The Role of Circulating Tumor Cells in Ovarian Cancer Dissemination
Abstract
Simple Summary
Abstract
1. Introduction
2. Biology of Circulating Tumor Cells
2.1. The Ever-Changing Phenotype of CTCs
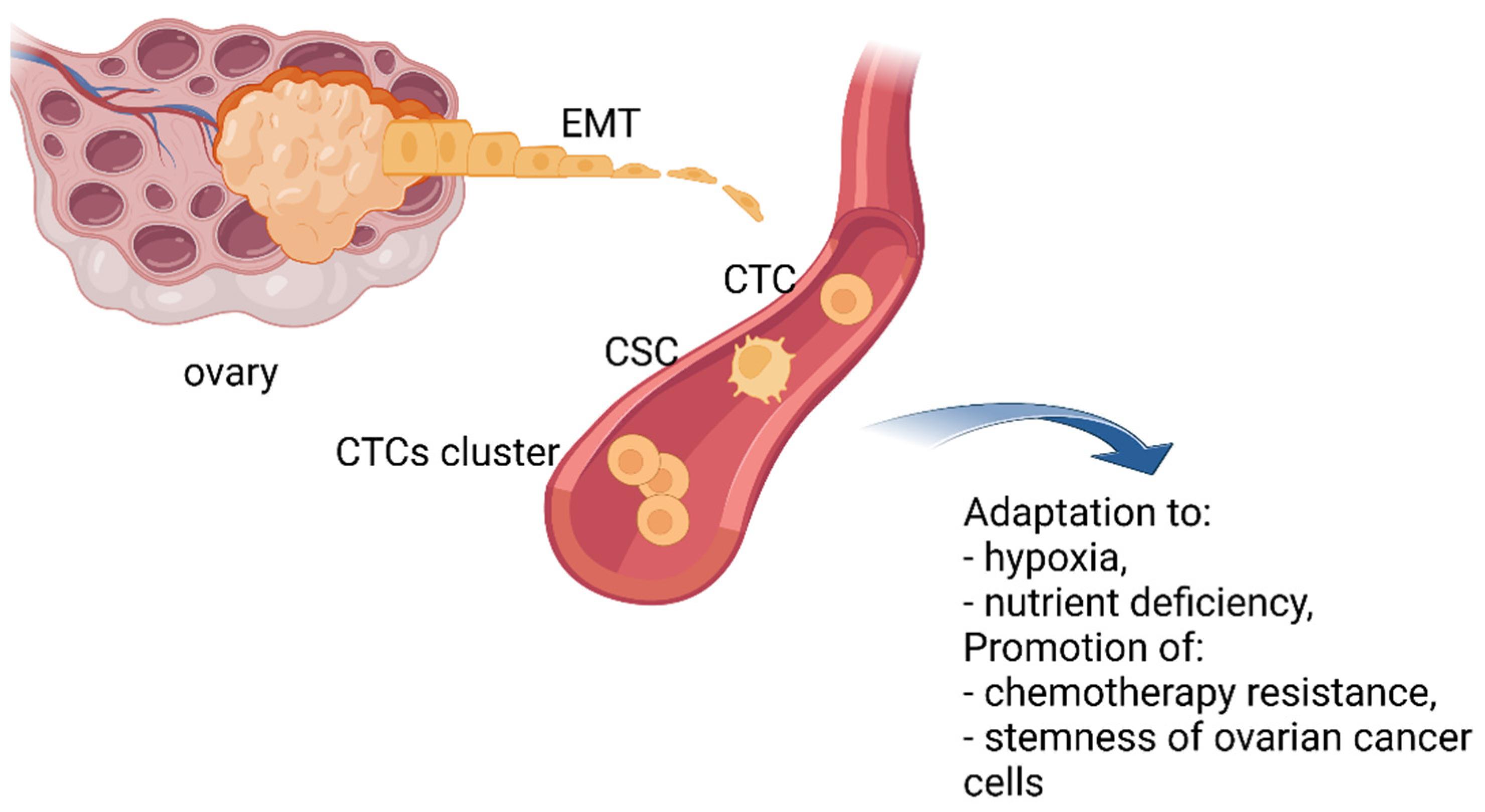
2.2. CTCs Clusters
2.3. CTCs and Cancer Stem Cells
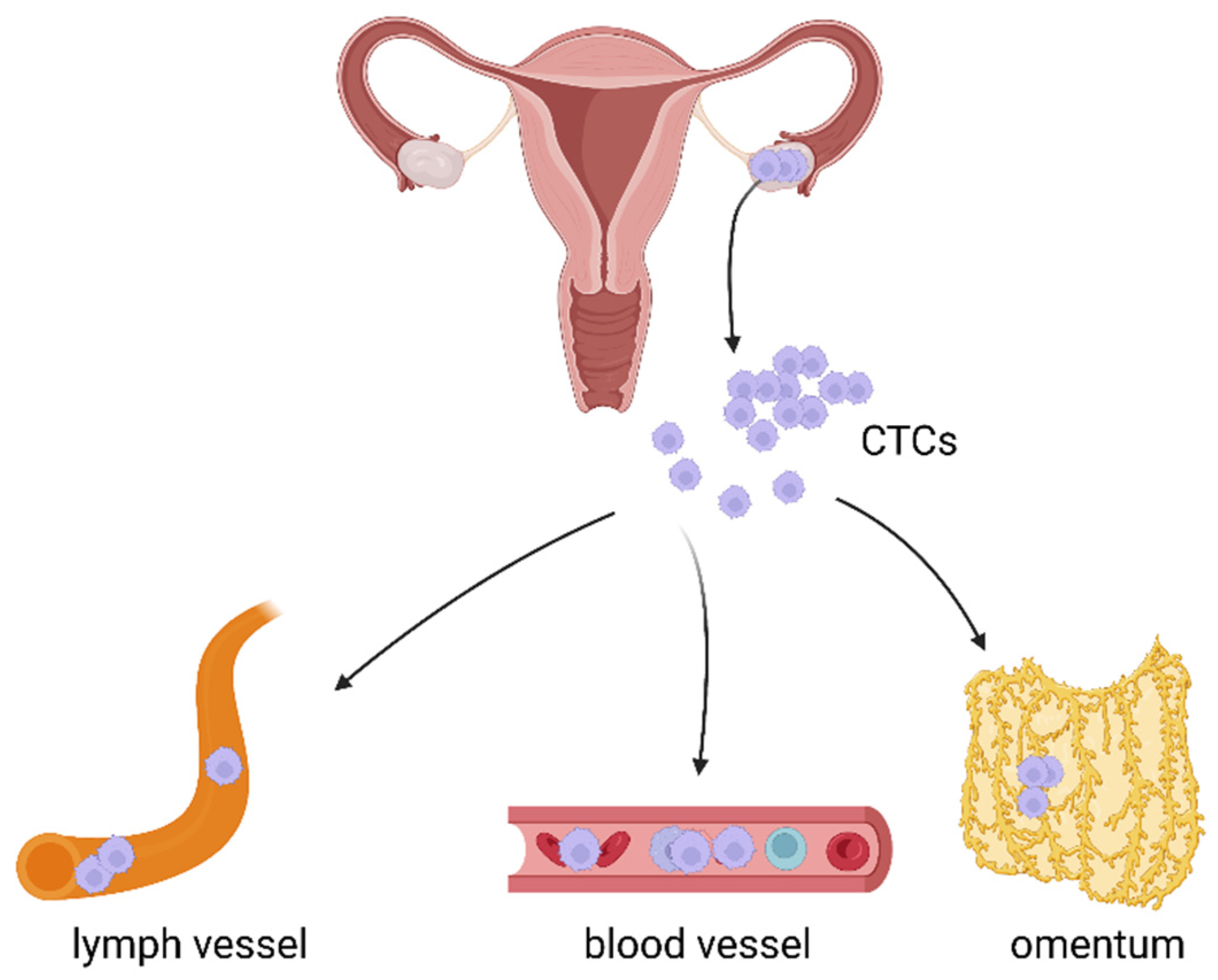
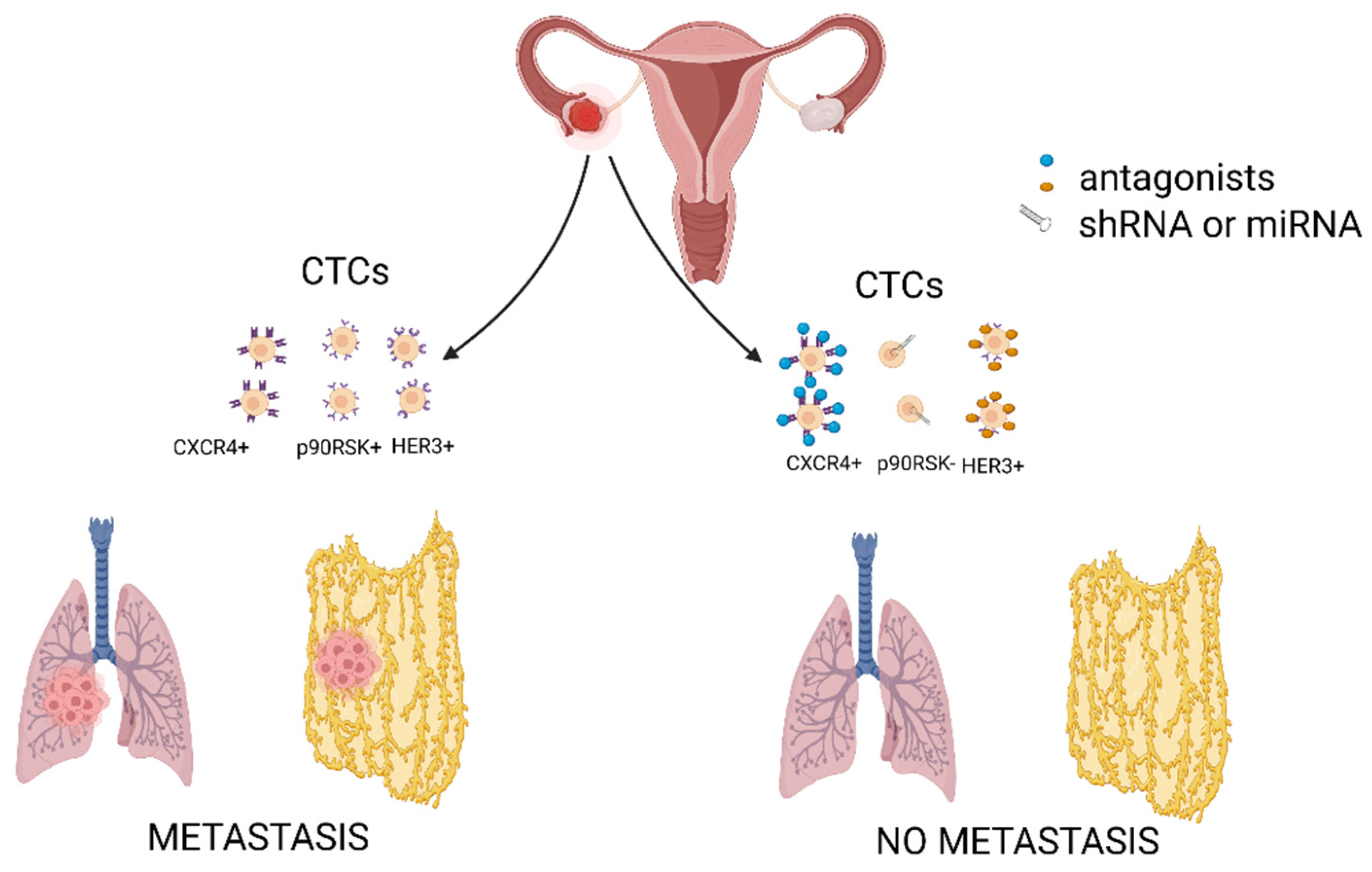
3. The Role of CTCs in Haematogenous Metastasis of Ovarian Cancer
4. Clinical Relevance of CTCs in Ovarian Cancer
5. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Siegel, R.L.; Miller, K.D.; Jemal, A. Cancer Statistics, 2020. CA. Cancer J. Clin. 2020, 70, 7–30. [Google Scholar] [CrossRef] [PubMed]
- Momenimovahed, Z.; Tiznobaik, A.; Taheri, S.; Salehiniya, H. Ovarian Cancer in the World: Epidemiology and Risk Factors. Int. J. Womens Health 2019, 11, 287–299. [Google Scholar] [CrossRef] [PubMed]
- Howlader, N.; Noone, A.; Krapcho, M.; Miller, D.; Brest, A.; Yu, M. SEER Cancer Statistics Review, 1975-2018, National Cancer Institute. Bethesda. Available online: https://seer.cancer.gov/csr/1975_2018/, (accessed on 31 October 2022).
