RBM22, a Key Player of Pre-mRNA Splicing and Gene Expression Regulation, Is Altered in Cancer
Simple Summary
Abstract
1. Introduction
2. The RNA-Binding Motif (RBM) Family
2.1. The Unity of the Family: The RNA-Recognition Motif
2.2. The Functions of RBM Proteins in RNA-Related Metabolism
3. RBM22 Is an RNA-Binding Protein
3.1. Structural Features of RBM22
3.2. RBM22 Is Essential for Gene Expression through Transcription Regulation and Pre-mRNA Splicing
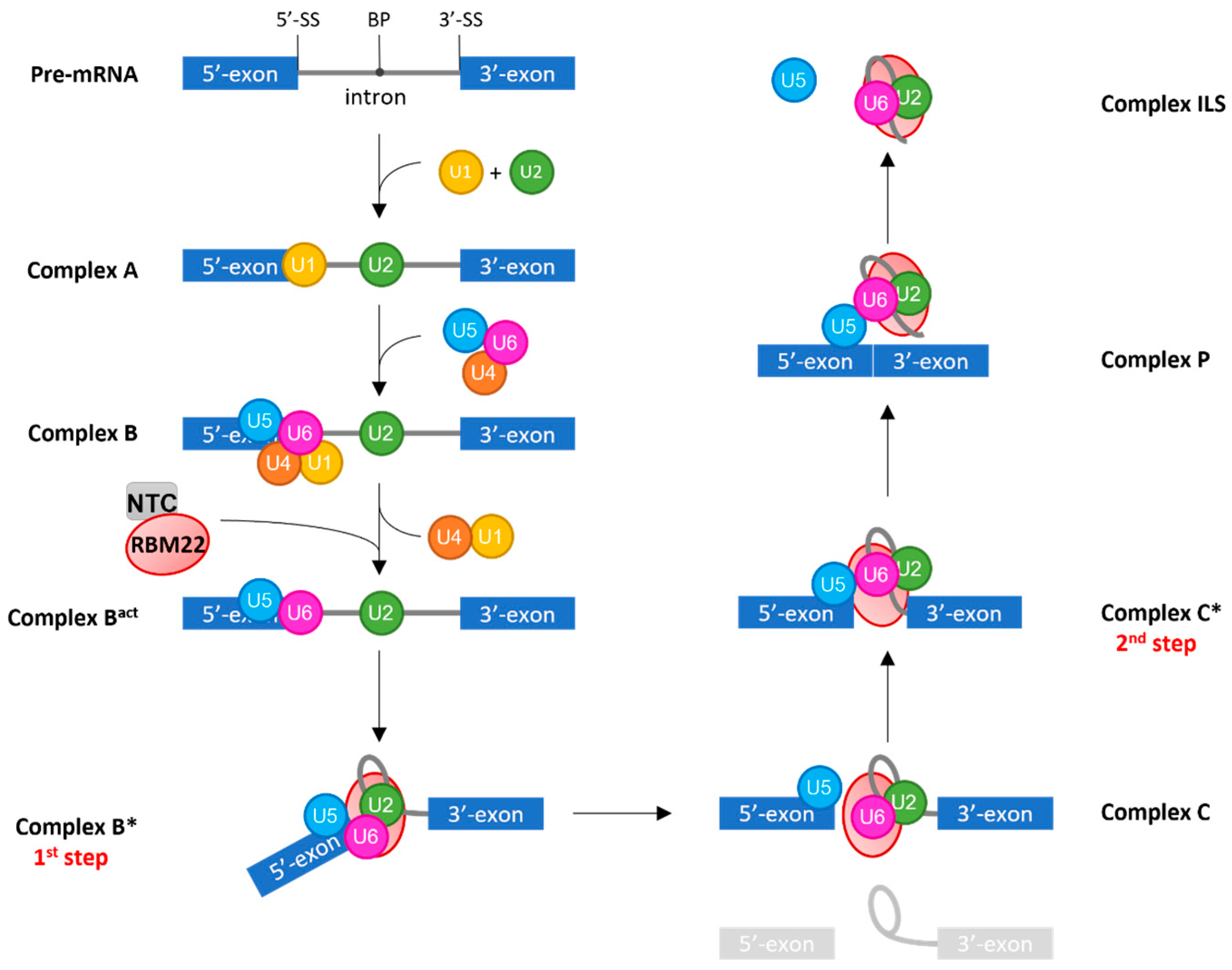
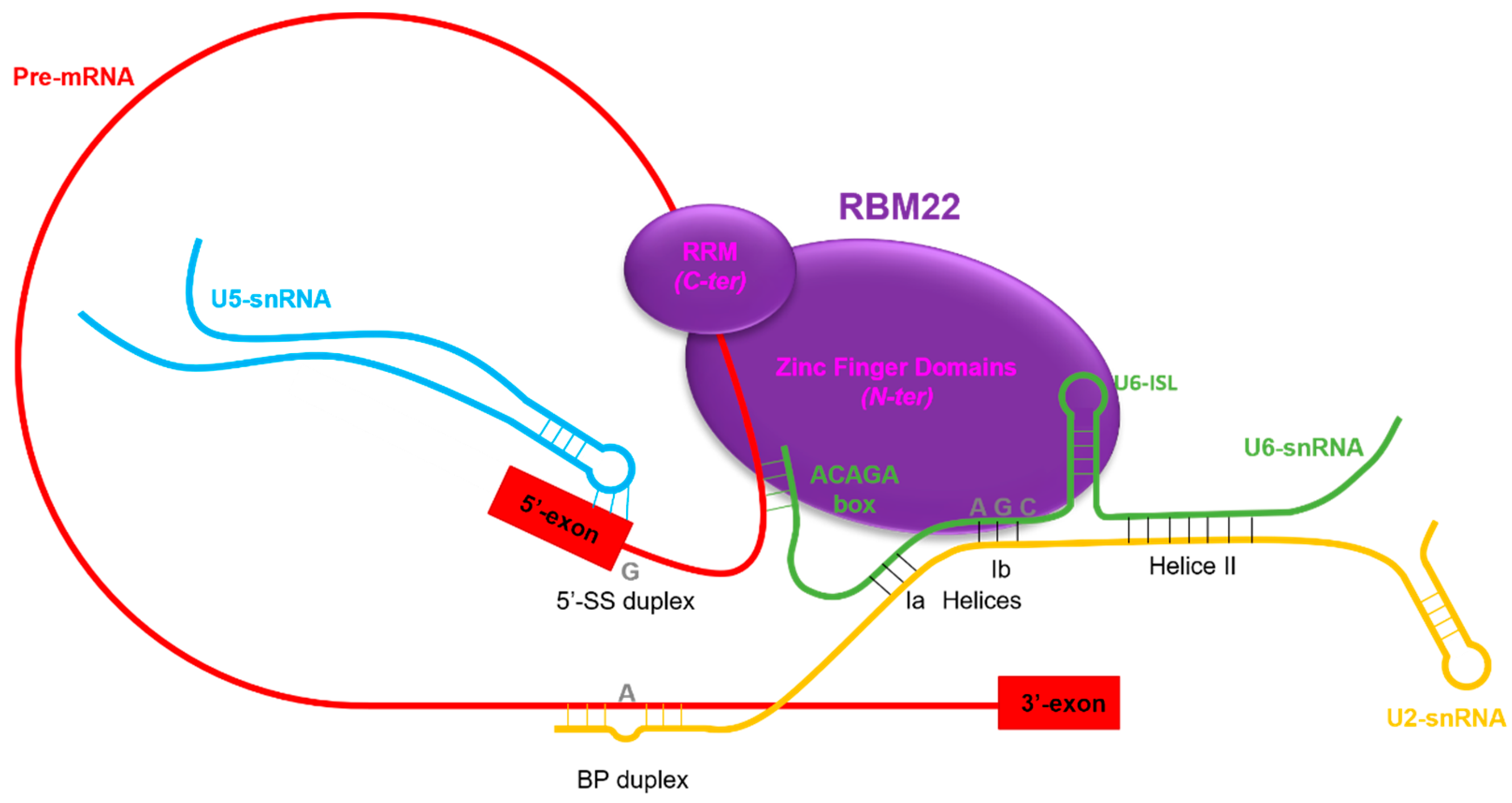
3.2.1. RBM22 Stabilizes the Catalytic Core of the Spliceosome
3.2.2. RBM22 Organizes Several Protein Components around the Catalytic Core
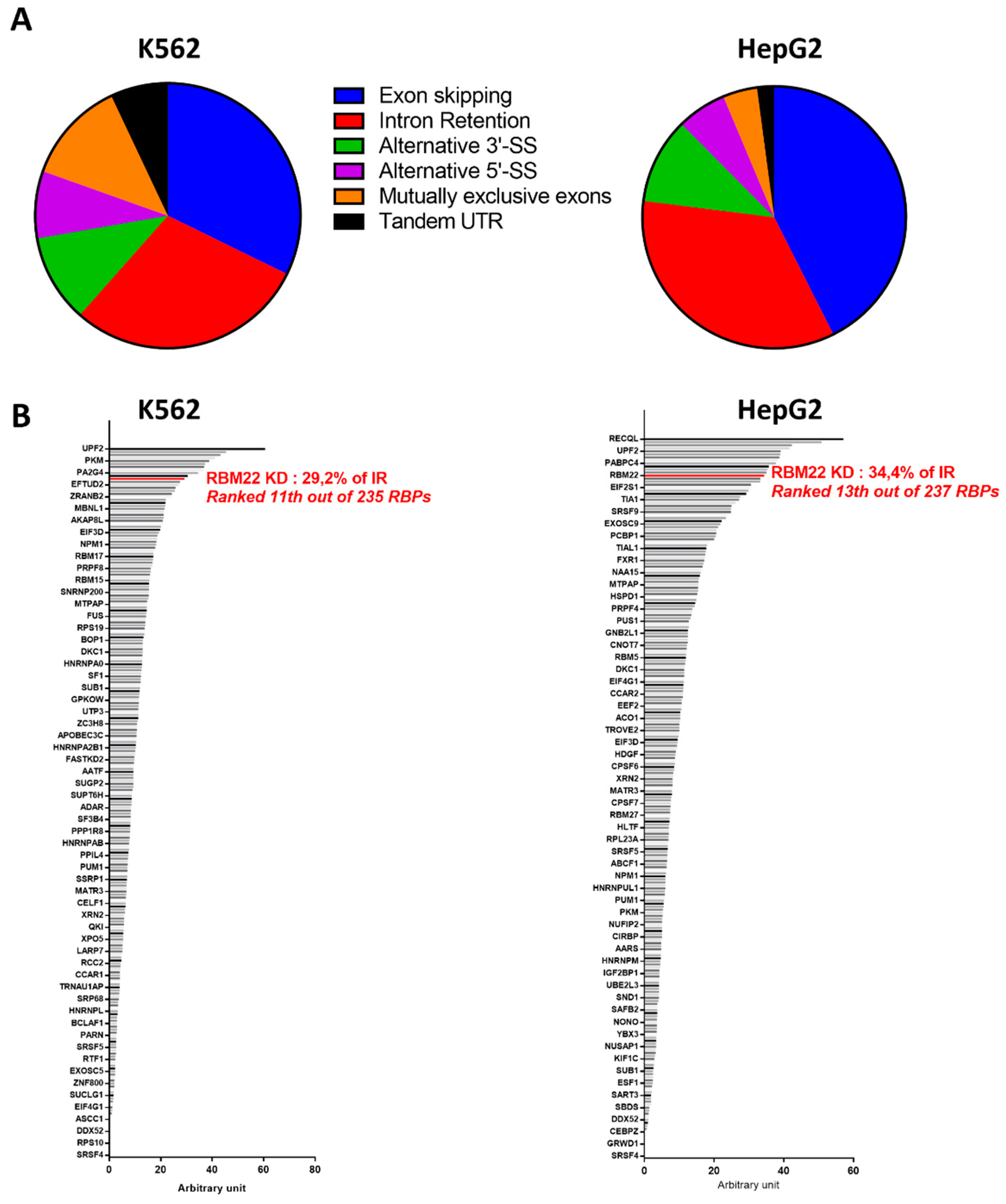
3.2.3. RBM22 Depletion Impacts Pre-mRNA Splicing
3.2.4. Emerging Evidence of Gene Regulation by RBM22
3.3. Implication of RBM22 in Ca2+-Dependent Regulation of mRNA Splicing
4. The Role of RBM22 in Mitosis and Differentiation
4.1. Spatio-Temporal Expression of RBM22
4.2. Experimental Depletion of RBM22 Results in Mitotic and Differentiation Defects
4.3. Implication of RBM22 in Human Diseases and Cancer: State-of-the-Art and Perspectives
5. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Alberts, B.; Johnson, A.; Lewis, J.; Raff, M.; Roberts, K.; Walter, P. The RNA World and the Origins of Life. In Molecular Biology of the Cell, 4th ed.; Garland Science: New York, NY, USA, 2002. [Google Scholar]
- Corley, M.; Burns, M.C.; Yeo, G.W. How RNA-Binding Proteins Interact with RNA: Molecules and Mechanisms. Mol. Cell 2020, 78, 9–29. [Google Scholar] [CrossRef] [PubMed]
- Gerstberger, S.; Hafner, M.; Tuschl, T. A Census of Human RNA-Binding Proteins. Nat. Rev. Genet. 2014, 15, 829–845. [Google Scholar] [CrossRef] [PubMed]
- Fredericks, A.M.; Cygan, K.J.; Brown, B.A.; Fairbrother, W.G. RNA-Binding Proteins: Splicing Factors and Disease. Biomolecules 2015, 5, 893–909. [Google Scholar] [CrossRef]
- Prashad, S.; Gopal, P.P. RNA-Binding Proteins in Neurological Development and Disease. RNA Biol. 2021, 18, 972–987. [Google Scholar] [CrossRef] [PubMed]
- Zealy, R.W.; Wrenn, S.P.; Davila, S.; Min, K.-W.; Yoon, J.-H. MicroRNA-Binding Proteins: Specificity and Function. Wiley Interdiscip. Rev. RNA 2017, 8, 1414. [Google Scholar] [CrossRef]
- Lunde, B.M.; Moore, C.; Varani, G. RNA-Binding Proteins: Modular Design for Efficient Function. Nat. Rev. Mol. Cell Biol. 2007, 8, 479–490. [Google Scholar] [CrossRef]
- Sutherland, L.C.; Rintala-Maki, N.D.; White, R.D.; Morin, C.D. RNA Binding Motif (RBM) Proteins: A Novel Family of Apoptosis Modulators? J. Cell. Biochem. 2005, 94, 5–24. [Google Scholar] [CrossRef]
- Pomeranz-Krummel, D.; Nagai, K. RNA-Binding Domains in Proteins. Encycl. Genet. 2001, 1733–1735. [Google Scholar] [CrossRef]
- Cléry, A.; Blatter, M.; Allain, F.H.-T. RNA Recognition Motifs: Boring? Not Quite. Curr. Opin. Struct. Biol. 2008, 18, 290–298. [Google Scholar] [CrossRef]
- Daubner, G.M.; Cléry, A.; Allain, F.H.-T. RRM-RNA Recognition: NMR or Crystallography…and New Findings. Curr. Opin. Struct. Biol. 2013, 23, 100–108. [Google Scholar] [CrossRef]
- Singh, M.; Wang, Z.; Koo, B.-K.; Patel, A.; Cascio, D.; Collins, K.; Feigon, J. Structural Basis for Telomerase RNA Recognition and RNP Assembly by the Holoenzyme La Family Protein P65. Mol. Cell 2012, 47, 16–26. [Google Scholar] [CrossRef] [PubMed]
- Skrisovska, L.; Bourgeois, C.F.; Stefl, R.; Grellscheid, S.-N.; Kister, L.; Wenter, P.; Elliott, D.J.; Stevenin, J.; Allain, F.H.-T. The Testis-Specific Human Protein RBMY Recognizes RNA through a Novel Mode of Interaction. EMBO Rep. 2007, 8, 372–379. [Google Scholar] [CrossRef] [PubMed]
- Jacks, A.; Babon, J.; Kelly, G.; Manolaridis, I.; Cary, P.D.; Curry, S.; Conte, M.R. Structure of the C-Terminal Domain of Human La Protein Reveals a Novel RNA Recognition Motif Coupled to a Helical Nuclear Retention Element. Structure 2003, 11, 833–843. [Google Scholar] [CrossRef]
- Volpon, L.; D’Orso, I.; Young, C.R.; Frasch, A.C.; Gehring, K. NMR Structural Study of TcUBP1, a Single RRM Domain Protein from Trypanosoma Cruzi: Contribution of a Beta Hairpin to RNA Binding. Biochemistry 2005, 44, 3708–3717. [Google Scholar] [CrossRef] [PubMed]
- Blatter, M.; Dunin-Horkawicz, S.; Grishina, I.; Maris, C.; Thore, S.; Maier, T.; Bindereif, A.; Bujnicki, J.M.; Allain, F.H.-T. The Signature of the Five-Stranded VRRM Fold Defined by Functional, Structural and Computational Analysis of the HnRNP L Protein. J. Mol. Biol. 2015, 427, 3001–3022. [Google Scholar] [CrossRef]
- Afroz, T.; Cienikova, Z.; Cléry, A.; Allain, F.H.T. One, Two, Three, Four! How Multiple RRMs Read the Genome Sequence. Meth. Enzymol. 2015, 558, 235–278. [Google Scholar] [CrossRef]
- Sickmier, E.A.; Frato, K.E.; Shen, H.; Paranawithana, S.R.; Green, M.R.; Kielkopf, C.L. Structural Basis for Polypyrimidine Tract Recognition by the Essential Pre-MRNA Splicing Factor U2AF65. Mol. Cell 2006, 23, 49–59. [Google Scholar] [CrossRef]
- Jenkins, H.T.; Malkova, B.; Edwards, T.A. Kinked β-Strands Mediate High-Affinity Recognition of MRNA Targets by the Germ-Cell Regulator DAZL. Proc. Natl. Acad. Sci. USA 2011, 108, 18266–18271. [Google Scholar] [CrossRef]
- Price, S.R.; Evans, P.R.; Nagai, K. Crystal Structure of the Spliceosomal U2B’-U2A’ Protein Complex Bound to a Fragment of U2 Small Nuclear RNA. Nature 1998, 394, 645–650. [Google Scholar] [CrossRef]
- Allain, F.H.; Bouvet, P.; Dieckmann, T.; Feigon, J. Molecular Basis of Sequence-Specific Recognition of Pre-Ribosomal RNA by Nucleolin. EMBO J. 2000, 19, 6870–6881. [Google Scholar] [CrossRef]
- Calero, G.; Wilson, K.F.; Ly, T.; Rios-Steiner, J.L.; Clardy, J.C.; Cerione, R.A. Structural Basis of M7GpppG Binding to the Nuclear Cap-Binding Protein Complex. Nat. Struct. Biol. 2002, 9, 912–917. [Google Scholar] [CrossRef] [PubMed]
- Mazza, C.; Segref, A.; Mattaj, I.W.; Cusack, S. Large-Scale Induced Fit Recognition of an m(7)GpppG Cap Analogue by the Human Nuclear Cap-Binding Complex. EMBO J. 2002, 21, 5548–5557. [Google Scholar] [CrossRef] [PubMed]
- Johansson, C.; Finger, L.D.; Trantirek, L.; Mueller, T.D.; Kim, S.; Laird-Offringa, I.A.; Feigon, J. Solution Structure of the Complex Formed by the Two N-Terminal RNA-Binding Domains of Nucleolin and a Pre-RRNA Target. J. Mol. Biol. 2004, 337, 799–816. [Google Scholar] [CrossRef] [PubMed]
