T2-Weighted MRI Radiomic Features Predict Prostate Cancer Presence and Eventual Biochemical Recurrence
Abstract
:Simple Summary
Abstract
1. Introduction
2. Materials and Methods
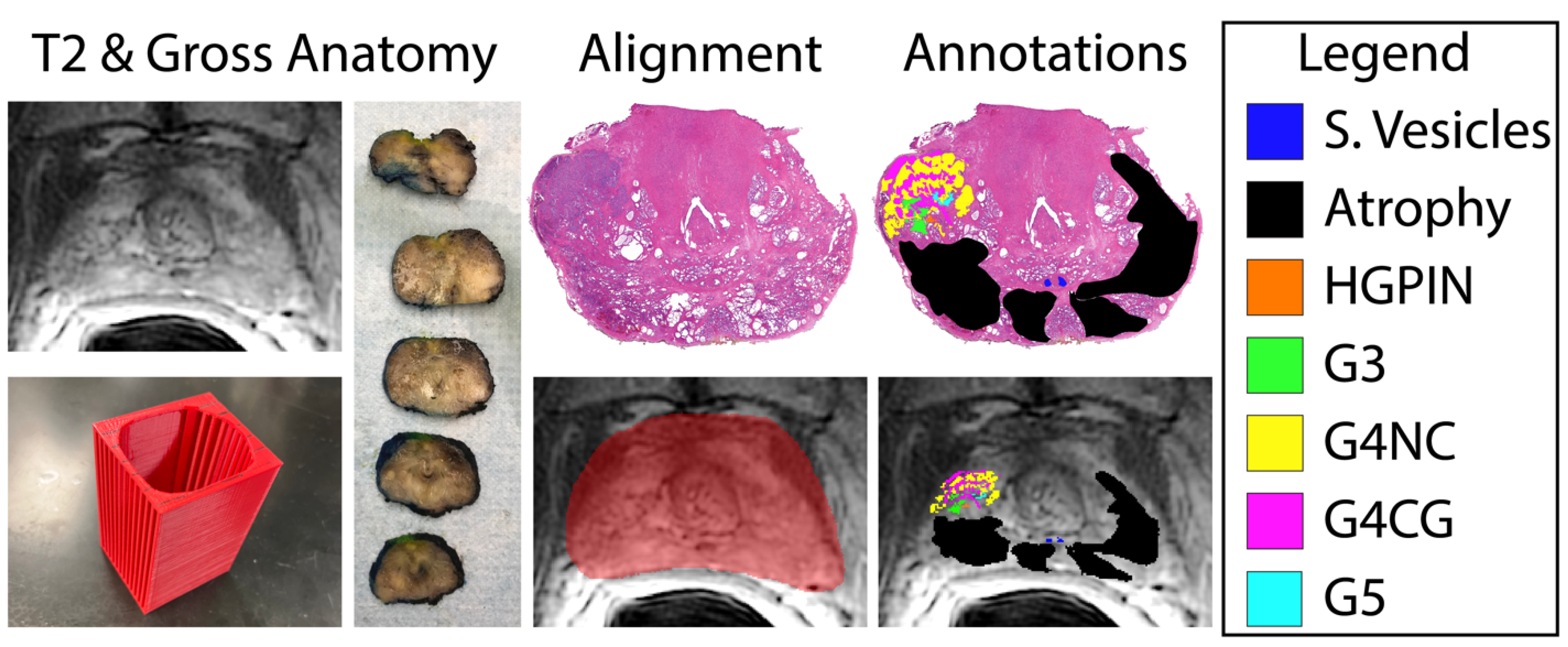
2.1. Patient Population and Data Acquisition
2.2. Histological Analysis
2.3. Histology and MRI Co-Registration
2.4. Radiomic Feature Extraction and Statistical Analyses
3. Results
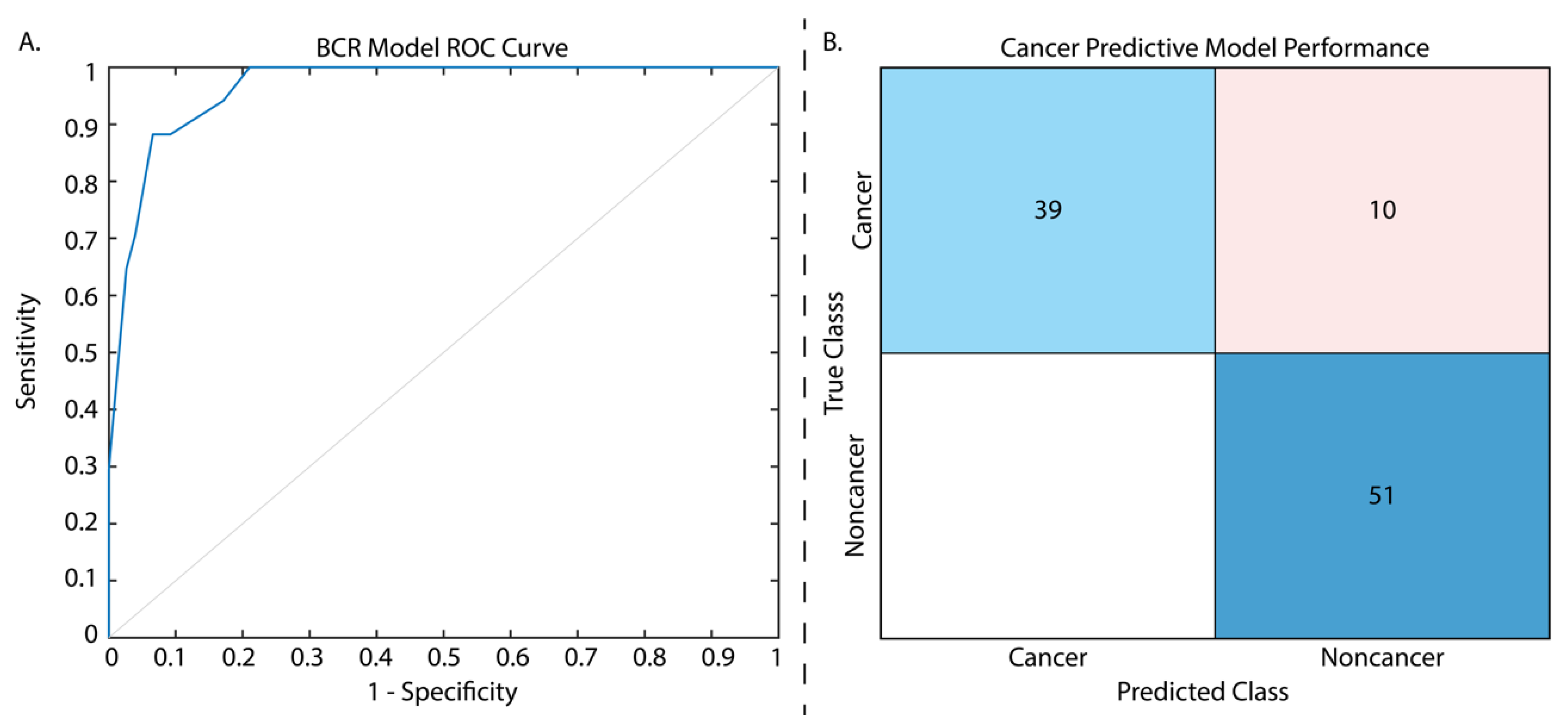
3.1. Biochemical Recurrence Regression Model
3.2. Cancer/Non-Cancer Classification Model
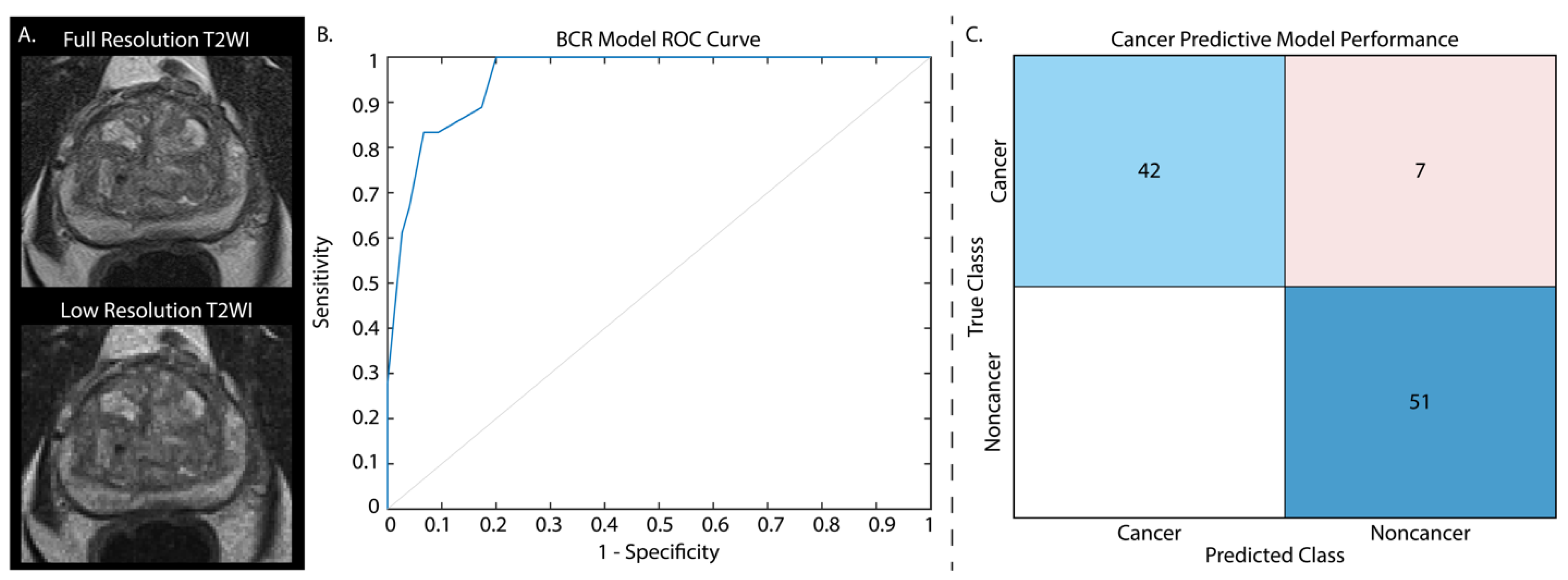
3.3. Low-Resolution Image Assessment
4. Discussion
Limitations
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Siegel, R.L.; Miller, K.D.; Wagle, N.S.; Jemal, A. Cancer statistics, 2023. CA Cancer J. Clin. 2023, 73, 17–48. [Google Scholar] [CrossRef] [PubMed]
- Amaro, A.; Esposito, A.I.; Gallina, A.; Nees, M.; Angelini, G.; Albini, A.; Pfeffer, U. Validation of proposed prostate cancer biomarkers with gene expression data: A long road to travel. Cancer Metastasis Rev. 2014, 33, 657–671. [Google Scholar] [CrossRef]
- Mir, M.C.; Li, J.; Klink, J.C.; Kattan, M.W.; Klein, E.A.; Stephenson, A.J. Optimal definition of biochemical recurrence after radical prostatectomy depends on pathologic risk factors: Identifying candidates for early salvage therapy. Eur. Urol. 2014, 66, 204–210. [Google Scholar] [CrossRef]
- Sokoll, L.J.; Zhang, Z.; Chan, D.W.; Reese, A.C.; Bivalacqua, T.J.; Partin, A.W.; Walsh, P.C. Do Ultrasensitive Prostate Specific Antigen Measurements Have a Role in Predicting Long-Term Biochemical Recurrence-Free Survival in Men after Radical Prostatectomy? J. Urol. 2016, 195, 330–336. [Google Scholar] [CrossRef] [PubMed]
- Hambrock, T.; Somford, D.M.; Huisman, H.J.; van Oort, I.M.; Witjes, J.A.; Hulsbergen-van de Kaa, C.A.; Scheenen, T.; Barentsz, J.O. Relationship between Apparent Diffusion Coefficients at 3.0-T MR Imaging and Gleason Grade in Peripheral Zone Prostate Cancer. Radiology 2011, 259, 11091409. [Google Scholar] [CrossRef] [PubMed]
- Vos, E.K.; Kobus, T.; Litjens, G.J.S.; Hambrock, T.; Hulsbergen-Van De Kaa, C.A.; Barentsz, J.O.; Maas, M.C.; Scheenen, T.W.J. Multiparametric Magnetic Resonance Imaging for Discriminating Low-Grade from High-Grade Prostate Cancer. Investig. Radiol. 2015, 50, 490–497. [Google Scholar] [CrossRef] [PubMed]
- Wichtmann, B.D.; Zöllner, F.G.; Attenberger, U.I.; Schönberg, S.O. Multiparametric MRI in the Diagnosis of Prostate Cancer: Physical Foundations, Limitations, and Prospective Advances of Diffusion-Weighted MRI. Rofo 2021, 193, 399–409. [Google Scholar] [CrossRef] [PubMed]
- Barentsz, J.O.; Richenberg, J.; Clements, R.; Choyke, P.; Verma, S.; Villeirs, G.; Rouviere, O.; Logager, V.; Fütterer, J.J. ESUR prostate MR guidelines 2012. Eur. Radiol. 2012, 22, 746–757. [Google Scholar] [CrossRef]
- Kohestani, K.; Wallström, J.; Dehlfors, N.; Sponga, O.M.; Månsson, M.; Josefsson, A.; Carlsson, S.; Hellström, M.; Hugosson, J. Performance and inter-observer variability of prostate MRI (PI-RADS version 2) outside high-volume centres. Scand J. Urol. 2019, 53, 304–311. [Google Scholar] [CrossRef] [PubMed]
