Use of Telemedicine to Improve Cognitive Functions and Psychological Well-Being in Patients with Breast Cancer: A Systematic Review of the Current Literature
Abstract
Simple Summary
Abstract
1. Introduction
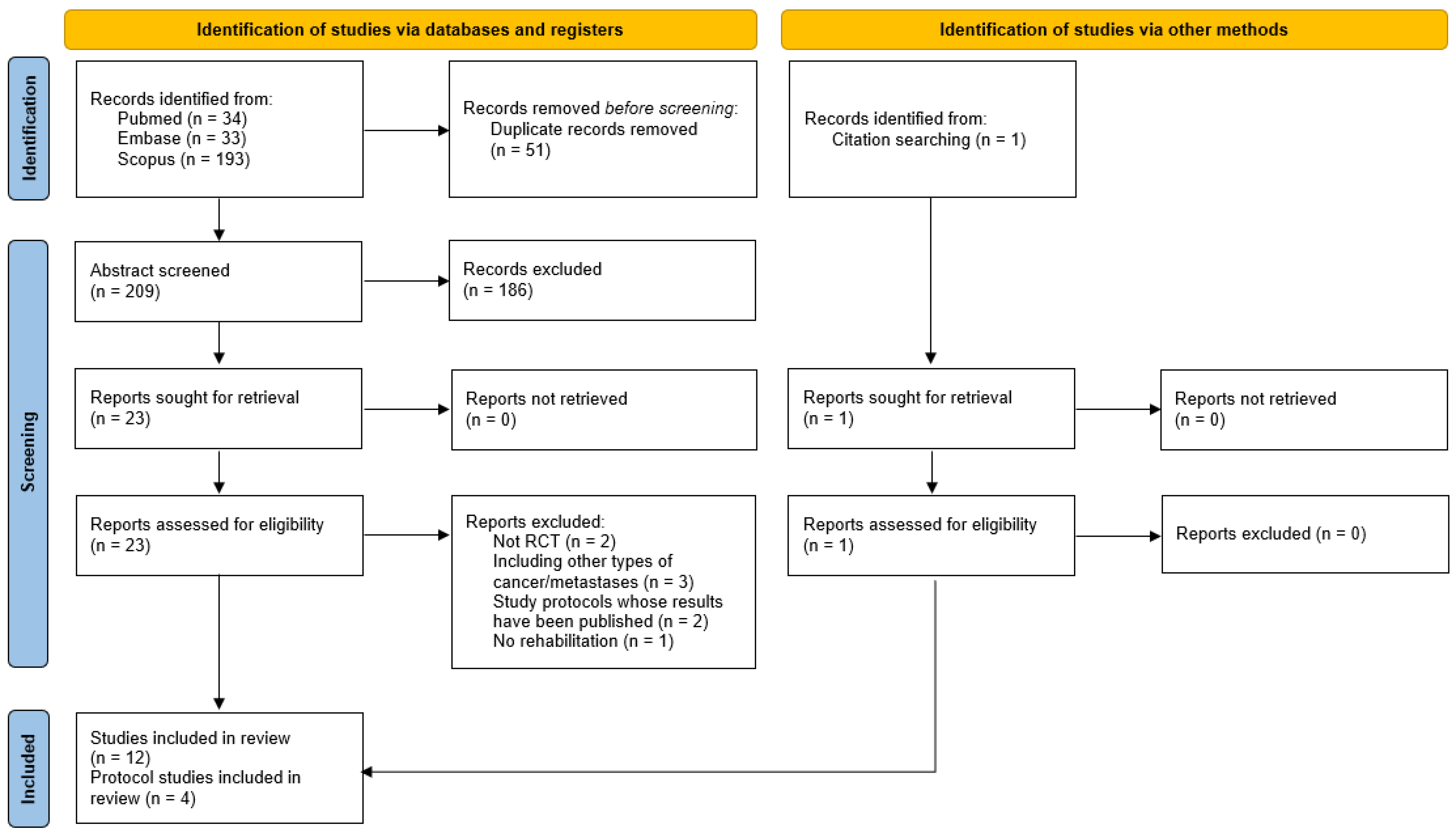
2. Materials and Methods
- To be eligible, studies had to meet the following criteria:
- Being concluded or planned randomized controlled trials (RCTs);
- Assessing the impact of telemedicine in patients treated for early breast cancer or breast cancer survivors after the completion of treatment;
- Reporting a cognitive test or psychological scales as primary or secondary outcomes;
- Using telemedicine for evaluation or rehabilitation;
- Being written in English;
- Being published in an English language journal after 2000.
