Multi-Parameter Analysis of Disseminated Tumor Cells (DTCs) in Early Breast Cancer Patients with Hormone-Receptor-Positive Tumors
Abstract
Simple Summary
Abstract
1. Introduction
2. Materials and Methods
2.1. Study Population and Informed Consent
2.2. Bone Marrow Aspirates
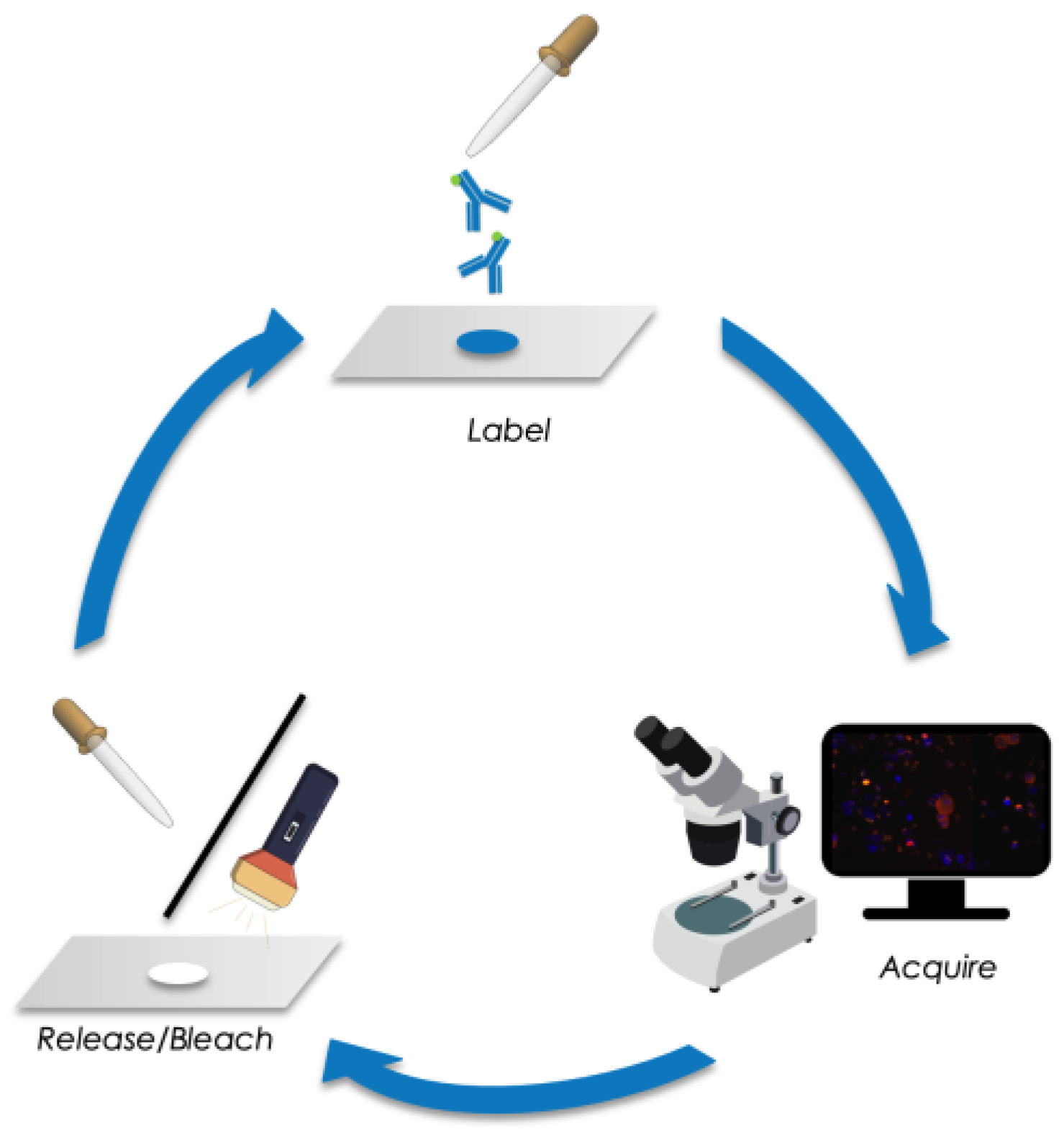
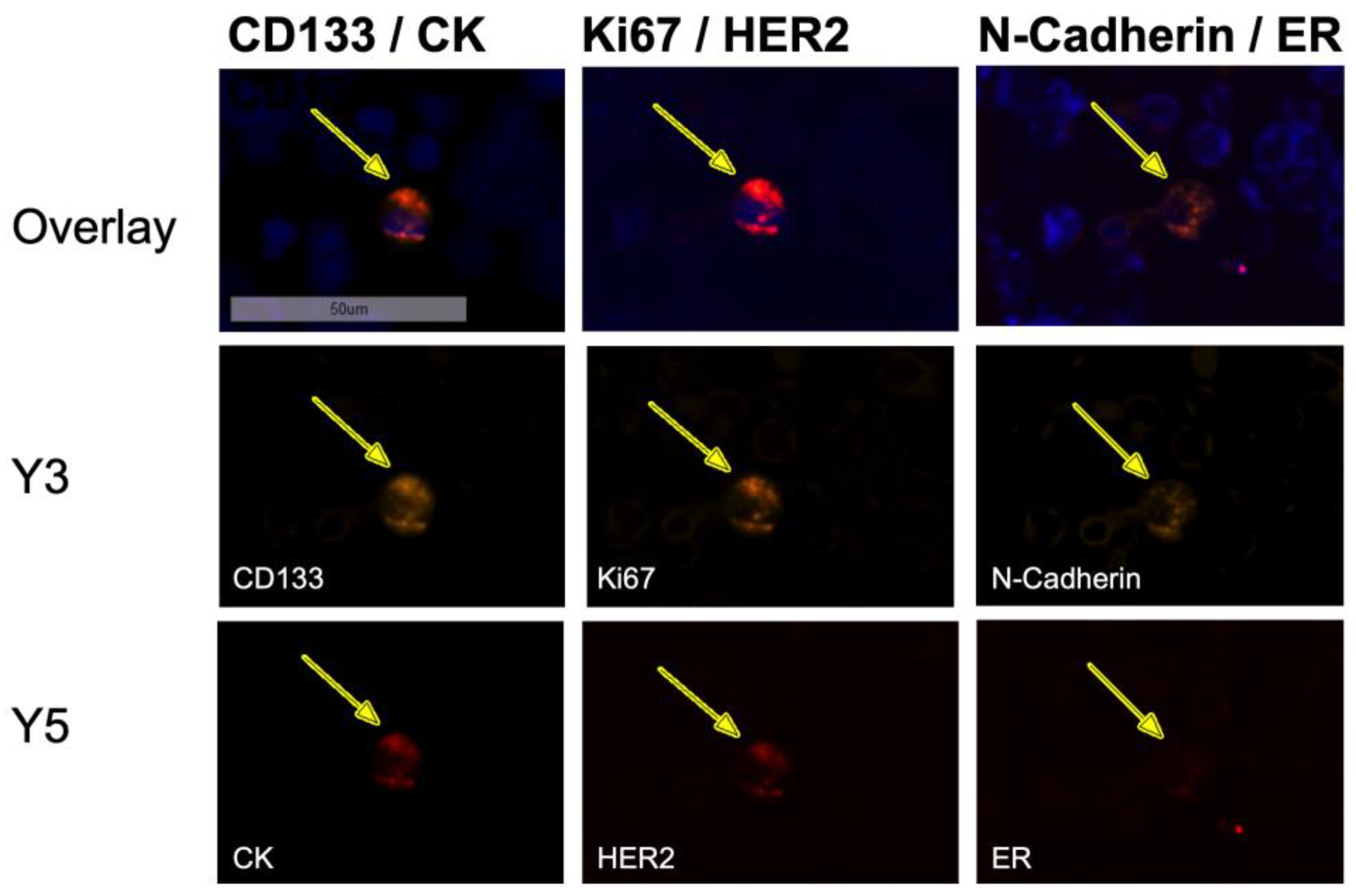
2.3. Sequential Immunofluorescence Staining and DTC Imaging
2.4. Cell Culture for Reference Slides
2.5. Statistical Analysis
3. Results
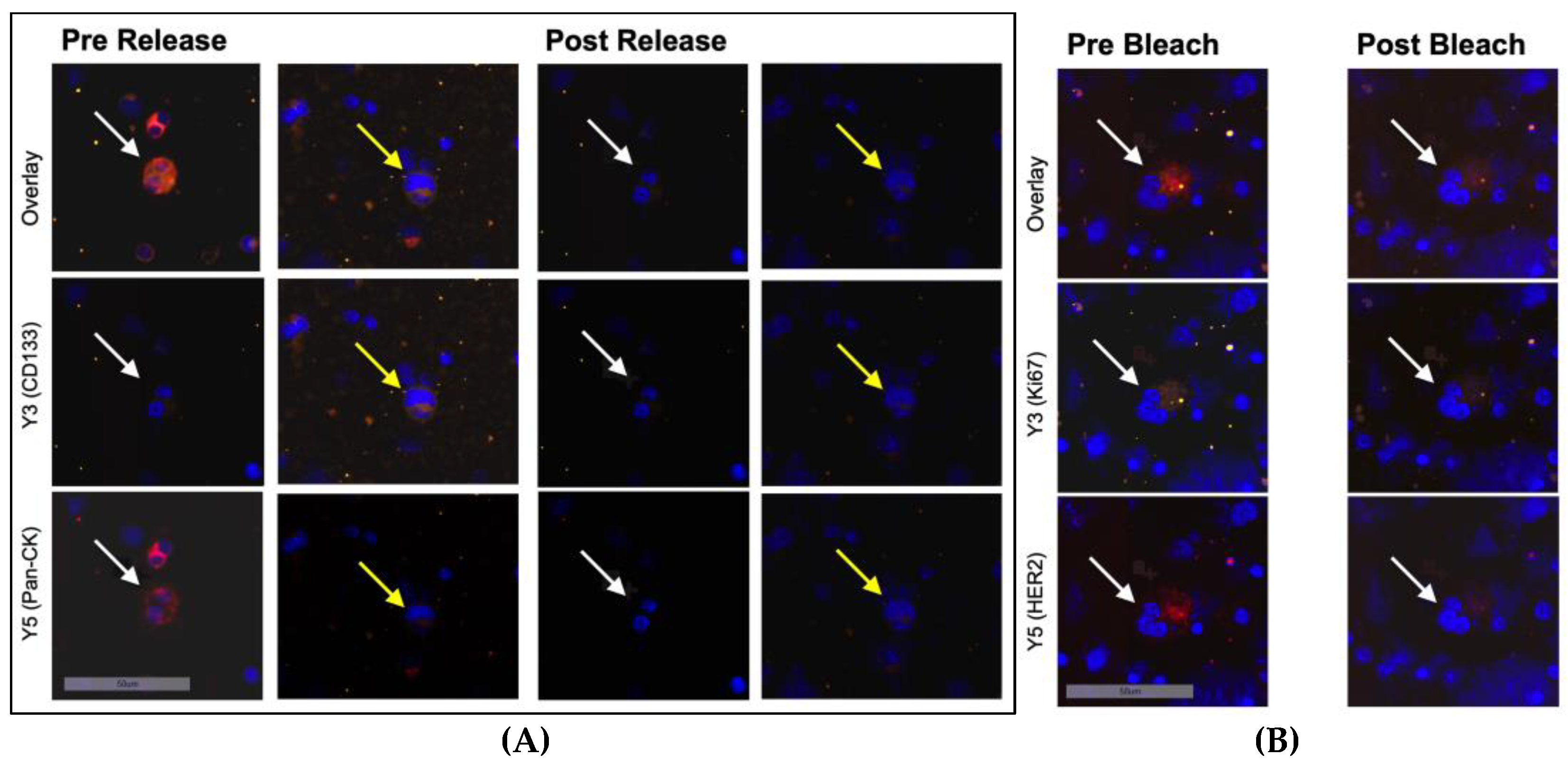
3.1. Staining of Reference Slides Using Sequential Immunofluorescence
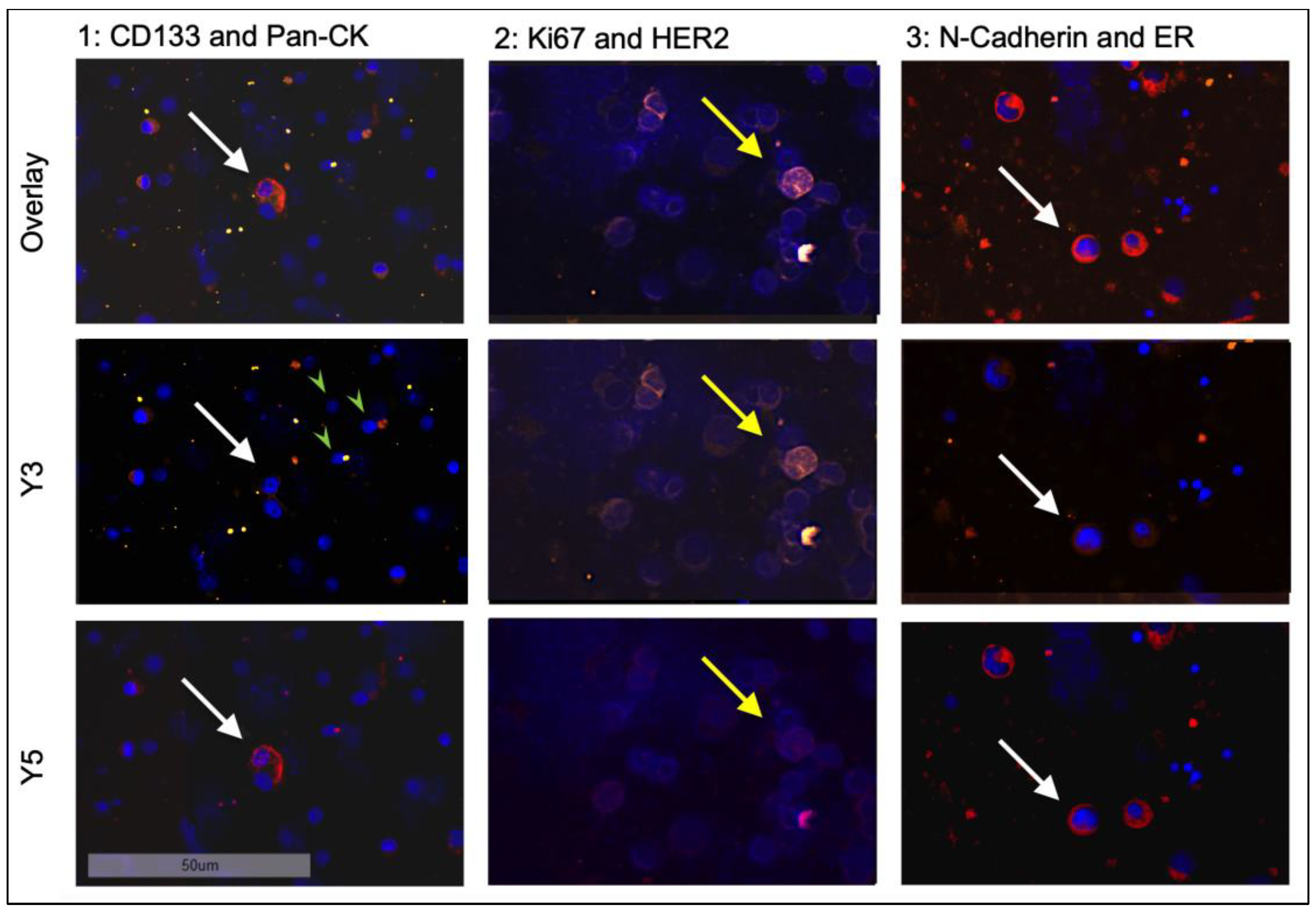
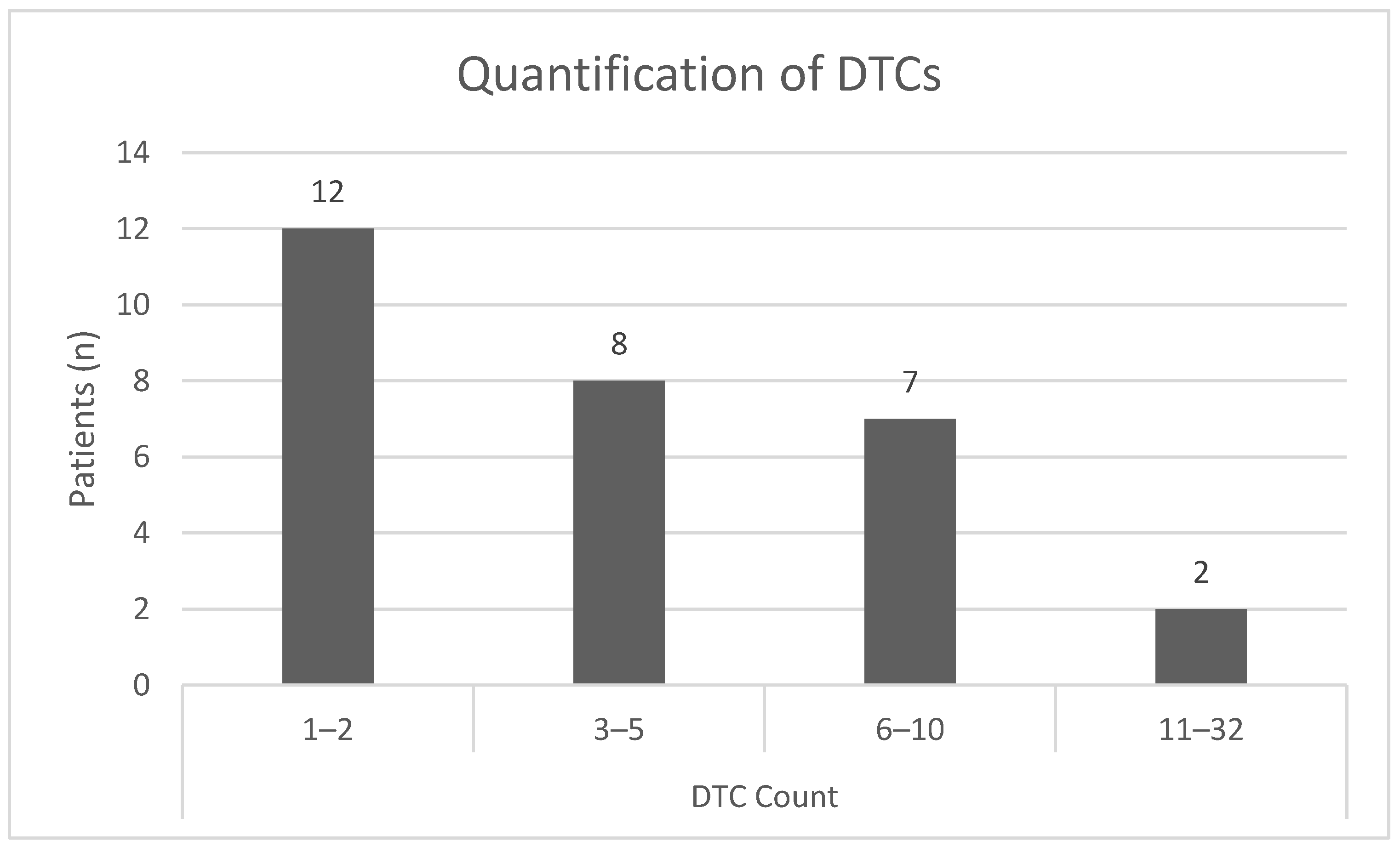
3.2. Identification and Quantification of DTC-Subtypes in HR+ Patients
3.3. DTC Subpopulations among Luminal A and Luminal B Tumors
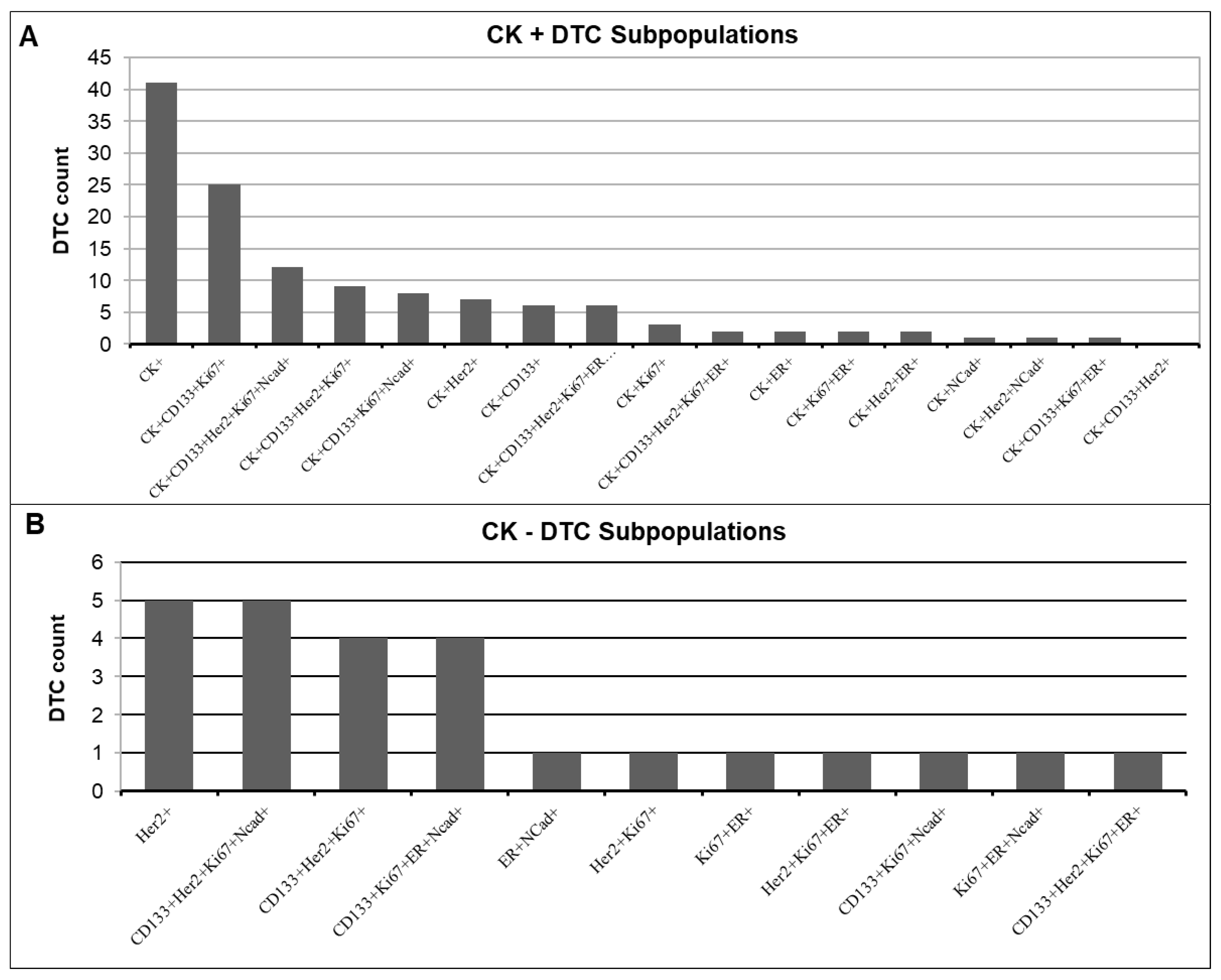
3.4. Detection of CK-Positive and CK-Negative DTC Subpopulations
3.5. Classification of DTCs According to Phenotypes
3.6. DTC-Subtypes and Clinical Data
3.7. Receptor Discordance between DTCs and Tumor Tissue Biopsy
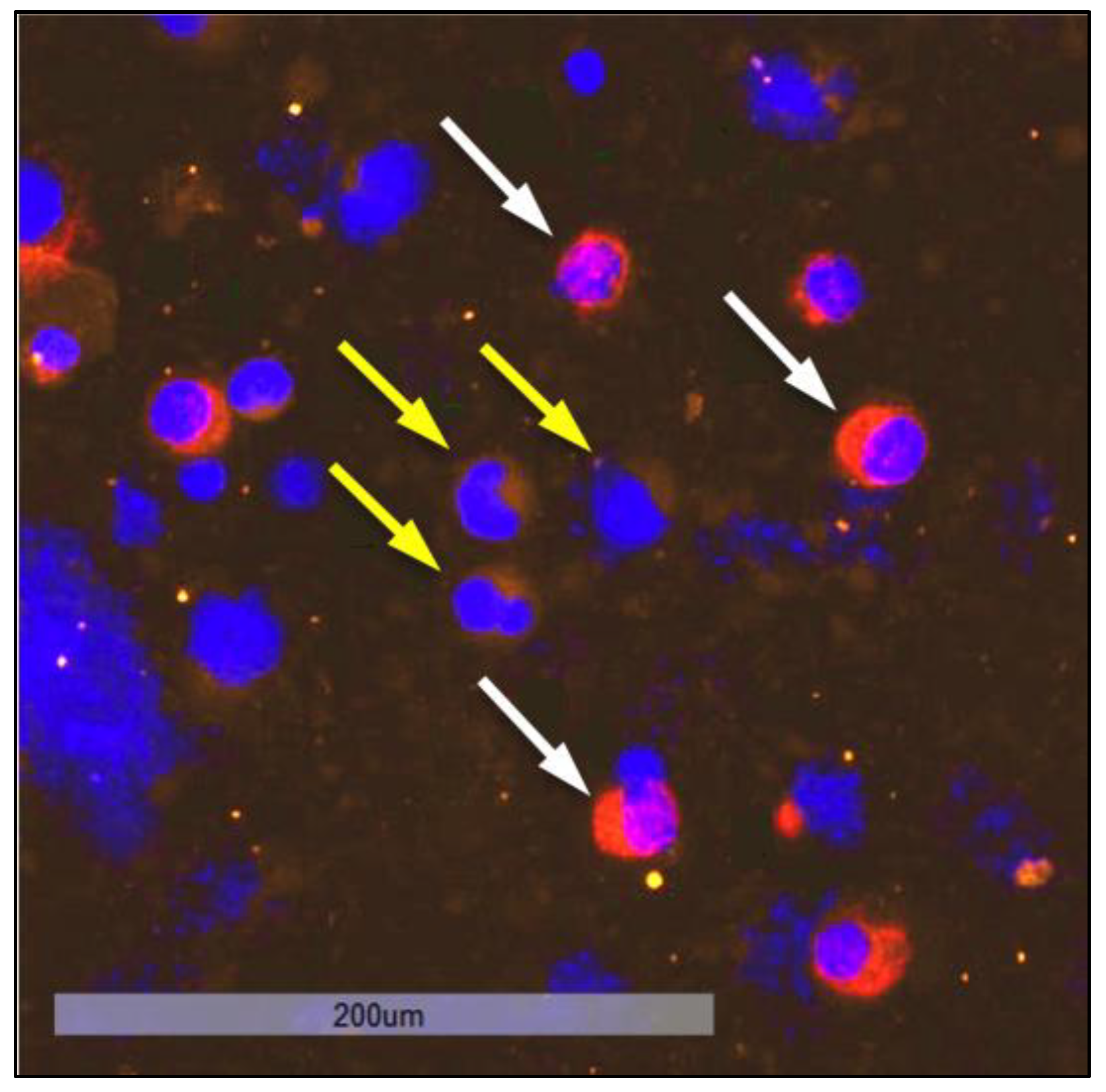
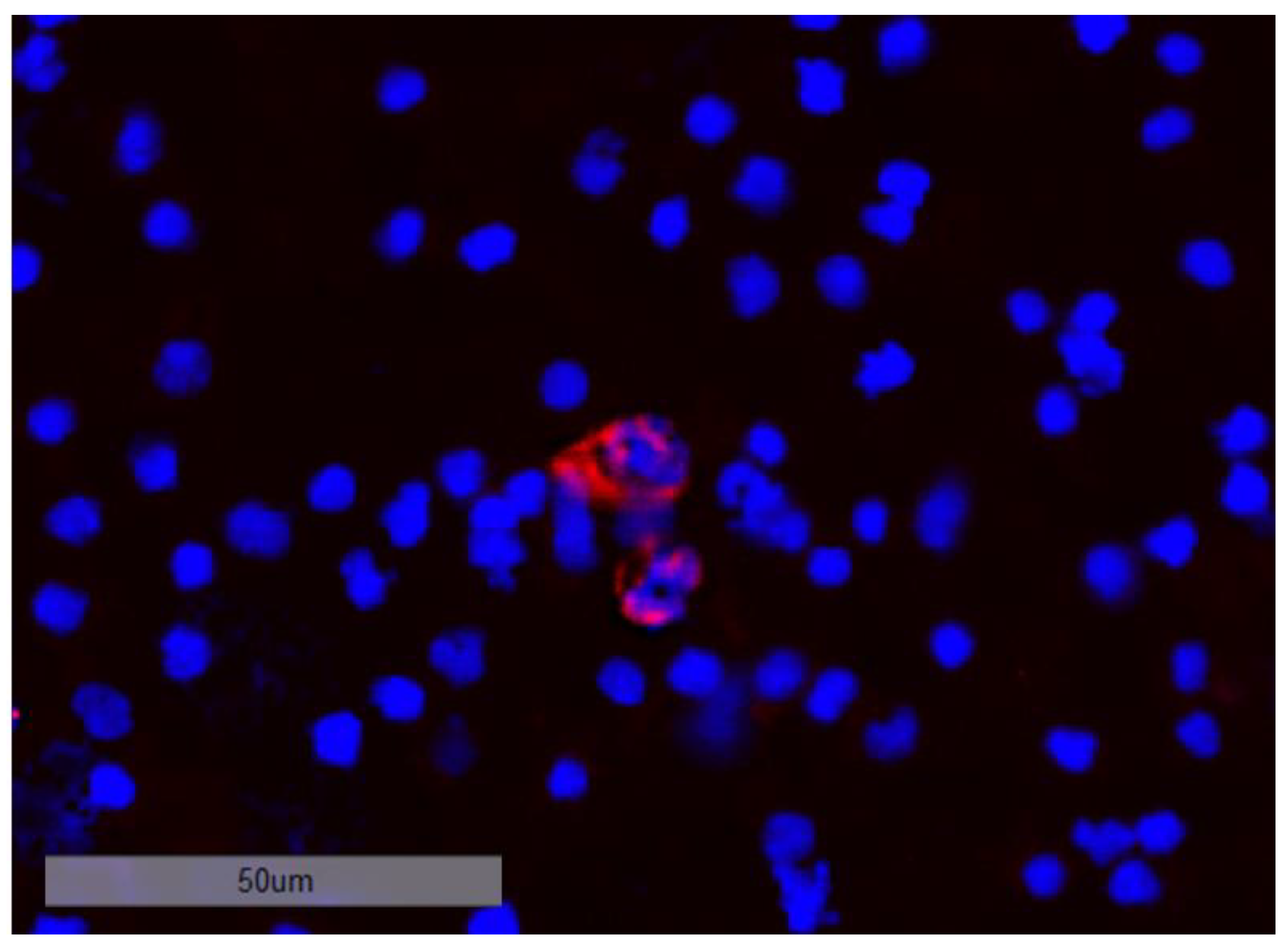
3.8. Investigation of DTC Clusters
3.9. DTC Subtype Detection vs. Standard Detection of CK+ Cells and Clinical Follow-Up
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Riggio, A.I.; Varley, K.E.; Welm, A.L. The lingering mysteries of metastatic recurrence in breast cancer. Br. J. Cancer 2021, 124, 13–26. [Google Scholar] [CrossRef] [PubMed]
