Impact of BRCA Status on Reproductive Decision-Making and Self-Concept: A Mixed-Methods Study Informing the Development of Tailored Interventions
Abstract
:Simple Summary
Abstract
1. Introduction
2. Materials and Methods
2.1. Participants and Procedures
2.2. Analysis
3. Results
3.1. Clinical and Reproductive Characteristics of BRCA+ Women
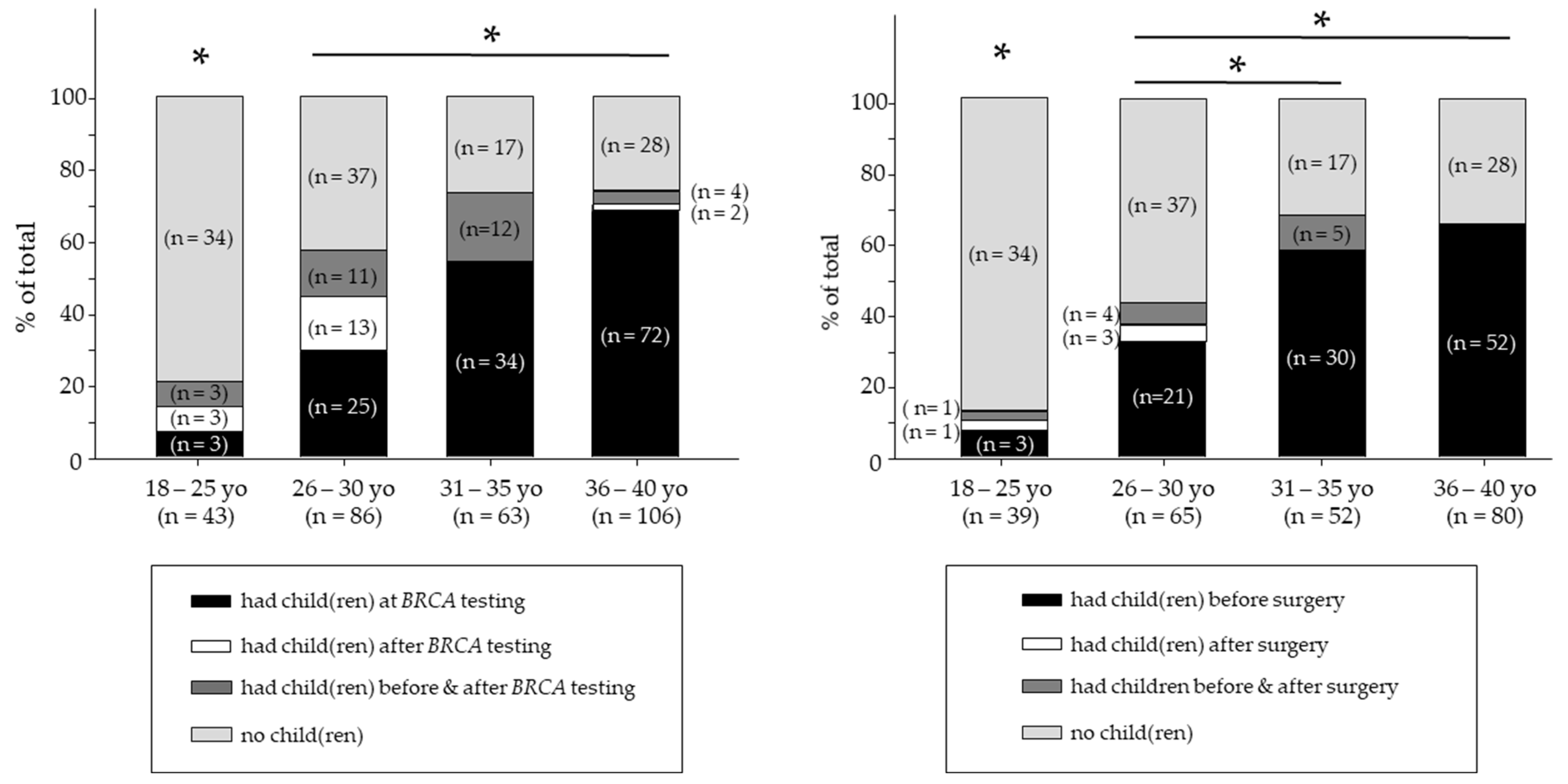
3.2. Relationship between Timing of BRCA Testing, Childbearing Status, and Reproductive Decisions
3.3. Relationship between Risk-Reducing Surgery, Childbearing Status, and Reproductive Decisions
3.4. Self-Concept in BRCA+ Women
3.5. Qualitative Interviews on Learning BRCA Status and Reproductive Decision-Making
3.6. Qualitative Interview Results on Risk-Reducing Surgery and Reproductive Decision-Making
3.7. Self-Concept Related to BRCA Status, Childbearing, and Risk-Reducing Surgery Decisions
3.8. Explanatory Findings Integrating Quantitative and Qualitative Results
4. Discussion
Study Limitations and Future Directions
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Mor, P.; Brennenstuhl, S.; Metcalfe, K.A. Uptake of Preimplantation Genetic Diagnosis in Female BRCA1 and BRCA2 Mutation Carriers. J. Genet. Couns. 2018, 27, 1386–1394. [Google Scholar] [CrossRef] [PubMed]
- Haddad, J.M.; Robison, K.; Beffa, L.; Laprise, J.; ScaliaWilbur, J.; Raker, C.A.; Clark, M.A.; Hofstatter, E.; Dalela, D.; Brown, A.; et al. Family planning in carriers of BRCA1 and BRCA2 pathogenic variants. J. Genet. Couns. 2021, 30, 1570–1581. [Google Scholar] [CrossRef] [PubMed]