3. Results
3.1. Included Studies and Protocols-Sample Characteristics
3.1.1. Patients’ Cancer Stages in the Included Studies
3.1.2. Characteristics of the Telemedicine Programs and of the Control Conditions Applied in the Included Studies
3.2. Effects on Cognition
Effects on Cognition: Study Protocols
3.3. Psychological Effects
3.3.1. Quality of Life
3.3.2. Sleep
3.3.3. Fatigue
3.3.4. Anxiety, Depression, and Distress
3.3.5. Pain
3.3.6. Psychological Effects: Study Protocols
4. Discussion
5. Considerations on Mediating Factors and Unmet Needs
6. Conclusions and Future Perspectives
Author Contributions
Funding
Conflicts of Interest
References
- Azamjah, N.; Soltan, Y.-Z.; Zayeri, F. Global Trend of Breast Cancer Mortality Rate: A 25-Year Study. Asian Pac. J. Cancer Prev. 2019, 20, 2015–2020. [Google Scholar] [CrossRef] [PubMed]
- Miller, K.D.; Nogueira, L.; Mariotto, A.B.; Rowland, J.H.; Yabroff, K.R.; Alfano, C.M.; Jemal, A.; Kramer, J.L.; Siegel, R.L. Cancer treatment and survivorship statistics, 2019, CA. Cancer treatment and survivorship statistics, 2019. CA Cancer J. Clin. 2019, 69, 363–385. [Google Scholar] [CrossRef] [PubMed]
- Oh, P.J.; Cho, J.R. Changes in Fatigue, Psychological Distress, and Quality of Life after Chemotherapy in Women with Breast Cancer: A Prospective Study. Cancer Nurs. 2018, 43, E54–E60. [Google Scholar] [CrossRef] [PubMed]
- Wefel, J.S.; Vardy, J.; Ahles, T.; Schagen, S.B. International Cognition and Cancer Task Force recommendations to harmonise studies of cognitive function in patients with cancercancer. Lancet Oncol. 2011, 12, 703–708. [Google Scholar] [CrossRef]
- Zwart, W.; Terra, H.; Linn, S.C.; Schagen, S.B. Cognitive effects of endocrine therapy for breast cancer: Keep calm and carry on? Nat. Rev. Clin. Oncol. 2015, 12, 597–606. [Google Scholar] [CrossRef]
- Buchanan, N.D.; Dasari, S.; Rodriguez, J.L.; Smith, J.L.; Hodgson, M.E.; Weinberg, C.R.; Sandler, D.P. Post-treatment Neurocognition and Psychosocial Care among Breast Cancer Survivors. Am. J. Prev. Med. 2015, 49, S498–S508. [Google Scholar] [CrossRef]
- Berndt, U.; Leplow, B.; Schoenfeld, R.; Lantzsch, T.; Grosse, R.; Thomssen, C. Memory and Spatial Cognition in Breast Cancer Patients Undergoing Adjuvant Endocrine Therapyy. Breast Care 2016, 11, 240–246. [Google Scholar] [CrossRef]
- Collins, B.; Mackenzie, J.; Stewart, A.; Bielajew, C.; Verma, S. Cognitive effects of hormonal therapy in early stage breast cancer patients: A prospective study. Psychooncology 2008, 18, 811–821. [Google Scholar] [CrossRef]
- Chen, X.; Li, J.; Zhang, J.; He, X.; Zhu, C.; Zhang, L.; Hu, X.; Wang, K. Impairment of the executive attention network in premenopausal women with hormone receptor-positive breast cancer treated with tamoxifen. Psychoneuroendocrinology 2017, 75, 116–123. [Google Scholar] [CrossRef]
- Asegaonkar, S.B.; Asegaonkar, B.N.; Takalkar, U.V.; Advani, S.; Thorat, A.P. C-Reactive Protein and Breast Cancer: New Insights from Old Molecule. Int. J. Breast Cancer 2015, 2015, 145647. [Google Scholar] [CrossRef]
- Cheung, Y.T.; Lim, S.R.; Ho, H.K.; Chan, A. Cytokines as mediators of chemotherapy-associated cognitive changes: Current evidence, limitations and directions for future research. PLoS ONE 2013, 8, e81234. [Google Scholar] [CrossRef]
- Gaman, A.M.; Uzoni, A.; Popa, A.-W.; Andrei, A.; Petcu, E.B. The role of oxidative stress in etiopathogenesis of chemotherapy induced cognitive impairment (CICI)-“Chemobrain”. Aging Dis. 2016, 7, 307–317. [Google Scholar] [CrossRef] [PubMed]
- Pullens, J.J.M.; De Vries, J.; Roukema, J.A. Subjective cognitive dysfunction in breast cancer patients: A systematic reviewreview. Psychooncology 2010, 19, 1127–1138. [Google Scholar] [CrossRef] [PubMed]
- Leedham, B.; Ganz, P.A. Psychosocial concerns and quality of life in breast cancer survivors. Cancer Investig. 1999, 17, 342–348. [Google Scholar] [CrossRef] [PubMed]