- Ramamoorthi, G.; Kodumudi, K.; Gallen, C.; Zachariah, N.N.; Basu, A.; Albert, G.; Beyer, A.; Snyder, C.; Wiener, D.; Costa, R.L.B.; et al. Disseminated cancer cells in breast cancer: Mechanism of dissemination and dormancy and emerging insights on therapeutic opportunities. Semin. Cancer Biol. 2022, 78, 78–89. [Google Scholar] [CrossRef] [PubMed]
- Pantel, K.; Schlimok, G.; Braun, S.; Kutter, D.; Lindemann, F.; Schaller, G.; Funke, I.; Izbicki, J.R.; Riethmüller, G. Differential expression of proliferation-associated molecules in individual micrometastatic carcinoma cells. J. Natl. Cancer Inst. 1993, 85, 1419–1424. [Google Scholar] [CrossRef]
- Braun, S.; Vogl, F.D.; Naume, B.; Janni, W.; Osborne, M.P.; Coombes, R.C.; Schlimok, G.; Diel, I.J.; Gerber, B.; Gebauer, G.; et al. A pooled analysis of bone marrow micrometastasis in breast cancer. N. Engl. J. Med. 2005, 353, 793–802. [Google Scholar] [CrossRef] [PubMed]
- Janni, W.; Vogl, F.D.; Wiedswang, G.; Synnestvedt, M.; Fehm, T.; Jückstock, J.; Borgen, E.; Rack, B.; Braun, S.; Sommer, H.; et al. Persistence of disseminated tumor cells in the bone marrow of breast cancer patients predicts increased risk for relapse--a European pooled analysis. Clin. Cancer Res. Off. J. Am. Assoc. Cancer Res. 2011, 17, 2967–2976. [Google Scholar] [CrossRef] [PubMed]
- Hartkopf, A.D.; Taran, F.-A.; Wallwiener, M.; Hahn, M.; Becker, S.; Solomayer, E.-F.; Brucker, S.Y.; Fehm, T.N.; Wallwiener, D. Prognostic relevance of disseminated tumour cells from the bone marrow of early stage breast cancer patients—Results from a large single-centre analysis. Eur. J. Cancer 2014, 50, 2550–2559. [Google Scholar] [CrossRef] [PubMed]