- Dean, M.; Rauscher, E.A. Men’s and Women’s Approaches to Disclosure About BRCA-Related Cancer Risks and Family Planning Decision-Making. Qual. Health Res. 2018, 28, 2155–2168. [Google Scholar] [CrossRef] [PubMed]
- Donnelly, L.S.; Watson, M.; Moynihan, C.; Bancroft, E.; Evans, D.G.; Eeles, R.; Lavery, S.; Ormondroyd, E. Reproductive decision-making in young female carriers of a BRCA mutation. Hum. Reprod. 2013, 28, 1006–1012. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- National Cancer Institute. Survelillance, Epidemiology, and End Resultd Program. Cancer Stat Facts. Available online: https://seer.cancer.gov/statfacts/ (accessed on 6 January 2022).
- Kuchenbaecker, K.B.; Hopper, J.L.; Barnes, D.R.; Phillips, K.-A.; Mooij, T.M.; Roos-Blom, M.-J.; Jervis, S.; Van Leeuwen, F.E.; Milne, R.L.; Andrieu, N.; et al. Risks of Breast, Ovarian, and Contralateral Breast Cancer for BRCA1 and BRCA2 Mutation Carriers. JAMA 2017, 317, 2402–2416. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- American Cancer Institute. BRCA Mutations: Cancer Risk and Genetic Testing Fact Sheet. Available online: https://www.cancer.gov/about-cancer/causes-prevention/genetics/brca-fact-sheet#r2 (accessed on 14 December 2021).
- Clinical Practice Guidelines in Oncology (NCCN Guidelines), Genetic/Familial High-Risk Assessment; Breast, Ovarian and Pancreatic. Version 1.2022—11 August 2021. Available online: https://www.nccn.org/professionals/physician_gls/pdf/genetics_bop.pdf (accessed on 10 December 2021).
