Treatment Outcomes and Risk Factors for Incomplete Treatment after Definitive Chemoradiotherapy for Non-Resectable or Metastatic Esophageal Cancer
Abstract
:Simple Summary
Abstract
1. Introduction
2. Materials and Methods
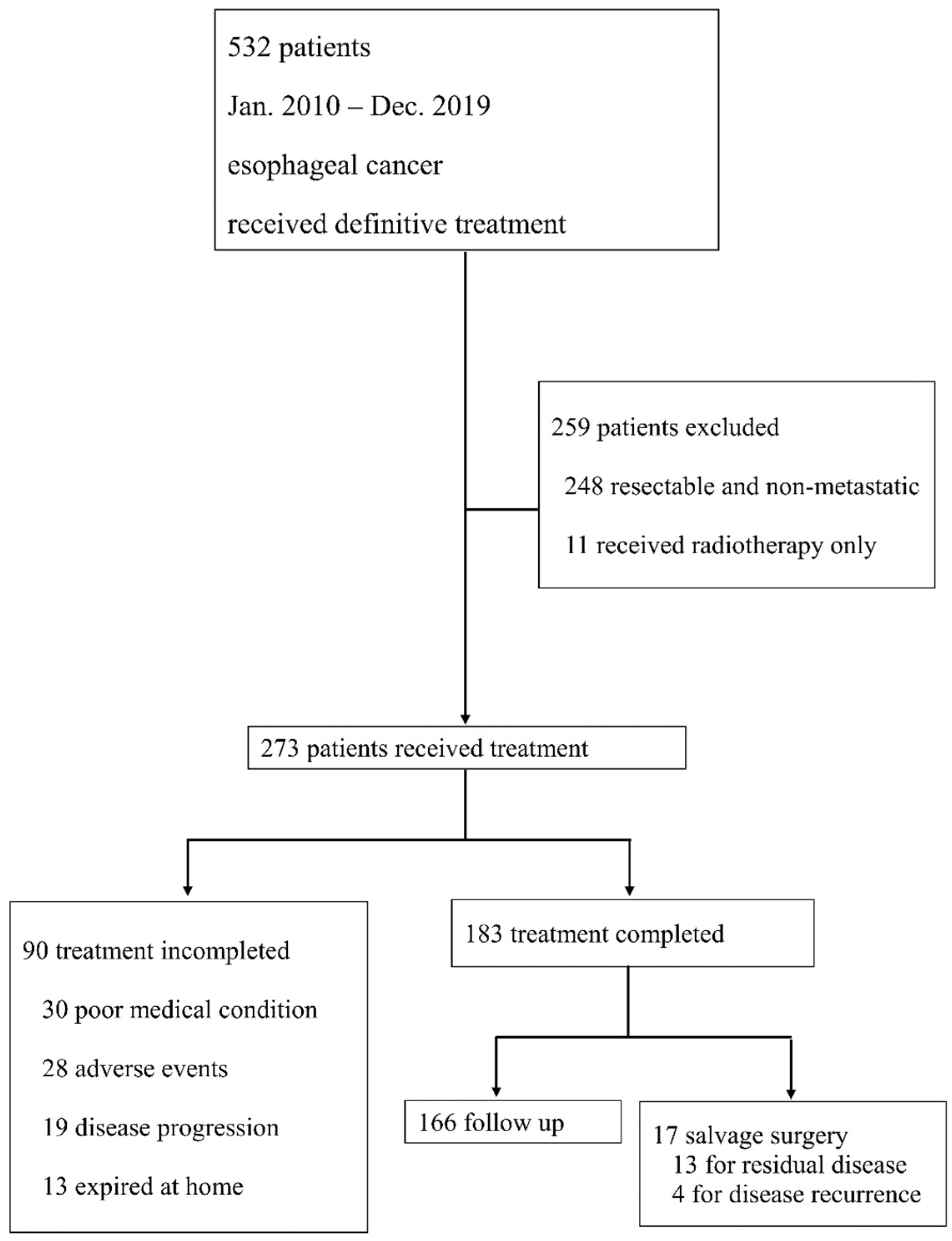
2.1. Study Population
2.2. Staging Evaluations
2.3. Definitive Chemoradiotherapy and Surgery
2.4. Statistics
3. Results
3.1. Clinicopathological Characteristics of the Study Patients
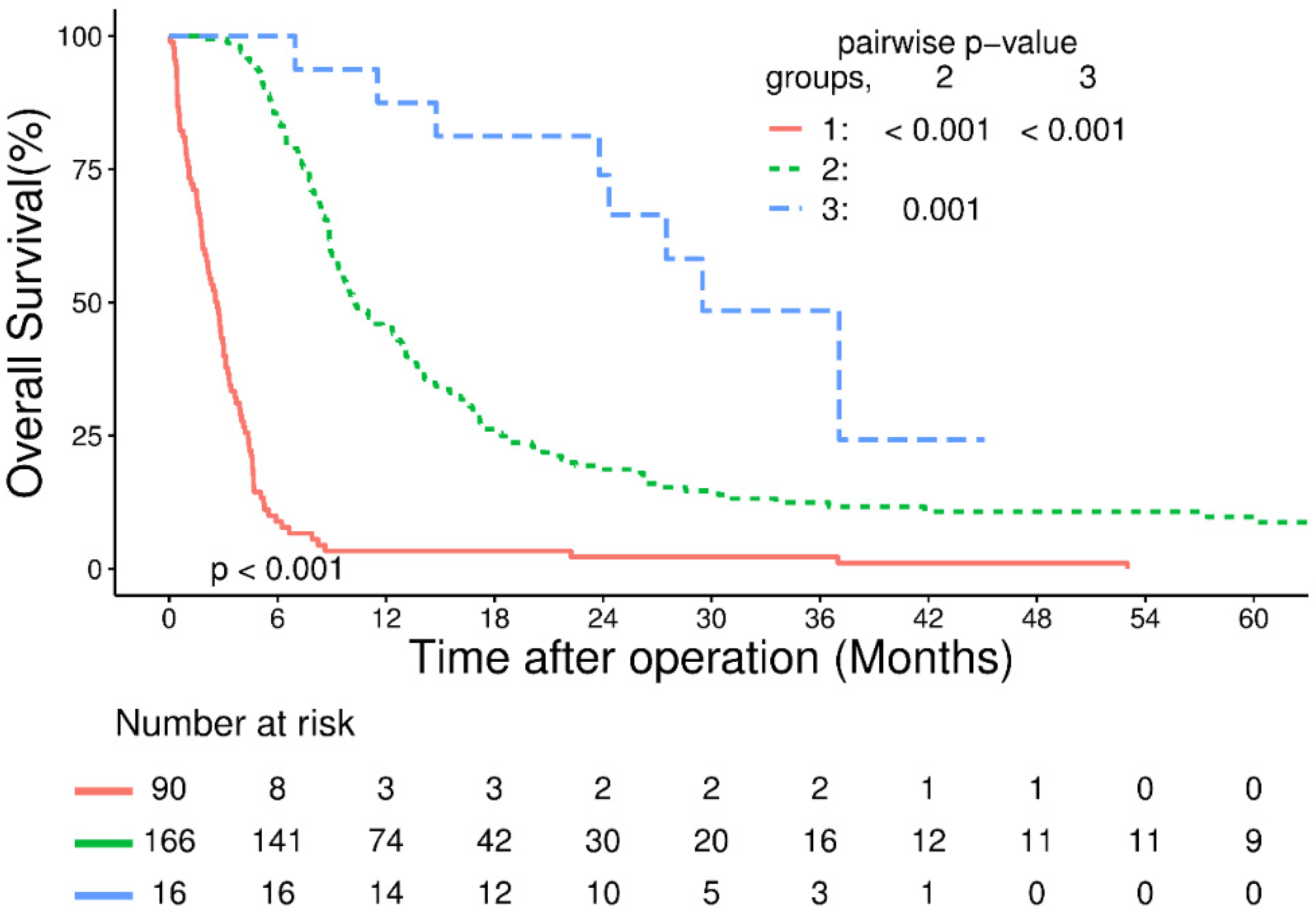
3.2. Survival and Prognostic Factors for Patients Receiving Surgical and Non-Surgical Treatment
3.3. Risk Factors for Incomplete Definitive Chemoradiotherapy
4. Discussion
4.1. Survival and Prognosticators of Patients Receiving Definitive Chemoradiotherapy
4.2. Prognostic Effect of Salvage Esophagectomy
4.3. Optimization of Treatment Plans
4.4. Brief Summary
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Ishida, K.; Ando, N.; Yamamoto, S.; Ide, H.; Shinoda, M. Phase II study of cisplatin and 5-fluorouracil with concurrent radiotherapy in advanced squamous cell carcinoma of the esophagus: A Japan Esophageal Oncology Group (JEOG)/Japan Clinical Oncology Group trial (JCOG9516). Jpn. J. Clin. Oncol. 2004, 34, 615–619. [Google Scholar] [CrossRef]
- Ohtsu, A.; Boku, N.; Muro, K.; Chin, K.; Muto, M.; Yoshida, S.; Satake, M.; Ishikura, S.; Ogino, T.; Miyata, Y.; et al. Definitive chemoradiotherapy for T4 and/or M1 lymph node squamous cell carcinoma of the esophagus. J. Clin. Oncol. 1999, 17, 2915–2921. [Google Scholar] [CrossRef]
- Minsky, B.D.; Pajak, T.F.; Ginsberg, R.J.; Pisansky, T.M.; Martenson, J.; Komaki, R.; Okawara, G.; Rosenthal, S.A.; Kelsen, D.P. INT 0123 (Radiation Therapy Oncology Group 94-05) Phase III Trial of Combined-Modality Therapy for Esophageal Cancer: High-Dose Versus Standard-Dose Radiation Therapy. J. Clin. Oncol. 2002, 20, 1167–1174. [Google Scholar] [CrossRef]
