Antitumor Properties of Curcumin in Breast Cancer Based on Preclinical Studies: A Systematic Review
Abstract
:Simple Summary
Abstract
1. Introduction
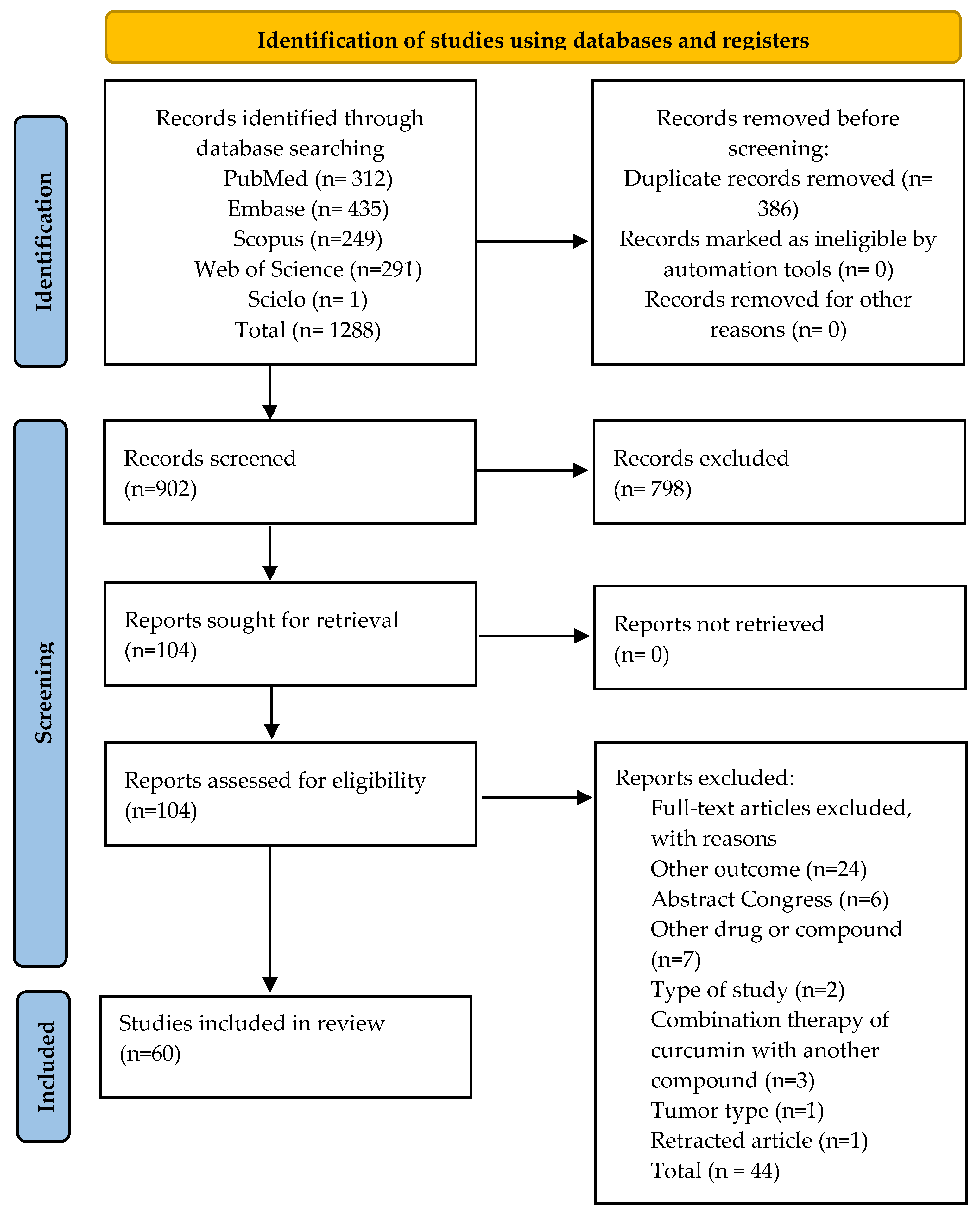
2. Materials and Methods
2.1. Protocol and Registration
2.2. Search Strategy and Eligibility Criteria
- Patient: laboratory animals with induced breast cancer (all species).
- Intervention(s): curcumin
- Comparator(s): control group or comparison with no treatment, treatment with other drugs, and/or traditional radiotherapy or chemotherapy regimens.
- Outcomes: antitumor activity (reduction in tumor volume and dimensions) in in vitro studies.
Definitions
2.3. Review Process
2.4. Training of Reviewers
2.5. Evidence Synthesis
3. Results
3.1. General Characteristics of the Studies
3.2. Summary of the Results
3.2.1. In Vitro Studies
3.2.2. In Vitro Cell Proliferation
| Author/Year/Country | Type of Cell/Model | Intervention | Outcomes | Conflict of Interest | |||
|---|---|---|---|---|---|---|---|
| Antitumor Activity | |||||||
| Concentration (Component) | Treatment Duration | Cell Proliferation In Vitro | Cell Viability | Apoptosis and/or Cell Cycle Interruption | |||
| Abbaspour and Afshar, 2018 [27] Iran | MCF-7 Human | Curcumin at 10, 20 and 30 μg/mL | 24, 48, and 72 h | MTT assay ↓ cell proliferation owing to downregulation of ODC1 and ADA gene expression. | MTT assay ↓ viability of cells in a time- and dose-dependent manner. | Not reported | None |
| Abuelba et al., 2015 [28] Romania | MDA-MB-231 Human | Curcumin at 15–19 μM | 24, 48, and 72 h | MTT assay ↓ cell proliferation upon treatment with 15 μM curcumin. | MTT assay ↓ cell viability by up to 25% upon treatment with 15 μM curcumin. | MTT assay Pro-apoptotic effects on MDA-MB-231 cells cultured in a single layer, without photoactivation. | None |
| Bimonte et al., 2015 [7] Italy | MDA.MB231 Human | Curcumin at 10 and 50 μM | 48 h | MTT assay Inhibition of breast cancer cell migration in 48 h. ↓ cell proliferation (p < 0.05). | Not reported | Flow cytometry Curcumin (10 μM) ↑ apoptosis (p < 0.0001). | None |
| Calaf et al., 2018 [31] Chile | MCF7 MDA-MB-231 Human | Curcumin at 30 µM | 48 h | Not reported | Not reported | Flow cytometry Apoptosis MDA-MB-231: 14.2% MCF7: 4.6% | None |
| Chiu and Su, 2009 [33] China | MDA-MB-231 Human | Curcumin at 10, 20, and 30 μg/mL | 48 h | MTT Assay ↓ proliferation of MDA-MB-231 cells via p21 expression. | Not reported | Flow cytometry Curcumin induced apoptosis via positive regulation of the Bax:Bcl-2 ratio. | None |
| Choudhuri et al., 2002 [34] India | MCF-7 Human | Curcumin at 10 and 25 μM | 24 h | Quantitative image analysis Cessation of cell growth followed by significant cell death. Optimal inhibition was obtained upon treatment with 25 μM curcumin. | Not reported | Quantitative image analysis techniques Curcumin induced apoptosis. | None |
| Coker-Gurkan et al., 2019 [35] Turkey | T47D Human | Curcumin at 30 µM | 24 and 48 h | Not reported | MTT assay ↓ cell viability by 48% and 60% upon treatment with 20 µM curcumin (p < 0.0024). | Double staining with Annexin-V/PI Curcumin induced apoptosis in 10.9% and 5.2% of the cell populations. | None |
| Coker-Gurkan et al., 2018 [36] Turkey | MCF-7 MDA-MB-231 Human | Curcumin at 30 µM | 24 and 48 h | Not reported | MTT assay ↓ cell viability MCF-7 cells by 49% and of MDA-MB-453 cells by 48% upon treatment for 24 h with 20 µM curcumin | MTT assay Curcumin induced apoptotic cell death. | None |
| Fan et al., 2016 [37] China | MDA-MB-231 Human | Curcumin at 50 μg/mL | 24 h | Not reported | MTT assay ↓ cell viability (% NR) (P:NR) | MTT assay Curcumin induced apoptosis. | None |
| Ghosh et al., 2021 [39] India | MDA-MB 231 Human | Curcumin at 50 μg/mL Nanostructured platform Nanoparticles, MSN-Curcumin (MSN-C), and MSN-Hyaluronic acid-Curcumin (MSN-HA-C) | 48 h | MTT Assay MSN-HA-C blocked cell proliferation, in contrast to free curcumin. The treatment agent exhibited anticancer properties at 20 μg/mL. | Not reported | MTT assay Cell death MSN-HA-C: 58% MSN-C: 34% (with equivalent dose of 12 μg/mL curcumin). MDA-MB-231 cycle arrest ↓ G1-phase cells: 32.5% Control: 54.6% G2/M phase cells: 37.8% Controls: 11.4%. | None |
| Hashemzehi et al., 2018 [42] Iran | MCF-7 Human | Curcumin at 1 mM Nanostructured platform Nano-curcumin—phytosomalcurcumin | 24 h | Transwell assay ↓ cell invasion MTT assay ↓ cell growth in a dose-dependent manner. | Not reported | Not reported | None |
| He et al., 2019 [43] China | 4T1 Mouse | Curcumin at 50 μg/mL Nanostructured platform Polymeric micellar NPs [amphiphilic diblock copolymer—mPEG-b-PLG (Se) -TP] | 48 h | Not reported | MTT assay ↓ of cell viability upon treatment with CUR-NP and Free CUR: 15% | Not reported | None |
| Hu et al., 2018 [44] China | T47D, MCF7 Human | Curcumin at 10 or 30 µM | 72 h | MTT assay ↓ cell proliferation | Not reported | Flow cytometry Apoptosis T47D cells: 13.87% and 30.09%. MCF7 cells: 15.14% and 35.04%. | None |
| Hua et al., 2010 [45] China | MDA-MB-435 Human | Curcumin at 10, 25, 50, and 75 μM | 12, 24, or 48 h. | MTT assay ↓ cell proliferation, inducing arrest in the G1 phase. | Not reported | Not reported | NR |
| Ji et al., 2020 [47] China | MDA-MB-231 Human | Curcumin at 50 μg/mL | 24 h | Not reported | Not reported | Flow cytometry Apoptosis HA@CUR-NCs 80%. | None |
| Jiang et al., 2013 [48] China | MCF-7/LCC2 and LCC9 Human | Curcumin at 10 and 30 μM | 24, 48, 72, and 96 h | Colony formation assay ↓ colony formation Complete suppression of colony formation upon treatment with 30 μM curcumin. | Not reported | Annexin-V/PI staining and flow cytometry 30 μM curcumin caused a significant increase (28.72% in MCF-7 cells, 31.36% in MCF-7/LCC2 cells, and 34.70% in MCF-7/LCC9 cells) in the percentage of late apoptotic cells. | None |
| Jin et al., 2017 [49] China and USA | MCF-7 Human | Curcumin at 10 µg/mL Nanostructured platform CUR-NP; GE11-CUR-NP; Free CUR | 24 h | Not reported | Nanostructured platform CUR-NP, GE11-CUR-NP, and Free CUR | Flow cytometry Apoptosis CUR-NP: 14.9%; GE11-CUR-NP: 18.9%; Free CUR 11.0%. | None |
| Jung et al., 2018 [50] South Korea | MDA-MB-468 Human | Curcumin at 5 and 10 μM | 72 and 96 h | Colony formation assay ↓ number of colonies over 2 weeks to 36.9 ± 7.7% upon treatment with 5 µM curcumin. | Unclear method ↓ significantly decreased cell viability (41.5 ± 2.8% of basal level) upon treatment with 10 µM curcumin | Not reported | None |
| Kim et al., 2012 [51] Coreia do Sul | MCF-7 Human | Curcumin at 1, 5, 10, 30, and 50 μM | 24 h | Not reported | MTT assay Curcumin exerted no effect on the viability of MCF-7 cells | Not reported | None |
| Kumari et al., 2017 [52] India | MDA-MB-231 Human | Curcumin at 50 and 100 μg/mL Nanostructured platform free CUR and CUR-mPEG-PLA-Ch micelles | 24 h | Not reported | MTT assay CUR: 55.26 ± 3.7% Free CUR: 66.84 ± 2.4% (p = 0.079) | Not reported | None |
| Kumari et al., 2020 [53] India | MDA-MB-231 Human 4T1 Mouse | Curcumin at 50 μg/mL Nanostructured platform CUR treatment (Free CUR group—24 μg/mL) and CUR-HSA-DOPE NPs treatment (CUR-HSA-DOPE group) | 6 and 24 h | Not reported | MTT Assay MDA-MB-231 Cur-HSA-DOPE NPs 24.34 ± 6.1% and 33.99 ± 4.5% free CUR 34.87 ± 4.9% and 43.12 ± 2.4% 50 μg/mL curcumin 4T1 CUR-HSA-DOPE NPs 25.2 ± 5.8% and 11.9 ± 8.6% free CUR 34.5 ± 6.6% and 48.3 ± 7.2% 50 μg of curcumin | Immunofluorescence TUNEL assay ↑ Apotosis CUR-HSA-DOPE NPs | None |