- Vargas, H.A.; Hötker, A.M.; Goldman, D.A.; Moskowitz, C.S.; Gondo, T.; Matsumoto, K.; Ehdaie, B.; Woo, S.; Fine, S.W.; Reuter, V.E.; et al. Updated prostate imaging reporting and data system (PIRADS v2) recommendations for the detection of clinically significant prostate cancer using multiparametric MRI: Critical evaluation using whole-mount pathology as standard of reference. Eur. Radiol. 2016, 26, 1606–1612. [Google Scholar] [CrossRef]
- Weinreb, J.C.; Barentsz, J.O.; Choyke, P.L.; Cornud, F.; Haider, M.A.; Macura, K.J.; Margolis, D.; Schnall, M.D.; Shtern, F.; Tempany, C.M.; et al. PI-RADS Prostate Imaging—Reporting and Data System: 2015, Version 2. Eur. Urol. 2016, 69, 16–40. [Google Scholar] [CrossRef]
- Bergaglio, C.; Giasotto, V.; Marcenaro, M.; Barra, S.; Turazzi, M.; Bauckneht, M.; Casaleggio, A.; Sciabà, F.; Terrone, C.; Mantica, G.; et al. The Role of mpMRI in the Assessment of Prostate Cancer Recurrence Using the PI-RR System: Diagnostic Accuracy and Interobserver Agreement in Readers with Different Expertise. Diagnostics 2023, 13, 387. [Google Scholar] [CrossRef]
- Gaur, S.; Turkbey, B. Prostate MR Imaging for Posttreatment Evaluation and Recurrence. Radiol. Clin. N. Am. 2018, 56, 263–275. [Google Scholar] [CrossRef]
- Manceau, C.; Beauval, J.B.; Lesourd, M.; Almeras, C.; Aziza, R.; Gautier, J.R.; Loison, G.; Salin, A.; Tollon, C.; Soulié, M.; et al. MRI Characteristics Accurately Predict Biochemical Recurrence after Radical Prostatectomy. J. Clin. Med. 2020, 9, 3841. [Google Scholar] [CrossRef] [PubMed]
- Epstein, J.I.; Zelefsky, M.J.; Sjoberg, D.D.; Nelson, J.B.; Egevad, L.; Magi-Galluzzi, C.; Vickers, A.J.; Parwani, A.V.; Reuter, V.E.; Fine, S.W.; et al. A Contemporary Prostate Cancer Grading System: A Validated Alternative to the Gleason Score. Eur. Urol. 2016, 69, 435. [Google Scholar] [CrossRef] [PubMed]
- Merriman, K.M.; Harmon, S.A.; Belue, M.J.; Yilmaz, E.C.; Blake, Z.; Lay, N.S.; Phelps, T.E.; Merino, M.J.; Parnes, H.L.; Law, Y.M.; et al. Comparison of MRI-Based Staging and Pathologic Staging for Predicting Biochemical Recurrence of Prostate Cancer After Radical Prostatectomy. AJR Am. J. Roentgenol. 2023. Online ahead of print. [Google Scholar]
- Jambor, I.; Falagario, U.; Ratnani, P.; Perez, I.M.; Demir, K.; Merisaari, H.; Sobotka, S.; Haines, G.K.; Martini, A.; Beksac, A.T.; et al. Prediction of biochemical recurrence in prostate cancer patients who underwent prostatectomy using routine clinical prostate multiparametric MRI and decipher genomic score. J. Magn. Reson. Imaging 2020, 51, 1075–1085. [Google Scholar] [CrossRef] [PubMed]
- Delgadillo, R.; Ford, J.C.; Abramowitz, M.C.; Dal Pra, A.; Pollack, A.; Stoyanova, R. The role of radiomics in prostate cancer radiotherapy. Strahlenther. Onkol. 2020, 196, 900–912. [Google Scholar] [CrossRef]
- Stoyanova, R.; Takhar, M.; Tschudi, Y.; Ford, J.C.; Solórzano, G.; Erho, N.; Balagurunathan, Y.; Punnen, S.; Davicioni, E.; Gillies, R.J.; et al. Prostate cancer radiomics and the promise of radiogenomics. Transl. Cancer Res. 2016, 5, 432–447. [Google Scholar] [CrossRef] [PubMed]