- van Helmondt, S.J.; van der Lee, M.L.; van Woezik, R.A.M.; Lodder, P.; de Vries, J. No effect of CBT-based online self-help training to reduce fear of cancer recurrence: First results of the CAREST multicenter randomized controlled trial. Psychooncology 2019, 29, 86–97. [Google Scholar] [CrossRef] [PubMed]
- Lueboonthavatchai, P. Prevalence and psychosocial factors of anxiety and depression in breast cancer patients. J. Med Assoc. Thail. 2007, 90, 2164–2174. [Google Scholar]
- Tsaras, K.; Papathanasiou, I.V.; Mitsi, D.; Veneti, A.; Kelesi, M.; Zyga, S.; Fradelos, E.C. Assessment of depression and anxiety in breast cancer patients: Prevalence and associated Factors. Asian Pac. J. Cancer Prev. 2018, 19, 1661–1669. [Google Scholar] [CrossRef]
- Izci, F.; Ilgun, A.S.; Findikli, E.; Ozmen, V. Psychiatric Symptoms and Psychosocial Problems in Patients with Breast Cancer. J. Breast Health 2016, 12, 94–101. [Google Scholar] [CrossRef]
- Triberti, S.; Savioni, L.; Sebri, V.; Pravettoni, G. eHealth for improving quality of life in breast cancer patients: A systematic review. Cancer Treat. Rev. 2019, 74, 1–14. [Google Scholar] [CrossRef]
- Perez-Tejada, J.; Labaka, A.; Pascual-Sagastizabal, E.; Garmendia, L.; Iruretagoyena, A.; Arregi, A. Predictors of psychological distress in breast cancer survivors: A biopsychosocial approach. Eur. J. Cancer Care 2019, 28, e13166. [Google Scholar] [CrossRef]
- Whittaker, A.L.; George, R.P.; O’Malley, L. Prevalence of cognitive impairment following chemotherapy treatment for breast cancer: A systematic review and meta-analysis. Sci. Rep. 2022, 12, 1–22. [Google Scholar] [CrossRef]
- Oksman, E.; Linna, M.; Hörhammer, I.; Lammintakanen, J.; Talja, M. Cost-effectiveness analysis for a tele-based health coaching program for chronic disease in primary care. BMC Health Serv. Res. 2017, 17, 1–7. [Google Scholar] [CrossRef] [PubMed]
- Ambrosino, N.; Fracchia, C. The role of tele-medicine in patients with respiratory diseases. Expert Rev. Respir. Med. 2017, 11, 893–900. [Google Scholar] [CrossRef]
- Maresca, G.; Maggio, M.G.; De Luca, R.; Manuli, A.; Tonin, P.; Pignolo, L.; Calabrò, R.S. Tele-Neuro-Rehabilitation in Italy: State of the Art and Future Perspectives. Front. Neurol. 2020, 11. [Google Scholar] [CrossRef] [PubMed]
- von Storch, K.; Graaf, E.; Wunderlich, M.; Rietz, C.; Polidori, M.C.; Woopen, C. Telemedicine-Assisted Self-Management Program for Type 2 Diabetes Patients. Diabetes Technol. Ther. 2019, 21, 514–521. [Google Scholar] [CrossRef] [PubMed]
- Koç, Z.; Kaplan, E.; Tanrıverdi, D. The effectiveness of telehealth programs on the mental health of women with breast cancer: A systematic review. J. Telemed. Telecare 2022. [Google Scholar] [CrossRef] [PubMed]
- Damholdt, M.; Mehlsen, M.; O’Toole, M.; Andreasen, R.; Pedersen, A.; Zachariae, R. Web-based cognitive training for breast cancer survivors with cognitive complaints—A randomized controlled trial. Psychooncology 2016, 25, 1293–1300. [Google Scholar] [CrossRef]
- Galiano-Castillo, N.; Ariza-García, A.; Cantarero-Villanueva, I.; Fernández-Lao, C.; Díaz-Rodríguez, L.; Legerén-Alvarez, M.; Sánchez-Salado, C.; Del-Moral-Avila, R.; Arroyo-Morales, M. Telehealth system (e-CUIDATE) to improve quality of life in breast cancer survivors: Rationale and study protocol for a randomized clinical trial. Trials 2013, 14, 187. [Google Scholar] [CrossRef]
- Freeman, L.W.; White, R.; Ratcliff, C.G.; Sutton, S.; Stewart, M.; Palmer, J.L.; Link, J.; Cohen, L. A randomized trial comparing live and telemedicine deliveries of an imagery-based behavioral intervention for breast cancer survivors: Reducing symptoms and barriers tocare. Psychooncology 2014, 24, 910–918. [Google Scholar] [CrossRef]
- Page, M.J.; E McKenzie, J.; Bossuyt, P.M.; Boutron, I.; Hoffmann, T.C.; Mulrow, C.D.; Shamseer, L.; Tetzlaff, J.M.; Moher, D. Updating guidance for reporting systematic reviews: Development of the PRISMA 2020 statement. J. Clin. Epidemiol. 2021, 134, 103–112. [Google Scholar] [CrossRef]