| Kumari et al., 2016 [54] India | MDA-MB-231 Human | Curcumin at 50 μg/mL Curcumin and curcumin-loaded nanoparticles (curcumin in mPEG-PLA micelles) (CUR-HSA-DOPE NPs) | 24 h | Not reported | MTT Assay CUR-mPEG-PLA231 35.1 ± 8.5 free CUR 65.7 ± 1.0% 50 μg/mL | Not reported | None |
| Laha et al., 2018 [55] India and USA | MDA-MB-468 Human | Curcumin at 20, 40, 60, 80, 100, and 120 mM | 12 and 24 h | Not reported | Not reported | Annexin V-FITC staining Apoptotic cells: 25% and 91%. | None |
| Lai et al., 2012 [56] China | MCF-7, BT-474, MDA-MB-231, and normal breast cells Human | Curcumin at 10 μg/mL | 72 h | Colorimetric analysis of sulforhodamine B ↓ cell proliferation (MCF-7, BT-474, and MDA-MB-231 cells). | Not reported | Not reported | None |
| Li et al., 2018 [3] China | MDA-MB-231 Human | Curcumin at 10 g/mL Nanostructured platform curcumin and curcumin nanoparticle MSN/IR780-PEI-FA 160 mg/kg | 24 and 48 h | Not reported | Not reported | Flow cytometry CUR and free MSN/CUR induced the G2/M phase of the cell cycle. | |
| Liu et al., 2013 [58] China | 4T1 Mouse | Curcumin at 100 μg/mL Nanostructured platform Nanoparticle with self-assembled polymeric micelles (CUR-M) loaded with curcumin (CUR) | 48 h | Not reported | MTT assay Both CUR-M and Free CUR drastically inhibited cell growth in a dose-dependent manner. | TUNEL assay by immunofluorescence staining Apoptotic index CUR-M: 15.77 ± 2.74%, Free CUR: 9.42 ± 2.13% p < 0.001) | None |
| Liu et al., 2009 [58] China | MDA-MB-231 Human | Curcumin at 1, 1.25, 2.5, 5, 10, and 20 mg/mL | 24 and 48 h | Method (NR) Inhibition of cell growth by 60–70% with 1.25 mg/mL curcumin. Inhibition of cell growth by 50–60% with 2.5 mg/mL curcumin. | Not reported | Not reported | NR |
| Lv et al., 2014 [61] China | MDA-MB-231 Human | Curcumin at 1–100 μL | 24 and 48 h | Not reported | MTT assay ↓ significant reduction in the number of viable cells in a time- and dose-dependent manner. | Flow cytometry of fixed nuclei ↑ in the number of apoptotic cells in a dose-dependent manner. | None |
| Masuelli et al., 2013 [63] Italy | MDA-MB-231, MDA -MB-435, MDA-MB-453, MDA-MB-468, T-47D, MCF7, BT-474, SK-BR-3 Human Mammary cancer cells (H-2”) (TUBO) Humanized mouse Mammary cancer cells (H-2”) (TUBO) Mouse | Curcumin 6 to 50 pM | 24 and 48 h | Not reported | Not reported | Pro-apoptotic Bax and anti-apoptotic Bcl-2 expression CUR induced apoptosis in all investigated cell types. | None |
| Mehta et al., 1997 [64] USA | MCF7 Human | Curcumin 1 to 3 μg/mL | 72 h | [3H]thymidine incorporation and flow cytometry. Cell growth inhibition in a time- and dose-dependent manner, correlated with the inhibition of ornithine decarboxylase activity. | Not reported | Flow cytometry Curcumin-induced cell death was not due to apoptosis or any significant change in the expression of apoptosis-related genes, including the Bcl-2, p53, cyclin B, and transglutaminase genes. | NR |
| Montazeri et al., 2017 [65] Iran | MCF7 Human | Curcumin at 23, 17, and 14 µM Dendrosomal curcumin (DNC) for 48 h (28–35 μM) and 72 h (23–25 μM) | 24, 48, and 72 h | Not reported | Not reported | Flow cytometry Total apoptosis by DNC: 24 h: 30.34 ± 0.011% 48 h: 33.83 ± 0.005% 72 h: 61.83 ± 0.009% | None |
| Mukhopadhyay et al., 2020 [67] India | MDA-MB-231 Human | 5 mg of curcumin Nanostructured platform Polymeric NPs PLGA/PVA with or without folate (F) | 24 h | Not reported | Not reported | Flow cytometry Apoptosis CUR-NP-F: 29% Free CUR: 20% | |
| Sarighieh et al., 2020 [70] Irã | MCF7 Human | Curcumin 5, 10, 20, 40, 80, and 160 μM | 24 h | Not reported | MTT assay Curcumin decreased the cell viability of MCF-7 cells. | Flow cytometry Apoptosis 24.6% | None |
| Sun Shih-Han et al., 2012 [73] Taiwan | MDA-MB-231/Her2 Human | Curcumin at 30 and 50 mM | 24 h | Not reported | Not reported | Flow cytometry Apoptosis occurred at a higher dosage (50 mM). | None |
| Sun Xiao-Dong et al., 2012 [74] China | MDA-MB-231 Human | Curcumin at 10, 20, and 30 μmol/mL | 48 h | MTT assay The inhibitory effect on MDA-MB-231 cell proliferation peaked upon treatment with 30 μmol/mL curcumin (p < 0.01). | Not reported | Flow cytometry Apoptosis control 2.76% and Curcumin 26.34%, 30 μmol/mL (p < 0.01). | None |
| Wang Xet al., 2017 [76] China | MCF-7 Human | Curcumin [0 (with DMSO vehicle), 0.5, 1.0, 2.0, 5.0, and 10.0 µM] | 24, 48, and 72 h | MTT assay ↓ cell growth (treatment with 0, 0.5, 1.0, 2.0, 5.0, and 10.0 µM curcumin). | Not reported | Flow cytometry Apoptotic cell death within 48 h upon treatment with 2 µM (p = 0.0021) and 5 µM (p = 0.0004) curcumin. | None |
| Yang et al., 2017 a [76] China | MCF-7 Human | Curcumin at 50 μm Nanostructured platform Micelle NPs (PPBV triblock copolymer) | 24 h | Not reported | Not reported | Flow cytometry Apoptotic cell death | |
| Younesian et al., 2017 [80] Irã | SKBR3 Human | Curcumin at 2.5, 10, 15, 20, 25, and 30 μM | 24, 48, and 72 h | Not reported | Not reported | Flow cytometry Apoptosis: 4.37% with 0 μM, 27.46% with 5 μM, 64.98% with 10 μM, 75.90% with 15 μM, and 76.92% with 20 μM curcumin. | None |
| Yu et al., 2021 [81] China | 4T1 Mouse | Curcumin at 5, 10, and 15 μM | 24 h | Not reported | MTT assay ↓ of cell viability by 16% using 15 μg/mL curcumin | Not reported | None |
| Zong et al., 2012 [84] China | MCF-7 Human | Curcumin at 10, 20, 50, and 100 μM | 48 h | MTT assay ↓ cell growth by 37%, 54%, and 73% using 20, 50, and 100 μM curcumin, respectively. | Not reported | Not reported | None |
| Author/Year/Country | Experimental Animal Model * | Intervention | Outcome | Conflicts of Interest | Ethical Approval | |
|---|---|---|---|---|---|---|
| Treatment Follow-Up | Dose (mg/kg)/Administration Route | Anti-Tumor Activity (Size or Volume of the Tumor) | ||||
| Abd-Ellatef et al., 2020 [38] Italy and Egypt | Balb/c/n = 8/JC/mouse/(1 × 107 cells)/mammary fat pad | VT: 50 mm3; three times (on days 1, 7, and 14); vehicle-free CUR: 10% DMSO suspension v/v Follow-up: 18 days Nanostructured platform Solid lipid nanoparticles (SLNs) with or without chitosan (CS) coating (cholesterol; trilaurin, butyl lactate, Epikuron® 200, Cremophor® RH60, sodium taurocholate, Pluronic® F68) | 5 mg/kg; Intravenous administration | CURC-CS-SLN and CURC ↓ VT (35%); Free CUR: no VT ↓; p < 0.01 | None | Yes |
| Alizadeh et al., 2015 [29] Iran | Balb/c/n = 8/Transplantation of spontaneous mouse mammary tumor/pieces < 0.3 cm3/subcutaneous administration in the left flank | 14 days after tumor induction; daily for 24 days Follow-up: 35 days Nanostructured platform Micelles/polymersomes NPs (PNP) [monomethoxyPEG (mPEG 2000), oleic acid (OA)] | Dose: (NR); Intraperitoneal administration | CUR-NP ↓ VT (80%); p < 0.05 | None | Yes |
