Combination, Modulation and Interplay of Modern Radiotherapy with the Tumor Microenvironment and Targeted Therapies in Pancreatic Cancer: Which Candidates to Boost Radiotherapy?
Abstract
Simple Summary
Abstract
1. Introduction
2. Radiotherapy Modalities and Perspectives in PDAC
2.1. Hypofractionated CRT
2.2. Hypofractionated Ablative RT (HFA-RT)
2.3. Isotoxic High-Dose SBRT (iHD-SBRT) and MR-Guided Stereotactic Ablative RT (SMART)
2.4. Intraoperative RT (IORT) and FLASH
2.5. Low-Dose Rate RT (LDR-RT)
2.6. Spatially Fractionated RT (SFRT)
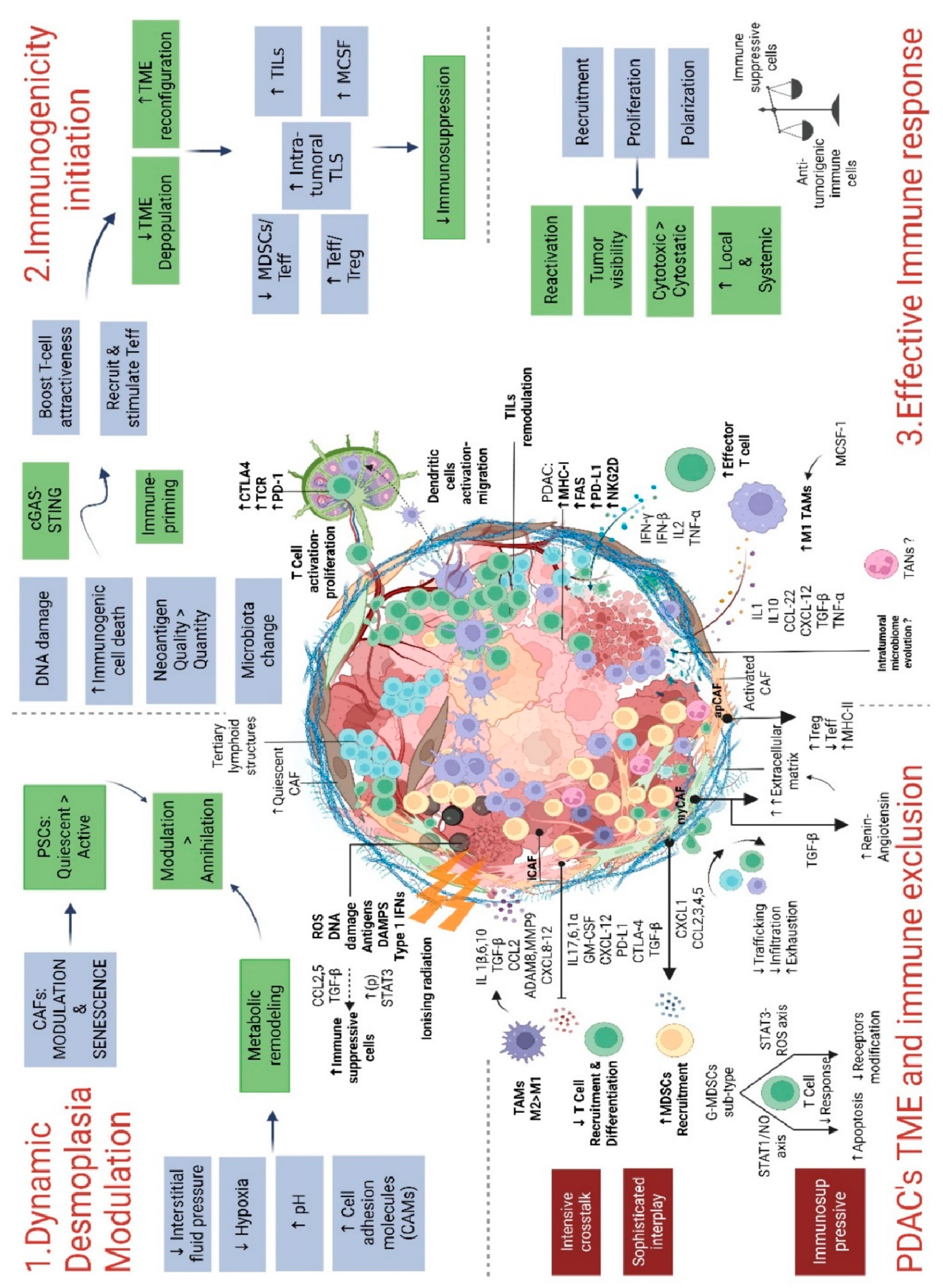
3. The Pancreatic Tumor Microenvironment and Its Modulation by RT
3.1. Stroma and Cancer-Associated Fibroblasts (CAFs)
Effects of RT
3.2. PDAC Vascular System
Effects of RT
3.3. Immune Cells
3.3.1. Tumor-Associated Macrophages (TAMs)
Effects of RT
3.3.2. Myeloid-Derived Suppressor Cells (MDSCs)
Effects of RT
3.3.3. Tumor-Associated Neutrophils (TANs)
Effect of RT
3.3.4. Tumor-Infiltrating Lymphocytes (TILs)
Effects of RT
3.4. Others
3.4.1. GMP-AMP synthase (cGAS)-stimulator of interferon genes (STING) pathway
3.4.2. Microbiome
4. Clinical Targeted Treatment Combinations including RT and Perspectives
4.1. Combination of Radiotherapy and Targeted Therapy in PDAC (tRT)
4.2. Selected Promising Clinical Perspectives of tRT in PDAC
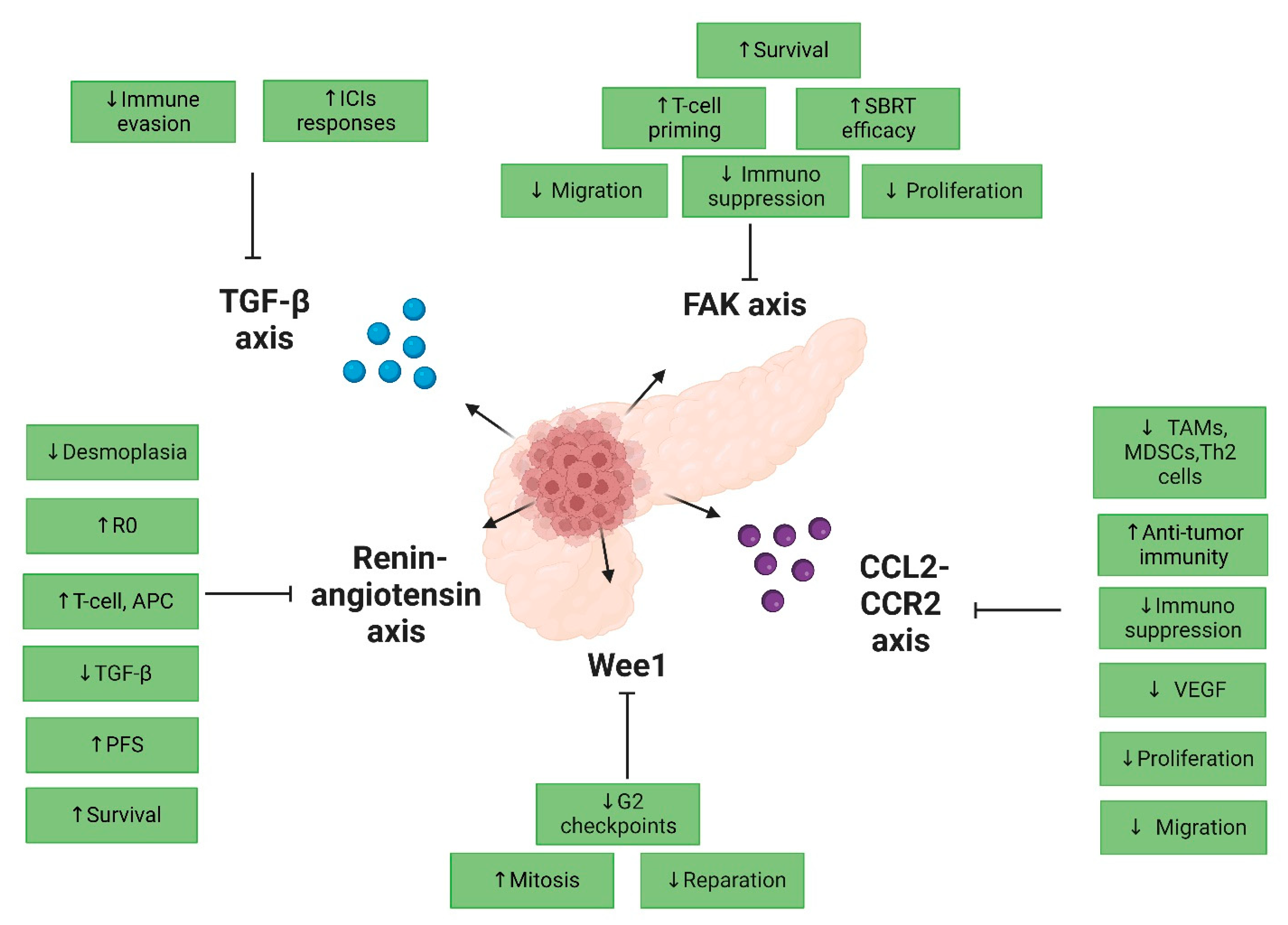
4.2.1. Targeting the CCL2 Axis
4.2.2. Targeting the TGF-β Axis
4.2.3. Targeting the Angiotensin Axis
4.2.4. Targeting the DNA Damage Response Axis
4.2.5. Targeting the FAK Axis
5. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Sung, H.; Ferlay, J.; Siegel, R.L.; Laversanne, M.; Soerjomataram, I.; Jemal, A.; Bray, F. Global Cancer Statistics 2020: GLOBOCAN Estimates of Incidence and Mortality Worldwide for 36 Cancers in 185 Countries. CA Cancer J. Clin. 2021, 71, 209–249. [Google Scholar] [CrossRef]
- Siegel, R.L.; Miller, K.D.; Fuchs, H.E.; Jemal, A. Cancer Statistics, 2021. CA Cancer J. Clin. 2021, 71, 7–33. [Google Scholar] [CrossRef] [PubMed]
- Christenson, E.S.; Jaffee, E.; Azad, N.S. Current and emerging therapies for patients with advanced pancreatic ductal adenocarcinoma: A bright future. Lancet Oncol. 2020, 21, e135–e145. [Google Scholar] [CrossRef] [PubMed]
- Tempero, M.A.; Malafa, M.P.; Al-Hawary, M. Pancreatic Adenocarcinoma. In NCCN Guidelines, 1st ed.; 2022; Available online: http://www.nccn.org/professionals/physician_gls/pdf/pancreatic.pdf (accessed on 20 July 2022).
