Association between Plasminogen Activator Inhibitor-1 and Osimertinib Tolerance in EGFR-Mutated Lung Cancer via Epithelial–Mesenchymal Transition
Abstract
Simple Summary
Abstract
1. Introduction
2. Materials and Methods
2.1. Cell Lines
2.2. Patients
2.3. Cell Culture
2.4. Reagents
2.5. Quantitative RealTime PCR (qRT-PCR)
2.6. Quantification of PAI-1 Protein
2.7. Immunohistochemical Staining
2.8. Generation of EGFR-TKI-Tolerant Cells
2.9. EGFR T790M, C797S Mutation, and Gene Copy Number Analyses
2.10. Validation of PAI-1-siRNA Knockdown Efficacy
2.11. Proliferation Assay
2.12. Analysis of PAI-1- or EMT-Related Gene Expression
2.13. Microarray
2.14. Animal Experimental Procedure
2.15. Statistical Analysis
3. Results
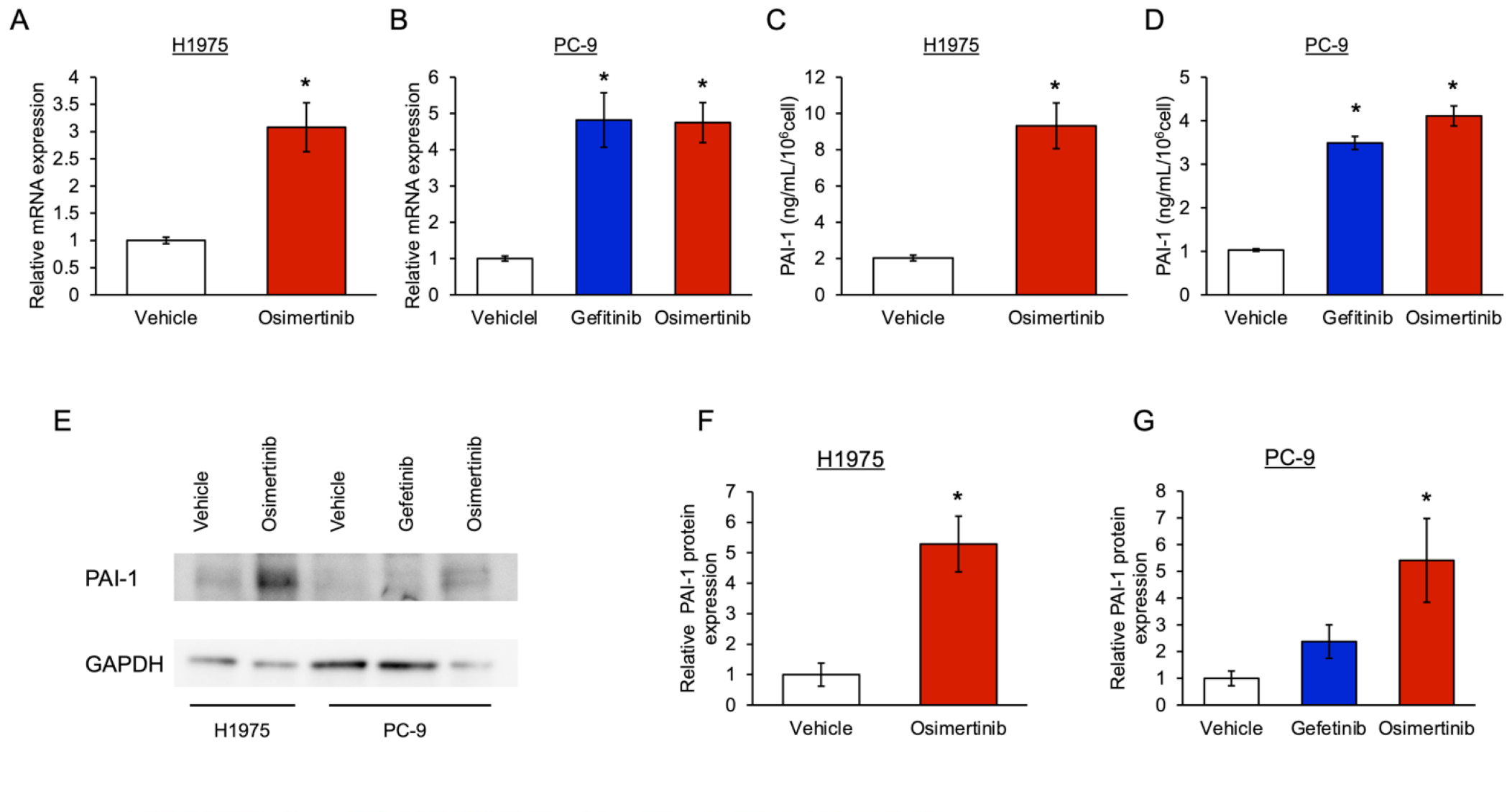
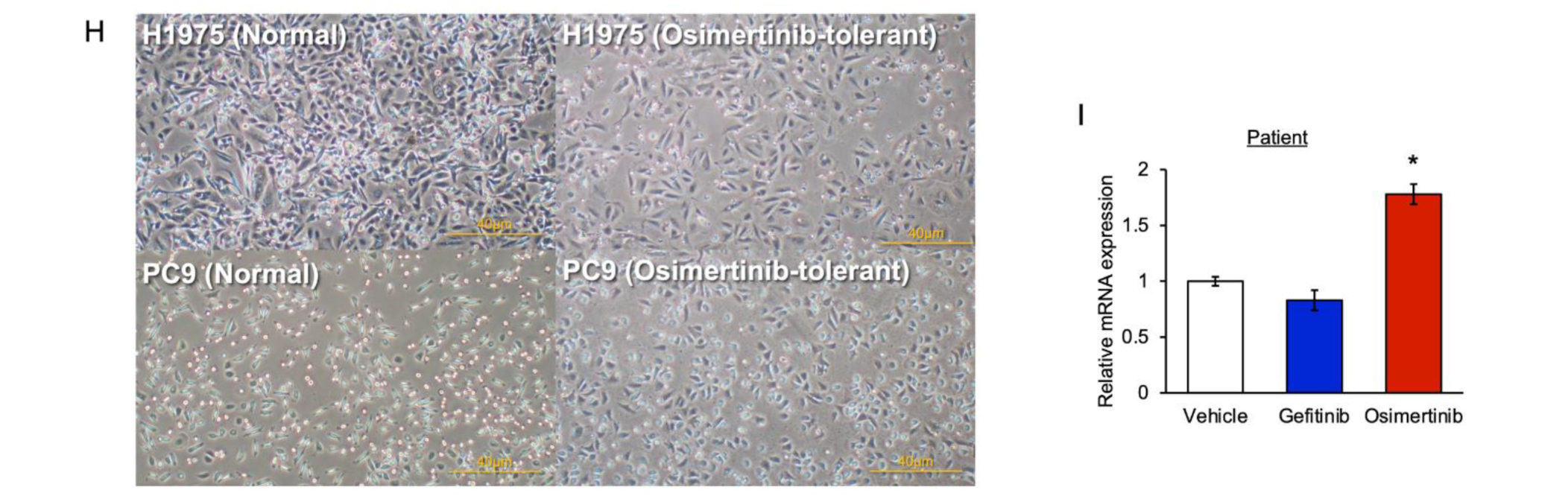
3.1. The Level of PAI-1 Increased in EGFR-Mutated Cancer Cells with Tolerance to EGFR-TKI
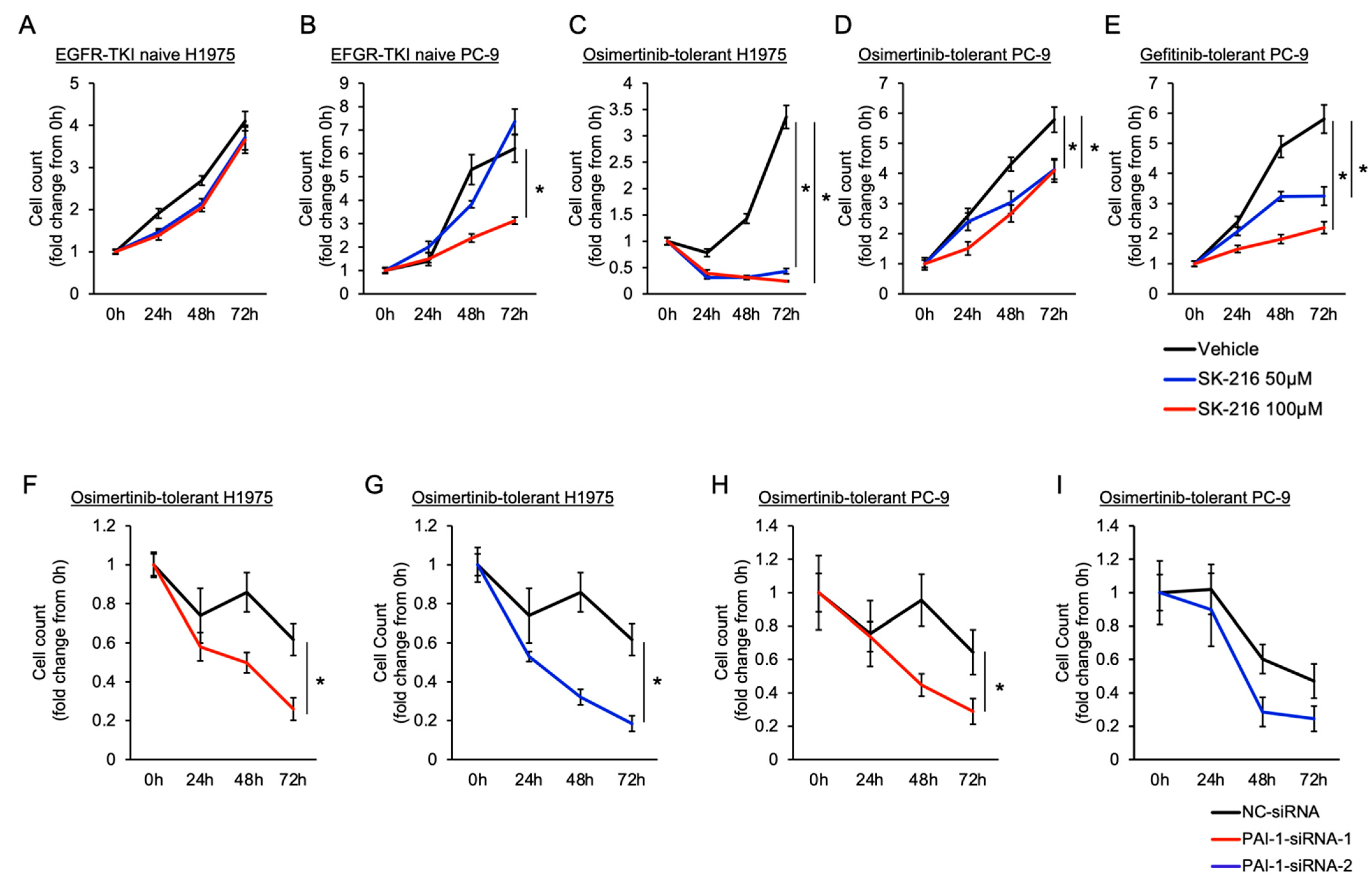
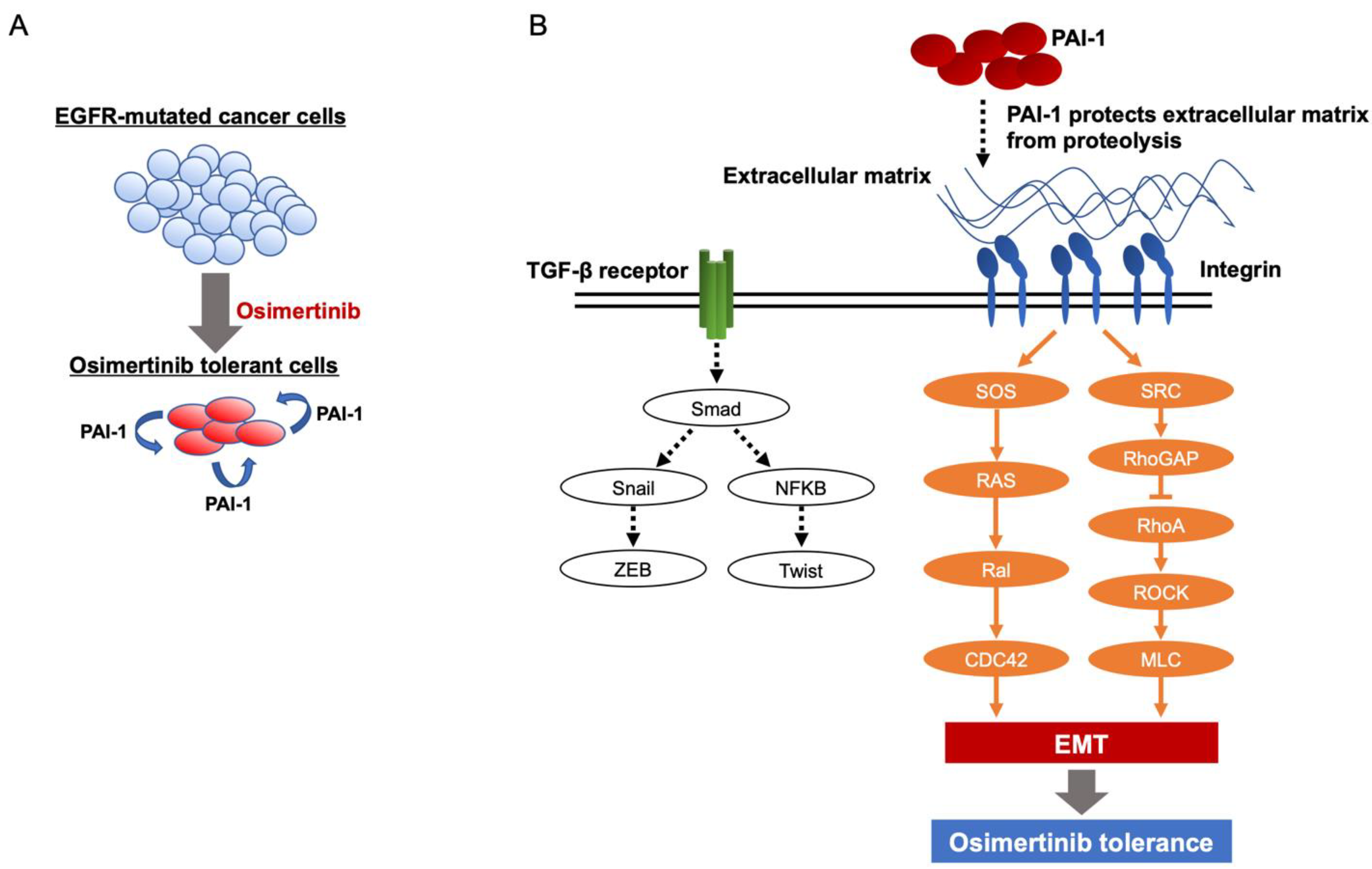
3.2. Involvement of PAI-1 in Tolerance to EGFR-TKI in EGFR-Mutated Lung Cancer Cells
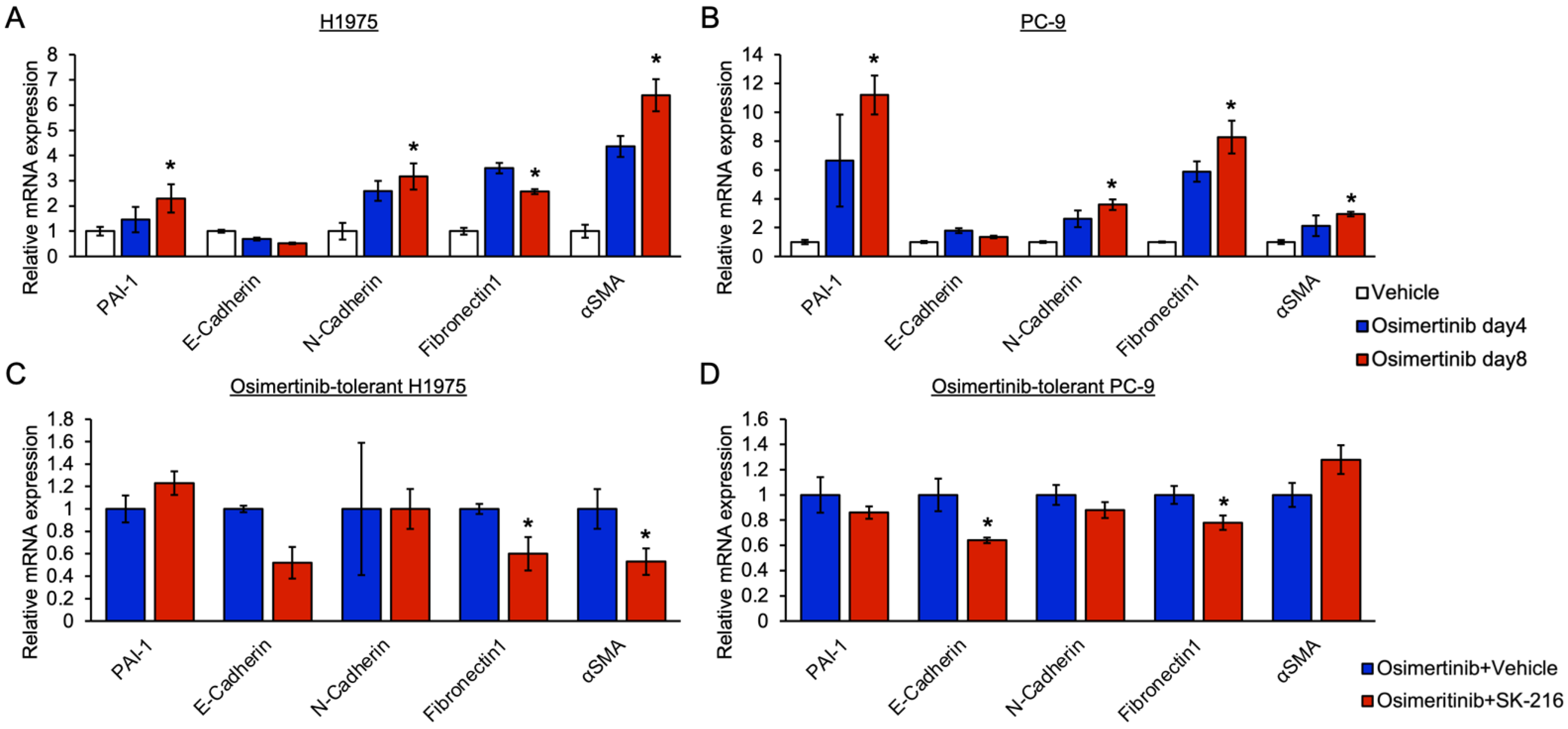
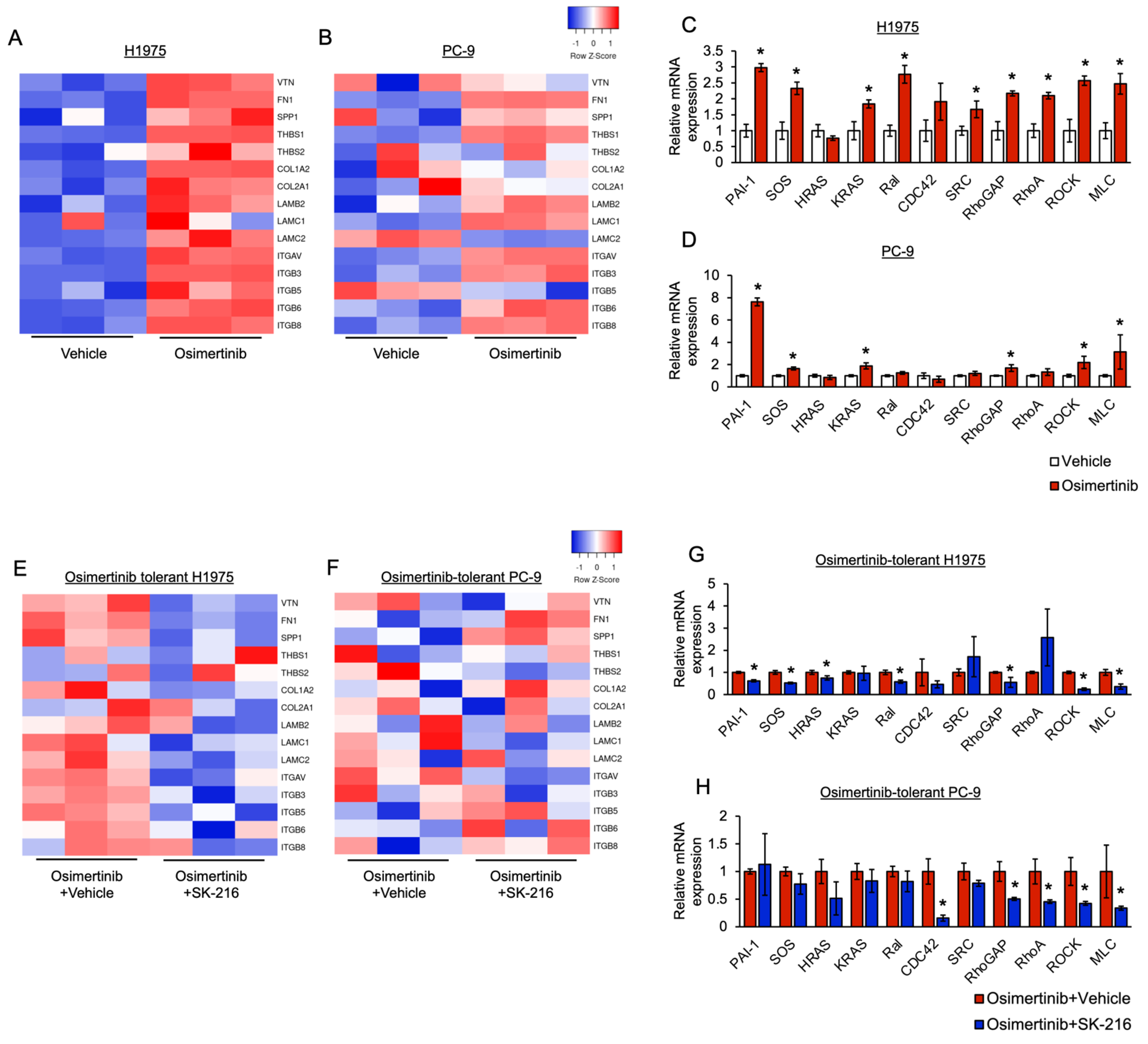
3.3. PAI-1 Was Found to Be Involved in Tolerance to Osimertinib in EGFR-Mutated Lung Cancer via Its Association with EMT
3.4. PAI-1 Was Found to Be Involved in Tolerance to Osimertinib via Its Association with Integrin-Initiated EMT
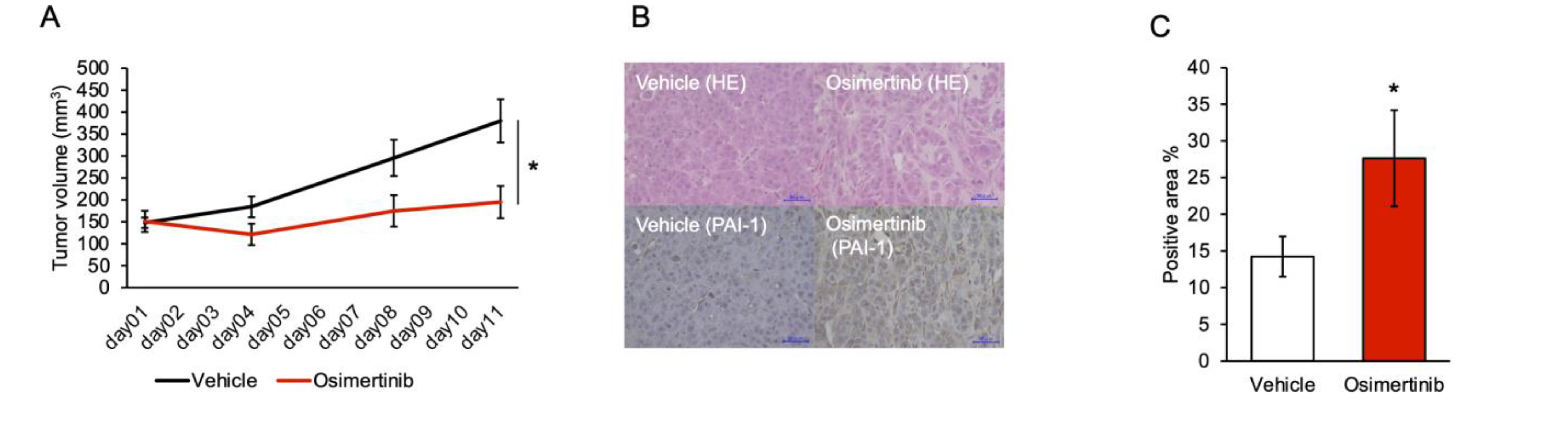
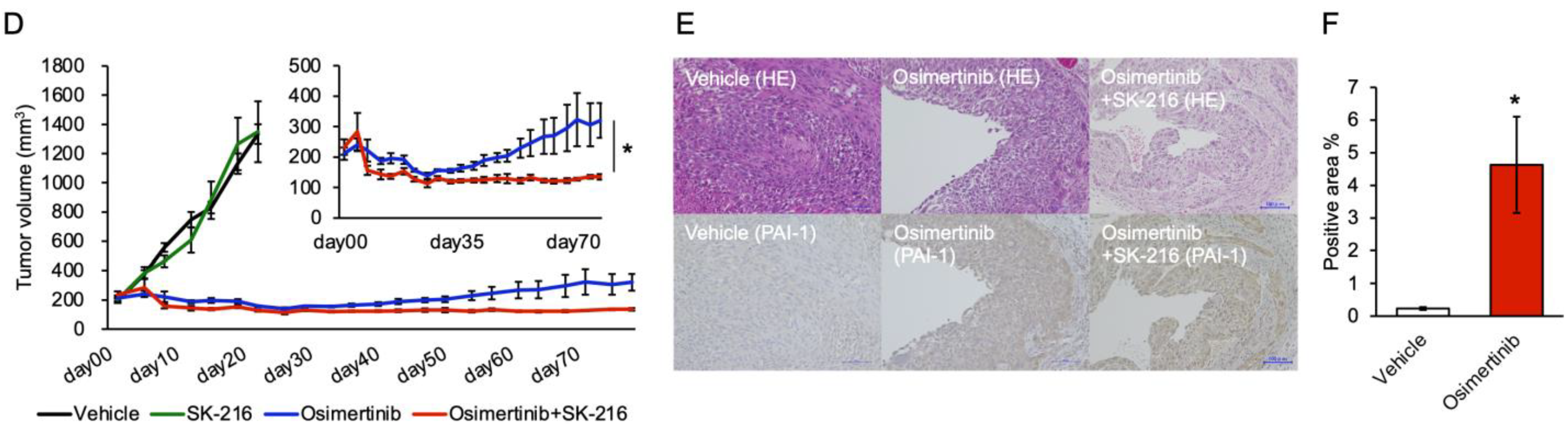
3.5. PAI-1 Inhibition Limited Regrowth of Osimertinib-Treated Tumors in the Subcutaneous Tumor Model
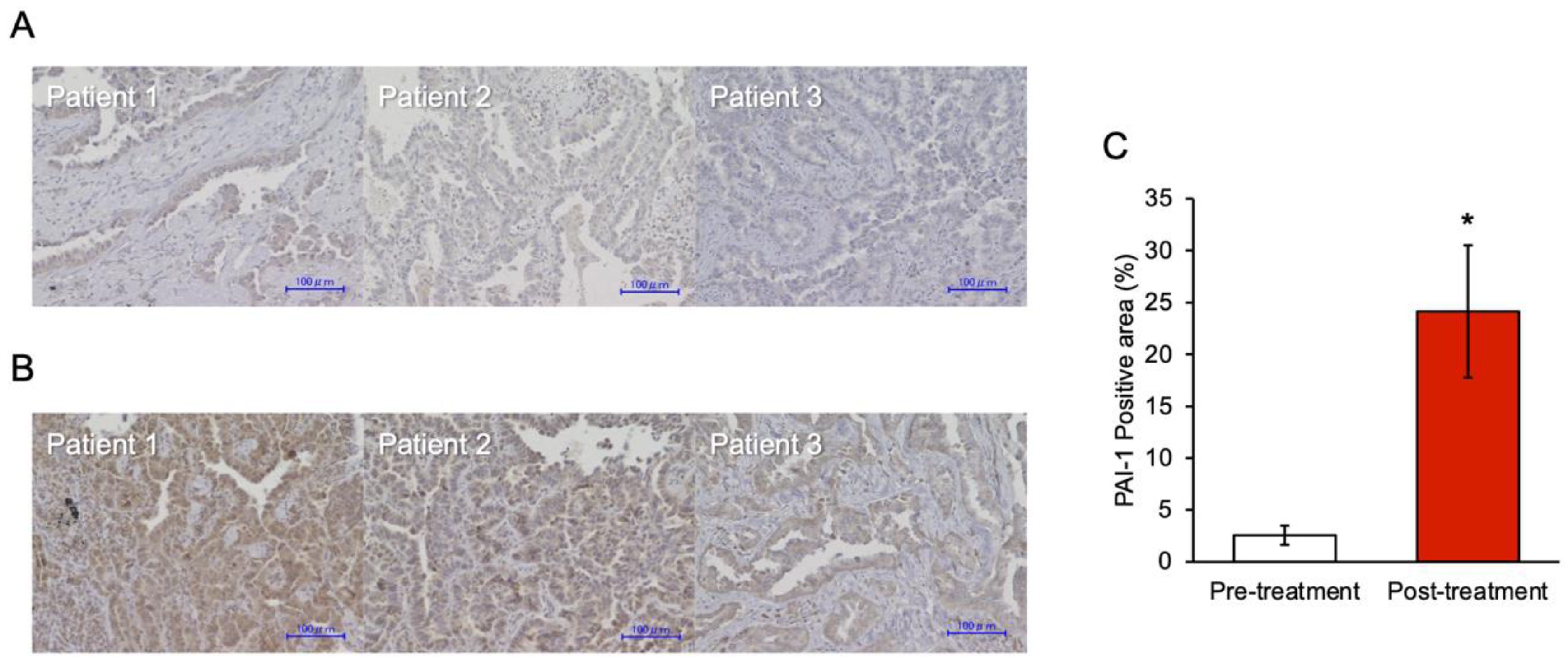
3.6. PAI-1 Expression in Tumor Specimens at the Time of Acquired Resistance to EGFR-TKIs
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Sung, H.; Ferlay, J.; Siegel, R.L.; Laversanne, M.; Soerjomataram, I.; Jemal, A.; Bray, F. Global Cancer Statistics 2020: GLOBOCAN Estimates of Incidence and Mortality Worldwide for 36 Cancers in 185 Countries. CA Cancer J. Clin. 2021, 71, 209–249. [Google Scholar] [CrossRef] [PubMed]
- Toyoda, Y.; Nakayama, T.; Ioka, A.; Tsukuma, H. Trends in Lung Cancer Incidence by Histological Type in Osaka, Japan. Jpn. J. Clin. Oncol. 2008, 38, 534–539. [Google Scholar] [CrossRef] [PubMed]
