CONUT Score: A New Tool for Predicting Prognosis in Patients with Advanced Thyroid Cancer Treated with TKI
Abstract
:Simple Summary
Abstract
1. Introduction
2. Materials and Methods
2.1. Study Population
2.2. Assessments and Definitions
- for serum albumin levels >3.5, between 3.0 and 3.49, between 2.5 and 2.99 and <2.5 g/dl, 0, 2, 4, and 6 points were assigned, respectively.
- for serum total cholesterol levels >180, between 140 and 179, between 100 and 139 and <100 mg/dl, 0, 1, 2, and 3 points were assigned, respectively.
- for serum total lymphocyte count >1600, between 1200 and 1599, between 800 and 1199 and <800 / mm3, 0, 1, 2, and 3 points were assigned, respectively.
2.3. Statistical Analysis
3. Results
3.1. Baseline CONUT Score (before TKI Treatment)
3.2. TKI Treatment
3.3. Correlation between Baseline CONUT Score and Response to TKI Treatment
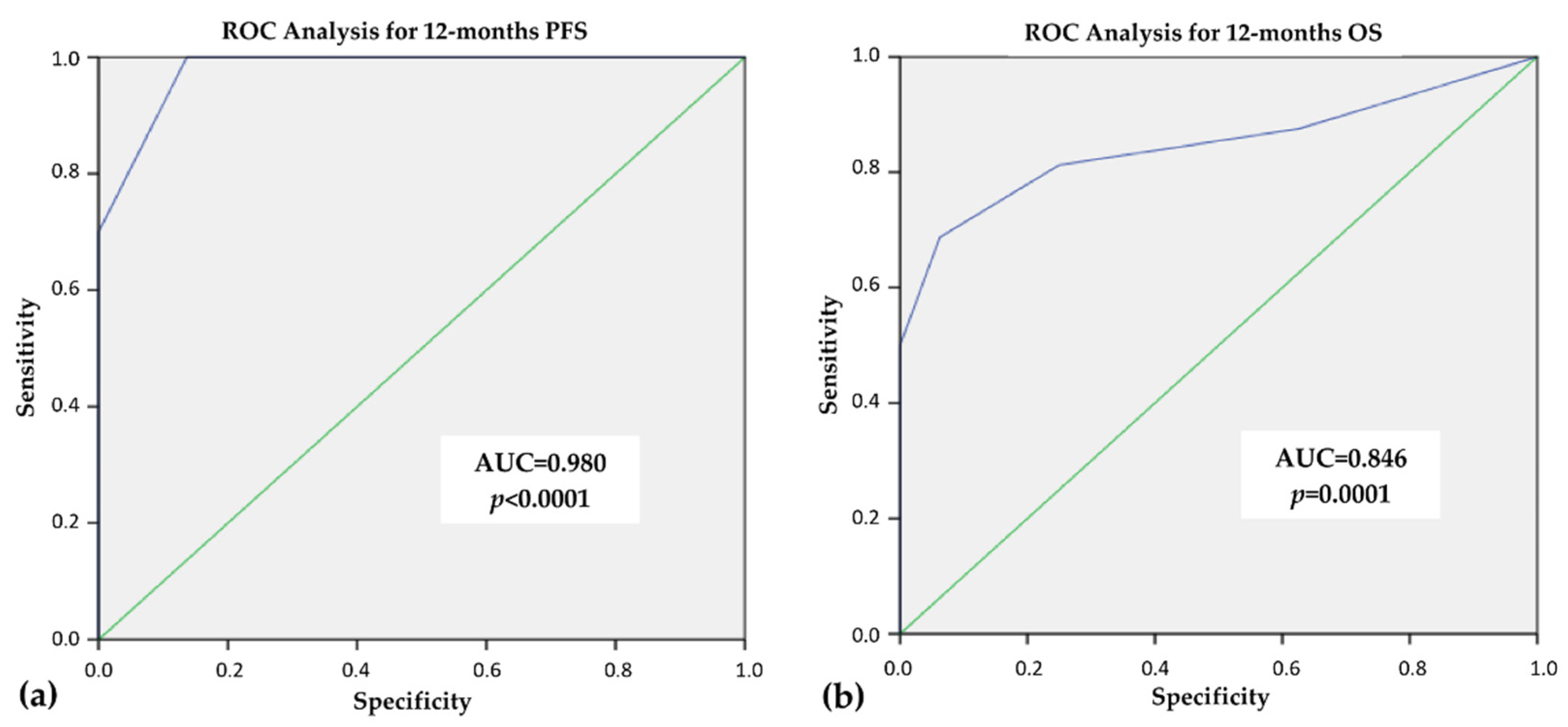
3.4. ROC Analysis for 12 Months—PFS and OS
3.5. Clinical-Pathological Features in Thyroid Cancer Patients with CONUT Score <3 (Group 1) and ≥3 (Group 2)
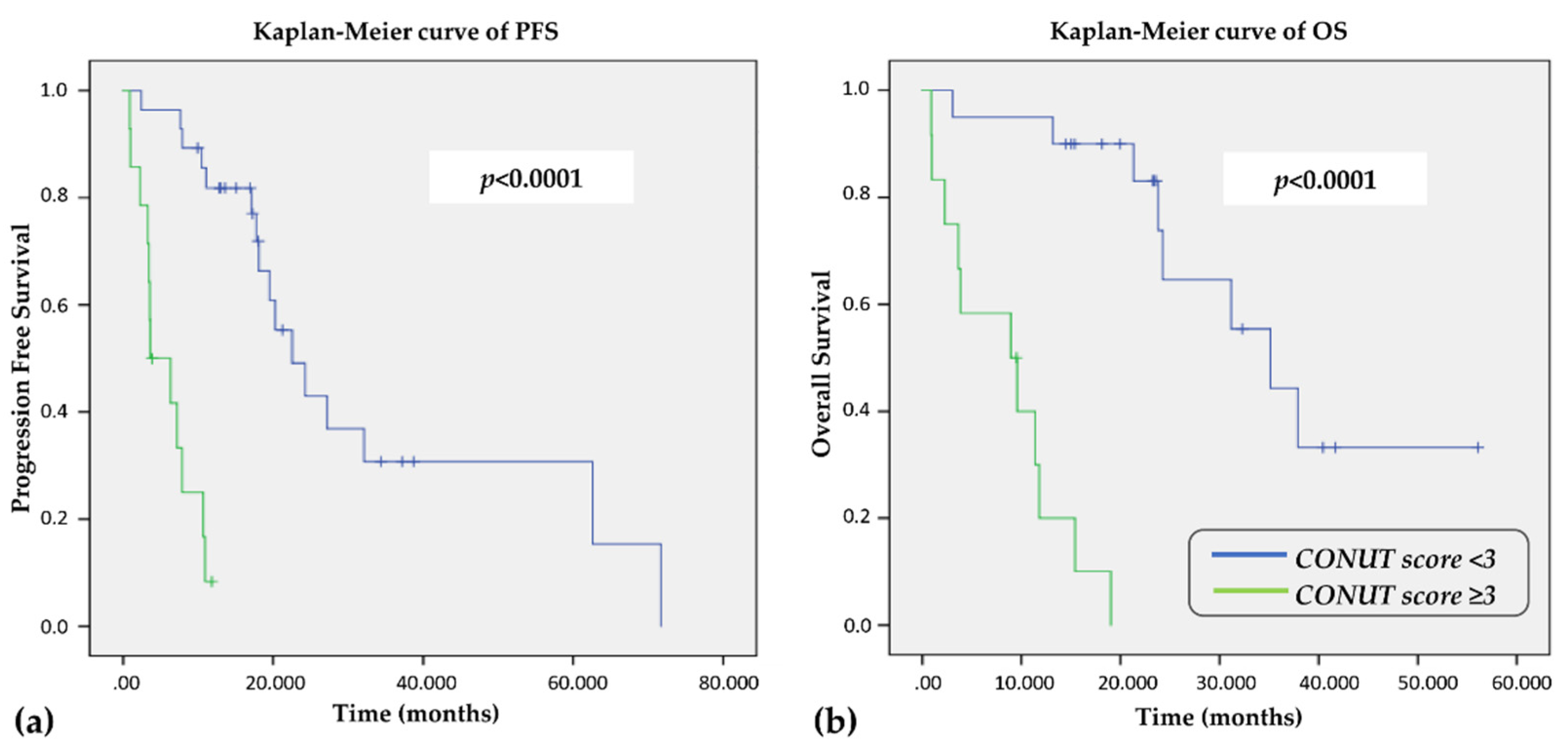
3.6. Clinical Outcomes According to CONUT Score
3.7. Univariate and Multivariate Analysis for PFS and OS
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Gotink, K.J.; Verheul, H.M. Anti-angiogenic tyrosine kinase inhibitors: What is their mechanism of action? Angiogenesis 2010, 13, 1–14. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Roskoski, R., Jr. Properties of FDA-approved small molecule protein kinase inhibitors: A 2021 update. Pharmacol. Res. 2021, 165, 105463. [Google Scholar] [CrossRef] [PubMed]
- Wells, S.A., Jr.; Robinson, B.G.; Gagel, R.F.; Dralle, H.; Fagin, J.A.; Santoro, M.; Schlumberger, M.J.; Read, J.; Langmuir, P.; Ryan, A.J.; et al. Vandetanib in patients with locally advanced or metastatic medullary thyroid cancer: A randomized, double-blind phase III trial. J. Clin. Oncol. 2012, 30, 134–141. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Elisei, R.; Schlumberger, M.; Müller, S.P.; Schöffski, P.; Brose, M.S.; Shah, M.H.; Sherman, S.I.; Yaron, Y.; Ball, D.; Nelkin, B.; et al. Cabozantinib in progressive medullary thyroid cancer. J. Clin. Oncol. 2013, 31, 3639–3646. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Brose, M.S.; Nutting, C.M.; Jarzab, B.; Elisei, R.; Siena, S.; Bastholt, L.; Schlumberger, M.J.; Kappeler, C.; Peña, C.; Molnár, I.; et al. Sorafenib in radioactive iodine-refractory, locally advanced or metastatic differentiated thyroid cancer: A randomized, double-blind, phase 3 trial. Lancet 2014, 384, 319–328. [Google Scholar] [CrossRef] [Green Version]
