Recent Pre-Clinical Advancements in Nuclear Medicine: Pioneering the Path to a Limitless Future
Abstract
:Simple Summary
Abstract
1. Introduction
2. Pre-Existing-Based Molecules
2.1. Repurposing and Optimization of FDA-Approved Drugs
2.1.1. Somatostatin Receptor 2 (SSTR2) Targeting
DOTA-TATE-Based Radiotracer
SARTATE-Based Radiotracer
Radiotracers Based on SSTR2 Antagonists
| Radiotracer | Type of Molecule | Disease Model | Study | Reference |
|---|---|---|---|---|
| 153Sm-DOTA-TATE | Agonist | CA20948, pancreatic tumor | Pre-clinical | [34] |
| 225Ac-DOTA-TATE | Agonist | H727 and H69 cells, Lung neuroendocrine neoplasms | Pre-clinical | [36] |
| 212Pb-DOTAMTATE | Agonist | AR42J, pancreatic tumor Neuro-endocrine tumors | Pre-clinical First in humans | [37] [38] |
| 64Cu-SARTATE | Agonist | AR42J, pancreatic tumor Neuro-endocrine tumors | Pre-clinical First in humans | [39] [40] |
| 67Cu-SARTATE | Agonist | AR42J, pancreatic tumor (metastasis) IMR32, neuroblastoma Multifocal Meningioma | Pre-clinical Pre-clinical Clinical | [41] [42] [44] |
| 177Lu-DOTA-LM3 | Antagonist | AR42J, pancreatic tumor Neuro-endocrine tumors | Pre-clinical First in humans | [46] [47] |
| 161Tb-DOTA-LM3 | Antagonist | AR42J, pancreatic tumor | Pre-clinical | [46] |
| 177Lu-AAZTA5-LM4 | Antagonist | HEK293-SST2R transfected cells | Pre-clinical | [49] |
| 177Lu-satoreotide tetraxetan | Antagonist | AR42J, pancreatic tumor | Pre-clinical | [48] |
2.1.2. Prostate-Membrane-Specific Antigen (PSMA) Targeting
Repurposing of 68Ga-PSMA-11/177Lu-PSMA-617
Development of 225Ac-PSMA-617
Other Agents Currently under Development
Modifying PSMA-Agents as a Valuable Strategy to Enhance Therapy Efficacy
Development of 18F-PSMA-1007 for Imaging
| Radiotracer | Type of Molecule | Disease Model | Study | Reference |
|---|---|---|---|---|
| 225Ac-PSMA-617 | Peptide | mCRPC mCRPC mCRPC mCRPC | Clinical study (51 patients) Clinical study (26 patients) Clinical study (73 patients) Clinical study (3 cohorts; 91, 40, 18 patients) -Tandem with 177Lu-PSMA-617 | [66] [67] [68] [69] |
| 177Lu-L1 | Peptide | PC3 PIP (PC xenografts) | Pre-clinical | [72] |
| 225Ac-L1 | Peptide | PC3 PIP (PC xenografts) | Pre-clinical | [73] |
| 213Bi-L1 | Peptide | PC3 PIP (PC xenografts) | Pre-clinical | [73] |
| 211At-3-Lu | Peptide | PC3 PIP (PC xenografts) | Pre-clinical | [74] |
| 177Lu-EB-PSMA-617 | Peptide with EB moiety | mCRPC mCRPC HepG2 xenografts, HCC Adenoid cystic carcinoma | Clinical study (4 patients) Clinical study (28 patients) Pre-clinical Clinical (30 patients) | [77] [80] [57] [81] |
| 177Lu-LNC1003 | Peptide with EB moiety | 22Rv (PC xenografts) | Pre-clinical | [82] |
| 225Ac-PSMA-TO-1 | Peptide + albumin binder (naphthyl group) | C4-2 (PC xenografts) | Pre-clinical | [83] |
| 225Ac-PSMA-DA1 | Peptide + albumin binder (iodophenylbutyric acid derivative) | LNCaP (PC xenografts) LNCaP (prostate xenografts) | Pre-clinical Pre-clinical | [84] [85] |
| 227Th-BAY 2315497 | Antibody | PC cell lines and PDXs VCap, ST1273 (PC xenografts) | Pre-clinical Pre-clinical | [87] [88] |
2.2. Molecules Expected to Be Translated Soon into Clinical Practice
2.2.1. Fibroblast Activation Protein Inhibitor (FAPI)
Development of β- and α-Emitter Labeled FAPIs
Modification of FAPIs to Enhance Tumor Targeting
| Radiotracer | Type of Molecule | Disease Model | Study | Reference |
|---|---|---|---|---|
| 90Y-FAPI-04 | Quinoline-based | HT-1080 xenografts (fibrosarcoma) Patient: metastatic breast cancer | Pre-clinical and first-in-human studies | [107] |
| 177Lu-FAPI-04 | Quinoline-based | Metastatic advanced stage cancer | Clinical (4 patients) | [112] |
| 131I-FAPI-04 | Quinoline-based | U87MG xenografts(glioblastoma) | Pre-clinical | [110] |
| 211At-FAPI-04 | Quinoline-based | U87MG xenografts(glioblastoma) | Pre-clinical | [111] |
| 225Ac-FAPI04 | Quinoline-based | PANC-1, MIA PaCa-2 xenografts (PDAC) | Pre-clinical | [108] |
| 177Lu-FAPI-46 | Quinoline-based | PANC-1 xenografts (PDAC) Relapsed or refractory cancers (10 patients) | Pre-clinical Clinical | [114] [115] |
| 90Y-FAPI-46 | Quinoline-based | Solid tumors (110 patients) | Clinical | [116] |
| 225Ac-FAPI-46 | Quinoline-based | PANC-1 xenografts (PDAC) | Pre-clinical | [114] |
| 177Lu-FAP-2286 | Quinoline-based | HEK293 and PDX (sarcoma) | Pre-clinical | [117] |
| 177Lu-DOTA.(SA.FAPI)2 | Bivalent FAP | Different tumors (7 patients) Thyroid cancer (15 radioiodine-refractory patients) | First in humans First in humans | [119] [120] |
| 177Lu-ND-bisFAPI | Bivalent FAP | A549 xenografts (lung cancer) | Pre-clinical | [121] |
| 177Lu-EB-FAPI-B1 | Quinoline-based and modified with EB | U87MG xenografts(glioblastoma) | Pre-clinical | [122] |
| 177Lu-TEFAPI06/07 | Quinoline-based and modified with albumin-binder | HT-1080 xenografts (fibrosarcoma) | Pre-clinical | [123] |
| 177Lu-FSDD0I | Quinoline-based and modified with IPBA | PDXs (HCC) | Pre-clinical | [124] |
2.2.2. Gastrin-Releasing Peptide Receptor (GRPR)
3. Major Recent Advances in the Theranostics Field
3.1. New Theranostic Agents for Solid Tumors
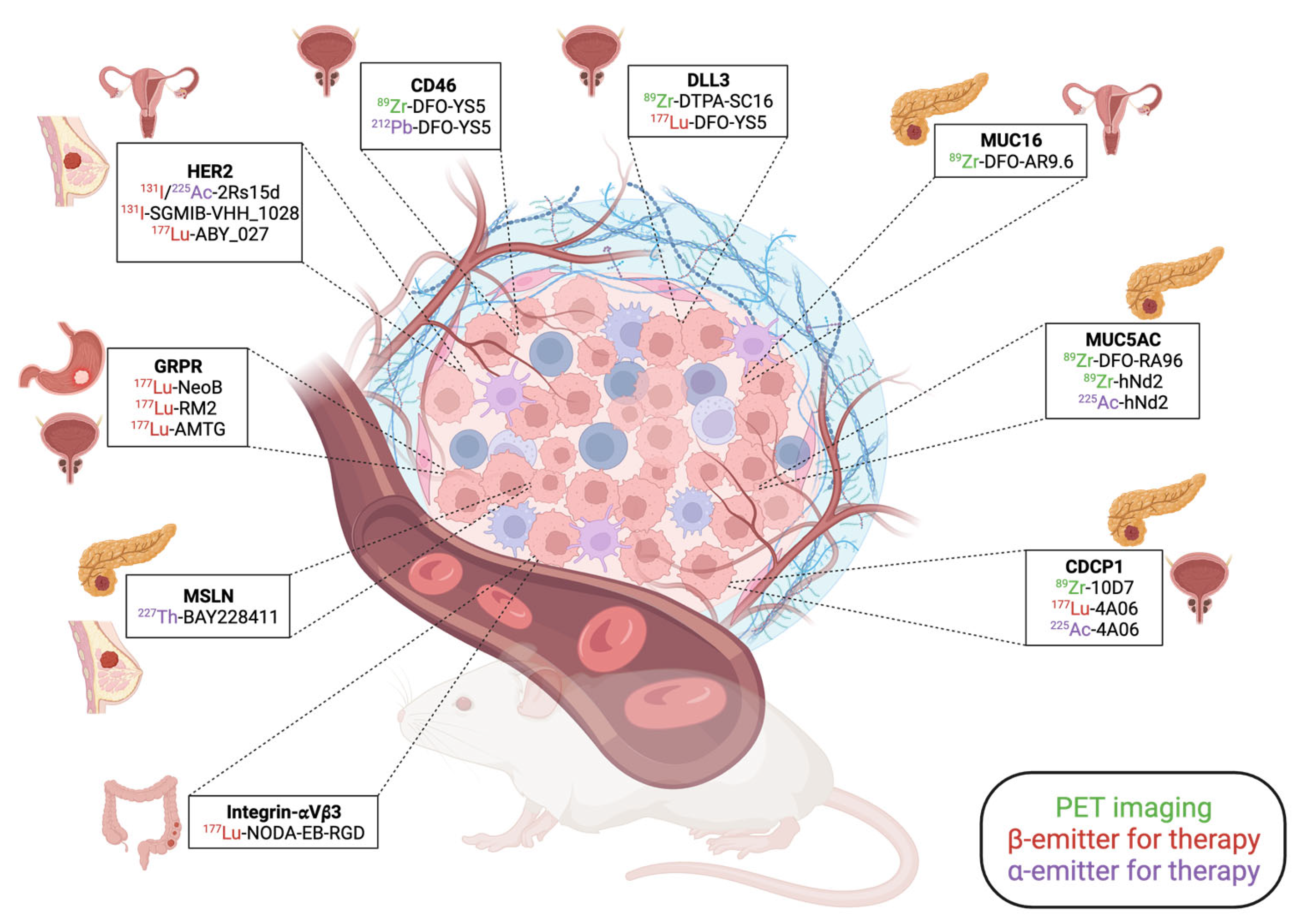
3.1.1. Human Epidermal Growth Factor Receptor 2 (HER2) Targeting
3.1.2. Recent Developments for Prostate Cancer
CD46 as an Alternative to PSMA?