- Rose, P.G.; Piver, M.S.; Tsukada, Y.; Lau, T. Metastatic Patterns in Histologic Variants of Ovarian Cancer. An Autopsy Study. Cancer 1989, 64, 1508–1513. [Google Scholar] [CrossRef]
- Güth, U.; Arndt, V.; Stadlmann, S.; Huang, D.J.; Singer, G. Epidemiology in Ovarian Carcinoma: Lessons from Autopsy. Gynecol. Oncol. 2015, 138, 417–420. [Google Scholar] [CrossRef] [PubMed]
- Lengyel, E. Ovarian Cancer Development and Metastasis. Am. J. Pathol. 2010, 177, 1053–1064. [Google Scholar] [CrossRef]
- Obermayr, E.; Reiner, A.; Brandt, B.; Braicu, E.I.; Reinthaller, A.; Loverix, L.; Concin, N.; Woelber, L.; Mahner, S.; Sehouli, J.; et al. The Long-Term Prognostic Significance of Circulating Tumor Cells in Ovarian Cancer—A Study of the OVCAD Consortium. Cancers 2021, 13, 2613. [Google Scholar] [CrossRef]
- Kolostova, K.; Pinkas, M.; Jakabova, A.; Pospisilova, E.; Svobodova, P.; Spicka, J.; Cegan, M.; Matkowski, R.; Bobek, V. Molecular Characterization of Circulating Tumor Cells in Ovarian Cancer. Am. J. Cancer Res. 2016, 6, 973–980. [Google Scholar]
- Pearl, M.L.; Dong, H.; Tulley, S.; Zhao, Q.; Golightly, M.; Zucker, S.; Chen, W.-T. Treatment Monitoring of Patients with Epithelial Ovarian Cancer Using Invasive Circulating Tumor Cells (ICTCs). Gynecol. Oncol. 2015, 137, 229–238. [Google Scholar] [CrossRef] [PubMed]
- Tarin, D.; Price, J.E.; Kettlewell, M.G.; Souter, R.G.; Vass, A.C.; Crossley, B. Mechanisms of Human Tumor Metastasis Studied in Patients with Peritoneovenous Shunts. Cancer Res. 1984, 44, 3584–3592. [Google Scholar]
- Lou, E.; Vogel, R.I.; Teoh, D.; Hoostal, S.; Grad, A.; Gerber, M.; Monu, M.; Łukaszewski, T.; Deshpande, J.; Linden, M.A.; et al. Assessment of Circulating Tumor Cells as a Predictive Biomarker of Histology in Women With Suspected Ovarian Cancer. Lab. Med. 2018, 49, 134–139. [Google Scholar] [CrossRef]
- Banys-Paluchowski, M.; Fehm, T.; Neubauer, H.; Paluchowski, P.; Krawczyk, N.; Meier-Stiegen, F.; Wallach, C.; Kaczerowsky, A.; Gebauer, G. Clinical Relevance of Circulating Tumor Cells in Ovarian, Fallopian Tube and Peritoneal Cancer. Arch. Gynecol. Obstet. 2020, 301, 1027–1035. [Google Scholar] [CrossRef] [PubMed]
- Yousefi, M.; Dehghani, S.; Nosrati, R.; Ghanei, M.; Salmaninejad, A.; Rajaie, S.; Hasanzadeh, M.; Pasdar, A. Current Insights into the Metastasis of Epithelial Ovarian Cancer—Hopes and Hurdles. Cell. Oncol. 2020, 43, 515–538. [Google Scholar] [CrossRef]
- Zeng, L.; Liang, X.; Liu, Q.; Yang, Z. The Predictive Value of Circulating Tumor Cells in Ovarian Cancer: A Meta Analysis. Int. J. Gynecol. Cancer 2017, 27, 1109–1117. [Google Scholar] [CrossRef]
- Lee, M.; Kim, E.J.; Cho, Y.; Kim, S.; Chung, H.H.; Park, N.H.; Song, Y.-S. Predictive Value of Circulating Tumor Cells (CTCs) Captured by Microfluidic Device in Patients with Epithelial Ovarian Cancer. Gynecol. Oncol. 2017, 145, 361–365. [Google Scholar] [CrossRef]
- Poveda, A.; Kaye, S.B.; McCormack, R.; Wang, S.; Parekh, T.; Ricci, D.; Lebedinsky, C.A.; Tercero, J.C.; Zintl, P.; Monk, B.J. Circulating Tumor Cells Predict Progression Free Survival and Overall Survival in Patients with Relapsed/Recurrent Advanced Ovarian Cancer. Gynecol. Oncol. 2011, 122, 567–572. [Google Scholar] [CrossRef]
- Pearl, M.L.; Zhao, Q.; Yang, J.; Dong, H.; Tulley, S.; Zhang, Q.; Golightly, M.; Zucker, S.; Chen, W.-T. Prognostic Analysis of Invasive Circulating Tumor Cells (ICTCs) in Epithelial Ovarian Cancer. Gynecol. Oncol. 2014, 134, 581–590. [Google Scholar] [CrossRef]
- Chebouti, I.; Kasimir-Bauer, S.; Buderath, P.; Wimberger, P.; Hauch, S.; Kimmig, R.; Kuhlmann, J.D. EMT-like Circulating Tumor Cells in Ovarian Cancer Patients Are Enriched by Platinum-Based Chemotherapy. Oncotarget 2017, 8, 48820–48831. [Google Scholar] [CrossRef] [PubMed]
- Bregenzer, M.E.; Horst, E.N.; Mehta, P.; Novak, C.M.; Repetto, T.; Snyder, C.S.; Mehta, G. Tumor Modeling Maintains Diverse Pathology in Vitro. Ann. Transl. Med. 2019, 7, S262. [Google Scholar] [CrossRef] [PubMed]
- Cheng, H.; Wang, S.; Luan, W.; Ye, X.; Dou, S.; Tang, Z.; Zhu, H.; Lin, P.P.; Li, Y.; Cui, H.; et al. Combined Detection and Subclass Characteristics Analysis of CTCs and CTECs by SE-IFISH in Ovarian Cancer. Chin. J. Cancer Res. 2021, 33, 256–270. [Google Scholar] [CrossRef] [PubMed]
- Bankó, P.; Lee, S.Y.; Nagygyörgy, V.; Zrínyi, M.; Chae, C.H.; Cho, D.H.; Telekes, A. Technologies for Circulating Tumor Cell Separation from Whole Blood. J. Hematol. Oncol. 2019, 12, 48. [Google Scholar] [CrossRef] [PubMed]
- Kolostova, K.; Matkowski, R.; Jędryka, M.; Soter, K.; Cegan, M.; Pinkas, M.; Jakabova, A.; Pavlasek, J.; Spicka, J.; Bobek, V. The Added Value of Circulating Tumor Cells Examination in Ovarian Cancer Staging. Am. J. Cancer Res. 2015, 5, 3363–3375. [Google Scholar]