- Wang, Y.; Deng, W.; Li, N.; Neri, S.; Sharma, A.; Jiang, W.; Lin, S.H. Combining Immunotherapy and Radiotherapy for Cancer Treatment: Current Challenges and Future Directions. Front. Pharmacol. 2018, 9, 185. [Google Scholar] [CrossRef] [PubMed]
- Schizas, D.; Charalampakis, N.; Kole, C.; Economopoulou, P.; Koustas, E.; Gkotsis, E.; Ziogas, D.; Psyrri, A.; Karamouzis, M.V. Immunotherapy for pancreatic cancer: A 2020 update. Cancer Treat. Rev. 2020, 86, 102016. [Google Scholar] [CrossRef] [PubMed]
- Neoptolemos, J.P.; Kleeff, J.; Michl, P.; Costello, E.; Greenhalf, W.; Palmer, D.H. Therapeutic developments in pancreatic cancer: Current and future perspectives. Nat. Rev. Gastroenterol. Hepatol. 2018, 15, 333–348. [Google Scholar] [CrossRef]
- Cellini, F.; Arcelli, A.; Simoni, N.; Caravatta, L.; Buwenge, M.; Calabrese, A.; Brunetti, O.; Genovesi, D.; Mazzarotto, R.; Deodato, F.; et al. Basics and Frontiers on Pancreatic Cancer for Radiation Oncology: Target Delineation, SBRT, SIB technique, MRgRT, Particle Therapy, Immunotherapy and Clinical Guidelines. Cancers 2020, 12, 1729. [Google Scholar] [CrossRef]
- Iacobuzio-Donahue, C.A.; Fu, B.; Yachida, S.; Luo, M.; Abe, H.; Henderson, C.M.; Vilardell, F.; Wang, Z.; Keller, J.W.; Banerjee, P.; et al. DPC4 Gene Status of the Primary Carcinoma Correlates with Patterns of Failure in Patients with Pancreatic Cancer. J. Clin. Oncol. 2009, 27, 1806–1813. [Google Scholar] [CrossRef]
- Badiyan, S.N.; Molitoris, J.K.; Chuong, M.D.; Regine, W.F.; Kaiser, A. The Role of Radiation Therapy for Pancreatic Cancer in the Adjuvant and Neoadjuvant Settings. Surg. Oncol. Clin. North Am. 2017, 26, 431–453. [Google Scholar] [CrossRef]
- Bouchart, C.; Navez, J.; Closset, J.; Hendlisz, A.; Van Gestel, D.; Moretti, L.; Van Laethem, J.L. Novel strategies using modern radiotherapy to improve pancreatic cancer outcomes: Toward a new standard? Ther. Adv. Med. Oncol. 2020, 12, 1758835920936093. [Google Scholar] [CrossRef]
- Skorupan, N.; Dominguez, M.P.; Ricci, S.L.; Alewine, C. Clinical Strategies Targeting the Tumor Microenvironment of Pancreatic Ductal Adenocarcinoma. Cancers 2022, 14, 4209. [Google Scholar] [CrossRef]
- Hu, Z.I.; Shia, J.; Stadler, Z.K.; Varghese, A.M.; Capanu, M.; Salo-Mullen, E.; Lowery, M.A.; Diaz, L.A.; Mandelker, D.; Yu, K.H.; et al. Evaluating Mismatch Repair Deficiency in Pancreatic Adenocarcinoma: Challenges and Recommendations. Clin. Cancer Res. 2018, 24, 1326–1336. [Google Scholar] [CrossRef] [PubMed]
- Lawlor, R.T.; Mattiolo, P.; Mafficini, A.; Hong, S.-M.; Piredda, M.L.; Taormina, S.V.; Malleo, G.; Marchegiani, G.; Pea, A.; Salvia, R.; et al. Tumor Mutational Burden as a Potential Biomarker for Immunotherapy in Pancreatic Cancer: Systematic Review and Still-Open Questions. Cancers 2021, 13, 3119. [Google Scholar] [CrossRef]
- Principe, D.R.; Korc, M.; Kamath, S.D.; Munshi, H.G.; Rana, A. Trials and tribulations of pancreatic cancer immunotherapy. Cancer Lett. 2021, 504, 1–14. [Google Scholar] [CrossRef]
- Kuznetsova, A.; Popova, O.; Panchenkov, D.; Dyuzheva, T.; Ivanov, A. Pancreatic ductal adenocarcinoma: Tumor microenvironment and problems in the development of novel therapeutic strategies. Clin. Exp. Med. 2022, 13, 1–25. [Google Scholar] [CrossRef]
- Lutz, E.; Yeo, C.J.; Lillemoe, K.D.; Bieddrzycki, B.; Kobrin, B.; Herman, J.; Sugar, J.; Piantadosi, S.; Cameron, L.J.; Solt, S.; et al. A lethally irradiated allogeneic granulocyte-macrophage colony stimulating factor-secreting tumor vaccine for pancreatic adenocarcinoma. A Phase II trial of safety, efficacy, and immune activation. Ann. Surg. 2011, 253, 328–335. [Google Scholar]
- Picozzi, V.J.; Abrams, R.A.; Decker, P.A.; Traverso, W.; O’Reilly, E.M.; Greeno, E.; Martin, R.C.; Wilfong, L.S.; Rothenberg, M.L.; Posner, M.C.; et al. Multicenter phase II trial of adjuvant therapy for resected pancreatic cancer using cisplatin, 5-fluorouracil, and interferon-alfa-2b-based chemoradiation: ACOSOG Trial Z05031. Ann. Oncol. 2011, 22, 348–354. [Google Scholar] [CrossRef]
- Crane, C.H.; Varadhachary, G.R.; Yordy, J.S.; Staerkel, G.A.; Javle, M.M.; Safran, H.; Haque, W.; Hobbs, B.D.; Krishnan, S.; Fleming, J.B.; et al. Phase II trial of cetuximab, gemcitabine, and oxaliplatin followed by chemoradiation with cetuximab for locally advanced (T4) pancreatic adenocarcinoma: Correlation of Smad4(Dpc4) immunostaining with pattern of disease progression. J. Clin. Oncol. 2011, 29, 3037–3043. [Google Scholar] [CrossRef]
- Hardacre, J.M.; Mulcahy, M.; Small, W.; Talamonti, M.; Obel, J.; Krishnamurthi, S.; Rocha-Lima, C.S.; Lenz, H.J.; Chiorean, E.G. Addition of algenpantucel-L immunotherapy to standard adjuvant therapy for pancreatic cancer: A phase 2 study. J. Gastrointest Surg. 2013, 17, 94–100. [Google Scholar] [CrossRef]
- Chan, E.; Arlinghaus, L.R.; Cardin, D.B.; Goff, L.; Berlin, J.D.; Parikh, A.; Abramson, R.G.; Yankeelov, T.E.; Hiebert, S.; Merchant, N.; et al. Phase I trial of vorinostat added to chemoradiation with capecitabine in pancreatic cancer. Radiother. Oncol. 2016, 119, 312–318. [Google Scholar] [CrossRef]
- Brar, G.; Xie, C.; Floudas, C.; Morelli, M.P.; Fioravanti, S.; Walker, M.; Mabry-Hrones, D.; Jones, J.C.; Greten, T.F. Immune checpoint inhibition (ICI) in combination with SBRT in patients with advanced pancreatic adenocarcinoma (aPDAC). JCO 2019, 37, 192. [Google Scholar] [CrossRef]
- Cuneo, K.C.; Morgan, M.A.; Sahai, V.; Schipper, M.J.; Parsels, L.A.; Parsels, J.D.; Devasia, T.; Al-Hawaray, M.; Cho, C.S.; Nathan, H.; et al. Dose Escalation Trial of the Wee1 Inhibitor Adavosertib (AZD1775) in Combination With Gemcitabine and Radiation for Patients With Locally Advanced Pancreatic Cancer. J. Clin. Oncol. 2019, 37, 2643–2650. [Google Scholar] [CrossRef] [PubMed]
- Murphy, J.E.; Wo, J.Y.; Ryan, D.P.; Clark, J.W.; Jiang, W.; Yeap, B.Y.; Drapek, L.C.; Ly., L.; Baglini, C.V.; Blaszkowsky, L.S.; et al. Total Neoadjuvant Therapy With FOLFIRINOX in Combination With Losartan Followed by Chemoradiotherapy for Locally Advanced Pancreatic Cancer: A Phase 2 Clinical Trial. JAMA Oncol. 2019, 5, 1020–1027. [Google Scholar] [CrossRef]
- Lin, C.; Verma, V.; Lazenby, A.; Ly, Q.P.; Berim, L.D.; Schwarz, J.K.; Madiyalakan, M.; Nicodemus, C.F.; Hollingsworth, M.A.; Meza, J.L.; et al. Phase I/II Trial of Neoadjuvant Oregovomab-based Chemoimmunotherapy Followed by Stereotactic Body Radiotherapy and Nelfinavir For Locally Advanced Pancreatic Adenocarcinoma. Am. J. Clin. Oncol. 2019, 42, 755–760. [Google Scholar] [CrossRef] [PubMed]
- Tuli, R.; Nissen, N.; Lo, S.; Tighiouart, M.; Placencio, V.; Hendifar, A. Abstract B58: A phase I/II study of durvalumab and stereotactic radiotherapy in locally advanced pancreatic cancer. Cancer Res. 2019, 79, B58. [Google Scholar] [CrossRef]
- Xie, C.; Duffy, A.G.; Brar, G.; Fioravanti, S.; Mabry-Hrones, D.; Walker, M.; Bonilla, C.M.; Wood, B.J.; Citrin, D.E.; Ramirez, E.M.G.; et al. Immune Checkpoint Blockade in Combination with Stereotactic Body Radiotherapy in Patients with Metastatic Pancreatic Ductal Adenocarcinoma. Clin. Cancer Res. 2020, 26, 2318–2326. [Google Scholar] [CrossRef]
- Parikh, A.R.; Szabolcs, A.; Allen, J.N.; Clark, J.W.; Wo, J.Y.; Raabe, M.; Thel, H.; Hoyos, D.; Mehta, A.; Arshad, S.; et al. Radiation therapy enhances immunotherapy response in microsatellite stable colorectal and pancreatic adenocarcinoma in a phase II trial. Nat. Cancer 2021, 2, 1124–1135. [Google Scholar] [CrossRef]
- Poklepovic, A.S.; Fields, E.C.; Bandyopadhyay, D.; Tombes, M.B.; Kmieciak, M.; McGuire, W.P.; Gordon, S.W.; Kaplan, B.J.; Myers, J.L.; Matin, K.; et al. A phase 1 study of neoadjuvant chemotherapy followed by concurrent chemoradiation with gemcitabine, sorafenib, and vorinostat in pancreatic cancer. J. Clin. Oncol. 2021, 39, e16268. [Google Scholar] [CrossRef]
- Rahma, O.E.; Katz, M.H.G.; Wolpin, B.M.; Dias-Costa, A.; Nowak, J.; Rodig, S.J.; Dougan, S.; Bekaii-Saab, T.S.; Stucky, C.-C.H.; Elias, R.; et al. Randomized multicenter phase Ib/II study of neoadjuvant chemoradiation therapy (CRT) alone or in combination with pembrolizumab in patients with resectable or borderline resectable pancreatic cancer. J. Clin. Oncol. 2021, 39, 4128. [Google Scholar] [CrossRef]