- Upadhyay, S.K.; Mackereth, C.D. Structural Basis of UCUU RNA Motif Recognition by Splicing Factor RBM20. Nucleic Acids Res. 2020, 48, 4538–4550. [Google Scholar] [CrossRef] [PubMed]
- Nowacka, M.; Boccaletto, P.; Jankowska, E.; Jarzynka, T.; Bujnicki, J.M.; Dunin-Horkawicz, S. RRMdb—An Evolutionary-Oriented Database of RNA Recognition Motif Sequences. Database 2019, 2019, bay148. [Google Scholar] [CrossRef]
- Farina, B.; Fattorusso, R.; Pellecchia, M. Targeting Zinc Finger Domains with Small Molecules: Solution Structure and Binding Studies of the RanBP2-Type Zinc Finger of RBM5. Chembiochem 2011, 12, 2837–2845. [Google Scholar] [CrossRef]
- Agafonov, D.E.; Deckert, J.; Wolf, E.; Odenwälder, P.; Bessonov, S.; Will, C.L.; Urlaub, H.; Lührmann, R. Semiquantitative Proteomic Analysis of the Human Spliceosome via a Novel Two-Dimensional Gel Electrophoresis Method. Mol. Cell. Biol. 2011, 31, 2667–2682. [Google Scholar] [CrossRef]
- Zhang, X.; Yan, C.; Hang, J.; Finci, L.I.; Lei, J.; Shi, Y. An Atomic Structure of the Human Spliceosome. Cell 2017, 169, 918–929. [Google Scholar] [CrossRef]
- Hachet, O.; Ephrussi, A. Splicing of Oskar RNA in the Nucleus Is Coupled to Its Cytoplasmic Localization. Nature 2004, 428, 959–963. [Google Scholar] [CrossRef]
- Dreumont, N.; Bourgeois, C.F.; Lejeune, F.; Liu, Y.; Ehrmann, I.E.; Elliott, D.J.; Stévenin, J. Human RBMY Regulates Germline-Specific Splicing Events by Modulating the Function of the Serine/Arginine-Rich Proteins 9G8 and Tra2-β. J. Cell Sci. 2010, 123, 40–50. [Google Scholar] [CrossRef]
- Yang, J.; Hung, L.-H.; Licht, T.; Kostin, S.; Looso, M.; Khrameeva, E.; Bindereif, A.; Schneider, A.; Braun, T. RBM24 Is a Major Regulator of Muscle-Specific Alternative Splicing. Dev. Cell 2014, 31, 87–99. [Google Scholar] [CrossRef] [PubMed]
- Arya, A.D.; Wilson, D.I.; Baralle, D.; Raponi, M. RBFOX2 Protein Domains and Cellular Activities. Biochem. Soc. Trans. 2014, 42, 1180–1183. [Google Scholar] [CrossRef] [PubMed]
- Coomer, A.O.; Black, F.; Greystoke, A.; Munkley, J.; Elliott, D.J. Alternative Splicing in Lung Cancer. Biochim. Biophys. Acta Gene Regul. Mech. 2019, 1862, 194388. [Google Scholar] [CrossRef]
- Silla, T.; Schmid, M.; Dou, Y.; Garland, W.; Milek, M.; Imami, K.; Johnsen, D.; Polak, P.; Andersen, J.S.; Selbach, M.; et al. The Human ZC3H3 and RBM26/27 Proteins Are Critical for PAXT-Mediated Nuclear RNA Decay. Nucleic Acids Res. 2020, 48, 2518–2530. [Google Scholar] [CrossRef]
- Al-Astal, H.I.M.; Massad, M.; AlMatar, M.; Ekal, H. Cellular Functions of RNA-Binding Motif Protein 3 (RBM3): Clues in Hypothermia, Cancer Biology and Apoptosis. Protein Pept. Lett. 2016, 23, 828–835. [Google Scholar] [CrossRef] [PubMed]
- Elliott, D.J. The Role of Potential Splicing Factors Including RBMY, RBMX, HnRNPG-T and STAR Proteins in Spermatogenesis. Int. J. Androl. 2004, 27, 328–334. [Google Scholar] [CrossRef]
- Abid, S.; Sagare-Patil, V.; Gokral, J.; Modi, D. Cellular Ontogeny of RBMY during Human Spermatogenesis and Its Role in Sperm Motility. J. Biosci. 2013, 38, 85–92. [Google Scholar] [CrossRef]
- Yan, Y.; Yang, X.; Liu, Y.; Shen, Y.; Tu, W.; Dong, Q.; Yang, D.; Ma, Y.; Yang, Y. Copy Number Variation of Functional RBMY1 Is Associated with Sperm Motility: An Azoospermia Factor-Linked Candidate for Asthenozoospermia. Hum. Reprod. 2017, 32, 1521–1531. [Google Scholar] [CrossRef]
- Yan, Q.; Zeng, P.; Zhou, X.; Zhao, X.; Chen, R.; Qiao, J.; Feng, L.; Zhu, Z.; Zhang, G.; Chen, C. RBMX Suppresses Tumorigenicity and Progression of Bladder Cancer by Interacting with the HnRNP A1 Protein to Regulate PKM Alternative Splicing. Oncogene 2021, 40, 2635–2650. [Google Scholar] [CrossRef]
- Martínez-Arribas, F.; Agudo, D.; Pollán, M.; Gómez-Esquer, F.; Díaz-Gil, G.; Lucas, R.; Schneider, J. Positive Correlation between the Expression of X-Chromosome RBM Genes (RBMX, RBM3, RBM10) and the Proapoptotic Bax Gene in Human Breast Cancer. J. Cell. Biochem. 2006, 97, 1275–1282. [Google Scholar] [CrossRef]
- Tsend-Ayush, E.; O’Sullivan, L.A.; Grützner, F.S.; Onnebo, S.M.N.; Lewis, R.S.; Delbridge, M.L.; Marshall Graves, J.A.; Ward, A.C. RBMX Gene Is Essential for Brain Development in Zebrafish. Dev. Dyn. 2005, 234, 682–688. [Google Scholar] [CrossRef] [PubMed]
- Matsunaga, S.; Takata, H.; Morimoto, A.; Hayashihara, K.; Higashi, T.; Akatsuchi, K.; Mizusawa, E.; Yamakawa, M.; Ashida, M.; Matsunaga, T.M.; et al. RBMX: A Regulator for Maintenance and Centromeric Protection of Sister Chromatid Cohesion. Cell Rep. 2012, 1, 299–308. [Google Scholar] [CrossRef] [PubMed]
- Cho, Y.; Ideue, T.; Nagayama, M.; Araki, N.; Tani, T. RBMX Is a Component of the Centromere Noncoding RNP Complex Involved in Cohesion Regulation. Genes Cells 2018, 23, 172–184. [Google Scholar] [CrossRef]
- Zheng, T.; Zhou, H.; Li, X.; Peng, D.; Yang, Y.; Zeng, Y.; Liu, H.; Ren, J.; Zhao, Y. RBMX Is Required for Activation of ATR on Repetitive DNAs to Maintain Genome Stability. Cell Death Differ. 2020, 27, 3162–3176. [Google Scholar] [CrossRef]
- Smart, F.; Aschrafi, A.; Atkins, A.; Owens, G.C.; Pilotte, J.; Cunningham, B.A.; Vanderklish, P.W. Two Isoforms of the Cold-Inducible MRNA-Binding Protein RBM3 Localize to Dendrites and Promote Translation. J. Neurochem. 2007, 101, 1367–1379. [Google Scholar] [CrossRef] [PubMed]
- Sureban, S.M.; Ramalingam, S.; Natarajan, G.; May, R.; Subramaniam, D.; Bishnupuri, K.S.; Morrison, A.R.; Dieckgraefe, B.K.; Brackett, D.J.; Postier, R.G.; et al. Translation Regulatory Factor RBM3 Is a Proto-Oncogene That Prevents Mitotic Catastrophe. Oncogene 2008, 27, 4544–4556. [Google Scholar] [CrossRef] [PubMed]
- Ushio, A.; Eto, K. RBM3 Expression Is Upregulated by NF-ΚB P65 Activity, Protecting Cells from Apoptosis, during Mild Hypothermia. J. Cell. Biochem. 2018, 119, 5734–5749. [Google Scholar] [CrossRef]
- Ehlén, Å.; Nodin, B.; Rexhepaj, E.; Brändstedt, J.; Uhlén, M.; Alvarado-Kristensson, M.; Pontén, F.; Brennan, D.J.; Jirström, K. RBM3-Regulated Genes Promote DNA Integrity and Affect Clinical Outcome in Epithelial Ovarian Cancer. Transl. Oncol. 2011, 4, 212–221. [Google Scholar] [CrossRef]
- Liu, Y.; Hu, W.; Murakawa, Y.; Yin, J.; Wang, G.; Landthaler, M.; Yan, J. Cold-Induced RNA-Binding Proteins Regulate Circadian Gene Expression by Controlling Alternative Polyadenylation. Sci. Rep. 2013, 3, 2054. [Google Scholar] [CrossRef]
