Standardization of Body Composition Status in Patients with Advanced Urothelial Tumors: The Role of a CT-Based AI-Powered Software for the Assessment of Sarcopenia and Patient Outcome Correlation
Abstract
Simple Summary
Abstract
1. Introduction
2. Materials and Methods
2.1. Study Design
2.2. Image Acquisition and Analysis
2.3. Sarcopenia and Response to Therapy Definition
2.4. Statistical Analysis
3. Results
3.1. Demographic, Tumor- and Sarcopenia-Related Characteristics of the Study Population
3.2. Correlation between AI Skeletal Muscle Index (SMI-L3) and Anthropomorphic Sarcopenia-Related Variables Pre-/Post-Systemic Treatment
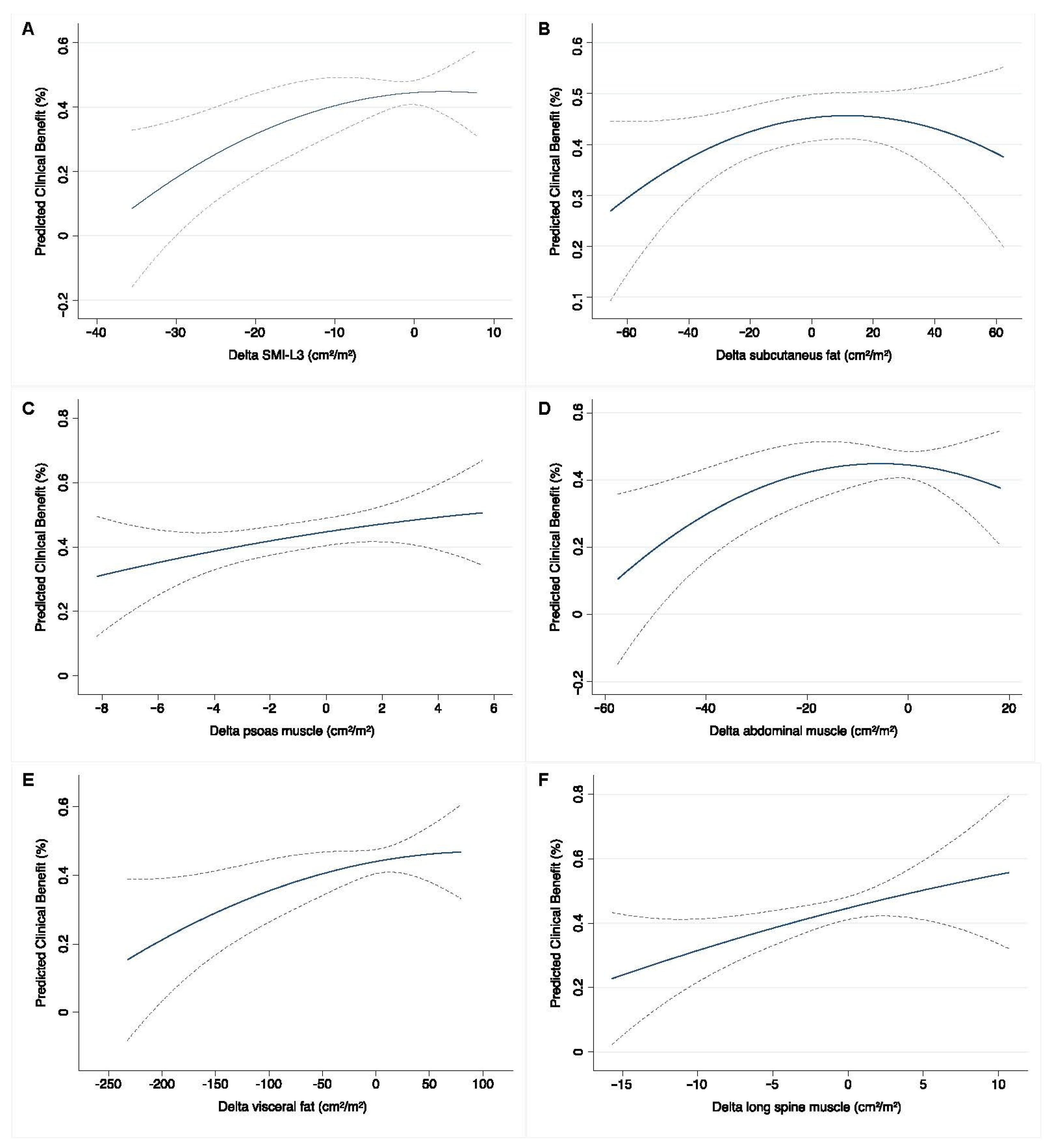
3.3. Baseline and Early Predictors of Clinical Benefit Measured at the Completion of Systemic Therapy
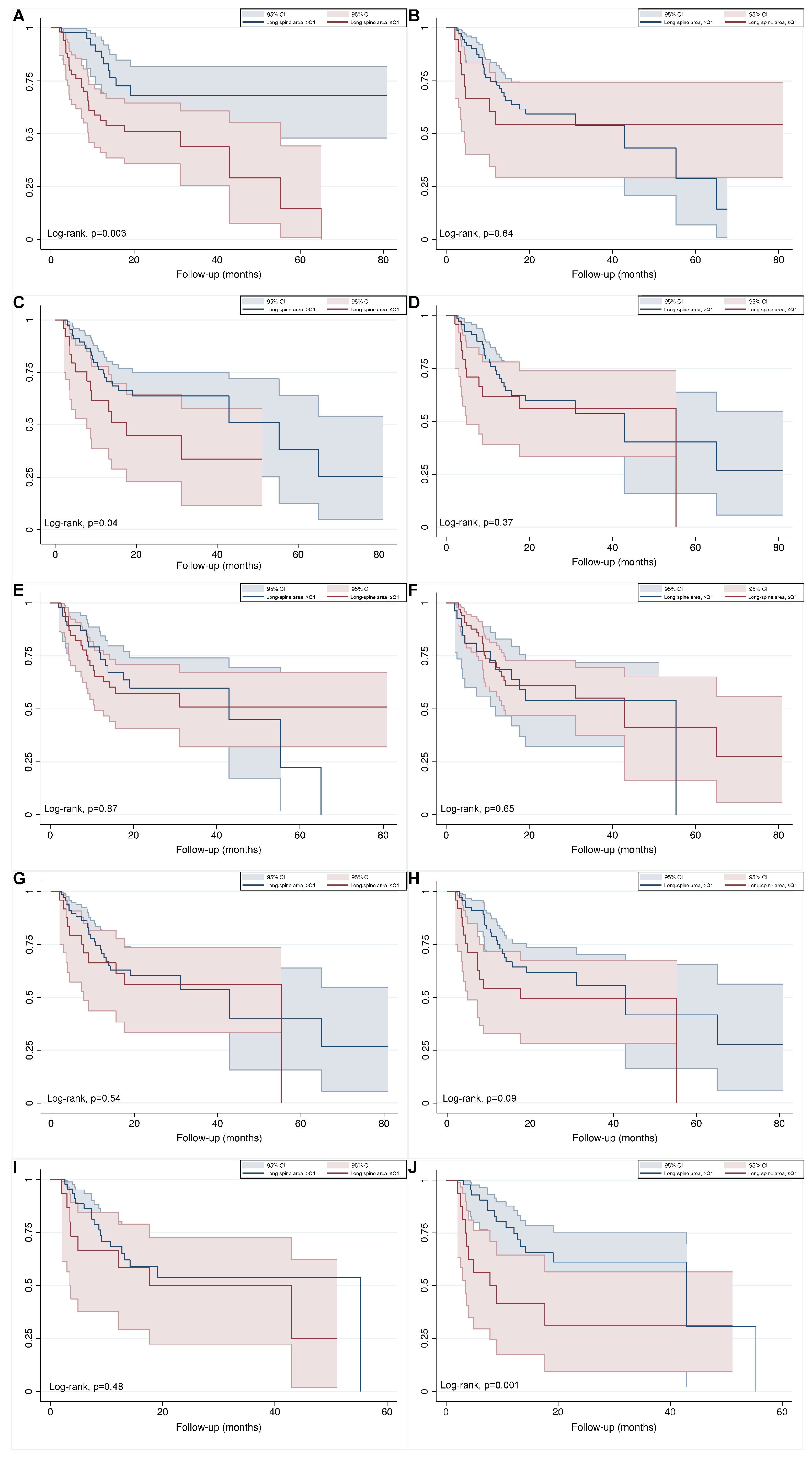
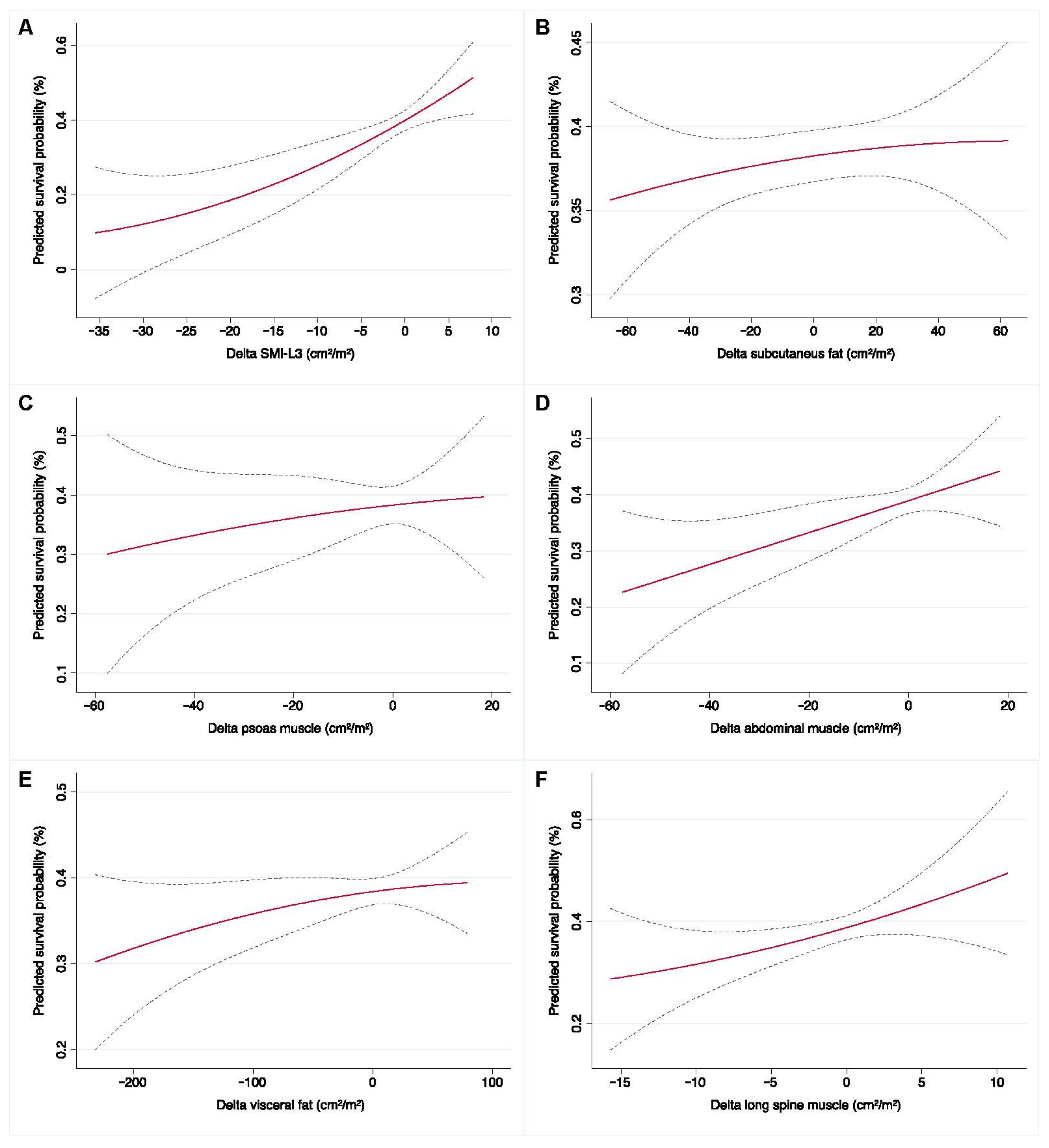
3.4. Baseline and Early Determinates for Overall Survival
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Muscaritoli, M.; Lucia, S.; Farcomeni, A.; Lorusso, V.; Saracino, V.; Barone, C.; Plastino, F.; Gori, S.; Magarotto, R.; Carteni, G.; et al. Prevalence of malnutrition in patients at first medical oncology visit: The PreMiO study. Oncotarget 2017, 8, 79884–79896. [Google Scholar] [CrossRef] [PubMed]
- Cederholm, T.; Jensen, G.L.; Correia, M.; Gonzalez, M.C.; Fukushima, R.; Higashiguchi, T.; Baptista, G.; Barazzoni, R.; Blaauw, R.; Coats, A.J.S.; et al. GLIM criteria for the diagnosis of malnutrition—A consensus report from the global clinical nutrition community. J. Cachexia Sarcopenia Muscle 2019, 10, 207–217. [Google Scholar] [CrossRef]
- Serinelli, S.; Panebianco, V.; Martino, M.; Battisti, S.; Rodacki, K.; Marinelli, E.; Zaccagna, F.; Semelka, R.C.; Tomei, E. Accuracy of MRI skeletal age estimation for subjects 12–19. Potential use for subjects of unknown age. Int. J. Legal Med. 2015, 129, 609–617. [Google Scholar] [CrossRef] [PubMed]
- Taguchi, S.; Nakagawa, T.; Uemura, Y.; Akamatsu, N.; Gonoi, W.; Naito, A.; Kawai, T.; Kume, H.; Fukuhara, H. Comparison of major definitions of sarcopenia based on the skeletal muscle index in patients with urothelial carcinoma. Future Oncol. 2021, 17, 197–203. [Google Scholar] [CrossRef] [PubMed]
- Chen, H.-W.; Chen, Y.-C.; Yang, L.-H.; Shih, M.-C.P.; Li, C.-C.; Chueh, K.-S.; Wu, W.-J.; Juan, Y.-S. Impact of cachexia on oncologic outcomes of sarcopenic patients with upper tract urothelial carcinoma after radical nephroureterectomy. PLoS ONE 2021, 16, e0250033. [Google Scholar] [CrossRef]
- Richenberg, J.; Løgager, V.; Panebianco, V.; Rouviere, O.; Villeirs, G.; Schoots, I.G. The primacy of multiparametric MRI in men with suspected prostate cancer. Eur. Radiol. 2019, 29, 6940–6952. [Google Scholar] [CrossRef]
- Ferro, M.; Babă, D.-F.; de Cobelli, O.; Musi, G.; Lucarelli, G.; Terracciano, D.; Porreca, A.; Busetto, G.M.; Del Giudice, F.; Soria, F.; et al. Neutrophil percentage-to-albumin ratio predicts mortality in bladder cancer patients treated with neoadjuvant chemotherapy followed by radical cystectomy. Future Sci. OA 2021, 7, FSO709. [Google Scholar] [CrossRef]