| Bansal et al., 2014 [30] USA | Female ACI mice/ 5 to 6 weeks old/mammary tumorigenesis mediated by 17β-estradiol (E2)/9 mg of E2/back | 4 days after tumor induction/ Curcumin implants (n = 6) Curcumin diet (n = 6) Follow-up: 6 months | Curcumin 1000 ppm via diet Two 2 cm implants, 200 mg/implant, 20% p/p drug load 10.9 mg of curcumin for 25 days subcutaneous administration | Curcumin implant: ↓ VT (35%) Curcumin administration via diet: ineffective | None | Yes |
| Bimonte et al., 2015 [7] Italy | Foxn1 nu/nu female mice/n = 16, 6-to-8-week-old/human breast cancer cell line MDA.MB231/2.5 × 106 cells/right flank | After reaching 30–60 mm3, normal diet (n = 8) and diet containing 0.6% curcumin were administered (n = 8). Follow-up: 6 weeks | 0.6% Curcumin administration via diet | ↓ VT (% NR) (p = 0.0195) | None | NR |
| Chen et al., 2017 [32] China | Balb/c/n = 5/BT-549/human (2 × 106 cells)/subcutaneous administration in the right upper thigh | 200 mm3 VT 35 mg/kg; Fourteen days, every 2 days Intratumoral—Vehicle Free CUR: NR Follow-up: 30 days Nanostructured platform Micelle NPs [POCA4C6 (phosphorylated calixarene) micelles—PM] | 5 mg/kg; Intratumoral administration | CUR-NP ↓ VT (60%); Free CUR: ↓ VT (34%); p < 0.05 | None | Yes |
| Ghosh et al., 2021 [39] India | Swiss albino mice/3 groups (n = 5)/MCF-7 and MDA-MB 231 cells (human)/vein | Alternating days after tumor induction Follow-up: 2 weeks Nanostructured platform Nanoparticles: MSN-Curcumin (MSN-C) and MSN-Hyaluronic acid-Curcumin (MSN-HA-C) | 10 mg/kg; intravenous administration | MSN-HA-C ↓ VT (% NR); p < 0.05 | None | Yes |
| Greish et al., 2018 [40] Bahrain | Balb/c/n = 5/4T1/mouse/(1 × 106 cells)/bilaterally on flanks | VT: 100 mm3; frequency of treatment: unclear; Treatment: 10 days Follow-up: 9 days Nanostructured platform Micelles (curcumin-metal complex and SMA) | 10 and 20 mg/kg; Intravenous administration | CUR-NP-10 mg/kg ↓ VT (61%); CUR-NP-20 mg/kg ↓ VT (92%); p < 0.05 | None | NR |
| Grill et al., 2018 [41] Estados Unidos | Balb-neuT mice/n = NR/HER-2-positive breast cancer cells/ten breast pads | At 2, 4, 7, or 12 weeks of age, and once a month thereafter Follow-up: 24 weeks Nanostructured platform Curcumin-loaded microparticles Curcumin (20 mg) and PLGA (20 mg) | 140 mg of microparticles, corresponding to 58.2 mg of curcumin/administered via subcutaneous injection | Curcumin MP ↓ VT (60%); p < 0.05 | None | Yes |
| Hashemzehi et al., 2018 [42] Iran | Balb mice/n = 4/MCF-7 cells (human)/flanks | VT: 100 mm3; 7 days after tumor induction Follow-up: 22 days Nanostructured platform Nanocurcumin—phytosomal curcumin | Dose: (NR); NR | Curcumin ↓ VT (22.2%) Curcumin + 5-FU ↓ VT (53.3%) | None | Yes |
| He et al., 2019 [43] China | Balb/c/n = 6/4T1/mouse/(1 × 106 cells)/subcutaneous administration in right back | VT: 100 mm3 Every 4 days for 4 times Free CUR: (NM) Follow-up: 21 days Nanostructured platform Polymeric micellar NPs [amphiphilic diblock copolymer—mPEG-b-PLG (Se)-TP] | 5 mg/kg; Intravenous administration | CUR-NP ↓ VT (62.9%); Free CUR: ↓ VT (55%); p < 0.05 | None | Yes |
| Huang et al., 2020 [46] China | Balb/c/n = 5/4T1/mouse/NR/Flank mice | VT: 40–50 mm3/every 2 days for 5 times Follow-up: 16 days Nanostructured platform | 50 mg/kg; Intravenous | CUR-NP ↓ VT (38%); p < 0.05 | None | Yes |
| Ji et al., 2020 [47] China | Balb/c/n = 5/4T1/mouse/(1 × 106 cells)/subcutaneous administration in the right flank | Polymeric NPs (HA-CHEMS); pH-sensitive First day of treatment: NR; Every 2 days Vehicle-free CUR: (NM) Follow-up: 10 days Nanostructured platform Nanocrystal NPs with or without HA | 5 mg/kg; Intravenous | HA@CUR-NCs ↓ VT (86%); CUR-NP ↓ VT (39%); Free CUR: ↓ VT (21%); p < 0.05 | None | Yes |
| Jin et al., 2017 [49] China and USA | Balb/c nude rats/n = 5/MCF-7/human/(1 × 107 cells)/subcutaneous administration in the dorsal flank | 7 days after tumor induction; every 24 h for 20 times Free CUR: NR Follow-up: 3 weeks Nanostructured platform Polymeric NPs with or without EGFR-targeting peptides (GE11) (PLGA-PEG); | 5 mg/kg; Intravenous administration | CUR-NP-GE11 and CUR-NP ↓ VT (80%); Free CUR: sem VT ↓; p < 0.05 | None | Yes |
| Jung et al., 2018 [50] República da Coréia | Balb/c nude rats/n = 4/MDA-MB-468 cells/human/(5 × 106 cells)/right shoulder | VT: 50 mm3; three times a week; eight injections in all Follow-up: NR Nanostructured platform CUR-NP e EGF-CUR-NP | 10 mg/kg; Intraperitoneal administration | CUR-NP-EGFR ↓ VT (59.1%); CUR-NP no ↓ VT; p < 0.05 | None | Yes |
| Kumari et al., 2020 [52] India | Balb/c mice/n = 18/Mouse (4T1)/50 μL, 1 × 106 cells/subcutaneous administration in left flank | VT: 50 mm3; Follow-up: 21 days Nanostructured platform CUR treatment (Free CUR group (0–24 μg/mL)) and CUR-HSA-DOPE NPs treatment (CUR-HSA-DOPE group) | 25 mg/kg; Intravenous administration | CUR-HSA-DOPE ↑ VT (80.41%); Free CUR ↑ VT (86.30%) | None | Yes |
| Laha et al., 2018 [55] India and USA | Balb/c/n = 6/4T1/mouse/NR/mammary fat pad | 10 days after tumor induction; every 5 days for four times Follow-up: 20 days Nanostructured platform Metal organic frameworks NPs (IRMOF-3) with or without folic acid (FA) [(Zn(NO3)2; NH2-H2 BDC] | 2 mg/kg (* unclear); Route of administration: (NM) | CUR-NP-FA ↓ VT (61%); CUR-NP ↓ VT (44%); p < 0.05 | None | Yes |
| Lai et al., 2012 [56] Taiwan | Nude mice/n = 16/BT-474 cells overexpressing HER-2 (1 × 107)/right flank subcutaneous route of administration | 21–28 days after xenograft inoculation. VT:50–100 mm3 Follow-up: after 4 weeks | 45 mg/kg curcumin injected intra-peritoneally | Herceptin and curcumin VT 34.1 ± 25.0 mm3 Curcumin VT 63.6 ± 25.7 mm3 p = 0.079 | ||
| Li et al., 2018 [3] China | Balb/c/n = 4/MDA-MB-231/human/(1 × 107 cells)/subcutaneous administration | Tumor diameter: 4 mm; every 3 days for six times in all Free CUR: NR Follow-up: 18 days Nanostructured platform Mesoporous silica nanoparticles with hyaluronan (MSN-HA) or polyethyleneimine-folic acid (MSN-PEI-FA). | 8 mg/kg; Intravenous administration | CUR-NP-PEI-HA ↓ VT (50%); Free CUR: no VT ↓; p < 0.01 | None | Yes |
| Lin et al., 2016 [57] China | Balb/c nude mice/n = 6/MCF-7/ human/(NM)/Subcutaneous administration in the right axilla | First day of treatment: NR once every 3 days for 15 days Vehicle-Free CUR: (NM) Follow-up: 15 days Nanostructured platform Lipid-based NPs (NLC) with or without folate coating (FA) (PEG-DSPE, soy lecithin, castor oil, Tween 80, and Precirol ATO-5) | Dose: NR; Intravenous administration | CUR-NP-FA ↓ VT (~83%); CUR-NP ↓ VT (~66%); Free CUR: ↓ VT (31%) | None | NR |
| Liu et al., 2013 [58] China | Balb/c mice n = 12; 6 per group/4T1/5 × 105 cells/right flank/subcutaneous administration | From day 4, palpable tumors were daily injected with the treatment agent intravenously for 10 days Follow-up: 25 days Nanostructured platform Self-assembled polymeric micelles (CUR-M) loaded with curcumin (CUR) | CUR-M (30 mg/kg body weight) Free CUR (30 mg/kg body weight) | CUR-M ↓ VT (68%); p < 0.01 Free CUR: sem ↓ VT (35%) | None | Yes |
| Lv et al., 2014 [61] China | Balb/c nude mice/n = 8 per group /MCF-7 and MDA-MB-231/2 × 106 cells/subcutaneous administration in the back | After reaching 60 mm3/treatment days alternating Follow-up: 4 weeks | Curcumin 50 µg/kg, 200 µg/kg Intraperitoneal injections | Cur 50 µg/kg ↓ VT (54%); p < 0.05 Cur 200 µg/kg ↓ (73%); p < 0.05 VT | None | Yes |
