Physical Activity and Cancer Care—A Review
Abstract
:Simple Summary
Abstract
1. Introduction
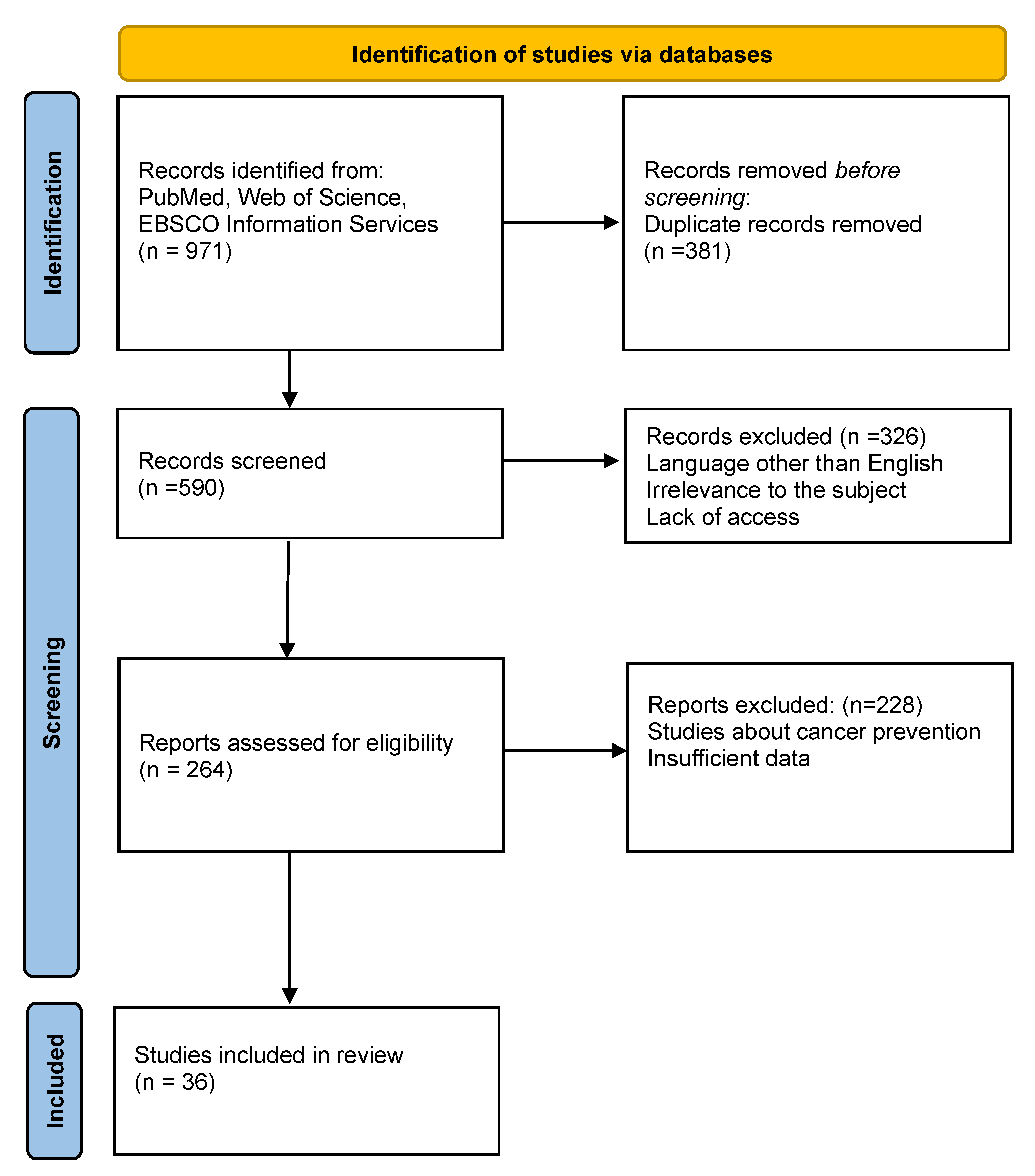
2. Material and Methods
3. Results
3.1. Side Effects of Cancer Treatment
3.2. Fatigue
3.3. Quality of Life
3.4. Mental Health
3.5. Physical Fitness, Muscle Strength, Impact on Body Weight
3.6. Mortality and Longer Survival
3.7. Recurrence
| Author (Ref.) | Type of Cancer, Number of Patients | Intervention Type, Intensity | Frequency, Duration | Main Findings |
|---|---|---|---|---|
| Singh B. et al. [3] | colorectal cancer (n = 670) | supervised and unsupervised aerobic and combined exercise | pre-treatment supervised: 1 session per week, unsupervised: 3–7 times per week for 4 weeks; during chemotherapy and post-treatment: from 1 to 7 sessions per week for 7 days to 6 months. | Superior to UC for: QoL, aerobic fitness, sleep, fatigue, reduced body fat, depression, upper-body strength (p < 0.05) |
| An K-Y. et al. [8] | breast cancer (n = 301) | CARE | 25–30 min of aerobic exercise (n = 96), 50–60 min (n = 101), a combined dose of 50–60 min of aerobic and resistance exercise (n = 104), median of 17 weeks | The “combined” group was superior for: fatigue, upper and lower body endurance, body fat percentage—12-month follow up, CARE after chemotherapy may be optimal for longer-term health outcomes |
| Cannioto R.A. et al. [9] | breast cancer (n = 1340) | DELCaP [38] | from minimum PAGAs (the MET [41] hour equivalent of 150 min to moderate intensity RPA per week) to exceeding the minimum recommended range | 1 year after diagnosis: reduction in recurrence and mortality |
| Parker N.H. et al. [18] | pancreatic cancer (n = 50) | aerobic exercise, full-body strengthening exercises | 60 min/week—moderate intensity aerobic PA; 60 min/week of strengthening exercises, 7–25 weeks | Exercise recommendations for cancer survivors are important, but in order to reduce barriers to participation, further efforts are needed. |