- Lee, V.; Ding, D.; Rodriguez, C.; Onners, B.; Narang, A.; Meyer, J.; Herman, J.M.; Hacker-Prietz, A.; Burkhart, R.A.; Burns, W.; et al. A phase 2 study of cyclophosphamide (CY), GVAX, pembrolizumab (Pembro), and stereotactic body radiation (SBRT) in patients (pts) with locally advanced pancreas cancer (LAPC). J. Clin. Oncol. 2021, 39, 4134. [Google Scholar] [CrossRef]
- Halfdanarson, T.R.; Foster, N.R.; Kim, G.P.; Haddock, M.G.; Dakhil, S.R.; Behrens, R.J.; Alberts, S.R. N064A (Alliance): Phase II Study of Panitumumab, Chemotherapy, and External Beam Radiation in Patients with Locally Advanced Pancreatic Adenocarcinoma. Oncologist 2022, 27, 534-e546. [Google Scholar] [CrossRef] [PubMed]
- Zhu, X.; Cao, Y.; Liu, W.; Ju, W.; Ju, X.; Zhao, X.; Jiang, L.; Ye, Y.; Jin, G.; Zhang, H. Stereotactic body radiotherapy plus pembrolizumab and trametinib versus stereotactic body radiotherapy plus gemcitabine for locally recurrent pancreatic cancer after surgical resection: An open-label, randomised, controlled, phase 2 trial. Lancet Oncol. 2022, 23, e105–e115. [Google Scholar] [CrossRef]
- Chen, I.M.; Johansen, J.S.; Theile, S.; Hjaltelin, J.X.; Novitski, S.I.; Brunak, S.; Hasselby, J.P.; Willemoe, G.L.; Lorentzen, T.; Madsen, K.; et al. Randomized Phase II Study of Nivolumab With or Without Ipilimumab Combined With Stereotactic Body Radiotherapy for Refractory Metastatic Pancreatic Cancer (CheckPAC). J. Clin. Oncol. 2022, 40, 3180–3189. [Google Scholar] [CrossRef] [PubMed]
- Liermann, J.; Munter, M.; Naumann, P.; Abdollahi, A.; Krempien, R.; Debus, J. Cetuximab, gemcitabine and radiotherapy in locally advanced pancreatic cancer: Long-term results of the randomized controlled phase II PARC trial. Clin. Transl. Radiat. Oncol. 2022, 34, 15–22. [Google Scholar] [CrossRef] [PubMed]
- Hewitt, D.B.; Nissen, N.; Hatoum, H.; Musher, B.; Seng, J.; Coveler, A.L.; Al-Rajabi, R.; Yeo, C.J.; Leiby, B.; Banks, J.; et al. A Phase 3 Randomized Clinical Trial of Chemotherapy With or Without Algenpantucel-L (HyperAcute-Pancreas) Immunother-apy in Subjects With Borderline Resectable or Locally Advanced Unresectable Pancreatic Cancer. Ann. Surg. 2022, 275, 45–53. [Google Scholar] [CrossRef]
- Palta, M.; Godfrey, D.; Goodman, K.A.; Hoffe, S.; Dawson, L.A.; Dessert, D.; Hall, W.A.; Herman, J.M.; Khorana, A.A.; Merchant, N.; et al. Radiation Therapy for Pancreatic Cancer: Executive Summary of an ASTRO Clinical Practice Guideline. Pract. Radiat. Oncol. 2019, 9, 322–332. [Google Scholar] [CrossRef]
- Khorana, A.A.; McKernin, S.E.; Berlin, J.; Hong, T.S.; Maitra, A.; Moravek, C.; Mumber, M.; Schulick, R.; Zeh, H.J.; Katz, M.H. Potentially Curable Pancreatic Adenocarcinoma: ASCO Clinical Practice Guideline Update. J. Clin. Oncol. 2019, 37, 2082–2088. [Google Scholar] [CrossRef]
- Fietkau, R.; Ghadimi, M.; Grützmann, R.; A Wittel, U.; Jacobasch, L.; Uhl, W.; Croner, R.S.; Bechstein, W.O.; Neumann, U.P.; Waldschmidt, D.; et al. Randomized phase III trial of induction chemotherapy followed by chemoradiotherapy or chemotherapy alone for nonresectable locally advanced pancreatic cancer: First results of the CONKO-007 trial. J. Clin. Oncol. 2022, 40, 4008. [Google Scholar] [CrossRef]
- Hammel, P.; Huguet, F.; van Laethem, J.L.; Goldstein, D.; Glimelius, B.; Artru, P.; Borbath, I.; Bouché, O.; Shannon, J.; Louvet, C.; et al. Effect of Chemoradiotherapy vs Chemotherapy on Survival in Patients With Locally Advanced Pancreatic Cancer Controlled After 4 Months of Gemcitabine With or Without Erlotinib: The LAP07 Randomized Clinical Trial. JAMA 2016, 315, 1844–1853. [Google Scholar] [CrossRef]
- Bouchart, C.; Engelholm, J.-L.; Closset, J.; Navez, J.; Loi, P.; Gökburun, Y.; De Grez, T.; Mans, L.; Hendlisz, A.; Bali, M.A.; et al. Isotoxic high-dose stereotactic body radiotherapy integrated in a total multimodal neoadjuvant strategy for the treatment of localized pancreatic ductal adenocarcinoma. Ther. Adv. Med. Oncol. 2021, 13, 17588359211045860. [Google Scholar] [CrossRef]
- Parikh, P.; Lee, P.; Low, D.; Kim, J.; Mittauer, K.; Bassetti, M.; Glide-Hurst, C.; Raldow, A.; Yang, Y.; Portelance, L.; et al. Stereotactic MR-Guided On-Table Adaptive Radiation Therapy (SMART) for Patients with Borderline or Locally Advanced Pancreatic Cancer: Primary Endpoint Outcomes of a Prospective Phase II Multi-Center International Trial. Int. J. Radiat. Oncol. 2022, 114, 1062–1063. [Google Scholar] [CrossRef]
- Versteijne, E.; van Dam, J.L.; Suker, M.; Janssen, Q.P.; Groothuis, K.; Akkermans-Vogelaar, J.M.; Besselink, M.G.; Bonsing, B.A.; Buijsen, J.; Busch, O.R.; et al. Neoadjuvant Chemoradiotherapy Versus Upfront Surgery for Resectable and Borderline Resectable Pancreatic Cancer: Long-Term Results of the Dutch Randomized PREOPANC Trial. J. Clin. Oncol. 2022, 40, 1220–1230. [Google Scholar] [CrossRef] [PubMed]
- Reyngold, M.; O’Reilly, E.M.; Varghese, A.M.; Fiasconaro, M.; Zinovoy, M.; Romesser, P.B.; Wu, A.; Hajj, C.; Cuaron, J.J.; Tuli, R.; et al. Association of Ablative Radiation Therapy With Survival Among Patients With Inoperable Pancreatic Cancer. JAMA Oncol. 2021, 7, 735. [Google Scholar] [CrossRef]
- Arcelli, A.; Guido, A.; Buwenge, M.; Simoni, N.; Mazzarotto, R.; Macchia, G.; Deodato, F.; Cilla, S.; Bonomo, P.; Scotti, V.; et al. Higher Biologically Effective Dose Predicts Survival in SBRT of Pancreatic Cancer: A Multicentric Analysis (PAULA-1). Anticancer. Res. 2019, 40, 465–472. [Google Scholar] [CrossRef]
- Krishnan, S.; Chadha, A.S.; Suh, Y.; Chen, H.-C.; Rao, A.; Das, P.; Minsky, B.D.; Mahmood, U.; Delclos, M.E.; Sawakuchi, G.O.; et al. Focal Radiation Therapy Dose Escalation Improves Overall Survival in Locally Advanced Pancreatic Cancer Patients Receiving Induction Chemotherapy and Consolidative Chemoradiation. Int. J. Radiat. Oncol. 2015, 94, 755–765. [Google Scholar] [CrossRef] [PubMed]
- Rudra, S.; Jiang, N.; Rosenberg, S.A.; Olsen, J.R.; Roach, M.; Wan, L.; Portelance, L.; Mellon, E.A.; Bruynzeel, A.; Lagerwaard, F.; et al. Using adaptive magnetic resonance image-guided radiation therapy for treatment of inoperable pancreatic cancer. Cancer Med. 2019, 8, 2123–2132. [Google Scholar] [CrossRef]
- Manderlier, M.; Navez, J.; Hein, M.; Engelholm, J.L.; Closset, J.; Bali, M.A.; Van Gestel, D.; Moretti, L.; Van Laethem, J.-L.; Bouchart, C. Isotoxic High-Dose Stereotactic Body Radiotherapy (iHD-SBRT) Versus Conventional Chemoradiotherapy for Localized Pancreatic Cancer: A Single Cancer Center Evaluation. Cancers 2022, 14, 5730. [Google Scholar] [CrossRef]
- Erasme University Hospital. Preoperative mFOLFIRINOX (or Gem-Nab-P) +/− Isotoxic High-Dose Stereotactic Body Radiation Therapy (iHD-SBRT) for Borderline (or at Risk) Resectable Pancreatic Adenocarcinoma: A Randomised Comparative Multicentre Phase II Study (STEREOPAC) [Internet]. 2021. Available online: https://clinicaltrials.gov/ct2/show/NCT05083247 (accessed on 18 November 2022).
- Reyngold, M.; Parikh, P.; Crane, C.H. Ablative radiation therapy for locally advanced pancreatic cancer: Techniques and results. Radiat. Oncol. 2019, 14, 95. [Google Scholar] [CrossRef]
- Viewray Inc. Locally Advanced Pancreatic Cancer Treated With ABLAtivE Stereotactic MRI-Guided Adaptive Radiation Therapy (LAP-ABLATE) [Internet]. 2022. Available online: https://clinicaltrials.gov/ct2/show/NCT05585554 (accessed on 18 November 2022).
- Krempien, R.; Roeder, F. Intraoperative radiation therapy (IORT) in pancreatic cancer. Radiat. Oncol. 2017, 12, 8. [Google Scholar] [CrossRef] [PubMed]
- Calvo, F.A.; Asencio, J.M.; Roeder, F.; Krempien, R.; Poortmans, P.; Hensley, F.W.; Krengli, M. ESTRO IORT Task Force/ACROP recommendations for intraoperative radiation therapy in borderline-resected pancreatic cancer. Clin. Transl. Radiat. Oncol. 2020, 23, 91–99. [Google Scholar] [CrossRef]
- Hong, T.S. PACER (Pancreatic AdenoCarcinoma with Electron Intraoperative Radiation Therapy): A Phase II Study of Electron Beam Intraoperative Radiation Therapy Following Chemoradiation in Patients with Pancreatic Cancer with Vascular Involvement [Internet]. 2022. Available online: https://clinicaltrials.gov/ct2/show/NCT03716531 (accessed on 18 November 2022).