DLL3 as an Emerging Target in PCs
3.1.3. Recent Developments for PDAC
MUC16
MUC5AC
CDCP1
3.2. Development of Bispecific Molecules
3.2.1. PSMA-FAPI
3.2.2. PSMA-GRPR
3.2.3. Other Developments of Dual-Targeting Molecules
3.3. Combination of PRRT with Immunotherapies
3.3.1. Combination with FDA-Approved Radiotracers
DOTA-TATE and Immunotherapies
PSMA and Immunotherapies
3.3.2. Other Promising Combinations Recently Evaluated in Pre-Clinical Studies
Mesothelin (MSLN) and Combination Therapy
Integrin αvβ3
NM600
Other Combinations with ICIs
4. Conclusions
Author Contributions
Funding
Conflicts of Interest
Abbreviations
| CDC1P | CUB domain containing protein 1 |
| CRPC | castration-resistant prostate cancer |
| CTLA-4 | cytotoxic T-lymphocyte antigen 4 |
| FAPI | fibroblast activation protein inhibitor |
| FDA | Food and Drug Administration |
| GBM | glioblastoma multiforme |
| GRPR | gastrin-releasing peptide receptor |
| HER2 | Human Epidermal Growth Factor Receptor 2 |
| ICI | immune checkpoint inhibitor |
| LET | linear energy transfer |
| LNCaP | Lymph Node Carcinoma of the Prostate |
| MSLN | mesothelin |
| MUC16 | mucin-16 |
| MUC5AC | mucin-5AC |
| NET | neuro-endocrine tumor |
| OS | overall survival |
| PC | prostate cancer |
| PDAC | pancreatic ductal adenocarcinoma |
| PDX | patient-derived xenograft |
| PD-1 | programmed cell death 1 |
| PD-L1 | programmed cell death ligand 1 |
| PET | positron emission tomography |
| PFS | progression-free survival |
| PRRT | peptide receptor radionuclide therapy |
| PSMA | prostate-specific membrane antigen |
| RT | radiotherapy |
| SARTATE | sarcophagine octreotate |
| SSTR2 | somatostatin receptor 2 |
| TKI | tyrosine kinase inhibitor |
References
- Strebhardt, K.; Ullrich, A. Paul Ehrlich’s Magic Bullet Concept: 100 Years of Progress. Nat. Rev. Cancer 2008, 8, 473–480. [Google Scholar] [CrossRef] [PubMed]
- Schwartz, R.S. Paul Ehrlich’s Magic Bullets. N. Engl. J. Med. 2004, 350, 1079–1080. [Google Scholar] [CrossRef] [PubMed]
- Sapio, L.; Naviglio, S. Innovation through Tradition: The Current Challenges in Cancer Treatment. Int. J. Mol. Sci. 2022, 23, 5296. [Google Scholar] [CrossRef] [PubMed]
- Falzone, L.; Salomone, S.; Libra, M. Evolution of Cancer Pharmacological Treatments at the Turn of the Third Millennium. Front. Pharmacol. 2018, 9, 1300. [Google Scholar] [CrossRef] [PubMed]
- Bissery, M.C.; Guénard, D.; Guéritte-Voegelein, F.; Lavelle, F. Experimental Antitumor Activity of Taxotere (RP 56976, NSC 628503), a Taxol Analogue. Cancer Res. 1991, 51, 4845–4852. [Google Scholar] [PubMed]
- Ojima, I.; Lichtenthal, B.; Lee, S.; Wang, C.; Wang, X. Taxane Anticancer Agents: A Patent Perspective. Expert Opin. Ther. Pat. 2016, 26, 1–20. [Google Scholar] [CrossRef] [PubMed]
- Hartmann, J.T.; Haap, M.; Kopp, H.-G.; Lipp, H.-P. Tyrosine Kinase Inhibitors—A Review on Pharmacology, Metabolism and Side Effects. Curr. Drug Metab. 2009, 10, 470–481. [Google Scholar] [CrossRef] [PubMed]
- Solassol, I.; Pinguet, F.; Quantin, X. FDA- and EMA-Approved Tyrosine Kinase Inhibitors in Advanced EGFR-Mutated Non-Small Cell Lung Cancer: Safety, Tolerability, Plasma Concentration Monitoring, and Management. Biomolecules 2019, 9, 668. [Google Scholar] [CrossRef]
- Waldman, A.D.; Fritz, J.M.; Lenardo, M.J. A Guide to Cancer Immunotherapy: From T Cell Basic Science to Clinical Practice. Nat. Rev. Immunol. 2020, 20, 651–668. [Google Scholar] [CrossRef]
- Twomey, J.D.; Zhang, B. Cancer Immunotherapy Update: FDA-Approved Checkpoint Inhibitors and Companion Diagnostics. AAPS J. 2021, 23, 39. [Google Scholar] [CrossRef]
- Debien, V.; De Caluwé, A.; Wang, X.; Piccart-Gebhart, M.; Tuohy, V.K.; Romano, E.; Buisseret, L. Immunotherapy in Breast Cancer: An Overview of Current Strategies and Perspectives. npj Breast Cancer 2023, 9, 7. [Google Scholar] [CrossRef] [PubMed]
- Emran, T.B.; Shahriar, A.; Mahmud, A.R.; Rahman, T.; Abir, M.H.; Siddiquee, M.F.-R.; Ahmed, H.; Rahman, N.; Nainu, F.; Wahyudin, E.; et al. Multidrug Resistance in Cancer: Understanding Molecular Mechanisms, Immunoprevention and Therapeutic Approaches. Front. Oncol. 2022, 12, 891652. [Google Scholar] [CrossRef] [PubMed]
- Montemagno, C.; Pagès, G. Resistance to Anti-Angiogenic Therapies: A Mechanism Depending on the Time of Exposure to the Drugs. Front. Cell. Dev. Biol. 2020, 8, 584. [Google Scholar] [CrossRef] [PubMed]
- Cree, I.A.; Charlton, P. Molecular Chess? Hallmarks of Anti-Cancer Drug Resistance. BMC Cancer 2017, 17, 10. [Google Scholar] [CrossRef] [PubMed]
- Gomes Marin, J.F.; Nunes, R.F.; Coutinho, A.M.; Zaniboni, E.C.; Costa, L.B.; Barbosa, F.G.; Queiroz, M.A.; Cerri, G.G.; Buchpiguel, C.A. Theranostics in Nuclear Medicine: Emerging and Re-Emerging Integrated Imaging and Therapies in the Era of Precision Oncology. Radio Graph. 2020, 40, 1715–1740. [Google Scholar] [CrossRef] [PubMed]
- Herrmann, K.; Krause, B.J.; Hadaschik, B.; Kunikowska, J.; van Poppel, H.; N’Dow, J.; Sartor, O.; Oyen, W.J.G. Nuclear Medicine Theranostics Comes of Age. Lancet Oncol. 2021, 22, 1497–1498. [Google Scholar] [CrossRef] [PubMed]
- Te Beek, E.T.; Teunissen, J.J.M.; Postema, J.W.A.; Lafeber, A.; ten Broek, M.R.J. Precision Medicine and Theranostics Using Radiopharmaceuticals in Oncology. Br. J. Clin. Pharmacol. 2022, 88, 359–361. [Google Scholar] [CrossRef]
- Fallah, J.; Agrawal, S.; Gittleman, H.; Fiero, M.H.; Subramaniam, S.; John, C.; Chen, W.; Ricks, T.K.; Niu, G.; Fotenos, A.; et al. FDA Approval Summary: Lutetium Lu 177 Vipivotide Tetraxetan for Patients with Metastatic Castration-Resistant Prostate Cancer. Clin. Cancer Res. 2023, 29, 1651–1657. [Google Scholar] [CrossRef]
- Hennrich, U.; Kopka, K. Lutathera®: The First FDA- and EMA-Approved Radiopharmaceutical for Peptide Receptor Radionuclide Therapy. Pharmaceuticals 2019, 12, 114. [Google Scholar] [CrossRef]
- Caers, J.; Duray, E.; Vrancken, L.; Marcion, G.; Bocuzzi, V.; De Veirman, K.; Krasniqi, A.; Lejeune, M.; Withofs, N.; Devoogdt, N.; et al. Radiotheranostic Agents in Hematological Malignancies. Front. Immunol. 2022, 13, 911080. [Google Scholar] [CrossRef]
- Fani, M.; Peitl, P.K.; Velikyan, I. Current Status of Radiopharmaceuticals for the Theranostics of Neuroendocrine Neoplasms. Pharmaceuticals 2017, 10, 30. [Google Scholar] [CrossRef] [PubMed]
- Vahidfar, N.; Aghanejad, A.; Ahmadzadehfar, H.; Farzanehfar, S.; Eppard, E. Theranostic Advances in Breast Cancer in Nuclear Medicine. Int. J. Mol. Sci. 2021, 22, 4597. [Google Scholar] [CrossRef] [PubMed]
- Echavidre, W.; Picco, V.; Faraggi, M.; Montemagno, C. Integrin-Avβ3 as a Therapeutic Target in Glioblastoma: Back to the Future? Pharmaceutics 2022, 14, 1053. [Google Scholar] [CrossRef] [PubMed]
- Montemagno, C.; Cassim, S.; De Leiris, N.; Durivault, J.; Faraggi, M.; Pagès, G. Pancreatic Ductal Adenocarcinoma: The Dawn of the Era of Nuclear Medicine? Int. J. Mol. Sci. 2021, 22, 6413. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Z.; Liu, X.; Chen, D.; Yu, J. Radiotherapy Combined with Immunotherapy: The Dawn of Cancer Treatment. Signal Transduct. Target Ther. 2022, 7, 258. [Google Scholar] [CrossRef] [PubMed]
- Strosberg, J.; El-Haddad, G.; Wolin, E.; Hendifar, A.; Yao, J.; Chasen, B.; Mittra, E.; Kunz, P.L.; Kulke, M.H.; Jacene, H.; et al. Phase 3 Trial of 177Lu-Dotatate for Midgut Neuroendocrine Tumors. N. Engl. J. Med. 2017, 376, 125–135. [Google Scholar] [CrossRef] [PubMed]
- Sartor, O.; de Bono, J.; Chi, K.N.; Fizazi, K.; Herrmann, K.; Rahbar, K.; Tagawa, S.T.; Nordquist, L.T.; Vaishampayan, N.; El-Haddad, G.; et al. Lutetium-177–PSMA-617 for Metastatic Castration-Resistant Prostate Cancer. N. Engl. J. Med. 2021, 385, 1091–1103. [Google Scholar] [CrossRef] [PubMed]
- Hennrich, U.; Eder, M. [68Ga]Ga-PSMA-11: The First FDA-Approved 68Ga-Radiopharmaceutical for PET Imaging of Prostate Cancer. Pharmaceuticals 2021, 14, 713. [Google Scholar] [CrossRef]
- Hennrich, U.; Benešová, M. [68Ga]Ga-DOTA-TOC: The First FDA-Approved 68Ga-Radiopharmaceutical for PET Imaging. Pharmaceuticals 2020, 13, 38. [Google Scholar] [CrossRef]
- Mejia, A.; Vivian, E.; Nwogu, C.; Shah, J.; Longoria, R.; Vo, A.; Shahin, I.; Verma, J.; Bageac, A. Peptide Receptor Radionuclide Therapy Implementation and Results in a Predominantly Gastrointestinal Neuroendocrine Tumor Population. Medicine 2022, 101, e28970. [Google Scholar] [CrossRef]
- Baldelli, R.; Barnabei, A.; Rizza, L.; Isidori, A.M.; Rota, F.; Di Giacinto, P.; Paoloni, A.; Torino, F.; Corsello, S.M.; Lenzi, A.; et al. Somatostatin Analogs Therapy in Gastroenteropancreatic Neuroendocrine Tumors: Current Aspects and New Perspectives. Front. Endocrinol. 2014, 5, 7. [Google Scholar] [CrossRef] [PubMed]
- McClellan, K.; Chen, E.Y.; Kardosh, A.; Lopez, C.D.; Del Rivero, J.; Mallak, N.; Rocha, F.G.; Koethe, Y.; Pommier, R.; Mittra, E.; et al. Therapy Resistant Gastroenteropancreatic Neuroendocrine Tumors. Cancers 2022, 14, 4769. [Google Scholar] [CrossRef] [PubMed]
- Seifert, V.; Richter, S.; Bechmann, N.; Bachmann, M.; Ziegler, C.G.; Pietzsch, J.; Ullrich, M. HIF2alpha-Associated Pseudohypoxia Promotes Radioresistance in Pheochromocytoma: Insights from 3D Models. Cancers 2021, 13, 385. [Google Scholar] [CrossRef] [PubMed]