- Schlumberger, M.; Tahara, M.; Wirth, L.J.; Robinson, B.; Brose, M.S.; Elisei, R.; Sherman, S.I.; Dutcus, C.E.; de las Heras, B.; Zhu, J.; et al. Lenvatinib versus placebo in radioiodine-refractory thyroid cancer. N. Engl. J. Med. 2015, 372, 621–630. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Subbiah, V.; Kreitman, R.J.; Wainberg, Z.A.; Cho, J.Y.; Schellens, J.H.M.; Soria, J.C.; Wen, P.Y.; Zielinski, C.; Cabanillas, M.E.; Urbanowitz, G.; et al. Dabrafenib and Trametinib Treatment in Patients With Locally Advanced or Metastatic BRAF V600-Mutant Anaplastic Thyroid Cancer. J. Clin. Oncol. 2018, 36, 7–13. [Google Scholar] [CrossRef] [Green Version]
- Cabanillas, M.E.; Ryder, M.; Jimenez, C. Targeted Therapy for Advanced Thyroid Cancer: Kinase Inhibitors and Beyond. Endocr. Rev. 2019, 40, 1573–1604. [Google Scholar] [CrossRef] [Green Version]
- Reber, E.; Schönenberger, K.A.; Vasiloglou, M.F.; Stanga, Z. Nutritional Risk Screening in Cancer Patients: The First Step Toward Better Clinical Outcome. Front. Nutr. 2021, 8, 152. [Google Scholar] [CrossRef]
- Wu, N.; Chen, G.; Hu, H.; Pang, L.; Chen, Z. Low pretherapeutic serum albumin as a risk factor for poor outcome in esophageal squamous cell carcinomas. Nutr. Cancer 2015, 67, 481–485. [Google Scholar] [CrossRef]
- Cengiz, O.; Kocer, B.; Surmeli, S.; Santicky, M.J.; Soran, A. Are pretreatment serum albumin and cholesterol levels prognostic tools in patients with colorectal carcinoma? Med. Sci. Monit. Basic Res. 2006, 12, 240–247. [Google Scholar]
- Onate-Ocana, L.F.; Aiello-Crocifoglio, V.; Gallardo-Rincon, D.; Herrera-Goepfert, R.; Brom-Valladares, R.; Carrillo, J.F.; Cervera, E.; Mohar-Betancourt, A. Serum albumin as a significant prognostic factor for patients with gastric carcinoma. Ann. Surg. Oncol. 2007, 14, 381–389. [Google Scholar] [CrossRef] [PubMed]
- Sun, K.; Chen, S.; Xu, J.; Li, G.; He, Y. The prognostic significance of the prognostic nutritional index in cancer: A systematic review and meta-analysis. J. Cancer Res. Clin. Oncol. 2014, 140, 1537–1549. [Google Scholar] [CrossRef] [PubMed]
- Arfsten, H.; Cho, A.; Prausmüller, S.; Spinka, G.; Novak, J.; Goliasch, G.; Pavo, N.; Preusser, M.; Hengstenberg, C.; Hülsmann, M.; et al. Inflammation-Based Scores as a Common Tool for Prognostic Assessment in Heart Failure or Cancer. Front. Cardiovasc. Med. 2021, 8, 725903. [Google Scholar] [CrossRef] [PubMed]
