Homologous Recombination Repair Gene Alterations Are Associated with Tumor Mutational Burden and Survival of Immunotherapy
Abstract
:Simple Summary
Abstract
1. Introduction
2. Materials and Methods
2.1. Patient Population
2.2. Cancer Genomic Testing with an NGS-Based Multiplex Gene Panel Assay (FoundationOne® CDx)
2.3. Statistical Analysis and Generating Figures
3. Results
3.1. Patients’ Characteristics
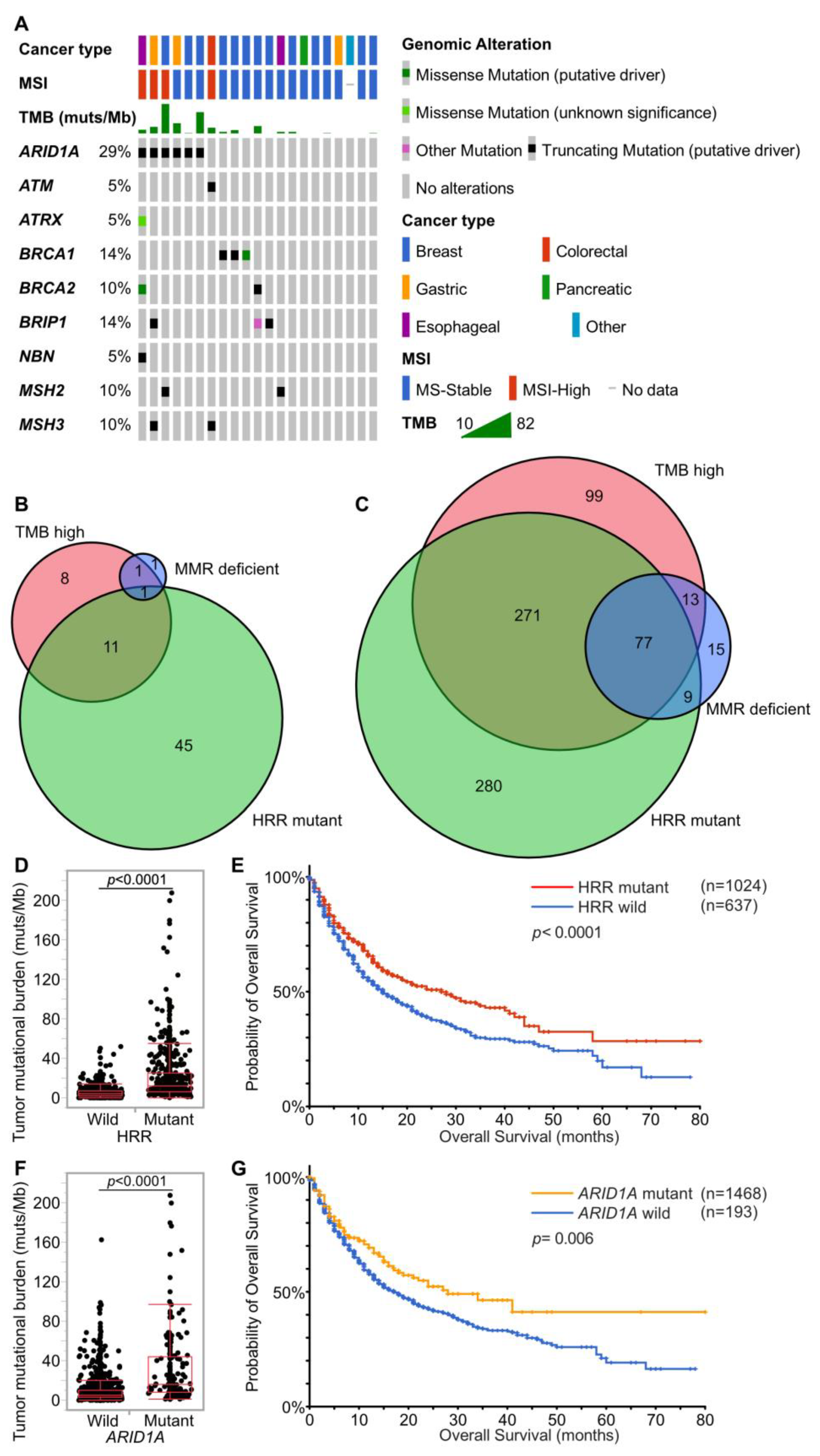
3.2. Genomic Characterization
3.3. Cases with MMR Deficient (dMMR), HRR Mutant (HRRm), MSI-High, and TMB-High
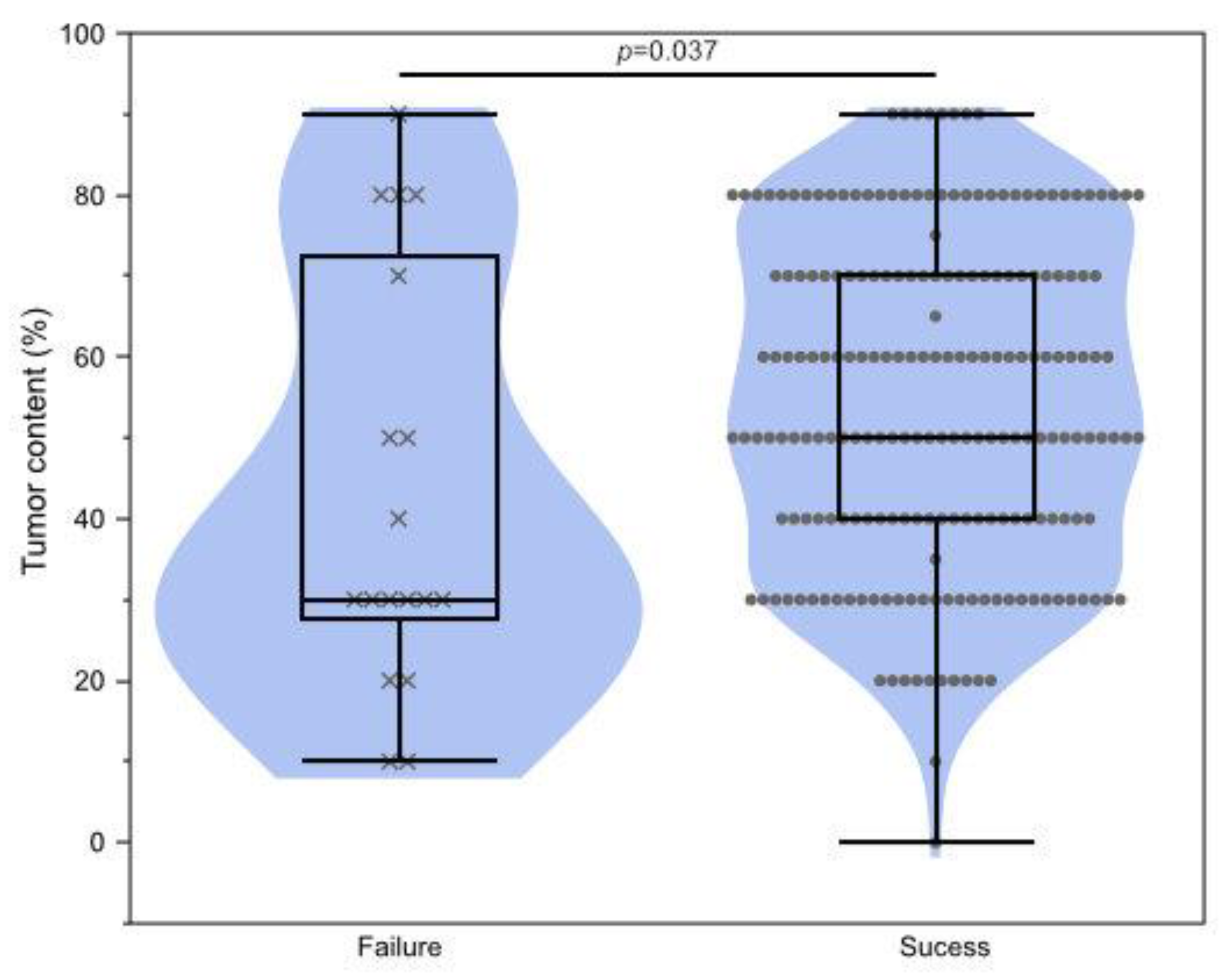
3.4. Clinical Factors Affecting Testing Failure
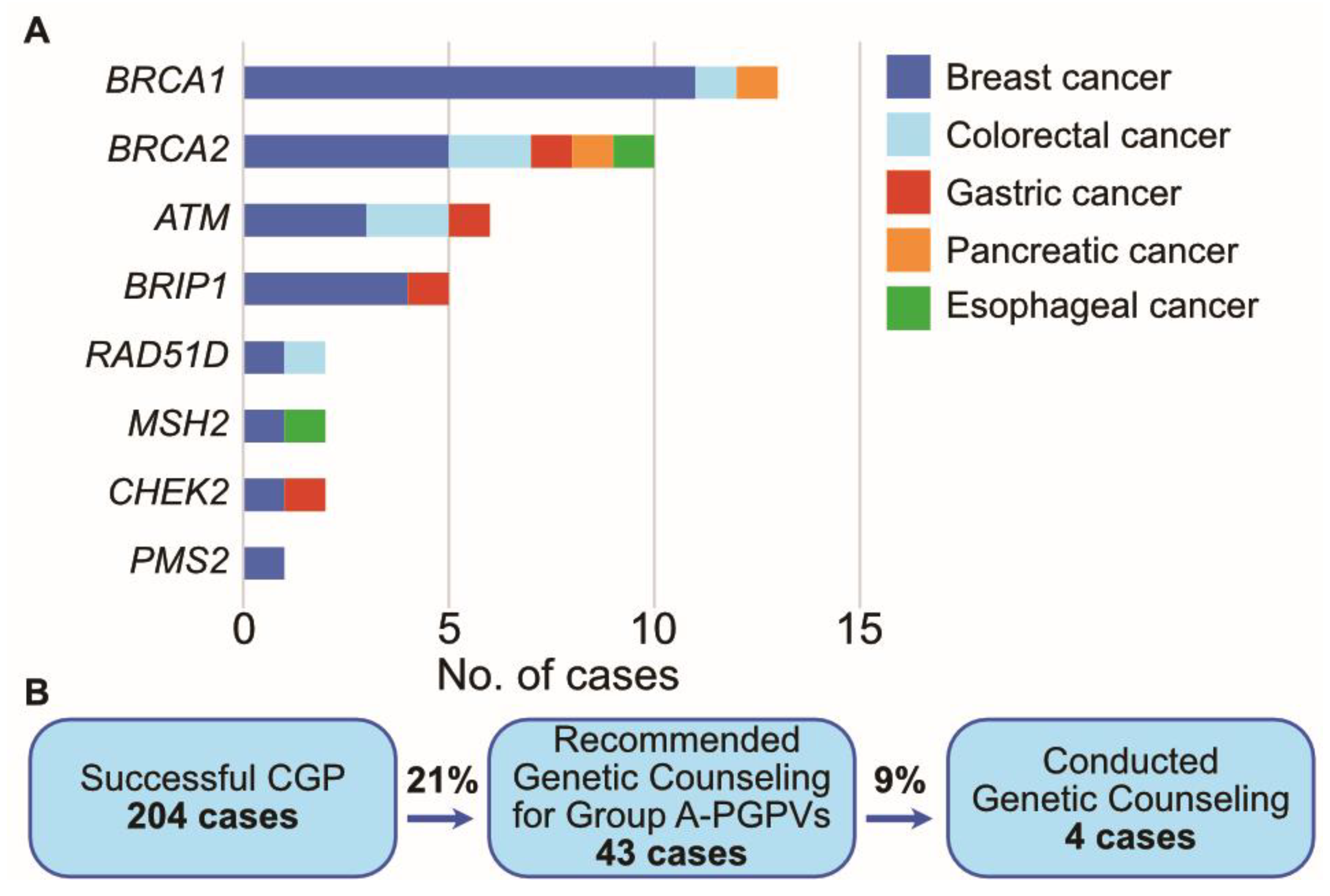
3.5. Presumed Germline Pathogenic Variants (PGPVs) Assessment and the Low Rate of Germline Testing in Patients with PGPVs
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
Abbreviations
| Genes | |
| APC | APC regulator of WNT signaling pathway |
| ARID1A | AT-Rich Interaction Domain 1A |
| BRCA1 | BRCA1 DNA repair associated |
| BRCA2 | BRCA2 DNA repair associated |
| CDK4 | cyclin dependent kinase 4 |
| CDK12 | cyclin dependent kinase 12 |
| CDKN2A | cyclin dependent kinase inhibitor 2A |
| CDKN2B | cyclin dependent kinase inhibitor 2B |