- Maggi, M.; Panebianco, V.; Mosca, A.; Salciccia, S.; Gentilucci, A.; Di Pierro, G.; Busetto, G.M.; Barchetti, G.; Campa, R.; Sperduti, I.; et al. Prostate Imaging Reporting and Data System 3 Category Cases at Multiparametric Magnetic Resonance for Prostate Cancer: A Systematic Review and Meta-analysis. Eur. Urol. Focus 2020, 6, 463–478. [Google Scholar] [CrossRef]
- Panebianco, V.; Sciarra, A.; Marcantonio, A.; Forte, V.; Biondi, T.; Laghi, A.; Catalano, C. Conventional imaging and multiparametric magnetic resonance (MRI, MRS, DWI, MRP) in the diagnosis of prostate cancer. Q. J. Nucl. Med. Mol. Imaging 2012, 56, 331–342. [Google Scholar]
- Sciarra, A.; Panebianco, V.; Ciccariello, M.; Salciccia, S.; Lisi, D.; Osimani, M.; Alfarone, A.; Gentilucci, A.; Parente, U.; Passariello, R.; et al. Magnetic Resonance Spectroscopic Imaging (1 H-MRSI) and Dynamic Contrast-Enhanced Magnetic Resonance (DCE-MRI): Pattern Changes from Inflammation to Prostate Cancer. Cancer Investig. 2010, 28, 424–432. [Google Scholar] [CrossRef]
- Pavone, P.; Laghi, A.; Panebianco, V.; Catalano, C.; Lobina, L.; Passariello, R. MR cholangiography: Techniques and clinical applications. Eur. Radiol. 1998, 8, 901–910. [Google Scholar] [CrossRef] [PubMed]
- Di Silverio, F.; Sciarra, A.; Parente, U.; Andrea, A.; Von Heland, M.; Panebianco, V.; Passariello, R. Neoadjuvant Therapy with Sorafenib in Advanced Renal Cell Carcinoma with Vena Cava Extension Submitted to Radical Nephrectomy. Urol. Int. 2008, 80, 451–453. [Google Scholar] [CrossRef] [PubMed]
- Sciarra, A.; Panebianco, V.; Cattarino, S.; Busetto, G.M.; De Berardinis, E.; Ciccariello, M.; Gentile, V.; Salciccia, S. Multiparametric magnetic resonance imaging of the prostate can improve the predictive value of the urinary prostate cancer antigen 3 test in patients with elevated prostate-specific antigen levels and a previous negative biopsy. BJU Int. 2012, 110, 1661–1665. [Google Scholar] [CrossRef] [PubMed]
- Mourtzakis, M.; Prado, C.M.; Lieffers, J.R.; Reiman, T.; McCargar, L.J.; Baracos, V.E. A practical and precise approach to quantification of body composition in cancer patients using computed tomography images acquired during routine care. Appl. Physiol. Nutr. Metab. 2008, 33, 997–1006. [Google Scholar] [CrossRef] [PubMed]
- Sánchez-Torralvo, F.J.; Ruiz-García, I.; Contreras-Bolívar, V.; González-Almendros, I.; Ruiz-Vico, M.; Abuín-Fernández, J.; Barrios, M.; Alba, E.; Olveira, G. CT-Determined Sarcopenia in GLIM-Defined Malnutrition and Prediction of 6-Month Mortality in Cancer Inpatients. Nutrients 2021, 13, 2647. [Google Scholar] [CrossRef] [PubMed]
- Bedrikovetski, S.; Seow, W.; Kroon, H.M.; Traeger, L.; Moore, J.W.; Sammour, T. Artificial intelligence for body composition and sarcopenia evaluation on computed tomography: A systematic review and meta-analysis. Eur. J. Radiol. 2022, 149, 110218. [Google Scholar] [CrossRef]