- Hartkopf, A.D.; Brucker, S.Y.; Taran, F.-A.; Harbeck, N.; von Au, A.; Naume, B.; Pierga, J.-Y.; Hoffmann, O.; Beckmann, M.W.; Rydén, L.; et al. Disseminated tumour cells from the bone marrow of early breast cancer patients: Results from an international pooled analysis. Eur. J. Cancer 2021, 154, 128–137. [Google Scholar] [CrossRef]
- Nap, M.; van Wel, T.; Andrés, C.; Bellanger, L.; Bodenmüller, H.; Bonfrer, H.; Brundell, J.; Einarsson, R.; Erlandsson, A.; Johansson, A.; et al. Immunohistochemical profiles of 30 monoclonal antibodies against cytokeratins 8, 18 and 19. Second report of the TD5 workshop. Tumour Biol. J. Int. Soc. Oncodev. Biol. Med. 2001, 22, 4–10. [Google Scholar] [CrossRef]
- Vincent-Salomon, A.; Bidard, F.C.; Pierga, J.Y. Bone marrow micrometastasis in breast cancer: Review of detection methods, prognostic impact and biological issues. J. Clin. Pathol. 2008, 61, 570–576. [Google Scholar] [CrossRef]
- Fehm, T.; Braun, S.; Muller, V.; Janni, W.; Gebauer, G.; Marth, C.; Schindlbeck, C.; Wallwiener, D.; Borgen, E.; Naume, B.; et al. A concept for the standardized detection of disseminated tumor cells in bone marrow from patients with primary breast cancer and its clinical implementation. Cancer 2006, 107, 885–892. [Google Scholar] [CrossRef]
- Adjuvant bisphosphonate treatment in early breast cancer: Meta-analyses of individual patient data from randomised trials. Lancet 2015, 386, 1353–1361. [CrossRef] [PubMed]
- Hong, D.; Fritz, A.J.; Zaidi, S.K.; van Wijnen, A.J.; Nickerson, J.A.; Imbalzano, A.N.; Lian, J.B.; Stein, J.L.; Stein, G.S. Epithelial-to-mesenchymal transition and cancer stem cells contribute to breast cancer heterogeneity. J. Cell. Physiol. 2018, 233, 9136–9144. [Google Scholar] [CrossRef] [PubMed]