- Saito, M.; Shiraishi, K.; Kunitoh, H.; Takenoshita, S.; Yokota, J.; Kohno, T. Gene Aberrations for Precision Medicine against Lung Adenocarcinoma. Cancer Sci. 2016, 107, 713–720. [Google Scholar] [CrossRef] [PubMed]
- Maemondo, M.; Inoue, A.; Kobayashi, K.; Sugawara, S.; Oizumi, S.; Isobe, H.; Gemma, A.; Harada, M.; Yoshizawa, H.; Kinoshita, I.; et al. Gefitinib or Chemotherapy for Non-Small-Cell Lung Cancer with Mutated EGFR. N. Engl. J. Med. 2010, 362, 2380–2388. [Google Scholar] [CrossRef]
- Zhou, C.; Wu, Y.-L.; Chen, G.; Feng, J.; Liu, X.-Q.; Wang, C.; Zhang, S.; Wang, J.; Zhou, S.; Ren, S.; et al. Erlotinib versus Chemotherapy as First-Line Treatment for Patients with Advanced EGFR Mutation-Positive Non-Small-Cell Lung Cancer (OPTIMAL, CTONG-0802): A Multicentre, Open-Label, Randomised, Phase 3 Study. Lancet Oncol. 2011, 12, 735–742. [Google Scholar] [CrossRef]
- Wu, Y.-L.; Zhou, C.; Hu, C.-P.; Feng, J.; Lu, S.; Huang, Y.; Li, W.; Hou, M.; Shi, J.H.; Lee, K.Y.; et al. Afatinib versus Cisplatin plus Gemcitabine for First-Line Treatment of Asian Patients with Advanced Non-Small-Cell Lung Cancer Harbouring EGFR Mutations (LUX-Lung 6): An Open-Label, Randomised Phase 3 Trial. Lancet Oncol. 2014, 15, 213–222. [Google Scholar] [CrossRef] [PubMed]
- Ettinger, D.S.; Wood, D.E.; Aisner, D.L.; Akerley, W.; Bauman, J.R.; Bharat, A.; Bruno, D.S.; Chang, J.Y.; Chirieac, L.R.; D’Amico, T.A.; et al. Non-Small Cell Lung Cancer, Version 3.2022, NCCN Clinical Practice Guidelines in Oncology. J. Natl. Compr. Canc. Netw. 2022, 20, 497–530. [Google Scholar] [CrossRef]
- Nagano, T.; Tachihara, M.; Nishimura, Y. Mechanism of Resistance to Epidermal Growth Factor Receptor-Tyrosine Kinase Inhibitors and a Potential Treatment Strategy. Cells 2018, 7, 212. [Google Scholar] [CrossRef]
- Mok, T.S.; Wu, Y.-L.; Ahn, M.-J.; Garassino, M.C.; Kim, H.R.; Ramalingam, S.S.; Shepherd, F.A.; He, Y.; Akamatsu, H.; Theelen, W.S.M.E.; et al. Osimertinib or Platinum-Pemetrexed in EGFR T790M-Positive Lung Cancer. N. Engl. J. Med. 2017, 376, 629–640. [Google Scholar] [CrossRef]
- Soria, J.-C.; Ohe, Y.; Vansteenkiste, J.; Reungwetwattana, T.; Chewaskulyong, B.; Lee, K.H.; Dechaphunkul, A.; Imamura, F.; Nogami, N.; Kurata, T.; et al. Osimertinib in Untreated EGFR-Mutated Advanced Non-Small-Cell Lung Cancer. N. Engl. J. Med. 2018, 378, 113–125. [Google Scholar] [CrossRef]