- Skrovanek, E.; Dunbar-Jacob, J.; Dunwoody, C.; Wesmiller, S. Integrative Review of Reproductive Decision Making of Women Who Are BRCA Positive. J. Obstet. Gynecol. Neonatal Nurs. 2020, 49, 525–536. [Google Scholar] [CrossRef] [PubMed]
- Hesse-Biber, S.; An, C. Genetic Testing and Post-Testing Decision Making among BRCA-Positive Mutation Women: A Psychosocial Approach. J. Genet. Couns. 2016, 25, 978–992. [Google Scholar] [CrossRef]
- Dwyer, A.A.; Hesse-Biber, S.; Flynn, B.; Remick, S. Parent of Origin Effects on Family Communication of Risk in BRCA+ Women: A Qualitative Investigation of Human Factors in Cascade Screening. Cancers 2020, 12, 2316. [Google Scholar] [CrossRef]
- Hesse-Biber, S.; Dwyer, A.A.; Yi, S. Parent of origin differences in psychosocial burden and approach to BRCA risk management. Breast J. 2020, 26, 734–738. [Google Scholar] [CrossRef] [PubMed]
- Seven, M.; Shah, L.L.; Daack-Hirsch, S.; Yazici, H. Experiences of BRCA1/2 Gene Mutation-Positive Women With Cancer in Communicating Genetic Risk to Their Relatives. Cancer Nurs. 2021, 44, E142–E150. [Google Scholar] [CrossRef] [PubMed]
- Mella, S.; Muzzatti, B.; Dolcetti, R.; Annunziata, M.A. Emotional impact on the results of BRCA1 and BRCA2 genetic test: An observational retrospective study. Hered. Cancer Clin. Pract. 2017, 15, 16. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Esplen, M.J.; Stuckless, N.; Hunter, J.; Liede, A.; Metcalfe, K.; Glendon, G.; Narod, S.; Butler, K.; Scott, J.; Irwin, E. The BRCA Self-Concept Scale: A new instrument to measure self-concept in BRCA1/2 mutation carriers. Psycho-Oncology 2009, 18, 1216–1229. [Google Scholar] [CrossRef] [PubMed]
- Lambertini, M.; Ameye, L.; Hamy, A.-S.; Zingarello, A.; Poorvu, P.D.; Carrasco, E.; Grinshpun, A.; Han, S.; Rousset-Jablonski, C.; Ferrari, A.; et al. Pregnancy After Breast Cancer in Patients With Germline BRCA Mutations. J. Clin. Oncol. 2020, 38, 3012–3023. [Google Scholar] [CrossRef] [PubMed]
- Condorelli, M.; Bruzzone, M.; Ceppi, M.; Ferrari, A.; Grinshpun, A.; Hamy, A.; de Azambuja, E.; Carrasco, E.; Peccatori, F.; Di Meglio, A.; et al. Safety of assisted reproductive techniques in young women harboring germline pathogenic variants in BRCA1/2 with a pregnancy after prior history of breast cancer. ESMO Open 2021, 6, 100300. [Google Scholar] [CrossRef] [PubMed]
- Hacker, E.D.; McCarthy, A.M.; Devon, H. Precision Health: Emerging Science for Nursing Research. Nurs. Outlook 2019, 67, 287–289. [Google Scholar] [CrossRef] [Green Version]
- Ryan, J.C.; Viana, J.N.; Sellak, H.; Gondalia, S.; O’Callaghan, N. Defining precision health: A scoping review protocol. BMJ Open 2021, 11, e044663. [Google Scholar] [CrossRef] [PubMed]
- Chadwick, A.; Frazier, A.; Khan, T.W.; Young, E. Understanding the Psychological, Physiological, and Genetic Factors Affecting Precision Pain Medicine: A Narrative Review. J. Pain Res. 2021, 14, 3145–3161. [Google Scholar] [CrossRef]
- Hekler, E.; Tiro, J.A.; Hunter, C.M.; Nebeker, C. Precision Health: The Role of the Social and Behavioral Sciences in Advancing the Vision. Ann. Behav. Med. 2020, 54, 805–826. [Google Scholar] [CrossRef]
- The Strengthening the Reporting of Observational Studies in Epidemiology (STROBE) Statement: Guidelines for Reporting Observational Studies. Available online: https://www.equator-network.org/reporting-guidelines/strobe/ (accessed on 5 January 2022).
- The American College of Obstetricians and Gynecologists Committee on Gynecologic Practice, Female Age-Related Fertility Decline, Committee Opinion, Number 589 (Replaces Committee Opinion Number 413, August 2008. Reaffirmed 2020). Available online: https://www.acog.org/clinical/clinical-guidance/committee-opinion/articles/2014/03/female-age-related-fertility-decline (accessed on 26 February 2022).