- Li, H.; Fang, Y.; Gu, D.; Du, M.; Zhang, Z.; Sun, L.; Zhou, G.; Ye, J. Paclitaxel and cisplatin combined with concurrent involved-field irradiation in definitive chemoradiotherapy for locally advanced esophageal squamous cell carcinoma: A phase II clinical trial. Radiat. Oncol. 2022, 17, 105. [Google Scholar] [CrossRef]
- Cooper, J.S.; Guo, M.D.; Herskovic, A.; Macdonald, J.S.; Martenson, J.A., Jr.; Al-Sarraf, M.; Byhardt, R.; Russell, A.H.; Beitler, J.J.; Spencer, S.; et al. Chemoradiotherapy of locally advanced esophageal cancer: Long-term follow-up of a prospective randomized trial (RTOG 85-01). Radiation Therapy Oncology Group. JAMA 1999, 281, 1623–1627. [Google Scholar] [CrossRef]
- Herskovic, A.; Martz, K.; Al-Sarraf, M.; Leichman, L.; Brindle, J.; Vaitkevicius, V.; Cooper, J.; Byhardt, R.; Davis, L.; Emami, B. Combined chemotherapy and radiotherapy compared with radiotherapy alone in patients with cancer of the esophagus. N. Engl. J. Med. 1992, 326, 1593–1598. [Google Scholar] [CrossRef]
- Edge, S.B.; Compton, C.C. The American Joint Committee on Cancer: The 7th edition of the AJCC cancer staging manual and the future of TNM. Ann. Surg. Oncol. 2010, 17, 1471–1474. [Google Scholar] [CrossRef]
- Eisenhauer, E.A.; Therasse, P.; Bogaerts, J.; Schwartz, L.H.; Sargent, D.; Ford, R.; Dancey, J.; Arbuck, S.; Gwyther, S.; Mooney, M.; et al. New response evaluation criteria in solid tumours: Revised RECIST guideline (version 1.1). Eur. J. Cancer 2009, 45, 228–247. [Google Scholar] [CrossRef]
- Common Terminology Criteria for Adverse Events, Version 5.0. Available online: https://ctep.cancer.gov/protocoldevelopment/electronic_applications/ctc.htm#ctc_50 (accessed on 2 November 2023).