- Li, X.-F.; Shen, W.-Z.; Jin, X.; Ren, P.; Zhang, J. Let-7c Regulated Epithelial-Mesenchymal Transition Leads to Osimertinib Resistance in NSCLC Cells with EGFR T790M Mutations. Sci. Rep. 2020, 10, 11236. [Google Scholar] [CrossRef]
- Qin, Q.; Li, X.; Liang, X.; Zeng, L.; Wang, J.; Sun, L.; Zhong, D. Targeting the EMT Transcription Factor Snail Overcomes Resistance to Osimertinib in EGFR-Mutant Non-Small Cell Lung Cancer. Thorac. Cancer 2021, 12, 1708–1715. [Google Scholar] [CrossRef] [PubMed]
- Jiang, Y.; Zhuo, X.; Wu, Y.; Fu, X.; Mao, C. PAR2 Blockade Reverses Osimertinib Resistance in Non-Small-Cell Lung Cancer Cells via Attenuating ERK-Mediated EMT and PD-L1 Expression. Biochim. Biophys. Acta Mol. Cell Res. 2022, 1869, 119144. [Google Scholar] [CrossRef]
- Fukuda, K.; Takeuchi, S.; Arai, S.; Kita, K.; Tanimoto, A.; Nishiyama, A.; Yano, S. Glycogen Synthase Kinase-3 Inhibition Overcomes Epithelial-Mesenchymal Transition-Associated Resistance to Osimertinib in EGFR-Mutant Lung Cancer. Cancer Sci. 2020, 111, 2374–2384. [Google Scholar] [CrossRef]
- Ji, W.; Choi, Y.J.; Kang, M.-H.; Sung, K.J.; Kim, D.H.; Jung, S.; Choi, C.-M.; Lee, J.C.; Rho, J.K. Efficacy of the CDK7 Inhibitor on EMT-Associated Resistance to 3rd Generation EGFR-TKIs in Non-Small Cell Lung Cancer Cell Lines. Cells 2020, 9, E2596. [Google Scholar] [CrossRef]
- Yochum, Z.A.; Cades, J.; Wang, H.; Chatterjee, S.; Simons, B.W.; O’Brien, J.P.; Khetarpal, S.K.; Lemtiri-Chlieh, G.; Myers, K.V.; Huang, E.H.-B.; et al. Targeting the EMT Transcription Factor TWIST1 Overcomes Resistance to EGFR Inhibitors in EGFR-Mutant Non-Small-Cell Lung Cancer. Oncogene 2019, 38, 656–670. [Google Scholar] [CrossRef]
- Jiang, X.-M.; Xu, Y.-L.; Yuan, L.-W.; Zhang, L.; Huang, M.-Y.; Ye, Z.-H.; Su, M.-X.; Chen, X.-P.; Zhu, H.; Ye, R.D.; et al. TGFβ2-Mediated Epithelial-Mesenchymal Transition and NF-ΚB Pathway Activation Contribute to Osimertinib Resistance. Acta Pharmacol. Sin. 2021, 42, 451–459. [Google Scholar] [CrossRef] [PubMed]
- Namba, K.; Shien, K.; Takahashi, Y.; Torigoe, H.; Sato, H.; Yoshioka, T.; Takeda, T.; Kurihara, E.; Ogoshi, Y.; Yamamoto, H.; et al. Activation of AXL as a Preclinical Acquired Resistance Mechanism Against Osimertinib Treatment in EGFR-Mutant Non-Small Cell Lung Cancer Cells. Mol. Cancer Res. 2019, 17, 499–507. [Google Scholar] [CrossRef] [PubMed]