- Dong, W.; Dai, Z.-H.; Liu, F.-C.; Guo, X.-G.; Ge, C.-M.; Ding, J.; Liu, H.; Yang, F. The RNA-Binding Protein RBM3 Promotes Cell Proliferation in Hepatocellular Carcinoma by Regulating Circular RNA SCD-CircRNA 2 Production. EBioMedicine 2019, 45, 155–167. [Google Scholar] [CrossRef]
- Yan, J.; Goerne, T.; Zelmer, A.; Guzman, R.; Kapfhammer, J.P.; Wellmann, S.; Zhu, X. The RNA-Binding Protein RBM3 Promotes Neural Stem Cell (NSC) Proliferation Under Hypoxia. Front. Cell Dev. Biol. 2019, 7, 288. [Google Scholar] [CrossRef] [PubMed]
- Wang, Y.; Chen, D.; Qian, H.; Tsai, Y.S.; Shao, S.; Liu, Q.; Dominguez, D.; Wang, Z. The Splicing Factor RBM4 Controls Apoptosis, Proliferation, and Migration to Suppress Tumor Progression. Cancer Cell 2014, 26, 374–389. [Google Scholar] [CrossRef] [PubMed]
- Wang, W.-Y.; Quan, W.; Yang, F.; Wei, Y.-X.; Chen, J.-J.; Yu, H.; Xie, J.; Zhang, Y.; Li, Z.-F. RBM4 Modulates the Proliferation and Expression of Inflammatory Factors via the Alternative Splicing of Regulatory Factors in HeLa Cells. Mol. Genet. Genom. 2020, 295, 95–106. [Google Scholar] [CrossRef] [PubMed]
- Lin, J.-C.; Tarn, W.-Y. Multiple Roles of RBM4 in Muscle Cell Differentiation. Front. Biosci. (Schol. Ed.) 2012, 4, 181–189. [Google Scholar] [CrossRef] [PubMed]
- Tarn, W.-Y.; Kuo, H.-C.; Yu, H.-I.; Liu, S.-W.; Tseng, C.-T.; Dhananjaya, D.; Hung, K.-Y.; Tu, C.-C.; Chang, S.-H.; Huang, G.-J.; et al. RBM4 Promotes Neuronal Differentiation and Neurite Outgrowth by Modulating Numb Isoform Expression. Mol. Biol. Cell 2016, 27, 1676–1683. [Google Scholar] [CrossRef]
- Lin, J.-C.; Yan, Y.-T.; Hsieh, W.-K.; Peng, P.-J.; Su, C.-H.; Tarn, W.-Y. RBM4 Promotes Pancreas Cell Differentiation and Insulin Expression. Mol. Cell. Biol. 2013, 33, 319–327. [Google Scholar] [CrossRef]
- Huang, Y.; McNeil, G.P.; Jackson, F.R. Translational Regulation of the DOUBLETIME/CKIδ/ε Kinase by LARK Contributes to Circadian Period Modulation. PLoS Genet. 2014, 10, e1004536. [Google Scholar] [CrossRef]
- Bechara, E.G.; Sebestyén, E.; Bernardis, I.; Eyras, E.; Valcárcel, J. RBM5, 6, and 10 Differentially Regulate NUMB Alternative Splicing to Control Cancer Cell Proliferation. Mol. Cell 2013, 52, 720–733. [Google Scholar] [CrossRef]
- Mourtada-Maarabouni, M.; Keen, J.; Clark, J.; Cooper, C.S.; Williams, G.T. Candidate Tumor Suppressor LUCA-15/RBM5/H37 Modulates Expression of Apoptosis and Cell Cycle Genes. Exp. Cell Res. 2006, 312, 1745–1752. [Google Scholar] [CrossRef]
- O’Bryan, M.K.; Clark, B.J.; McLaughlin, E.A.; D’Sylva, R.J.; O’Donnell, L.; Wilce, J.A.; Sutherland, J.; O’Connor, A.E.; Whittle, B.; Goodnow, C.C.; et al. RBM5 Is a Male Germ Cell Splicing Factor and Is Required for Spermatid Differentiation and Male Fertility. PLoS Genet. 2013, 9, e1003628. [Google Scholar] [CrossRef]
- Xi, P.-W.; Zhang, X.; Zhu, L.; Dai, X.-Y.; Cheng, L.; Hu, Y.; Shi, L.; Wei, J.-F.; Ding, Q. Oncogenic Action of the Exosome Cofactor RBM7 by Stabilization of CDK1 MRNA in Breast Cancer. NPJ Breast Cancer 2020, 6, 58. [Google Scholar] [CrossRef] [PubMed]
- McSweeney, C.; Dong, F.; Chen, M.; Vitale, J.; Xu, L.; Crowley, N.; Luscher, B.; Zou, D.; Mao, Y. Full Function of Exon Junction Complex Factor, Rbm8a, Is Critical for Interneuron Development. Transl. Psychiatry 2020, 10, 379. [Google Scholar] [CrossRef] [PubMed]
- Le Hir, H.; Gatfield, D.; Braun, I.C.; Forler, D.; Izaurralde, E. The Protein Mago Provides a Link between Splicing and MRNA Localization. EMBO Rep. 2001, 2, 1119–1124. [Google Scholar] [CrossRef]
- Lee, H.C.; Choe, J.; Chi, S.-G.; Kim, Y.K. Exon Junction Complex Enhances Translation of Spliced MRNAs at Multiple Steps. Biochem. Biophys. Res. Commun. 2009, 384, 334–340. [Google Scholar] [CrossRef] [PubMed]
- Ishigaki, Y.; Nakamura, Y.; Tatsuno, T.; Hashimoto, M.; Shimasaki, T.; Iwabuchi, K.; Tomosugi, N. Depletion of RNA-Binding Protein RBM8A (Y14) Causes Cell Cycle Deficiency and Apoptosis in Human Cells. Exp. Biol. Med. 2013, 238, 889–897. [Google Scholar] [CrossRef]
- Lu, C.-C.; Lee, C.-C.; Tseng, C.-T.; Tarn, W.-Y. Y14 Governs P53 Expression and Modulates DNA Damage Sensitivity. Sci. Rep. 2017, 7, 45558. [Google Scholar] [CrossRef]
- Zou, D.; McSweeney, C.; Sebastian, A.; Reynolds, D.J.; Dong, F.; Zhou, Y.; Deng, D.; Wang, Y.; Liu, L.; Zhu, J.; et al. A Critical Role of RBM8a in Proliferation and Differentiation of Embryonic Neural Progenitors. Neural Dev. 2015, 10, 18. [Google Scholar] [CrossRef]
- Chuang, T.-W.; Lu, C.-C.; Su, C.-H.; Wu, P.-Y.; Easwvaran, S.; Lee, C.-C.; Kuo, H.-C.; Hung, K.-Y.; Lee, K.-M.; Tsai, C.-Y.; et al. The RNA Processing Factor Y14 Participates in DNA Damage Response and Repair. iScience 2019, 13, 402–415. [Google Scholar] [CrossRef]
- Venables, J.P.; Lapasset, L.; Gadea, G.; Fort, P.; Klinck, R.; Irimia, M.; Vignal, E.; Thibault, P.; Prinos, P.; Chabot, B.; et al. MBNL1 and RBFOX2 Cooperate to Establish a Splicing Programme Involved in Pluripotent Stem Cell Differentiation. Nat. Commun. 2013, 4, 2480. [Google Scholar] [CrossRef]
- Jung, J.H.; Lee, H.; Zeng, S.X.; Lu, H. RBM10, a New Regulator of P53. Cells 2020, 9, 2107. [Google Scholar] [CrossRef]
- Cao, Y.; Di, X.; Zhang, Q.; Li, R.; Wang, K. RBM10 Regulates Tumor Apoptosis, Proliferation, and Metastasis. Front. Oncol. 2021, 11, 603932. [Google Scholar] [CrossRef] [PubMed]
- Kunimoto, H.; Inoue, A.; Kojima, H.; Yang, J.; Zhao, H.; Tsuruta, D.; Nakajima, K. RBM10 Regulates Centriole Duplication in HepG2 Cells by Ectopically Assembling PLK4-STIL Complexes in the Nucleus. Genes Cells 2020, 25, 100–110. [Google Scholar] [CrossRef] [PubMed]
- Jung, J.H.; Lee, H.; Cao, B.; Liao, P.; Zeng, S.X.; Lu, H. RNA-Binding Motif Protein 10 Induces Apoptosis and Suppresses Proliferation by Activating P53. Oncogene 2020, 39, 1031–1040. [Google Scholar] [CrossRef] [PubMed]
- Pedrotti, S.; Busà, R.; Compagnucci, C.; Sette, C. The RNA Recognition Motif Protein RBM11 Is a Novel Tissue-Specific Splicing Regulator. Nucleic Acids Res. 2012, 40, 1021–1032. [Google Scholar] [CrossRef] [PubMed]
- Li, J.; Wang, C.; Feng, G.; Zhang, L.; Chen, G.; Sun, H.; Wang, J.; Zhang, Y.; Zhou, Q.; Li, W. Rbm14 Maintains the Integrity of Genomic DNA during Early Mouse Embryogenesis via Mediating Alternative Splicing. Cell Prolif. 2020, 53, e12724. [Google Scholar] [CrossRef] [PubMed]