- Vozenin, M.-C.; Bourhis, J.; Durante, M. Towards clinical translation of FLASH radiotherapy. Nat. Rev. Clin. Oncol. 2022, 19, 791–803. [Google Scholar] [CrossRef]
- Okoro, C.M.; Schüler, E.; Taniguchi, C.M. The Therapeutic Potential of FLASH-RT for Pancreatic Cancer. Cancers 2022, 14, 1167. [Google Scholar] [CrossRef]
- Wen, X.; Qiu, H.; Shao, Z.; Liu, G.; Liu, N.; Chen, A.; Zhang, X.; Ding, X.; Zhang, L. Pulsed low-dose rate radiotherapy has an improved therapeutic effect on abdominal and pelvic malignancies. J. Zhejiang Univ. B 2021, 22, 774–781. [Google Scholar] [CrossRef]
- Martin, L.M.; Marples, B.; Lynch, T.H.; Hollywood, D.; Marignol, L. Exposure to low dose ionising radiation: Molecular and clinical consequences. Cancer Lett. 2013, 338, 209–218. [Google Scholar] [CrossRef]
- Yan, W.; Khan, M.K.; Wu, X.; Simone, C.B.; Fan, J.; Gressen, E.; Zhang, X.; Limoli, C.L.; Bahig, H.; Tubin, S.; et al. Spatially fractionated radiation therapy: History, present and the future. Clin. Transl. Radiat. Oncol. 2019, 20, 30–38. [Google Scholar] [CrossRef]
- Tubin, S.; Popper, H.H.; Brcic, L. Novel stereotactic body radiation therapy (SBRT)-based partial tumor irradiation targeting hypoxic segment of bulky tumors (SBRT-PATHY): Improvement of the radiotherapy outcome by exploiting the bystander and abscopal effects. Radiat. Oncol. 2019, 14, 21. [Google Scholar] [CrossRef]
- Tubin, S.; Gupta, S.; Grusch, M.; Popper, H.H.; Brcic, L.; Ashdown, M.L.; Khleif, S.N.; Peter-Vörösmarty, B.; Hyden, M.; Negrini, S.; et al. Shifting the Immune-Suppressive to Predominant Immune-Stimulatory Radiation Effects by SBRT-PArtial Tumor Irradiation Targeting HYpoxic Segment (SBRT-PATHY). Cancers 2020, 13, 50. [Google Scholar] [CrossRef]
- Thorsson, V.; Gibbs, D.L.; Brown, S.D.; Wolf, D.; Bortone, D.S.; Ouyang, T.H.; Porta-Pardo, E.; Gao, G.F.; Plaisier, C.L.; Eddy, J.A.; et al. The Immune Landscape of Cancer. Immunity 2018, 48, 812–830.e14. [Google Scholar] [CrossRef]
- Muller, M.; Haghnejad, V.; Schaefer, M.; Gauchotte, G.; Caron, B.; Peyrin-Biroulet, L.; Bronowicki, J.-P.; Neuzillet, C.; Lopez, A. The Immune Landscape of Human Pancreatic Ductal Carcinoma: Key Players, Clinical Implications, and Challenges. Cancers 2022, 14, 995. [Google Scholar] [CrossRef]
- Ansems, M.; Span, P. The tumor microenvironment and radiotherapy response; a central role for cancer-associated fibroblasts. Clin. Transl. Radiat. Oncol. 2020, 22, 90–97. [Google Scholar] [CrossRef]
- Mei, L.; Du, W.; Ma, W.W. Targeting stromal microenvironment in pancreatic ductal adenocarcinoma: Controversies and promises. J. Gastrointest. Oncol. 2016, 7, 487–494. [Google Scholar] [CrossRef]
- Duluc, C.; Moatassim-Billah, S.; Chalabi-Dchar, M.; Perraud, A.; Samain, R.; Breibach, F.; Gayral, M.; Cordelier, P.; Delisle, M.; Bousquet-Dubouch, M.; et al. Pharmacological targeting of the protein synthesis mTOR /4E- BP 1 pathway in cancer-associated fibroblasts abrogates pancreatic tumour chemoresistance. EMBO Mol. Med. 2015, 7, 735–753. [Google Scholar] [CrossRef]
- Whatcott, C.J.; Diep, C.H.; Jiang, P.; Watanabe, A.; LoBello, J.; Sima, C.; Hostetter, G.; Shepard, H.M.; Von Hoff, D.D.; Han, H. Desmoplasia in Primary Tumors and Metastatic Lesions of Pancreatic Cancer. Clin. Cancer Res. 2015, 21, 3561–3568. [Google Scholar] [CrossRef]
- Lander, V.E.; Belle, J.I.; Kingston, N.L.; Herndon, J.M.; Hogg, G.D.; Liu, X.; Kang, L.-I.; Knolhoff, B.L.; Bogner, S.J.; Baer, J.M.; et al. Stromal Reprogramming by FAK Inhibition Overcomes Radiation Resistance to Allow for Immune Priming and Response to Checkpoint Blockade. Cancer Discov. 2022, 12, 2774–2799. [Google Scholar] [CrossRef]
- Bulle, A.; Lim, K.-H. Beyond just a tight fortress: Contribution of stroma to epithelial-mesenchymal transition in pancreatic cancer. Signal Transduct. Target. Ther. 2020, 5, 249. [Google Scholar] [CrossRef]
- Moffitt, R.A.; Marayati, R.; Flate, E.L.; Volmar, K.E.; Loeza, S.G.H.; Hoadley, K.A.; Rashid, N.U.; Williams, L.A.; Eaton, S.C.; Chung, A.H.; et al. Virtual microdissection identifies distinct tumor- and stroma-specific subtypes of pancreatic ductal adenocarcinoma. Nat. Genet. 2015, 47, 1168–1178. [Google Scholar] [CrossRef]
- Polani, F.; Grierson, P.M.; Lim, K.-H. Stroma-targeting strategies in pancreatic cancer: Past lessons, challenges and prospects. World J. Gastroenterol. 2021, 27, 2105–2121. [Google Scholar] [CrossRef]
- Sodergren, M.H.; Mangal, N.; Wasan, H.; Sadanandam, A.; Balachandran, V.P.; Jiao, L.R.; Habib, N. Immunological combination treatment holds the key to improving survival in pancreatic cancer. J. Cancer Res. Clin. Oncol. 2020, 146, 2897–2911. [Google Scholar] [CrossRef]
- Tan, E.; El-Rayes, B. Pancreatic Cancer and Immunotherapy: Resistance Mechanisms and Proposed Solutions. J. Gastrointest. Cancer 2018, 50, 1–8. [Google Scholar] [CrossRef]
- Jiang, H.; Liu, X.; Knolhoff, B.L.; Hegde, S.; Lee, K.B.; Jiang, H.; Fields, R.C.; Pachter, J.A.; Lim, K.-H.; DeNardo, D.G. Development of resistance to FAK inhibition in pancreatic cancer is linked to stromal depletion. Gut 2019, 69, 122–132. [Google Scholar] [CrossRef]
- Kalluri, R. The biology and function of fibroblasts in cancer. Nat. Rev. Cancer 2016, 16, 582–598. [Google Scholar] [CrossRef]
- Elyada, E.; Bolisetty, M.; Laise, P.; Flynn, W.F.; Courtois, E.T.; Burkhart, R.A.; Teinor, J.A.; Belleau, P.; Biffi, G.; Lucito, M.S.; et al. Cross-Species Single-Cell Analysis of Pancreatic Ductal Adenocarcinoma Reveals Antigen-Presenting Cancer-Associated Fibroblasts. Cancer Discov. 2019, 9, 1102–1123. [Google Scholar] [CrossRef] [PubMed]
- Djurec, M.; Graña, O.; Lee, A.; Troulé, K.; Espinet, E.; Cabras, L.; Navas, C.; Blasco, M.T.; Martín-Díaz, L.; Burdiel, M.; et al. Saa3 is a key mediator of the protumorigenic properties of cancer-associated fibroblasts in pancreatic tumors. Proc. Natl. Acad. Sci. USA 2018, 115, E1147–E1156. [Google Scholar] [CrossRef]
- Özdemir, B.C.; Pentcheva-Hoang, T.; Carstens, J.L.; Zheng, X.; Wu, C.C.; Simpson, T.R.; Laklai, H.; Sugimoto, H.; Kahlert, C.; De Jesus-Acosta, A.; et al. Faculty Opinions recommendation of Depletion of carcinoma-associated fibroblasts and fibrosis induces immunosuppression and accelerates pancreas cancer with reduced survival. Cancer Cell 2014, 25, 719–734. [Google Scholar] [CrossRef]
- Domen, A.; Quatannens, D.; Zanivan, S.; Deben, C.; Van Audenaerde, J.; Smits, E.; Wouters, A.; Lardon, F.; Roeyen, G.; Verhoeven, Y.; et al. Cancer-Associated Fibroblasts as a Common Orchestrator of Therapy Resistance in Lung and Pancreatic Cancer. Cancers 2021, 13, 987. [Google Scholar] [CrossRef]
- Fiori, M.E.; Di Franco, S.; Villanova, L.; Bianca, P.; Stassi, G.; De Maria, R. Cancer-associated fibroblasts as abettors of tumor progression at the crossroads of EMT and therapy resistance. Mol. Cancer 2019, 18, 70. [Google Scholar] [CrossRef]
- Erkan, M.; Kleeff, J.; Gorbachevski, A.; Reiser, C.; Mitkus, T.; Esposito, I.; Giese, T.; Büchler, M.W.; Giese, N.A.; Friess, H. Periostin Creates a Tumor-Supportive Microenvironment in the Pancreas by Sustaining Fibrogenic Stellate Cell Activity. Gastroenterology 2007, 132, 1447–1464. [Google Scholar] [CrossRef]
- Mills, B.N.; Qiu, H.; Drage, M.G.; Chen, C.; Mathew, J.S.; Garrett-Larsen, J.; Ye, J.; Uccello, T.P.; Murphy, J.D.; Belt, B.A.; et al. Modulation of the Human Pancreatic Ductal Adenocarcinoma Immune Microenvironment by Stereotactic Body Radiotherapy. Clin. Cancer Res. 2022, 28, 150–162. [Google Scholar] [CrossRef]
- Wang, Z.; Tang, Y.; Tan, Y.; Wei, Q.; Yu, W. Cancer-associated fibroblasts in radiotherapy: Challenges and new opportunities. Cell Commun. Signal. 2019, 17, 47. [Google Scholar] [CrossRef]
- Hellevik, T.; Berzaghi, R.; Lode, K.; Islam, A.; Martinez-Zubiaurre, I. Immunobiology of cancer-associated fibroblasts in the context of radiotherapy. J. Transl. Med. 2021, 19, 437. [Google Scholar] [CrossRef]
- Ohuchida, K.; Mizumoto, K.; Murakami, M.; Qian, L.-W.; Sato, N.; Nagai, E.; Matsumoto, K.; Nakamura, T.; Tanaka, M. Radiation to Stromal Fibroblasts Increases Invasiveness of Pancreatic Cancer Cells through Tumor-Stromal Interactions. Cancer Res. 2004, 64, 3215–3222. [Google Scholar] [CrossRef]
- Steer, A.; Cordes, N.; Jendrossek, V.; Klein, D. Impact of Cancer-Associated Fibroblast on the Radiation-Response of Solid Xenograft Tumors. Front. Mol. Biosci. 2019, 6, 70. [Google Scholar] [CrossRef] [PubMed]
- Annese, T.; Tamma, R.; Ruggieri, S.; Ribatti, D. Angiogenesis in Pancreatic Cancer: Pre-Clinical and Clinical Studies. Cancers 2019, 11, 381. [Google Scholar] [CrossRef]
- Barău, A.; Ruiz-Sauri, A.; Valencia, G.; Gómez-Mateo, M.D.C.; Sabater, L.; Ferrandez, A.; Llombart-Bosch, A. High microvessel density in pancreatic ductal adenocarcinoma is associated with high grade. Virchows Archiv 2013, 462, 541–546. [Google Scholar] [CrossRef] [PubMed]