- Chebouti, I.; Kuhlmann, J.D.; Buderath, P.; Weber, S.; Wimberger, P.; Bokeloh, Y.; Hauch, S.; Kimmig, R.; Kasimir-Bauer, S. ERCC1-Expressing Circulating Tumor Cells as a Potential Diagnostic Tool for Monitoring Response to Platinum-Based Chemotherapy and for Predicting Post-Therapeutic Outcome of Ovarian Cancer. Oncotarget 2017, 8, 24303–24313. [Google Scholar] [CrossRef]
- Blassl, C.; Kuhlmann, J.D.; Webers, A.; Wimberger, P.; Fehm, T.; Neubauer, H. Gene Expression Profiling of Single Circulating Tumor Cells in Ovarian Cancer—Establishment of a Multi-Marker Gene Panel. Mol. Oncol. 2016, 10, 1030–1042. [Google Scholar] [CrossRef]
- Yousefi, M.; Rajaie, S.; Keyvani, V.; Bolandi, S.; Hasanzadeh, M.; Pasdar, A. Clinical Significance of Circulating Tumor Cell Related Markers in Patients with Epithelial Ovarian Cancer before and after Adjuvant Chemotherapy. Sci. Rep. 2021, 11, 10524. [Google Scholar] [CrossRef] [PubMed]
- Asante, D.-B.; Calapre, L.; Ziman, M.; Meniawy, T.M.; Gray, E.S. Liquid Biopsy in Ovarian Cancer Using Circulating Tumor DNA and Cells: Ready for Prime Time? Cancer Lett. 2020, 468, 59–71. [Google Scholar] [CrossRef] [PubMed]
- Genna, A.; Vanwynsberghe, A.M.; Villard, A.V.; Pottier, C.; Ancel, J.; Polette, M.; Gilles, C. EMT-Associated Heterogeneity in Circulating Tumor Cells: Sticky Friends on the Road to Metastasis. Cancers 2020, 12, 1632. [Google Scholar] [CrossRef]
- Loret, N.; Denys, H.; Tummers, P.; Berx, G. The Role of Epithelial-to-Mesenchymal Plasticity in Ovarian Cancer Progression and Therapy Resistance. Cancers 2019, 11, 838. [Google Scholar] [CrossRef]
- Thankamony, A.P.; Saxena, K.; Murali, R.; Jolly, M.K.; Nair, R. Cancer Stem Cell Plasticity—A Deadly Deal. Front. Mol. Biosci. 2020, 7, 79. [Google Scholar] [CrossRef]
- Schuster, E.; Taftaf, R.; Reduzzi, C.; Albert, M.K.; Romero-Calvo, I.; Liu, H. Better Together: Circulating Tumor Cell Clustering in Metastatic Cancer. Trends Cancer 2021, 7, 1020–1032. [Google Scholar] [CrossRef]
- Amintas, S.; Bedel, A.; Moreau-Gaudry, F.; Boutin, J.; Buscail, L.; Merlio, J.-P.; Vendrely, V.; Dabernat, S.; Buscail, E. Circulating Tumor Cell Clusters: United We Stand Divided We Fall. Int. J. Mol. Sci. 2020, 21, 2653. [Google Scholar] [CrossRef]
- Gkountela, S.; Castro-Giner, F.; Szczerba, B.M.; Vetter, M.; Landin, J.; Scherrer, R.; Krol, I.; Scheidmann, M.C.; Beisel, C.; Stirnimann, C.U.; et al. Circulating Tumor Cell Clustering Shapes DNA Methylation to Enable Metastasis Seeding. Cell 2019, 176, 98–112.e14. [Google Scholar] [CrossRef] [PubMed]
- Nelep, C.; Eberhardt, J. Automated Rare Single Cell Picking with the ALS CellcelectorTM. Cytom. Part A 2018, 93, 1267–1270. [Google Scholar] [CrossRef] [PubMed]
- Virant-Klun, I.; Zech, N.; Rožman, P.; Vogler, A.; Cvjetičanin, B.; Klemenc, P.; Maličev, E.; Meden-Vrtovec, H. Putative Stem Cells with an Embryonic Character Isolated from the Ovarian Surface Epithelium of Women with No Naturally Present Follicles and Oocytes. Differentiation 2008, 76, 843–856. [Google Scholar] [CrossRef]
- Parte, S.; Bhartiya, D.; Telang, J.; Daithankar, V.; Salvi, V.; Zaveri, K.; Hinduja, I. Detection, Characterization, and Spontaneous Differentiation In Vitro of Very Small Embryonic-Like Putative Stem Cells in Adult Mammalian Ovary. Stem Cells Dev. 2011, 20, 1451–1464. [Google Scholar] [CrossRef] [PubMed]
- Liao, J.; Qian, F.; Tchabo, N.; Mhawech-Fauceglia, P.; Beck, A.; Qian, Z.; Wang, X.; Huss, W.J.; Lele, S.B.; Morrison, C.D.; et al. Ovarian Cancer Spheroid Cells with Stem Cell-like Properties Contribute to Tumor Generation, Metastasis and Chemotherapy Resistance through Hypoxia-Resistant Metabolism. PLoS ONE 2014, 9, e84941. [Google Scholar] [CrossRef]
- Keyvani, V.; Farshchian, M.; Esmaeili, S.-A.; Yari, H.; Moghbeli, M.; Nezhad, S.-R.K.; Abbaszadegan, M.R. Ovarian Cancer Stem Cells and Targeted Therapy. J. Ovarian Res. 2019, 12, 120. [Google Scholar] [CrossRef] [PubMed]
- Auersperg, N. The Stem-Cell Profile of Ovarian Surface Epithelium Is Reproduced in the Oviductal Fimbriae, with Increased Stem-Cell Marker Density in Distal Parts of the Fimbriae. Int. J. Gynecol. Pathol. 2013, 32, 444–453. [Google Scholar] [CrossRef]
- Bapat, S.A.; Mali, A.M.; Koppikar, C.B.; Kurrey, N.K. Stem and Progenitor-Like Cells Contribute to the Aggressive Behavior of Human Epithelial Ovarian Cancer. Cancer Res. 2005, 65, 3025–3029. [Google Scholar] [CrossRef]
- Hu, L.; McArthur, C.; Jaffe, R.B. Ovarian Cancer Stem-like Side-Population Cells Are Tumourigenic and Chemoresistant. Br. J. Cancer 2010, 102, 1276–1283. [Google Scholar] [CrossRef]
- Muñoz-Galván, S.; Carnero, A. Targeting Cancer Stem Cells to Overcome Therapy Resistance in Ovarian Cancer. Cells 2020, 9, 1402. [Google Scholar] [CrossRef]
- Kim, M.; Suh, D.H.; Choi, J.Y.; Bu, J.; Kang, Y.-T.; Kim, K.; No, J.H.; Kim, Y.B.; Cho, Y.-H. Post-Debulking Circulating Tumor Cell as a Poor Prognostic Marker in Advanced Stage Ovarian Cancer. Medicine 2019, 98, e15354. [Google Scholar] [CrossRef] [PubMed]
