The First Cold Atmospheric Plasma Phase I Clinical Trial for the Treatment of Advanced Solid Tumors: A Novel Treatment Arm for Cancer
Abstract
Simple Summary
Abstract
1. Introduction
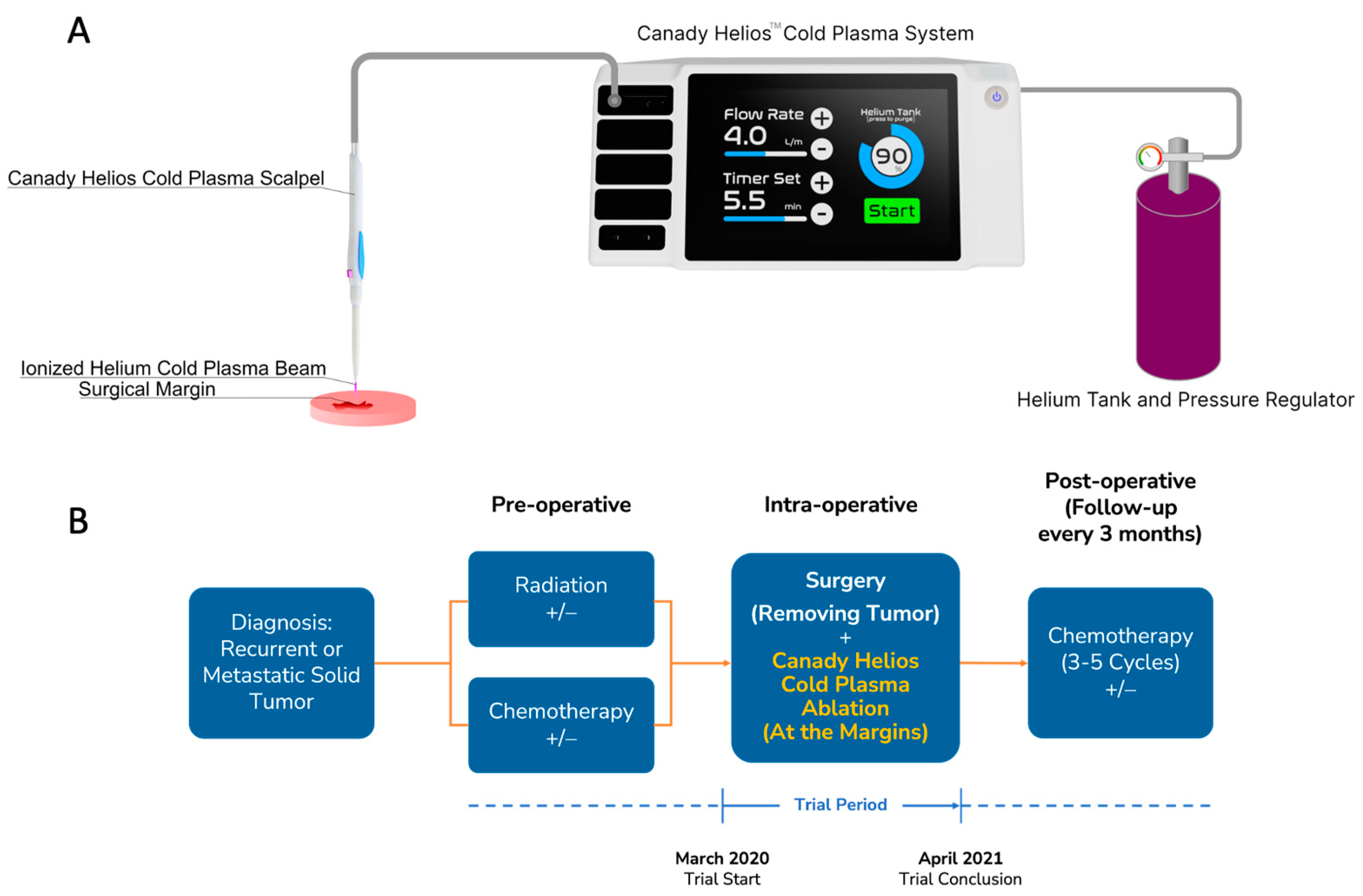
2. Materials and Methods
2.1. Trial Oversight
2.2. Patients
2.3. Safety Assessment
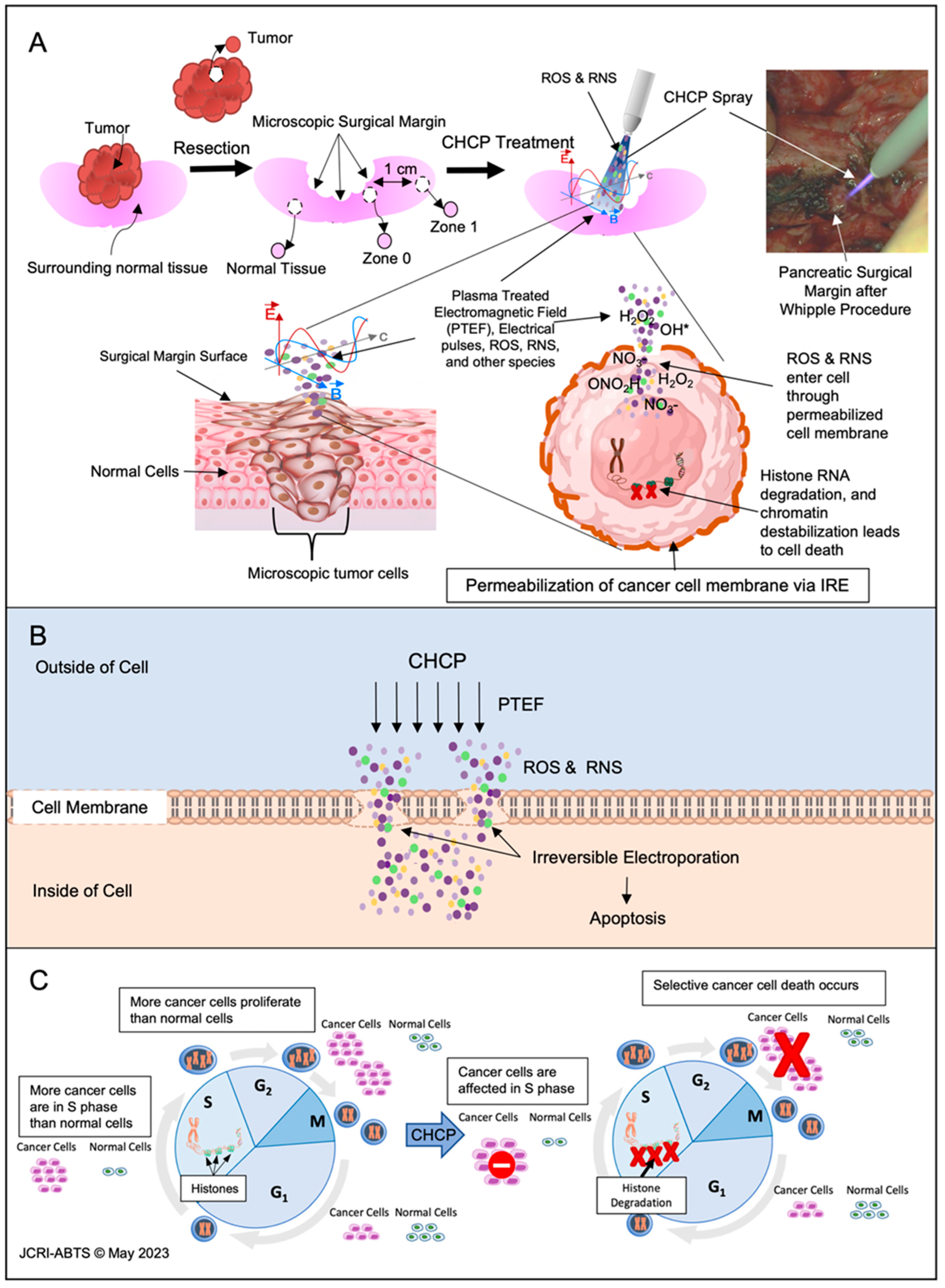
2.4. Pathology
2.5. Primary Culture
2.6. Quantitative Confocal Immunofluorescence Analysis
2.7. Statistical Analysis
3. Results
3.1. Patient Demographics and Characteristics
3.2. Safety Outcomes
3.2.1. Intra-Operative Physiological Data and CHCP Temperature
3.2.2. Post-Operative Adverse Events
3.3. Efficacy Outcomes
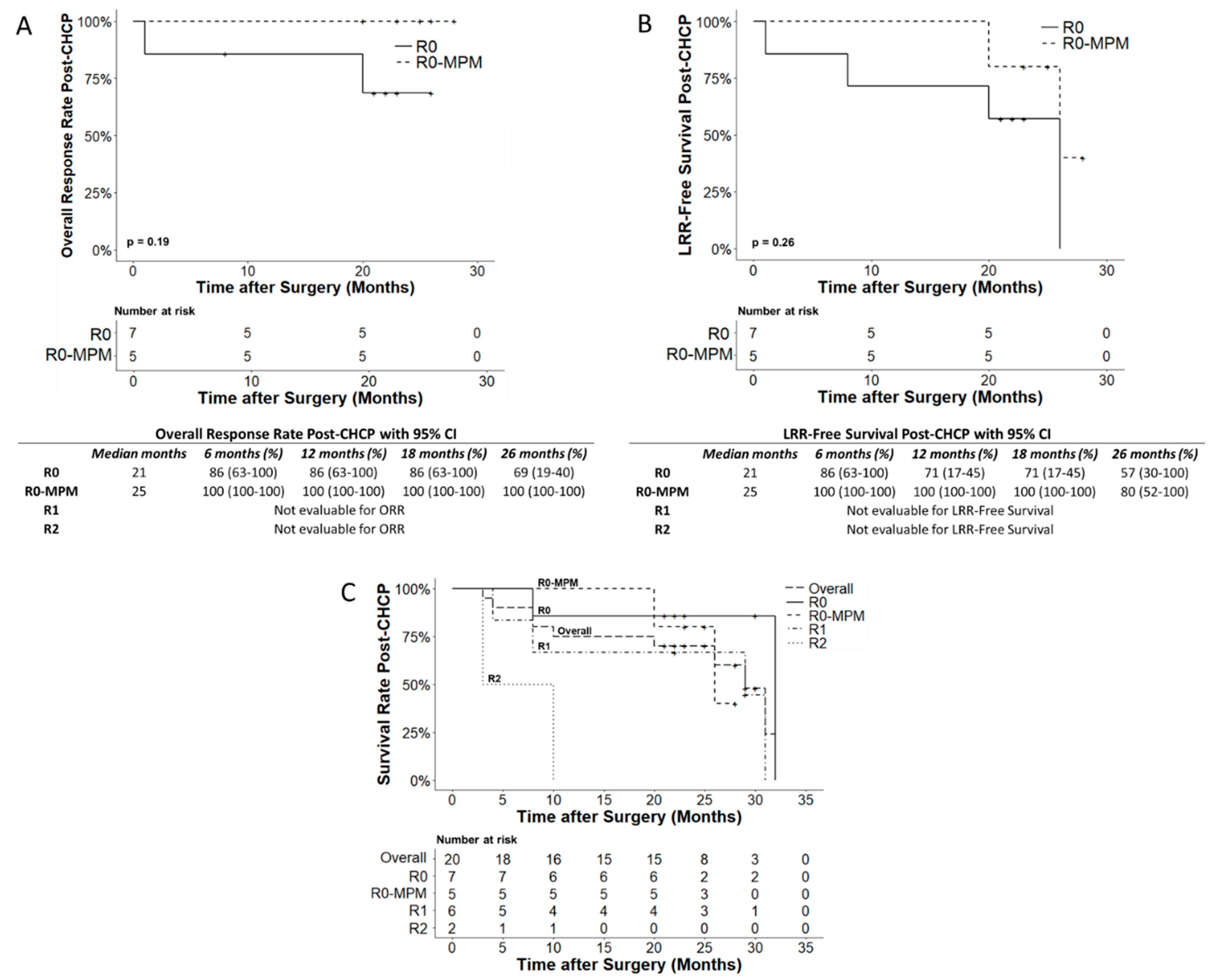
3.3.1. Recurrence and Overall Survival
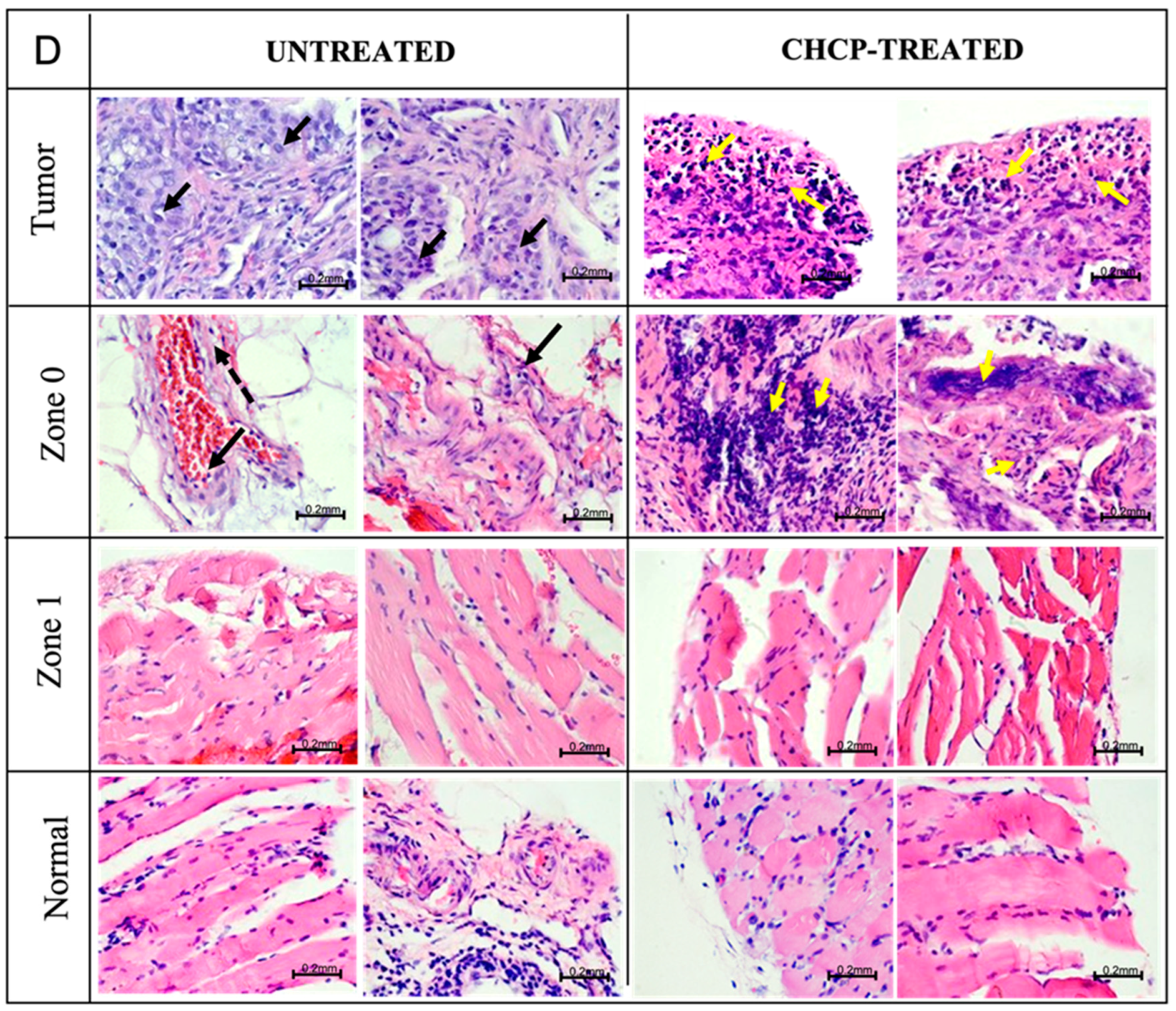
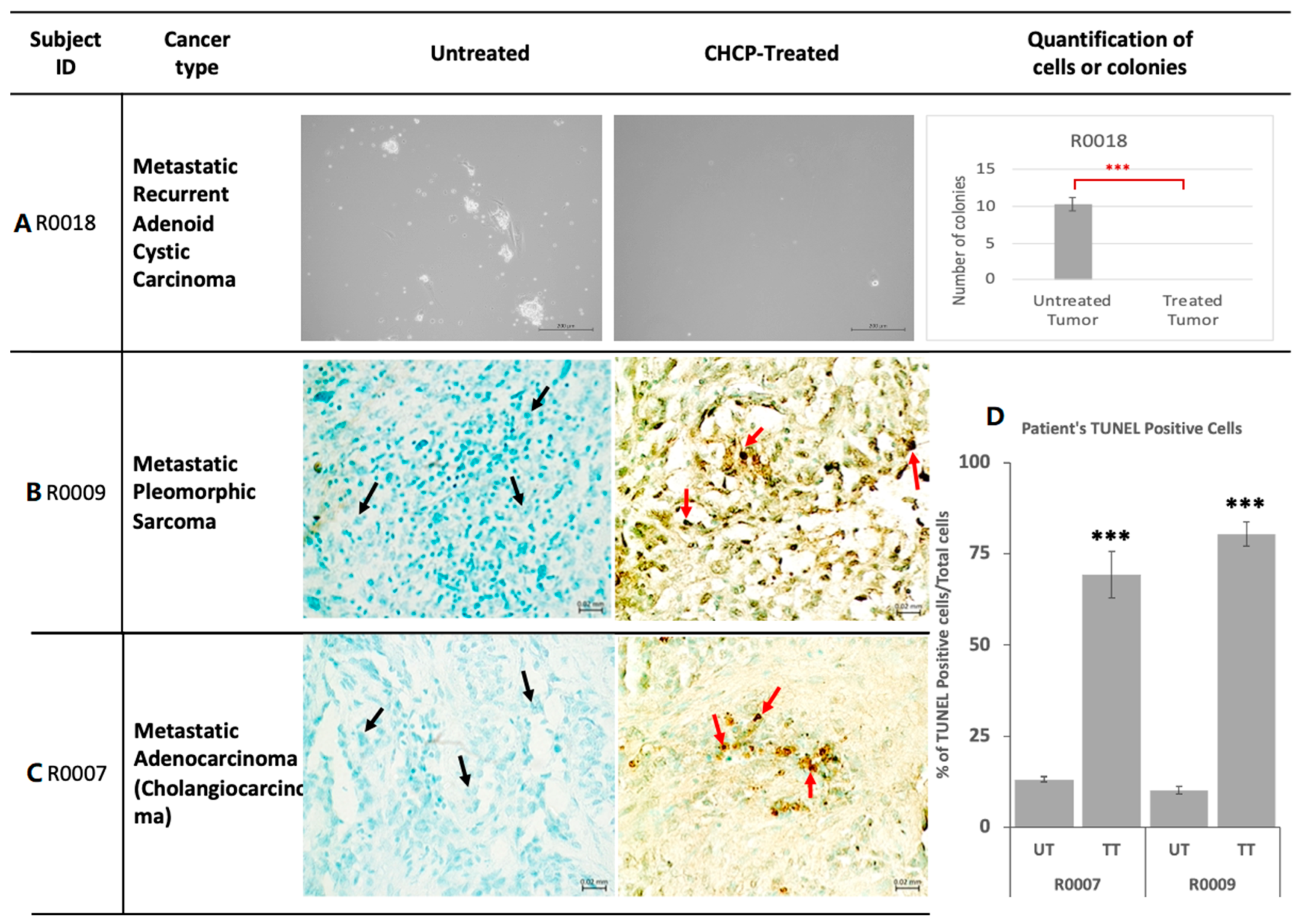
3.3.2. Histopathology Evaluation
3.3.3. Histopathology Findings
3.3.4. Primary Tissue Culture
3.3.5. Apoptotic DNA Damage Analysis by TUNEL Assay
3.3.6. Confocal Immunofluorescence Analysis
3.3.7. Gene Expression Analysis
3.3.8. Regulation of Protein Expression
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Siegel, R.L.; Miller, K.D.; Fuchs, H.E.; Jemal, A. Cancer Statistics, 2021. CA Cancer J. Clin. 2021, 71, 7–33. [Google Scholar] [CrossRef] [PubMed]
- Miller, K.D.; Nogueira, L.; Devasia, T.; Mariotto, A.T.B.; Yabroff, K.R.; Jemal, A.; Kramer, J.; Siegel, R.L. Cancer treatment and survivorship statistics, 2022. CA Cancer J. Clin. 2022, 72, 409–436. [Google Scholar] [CrossRef] [PubMed]
- Sanoff, H.K. Improving Treatment Approaches for Rectal Cancer. N. Engl. J. Med. 2022, 386, 2425–2426. [Google Scholar] [CrossRef] [PubMed]
- Mahvi, D.A.; Liu, R.; Grinstaff, M.W.; Colson, Y.L.; Raut, C.P. Local Cancer Recurrence: The Realities, Challenges, and Opportunities for New Therapies. CA Cancer J. Clin. 2018, 68, 488–505. [Google Scholar] [CrossRef] [PubMed]
- Buchholz, T.A.; Ali, S.; Hunt, K.K. Multidisciplinary Management of Locoregional Recurrent Breast Cancer. J. Clin. Oncol. 2020, 38, 2321–2328. [Google Scholar] [CrossRef]