- Picchio, M.; Mapelli, P.; Panebianco, V.; Castellucci, P.; Incerti, E.; Briganti, A.; Gandaglia, G.; Kirienko, M.; Barchetti, F.; Nanni, C.; et al. Imaging biomarkers in prostate cancer: Role of PET/CT and MRI. Eur. J. Nucl. Med. Mol. Imaging 2015, 42, 644–655. [Google Scholar] [CrossRef]
- Ha, J.; Park, T.; Kim, H.-K.; Shin, Y.; Ko, Y.; Kim, D.W.; Sung, Y.S.; Lee, J.; Ham, S.J.; Khang, S.; et al. Development of a fully automatic deep learning system for L3 selection and body composition assessment on computed tomography. Sci. Rep. 2021, 11, 21656. [Google Scholar] [CrossRef]
- Fearon, K.; Strasser, F.; Anker, S.D.; Bosaeus, I.; Bruera, E.; Fainsinger, R.L.; Jatoi, A.; Loprinzi, C.; MacDonald, N.; Mantovani, G.; et al. Definition and classification of cancer cachexia: An international consensus. Lancet Oncol. 2011, 12, 489–495. [Google Scholar] [CrossRef]
- Eisenhauer, E.A.; Therasse, P.; Bogaerts, J.; Schwartz, L.H.; Sargent, D.; Ford, R.; Dancey, J.; Arbuck, S.; Gwyther, S.; Mooney, M.; et al. New response evaluation criteria in solid tumours: Revised RECIST guideline (version 1.1). Eur. J. Cancer 2009, 45, 228–247. [Google Scholar] [CrossRef]
- Delgado, A.; Guddati, A.K. Clinical endpoints in oncology—A primer. Am. J. Cancer Res. 2021, 11, 1121–1131. [Google Scholar]
- Salciccia, S.; Del Giudice, F.; Gentile, V.; Mastroianni, C.M.; Pasculli, P.; Di Lascio, G.; Ciardi, M.R.; Sperduti, I.; Maggi, M.; De Berardinis, E.; et al. Interplay between male testosterone levels and the risk for subsequent invasive respiratory assistance among COVID-19 patients at hospital admission. Endocrine 2020, 70, 206–210. [Google Scholar] [CrossRef] [PubMed]
- Busetto, G.M.; Giovannone, R.; Antonini, G.; Rossi, A.; Del Giudice, F.; Tricarico, S.; Ragonesi, G.; Gentile, V.; De Berardinis, E. Short-term pretreatment with a dual 5α-reductase inhibitor before bipolar transurethral resection of the prostate (B-TURP): Evaluation of prostate vascularity and decreased surgical blood loss in large prostates: Short-term treatment with dutasteride before B-TURP. BJU Int. 2015, 116, 117–123. [Google Scholar] [CrossRef] [PubMed]
- Giovannone, R.; Busetto, G.M.; Antonini, G.; De Cobelli, O.; Ferro, M.; Tricarico, S.; Del Giudice, F.; Ragonesi, G.; Conti, S.L.; Lucarelli, G.; et al. Hyperhomocysteinemia as an Early Predictor of Erectile Dysfunction: International Index of Erectile Function (IIEF) and Penile Doppler Ultrasound Correlation with Plasma Levels of Homocysteine. Medicine 2015, 94, e1556. [Google Scholar] [CrossRef]
- Caan, B.J.; Feliciano, E.M.C.; Prado, C.M.; Alexeeff, S.; Kroenke, C.H.; Bradshaw, P.; Quesenberry, C.P.; Weltzien, E.K.; Castillo, A.L.; Olobatuyi, T.A.; et al. Association of Muscle and Adiposity Measured by Computed Tomography with Survival in Patients with Nonmetastatic Breast Cancer. JAMA Oncol. 2018, 4, 798. [Google Scholar] [CrossRef] [PubMed]