- Aktas, B.; Kasimir-Bauer, S.; Heubner, M.; Kimmig, R.; Wimberger, P. Molecular Profiling and Prognostic Relevance of Circulating Tumor Cells in the Blood of Ovarian Cancer Patients at Primary Diagnosis and after Platinum-Based Chemotherapy. Int. J. Gynecol. Cancer 2011, 21, 822–830. [Google Scholar] [CrossRef] [PubMed]
- Ma, J.; Yang, J.; Jin, Y.; Cheng, S.; Huang, S.; Zhang, N.; Wang, Y. Artificial Intelligence Based on Blood Biomarkers Including CTCs Predicts Outcomes in Epithelial Ovarian Cancer: A Prospective Study. Onco. Targets. Ther. 2021, 14, 3267–3280. [Google Scholar] [CrossRef] [PubMed]
- Motohara, T.; Fujimoto, K.; Tayama, S.; Narantuya, D.; Sakaguchi, I.; Tashiro, H.; Katabuchi, H. CD44 Variant 6 as a Predictive Biomarker for Distant Metastasis in Patients With Epithelial Ovarian Cancer. Obstet. Gynecol. 2016, 127, 1003–1011. [Google Scholar] [CrossRef] [PubMed]
- Bai, S.; Ingrfam, P.; Chen, Y.-C.; Deng, N.; Pearson, A.; Niknafs, Y.S.; O’Hayer, P.; Wang, Y.; Zhang, Z.-Y.; Boscolo, E.; et al. EGFL6 Regulates the Asymmetric Division, Maintenance, and Metastasis of ALDH+ Ovarian Cancer Cells. Cancer Res. 2016, 76, 6396–6409. [Google Scholar] [CrossRef] [PubMed]
- Zhang, X.; Li, H.; Yu, X.; Li, S.; Lei, Z.; Li, C.; Zhang, Q.; Han, Q.; Li, Y.; Zhang, K.; et al. Analysis of Circulating Tumor Cells in Ovarian Cancer and Their Clinical Value as a Biomarker. Cell. Physiol. Biochem. 2018, 48, 1983–1994. [Google Scholar] [CrossRef]
- Zuo, L.; Li, X.; Zhu, H.; Li, A.; Wang, Y. Expression of MiR-181a in Circulating Tumor Cells of Ovarian Cancer and Its Clinical Application. ACS Omega 2021, 6, 22011–22019. [Google Scholar] [CrossRef]
- Yang, J.; Ma, J.; Jin, Y.; Cheng, S.; Huang, S.; Zhang, N.; Wang, Y. Development and Validation for Prognostic Nomogram of Epithelial Ovarian Cancer Recurrence Based on Circulating Tumor Cells and Epithelial-Mesenchymal Transition. Sci. Rep. 2021, 11, 6540. [Google Scholar] [CrossRef]
- Marth, C.; Kisic, J.; Kaern, J.; Tropé, C.; Fodstad, Ø. Circulating Tumor Cells in the Peripheral Blood and Bone Marrow of Patients with Ovarian Carcinoma Do Not Predict Prognosis. Cancer 2002, 94, 707–712. [Google Scholar] [CrossRef]
- Pradeep, S.; Kim, S.W.; Wu, S.Y.; Nishimura, M.; Chaluvally-Raghavan, P.; Miyake, T.; Pecot, C.V.; Kim, S.-J.; Choi, H.J.; Bischoff, F.Z.; et al. Hematogenous Metastasis of Ovarian Cancer: Rethinking Mode of Spread. Cancer Cell 2014, 26, 77–91. [Google Scholar] [CrossRef]
- Izraely, S.; Witz, I.P. Site-Specific Metastasis: A Cooperation between Cancer Cells and the Metastatic Microenvironment. Int. J. Cancer 2021, 148, 1308–1322. [Google Scholar] [CrossRef] [PubMed]
- Cîrstea, A.E.; Stepan, A.E.; Mărgăritescu, C.; Zăvoi, R.E.; Olimid, D.A.; Simionescu, C.E. The Immunoexpression of EGFR, HER2 and HER3 in Malignant Serous Ovarian Tumors. Rom. J. Morphol. Embryol. 2017, 58, 1269–1273. [Google Scholar] [PubMed]
- Mizuno, T.; Kojima, Y.; Yonemori, K.; Yoshida, H.; Sugiura, Y.; Ohtake, Y.; Okuma, H.; Nishikawa, T.; Tanioka, M.; Sudo, K.; et al. Neoadjuvant Chemotherapy Promotes the Expression of HER3 in Patients with Ovarian Cancer. Oncol. Lett. 2020, 20, 336. [Google Scholar] [CrossRef] [PubMed]
- Figueras, A.; Alsina-Sanchís, E.; Lahiguera, Á.; Abreu, M.; Muinelo-Romay, L.; Moreno-Bueno, G.; Casanovas, O.; Graupera, M.; Matias-Guiu, X.; Vidal, A.; et al. A Role for CXCR4 in Peritoneal and Hematogenous Ovarian Cancer Dissemination. Mol. Cancer Ther. 2018, 17, 532–543. [Google Scholar] [CrossRef]
- Liu, W.; Wang, W.; Zhang, N.; Di, W. The Role of CCL20-CCR6 Axis in Ovarian Cancer Metastasis. OncoTargets Ther. 2020, 13, 12739–12750. [Google Scholar] [CrossRef]
- Zou, W.; Wicha, M.S. Chemokines and Cellular Plasticity of Ovarian Cancer Stem Cells. Oncoscience 2015, 2, 615–616. [Google Scholar] [CrossRef]
- Long, H.; Xiang, T.; Qi, W.; Huang, J.; Chen, J.; He, L.; Liang, Z.; Guo, B.; Li, Y.; Xie, R.; et al. CD133+ Ovarian Cancer Stem-like Cells Promote Non-Stem Cancer Cell Metastasis via CCL5 Induced Epithelial-Mesenchymal Transition. Oncotarget 2015, 6, 5846–5859. [Google Scholar] [CrossRef]
- Wang, Q.; Tang, Y.; Yu, H.; Yin, Q.; Li, M.; Shi, L.; Zhang, W.; Li, D.; Li, L. CCL18 from Tumor-Cells Promotes Epithelial Ovarian Cancer Metastasis via MTOR Signaling Pathway. Mol. Carcinog. 2016, 55, 1688–1699. [Google Scholar] [CrossRef]
- Johnson, E.L.; Singh, R.; Johnson-Holiday, C.M.; Grizzle, W.E.; Partridge, E.E.; Lillard, J.W.; Singh, S. CCR9 Interactions Support Ovarian Cancer Cell Survival and Resistance to Cisplatin-Induced Apoptosis in a PI3K-Dependent and FAK-Independent Fashion. J. Ovarian Res. 2010, 3, 15. [Google Scholar] [CrossRef]