- Chen, G.; Zhang, D.; Zhang, L.; Feng, G.; Zhang, B.; Wu, Y.; Li, W.; Zhang, Y.; Hu, B. RBM14 Is Indispensable for Pluripotency Maintenance and Mesoderm Development of Mouse Embryonic Stem Cells. Biochem. Biophys. Res. Commun. 2018, 501, 259–265. [Google Scholar] [CrossRef] [PubMed]
- Kai, M. Roles of RNA-Binding Proteins in DNA Damage Response. Int. J. Mol. Sci. 2016, 17, 310. [Google Scholar] [CrossRef]
- Zhang, L.; Tran, N.-T.; Su, H.; Wang, R.; Lu, Y.; Tang, H.; Aoyagi, S.; Guo, A.; Khodadadi-Jamayran, A.; Zhou, D.; et al. Cross-Talk between PRMT1-Mediated Methylation and Ubiquitylation on RBM15 Controls RNA Splicing. eLife 2015, 4, e07938. [Google Scholar] [CrossRef]
- Lindtner, S.; Zolotukhin, A.S.; Uranishi, H.; Bear, J.; Kulkarni, V.; Smulevitch, S.; Samiotaki, M.; Panayotou, G.; Felber, B.K.; Pavlakis, G.N. RNA-Binding Motif Protein 15 Binds to the RNA Transport Element RTE and Provides a Direct Link to the NXF1 Export Pathway. J. Biol. Chem. 2006, 281, 36915–36928. [Google Scholar] [CrossRef]
- Hu, M.; Yang, Y.; Ji, Z.; Luo, J. RBM15 Functions in Blood Diseases. Curr. Cancer Drug Targets 2016, 16, 579–585. [Google Scholar] [CrossRef]
- Ma, X.; Renda, M.J.; Wang, L.; Cheng, E.-C.; Niu, C.; Morris, S.W.; Chi, A.S.; Krause, D.S. Rbm15 Modulates Notch-Induced Transcriptional Activation and Affects Myeloid Differentiation. Mol. Cell. Biol. 2007, 27, 3056–3064. [Google Scholar] [CrossRef] [PubMed]
- Jin, S.; Mi, Y.; Song, J.; Zhang, P.; Liu, Y. PRMT1-RBM15 Axis Regulates Megakaryocytic Differentiation of Human Umbilical Cord Blood CD34+ Cells. Exp. Ther. Med. 2018, 15, 2563–2568. [Google Scholar] [CrossRef] [PubMed]
- Chen, X.-Y.; Zhang, J.; Zhu, J.-S. The Role of M6A RNA Methylation in Human Cancer. Mol. Cancer 2019, 18, 103. [Google Scholar] [CrossRef] [PubMed]
- Gregersen, L.H.; Mitter, R.; Ugalde, A.P.; Nojima, T.; Proudfoot, N.J.; Agami, R.; Stewart, A.; Svejstrup, J.Q. SCAF4 and SCAF8, MRNA Anti-Terminator Proteins. Cell 2019, 177, 1797–1813. [Google Scholar] [CrossRef]
- De Maio, A.; Yalamanchili, H.K.; Adamski, C.J.; Gennarino, V.A.; Liu, Z.; Qin, J.; Jung, S.Y.; Richman, R.; Orr, H.; Zoghbi, H.Y. RBM17 Interacts with U2SURP and CHERP to Regulate Expression and Splicing of RNA-Processing Proteins. Cell Rep. 2018, 25, 726–736.e7. [Google Scholar] [CrossRef]
- Villamizar, O.; Chambers, C.B.; Riberdy, J.M.; Persons, D.A.; Wilber, A. Long Noncoding RNA Saf and Splicing Factor 45 Increase Soluble Fas and Resistance to Apoptosis. Oncotarget 2016, 7, 13810–13826. [Google Scholar] [CrossRef]
- Lu, J.; Li, Q.; Cai, L.; Zhu, Z.; Guan, J.; Wang, C.; Xia, J.; Xia, L.; Wen, M.; Zheng, W.; et al. RBM17 Controls Apoptosis and Proliferation to Promote Glioma Progression. Biochem. Biophys. Res. Commun. 2018, 505, 20–28. [Google Scholar] [CrossRef]
- Martín, E.; Vivori, C.; Rogalska, M.; Herrero-Vicente, J.; Valcárcel, J. Alternative Splicing Regulation of Cell-Cycle Genes by SPF45/SR140/CHERP Complex Controls Cell Proliferation. RNA 2021, 27, 1557–1576. [Google Scholar] [CrossRef]
- Amsterdam, A.; Nissen, R.M.; Sun, Z.; Swindell, E.C.; Farrington, S.; Hopkins, N. Identification of 315 Genes Essential for Early Zebrafish Development. Proc. Natl. Acad. Sci. USA 2004, 101, 12792–12797. [Google Scholar] [CrossRef]
- Lorenzen, J.A.; Bonacci, B.B.; Palmer, R.E.; Wells, C.; Zhang, J.; Haber, D.A.; Goldstein, A.M.; Mayer, A.N. Rbm19 Is a Nucleolar Protein Expressed in Crypt/Progenitor Cells of the Intestinal Epithelium. Gene Expr. Patterns 2005, 6, 45–56. [Google Scholar] [CrossRef]
- Saijou, E.; Fujiwara, T.; Suzaki, T.; Inoue, K.; Sakamoto, H. RBD-1, a Nucleolar RNA-Binding Protein, Is Essential for Caenorhabditis Elegans Early Development through 18S Ribosomal RNA Processing. Nucleic Acids Res. 2004, 32, 1028–1036. [Google Scholar] [CrossRef] [PubMed]
- Zhang, J.; Tomasini, A.J.; Mayer, A.N. RBM19 Is Essential for Preimplantation Development in the Mouse. BMC Dev. Biol. 2008, 8, 115. [Google Scholar] [CrossRef] [PubMed]
- Watanabe, T.; Kimura, A.; Kuroyanagi, H. Alternative Splicing Regulator RBM20 and Cardiomyopathy. Front. Mol. Biosci. 2018, 5, 105. [Google Scholar] [CrossRef] [PubMed]
- Yashiro, Y.; Tomita, K. Function and Regulation of Human Terminal Uridylyltransferases. Front. Genet. 2018, 9, 538. [Google Scholar] [CrossRef] [PubMed]
- Li, W.; Li, W.; Laishram, R.S.; Hoque, M.; Ji, Z.; Tian, B.; Anderson, R.A. Distinct Regulation of Alternative Polyadenylation and Gene Expression by Nuclear Poly(A) Polymerases. Nucleic Acids Res. 2017, 45, 8930–8942. [Google Scholar] [CrossRef] [PubMed]
- Rasche, N.; Dybkov, O.; Schmitzová, J.; Akyildiz, B.; Fabrizio, P.; Lührmann, R. Cwc2 and Its Human Homologue RBM22 Promote an Active Conformation of the Spliceosome Catalytic Centre. EMBO J. 2012, 31, 1591–1604. [Google Scholar] [CrossRef]
- Zhan, X.; Yan, C.; Zhang, X.; Lei, J.; Shi, Y. Structure of a Human Catalytic Step I Spliceosome. Science 2018, 359, 537–545. [Google Scholar] [CrossRef]
- Sun, C.; Rigo, N.; Fabrizio, P.; Kastner, B.; Lührmann, R. A Protein Map of the Yeast Activated Spliceosome as Obtained by Electron Microscopy. RNA 2016, 22, 1427–1440. [Google Scholar] [CrossRef]
- Liu, S.; Li, X.; Zhang, L.; Jiang, J.; Hill, R.C.; Cui, Y.; Hansen, K.C.; Zhou, Z.H.; Zhao, R. Structure of the Yeast Spliceosomal Postcatalytic P Complex. Science 2017, 358, 1278–1283. [Google Scholar] [CrossRef]
- Wan, R.; Yan, C.; Bai, R.; Lei, J.; Shi, Y. Structure of an Intron Lariat Spliceosome from Saccharomyces Cerevisiae. Cell 2017, 171, 120–132.e12. [Google Scholar] [CrossRef]
- Kittler, R.; Putz, G.; Pelletier, L.; Poser, I.; Heninger, A.-K.; Drechsel, D.; Fischer, S.; Konstantinova, I.; Habermann, B.; Grabner, H.; et al. An Endoribonuclease-Prepared SiRNA Screen in Human Cells Identifies Genes Essential for Cell Division. Nature 2004, 432, 1036. [Google Scholar] [CrossRef] [PubMed]
- Ebert, B.L.; Pretz, J.; Bosco, J.; Chang, C.Y.; Tamayo, P.; Galili, N.; Raza, A.; Root, D.E.; Attar, E.; Ellis, S.R.; et al. Identification of RPS14 as a 5q- Syndrome Gene by RNA Interference Screen. Nature 2008, 451, 335–339. [Google Scholar] [CrossRef] [PubMed]
