Safety and Efficacy of Transoral Robotic Thyroidectomy for Thyroid Tumor: A Systematic Review and Meta-Analysis
Abstract
:Simple Summary
Abstract
1. Introduction
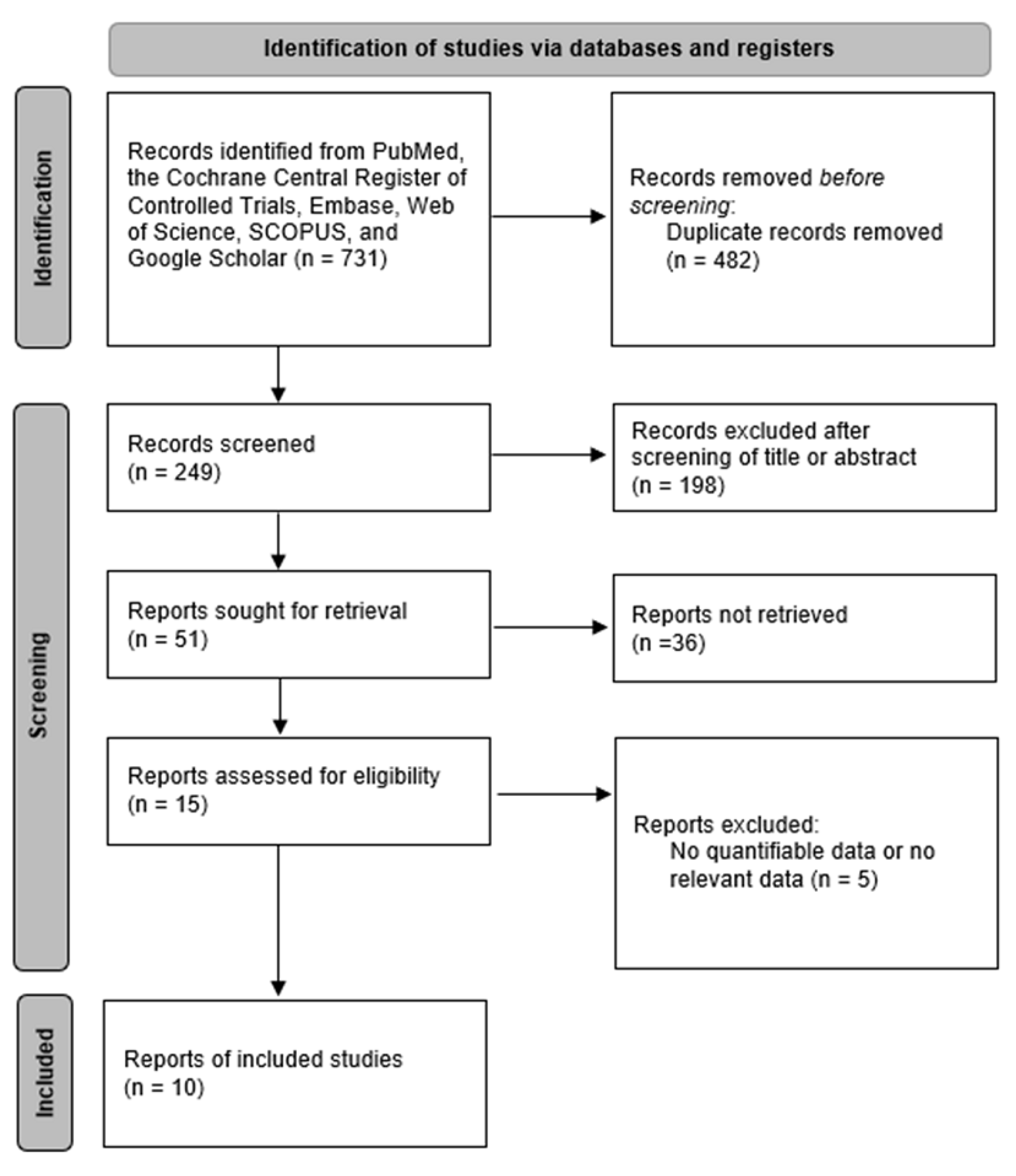
2. Materials and Methods
2.1. Search Strategy
2.2. Data Extraction and Risk of Bias Assessment
2.3. Statistical Analysis
3. Results
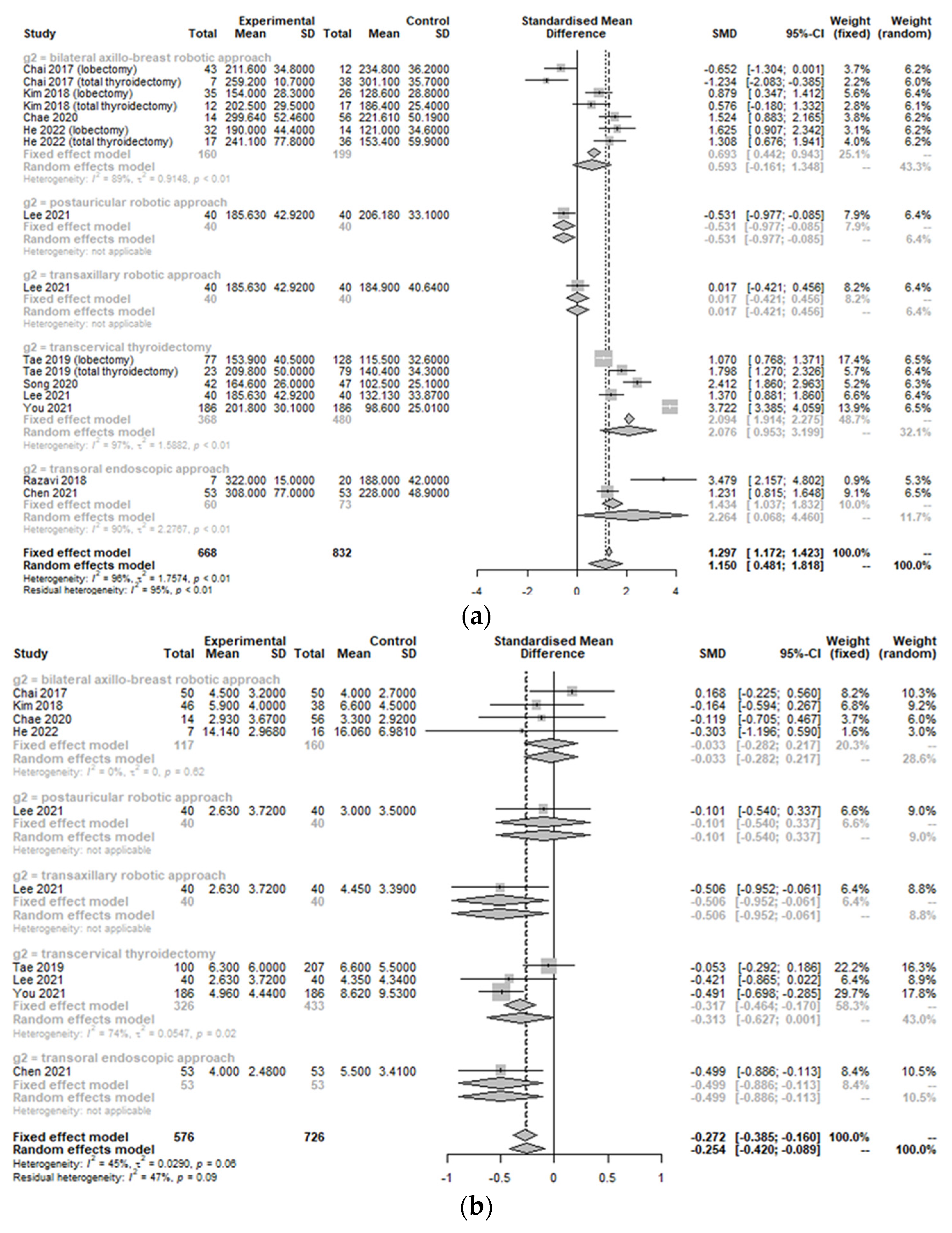
3.1. Operation-Related Outcomes between Treatment Group and Control Group
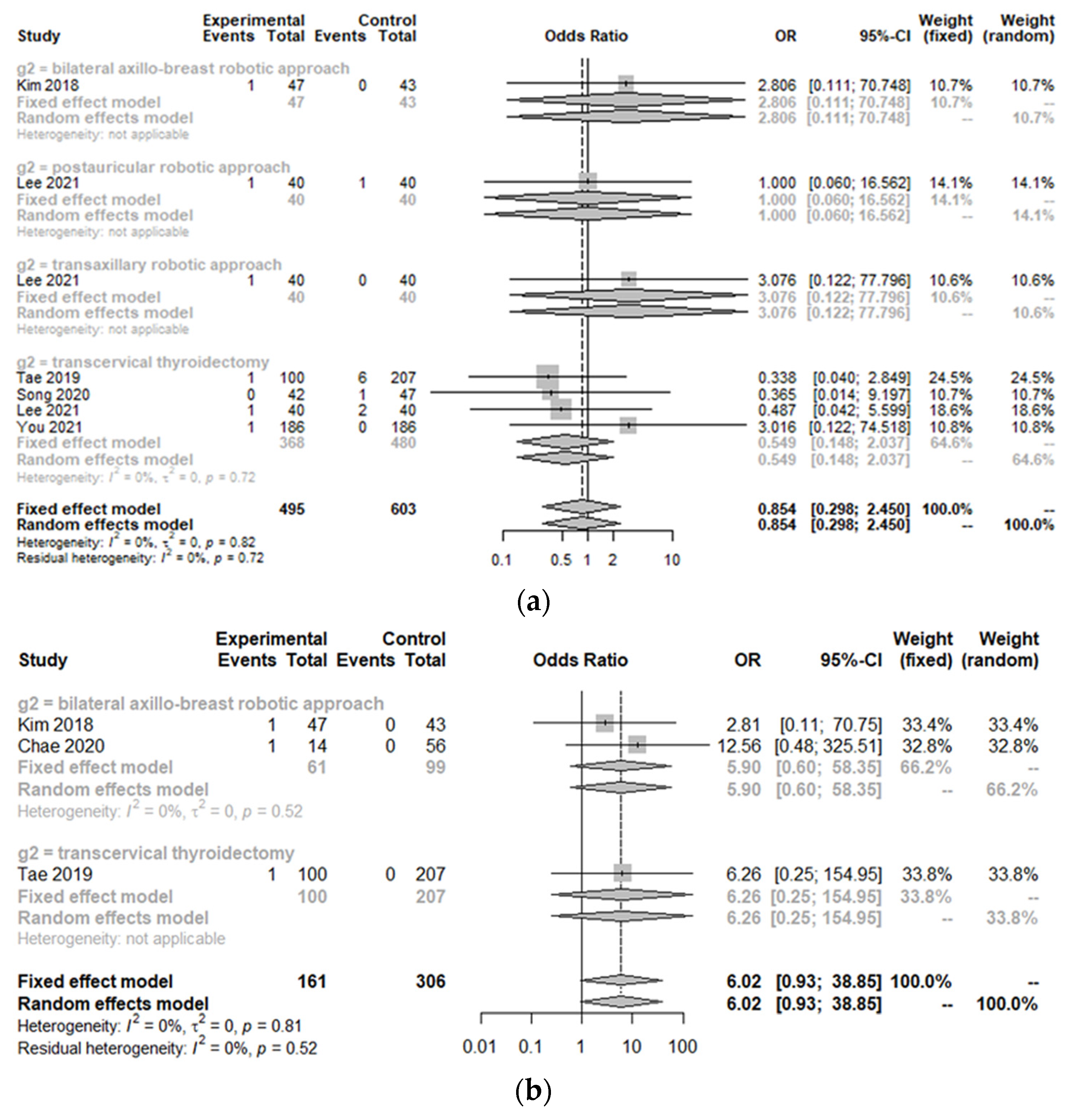
3.2. Peri-Operative Complications between the Treatment Group and Control Group
3.3. Postoperative Outcomes between the Treatment Group and Control Group
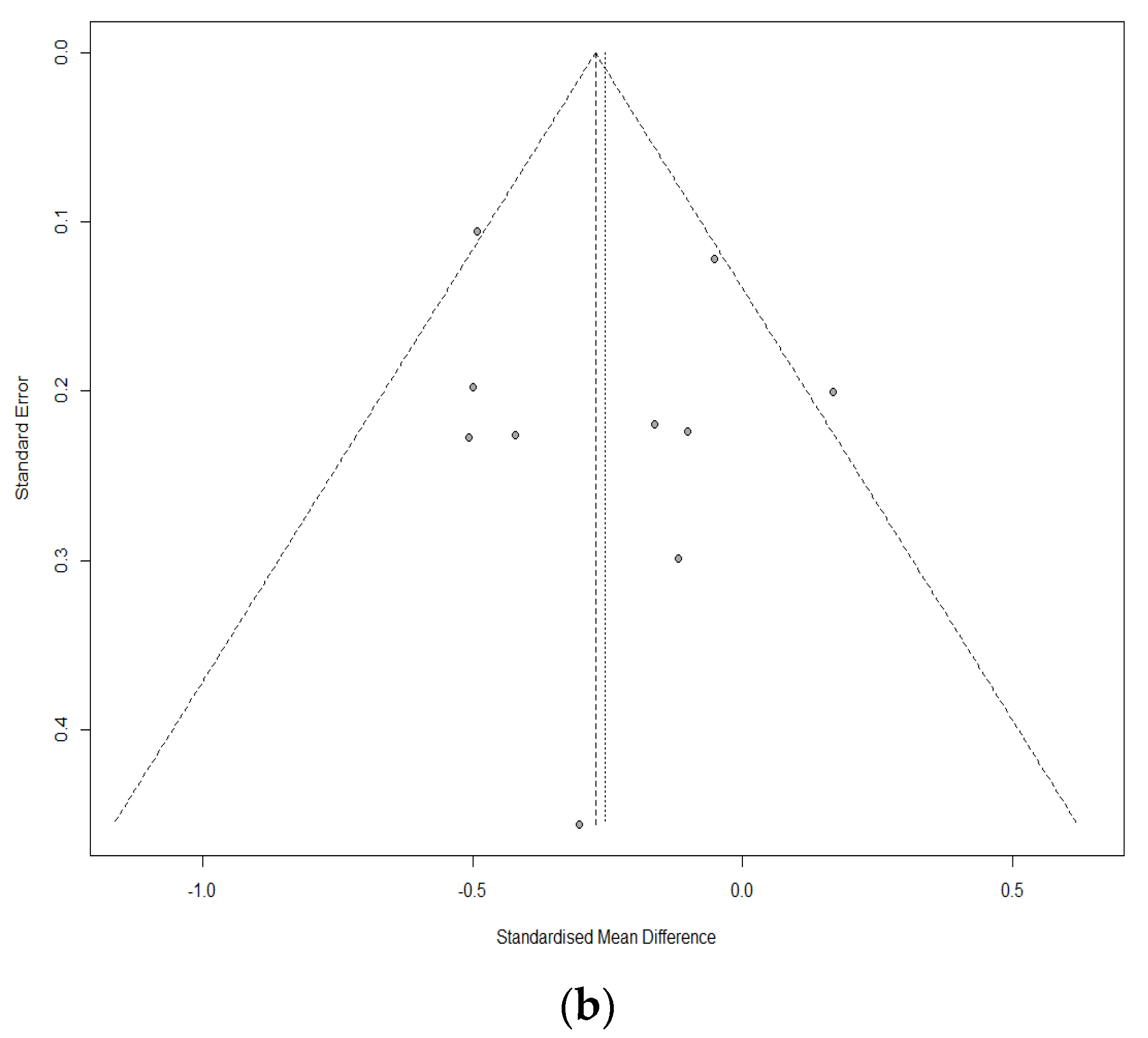
3.4. Sensitivity Analyses
4. Discussion
5. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Haugen, B.R.; Alexander, E.K.; Bible, K.C.; Doherty, G.M.; Mandel, S.J.; Nikiforov, Y.E.; Pacini, F.; Randolph, G.W.; Sawka, A.M.; Schlumberger, M.; et al. 2015 American Thyroid Association Management Guidelines for Adult Patients with Thyroid Nodules and Differentiated Thyroid Cancer: The American Thyroid Association Guidelines Task Force on Thyroid Nodules and Differentiated Thyroid Cancer. Thyroid 2016, 26, 1–133. [Google Scholar] [CrossRef] [PubMed]
- De Vries, L.H.; Aykan, D.; Lodewijk, L.; Damen, J.A.A.; Borel Rinkes, I.H.M.; Vriens, M.R. Outcomes of Minimally Invasive Thyroid Surgery—A Systematic Review and Meta-Analysis. Front. Endocrinol. 2021, 12, 719397. [Google Scholar] [CrossRef] [PubMed]
- Vaccarella, S.; Franceschi, S.; Bray, F.; Wild, C.P.; Plummer, M.; Dal Maso, L. Worldwide Thyroid-Cancer Epidemic? The Increasing Impact of Overdiagnosis. N. Engl. J. Med. 2016, 375, 614–617. [Google Scholar] [CrossRef] [PubMed]
- Jantharapattana, K.; Maethasith, J. Transaxillary gasless endoscopic thyroidectomy versus conventional open thyroidectomy: A randomized study. Eur. Arch. Otorhinolaryngol. 2017, 274, 495–500. [Google Scholar] [CrossRef] [PubMed]
- Tae, K.; Ji, Y.B.; Song, C.M.; Ryu, J. Robotic and Endoscopic Thyroid Surgery: Evolution and Advances. Clin. Exp. Otorhinolaryngol. 2019, 12, 1–11. [Google Scholar] [CrossRef]
- Choi, Y.; Lee, J.H.; Kim, Y.H.; Lee, Y.S.; Chang, H.S.; Park, C.S.; Roh, M.R. Impact of postthyroidectomy scar on the quality of life of thyroid cancer patients. Ann. Dermatol. 2014, 26, 693–699. [Google Scholar] [CrossRef]