| Lv et al., 2015 [60] China | Kunming mice/n = 6/EMT6/mouse/(1.0 × 107 cells/mL)/Subcutaneous administration | VT: 300 mm3; daily for 9 days Vehicle-free CUR: cremophor EL:dehydrated alcohol (1:1, v/v) and diluted with saline solution Follow-up: 14 days Nanostructured platform Polymeric NPs (PEG-PCDA) with or without biotin | 10 mg/kg; Intravenous administration | CUR-NP ↓ VT (69%); CUR-NP-biotin ↓ VT (79%); Free CUR: ↓ TV (32%); p < 0.05 | NR | Yes |
| Mahalunkar et al., 2019 [62] India, Germany and Norway | Balb/c/n = 6/4T1/mouse/(1 × 105 cells)/Mammary fat pad | First day of treatment: (NM) Twice a week for 2 weeks Vehicle-free CUR: (NM) Follow-up: 21 days Nanostructured platform Metallic gold NPs (CurAu-PVP) with folic acid (FA) (HAuCl4 and PVP polymer) | 10 mg/kg; Intratumoral administration | CUR-NP-FA ↓ VT (51%); Free CUR: no ↓ VT; p < 0.006 | None | Yes |
| Masuelli et al., 2013 [63] Italy | Transgenic BALB-neuT mouse/n = 5 per group /NR | After the diameter reached 15 mm, CUR (2 mg in 50 |.il oil with), with oil (50 |.il) or water (50 |.il) was administered three times a week. Follow-up: 30 weeks | Curcumin 6–50 µM Oral administration | No mice treated with CUR exhibited tumor growth at week 22, (p < 0.01). Cur ↓ VT (52%) (p < 0.05) | None | NR |
| Mukerjee et al., 2016 [66] USA | Balb/c nude rats/n = 8/MCF10CA1a/human/(3 × 106 cells)/flank | VT: 70 mm3; Three times a week for 30 days Follow-up: 32 days Nanostructured platform Polymeric NPs [PLGA/PVA with or without antibody targeting (AnxA2)] | 20 mg/kg; Intravenous administration | CUR-NP-AnxA2 ↓ VT (44.0%); CUR-NP ↓ VT (33.5%); p < 0.05 CUR-NP-AnxA2 ↓ PT (53.0%); CUR-NP ↓ PT (30%); p < 0.05 | NR | NR |
| Mukhopadhyay et al., 2020 [67] India | Balb/c nude rat/n = 5/MDA-MB-231/human/(5 × 106 cells)/Right flank | 8 days after induction; three times a week Follow-up: 29 days Nanostructured platform Polymeric NPs [PLGA/PVA with or without folate (F)] | 20 mg/kg Route of administration: unclear | CUR-NP-F ↓ VT (90%); CUR-NP ↓ VT (75%); p < 0.05 | NR | Yes |
| Pal et al., 2019 [68] India | Balb/c mice/n = 5 per group /human MCF-7, MDA-MB-231, MDA-MB-468, and murine 4T1/100 L/abdominal skin | Treatment for 20 days at 3-day intervals after 10 days of tumor implantation Follow-up: 30 days Nanostructured platform Synthesis of curcumin-loaded microsphere (10% by weight polymer) PLGA@CCM@FA | 2000 µg/kg Route of administration: unclear | PLGA—VT 0.092 mm3 ↓ VT (25%) PLGA @ CCM—VT 0.064 mm3 ↓ VT (48%) PLGA @ CCM @FA-VT—0.031 mm3 ↓ VT (75%) | NR | NR |
| Sahne et al., 2019 [69] Irã | Balb/c/n = 4/4T1/mouse/NR/ssubcutaneous administration in the flank | VT: 50–100 mm3; daily follow-up: 3 weeks Nanostructured platform Graphene oxide NPs (GO NPs with CMC, PVP, PEG, and FA) | 4 mg/kg; Intravenous administration | CUR-NP-FA ↓ VT (86%); p < 0.05 | None | Yes |
| Shiri et al., 2015 [71] Irã | Balb/c/n = 9/4T1/mouse/(1 × 106 cells)/left flank | Third day after tumor induction Follow-up: 35 days Nanostructured platform Dendrosome NPs (DNC) [composition: not mentioned (patent number: 71753)]. | 40 or 80 mg/kg Route of administration: NR | NP-40 mg/kg ↓ VT (72%); NP-80 mg/kg ↓ VT (76%); p < 0.05 NP-40 mg/kg ↓ VT (61%); NP-80 mg/kg ↓ VT (64%); p < 0.05 | NR | Yes |
| Shukla et al., 2017 [72] India | Balb/c mice/n = 3/(1 × 106 cells)/subcutaneous administration in hind skin | 10 days from tumor inoculation; daily administration for 28 days: gum acacia (1%, w/v). Follow-up: 42 days Nanostructured platform Lipid-based CPC-SNEDDS NPs (Phospholipid, castor oil, Tween 80, and PEG 400) | 100 mg/kg; oral | 1) CUR-NP ↓ VT (58.9%); Free CUR ↓ VT (29.5%); p < 0.001 | None | Yes |
| Vakilinezhad et al., 2019 [75] Irã | Sprague–Dawley rats/n = 6/Chemically-induced mammary tumors (MNU) | 4 months after tumor induction; Once a week for 4 weeks Free curing vehicle: aqueous suspension Follow-up: 20 weeks Nanostructured platform Polymeric NPs (PLGA-PVA) | 2.5 mg; Intravenous | CUR-NP ↓ VT (20%); Free CUR: ↓ VT (16%); p < 0.05 | None | Yes |
| Wang et al., 2018 [77] China | Nude mice/n = (NM)/MDA-MB-231/human/(1.5 × 106 cells)/subcutaneous | 2 months after tumor induction; daily Free CUR: (NM) Follow-up: 2 weeks Nanostructured platform Polymeric NPs (MPEG-PCL) | 1 × 10−3 M; Intravenous administration | CUR-NP ↓ VT (82%); Free CUR: ↓ VT (49%); p < 0.01 | None | Yes |
| Yang et al., 2017 a [78] China | Balb/c nude mice/n = 5 MCF-7/human/(1 × 107 cells)/subcutaneous administration in the flank | VT: 200 mm3 Every other day, five times; total duration: 20 days Free CUR vehicle: NR Follow-up: 20 days Nanostructured platform Hybrid NPs [PLGA NPs coated with a modified hyaluronic acid (HA hybrid)] | 15 mg/kg; Intravenous | HA-Hybrid NPs/CUR ↓ VT (43.8%, day 12); ↓ VT (24%, day 20); p < 0.05 | NR | Yes |
| Yang et al., 2017 b [79] China | Balb/c nude mice/n = 5 MCF-7/human/(1 × 107 cells)/subcutaneous administration in the flank | VT: 200 mm3 Every other day, five times; total duration: 20 days Free CUR vehicle: NR Follow-up: 20 days Nanostructured platform Micelle NPs (PPBV triblock copolymer) | 10 mg/kg; Intravenous | PPBV micelles/CUR ↓ VT (58.5%, day 12); ↓ VT (28.9%, day 20); p < 0.05 | NR | Yes |
| Yu et al., 2014 [82] China | Balb/c nude mice/n = 5/MCF-7/human/(3 × 106 cells)/subcutaneous administration in the right flank | VT: 100–400 mm3; Every other day for 5 times for 24 days in all Follow-up: 25 days Nanostructured platform Micelle NPs (MPEG-PLA with or without PAE) | 40 mg/kg; Intravenous administration | CUR-NP-PAE ↓ VT (65.6%); CUR-NP ↓ VT (47.1%); p < 0.05 | NR | Yes |
| Yu et al., 2021 [81] China | Balb/c mice/ murine 4T1/NR/intradermal administration in the back of the neck | VT: 150–200 mm3, administration via tail vein every 3 days; 14 days in all Follow-up: 16 days Nanostructured platform curcumin (CUR), zeolitic imidazolate framework-8 nanoparticles (ZIF-8), and hyaluronic acid (HA) | CUR@ZIF-8 19.6 mg of CUR@ZIF-8@ HA 20.9 mg Intravenous administration | CUR@ZIF-8 ↓ VT (12.5%); CUR@ZIF-8@HA ↓ VT (62.5%); | None | Yes |
| Yuan et al., 2018 [83] China | Balb/c nude mice/n = 6/MCF-7/human/(3 × 106 cells)/right flank | VT: 100 mm3; every other day, four times Follow-up: 18 days Nanostructured platform Polymeric NPs (mPEG-PLGA-Pglu) | 2.5 mg/kg; intravenous administration | CUR-NP ↓ VT (28.0%); p < 0.05 CUR-NP ↓ PT (22.5%); p < 0.05 | None | Yes |
3.2.3. Cell Viability
3.2.4. Apoptosis and/or Interruption of Cell Cycle
3.2.5. Animal Studies
3.3. Conflict of Interest and Ethics Committee Approval
3.4. Overall Quality of Evidence
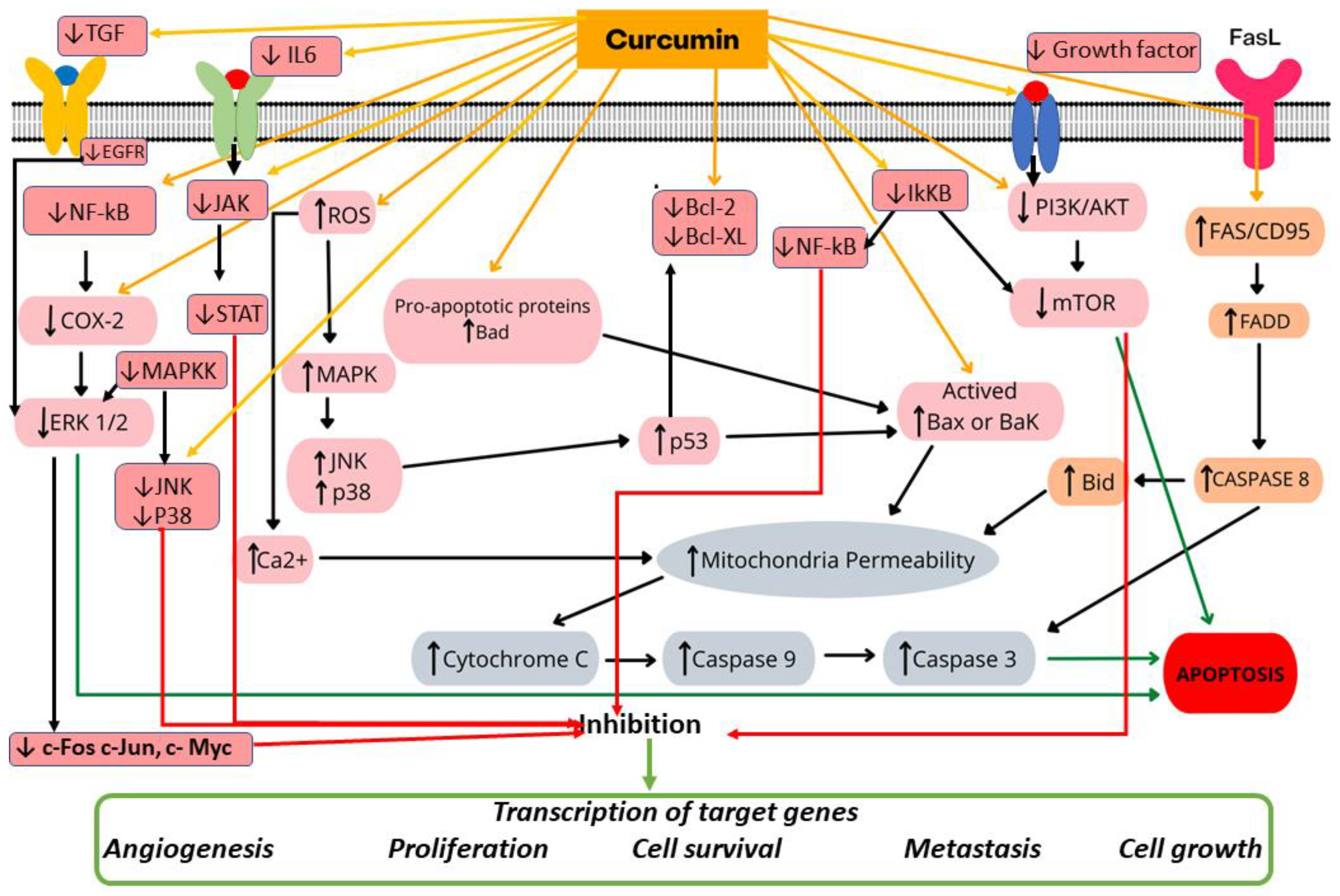
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Momenimovahed, Z.; Salehiniya, H. Epidemiological characteristics of and risk factors for breast cancer in the world. Breast Cancer (Dove Med. Press) 2019, 11, 151–164. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Sung, H.; Ferlay, J.; Siegel, R.L.; Laversanne, M.; Soerjomataram, I.; Jemal, A.; Bray, F. Global Cancer Statistics 2020: GLOBOCAN Estimates of Incidence and Mortality Worldwide for 36 Cancers in 185 Countries. CA A Cancer J. Clin. 2021, 71, 209–249. [Google Scholar] [CrossRef] [PubMed]