- Krzyzanowska, M.K.; A Julian, J.; Gu, C.-S.; Powis, M.; Li, Q.; Enright, K.; Howell, D.; Earle, C.C.; Gandhi, S.; Rask, S.; et al. Remote, proactive, telephone based management of toxicity in outpatients during adjuvant or neoadjuvant chemotherapy for early stage breast cancer: Pragmatic, cluster randomised trial. BMJ 2021, 375, e066588. [Google Scholar] [CrossRef] [PubMed]
- Lozano-Lozano, M.; Martín-Martín, L.; Galiano-Castillo, N.; Fernández-Lao, C.; Cantarero-Villanueva, I.; López-Barajas, I.B.; Arroyo-Morales, M. Mobile health and supervised rehabilitation versus mobile health alone in breast cancer survivors: Randomized controlled trial. Ann. Phys. Rehabil. Med. 2019, 63, 316–324. [Google Scholar] [CrossRef] [PubMed]
- Lozano-Lozano, M.; Galiano-Castillo, N.; Gonzalez-Santos, A.; Ortiz-Comino, L.; Sampedro-Pilegaard, M.; Martín-Martín, L.; Arroyo-Morales, M. Effect of mHealth plus occupational therapy on cognitive function, mood and physical function in people after cancer: Secondary analysis of a randomized controlled trial. Ann. Phys. Rehabil. Med. 2023, 66, 101681. [Google Scholar] [CrossRef] [PubMed]
- Admiraal, J.M.; van der Velden, A.W.; Geerling, J.I.; Burgerhof, J.G.; Bouma, G.; Walenkamp, A.M.; de Vries, E.; Schröder, C.P.; Reyners, A.K. Web-Based Tailored Psychoeducation for Breast Cancer Patients at the Onset of the Survivorship Phase: A Multicenter Randomized Controlled Trial. J. Pain Symptom Manag. 2017, 54, 466–475. [Google Scholar] [CrossRef] [PubMed]
- Galiano-Castillo, N.; Arroyo-Morales, M.; Lozano-Lozano, M.; Fernández-Lao, C.; Martín-Martín, L.; Del-Moral-Ávila, R.; Cantarero-Villanueva, I. Effect of an Internet-based telehealth system on functional capacity and cognition in breast cancer survivors: A secondary analysis of a randomized controlled trial. Support. Care Cancer 2017, 25, 3551–3559. [Google Scholar] [CrossRef]
- Galiano-Castillo, N.; Cantarero-Villanueva, I.; Fernández-Lao, C.; Ariza-García, A.; Díaz-Rodríguez, L.; Del-Moral-Ávila, R.; Arroyo-Morales, M. Telehealth system: A randomized controlled trial evaluating the impact of an internet-based exercise intervention on quality of life, pain, muscle strength, and fatigue in breast cancer survivors. Cancer 2016, 122, 3166–3174. [Google Scholar] [CrossRef] [PubMed]
- Zachariae, R.; Amidi, A.; Damholdt, M.F.; Clausen, C.D.R.; Dahlgaard, J.; Lord, H.; Thorndike, F.P.; Ritterband, L.M. Internet-Delivered Cognitive-Behavioral Therapy for Insomnia in Breast Cancer Survivors: A Randomized Controlled Trial. Gynecol. Oncol. 2018, 110, 880–887. [Google Scholar] [CrossRef]
- Bandani-Susan, B.; Montazeri, A.; Haghighizadeh, M.H.; Araban, M. The effect of mobile health educational intervention on body image and fatigue in breast cancer survivors: A randomized controlled trial. Ir. J. Med Sci. 2021, 191, 1599–1605. [Google Scholar] [CrossRef]
- Berg, S.W.V.D.; Gielissen, M.F.; Custers, J.A.; Van Der Graaf, W.T.; Ottevanger, P.B.; Prins, J.B. BREATH: Web-Based Self-Management for Psychological Adjustment after Primary Breast Cancer—Results of a Multicenter Randomized Controlled Trial. J. Clin. Oncol. 2015, 33, 2763–2771. [Google Scholar] [CrossRef]
- Oswald, L.B.; Morales-Cruz, J.; Eisel, S.L.; Del Rio, J.; Hoogland, A.I.; Ortiz-Rosado, V.; Soto-Lopez, G.; Rodriguez-Rivera, E.; Savard, J.; Castro, E.; et al. Pilot randomized controlled trial of eHealth cognitive-behavioral therapy for insomnia among Spanish-speaking breast cancer survivors. J. Behav. Med. 2022, 45, 503–508. [Google Scholar] [CrossRef]
- Carlson, L.E.; Subnis, U.B.; Piedalue, K.L.; Vallerand, J.; Speca, M.; Lupichuk, S.; Tang, P.; Faris, P.; Wolever, R.Q. The ONE-MIND Study: Rationale and protocol for assessing the effects of ONlinE MINDfulness-based cancer recovery for the prevention of fatigue and other common side effects during chemotherapy. Eur. J. Cancer Care 2019, 28, e13074. [Google Scholar] [CrossRef] [PubMed]