- Jin, X.; Dai, L.; Ma, Y.; Wang, J.; Liu, Z. Implications of HIF-1α in the tumorigenesis and progression of pancreatic cancer. Cancer Cell Int. 2020, 20, 273. [Google Scholar] [CrossRef] [PubMed]
- Tao, J.; Yang, G.; Zhou, W.; Qiu, J.; Chen, G.; Luo, W.; Zhao, F.; You, L.; Zheng, L.; Zhang, T.; et al. Targeting hypoxic tumor microenvironment in pancreatic cancer. J. Hematol. Oncol. 2021, 14, 14. [Google Scholar] [CrossRef]
- Li, S.; Xu, H.-X.; Wu, C.-T.; Wang, W.-Q.; Jin, W.; Gao, H.-L.; Li, H.; Zhang, S.-R.; Xu, J.-Z.; Qi, Z.-H.; et al. Angiogenesis in pancreatic cancer: Current research status and clinical implications. Angiogenesis 2018, 22, 15–36. [Google Scholar] [CrossRef]
- Saiyin, H.; Ardito-Abraham, C.M.; Wu, Y.; Wei, Y.; Fang, Y.; Han, X.; Li, J.; Zhou, P.; Yi, Q.; Maitra, A.; et al. Identification of novel vascular projections with cellular trafficking abilities on the microvasculature of pancreatic ductal adenocarcinoma. J. Pathol. 2015, 236, 142–154. [Google Scholar] [CrossRef]
- Langley, R.; Bump, E.; Quartuccio, S.; Medeiros, D.; Braunhut, S. Radiation-induced apoptosis in microvascular endothelial cells. Br. J. Cancer 1997, 75, 666–672. [Google Scholar] [CrossRef]
- Huang, S.; Ma, H. Effect of radiotherapy on angiogenesis of human pancreatic cancer transplanted tumor in nude mice. Chinese-German J. Clin. Oncol. 2012, 11, 635–637. [Google Scholar] [CrossRef]
- Ye, J.; Mills, B.N.; Zhao, T.; Han, B.J.; Murphy, J.D.; Patel, A.P.; Johnston, C.J.; Lord, E.M.; Belt, B.A.; Linehan, D.C.; et al. Assessing the Magnitude of Immunogenic Cell Death Following Chemotherapy and Irradiation Reveals a New Strategy to Treat Pancreatic Cancer. Cancer Immunol. Res. 2020, 8, 94–107. [Google Scholar] [CrossRef] [PubMed]
- Carstens, J.L.; de Sampaio, P.C.; Yang, D.; Barua, S.; Wang, H.; Rao, A.; Allison, J.P.; LeBleu, V.S.; Kalluri, R. Spatial computation of intratumoral T cells correlates with survival of patients with pancreatic cancer. Nat. Commun. 2017, 8, 15095. [Google Scholar] [CrossRef]
- Yang, S.; Liu, Q.; Liao, Q. Tumor-Associated Macrophages in Pancreatic Ductal Adenocarcinoma: Origin, Polarization, Function, and Reprogramming. Front. Cell Dev. Biol. 2020, 8, 607209. [Google Scholar] [CrossRef] [PubMed]
- Principe, D.R.; DeCant, B.; Mascariñas, E.; Wayne, E.A.; Diaz, A.M.; Akagi, N.; Hwang, R.; Pasche, B.; W Dawson, D.; Grippo, P.J.; et al. TGFβ Signaling in the Pancreatic Tumor Microenvironment Promotes Fibrosis and Immune Evasion to Facilitate Tumorigenesis. Cancer Res. 2016, 76, 2525–2539. [Google Scholar] [CrossRef] [PubMed]
- Matsuo, Y.; Ochi, N.; Sawai, H.; Yasuda, A.; Takahashi, H.; Funahashi, H.; Takeyama, H.; Tong, Z.; Guha, S. CXCL8/IL-8 and CXCL12/SDF-1α Co-operatively Promote Invasiveness and Angiogenesis in Pancreatic Cancer. Int. J. Cancer J. Int. Cancer 2009, 124, 853–861. [Google Scholar] [CrossRef] [PubMed]
- Puolakkainen, P.; Koski, A.; Vainionpää, S.; Shen, Z.; Repo, H.; Kemppainen, E.; Mustonen, H.; Seppänen, H. Anti-inflammatory macrophages activate invasion in pancreatic adenocarcinoma by increasing the MMP9 and ADAM8 expression. Med. Oncol. 2014, 31, 884. [Google Scholar] [CrossRef]
- Poh, A.; Ernst, M. Tumor-Associated Macrophages in Pancreatic Ductal Adenocarcinoma: Therapeutic Opportunities and Clinical Challenges. Cancers 2021, 13, 2860. [Google Scholar] [CrossRef]
- Seifert, L.; Werba, G.; Tiwari, S.; Ly, N.N.G.; Nguy, S.; Alothman, S.; Alqunaibit, D.; Avanzi, A.; Daley, D.; Barilla, R.; et al. Radiation Therapy Induces Macrophages to Suppress T-Cell Responses against Pancreatic Tumors in Mice. Gastroenterology 2016, 150, 1659–1672.e5. [Google Scholar] [CrossRef]
- Yang, X.; Lu, Y.; Hang, J.; Zhang, J.; Zhang, T.; Huo, Y.; Liu, J.; Lai, S.; Luo, D.; Wang, L.; et al. Lactate-Modulated Immunosuppression of Myeloid-Derived Suppressor Cells Contributes to the Radioresistance of Pancreatic Cancer. Cancer Immunol. Res. 2020, 8, 1440–1451. [Google Scholar] [CrossRef]
- Oweida, A.J.; Mueller, A.C.; Piper, M.; Milner, D.; Van Court, B.; Bhatia, S.; Phan, A.; Bickett, T.; Jordan, K.; Proia, T.; et al. Response to radiotherapy in pancreatic ductal adenocarcinoma is enhanced by inhibition of myeloid-derived suppressor cells using STAT3 anti-sense oligonucleotide. Cancer Immunol. Immunother. 2020, 70, 989–1000. [Google Scholar] [CrossRef]
- Kalbasi, A.; Komar, C.; Tooker, G.M.; Liu, M.; Lee, J.W.; Gladney, W.L.; Ben-Josef, E.; Beatty, G.L. Tumor-Derived CCL2 Mediates Resistance to Radiotherapy in Pancreatic Ductal Adenocarcinoma. Clin. Cancer Res. 2017, 23, 137–148. [Google Scholar] [CrossRef] [PubMed]
- Beach, C.; MacLean, D.; Majorova, D.; Arnold, J.N.; Olcina, M.M. The effects of radiation therapy on the macrophage response in cancer. Front. Oncol. 2022, 12, 1020606. [Google Scholar] [CrossRef]
- Wang, J.; Saung, M.T.; Li, K.; Fu, J.; Fujiwara, K.; Niu, N.; Muth, S.; Wang, J.; Xu, Y.; Zheng, L.; et al. CCR2/CCR5 inhibitor permits the radiation-induced effector T cell infiltration in pancreatic adenocarcinoma. J. Exp. Med. 2022, 219, e20211631. [Google Scholar] [CrossRef] [PubMed]
- Jiménez-Cortegana, C.; Galassi, C.; Klapp, V.; Gabrilovich, D.I.; Galluzzi, L. Myeloid-Derived Suppressor Cells and Radiotherapy. Cancer Immunol. Res. 2022, 10, 545–557. [Google Scholar] [CrossRef] [PubMed]
- Thyagarajan, A.; Alshehri, M.S.A.; Miller, K.L.; Sherwin, C.M.; Travers, J.B.; Sahu, R.P. Myeloid-Derived Suppressor Cells and Pancreatic Cancer: Implications in Novel Therapeutic Approaches. Cancers 2019, 11, 1627. [Google Scholar] [CrossRef]
- Bronte, V.; Brandau, S.; Chen, S.-H.; Colombo, M.P.; Frey, A.B.; Greten, T.F.; Mandruzzato, S.; Murray, P.J.; Ochoa, A.; Ostrand-Rosenberg, S.; et al. Recommendations for myeloid-derived suppressor cell nomenclature and characterization standards. Nat. Commun. 2016, 7, 12150. [Google Scholar] [CrossRef]
- Qin, C.; Yang, G.; Yang, J.; Ren, B.; Wang, H.; Chen, G.; Zhao, F.; You, L.; Wang, W.; Zhao, Y. Metabolism of pancreatic cancer: Paving the way to better anticancer strategies. Mol. Cancer 2020, 19, 50. [Google Scholar] [CrossRef]
- Jin, L.; Kim, H.; Shi, J. Neutrophil in the Pancreatic Tumor Microenvironment. Biomolecules 2021, 11, 1170. [Google Scholar] [CrossRef]
- Fridlender, Z.G.; Sun, J.; Kim, S.; Kapoor, V.; Cheng, G.; Ling, L.; Albelda, S.M. Polarization of tumor-associated neutrophil phenotype by TGF-beta: ‘N1’ versus ‘N2’ TAN. Cancer Cell 2009, 16, 183–194. [Google Scholar] [CrossRef]
- Brinkmann, V.; Reichard, U.; Goosmann, C.; Fauler, B.; Uhlemann, Y.; Weiss, D.S.; Weinrauch, Y.; Zychlinsky, A. Neutrophil extracellular traps kill bacteria. Science 2004, 303, 1532–1535. [Google Scholar] [CrossRef]
- Kajioka, H.; Kagawa, S.; Ito, A.; Yoshimoto, M.; Sakamoto, S.; Kikuchi, S.; Kuroda, S.; Yoshida, R.; Umeda, Y.; Noma, K.; et al. Targeting neutrophil extracellular traps with thrombomodulin prevents pancreatic cancer metastasis. Cancer Lett. 2020, 497, 1–13. [Google Scholar] [CrossRef] [PubMed]
- Yang, L.; Liu, Q.; Zhang, X.; Liu, X.; Zhou, B.; Chen, J.; Huang, D.; Li, J.; Li, H.; Chen, F.; et al. DNA of neutrophil extracellular traps promotes cancer metastasis via CCDC25. Nature 2020, 583, 133–138. [Google Scholar] [CrossRef] [PubMed]
- Nielsen, S.R.; Strøbech, J.E.; Horton, E.R.; Jackstadt, R.; Laitala, A.; Bravo, M.C.; Maltese, G.; RD Jensen, A.; Rueten, R.; Erler, J.T.; et al. Suppression of tumor-associated neutrophils by lorlatinib attenuates pancreatic cancer growth and improves treatment with immune checkpoint blockade. Nat. Commun. 2021, 12, 3414. [Google Scholar] [CrossRef] [PubMed]
- Iwai, N.; Okuda, T.; Sakagami, J.; Harada, T.; Ohara, T.; Taniguchi, M.; Sakai, H.; Oka, K.; Hara, T.; Tsuji, T.; et al. Neutrophil to lymphocyte ratio predicts prognosis in unresectable pancreatic cancer. Sci. Rep. 2020, 10, 18758. [Google Scholar] [CrossRef] [PubMed]
- Reddy, A.V.; Hill, C.S.; Sehgal, S.; He, J.; Zheng, L.; Herman, J.M.; Meyer, J.; Narang, A.K. High neutrophil-to-lymphocyte ratio following stereotactic body radiation therapy is associated with poor clinical outcomes in patients with borderline resectable and locally advanced pancreatic cancer. J. Gastrointest. Oncol. 2022, 13, 368–379. [Google Scholar] [CrossRef] [PubMed]
- Wolfe, A.R.; Siedow, M.; Nalin, A.; DiCostanzo, D.; Miller, E.D.; Diaz, D.A.; Arnett, A.; Cloyd, J.M.; Dillhoff, M.; Ejaz, A.; et al. Increasing neutrophil-to-lymphocyte ratio following radiation is a poor prognostic factor and directly correlates with splenic radiation dose in pancreatic cancer. Radiother. Oncol. 2021, 158, 207–214. [Google Scholar] [CrossRef]