- Ignacio de Ulíbarri, J.; González-Madroño, A.; De Villar, N.G.; González, P.; Gonzalez, B.; Mancha, A.; Rodriguez, F.; Fernandez, G. CONUT: A tool for Controlling Nutritional Status. First validation in a hospital population. Nutr. Hosp. 2005, 20, 38–45. [Google Scholar] [PubMed]
- Shirakabe, A.; Hata, N.; Kobayashi, N.; Okazaki, H.; Matsushita, M.; Shibata, Y.; Nishigoori, S.; Uchiyama, S.; Asai, K.; Shimizu, W. The prognostic impact of malnutrition in patients with severely decompensated acute heart failure, as assessed using the Prognostic Nutritional Index (PNI) and Controlling Nutritional Status (CONUT) score. Heart Vessel. 2018, 33, 134–144. [Google Scholar] [CrossRef]
- Fukushima, K.; Ueno, Y.; Kawagishi, N.; Kondo, Y.; Inoue, J.; Kakazu, E.; Ninomiya, M.; Wakui, Y.; Saito, N.; Satomi, S.; et al. The nutritional index ‘CONUT’ is useful for predicting long-term prognosis of patients with end-stage liver diseases. Tohoku J. Exp. Med. 2011, 224, 215–219. [Google Scholar] [CrossRef] [Green Version]
- Sun, X.; Luo, L.; Zhao, X.; Ye, P. Controlling Nutritional Status (CONUT) score as a predictor of all-cause mortality in elderly hypertensive patients: A prospective follow-up study. BMJ Open 2017, 7, 015649. [Google Scholar] [CrossRef] [PubMed]
- Zhou, H.; Chao, W.; Cui, L.; Li, M.; Zou, Y.; Yang, M. Controlling Nutritional Status (CONUT) score as immune-nutritional predictor of outcomes in patients undergoing peritoneal dialysis. Clin. Nutr. 2020, 39, 2564–2570. [Google Scholar] [CrossRef]
- Yılmaz, A.; Tekin, S.B.; Bilici, M.; Yılmaz, H. The Significance of Controlling Nutritional Status (CONUT) Score as a Novel Prognostic Parameter in Small Cell Lung Cancer. Lung 2020, 198, 695–704. [Google Scholar] [CrossRef]
- Zhang, Y.; Zhang, X. Controlling nutritional status score, a promising prognostic marker in patients with gastrointestinal cancers after surgery: A systematic review and meta-analysis. Int. J. Surg. 2018, 55, 39–45. [Google Scholar] [CrossRef] [PubMed]
- Takagi, K.; Buettner, S.; Ijzermans, J.N.M. Prognostic significance of the controlling nutritional status (CONUT) score in patients with colorectal cancer: A systematic review and meta-analysis. Int. J. Surg. 2020, 78, 91–96. [Google Scholar] [CrossRef] [PubMed]