- González, Á.S.; Lopez-Garzon, M.; Sánchez-Salado, C.; Postigo-Martin, P.; Lozano-Lozano, M.; Galiano-Castillo, N.; Fernández-Lao, C.; Castro-Martín, E.; Gallart-Aragón, T.; Legerén-Álvarez, M.; et al. A Telehealth-Based Cognitive-Adaptive Training (e-OTCAT) to Prevent Cancer and Chemotherapy-Related Cognitive Impairment in Women with Breast Cancer: Protocol for a Randomized Controlled Trial. Int. J. Environ. Res. Public Health 2022, 19, 7147. [Google Scholar] [CrossRef] [PubMed]
- Lidington, E.; E McGrath, S.; Noble, J.; Stanway, S.; Lucas, A.; Mohammed, K.; van der Graaf, W.; Husson, O. Evaluating a digital tool for supporting breast cancer patients: A randomized controlled trial protocol (ADAPT). Trials 2020, 21, 1–10. [Google Scholar] [CrossRef] [PubMed]
- Krusche, A.; Bradbury, K.; Corbett, T.; Barnett, J.; Stuart, B.; Yao, G.L.; Bacon, R.; Böhning, D.; Cheetham-Blake, T.; Eccles, D.; et al. Renewed: Protocol for a randomised controlled trial of a digital intervention to support quality of life in cancer survivors. BMJ Open 2019, 9, e024862. [Google Scholar] [CrossRef]
- Tombaugh, T.N. Trail Making Test A and B: Normative data stratified by age and education. Arch. Clin. Neuropsychol. 2004, 19, 203–214. [Google Scholar] [CrossRef] [PubMed]
- Crawford, J.R.; Obonsawin, M.C.; Allan, K.M. PASAT and Components of WAIS-R Performance: Convergent and Discriminant Validity. Neuropsychol. Rehabil. 1998, 8, 255–272. [Google Scholar] [CrossRef]
- Jacobs, S.R.; Jacobsen, P.B.; Booth-Jones, M.; Wagner, L.I.; Anasetti, C. Evaluation of the Functional Assessment of Cancer Therapy Cognitive Scale with Hematopoetic Stem Cell Transplant Patients. J. Pain Symptom Manag. 2007, 33, 13–23. [Google Scholar] [CrossRef]
- Scott, J.; Teasdale, J.D.; Paykel, E.S.; Johnson, A.L.; Abbott, R.; Hayhurst, H.; Moore, R.; Garland, A. Effects of Cognitive Therapy on Psychological Symptoms and Social Functioning in Residual Depression. Focus 2005, 3, 122–130. [Google Scholar] [CrossRef]
- Bourne, L.E., Jr.; Yaroush, R.A. Stress and cognition: A cognitive psychological perspective. NASA Tech. Reports Serv. 2003. [Google Scholar]
- Fobair, P.; Stewart, S.L.; Chang, S.; D’Onofrio, C.; Banks, P.J.; Bloom, J.R. Body image and sexual problems in young women with breast cancer. Psychooncology 2005, 15, 579–594. [Google Scholar] [CrossRef]
- Carroll, J.E.; Van Dyk, K.; Bower, J.E.; Scuric, Z.; Ms, L.P.; Schiestl, R.; Irwin, M.; Ganz, P.A. Cognitive performance in survivors of breast cancer and markers of biological aging. Cancer 2018, 125, 298–306. [Google Scholar] [CrossRef] [PubMed]
- Wagner, A.D. Sex differences in cancer chemotherapy effects, and why we need to reconsider BSA-based dosing of chemotherapy. ESMO Open 2020, 5, e000770. [Google Scholar] [CrossRef] [PubMed]
- Ganz, P.A.; Van Dyk, K. Cognitive Impairment in Patients with Breast Cancer: Understanding the Impact of Chemotherapy and Endocrine Therapy. J. Clin. Oncol. 2020, 38, 1871–1874. [Google Scholar] [CrossRef]
- Cheung, Y.T.; Ng, T.; Shwe, M.; Ho, H.K.; Foo, K.M.; Cham, M.T.; Lee, J.A.; Fan, G.; Tan, Y.P.; Yong, W.S.; et al. Association of proinflammatory cytokines and chemotherapy-associated cognitive impairment in breast cancer patients: A multi-centered, prospective, cohort study. Ann. Oncol. 2015, 26, 1446–1451. [Google Scholar] [CrossRef] [PubMed]
- Meyers, C.A.; Albitar, M.; Estey, E. Cognitive impairment, fatigue, and cytokine levels in patients with acute myelogenous leukemia or myelodysplastic syndrome. Cancer 2005, 104, 788–793. [Google Scholar] [CrossRef]
- Carroll, J.E.; Nakamura, Z.M.; Small, B.J.; Zhou, X.; Cohen, H.J.; Ahles, T.A.; Ahn, J.; Bethea, T.N.; Extermann, M.; Graham, D.; et al. Elevated C-Reactive Protein and Subsequent Patient-Reported Cognitive Problems in Older Breast Cancer Survivors: The Thinking and Living with Cancer Study. J. Clin. Oncol. 2023, 41, 295–306. [Google Scholar] [CrossRef]