- Cihoric, N.; Tsikkinis, A.; Filipovic, N.; Jeremic, B. Treatment options for isolated locoregional recurrences of nonsmall cell lung cancer after surgery: Yes, radiation therapy too! Eur. Respir. J. 2016, 48, 276–278. [Google Scholar] [CrossRef]
- Laroussi, M.; Lu, X.; Keidar, M. Perspective: The physics, diagnostics, and applications of atmospheric pressure low temperature plasma sources used in plasma medicine. J. Appl. Phys. 2017, 122. [Google Scholar] [CrossRef]
- Charles, F.; Morrison, J. Electrosurgical Method and Apparatus for Initiating An Electrical Discharge in An Inert Gas Flow. U.S. Patent 4,040,426, 9 August 1977. [Google Scholar]
- Canady, J. Surgical Coagulation Device. U.S. Patent 5,207,675, 4 May 1993. [Google Scholar]
- Babington, P.; Rajjoub, K.; Canady, J.; Siu, A.; Keidar, M.; Sherman, J.H. Use of cold atmospheric plasma in the treatment of cancer. Biointerphases 2015, 10, 029403. [Google Scholar] [CrossRef] [PubMed]
- Laroussi, M. Low Temperature Plasma-Based Sterilization: Overview and State-of-the-Art. Plasma Process. Polym. 2005, 2, 391–400. [Google Scholar] [CrossRef]
- Yan, D.; Wang, Q.; Adhikari, M.; Malyavko, A.; Lin, L.; Zolotukhin, D.B.; Yao, X.; Kirschner, M.; Sherman, J.H.; Keidar, M. A Physically Triggered Cell Death via Transbarrier Cold Atmospheric Plasma Cancer Treatment. ACS Appl. Mater. Interfaces 2020, 12, 34548–34563. [Google Scholar] [CrossRef]
- Haralambiev, L.; Nitsch, A.; Einenkel, R.; Muzzio, D.O.; Gelbrich, N.; Burchardt, M.; Zygmunt, M.; Ekkernkamp, A.; Stope, M.B.; Gumbel, D. The Effect of Cold Atmospheric Plasma on the Membrane Permeability of Human Osteosarcoma Cells. Anticancer Res. 2020, 40, 841–846. [Google Scholar] [CrossRef]
- Ly, L.; Cheng, X.; Murthy, S.R.K.; Jones, O.Z.; Zhuang, T.; Gitelis, S.; Blank, A.T.; Nissan, A.; Adileh, M.; Colman, M.; et al. Canady Cold Helios Plasma Reduces Soft Tissue Sarcoma Viability by Inhibiting Proliferation, Disrupting Cell Cycle, and Inducing Apoptosis: A Preliminary Report. Molecules 2022, 27, 4168. [Google Scholar] [CrossRef]
- Gjika, E.; Pal-Ghosh, S.; Kirschner, M.E.; Lin, L.; Sherman, J.H.; Stepp, M.A.; Keidar, M. Combination Therapy of Cold Atmospheric Plasma (CAP) With Temozolomide in the Treatment of U87MG Glioblastoma Cells. Sci. Rep. 2020, 10, 16495. [Google Scholar] [CrossRef] [PubMed]