- Suda, K.; Mitsudomi, T. Drug Tolerance to EGFR Tyrosine Kinase Inhibitors in Lung Cancers with EGFR Mutations. Cells 2021, 10, 1590. [Google Scholar] [CrossRef]
- Sharma, S.V.; Lee, D.Y.; Li, B.; Quinlan, M.P.; Takahashi, F.; Maheswaran, S.; McDermott, U.; Azizian, N.; Zou, L.; Fischbach, M.A.; et al. A Chromatin-Mediated Reversible Drug-Tolerant State in Cancer Cell Subpopulations. Cell 2010, 141, 69–80. [Google Scholar] [CrossRef]
- Raoof, S.; Mulford, I.J.; Frisco-Cabanos, H.; Nangia, V.; Timonina, D.; Labrot, E.; Hafeez, N.; Bilton, S.J.; Drier, Y.; Ji, F.; et al. Targeting FGFR Overcomes EMT-Mediated Resistance in EGFR Mutant Non-Small Cell Lung Cancer. Oncogene 2019, 38, 6399–6413. [Google Scholar] [CrossRef] [PubMed]
- Ramirez, M.; Rajaram, S.; Steininger, R.J.; Osipchuk, D.; Roth, M.A.; Morinishi, L.S.; Evans, L.; Ji, W.; Hsu, C.-H.; Thurley, K.; et al. Diverse Drug-Resistance Mechanisms Can Emerge from Drug-Tolerant Cancer Persister Cells. Nat. Commun. 2016, 7, 10690. [Google Scholar] [CrossRef] [PubMed]
- Iwaki, T.; Urano, T.; Umemura, K. PAI-1, Progress in Understanding the Clinical Problem and Its Aetiology. Br. J. Haematol. 2012, 157, 291–298. [Google Scholar] [CrossRef]
- Fang, H.; Placencio, V.R.; DeClerck, Y.A. Protumorigenic Activity of Plasminogen Activator Inhibitor-1 through an Antiapoptotic Function. J. Natl. Cancer Inst. 2012, 104, 1470–1484. [Google Scholar] [CrossRef] [PubMed]
- Rømer, M.U.; Larsen, L.; Offenberg, H.; Brünner, N.; Lademann, U.A. Plasminogen Activator Inhibitor 1 Protects Fibrosarcoma Cells from Etoposide-Induced Apoptosis through Activation of the PI3K/Akt Cell Survival Pathway. Neoplasia 2008, 10, 1083–1091. [Google Scholar] [CrossRef] [PubMed]
- Masuda, T.; Nakashima, T.; Namba, M.; Yamaguchi, K.; Sakamoto, S.; Horimasu, Y.; Miyamoto, S.; Iwamoto, H.; Fujitaka, K.; Miyata, Y.; et al. Inhibition of PAI-1 Limits Chemotherapy Resistance in Lung Cancer through Suppressing Myofibroblast Characteristics of Cancer-Associated Fibroblasts. J. Cell Mol. Med. 2019, 23, 2984–2994. [Google Scholar] [CrossRef]