- Juarez, M.C.; Ishii, L.; Nellis, J.C.; Bater, K.; Huynh, P.P.; Fung, N.; Darrach, H.; Russell, J.O.; Ishii, M. Objectively measuring social attention of thyroid neck scars and transoral surgery using eye tracking. Laryngoscope 2019, 129, 2789–2794. [Google Scholar] [CrossRef]
- Hüscher, C.S.; Chiodini, S.; Napolitano, C.; Recher, A. Endoscopic right thyroid lobectomy. Surg. Endosc. 1997, 11, 877. [Google Scholar] [CrossRef]
- Miccoli, P.; Berti, P.; Conte, M.; Bendinelli, C.; Marcocci, C. Minimally invasive surgery for thyroid small nodules: Preliminary report. J. Endocrinol. Investig. 1999, 22, 849–851. [Google Scholar] [CrossRef]
- Ikeda, Y.; Takami, H.; Sasaki, Y.; Kan, S.; Niimi, M. Endoscopic neck surgery by the axillary approach. J. Am. Coll. Surg. 2000, 191, 336–340. [Google Scholar] [CrossRef]
- Lee, K.E.; Rao, J.; Youn, Y.K. Endoscopic thyroidectomy with the da Vinci robot system using the bilateral axillary breast approach (BABA) technique: Our initial experience. Surg. Laparosc. Endosc. Percutan. Tech. 2009, 19, e71–e75. [Google Scholar] [CrossRef] [PubMed]
- Kim, W.W.; Kim, J.S.; Hur, S.M.; Kim, S.H.; Lee, S.K.; Choi, J.H.; Kim, S.; Lee, J.E.; Kim, J.H.; Nam, S.J.; et al. Is robotic surgery superior to endoscopic and open surgeries in thyroid cancer? World J. Surg. 2011, 35, 779–784. [Google Scholar] [CrossRef] [PubMed]
- Luna-Ortiz, K.; Gómez-Pedraza, A.; Anuwong, A. Lessons Learned from the Transoral Endoscopic Thyroidectomy with Vestibular Approach (TOETVA) for the Treatment of Thyroid Carcinoma. Ann. Surg. Oncol. 2020, 27, 1356–1360. [Google Scholar] [CrossRef] [PubMed]
- Anuwong, A. Transoral Endoscopic Thyroidectomy Vestibular Approach: A Series of the First 60 Human Cases. World J. Surg. 2016, 40, 491–497. [Google Scholar] [CrossRef]
- Dionigi, G.; Bacuzzi, A.; Lavazza, M.; Inversini, D.; Pappalardo, V.; Boni, L.; Rausei, S.; Barczynski, M.; Tufano, R.P.; Kim, H.Y.; et al. Transoral endoscopic thyroidectomy via vestibular approach: Operative steps and video. Gland Surg. 2016, 5, 625–627. [Google Scholar] [CrossRef]
- Chang, E.H.E.; Kim, H.Y.; Koh, Y.W.; Chung, W.Y. Overview of robotic thyroidectomy. Gland Surg. 2017, 6, 218–228. [Google Scholar] [CrossRef] [PubMed]
- Chai, Y.J.; Kim, H.Y.; Kim, H.K.; Jun, S.H.; Dionigi, G.; Anuwong, A.; Richmon, J.D.; Tufano, R.P. Comparative analysis of 2 robotic thyroidectomy procedures: Transoral versus bilateral axillo-breast approach. Head Neck 2018, 40, 886–892. [Google Scholar] [CrossRef]