- Cuocolo, R.; Stanzione, A.; Ponsiglione, A.; Romeo, V.; Verde, F.; Creta, M.; La Rocca, R.; Longo, N.; Pace, L.; Imbriaco, M. Clinically significant prostate cancer detection on MRI: A radiomic shape features study. Eur. J. Radiol. 2019, 116, 144–149. [Google Scholar] [CrossRef] [PubMed]
- Merisaari, H.; Taimen, P.; Shiradkar, R.; Ettala, O.; Pesola, M.; Saunavaara, J.; Boström, P.J.; Madabhushi, A.; Aronen, H.J.; Jambor, I. Repeatability of radiomics and machine learning for DWI: Short-term repeatability study of 112 patients with prostate cancer. Magn. Reson. Med. 2020, 83, 2293–2309. [Google Scholar] [CrossRef] [PubMed]
- Varghese, B.; Chen, F.; Hwang, D.; Palmer, S.L.; De Castro Abreu, A.L.; Ukimura, O.; Aron, M.; Gill, I.; Duddalwar, V.; Pandey, G. Objective risk stratification of prostate cancer using machine learning and radiomics applied to multiparametric magnetic resonance images. Sci. Rep. 2019, 9, 1570. [Google Scholar] [CrossRef]
- Bertelli, E.; Mercatelli, L.; Marzi, C.; Pachetti, E.; Baccini, M.; Barucci, A.; Colantonio, S.; Gherardini, L.; Lattavo, L.; Pascali, M.A.; et al. Machine and Deep Learning Prediction Of Prostate Cancer Aggressiveness Using Multiparametric MRI. Front. Oncol. 2021, 11, 802964. [Google Scholar] [CrossRef] [PubMed]
- Chaddad, A.; Kucharczyk, M.J.; Niazi, T. Multimodal Radiomic Features for the Predicting Gleason Score of Prostate Cancer. Cancers 2018, 10, 249. [Google Scholar] [CrossRef] [PubMed]
- Chaddad, A.; Niazi, T.; Probst, S.; Bladou, F.; Anidjar, M.; Bahoric, B. Predicting Gleason Score of Prostate Cancer Patients Using Radiomic Analysis. Front. Oncol. 2018, 8, 630. [Google Scholar] [CrossRef] [PubMed]
- Nai, Y.H.; Cheong, D.L.H.; Roy, S.; Kok, T.; Stephenson, M.C.; Schaefferkoetter, J.; Totman, J.J.; Conti, M.; Eriksson, L.; Robins, E.G.; et al. Comparison of quantitative parameters and radiomic features as inputs into machine learning models to predict the Gleason score of prostate cancer lesions. Magn. Reson. Imaging 2023, 100, 64–72. [Google Scholar] [CrossRef]
- Bernatz, S.; Ackermann, J.; Mandel, P.; Kaltenbach, B.; Zhdanovich, Y.; Harter, P.N.; Döring, C.; Hammerstingl, R.; Bodelle, B.; Smith, K.; et al. Comparison of machine learning algorithms to predict clinically significant prostate cancer of the peripheral zone with multiparametric MRI using clinical assessment categories and radiomic features. Eur. Radiol. 2020, 30, 6757–6769. [Google Scholar] [CrossRef] [PubMed]
- Fan, X.; Xie, N.; Chen, J.; Li, T.; Cao, R.; Yu, H.; He, M.; Wang, Z.; Wang, Y.; Liu, H.; et al. Multiparametric MRI and Machine Learning Based Radiomic Models for Preoperative Prediction of Multiple Biological Characteristics in Prostate Cancer. Front. Oncol. 2022, 12, 839621. [Google Scholar] [CrossRef]
- Yu, Y.; He, Z.; Ouyang, J.; Tan, Y.; Chen, Y.; Gu, Y.; Mao, L.; Ren, W.; Wang, J.; Lin, L.; et al. Magnetic resonance imaging radiomics predicts preoperative axillary lymph node metastasis to support surgical decisions and is associated with tumor microenvironment in invasive breast cancer: A machine learning, multicenter study. EBioMedicine 2021, 69, 103460. [Google Scholar] [CrossRef]