- Kang, J.; Kim, W.; Kwon, T.; Youn, H.; Kim, J.S.; Youn, B. Plasminogen Activator Inhibitor-1 Enhances Radioresistance and Aggressiveness of Non-Small Cell Lung Cancer Cells. Oncotarget 2016, 7, 23961–23974. [Google Scholar] [CrossRef]
- Varghese, F.; Bukhari, A.B.; Malhotra, R.; De, A. IHC Profiler: An Open Source Plugin for the Quantitative Evaluation and Automated Scoring of Immunohistochemistry Images of Human Tissue Samples. PLoS ONE 2014, 9, e96801. [Google Scholar] [CrossRef]
- Murakami, A.; Takahashi, F.; Nurwidya, F.; Kobayashi, I.; Minakata, K.; Hashimoto, M.; Nara, T.; Kato, M.; Tajima, K.; Shimada, N.; et al. Hypoxia Increases Gefitinib-Resistant Lung Cancer Stem Cells through the Activation of Insulin-like Growth Factor 1 Receptor. PLoS ONE 2014, 9, e86459. [Google Scholar] [CrossRef]
- Ge, S.X.; Son, E.W.; Yao, R. IDEP: An Integrated Web Application for Differential Expression and Pathway Analysis of RNA-Seq Data. BMC Bioinform. 2018, 19, 534. [Google Scholar] [CrossRef] [PubMed]
- Kanehisa, M.; Furumichi, M.; Sato, Y.; Ishiguro-Watanabe, M.; Tanabe, M. KEGG: Integrating Viruses and Cellular Organisms. Nucleic. Acids Res. 2021, 49, D545–D551. [Google Scholar] [CrossRef] [PubMed]
- Takayama, Y.; Hattori, N.; Hamada, H.; Masuda, T.; Omori, K.; Akita, S.; Iwamoto, H.; Fujitaka, K.; Kohno, N. Inhibition of PAI-1 Limits Tumor Angiogenesis Regardless of Angiogenic Stimuli in Malignant Pleural Mesothelioma. Cancer Res. 2016, 76, 3285–3294. [Google Scholar] [CrossRef] [PubMed]
- Masuda, T.; Hattori, N.; Senoo, T.; Akita, S.; Ishikawa, N.; Fujitaka, K.; Haruta, Y.; Murai, H.; Kohno, N. SK-216, an Inhibitor of Plasminogen Activator Inhibitor-1, Limits Tumor Progression and Angiogenesis. Mol. Cancer Ther. 2013, 12, 2378–2388. [Google Scholar] [CrossRef]
- Pappot, H.; Pedersen, A.N.; Brünner, N.; Christensen, I.J. The Complex between Urokinase (UPA) and Its Type-1 Inhibitor (PAI-1) in Pulmonary Adenocarcinoma: Relation to Prognosis. Lung Cancer 2006, 51, 193–200. [Google Scholar] [CrossRef] [PubMed]
- Foekens, J.A.; Peters, H.A.; Look, M.P.; Portengen, H.; Schmitt, M.; Kramer, M.D.; Brünner, N.; Jänicke, F.; Meijer-van Gelder, M.E.; Henzen-Logmans, S.C.; et al. The Urokinase System of Plasminogen Activation and Prognosis in 2780 Breast Cancer Patients. Cancer Res. 2000, 60, 636–643. [Google Scholar]