- Van Nostrand, E.L.; Freese, P.; Pratt, G.A.; Wang, X.; Wei, X.; Xiao, R.; Blue, S.M.; Chen, J.-Y.; Cody, N.A.L.; Dominguez, D.; et al. A Large-Scale Binding and Functional Map of Human RNA-Binding Proteins. Nature 2020, 583, 711–719. [Google Scholar] [CrossRef] [PubMed]
- Xiao, R.; Chen, J.-Y.; Liang, Z.; Luo, D.; Chen, G.; Lu, Z.J.; Chen, Y.; Zhou, B.; Li, H.; Du, X.; et al. Pervasive Chromatin-RNA Binding Protein Interactions Enable RNA-Based Regulation of Transcription. Cell 2019, 178, 107–121.e18. [Google Scholar] [CrossRef]
- Kim, Y.-O.; Park, S.-J.; Balaban, R.S.; Nirenberg, M.; Kim, Y. A Functional Genomic Screen for Cardiogenic Genes Using RNA Interference in Developing Drosophila Embryos. Proc. Natl. Acad. Sci. USA 2004, 101, 159–164. [Google Scholar] [CrossRef]
- Dowhan, D.H.; Hong, E.P.; Auboeuf, D.; Dennis, A.P.; Wilson, M.M.; Berget, S.M.; O’Malley, B.W. Steroid Hormone Receptor Coactivation and Alternative RNA Splicing by U2AF65-Related Proteins CAPERalpha and CAPERbeta. Mol. Cell 2005, 17, 429–439. [Google Scholar] [CrossRef]
- Grifone, R.; Shao, M.; Saquet, A.; Shi, D.-L. RNA-Binding Protein Rbm24 as a Multifaceted Post-Transcriptional Regulator of Embryonic Lineage Differentiation and Cellular Homeostasis. Cells 2020, 9, 1891. [Google Scholar] [CrossRef]
- Li, N.; Du, H.; Ren, R.; Wang, Y.; Xu, Z. Alternative Splicing of Cdh23 Exon 68 Is Regulated by RBM24, RBM38, and PTBP1. Neural Plast. 2020, 2020, 8898811. [Google Scholar] [CrossRef]
- Xu, E.; Zhang, J.; Zhang, M.; Jiang, Y.; Cho, S.-J.; Chen, X. RNA-Binding Protein RBM24 Regulates P63 Expression via MRNA Stability. Mol. Cancer Res. 2014, 12, 359–369. [Google Scholar] [CrossRef][Green Version]
- Zhang, M.; Zhang, Y.; Xu, E.; Mohibi, S.; de Anda, D.M.; Jiang, Y.; Zhang, J.; Chen, X. Rbm24, a Target of P53, Is Necessary for Proper Expression of P53 and Heart Development. Cell Death Differ. 2018, 25, 1118–1130. [Google Scholar] [CrossRef]
- Yin, Y.-W.; Liu, K.-L.; Lu, B.-S.; Li, W.; Niu, Y.-L.; Zhao, C.-M.; Yang, Z.; Guo, P.-Y.; Qi, J.-C. RBM24 Exacerbates Bladder Cancer Progression by Forming a Runx1t1/TCF4/MiR-625-5p Feedback Loop. Exp. Mol. Med. 2021, 53, 933–946. [Google Scholar] [CrossRef] [PubMed]
- Zhou, A.; Ou, A.C.; Cho, A.; Benz, E.J.; Huang, S.-C. Novel Splicing Factor RBM25 Modulates Bcl-x Pre-MRNA 5′ Splice Site Selection. Mol. Cell. Biol. 2008, 28, 5924–5936. [Google Scholar] [CrossRef] [PubMed]
- Carlson, S.M.; Soulette, C.M.; Yang, Z.; Elias, J.E.; Brooks, A.N.; Gozani, O. RBM25 Is a Global Splicing Factor Promoting Inclusion of Alternatively Spliced Exons and Is Itself Regulated by Lysine Mono-Methylation. J. Biol. Chem. 2017, 292, 13381–13390. [Google Scholar] [CrossRef] [PubMed]
- Ge, Y.; Schuster, M.B.; Pundhir, S.; Rapin, N.; Bagger, F.O.; Sidiropoulos, N.; Hashem, N.; Porse, B.T. The Splicing Factor RBM25 Controls MYC Activity in Acute Myeloid Leukemia. Nat. Commun. 2019, 10, 172. [Google Scholar] [CrossRef] [PubMed]
- Damianov, A.; Kann, M.; Lane, W.S.; Bindereif, A. Human RBM28 Protein Is a Specific Nucleolar Component of the Spliceosomal SnRNPs. Biol. Chem. 2006, 387, 1455–1460. [Google Scholar] [CrossRef]
- Li, W.; Feng, Y.; Chen, A.; Li, T.; Huang, S.; Liu, J.; Liu, X.; Liu, Y.; Gao, J.; Yan, D.; et al. Elmod3 Knockout Leads to Progressive Hearing Loss and Abnormalities in Cochlear Hair Cell Stereocilia. Hum. Mol. Genet. 2019, 28, 4103–4112. [Google Scholar] [CrossRef]
- Turn, R.E.; Hu, Y.; Dewees, S.I.; Devi, N.; East, M.P.; Hardin, K.R.; Khatib, T.; Linnert, J.; Wolfrum, U.; Lim, M.J.; et al. The ARF GAPs ELMOD1 and ELMOD3 Act at the Golgi and Cilia to Regulate Ciliogenesis and Ciliary Protein Traffic. Mol. Biol. Cell 2022, 33, mbcE21090443. [Google Scholar] [CrossRef]
- Will, C.L.; Schneider, C.; Hossbach, M.; Urlaub, H.; Rauhut, R.; Elbashir, S.; Tuschl, T.; LÜhrmann, R. The Human 18S U11/U12 SnRNP Contains a Set of Novel Proteins Not Found in the U2-Dependent Spliceosome. RNA 2004, 10, 929–941. [Google Scholar] [CrossRef]
- Heinicke, L.A.; Nabet, B.; Shen, S.; Jiang, P.; van Zalen, S.; Cieply, B.; Russell, J.E.; Xing, Y.; Carstens, R.P. The RNA Binding Protein RBM38 (RNPC1) Regulates Splicing during Late Erythroid Differentiation. PLoS ONE 2013, 8, e78031. [Google Scholar] [CrossRef]
- She, X.; Lin, Y.; Liang, R.; Liu, Z.; Gao, X.; Ye, J. RNA-Binding Motif Protein 38 as a Potential Biomarker and Therapeutic Target in Cancer. OncoTargets Ther. 2020, 13, 13225–13236. [Google Scholar] [CrossRef]
- Zou, C.; Wan, Y.; He, L.; Zheng, J.H.; Mei, Y.; Shi, J.; Zhang, M.; Dong, Z.; Zhang, D. RBM38 in Cancer: Role and Mechanism. Cell. Mol. Life Sci. 2021, 78, 117–128. [Google Scholar] [CrossRef] [PubMed]
- Cho, S.-J.; Teng, I.-F.; Zhang, M.; Yin, T.; Jung, Y.-S.; Zhang, J.; Chen, X. Hypoxia-Inducible Factor 1 Alpha Is Regulated by RBM38, a RNA-Binding Protein and a P53 Family Target, via MRNA Translation. Oncotarget 2015, 6, 305–316. [Google Scholar] [CrossRef] [PubMed]
- Miyamoto, S.; Hidaka, K.; Jin, D.; Morisaki, T. RNA-Binding Proteins Rbm38 and Rbm24 Regulate Myogenic Differentiation via P21-Dependent and -Independent Regulatory Pathways. Genes Cells 2009, 14, 1241–1252. [Google Scholar] [CrossRef] [PubMed]
- Alvarez-Dominguez, J.R.; Zhang, X.; Hu, W. Widespread and Dynamic Translational Control of Red Blood Cell Development. Blood 2017, 129, 619–629. [Google Scholar] [CrossRef] [PubMed]
- Ding, Z.; Yang, H.-W.; Xia, T.-S.; Wang, B.; Ding, Q. Integrative Genomic Analyses of the RNA-Binding Protein, RNPC1, and Its Potential Role in Cancer Prediction. Int. J. Mol. Med. 2015, 36, 473–484. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Wang, E.; Lu, S.X.; Pastore, A.; Chen, X.; Imig, J.; Chun-Wei Lee, S.; Hockemeyer, K.; Ghebrechristos, Y.E.; Yoshimi, A.; Inoue, D.; et al. Targeting an RNA-Binding Protein Network in Acute Myeloid Leukemia. Cancer Cell 2019, 35, 369–384.e7. [Google Scholar] [CrossRef]
- Suvorova, E.S.; Croken, M.; Kratzer, S.; Ting, L.-M.; Conde de Felipe, M.; Balu, B.; Markillie, M.L.; Weiss, L.M.; Kim, K.; White, M.W. Discovery of a Splicing Regulator Required for Cell Cycle Progression. PLoS Genet. 2013, 9, e1003305. [Google Scholar] [CrossRef]
- Fukuda, T.; Naiki, T.; Saito, M.; Irie, K. HnRNP K Interacts with RNA Binding Motif Protein 42 and Functions in the Maintenance of Cellular ATP Level during Stress Conditions. Genes Cells 2009, 14, 113–128. [Google Scholar] [CrossRef]