| ERBB2 | erb-b2 receptor tyrosine kinase 2 |
| KRAS | KRAS proto-oncogene, GTPase |
| MDM2 | MDM2 proto-oncogene |
| MSH2 | MutS Homolog 2 |
| MYC | MYC proto-oncogene, bHLH transcription factor |
| PIK3CA | phosphatidylinositol-4,5-bisphosphate 3-kinase catalytic subunit alpha |
| SMAD4 | SMAD family member 4 |
| TP53 | tumor protein p53 |
| Cellular process/components/genetics | |
| PD-1 | programmed cell death protein 1 |
| PD-L1 | programmed cell death 1 ligand 1 |
| HRR | homologous recombination repair |
| HRD | homologous recombination deficiency |
| NHEJ | non-homologous end joining |
| DSB | double-strand break |
| PARP | poly (ADP-ribose) polymerase |
| MMR | mismatch repair |
| SWI/SNF | switch/sucrose non-fermenting |
| SUB | nucleotide substitution |
| Indel | insertion and deletion |
| CNA | copy number alteration |
| MSI | microsatellite instability |
| TMB | tumor mutational burden |
| ctDNA | circulating tumor DNA |
| PGPV | presumed germline pathogenic variant |
| PGV | pathogenic germline variant |
| Technologies/Organizations/Regulations | |
| NGS | next-generation sequencing |
| CGP | comprehensive genomic profiling |
| NCCN | National Comprehensive Cancer Network |
| MHLW | The Ministry of Health, Labor, and Welfare |