- Chen, Z.; Simonyan, H.; Cheng, X.; Gjika, E.; Lin, L.; Canady, J.; Sherman, J.H.; Young, C.; Keidar, M. A Novel Micro Cold Atmospheric Plasma Device for Glioblastoma Both In Vitro and In Vivo. Cancers 2017, 9, 61. [Google Scholar] [CrossRef] [PubMed]
- Liu, J.-R.; Wu, Y.-M.; Xu, G.-M.; Gao, L.-G.; Ma, Y.; Shi, X.-M.; Zhang, G.-J. Low-temperature plasma induced melanoma apoptosis by triggering a p53/PIGs/caspase-dependent pathway in vivo and in vitro. J. Phys. D Appl. Phys. 2019, 52, 315204. [Google Scholar] [CrossRef]
- Cheng, X.; Rowe, W.; Ly, L.; Shashurin, A.; Zhuang, T.; Wigh, S.; Basadonna, G.; Trink, B.; Keidar, M.; Canady, J. Treatment of Triple-Negative Breast Cancer Cells with the Canady Cold Plasma Conversion System: Preliminary Results. Plasma 2018, 1, 218–228. [Google Scholar] [CrossRef]
- Ly, L.; Cheng, X.; Murthy, S.R.K.; Zhuang, T.; Jones, O.Z.; Basadonna, G.; Keidar, M.; Canady, J. Canady Cold Plasma Conversion System Treatment: An effective Inhibitor of Cell Viability In Breast Cancer Molecular Subtypes. Clin. Plasma Med. 2020, 19, 100109. [Google Scholar] [CrossRef]
- Sun, C.; Chesnokov, V.; Larson, G.; Itakura, K. Glucosamine Enhances TRAIL-Induced Apoptosis in the Prostate Cancer Cell Line DU145. Medicines 2019, 6, 104. [Google Scholar] [CrossRef]
- Griseti, E.; Kolosnjaj-Tabi, J.; Gibot, L.; Fourquaux, I.; Rols, M.P.; Yousfi, M.; Merbahi, N.; Golzio, M. Pulsed Electric Field Treatment Enhances the Cytotoxicity of Plasma-Activated Liquids in a Three-Dimensional Human Colorectal Cancer Cell Model. Sci. Rep. 2019, 9, 7583. [Google Scholar] [CrossRef]
- Ma, J.; Yu, K.N.; Cheng, C.; Ni, G.; Shen, J.; Han, W. Targeting Nrf2-mediated heme oxygenase-1 enhances non-thermal plasma-induced cell death in non-small-cell lung cancer A549 cells. Arch. Biochem. Biophys. 2018, 658, 54–65. [Google Scholar] [CrossRef] [PubMed]
- Rowe, W.; Cheng, X.; Ly, L.; Zhuang, T.; Basadonna, G.; Trink, B.; Keidar, M.; Canady, J. The Canady Helios Cold Plasma Scalpel Significantly Decreases Viability in Malignant Solid Tumor Cells in a Dose-Dependent Manner. Plasma 2018, 1, 177–188. [Google Scholar] [CrossRef]