- Cioffi, M.; D’Alterio, C.; Camerlingo, R.; Tirino, V.; Consales, C.; Riccio, A.; Ieranò, C.; Cecere, S.C.; Losito, N.S.; Greggi, S.; et al. Identification of a Distinct Population of CD133(+)CXCR4(+) Cancer Stem Cells in Ovarian Cancer. Sci. Rep. 2015, 5, 10357. [Google Scholar] [CrossRef]
- Johnson, E.L.; Singh, R.; Singh, S.; Johnson-Holiday, C.M.; Grizzle, W.E.; Partridge, E.E.; Lillard, J.W. CCL25-CCR9 Interaction Modulates Ovarian Cancer Cell Migration, Metalloproteinase Expression, and Invasion. World J. Surg. Oncol. 2010, 8, 62. [Google Scholar] [CrossRef]
- Torchiaro, E.; Lorenzato, A.; Olivero, M.; Valdembri, D.; Gagliardi, P.A.; Gai, M.; Erriquez, J.; Serini, G.; Di Renzo, M.F. Peritoneal and Hematogenous Metastases of Ovarian Cancer Cells Are Both Controlled by the P90RSK through a Self-Reinforcing Cell Autonomous Mechanism. Oncotarget 2016, 7, 712–728. [Google Scholar] [CrossRef] [PubMed]
- Coffman, L.G.; Burgos-Ojeda, D.; Wu, R.; Cho, K.; Bai, S.; Buckanovich, R.J. New Models of Hematogenous Ovarian Cancer Metastasis Demonstrate Preferential Spread to the Ovary and a Requirement for the Ovary for Abdominal Dissemination. Transl. Res. 2016, 175, 92–102.e2. [Google Scholar] [CrossRef] [PubMed]
- Thomakos, N.; Diakosavvas, M.; Machairiotis, N.; Fasoulakis, Z.; Zarogoulidis, P.; Rodolakis, A. Rare Distant Metastatic Disease of Ovarian and Peritoneal Carcinomatosis: A Review of the Literature. Cancers 2019, 11, 1044. [Google Scholar] [CrossRef] [PubMed]
- Li, J.; Yue, H.; Li, W.; Zhu, G.; Zhu, T.; Chen, R.; Lu, X. Bevacizumab Confers Significant Improvements in Survival for Ovarian Cancer Patients with Low MiR-25 Expression and High MiR-142 Expression. J. Ovarian Res. 2021, 14, 166. [Google Scholar] [CrossRef]
- Yue, H.; Wang, J.; Chen, R.; Hou, X.; Li, J.; Lu, X. Gene Signature Characteristic of Elevated Stromal Infiltration and Activation Is Associated with Increased Risk of Hematogenous and Lymphatic Metastasis in Serous Ovarian Cancer. BMC Cancer 2019, 19, 1266. [Google Scholar] [CrossRef]
- Erdem, B.; Yüksel, I.T.; Peker, N.; Ulukent, S.C.; Aşıcıoğlu, O.; Özaydin, I.Y.; Ülker, V.; Akbayir, O. Evaluation of Factors Affecting Lymph Node Metastasis in Clinical Stage I-II Epithelial Ovarian Cancer. Oncol. Res. Treat. 2018, 41, 444–448. [Google Scholar] [CrossRef]
- Fan, T.; Zhao, Q.; Chen, J.J.; Chen, W.-T.; Pearl, M.L. Clinical Significance of Circulating Tumor Cells Detected by an Invasion Assay in Peripheral Blood of Patients with Ovarian Cancer. Gynecol. Oncol. 2009, 112, 185–191. [Google Scholar] [CrossRef]
- Andrusiewicz, M.; Szczerba, A.; Wołuń-Cholewa, M.; Warchoł, W.; Nowak-Markwitz, E.; Gąsiorowska, E.; Adamska, K.; Jankowska, A. CGB and GNRH1 expression analysis as a method of tumor cells metastatic spread detection in patients with gynecological malignances. J. Transl. Med. 2011, 9, 130. [Google Scholar] [CrossRef]
- Wang, T.; Gao, Y.; Wang, X.; Tian, J.; Li, Y.; Yu, B.; Huang, L.H.; Liang, H.; Irwin, D.M.; Ta, H.; et al. Establishment of an optimized CTC detection model consisting of EpCAM, MUC1 and WT1 in epithelial ovarian cancer and its correlation with clinical characteristics. Chin. J. Cancer Res. 2022, 34, 95–108. [Google Scholar] [CrossRef]
| Author and Year of the Study | Patients Number | Blood Amount | CTCs Detection Method | CTCs Clinical Significance |
|---|---|---|---|---|
| Marth C. et al., 2002 [50] | 90 | 40 mL | Microbeads coated with MOC-31 antibody. | CTCs were detected in 12% of patients. |
| Enrichment with magnetic beads coupled with EGP-2 antibody. | CTCs rate varied between 10 and 150 tumor cells per 106 MNC. | |||
| Fan et al., 2009 [69] | 66 | 5–20 mL | Ficoll density gradient centrifugation followed by cell invasion assay that enriches and identifies tumor cells with a cell adhesion matrix (Vita-Assay™). | CTCs were detected in 60.6% of patients. |
| -10% in the early stage; | ||||
| -73.1% in the late stage. | ||||
| CTCs significantly correlated with decreased disease-free survival. | ||||
| Aktas B. et al., 2011 [43] | 122 | 10 mL | Immunomagnetically enriched tumor cells with antibody mixture (anti-GA 73.3 and anti-MUC1 antibodies). | Before surgery, CTCs were detected in 19% of patients. |
| Analysis of tumor-associated mRNA performed by multiplex PCR for: HER2, MUC1, and GA 733-2. | After chemotherapy CTCs were detected in 27% of patients. | |||
| CTCs positivity significantly correlated with shorter overall survival before surgery and after chemotherapy. | ||||
| Poveda A. et al., 2011 [16] | 216 | 10 mL | CellSearch system. | CTCs were detected in 51.4% of patients. |
| CTCs identified as EpCAM+, cytokeratin+, CD45−, and positive for the nuclear stain. | Prior to the start of therapy, ≥ 2 CTCs were identified in 14.4% of patients. | |||
| Patients with ≥2 CTCs prior to therapy had a significantly higher risk for progression and death. | ||||