- Del Giudice, F.; Leonardo, C.; Simone, G.; Pecoraro, M.; De Berardinis, E.; Cipollari, S.; Flammia, S.; Bicchetti, M.; Busetto, G.M.; Chung, B.I.; et al. Preoperative detection of Vesical Imaging-Reporting and Data System (VI-RADS) score 5 reliably identifies extravesical extension of urothelial carcinoma of the urinary bladder and predicts significant delayed time to cystectomy: Time to reconsider the nee: VI-RADS score 5 may avoid deep primary TURBT. BJU Int. 2020, 126, 610–619. [Google Scholar] [CrossRef]
- Fukushima, H.; Takemura, K.; Suzuki, H.; Koga, F. Impact of Sarcopenia as a Prognostic Biomarker of Bladder Cancer. Int. J. Mol. Sci. 2018, 19, 2999. [Google Scholar] [CrossRef]
- Huang, Y.-T.; Tsai, Y.-S.; Lin, P.-C.; Yeh, Y.-M.; Hsu, Y.-T.; Wu, P.-Y.; Shen, M.-R. The Value of Artificial Intelligence-Assisted Imaging in Identifying Diagnostic Markers of Sarcopenia in Patients with Cancer. Dis. Markers 2022, 2022, 1819841. [Google Scholar] [CrossRef]
- Jang, W.; Jeong, C.; Kwon, K.; Yoon, T.I.; Yi, O.; Kim, K.W.; Yang, S.-O.; Lee, J. Artificial intelligence for predicting five-year survival in stage IV metastatic breast cancer patients: A focus on sarcopenia and other host factors. Front. Physiol. 2022, 13, 977189. [Google Scholar] [CrossRef]
- Chung, H.; Ko, Y.; Lee, I.; Hur, H.; Huh, J.; Han, S.; Kim, K.W.; Lee, J. Prognostic artificial intelligence model to predict 5 year survival at 1 year after gastric cancer surgery based on nutrition and body morphometry. J. Cachexia Sarcopenia Muscle 2023, 14, 847–859. [Google Scholar] [CrossRef]
- Kim, S.I.; Chung, J.Y.; Paik, H.; Seol, A.; Yoon, S.H.; Kim, T.M.; Kim, H.S.; Chung, H.H.; Cho, J.Y.; Kim, J.-W.; et al. Prognostic role of computed tomography-based, artificial intelligence-driven waist skeletal muscle volume in uterine endometrial carcinoma. Insights Imaging 2021, 12, 192. [Google Scholar] [CrossRef] [PubMed]
- Han, Q.; Kim, S.I.; Yoon, S.H.; Kim, T.M.; Kang, H.-C.; Kim, H.J.; Cho, J.Y.; Kim, J.-W. Impact of Computed Tomography-Based, Artificial Intelligence-Driven Volumetric Sarcopenia on Survival Outcomes in Early Cervical Cancer. Front. Oncol. 2021, 11, 741071. [Google Scholar] [CrossRef] [PubMed]
- Stangl-Kremser, J.; Mari, A.; Lai, L.Y.; Lee, C.T.; Vince, R.; Zaslavsky, A.; Salami, S.S.; Fajkovic, H.; Shariat, S.F.; Palapattu, G.S. Sarcopenic Obesity and its Prognostic Impact on Urological Cancers: A Systematic Review. J. Urol. 2021, 206, 854–865. [Google Scholar] [CrossRef] [PubMed]
- Wu, E.; Hadjiiski, L.M.; Samala, R.K.; Chan, H.-P.; Cha, K.H.; Richter, C.; Cohan, R.H.; Caoili, E.M.; Paramagul, C.; Alva, A.; et al. Deep Learning Approach for Assessment of Bladder Cancer Treatment Response. Tomography 2019, 5, 201–208. [Google Scholar] [CrossRef]
- Roblot, V.; Giret, Y.; Mezghani, S.; Auclin, E.; Arnoux, A.; Oudard, S.; Duron, L.; Fournier, L. Validation of a deep learning segmentation algorithm to quantify the skeletal muscle index and sarcopenia in metastatic renal carcinoma. Eur. Radiol. 2022, 32, 4728–4737. [Google Scholar] [CrossRef]