- Kim, W.W.; Lee, J.; Jung, J.H.; Park, H.Y.; Tufano, R.P.; Kim, H.Y. A comparison study of the transoral and bilateral axillo-breast approaches in robotic thyroidectomy. J. Surg. Oncol. 2018, 118, 381–387. [Google Scholar] [CrossRef]
- You, J.Y.; Kim, H.Y.; Chai, Y.J.; Kim, H.K.; Anuwong, A.; Tufano, R.P.; Dionigi, G. Transoral Robotic Thyroidectomy Versus Conventional Open Thyroidectomy: Comparative Analysis of Surgical Outcomes in Thyroid Malignancies. J. Laparoendosc. Adv. Surg. Tech. A 2019, 29, 796–800. [Google Scholar] [CrossRef]
- Sun, H.; Dionigi, G. Applicability of transoral robotic thyroidectomy: Is it the final solution? J. Surg. Oncol. 2019, 119, 541–542. [Google Scholar] [CrossRef]
- Chen, Y.H.; Kim, H.Y.; Anuwong, A.; Huang, T.S.; Duh, Q.Y. Transoral robotic thyroidectomy versus transoral endoscopic thyroidectomy: A propensity-score-matched analysis of surgical outcomes. Surg. Endosc. 2021, 35, 6179–6189. [Google Scholar] [CrossRef] [PubMed]
- Kim, H.K.; Kim, H.Y.; Chai, Y.J.; Dionigi, G.; Berber, E.; Tufano, R.P. Transoral Robotic Thyroidectomy: Comparison of Surgical Outcomes Between the da Vinci Xi and Si. Surg. Laparosc. Endosc. Percutan. Tech. 2018, 28, 404–409. [Google Scholar] [CrossRef]
- Paek, S.H.; Kang, K.H. Robotic thyroidectomy and cervical neck dissection for thyroid cancer. Gland Surg. 2016, 5, 342–351. [Google Scholar] [CrossRef] [PubMed]
- Razavi, C.R.; Khadem, M.G.A.; Fondong, A.; Clark, J.H.; Richmon, J.D.; Tufano, R.P.; Russell, J.O. Early outcomes in transoral vestibular thyroidectomy: Robotic versus endoscopic techniques. Head Neck 2018, 40, 2246–2253. [Google Scholar] [CrossRef] [PubMed]
- Tae, K.; Ji, Y.B.; Song, C.M.; Park, J.S.; Park, J.H.; Kim, D.S. Safety and efficacy of transoral robotic and endoscopic thyroidectomy: The first 100 cases. Head Neck 2020, 42, 321–329. [Google Scholar] [CrossRef]
- Chae, S.; Min, S.Y.; Park, W.S. Comparison Study of Robotic Thyroidectomies Through a Bilateral Axillo-Breast Approach and a Transoral Approach. J. Laparoendosc. Adv. Surg. Tech. A 2020, 30, 175–182. [Google Scholar] [CrossRef]
- Song, C.M.; Park, J.S.; Park, H.J.; Tae, K. Voice outcomes of transoral robotic thyroidectomy: Comparison with conventional trans-cervical thyroidectomy. Oral Oncol. 2020, 107, 104748. [Google Scholar] [CrossRef]