| C-CAT | Center for Cancer Genomics and Advanced Therapeutics |
| ACMG | American College of Medical Genetics |
| ESMO | European Society for Medical Oncology |
| CAT | College of American Pathologists |
| CLIA | Clinical Laboratory Improvement Amendments |
References
- Aldea, M.; Friboulet, L.; Apcher, S.; Jaulin, F.; Mosele, F.; Sourisseau, T.; Soria, J.C.; Nikolaev, S.; André, F. Precision Medicine in the Era of Multi-Omics: Can the Data Tsunami Guide Rational Treatment Decision? ESMO Open 2023, 8, 101642. [Google Scholar] [CrossRef] [PubMed]
- Jurisic, V. Multiomic Analysis of Cytokines in Immuno-Oncology. Expert Rev. Proteom. 2020, 17, 663–674. [Google Scholar] [CrossRef]
- Pishvaian, M.J.; Blais, E.M.; Brody, J.R.; Lyons, E.; DeArbeloa, P.; Hendifar, A.; Mikhail, S.; Chung, V.; Sahai, V.; Sohal, D.P.S.; et al. Overall Survival in Patients with Pancreatic Cancer Receiving Matched Therapies Following Molecular Profiling: A Retrospective Analysis of the Know Your Tumor Registry Trial. Lancet Oncol. 2020, 21, 508–518. [Google Scholar] [CrossRef] [PubMed]
- NCCN Clinical Practice Guideline: Pancreatic Adenocarcinoma. Version 4. 2023. Available online: https://www.nccn.org/guidelines/guidelines-detail?category=1&id=1455 (accessed on 15 August 2023).