- Feil, L.; Koch, A.; Utz, R.; Ackermann, M.; Barz, J.; Stope, M.; Kramer, B.; Wallwiener, D.; Brucker, S.Y.; Weiss, M. Cancer-Selective Treatment of Cancerous and Non-Cancerous Human Cervical Cell Models by a Non-Thermally Operated Electrosurgical Argon Plasma Device. Cancers 2020, 12, 1037. [Google Scholar] [CrossRef] [PubMed]
- Guerrero-Preston, R.; Ogawa, T.; Uemura, M.; Shumulinsky, G.; Valle, B.L.; Pirini, F.; Ravi, R.; Sidransky, D.; Keidar, M.; Trink, B. Cold Atmospheric Plasma Treatment Selectively Targets Head and Neck Squamous Cell Carcinoma Cells. Int. J. Mol. Med. 2014, 34, 941–946. [Google Scholar] [CrossRef]
- Yan, D.; Sherman, J.H.; Keidar, M. Cold atmospheric plasma, a novel promising anti-cancer treatment modality. Oncotarget 2017, 8, 15977–15995. [Google Scholar] [CrossRef]
- Vaquero, J.; Judée, F.; Vallette, M.; Decauchy, H.; Arbelaiz, A.; Aoudjehane, L.; Scatton, O.; Gonzalez-Sanchez, E.; Merabtene, F.; Augustin, J.; et al. Cold-Atmospheric Plasma Induces Tumor Cell Death in Preclinical In Vivo and In Vitro Models of Human Cholangiocarcinoma. Cancers 2020, 12, 1280. [Google Scholar] [CrossRef] [PubMed]
- Ly, L.; Jones, S.; Shashurin, A.; Zhuang, T.; Rowe, W.; Cheng, X.; Wigh, S.; Naab, T.; Keidar, M.; Canady, J. A New Cold Plasma Jet: Performance Evaluation of Cold Plasma, Hybrid Plasma and Argon Plasma Coagulation. Plasma 2018, 1, 189–200. [Google Scholar] [CrossRef]
- Canady, J.; Shashurin, A.; Keidar, M.; Zhuang, T. Integrated cold plasma and high frequency plasma electrosurgical system and method. U.S. Patent No. 9,999,462, 19 June 2018. [Google Scholar]
- Cheng, X.; Murthy, S.R.K.; Zhuang, T.; Ly, L.; Jones, O.; Basadonna, G.; Keidar, M.; Kanaan, Y.; Canady, J. Canady Helios Cold Plasma Induces Breast Cancer Cell Death by Oxidation of Histone mRNA. Int. J. Mol. Sci. 2021, 22, 9578. [Google Scholar] [CrossRef]
- Elbedour, A.; Cheng, X.; Murthy, S.R.K.; Zhuang, T.; Ly, L.; Jones, O.; Basadonna, G.; Keidar, M.; Canady, J. The Granger Causal Effects of Canady Helios Cold Plasma on the Inhibition of Breast Cancer Cell Proliferation. Appl. Sci. 2022, 12, 4622. [Google Scholar] [CrossRef]