- Li, N.; Wang, Z.; Zhang, Y.; Zhang, K.; Xie, J.; Liu, Y.; Li, W.; Feng, N. Curcumin-loaded redox-responsive mesoporous silica nanoparticles for targeted breast cancer therapy. Artif. Cells Nanomed. Biotechnol. 2018, 46, 921–935. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Inotai, A.; Ágh, T.; Maris, R.; Erdősi, D.; Kovács, S.; Kaló, Z.; Senkus, E. Systematic review of real-world studies evaluating the impact of medication non-adherence to endocrine therapies on hard clinical endpoints in patients with non-metastatic breast cancer. Cancer Treat. Rev. 2021, 100, 102264. [Google Scholar] [CrossRef]
- Terret, C.; Russo, C. Pharmacotherapeutic Management of Breast Cancer in Elderly Patients: The Promise of Novel Agents. Drugs Aging 2018, 35, 93–115. [Google Scholar] [CrossRef]
- Sen, G.S.; Mohanty, S.; Hossain, D.M.S.; Bhattacharyya, S.; Banerjee, S.; Chakraborty, J.; Saha, S.; Ray, P.; Bhattacharjee, P.; Mandal, D.; et al. Curcumin enhances the efficacy of chemotherapy by tailoring p65NFκB-p300 cross-talk in favor of p53–p300 in breast cancer. J. Biol. Chem. 2011, 286, 42232–42247. [Google Scholar] [CrossRef] [Green Version]
- Bimonte, S.; Barbieri, A.; Palma, G.; Rea, D.; Luciano, A.; D’Aiuto, M.; Arra, C.; Izzo, F. Dissecting the role of curcumin in tumour growth and angiogenesis in mouse model of human breast cancer. BioMed Res. Int. 2015, 2015, 878134. [Google Scholar] [CrossRef]
- Witika, B.A.; Makoni, P.A.; Matafwali, S.K.; Mweetwa, L.L.; Shandele, G.C.; Walker, R.B. Enhancement of Biological and Pharmacological Properties of an Encapsulated Polyphenol: Curcumin. Molecules 2021, 26, 4244. [Google Scholar] [CrossRef]
- Farghadani, R.; Naidu, R. Curcumin: Modulator of Key Molecular Signaling Pathways in Hormone-Independent Breast Cancer. Cancers 2021, 13, 3427. [Google Scholar] [CrossRef]
- Farghadani, R.; Naidu, R. Curcumin as an Enhancer of Therapeutic Efficiency of Chemotherapy Drugs in Breast Cancer. Int. J. Mol. Sci. 2022, 23, 2144. [Google Scholar] [CrossRef]
- Sinha, D.; Biswas, J.; Sung, B.; Aggarwal, B.B.; Bishayee, A. Chemopreventive and chemotherapeutic potential of curcumin in breast cancer. Curr. Drug Targets 2012, 13, 1799–1819. [Google Scholar] [CrossRef] [PubMed]
- Song, X.; Zhang, M.; Dai, E.; Luo, Y. Molecular targets of curcumin in breast cancer (Review). Mol. Med. Rep. 2019, 19, 23–29. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Ombredane, A.S.; Silva, V.R.P.; Andrade, L.R.; Pinheiro, W.O.; Simonelly, M.; Oliveira, J.V.; Pinheiro, A.C.; Gonçalves, G.F.; Felice, G.J.; Garcia, M.P.; et al. In Vivo Efficacy and Toxicity of Curcumin Nanoparticles in Breast Cancer Treatment: A Systematic Review. Front. Oncol. 2021, 11, 612903. [Google Scholar] [CrossRef] [PubMed]
- Gianfredi, V.; Nucci, D.; Vannini, S.; Villarini, M.; Moretti, M. In vitro Biological Effects of Sulforaphane (SFN), Epigallocatechin-3-gallate (EGCG), and Curcumin on Breast Cancer Cells: A Systematic Review of the Literature. Nutr. Cancer 2017, 69, 969–978. [Google Scholar] [CrossRef] [PubMed]
- Moher, D.; Liberati, A.; Tetzlaff, J.; Altman, D.G. Preferred reporting items for systematic reviews and meta-analyses: The PRISMA statement. BMJ 2009, 339, b2535. [Google Scholar] [CrossRef] [Green Version]
- de Vries, R.B.M.; Hooijmans, C.R.; Langendam, M.W.; van Luijk, J.; Leenaars, M.; Ritskes-Hoitinga, M.; Wever, K.E. A protocol format for the preparation, registration and publication of systematic reviews of animal intervention studies. Evid. Based Preclin. Med. 2015, 2, e00007. [Google Scholar] [CrossRef]
- Yang, N.; Ray, S.D.; Krafts, K. Cell Proliferation. In Encyclopedia of Toxicology, 3rd ed.; Wexler, P., Ed.; Academic Press: Oxford, UK, 2014. [Google Scholar]
- Peng, Y.; Li, J.; Zhu, L. Chapter 8—Cancer and non-coding RNAs. In Nutritional Epigenomics; Ferguson, B.S., Ed.; Academic Press: Oxford, UK, 2019; Volume 14, pp. 119–132. [Google Scholar]
- Fang, I.J.; Trewyn, B.G. Application of mesoporous silica nanoparticles in intracellular delivery of molecules and proteins. Methods Enzymol. 2012, 508, 41–59. [Google Scholar]
- Khan, Y. Characterizing the Properties of Tissue Constructs for Regenerative Engineering; Elsevier: Amsterdam, The Netherlands, 2019. [Google Scholar] [CrossRef]
- Brown, D.A.; Yang, N.; Ray, S.D. Apoptosis. In Encyclopedia of Toxicology, 3rd ed.; Wexler, P., Ed.; Academic Press: Oxford, UK, 2014. [Google Scholar]
- Wong, R.S. Apoptosis in cancer: From pathogenesis to treatment. J. Exp. Clin. Cancer Res. 2011, 30, 87. [Google Scholar] [CrossRef] [Green Version]
- Noll, M.; Wedderkopp, N.; Mendonça, C.R.; Kjaer, P. Motor performance and back pain in children and adolescents: A systematic review and meta-analysis protocol. Syst. Rev. 2020, 9, 212. [Google Scholar] [CrossRef]
- Noll, M.; Kjaer, P.; Mendonça, C.R.; Wedderkopp, N. Motor performance and back pain in children and adolescents: A systematic review. Eur. J. Pain 2022, 26, 77–102. [Google Scholar] [CrossRef]
- Pavan, L.M.C.; Rêgo, D.F.; Elias, S.T.; De Luca Canto, G.; Guerra, E.N.S. In vitro Anti-Tumor Effects of Statins on Head and Neck Squamous Cell Carcinoma: A Systematic Review. PLoS ONE 2015, 10, e0130476. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Wei, D.; Tang, K.; Wang, Q.; Estill, J.; Yao, L.; Wang, X.; Chen, Y.; Yang, K. The use of GRADE approach in systematic reviews of animal studies. J. Evid.-Based Med. 2016, 9, 98–104. [Google Scholar] [CrossRef] [PubMed]
- Abbaspour, H.; Safipour Afshar, A. Curcumin inhibits the expression of ornithine decarboxylase and adenosine deaminase genes in MCF-7 human breast cancer cells. Arch. Biol. Sci. 2018, 70, 639–645. [Google Scholar] [CrossRef]
- Abuelba, H.; Cotrutz, C.E.; Stoica, B.A.; Stoica, L.; Olinici, D.; Petreuş, T. In vitro evaluation of curcumin effects on breast adenocarcinoma 2D and 3D cell cultures. Rom. J. Morphol. Embryol. = Rev. Roum. De Morphol. Et Embryol. 2015, 56, 71–76. [Google Scholar]
- Alizadeh, A.M.; Sadeghizadeh, M.; Najafi, F.; Ardestani, S.K.; Erfani-Moghadam, V.; Khaniki, M.; Rezaei, A.; Zamani, M.; Khodayari, S.; Khodayari, H.; et al. Encapsulation of Curcumin in Diblock Copolymer Micelles for Cancer Therapy. BioMed Res. Int. 2015, 2015, 824746. [Google Scholar] [CrossRef] [PubMed]
- Bansal, S.S.; Kausar, H.; Vadhanam, M.V.; Ravoori, S.; Pan, J.; Rai, S.N.; Gupta, R.C. Curcumin implants, not curcumin diet, inhibit estrogen-induced mammary carcinogenesis in ACI rats. Cancer Prev. Res. 2014, 7, 456–465. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Calaf, G.M.; Ponce-Cusi, R.; Carrión, F. Curcumin and paclitaxel induce cell death in breast cancer cell lines. Oncol. Rep. 2018, 40, 2381–2388. [Google Scholar] [CrossRef]
- Chen, W.; Li, L.; Zhang, X.; Liang, Y.-k.; Pu, Z.; Wang, L.; Mo, J.J.D.D. Curcumin: A calixarene derivative micelle potentiates anti-breast cancer stem cells effects in xenografted, triple-negative breast cancer mouse models. Drug Deliv. 2017, 24, 1470–1481. [Google Scholar] [CrossRef] [Green Version]
- Chiu, T.L.; Su, C.C. Curcumin inhibits proliferation and migration by increasing the Bax to Bcl-2 ratio and decreasing NF-kappaBp65 expression in breast cancer MDA-MB-231 cells. Int. J. Mol. Med. 2009, 23, 469–475. [Google Scholar]