- Sunami, K.; Ichikawa, H.; Kubo, T.; Kato, M.; Fujiwara, Y.; Shimomura, A.; Koyama, T.; Kakishima, H.; Kitami, M.; Matsushita, H.; et al. Feasibility and Utility of a Panel Testing for 114 Cancer-Associated Genes in a Clinical Setting: A Hospital-Based Study. Cancer Sci. 2019, 110, 1480–1490. [Google Scholar] [CrossRef] [PubMed]
- Janjigian, Y.Y.; Shitara, K.; Moehler, M.; Garrido, M.; Salman, P.; Shen, L.; Wyrwicz, L.; Yamaguchi, K.; Skoczylas, T.; Campos Bragagnoli, A.; et al. First-Line Nivolumab plus Chemotherapy versus Chemotherapy Alone for Advanced Gastric, Gastro-Oesophageal Junction, and Oesophageal Adenocarcinoma (CheckMate 649): A Randomised, Open-Label, Phase 3 Trial. Lancet 2021, 398, 27–40. [Google Scholar] [CrossRef] [PubMed]
- Shitara, K.; Ajani, J.A.; Moehler, M.; Garrido, M.; Gallardo, C.; Shen, L.; Yamaguchi, K.; Wyrwicz, L.; Skoczylas, T.; Bragagnoli, A.C.; et al. Nivolumab plus Chemotherapy or Ipilimumab in Gastro-Oesophageal Cancer. Nature 2022, 603, 942–948. [Google Scholar] [CrossRef]
- Marabelle, A.; Le, D.T.; Ascierto, P.A.; Di Giacomo, A.M.; de Jesus-Acosta, A.; Delord, J.P.; Geva, R.; Gottfried, M.; Penel, N.; Hansen, A.R.; et al. Efficacy of Pembrolizumab in Patients with Noncolorectal High Microsatellite Instability/ Mismatch Repair–Deficient Cancer: Results from the Phase II KEYNOTE-158 Study. J. Clin. Oncol. 2020, 38, 1–10. [Google Scholar] [CrossRef]
- Overman, M.J.; Lonardi, S.; Yeung, K.; Wong, M.; Lenz, H.-J.; Gelsomino, F.; Aglietta, M.; Morse, M.A.; Cutsem, E.V.; Mcdermott, R.; et al. Durable Clinical Benefit with Nivolumab Plus Ipilimumab in DNA Mismatch Repair–Deficient/Microsatellite Instability–High Metastatic Colorectal Cancer. J. Clin. Oncol. 2018, 36, 773–779. [Google Scholar] [CrossRef]
- Marabelle, A.; Fakih, M.; Lopez, J.; Shah, M.; Shapira-Frommer, R.; Nakagawa, K.; Chung, H.C.; Kindler, H.L.; Lopez-Martin, J.A.; Miller, W.H.; et al. Association of Tumour Mutational Burden with Outcomes in Patients with Advanced Solid Tumours Treated with Pembrolizumab: Prospective Biomarker Analysis of the Multicohort, Open-Label, Phase 2 KEYNOTE-158 Study. Lancet Oncol. 2020, 2045, 1353–1365. [Google Scholar] [CrossRef]
- Spencer, K.R.; Wang, J.; Silk, A.W.; Ganesan, S.; Kaufman, H.L.; Mehnert, J.M. Biomarkers for Immunotherapy: Current Developments and Challenges. Am. Soc. Clin. Oncol. Educ. B. 2016, 36, e493–e503. [Google Scholar] [CrossRef]
- Bird, B.H.; Nally, K.; Ronan, K.; Clarke, G.; Amu, S.; Almeida, A.S.; Flavin, R.; Finn, S. Cancer Immunotherapy with Immune Checkpoint Inhibitors-Biomarkers of Response and Toxicity; Current Limitations and Future Promise. Diagnostics 2022, 12, 124. [Google Scholar] [CrossRef] [PubMed]
- Lu, S.; Stein, J.E.; Rimm, D.L.; Wang, D.W.; Bell, J.M.; Johnson, D.B.; Sosman, J.A.; Schalper, K.A.; Anders, R.A.; Wang, H.; et al. Comparison of Biomarker Modalities for Predicting Response to PD-1/PD-L1 Checkpoint Blockade: A Systematic Review and Meta-Analysis. JAMA Oncol. 2019, 5, 1195–1204. [Google Scholar] [CrossRef] [PubMed]
- Yamamoto, H.; Hirasawa, A. Homologous Recombination Deficiencies and Hereditary Tumors. Int. J. Mol. Sci. 2022, 23, 348. [Google Scholar] [CrossRef] [PubMed]
- Cortesi, L.; Piombino, C.; Toss, A. Germline Mutations in Other Homologous Recombination Repair-Related Genes than Brca1/2: Predictive or Prognostic Factors? J. Pers. Med. 2021, 11, 245. [Google Scholar] [CrossRef] [PubMed]