- Gillies, R.J.; Schabath, M.B. Radiomics Improves Cancer Screening and Early Detection. Cancer Epidemiol. Biomark. Prev. 2020, 29, 2556–2567. [Google Scholar] [CrossRef] [PubMed]
- McGarry, S.D.; Hurrell, S.L.; Kaczmarowski, A.L.; Cochran, E.J.; Connelly, J.; Rand, S.D.; Schmainda, K.M.; LaViolette, P.S. Magnetic Resonance Imaging-Based Radiomic Profiles Predict Patient Prognosis in Newly Diagnosed Glioblastoma Before Therapy. Tomography 2016, 2, 223–228. [Google Scholar] [CrossRef]
- Muller, B.G.; Shih, J.H.; Sankineni, S.; Marko, J.; Rais-Bahrami, S.; George, A.K.; de la Rosette, J.J.; Merino, M.J.; Wood, B.J.; Pinto, P.; et al. Prostate Cancer: Interobserver Agreement and Accuracy with the Revised Prostate Imaging Reporting and Data System at Multiparametric MR Imaging. Radiology 2015, 277, 741–750. [Google Scholar] [CrossRef]
- Westphalen, A.C.; McCulloch, C.E.; Anaokar, J.M.; Arora, S.; Barashi, N.S.; Barentsz, J.O.; Bathala, T.K.; Bittencourt, L.K.; Booker, M.T.; Braxton, V.G.; et al. Variability of the Positive Predictive Value of PI-RADS for Prostate MRI across 26 Centers: Experience of the Society of Abdominal Radiology Prostate Cancer Disease-focused Panel. Radiology 2020, 296, 76–84. [Google Scholar] [CrossRef] [PubMed]
- van der Slot, M.A.; Hollemans, E.; den Bakker, M.A.; Hoedemaeker, R.; Kliffen, M.; Budel, L.M.; Goemaere, N.N.T.; van Leenders, G.J.L.H. Inter-observer variability of cribriform architecture and percent Gleason pattern 4 in prostate cancer: Relation to clinical outcome. Virchows Arch. 2021, 478, 249–256. [Google Scholar] [CrossRef]
- McGarry, S.D.; Bukowy, J.D.; Iczkowski, K.A.; Lowman, A.K.; Brehler, M.; Bobholz, S.; Nencka, A.; Barrington, A.; Jacobsohn, K.; Unteriner, J.; et al. Radio-pathomic mapping model generated using annotations from five pathologists reliably distinguishes high-grade prostate cancer. J. Med. Imaging 2020, 7, 054501. [Google Scholar] [CrossRef]
- Niazi, M.K.K.; Parwani, A.V.; Gurcan, M.N. Digital pathology and artificial intelligence. Lancet Oncol. 2019, 20, e253–e261. [Google Scholar] [CrossRef]
- American Urological Association. Prostate-specific antigen (PSA) best practice policy. American Urological Association (AUA). Oncology 2000, 14, 267–272+277–268+280 passim. [Google Scholar]
- Swanson, G.P.; Chen, W.; Trevathan, S.; Hermans, M. Long-Term Follow-Up after Prostatectomy for Prostate Cancer and the Need for Active Monitoring. Prostate Cancer 2020, 2020, 7196189. [Google Scholar] [CrossRef]
- Sood, A.; Jeong, W.; Peabody, J.O.; Hemal, A.K.; Menon, M. Robot-Assisted Radical Prostatectomy: Inching toward Gold Standard. Urol. Clin. N. Am. 2014, 41, 473–484. [Google Scholar] [CrossRef] [PubMed]
- Menon, M.; Hemal, A.K. Vattikuti Institute prostatectomy: A technique of robotic radical prostatectomy: Experience in more than 1000 cases. J. Endourol. 2004, 18, 611–619. [Google Scholar] [CrossRef] [PubMed]
- Shah, V.; Pohida, T.; Turkbey, B.; Mani, H.; Merino, M.; Pinto, P.A.; Choyke, P.; Bernardo, M. A method for correlating in vivo prostate magnetic resonance imaging and histopathology using individualized magnetic resonance -based molds. Rev. Sci. Instrum. 2009, 80, 104301. [Google Scholar] [CrossRef] [PubMed]