- Choudhuri, T.; Pal, S.; Agwarwal, M.L.; Das, T.; Sa, G. Curcumin induces apoptosis in human breast cancer cells through p53-dependent Bax induction. FEBS Lett. 2002, 512, 334–340. [Google Scholar] [CrossRef] [Green Version]
- Coker-Gurkan, A.; Bulut, D.; Genc, R.; Arisan, E.D.; Obakan-Yerlikaya, P.; Palavan-Unsal, N. Curcumin prevented human autocrine growth hormone (GH) signaling mediated NF-κB activation and miR-183-96-182 cluster stimulated epithelial mesenchymal transition in T47D breast cancer cells. Mol. Biol. Rep. 2019, 46, 355–369. [Google Scholar] [CrossRef] [PubMed]
- Coker-Gurkan, A.; Celik, M.; Ugur, M.; Arisan, E.D.; Obakan-Yerlikaya, P.; Durdu, Z.B.; Palavan-Unsal, N. Curcumin inhibits autocrine growth hormone-mediated invasion and metastasis by targeting NF-κB signaling and polyamine metabolism in breast cancer cells. Amino Acids 2018, 50, 1045–1069. [Google Scholar] [CrossRef] [PubMed]
- Fan, H.; Liang, Y.; Jiang, B.; Li, X.; Xun, H.; Sun, J.; He, W.; Lau, H.T.; Ma, X. Curcumin inhibits intracellular fatty acid synthase and induces apoptosis in human breast cancer MDA-MB-231 cells. Oncol. Rep. 2016, 35, 2651–2656. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Fathy Abd-Ellatef, G.E.; Gazzano, E.; Chirio, D.; Hamed, A.R.; Belisario, D.C.; Zuddas, C.; Peira, E.; Rolando, B.; Kopecka, J.; Assem Said Marie, M.; et al. Curcumin-Loaded Solid Lipid Nanoparticles Bypass P-Glycoprotein Mediated Doxorubicin Resistance in Triple Negative Breast Cancer Cells. Pharmaceutics 2020, 12, 96. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Ghosh, S.; Dutta, S.; Sarkar, A.; Kundu, M.; Sil, P.C. Targeted delivery of curcumin in breast cancer cells via hyaluronic acid modified mesoporous silica nanoparticle to enhance anticancer efficiency. Colloids Surf. B Biointerfaces 2021, 197, 111404. [Google Scholar] [CrossRef] [PubMed]
- Greish, K.; Pittalà, V.; Taurin, S.; Taha, S.; Bahman, F.; Mathur, A.; Jasim, A.; Mohammed, F.; El-Deeb, I.M.; Fredericks, S.; et al. Curcumin-Copper Complex Nanoparticles for the Management of Triple-Negative Breast Cancer. Nanomaterials 2018, 8, 884. [Google Scholar] [CrossRef] [Green Version]
- Grill, A.E.; Shahani, K.; Koniar, B.; Panyam, J. Chemopreventive efficacy of curcumin-loaded PLGA microparticles in a transgenic mouse model of HER-2-positive breast cancer. Drug Deliv. Transl. Res. 2018, 8, 329–341. [Google Scholar] [CrossRef] [PubMed]
- Hashemzehi, M.; Behnam-Rassouli, R.; Hassanian, S.M.; Moradi-Binabaj, M.; Moradi-Marjaneh, R.; Rahmani, F.; Fiuji, H.; Jamili, M.; Mirahmadi, M.; Boromand, N.; et al. Phytosomal-curcumin antagonizes cell growth and migration, induced by thrombin through AMP-Kinase in breast cancer. J. Cell Biochem. 2018, 119, 5996–6007. [Google Scholar] [CrossRef]
- He, H.; Zhuang, W.; Ma, B.; Su, X.; Yu, T.; Hu, J.; Chen, L.; Peng, R.; Li, G.; Wang, Y. Oxidation-Responsive and Aggregation-Induced Emission Polymeric Micelles with Two-Photon Excitation for Cancer Therapy and Bioimaging. ACS Biomater. Sci. Eng. 2019, 5, 2577–2586. [Google Scholar] [CrossRef]
- Hu, S.; Xu, Y.; Meng, L.; Huang, L.; Sun, H. Curcumin inhibits proliferation and promotes apoptosis of breast cancer cells. Exp. Ther. Med. 2018, 16, 1266–1272. [Google Scholar] [CrossRef]
- Hua, W.F.; Fu, Y.S.; Liao, Y.J.; Xia, W.J.; Chen, Y.C.; Zeng, Y.X.; Kung, H.F.; Xie, D. Curcumin induces down-regulation of EZH2 expression through the MAPK pathway in MDA-MB-435 human breast cancer cells. Eur. J. Pharmacol. 2010, 637, 16–21. [Google Scholar] [CrossRef] [PubMed]
- Huang, C.; Chen, F.; Zhang, L.; Yang, Y.; Yang, X.; Pan, W. (99 m)Tc Radiolabeled HA/TPGS-Based Curcumin-Loaded Nanoparticle for Breast Cancer Synergistic Theranostics: Design, in vitro and in vivo Evaluation. Int. J. Nanomed. 2020, 15, 2987–2998. [Google Scholar] [CrossRef] [PubMed]
- Ji, P.; Wang, L.; Chen, Y.; Wang, S.; Wu, Z.; Qi, X. Hyaluronic acid hydrophilic surface rehabilitating curcumin nanocrystals for targeted breast cancer treatment with prolonged biodistribution. Biomater. Sci. 2020, 8, 462–472. [Google Scholar] [CrossRef] [PubMed]
- Jiang, M.; Huang, O.; Zhang, X.; Xie, Z.; Shen, A.; Liu, H.; Geng, M.; Shen, K. Curcumin induces cell death and restores tamoxifen sensitivity in the antiestrogen-resistant breast cancer cell lines MCF-7/LCC2 and MCF-7/LCC9. Molecules 2013, 18, 701–720. [Google Scholar] [CrossRef] [Green Version]
- Jin, H.; Pi, J.; Zhao, Y.; Jiang, J.; Li, T.; Zeng, X.; Yang, P.; Evans, C.E.; Cai, J. EGFR-targeting PLGA-PEG nanoparticles as a curcumin delivery system for breast cancer therapy. Nanoscale 2017, 9, 16365–16374. [Google Scholar] [CrossRef]
- Jung, K.H.; Lee, J.H.; Park, J.W.; Kim, D.H.; Moon, S.H.; Cho, Y.S.; Lee, K.H. Targeted therapy of triple negative MDA-MB-468 breast cancer with curcumin delivered by epidermal growth factor-conjugated phospholipid nanoparticles. Oncol. Lett. 2018, 15, 9093–9100. [Google Scholar] [CrossRef] [Green Version]
- Kim, J.M.; Noh, E.M.; Kwon, K.B.; Kim, J.S.; You, Y.O.; Hwang, J.K.; Hwang, B.M.; Kim, B.S.; Lee, S.H.; Lee, S.J.; et al. Curcumin suppresses the TPA-induced invasion through inhibition of PKCα-dependent MMP-expression in MCF-7 human breast cancer cells. Phytomedicine 2012, 19, 1085–1092. [Google Scholar] [CrossRef]
- Kumari, P.; Muddineti, O.S.; Rompicharla, S.V.; Ghanta, P.; Karthik, B.B.N.A.; Ghosh, B.; Biswas, S. Cholesterol-conjugated poly (D, L-lactide)-based micelles as a nanocarrier system for effective delivery of curcumin in cancer therapy. Drug Deliv. 2017, 24, 209–223. [Google Scholar] [CrossRef] [Green Version]
- Kumari, P.; Paul, M.; Bobde, Y.; Soniya, K.; Kiran Rompicharla, S.V.; Ghosh, B.; Biswas, S. Albumin-based lipoprotein nanoparticles for improved delivery and anticancer activity of curcumin for cancer treatment. Nanomedicine 2020, 15, 2851–2869. [Google Scholar] [CrossRef]
- Kumari, P.; Swami, M.O.; Nadipalli, S.K.; Myneni, S.; Ghosh, B.; Biswas, S. Curcumin Delivery by Poly(Lactide)-Based Co-Polymeric Micelles: An In Vitro Anticancer Study. Pharm. Res. 2016, 33, 826–841. [Google Scholar] [CrossRef]
- Laha, D.; Pal, K.; Ray Chowdhuri, A.; Parida, P.; Sahu, S.; Jana, K.; Karmakar, P. Fabrication of curcumin loaded folic acid tagged metal organic framework for triple negative breast cancer therapy in in vitro and in vivo system. New J. Chem. 2018, 43, 217–229. [Google Scholar] [CrossRef]
- Lai, H.-W.; Chien, S.-Y.; Kuo, S.-J.; Tseng, L.-M.; Lin, H.-Y.; Chi, C.-W.; Chen, D.-R. The Potential Utility of Curcumin in the Treatment of HER-2-Overexpressed Breast Cancer: An In Vitro and In Vivo Comparison Study with Herceptin. Evid. Based Complement. Altern. Med. 2012, 2012, 486568. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Lin, M.; Teng, L.; Wang, Y.; Zhang, J.; Sun, X. Curcumin-guided nanotherapy: A lipid-based nanomedicine for targeted drug delivery in breast cancer therapy. Drug Deliv. 2016, 23, 1420–1425. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Liu, L.; Sun, L.; Wu, Q.; Guo, W.; Li, L.; Chen, Y.; Li, Y.; Gong, C.; Qian, Z.; Wei, Y. Curcumin loaded polymeric micelles inhibit breast tumor growth and spontaneous pulmonary metastasis. Int. J. Pharm. 2013, 443, 175–182. [Google Scholar] [CrossRef] [PubMed]