- Lee, D.W.; Bang, H.S.; Jeong, J.H.; Kwak, S.G.; Choi, Y.Y.; Tae, K. Cosmetic outcomes after transoral robotic thyroidectomy: Comparison with transaxillary, postauricular, and conventional approaches. Oral Oncol. 2021, 114, 105139. [Google Scholar] [CrossRef]
- You, J.Y.; Kim, H.Y.; Park, D.W.; Yang, H.W.; Kim, H.K.; Dionigi, G.; Tufano, R.P. Transoral robotic thyroidectomy versus conventional open thyroidectomy: Comparative analysis of surgical outcomes using propensity score matching. Surg. Endosc. 2021, 35, 124–129. [Google Scholar] [CrossRef]
- He, Q.; Zhu, J.; Li, X.; Wang, M.; Wang, G.; Zhou, P.; Wang, D.; Liu, C.; Zheng, L.; Zhuang, D.; et al. A comparative study of two robotic thyroidectomy procedures: Transoral vestibular versus bilateral axillary-breast approach. BMC Surg. 2022, 22, 173. [Google Scholar] [CrossRef]
- Kim, D.H.; Kim, S.W.; Basurrah, M.A.; Hwang, S.H. Clinical and laboratory features for various criteria of eosinophilic chronic rhinosinusitis: A systematic review and meta-analysis. Clin. Exp. Otorhinolaryngol. 2022, 15, 230–246. [Google Scholar] [CrossRef] [PubMed]
- Higgins, J.P.; Altman, D.G.; Gøtzsche, P.C.; Jüni, P.; Moher, D.; Oxman, A.D.; Savovic, J.; Schulz, K.F.; Weeks, L.; Sterne, J.A. The Cochrane Collaboration’s tool for assessing risk of bias in randomised trials. BMJ 2011, 343, d5928. [Google Scholar] [CrossRef] [PubMed]
- Wang, Y.C.; Liu, K.; Xiong, J.J.; Zhu, J.Q. Robotic thyroidectomy versus conventional open thyroidectomy for differentiated thyroid cancer: Meta-analysis. J. Laryngol. Otol. 2015, 129, 558–567. [Google Scholar] [CrossRef] [PubMed]
- Pan, J.H.; Zhou, H.; Zhao, X.X.; Ding, H.; Wei, L.; Qin, L.; Pan, Y.L. Robotic thyroidectomy versus conventional open thyroidectomy for thyroid cancer: A systematic review and meta-analysis. Surg. Endosc. 2017, 31, 3985–4001. [Google Scholar] [CrossRef]
- Son, S.K.; Kim, J.H.; Bae, J.S.; Lee, S.H. Surgical safety and oncologic effectiveness in robotic versus conventional open thyroidectomy in thyroid cancer: A systematic review and meta-analysis. Ann. Surg. Oncol. 2015, 22, 3022–3032. [Google Scholar] [CrossRef]
- Jackson, N.R.; Yao, L.; Tufano, R.P.; Kandil, E.H. Safety of robotic thyroidectomy approaches: Meta-analysis and systematic review. Head Neck 2014, 36, 137–143. [Google Scholar] [CrossRef]