- Cox, R.W. AFNI: Software for analysis and visualization of functional magnetic resonance neuroimages. Comput. Biomed. Res. 1996, 29, 162–173. [Google Scholar] [CrossRef] [PubMed]
- Hurrell, S.L.; McGarry, S.D.; Kaczmarowski, A.; Iczkowski, K.A.; Jacobsohn, K.; Hohenwalter, M.D.; Hall, W.A.; See, W.A.; Banerjee, A.; Charles, D.K.; et al. Optimized b-value selection for the discrimination of prostate cancer grades, including the cribriform pattern, using diffusion weighted imaging. J. Med. Imaging 2017, 5, 162–173. [Google Scholar] [CrossRef] [PubMed]
- McGarry, S.D.; Hurrell, S.L.; Iczkowski, K.A.; Hall, W.; Kaczmarowski, A.L.; Banerjee, A.; Keuter, T.; Jacobsohn, K.; Bukowy, J.D.; Nevalainen, M.T.; et al. Radio-pathomic Maps of Epithelium and Lumen Density Predict the Location of High-Grade Prostate Cancer. Int. J. Radiat. Oncol. Biol. Phys. 2018, 101, 1179–1187. [Google Scholar] [CrossRef] [PubMed]
- McGarry, S.D.; Bukowy, J.D.; Iczkowski, K.A.; Unteriner, J.G.; Duvnjak, P.; Lowman, A.K.; Jacobsohn, K.; Hohenwalter, M.; Griffin, M.O.; Barrington, A.W.; et al. Gleason probability maps: A radiomics tool for mapping prostate cancer likelihood in mri space. Tomography 2019, 5, 127–134. [Google Scholar] [CrossRef]
- Iczkowski, K.A.; Torkko, K.C.; Kotnis, G.R.; Wilson, R.S.; Huang, W.; Wheeler, T.M.; Abeyta, A.M.; La Rosa, F.G.; Cook, S.; Werahera, P.N.; et al. Digital quantification of five high-grade prostate cancer patterns, including the cribriform pattern, and their association with adverse outcome. Am. J. Clin. Pathol. 2011, 136, 98–107. [Google Scholar] [CrossRef]
- Iczkowski, K.A.; Paner, G.P.; Van der Kwast, T. The New Realization About Cribriform Prostate Cancer. Adv. Anat. Pathol. 2018, 25, 31–37. [Google Scholar] [CrossRef]
- Kweldam, C.F.; Wildhagen, M.F.; Steyerberg, E.W.; Bangma, C.H.; Van Der Kwast, T.H.; Van Leenders, G.J.L.H. Cribriform growth is highly predictive for postoperative metastasis and disease-specific death in Gleason score 7 prostate cancer. Mod. Pathol. 2015, 28, 457–464. [Google Scholar] [CrossRef] [PubMed]
- Montironi, R.; Cimadamore, A.; Gasparrini, S.; Mazzucchelli, R.; Santoni, M.; Massari, F.; Cheng, L.; Lopez-Beltran, A.; Scarpelli, M. Prostate cancer with cribriform morphology: Diagnosis, aggressiveness, molecular pathology and possible relationships with intraductal carcinoma. Expert Rev. Anticancer Ther. 2018, 18, 685–693. [Google Scholar] [CrossRef] [PubMed]
- Bobholz, S.A.; Lowman, A.K.; Barrington, A.; Brehler, M.; McGarry, S.; Cochran, E.J.; Connelly, J.; Mueller, W.M.; Agarwal, M.; O’Neill, D.; et al. Radiomic Features of Multiparametric MRI Present Stable Associations with Analogous Histological Features in Patients with Brain Cancer. Tomography 2020, 6, 60–169. [Google Scholar] [CrossRef]
- McGarry, S.D.; Brehler, M.; Bukowy, J.D.; Lowman, A.K.; Bobholz, S.A.; Duenweg, S.R.; Banerjee, A.; Hurrell, S.L.; Malyarenko, D.; Chenevert, T.L.; et al. Multi-Site Concordance of Diffusion-Weighted Imaging Quantification for Assessing Prostate Cancer Aggressiveness. J. Magn. Reson. Imaging 2021, 55, 1745–1758. [Google Scholar] [CrossRef] [PubMed]