- Asher, A.; Myers, J.S. The effect of cancer treatment on cognitive function. Clin. Adv. Hematol. Oncol. 2015. [Google Scholar]
- Mandelblatt, J.S.; Stern, R.A.; Luta, G.; McGuckin, M.; Clapp, J.D.; Hurria, A.; Jacobsen, P.B.; Faul, L.A.; Isaacs, C.; Denduluri, N.; et al. Cognitive Impairment in Older Patients with Breast Cancer before Systemic Therapy: Is There an Interaction between Cancer and Comorbidity? J. Clin. Oncol. 2014, 32, 1909–1918. [Google Scholar] [CrossRef]
- Irwin, M.R.; Cole, S.W. Reciprocal regulation of the neural and innate immune systems. Nat. Rev. Immunol. 2011, 11, 625–632. [Google Scholar] [CrossRef]
- Mishra, J.; Gazzaley, A. Harnessing the neuroplastic potential of the human brain & the future of cognitive rehabilitation. Front. Hum. Neurosci. 2014, 8, 218. [Google Scholar] [CrossRef]
- Koutrouli, N.; Anagnostopoulos, F.; Potamianos, G. Posttraumatic Stress Disorder and Posttraumatic Growth in Breast Cancer Patients: A Systematic Review. Women Health 2012, 52, 503–516. [Google Scholar] [CrossRef] [PubMed]
- Runowicz, C.D.; Leach, C.R.; Henry, N.L.; Henry, K.S.; Mackey, H.T.; Cowens-Alvarado, R.L.; Cannady, R.S.; Pratt-Chapman, M.; Edge, S.B.; Jacobs, L.A.; et al. American Cancer Society/American Society of Clinical Oncology Breast Cancer Survivorship Care Guideline. CA A Cancer J. Clin. 2015, 66, 43–73. [Google Scholar] [CrossRef] [PubMed]
| (A) | |||||||||
|---|---|---|---|---|---|---|---|---|---|
| Author and Year | Sample Size | Age | Cancer Stage | Treatment | Type of Telemedicine Applied | Control | Duration | Outcome Variables | Results |
| Admiraal et al., 2017 [27] | 139 | C: 53.2 ± 8.5; T: 53.1 ± 9.8 | I–III | Completed curative-intent primary treatment (surgery + chemotherapy) within the past six months | ENCOURAGE program: Psycho-education, problem-solving strategies for coping | Standard care | 12 weeks | EORTC, QoL_Questionnaire, optimism and control over the future scale, Dutch Distress Thermometer, 47-item problem list | Increased optimism and control over the future |
| Bandani-Susan et al., 2021 [31] | 38 | 46.34 ± 9.96 (C: 45.89 ± 7.64; T: 46.79 ± 12.28) | NA | Ongoing chemotherapy, radiotherapy, hormone therapy and/or brachytherapy | Educational messages, cognitive behavioral therapy for improving body image, and cognitive restructuring | Waitlist | 49 days | CFS; Body Image Concern Inventory | Significant difference in quality of life (body image and cancer-related fatigue) |
| Damholdt et al., 2016 [21] | 157 | C: 54.56 ± 8.74; T: 54.98 ± 8.51 | 0-III | Ongoing chemotherapy, radiotherapy, or hormonal therapy | HappyNeuron Pro: Web-based program for cognitive training focused on six cognitive domains (attention, processing speed, learning, memory, working memory, problem-solving) | Waitlist | 6 weeks | PASAT, RAVLT, Digit Span Forwards, Digit Span Backwards, Digit Ordering, Letter Fluency Test, 20 Questions Test, Cognitive Estimation Task, BDI, Whitely-7, SCL-ANX4 from Symptoms Checklist-92, self-reported benefit from the training | Improvements in verbal learning and working memory |
| Freeman et al., 2014 [23] | 102 | C: 55.28 ± 7.90; LD: 55.44 ± 8.08; T: 55.57 ± 9.88 | 0-IV | Completed main treatment from at least 6 weeks | Videoconference groups, education on the mind–body connection and on mental imagery | Live delivered (LD) psycho-educational groups; waitlist | 5 weeks | Medical Outcomes Study survey (SF-36); FACT-B; FACIT-Fatigue Scale; FACT-Cog; Functional Assessment of Chronic Illness Therapy; Brief Symptom Inventory-GSI; Pittsburgh Sleep Quality Index | Improvement in fatigue, cognitive dysfunction, sleep disturbance, and health-related and breast cancer-related QoL for LD and TD groups compared to waitlist |