- Zou, H.; Yang, Z.; Chan, Y.-S.; Yeung, S.-K.A.; Alam, M.K.; Si, T.; Xu, T.; Yang, M. Single cell analysis of mechanical properties and EMT-related gene expression profiles in cancer fingers. iScience 2022, 25, 103917. [Google Scholar] [CrossRef] [PubMed]
- Jørgensen, C.L.T.; Forsare, C.; Bendahl, P.-O.; Falck, A.-K.; Fernö, M.; Lövgren, K.; Aaltonen, K.; Rydén, L. Expression of epithelial-mesenchymal transition-related markers and phenotypes during breast cancer progression. Breast Cancer Res. Treat. 2020, 181, 369–381. [Google Scholar] [CrossRef] [PubMed]
- Voutsadakis, I.A. Epithelial-Mesenchymal Transition (EMT) and Regulation of EMT Factors by Steroid Nuclear Receptors in Breast Cancer: A Review and in Silico Investigation. J. Clin. Med. 2016, 5, 11. [Google Scholar] [CrossRef] [PubMed]
- Goldhirsch, A.; Wood, W.C.; Coates, A.S.; Gelber, R.D.; Thürlimann, B.; Senn, H.-J. Strategies for subtypes--dealing with the diversity of breast cancer: Highlights of the St. Gallen International Expert Consensus on the Primary Therapy of Early Breast Cancer 2011. Ann. Oncol. Off. J. Eur. Soc. Med. Oncol. 2011, 22, 1736–1747. [Google Scholar] [CrossRef] [PubMed]
- Jäger, B.A.S.; Finkenzeller, C.; Bock, C.; Majunke, L.; Jueckstock, J.K.; Andergassen, U.; Neugebauer, J.K.; Pestka, A.; Friedl, T.W.P.; Jeschke, U.; et al. Estrogen Receptor and HER2 Status on Disseminated Tumor Cells and Primary Tumor in Patients with Early Breast Cancer. Transl. Oncol. 2015, 8, 509–516. [Google Scholar] [CrossRef] [PubMed]