- Terasaki, F.; Sugiura, T.; Okamura, Y.; Ito, T.; Yamamoto, Y.; Ashida, R.; Uesaka, K.; Ohgi, K. The preoperative controlling nutritional status (CONUT) score is an independent prognostic marker for pancreatic ductal adenocarcinoma. Updates Surg. 2021, 73, 251–259. [Google Scholar] [CrossRef] [PubMed]
- Li, Y.; Zhang, C.; Ji, R.; Lu, H.; Zhang, W.; Li, L.L.; He, A.; Liu, R.; Qian, H. Prognostic significance of the controlling nutritional status (CONUT) score in epithelial ovarian cancer. Int. J. Gynecol. Cancer 2020, 30, 74–82. [Google Scholar] [CrossRef] [PubMed]
- Li, W.; Li, M.; Wang, T.; Ma, G.; Deng, Y.; Pu, D.; Zhou, Q.; Liu, Z.; Wu, Q.; Liuet, X. Controlling Nutritional Status (CONUT) score is a prognostic factor in patients with resected breast cancer. Sci. Rep. 2020, 10, 6633. [Google Scholar] [CrossRef]
- Niu, X.; Zhu, Z.; Bao, J. Prognostic significance of pretreatment controlling nutritional status score in urological cancers: A systematic review and meta-analysis. Cancer Cell Int. 2021, 21, 126. [Google Scholar] [CrossRef]
- Ahn, S.S.; Yoo, J.; Jung, S.M.; Song, J.J.; Park, Y.B.; Lee, S.W. Comparison of the Clinical Implications among Five Different Nutritional Indices in Patients with Lupus Nephritis. Nutrients 2019, 11, 1456. [Google Scholar] [CrossRef] [Green Version]
- Cabré, M.; Ferreiro, C.; Arus, M.; Roca, M.; Palomera, E.; Serra-Prat, M. Evaluation of CONUT for clinical malnutrition detection and shorttermprognostic assessment in hospitalized elderly people. J. Nutr. Health Aging 2015, 19, 729–733. [Google Scholar] [CrossRef]
- Shimose, S.; Kawaguchi, T.; Iwamoto, H.; Tanaka, M.; Miyazaki, K.; Ono, M.; Torimura, T.; Koga, H.; Noguchi, K.; Yokokura, Y.; et al. Controlling Nutritional Status (CONUT) Score is Associated with Overall Survival in Patients with Unresectable Hepatocellular Carcinoma Treated with Lenvatinib: A Multicenter Cohort Study. Nutrients 2020, 12, 1076. [Google Scholar] [CrossRef]
- Coussens, L.M.; Werb, Z. Inflammation and cancer. Nature 2002, 420, 860–867. [Google Scholar] [CrossRef]
- Zitvogel, L.; Pietrocola, F.; Kroemer, G. Nutrition, inflammation and cancer. Nat. Immunol. 2017, 18, 843–850. [Google Scholar] [CrossRef] [PubMed]
- Diakos, C.I.; Charles, K.A.; McMillan, D.C.; Clarke, S.J. Cancer-related inflammation and treatment effectiveness. Lancet Oncol. 2014, 15, 493–503. [Google Scholar] [CrossRef]
- Mantzorou, M.; Koutelidakis, A.; Theocharis, S.; Giaginis, C. Clinical Value of Nutritional Status in Cancer: What is its Impact and how it Affects Disease Progression and Prognosis? Nutr. Cancer 2017, 69, 1151–1176. [Google Scholar] [CrossRef] [PubMed]