| Galiano-Castillo et al., 2016 [29] | 81 | C: 49.2 ± 7.9; T: 47.4 ± 9.6 | I-III | Completed adjuvant therapy except hormone treatment | e-CUIDATE: Online system for the remote administration of physical exercises focused on resistance, aerobic, mobility, and stretching | Written recommendation on stress management, physical fitness | 8 weeks | EORTC QLQ-C30; Brief Pain Inventory short form; Piper Fatigue Scale-revised | Improvements in global health status, pain severity, interference, and total fatigue |
| Galiano-Castillo et al., 2017 [28] | 81 | 48.30 ± 8.80 | I-III | Completed adjuvant therapy except hormone treatment | e-CUIDATE: Online system for the remote administration of physical | Written recommendation on stress management, physical fitness | 8 weeks | 6 Min Walk Test; Auditory Consonant Trigrams; TMT | Improvements in functional capacity and cognitive functioning maintained at 6 months |
| Krzyzanowska et al., 2021 [25] | 561 | 55.7 * | I-IV | Starting adjuvant or neoadjuvant chemotherapy | Symptom Self-Management Booklet-patient edition + follow-up calls to address the presence of chemotherapy side effects | Standard care | Duration of chemotherapy cycle | NCI PRO-CTCAE; Stanford self-management self-efficacy scale; European quality-of-life; Patient Health Questionnaire 9; VAS; Generalized anxiety disorder; FACT-B | No differences in self-efficacy, anxiety, or depression |
| Lozano-Lozano et al., 2020 [34] | 80 | C: 49.76 ± 8.42; T: 53.40 ± 8.66 | I-III | Completed adjuvant therapy except hormonal therapy | BENECA mHEALTH + rehabilitation: Supervised used of the BENECA app occupational therapy focused on reduction of fatigue and improvement of processing speed, working memory, and attention | BENECA mHEALTH: App for recommendation on physical activity and nutrition | 8 weeks | EORTC- QoL Questionnaire; Breast Cancer-Specific Quality of Life Questionnaire; | Improvement in QoL, maintained after 6 months |
| Lozano-Lozano et al., 2022 [26] | 80 | C: 49.76 ± 8.42; T: 53.40 ± 8.66 | I-III | Completed chemotherapy, could continue therapy with hormone | BENECA mHEALTH + rehabilitation: Supervised used of the BENECA app for recommendation on physical activity and nutrition; occupational therapy focused on reduction of fatigue and improvement of processing speed, working memory and attention | BENECA mHEALTH: App for recommendation on physical activity and nutrition | 8 weeks | TMT; WAIS; Hospital Anxiety and Depression Scale; Brief pain inventory; Piper Fatigue Scale-revised; 6 Min Walk Test | Greater improvements in selective attention, working memory, and processing speed, anxiety, and functional capacity at 8 weeks and 6 months. Fatigue perception and pain were also improved |
| Oswald et al., 2022 [33] | 30 | 58.44 ± 9.22 (C: 56.90 ± 8.91; T: 59.98 ± 9.58) | NA | Completed primary cancer treatment | CBT-I: Group sessions of cognitive behavioral therapy focused on sleep education, sleep restriction, stimulus control, sleep hygiene, and sleep medications, cognitive restructuring, and relapse prevention | Waitlist | 6 weeks | Treatment Perceptions Questionnaire; 7-item Insomnia Severity Index; Pittsburgh Sleep Quality Index | Improvements in insomnia symptoms, sleep disturbance, and sleep efficiency compared to the control group |
| van den Berg et al., 2015 [32] | 150 | C: 50.18 ± 9.15; T: 51.44 ± 8.30 | NA | Completed primary cancer treatment (surgery plus adjuvant chemotherapy and/or radiotherapy) | BREATH: Cognitive behavioral therapy online self-help program for the four phases of adjustment to breast cancer (looking back, emotional processing, strengthening, and looking ahead) | Standard care | 16 weeks | Symptom Checklist-90; Cancer Empowerment Questionnaire; Hospital Anxiety and Depression Scale; EORTC Quality of Life Questionnaire Core 30; Distress Thermometer; Illness Cognitions Questionnaire; Remoralization Scale; Mastery Scale; Positive Adjustment Questionnaire; Self-Efficacy Scale; Cancer Worry Scale; Cancer Acceptance Scale; Checklist Individual Strength-Fatigue; Openness to discuss hereditary cancer in the family; Big Five Inventory | Reduced distress. Not persistent at follow-up |