- Cheung, H.; Wang, Y.; Chang, S.L.; Khandwala, Y.S.; Del Giudice, F.; Chung, B.I. Adoption of Robot-Assisted Partial Nephrectomies: A Population-Based Analysis of U.S. Surgeons from 2004 to 2013. J. Endourol. 2017, 31, 886–892. [Google Scholar] [CrossRef]
- Cruz-Jentoft, A.J.; Landi, F.; Schneider, S.M.; Zúñiga, C.; Arai, H.; Boirie, Y.; Chen, L.-K.; Fielding, R.A.; Martin, F.C.; Michel, J.-P.; et al. Prevalence of and interventions for sarcopenia in ageing adults: A systematic review. Report of the International Sarcopenia Initiative (EWGSOP and IWGS). Age Ageing 2014, 43, 748–759. [Google Scholar] [CrossRef]
- Papadopoulou, S.K. Sarcopenia: A Contemporary Health Problem among Older Adult Populations. Nutrients 2020, 12, 1293. [Google Scholar] [CrossRef]
- Pacifico, J.; Geerlings, M.A.; Reijnierse, E.M.; Phassouliotis, C.; Lim, W.K.; Maier, A.B. Prevalence of sarcopenia as a comorbid disease: A systematic review and meta-analysis. Exp. Gerontol. 2020, 131, 110801. [Google Scholar] [CrossRef]
- Zhang, X.; Zhao, Y.; Chen, S.; Shao, H. Anti-diabetic drugs and sarcopenia: Emerging links, mechanistic insights, and clinical implications. J. Cachexia Sarcopenia Muscle 2021, 12, 1368–1379. [Google Scholar] [CrossRef]
- Campins, L.; Camps, M.; Riera, A.; Pleguezuelos, E.; Yébenes, J.C.; Serra-Prat, M. Oral Drugs Related with Muscle Wasting and Sarcopenia. A Review. Pharmacology 2017, 99, 1–8. [Google Scholar] [CrossRef] [PubMed]
- Yumioka, T.; Honda, M.; Nishikawa, R.; Teraoka, S.; Kimura, Y.; Iwamoto, H.; Morizane, S.; Hikita, K.; Takenaka, A. Sarcopenia as a significant predictive factor of neutropenia and overall survival in urothelial carcinoma patients underwent gemcitabine and cisplatin or carboplatin. Int. J. Clin. Oncol. 2020, 25, 158–164. [Google Scholar] [CrossRef]
- Shimizu, R.; Honda, M.; Teraoka, S.; Yumioka, T.; Yamaguchi, N.; Kawamoto, B.; Iwamoto, H.; Morizane, S.; Hikita, K.; Takenaka, A. Sarcopenia is associated with survival in patients with urothelial carcinoma treated with systemic chemotherapy. Int. J. Clin. Oncol. 2022, 27, 175–183. [Google Scholar] [CrossRef] [PubMed]
- Nicolazzo, C.; Busetto, G.M.; Del Giudice, F.; Sperduti, I.; Giannarelli, D.; Gradilone, A.; Gazzaniga, P.; de Berardinis, E.; Raimondi, C. The long-term prognostic value of survivin expressing circulating tumor cells in patients with high-risk non-muscle invasive bladder cancer (NMIBC). J. Cancer Res. Clin. Oncol. 2017, 143, 1971–1976. [Google Scholar] [CrossRef]
- Del Giudice, F.; Pecoraro, M.; Vargas, H.A.; Cipollari, S.; De Berardinis, E.; Bicchetti, M.; Chung, B.I.; Catalano, C.; Narumi, Y.; Catto, J.W.F.; et al. Systematic Review and Meta-Analysis of Vesical Imaging-Reporting and Data System (VI-RADS) Inter-Observer Reliability: An Added Value for Muscle Invasive Bladder Cancer Detection. Cancers 2020, 12, 2994. [Google Scholar] [CrossRef] [PubMed]