- Pai, C.P.; Hsu, P.K.; Chien, L.I.; Huang, C.S.; Hsu, H.S. Clinical outcome of patients after recurrent laryngeal nerve lymph node dissection for oesophageal squamous cell carcinoma. Interact. Cardiovasc. Thorac. Surg. 2022, 34, 393–401. [Google Scholar] [CrossRef]
- George, B.; Seals, S.; Aban, I. Survival analysis and regression models. J. Nucl. Cardiol. 2014, 21, 686–694. [Google Scholar] [CrossRef]
- Ochi, M.; Murakami, Y.; Nishibuchi, I.; Kubo, K.; Imano, N.; Takeuchi, Y.; Kimura, T.; Hamai, Y.; Emi, M.; Okada, M.; et al. Long-term results of definitive chemoradiotherapy for unresectable locally advanced esophageal squamous cell carcinoma. J. Radiat. Res. 2021, 62, 142–148. [Google Scholar] [CrossRef] [PubMed]
- Stahl, M.; Stuschke, M.; Lehmann, N.; Meyer, H.J.; Walz, M.K.; Seeber, S.; Klump, B.; Budach, W.; Teichmann, R.; Schmitt, M.; et al. Chemoradiation with and without surgery in patients with locally advanced squamous cell carcinoma of the esophagus. J. Clin. Oncol. 2005, 23, 2310–2317. [Google Scholar] [CrossRef] [PubMed]
- Shinoda, M.; Ando, N.; Kato, K.; Ishikura, S.; Kato, H.; Tsubosa, Y.; Minashi, K.; Okabe, H.; Kimura, Y.; Kawano, T.; et al. Randomized study of low-dose versus standard-dose chemoradiotherapy for unresectable esophageal squamous cell carcinoma (JCOG0303). Cancer Sci. 2015, 106, 407–412. [Google Scholar] [CrossRef] [PubMed]
- Chao, Y.K.; Chan, S.C.; Chang, H.K.; Liu, Y.H.; Wu, Y.C.; Hsieh, M.J.; Tseng, C.K.; Liu, H.P. Salvage surgery after failed chemoradiotherapy in squamous cell carcinoma of the esophagus. Eur. J. Surg. Oncol. 2009, 35, 289–294. [Google Scholar] [CrossRef]
- Booka, E.; Haneda, R.; Ishii, K.; Kawakami, T.; Tsushima, T.; Yasui, H.; Onoe, T.; Ogawa, H.; Tsubosa, Y. Appropriate Candidates for Salvage Esophagectomy of Initially Unresectable Locally Advanced T4 Esophageal Squamous Cell Carcinoma. Ann. Surg. Oncol. 2020, 27, 3163–3170. [Google Scholar] [CrossRef]
- Yokota, T.; Kato, K.; Hamamoto, Y.; Tsubosa, Y.; Ogawa, H.; Ito, Y.; Hara, H.; Ura, T.; Kojima, T.; Chin, K.; et al. A 3-Year Overall Survival Update from a Phase 2 Study of Chemoselection With DCF and Subsequent Conversion Surgery for Locally Advanced Unresectable Esophageal Cancer. Ann. Surg. Oncol. 2020, 27, 460–467. [Google Scholar] [CrossRef]
- Faiz, Z.; Dijksterhuis, W.P.M.; Burgerhof, J.G.; Muijs, C.T.; Mul, V.E.M.; Wijnhoven, B.P.L.; Smit, J.K.; Plukker, J.T.M. A meta-analysis on salvage surgery as a potentially curative procedure in patients with isolated local recurrent or persistent esophageal cancer after chemoradiotherapy. Eur. J. Surg. Oncol. 2019, 45, 931–940. [Google Scholar] [CrossRef]
- Nishimura, Y.; Hiraoka, M.; Koike, R.; Nakamatsu, K.; Itasaka, S.; Kawamura, M.; Negoro, Y.; Araki, N.; Ishikawa, H.; Fujii, T.; et al. Long-term follow-up of a randomized Phase II study of cisplatin/5-FU concurrent chemoradiotherapy for esophageal cancer (KROSG0101/JROSG021). Jpn. J. Clin. Oncol. 2012, 42, 807–812. [Google Scholar] [CrossRef]
- Yao, B.; Tan, B.; Wang, C.; Song, Q.; Wang, J.; Guan, S.; Jia, Y.; Ma, Y.; Huang, X.; Sun, Y.; et al. Comparison of Definitive Chemoradiotherapy in Locally Advanced Esophageal Squamous Cell Carcinoma. Ann. Surg. Oncol. 2016, 23, 2367–2372. [Google Scholar] [CrossRef]