- Liu, J.; Song, T.; Xu, M. Minimally invasive video-assisted versus conventional open thyroidectomy: A systematic review of available data. Surg. Today 2012, 42, 848–856. [Google Scholar] [CrossRef]
- Sun, G.H.; Peress, L.; Pynnonen, M.A. Systematic review and meta-analysis of robotic vs conventional thyroidectomy approaches for thyroid disease. Otolaryngol. Head Neck Surg. 2014, 150, 520–532. [Google Scholar] [CrossRef]
- Kandil, E.; Hammad, A.Y.; Walvekar, R.R.; Hu, T.; Masoodi, H.; Mohamed, S.E.; Deniwar, A.; Stack, B.C., Jr. Robotic Thyroidectomy Versus Nonrobotic Approaches: A Meta-Analysis Examining Surgical Outcomes. Surg. Innov. 2016, 23, 317–325. [Google Scholar] [CrossRef]
- Jiang, W.J.; Yan, P.J.; Zhao, C.L.; Si, M.B.; Tian, W.; Zhang, Y.J.; Tian, H.W.; Feng, S.W.; Han, C.W.; Yang, J.; et al. Comparison of total endoscopic thyroidectomy with conventional open thyroidectomy for treatment of papillary thyroid cancer: A systematic review and meta-analysis. Surg. Endosc. 2020, 34, 1891–1903. [Google Scholar] [CrossRef]
- Chen, C.; Huang, S.; Huang, A.; Jia, Y.; Wang, J.; Mao, M.; Zhou, J.; Wang, L. Total endoscopic thyroidectomy versus conventional open thyroidectomy in thyroid cancer: A systematic review and meta-analysis. Ther. Clin. Risk Manag. 2018, 14, 2349–2361. [Google Scholar] [CrossRef] [PubMed]
- Lang, B.H.; Wong, C.K.; Tsang, J.S.; Wong, K.P. A systematic review and meta-analysis comparing outcomes between robotic-assisted thyroidectomy and non-robotic endoscopic thyroidectomy. J. Surg. Res. 2014, 191, 389–398. [Google Scholar] [CrossRef] [PubMed]
- Xing, Z.; Qiu, Y.; Abuduwaili, M.; Xia, B.; Fei, Y.; Zhu, J.; Su, A. Surgical outcomes of different approaches in robotic assisted thyroidectomy for thyroid cancer: A systematic review and Bayesian network meta-analysis. Int. J. Surg. 2021, 89, 105941. [Google Scholar] [CrossRef] [PubMed]
- Yu, H.W.; Yi, J.W.; Seong, C.Y.; Kim, J.K.; Bae, I.E.; Kwon, H.; Chai, Y.J.; Kim, S.J.; Choi, J.Y.; Lee, K.E. Development of a surgical training model for bilateral axillo-breast approach robotic thyroidectomy. Surg. Endosc. 2018, 32, 1360–1367. [Google Scholar] [CrossRef]
- Ohgami, M.; Ishii, S.; Arisawa, Y.; Ohmori, T.; Noga, K.; Furukawa, T.; Kitajima, M. Scarless endoscopic thyroidectomy: Breast approach for better cosmesis. Surg. Laparosc. Endosc. Percutan. Tech. 2000, 10, 1–4. [Google Scholar] [CrossRef]