- van Griethuysen, J.J.M.; Fedorov, A.; Parmar, C.; Hosny, A.; Aucoin, N.; Narayan, V.; Beets-Tan, R.G.H.; Fillion-Robin, J.C.; Pieper, S.; Aerts, H.J.W.L. Computational Radiomics System to Decode the Radiographic Phenotype. Cancer Res. 2017, 77, e104–e107. [Google Scholar] [CrossRef] [PubMed]
- Cookson, M.S.; Aus, G.; Burnett, A.L.; Canby-Hagino, E.D.; D’Amico, A.V.; Dmochowski, R.R.; Eton, D.T.; Forman, J.D.; Goldenberg, S.L.; Hernandez, J.; et al. Variation in the definition of biochemical recurrence in patients treated for localized prostate cancer: The American Urological Association Prostate Guidelines for Localized Prostate Cancer Update Panel report and recommendations for a standard in the reporting of surgical outcomes. J. Urol. 2007, 177, 540–545. [Google Scholar] [CrossRef] [PubMed]
- Heidenreich, A.; Bastian, P.J.; Bellmunt, J.; Bolla, M.; Joniau, S.; van der Kwast, T.; Mason, M.; Matveev, V.; Wiegel, T.; Zattoni, F.; et al. EAU guidelines on prostate cancer. Part II: Treatment of advanced, relapsing, and castration-resistant prostate cancer. Eur. Urol. 2014, 65, 467–479. [Google Scholar] [CrossRef]
- Amling, C.L.; Blute, M.L.; Bergstralh, E.J.; Seay, T.M.; Slezak, J.; Zincke, H. Long-term hazard of progression after radical prostatectomy for clinically localized prostate cancer: Continued risk of biochemical failure after 5 years. J. Urol. 2000, 164, 101–105. [Google Scholar] [CrossRef] [PubMed]
- Brockman, J.A.; Alanee, S.; Vickers, A.J.; Scardino, P.T.; Wood, D.P.; Kibel, A.S.; Lin, D.W.; Bianco, F.J.; Rabah, D.M.; Klein, E.A.; et al. Nomogram predicting prostate cancer-specific mortality for men with biochemical recurrence after radical prostatectomy. Eur. Urol. 2015, 67, 1160–1167. [Google Scholar] [CrossRef] [PubMed]
- Ozkan, T.A.; Eruyar, A.T.; Cebeci, O.O.; Memik, O.; Ozcan, L.; Kuskonmaz, I. Interobserver variability in Gleason histological grading of prostate cancer. Scand. J. Urol. 2016, 50, 420–424. [Google Scholar] [CrossRef]
- An, P.; Lin, Y.; Hu, Y.; Qin, P.; Ye, Y.; Gu, W.; Li, X.; Song, P.; Feng, G. Predicting Model of Biochemical Recurrence of Prostate Carcinoma (PCa-BCR) Using MR Perfusion-Weighted Imaging-Based Radiomics. Technol. Cancer Res. Treat. 2023, 22, 15330338231166766. [Google Scholar] [CrossRef]
- Zhong, Q.Z.; Long, L.H.; Liu, A.; Li, C.M.; Xiu, X.; Hou, X.Y.; Wu, Q.H.; Gao, H.; Xu, Y.G.; Zhao, T.; et al. Radiomics of Multiparametric MRI to Predict Biochemical Recurrence of Localized Prostate Cancer After Radiation Therapy. Front. Oncol. 2020, 10, 731. [Google Scholar] [CrossRef]
- Zhu, X.; Liu, Z.; He, J.; Li, Z.; Huang, Y.; Lu, J. MRI-Derived Radiomics Model to Predict the Biochemical Recurrence of Prostate Cancer Following Seed Brachytherapy. Arch. Esp. Urol. 2023, 76, 264–269. [Google Scholar] [CrossRef]
- Algohary, A.; Viswanath, S.; Shiradkar, R.; Ghose, S.; Pahwa, S.; Moses, D.; Jambor, I.; Shnier, R.; Böhm, M.; Haynes, A.M.; et al. Radiomic features on MRI enable risk categorization of prostate cancer patients on active surveillance: Preliminary findings. J. Magn. Reson. Imaging 2018, 48, 818–828. [Google Scholar] [CrossRef]