- Trinh, H.; Dzul, S.P.; Hyder, J.; Hyejeong, J.; Jang, H.; Kim, S.; Flowers, J.; Miller, S.; Chen, J.; Winer, I. Prognostic value of changes in neutrophil-to-lymphocyte ratio (NLR), platelet-to-lymphocyte ratio (PLR) and lymphocyte-to-monocyte ratio (LMR) for patients with cervical cancer undergoing definitive chemoradiotherapy (dCRT). Clin. Chim. Acta 2020, 510, 711–716. [Google Scholar] [CrossRef] [PubMed]
- Chen, Y.; Wang, W.; Zhang, X.; Xiangyang, Y.; Xi, K.; Wen, Y.; Zhang, L.; Wang, G.; Feng, X. Prognostic significance of combined preoperative platelet-to-lymphocyte ratio and lymphocyte-to-monocyte ratio in patients undergoing surgery with stage IB non-small-cell lung cancer. Cancer Manag. Res. 2018, 10, 5411–5422. [Google Scholar] [CrossRef] [Green Version]
- Lee, S.M.; Russell, A.; Hellawell, G. Predictive value of pretreatment inflammation-based prognostic scores (neutrophil-to-lymphocyte ratio, platelet-to-lymphocyte ratio, and lymphocyte-to-monocyte ratio) for invasive bladder carcinoma. Korean J. Urol. 2015, 56, 749–755. [Google Scholar] [CrossRef] [Green Version]
- Yan, L.; Nakamura, T.; Casadei-Gardini, A.; Bruixola, G.; Huang, Y.L.; Hu, Z.D. Long-term and short-term prognostic value of the prognostic nutritional index in cancer: A narrative review. Ann. Transl. Med. 2021, 9, 1630. [Google Scholar] [CrossRef]
- Kheirouri, S.; Alizadeh, M. Prognostic Potential of the Preoperative Controlling Nutritional Status (CONUT) Score in Predicting Survival of Patients with Cancer: A Systematic Review. Adv. Nutr. 2021, 12, 234–250. [Google Scholar] [CrossRef]
- Matsukawa, T.; Suto, K.; Kanaya, M.; Izumiyama, K. North Japan Hematology Study Group (NJHSG). Validation and comparison of prognostic values of GNRI, PNI, and CONUT in newly diagnosed diffuse large B cell lymphoma. Ann. Hematol. 2020, 99, 2859–2868. [Google Scholar] [CrossRef]
- Okamoto, S.; Ureshino, H.; Kidoguchi, K.; Kusaba, K.; Kizuka-Sano, H.; Sano, H.; Kimura, S.; Kojima, K.; Kawaguchi, A.; Sueoka, E.; et al. Clinical impact of the CONUT score in patients with multiple myeloma. Ann. Hematol. 2020, 99, 113–119. [Google Scholar] [CrossRef]