- Conroy, T.; Galais, M.P.; Raoul, J.L.; Bouché, O.; Gourgou-Bourgade, S.; Douillard, J.Y.; Etienne, P.L.; Boige, V.; Martel-Lafay, I.; Michel, P.; et al. Definitive chemoradiotherapy with FOLFOX versus fluorouracil and cisplatin in patients with oesophageal cancer (PRODIGE5/ACCORD17): Final results of a randomised, phase 2/3 trial. Lancet Oncol. 2014, 15, 305–314. [Google Scholar] [CrossRef]
- Hulshof, M.C.; Geijsen, E.D.; Rozema, T.; Oppedijk, V.; Buijsen, J.; Neelis, K.J.; Nuyttens, J.J.; van der Sangen, M.J.; Jeene, P.M.; Reinders, J.G.; et al. Randomized Study on Dose Escalation in Definitive Chemoradiation for Patients with Locally Advanced Esophageal Cancer (ARTDECO Study). J. Clin. Oncol. 2021, 39, 2816–2824. [Google Scholar] [CrossRef] [PubMed]
- Crehange, G.; M’vondo, C.; Bertaut, A.; Pereira, R.; Rio, E.; Peiffert, D.; Gnep, K.; Benezery, K.; Ronchin, P.; Noel, G.; et al. Exclusive Chemoradiotherapy with or Without Radiation Dose Escalation in Esophageal Cancer: Multicenter Phase 2/3 Randomized Trial CONCORDE (PRODIGE-26). Int. J. Radiat. Oncol. Biol. Phys. 2021, 111, S5. [Google Scholar] [CrossRef]
- Bridges, S.; Thomas, B.; Radhakrishna, G.; Hawkins, M.; Holborow, A.; Hurt, C.; Mukherjee, S.; Nixon, L.; Crosby, T.; Gwynne, S. SCOPE 2—Still Answering the Unanswered Questions in Oesophageal Radiotherapy? SCOPE 2: A Randomised Phase II/III Trial to Study Radiotherapy Dose Escalation in Patients with Oesophageal Cancer Treated with Definitive Chemoradiation with an Embedded Phase II Trial for Patients with a Poor Early Response using Positron Emission Tomography/Computed Tomography. Clin. Oncol. 2022, 34, e269–e280. [Google Scholar]
- Mikhail, S.; Wei, L.; Salem, M.E.; Bekaii-Saab, T. Outcomes of definitive chemoradiation in patients with esophageal cancer. Dis. Esophagus. 2017, 30, 1–7. [Google Scholar] [CrossRef] [PubMed]
- Cheng, C.; Wen, Y.W.; Tsai, C.Y.; Chao, Y.K. Impact of Child-Pugh class A liver cirrhosis on perioperative outcomes of patients with oesophageal cancer: A propensity score-matched analysis. Eur. J. Cardiothorac. Surg. 2020, 59, 395–402. [Google Scholar] [CrossRef]
- Katano, A.; Yamashita, H.; Nakagawa, K. Successful definitive concurrent chemoradiotherapy in a patient with esophageal cancer and Child-Pugh B cirrhosis of the liver. J. Cancer Res. Ther. 2019, 15, 255–257. [Google Scholar]
- Trivin, F.; Boucher, E.; Vauléon, E.; Cumin, I.; Le Prisé, E.; Audrain, O.; Raoul, J.L. Management of esophageal carcinoma associated with cirrhosis: A retrospective case-control analysis. J. Oncol. 2009, 2009, 173421. [Google Scholar] [CrossRef]
- Huang, T.T.; Li, S.H.; Chen, Y.H.; Lu, H.I.; Lo, C.M.; Fang, F.M.; Chou, S.Y.; Chiu, Y.C.; Chou, Y.P.; Wang, Y.M. Definitive chemoradiotherapy for clinical T4b esophageal cancer—Treatment outcomes, failure patterns, and prognostic factors. Radiother. Oncol. 2021, 157, 56–62. [Google Scholar] [CrossRef]
- Sugawara, K.; Yagi, K.; Okumura, Y.; Nishida, M.; Aikou, S.; Yamashita, H.; Yamashita, H.; Seto, Y. Long-term outcomes of multimodal therapy combining definitive chemoradiotherapy and salvage surgery for T4 esophageal squamous cell carcinoma. Int. J. Clin. Oncol. 2020, 25, 552–560. [Google Scholar] [CrossRef]
- Chen, B.; Deng, M.; Yang, C.; Dragomir, M.P.; Zhao, L.; Bai, K.; Xi, M.; Hu, Y.; Zhu, Y.; Li, Q. High incidence of esophageal fistula on patients with clinical T4b esophageal squamous cell carcinoma who received chemoradiotherapy: A retrospective analysis. Radiother. Oncol. 2021, 158, 191–199. [Google Scholar] [CrossRef]
- Tachimori, Y. Role of salvage esophagectomy after definitive chemoradiotherapy. Gen. Thorac. Cardiovasc. Surg. 2009, 57, 71–78. [Google Scholar] [CrossRef] [PubMed]