- Krishnamurthy, S.; Bischoff, F.; Ann Mayer, J.; Wong, K.; Pham, T.; Kuerer, H.; Lodhi, A.; Bhattacharyya, A.; Hall, C.; Lucci, A. Discordance in HER2 gene amplification in circulating and disseminated tumor cells in patients with operable breast cancer. Cancer Med. 2013, 2, 226–233. [Google Scholar] [CrossRef]
- Krawczyk, N.; Banys, M.; Neubauer, H.; Solomayer, E.-F.; Gall, C.; Hahn, M.; Becker, S.; Bachmann, R.; Wallwiener, D.; Fehm, T. HER2 status on persistent disseminated tumor cells after adjuvant therapy may differ from initial HER2 status on primary tumor. Anticancer Res. 2009, 29, 4019–4024. [Google Scholar]
- Lüönd, F.; Sugiyama, N.; Bill, R.; Bornes, L.; Hager, C.; Tang, F.; Santacroce, N.; Beisel, C.; Ivanek, R.; Bürglin, T.; et al. Distinct contributions of partial and full EMT to breast cancer malignancy. Dev. Cell 2021, 56, 3203–3221.e11. [Google Scholar] [CrossRef]
- Felipe Lima, J.; Nofech-Mozes, S.; Bayani, J.; Bartlett, J.M.S. EMT in Breast Carcinoma—A Review. J. Clin. Med. 2016, 5, 65. [Google Scholar] [CrossRef] [PubMed]
- Thiery, J.P.; Lim, C.T. Tumor dissemination: An EMT affair. Cancer Cell 2013, 23, 272–273. [Google Scholar] [CrossRef] [PubMed]
- Nel, I.; Herzog, H.; Aktas, B. Combined Analysis of Disseminated Tumor Cells (DTCs) and Circulating Tumor DNA (ctDNA) in a Patient Suffering from Triple Negative Breast Cancer Revealed Elevated Risk. Front. Biosci. Landmark Ed. 2022, 27, 208. [Google Scholar] [CrossRef] [PubMed]
- R Foundation for Statistical Computing. R: A Language and Environment for Statistical Computing; R Core Team: Vienna, Austria; Available online: https://www.r-project.org/ (accessed on 14 October 2022).
