mettmann, V., Sorg, B., & Hecker-Nolting, S. (2023). Establishment, Maintenance, and Performance of the Cooperative Osteosarcoma Study Group (COSS). Cancers, 15(5), 1520. https://doi.org/10.3390/cancers15051520