- Vermeulen, K.; Van de Voorde, M.; Segers, C.; Coolkens, A.; Rodriguez Pérez, S.; Daems, N.; Duchemin, C.; Crabbé, M.; Opsomer, T.; Saldarriaga Vargas, C.; et al. Exploring the Potential of High-Molar-Activity Samarium-153 for Targeted Radionuclide Therapy with [153Sm]Sm-DOTA-TATE. Pharmaceutics 2022, 14, 2566. [Google Scholar] [CrossRef] [PubMed]
- Kunikowska, J.; Królicki, L. Targeted α-Emitter Therapy of Neuroendocrine Tumors. Semin. Nucl. Med. 2020, 50, 171–176. [Google Scholar] [CrossRef] [PubMed]
- Tafreshi, N.K.; Pandya, D.N.; Tichacek, C.J.; Budzevich, M.M.; Wang, Z.; Reff, J.N.; Engelman, R.W.; Boulware, D.C.; Chiappori, A.A.; Strosberg, J.R.; et al. Preclinical Evaluation of [225Ac]Ac-DOTA-TATE for Treatment of Lung Neuroendocrine Neoplasms. Eur. J. Nucl. Med. Mol. Imaging 2021, 48, 3408–3421. [Google Scholar] [CrossRef] [PubMed]
- Stallons, T.A.R.; Saidi, A.; Tworowska, I.; Delpassand, E.S.; Torgue, J.J. Preclinical Investigation of 212Pb-DOTAMTATE for Peptide Receptor Radionuclide Therapy in a Neuroendocrine Tumor Model. Mol. Cancer Ther. 2019, 18, 1012–1021. [Google Scholar] [CrossRef] [PubMed]
- Delpassand, E.S.; Tworowska, I.; Esfandiari, R.; Torgue, J.; Hurt, J.; Shafie, A.; Núñez, R. Targeted α-Emitter Therapy with 212Pb-DOTAMTATE for the Treatment of Metastatic SSTR-Expressing Neuroendocrine Tumors: First-in-Humans Dose-Escalation Clinical Trial. J. Nucl. Med. 2022, 63, 1326–1333. [Google Scholar] [CrossRef]
- Paterson, B.M.; Roselt, P.; Denoyer, D.; Cullinane, C.; Binns, D.; Noonan, W.; Jeffery, C.M.; Price, R.I.; White, J.M.; Hicks, R.J.; et al. PET Imaging of Tumours with a 64Cu Labeled Macrobicyclic Cage Amine Ligand Tethered to Tyr3-Octreotate. Dalton Trans. 2014, 43, 1386–1396. [Google Scholar] [CrossRef]
- Hicks, R.J.; Jackson, P.; Kong, G.; Ware, R.E.; Hofman, M.S.; Pattison, D.A.; Akhurst, T.A.; Drummond, E.; Roselt, P.; Callahan, J.; et al. 64Cu-SARTATE PET Imaging of Patients with Neuroendocrine Tumors Demonstrates High Tumor Uptake and Retention, Potentially Allowing Prospective Dosimetry for Peptide Receptor Radionuclide Therapy. J. Nucl. Med. 2019, 60, 777–785. [Google Scholar] [CrossRef]
- Cullinane, C.; Jeffery, C.M.; Roselt, P.D.; van Dam, E.M.; Jackson, S.; Kuan, K.; Jackson, P.; Binns, D.; van Zuylekom, J.; Harris, M.J.; et al. Peptide Receptor Radionuclide Therapy with 67Cu-CuSarTATE Is Highly Efficacious Against a Somatostatin-Positive Neuroendocrine Tumor Model. J. Nucl. Med. 2020, 61, 1800–1805. [Google Scholar] [CrossRef] [PubMed]
- Dearling, J.L.J.; van Dam, E.M.; Harris, M.J.; Packard, A.B. Detection and Therapy of Neuroblastoma Minimal Residual Disease Using [64/67Cu]Cu-SARTATE in a Preclinical Model of Hepatic Metastases. EJNMMI Res. 2021, 11, 20. [Google Scholar] [CrossRef] [PubMed]
- Jha, A.; Patel, M.; Carrasquillo, J.A.; Chen, C.C.; Millo, C.; Maass-Moreno, R.; Ling, A.; Lin, F.I.; Lechan, R.M.; Hope, T.A.; et al. Choice Is Good at Times: The Emergence of [64Cu]Cu-DOTATATE–Based Somatostatin Receptor Imaging in the Era of [68Ga]Ga-DOTATATE. J. Nucl. Med. 2022, 63, 1300–1301. [Google Scholar] [CrossRef] [PubMed]
- Bailey, D.L.; Willowson, K.P.; Harris, M.; Biggin, C.; Aslani, A.; Lengkeek, N.A.; Stoner, J.; Eslick, M.E.; Marquis, H.; Parker, M.; et al. 64Cu Treatment Planning and 67Cu Therapy with Radiolabeled [64Cu/67Cu]MeCOSar-Octreotate in Subjects with Unresectable Multifocal Meningioma: Initial Results for Human Imaging, Safety, Biodistribution, and Radiation Dosimetry. J. Nucl. Med. 2023, 64, 704–710. [Google Scholar] [CrossRef] [PubMed]
- Wild, D.; Fani, M.; Fischer, R.; Del Pozzo, L.; Kaul, F.; Krebs, S.; Fischer, R.; Rivier, J.E.F.; Reubi, J.C.; Maecke, H.R.; et al. Comparison of Somatostatin Receptor Agonist and Antagonist for Peptide Receptor Radionuclide Therapy: A Pilot Study. J. Nucl. Med. 2014, 55, 1248–1252. [Google Scholar] [CrossRef] [PubMed]
- Borgna, F.; Haller, S.; Rodriguez, J.M.M.; Ginj, M.; Grundler, P.V.; Zeevaart, J.R.; Köster, U.; Schibli, R.; van der Meulen, N.P.; Müller, C. Combination of Terbium-161 with Somatostatin Receptor Antagonists-a Potential Paradigm Shift for the Treatment of Neuroendocrine Neoplasms. Eur. J. Nucl. Med. Mol. Imaging 2022, 49, 1113–1126. [Google Scholar] [CrossRef] [PubMed]
- Baum, R.P.; Zhang, J.; Schuchardt, C.; Müller, D.; Mäcke, H. First-in-Humans Study of the SSTR Antagonist 177Lu-DOTA-LM3 for Peptide Receptor Radionuclide Therapy in Patients with Metastatic Neuroendocrine Neoplasms: Dosimetry, Safety, and Efficacy. J. Nucl. Med. 2021, 62, 1571–1581. [Google Scholar] [CrossRef] [PubMed]
- Plas, P.; Limana, L.; Carré, D.; Thiongane, A.; Raguin, O.; Mansi, R.; Meyer-Losic, F.; Lezmi, S. Comparison of the Anti-Tumour Activity of the Somatostatin Receptor (SST) Antagonist [177Lu]Lu-Satoreotide Tetraxetan and the Agonist [177Lu]Lu-DOTA-TATE in Mice Bearing AR42J SST2-Positive Tumours. Pharmaceuticals 2022, 15, 1085. [Google Scholar] [CrossRef]
- Nock, B.A.; Kanellopoulos, P.; Moon, E.S.; Rouchota, M.; Loudos, G.; Ballal, S.; Yadav, M.P.; Bal, C.; Mishra, P.; Sheokand, P.; et al. [111In]In/[177Lu]Lu-AAZTA5-LM4 SST2R-Antagonists in Cancer Theranostics: From Preclinical Testing to First Patient Results. Pharmaceutics 2023, 15, 776. [Google Scholar] [CrossRef]
- Baum, R.P.; Singh, A.; Kulkarni, H.R.; Bernhardt, P.; Rydén, T.; Schuchardt, C.; Gracheva, N.; Grundler, P.V.; Köster, U.; Müller, D.; et al. First-in-Humans Application of 161Tb: A Feasibility Study Using 161Tb-DOTATOC. J. Nucl. Med. 2021, 62, 1391–1397. [Google Scholar] [CrossRef]
- Hennrich, U.; Eder, M. [177Lu]Lu-PSMA-617 (PluvictoTM): The First FDA-Approved Radiotherapeutical for Treatment of Prostate Cancer. Pharmaceuticals 2022, 15, 1292. [Google Scholar] [CrossRef] [PubMed]
- Stuparu, A.D.; Capri, J.R.; Meyer, C.A.L.; Le, T.M.; Evans-Axelsson, S.L.; Current, K.; Lennox, M.; Mona, C.E.; Fendler, W.P.; Calais, J.; et al. Mechanisms of Resistance to Prostate-Specific Membrane Antigen-Targeted Radioligand Therapy in a Mouse Model of Prostate Cancer. J. Nucl. Med. 2021, 62, 989–995. [Google Scholar] [CrossRef] [PubMed]
- Lauri, C.; Chiurchioni, L.; Russo, V.M.; Zannini, L.; Signore, A. PSMA Expression in Solid Tumors beyond the Prostate Gland: Ready for Theranostic Applications? J. Clin. Med. 2022, 11, 6590. [Google Scholar] [CrossRef] [PubMed]
- Patel, D.; Loh, H.; Le, K.; Stevanovic, A.; Mansberg, R. Incidental Detection of Hepatocellular Carcinoma on 68Ga-Labeled Prostate-Specific Membrane Antigen PET/CT. Clin. Nucl. Med. 2017, 42, 881. [Google Scholar] [CrossRef]
- Kesler, M.; Levine, C.; Hershkovitz, D.; Mishani, E.; Menachem, Y.; Lerman, H.; Zohar, Y.; Shibolet, O.; Even-Sapir, E. 68Ga-Labeled Prostate-Specific Membrane Antigen Is a Novel PET/CT Tracer for Imaging of Hepatocellular Carcinoma: A Prospective Pilot Study. J. Nucl. Med. 2019, 60, 185–191. [Google Scholar] [CrossRef] [PubMed]
- Lu, Q.; Long, Y.; Fan, K.; Shen, Z.; Gai, Y.; Liu, Q.; Jiang, D.; Cai, W.; Wan, C.; Lan, X. PET Imaging of Hepatocellular Carcinoma by Targeting Tumor-Associated Endothelium Using [68Ga]Ga-PSMA-617. Eur. J. Nucl. Med. Mol. Imaging 2022, 49, 4000–4013. [Google Scholar] [CrossRef] [PubMed]
- Lu, Q.; Long, Y.; Gai, Y.; Liu, Q.; Jiang, D.; Lan, X. [177Lu]Lu-PSMA-617 Theranostic Probe for Hepatocellular Carcinoma Imaging and Therapy. Eur. J. Nucl. Med. Mol. Imaging 2023, 50, 2342–2352. [Google Scholar] [CrossRef]
- Hirmas, N.; Leyh, C.; Sraieb, M.; Barbato, F.; Schaarschmidt, B.M.; Umutlu, L.; Nader, M.; Wedemeyer, H.; Ferdinandus, J.; Rischpler, C.; et al. 68Ga-PSMA-11 PET/CT Improves Tumor Detection and Impacts Management in Patients with Hepatocellular Carcinoma. J. Nucl. Med. 2021, 62, 1235–1241. [Google Scholar] [CrossRef]
- Weller, M.; Le Rhun, E.; Preusser, M.; Tonn, J.-C.; Roth, P. How We Treat Glioblastoma. ESMO Open 2019, 4, e000520. [Google Scholar] [CrossRef]
- Holzgreve, A.; Biczok, A.; Ruf, V.C.; Liesche-Starnecker, F.; Steiger, K.; Kirchner, M.A.; Unterrainer, M.; Mittlmeier, L.; Herms, J.; Schlegel, J.; et al. PSMA Expression in Glioblastoma as a Basis for Theranostic Approaches: A Retrospective, Correlational Panel Study Including Immunohistochemistry, Clinical Parameters and PET Imaging. Front. Oncol. 2021, 11, 646387. [Google Scholar] [CrossRef]
- Saffar, H.; Noohi, M.; Tavangar, S.M.; Saffar, H.; Azimi, S. Expression of Prostate-Specific Membrane Antigen (PSMA) in Brain Glioma and Its Correlation with Tumor Grade. Iran. J. Pathol. 2018, 13, 45–53. [Google Scholar] [PubMed]
- Traub-Weidinger, T.; Poetsch, N.; Woehrer, A.; Klebermass, E.-M.; Bachnik, T.; Preusser, M.; Mischkulnig, M.; Kiesel, B.; Widhalm, G.; Mitterhauser, M.; et al. PSMA Expression in 122 Treatment Naive Glioma Patients Related to Tumor Metabolism in 11C-Methionine PET and Survival. J. Pers. Med. 2021, 11, 624. [Google Scholar] [CrossRef] [PubMed]
- More, S.; Naiker, T.; Jacobs, N.; Oompie, F.; Prasad, V. Short-Interval, Low-Dose [ 177 Lu]Lu-Prostate-Specific Membrane Antigen in the Treatment of Refractory Glioblastoma. Clin. Nucl. Med. 2023, 48, e217–e218. [Google Scholar] [CrossRef] [PubMed]