- Sakakibara, T.; Hibi, K.; Kodera, Y.; Ito, K.; Akiyama, S.; Nakao, A. Plasminogen Activator Inhibitor-1 as a Potential Marker for the Malignancy of Esophageal Squamous Cell Carcinoma. Clin. Cancer Res. 2004, 10, 1375–1378. [Google Scholar] [CrossRef]
- Heiss, M.M.; Allgayer, H.; Gruetzner, K.U.; Babic, R.; Jauch, K.W.; Schildberg, F.W. Clinical Value of Extended Biologic Staging by Bone Marrow Micrometastases and Tumor-Associated Proteases in Gastric Cancer. Ann. Surg. 1997, 226, 736–744, discussion 744–745. [Google Scholar] [CrossRef]
- Ohba, K.; Miyata, Y.; Kanda, S.; Koga, S.; Hayashi, T.; Kanetake, H. Expression of Urokinase-Type Plasminogen Activator, Urokinase-Type Plasminogen Activator Receptor and Plasminogen Activator Inhibitors in Patients with Renal Cell Carcinoma: Correlation with Tumor Associated Macrophage and Prognosis. J. Urol. 2005, 174, 461–465. [Google Scholar] [CrossRef]
- Ren, J.; Chen, Y.; Song, H.; Chen, L.; Wang, R. Inhibition of ZEB1 Reverses EMT and Chemoresistance in Docetaxel-Resistant Human Lung Adenocarcinoma Cell Line. J. Cell Biochem. 2013, 114, 1395–1403. [Google Scholar] [CrossRef]
- Yang, J.; Antin, P.; Berx, G.; Blanpain, C.; Brabletz, T.; Bronner, M.; Campbell, K.; Cano, A.; Casanova, J.; Christofori, G.; et al. Guidelines and Definitions for Research on Epithelial–Mesenchymal Transition. Nat. Rev. Mol. Cell Biol. 2020, 21, 341–352. [Google Scholar] [CrossRef]
- Senoo, T.; Hattori, N.; Tanimoto, T.; Furonaka, M.; Ishikawa, N.; Fujitaka, K.; Haruta, Y.; Murai, H.; Yokoyama, A.; Kohno, N. Suppression of Plasminogen Activator Inhibitor-1 by RNA Interference Attenuates Pulmonary Fibrosis. Thorax 2010, 65, 334–340. [Google Scholar] [CrossRef]
- Omori, K.; Hattori, N.; Senoo, T.; Takayama, Y.; Masuda, T.; Nakashima, T.; Iwamoto, H.; Fujitaka, K.; Hamada, H.; Kohno, N. Inhibition of Plasminogen Activator Inhibitor-1 Attenuates Transforming Growth Factor-β-Dependent Epithelial Mesenchymal Transition and Differentiation of Fibroblasts to Myofibroblasts. PLoS ONE 2016, 11, e0148969. [Google Scholar] [CrossRef]
- Yang, X.H.; Flores, L.M.; Li, Q.; Zhou, P.; Xu, F.; Krop, I.E.; Hemler, M.E. Disruption of Laminin-Integrin-CD151-Focal Adhesion Kinase Axis Sensitizes Breast Cancer Cells to ErbB2 Antagonists. Cancer Res. 2010, 70, 2256–2263. [Google Scholar] [CrossRef]