- The Jamovi Project; Version 2.3; Jamovi: Sydney, Australia, 2022; Available online: https://www.jamovi.org (accessed on 14 October 2022).
- Fehm, T.; Hoffmann, O.; Aktas, B.; Becker, S.; Solomayer, E.F.; Wallwiener, D.; Kimmig, R.; Kasimir-Bauer, S. Detection and characterization of circulating tumor cells in blood of primary breast cancer patients by RT-PCR and comparison to status of bone marrow disseminated cells. Breast Cancer Res. BCR 2009, 11, R59. [Google Scholar] [CrossRef] [PubMed]
- Andergassen, U.; Vogl, A.; Mumm, J.-N.; Kölbl, A.C.; Hutter, S.; Rack, B.; Friese, K.; Jeschke, U. Immunocytochemical Characterization of Disseminated Tumour Cells from Bone Marrow of Breast Cancer Patients. Anticancer Res. 2016, 36, 3217–3222. [Google Scholar] [PubMed]
- Rack, B.; Zombirt, E.; Trapp, E.; Jückstock, J.; Andergassen, U.; Neugebauer, J.; Kost, B.; Weissenbacher, T.; Jeschke, U.; Schindlbeck, C.; et al. Comparison of HER2 Expression in Primary Tumor and Disseminated Tumor Cells in the Bone Marrow of Breast Cancer Patients. Oncology 2016, 90, 232–238. [Google Scholar] [CrossRef] [PubMed]
- Effenberger, K.E.; Borgen, E.; Eulenburg, C.Z.; Bartkowiak, K.; Grosser, A.; Synnestvedt, M.; Kaaresen, R.; Brandt, B.; Nesland, J.M.; Pantel, K.; et al. Detection and clinical relevance of early disseminated breast cancer cells depend on their cytokeratin expression pattern. Breast Cancer Res. Treat. 2011, 125, 729–738. [Google Scholar] [CrossRef]
- Balic, M.; Lin, H.; Young, L.; Hawes, D.; Giuliano, A.; McNamara, G.; Datar, R.H.; Cote, R.J. Most early disseminated cancer cells detected in bone marrow of breast cancer patients have a putative breast cancer stem cell phenotype. Clin. Cancer Res. Off. J. Am. Assoc. Cancer Res. 2006, 12, 5615–5621. [Google Scholar] [CrossRef]
- Fehm, T.; Krawczyk, N.; Solomayer, E.-F.; Becker-Pergola, G.; Dürr-Störzer, S.; Neubauer, H.; Seeger, H.; Staebler, A.; Wallwiener, D.; Becker, S. ERalpha-status of disseminated tumour cells in bone marrow of primary breast cancer patients. Breast Cancer Res. BCR 2008, 10, R76. [Google Scholar] [CrossRef]
- Axelrod, H.D.; Pienta, K.J.; Valkenburg, K.C. Optimization of Immunofluorescent Detection of Bone Marrow Disseminated Tumor Cells. Biol. Proced. Online 2018, 20, 13. [Google Scholar] [CrossRef]
- Vidula, N.; Greenberg, S.; Petrillo, L.; Hwang, J.; Melisko, M.; Goga, A.; Moasser, M.; Magbanua, M.; Park, J.W.; Rugo, H.S. Evaluation of disseminated tumor cells and circulating tumor cells in patients with breast cancer receiving adjuvant zoledronic acid. NPJ Breast Cancer 2021, 7, 113. [Google Scholar] [CrossRef] [PubMed]
- Rack, B.; Jückstock, J.; Günthner-Biller, M.; Andergassen, U.; Neugebauer, J.; Hepp, P.; Schoberth, A.; Mayr, D.; Zwingers, T.; Schindlbeck, C.; et al. Trastuzumab clears HER2/neu-positive isolated tumor cells from bone marrow in primary breast cancer patients. Arch. Gynecol. Obstet. 2012, 285, 485–492. [Google Scholar] [CrossRef] [PubMed]
- Kallergi, G.; Hoffmann, O.; Bittner, A.-K.; Papadimitriou, L.; Katsarou, S.D.; Zacharopoulou, N.; Zervakis, M.; Sfakianakis, S.; Stournaras, C.; Georgoulias, V.; et al. CXCR4 and JUNB double-positive disseminated tumor cells are detected frequently in breast cancer patients at primary diagnosis. Ther. Adv. Med. Oncol. 2020, 12, 1758835919895754. [Google Scholar] [CrossRef] [PubMed]
- Tjensvoll, K.; Nordgård, O.; Skjæveland, M.; Oltedal, S.; Janssen, E.A.M.; Gilje, B. Detection of disseminated tumor cells in bone marrow predict late recurrences in operable breast cancer patients. BMC Cancer 2019, 19, 1131. [Google Scholar] [CrossRef] [PubMed]
- Scholzen, T.; Gerdes, J. The Ki-67 protein: From the known and the unknown. J. Cell. Physiol. 2000, 182, 311–322. [Google Scholar] [CrossRef]
- Kumar, A.; Golani, A.; Kumar, L.D. EMT in breast cancer metastasis: An interplay of microRNAs, signaling pathways and circulating tumor cells. Front. Biosci. Landmark Ed. 2020, 25, 979–1010. [Google Scholar] [CrossRef]
- Fridrichova, I.; Kalinkova, L.; Ciernikova, S. Clinical Relevancy of Circulating Tumor Cells in Breast Cancer: Epithelial or Mesenchymal Characteristics, Single Cells or Clusters? Int. J. Mol. Sci. 2022, 23, 12141. [Google Scholar] [CrossRef]
- Nieto, M.A.; Huang, R.Y.-J.; Jackson, R.A.; Thiery, J.P. EMT: 2016. Cell 2016, 166, 21–45. [Google Scholar] [CrossRef]
- Wang, H.; Yu, C.; Gao, X.; Welte, T.; Muscarella, A.M.; Tian, L.; Zhao, H.; Zhao, Z.; Du, S.; Tao, J.; et al. The osteogenic niche promotes early-stage bone colonization of disseminated breast cancer cells. Cancer Cell 2015, 27, 193–210. [Google Scholar] [CrossRef]
- Magbanua, M.J.M.; Rugo, H.S.; Hauranieh, L.; Roy, R.; Scott, J.H.; Lee, J.C.; Hsiao, F.; Sosa, E.V.; Van’t Veer, L.; Esserman, L.J.; et al. Genomic and expression profiling reveal molecular heterogeneity of disseminated tumor cells in bone marrow of early breast cancer. NPJ Breast Cancer 2018, 4, 31. [Google Scholar] [CrossRef] [PubMed]
- Barzegar Behrooz, A.; Syahir, A.; Ahmad, S. CD133: Beyond a cancer stem cell biomarker. J. Drug Target. 2019, 27, 257–269. [Google Scholar] [CrossRef] [PubMed]
- Zhang, X.H.-F.; Jin, X.; Malladi, S.; Zou, Y.; Wen, Y.H.; Brogi, E.; Smid, M.; Foekens, J.A.; Massagué, J. Selection of bone metastasis seeds by mesenchymal signals in the primary tumor stroma. Cell 2013, 154, 1060–1073. [Google Scholar] [CrossRef] [PubMed]