- Rahbar, K.; Bode, A.; Weckesser, M.; Avramovic, N.; Claesener, M.; Stegger, L.; Bögemann, M. Radioligand Therapy With 177Lu-PSMA-617 as A Novel Therapeutic Option in Patients With Metastatic Castration Resistant Prostate Cancer. Clin. Nucl. Med. 2016, 41, 522–528. [Google Scholar] [CrossRef] [PubMed]
- Khreish, F.; Ebert, N.; Ries, M.; Maus, S.; Rosar, F.; Bohnenberger, H.; Stemler, T.; Saar, M.; Bartholomä, M.; Ezziddin, S. 225Ac-PSMA-617/177Lu-PSMA-617 Tandem Therapy of Metastatic Castration-Resistant Prostate Cancer: Pilot Experience. Eur. J. Nucl. Med. Mol. Imaging 2020, 47, 721–728. [Google Scholar] [CrossRef]
- Kratochwil, C.; Bruchertseifer, F.; Rathke, H.; Hohenfellner, M.; Giesel, F.L.; Haberkorn, U.; Morgenstern, A. Targeted α-Therapy of Metastatic Castration-Resistant Prostate Cancer with 225Ac-PSMA-617: Swimmer-Plot Analysis Suggests Efficacy Regarding Duration of Tumor Control. J. Nucl. Med. 2018, 59, 795–802. [Google Scholar] [CrossRef] [PubMed]
- Feuerecker, B.; Tauber, R.; Knorr, K.; Heck, M.; Beheshti, A.; Seidl, C.; Bruchertseifer, F.; Pickhard, A.; Gafita, A.; Kratochwil, C.; et al. Activity and Adverse Events of Actinium-225-PSMA-617 in Advanced Metastatic Castration-Resistant Prostate Cancer After Failure of Lutetium-177-PSMA. Eur. Urol. 2021, 79, 343–350. [Google Scholar] [CrossRef] [PubMed]
- Sathekge, M.; Bruchertseifer, F.; Vorster, M.; Lawal, I.O.; Knoesen, O.; Mahapane, J.; Davis, C.; Reyneke, F.; Maes, A.; Kratochwil, C.; et al. Predictors of Overall and Disease-Free Survival in Metastatic Castration-Resistant Prostate Cancer Patients Receiving 225Ac-PSMA-617 Radioligand Therapy. J. Nucl. Med. 2020, 61, 62–69. [Google Scholar] [CrossRef]
- Langbein, T.; Kulkarni, H.R.; Schuchardt, C.; Mueller, D.; Volk, G.F.; Baum, R.P. Salivary Gland Toxicity of PSMA-Targeted Radioligand Therapy with 177Lu-PSMA and Combined 225Ac- and 177Lu-Labeled PSMA Ligands (TANDEM-PRLT) in Advanced Prostate Cancer: A Single-Center Systematic Investigation. Diagnostics 2022, 12, 1926. [Google Scholar] [CrossRef]
- Ballal, S.; Yadav, M.P.; Sahoo, R.K.; Tripathi, M.; Dwivedi, S.N.; Bal, C. 225 Ac-PSMA-617-Targeted Alpha Therapy for the Treatment of Metastatic Castration-Resistant Prostate Cancer: A Systematic Review and Meta-Analysis. Prostate 2021, 81, 580–591. [Google Scholar] [CrossRef]
- Ma, J.; Li, L.; Liao, T.; Gong, W.; Zhang, C. Efficacy and Safety of 225Ac-PSMA-617-Targeted Alpha Therapy in Metastatic Castration-Resistant Prostate Cancer: A Systematic Review and Meta-Analysis. Front. Oncol. 2022, 12, 796657. [Google Scholar] [CrossRef] [PubMed]
- Banerjee, S.R.; Kumar, V.; Lisok, A.; Chen, J.; Minn, I.; Brummet, M.; Boinapally, S.; Cole, M.; Ngen, E.; Wharram, B.; et al. 177Lu-Labeled Low-Molecular-Weight Agents for PSMA-Targeted Radiopharmaceutical Therapy. Eur. J. Nucl. Med. Mol. Imaging 2019, 46, 2545–2557. [Google Scholar] [CrossRef] [PubMed]
- Banerjee, S.R.; Lisok, A.; Minn, I.; Josefsson, A.; Kumar, V.; Brummet, M.; Boinapally, S.; Brayton, C.; Mease, R.C.; Sgouros, G.; et al. Preclinical Evaluation of 213Bi- and 225Ac-Labeled Low-Molecular-Weight Compounds for Radiopharmaceutical Therapy of Prostate Cancer. J. Nucl. Med. 2021, 62, 980–988. [Google Scholar] [CrossRef] [PubMed]
- Mease, R.C.; Kang, C.M.; Kumar, V.; Banerjee, S.R.; Minn, I.; Brummet, M.; Gabrielson, K.L.; Feng, Y.; Park, A.; Kiess, A.P.; et al. An Improved 211At-Labeled Agent for PSMA-Targeted α-Therapy. J. Nucl. Med. 2022, 63, 259–267. [Google Scholar] [CrossRef] [PubMed]
- Lepareur, N.; Lacœuille, F.; Bouvry, C.; Hindré, F.; Garcion, E.; Chérel, M.; Noiret, N.; Garin, E.; Knapp, F.F.R. Rhenium-188 Labeled Radiopharmaceuticals: Current Clinical Applications in Oncology and Promising Perspectives. Front. Med. 2019, 6, 132. [Google Scholar] [CrossRef] [PubMed]
- Cardinale, J.; Giesel, F.L.; Wensky, C.; Rathke, H.G.; Haberkorn, U.; Kratochwil, C. PSMA-GCK01: A Generator-Based 99mTc/188Re Theranostic Ligand for the Prostate-Specific Membrane Antigen. J. Nucl. Med. 2023, 64, 1069–1075. [Google Scholar] [CrossRef] [PubMed]
- Zang, J.; Fan, X.; Wang, H.; Liu, Q.; Wang, J.; Li, H.; Li, F.; Jacobson, O.; Niu, G.; Zhu, Z.; et al. First-in-Human Study of 177Lu-EB-PSMA-617 in Patients with Metastatic Castration-Resistant Prostate Cancer. Eur. J. Nucl. Med. Mol. Imaging 2019, 46, 148–158. [Google Scholar] [CrossRef]
- Liu, Q.; Zang, J.; Sui, H.; Ren, J.; Guo, H.; Wang, H.; Wang, R.; Jacobson, O.; Zhang, J.; Cheng, Y.; et al. Peptide Receptor Radionuclide Therapy of Late-Stage Neuroendocrine Tumor Patients with Multiple Cycles of 177Lu-DOTA-EB-TATE. J. Nucl. Med. 2021, 62, 386–392. [Google Scholar] [CrossRef]
- Zhang, J.; Wang, H.; Jacobson, O.; Cheng, Y.; Niu, G.; Li, F.; Bai, C.; Zhu, Z.; Chen, X. Safety, Pharmacokinetics, and Dosimetry of a Long-Acting Radiolabeled Somatostatin Analog 177Lu-DOTA-EB-TATE in Patients with Advanced Metastatic Neuroendocrine Tumors. J. Nucl. Med. 2018, 59, 1699–1705. [Google Scholar] [CrossRef]
- Zang, J.; Liu, Q.; Sui, H.; Wang, R.; Jacobson, O.; Fan, X.; Zhu, Z.; Chen, X. 177Lu-EB-PSMA Radioligand Therapy with Escalating Doses in Patients with Metastatic Castration-Resistant Prostate Cancer. J. Nucl. Med. 2020, 61, 1772–1778. [Google Scholar] [CrossRef]
- Wang, G.; Zhou, M.; Zang, J.; Jiang, Y.; Chen, X.; Zhu, Z.; Chen, X. A Pilot Study of 68 Ga-PSMA-617 PET/CT Imaging and 177Lu-EB-PSMA-617 Radioligand Therapy in Patients with Adenoid Cystic Carcinoma. EJNMMI Res. 2022, 12, 52. [Google Scholar] [CrossRef] [PubMed]
- Wen, X.; Xu, P.; Zeng, X.; Liu, J.; Du, C.; Zeng, X.; Cheng, X.; Wang, X.; Liang, Y.; Zhao, T.; et al. Development of [177Lu]Lu-LNC1003 for Radioligand Therapy of Prostate Cancer with a Moderate Level of PSMA Expression. Eur. J. Nucl. Med. Mol. Imaging 2023, 50, 2846–2860. [Google Scholar] [CrossRef] [PubMed]
- Meyer, C.; Prasad, V.; Stuparu, A.; Kletting, P.; Glatting, G.; Miksch, J.; Solbach, C.; Lueckerath, K.; Nyiranshuti, L.; Zhu, S.; et al. Comparison of PSMA-TO-1 and PSMA-617 Labeled with Gallium-68, Lutetium-177 and Actinium-225. EJNMMI Res. 2022, 12, 65. [Google Scholar] [CrossRef] [PubMed]
- Iikuni, S.; Tarumizu, Y.; Nakashima, K.; Higaki, Y.; Ichikawa, H.; Watanabe, H.; Ono, M. Radiotheranostics Using a Novel 225Ac-Labeled Radioligand with Improved Pharmacokinetics Targeting Prostate-Specific Membrane Antigen. J. Med. Chem. 2021, 64, 13429–13438. [Google Scholar] [CrossRef] [PubMed]
- Tsuchihashi, S.; Nakashima, K.; Tarumizu, Y.; Ichikawa, H.; Jinda, H.; Watanabe, H.; Ono, M. Development of Novel 111In/225Ac-Labeled Agent Targeting PSMA for Highly Efficient Cancer Radiotheranostics. J. Med. Chem. 2023, 66, 8043–8053. [Google Scholar] [CrossRef]
- Tagawa, S.T.; Milowsky, M.I.; Morris, M.; Vallabhajosula, S.; Christos, P.; Akhtar, N.H.; Osborne, J.; Goldsmith, S.J.; Larson, S.; Taskar, N.P.; et al. Phase II Study of Lutetium-177-Labeled Anti-Prostate-Specific Membrane Antigen Monoclonal Antibody J591 for Metastatic Castration-Resistant Prostate Cancer. Clin. Cancer Res. 2013, 19, 5182–5191. [Google Scholar] [CrossRef] [PubMed]
- Hammer, S.; Hagemann, U.B.; Zitzmann-Kolbe, S.; Larsen, A.; Ellingsen, C.; Geraudie, S.; Grant, D.; Indrevoll, B.; Smeets, R.; von Ahsen, O.; et al. Preclinical Efficacy of a PSMA-Targeted Thorium-227 Conjugate (PSMA-TTC), a Targeted Alpha Therapy for Prostate Cancer. Clin. Cancer Res. 2020, 26, 1985–1996. [Google Scholar] [CrossRef] [PubMed]
- Hammer, S.; Schlicker, A.; Zitzmann-Kolbe, S.; Baumgart, S.; Hagemann, U.B.; Scholz, A.; Haendler, B.; Lejeune, P.; Karlsson, J.; Ellingsen, C.; et al. Darolutamide Potentiates the Antitumor Efficacy of a PSMA-Targeted Thorium-227 Conjugate by a Dual Mode of Action in Prostate Cancer Models. Clin. Cancer Res. 2021, 27, 4367–4378. [Google Scholar] [CrossRef]
- Zippel, C.; Ronski, S.C.; Bohnet-Joschko, S.; Giesel, F.L.; Kopka, K. Current Status of PSMA-Radiotracers for Prostate Cancer: Data Analysis of Prospective Trials Listed on ClinicalTrials.Gov. Pharmaceuticals 2020, 13, 12. [Google Scholar] [CrossRef]
- Olivier, P.; Giraudet, A.-L.; Skanjeti, A.; Merlin, C.; Weinmann, P.; Rudolph, I.; Hoepping, A.; Gauthé, M. Phase III Study of 18F-PSMA-1007 Versus 18F-Fluorocholine PET/CT for Localization of Prostate Cancer Biochemical Recurrence: A Prospective, Randomized, Crossover Multicenter Study. J. Nucl. Med. 2023, 64, 579–585. [Google Scholar] [CrossRef]
- Fendler, W.P.; Eiber, M.; Beheshti, M.; Bomanji, J.; Calais, J.; Ceci, F.; Cho, S.Y.; Fanti, S.; Giesel, F.L.; Goffin, K.; et al. PSMA PET/CT: Joint EANM Procedure Guideline/SNMMI Procedure Standard for Prostate Cancer Imaging 2.0. Eur. J. Nucl. Med. Mol. Imaging 2023, 50, 1466–1486. [Google Scholar] [CrossRef] [PubMed]
- Schwarzenboeck, S.M.; Rauscher, I.; Bluemel, C.; Fendler, W.P.; Rowe, S.P.; Pomper, M.G.; Afshar-Oromieh, A.; Herrmann, K.; Eiber, M. PSMA Ligands for PET Imaging of Prostate Cancer. J. Nucl. Med. 2017, 58, 1545–1552. [Google Scholar] [CrossRef] [PubMed]