- Chen, Y.; Lu, Y.; Wang, Y.; Yang, H.; Xia, Y.; Chen, M.; Song, H.; Li, T.; Li, D.; Wang, J.; et al. Comparison of salvage chemoradiation versus salvage surgery for recurrent esophageal squamous cell carcinoma after definitive radiochemotherapy or radiotherapy alone. Dis. Esophagus 2014, 27, 134–140. [Google Scholar] [CrossRef] [PubMed]
- Kumagai, K.; Mariosa, D.; Tsai, J.A.; Nilsson, M.; Ye, W.; Lundell, L.; Rouvelas, I. Systematic review and meta-analysis on the significance of salvage esophagectomy for persistent or recurrent esophageal squamous cell carcinoma after definitive chemoradiotherapy. Dis. Esophagus. 2016, 29, 734–739. [Google Scholar] [CrossRef] [PubMed]
- Mori, K.; Sugawara, K.; Aikou, S.; Yamashita, H.; Yamashita, K.; Ogura, M.; Chin, K.; Watanabe, M.; Matsubara, H.; Toh, Y.; et al. Esophageal cancer patients’ survival after complete response to definitive chemoradiotherapy: A retrospective analysis. Esophagus 2021, 18, 629–637. [Google Scholar] [CrossRef]
- Wang, S.; Tachimori, Y.; Hokamura, N.; Igaki, H.; Nakazato, H.; Kishino, T. Prognostic analysis of salvage esophagectomy after definitive chemoradiotherapy for esophageal squamous cell carcinoma: The importance of lymphadenectomy. J. Thorac. Cardiovasc. Surg. 2014, 147, 1805–1811. [Google Scholar] [CrossRef]
- Kiyozumi, Y.; Yoshida, N.; Ishimoto, T.; Yagi, T.; Koga, Y.; Uchihara, T.; Sawayama, H.; Hiyoshi, Y.; Iwatsuki, M.; Baba, Y.; et al. Prognostic Factors of Salvage Esophagectomy for Residual or Recurrent Esophageal Squamous Cell Carcinoma After Definitive Chemoradiotherapy. World J. Surg. 2018, 42, 2887–2893. [Google Scholar] [CrossRef]
- Okamura, A.; Hayami, M.; Kozuki, R.; Takahashi, K.; Toihata, T.; Imamura, Y.; Mine, S.; Watanabe, M. Salvage esophagectomy for initially unresectable locally advanced T4 esophageal squamous cell carcinoma. Esophagus 2020, 17, 59–66. [Google Scholar]
- Sugimura, K.; Miyata, H.; Shinno, N.; Ushigome, H.; Asukai, K.; Hara, H.; Hasegawa, S.; Yamada, D.; Yamamoto, K.; Haraguchi, N.; et al. Prognostic Impact of Postoperative Complications following Salvage Esophagectomy for Esophageal Cancer after Definitive Chemoradiotherapy. Oncology 2020, 98, 280–288. [Google Scholar] [CrossRef]
- Takemura, M.; Takii, M.; Gyobu, K.; Oshima, T.; Takada, N.; Iwasaki, Y. Outcomes of Salvage Thoracoscopic Esophagectomy after Definitive Radiochemotherapy for Clinical Stage IVa Esophageal Cancer. Gan Kagaku Ryoho. Cancer Chemother. 2020, 47, 1848–1850. [Google Scholar]
| All Patients (N = 273) | dCRT Incomplete (N = 90) | dCRT Complete (N = 183) | |||||
|---|---|---|---|---|---|---|---|
| p * | Total | Total | Salvage Operation | p ** | |||
| Group 1 | No (N = 166) Group 2 | Yes (N = 17) Group 3 | |||||
| Age (years; median, IQR) | 59.0 (52.0–67.0) | 0.282 | 61.0 (52.0–68.0) | 58.0 (52.0–66.0) | 58.0 (51.8–66.3) | 60.0 (54.0–66.5) | 0.886 |
| Sex (%) | 0.287 | 0.611 | |||||
| Male | 251 (91.9) | 85 (94.4) | 166 (90.7) | 150 (90.4) | 16 (94.1) | ||
| Female | 22 (8.1) | 5 (5.6) | 17 (9.3) | 16 (9.6) | 1 (5.9) | ||
| BMI (median, IQR) | 21.0 (19.0–23.7) | 0.087 | 20.4 (17.9–23.2) | 21.2 (19.3–23.8) | 21.1 (19.2–23.8) | 23.2 (20.3–25.2) | 0.091 |
| Smoking (%) | 0.065 | 0.883 | |||||
| Never | 53 (20.2) | 23 (25.8) | 30 (16.4) | 27 (16.3) | 3 (17.6) | ||
| Former/current | 219 (79.6) | 66 (74.2) | 153 (83.6) | 139 (83.7) | 14 (82.4) | ||