| Patients with elevated baseline CTCs had a significantly higher risk of progression and death, respectively. | ||||
| Pearl et al., 2014 [17] | 129 | 2–20 mL | Cell adhesion matrix (CAM)-based functional cell enrichment and identification platform. | 1.2% sensitivity, 95.1% specificity, and 77.8% positive predictive value (PPV) of iCTCs in detecting patients with stage I and II EOC malignancy. |
| iCTCs identified as epithelial (Epi+)-positive and hematopoietic lineage (HL-)-negative when analyzed by flow cytometry and fluorescent microscopy imaging. | 83% sensitivity and 97.3% PPV of iCTCs in detecting all stages of EOC malignancy. | |||
| Pearl M. et al., 2015 [9] | 123 | 2–20 mL | Cell adhesion matrix (CAM)-based platform to isolate invasive CTCs (iCTCs). | iCTCs were detected in 85.3% of patients. |
| -Positive predictive value (PPV) of iCTCs was 90%, | ||||
| iCTCs identified as epithelial (Epi+)-positive and hematopoietic lineage (HL-)-negative when analyzed by flow cytometry and fluorescent microscopy imaging, | -Negative predictive values (NPV) of iCTCs was 80.6%. | |||
| Increases in iCTCs (79.5%) were more sensitive than increases in CA125 (67.6%) to predict progressive disease or relapse. | ||||
| Kolostova K. et al., 2015 [22] | 118 | 8 mL | MetaCell: size-based enrichment based on filtration. | CTCs were detected in 65.2% of patients. |
| CTCs identified as cells with: (i) nuclear size ≥10 μm), (ii) irregular nuclear contour, (iii) visible cytoplasm, (iv) prominent nucleoli, (v) high nuclear-cytoplasmic ratio, (vi) proliferating, (vii) growing in 3D layers. | CTCs correlated with the presence of ascites, peritoneal carcinomatosis, and residual disease. | |||
| Blassl et al., 2016 [24] | 10 | 5 mL | AdnaTest OvarianCancerSelect. | CTCs presence correlated with decreased overall survival. |
| AdnaTest EMT-1/StemCellDetect. | CTCs with epithelial–mesenchymal-transition (EMT) or stem-like traits were pointed to be involved in metastatic progression and recurrence. | |||
| Chebouti et al., 2017 [18] | 91 | 5 mL | AdnaTest OvarianCancer Detect. | Detection rate for epithelial CTCs was 18%. |
| AdnaTest EMT-1 Detect. | Detection rate for EMT-like CTCs was 30%. | |||
| Analysis of EpCAM, Muc-1, and CA125 and the EMT-associated transcripts: PI3Kα, Akt-2, and Twist. | PI3K+ EMT-like CTCs, in combination with epithelial CTCs, indicated decreased OS for FIGO I-III patients with residual tumor burden after surgery. | |||
| Epithelial CTCs alone significantly correlated with decreased PFS and OS. | ||||
| Chebouti et al., 2017 [23] | 65 | 10 mL | AdnaTest Ovarian Cancer. | ERCC1+CTCs were detected in 15% of patients at primary diagnosis and in 12% after chemotherapy. |
| ERCC1+CTCs after chemotherapy correlated with platinum resistance and reduced PFS and OS. | ||||
| Tumor-associated transcripts: EpCAM, MUC-1, and CA-125. ERCC1 was investigated by RT-PCR. | ERCC1+CTCs persistence indicated poor post-therapeutic outcome. | |||
| Lee M. et al., 2017 [15] | 54 | 10 mL | Biotin-doped. | CTCs were detected in 98.1% of cases. |
| Ppy-deposited microfluidic system with streptavidin. | Newly diagnosed patients’ median counts of single CTCs and CTC clusters were 4 and 1, respectively. | |||
| Antibodies mixture directed against: EpCAM, TROP-2, EGFR, vimentin, and N-cadherin. | In primary and recurrent disease, median counts of CTCs clusters were 1 and 1, respectively. | |||
| CTCs identified as EpCAM-positive and DAPI-positive, and CD45-negative cells. | In newly diagnosed patients with CTCs counts ≥ 3, PFS was significantly shorter. CTCs clusters positivity correlated with platinum resistance. | |||
| Lou E. et al., 2018 [11] | 29 | 7.5 mL | Positive selection with magnetic beads conjugated to an anti-EpCAM antibody. | CTCs were detected in 17.2% of patients. |
| CTC enumeration with DAPI, anti-CD45, and an anti-cytokeratin cocktail ( CK8, CK18, and CK19). | CTCs correlated with higher stage (FIGO stage III or IV) of tumor. | |||
| CTCs identified as EpCAM-positive, CK-positive, DAPI-positive, and CD45-negative by the morphology of a single intact carcinoma cell (no cell clusters identified). | ||||
| Zhang X. et al., 2018 [47] | 109 | 5 mL | Magnetic separation with beads coated with EpCAM, HER2, and MUC1 antibodies. | CTCs were detected in blood of 90% of newly diagnosed patients: |
| - Average CTCs number: 264 (range 0–1929); | ||||
| RT-PCR analysis of EpCAM, HER2, and MUC1 expression. | - CTCs detected in 82%, 85%, 91%, and 100% of cases at stages I, II, III, and IV, respectively. | |||
| CTCs were detected in 91% of patients after the treatment: | ||||
| - Average CTCs number: 314 (range 0–1822). | ||||
| Expression of EpCAM and HER in CTCs was correlated with resistance to chemotherapy. | ||||
| Expression of EpCAM in CTCs before the treatment was correlated with overall survival. | ||||