- Chandekar, K.R.; Singh, H.; Kumar, R.; Kumar, S.; Kakkar, N.; Mittal, B.R.; Singh, S.K. Comparison of 18 F-PSMA-1007 PET/CT With 68 Ga-PSMA-11 PET/CT for Initial Staging in Intermediate- and High-Risk Prostate Cancer. Clin. Nucl. Med. 2023, 48, e1–e8. [Google Scholar] [CrossRef] [PubMed]
- Kuten, J.; Fahoum, I.; Savin, Z.; Shamni, O.; Gitstein, G.; Hershkovitz, D.; Mabjeesh, N.J.; Yossepowitch, O.; Mishani, E.; Even-Sapir, E. Head-to-Head Comparison of 68Ga-PSMA-11 with 18F-PSMA-1007 PET/CT in Staging Prostate Cancer Using Histopathology and Immunohistochemical Analysis as a Reference Standard. J. Nucl. Med. 2020, 61, 527–532. [Google Scholar] [CrossRef] [PubMed]
- Alberts, I.L.; Seide, S.E.; Mingels, C.; Bohn, K.P.; Shi, K.; Zacho, H.D.; Rominger, A.; Afshar-Oromieh, A. Comparing the Diagnostic Performance of Radiotracers in Recurrent Prostate Cancer: A Systematic Review and Network Meta-Analysis. Eur. J. Nucl. Med. Mol. Imaging 2021, 48, 2978–2989. [Google Scholar] [CrossRef] [PubMed]
- Kirchner, M.A.; Holzgreve, A.; Brendel, M.; Orth, M.; Ruf, V.C.; Steiger, K.; Pötter, D.; Gold, L.; Unterrainer, M.; Mittlmeier, L.M.; et al. PSMA PET Imaging in Glioblastoma: A Preclinical Evaluation and Theranostic Outlook. Front. Oncol. 2021, 11, 774017. [Google Scholar] [CrossRef] [PubMed]
- Xin, L.; Gao, J.; Zheng, Z.; Chen, Y.; Lv, S.; Zhao, Z.; Yu, C.; Yang, X.; Zhang, R. Fibroblast Activation Protein-α as a Target in the Bench-to-Bedside Diagnosis and Treatment of Tumors: A Narrative Review. Front. Oncol. 2021, 11, 648187. [Google Scholar] [CrossRef]
- Li, M.; Cheng, X.; Rong, R.; Gao, Y.; Tang, X.; Chen, Y. High Expression of Fibroblast Activation Protein (FAP) Predicts Poor Outcome in High-Grade Serous Ovarian Cancer. BMC Cancer 2020, 20, 1032. [Google Scholar] [CrossRef]
- Liu, F.; Qi, L.; Liu, B.; Liu, J.; Zhang, H.; Che, D.; Cao, J.; Shen, J.; Geng, J.; Bi, Y.; et al. Fibroblast Activation Protein Overexpression and Clinical Implications in Solid Tumors: A Meta-Analysis. PLoS ONE 2015, 10, e0116683. [Google Scholar] [CrossRef]
- Cheng, Z.; Wang, S.; Xu, S.; Du, B.; Li, X.; Li, Y. FAPI PET/CT in Diagnostic and Treatment Management of Colorectal Cancer: Review of Current Research Status. J. Clin. Med. 2023, 12, 577. [Google Scholar] [CrossRef]
- Dong, Y.; Zhou, H.; Alhaskawi, A.; Wang, Z.; Lai, J.; Yao, C.; Liu, Z.; Hasan Abdullah Ezzi, S.; Goutham Kota, V.; Hasan Abdulla Hasan Abdulla, M.; et al. The Superiority of Fibroblast Activation Protein Inhibitor (FAPI) PET/CT Versus FDG PET/CT in the Diagnosis of Various Malignancies. Cancers 2023, 15, 1193. [Google Scholar] [CrossRef] [PubMed]
- Huang, R.; Pu, Y.; Huang, S.; Yang, C.; Yang, F.; Pu, Y.; Li, J.; Chen, L.; Huang, Y. FAPI-PET/CT in Cancer Imaging: A Potential Novel Molecule of the Century. Front. Oncol. 2022, 12, 854658. [Google Scholar] [CrossRef] [PubMed]
- Röhrich, M.; Naumann, P.; Giesel, F.L.; Choyke, P.L.; Staudinger, F.; Wefers, A.; Liew, D.P.; Kratochwil, C.; Rathke, H.; Liermann, J.; et al. Impact of 68Ga-FAPI PET/CT Imaging on the Therapeutic Management of Primary and Recurrent Pancreatic Ductal Adenocarcinomas. J. Nucl. Med. 2021, 62, 779–786. [Google Scholar] [CrossRef] [PubMed]
- Ding, J.; Qiu, J.; Zhang, T.; Huo, L. The Performance of 68Ga-FAPI-04 PET/CT in Patients with Resectable Primary Pancreatic Ductal Carcinoma. J. Nucl. Med. 2022, 63, 2379. [Google Scholar]
- Wang, H.; Zhu, W.; Ren, S.; Kong, Y.; Huang, Q.; Zhao, J.; Guan, Y.; Jia, H.; Chen, J.; Lu, L.; et al. 68Ga-FAPI-04 Versus 18F-FDG PET/CT in the Detection of Hepatocellular Carcinoma. Front. Oncol. 2021, 11, 693640. [Google Scholar] [CrossRef] [PubMed]
- Wu, M.; Wang, X.; Yang, X.; Wang, Y.; Zhao, H.; Huo, L. 68Ga-FAPI and 18F-FDG PET/CT in Evaluation of Primary and Metastatic Lesions in Late-Stage Hepatocellular Carcinoma. J. Nucl. Med. 2022, 63, 2373. [Google Scholar]
- Lindner, T.; Loktev, A.; Altmann, A.; Giesel, F.; Kratochwil, C.; Debus, J.; Jäger, D.; Mier, W.; Haberkorn, U. Development of Quinoline Based Theranostic Ligands for the Targeting of Fibroblast Activation Protein. J. Nucl. Med. 2018, 59, 1415–1422. [Google Scholar] [CrossRef] [PubMed]
- Watabe, T.; Liu, Y.; Kaneda-Nakashima, K.; Shirakami, Y.; Lindner, T.; Ooe, K.; Toyoshima, A.; Nagata, K.; Shimosegawa, E.; Haberkorn, U.; et al. Theranostics Targeting Fibroblast Activation Protein in the Tumor Stroma: 64Cu- and 225Ac-Labeled FAPI-04 in Pancreatic Cancer Xenograft Mouse Models. J. Nucl. Med. 2020, 61, 563–569. [Google Scholar] [CrossRef]
- Zhang, H.; An, J.; Wu, P.; Zhang, C.; Zhao, Y.; Tan, D.; Shi, C.; Ge, X. The Application of [68Ga]-Labeled FAPI-04 PET/CT for Targeting and Early Detection of Pancreatic Carcinoma in Patient-Derived Orthotopic Xenograft Models. Contrast Media Mol. Imaging 2022, 2022, 6596702. [Google Scholar] [CrossRef]
- Ma, H.; Li, F.; Shen, G.; Cai, H.; Liu, W.; Lan, T.; Yang, Y.; Yang, J.; Liao, J.; Liu, N. Synthesis and Preliminary Evaluation of 131I-Labeled FAPI Tracers for Cancer Theranostics. Mol. Pharm. 2021, 18, 4179–4187. [Google Scholar] [CrossRef]
- Ma, H.; Li, F.; Shen, G.; Pan, L.; Liu, W.; Liang, R.; Lan, T.; Yang, Y.; Yang, J.; Liao, J.; et al. In Vitro and in Vivo Evaluation of 211At-Labeled Fibroblast Activation Protein Inhibitor for Glioma Treatment. Bioorg. Med. Chem. 2022, 55, 116600. [Google Scholar] [CrossRef] [PubMed]
- Kuyumcu, S.; Kovan, B.; Sanli, Y.; Buyukkaya, F.; Has Simsek, D.; Özkan, Z.G.; Isik, E.G.; Ekenel, M.; Turkmen, C. Safety of Fibroblast Activation Protein-Targeted Radionuclide Therapy by a Low-Dose Dosimetric Approach Using 177Lu-FAPI04. Clin. Nucl. Med. 2021, 46, 641–646. [Google Scholar] [CrossRef] [PubMed]
- Loktev, A.; Lindner, T.; Burger, E.-M.; Altmann, A.; Giesel, F.; Kratochwil, C.; Debus, J.; Marmé, F.; Jäger, D.; Mier, W.; et al. Development of Fibroblast Activation Protein–Targeted Radiotracers with Improved Tumor Retention. J. Nucl. Med. 2019, 60, 1421–1429. [Google Scholar] [CrossRef] [PubMed]
- Liu, Y.; Watabe, T.; Kaneda-Nakashima, K.; Shirakami, Y.; Naka, S.; Ooe, K.; Toyoshima, A.; Nagata, K.; Haberkorn, U.; Kratochwil, C.; et al. Fibroblast Activation Protein Targeted Therapy Using [177Lu]FAPI-46 Compared with [225Ac]FAPI-46 in a Pancreatic Cancer Model. Eur. J. Nucl. Med. Mol. Imaging 2022, 49, 871–880. [Google Scholar] [CrossRef] [PubMed]
- Assadi, M.; Rekabpour, S.J.; Jafari, E.; Divband, G.; Nikkholgh, B.; Amini, H.; Kamali, H.; Ebrahimi, S.; Shakibazad, N.; Jokar, N.; et al. Feasibility and Therapeutic Potential of 177Lu-Fibroblast Activation Protein Inhibitor-46 for Patients With Relapsed or Refractory Cancers: A Preliminary Study. Clin. Nucl. Med. 2021, 46, e523–e530. [Google Scholar] [CrossRef]
- Fendler, W.P.; Pabst, K.M.; Kessler, L.; Fragoso Costa, P.; Ferdinandus, J.; Weber, M.; Lippert, M.; Lueckerath, K.; Umutlu, L.; Kostbade, K.; et al. Safety and Efficacy of 90Y-FAPI-46 Radioligand Therapy in Patients with Advanced Sarcoma and Other Cancer Entities. Clin. Cancer Res. 2022, 28, 4346–4353. [Google Scholar] [CrossRef]
- Zboralski, D.; Hoehne, A.; Bredenbeck, A.; Schumann, A.; Nguyen, M.; Schneider, E.; Ungewiss, J.; Paschke, M.; Haase, C.; von Hacht, J.L.; et al. Preclinical Evaluation of FAP-2286 for Fibroblast Activation Protein Targeted Radionuclide Imaging and Therapy. Eur. J. Nucl. Med. Mol. Imaging 2022, 49, 3651–3667. [Google Scholar] [CrossRef]
- Ballal, S.; Yadav, M.P.; Roesch, F.; Wakade, N.; Raju, S.; Sheokand, P.; Mishra, P.; Moon, E.S.; Tripathi, M.; Martin, M.; et al. Head-to-Head Comparison between [68Ga]Ga-DOTA.SA.FAPi and [18F]F-FDG PET/CT Imaging in Patients with Breast Cancer. Pharmaceuticals 2023, 16, 521. [Google Scholar] [CrossRef]
- Ballal, S.; Yadav, M.P.; Moon, E.S.; Kramer, V.S.; Roesch, F.; Kumari, S.; Bal, C. First-In-Human Results on the Biodistribution, Pharmacokinetics, and Dosimetry of [177Lu]Lu-DOTA.SA.FAPi and [177Lu]Lu-DOTAGA.(SA.FAPi)2. Pharmaceuticals 2021, 14, 1212. [Google Scholar] [CrossRef]
- Ballal, S.; Yadav, M.P.; Moon, E.S.; Roesch, F.; Kumari, S.; Agarwal, S.; Tripathi, M.; Sahoo, R.K.; Mangu, B.S.; Tupalli, A.; et al. Novel Fibroblast Activation Protein Inhibitor-Based Targeted Theranostics for Radioiodine-Refractory Differentiated Thyroid Cancer Patients: A Pilot Study. Thyroid 2022, 32, 65–77. [Google Scholar] [CrossRef]
- Li, H.; Ye, S.; Li, L.; Zhong, J.; Yan, Q.; Zhong, P.; Feng, P.; Hu, K. 18F- or 177Lu-Labeled Bivalent Ligand of Fibroblast Activation Protein with High Tumor Uptake and Retention. Eur. J. Nucl. Med. Mol. Imaging 2022, 49, 2705–2715. [Google Scholar] [CrossRef] [PubMed]
- Wen, X.; Xu, P.; Shi, M.; Liu, J.; Zeng, X.; Zhang, Y.; Shi, C.; Li, J.; Guo, Z.; Zhang, X.; et al. Evans Blue-Modified Radiolabeled Fibroblast Activation Protein Inhibitor as Long-Acting Cancer Therapeutics. Theranostics 2022, 12, 422–433. [Google Scholar] [CrossRef] [PubMed]
- Xu, M.; Zhang, P.; Ding, J.; Chen, J.; Huo, L.; Liu, Z. Albumin Binder-Conjugated Fibroblast Activation Protein Inhibitor Radiopharmaceuticals for Cancer Therapy. J. Nucl. Med. 2022, 63, 952–958. [Google Scholar] [CrossRef] [PubMed]