- Peters, S.J.; Vanhaecke, T.; Papeleu, P.; Rogiers, V.; Haagsman, H.P.; van Norren, K. Co-culture of primary rat hepatocytes with rat liver epithelial cells enhances interleukin-6-induced acute-phase protein response. Cell Tissue Res. 2010, 340, 451–457. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Honda, H.; Qureshi, A.R.; Heimburger, O.; Barany, P.; Wang, K.; Pecoits-Filho, R.; Lindholm, B.; Stenvinkel, P. Serum albumin, C-reactive protein, interleukin 6, and fetuin a as predictors of malnutrition, cardiovascular disease, and mortality in patients with ESRD. Am. J. Kidney Dis. 2006, 47, 139–148. [Google Scholar] [CrossRef] [PubMed]
- Op den Kamp, C.M.; Langen, R.C.; Snepvangers, F.J.; de Theije, C.C.; Schellekens, J.M.; Laugs, F.; Schols, A.M.; Dingemans, A.-M.C. Nuclear transcription factor κ B activation and protein turnover adaptations in skeletal muscle of patients with progressive stages of lung cancer cachexia. Am. J. Clin. Nutr. 2013, 98, 738–748. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Banks, W.A. Anorectic effects of circulating cytokines: Role of the vascular blood-brain barrier. Nutrition 2001, 17, 434–437. [Google Scholar] [CrossRef]
- Law, D.K.; Dudrick, S.J.; Abdou, N.I. Immunocompetence of patients with protein-calorie malnutrition. The effects of nutritional repletion. Ann. Intern. Med. 1973, 79, 545–550. [Google Scholar] [CrossRef] [PubMed]
- Menendez, J.A.; Lupu, R. Fatty acid synthase and the lipogenic phenotype in cancer pathogenesis. Nat. Rev. Cancer 2007, 7, 763–777. [Google Scholar] [CrossRef] [PubMed]
- Vitols, S.; Gahrton, G.; Björkholm, M.; Peterson, C. Hypocholesterolaemia in malignancy due to elevated low-density-lipoprotein-receptor activity in tumour cells: Evidence from studies in patients with leukaemia. Lancet 1985, 2, 1150–1154. [Google Scholar] [CrossRef]
- Henriksson, P.; Eriksson, M.; Ericsson, S.; Rudling, M.; Berglund, L.; Angelin, B. Hypocholesterolaemia and increased elimination of low-density lipoproteins in metastatic cancer of the prostate. Lancet 1989, 2, 1178–1180. [Google Scholar] [CrossRef]
- Okuyama, H.; Ichikawa, Y.; Sun, Y.; Hamazaki, T.; Lands, W.E. Cancer and all-cause mortalities are lower in the higher total cholesterol groups among general populations. World Rev. Nutr. Diet. 2007, 96, 37–54. [Google Scholar]
- Li, J.R.; Zhang, Y.; Zheng, J.L. Decreased pretreatment serum cholesterol level is related with poor prognosis in resectable non-small cell lung cancer. Int. J. Clin. Exp. Pathol. 2015, 8, 11877–11883. [Google Scholar]
- Shin, H.J.; Roh, C.K.; Son, S.Y.; Hoon, H.; Han, S.U. Prognostic value of hypocholesterolemia in patients with gastric cancer. Asian J. Surg. 2021, 44, 72–79. [Google Scholar] [CrossRef] [PubMed]