- Jones, O.; Cheng, X.; Murthy, S.R.K.; Ly, L.; Zhuang, T.; Basadonna, G.; Keidar, M.; Canady, J. The Synergistic Effect of Canady Helios Cold Atmospheric Plasma and a FOLFIRINOX Regimen for the Treatment of Cholangiocarcinoma In Vitro. Sci. Rep. 2021, 11, 8967. [Google Scholar] [CrossRef]
- Murthy, S.R.K.; Cheng, X.; Zhuang, T.; Ly, L.; Jones, O.; Basadonna, G.; Keidar, M.; Canady, J. BCL2A1 regulates Canady Helios Cold Plasma-induced cell death in triple-negative breast cancer. Sci. Rep. 2022, 12, 4038. [Google Scholar] [CrossRef]
- Schwartz, L.H.; Litiere, S.; de Vries, E.; Ford, R.; Gwyther, S.; Mandrekar, S.; Shankar, L.; Bogaerts, J.; Chen, A.; Dancey, J.; et al. RECIST 1.1-Update and clarification: From the RECIST committee. Eur. J. Cancer 2016, 62, 132–137. [Google Scholar] [CrossRef] [PubMed]
- Badgwell, B.; Krouse, R.; Cormier, J.; Guevara, C.; Klimberg, V.S.; Ferrell, B. Frequent and early death limits quality of life assessment in patients with advanced malignancies evaluated for palliative surgical intervention. Ann. Surg. Oncol. 2012, 19, 3651–3658. [Google Scholar] [CrossRef] [PubMed]
- Neumann, E.; Rosenheck, K. Permeability changes induced by electric impulses in vesicular membranes. J. Membr. Biol. 1972, 10, 279–290. [Google Scholar] [CrossRef] [PubMed]
- Escoffre, J.M.; Portet, T.; Wasungu, L.; Teissié, J.; Dean, D.; Rols, M.P. What is (still not) known of the mechanism by which electroporation mediates gene transfer and expression in cells and tissues. Mol. Biotechnol. 2009, 41, 286–295. [Google Scholar] [CrossRef]
- Teissie, J.; Golzio, M.; Rols, M.P. Mechanisms of cell membrane electropermeabilization: A minireview of our present (lack of ?) knowledge. Biochim. Biophys. Acta 2005, 1724, 270–280. [Google Scholar] [CrossRef]
- Volotskova, O.; Hawley, T.S.; Stepp, M.A.; Keidar, M. Targeting the cancer cell cycle by cold atmospheric plasma. Sci. Rep. 2012, 2, 636. [Google Scholar] [CrossRef]