| ECOG (%) | <0.001 | 0.580 | |||||
| 0 | 142 (52.0) | 29 (32.2) | 113 (61.7) | 101 (60.8) | 12 (70.6) | ||
| 1 | 106 (38.8) | 43 (47.8) | 63 (34.4) | 58 (34.9) | 5 (29.4) | ||
| ≥2 | 25 (9.2) | 18 (20.0) | 7 (3.8) | 7 (4.2) | 0 (0) | ||
| Cirrhosis (%) | 0.007 | 0.355 | |||||
| Child–Pugh A | 10 (11.2) | 8 (4.4) | 8 (4.8) | 0 (0) | |||
| Child–Pugh B | 2 (2.2) | 0 (0) | 0 (0) | 0 (0) | |||
| Charlson comorbidity index | 3.0 (2.0–4.0) | 0.131 | 3.0 (2.0–4.3) | 3.0 (2.0–4.0) | 3.0 (2.0–4.0) | 2.0 (2.0–3.5) | 0.703 |
| Laboratory data (median, IQR) | |||||||
| Hemoglobin (g/dL) | 12.4 (10.9–13.6) | 0.003 | 11.9 (10.1–13.3) | 12.6 (11.3–13.8) | 12.6 (11.2–13.8) | 13.5 (12.3–14.1) | 0.150 |
| Albumin (g/dL) | 3.7 (3.3–4.0) | <0.001 | 3.5 (3.0–3.9) | 3.8 (3.4–4.1) | 3.8 (3.4–4.0) | 3.9 (3.6–4.3) | 0.017 |
| Neutrophil (cells/µL) | 5427.1 (4126.0–7392.6) | 0.109 | 5505.2 (4203.7–8296.7) | 5217.2 (4002.3–7145.7) | 5544.0 (4301.5–7204.4) | 3938.7 (2989.2–5036.8) | 0.018 |
| Lymphocyte (cells/µL) | 1504.4 (1124.3–1850.0) | 0.009 | 1354.2 (1024.6–1731.3) | 1572.6 (1163.6–1894.8) | 1548.6 (1153.1–1862.3) | 1674.8 (1405.9–2154.4) | 0.136 |
| Monocyte (cells/µL) | 633.2 (490.2–861.6) | 0.509 | 636.0 (463.3–910.6) | 629.9 (501.0–846.3) | 633.9 (520.4–859.5) | 555.0 (458.3–763.4) | 0.692 |
| Platelet (counts/µL) | 279,000.0 (217,000.0–342,000.0) | 0.191 | 285,000.0 (186,500.0–346,000.0) | 277,000.0 (227,000.0–341,000.0) | 279,000.0 (228,000.0–345,000.0) | 248,000.0 (222,000.0–383,000.0) | 0.081 |
| NLR (IQR) | 3.7 (2.6–5.5) | 0.001 | 4.5 (2.9–7.2) | 3.5 (2.6–4.9) | 3.6 (2.7–5.0) | 2.2 (1.7–3.4) | 0.002 |
| LMR (IQR) | 2.2 (1.6–3.0) | 0.009 | 1.9 (1.4–2.8) | 2.4 (1.7–3.2) | 2.2 (1.7–3.1) | 3.5 (2.3–3.8) | 0.012 |
| PLR (IQR) | 184.1 (134.2–255.4) | 0.376 | 190.9 (131.2–277.8) | 178.3 (135.6–238.9) | 184.5 (139.6–252.0) | 147.8 (100.2–182.4) | 0.008 |
| Tumor location (%) | 0.610 | 0.867 | |||||
| Cervical | 24 (8.8) | 8 (8.9) | 16 (8.7) | 15 (9.0) | 1 (5.9) | ||
| Upper | 69 (25.3) | 26 (28.9) | 43 (23.5) | 40 (24.1) | 3 (17.6) | ||
| Middle | 99 (36.3) | 32 (35.6) | 67 (36.6) | 59 (35.5) | 8 (47.1) | ||
| Lower | 72 (26.4) | 23 (25.6) | 49 (26.8) | 45 (27.1) | 4 (23.5) | ||
| EGJ | 9 (3.3) | 1 (1.1) | 8 (4.4) | 7 (4.2) | 1 (5.9) | ||
| cT stage (%) | 0.004 | 0.758 | |||||
| 1 | 5 (1.8) | 0 (0) | 5 (2.7) | 4 (2.4) | 1 (5.9) | ||
| 2 | 30 (11.0) | 8 (8.9) | 22 (12.0) | 20 (12.0) | 2 (11.8) | ||
| 3 | 146 (53.5) | 39 (43.3) | 107 (58.5) | 96 (57.8) | 11 (64.7) | ||
| 4 | 87 (31.9) | 42 (46.7) | 45 (24.6) | 42 (25.3) | 3 (17.6) | ||
| Cannot be assessed | 5 (1.8) | 1 (1.1) | 4 (2.2) | 4 (2.4) | 0 (0) | ||
| cN stage (%) | 0.648 | 0.511 | |||||
| 0 | 17 (6.2) | 6 (6.7) | 11 (6.0) | 11 (6.6) | 0 (0) | ||
| 1 | 61 (22.3) | 20 (22.2) | 41 (22.4) | 38 (22.9) | 3 (17.6) | ||
| 2 | 74 (27.1) | 26 (28.9) | 48 (26.2) | 41 (24.7) | 7 (41.2) | ||
| 3 | 120 (44.0) | 37 (41.1) | 83 (45.4) | 76 (45.8) | 7 (41.2) | ||
| Cannot be assessed | 1 (0.4) | 1 (1.1) | 0 (0.0) | 0 (0.0) | 0 (0.0) | ||
| cM stage (%) | 0.567 | 0.766 | |||||
| 0 | 67 (24.5) | 24 (26.7) | 43 (23.5) | 40 (24.1) | 3 (17.6) | ||
| 1 | 206 (75.5) | 66 (73.3) | 140 (76.5) | 126 (75.9) | 14 (82.4) | ||
| Histology (%) | 0.621 | 0.715 | |||||
| SCC | 249 (91.2) | 81 (90.0) | 168 (91.8) | 152 (91.6) | 16 (94.1) | ||
| ADC | 24 (8.8) | 9 (10.0) | 15 (8.2) | 14 (8.4) | 1 (5.9) | ||
| MDT conference (%) | <0.001 | <0.001 | |||||
| 0 | 109 (39.9) | 46 (51.1) | 63 (34.4) | 62 (37.3) | 1 (5.9) | ||