- Shen, J.; Peng, Y.; Wei, L.; Zhang, W.; Yang, L.; Lan, L.; Kapoor, P.; Ju, Z.; Mo, Q.; Shih, I.M.; et al. ARID1A Deficiency Impairs the DNA Damage Checkpoint and Sensitizes Cells to PARP Inhibitors. Cancer Discov. 2015, 5, 752–767. [Google Scholar] [CrossRef]
- Mandal, J.; Mandal, P.; Wang, T.L.; Shih, I.M. Treating ARID1A Mutated Cancers by Harnessing Synthetic Lethality and DNA Damage Response. J. Biomed. Sci. 2022, 29, 71. [Google Scholar] [CrossRef]
- Shen, J.; Ju, Z.; Zhao, W.; Wang, L.; Peng, Y.; Ge, Z.; Nagel, Z.D.; Zou, J.; Wang, C.; Kapoor, P.; et al. ARID1A Deficiency Promotes Mutability and Potentiates Therapeutic Antitumor Immunity Unleashed by Immune Checkpoint Blockade. Nat. Med. 2018, 24, 556–562. [Google Scholar] [CrossRef]
- Gao, J.; Aksoy, B.A.; Dogrusoz, U.; Dresdner, G.; Gross, B.; Sumer, S.O.; Sun, Y.; Jacobsen, A.; Sinha, R.; Larsson, E.; et al. Integrative Analysis of Complex Cancer Genomics and Clinical Profiles Using the CBioPortal. Sci. Signal. 2013, 6, pl1. [Google Scholar] [CrossRef]
- Cerami, E.; Gao, J.; Dogrusoz, U.; Gross, B.E.; Sumer, S.O.; Aksoy, B.A.; Jacobsen, A.; Byrne, C.J.; Heuer, M.L.; Larsson, E.; et al. The CBio Cancer Genomics Portal: An Open Platform for Exploring Multidimensional Cancer Genomics Data. Cancer Discov. 2012, 2, 401–404. [Google Scholar] [CrossRef]
- Samstein, R.M.; Lee, C.H.; Shoushtari, A.N.; Hellmann, M.D.; Shen, R.; Janjigian, Y.Y.; Barron, D.A.; Zehir, A.; Jordan, E.J.; Omuro, A.; et al. Tumor Mutational Load Predicts Survival after Immunotherapy across Multiple Cancer Types. Nat. Genet. 2019, 51, 202–206. [Google Scholar] [CrossRef]
- Kawaji, H.; Kubo, M.; Yamashita, N.; Yamamoto, H.; Kai, M.; Kajihara, A.; Yamada, M.; Kurata, K.; Kaneshiro, K.; Harada, Y.; et al. Comprehensive Molecular Profiling Broadens Treatment Options for Breast Cancer Patients. Cancer Med. 2020, 10, 529–539. [Google Scholar] [CrossRef] [PubMed]
- Li, M.M.; Chao, E.; Esplin, E.D.; Miller, D.T.; Nathanson, K.L.; Plon, S.E.; Scheuner, M.T.; Stewart, D.R. Points to Consider for Reporting of Germline Variation in Patients Undergoing Tumor Testing: A Statement of the American College of Medical Genetics and Genomics (ACMG). Genet. Med. 2020, 22, 1142–1148. [Google Scholar] [CrossRef]
- Mandelker, D.; Donoghue, M.; Talukdar, S.; Bandlamudi, C.; Srinivasan, P.; Vivek, M.; Jezdic, S.; Hanson, H.; Snape, K.; Kulkarni, A.; et al. Germline-Focussed Analysis of Tumour-Only Sequencing: Recommendations from the ESMO Precision Medicine Working Group. Ann. Oncol. 2019, 30, 1221–1231. [Google Scholar] [CrossRef] [PubMed]
- Milbury, C.A.; Creeden, J.; Yip, W.K.; Smith, D.L.; Pattani, V.; Maxwell, K.; Sawchyn, B.; Gjoerup, O.; Meng, W.; Skoletsky, J.; et al. Clinical and Analytical Validation of FoundationOne®CDx, a Comprehensive Genomic Profiling Assay. for Solid Tumors. PLoS ONE 2022, 17, e0264138. [Google Scholar] [CrossRef] [PubMed]
- Wu, Y.M.; Cieślik, M.; Lonigro, R.J.; Vats, P.; Reimers, M.A.; Cao, X.; Ning, Y.; Wang, L.; Kunju, L.P.; de Sarkar, N.; et al. Inactivation of CDK12 Delineates a Distinct Immunogenic Class of Advanced Prostate Cancer. Cell 2018, 173, 1770–1782.e14. [Google Scholar] [CrossRef]
- Antonarakis, E.S.; Isaacsson Velho, P.; Fu, W.; Wang, H.; Agarwal, N.; Santos, V.S.; Maughan, B.L.; Pili, R.; Adra, N.; Sternberg, C.N.; et al. CDK12 -Altered Prostate Cancer: Clinical Features and Therapeutic Outcomes to Standard Systemic Therapies, Poly (ADP-Ribose) Polymerase Inhibitors, and PD-1 Inhibitors. JCO Precis. Oncol. 2020, 4, 370–381. [Google Scholar] [CrossRef]