- Lee, H.; Jeong, C.W.; Kwak, C.; Kim, H.H.; Seo, S.I.; Lee, H.M.; Byun, S.S.; Lee, S.C.; Hong, S.K.; Lee, S.E.; et al. Preoperative Cholesterol Level Is Associated With Worse Pathological Outcomes and Postoperative Survival in Localized Renal Cell Carcinoma Patients: A Propensity Score-Matched Study. Clin. Genitourin. Cancer 2017, 15, 935–941. [Google Scholar] [CrossRef] [PubMed]
- Kang, R.; Li, P.; Wang, T.; Li, X.; Wei, Z.; Zhang, Z.; Chen, X.F.; Xu, H.; Huang, C.; Bu, G.; et al. Apolipoprotein E epsilon 2 allele and low serum cholesterol as risk factors for gastric cancer in a Chinese Han population. Sci. Rep. 2016, 6, 19930. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Chiba, T.; Ohtani, H.; Mizoi, T.; Naito, Y.; Sato, E.; Nagura, H.; Satomi, S.; Ohuchi, K.; Shiiba, K.; Kurokawa, Y.; et al. Intraepithelial CD8+ T-cell-count becomes a prognostic factor after a longer follow-up period in human colorectal carcinoma: Possible association with suppression of micrometastasis. Br. J. Cancer 2004, 91, 1711–1717. [Google Scholar] [CrossRef]
- Nakano, O.; Sato, M.; Naito, Y.; Suzuki, K.; Orikasa, S.; Aizawa, M.; Ohtani, H.; Suzuki, Y.; Shintaku, I.; Nagura, H. Proliferative activity of intratumoral CD8(+) T-lymphocytes as a prognostic factor in human renal cell carcinoma: Clinicopathologic demonstration of antitumor immunity. Cancer Res. 2001, 61, 5132–5136. [Google Scholar]
- Schumacher, K.; Haensch, W.; Röefzaad, C.; Schlag, P.M. Prognostic significance of activated CD8(+) T cell infiltrations within esophageal carcinomas. Cancer Res. 2001, 61, 3932–3936. [Google Scholar]
| All Patients | DTC | PDTC | MTC | |
|---|---|---|---|---|
| (n = 42) | (n = 28) | (n = 9) | (n = 5) | |
| Gender n (%) | ||||
| F | 23 (54.8%) | 18 (64.3%) | 3 (33.3%) | 2 (40%) |
| M | 19 (45.2%) | 10 (35.7%) | 6 (66.7%) | 3 (60%) |
| Age at the time of TKI treatment (years) Median (range) | 69 (30–9) | 71.6 (43–87) | 67.2 (51.2–96.2) | 51.4 (30–80) |
| Time-lapse between cancer diagnosis and TKI treatment (years) Median (range) | 5.4 (0.06–15.4) | 5.71 (0.09–15.4) | 1.96 (0.06–12) | 7.7 (1.1–9.9) |
| Time-lapse between appearance of metastases and TKI treatment (years) Median (range) | 2.8 (0–14.2) | 3.2 (0.03–14.2) | 1.2 (0–12) | 1.1 (0.6–4.9) |
| Numbers of anatomical site involved n (%) | ||||
| 1 | 5 (11.9%) | 4 (14.3%) | 1 (11.1%) | 0 (0%) |
| 2 | 10 (23.8%) | 8 (28.6%) | 1 (11.1%) | 1 (20%) |
| ≥3 | 27 (64.3%) | 16 (57.1%) | 7 (77.8%) | 4 (80%) |