- Feng, H.; Liu, J.; Qiu, Y.; Liu, Y.; Saiyin, H.; Liang, X.; Zheng, F.; Wang, Y.; Jiang, D.; Wang, Y.; et al. RNA-Binding Motif Protein 43 (RBM43) Suppresses Hepatocellular Carcinoma Progression through Modulation of Cyclin B1 Expression. Oncogene 2020, 39, 5495–5506. [Google Scholar] [CrossRef]
- Iwamori, T.; Lin, Y.-N.; Ma, L.; Iwamori, N.; Matzuk, M.M. Identification and Characterization of RBM44 as a Novel Intercellular Bridge Protein. PLoS ONE 2011, 6, e17066. [Google Scholar] [CrossRef]
- del Río-Moreno, M.; Alors-Pérez, E.; González-Rubio, S.; Ferrín, G.; Reyes, O.; Rodríguez-Perálvarez, M.; Sánchez-Frías, M.E.; Sánchez-Sánchez, R.; Ventura, S.; López-Miranda, J.; et al. Dysregulation of the Splicing Machinery Is Associated to the Development of Nonalcoholic Fatty Liver Disease. J. Clin. Endocrinol. Metab. 2019, 104, 3389–3402. [Google Scholar] [CrossRef] [PubMed]
- Chen, X.; Yang, Z.; Wang, W.; Qian, K.; Liu, M.; Wang, J.; Wang, M. Structural Basis for RNA Recognition by the N-Terminal Tandem RRM Domains of Human RBM45. Nucleic Acids Res. 2021, 49, 2946–2958. [Google Scholar] [CrossRef]
- Wang, C.; Chen, Y.; Deng, H.; Gao, S.; Li, L. Rbm46 Regulates Trophectoderm Differentiation by Stabilizing Cdx2 MRNA in Early Mouse Embryos. Stem Cells Dev. 2015, 24, 904–915. [Google Scholar] [CrossRef] [PubMed]
- Dai, X.; Cheng, X.; Huang, J.; Gao, Y.; Wang, D.; Feng, Z.; Zhai, G.; Lou, Q.; He, J.; Wang, Z.; et al. Rbm46, a Novel Germ Cell-Specific Factor, Modulates Meiotic Progression and Spermatogenesis. Biol. Reprod. 2021, 104, 1139–1153. [Google Scholar] [CrossRef] [PubMed]
- Shen, D.-J.; Jiang, Y.-H.; Li, J.-Q.; Xu, L.-W.; Tao, K.-Y. The RNA-Binding Protein RBM47 Inhibits Non-Small Cell Lung Carcinoma Metastasis through Modulation of AXIN1 MRNA Stability and Wnt/β-Catentin Signaling. Surg. Oncol. 2020, 34, 31–39. [Google Scholar] [CrossRef] [PubMed]
- Wang, K.; Huang, C.; Jiang, T.; Chen, Z.; Xue, M.; Zhang, Q.; Zhang, J.; Dai, J. RNA-Binding Protein RBM47 Stabilizes IFNAR1 MRNA to Potentiate Host Antiviral Activity. EMBO Rep. 2021, 22, e52205. [Google Scholar] [CrossRef]
- Shivalingappa, P.K.M.; Sharma, V.; Shiras, A.; Bapat, S.A. RNA Binding Motif 47 (RBM47): Emerging Roles in Vertebrate Development, RNA Editing and Cancer. Mol. Cell. Biochem. 2021, 476, 4493–4505. [Google Scholar] [CrossRef]
- Bai, F.; Corll, J.; Shodja, D.N.; Davenport, R.; Feng, G.; Mudunkothge, J.; Brigolin, C.J.; Martin, F.; Spielbauer, G.; Tseung, C.-W.; et al. RNA Binding Motif Protein 48 Is Required for U12 Splicing and Maize Endosperm Differentiation. Plant. Cell 2019, 31, 715–733. [Google Scholar] [CrossRef]
- Li, Z.; Guo, Q.; Zhang, J.; Fu, Z.; Wang, Y.; Wang, T.; Tang, J. The RNA-Binding Motif Protein Family in Cancer: Friend or Foe? Front. Oncol. 2021, 11, 4646. [Google Scholar] [CrossRef]
- Lu, P.; Lu, G.; Yan, C.; Wang, L.; Li, W.; Yin, P. Structure of the MRNA Splicing Complex Component Cwc2: Insights into RNA Recognition. Biochem. J. 2012, 441, 591–597. [Google Scholar] [CrossRef]
- McGrail, J.C.; Krause, A.; O’Keefe, R.T. The RNA Binding Protein Cwc2 Interacts Directly with the U6 SnRNA to Link the Nineteen Complex to the Spliceosome during Pre-MRNA Splicing. Nucleic Acids Res. 2009, 37, 4205–4217. [Google Scholar] [CrossRef] [PubMed]
- Hung, M.-L.; Chao, P.; Chang, K.-Y. DsRBM1 and a Proline-Rich Domain of RNA Helicase A Can Form a Composite Binder to Recognize a Specific DsDNA. Nucleic Acids Res. 2003, 31, 5741–5753. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Li, Q.; Fan, T.; Pang, T.; Yuan, W.; Han, Z. Effect of Proline Rich Domain of an RNA-Binding Protein Sam68 in Cell Growth Process, Death and B Cell Signal Transduction. Chin. Med. J. 2006, 119, 1536–1542. [Google Scholar] [CrossRef] [PubMed]
- Wang, X.; Wang, D.; Zhao, J.; Qu, M.; Zhou, X.; He, H.; He, R. The Proline-Rich Domain and the Microtubule Binding Domain of Protein Tau Acting as RNA Binding Domains. Protein Pept. Lett. 2006, 13, 679–685. [Google Scholar] [CrossRef] [PubMed]
- Schmitzová, J.; Rasche, N.; Dybkov, O.; Kramer, K.; Fabrizio, P.; Urlaub, H.; Lührmann, R.; Pena, V. Crystal Structure of Cwc2 Reveals a Novel Architecture of a Multipartite RNA-Binding Protein. EMBO J. 2012, 31, 2222–2234. [Google Scholar] [CrossRef] [PubMed]
- Proudfoot, N.J.; Furger, A.; Dye, M.J. Integrating MRNA Processing with Transcription. Cell 2002, 108, 501–512. [Google Scholar] [CrossRef]
- Wahl, M.C.; Will, C.L.; Lührmann, R. The Spliceosome: Design Principles of a Dynamic RNP Machine. Cell 2009, 136, 701–718. [Google Scholar] [CrossRef]
- Will, C.L.; Lührmann, R. Spliceosome Structure and Function. Cold Spring Harb. Perspect. Biol. 2011, 3, a003707. [Google Scholar] [CrossRef]
- Hogg, R.; de Almeida, R.A.; Ruckshanthi, J.P.D.; O’Keefe, R.T. Remodeling of U2-U6 SnRNA Helix I during Pre-MRNA Splicing by Prp16 and the NineTeen Complex Protein Cwc2. Nucleic Acids Res. 2014, 42, 8008–8023. [Google Scholar] [CrossRef]
- Steitz, T.A.; Steitz, J.A. A General Two-Metal-Ion Mechanism for Catalytic RNA. Proc. Natl. Acad. Sci. USA 1993, 90, 6498–6502. [Google Scholar] [CrossRef]
- de Almeida, R.A.; O’Keefe, R.T. The NineTeen Complex (NTC) and NTC-Associated Proteins as Targets for Spliceosomal ATPase Action during Pre-MRNA Splicing. RNA Biol. 2015, 12, 109–114. [Google Scholar] [CrossRef] [PubMed]
- Bertram, K.; Agafonov, D.E.; Liu, W.-T.; Dybkov, O.; Will, C.L.; Hartmuth, K.; Urlaub, H.; Kastner, B.; Stark, H.; Lührmann, R. Cryo-EM Structure of a Human Spliceosome Activated for Step 2 of Splicing. Nature 2017, 542, 318–323. [Google Scholar] [CrossRef] [PubMed]
- Hogg, R.; McGrail, J.C.; O’Keefe, R.T. The Function of the NineTeen Complex (NTC) in Regulating Spliceosome Conformations and Fidelity During Pre-MRNA Splicing. Biochem. Soc. Trans. 2010, 38, 1110–1115. [Google Scholar] [CrossRef] [PubMed]
- Kastner, B.; Will, C.L.; Stark, H.; Lührmann, R. Structural Insights into Nuclear Pre-MRNA Splicing in Higher Eukaryotes. Cold Spring Harb. Perspect. Biol. 2019, 11, a032417. [Google Scholar] [CrossRef] [PubMed]
- van der Feltz, C.; Nikolai, B.; Schneider, C.; Paulson, J.C.; Fu, X.; Hoskins, A.A. Saccharomyces Cerevisiae Ecm2 Modulates the Catalytic Steps of Pre-MRNA Splicing. RNA 2021, 27, 591–603. [Google Scholar] [CrossRef] [PubMed]