- Liu, Q.; Loo, W.T.; Sze, S.C.; Tong, Y. Curcumin inhibits cell proliferation of MDA-MB-231 and BT-483 breast cancer cells mediated by down-regulation of NFkappaB, cyclinD and MMP-1 transcription. Phytomedicine 2009, 16, 916–922. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Lv, L.; Guo, Y.; Shen, Y.; Liu, J.; Zhang, W.; Zhou, D.; Guo, S. Intracellularly Degradable, Self-Assembled Amphiphilic Block Copolycurcumin Nanoparticles for Efficient In Vivo Cancer Chemotherapy. Adv. Healthc. Mater. 2015, 4, 1496–1501. [Google Scholar] [CrossRef] [Green Version]
- Lv, Z.D.; Liu, X.P.; Zhao, W.J.; Dong, Q.; Li, F.N.; Wang, H.B.; Kong, B. Curcumin induces apoptosis in breast cancer cells and inhibits tumor growth in vitro and in vivo. Int. J. Clin. Exp. Pathol. 2014, 7, 2818–2824. [Google Scholar]
- Mahalunkar, S.; Yadav, A.S.; Gorain, M.; Pawar, V.; Braathen, R.; Weiss, S.; Bogen, B.; Gosavi, S.W.; Kundu, G.C. Functional design of pH-responsive folate-targeted polymer-coated gold nanoparticles for drug delivery and in vivo therapy in breast cancer. Int. J. Nanomed. 2019, 14, 8285–8302. [Google Scholar] [CrossRef] [Green Version]
- Masuelli, L.; Benvenuto, M.; Fantini, M.; Marzocchella, L.; Sacchetti, P.; Di Stefano, E.; Tresoldi, I.; Izzi, V.; Bernardini, R.; Palumbo, C.; et al. Curcumin induces apoptosis in breast cancer cell lines and delays the growth of mammary tumors in neu transgenic mice. J. Biol. Regul. Homeost. Agents 2013, 27, 105–119. [Google Scholar]
- Mehta, K.; Pantazis, P.; McQueen, T.; Aggarwal, B.B. Antiproliferative effect of curcumin (diferuloylmethane) against human breast tumor cell lines. Anti-Cancer Drugs 1997, 8, 470–481. [Google Scholar] [CrossRef]
- Montazeri, M.; Pilehvar-Soltanahmadi, Y.; Mohaghegh, M.; Panahi, A.; Khodi, S.; Zarghami, N.; Sadeghizadeh, M. Antiproliferative and Apoptotic Effect of Dendrosomal Curcumin Nanoformulation in P53 Mutant and Wide-Type Cancer Cell Lines. Anticancer Agents Med. Chem. 2017, 17, 662–673. [Google Scholar] [CrossRef]
- Mukerjee, A.; Ranjan, A.P.; Vishwanatha, J.K. Targeted Nanocurcumin Therapy Using Annexin A2 Anitbody Improves Tumor Accumulation and Therapeutic Efficacy Against Highly Metastatic Breast Cancer. J. Biomed. Nanotechnol. 2016, 12, 1374–1392. [Google Scholar] [CrossRef]
- Mukhopadhyay, R.; Sen, R.; Paul, B.; Kazi, J.; Ganguly, S.; Debnath, M.C. Gemcitabine Co-Encapsulated with Curcumin in Folate Decorated PLGA Nanoparticles; a Novel Approach to Treat Breast Adenocarcinoma. Pharm. Res. 2020, 37, 56. [Google Scholar] [CrossRef]
- Pal, K.; Laha, D.; Parida, P.K.; Roy, S.; Bardhan, S.; Dutta, A.; Jana, K.; Karmakar, P. An In Vivo Study for Targeted Delivery of Curcumin in Human Triple Negative Breast Carcinoma Cells Using Biocompatible PLGA Microspheres Conjugated with Folic Acid. J. Nanosci. Nanotechnol. 2019, 19, 3720–3733. [Google Scholar] [CrossRef]
- Sahne, F.; Mohammadi, M.; Najafpour, G.D. Single-Layer Assembly of Multifunctional Carboxymethylcellulose on Graphene Oxide Nanoparticles for Improving in Vivo Curcumin Delivery into Tumor Cells. ACS Biomater. Sci. Eng. 2019, 5, 2595–2609. [Google Scholar] [CrossRef]
- Sarighieh, M.A.; Montazeri, V.; Shadboorestan, A.; Ghahremani, M.H.; Ostad, S.N. The Inhibitory Effect of Curcumin on Hypoxia Inducer Factors (Hifs) as a Regulatory Factor in the Growth of Tumor Cells in Breast Cancer Stem-Like Cells. Drug Res. 2020, 70, 512–518. [Google Scholar] [CrossRef]
- Shiri, S.; Alizadeh, A.M.; Baradaran, B.; Farhanghi, B.; Shanehbandi, D.; Khodayari, S.; Khodayari, H.; Tavassoli, A. Dendrosomal curcumin suppresses metastatic breast cancer in mice by changing m1/m2 macrophage balance in the tumor microenvironment. Asian Pac. J. Cancer Prev. 2015, 16, 3917–3922. [Google Scholar] [CrossRef] [Green Version]
- Shukla, M.; Jaiswal, S.; Sharma, A.; Srivastava, P.K.; Arya, A.; Dwivedi, A.K.; Lal, J. A combination of complexation and self-nanoemulsifying drug delivery system for enhancing oral bioavailability and anticancer efficacy of curcumin. Drug Dev. Ind. Pharm. 2017, 43, 847–861. [Google Scholar] [CrossRef]
- Sun, S.H.; Huang, H.C.; Huang, C.; Lin, J.K. Cycle arrest and apoptosis in MDA-MB-231/Her2 cells induced by curcumin. Eur. J. Pharmacol. 2012, 690, 22–30. [Google Scholar] [CrossRef]
- Sun, X.D.; Liu, X.E.; Huang, D.S. Curcumin induces apoptosis of triple-negative breast cancer cells by inhibition of EGFR expression. Mol. Med. Rep. 2012, 6, 1267–1270. [Google Scholar] [CrossRef] [Green Version]
- Vakilinezhad, M.A.; Amini, A.; Dara, T.; Alipour, S. Methotrexate and Curcumin co-encapsulated PLGA nanoparticles as a potential breast cancer therapeutic system: In vitro and in vivo evaluation. Colloids Surfaces. B Biointerfaces 2019, 184, 110515. [Google Scholar] [CrossRef] [PubMed]
- Wang, X.; Hang, Y.; Liu, J.; Hou, Y.; Wang, N.; Wang, M. Anticancer effect of curcumin inhibits cell growth through miR-21/PTEN/Akt pathway in breast cancer cell. Oncol. Lett. 2017, 13, 4825–4831. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Wang, Y.; Luo, Z.; Wang, Z.; You, M.; Xie, S.; Peng, Y.; Yang, H. Effect of curcumin-loaded nanoparticles on mitochondrial dysfunctions of breast cancer cells. J. Nanopart. Res. 2018, 20, 1–11. [Google Scholar] [CrossRef]
- Yang, Z.; Sun, N.; Cheng, R.; Zhao, C.; Liu, J.; Tian, Z. Hybrid nanoparticles coated with hyaluronic acid lipoid for targeted co-delivery of paclitaxel and curcumin to synergistically eliminate breast cancer stem cells. J. Mater. Chem. B 2017, 5, 6762–6775. [Google Scholar] [CrossRef]
- Yang, Z.; Sun, N.; Cheng, R.; Zhao, C.; Liu, Z.; Li, X.; Liu, J.; Tian, Z. pH multistage responsive micellar system with charge-switch and PEG layer detachment for co-delivery of paclitaxel and curcumin to synergistically eliminate breast cancer stem cells. Biomaterials 2017, 147, 53–67. [Google Scholar] [CrossRef]
- Younesian, O.; Kazerouni, F.; Dehghan-Nayeri, N.; Omrani, D.; Rahimipour, A.; Shanaki, M.; Rezapour Kalkhoran, M.; Cheshmi, F. Effect of Curcumin on Fatty Acid Synthase Expression and Enzyme Activity in Breast Cancer Cell Line SKBR3. Int. J. Cancer Manag. 2017, 10, e8173. [Google Scholar] [CrossRef]
- Yu, S.; Wang, S.; Xie, Z.; Yu, S.; Li, L.; Xiao, H.; Song, Y. Hyaluronic acid coating on the surface of curcumin-loaded ZIF-8 nanoparticles for improved breast cancer therapy: An in vitro and in vivo study. Colloids Surf. B Biointerfaces 2021, 203, 111759. [Google Scholar] [CrossRef]
- Yu, Y.; Zhang, X.; Qiu, L. The anti-tumor efficacy of curcumin when delivered by size/charge-changing multistage polymeric micelles based on amphiphilic poly(β-amino ester) derivates. Biomaterials 2014, 35, 3467–3479. [Google Scholar] [CrossRef]
- Yuan, J.D.; ZhuGe, D.L.; Tong, M.Q.; Lin, M.T.; Xu, X.F.; Tang, X.; Zhao, Y.Z.; Xu, H.L. pH-sensitive polymeric nanoparticles of mPEG-PLGA-PGlu with hybrid core for simultaneous encapsulation of curcumin and doxorubicin to kill the heterogeneous tumour cells in breast cancer. Artif. Cells Nanomed. 2018, 46, 302–313. [Google Scholar] [CrossRef] [Green Version]