- Panebianco, V.; Pecoraro, M.; Del Giudice, F.; Takeuchi, M.; Muglia, V.F.; Messina, E.; Cipollari, S.; Giannarini, G.; Catalano, C.; Narumi, Y. VI-RADS for Bladder Cancer: Current Applications and Future Developments. J. Magn. Reson. Imaging 2022, 55, 23–36. [Google Scholar] [CrossRef] [PubMed]
- Basile, D.; Corvaja, C.; Caccialanza, R.; Aprile, G. Sarcopenia: Looking to muscle mass to better manage pancreatic cancer patients. Curr. Opin. Support. Palliat. Care 2019, 13, 279–285. [Google Scholar] [CrossRef]
- Kurk, S.A.; Peeters, P.H.M.; Dorresteijn, B.; De Jong, P.A.; Jourdan, M.; Creemers, G.-J.M.; Erdkamp, F.L.G.; De Jongh, F.E.; Kint, P.A.M.; Poppema, B.J.; et al. Loss of skeletal muscle index and survival in patients with metastatic colorectal cancer: Secondary analysis of the phase 3 CAIRO3 trial. Cancer Med. 2020, 9, 1033–1043. [Google Scholar] [CrossRef]
- Panebianco, V.; Grazhdani, H.; Iafrate, F.; Petroni, M.; Anzidei, M.; Laghi, A.; Passariello, R. 3D CT protocol in the assessment of the esophageal neoplastic lesions: Can it improve TNM staging? Eur. Radiol. 2006, 16, 414–421. [Google Scholar] [CrossRef]
- Iwase, T.; Wang, X.; Shrimanker, T.V.; Kolonin, M.G.; Ueno, N.T. Body composition and breast cancer risk and treatment: Mechanisms and impact. Breast Cancer Res. Treat. 2021, 186, 273–283. [Google Scholar] [CrossRef]
- Ubachs, J.; Ziemons, J.; Minis-Rutten, I.J.G.; Kruitwagen, R.F.P.M.; Kleijnen, J.; Lambrechts, S.; Olde Damink, S.W.M.; Rensen, S.S.; Van Gorp, T. Sarcopenia and ovarian cancer survival: A systematic review and meta-analysis. J. Cachexia Sarcopenia Muscle 2019, 10, 1165–1174. [Google Scholar] [CrossRef]
- Jung, J.; Lee, E.; Shim, H.; Park, J.-H.; Eom, H.-S.; Lee, H. Prediction of clinical outcomes through assessment of sarcopenia and adipopenia using computed tomography in adult patients with acute myeloid leukemia. Int. J. Hematol. 2021, 114, 44–52. [Google Scholar] [CrossRef]
- Cecchini, S.; Cavazzini, E.; Marchesi, F.; Sarli, L.; Roncoroni, L. Computed Tomography Volumetric Fat Parameters versus Body Mass Index for Predicting Short-term Outcomes of Colon Surgery. World J. Surg. 2011, 35, 415–423. [Google Scholar] [CrossRef]
- Wang, B.; Torriani, M. Artificial Intelligence in the Evaluation of Body Composition. Semin. Musculoskelet. Radiol. 2020, 24, 030–037. [Google Scholar] [CrossRef]
- Chung, H.; Jo, Y.; Ryu, D.; Jeong, C.; Choe, S.; Lee, J. Artificial-intelligence-driven discovery of prognostic biomarker for sarcopenia. J. Cachexia Sarcopenia Muscle 2021, 12, 2220–2230. [Google Scholar] [CrossRef] [PubMed]
- Bhinder, B.; Gilvary, C.; Madhukar, N.S.; Elemento, O. Artificial Intelligence in Cancer Research and Precision Medicine. Cancer Discov. 2021, 11, 900–915. [Google Scholar] [CrossRef] [PubMed]
- Somasundaram, E.; Castiglione, J.A.; Brady, S.L.; Trout, A.T. Defining Normal Ranges of Skeletal Muscle Area and Skeletal Muscle Index in Children on CT Using an Automated Deep Learning Pipeline: Implications for Sarcopenia Diagnosis. Am. J. Roentgenol. 2022, 219, 326–336. [Google Scholar] [CrossRef] [PubMed]