- Schmitzová, J.; Pena, V. Emerging Views about the Molecular Structure of the Spliceosomal Catalytic Center. RNA Biol. 2012, 9, 1311–1318. [Google Scholar] [CrossRef][Green Version]
- Chan, S.-P.; Kao, D.-I.; Tsai, W.-Y.; Cheng, S.-C. The Prp19p-Associated Complex in Spliceosome Activation. Science 2003, 302, 279–282. [Google Scholar] [CrossRef]
- Chan, S.-P.; Cheng, S.-C. The Prp19-Associated Complex Is Required for Specifying Interactions of U5 and U6 with Pre-MRNA during Spliceosome Activation. J. Biol. Chem. 2005, 280, 31190–31199. [Google Scholar] [CrossRef]
- Chu, H.; Perea, W.; Greenbaum, N.L. Role of the Central Junction in Folding Topology of the Protein-Free Human U2-U6 SnRNA Complex. RNA 2020, 26, 836–850. [Google Scholar] [CrossRef]
- Xu, D.; Friesen, J.D. Splicing Factor Slt11p and Its Involvement in Formation of U2/U6 Helix II in Activation of the Yeast Spliceosome. Mol. Cell. Biol. 2001, 21, 1011–1023. [Google Scholar] [CrossRef][Green Version]
- Park, J.W.; Parisky, K.; Celotto, A.M.; Reenan, R.A.; Graveley, B.R. Identification of Alternative Splicing Regulators by RNA Interference in Drosophila. Proc. Natl. Acad. Sci. USA 2004, 101, 15974–15979. [Google Scholar] [CrossRef] [PubMed]
- Kung, J.T.; Kesner, B.; An, J.Y.; Ahn, J.Y.; Cifuentes-Rojas, C.; Colognori, D.; Jeon, Y.; Szanto, A.; del Rosario, B.C.; Pinter, S.F.; et al. Locus-Specific Targeting to the X Chromosome Revealed by the RNA Interactome of CTCF. Mol. Cell 2015, 57, 361–375. [Google Scholar] [CrossRef] [PubMed]
- Debaize, L.; Jakobczyk, H.; Avner, S.; Gaudichon, J.; Rio, A.-G.; Sérandour, A.A.; Dorsheimer, L.; Chalmel, F.; Carroll, J.S.; Zörnig, M.; et al. Interplay between Transcription Regulators RUNX1 and FUBP1 Activates an Enhancer of the Oncogene C-KIT and Amplifies Cell Proliferation. Nucleic Acids Res. 2018, 46, 11214–11228. [Google Scholar] [CrossRef]
- Debaize, L.; Troadec, M.-B. The Master Regulator FUBP1: Its Emerging Role in Normal Cell Function and Malignant Development. Cell. Mol. Life Sci. 2019, 76, 259–281. [Google Scholar] [CrossRef] [PubMed]
- Sigova, A.A.; Abraham, B.J.; Ji, X.; Molinie, B.; Hannett, N.M.; Guo, Y.E.; Jangi, M.; Giallourakis, C.C.; Sharp, P.A.; Young, R.A. Transcription Factor Trapping by RNA in Gene Regulatory Elements. Science 2015, 350, 978–981. [Google Scholar] [CrossRef] [PubMed]
- Ghosh, A.; Ginty, D.D.; Bading, H.; Greenberg, M.E. Calcium Regulation of Gene Expression in Neuronal Cells. J. Neurobiol. 1994, 25, 294–303. [Google Scholar] [CrossRef] [PubMed]
- Krebs, J. Calmodulin-Dependent Protein Kinase IV: Regulation of Function and Expression. Biochim. Biophys. Acta 1998, 1448, 183–189. [Google Scholar] [CrossRef]
- Krebs, J. The Influence of Calcium Signaling on the Regulation of Alternative Splicing. Biochim. Biophys. Acta (BBA)-Mol. Cell Res. 2009, 1793, 979–984. [Google Scholar] [CrossRef]
- Montaville, P.; Dai, Y.; Cheung, C.Y.; Giller, K.; Becker, S.; Michalak, M.; Webb, S.E.; Miller, A.L.; Krebs, J. Nuclear Translocation of the Calcium-Binding Protein ALG-2 Induced by the RNA-Binding Protein RBM22. Biochim. Biophys. Acta 2006, 1763, 1335–1343. [Google Scholar] [CrossRef]
- Janowicz, A.; Michalak, M.; Krebs, J. Stress Induced Subcellular Distribution of ALG-2, RBM22 and HSlu7. Biochim. Biophys. Acta (BBA)-Mol. Cell Res. 2011, 1813, 1045–1049. [Google Scholar] [CrossRef]
- Fagerberg, L.; Hallström, B.M.; Oksvold, P.; Kampf, C.; Djureinovic, D.; Odeberg, J.; Habuka, M.; Tahmasebpoor, S.; Danielsson, A.; Edlund, K.; et al. Analysis of the Human Tissue-Specific Expression by Genome-Wide Integration of Transcriptomics and Antibody-Based Proteomics. Mol. Cell Proteom. 2014, 13, 397–406. [Google Scholar] [CrossRef] [PubMed]
- He, F.; Wang, C.-T.; Gou, L.-T. RNA-Binding Motif Protein RBM22 Is Required for Normal Development of Zebrafish Embryos. Genet. Mol. Res. 2009, 8, 1466–1473. [Google Scholar] [CrossRef]
- Yamauchi, T.; Masuda, T.; Canver, M.C.; Seiler, M.; Semba, Y.; Shboul, M.; Al-Raqad, M.; Maeda, M.; Schoonenberg, V.A.C.; Cole, M.A.; et al. Genome-Wide CRISPR-Cas9 Screen Identifies Leukemia-Specific Dependence on a Pre-MRNA Metabolic Pathway Regulated by DCPS. Cancer Cell 2018, 33, 386–400.e5. [Google Scholar] [CrossRef] [PubMed]
- Tate, J.G.; Bamford, S.; Jubb, H.C.; Sondka, Z.; Beare, D.M.; Bindal, N.; Boutselakis, H.; Cole, C.G.; Creatore, C.; Dawson, E.; et al. COSMIC: The Catalogue of Somatic Mutations in Cancer. Nucleic Acids Res. 2019, 47, D941–D947. [Google Scholar] [CrossRef] [PubMed]
- Bataller, B.; Javier, F. Análisis Transcriptómico del Espliceosoma en Tejido Cardíaco de Pacientes con Insuficiencia Cardiaca. Relación con la Función Ventricular. Master’s Thesis, Universitat Politècnica de València, Valencia, Spain, 2019. [Google Scholar]
- Gurumayum, S.; Jiang, P.; Hao, X.; Campos, T.L.; Young, N.D.; Korhonen, P.K.; Gasser, R.B.; Bork, P.; Zhao, X.-M.; He, L.; et al. OGEE v3: Online GEne Essentiality Database with Increased Coverage of Organisms and Human Cell Lines. Nucleic Acids Res. 2021, 49, D998–D1003. [Google Scholar] [CrossRef]
- Chan, S.; Sridhar, P.; Kirchner, R.; Lock, Y.J.; Herbert, Z.; Buonamici, S.; Smith, P.; Lieberman, J.; Petrocca, F. Basal-A Triple-Negative Breast Cancer Cells Selectively Rely on RNA Splicing for Survival. Mol. Cancer Ther. 2017, 16, 2849–2861. [Google Scholar] [CrossRef]
- Fuentes-Fayos, A.C.; Vázquez-Borrego, M.C.; Jiménez-Vacas, J.M.; Bejarano, L.; Pedraza-Arévalo, S.; L-López, F.; Blanco-Acevedo, C.; Sánchez-Sánchez, R.; Reyes, O.; Ventura, S.; et al. Splicing Machinery Dysregulation Drives Glioblastoma Development/Aggressiveness: Oncogenic Role of SRSF3. Brain 2020, 143, 3273–3293. [Google Scholar] [CrossRef]
- Konishi, H.; Kashima, S.; Goto, T.; Ando, K.; Sakatani, A.; Tanaka, H.; Ueno, N.; Moriichi, K.; Okumura, T.; Fujiya, M. The Identification of RNA-Binding Proteins Functionally Associated with Tumor Progression in Gastrointestinal Cancer. Cancers 2021, 13, 3165. [Google Scholar] [CrossRef]
- Mao, C.-G.; Jiang, S.-S.; Shen, C.; Long, T.; Jin, H.; Tan, Q.-Y.; Deng, B. BCAR1 Promotes Proliferation and Cell Growth in Lung Adenocarcinoma via Upregulation of POLR2A. Thorac. Cancer 2020, 11, 3326–3336. [Google Scholar] [CrossRef]
- Boultwood, J.; Pellagatti, A.; Cattan, H.; Lawrie, C.H.; Giagounidis, A.; Malcovati, L.; Della Porta, M.G.; Jädersten, M.; Killick, S.; Fidler, C.; et al. Gene Expression Profiling of CD34+ Cells in Patients with the 5q- Syndrome. Br. J. Haematol. 2007, 139, 578–589. [Google Scholar] [CrossRef]