| Kim M. et al., 2019 [42] | 30 | 5 mL | Tapered-slit filter (TSF) platform. | Postoperative CTCs were more frequently detected in women with lymph node involvement: 100% vs. 30.0%. |
| CTCs defined as (DAPI)-positive, (CD45)-negative, CK 9-positive, and EpCAM-positive, and using morphological criteria: higher nucleus-to-cytoplasm ratio, larger size, and higher degree of irregularity than observed in the background blood cell. | ||||
| Banys-Paluchowski M. et al., 2020 [12] | 43 | 7.5 mL | CellSearch™ system (magnetic separation with beads coated with EpCAM). | Positive rate of CTCs: 27%. |
| CTCs status before the start of systemic therapy correlated with clinical outcome. | ||||
| CTCs stained with several antibodies. | ||||
| Zuo Li et al., 2021 [48] | 30 | 7 mL | Magnetic separation with beads coated with EpCAM. | Expression level of miR181 in CTCs was related to: |
| - The stage of OC (in stages III and IV significantly higher than in stages I and II); | ||||
| miR181a expression determined by RT-PCR. | - The presence of lymphatic metastasis. | |||
| Obermayr E. et al., 2021 [7] | 105 | 25 mL | Gradient centrifugation. | CTCs were detected in 24.5% of patients before the treatment. CTCs were detected in 20.4% of the patients after adjuvant treatment (follow-up patients). |
| PPIC expression determined by IF and RT-PCR. | CTCs in follow-up patients were correlated with: | |||
| - Age; | ||||
| - Resistance to platinum-based chemotherapy; | ||||
| - FIGO stage at borderline significance. | ||||
| Patients with PPIC-positive CTCs were characterized by significantly shorter disease-free survival than PPIC-negative patients (median PFS 11 vs. 21 months) and shorter overall survival. | ||||
| Presence of CTCs in patients after chemotherapy was associated with: | ||||
| - Increased mortality; | ||||
| - Higher risk of recurrence; | ||||
| - Increased mortality after 5 survived years. | ||||
| Yang J. et al., 2021 [49] | 181 | 5 mL | Nanofiltration technology. | CTC counts: 8.70 ± 5.69 |
| - M-CTC/total CTCs percentage: 0.24 ± 0.19; | ||||
| Epithelial E-CTCs (EpCAM, CK8/18/19), mesenchymal M-CTCs (vimentin, Twist), and epithelial/mesenchymal hybrid CTCs identified by RNA-In Situ hybridization (RNA-ISH) method. | - E-CTC/total CTCs percentage: 0.57 ± 0.25; | |||
| - Hybrids/total CTCs percentage: 0.19 ± 0.11. | ||||
| Increase in recurrence rate: | ||||
| - CTCs ≥ 5–1.98-fold increase; | ||||
| - CTCs < 5–1.24-fold increase; | ||||
| - M-CTC < 0.1–1.43-fold increase. | ||||
| Cheng H. et al., 2021 [20] | 20 | 5 mL | Negative selection of leukocytes with immunomagnetic beads (anti-CD45). | CTCs were detected in 95.0% of patients. |
| The cell size, quantified immunostaining intensity of CA125 and HE4, and ploidy of Chr8. | Total number of CTCs: 8.5 cells. | |||
| Ma et al., 2021 [44] | 156 | 5 mL | Can PatrolTM technique followed by RNA-ISH with probes for mesenchymal molecules (Vimentin and Twist) and epithelial cell adhesion molecules (CK8/18/19 and EpCAM). | CTC counts and M-CTC percentage provided significantly great prediction values for clinical stages, platinum resistance, and survival. |
| Wang et al., 2022 [71] | 160 | 5 ml | Immunomagnetic beads targeting epithelial cell surface antigens (EpCAM and MUC1) and RT-PCR (detecting EpCAM, MUC1, and WT1). | Specificity of the CTCs detection was significantly higher than CA125 (92.2% vs. 82.2%). |
| Detection rate of CTCs was higher than the positive rate of CA125 (74.5% vs. 58.2%) in early-stage patients. | ||||
| CTCs detection rate was significantly higher in patients with ascitic volume ≥500 mL. | ||||
| The detection rate of CTCs EpCAM+ and CTCs MUC1+ was significantly higher in chemo-resistant patients (26.3% vs. 11.9%; 26.4% vs. 13.4%). | ||||
| The median progression-free survival time for CTCs MUC1+ patients trended to be longer than CTCs MUC1− patients and overall survival was shorter in CTCs MUC1+ patients. |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Szczerba, A.; Śliwa, A.; Pieta, P.P.; Jankowska, A. The Role of Circulating Tumor Cells in Ovarian Cancer Dissemination. Cancers 2022, 14, 6030. https://doi.org/10.3390/cancers14246030
Szczerba A, Śliwa A, Pieta PP, Jankowska A. The Role of Circulating Tumor Cells in Ovarian Cancer Dissemination. Cancers. 2022; 14(24):6030. https://doi.org/10.3390/cancers14246030
Chicago/Turabian StyleSzczerba, Anna, Aleksandra Śliwa, Pawel P. Pieta, and Anna Jankowska. 2022. "The Role of Circulating Tumor Cells in Ovarian Cancer Dissemination" Cancers 14, no. 24: 6030. https://doi.org/10.3390/cancers14246030
APA StyleSzczerba, A., Śliwa, A., Pieta, P. P., & Jankowska, A. (2022). The Role of Circulating Tumor Cells in Ovarian Cancer Dissemination. Cancers, 14(24), 6030. https://doi.org/10.3390/cancers14246030