- Hsieh, H.-F.; Shannon, S.E. Three Approaches to Qualitative Content Analysis. Qual. Health Res. 2005, 15, 1277–1288. [Google Scholar] [CrossRef] [PubMed]
- IBM SPSS Statistics for Windows; Version 27.0; IBM Corp: Armonk, NY, USA, 2020.
- Bryant, A.; Charmaz, K. Grounded Theory in Historical Perspective: An Epistemological Account. In The SAGE Handbook of Grounded Theory; Bryant, A., Charmaz, K., Eds.; SAGE Publications, Inc.: Thousand Oaks, CA, USA, 2012; pp. 31–57. [Google Scholar]
- Hesse-Biber, S. Gender Differences in Psychosocial and Medical Outcomes Stemming From Testing Positive for the BRCA1/2 Genetic Mutation for Breast Cancer: An Explanatory Sequential Mixed Methods Study. J. Mix. Methods Res. 2018, 12, 280–304. [Google Scholar] [CrossRef]
- Chan, J.L.; Johnson, L.N.C.; Sammel, M.D.; DiGiovanni, L.; Voong, C.; Domchek, S.M.; Gracia, C.R. Reproductive Decision-Making in Women with BRCA1/2 Mutations. J. Genet. Couns. 2017, 26, 594–603. [Google Scholar] [CrossRef] [PubMed]
- Vodermaier, A.; Esplen, M.J.; Maheu, C. Can self-esteem, mastery and perceived stigma predict long-term adjustment in women carrying a BRCA1/2-mutation? Evidence from a multi-center study. Fam. Cancer 2010, 9, 305–311. [Google Scholar] [CrossRef] [PubMed]
- Reumkens, K.; Tummers, M.H.E.; Severijns, Y.; Gietel-Habets, J.J.G.; van Kuijk, S.M.J.; Aalfs, C.M.; van Asperen, C.J.; Ausems, M.G.E.M.; Collée, M.; Dommering, C.J.; et al. Reproductive decision-making in the context of hereditary cancer: The effects of an online decision aid on informed decision-making. J. Community Genet. 2021, 12, 101–110. [Google Scholar] [CrossRef] [PubMed]
- Lambertini, M.; Di Maio, M.; Poggio, F.; Pagani, O.; Curigliano, G.; Del Mastro, L.; Paluch-Shimon, S.; Loibl, S.; Partridge, A.H.; Azim, J.H.A.; et al. Knowledge, attitudes and practice of physicians towards fertility and pregnancy-related issues in youngBRCA-mutated breast cancer patients. Reprod. Biomed. Online 2019, 38, 835–844. [Google Scholar] [CrossRef] [Green Version]
- Baroutsou, V.; Underhill-Blazey, M.; Appenzeller-Herzog, C.; Katapodi, M. Interventions Facilitating Family Communication of Genetic Testing Results and Cascade Screening in Hereditary Breast/Ovarian Cancer or Lynch Syndrome: A Systematic Review and Meta-Analysis. Cancers 2021, 13, 925. [Google Scholar] [CrossRef]
- Heijer, M.D.; Seynaeve, C.; Vanheusden, K.; Duivenvoorden, H.J.; Vos, J.; Bartels, C.C.M.; Menke-Pluymers, M.B.E.; Tibben, A. The contribution of self-esteem and self-concept in psychological distress in women at risk of hereditary breast cancer. Psycho-Oncology 2011, 20, 1170–1175. [Google Scholar] [CrossRef] [PubMed]