| 255 | 53.1 ± 8.8 (C: 52.9 ± 8.9; T: 53.2 ± 8.8) | I-III | Ongoing chemotherapy, radiotherapy, or endocrine therapy | SHUTi: Automated interactive cognitive behavioral therapy for insomnia focused on sleep restriction and stimulus control, cognitive restructuring, sleep hygiene, and relapse prevention | Waitlist | 6 weeks | Sleep diary; Insomnia Severity Index; Pittsburgh Sleep Quality Index; Functional Assessment of Chronic Illness Therapy for Fatigue | iCBT-I groups showed improvements in sleep-related outcomes which were maintained at 15 weeks follow-up | |
| (B) | |||||||||
| Author and Year | Sample Size | Age | Cancer Stage | Treatment | Type of Telemedicine Applied | Control | Duration | Outcome Variables | |
| Carlson et al., 2019 [35] | 178 | >18 | I–III | Scheduled for chemotherapy | Mindfulness-based cancer recovery, online group | Standard care, waitlist | 12 weeks | Brief Screen for Cognitive Impairment; Functional Assessment of Chronic Illness Therapy—Fatigue; Pittsburgh Sleep Quality Index; Brief Pain Inventory; Osoba Nausea and Vomiting Module; FACT—General; Calgary Symptoms of Stress Inventory; Profile of Mood States—Short Form; FACT—Cog; Sustained Attention to Response Task; blood counts | |
| González-Santos et al., 2022 [36] | 98 | >18 | I–III | Scheduled for chemotherapy | e-OTCAT program: Cognitive training using paper-pencil exercises and the NeuroNation mobile app | Standard care, provision of educational handbook on cancer treatment side effects | 12 weeks | FACT-Cog; TMT; WAIS; Hospital Anxiety and Depression Scale; Piper Fatigue Scale-Revised; Pittsburgh Sleep Quality Index; EORTC-Quality of Life Questionnaire Core; Canadian Occupational Performance Measure | |
| Krusche et al., 2019 [38] | 2500 | >18 | NA | Finished primary cancer treatment within prior ten years | Renewed: Software addressing four main areas (physical activity, stress reduction, diet improvement, weight loss) | Standard care, provision of educational resources | NA | QoL; fear of relapses; anxiety and depression; website satisfaction and usage | |
| Lidington et al., 2020 [37] | 122 | >18 | Early stage | Non-specified anticancer treatment | OWise: Online tool offering tailored medical information, medical terms glossary, useful links to local resources, tracking tool for symptoms, and a consultation recording device | Standard care, provision of educational resources | NA | Hospital Anxiety and Depression Scale; EORTC QLQ C-30; EuroQol 5-Dimension 5-Level questionnaire (EQ-5D-5L) | |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Giustiniani, A.; Danesin, L.; Pezzetta, R.; Masina, F.; Oliva, G.; Arcara, G.; Burgio, F.; Conte, P. Use of Telemedicine to Improve Cognitive Functions and Psychological Well-Being in Patients with Breast Cancer: A Systematic Review of the Current Literature. Cancers 2023, 15, 1353. https://doi.org/10.3390/cancers15041353
Giustiniani A, Danesin L, Pezzetta R, Masina F, Oliva G, Arcara G, Burgio F, Conte P. Use of Telemedicine to Improve Cognitive Functions and Psychological Well-Being in Patients with Breast Cancer: A Systematic Review of the Current Literature. Cancers. 2023; 15(4):1353. https://doi.org/10.3390/cancers15041353
Chicago/Turabian StyleGiustiniani, Andreina, Laura Danesin, Rachele Pezzetta, Fabio Masina, Giulia Oliva, Giorgio Arcara, Francesca Burgio, and Pierfranco Conte. 2023. "Use of Telemedicine to Improve Cognitive Functions and Psychological Well-Being in Patients with Breast Cancer: A Systematic Review of the Current Literature" Cancers 15, no. 4: 1353. https://doi.org/10.3390/cancers15041353
APA StyleGiustiniani, A., Danesin, L., Pezzetta, R., Masina, F., Oliva, G., Arcara, G., Burgio, F., & Conte, P. (2023). Use of Telemedicine to Improve Cognitive Functions and Psychological Well-Being in Patients with Breast Cancer: A Systematic Review of the Current Literature. Cancers, 15(4), 1353. https://doi.org/10.3390/cancers15041353