- Reddy, A.V.; Hill, C.S.; Sehgal, S.; Zheng, L.; He, J.; Laheru, D.A.; De Jesus-Acosta, A.; Herman, J.M.; Meyer, J.; Narang, A.K. Post-radiation neutrophil-to-lymphocyte ratio is a prognostic marker in patients with localized pancreatic adenocarcinoma treated with anti-PD-1 antibody and stereotactic body radiation therapy. Radiat. Oncol. J. 2022, 40, 111–119. [Google Scholar] [CrossRef]
- Liu, Q.; Hao, Y.; Du, R.; Hu, D.; Xie, J.; Zhang, J.; Deng, G.; Liang, N.; Tian, T.; Käsmann, L.; et al. Radiotherapy programs neutrophils to an antitumor phenotype by inducing mesenchymal-epithelial transition. Transl. Lung Cancer Res. 2021, 10, 1424–1443. [Google Scholar] [CrossRef]
- Li, T.J.; Wang, W.Q.; Yu, X.J.; Liu, L. Killing the ‘BAD’: Challenges for immunotherapy in pancreatic cancer. Biochim Biophys Acta Rev. Cancer. 2020, 1874, 188384. [Google Scholar] [CrossRef]
- Zhou, Q.; Tao, X.; Xia, S.; Guo, F.; Pan, C.; Xiang, H.; Shang, D. T Lymphocytes: A Promising Immunotherapeutic Target for Pancreatitis and Pancreatic Cancer? Front. Oncol. 2020, 10, 382. [Google Scholar] [CrossRef]
- Thind, K.; Padrnos, L.J.; Ramanathan, R.K.; Borad, M.J. Immunotherapy in pancreatic cancer treatment: A new frontier. Ther. Adv. Gastroenterol. 2016, 10, 168–194. [Google Scholar] [CrossRef] [PubMed]
- Basso, D.; Fogar, P.; Falconi, M.; Fadi, E.; Sperti, C.; Frasson, C.; Greco, E.; Tamburrino, D.; Teolato, S.; Moz, S.; et al. Pancreatic Tumors and Immature Immunosuppressive Myeloid Cells in Blood and Spleen: Role of Inhibitory Co-Stimulatory Molecules PDL1 and CTLA4. An In Vivo and In Vitro Study. PLoS ONE 2013, 8, e54824. [Google Scholar] [CrossRef] [PubMed]
- Ryschich, E.; Nötzel, T.; Hinz, U.; Autschbach, F.; Ferguson, J.; Simon, I.; Weitz, J.; Fröhlich, B.; Klar, E.; Büchler, M.W.; et al. Control of T-Cell–Mediated Immune Response by HLA Class I in Human Pancreatic Carcinoma. Clin. Cancer Res. 2005, 11, 498–504. [Google Scholar] [CrossRef] [PubMed]
- Ahmed, A.; Köhler, S.; Klotz, R.; Giese, N.; Hackert, T.; Springfeld, C.; Zörnig, I.; Jäger, D.; Halama, N. Tertiary lymphoid structures and their association to immune phenotypes and circulatory IL2 levels in pancreatic ductal adenocarcinoma. Oncoimmunology 2022, 11, 2027148. [Google Scholar] [CrossRef]
- Bai, M.; Zheng, Y.; Liu, H.; Su, B.; Zhan, Y.; He, H. CXCR5+ CD8+ T cells potently infiltrate pancreatic tumors and present high functionality. Exp. Cell Res. 2017, 361, 39–45. [Google Scholar] [CrossRef] [PubMed]
- Australian Pancreatic Cancer Genome Initiative; Balachandran, V.P.; Łuksza, M.; Zhao, J.N.; Makarov, V.; Moral, J.A.; Remark, R.; Herbst, B.; Askan, G.; Bhanot, U.; et al. Identification of unique neoantigen qualities in long-term survivors of pancreatic cancer. Nature 2017, 551, 512–516. [Google Scholar] [CrossRef]
- Mota Reyes, C.; Demir, E.; Çifcibaşı, K.; Istvanffy, R.; Friess, H.; Demir, I.E. Regulatory T Cells in Pancreatic Cancer: Of Mice and Men. Cancers 2022, 14, 4582. [Google Scholar] [CrossRef]
- Li, C.; Jiang, P.; Wei, S.; Xu, X.; Wang, J. Regulatory T cells in tumor microenvironment: New mechanisms, potential therapeutic strategies and future prospects. Mol. Cancer 2020, 19, 116. [Google Scholar] [CrossRef]
- Hiraoka, N.; Ino, Y.; Yamazaki-Itoh, R.; Kanai, Y.; Kosuge, T.; Shimada, K. Intratumoral tertiary lymphoid organ is a favourable prognosticator in patients with pancreatic cancer. Br. J. Cancer 2015, 112, 1782–1790. [Google Scholar] [CrossRef]
- Fincham, R.E.A.; Delvecchio, F.R.; Goulart, M.R.; Yeong, J.P.S.; Kocher, H.M. Natural killer cells in pancreatic cancer stroma. World J. Gastroenterol. 2021, 27, 3483–3501. [Google Scholar] [CrossRef]
- Peng, Y.-P.; Xi, C.-H.; Zhu, Y.; Yin, L.-D.; Wei, J.-S.; Zhang, J.-J.; Liu, X.-C.; Guo, S.; Fu, Y.; Miao, Y. Altered expression of CD226 and CD96 on natural killer cells in patients with pancreatic cancer. Oncotarget 2016, 7, 66586–66594. [Google Scholar] [CrossRef]
- Peng, Y.-P.; Zhang, J.-J.; Liang, W.-B.; Tu, M.; Lu, Z.-P.; Wei, J.-S.; Jiang, K.-R.; Gao, W.-T.; Wu, J.-L.; Xu, Z.-K.; et al. Elevation of MMP-9 and IDO induced by pancreatic cancer cells mediates natural killer cell dysfunction. BMC Cancer 2014, 14, 738. [Google Scholar] [CrossRef]
- Crocenzi, T.; Cottam, B.; Newell, P.; Wolf, R.F.; Hansen, P.D.; Hammill, C.; Solhjem, M.C.; To, Y.-Y.; Greathouse, A.; Tormoen, G.; et al. A hypofractionated radiation regimen avoids the lymphopenia associated with neoadjuvant chemoradiation therapy of borderline resectable and locally advanced pancreatic adenocarcinoma. J. Immunother. Cancer 2016, 4, 45. [Google Scholar] [CrossRef] [PubMed]
- Farren, M.R.; Sayegh, L.; Ware, M.B.; Chen, H.-R.; Gong, J.; Liang, Y.; Krasinskas, A.; Maithel, S.K.; Zaidi, M.; Sarmiento, J.M.; et al. Immunologic alterations in the pancreatic cancer microenvironment of patients treated with neoadjuvant chemotherapy and radiotherapy. J. Clin. Investig. 2020, 5, e130362. [Google Scholar] [CrossRef] [PubMed]
- Lee, Y.S.; Kim, H.S.; Cho, Y.; Lee, I.J.; Kim, H.J.; Lee, D.E.; Kang, H.W.; Park, J.S. Intraoperative radiation therapy induces immune response activity after pancreatic surgery. BMC Cancer 2021, 21, 1097. [Google Scholar] [CrossRef] [PubMed]
- Chamma, H.; Vila, I.K.; Taffoni, C.; Turtoi, A.; Laguette, N. Activation of STING in the pancreatic tumor microenvironment: A novel therapeutic opportunity. Cancer Lett. 2022, 538, 215694. [Google Scholar] [CrossRef] [PubMed]
- Mohseni, G.; Li, J.; Ariston Gabriel, A.N.; Du, L.; Wang, Y.; Wang, C. The Function of cGAS-STING Pathway in Treatment of Pancreatic Cancer. Front. Immunol. 2021, 12, 781032. [Google Scholar] [CrossRef]
- Kabashima, A.; Matsuo, Y.; Ito, S.; Akiyama, Y.; Ishii, T.; Shimada, S.; Masamune, A.; Tanabe, M.; Tanaka, S. cGAS-STING signaling encourages immune cell overcoming of fibroblast barricades in pancreatic cancer. Sci. Rep. 2022, 12, 10466. [Google Scholar] [CrossRef]
- Baird, J.R.; Friedman, D.; Cottam, B.; Dubensky, T.W., Jr.; Kanne, D.B.; Bambina, S.; Bahjat, K.; Crittenden, M.R.; Gough, M.J. Radiotherapy Combined with Novel STING-Targeting Oligonucleotides Results in Regression of Established Tumors. Cancer Res. 2016, 76, 50–61. [Google Scholar] [CrossRef]
- Storozynsky, Q.; Hitt, M.M. The Impact of Radiation-Induced DNA Damage on cGAS-STING-Mediated Immune Responses to Cancer. Int. J. Mol. Sci. 2020, 21, 8877. [Google Scholar] [CrossRef]
- Vanpouille-Box, C.; Alard, A.; Aryankalayil, M.J.; Sarfraz, Y.; Diamond, J.M.; Schneider, R.J.; Inghirami, G.; Coleman, C.N.; Formenti, S.C.; Demaria, S. DNA exonuclease Trex1 regulates radiotherapy-induced tumour immunogenicity. Nat. Commun. 2017, 8, 15618. [Google Scholar] [CrossRef]
- Vanpouille-Box, C.; Formenti, S.C.; Demaria, S. Toward Precision Radiotherapy for Use with Immune Checkpoint Blockers. Clin. Cancer Res. 2018, 24, 259–265. [Google Scholar] [CrossRef] [PubMed]
- Arsenijevic, T.; Nicolle, R.; Bouchart, C.; D’Haene, N.; Demetter, P.; Puleo, F.; Van Laethem, J.-L. Pancreatic Cancer Meets Human Microbiota: Close Encounters of the Third Kind. Cancers 2021, 13, 1231. [Google Scholar] [CrossRef]
- Stasiewicz, M.; Kwaśniewski, M.; Karpiński, T. Microbial Associations with Pancreatic Cancer: A New Frontier in Biomarkers. Cancers 2021, 13, 3784. [Google Scholar] [CrossRef]
- Aykut, B.; Pushalkar, S.; Chen, R.; Li, Q.; Abengozar, R.; Kim, J.I.; Shadaloey, S.A.; Wu, D.; Preiss, P.; Verma, N.; et al. The fungal mycobiome promotes pancreatic oncogenesis via activation of MBL. Nature 2019, 574, 264–267. [Google Scholar] [CrossRef]
- Pushalkar, S.; Hundeyin, M.; Daley, D.; Zambirinis, C.P.; Kurz, E.; Mishra, A.; Mohan, N.; Aykut, B.; Usyk, M.; Torres, L.E.; et al. The Pancreatic Cancer Microbiome Promotes Oncogenesis by Induction of Innate and Adaptive Immune Suppression. Cancer Discov. 2018, 8, 403–416. [Google Scholar] [CrossRef] [PubMed]
- Liu, J.; Liu, C.; Yue, J. Radiotherapy and the gut microbiome: Facts and fiction. Radiat. Oncol. 2021, 16, 9. [Google Scholar] [CrossRef] [PubMed]
- Leroux, C.; Konstantinidou, G. Targeted Therapies for Pancreatic Cancer: Overview of Current Treatments and New Opportunities for Personalized Oncology. Cancers 2021, 13, 799. [Google Scholar] [CrossRef]
- Zhang, Z.; Liu, X.; Chen, D.; Yu, J. Radiotherapy combined with immunotherapy: The dawn of cancer treatment. Signal Transduct. Target. Ther. 2022, 7, 258. [Google Scholar] [CrossRef] [PubMed]
- Antonia, S.J.; Villegas, A.; Daniel, D.; Vicente, D.; Murakami, S.; Hui, R.; Yokoi, T.; Chiappori, A.; Lee, K.H.; De Wit, M.; et al. Durvalumab after Chemoradiotherapy in Stage III Non-Small-Cell Lung Cancer. N. Engl. J. Med. 2017, 377, 1919–1929. [Google Scholar] [CrossRef]