- Prata, F.; Anceschi, U.; Cordelli, E.; Faiella, E.; Civitella, A.; Tuzzolo, P.; Iannuzzi, A.; Ragusa, A.; Esperto, F.; Prata, S.M.; et al. Radiomic Machine-Learning Analysis of Multiparametric Magnetic Resonance Imaging in the Diagnosis of Clinically Significant Prostate Cancer: New Combination of Textural and Clinical Features. Curr. Oncol. 2023, 30, 2021–2031. [Google Scholar] [CrossRef]
- He, D.; Wang, X.; Fu, C.; Wei, X.; Bao, J.; Ji, X.; Bai, H.; Xia, W.; Gao, X.; Huang, Y.; et al. MRI-based radiomics models to assess prostate cancer, extracapsular extension and positive surgical margins. Cancer Imaging 2021, 21, 46. [Google Scholar] [CrossRef] [PubMed]
- Algohary, A.; Shiradkar, R.; Pahwa, S.; Purysko, A.; Verma, S.; Moses, D.; Shnier, R.; Haynes, A.M.; Delprado, W.; Thompson, J.; et al. Combination of Peri-Tumoral and Intra-Tumoral Radiomic Features on Bi-Parametric MRI Accurately Stratifies Prostate Cancer Risk: A Multi-Site Study. Cancers 2020, 12, 2200. [Google Scholar] [CrossRef]
- Shiradkar, R.; Ghose, S.; Jambor, I.; Taimen, P.; Ettala, O.; Purysko, A.S.; Madabhushi, A. Radiomic features from pretreatment biparametric MRI predict prostate cancer biochemical recurrence: Preliminary findings. J. Magn. Reson. Imaging 2018, 48, 1626–1636. [Google Scholar] [CrossRef] [PubMed]
- Lee, H.W.; Kim, E.; Na, I.; Kim, C.K.; Seo, S.I.; Park, H. Novel Multiparametric Magnetic Resonance Imaging-Based Deep Learning and Clinical Parameter Integration for the Prediction of Long-Term Biochemical Recurrence-Free Survival in Prostate Cancer after Radical Prostatectomy. Cancers 2023, 15, 3416. [Google Scholar] [CrossRef]
- Dhatt, R.; Choy, S.; Co, S.J.; Ischia, J.; Kozlowski, P.; Harris, A.C.; Jones, E.C.; Black, P.C.; Goldenberg, S.L.; Chang, S.D. MRI of the Prostate With and Without Endorectal Coil at 3 T: Correlation With Whole-Mount Histopathologic Gleason Score. AJR Am. J. Roentgenol. 2020, 215, 133–141. [Google Scholar] [CrossRef]
- Gawlitza, J.; Reiss-Zimmermann, M.; Thörmer, G.; Schaudinn, A.; Linder, N.; Garnov, N.; Horn, L.C.; Minh, D.H.; Ganzer, R.; Stolzenburg, J.U.; et al. Impact of the use of an endorectal coil for 3 T prostate MRI on image quality and cancer detection rate. Sci. Rep. 2017, 7, 40640. [Google Scholar] [CrossRef] [PubMed]
- Lee, G.; Oto, A.; Giurcanu, M. Prostate MRI: Is Endorectal Coil Necessary?-A Review. Life 2022, 12, 569. [Google Scholar] [CrossRef] [PubMed]
- Turkbey, B.; Merino, M.J.; Gallardo, E.C.; Shah, V.; Aras, O.; Bernardo, M.; Mena, E.; Daar, D.; Rastinehad, A.R.; Linehan, W.M.; et al. Comparison of endorectal coil and nonendorectal coil T2W and diffusion-weighted MRI at 3 Tesla for localizing prostate cancer: Correlation with whole-mount histopathology. J. Magn. Reson. Imaging 2014, 39, 1443–1448. [Google Scholar] [CrossRef] [PubMed]
| Clinical Feature | Training (n = 186) | Testing (n = 93) | Total (n = 279) |
|---|---|---|---|
| Age at RP, years (mean ± SD) | 62 ± 6 | 62 ± 7 | 62 ± 6 |
| Race (n, %) | |||
| African American | 11 (6) | 5 (5) | 16 (6) |
| White/Caucasian | 91 (49) | 43 (46) | 134 (48) |
| Asian | 12 (6) | 5 (5) | 17 (6) |
| Other | 3 (2) | 0 (0) | 3 (1) |
| Missing | 80 (43) | 45 (48) | 125 (45) |
| Preoperative PSA, ng/mL (n, %) | |||