- Huang, C.; Park, C.C.; Hilsenbeck, S.G.; Ward, R.; Rimawi, M.F.; Wang, Y.-C.; Shou, J.; Bissell, M.J.; Osborne, C.K.; Schiff, R. Β1 Integrin Mediates an Alternative Survival Pathway in Breast Cancer Cells Resistant to Lapatinib. Breast Cancer Res. 2011, 13, R84. [Google Scholar] [CrossRef]
- Guo, W.; Giancotti, F.G. Integrin Signalling during Tumour Progression. Nat. Rev. Mol. Cell Biol. 2004, 5, 816–826. [Google Scholar] [CrossRef]
- Wang, C.; Wang, T.; Lv, D.; Li, L.; Yue, J.; Chen, H.-Z.; Xu, L. Acquired Resistance to EGFR TKIs Mediated by TGFβ1/Integrin Β3 Signaling in EGFR-Mutant Lung Cancer. Mol. Cancer Ther. 2019, 18, 2357–2367. [Google Scholar] [CrossRef]
- Ciambrone, G.J.; McKeown-Longo, P.J. Plasminogen Activator Inhibitor Type I Stabilizes Vitronectin-Dependent Adhesions in HT-1080 Cells. J. Cell Biol. 1990, 111, 2183–2195. [Google Scholar] [CrossRef]
- Burridge, K.; Fath, K.; Kelly, T.; Nuckolls, G.; Turner, C. Focal Adhesions: Transmembrane Junctions between the Extracellular Matrix and the Cytoskeleton. Annu. Rev. Cell Biol. 1988, 4, 487–525. [Google Scholar] [CrossRef]
- Ghosh, A.K.; Vaughan, D.E. PAI-1 in Tissue Fibrosis. J. Cell Physiol. 2012, 227, 493–507. [Google Scholar] [CrossRef]
- Ge, X.; Zhang, Y.; Huang, F.; Wu, Y.; Pang, J.; Li, X.; Fan, F.; Liu, H.; Li, S. EGFR Tyrosine Kinase Inhibitor Almonertinib Induces Apoptosis and Autophagy Mediated by Reactive Oxygen Species in Non-Small Cell Lung Cancer Cells. Hum. Exp. Toxicol. 2021, 40, S49–S62. [Google Scholar] [CrossRef]
- Swiatkowska, M.; Szemraj, J.; Al-Nedawi, K.N.I.; Pawłowska, Z. Reactive Oxygen Species Upregulate Expression of PAI-1 in Endothelial Cells. Cell Mol. Biol. Lett. 2002, 7, 1065–1071. [Google Scholar]
- Arasada, R.R.; Shilo, K.; Yamada, T.; Zhang, J.; Yano, S.; Ghanem, R.; Wang, W.; Takeuchi, S.; Fukuda, K.; Katakami, N.; et al. Notch3-Dependent β-Catenin Signaling Mediates EGFR TKI Drug Persistence in EGFR Mutant NSCLC. Nat. Commun. 2018, 9, 3198. [Google Scholar] [CrossRef]
| Patient 1 | Patient 2 | Patient 3 | Patient 4 | Patient 5 | |
|---|---|---|---|---|---|
| Sex | F | M | M | M | M |
| Age (years) | 66 | 65 | 52 | 66 | 63 |
| Histological classification | ad | ad | ad | ad | ad |
| Clinical stage at diagnosis | IIIA | IIA | IIB | I | IIIB |