- Kim, H.; Ahn, S.; Kim, H.; Hong, J.Y.; Lee, J.; Park, S.H.; Park, J.O.; Park, Y.S.; Lim, H.Y.; Kang, W.K.; et al. The Prevalence of Homologous Recombination Deficiency (HRD) in Various Solid Tumors and the Role of HRD as a Single Biomarker to Immune Checkpoint Inhibitors. J. Cancer Res. Clin. Oncol. 2021, 148, 2427–2435. [Google Scholar] [CrossRef]
- Okamura, R.; Kato, S.; Lee, S.; Jimenez, R.E.; Sicklick, J.K.; Kurzrock, R. ARID1A Alterations Function as a Biomarker for Longer Progression-Free Survival after Anti-PD-1/PD-L1 Immunotherapy. J. Immunother. Cancer 2020, 8, e000438. [Google Scholar] [CrossRef]
- Hugo, W.; Zaretsky, J.M.; Sun, L.; Song, C.; Moreno, B.H.; Hu-Lieskovan, S.; Berent-Maoz, B.; Pang, J.; Chmielowski, B.; Cherry, G.; et al. Genomic and Transcriptomic Features of Response to Anti-PD-1 Therapy in Metastatic Melanoma. Cell 2016, 165, 35–44. [Google Scholar] [CrossRef]
- Monk, B.J.; Colombo, N.; Oza, A.M.; Fujiwara, K.; Birrer, M.J.; Randall, L.; Poddubskaya, E.V.; Scambia, G.; Shparyk, Y.V.; Lim, M.C.; et al. Chemotherapy with or without Avelumab Followed by Avelumab Maintenance versus Chemotherapy Alone in Patients with Previously Untreated Epithelial Ovarian Cancer (JAVELIN Ovarian 100): An Open-Label, Randomised, Phase 3 Trial. Lancet Oncol. 2021, 22, 1275–1289. [Google Scholar] [CrossRef]
- Weigelt, B.; Comino-Méndez, I.; De Bruijn, I.; Tian, L.; Meisel, J.L.; García-Murillas, I.; Fribbens, C.; Cutts, R.; Martelotto, L.G.; Ng, C.K.Y.; et al. Diverse BRCA1 and BRCA2 Reversion Mutations in Circulating Cell-Free DNA of Therapy-Resistant Breast or Ovarian Cancer. Clin. Cancer Res. 2017, 23, 6708–6720. [Google Scholar] [CrossRef] [PubMed]
- Lin, K.K.; Harrell, M.I.; Oza, A.M.; Oaknin, A.; Ray-Coquard, I.; Tinker, A.V.; Helman, E.; Radke, M.R.; Say, C.; Vo, L.T.; et al. BRCA Reversion Mutations in Circulating Tumor DNA Predict Primary and Acquired Resistance to the PARP Inhibitor Rucaparib in High-Grade Ovarian Carcinoma. Cancer Discov. 2019, 9, 210–219. [Google Scholar] [CrossRef] [PubMed]
- Peyraud, F.; Italiano, A. Combined Parp Inhibition and Immune Checkpoint Therapy in Solid Tumors. Cancers 2020, 12, 1502. [Google Scholar] [CrossRef] [PubMed]
- Maio, M.; Shapira-Frommer, R.; Yap, T.A.; Ciuleanu, T.; Gomez, H.; Hill, A.; Lugowska, I.; Ozyilkan, O.; Vera, K.; Im, S.-A.; et al. Abstract CT178: Olaparib plus Pembrolizumab in Patients with Previously Treated Advanced Solid Tumors with Homologous Recombination Repair Mutation (HRRm) and/or Homologous Recombination Deficiency (HRD): Initial Results of the Phase 2 KEYLYNK-007 Study. Cancer Res. 2021, 81, CT178. [Google Scholar] [CrossRef]
- Obradovic, J.; Todosijevic, J.; Jurisic, V. Application of the Conventional and Novel Methods in Testing EGFR Variants for NSCLC Patients in the Last 10 Years through Different Regions: A Systematic Review. Mol. Biol. Rep. 2021, 48, 3593–3604. [Google Scholar] [CrossRef] [PubMed]
- Chen, H.; Luthra, R.; Goswami, R.S.; Singh, R.R.; Roy-Chowdhuri, S. Analysis of Pre-Analytic Factors Affecting the Success of Clinical next-Generation Sequencing of Solid Organ Malignancies. Cancers 2015, 7, 1699–1715. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Q.; Luo, J.; Wu, S.; Si, H.; Gao, C.; Xu, W.; Abdullah, S.E.; Higgs, B.W.; Dennis, P.A.; van der Heijden, M.S.; et al. Prognostic and Predictive Impact of Circulating Tumor DNA in Patients with Advanced Cancers Treated with Immune Checkpoint Blockade. Cancer Discov. 2020, 10, 1842–1853. [Google Scholar] [CrossRef]