- Meng, L.; Fang, J.; Zhao, L.; Wang, T.; Yuan, P.; Zhao, Z.; Zhuang, R.; Lin, Q.; Chen, H.; Chen, X.; et al. Rational Design and Pharmacomodulation of Protein-Binding Theranostic Radioligands for Targeting the Fibroblast Activation Protein. J. Med. Chem. 2022, 65, 8245–8257. [Google Scholar] [CrossRef] [PubMed]
- Sano, H.; Feighner, S.D.; Hreniuk, D.L.; Iwaasa, H.; Sailer, A.W.; Pan, J.; Reitman, M.L.; Kanatani, A.; Howard, A.D.; Tan, C.P. Characterization of the Bombesin-like Peptide Receptor Family in Primates. Genomics 2004, 84, 139–146. [Google Scholar] [CrossRef] [PubMed]
- Gonzalez, N.; Moody, T.W.; Igarashi, H.; Ito, T.; Jensen, R.T. Bombesin-Related Peptides and Their Receptors: Recent Advances in Their Role in Physiology and Disease States. Curr. Opin. Endocrinol. Diabetes Obes. 2008, 15, 58–64. [Google Scholar] [CrossRef]
- Cornelio, D.B.; Roesler, R.; Schwartsmann, G. Gastrin-Releasing Peptide Receptor as a Molecular Target in Experimental Anticancer Therapy. Ann. Oncol. 2007, 18, 1457–1466. [Google Scholar] [CrossRef] [PubMed]
- Mansi, R.; Nock, B.A.; Dalm, S.U.; Busstra, M.B.; van Weerden, W.M.; Maina, T. Radiolabeled Bombesin Analogs. Cancers 2021, 13, 5766. [Google Scholar] [CrossRef]
- Kurth, J.; Krause, B.J.; Schwarzenböck, S.M.; Bergner, C.; Hakenberg, O.W.; Heuschkel, M. First-in-Human Dosimetry of Gastrin-Releasing Peptide Receptor Antagonist [177Lu]Lu-RM2: A Radiopharmaceutical for the Treatment of Metastatic Castration-Resistant Prostate Cancer. Eur. J. Nucl. Med. Mol. Imaging 2020, 47, 123–135. [Google Scholar] [CrossRef]
- Stoykow, C.; Erbes, T.; Maecke, H.R.; Bulla, S.; Bartholomä, M.; Mayer, S.; Drendel, V.; Bronsert, P.; Werner, M.; Gitsch, G.; et al. Gastrin-Releasing Peptide Receptor Imaging in Breast Cancer Using the Receptor Antagonist 68Ga-RM2 And PET. Theranostics 2016, 6, 1641–1650. [Google Scholar] [CrossRef]
- Kähkönen, E.; Jambor, I.; Kemppainen, J.; Lehtiö, K.; Grönroos, T.J.; Kuisma, A.; Luoto, P.; Sipilä, H.J.; Tolvanen, T.; Alanen, K.; et al. In Vivo Imaging of Prostate Cancer Using [68Ga]-Labeled Bombesin Analog BAY86-7548. Clin. Cancer Res. 2013, 19, 5434–5443. [Google Scholar] [CrossRef] [PubMed]
- Djaileb, L.; Morgat, C.; van der Veldt, A.; Virgolini, I.; Cortes, F.; Demange, A.; Orlandi, F.; Wegener, A. Preliminary Diagnostic Performance of [68Ga]-NeoBOMB1 in Patients with Gastrin-Releasing Peptide Receptor-Positive Breast, Prostate, Colorectal or Lung Tumors (NeoFIND). J. Nucl. Med. 2020, 61, 346. [Google Scholar]
- Gruber, L.; Decristoforo, C.; Uprimny, C.; Hohenberger, P.; Schoenberg, S.O.; Orlandi, F.; Mariani, M.F.; Manzl, C.; Kasseroler, M.T.; Tilg, H.; et al. Imaging Properties and Tumor Targeting of 68Ga-NeoBOMB1, a Gastrin-Releasing Peptide Receptor Antagonist, in GIST Patients. Biomedicines 2022, 10, 2899. [Google Scholar] [CrossRef] [PubMed]
- Baratto, L.; Song, H.; Duan, H.; Hatami, N.; Bagshaw, H.P.; Buyyounouski, M.; Hancock, S.; Shah, S.; Srinivas, S.; Swift, P.; et al. PSMA- and GRPR-Targeted PET: Results from 50 Patients with Biochemically Recurrent Prostate Cancer. J. Nucl. Med. 2021, 62, 1545–1549. [Google Scholar] [CrossRef] [PubMed]
- Roivainen, A.; Kähkönen, E.; Luoto, P.; Borkowski, S.; Hofmann, B.; Jambor, I.; Lehtiö, K.; Rantala, T.; Rottmann, A.; Sipilä, H.; et al. Plasma Pharmacokinetics, Whole-Body Distribution, Metabolism, and Radiation Dosimetry of 68Ga Bombesin Antagonist BAY 86-7548 in Healthy Men. J. Nucl. Med. 2013, 54, 867–872. [Google Scholar] [CrossRef] [PubMed]
- Dalm, S.U.; Bakker, I.L.; de Blois, E.; Doeswijk, G.N.; Konijnenberg, M.W.; Orlandi, F.; Barbato, D.; Tedesco, M.; Maina, T.; Nock, B.A.; et al. 68Ga/177Lu-NeoBOMB1, a Novel Radiolabeled GRPR Antagonist for Theranostic Use in Oncology. J. Nucl. Med. 2017, 58, 293–299. [Google Scholar] [CrossRef]
- Montemagno, C.; Raes, F.; Ahmadi, M.; Bacot, S.; Debiossat, M.; Leenhardt, J.; Boutonnat, J.; Orlandi, F.; Barbato, D.; Tedesco, M.; et al. In Vivo Biodistribution and Efficacy Evaluation of NeoB, a Radiotracer Targeted to GRPR, in Mice Bearing Gastrointestinal Stromal Tumor. Cancers 2021, 13, 1051. [Google Scholar] [CrossRef]
- Ruigrok, E.A.M.; Verhoeven, M.; Konijnenberg, M.W.; de Blois, E.; de Ridder, C.M.A.; Stuurman, D.C.; Bertarione, L.; Rolfo, K.; de Jong, M.; Dalm, S.U. Safety of [177Lu]Lu-NeoB Treatment: A Preclinical Study Characterizing Absorbed Dose and Acute, Early, and Late Organ Toxicity. Eur. J. Nucl. Med. Mol. Imaging 2022, 49, 4440–4451. [Google Scholar] [CrossRef]
- Advanced Accelerator Applications. A Phase I/IIa Open-Label, Multi-Center Study to Evaluate the Safety, Tolerability, Whole-Body Distribution, Radiation Dosimetry and Anti-Tumor Activity of [177Lu]-NeoB Administered in Patients with Advanced Solid Tumors Known to Overexpress Gastrin-Releasing Peptide Receptor (GRPR). 2023. Available online: https://www.novartis.com/clinicaltrials/study/nct03872778 (accessed on 20 August 2023).
- Yan, M.; Schwaederle, M.; Arguello, D.; Millis, S.Z.; Gatalica, Z.; Kurzrock, R. HER2 Expression Status in Diverse Cancers: Review of Results from 37,992 Patients. Cancer Metastasis Rev. 2015, 34, 157–164. [Google Scholar] [CrossRef]
- Greenblatt, K.; Khaddour, K. Trastuzumab. In StatPearls; StatPearls Publishing: Treasure Island, FL, USA, 2023. [Google Scholar]
- Ramezani, M.; Siami, S.; Rezaei, M.; Khazaei, S.; Sadeghi, M. An Immunohistochemical Study of HER2 Expression in Primary Brain Tumors. Biomedicine 2020, 10, 21–27. [Google Scholar] [CrossRef]
- Indini, A.; Rijavec, E.; Grossi, F. Trastuzumab Deruxtecan: Changing the Destiny of HER2 Expressing Solid Tumors. Int. J. Mol. Sci. 2021, 22, 4774. [Google Scholar] [CrossRef] [PubMed]
- Keyaerts, M.; Xavier, C.; Heemskerk, J.; Devoogdt, N.; Everaert, H.; Ackaert, C.; Vanhoeij, M.; Duhoux, F.P.; Gevaert, T.; Simon, P.; et al. Phase I Study of 68Ga-HER2-Nanobody for PET/CT Assessment of HER2 Expression in Breast Carcinoma. J. Nucl. Med. 2016, 57, 27–33. [Google Scholar] [CrossRef] [PubMed]
- Miao, H.; Sun, Y.; Jin, Y.; Hu, X.; Song, S.; Zhang, J. Application of a Novel 68Ga-HER2 Affibody PET/CT Imaging in Breast Cancer Patients. Front. Oncol. 2022, 12, 894767. [Google Scholar] [CrossRef] [PubMed]
- Altunay, B.; Morgenroth, A.; Beheshti, M.; Vogg, A.; Wong, N.C.L.; Ting, H.H.; Biersack, H.-J.; Stickeler, E.; Mottaghy, F.M. HER2-Directed Antibodies, Affibodies and Nanobodies as Drug-Delivery Vehicles in Breast Cancer with a Specific Focus on Radioimmunotherapy and Radioimmunoimaging. Eur. J. Nucl. Med. Mol. Imaging 2021, 48, 1371–1389. [Google Scholar] [CrossRef] [PubMed]
- Puttemans, J.; Dekempeneer, Y.; Eersels, J.L.; Hanssens, H.; Debie, P.; Keyaerts, M.; Windhorst, A.D.; van der Aa, F.; Lecocq, Q.; Breckpot, K.; et al. Preclinical Targeted α- and Β-Radionuclide Therapy in HER2-Positive Brain Metastasis Using Camelid Single-Domain Antibodies. Cancers 2020, 12, 1017. [Google Scholar] [CrossRef]
- Rodak, M.; Dekempeneer, Y.; Wojewódzka, M.; Caveliers, V.; Covens, P.; Miller, B.W.; Sevenois, M.B.; Bruchertseifer, F.; Morgenstern, A.; Lahoutte, T.; et al. Preclinical Evaluation of 225Ac-Labeled Single-Domain Antibody for the Treatment of HER2pos Cancer. Mol. Cancer Ther. 2022, 21, 1835–1845. [Google Scholar] [CrossRef] [PubMed]
- Feng, Y.; Meshaw, R.; McDougald, D.; Zhou, Z.; Zhao, X.-G.; Jannetti, S.A.; Reiman, R.E.; Pippen, E.; Marjoram, R.; Schaal, J.L.; et al. Evaluation of an 131I-Labeled HER2-Specific Single Domain Antibody Fragment for the Radiopharmaceutical Therapy of HER2-Expressing Cancers. Sci. Rep. 2022, 12, 3020. [Google Scholar] [CrossRef]
- Feng, Y.; Meshaw, R.; Zhao, X.-G.; Jannetti, S.; Vaidyanathan, G.; Zalutsky, M.R. Effective Treatment of Human Breast Carcinoma Xenografts with Single-Dose 211At-Labeled Anti-HER2 Single-Domain Antibody Fragment. J. Nucl. Med. 2023, 64, 124–130. [Google Scholar] [CrossRef]
- Zhao, L.; Gong, J.; Qi, Q.; Liu, C.; Su, H.; Xing, Y.; Zhao, J. 131I-Labeled Anti-HER2 Nanobody for Targeted Radionuclide Therapy of HER2-Positive Breast Cancer. Int. J. Nanomed. 2023, 18, 1915–1925. [Google Scholar] [CrossRef]
- Liu, Y.; Xu, T.; Vorobyeva, A.; Loftenius, A.; Bodenko, V.; Orlova, A.; Frejd, F.Y.; Tolmachev, V. Radionuclide Therapy of HER2-Expressing Xenografts Using [177Lu]Lu-ABY-027 Affibody Molecule Alone and in Combination with Trastuzumab. Cancers 2023, 15, 2409. [Google Scholar] [CrossRef]
- Abdulfatah, E.; Reichert, Z.R.; Davenport, M.S.; Chinnaiyan, A.M.; Dadhania, V.; Wang, X.; Mannan, R.; Kunju, L.P.; Hollenbeck, B.K.; Montgomery, J.S.; et al. De Novo Neuroendocrine Transdifferentiation in Primary Prostate Cancer-a Phenotype Associated with Advanced Clinico-Pathologic Features and Aggressive Outcome. Med. Oncol. 2021, 38, 26. [Google Scholar] [CrossRef] [PubMed]
- Merkens, L.; Sailer, V.; Lessel, D.; Janzen, E.; Greimeier, S.; Kirfel, J.; Perner, S.; Pantel, K.; Werner, S.; von Amsberg, G. Aggressive Variants of Prostate Cancer: Underlying Mechanisms of Neuroendocrine Transdifferentiation. J. Exp. Clin. Cancer Res. 2022, 41, 46. [Google Scholar] [CrossRef] [PubMed]