- Semmler, M.L.; Bekeschus, S.; Schafer, M.; Bernhardt, T.; Fischer, T.; Witzke, K.; Seebauer, C.; Rebl, H.; Grambow, E.; Vollmar, B.; et al. Molecular Mechanisms of the Efficacy of Cold Atmospheric Pressure Plasma (CAP) in Cancer Treatment. Cancers 2020, 12, 269. [Google Scholar] [CrossRef]
- Mizuno, K.; Yonetamari, K.; Shirakawa, Y.; Akiyama, T.; Ono, R. Anti-tumor immune response induced by nanosecond pulsed streamer discharge in mice. J. Phys. D Appl. Phys. 2017, 50, 12LT01. [Google Scholar] [CrossRef]

| Patient ID (Sex, Age) | Post-Operative Diagnosis | R0 | Post Operation ECOG ≤ 2 ASA ≤ 3 | LOS (Days) | OS Post Surgical Resection | Adverse Events with CHCP | Preoperative Staging Stage IV (Metastatic or Recurrent) | Intra- and Post-Operative Outcomes | Neo-Adjuvant Chemo-Therapy | Intra-Operative Chemo-Therapy | Adjuvant Chemo-Therapy |
|---|---|---|---|---|---|---|---|---|---|---|---|
| S0001 (F, 49) | Metastatic recurrent colon cancer | Yes | Yes | 7 | Died of disease (32 months) Lost follow-up (26 months) | None | Yes | No complications | CT | HIPEC, IORT | CT |
| S0002 (F, 44) | Metastatic recurrent ovarian cancer | No | Yes | 5 | Died of disease (29 months) | None | Yes | No complications | CT | None | CT |
| S0003 (F, 61) | Metastatic anal cancer to the liver | No | Yes | 5 | Died of disease (3 months) | None | Yes | No complications | CT | None | IMT |
| S0004 (M, 51) | Recurrent epithelioid peritoneal Mesothelioma | No | Yes | 1 | Died of disease (10 months) | None | Yes | No complications | CT | HIPEC | None |
| S0005 (M, 60) | Recurrent Colon Cancer | No | Yes | 6 | Died of disease (31 months) | None | Yes | No complications | CT | HIPEC | CT |
| R0002 (F, 85) | Metastatic myxofibrosarcoma left chest wall | Yes | Yes | 1 | Alive with disease (30 months) Recurrence of disease (20 months) at left chest wall resection site | None | Yes | No complications | RT, RT (internal), CT | None | RT |
| R0003 (F, 63) | Recurrent metastatic breast carcinoma to the right pelvis | No | Yes | 5 | Alive with disease (29 months) FDRS9 | None | Yes | No complications | RT, HT | None | RT, HT, IMT, CT |
| R0004 (F, 76) | Metastatic non-small cell lung carcinoma (NSCLC) to the left proximal femur | Yes | Yes | 4 | Died of disease (26 months) | None | Yes | No complications | None | None | RT, IMT |