| <6 | 77 (41) | 50 (54) | 127 (46) |
| ≥6–10 | 71 (38) | 23 (25) | 94 (34) |
| ≥10–20 | 31 (17) | 16 (17) | 47 (17) |
| ≥20–30 | 5 (3) | 2 (2) | 7 (3) |
| ≥30 | 2 (1) | 2 (2) | 4 (1) |
| Grade group at RP (n, %) | |||
| 6 | 18 (10) | 10 (11) | 28 (10) |
| 3 + 4 | 117 (63) | 52 (56) | 169 (61) |
| 4 + 3 | 26 (14) | 15 (16) | 41 (15) |
| 8 | 5 (3) | 4 (4) | 9 (3) |
| ≥9 | 20 (11) | 12 (13) | 32 (11) |
| Clinical Stage (n, %) | |||
| T1 | 145 (78) | 60 (65) | 205 (73) |
| T2 | 24 (13) | 22 (24) | 46 (16) |
| Missing | 11 (6) | 17 (18) | 28 (11) |
| Surgical Stage (n, %) | |||
| 2a,b | 102 (55) | 48 (52) | 150 (54) |
| 2c | 29 (16) | 12 (13) | 41 (15) |
| 3a,b | 52 (28) | 31 (33) | 83 (30) |
| Missing | 3 (1) | 2 (2) | 5(1) |
| Cribriform Pattern (n, %) (n = 257) | |||
| Presence | 75 (40) | 40 (43) | 115 (41) |
| Absence | 98 (53) | 44 (47) | 142 (51) |
| Missing | 13 (7) | 9 (10) | 22 (8) |
| Biochemical Recurrence (n, %) | 28 (15) | 18 (19) | 46 (16) |
| Time to BCR, years (mean, range) (n = 45) | 1.68 (0.1–5.0) | ||
| Follow-up time post RP, years (mean, range) | 2.1 (0.1–7.3) | ||
| Class | Radiomic Features | t-Value | p-Value |
|---|---|---|---|
| First Order Features | 90th Percentile | 2.38 | 0.02 * |
| Interquartile Range | 2.07 | 0.04 * | |
| Maximum | −1.87 | 0.06 + | |
| Mean | 2.31 | 0.02 * | |
| Mean Absolute Deviation | 2.16 | 0.03 * | |
| Range | −1.92 | 0.06+ | |
| Robust Mean Absolute Deviation | 2.25 | 0.03 * | |
| Root Mean Squared | 2.05 | 0.04 * | |
| Variance | 2.09 | 0.04 * | |
| GLSZM | Zone Entropy | −1.88 | 0.06 + |
| Model | Accuracy | Sensitivity | Specificity |
|---|---|---|---|
| BCR (Full-Res) | AUC = 0.97 | 0.88 | 0.93 |
| Cancer/Non-cancer (Full-Res) | ACC = 89.9% | 0.80 | 1 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Duenweg, S.R.; Bobholz, S.A.; Barrett, M.J.; Lowman, A.K.; Winiarz, A.; Nath, B.; Stebbins, M.; Bukowy, J.; Iczkowski, K.A.; Jacobsohn, K.M.; et al. T2-Weighted MRI Radiomic Features Predict Prostate Cancer Presence and Eventual Biochemical Recurrence. Cancers 2023, 15, 4437. https://doi.org/10.3390/cancers15184437
Duenweg SR, Bobholz SA, Barrett MJ, Lowman AK, Winiarz A, Nath B, Stebbins M, Bukowy J, Iczkowski KA, Jacobsohn KM, et al. T2-Weighted MRI Radiomic Features Predict Prostate Cancer Presence and Eventual Biochemical Recurrence. Cancers. 2023; 15(18):4437. https://doi.org/10.3390/cancers15184437
Chicago/Turabian StyleDuenweg, Savannah R., Samuel A. Bobholz, Michael J. Barrett, Allison K. Lowman, Aleksandra Winiarz, Biprojit Nath, Margaret Stebbins, John Bukowy, Kenneth A. Iczkowski, Kenneth M. Jacobsohn, and et al. 2023. "T2-Weighted MRI Radiomic Features Predict Prostate Cancer Presence and Eventual Biochemical Recurrence" Cancers 15, no. 18: 4437. https://doi.org/10.3390/cancers15184437
APA StyleDuenweg, S. R., Bobholz, S. A., Barrett, M. J., Lowman, A. K., Winiarz, A., Nath, B., Stebbins, M., Bukowy, J., Iczkowski, K. A., Jacobsohn, K. M., Vincent-Sheldon, S., & LaViolette, P. S. (2023). T2-Weighted MRI Radiomic Features Predict Prostate Cancer Presence and Eventual Biochemical Recurrence. Cancers, 15(18), 4437. https://doi.org/10.3390/cancers15184437