- Aggarwal, R.; Huang, J.; Alumkal, J.J.; Zhang, L.; Feng, F.Y.; Thomas, G.V.; Weinstein, A.S.; Friedl, V.; Zhang, C.; Witte, O.N.; et al. Clinical and Genomic Characterization of Treatment-Emergent Small-Cell Neuroendocrine Prostate Cancer: A Multi-Institutional Prospective Study. J. Clin. Oncol. 2018, 36, 2492–2503. [Google Scholar] [CrossRef] [PubMed]
- Su, Y.; Liu, Y.; Behrens, C.R.; Bidlingmaier, S.; Lee, N.-K.; Aggarwal, R.; Sherbenou, D.W.; Burlingame, A.L.; Hann, B.C.; Simko, J.P.; et al. Targeting CD46 for Both Adenocarcinoma and Neuroendocrine Prostate Cancer. JCI Insight 2018, 3, e121497. [Google Scholar] [CrossRef]
- Elvington, M.; Liszewski, M.K.; Atkinson, J.P. CD46 and Oncologic Interactions: Friendly Fire against Cancer. Antibodies 2020, 9, 59. [Google Scholar] [CrossRef] [PubMed]
- Do, M.-H.; Thanh, H.D.; To, P.K.; Kim, M.S.; Moon, C.; Jung, C. CD46 Protects the Bladder Cancer Cells from Cetuximab-Mediated Cytotoxicity. Sci. Rep. 2022, 12, 22420. [Google Scholar] [CrossRef] [PubMed]
- Wang, S.; Li, J.; Hua, J.; Su, Y.; Beckford-Vera, D.R.; Zhao, W.; Jayaraman, M.; Huynh, T.L.; Zhao, N.; Wang, Y.; et al. Molecular Imaging of Prostate Cancer Targeting CD46 Using ImmunoPET. Clin. Cancer Res. 2021, 27, 1305–1315. [Google Scholar] [CrossRef] [PubMed]
- Li, J.; Huang, T.; Hua, J.; Wang, Q.; Su, Y.; Chen, P.; Bidlingmaier, S.; Li, A.; Xie, Z.; Bidkar, A.P.; et al. CD46 Targeted 212Pb Alpha Particle Radioimmunotherapy for Prostate Cancer Treatment. J. Exp. Clin. Cancer Res. 2023, 42, 61. [Google Scholar] [CrossRef]
- Saunders, L.R.; Bankovich, A.J.; Anderson, W.C.; Aujay, M.A.; Bheddah, S.; Black, K.; Desai, R.; Escarpe, P.A.; Hampl, J.; Laysang, A.; et al. A DLL3-Targeted Antibody-Drug Conjugate Eradicates High-Grade Pulmonary Neuroendocrine Tumor-Initiating Cells in Vivo. Sci. Transl. Med. 2015, 7, 302ra136. [Google Scholar] [CrossRef]
- Puca, L.; Gavyert, K.; Sailer, V.; Conteduca, V.; Dardenne, E.; Sigouros, M.; Isse, K.; Kearney, M.; Vosoughi, A.; Fernandez, L.; et al. Delta-like Protein 3 Expression and Therapeutic Targeting in Neuroendocrine Prostate Cancer. Sci. Transl. Med. 2019, 11, eaav0891. [Google Scholar] [CrossRef]
- Cortinovis, D.L.; Colonese, F.; Abbate, M.I.; Sala, L.; Meazza Prina, M.; Cordani, N.; Sala, E.; Canova, S. Harnessing DLL3 Inhibition: From Old Promises to New Therapeutic Horizons. Front. Med. 2022, 9, 989405. [Google Scholar] [CrossRef] [PubMed]
- Sharma, S.K.; Pourat, J.; Abdel-Atti, D.; Carlin, S.D.; Piersigilli, A.; Bankovich, A.J.; Gardner, E.E.; Hamdy, O.; Isse, K.; Bheddah, S.; et al. Non-Invasive Interrogation of DLL3 Expression in Metastatic Small Cell Lung Cancer. Cancer Res. 2017, 77, 3931–3941. [Google Scholar] [CrossRef] [PubMed]
- Korsen, J.A.; Kalidindi, T.M.; Khitrov, S.; Samuels, Z.V.; Chakraborty, G.; Gutierrez, J.A.; Poirier, J.T.; Rudin, C.M.; Chen, Y.; Morris, M.J.; et al. Molecular Imaging of Neuroendocrine Prostate Cancer by Targeting Delta-Like Ligand 3. J. Nucl. Med. 2022, 63, 1401–1407. [Google Scholar] [CrossRef] [PubMed]
- Korsen, J.A.; Gutierrez, J.A.; Tully, K.M.; Carter, L.M.; Samuels, Z.V.; Khitrov, S.; Poirier, J.T.; Rudin, C.M.; Chen, Y.; Morris, M.J.; et al. Delta-like Ligand 3-Targeted Radioimmunotherapy for Neuroendocrine Prostate Cancer. Proc. Natl. Acad. Sci. USA 2022, 119, e2203820119. [Google Scholar] [CrossRef] [PubMed]
- Tully, K.M.; Tendler, S.; Carter, L.M.; Sharma, S.K.; Samuels, Z.V.; Mandleywala, K.; Korsen, J.A.; Delos Reyes, A.M.; Piersigilli, A.; Travis, W.D.; et al. Radioimmunotherapy Targeting Delta-like Ligand 3 in Small Cell Lung Cancer Exhibits Antitumor Efficacy with Low Toxicity. Clin. Cancer Res. 2022, 28, 1391–1401. [Google Scholar] [CrossRef] [PubMed]
- Charkhchi, P.; Cybulski, C.; Gronwald, J.; Wong, F.O.; Narod, S.A.; Akbari, M.R. CA125 and Ovarian Cancer: A Comprehensive Review. Cancers 2020, 12, 3730. [Google Scholar] [CrossRef] [PubMed]
- Fung, K.; Sharma, S.K.; Keinänen, O.; Roche, K.L.; Lewis, J.S.; Zeglis, B.M. A Molecularly Targeted Intraoperative Near-Infrared Fluorescence Imaging Agent for High-Grade Serous Ovarian Cancer. Mol. Pharm. 2020, 17, 3140–3147. [Google Scholar] [CrossRef] [PubMed]
- Chen, Y.; Clark, S.; Wong, T.; Chen, Y.; Chen, Y.; Dennis, M.S.; Luis, E.; Zhong, F.; Bheddah, S.; Koeppen, H.; et al. Armed Antibodies Targeting the Mucin Repeats of the Ovarian Cancer Antigen, MUC16, Are Highly Efficacious in Animal Tumor Models. Cancer Res. 2007, 67, 4924–4932. [Google Scholar] [CrossRef]
- Rao, T.D.; Fernández-Tejada, A.; Axelrod, A.; Rosales, N.; Yan, X.; Thapi, S.; Wang, A.; Park, K.J.; Nemieboka, B.; Xiang, J.; et al. Antibodies Against Specific MUC16 Glycosylation Sites Inhibit Ovarian Cancer Growth. ACS Chem. Biol. 2017, 12, 2085–2096. [Google Scholar] [CrossRef]
- Thomas, D.; Sagar, S.; Liu, X.; Lee, H.-R.; Grunkemeyer, J.A.; Grandgenett, P.M.; Caffrey, T.; O’Connell, K.A.; Swanson, B.; Marcos-Silva, L.; et al. Isoforms of MUC16 Activate Oncogenic Signaling through EGF Receptors to Enhance the Progression of Pancreatic Cancer. Mol. Ther. 2021, 29, 1557–1571. [Google Scholar] [CrossRef]
- Sharma, S.K.; Mack, K.N.; Piersigilli, A.; Pourat, J.; Edwards, K.J.; Keinänen, O.; Jiao, M.S.; Zhao, H.; White, B.; Brooks, C.L.; et al. ImmunoPET of Ovarian and Pancreatic Cancer with AR9.6, a Novel MUC16-Targeted Therapeutic Antibody. Clin. Cancer Res. 2022, 28, 948–959. [Google Scholar] [CrossRef] [PubMed]
- Manne, A.; Kasi, A.; Esnakula, A.K.; Paluri, R.K. Predictive Value of MUC5AC Signature in Pancreatic Ductal Adenocarcinoma: A Hypothesis Based on Preclinical Evidence. Int. J. Mol. Sci. 2023, 24, 8087. [Google Scholar] [CrossRef] [PubMed]
- Henry, K.E.; Shaffer, T.M.; Mack, K.N.; Ring, J.; Ogirala, A.; Klein-Scory, S.; Eilert-Micus, C.; Schmiegel, W.; Bracht, T.; Sitek, B.; et al. Exploiting the MUC5AC Antigen for Noninvasive Identification of Pancreatic Cancer. J. Nucl. Med. 2021, 62, 1384–1390. [Google Scholar] [CrossRef] [PubMed]
- Nakata, N.; Kobashi, N.; Okumura, Y.; Sato, M.; Matono, M.; Otsuki, K.; Tanaka, A.; Hayashi, A. Radiation Dosimetry and Efficacy of an 89Zr/225Ac-Labeled Humanized Anti-MUC5AC Antibody. Nucl. Med. Biol. 2022, 108–109, 33–43. [Google Scholar] [CrossRef] [PubMed]
- Miyazawa, Y.; Uekita, T.; Hiraoka, N.; Fujii, S.; Kosuge, T.; Kanai, Y.; Nojima, Y.; Sakai, R. CUB Domain-Containing Protein 1, a Prognostic Factor for Human Pancreatic Cancers, Promotes Cell Migration and Extracellular Matrix Degradation. Cancer Res. 2010, 70, 5136–5146. [Google Scholar] [CrossRef] [PubMed]
- Kryza, T.; Khan, T.; Puttick, S.; Li, C.; Sokolowski, K.A.; Tse, B.W.; Cuda, T.; Lyons, N.; Gough, M.; Yin, J.; et al. Effective Targeting of Intact and Proteolysed CDCP1 for Imaging and Treatment of Pancreatic Ductal Adenocarcinoma. Theranostics 2020, 10, 4116–4133. [Google Scholar] [CrossRef] [PubMed]
- Harrington, B.S.; He, Y.; Khan, T.; Puttick, S.; Conroy, P.J.; Kryza, T.; Cuda, T.; Sokolowski, K.A.; Tse, B.W.; Robbins, K.K.; et al. Anti-CDCP1 Immuno-Conjugates for Detection and Inhibition of Ovarian Cancer. Theranostics 2020, 10, 2095–2114. [Google Scholar] [CrossRef] [PubMed]
- Moroz, A.; Wang, Y.-H.; Sharib, J.M.; Wei, J.; Zhao, N.; Huang, Y.; Chen, Z.; Martinko, A.J.; Zhuo, J.; Lim, S.A.; et al. Theranostic Targeting of CUB Domain Containing Protein 1 (CDCP1) in Pancreatic Cancer. Clin. Cancer Res. 2020, 26, 3608–3615. [Google Scholar] [CrossRef]
- Zhao, N.; Chopra, S.; Trepka, K.; Wang, Y.-H.; Sakhamuri, S.; Hooshdaran, N.; Kim, H.; Zhou, J.; Lim, S.A.; Leung, K.K.; et al. CUB Domain-Containing Protein 1 (CDCP1) Is a Target for Radioligand Therapy in Castration-Resistant Prostate Cancer, Including PSMA Null Disease. Clin. Cancer Res. 2022, 28, 3066–3075. [Google Scholar] [CrossRef]
- Chopra, S.; Trepka, K.; Sakhamuri, S.; Carretero-González, A.; Zhu, J.; Egusa, E.; Zhou, J.; Leung, K.; Zhao, N.; Hooshdaran, N.; et al. Theranostic Targeting of CUB Domain-Containing Protein 1 (CDCP1) in Multiple Subtypes of Bladder Cancer. Clin. Cancer Res. 2023, 29, 1232–1242. [Google Scholar] [CrossRef]
- Wang, P.; Wang, S.; Liu, F.; Ren, Y.; Guo, Q.; Zhang, Q.; Hou, X.; Yao, Y.; Zhu, H.; Yang, Z. Preclinical Evaluation of a Fibroblast Activation Protein and a Prostate-Specific Membrane Antigen Dual-Targeted Probe for Noninvasive Prostate Cancer Imaging. Mol. Pharm. 2023, 20, 1415–1425. [Google Scholar] [CrossRef] [PubMed]
- Verena, A.; Zhang, Z.; Kuo, H.-T.; Merkens, H.; Zeisler, J.; Wilson, R.; Bendre, S.; Wong, A.A.W.L.; Bénard, F.; Lin, K.-S. Synthesis and Preclinical Evaluation of Three Novel 68Ga-Labeled Bispecific PSMA/FAP-Targeting Tracers for Prostate Cancer Imaging. Molecules 2023, 28, 1088. [Google Scholar] [CrossRef] [PubMed]
- Hu, K.; Li, L.; Huang, Y.; Ye, S.; Zhong, J.; Yan, Q.; Zhong, Y.; Fu, L.; Feng, P.; Li, H. Radiosynthesis and Preclinical Evaluation of Bispecific PSMA/FAP Heterodimers for Tumor Imaging. Pharmaceuticals 2022, 15, 383. [Google Scholar] [CrossRef] [PubMed]
- Boinapally, S.; Lisok, A.; Lofland, G.; Minn, I.; Yan, Y.; Jiang, Z.; Shin, M.J.; Merino, V.F.; Zheng, L.; Brayton, C.; et al. Hetero-Bivalent Agents Targeting FAP and PSMA. Eur. J. Nucl. Med. Mol. Imaging 2022, 49, 4369–4381. [Google Scholar] [CrossRef] [PubMed]