- Bettegowda, C.; Sausen, M.; Leary, R.J.; Kinde, I.; Wang, Y.; Agrawal, N.; Bartlett, B.R.; Wang, H.; Luber, B.; Alani, R.M.; et al. Detection of Circulating Tumor DNA in Early- and Late-Stage Human Malignancies. Sci. Transl. Med. 2014, 6, 224ra24. [Google Scholar] [CrossRef]
- Bando, H.; Kagawa, Y.; Kato, T.; Akagi, K.; Denda, T.; Nishina, T.; Komatsu, Y.; Oki, E.; Kudo, T.; Kumamoto, H.; et al. A Multicentre, Prospective Study of Plasma Circulating Tumour DNA Test for Detecting RAS Mutation in Patients with Metastatic Colorectal Cancer. Br. J. Cancer 2019, 120, 982–986. [Google Scholar] [CrossRef] [PubMed]
- Vidal, J.; Muinelo, L.; Dalmases, A.; Jones, F.; Edelstein, D.; Iglesias, M.; Orrillo, M.; Abalo, A.; Rodríguez, C.; Brozos, E.; et al. Plasma CtDNA RAS Mutation Analysis for the Diagnosis and Treatment Monitoring of Metastatic Colorectal Cancer Patients. Ann. Oncol. 2017, 28, 1325–1332. [Google Scholar] [CrossRef]
- Faust, N.; Muller, C.; Prenner, J.; Lee, S.M.; Kupfer, S.S. Low Rates of Genetic Counseling and Testing in Individuals at Risk for Lynch Syndrome Reported in the National Health Interview Survey. Gastroenterology 2020, 158, 1159–1161. [Google Scholar] [CrossRef] [PubMed]
- Jonassaint, C.R.; Santos, E.R.; Glover, C.M.; Payne, P.W.; Fasaye, G.A.; Oji-Njideka, N.; Hooker, S.; Hernandez, W.; Foster, M.W.; Kittles, R.A.; et al. Regional Differences in Awareness and Attitudes Regarding Genetic Testing for Disease Risk and Ancestry. Hum. Genet. 2010, 128, 249–260. [Google Scholar] [CrossRef] [PubMed]
- Rodon, J.; Soria, J.C.; Berger, R.; Miller, W.H.; Rubin, E.; Kugel, A.; Tsimberidou, A.; Saintigny, P.; Ackerstein, A.; Braña, I.; et al. Genomic and Transcriptomic Profiling Expands Precision Cancer Medicine: The WINTHER Trial. Nat. Med. 2019, 25, 751–758. [Google Scholar] [CrossRef] [PubMed]
- Van Tilburg, C.M.; Pfaff, E.; Pajtler, K.W.; Langenberg, K.P.S.; Fiesel, P.; Jones, B.C.; Balasubramanian, G.P.; Stark, S.; Johann, P.D.; Blattner-Johnson, M.; et al. The Pediatric Precision Oncology Inform Registry: Clinical Outcome and Benefit for Patients with Very High-Evidence Targets. Cancer Discov. 2021, 11, 2764–2779. [Google Scholar] [CrossRef] [PubMed]
- Ohmura, H.; Yamaguchi, K.; Hanamura, F.; Ito, M.; Makiyama, A.; Uchino, K.; Shimokawa, H.; Tamura, S.; Esaki, T.; Mitsugi, K.; et al. OX40 and LAG3 Are Associated with Better Prognosis in Advanced Gastric Cancer Patients Treated with Anti-Programmed Death-1 Antibody. Br. J. Cancer 2020, 122, 1507–1517. [Google Scholar] [CrossRef] [PubMed]
- Uehara, K.; Tanoue, K.; Yamaguchi, K.; Ohmura, H.; Ito, M.; Matsushita, Y.; Tsuchihashi, K.; Tamura, S.; Shimokawa, H.; Isobe, T.; et al. Preferential B Cell Differentiation by Combined Immune Checkpoint Blockade for Renal Cell Carcinoma Is Associated with Clinical Response and Autoimmune Reactions. Cancer Immunol. Immunother. 2023, 72, 3543–3558. [Google Scholar] [CrossRef]


| Number of Patients (%) | |
|---|---|
| Age, years, median (range) | 63 (22–92) |
| Sex | |
| Male | 54 (26%) |
| Female | 150 (74%) |
| Disease | |
| Breast cancer | 109 (53%) |
| Colorectal cancer | 26 (13%) |
| Gastric cancer | 19 (9%) |
| Pancreatic cancer | 17 (8%) |
| Esophageal cancer | 8 (4%) |