- Kim, R.S.; Avivar-Valderas, A.; Estrada, Y.; Bragado, P.; Sosa, M.S.; Aguirre-Ghiso, J.A.; Segall, J.E. Dormancy signatures and metastasis in estrogen receptor positive and negative breast cancer. PLoS ONE 2012, 7, e35569. [Google Scholar] [CrossRef]
- Borgen, E.; Rypdal, M.C.; Sosa, M.S.; Renolen, A.; Schlichting, E.; Lønning, P.E.; Synnestvedt, M.; Aguirre-Ghiso, J.A.; Naume, B. NR2F1 stratifies dormant disseminated tumor cells in breast cancer patients. Breast Cancer Res. BCR 2018, 20, 120. [Google Scholar] [CrossRef] [PubMed]
- Spiliotaki, M.; Mavroudis, D.; Kapranou, K.; Markomanolaki, H.; Kallergi, G.; Koinis, F.; Kalbakis, K.; Georgoulias, V.; Agelaki, S. Evaluation of proliferation and apoptosis markers in circulating tumor cells of women with early breast cancer who are candidates for tumor dormancy. Breast Cancer Res. BCR 2014, 16, 485. [Google Scholar] [CrossRef]
- Harper, K.L.; Sosa, M.S.; Entenberg, D.; Hosseini, H.; Cheung, J.F.; Nobre, R.; Avivar-Valderas, A.; Nagi, C.; Girnius, N.; Davis, R.J.; et al. Mechanism of early dissemination and metastasis in Her2+ mammary cancer. Nature 2016, 540, 588–592. [Google Scholar] [CrossRef]
- Fehm, T.; Müller, V.; Aktas, B.; Janni, W.; Schneeweiss, A.; Stickeler, E.; Lattrich, C.; Löhberg, C.R.; Solomayer, E.; Rack, B.; et al. HER2 status of circulating tumor cells in patients with metastatic breast cancer: A prospective, multicenter trial. Breast Cancer Res. Treat. 2010, 124, 403–412. [Google Scholar] [CrossRef]
- Pestrin, M.; Bessi, S.; Galardi, F.; Truglia, M.; Biggeri, A.; Biagioni, C.; Cappadona, S.; Biganzoli, L.; Giannini, A.; Di Leo, A. Correlation of HER2 status between primary tumors and corresponding circulating tumor cells in advanced breast cancer patients. Breast Cancer Res. Treat. 2009, 118, 523–530. [Google Scholar] [CrossRef]
- Meng, S.; Tripathy, D.; Shete, S.; Ashfaq, R.; Haley, B.; Perkins, S.; Beitsch, P.; Khan, A.; Euhus, D.; Osborne, C.; et al. HER-2 gene amplification can be acquired as breast cancer progresses. Proc. Natl. Acad. Sci. USA 2004, 101, 9393–9398. [Google Scholar] [CrossRef]
- Lang, J.E.; Mosalpuria, K.; Cristofanilli, M.; Krishnamurthy, S.; Reuben, J.; Singh, B.; Bedrosian, I.; Meric-Bernstam, F.; Lucci, A. HER2 status predicts the presence of circulating tumor cells in patients with operable breast cancer. Breast Cancer Res. Treat. 2009, 113, 501–507. [Google Scholar] [CrossRef]
- Weydandt, L.; Nel, I.; Kreklau, A.; Horn, L.-C.; Aktas, B. Heterogeneity between Core Needle Biopsy and Synchronous Axillary Lymph Node Metastases in Early Breast Cancer Patients-A Comparison of HER2, Estrogen and Progesterone Receptor Expression Profiles during Primary Treatment Regime. Cancers 2022, 14, 1863. [Google Scholar] [CrossRef] [PubMed]
- Kraemer, B.; Rothmund, R.; Banys, M.; Krawczyk, N.; Solomayer, E.-F.; Mack, C.; Wallwiener, D.; Fehm, T. Impaired bone microenvironment: Correlation between bone density and presence of disseminated tumor cells. Anticancer Res. 2011, 31, 4423–4428. [Google Scholar] [PubMed]
- Pierga, J.-Y.; Bonneton, C.; Vincent-Salomon, A.; de Cremoux, P.; Nos, C.; Blin, N.; Pouillart, P.; Thiery, J.-P.; Magdelénat, H. Clinical significance of immunocytochemical detection of tumor cells using digital microscopy in peripheral blood and bone marrow of breast cancer patients. Clin. Cancer Res. Off. J. Am. Assoc. Cancer Res. 2004, 10, 1392–1400. [Google Scholar] [CrossRef] [PubMed]
- Bidard, F.-C.; Peeters, D.J.; Fehm, T.; Nolé, F.; Gisbert-Criado, R.; Mavroudis, D.; Grisanti, S.; Generali, D.; Garcia-Saenz, J.A.; Stebbing, J.; et al. Clinical validity of circulating tumour cells in patients with metastatic breast cancer: A pooled analysis of individual patient data. Lancet Oncol. 2014, 15, 406–414. [Google Scholar] [CrossRef]
- Gebauer, G.; Fehm, T.; Merkle, E.; Beck, E.P.; Lang, N.; Jäger, W. Epithelial cells in bone marrow of breast cancer patients at time of primary surgery: Clinical outcome during long-term follow-up. J. Clin. Oncol. Off. J. Am. Soc. Clin. Oncol. 2001, 19, 3669–3674. [Google Scholar] [CrossRef]
- Braun, S.; Pantel, K.; Müller, P.; Janni, W.; Hepp, F.; Kentenich, C.R.; Gastroph, S.; Wischnik, A.; Dimpfl, T.; Kindermann, G.; et al. Cytokeratin-positive cells in the bone marrow and survival of patients with stage I, II, or III breast cancer. N. Engl. J. Med. 2000, 342, 525–533. [Google Scholar] [CrossRef]
- Wiedswang, G.; Borgen, E.; Kåresen, R.; Kvalheim, G.; Nesland, J.M.; Qvist, H.; Schlichting, E.; Sauer, T.; Janbu, J.; Harbitz, T.; et al. Detection of isolated tumor cells in bone marrow is an independent prognostic factor in breast cancer. J. Clin. Oncol. Off. J. Am. Soc. Clin. Oncol. 2003, 21, 3469–3478. [Google Scholar] [CrossRef]
- Diel, I.J.; Solomayer, E.F.; Costa, S.D.; Gollan, C.; Goerner, R.; Wallwiener, D.; Kaufmann, M.; Bastert, G. Reduction in new metastases in breast cancer with adjuvant clodronate treatment. N. Engl. J. Med. 1998, 339, 357–363. [Google Scholar] [CrossRef]
- Diel, I.J.; Jaschke, A.; Solomayer, E.F.; Gollan, C.; Bastert, G.; Sohn, C.; Schuetz, F. Adjuvant oral clodronate improves the overall survival of primary breast cancer patients with micrometastases to the bone marrow: A long-term follow-up. Ann. Oncol. Off. J. Eur. Soc. Med. Oncol. 2008, 19, 2007–2011. [Google Scholar] [CrossRef]