- Borrelli, P.; Kaboteh, R.; Enqvist, O.; Ulén, J.; Trägårdh, E.; Kjölhede, H.; Edenbrandt, L. Artificial intelligence-aided CT segmentation for body composition analysis: A validation study. Eur. Radiol. Exp. 2021, 5, 11. [Google Scholar] [CrossRef] [PubMed]
- Ferro, M.; de Cobelli, O.; Musi, G.; del Giudice, F.; Carrieri, G.; Busetto, G.M.; Falagario, U.G.; Sciarra, A.; Maggi, M.; Crocetto, F.; et al. Radiomics in prostate cancer: An up-to-date review. Ther. Adv. Urol. 2022, 14, 175628722211090. [Google Scholar] [CrossRef] [PubMed]
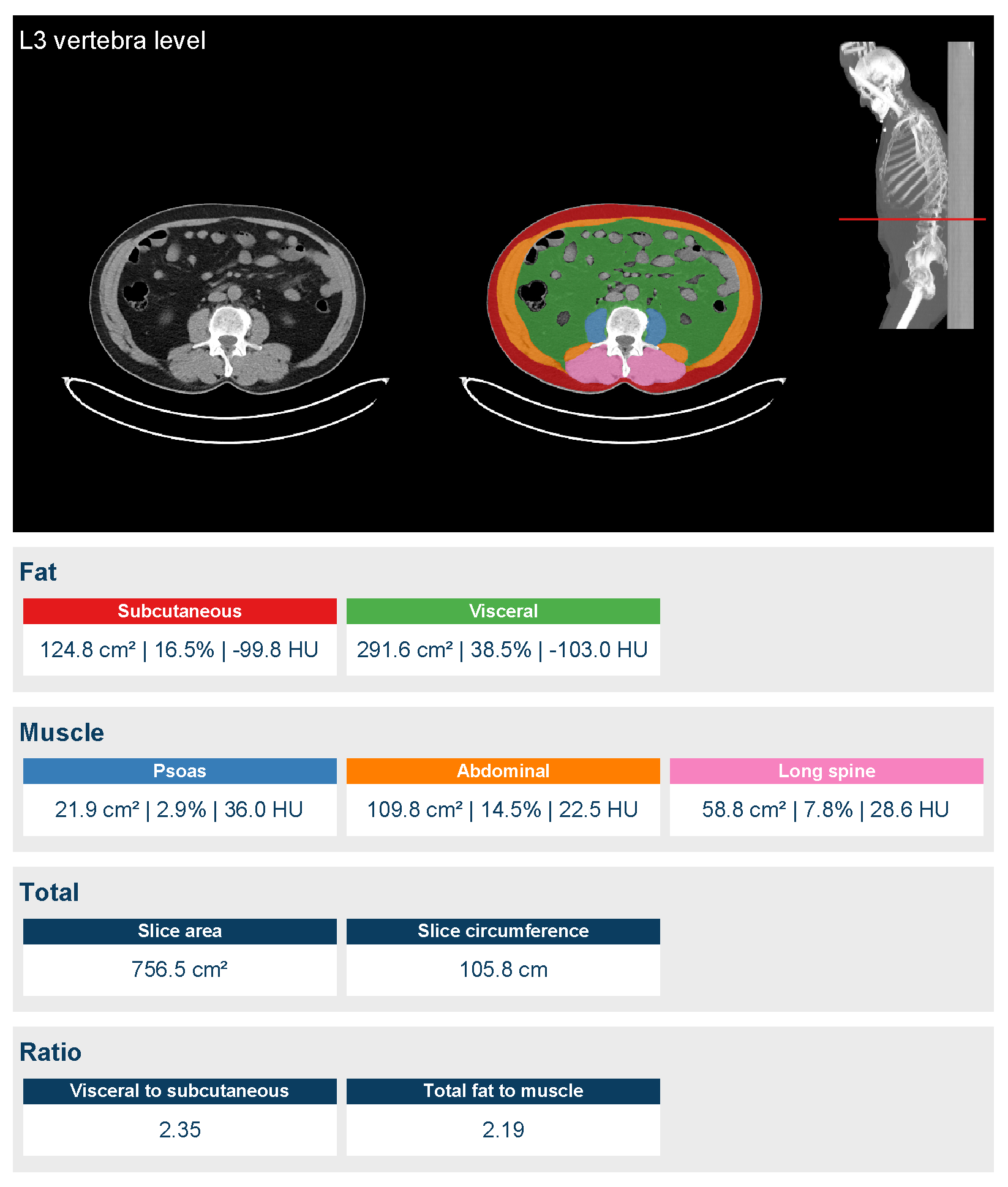

| Variables | No Sarcopenic Status, Baseline (By SMI-L3 Cut-Off) | Sarcopenic Status, Baseline (By SMI-L3 Cut-Off) | p-Value * | No SARCOPENIC Status, after CHT (By SMI-L3 Cut-Off) | Sarcopenic Status, after CHT (By SMI-L3 Cut-Off) | p-Value * |
|---|---|---|---|---|---|---|
| Sample size, n (%) | 46 (47.4) | 51 (52.6) | 45 (46.4) | 52 (53.6) | ||
| Demographics and tumor-related features | ||||||
| Age y, median (IQR) | 73 (64–76) | 69 (64–74) | 0.414 | 73 (66–76) | 68 (64–74) | 0.126 |
| Age y, n (%) | ||||||
| <70 y | 19 (41.3) | 30 (58.8) | 0.105 | 17 (37.8) | 32 (61.5) | 0.025 |
| ≥70 y | 27 (58.7) | 21 (42.2) | 28 (62.2) | 20 (38.5) | ||
| Gender, n (%) | ||||||
| Male | 36 (78.3) | 34 (66.7) | 0.259 | 11 (24.4) | 16 (30.8) | 0.506 |
| Female | 10 (27.7) | 17 (33.3) | 34 (75.6) | 36 (69.2) | ||
| ECOG PS, n (%) | ||||||
| <2 | 39 (84.8) | 40 (78.4) | 0.447 | 38 (84.4) | 41 (78.8) | 0.603 |
| ≥2 | 7 (15.2) | 11 (21.6) | 7 (15.6) | 11 (21.2) | ||
| n. of Medications, n (%) | ||||||
| <6 | 36 (78.3) | 42 (82.4) | 0.620 | 34 (75.6) | 44 (84.6) | 0.311 |
| ≥6 | 10 (21.7) | 9 (17.6) | 11 (24.4) | 8 (15.4) | ||
| Primary location n (%) | ||||||
| BCa | 24 (52.2) | 37 (72.5) | 0.049 | 24 (53.3) | 37 (71.2) | 0.096 |
| UTUC | 21 (45.7) | 12 (23.5) | 20 (44.4) | 13 (25.0) | ||
| Concomitant | 1 (2.2) | 2 (3.9) | 1 (2.2) | 2 (3.8) | ||
| Oncologic stage, n (%) | ||||||
| III | 22 (47.8) | 17 (33.3) | 0.155 | 22 (48.9) | 17 (32.7) | 0.146 |
| IV | 24 (52.2) | 34 (66.7) | 23 (51.1) | 35 (67.3) | ||
| Anthropometric measures | ||||||
| Height, m | 1.70 (1.66–1.75) | 1.70 (1.62–1.75) | 0.753 | 1.70 (1.64–1.73) | 1.70 (1.64–1.75) | 0.677 |
| Weight, kg | 77 (70–85.25) | 70 (60–75) | 0.001 | 77 (70–80.25) | 70 (60–80) | 0.005 |
| BMI, kg/m2 | 26.3 (24.85–27.75) | 24.2 (21.9–26.5) | 0.001 | 26.3 (25.3–29.3) | 23.5 (21.7–26.4) | 0.007 |
| SMA, cm2 | 179.7 (167.8–194) | 135.8 (114.8–155.2) | <0.0001 | 173.1 (157.2–193) | 133.7 (116–154.4) | <0.0001 |
| SMI-L3, (cm2/m2) | 62 (57.8–67.1) | 48.6 (43.1–53) | <0.0001 | 59.9 (57.3–63.8) | 47.9 (43.4–50.9) | <0.0001 |
| Subcutaneous fat, (cm2/m2) | 184.2 (133.6–221.8) | 135.2 (108.4–178.6) | 0.003 | 178.7 (130.7–215.1) | 143.1 (108.4–183.2) | 0.002 |