| R0005 (M, 61) | Metastatic renal cell carcinoma to the left clavicle | Yes | Yes | 2 | Alive (28 months) FDRS | None | Yes | No complications | RT | None | IMT |
| R0007 (M, 59) | Metastatic cholangiocarcinoma to the left distal humerus | No | Yes | 3 | Died of disease (8 months) | None | Yes | No complications | None | None | CT, CRT |
| R0008 (M, 58) | Metastatic Non-small cell Lung Carcinoma (NSCLC) to the right hip and spine. | Yes | Yes | 7 | Died of disease (8 months) | None | Yes | No complications | CRT, IMT, RT, CT | None | CT |
| R0009 (F, 77) | Pleomorphic sarcoma of left distal femur | Yes | Yes | 2 | Alive (25 months) FDRS | None | Yes | No complications | None | None | RT |
| R0011 (M, 67) | Metastatic Chordoma to right gluteal | Yes | Yes | 11 | Died of disease (20 months) | None | Yes | No complications | RT | None | RT |
| R0010 (M, 53) | Metastatic melanoma to the left pelvis | No | Yes | 18 | Died of disease (4 months) | None | Yes | Intraoperative venous bleeding. Emergent angiogram IR embolization 1st re-admission to hospital for concern of active bleeding in the surgical area. Treatment embolization of left illiac artery. 2nd re-admission to hospital for continued surgical incision drainage observation. New bleed ruled out. Treatment with silvadene dressing changes. | IMT | None | None |
| R0012 (M, 41) | Metastatic Pleomorphic Spindle cell sarcoma | Yes | Yes | 1 | Alive (23 months) FDRS | None | Yes | No complications | CRT | None | RT, CT |
| R0013 (F, 55) | Squamous cell Carcinoma (SCC) (History of Kidney Transplant) | Yes | Yes | 2 | Alive with disease (23 months) Recurrence of disease (1 month) in the hand and axillary lymph node. Receiving anti-rejection immunosuppression therapy for kidney transplant. | None | Yes | No complications | IMT | None | CT |
| R0014 (F, 77) | Metastatic angiosarcoma to contralateral breast right Axilla | Yes | Yes | 1 | Alive (23 months) FDRS | None | Yes | No complications | RT, HT, CT | None | CT |