| Patients with bone metastasis n (%) | 15 (35.7%) | 9 (32.1%) | 3 (33.3%) | 3 (60%) |
| Type of first-line TKI n (%) | ||||
| Lenvatinib | 16 (38.1%) | 10 (35.7%) | 6 (66.7%) | 0 (0%) |
| Sorafenib | 18 (42.8%) | 14 (50%) | 3 (33.3%) | 1 (20%) |
| Vandetanib | 4 (9.5%) | 2 (7.1%) | 0 (0%) | 2 (40%) |
| Motesanib | 4 (9.5%) | 2 (7.1%) | 0 (0%) | 2 (40%) |
| BMI (kg/m2) Median (range) | 26.5 (18.1–47) | 26.9 (18.7–44.1) | 24.6 (18–46.9) | 24.8 (22.7–34.4) |
| ECOG PS n (%) 1 | ||||
| 0 | 31 (86.1%) | 20 (83.33%) | 7 (87.5%) | 4 (100%) |
| 1 | 3 (8.3%) | 2 (8.33%) | 1 (12.5%) | 0 (0%) |
| 2 | 2 (5.6%) | 2 (8.33%) | 0 (0%) | 0 (0%) |
| Patients (n; %) | BR1 (n; %) | PFS 1 Median (Months) Range | OS 2 Median (Months) Range | |
|---|---|---|---|---|
| Group A (CONUT score 0–2) | 28 (66.7%) | PR 10/27 (37%) | 17.9 (2.4–71.7) | 23.5 (3.1–56.1) |
| SD 17/27 (63%) | ||||
| PD 0/27 (0%) | ||||
| Group B (CONUT score 3–4) | 9 (21.4%) | PR 3/8 (37.5%) | 3.6 (0.9–11.8) | 8.9 (0.9–19.0) |
| SD 2/8 (25%) | ||||
| PD 3/8 (37.5%) | ||||
| Group C (CONUT score 5–7) | 5 (11.9%) | PR 1/5 (20%) | 7.9 (2.2–10.9) | 9.6 (2.2–15.4) |
| SD 2/5 (40%) | ||||
| PD 2/5 (40%) | ||||
| p A vs. B = 0.002 | p A vs. B = 0.001 | p A vs. B = 0.0006 | ||
| p A vs. C = 0.003 | p A vs. C = 0.0224 | p A vs. C = 0.0028 | ||
| p B vs. C = 0.759 | p B vs. C = 0.737 | p B vs. C = 0.923 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Dalmiglio, C.; Brilli, L.; Campanile, M.; Ciuoli, C.; Cartocci, A.; Castagna, M.G. CONUT Score: A New Tool for Predicting Prognosis in Patients with Advanced Thyroid Cancer Treated with TKI. Cancers 2022, 14, 724. https://doi.org/10.3390/cancers14030724
Dalmiglio C, Brilli L, Campanile M, Ciuoli C, Cartocci A, Castagna MG. CONUT Score: A New Tool for Predicting Prognosis in Patients with Advanced Thyroid Cancer Treated with TKI. Cancers. 2022; 14(3):724. https://doi.org/10.3390/cancers14030724
Chicago/Turabian StyleDalmiglio, Cristina, Lucia Brilli, Michele Campanile, Cristina Ciuoli, Alessandra Cartocci, and Maria Grazia Castagna. 2022. "CONUT Score: A New Tool for Predicting Prognosis in Patients with Advanced Thyroid Cancer Treated with TKI" Cancers 14, no. 3: 724. https://doi.org/10.3390/cancers14030724
APA StyleDalmiglio, C., Brilli, L., Campanile, M., Ciuoli, C., Cartocci, A., & Castagna, M. G. (2022). CONUT Score: A New Tool for Predicting Prognosis in Patients with Advanced Thyroid Cancer Treated with TKI. Cancers, 14(3), 724. https://doi.org/10.3390/cancers14030724