- Jabbour, S.K.; Lee, K.H.; Frost, N.; Breder, V.; Kowalski, D.M.; Pollock, T.; Levchenko, E.; Reguart, N.; Martinez-Marti, A.; Houghton, B.; et al. Pembrolizumab Plus Concurrent Chemoradiation Therapy in Patients With Unresectable, Locally Advanced, Stage III Non–Small Cell Lung Cancer. JAMA Oncol. 2021, 7, 1351. [Google Scholar] [CrossRef] [PubMed]
- Kole, C.; Charalampakis, N.; Tsakatikas, S.; Frountzas, M.; Apostolou, K.; Schizas, D. Immunotherapy in Combination with Well-Established Treatment Strategies in Pancreatic Cancer: Current Insights. Cancer Manag. Res. 2022, 14, 1043–1061. [Google Scholar] [CrossRef] [PubMed]
- Barker, H.E.; Paget, J.T.E.; Khan, A.A.; Harrington, K.J. The tumour microenvironment after radiotherapy: Mechanisms of resistance and recurrence. Nat. Rev. Cancer 2015, 15, 409–425. [Google Scholar] [CrossRef]
- Caputo, C.; Pozzi, P.; Farolfi, T.; Passa, R.; Coppola, R.; Caracciolo, G. Nanotechnology and pancreatic cancer management: State of the art and further perspec-tives. World J. Gastrointest. Oncol. 2021, 13, 231–237. [Google Scholar] [CrossRef]
- Jia, M.; Zhang, D.; Zhang, C.; Li, C. Nanoparticle-based delivery systems modulate the tumor microenvironment in pancreatic cancer for enhanced therapy. J. Nanobiotechnology 2021, 19, 1–15. [Google Scholar] [CrossRef] [PubMed]
- Von Hoff, D.D.; Ervin, T.; Arena, F.P.; Chiorean, E.G.; Infante, J.; Moore, M.; Seay, T.; Tjulandin, S.A.; Ma, W.W.; Saleh, M.N.; et al. Increased survival in pancreatic cancer with nab-paclitaxel plus gemcitabine. N. Engl. J. Med. 2013, 369, 1691–1703. [Google Scholar] [CrossRef]
- Wang-Gillam, A.; Li, C.-P.; Bodoky, G.; Dean, A.; Shan, Y.-S.; Jameson, G.; Macarulla, T.; Lee, K.-H.; Cunningham, D.; Blanc, J.F.; et al. Nanoliposomal irinotecan with fluorouracil and folinic acid in metastatic pancreatic cancer after previous gemcitabine-based therapy (NAPOLI-1): A global, randomised, open-label, phase 3 trial. Lancet 2015, 387, 545–557. [Google Scholar] [CrossRef]
- Wang-Gillam, A.; Hubner, R.A.; Siveke, J.T.; Von Hoff, D.D.; Belanger, B.; de Jong, F.A.; Mirakhur, B.; Chen, L.-T. NAPOLI-1 phase 3 study of liposomal irinotecan in metastatic pancreatic cancer: Final overall survival analysis and characteristics of long-term survivors. Eur. J. Cancer 2019, 108, 78–87. [Google Scholar] [CrossRef]
- Chen, Y.; Yang, J.; Fu, S.; Wu, J. Gold Nanoparticles as Radiosensitizers in Cancer Radiotherapy. Int. J. Nanomed. 2020, 15, 9407–9430. [Google Scholar] [CrossRef]
- Wason, M.S.; Lu, H.; Yu, L.; Lahiri, S.K.; Mukherjee, D.; Shen, C.; Das, S.; Seal, S.; Zhao, J. Cerium oxide nanoparticles sensitize pancreatic cancer to radiation therapy through oxidative activation of the JNK apoptotic pathway. Cancers 2018, 10, 303. [Google Scholar] [CrossRef]
- Alhussan, A.; Palmerley, N.; Smazynski, J.; Karasinska, J.; Renouf, D.J.; Schaeffer, D.F.; Beckham, W.; Alexander, A.S.; Chithrani, D.B. Potential of Gold Nanoparticles in Current Radiotherapy Using a Co-Culture Model of Cancer Cells and Cancer Associated Fibroblasts. Cancers 2022, 14, 3586. [Google Scholar] [CrossRef]
- Bilynsky, C.; Millot, N.; Papa, A. Radiation nanosensitizers in cancer therapy—From preclinical discoveries to the outcomes of early clinical trials. Bioeng. Transl. Med. 2021, 7. [Google Scholar] [CrossRef]
- Nano-SMART: Nanoparticles with MR Guided SBRT in Centrally Located Lung Tumors and Pancreatic Cancer. Available online: https://clinicaltrials.gov/ct2/show/NCT04789486) (accessed on 18 January 2023).
- Nywening, T.M.; Wang-Gillam, A.; Sanford, D.E.; Belt, B.A.; Panni, R.Z.; Cusworth, B.M.; Toriola, A.T.; Nieman, R.K.; Worley, L.A.; Linehan, D.C.; et al. Targeting tumour-associated macrophages with CCR2 inhibition in combination with FOLFIRINOX in patients with borderline resectable and locally advanced pancreatic cancer: A single-centre, open-label, dose-finding, non-randomised, phase 1b trial. Lancet Oncol. 2016, 17, 651–662. [Google Scholar] [CrossRef]
- Sidney Kimmel Comprehensive Cancer Center at Johns Hopkins. A Phase I/II Trial of Combination Immunotherapy with Nivolumab and a CCR2/CCR5 Dual Antagonist (BMS-813160) with or without GVAX Following Chemotherapy and Radiotherapy for Locally Advanced Pancreatic Ductal Adenocarcinomas (PDACs). 2022. Available online: https://clinicaltrials.gov/ct2/show/NCT03767582 (accessed on 18 November 2022).
- Carr, M.I.; Chiu, L.-Y.; Guo, Y.; Xu, C.; Lazorchak, A.S.; Yu, H.; Qin, G.; Qi, J.; Marelli, B.; Lan, Y.; et al. DNA-PK Inhibitor Peposertib Amplifies Radiation-Induced Inflammatory Micronucleation and Enhances TGFβ/PD-L1 Targeted Cancer Immunotherapy. Mol. Cancer Res. 2022, 20, 568–582. [Google Scholar] [CrossRef]
- Strauss, J.; Heery, C.R.; Schlom, J.; Madan, R.A.; Cao, L.; Kang, Z. Phase I Trial of M7824 (MSB0011359C), a Bifunctional Fusion Protein Targeting PD-L1 and TGFβ, in Advanced Solid Tumors. Clin. Cancer Res. Off J. Am. Assoc. Cancer Res. 2018, 24, 1287–1295. [Google Scholar] [CrossRef]
- Metropulos, A.E.; Munshi, H.G.; Principe, D.R. The difficulty in translating the preclinical success of combined TGFβ and immune checkpoint inhibition to clinical trial. Ebiomedicine 2022, 86, 104380. [Google Scholar] [CrossRef]
- MD UR. A Phase IB/II Single-Arm Study of M7824 (MSB0011359C) in Combination With Gemcitabine in Adults With Previously Treated Advanced Adenocarcinoma of the Pancreas [Internet]. 2021. Available online: https://clinicaltrials.gov/ct2/show/NCT03451773 (accessed on 18 November 2022).
- Pinter, M.; Jain, R.K. Targeting the renin-angiotensin system to improve cancer treatment: Implications for immunotherapy. Sci. Transl. Med. 2017, 9, eaan5616. [Google Scholar] [CrossRef]
- George, A.J.; Thomas, W.G.; Hannan, R.D. The renin-angiotensin system and cancer: Old dog, new tricks. Nat. Rev. Cancer 2010, 10, 745–759. [Google Scholar] [CrossRef]
- Kumar, V.; Boucher, Y.; Liu, H.; Ferreira, D.; Hooker, J.; Catana, C.; Hoover, A.J.; Ritter, T.; Jain, R.K.; Guimaraes, A.R. Noninvasive Assessment of Losartan-Induced Increase in Functional Microvasculature and Drug Delivery in Pancreatic Ductal Adenocarcinoma. Transl. Oncol. 2016, 9, 431–437. [Google Scholar] [CrossRef]
- Chauhan, V.P.; Martin, J.D.; Liu, H.; Lacorre, D.A.; Jain, S.R.; Kozin, S.V.; Stylianopoulos, T.; Mousa, A.S.; Han, X.; Adstamongkonkul, P.; et al. Angiotensin inhibition enhances drug delivery and potentiates chemotherapy by decompressing tumour blood vessels. Nat. Commun. 2013, 4, 2516. [Google Scholar] [CrossRef]
- Liu, H.; Naxerova, K.; Pinter, M.; Incio, J.; Lee, H.; Shigeta, K.; Ho, W.W.; Crain, J.A.; Jacobson, A.; Michelakos, T.; et al. Use of Angiotensin System Inhibitors Is Associated with Immune Activation and Longer Survival in Nonmetastatic Pancreatic Ductal Adenocarcinoma. Clin. Cancer Res. 2017, 23, 5959–5969. [Google Scholar] [CrossRef]
- Krajewska, M.; Heijink, A.M.; Bisselink, Y.J.W.M.; I Seinstra, R.; Silljé, H.H.W.; E de Vries, E.G.; van Vugt, M.A. Forced activation of Cdk1 via wee1 inhibition impairs homologous recombination. Oncogene 2012, 32, 3001–3008. [Google Scholar] [CrossRef]
- Sørensen, C.S.; Syljuåsen, R.G.; Falck, J.; Schroeder, T.; Rönnstrand, L.; Khanna, K.K.; Zhou, B.-B.; Bartek, J.; Lukas, J. Chk1 regulates the S phase checkpoint by coupling the physiological turnover and ionizing radiation-induced accelerated proteolysis of Cdc25A. Cancer Cell 2003, 3, 247–258. [Google Scholar] [CrossRef]
- Kotsantis, P.; Silva, L.M.; Irmscher, S.; Jones, R.M.; Folkes, L.; Gromak, N.; Petermann, E. Increased global transcription activity as a mechanism of replication stress in cancer. Nat. Commun. 2016, 7, 13087. [Google Scholar] [CrossRef]
- Kausar, T.; Schreiber, J.S.; Karnak, D.; Parsels, L.A.; Parsels, J.D.; Davis, M.A.; Zhao, L.; Maybaum, J.; Lawrence, T.S.; Morgan, M.A. Sensitization of Pancreatic Cancers to Gemcitabine Chemoradiation by WEE1 Kinase Inhibition Depends on Homologous Recombination Repair. Neoplasia 2015, 17, 757–766. [Google Scholar] [CrossRef]
- Parsels, L.A.; Parsels, J.D.; Tanska, D.M.; Maybaum, J.; Lawrence, T.S.; Morgan, M.A. The contribution of DNA replication stress marked by high-intensity, pan-nuclear γH2AX staining to chemosensitization by CHK1 and WEE1 inhibitors. Cell Cycle 2018, 17, 1076–1086. [Google Scholar] [CrossRef]
- Mitra, S.K.; Hanson, D.A.; Schlaepfer, D.D. Focal adhesion kinase: In command and control of cell motility. Nat. Rev. Mol. Cell Biol. 2005, 6, 56–68. [Google Scholar] [CrossRef]
- Jiang, H.; Hegde, S.; DeNardo, D.G. Tumor-associated fibrosis as a regulator of tumor immunity and response to immunotherapy. Cancer Immunol. Immunother. 2017, 66, 1037–1048. [Google Scholar] [CrossRef]