| Visceral fat, (cm2/m2) | 207 (153.7–255.7) | 103 (67.9–175.7) | <0.0001 | 182.6 (140–218.4) | 137 (64.5–181.6) | <0.0001 |
| Psoas muscle, (cm2/m2) | 21.7 (19.4–24.6) | 16.5 (13.3–20) | <0.0001 | 20.8 (18–22.8) | 16.1 (13.5–19) | <0.0001 |
| Abdominal muscle, (cm2/m2) | 100.6 (89.6–109.2) | 71.5 (59.1–82.2) | <0.0001 | 93.4 (86.6–106.9) | 71.6 (59.7–80.9) | <0.0001 |
| Long spine muscle, (cm2/m2) | 60.3 (54.8–64.1) | 45.5 (40.8–55) | <0.0001 | 57.9 (51.4–62.8) | 46.4 (40.7–55.2) | <0.0001 |
| ∆_SMI-L3, mean (SD) | −1.86 (5.78) | |||||
| ∆_Subcutaneous fat mean (SD) | 0.25 (24.93) | |||||
| ∆_Visceral fat, mean (SD) | −4.93 (42.17) | |||||
| ∆_ Psoas muscle, mean (SD) | −0.85 (2.56) | |||||
| ∆_ Abdominal muscle, mean (SD) | −3.02 (10.74) | |||||
| ∆_ Long spine muscle, mean (SD) | −1.06 (3.39) | |||||
| Clinical outcomes | ||||||
| Clinical Benefit, n (%) | ||||||
| SD/PR/CR | 30 (65.2) | 25 (49) | 0.151 | 29 (64.4) | 26 (50.0) | 0.217 |
| PD | 16 (34.8) | 26 (51) | 16 (35.6) | 26 (50.0) | ||
| Survival n (%) | ||||||
| Deceased | 37 (80.4) | 23 (45.1) | 0.001 | 35 (77.8) | 25 (48.1) | 0.003 |
| Survivors | 9 (19.6) | 28 (54.9) | 10 (22.2) | 27 (51.9) | ||
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Borrelli, A.; Pecoraro, M.; Del Giudice, F.; Cristofani, L.; Messina, E.; Dehghanpour, A.; Landini, N.; Roberto, M.; Perotti, S.; Muscaritoli, M.; et al. Standardization of Body Composition Status in Patients with Advanced Urothelial Tumors: The Role of a CT-Based AI-Powered Software for the Assessment of Sarcopenia and Patient Outcome Correlation. Cancers 2023, 15, 2968. https://doi.org/10.3390/cancers15112968
Borrelli A, Pecoraro M, Del Giudice F, Cristofani L, Messina E, Dehghanpour A, Landini N, Roberto M, Perotti S, Muscaritoli M, et al. Standardization of Body Composition Status in Patients with Advanced Urothelial Tumors: The Role of a CT-Based AI-Powered Software for the Assessment of Sarcopenia and Patient Outcome Correlation. Cancers. 2023; 15(11):2968. https://doi.org/10.3390/cancers15112968
Chicago/Turabian StyleBorrelli, Antonella, Martina Pecoraro, Francesco Del Giudice, Leonardo Cristofani, Emanuele Messina, Ailin Dehghanpour, Nicholas Landini, Michela Roberto, Stefano Perotti, Maurizio Muscaritoli, and et al. 2023. "Standardization of Body Composition Status in Patients with Advanced Urothelial Tumors: The Role of a CT-Based AI-Powered Software for the Assessment of Sarcopenia and Patient Outcome Correlation" Cancers 15, no. 11: 2968. https://doi.org/10.3390/cancers15112968
APA StyleBorrelli, A., Pecoraro, M., Del Giudice, F., Cristofani, L., Messina, E., Dehghanpour, A., Landini, N., Roberto, M., Perotti, S., Muscaritoli, M., Santini, D., Catalano, C., & Panebianco, V. (2023). Standardization of Body Composition Status in Patients with Advanced Urothelial Tumors: The Role of a CT-Based AI-Powered Software for the Assessment of Sarcopenia and Patient Outcome Correlation. Cancers, 15(11), 2968. https://doi.org/10.3390/cancers15112968