| 1 | 112 (41.0) | 39 (43.3) | 73 (39.9) | 70 (42.2) | 3 (17.6) | ||
| ≥2 | 52 (19.1) | 5 (5.6) | 47 (25.7) | 34 (20.5) | 13 (76.5) | ||
| Radiation dose (cGy) (%) | |||||||
| ≤5040 | 125 (45.8) | <0.001 | 59 (65.6) | 66 (36.1) | 59 (35.5) | 7 (41.2) | 0.792 |
| >5040 | 148 (54.2) | 31 (34.4) | 117 (63.9) | 107 (64.5) | 10 (58.8) | ||
| Chemotherapy regimen (%) | 0.192 | 0.800 | |||||
| CDDP + 5-FU | 230 (84.2) | 77 (85.6) | 153 (83.6) | 138 (83.1) | 15 (88.2) | ||
| CDDP + 5-FU + Taxane | 10 (3.7) | 1 (1.1) | 9 (4.9) | 9 (5.4) | 0 (0) | ||
| Tegafur–Uracil | 7 (2.6) | 1 (1.1) | 6 (3.3) | 5 (3.0) | 1 (5.9) | ||
| 5-FU | 6 (2.2) | 3 (3.3) | 3 (1.6) | 3 (1.8) | 0 (0) | ||
| Carboplatin + Taxane | 4 (1.5) | 3 (3.3) | 1 (0.5) | 1 (0.6) | 0 (0) | ||
| Others *** | 16 (5.9) | 5 (5.6) | 11 (6.0) | 10 (6.0) | 1 (5.9) | ||
| Clinical response to therapy (%) | <0.001 | <0.001 | |||||
| CR | 16 (5.8) | 0 (0) | 16 (8.7) | 11 (6.6) | 5 (29.4) | ||
| PR | 112 (41.0) | 58 (64.4) | 54 (29.5) | 43 (25.9) | 11 (64.7) | ||
| SD | 23 (8.4) | 13 (14.4) | 10 (5.5) | 10 (6.0) | 0 (0) | ||
| PD | 119 (43.6) | 19 (21.1) | 100 (54.6) | 99 (59.6) | 1 (5.9) | ||
| Undefined | 3 (1.1) | 0 (0) | 3 (1.6) | 3 (1.8) | 0 (0) | ||
| Follow-up time (month; median, IQR) | 8.1 (3.9–16.1) | <0.001 | 2.6 (1.1–4.4) | 11.4 (7.7–21.7) | 10.2 (7.4–18.2) | 26.5 (18.5–33.3) | 0.071 |
| Variable | Univariate | Multivariate | ||||
|---|---|---|---|---|---|---|
| HR | 95% CI | p Value | HR | 95% CI | p Value | |
| Age (years; ≥60 vs. <60) | 0.84 | 0.65–1.08 | 0.181 | |||
| Sex (male vs. female) | 1.69 | 1.04–2.74 | 0.033 | 1.70 | 0.99–2.91 | 0.051 |
| Smoking (ever vs. never) | 0.77 | 0.56–1.05 | 0.102 | |||
| BMI (≥21 vs. <21) | 0.94 | 0.73–1.21 | 0.630 | |||
| ECOG (≥2 vs. 0–1) | 2.13 | 1.40–3.23 | <0.001 | 1.58 | 0.95–2.65 | 0.081 |
| CCI score (≥4 vs. <4) | 1.04 | 0.80–1.35 | 0.777 | |||
| Liver cirrhosis (yes vs. no) | 1.20 | 0.75–1.93 | 0.452 | |||
| Laboratory data | ||||||
| Hemoglobin (g/dL; ≥10 vs. <10) | 0.80 | 0.57–1.11 | 0.180 | |||
| Albumin (g/dL; ≥3.5 vs. <3.5) | 0.69 | 0.54–0.90 | 0.005 | 0.85 | 0.63–1.14 | 0.273 |
| NLR (≥3.4 vs. <3.4) | 1.41 | 1.09–1.82 | 0.009 | 1.02 1.23 | 0.98–1.06 0.93–1.63 | 0.295 0.152 |
| LMR (<2.4 vs. ≥2.4) | 1.23 | 0.96–1.59 | 0.109 | |||
| PLR (≥170 vs. <170) | 1.23 | 0.96–1.59 | 0.106 | |||
| Tumor location (Ce vs. Ut/Mt/Lt/EGJ) | 0.94 | 0.60–1.47 | 0.776 | |||
| c T stage (4b vs. 1/2/3/4a) | 1.48 | 1.12–1.96 | 0.005 | |||
| c N stage (3 vs. 0/1/2) | 1.35 | 1.05–1.75 | 0.020 | 1.38 | 1.06–1.80 | 0.019 |
| c M stage (1 vs. 0) | 0.83 | 0.62–1.10 | 0.190 | |||
| cT4b | ||||||
| Aorta | 1.41 | 0.88–2.25 | 0.152 | |||
| Airway | 1.65 | 1.23–2.21 | 0.001 | 1.63 | 1.17–2.25 | 0.003 |
| Distant metastasis | ||||||
| Retroperitoneal LN | 1.24 | 0.93–1.65 | 0.149 | |||
| Liver | 1.38 | 1.01–1.90 | 0.045 | 1.66 | 1.18–2.33 | 0.004 |
| Bone | 1.75 | 1.29–2.38 | <0.001 | 1.71 | 1.23–2.38 | 0.002 |
| Lung | 0.94 | 0.72–1.23 | 0.647 | |||
| Adrenal | 1.42 | 0.73–2.76 | 0.308 | |||
| Histopathology (SCC vs. ADC) | 1.24 | 0.80–1.93 | 0.341 | |||
| MDT (yes vs. no) | 0.82 | 0.63–1.05 | 0.117 | |||
| Clinical response | ||||||
| PD | 1 | 1 | ||||
| PR | 0.80 | 0.61–1.06 | 0.120 | 0.82 | 0.61–1.10 | 0.190 |
| SD | 1.06 | 0.67–1.67 | 0.807 | 1.05 | 0.60–1.83 | 0.863 |
| CR | 0.05 | 0.01–0.16 | <0.001 | 0.05 | 0.02–0.17 | <0.001 |
| Variable | Univariate | Multivariate | ||||
|---|---|---|---|---|---|---|