- Powles, T.; Paterson, A.; McCloskey, E.; Schein, P.; Scheffler, B.; Tidy, A.; Ashley, S.; Smith, I.; Ottestad, L.; Kanis, J. Reduction in bone relapse and improved survival with oral clodronate for adjuvant treatment of operable breast cancer ISRCTN83688026. Breast Cancer Res. BCR 2006, 8, R13. [Google Scholar] [CrossRef]

| Clinical Parameter | n (n = 29) | % |
|---|---|---|
| Age | ||
| 30 to ≤49 | 11 | 38% |
| 50 to ≤64 | 11 | 38% |
| 65 to ≤74 | 4 | 14% |
| ≥75 | 3 | 10% |
| Gender | ||
| female | 28 | 97% |
| male | 1 | 3% |
| Molecular subtypes | ||
| Luminal A | 17 | 59% |
| Luminal B/ HER2− | 6 | 21% |
| Luminal B/ HER2+ | 6 | 21% |
| Tumor size | ||
| pT1mic to T1c | 18 | 62% |
| pT2 | 7 | 24% |
| pT3 to pT3b | 3 | 10% |
| pT4d | 1 | 3% |
| Lymph node status | ||
| N0 | 17 | 59% |
| N1 | 7 | 24% |
| N2 | 3 | 10% |
| N3 | 2 | 7% |
| Neoadjuvant therapy | ||
| No neoadjuvant therapy | 18 | 62% |
| Chemotherapy combined with targeted antibodies | 3 | 10% |
| Endocrine therapy | 8 | 28% |
| Ki67 in % | ||
| ≤15 | 19 | 66% |
| 16–49 | 7 | 24% |
| ≥50 | 3 | 10% |
| Menopausal status | ||
| pre- | 12 | 41% |
| peri- | 5 | 17% |
| post- | 12 | 41% |
| Cells | ZR75-1 | T98G | Hematopoietic Cells |
|---|---|---|---|
| Pan-CK | positive | negative | negative |
| CD133 | negative | positive | negative |
| HER2 | positive | negative | negative |
| Ki67 | positive | positive | negative |
| ER | positive | negative | negative |
| N-Cadherin | negative | positive | negative |
| DAPI | positive | positive | positive |
| Pat.-ID | Subtype * Tumor | Total DTCs | Total CK+ DTC | Total HER2+ DTC | Total Ki67+ DTC | Total ER+ DTC | Total CD133+ DTC | Total Ncad+ DTC |
|---|---|---|---|---|---|---|---|---|
| 243 | 1 | 6 | 6 | 0 | 3 | 0 | 3 | 0 |
| 275 | 1 | 5 | 4 | 2 | 3 | 0 | 5 | 0 |
| 277 | 1 | 11 | 7 | 8 | 10 | 1 | 10 | 3 |
| 536 | 1 | 2 | 2 | 2 | 2 | 1 | 2 | 2 |
| 757 | 1 | 5 | 3 | 2 | 2 | 1 | 0 | 0 |
| 775 | 1 | 7 | 4 | 1 | 3 | 5 | 3 | 4 |
| 956 | 1 | 6 | 3 | 5 | 4 | 2 | 3 | 2 |
| 1116 | 1 | 32 | 28 | 4 | 8 | 0 | 8 | 5 |
| 1124 | 1 | 8 | 7 | 6 | 5 | 1 | 5 | 2 |
| 1348 | 1 | 3 | 3 | 2 | 3 | 1 | 3 | 1 |
| 1363 | 1 | 3 | 3 | 2 | 0 | 0 | 0 | 0 |
| 1442 | 1 | 1 | 1 | 1 | 0 | 0 | 0 | 0 |
| 1457 | 1 | 2 | 1 | 1 | 2 | 1 | 1 | 2 |
| 1559 | 1 | 5 | 4 | 2 | 3 | 1 | 3 | 1 |
| 1583 | 1 | 10 | 10 | 1 | 4 | 0 | 6 | 2 |
| 1824 | 1 | 10 | 8 | 5 | 10 | 1 | 10 | 5 |
| 1864 | 1 | 1 | 1 | 1 | 0 | 0 | 0 | 0 |
| sum | 17 | 117 | 95 | 45 | 62 | 15 | 62 | 29 |
| mean | 6.88 | 5.59 | 2.651 | 3.65 | 0.88 | 3.64 | 1.71 | |
| 542 | 2 | 5 | 3 | 0 | 4 | 2 | 3 | 3 |
| 563 | 2 | 1 | 1 | 1 | 1 | 1 | 1 | 0 |
| 878 | 2 | 2 | 2 | 0 | 1 | 0 | 1 | 0 |
| 1049 | 2 | 2 | 2 | 1 | 1 | 0 | 2 | 0 |
| 1519 | 2 | 1 | 1 | 0 | 1 | 1 | 0 | 0 |
| 1930 | 2 | 9 | 8 | 4 | 9 | 1 | 8 | 3 |
| sum | 6 | 20 | 17 | 6 | 17 | 5 | 15 | 6 |
| mean | 3.33 | 2.83 | 1.00 | 2.83 | 0.83 | 2.50 | 1.00 | |
| 130 | 3 | 5 | 5 | 0 | 1 | 0 | 0 | 0 |
| 196 | 3 | 1 | 1 | 0 | 0 | 0 | 0 | 0 |
| 535 | 3 | 4 | 4 | 3 | 4 | 1 | 3 | 2 |
| 552 | 3 | 2 | 2 | 0 | 1 | 1 | 1 | 0 |
| 763 | 3 | 2 | 2 | 2 | 2 | 2 | 2 | 2 |
| 876 | 3 | 2 | 2 | 0 | 0 | 0 | 0 | 1 |
| sum | 6 | 16 | 16 | 5 | 8 | 4 | 6 | 5 |
| mean | 2.67 | 2.67 | 0.83 | 1.33 | 0.67 | 1.00 | 0.84 | |
| Total | 29 | 153 | 128 | 56 | 87 | 24 | 83 | 40 |
| Ki67+ CK+ | HER2+ CK+ | CD133+ CK+ | ER+ CK+ | Ncad+ CK+ | |
| n DTCs | 68 | 39 | 69 | 15 | 28 |
| % of CK+ DTCs | 53 | 30 | 54 | 12 | 22 |
| % of total DTCs | 44 | 25 | 45 | 10 | 18 |
| Ki67 + CK− | HER2 + CK− | CD133+ CK− | ER+ CK− | Ncad+ CK− | |
| n DTCs | 19 | 17 | 15 | 9 | 12 |
| % of CK- DTCs | 76 | 68 | 60 | 36 | 48 |
| % of all DTCs | 12 | 11 | 10 | 6 | 8 |
| Receptor | No Neoadjuvant Therapy n (%) | Endocrine Therapy n (%) | Chemo-/Targeted Therapy n (%) |
|---|---|---|---|
| Discordance ER | 8 (44%) | 2 (25%) | 2 (66%) |
| Concordance ER | 10 (56%) | 6 (75%) | 1 (33%) |
| Discordance HER2 | 14 (78%) | 7 (87,5%) | 2 (66%) |
| Concordance HER2 | 4 (22%) | 1 (12,5%) | 1 (33%) |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
König, T.; Dogan, S.; Höhn, A.K.; Weydandt, L.; Aktas, B.; Nel, I. Multi-Parameter Analysis of Disseminated Tumor Cells (DTCs) in Early Breast Cancer Patients with Hormone-Receptor-Positive Tumors. Cancers 2023, 15, 568. https://doi.org/10.3390/cancers15030568
König T, Dogan S, Höhn AK, Weydandt L, Aktas B, Nel I. Multi-Parameter Analysis of Disseminated Tumor Cells (DTCs) in Early Breast Cancer Patients with Hormone-Receptor-Positive Tumors. Cancers. 2023; 15(3):568. https://doi.org/10.3390/cancers15030568
Chicago/Turabian StyleKönig, Theresa, Senol Dogan, Anne Kathrin Höhn, Laura Weydandt, Bahriye Aktas, and Ivonne Nel. 2023. "Multi-Parameter Analysis of Disseminated Tumor Cells (DTCs) in Early Breast Cancer Patients with Hormone-Receptor-Positive Tumors" Cancers 15, no. 3: 568. https://doi.org/10.3390/cancers15030568
APA StyleKönig, T., Dogan, S., Höhn, A. K., Weydandt, L., Aktas, B., & Nel, I. (2023). Multi-Parameter Analysis of Disseminated Tumor Cells (DTCs) in Early Breast Cancer Patients with Hormone-Receptor-Positive Tumors. Cancers, 15(3), 568. https://doi.org/10.3390/cancers15030568