- Giagounidis, A.A.; Germing, U.; Wainscoat, J.S.; Boultwood, J.; Aul, C. The 5q- Syndrome. Hematology 2004, 9, 271–277. [Google Scholar] [CrossRef] [PubMed]
- Liu, T.; Ahmed, T.; Krysiak, K.; Shirai, C.L.; Shao, J.; Nunley, R.; Bucala, R.; McKenzie, A.; Ndonwi, M.; Walter, M.J. Haploinsufficiency of Multiple Del(5q) Genes Induce B Cell Abnormalities in Mice. Leuk Res. 2020, 96, 106428. [Google Scholar] [CrossRef] [PubMed]
- Ge, Z.; Song, E.J.; Kawasawa, Y.I.; Li, J.; Dovat, S.; Song, C. WDR5 High Expression and Its Effect on Tumorigenesis in Leukemia. Oncotarget 2016, 7, 37740–37754. [Google Scholar] [CrossRef] [PubMed]

| IDs of RRM Families | |||||||
|---|---|---|---|---|---|---|---|
| RBMYA1 | 0 | ||||||
| RBMX | 0 | ||||||
| RBM7 | 72 | ||||||
| RBM8 | 60 | ||||||
| RBM11 | 72 | ||||||
| RBM18 | 129 | ||||||
| RBM23 | 32 | 88 | |||||
| RBM24 | 25 | ||||||
| RBM25 | 21 | ||||||
| RBM28 | 36 | 92 | 112 | ||||
| RBM34 | 41 | 98 | |||||
| RBM38 | 25 | ||||||
| RBM39 | 32 | 88 | 52 | ||||
| RBM44 | 210 | ||||||
| RBM45 | 414 | 149 | 235 | ||||
| RBM19 | 19 | 29 | 43 | 120 | 2 | 37 | 58 |
| RBM12 | 161 | 185 | 2 | 205 | |||
| RBMS | 10 | 133 | |||||
| RBM17 | 50 | ||||||
| RBM4 | 102 | ||||||
| RBM4B | 102 | ||||||
| RBM14 | 102 | ||||||
| RBM15 | 136 | 153 | 243 | ||||
| RBM16 | 23 | ||||||
| RBM22 | 77 | ||||||
| RBM26 | 87 | 172 | |||||
| RBM27 | 87 | 172 | |||||
| RBM30 | 102 | ||||||
| RBM46 | 20 | 82 | |||||
| RBM47 | 20 | 82 | |||||
| RBM5 | 130 | 213 | |||||
| RBM6 | 213 | ||||||
| RBM10 | 130 | 213 | |||||
| RBM40 | 113 | 169 | |||||
| RBM41 | 113 | ||||||
| RBM9 | 141 | ||||||
| RBM20 | 46 | ||||||
| RBM21 | 232 | ||||||
| RBM36 | 152 | ||||||
| RBM42 | 81 | ||||||
| RBM48 | 151 | ||||||
| Biological Processes | Biological Functions | |||||||
|---|---|---|---|---|---|---|---|---|
| mRNA Splicing | RNA Stability | RNA Transport | Translation | Apoptosis | Cell Cycle | Differentiation | Other Functions | |
| RBMY1A1 | [31] | Sperm cells [37] | Sperm motility [38,39] | |||||
| RBMX | [40] | [41] | Zebrafish brain development [42] Cohesion of sister chromatids [43,44] Genome maintenance [45] | |||||
| RBM3 | [46,47] | [41,48] | DNA Damage Response [49] Circadian cycle [50] Cell proliferation [51,52] | |||||
| RBM4 | [34,53,54] | [53] | Muscle [55] Neuronal [56] Pancreas [57] | Circadian cycle [58] | ||||
| RBM4B | Circadian cycle [58] | |||||||
| RBM5 | [34,59] | [8] | [60] | Sperm cells [61] | Cell proliferation [59] | |||
| RBM6 | [34,59] | [8] | Cell proliferation [59] | |||||
| RBM7 | [62] | |||||||
| RBM8A | [63] | [63] | [64] | [65] | [66,67] | [66,67] | Neural [68] | Cell proliferation [68] DNA damage signaling & response [67,69] Cortical development [63] |
| RBM9 | [33] | Pluripotent stem cells [70] | ||||||
| RBM10 | [34,59,71] | [8,72] | [73] | Cell proliferation [59,74] | ||||
| RBM11 | [75] | Neuro/Germinal [75] | ||||||
| RBM12 | ||||||||
| RBM13 | ||||||||
| RBM14 | [76] | Mouse embryo development [77] DNA damage response [76,78] | ||||||
| RBM15 | [79] | [80] | [81] | Blood cells [82,83] | RNA-methylation regulation [84] | |||
| RBM16 | Transcription [85] | |||||||
| RBM17 | [86] | [87,88] | Cell proliferation [88,89] | |||||
| RBM18 | ||||||||
| RBM19 | Digestive tube [90,91] | rRNA processing [92] Pre-implantation development of mouse embryo [93] | ||||||
| RBM20 | [94] | |||||||
| RBM21 | [95] | RNA poly-adenylation [96] | ||||||
| RBM22 | [29,97,98,99,100,101] | [102] | Erythroid [103] | Transcription regulation [104,105] Zebrafish development [90] Drosophila development [106] | ||||
| RBM23 | [107] | |||||||
| RBM24 | [108,109] | [110] | [111] | Embryo [108] Heart [111] | Cell proliferation [112] | |||
| RBM25 | [113,114,115] | [115] | Cell proliferation [115] | |||||
| RBM26 | [35] | |||||||
| RBM27 | [35] | |||||||
| RBM28 | [116] | |||||||
| RBM29 | Cell hair development in cochlea [117,118] | |||||||
| RBM30 | Circadian cycle [58] | |||||||
| RBM33 | ||||||||
| RBM34 | ||||||||
| RBM36 | [119] | |||||||
| RBM38 | [109,120] | [121,122] | [121,123] | [121] | Heart [124] Erythrocytes [120,125] | Cell proliferation [126] | ||
| RBM39 | [127] | |||||||
| RBM40 | [119] | |||||||
| RBM41 | ||||||||
| RBM42 | [128] | [129] | [128] | |||||
| RBM43 | [130] | [130] | Cell proliferation [130] | |||||
| RBM44 | Meiosis [131] | |||||||
| RBM45 | [132] | Neural [133] | ||||||
| RBM46 | [134] | Spermatogenesis [135] Mouse embryo development [134] | ||||||
| RBM47 | [136,137] | RNA editing [138] Cell proliferation [136] | ||||||
| RBM48 | [139] | |||||||
| From Complex | Protein Partner | References |
|---|---|---|
| NTC | Cdc5 | Zhang et al., 2017 [29] |
| Isy1 | McGrail et al., 2009 [142]; Hogg et al., 2014 [150] | |
| PRP19 | McGrail et al., 2009 [142] | |
| NTR | Aquarius | Bertram et al., 2017 [153]; Kastner et al., 2019 [155] |
| G10 | Bertram et al., 2017 [153]; Zhang et al., 2017 [29] | |
| PRP17 | Zhang et al., 2017 [29] | |
| SKIP | Bertram et al., 2017 [153]; Zhang et al., 2017 [29] | |
| Other | PPIL1 | Bertram et al., 2017 [153]; Zhang et al., 2017 [29]; Zhan et al., 2018 [98] |
| PRP8 | Bertram et al., 2017 [153] | |
| SLU7 | Xu et al., 2001 [161] |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Soubise, B.; Jiang, Y.; Douet-Guilbert, N.; Troadec, M.-B. RBM22, a Key Player of Pre-mRNA Splicing and Gene Expression Regulation, Is Altered in Cancer. Cancers 2022, 14, 643. https://doi.org/10.3390/cancers14030643
Soubise B, Jiang Y, Douet-Guilbert N, Troadec M-B. RBM22, a Key Player of Pre-mRNA Splicing and Gene Expression Regulation, Is Altered in Cancer. Cancers. 2022; 14(3):643. https://doi.org/10.3390/cancers14030643
Chicago/Turabian StyleSoubise, Benoît, Yan Jiang, Nathalie Douet-Guilbert, and Marie-Bérengère Troadec. 2022. "RBM22, a Key Player of Pre-mRNA Splicing and Gene Expression Regulation, Is Altered in Cancer" Cancers 14, no. 3: 643. https://doi.org/10.3390/cancers14030643
APA StyleSoubise, B., Jiang, Y., Douet-Guilbert, N., & Troadec, M.-B. (2022). RBM22, a Key Player of Pre-mRNA Splicing and Gene Expression Regulation, Is Altered in Cancer. Cancers, 14(3), 643. https://doi.org/10.3390/cancers14030643