- Kim, B.S.; Kang, K.H.; Kang, H.; Park, S.J. Central neck dissection using a bilateral axillo-breast approach for robotic thyroidectomy: Comparison with conventional open procedure after propensity score matching. Surg. Laparosc. Endosc. Percutan. Tech. 2014, 24, 67–72. [Google Scholar] [CrossRef]
- Tae, K.; Ji, Y.B.; Song, C.M.; Min, H.J.; Lee, S.H.; Kim, D.S. Robotic lateral neck dissection by a gasless unilateral axillobreast approach for differentiated thyroid carcinoma: Our early experience. Surg Laparosc. Endosc. Percutan. Tech. 2014, 24, e128-132. [Google Scholar] [CrossRef]
- Kang, S.W.; Lee, S.C.; Lee, S.H.; Lee, K.Y.; Jeong, J.J.; Lee, Y.S.; Nam, K.H.; Chang, H.S.; Chung, W.Y.; Park, C.S. Robotic thyroid surgery using a gasless, transaxillary approach and the da Vinci S system: The operative outcomes of 338 consecutive patients. Surgery 2009, 146, 1048–1055. [Google Scholar] [CrossRef]
- Sun, Z.; Xu, Y.; Li, D.M.; Wang, Z.N.; Zhu, G.L.; Huang, B.J.; Li, K.; Xu, H.M. Log odds of positive lymph nodes: A novel prognostic indicator superior to the number-based and the ratio-based N category for gastric cancer patients with R0 resection. Cancer 2010, 116, 2571–2580. [Google Scholar] [CrossRef]
- Dimofte, G.; Târcoveanu, E.; Taraşi, M.; Panait, C.; Lozneanu, G.; Nicolescu, S.; Porumb, V.; Grigoraş, O. Mean number of lymph nodes in colonic cancer specimen: Possible quality control index for surgical performance. Chirurgia 2011, 106, 759–764. [Google Scholar]
- Anuwong, A.; Ketwong, K.; Jitpratoom, P.; Sasanakietkul, T.; Duh, Q.Y. Safety and Outcomes of the Transoral Endoscopic Thyroidectomy Vestibular Approach. JAMA Surg. 2018, 153, 21–27. [Google Scholar] [CrossRef] [PubMed]
- Tae, K.; Ji, Y.B.; Jeong, J.H.; Lee, S.H.; Jeong, M.A.; Park, C.W. Robotic thyroidectomy by a gasless unilateral axillo-breast or axillary approach: Our early experiences. Surg. Endosc. 2011, 25, 221–228. [Google Scholar] [CrossRef] [PubMed]


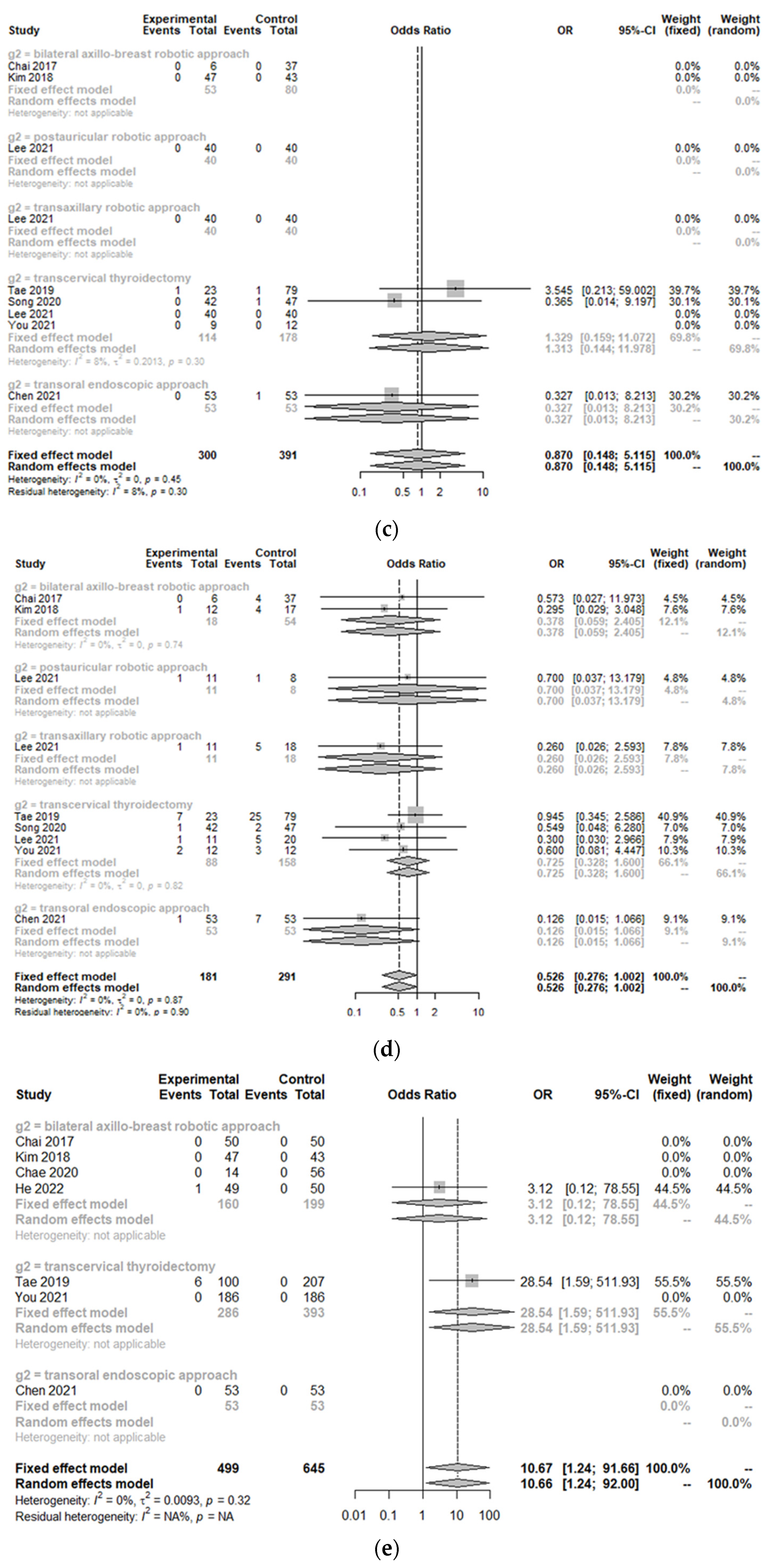
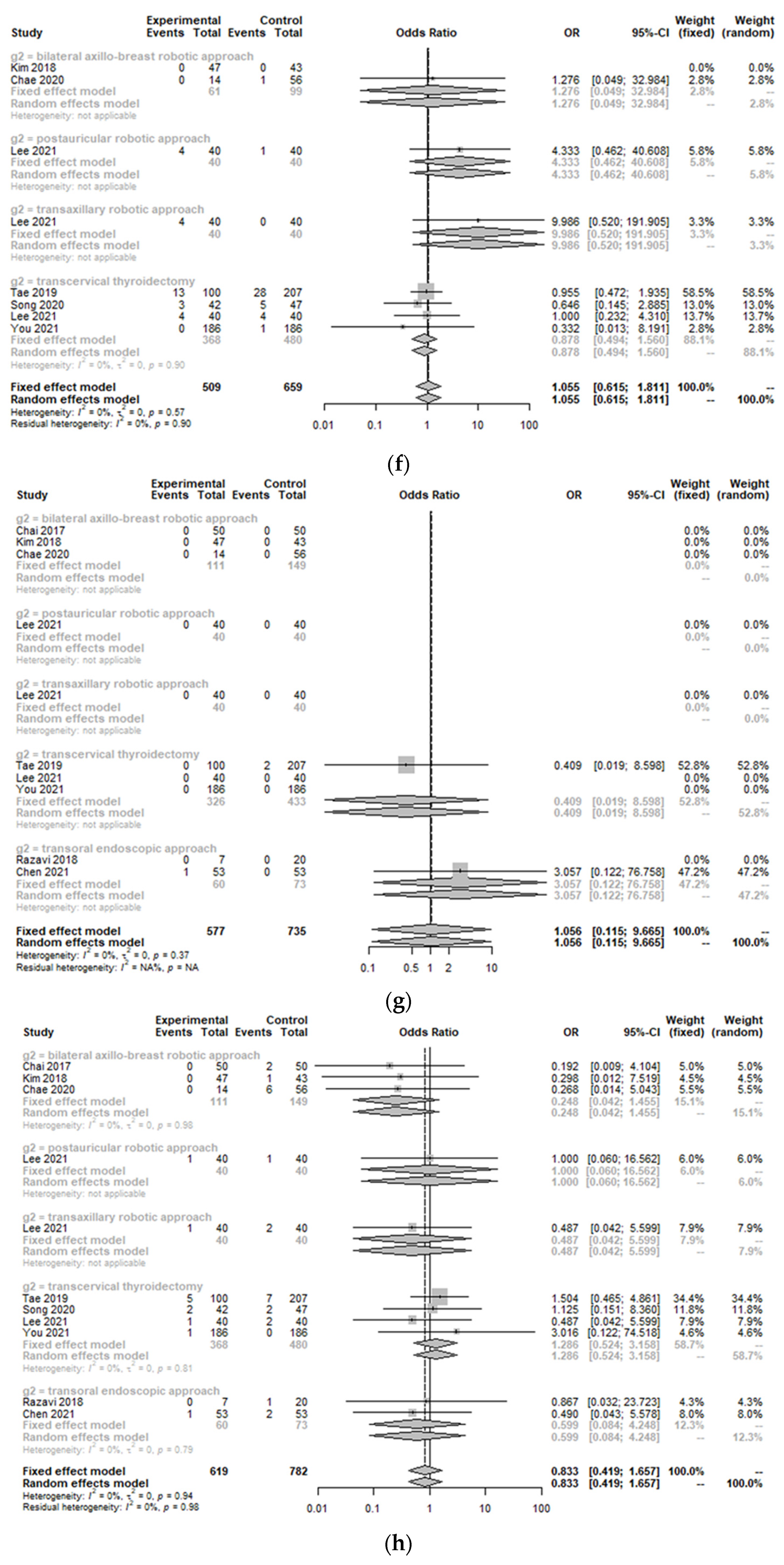
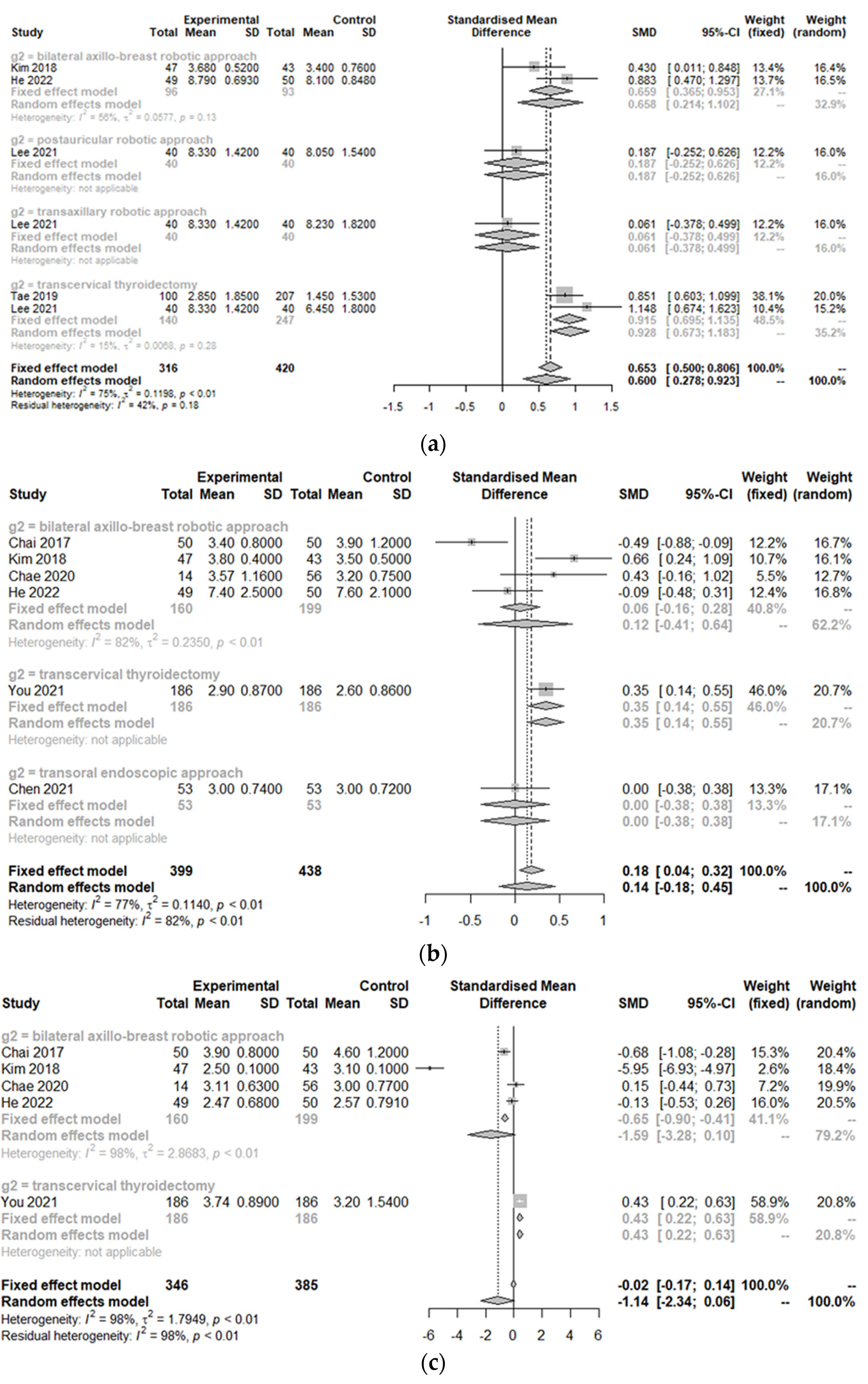
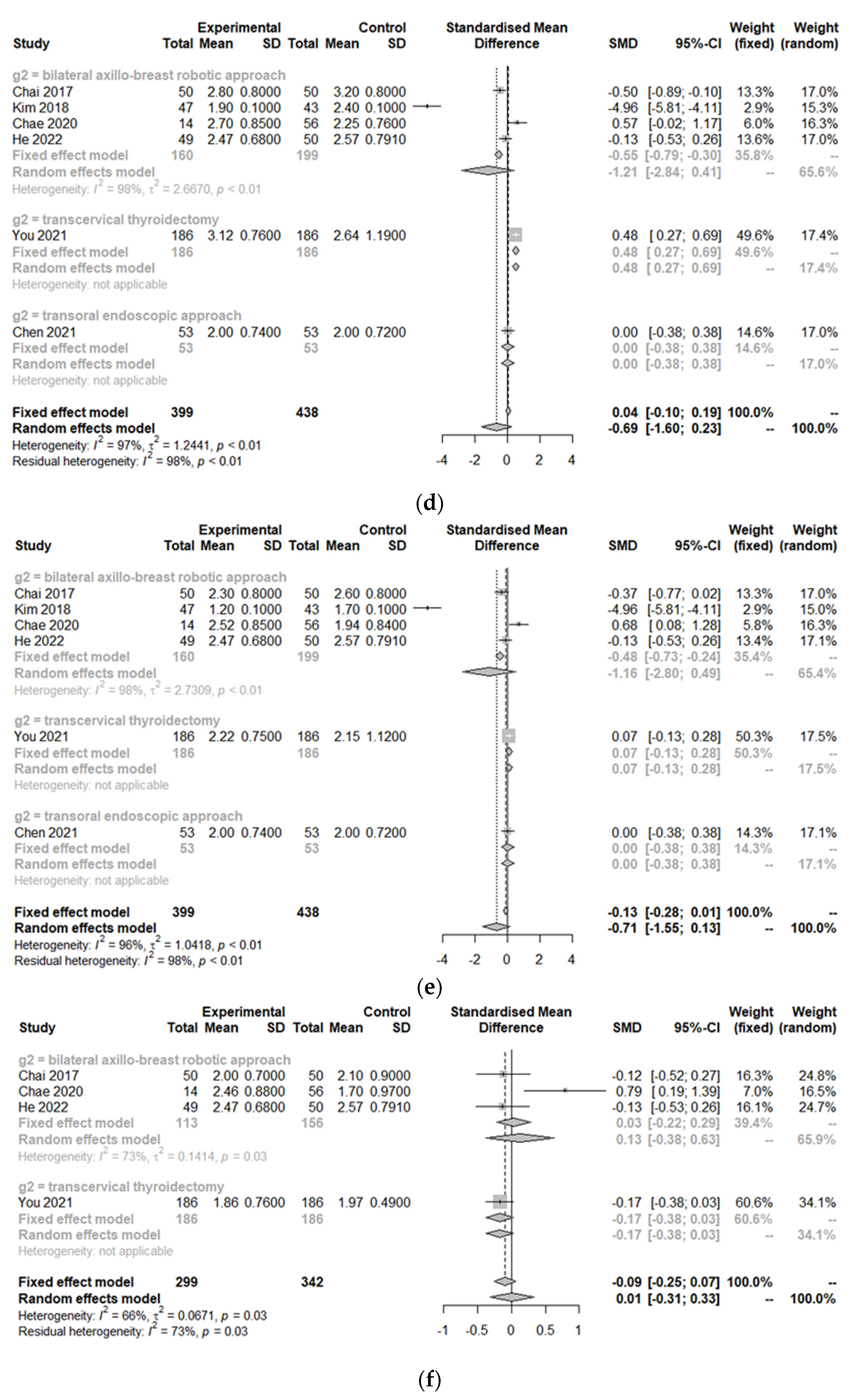
| Study | Design | Total Number of Patients (n) | Age of Patients with Robotic Approach (years, mean ± SD) | Sex (F/M) | Nation | Body Mass Index (kg/m2, mean ± SD) | Tumor Size (cm, mean ± SD) | Comparison | Control Group | Clinicopathology | Outcomes |
|---|---|---|---|---|---|---|---|---|---|---|---|