| EGFR mutation category | Exon 19 del | Exon 19 del | Exon 19 del | Exon 21 L858R | Exon 18 D719C |
| Tissue collection site (at diagnosis) | Lung | Lung | Lung | Lung | Lung |
| Method of tissue collection (at diagnosis) | Surgery | Surgery | Surgery | Surgery | Surgery |
| Tissue collection site (second biopsy) | Lung | Lung | Lung | Bone | Lymph node |
| Method of tissue collection (second biopsy) | Surgery | Surgery | Surgery | CT | Surgery |
| Presence of T790M mutation (second biopsy) | Yes | Yes | Yes | No | No |
| EGFR-TKI used before the second biopsy | Gefitinib | Gefitinib | Erlotinib | Afatinib | Afatinib |
| EGFR-TKI used after the second biopsy | Osimertinib | Osimertinib | Osimertinib | None | None |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Tokumo, K.; Masuda, T.; Nakashima, T.; Namba, M.; Yamaguchi, K.; Sakamoto, S.; Horimasu, Y.; Miyamoto, S.; Iwamoto, H.; Fujitaka, K.; et al. Association between Plasminogen Activator Inhibitor-1 and Osimertinib Tolerance in EGFR-Mutated Lung Cancer via Epithelial–Mesenchymal Transition. Cancers 2023, 15, 1092. https://doi.org/10.3390/cancers15041092
Tokumo K, Masuda T, Nakashima T, Namba M, Yamaguchi K, Sakamoto S, Horimasu Y, Miyamoto S, Iwamoto H, Fujitaka K, et al. Association between Plasminogen Activator Inhibitor-1 and Osimertinib Tolerance in EGFR-Mutated Lung Cancer via Epithelial–Mesenchymal Transition. Cancers. 2023; 15(4):1092. https://doi.org/10.3390/cancers15041092
Chicago/Turabian StyleTokumo, Kentaro, Takeshi Masuda, Taku Nakashima, Masashi Namba, Kakuhiro Yamaguchi, Shinjiro Sakamoto, Yasushi Horimasu, Shintaro Miyamoto, Hiroshi Iwamoto, Kazunori Fujitaka, and et al. 2023. "Association between Plasminogen Activator Inhibitor-1 and Osimertinib Tolerance in EGFR-Mutated Lung Cancer via Epithelial–Mesenchymal Transition" Cancers 15, no. 4: 1092. https://doi.org/10.3390/cancers15041092
APA StyleTokumo, K., Masuda, T., Nakashima, T., Namba, M., Yamaguchi, K., Sakamoto, S., Horimasu, Y., Miyamoto, S., Iwamoto, H., Fujitaka, K., Miyata, Y., Okada, M., Hamada, H., & Hattori, N. (2023). Association between Plasminogen Activator Inhibitor-1 and Osimertinib Tolerance in EGFR-Mutated Lung Cancer via Epithelial–Mesenchymal Transition. Cancers, 15(4), 1092. https://doi.org/10.3390/cancers15041092