- Sebri, V.; Triberti, S.; Pravettoni, G. Injured Self: Autobiographical Memory, Self-Concept, and Mental Health Risk in Breast Cancer Survivors. Front. Psychol. 2020, 11, 607514. [Google Scholar] [CrossRef]
| Quantitative Survey (N = 505) | Qualitative Interview (N = 40) | |
|---|---|---|
| n (%) | n (%) | |
| Age at the time of study | ||
| 18–25 yrs. | 19 (3.8%) | 8 (20%) |
| 26–30 yrs. | 42 (8.3%) | 5 (12.5%) |
| 31–35 yrs. | 71 (14.1%) | 8 (20%) |
| 36–40 yrs. | 96 (19%) | 7 (17.5%) |
| 41–50 yrs. | 156 (30.9%) | 7 (17.5%) |
| 51–60 yrs. | 86 (17%) | 3 (7.5%) |
| 61–70 yrs. | 35 (6.9%) | 2 (5%) |
| Race and ethnicity | ||
| White/Caucasian | 474 (93.9%) | 35 (87.5%) |
| Hispanic and/or Latino | 6 (1.2%) | 1 (2.5%) |
| Black/African-American | 4 (0.8%) | - |
| Asian/Asian-American | 5 (1.0%) | - |
| American Indian/Alaska Native | 2 (0.4%) | - |
| Mixed/Other | 12 (2.4%) | 4 (10%) |
| Marital status | ||
| married | 334 (66.1%) | 26 (65%) |
| single | 170 (33.7%) | 14 (35%) |
| not reported | 1 (<1%) | - |
| Education | ||
| high school | 47 (9.3%) | - |
| some college | 292 (57.8%) | 26 (65%) |
| college/advanced degree | 157 (31.1%) | 14 (35%) |
| not reported | 9 (1.8%) | - |
| Employment | ||
| full time | 298 (59%) | 30 (75%) |
| part-time | 85 (16.8%) | - |
| unemployed | 73 (14.5%) | 2 (5%) |
| student | 17 (3.4%) | 6 (15%) |
| retired | 31 (6.1%) | 2 (5%) |
| other | 1 (<1%) | - |
| Household income (annual) | ||
| >$126,000 | 127 (25.1%) | 6 (15%) |
| $76,000–125,000 | 140 (27.7%) | 18 (45%) |
| <$75,000 | 208 (41.1%) | 4 (10%) |
| not reported | 30 (5.9%) | 12 (30%) |
| Characteristics | n (%) |
|---|---|
| Personal history of cancer | |
| yes | 170 (33.7%) |
| no | 290 (57.4%) |
| not reported | 45 (8.9%) |
| Age at the time of the genetic testing | |
| Of reproductive age (≤40 yrs.) | 307 (60.7%) |
| 18–25 yrs. | 43/307 (14%) |
| 26–30 yrs. | 87/307 (28%) |
| 31–35 yrs. | 69/307 (22%) |
| 36–40 yrs. | 108/307 (36%) |
| Non-reproductive age (40+ yrs.) | 198 (39.2%) |
| 41–50 yrs. | 133/198 (67.1%) |
| 51–60 yrs. | 50/198 (25.2%) |
| 61–70 yrs. | 15/198 (7.5%) |
| Pathogenic BRCA variant | |
| BRCA1 | 236 (46.7%) |
| BRCA2 | 259 (51.3%) |
| BRCA1 & BRCA2 | 10 (2%) |
| History of risk-reducing surgery * | |
| yes | 317 (62.8%) |
| no | 97 (19.2%) |
| other surgery (not risk-reducing) | 39 (7.7%) |
| not reported | 52 (10.3%) |
| Children | |
| biological child(ren) | 324 (64.2%) |
| adopted child(ren) | 16 (3.2%) |
| no children | 165 (32.7%) |