| HR | 95% CI | p Value | HR | 95% CI | p Value | |
| Age (years; ≥60 vs. <60) | 1.51 | 0.91–2.50 | 0.114 | |||
| Sex (male vs. female) | 1.74 | 0.62–4.88 | 0.292 | |||
| Smoking (ever vs. never) | 0.56 | 0.30–1.04 | 0.067 | |||
| BMI (≥21 vs. <21) | 0.76 | 0.46–1.27 | 0.299 | |||
| ECOG (≥2 vs. 0–1) | 6.29 | 2.52–15.70 | < 0.001 | 5.23 | 1.95–14.02 | 0.001 |
| CCI score (≥4 vs. <4) | 1.55 | 0.92–2.63 | 0.102 | |||
| Laboratory data | ||||||
| Hemoglobin (g/dL; ≥10 vs. <10) | 0.44 | 0.23–0.84 | 0.014 | 0.64 | 0.31–1.34 | 0.236 |
| Albumin (g/dL; ≥3.5 vs. <3.5) | 0.49 | 0.29–0.82 | 0.006 | |||
| NLR (≥3.4 vs. <3.4) | 1.78 | 1.05–3.03 | 0.033 | 1.06 | 0.97–1.17 | 0.202 |
| LMR (<2.4 vs. ≥2.4) | 1.77 | 1.04–3.00 | 0.034 | 1.01 | 0.73–1.37 | 0.993 |
| PLR (≥170 vs. <170) | 1.44 | 0.85–2.42 | 0.174 | |||
| Tumor location (Ce vs. Ut/Mt/Lt/EGJ) | 1.02 | 0.42–2.48 | 0.968 | |||
| c T stage (4b vs. 1/2/3/4a) | 2.08 | 1.20–3.61 | 0.009 | |||
| c N stage (3 vs. 0/1/2) | 0.86 | 0.51–1.43 | 0.556 | |||
| c M stage (1 vs. 0) | 0.85 | 0.47–1.51 | 0.568 | |||
| cT4b | ||||||
| Aorta | 1.58 | 0.64–3.91 | 0.319 | |||
| Airway | 2.48 | 1.39–4.42 | 0.002 | 2.90 | 1.53–5.51 | 0.001 |
| Distant metastasis | ||||||
| Retroperitoneal LN | 1.25 | 0.71–2.22 | 0.443 | |||
| Liver | 1.72 | 0.92–3.20 | 0.089 | |||
| Bone | 2.40 | 1.32–4.36 | 0.004 | 2.18 | 1.11–4.30 | 0.024 |
| Lung | 1.08 | 0.63–1.86 | 0.786 | |||
| Adrenal | 1.37 | 0.38–4.99 | 0.631 | |||
| Histopathology (SCC vs. ADC) | 0.80 | 0.34–1.91 | 0.621 | |||
| MDT (yes vs. no) | 0.50 | 0.30–0.84 | 0.009 | 0.65 | 0.37–1.15 | 0.139 |
| Liver cirrhosis (yes vs. no) | 3.41 | 1.34–8.67 | 0.010 | 3.20 | 1.15–8.91 | 0.026 |
| dCRT Incomplete (N = 90) | dCRT Complete (N = 183) | |||
|---|---|---|---|---|
| Grade 3 | Grade 4 | Grade 3 | Grade 4 | |
| Hematological (%) | ||||
| Leukopenia | 15 (16.7) | 18 (20.0) | 48 (26.2) | 20 (10.9) |
| Anemia | 23 (25.6) | 2 (2.2) | 33 (18.0) | 2 (1.1) |
| Thrombocytopenia | 9 (10.0) | 14 (15.6) | 22 (12.0) | 5 (2.7) |
| Non-hematological (%) | ||||
| Respiratory system | 20 (22.2) | 13 (14.4) | 22 (12.0) | 9 (4.9) |
| Gastrointestinal tract | 23 (25.6) | 5 (5.5) | 12 (6.6) | 4 (2.2) |
| Renal insufficiency | 2 (2.2) | 2 (2.2) | 0 (0) | 1 (0.5) |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Pai, C.-P.; Chien, L.-I.; Huang, C.-S.; Hsu, H.-S.; Hsu, P.-K. Treatment Outcomes and Risk Factors for Incomplete Treatment after Definitive Chemoradiotherapy for Non-Resectable or Metastatic Esophageal Cancer. Cancers 2023, 15, 5421. https://doi.org/10.3390/cancers15225421
Pai C-P, Chien L-I, Huang C-S, Hsu H-S, Hsu P-K. Treatment Outcomes and Risk Factors for Incomplete Treatment after Definitive Chemoradiotherapy for Non-Resectable or Metastatic Esophageal Cancer. Cancers. 2023; 15(22):5421. https://doi.org/10.3390/cancers15225421
Chicago/Turabian StylePai, Chu-Pin, Ling-I Chien, Chien-Sheng Huang, Han-Shui Hsu, and Po-Kuei Hsu. 2023. "Treatment Outcomes and Risk Factors for Incomplete Treatment after Definitive Chemoradiotherapy for Non-Resectable or Metastatic Esophageal Cancer" Cancers 15, no. 22: 5421. https://doi.org/10.3390/cancers15225421
APA StylePai, C.-P., Chien, L.-I., Huang, C.-S., Hsu, H.-S., & Hsu, P.-K. (2023). Treatment Outcomes and Risk Factors for Incomplete Treatment after Definitive Chemoradiotherapy for Non-Resectable or Metastatic Esophageal Cancer. Cancers, 15(22), 5421. https://doi.org/10.3390/cancers15225421