| Soft tissue sarcoma | 7 (3%) |
| Other | 5 (2%) |
| Head and neck cancer | 3 (1%) |
| Cancer of unknown primary | 3 (1%) |
| Thyroid cancer | 2 (1%) |
| Lung cancer | 1 (0.5%) |
| Osteosarcoma | 1 (0.5%) |
| Brain cancer | 1 (0.5%) |
| Biliary tract cancer | 1 (0.5%) |
| Melanoma | 1 (0.5%) |
| TNM Staging | |
| I | 3 (1%) |
| II | 42 (36%) |
| III | 42 (36%) |
| IV | 117 (57%) |
| Sampling method | |
| Biopsy | 67 (33%) |
| Resection | 137 (67%) |
| Sampling site | |
| Primary | 158 (77%) |
| Metastatic | 46 (23%) |
| Prior chemotherapy | |
| Yes | 40 (20%) |
| Breast (n = 109) | Colorectal (n = 26) | Gastric (n = 19) | Pancreatic (n = 17) | ||||
|---|---|---|---|---|---|---|---|
| TP53 | 61 (56.0%) | TP53 | 23 (88.5%) | TP53 | 8 (42.1%) | KRAS | 16 (94.1%) |
| PIK3CA | 43 (39.4%) | APC | 19 (73.1%) | KRAS | 5 (26.3%) | TP53 | 13 (76.5%) |
| ERBB2 | 25 (22.9%) | KRAS | 10 (38.5%) | ARID1A | 4 (21.1%) | SMAD4 | 4 (23.5%) |
| MYC | 23 (21.1%) | SMAD4 | 6 (23.1%) | APC | 3 (15.8%) | CDKN2A | 3 (17.6%) |
| RAD21 | 23 (21.1%) | GNAS | 4 (15.4%) | ERBB2 | 3 (15.8%) | CDKN2B | 2 (11.8%) |
| PTEN | 20 (18.3%) | PIK3CA | 4 (15.4%) | MET | 2 (11.8%) | ||
| GATA3 | 15 (13.8%) | MLL2 | 3 (11.5%) | ||||
| TMB-High * | TMB-Low | p-Value ** | |
|---|---|---|---|
| MSI | |||
| MSI-high | 4 | 0 | <0.0001 |
| MS-stable | 17 | 166 | |
| MMR | |||
| MMR mutant | 2 | 1 | 0.0337 |
| MMR wild type | 19 | 165 | |
| HRR | |||
| HRR mutant | 12 | 45 | 0.0099 |
| HRR wild type | 9 | 121 |
| Failure (n = 18) | Success (n = 204) | p-Value * | |
|---|---|---|---|
| Sampling method | 0.60 | ||
| Biopsy | 7 | 67 | |
| Resection | 11 | 137 | |
| Sampling site | 0.76 | ||
| Primary site | 15 | 158 | |
| Metastatic site | 3 | 46 | |
| Prior chemotherapy | 0.22 | ||
| Yes | 6 | 40 | |
| No | 12 | 164 |
| Group A | |||||
| ATM | BRCA1 | BRCA2 | BRIP1 | CHEK2 | MLH1 |
| MSH2 | MSH6 | MUTYH * | PALB2 | PMS2 | RAD51C |
| RAD51D | RET | SDHB | SDHC | SDHD | TSC2 |
| VHL | |||||
| Group B | |||||
| APC | CDH1 | MEN1 | NF2 | PTEN | RB1 |
| SMAD4 | STK11 | TP53 | TSC1 | WT1 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Ito, M.; Kubo, M.; Kawaji, H.; Otsubo, Y.; Kurata, K.; Abutani, H.; Suyama, M.; Oda, Y.; Yoshizumi, T.; Nakamura, M.; et al. Homologous Recombination Repair Gene Alterations Are Associated with Tumor Mutational Burden and Survival of Immunotherapy. Cancers 2023, 15, 5608. https://doi.org/10.3390/cancers15235608
Ito M, Kubo M, Kawaji H, Otsubo Y, Kurata K, Abutani H, Suyama M, Oda Y, Yoshizumi T, Nakamura M, et al. Homologous Recombination Repair Gene Alterations Are Associated with Tumor Mutational Burden and Survival of Immunotherapy. Cancers. 2023; 15(23):5608. https://doi.org/10.3390/cancers15235608
Chicago/Turabian StyleIto, Mamoru, Makoto Kubo, Hitomi Kawaji, Yoshiki Otsubo, Kanako Kurata, Hikaru Abutani, Mikita Suyama, Yoshinao Oda, Tomoharu Yoshizumi, Masafumi Nakamura, and et al. 2023. "Homologous Recombination Repair Gene Alterations Are Associated with Tumor Mutational Burden and Survival of Immunotherapy" Cancers 15, no. 23: 5608. https://doi.org/10.3390/cancers15235608
APA StyleIto, M., Kubo, M., Kawaji, H., Otsubo, Y., Kurata, K., Abutani, H., Suyama, M., Oda, Y., Yoshizumi, T., Nakamura, M., & Baba, E. (2023). Homologous Recombination Repair Gene Alterations Are Associated with Tumor Mutational Burden and Survival of Immunotherapy. Cancers, 15(23), 5608. https://doi.org/10.3390/cancers15235608