| Stigma (Mean ± SD) | Vulnerability (Mean ± SD) | Mastery (Mean ± SD) | Total (Mean ± SD) | |
|---|---|---|---|---|
| Of reproductive age at the time of BRCA testing | ||||
| ≤40 years-old (n = 312) | 3.20 ± 1.2 | 4.58 ± 1.4 | 5.36 ± 1.1 | 4.11 ± 0.8 |
| 40+ years-old (n = 201) | 3.06 ± 1.4 | 4.22 ± 1.4 | 5.36 ± 1.1 | 3.95 ± 0.8 |
| Z (p value) | −1.41 (0.15) | −2.75 (0.006) | −0.01 (0.99) | −2.23 (0.02) |
| Personal history of cancer | ||||
| yes | 3.11 ± 1.3 | 4.62 ± 1.4 | 5.20 ± 1.1 | 4.05 ± 0.8 |
| no | 3.17 ± 1.3 | 4.32 ± 1.4 | 5.46 ± 1.1 | 4.05 ± 0.8 |
| Z (p value) | −0.50 (0.61) | −2.28 (0.02) | −2.84 (0.005) | −0.18 (0.85) |
| Pathogenic BRCA variant | ||||
| BRCA1 | 3.14 ± 1.3 | 4.38 ± 1.5 | 5.40 ± 1.2 | 4.03 ± 0.8 |
| BRCA2 | 3.19 ± 1.3 | 4.50 ± 1.3 | 5.33 ± 1.1 | 4.08 ± 0.8 |
| BRCA1 & BRCA2 | 2.80 ± 1.2 | 4.33 ± 1.3 | 5.25 ± 0.9 | 3.83 ± 0.7 |
| H (p value) | 0.71 (0.70) | 0.36 (0.83) | 1.52 (0.46) | 0.99 (0.60) |
| Childbearing status and BRCA testing † | ||||
| child(ren) before testing | 3.12 ± 1.3 | 4.61 ± 1.4 | 5.32 ± 1.1 | 4.08 ± 0.8 |
| child(ren) after testing | 3.49 ± 1.3 | 4.97 ± 1.3 | 5.41 ± 1.2 | 4.37 ± 0.7 |
| child(ren) before & after testing | 2.95 ± 1.3 | 4.56 ± 1.4 | 5.34 ± 1.1 | 3.97 ± 0.8 |
| no children | 3.23 ± 1.3 | 4.14 ± 1.4 | 5.39 ± 1.2 | 4.00 ± 0.8 |
| H (p value) | 2.22 (0.52) | 11.58 (0.009) a | 0.63 (0.88) | 3.47 (0.32) |
| Childbearing status and risk-reducing surgery †† | ||||
| child(ren) before surgery | 3.00 ± 1.3 | 4.59 ± 1.4 | 5.33 ± 1.1 | 4.02 ± 0.8 |
| child(ren) after surgery | 3.08 ± 1.8 | 4.73 ± 1.2 | 6.00 ± 0.6 | 4.25 ± 0.9 |
| child(ren) before & after surgery | 3.23 ± 0.9 | 4.54 ± 1.0 | 5.37 ± 1.1 | 4.12 ± 0.5 |
| no children | 3.23 ± 1.3 | 4.14 ± 1.4 | 5.39 ± 1.2 | 4.00 ± 0.8 |
| H (p-value) | 3.13 (0.37) | 7.81 (0.05) | 1.07 (0.78) | 0.61 (0.89) |
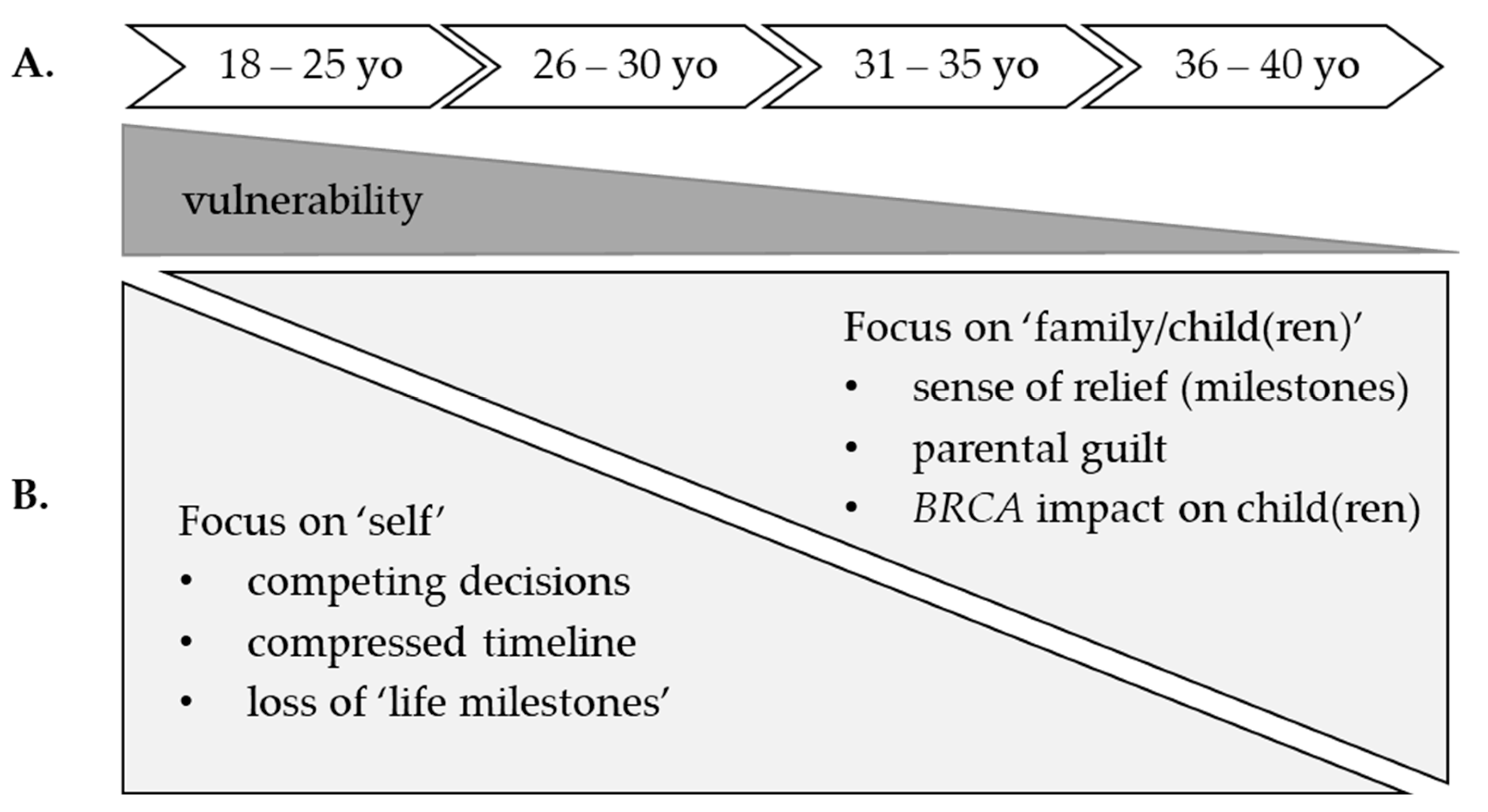
| Quantitative Survey Significant Findings | Qualitative Interviews Representative Quotes Depicting Women’s ‘Lived Experience’ |
| A. Childbearing status at the time of BRCA testing | |
| Group 1: “I was pretty upset for a few months, but I was also like ‘I’m done having my kids, I’ve done that. I’m done, I’ve nursed my kids… my babies’. You know, they’re growing up… I’m done with that part of my life.” (Interviewee #5) “I like always hated kids, but now... How do I know five years from now? I’m not gonna want a kid or have kids… Like a lot of my friends are older than me, and they are getting engaged, getting married, or they’re having kids. And I’m like crap, like, maybe I do want to do that... I feel like I would be thinking about that stuff a lot later if I didn’t know about BRCA.” (Interviewee #1) Group 2: “I’ve had friends that have said, ‘Well, I can’t believe that you’d want to have kids with that [BRCA].’ And I’m like…it kind of shocks me sometimes because I’m like, ‘Really’? You think this is the worst thing I could have?’ I kind of think that you know…I look at some of the other things you could have, and I’m like, ‘I’ll take my BRCA, and I’ll be happy about it.” (Interviewee #15) Group 3: “I didn’t know if I would have had a third child or not. I think going through this process… it was ultimately my choice to have another one, and I knew when I was turning thirty-five, I wasn’t gonna have that choice anymore. So, we… we ended up um… having another child eighteen months later…I always wanted a bunch of kids.” (Interviewee #6) Group 4: “My husband and I have been together for about 13 years now, and neither of us had ever had the inkling to want children in our entire life. We already knew, even before we found out about [BRCA], that we weren’t going to have kids. It kind of seals the deal for us, knowing that I could pass this on to them, and I definitely wouldn’t want to do that. So, it kind of helped solidify the decision not to have children for us…. I want to do a hysterectomy.” (Interviewee #20) |
| B. Weighing risk-reducing surgery and childbearing | |
| Group 1:“For me, I always knew that I would probably test [BRCA], but I had… it had always been in the back of my mind, and I wanted to finish my family first. Because I didn’t know necessarily what steps I would want to take once I found out. But at the same time, it was like… I have two kids that I have to worry about now that I need to figure out … My husband and I had a million and one conversations, and we decided to try for another child... If we didn’t get pregnant … [then] I was going to figure out my surgical option. So, we [had] another baby … I had my salpingectomy at six weeks post-partum. At thirty-four, I’m not ready to go through menopause, and so I’m going to breastfeed this baby until she’s six months old … and I’m hoping to do my mastectomy in December.” (Interviewee #12) Group 2: “My boyfriend and I... have a ring. We’re getting engaged in the next few weeks. So, I want to get married, and I know I want to have one kid. I’ve always felt that way. I just want to have one kid. So when I get that… in like, the next three to four years… not out of the way, but for lack of a better term. I then think that I will go ahead and have