| Chai 2017 | Retrospective review | 100 | 41.2 ± 9.4 | 93/7 | Korea | 22.8 ± 62.6 | 1.1 ± 0.8 | TORT | Robotic BABA | Benign, PTC | Operative time, postoperative pain (VAS), hospitalization day, number of retrieved LNs, hypoparathyroidism, VCP, infection |
| Kim 2018 | Prospective comparative study | 90 | 39.8 ± 10.7 | 83/7 | Korea | NA | 0.9 ± 0.5 | TORT | Robotic BABA | Benign, PTC, follicular neoplasm | Operative time, cosmetic satisfaction, postoperative pain (VAS) hospitalization day, number of retrieved LNs, metastatic LN, incidental parathyroidectomy, VCP, seroma, infection |
| Razavi 2018 | Retrospective review | 27 | 41.3 ± 15.8 | 23/4 | USA | 28.3 ± 8.1 | NA | TORT | Transoral endoscopic approach | TORT: Benign, NITNP, PTC Control: Benign, NITNP, PTC, Hürthle cell carcinoma | Operative time, VCP |
| Tae 2019 | Retrospective review | 307 | 45.5 ± 18.8 | 82/235 | Korea | 24.6 ± 3.7 | NA | TORT | Conventional OT | TORT: Benign, PTC, follicular, medullary carcinoma Control: Benign, PTC, follicular carcinoma | Operative time, cosmetic satisfaction, number of retrieved LNs, metastatic LN, incidental parathyroidectomy, hypoparathyroidism, VCP, hematoma, seroma, infection, skin flap perforation |
| Chae 2020 | Retrospective review | 70 | 40.88 ± 9.80 | 9/61 | Korea | 23.60 ± 4.31 | 0.75 ± 0.35 | TORT | Robotic BABA | PTC | Operative time, postoperative pain (VAS), hospitalization day, number of retrieved LNs, metastatic LN, incidental parathyroidectomy, VCP, seroma, infection |
| Song 2020 | Prospective comparative study | 89 | 44.0 ± 12.8 | NA | Korea | 24.1 ± 3.6 | 12.6 ± 12.1 | TORT | Conventional OT | Benign, well differentiated carcinoma | Operative time, number of hypoparathyroidisms, VCP, seroma, hematoma |
| Chen 2021 | Retrospective review | 106 | 43.38 ± 12.84 | 17/89 | Taiwan | 23.6 ± 3.56 | 2.19 ± 1.734 | TORT | Transoral endoscopic approach | Benign, indeterminate thyroid nodules, Graves’ disease, malignant or suspicious nodules | Operative time, postoperative pain (VAS), hospitalization day, number of retrieved LNs, metastatic LN, hypoparathyroidism, VCP, seroma, infection |
| Lee 2021 | Retrospective review | 160 | 46.48 ± 11.45 | 33/127 | Korea | 25.64 ± 3.79 | 1.02 ± 0.97 | TORT | Robotic trans-axillary approach, robotic PAA | Follicular neoplasm, benign, differentiated thyroid carcinoma | Operative time, cosmetic satisfaction, postoperative pain (VAS), hospitalization day, number of retrieved LNs, hypoparathyroidism, VCP, seroma, hematoma |