- Lundmark, F.; Abouzayed, A.; Mitran, B.; Rinne, S.S.; Varasteh, Z.; Larhed, M.; Tolmachev, V.; Rosenström, U.; Orlova, A. Heterodimeric Radiotracer Targeting PSMA and GRPR for Imaging of Prostate Cancer-Optimization of the Affinity towards PSMA by Linker Modification in Murine Model. Pharmaceutics 2020, 12, 614. [Google Scholar] [CrossRef] [PubMed]
- Lundmark, F.; Abouzayed, A.; Rinne, S.S.; Timofeev, V.; Sipkina, N.; Naan, M.; Kirichenko, A.; Vasyutina, M.; Ryzhkova, D.; Tolmachev, V.; et al. Preclinical Characterisation of PSMA/GRPR-Targeting Heterodimer [68Ga]Ga-BQ7812 for PET Diagnostic Imaging of Prostate Cancer: A Step towards Clinical Translation. Cancers 2023, 15, 442. [Google Scholar] [CrossRef] [PubMed]
- Danen, E.H.J. Integrin Signaling as a Cancer Drug Target. ISRN Cell Biol. 2013, 2013, e135164. [Google Scholar] [CrossRef]
- Ellert-Miklaszewska, A.; Poleszak, K.; Pasierbinska, M.; Kaminska, B. Integrin Signaling in Glioma Pathogenesis: From Biology to Therapy. Int. J. Mol. Sci. 2020, 21, 888. [Google Scholar] [CrossRef]
- Niu, G.; Chen, X. Why Integrin as a Primary Target for Imaging and Therapy. Theranostics 2011, 1, 30–47. [Google Scholar] [CrossRef]
- Zang, J.; Wen, X.; Lin, R.; Zeng, X.; Wang, C.; Shi, M.; Zeng, X.; Zhang, J.; Wu, X.; Zhang, X.; et al. Synthesis, Preclinical Evaluation and Radiation Dosimetry of a Dual Targeting PET Tracer [68Ga]Ga-FAPI-RGD. Theranostics 2022, 12, 7180–7190. [Google Scholar] [CrossRef]
- Zhao, L.; Wen, X.; Xu, W.; Pang, Y.; Sun, L.; Wu, X.; Xu, P.; Zhang, J.; Guo, Z.; Lin, Q.; et al. Clinical Evaluation of 68Ga-FAPI-RGD for Imaging of Fibroblast Activation Protein and Integrin Avβ3 in Various Cancer Types. J. Nucl. Med. 2023, 64, 1210–1217. [Google Scholar] [CrossRef] [PubMed]
- Wang, R.; Jakobsson, V.; Wang, J.; Zhao, T.; Peng, X.; Li, B.; Xue, J.; Liang, N.; Zhu, Z.; Chen, X.; et al. Dual Targeting PET Tracer [68Ga]Ga-FAPI-RGD in Patients with Lung Neoplasms: A Pilot Exploratory Study. Theranostics 2023, 13, 2979–2992. [Google Scholar] [CrossRef] [PubMed]
- Ayoub, N.M. Editorial: Novel Combination Therapies for the Treatment of Solid Cancers. Front. Oncol. 2021, 11, 708943. [Google Scholar] [CrossRef]
- Rajeev-Kumar, G.; Pitroda, S.P. Synergizing Radiotherapy and Immunotherapy: Current Challenges and Strategies for Optimization. Neoplasia 2023, 36, 100867. [Google Scholar] [CrossRef] [PubMed]
- Aicher, A.; Sindrilaru, A.; Crisan, D.; Thaiss, W.; Steinacker, J.; Beer, M.; Wiegel, T.; Scharffetter-Kochanek, K.; Beer, A.J.; Prasad, V. Short-Interval, Low-Dose Peptide Receptor Radionuclide Therapy in Combination with PD-1 Checkpoint Immunotherapy Induces Remission in Immunocompromised Patients with Metastatic Merkel Cell Carcinoma. Pharmaceutics 2022, 14, 1466. [Google Scholar] [CrossRef] [PubMed]
- Esfahani, S.A.; De Aguiar Ferreira, C.; Summer, P.; Mahmood, U.; Heidari, P. Addition of Peptide Receptor Radiotherapy to Immune Checkpoint Inhibition Therapy Improves Outcomes in Neuroendocrine Tumors. J. Nucl. Med. 2023, 64, 1056–1061. [Google Scholar] [CrossRef] [PubMed]
- Kim, C.; Liu, S.V.; Subramaniam, D.S.; Torres, T.; Loda, M.; Esposito, G.; Giaccone, G. Phase I Study of the 177Lu-DOTA0-Tyr3-Octreotate (Lutathera) in Combination with Nivolumab in Patients with Neuroendocrine Tumors of the Lung. J. Immunother. Cancer 2020, 8, e000980. [Google Scholar] [CrossRef] [PubMed]
- Aggarwal, R.R.; Luch Sam, S.; Koshkin, V.S.; Small, E.J.; Feng, F.Y.; de Kouchkovsky, I.; Kwon, D.H.; Friedlander, T.W.; Borno, H.; Bose, R.; et al. Immunogenic Priming with 177Lu-PSMA-617 plus Pembrolizumab in Metastatic Castration Resistant Prostate Cancer (MCRPC): A Phase 1b Study. JCO 2021, 39, 5053. [Google Scholar] [CrossRef]
- Czernin, J.; Current, K.; Mona, C.E.; Nyiranshuti, L.; Hikmat, F.; Radu, C.G.; Lückerath, K. Immune-Checkpoint Blockade Enhances 225Ac-PSMA617 Efficacy in a Mouse Model of Prostate Cancer. J. Nucl. Med. 2021, 62, 228–231. [Google Scholar] [CrossRef]
- Weidemann, S.; Gagelmann, P.; Gorbokon, N.; Lennartz, M.; Menz, A.; Luebke, A.M.; Kluth, M.; Hube-Magg, C.; Blessin, N.C.; Fraune, C.; et al. Mesothelin Expression in Human Tumors: A Tissue Microarray Study on 12,679 Tumors. Biomedicines 2021, 9, 397. [Google Scholar] [CrossRef]
- Montemagno, C.; Cassim, S.; Pouyssegur, J.; Broisat, A.; Pagès, G. From Malignant Progression to Therapeutic Targeting: Current Insights of Mesothelin in Pancreatic Ductal Adenocarcinoma. Int. J. Mol. Sci. 2020, 21, E4067. [Google Scholar] [CrossRef] [PubMed]
- Faust, J.R.; Hamill, D.; Kolb, E.A.; Gopalakrishnapillai, A.; Barwe, S.P. Mesothelin: An Immunotherapeutic Target beyond Solid Tumors. Cancers 2022, 14, 1550. [Google Scholar] [CrossRef] [PubMed]
- Hagemann, U.B.; Ellingsen, C.; Schuhmacher, J.; Kristian, A.; Mobergslien, A.; Cruciani, V.; Wickstroem, K.; Schatz, C.A.; Kneip, C.; Golfier, S.; et al. Mesothelin-Targeted Thorium-227 Conjugate (MSLN-TTC): Preclinical Evaluation of a New Targeted Alpha Therapy for Mesothelin-Positive Cancers. Clin. Cancer Res. 2019, 25, 4723–4734. [Google Scholar] [CrossRef]
- Lejeune, P.; Cruciani, V.; Berg-Larsen, A.; Schlicker, A.; Mobergslien, A.; Bartnitzky, L.; Berndt, S.; Zitzmann-Kolbe, S.; Kamfenkel, C.; Stargard, S.; et al. Immunostimulatory Effects of Targeted Thorium-227 Conjugates as Single Agent and in Combination with Anti-PD-L1 Therapy. J. Immunother. Cancer 2021, 9, e002387. [Google Scholar] [CrossRef] [PubMed]
- Zhao, L.; Chen, H.; Guo, Z.; Fu, K.; Yao, L.; Fu, L.; Guo, W.; Wen, X.; Jacobson, O.; Zhang, X. Targeted Radionuclide Therapy in Patient-Derived Xenografts Using 177Lu-EB-RGD. Mol. Cancer Ther. 2020, 19, 2034–2043. [Google Scholar] [CrossRef] [PubMed]
- Chen, H.; Zhao, L.; Fu, K.; Lin, Q.; Wen, X.; Jacobson, O.; Sun, L.; Wu, H.; Zhang, X.; Guo, Z.; et al. Integrin Avβ3-Targeted Radionuclide Therapy Combined with Immune Checkpoint Blockade Immunotherapy Synergistically Enhances Anti-Tumor Efficacy. Theranostics 2019, 9, 7948–7960. [Google Scholar] [CrossRef] [PubMed]
- Hernandez, R.; Grudzinski, J.J.; Aluicio-Sarduy, E.; Massey, C.F.; Pinchuk, A.N.; Bitton, A.N.; Patel, R.; Zhang, R.; Rao, A.V.; Iyer, G.; et al. 177Lu-NM600 Targeted Radionuclide Therapy Extends Survival in Syngeneic Murine Models of Triple-Negative Breast Cancer. J. Nucl. Med. 2020, 61, 1187–1194. [Google Scholar] [CrossRef]
- Jagodinsky, J.C.; Jin, W.J.; Bates, A.M.; Hernandez, R.; Grudzinski, J.J.; Marsh, I.R.; Chakravarty, I.; Arthur, I.S.; Zangl, L.M.; Brown, R.J.; et al. Temporal Analysis of Type 1 Interferon Activation in Tumor Cells Following External Beam Radiotherapy or Targeted Radionuclide Therapy. Theranostics 2021, 11, 6120–6137. [Google Scholar] [CrossRef]
- Patel, R.B.; Hernandez, R.; Carlson, P.; Grudzinski, J.; Bates, A.M.; Jagodinsky, J.C.; Erbe, A.; Marsh, I.R.; Arthur, I.; Aluicio-Sarduy, E.; et al. Low-Dose Targeted Radionuclide Therapy Renders Immunologically Cold Tumors Responsive to Immune Checkpoint Blockade. Sci. Transl. Med. 2021, 13, eabb3631. [Google Scholar] [CrossRef]
- Malo, M.E.; Allen, K.J.H.; Jiao, R.; Frank, C.; Rickles, D.; Dadachova, E. Mechanistic Insights into Synergy between Melanin-Targeting Radioimmunotherapy and Immunotherapy in Experimental Melanoma. Int. J. Mol. Sci. 2020, 21, 8721. [Google Scholar] [CrossRef]
- Rouanet, J.; Benboubker, V.; Akil, H.; Hennino, A.; Auzeloux, P.; Besse, S.; Pereira, B.; Delorme, S.; Mansard, S.; D’Incan, M.; et al. Immune Checkpoint Inhibitors Reverse Tolerogenic Mechanisms Induced by Melanoma Targeted Radionuclide Therapy. Cancer Immunol. Immunother. 2020, 69, 2075–2088. [Google Scholar] [CrossRef]
- Jiao, R.; Allen, K.J.H.; Malo, M.E.; Rickles, D.; Dadachova, E. Evaluating the Combination of Radioimmunotherapy and Immunotherapy in a Melanoma Mouse Model. Int. J. Mol. Sci. 2020, 21, 773. [Google Scholar] [CrossRef]
- Guzik, P.; Siwowska, K.; Fang, H.-Y.; Cohrs, S.; Bernhardt, P.; Schibli, R.; Müller, C. Promising Potential of [177Lu]Lu-DOTA-Folate to Enhance Tumor Response to Immunotherapy-a Preclinical Study Using a Syngeneic Breast Cancer Model. Eur. J. Nucl. Med. Mol. Imaging 2021, 48, 984–994. [Google Scholar] [CrossRef]
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Echavidre, W.; Fagret, D.; Faraggi, M.; Picco, V.; Montemagno, C. Recent Pre-Clinical Advancements in Nuclear Medicine: Pioneering the Path to a Limitless Future. Cancers 2023, 15, 4839. https://doi.org/10.3390/cancers15194839
Echavidre W, Fagret D, Faraggi M, Picco V, Montemagno C. Recent Pre-Clinical Advancements in Nuclear Medicine: Pioneering the Path to a Limitless Future. Cancers. 2023; 15(19):4839. https://doi.org/10.3390/cancers15194839
Chicago/Turabian StyleEchavidre, William, Daniel Fagret, Marc Faraggi, Vincent Picco, and Christopher Montemagno. 2023. "Recent Pre-Clinical Advancements in Nuclear Medicine: Pioneering the Path to a Limitless Future" Cancers 15, no. 19: 4839. https://doi.org/10.3390/cancers15194839
APA StyleEchavidre, W., Fagret, D., Faraggi, M., Picco, V., & Montemagno, C. (2023). Recent Pre-Clinical Advancements in Nuclear Medicine: Pioneering the Path to a Limitless Future. Cancers, 15(19), 4839. https://doi.org/10.3390/cancers15194839