- Tavora, B.; Reynolds, L.E.; Batista, S.; Demircioglu, F.; Fernandez, I.; Lechertier, T.; Lees, D.M.; Wong, P.-P.; Alexopoulou, A.; Elia, G.; et al. Endothelial-cell FAK targeting sensitizes tumours to DNA-Damaging therapy. Nature 2014, 514, 112–116. [Google Scholar] [CrossRef]
- Serrels, A.; Lund, T.; Serrels, B.; Byron, A.; McPherson, R.C.; von Kriegsheim, A.; Gómez-Cuadrado, L.; Canel, M.; Muir, M.; Ring, J.E.; et al. Nuclear FAK Controls Chemokine Transcription, Tregs, and Evasion of Anti-tumor Immunity. Cell 2015, 163, 160–173. [Google Scholar] [CrossRef]
- Jiang, H.; Hegde, S.; Knolhoff, B.L.; Zhu, Y.; Herndon, J.M.; Meyer, M.A.; Nywening, T.M.; Hawkins, W.G.; Shapiro, I.M.; Weaver, D.T.; et al. Targeting focal adhesion kinase renders pancreatic cancers responsive to checkpoint immunotherapy. Nat. Med. 2016, 22, 851–860. [Google Scholar] [CrossRef]
| Reference | Phase | Setting | N | Systemic Agents | RT Scheme | RT Timing/Immunotherapy | Primary Endpoint | Note |
|---|---|---|---|---|---|---|---|---|
| Clinical trials with published results | ||||||||
| Lutz et al. 2011 [17] | II | Adjuvant | 60 | GVAX (cancer vaccine) | 28 × 1.8 Gy (+ 5-FU) | After 1st dose | mDFS: 17.3 mo. | |
| Picozzi et al. 2011 (ACOSOG Z05031) [18] | II | Adjuvant | 89 | Cisplatin-5FU + INFα2b | 28 × 1.8 Gy | At cycle 1 | OS at 18 mo.: 69% | Toxicity failure (95% grade ≥3) |
| Crane et al. 2011 [19] | II | Unresectable LA | 69 | Gemcitabine + Oxaliplatin + Cetuximab | 28 × 1.8 Gy (+capecitabine) | After 4 cycles | 1y-OS: 66% | |
| Hardacre et al. 2013 [20] | II | Adjuvant | 70 | Gemcitabine + Algenpantucel-L (cancer vaccine) | 28 × 1.8 Gy (+ 5-FU) | At 3rd dose | 1y-DFS: 62% | |
| Chan et al. 2016 [21] | I | Neoadjuvant R/BR/LA | 21 | Vorinostat (HDAC inhibitor) | 10 × 3 Gy | Day 1 | MTD | mOS: 13 mo. |
| Brar et al. 2019 (abstract) [22] NCT02311361 | Ib/II | Unresectable + nonresponder to chemotherapy | 51/65 | Durvalumab +/− Tremelimumab +/− | 1 × 8 Gy or 5 × 5 Gy | Day 1 | Feasibility/Safety | Preliminary results |
| Cuneo et al. 2019 [23] | I | Neoadjuvant LA | 34 | Gemcitabine + Adavosertib (Wee1 inhibitor) | 25 × 2.1 Gy | At cycle 2 | MTD | mOS: 21.7 mo. |
| Murphy et al. 2019 [24] | II | Neoadjuvant LA | 49 | FFX + Losartan | 5 × 5 GyE (protons) or 10 × 3 Gy or 28 × 1.8 Gy (+capecitabine) | After cycle 8 | R0 RR: 61% | mOS: 31.4 mo. mPFS: 17.5 mo. |
| Lin et al. 2019 [25] | Ib/II | Neoadjuvant LA | 11 | Gemcitabine-5FU-leucovorin + Oregovomab (anti-CA-125) + Nelfinavir mesylate | 5 × 8 Gy | At week 11 | mPFS: 8.6 mo. | Closed prematurely due to outdated chemotherapy |
| Tuli et al. 2019 (abstract) [26] NCT03245541 | Ib/II | Neoadjuvant LA | 18/30 | Gem-Np + Durvalumab | 5 × 6.6 Gy | Day 8 | Safety, PFS, RR | Preliminary results – mPFS: 14 mo., RR: 50% |
| Xie et al. 2020 [27] | I | M+ (2nd line) | 59 | Durvalumab + Tremelimumab | 1 × 8 Gy or 5 × 5 Gy | Day 1 Day -3 to +1 | Safety | ORR: 5.1% |
| Parikh et al. 2021 [28] | II | M+ (MSS) | 25 | Nivolumab + Ipilimumab | 3 × 8 Gy (photons or protons) | At cycle 2 | DCR: 20% | ORR:12% |
| Poklepovic et al. 2021 (abstract) [29] NCT02349867 | I | Neoadjuvant R/BR/LA | 22 | Gemcitabine + Sorafenib + Vorinostat | 28 × 1.8 Gy | Concurrent | Recommended dose | Preliminary results |
| Rahma et al. 2021 (abstract) [30] | Ib/II | Neoadjuvant R/BR | 37 | Pembrolizumab +/− FFX | 28 × 1.8 Gy (+capecitabine) | Concurrent | Safety/TILs density | Preliminary results–mOS: 27.8 mo. TILs: No difference |
| Lee et al. 2021 (abstract) [31] NCT02648282 | II | Neoadjuvant LA | 58 | FFX or Gem-Np + GVAX + Pembrolizumab | 5 × 6.6 Gy | In cycle 2, concurrent | DMFS: 9.7 mo. (NS) | |
| Haldanarson et al. 2022 [32] (N064A Alliance) NCT00601627 | II | Unresectable LA | 52 | Panitumumab (EGFR inhibitor) + Gemcitabine | 28 × 1.8 Gy (+ 5FU) | Day 1 | 1 y-OS rate: 50% | OS and toxicity failure (88% grade ≥3) |
| Zhu et al. 2022 [33] | II | Post-op local recurrence (KRAS mut and PD-L1+) | 85 | Pembrolizumab + Trametinib | 5 × 7–8 Gy | 2 weeks before ICIs | mOS: 14.9 mo. | |
| Chen et al. 2022 [34] CHECKPAC | II | Refractory M+ | 84 | Nivolumab +/− Ipilimumab | 1 × 15 Gy (single lesion) | Day 1 | CBR: 37.2% for triple combination | ORR: 14% for the triple combination mOS: 3.8 mo. |
| Lierman et al. 2022 [35] (PARC) | rII | Inoperable PDAC | 68 | Gemcitabine +/− Cetuximab | 25 × 2.16 Gy | After 1 week | mPFS: 6.8 mo. | mOS: 14.2 mo. (NS) |
| Hewitt et al. 2022 [36] | III | Neoadjuvant BR/LA | 303 | FFX or Gem-Np +/− Algenpantucel-L | 28 × 1.8 Gy (+ 5-FU or capecitabine) | After 6 doses | mOS: 14.3 mo. (vs. 14.9 for SOC) | |
| Ongoing clinical trials | ||||||||
| NCT01072981 | III | Adjuvant | 722 | Gemcitabine +/−Algenpantucel-L | 28 × 1.8 Gy (+ 5-FU) | At 3rd dose | mOS | Recruitment completed |
| NCT03767582 | I/rII | Neoadjuvant LA | 30 | Chemotherapy + Nivolumab + CCR2/5 dual antagonist +/− GVAX | 5 × 6.6 Gy | 2–4 weeks after chemotherapy and 2–3 weeks before ICIs | Safety/TILs response | Recruiting |
NCT04331041 | rII | Neoadjuvant LA | 42 | FFX or Gem-Np +/− Defactinib (FAK inhibitor) | 5 × 10 Gy (MR-Linac) | Day 1, concurrent | PFS | Recruiting |
| NCT04172532 | Ib/rII | Neoadjuvant LA | 24 | Pebosertib (DNA-PK inhibitor) | 5 × ?Gy | Day 1 | MTD, PFS | Recruiting |
| NCT04106856 (SHAPER) | I | Neoadjuvant BR/LA | 20 | Losartan | 15 × ?Gy | Day 14 | Toxicity | Recruiting |
| NCT02305186 | Ib/II | Neoadjuvant R/BR | 68 | Pembrolizumab | 28 × 1.8Gy (+ capecitabine) | Concurrent | Number of TILs in resected tissue/Safety | Recruiting |
| NCT05411094 | I | Unresectable LA | 18 | Olaparib (PARP inhibitor) + Durvalumab | NR | In cycle 2, concurrent | MTD | Recruiting |
| NCT04247165 (LAPTOP) | Ib/II | BR/LA/M+ | 40 | Gem-Np + Nivolumab + Ipilimumab | 3 × 8 Gy (MR-Linac) | NR | Safety | Recruiting |
| NCT03915678 (AGADIR) | II | M+ solid tumors (including PDAC) | 247 | Atezolizumab + BDB001 (TLR 7/8 agonist) | 3–5 × 9–12 Gy | After 1st dose | ORR | Recruiting Basket trial |
| NCT05116917 (INFLUENCE) | II | M+ | 30 | Nivolumab + Ipilimumab + Influenza vaccine | 1 × 15 Gy | Day 1 | ORR | Recruiting |
| NCT05088889 | I | M+ | 10 | Nivolumab + Ipilimumab + | 3 × 8 Gy + 1 × 2 Gy for nonresponder | Day 1 | ORR | Recruiting |
| NCT04361162 | II | M+ (MSS) | 30 | Nivolumab + Ipilimumab | NR (3D) | Week 1 of 1st cycle | ORR | Active, not recruiting |
| NCT04050085 | I | Refractory M+ | 6 | Nivolumab + SD-101 (TLR-9 agonist) | 5 × 6–10 Gy | Day 1 | Safety | Active, not recruiting |
| NCT03490760 | II | Refractory M+ | 39 | Durvalumab | 3 × 8 Gy | At week 5 | PFS | Active, not recruiting |
| NCT03161379 | II | Neoadjuvant BR | 30 | Cyclophosphamide + GVAX + Nivolumab | 5 × 6.6 Gy | At 2nd dose | CD8 count (cell/ mm3) in the TME | Active, not recruiting |
| NCT03563248 (SU2C) | rII | Neoadjuvant BR/LA | 168 | FFX +/− Losartan +/− Nivolumab | NR (SBRT) | Concurrent | R0 RR | Active, not recruiting |
| NCT01595321 | I | Adjuvant | 19 | FFX + Cyclophosphamide + GVAX | 5 × 6.6 Gy | After 1st dose | Toxicity | Active, not recruiting |
| NCT04098432 | Ib/II | Neoadjuvant LA | 15 | FFX + Nivolumab | 4 × 8 Gy | Before ICI | PFS | Active, not recruiting |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Benkhaled, S.; Peters, C.; Jullian, N.; Arsenijevic, T.; Navez, J.; Van Gestel, D.; Moretti, L.; Van Laethem, J.-L.; Bouchart, C. Combination, Modulation and Interplay of Modern Radiotherapy with the Tumor Microenvironment and Targeted Therapies in Pancreatic Cancer: Which Candidates to Boost Radiotherapy? Cancers 2023, 15, 768. https://doi.org/10.3390/cancers15030768
Benkhaled S, Peters C, Jullian N, Arsenijevic T, Navez J, Van Gestel D, Moretti L, Van Laethem J-L, Bouchart C. Combination, Modulation and Interplay of Modern Radiotherapy with the Tumor Microenvironment and Targeted Therapies in Pancreatic Cancer: Which Candidates to Boost Radiotherapy? Cancers. 2023; 15(3):768. https://doi.org/10.3390/cancers15030768
Chicago/Turabian StyleBenkhaled, Sofian, Cedric Peters, Nicolas Jullian, Tatjana Arsenijevic, Julie Navez, Dirk Van Gestel, Luigi Moretti, Jean-Luc Van Laethem, and Christelle Bouchart. 2023. "Combination, Modulation and Interplay of Modern Radiotherapy with the Tumor Microenvironment and Targeted Therapies in Pancreatic Cancer: Which Candidates to Boost Radiotherapy?" Cancers 15, no. 3: 768. https://doi.org/10.3390/cancers15030768
APA StyleBenkhaled, S., Peters, C., Jullian, N., Arsenijevic, T., Navez, J., Van Gestel, D., Moretti, L., Van Laethem, J.-L., & Bouchart, C. (2023). Combination, Modulation and Interplay of Modern Radiotherapy with the Tumor Microenvironment and Targeted Therapies in Pancreatic Cancer: Which Candidates to Boost Radiotherapy? Cancers, 15(3), 768. https://doi.org/10.3390/cancers15030768