the surgery. So, sometime in my thirties, my early thirties… I want to have the surgery.” (Interviewee #13) Group 3: “That [not being able to breastfeed] was something else that I had to… like, mourn and come to terms with. You know? Having her [another child] after all of this and not having that option to breastfeed.” (Interviewee #8) Group 4: “And then, eventually I realized… you know what I mean? If I’m not going to have a baby… why? And with my horrible history… why not do it [surgery] you know?” (Interviewee #7) |
| C. BRCA+ status and self-concept | |
| “I feel like there’s multiple ticking time bombs. I feel like there’s a ticking time bomb related to my fertility and when I need to… need to meet someone, get married, have children.” (Interviewee #19) “I have no idea how to tell them [daughters]. What I do know is, I don’t want them [doctors] testing them [daughters] until they’re in their thirties, because I don’t want them [daughters] picking these guys that are the wrong guys and having children early because they’re worried about it [BRCA]” (Interviewee #3) “Thinking about, um, when to tell her [daughter]. When… when to push her to get tested? Things like that. It’s really about… like, when do you tell them? It’s almost like… you know, adopting a child. When do you tell them something like this?” (Interviewee #2) |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Hesse-Biber, S.; Seven, M.; Jiang, J.; Schaik, S.V.; Dwyer, A.A. Impact of BRCA Status on Reproductive Decision-Making and Self-Concept: A Mixed-Methods Study Informing the Development of Tailored Interventions. Cancers 2022, 14, 1494. https://doi.org/10.3390/cancers14061494
Hesse-Biber S, Seven M, Jiang J, Schaik SV, Dwyer AA. Impact of BRCA Status on Reproductive Decision-Making and Self-Concept: A Mixed-Methods Study Informing the Development of Tailored Interventions. Cancers. 2022; 14(6):1494. https://doi.org/10.3390/cancers14061494
Chicago/Turabian StyleHesse-Biber, Sharlene, Memnun Seven, Jing Jiang, Sara Van Schaik, and Andrew A. Dwyer. 2022. "Impact of BRCA Status on Reproductive Decision-Making and Self-Concept: A Mixed-Methods Study Informing the Development of Tailored Interventions" Cancers 14, no. 6: 1494. https://doi.org/10.3390/cancers14061494
APA StyleHesse-Biber, S., Seven, M., Jiang, J., Schaik, S. V., & Dwyer, A. A. (2022). Impact of BRCA Status on Reproductive Decision-Making and Self-Concept: A Mixed-Methods Study Informing the Development of Tailored Interventions. Cancers, 14(6), 1494. https://doi.org/10.3390/cancers14061494