- Zong, H.; Wang, F.; Fan, Q.X.; Wang, L.X. Curcumin inhibits metastatic progression of breast cancer cell through suppression of urokinase-type plasminogen activator by NF-kappa B signaling pathways. Mol. Biol. Rep. 2012, 39, 4803–4808. [Google Scholar] [CrossRef]
- Liu, H.-T.; Ho, Y.-S. Anticancer effect of curcumin on breast cancer and stem cells. Food Sci. Hum. Wellness 2018, 7, 134–137. [Google Scholar] [CrossRef]
- Abbasalizadeh, F.; Alizadeh, E.; Bagher Fazljou, S.M.; Torbati, M.; Akbarzadeh, A. Anticancer Effect of Alginate-Chitosan Hydrogel Loaded with Curcumin and Chrysin on Lung and Breast Cancer Cell Lines. Curr. Drug Deliv. 2021. [Google Scholar] [CrossRef]
- Saghatelyan, T.; Tananyan, A.; Janoyan, N.; Tadevosyan, A.; Petrosyan, H.; Hovhannisyan, A.; Hayrapetyan, L.; Arustamyan, M.; Arnhold, J.; Rotmann, A.R.; et al. Efficacy and safety of curcumin in combination with paclitaxel in patients with advanced, metastatic breast cancer: A comparative, randomized, double-blind, placebo-controlled clinical trial. Phytomedicine 2020, 70, 153218. [Google Scholar] [CrossRef]
- Stohs, S.J.; Ji, J.; Bucci, L.R.; Preuss, H.G. A Comparative Pharmacokinetic Assessment of a Novel Highly Bioavailable Curcumin Formulation with 95% Curcumin: A Randomized, Double-Blind, Crossover Study. J. Am. Coll. Nutr. 2018, 37, 51–598. [Google Scholar] [CrossRef] [Green Version]
- Stohs, S.J.; Chen, O.; Ray, S.D.; Ji, J.; Bucci, L.R.; Preuss, H.G. Highly Bioavailable Forms of Curcumin and Promising Avenues for Curcumin-Based Research and Application: A Review. Molecules 2020, 25, 1397. [Google Scholar] [CrossRef] [Green Version]
- Giordano, A.; Tommonaro, G. Curcumin and Cancer. Nutrients 2019, 5, 2376. [Google Scholar] [CrossRef] [Green Version]
- Mansouri, K.; Rasoulpoor, S.; Daneshkhah, A.; Abolfathi, S.; Salari, N.; Mohammadi, M.; Rasoulpoor, S.; Shabani, S. Clinical effects of curcumin in enhancing cancer therapy: A systematic review. BMC Cancer 2020, 24, 791. [Google Scholar] [CrossRef]
| Authors | Selection Bias | Performance Bias | Detection Bias | Attrition Bias | Reporting Bias | Other Biases | ||||
|---|---|---|---|---|---|---|---|---|---|---|
| 1 | 2 | 3 | 4 | 5 | 6 | 7 | 8 | 9 | 10 | |
| Abd-Ellatef et al., 2020 [38] |  |  |  |  |  |  |  |  |  |  |
| Alizadeh et al., 2015 [29] |  |  |  |  |  |  |  |  |  |  |
| Bansal et al., 2014 [30] |  |  |  |  |  |  |  |  |  |  |
| Bimonte et al., 2015 [7] |  |  |  |  |  |  |  |  |  |  |
| Chen et al., 2017 [32] |  |  |  |  |  |  |  |  |  |  |
| Ghosh et al., 2021 [39] |  |  |  |  |  |  |  |  |  |  |
| Greish et al., 2018 [40] |  |  |  |  |  |  |  |  |  |  |
| Grill et al., 2018 [41] |  |  |  |  |  |  |  |  |  |  |
| Hashemzehi et al., 2018 [42] |  |  |  |  |  |  |  |  |  |  |
| He et al., 2019 [43] |  |  |  |  |  |  |  |  |  |  |
| Huang et al., 2020 [46] China |  |  |  |  |  |  |  |  |  |  |
| Ji et al., 2020 [47] |  |  |  |  |  |  |  |  |  |  |
| Jin et al., 2017 [49] |  |  |  |  |  |  |  |  |  |  |
| Jung et al., 2018 [50] |  |  |  |  |  |  |  |  |  |  |
| Kumari et al., 2020 [52] |  |  |  |  |  |  |  |  |  |  |
| Laha et al., 2018 [55] |  |  |  |  |  |  |  |  |  |  |
| Lai et al., 2012 [56] |  |  |  |  |  |  |  |  |  |  |
| Li et al., 2018 [3] |  |  |  |  |  |  |  |  |  |  |
| Lin et al., 2016 [57] |  |  |  |  |  |  |  |  |  |  |
| Liu et al., 2013 [58] |  |  |  |  |  |  |  |  |  |  |
| Lv et al., 2014 [61] |  |  |  |  |  |  |  |  |  |  |
| Lv et al., 2015 [60] |  |  |  |  |  |  |  |  |  |  |
| Mahalunkar et al., 2019 [62] |  |  |  |  |  |  |  |  |  |  |
| Masuelli et al., 2013 [63] |  |  |  |  |  |  |  |  |  |  |
| Mukerjee et al., 2016 [66] USA |  |  |  |  |  |  |  |  |  |  |
| Mukhopadhyay et al., 2020 [67] |  |  |  |  |  |  |  |  |  |  |
| Pal et al., 2019 [68] |  |  |  |  |  |  |  |  |  |  |
| Sahne et al., 2019 [69] |  |  |  |  |  |  |  |  |  |  |
| Shiri et al., 2015 [71] |  |  |  |  |  |  |  |  |  |  |
| Shukla et al., 2017 [72] |  |  |  |  |  |  |  |  |  |  |
| Vakilinezhad et al., 2019 [75] |  |  |  |  |  |  |  |  |  |  |
| Wang et al., 2018 [77] |  |  |  |  |  |  |  |  |  |  |
| Yang et al., 2017 a [78] |  |  |  |  |  |  |  |  |  |  |
| Yang et al., 2017 b [79] |  |  |  |  |  |  |  |  |  |  |
| Yu et al., 2014 [82] |  |  |  |  |  |  |  |  |  |  |
| Yu et al., 2021 [81] |  |  |  |  |  |  |  |  |  |  |
| Yuan et al., 2018 [83] |  |  |  |  |  |  |  |  |  |  |
 NO
NO  UNCLEAR
UNCLEAR  . YES indicates low risk of bias; NO indicates high risk of bias; UNCLEAR indicates inability of bias assignment. The ten items assessed included: 1 Was the sequence of assignment generated and applied properly? 2 Were the groups similar at baseline, or were they adjusted for confounders in the analysis? 3 Was the allocation to the different groups adequately concealed? 4 Were the animals randomly housed during the experiment? 5 Were caregivers and/or investigators blinded to the intervention each animal received during the experiment? 6 Were the animals randomly selected for the evaluation of results? 7 Was the outcome assessor blinded? 8 Were data of incomplete results handled appropriately? 9 Are study reports exempt from selective result reporting? 10 Was the study apparently free from other problems that could cause a high risk of bias?
. YES indicates low risk of bias; NO indicates high risk of bias; UNCLEAR indicates inability of bias assignment. The ten items assessed included: 1 Was the sequence of assignment generated and applied properly? 2 Were the groups similar at baseline, or were they adjusted for confounders in the analysis? 3 Was the allocation to the different groups adequately concealed? 4 Were the animals randomly housed during the experiment? 5 Were caregivers and/or investigators blinded to the intervention each animal received during the experiment? 6 Were the animals randomly selected for the evaluation of results? 7 Was the outcome assessor blinded? 8 Were data of incomplete results handled appropriately? 9 Are study reports exempt from selective result reporting? 10 Was the study apparently free from other problems that could cause a high risk of bias?Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Barcelos, K.A.; Mendonça, C.R.; Noll, M.; Botelho, A.F.; Francischini, C.R.D.; Silva, M.A.M. Antitumor Properties of Curcumin in Breast Cancer Based on Preclinical Studies: A Systematic Review. Cancers 2022, 14, 2165. https://doi.org/10.3390/cancers14092165
Barcelos KA, Mendonça CR, Noll M, Botelho AF, Francischini CRD, Silva MAM. Antitumor Properties of Curcumin in Breast Cancer Based on Preclinical Studies: A Systematic Review. Cancers. 2022; 14(9):2165. https://doi.org/10.3390/cancers14092165
Chicago/Turabian StyleBarcelos, Kênia Alves, Carolina Rodrigues Mendonça, Matias Noll, Ana Flávia Botelho, Cristiane Raquel Dias Francischini, and Marco Augusto Machado Silva. 2022. "Antitumor Properties of Curcumin in Breast Cancer Based on Preclinical Studies: A Systematic Review" Cancers 14, no. 9: 2165. https://doi.org/10.3390/cancers14092165
APA StyleBarcelos, K. A., Mendonça, C. R., Noll, M., Botelho, A. F., Francischini, C. R. D., & Silva, M. A. M. (2022). Antitumor Properties of Curcumin in Breast Cancer Based on Preclinical Studies: A Systematic Review. Cancers, 14(9), 2165. https://doi.org/10.3390/cancers14092165