| You 2021 | Retrospective review | 372 | 43.1 ± 10.74 | 68/304 | Korea | 23.5 ± 3.82 | 0.70 ± 0.50 | TORT | Conventional OT | PTC | Operative time, postoperative pain (VAS), hospitalization day, number of retrieved LNs, metastatic LN, incidental parathyroidectomy, VCP, seroma, infection |
| He 2022 | Prospective comparative study | 99 | 44.6 ± 11.8 | 21/78 | China | 25.2 ± 14.2 | 3.50 ± 3.3 | TORT | Robotic BABA | PTC | Operative time, cosmetic satisfaction, postoperative pain (VAS), hospitalization day, number of retrieved LNs, metastatic LN, infection |
| Subgroups | Positive Lymph Node Number (SMD [95% CIs]; I2) | Incidence of Skin Flap Perforation (OR [95% CIs]; I2) | Incidental Parathyroidectomy (OR [95% CIs]; I2) |
|---|---|---|---|
| Overall | −0.0858 [−0.3441; 0.1725]; 66.7% | 6.0165 [0.9318; 38.8464]; 0.0% | 1.3392 [0.3981; 4.5049]; 0.0% |
| Robotic bilateral axillo-breast approach | N = 2 0.3221 [−0.6146; −0.0296]; 0.0% | N = 2 5.8977 [0.5962; 58.3455]; 0.0% | N = 2 1.3392 [0.3981; 4.5049]; 0.0% |
| Robotic postauricular approach | |||
| Conventional open thyroidectomy | N = 2 0.0629 [−0.2508; 0.3766]; 75.0% | N = 1 6.2563 [0.2526; 154.9504]; NA | |
| Transoral endoscopic approach | |||
| p value | 0.0785 | 0.9766 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Kang, Y.J.; Cho, J.-H.; Stybayeva, G.; Hwang, S.H. Safety and Efficacy of Transoral Robotic Thyroidectomy for Thyroid Tumor: A Systematic Review and Meta-Analysis. Cancers 2022, 14, 4230. https://doi.org/10.3390/cancers14174230
Kang YJ, Cho J-H, Stybayeva G, Hwang SH. Safety and Efficacy of Transoral Robotic Thyroidectomy for Thyroid Tumor: A Systematic Review and Meta-Analysis. Cancers. 2022; 14(17):4230. https://doi.org/10.3390/cancers14174230
Chicago/Turabian StyleKang, Yun Jin, Jin-Hee Cho, Gulnaz Stybayeva, and Se Hwan Hwang. 2022. "Safety and Efficacy of Transoral Robotic Thyroidectomy for Thyroid Tumor: A Systematic Review and Meta-Analysis" Cancers 14, no. 17: 4230. https://doi.org/10.3390/cancers14174230
APA StyleKang, Y. J., Cho, J.-H., Stybayeva, G., & Hwang, S. H. (2022). Safety and Efficacy of Transoral Robotic Thyroidectomy for Thyroid Tumor: A Systematic Review and Meta-Analysis. Cancers, 14(17), 4230. https://doi.org/10.3390/cancers14174230