| R0016 (F, 50) | Metastatic Non-Small Cell Lung Carcinoma (NSCLC) to Bone | No | Yes | 2 | Alive (22 months) FDRS | None | Yes | No complications | Internal RT (radioactive iodine); RT, CRT, and CT/IMT for NSCLC. | None | RT/IMT |
| R0017 (M, 26) | Metastatic Desmoplastic Small Round Cell Sarcoma of left inguinal soft tissue and testicle | Yes | Yes | 1 | Alive (22 months) FDRS | None | Yes | No complications | CT | None | CRT |
| R0018 (M, 69) | Metastatic Adenoid Cystic Carcinoma of the left submandibular gland | Yes | Yes | 28 | Alive (21 months) FDRS | None | Yes | Salivary leak. Return to the OR for Repair of floor of the mouth defect with buccal fat flap | RT, CT | None | None |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Canady, J.; Murthy, S.R.K.; Zhuang, T.; Gitelis, S.; Nissan, A.; Ly, L.; Jones, O.Z.; Cheng, X.; Adileh, M.; Blank, A.T.; et al. The First Cold Atmospheric Plasma Phase I Clinical Trial for the Treatment of Advanced Solid Tumors: A Novel Treatment Arm for Cancer. Cancers 2023, 15, 3688. https://doi.org/10.3390/cancers15143688
Canady J, Murthy SRK, Zhuang T, Gitelis S, Nissan A, Ly L, Jones OZ, Cheng X, Adileh M, Blank AT, et al. The First Cold Atmospheric Plasma Phase I Clinical Trial for the Treatment of Advanced Solid Tumors: A Novel Treatment Arm for Cancer. Cancers. 2023; 15(14):3688. https://doi.org/10.3390/cancers15143688
Chicago/Turabian StyleCanady, Jerome, Saravana R. K. Murthy, Taisen Zhuang, Steven Gitelis, Aviram Nissan, Lawan Ly, Olivia Z. Jones, Xiaoqian Cheng, Mohammad Adileh, Alan T. Blank, and et al. 2023. "The First Cold Atmospheric Plasma Phase I Clinical Trial for the Treatment of Advanced Solid Tumors: A Novel Treatment Arm for Cancer" Cancers 15, no. 14: 3688. https://doi.org/10.3390/cancers15143688
APA StyleCanady, J., Murthy, S. R. K., Zhuang, T., Gitelis, S., Nissan, A., Ly, L., Jones, O. Z., Cheng, X., Adileh, M., Blank, A. T., Colman, M. W., Millikan, K., O’Donoghue, C., Stenson, K. M., Ohara, K., Schtrechman, G., Keidar, M., & Basadonna, G. (2023). The First Cold Atmospheric Plasma Phase I Clinical Trial for the Treatment of Advanced Solid Tumors: A Novel Treatment Arm for Cancer. Cancers, 15(14), 3688. https://doi.org/10.3390/cancers15143688