| Dieli- Conwright C.M. et al. [23] | breast cancer (n = 100), overweight and obese survivors | combined aerobic and resistance exercise | 16 weeks, 150 min of aerobic exercise with 2–3 days of resistance exercise training/week | Superior to usual care for QoL, fatigue, depression, muscular strength (p < 0.001). Three-month follow-up: outcomes remained improved. |
| Li W et al. [26] | childhood cancer (n = 222) | adventure-based training program: ice-breaking and teambuilding games, shuttle runs, rock climbing, high- and low-level rope courses and descending | 4 training days: 2 weeks, 2, 4, and 6 months after randomisation | Significantly lower levels of CRF (p < 0.001), higher levels of physical activity (p < 0.001), QoL (p < 0.01) |
| Jones T.L. et al. [27] | ovarian cancer (20 articles with sample sizes from 10 to 7022) | aerobic | 3 to 26-week intervention, from 75 min/week to 225 min/week | Higher health-related QoL, decreased levels of anxiety and depression, improvement in fatigue, physical and psychological health |
| Galvao D. et al. [31] | prostate cancer with bone metastases (n = 103) | resistance, aerobic, flexibility exercise | 3 times per week, 60 min session, for 3 months | After 3 months—improved self-reported physical function, muscle strength. No changes for total body fat mass, fatigue (p = 0.964). |
| Cataldi et al. [32] | breast cancer (n = 3) | each session: cardiorespiratory, resistance, flexibility, postural education exercises | 8-week programme, 60 min of exercise, 2 days per week | Measures of fatigue have decreased (p < 0.001), exercises improved physical fitness, functional capacity, capability to manage emotional life (p = 0.003), helped with dealing with the physiological and psychological side effects. |
| Di Maso et al. [35] | prostate cancer (n = 777) | occupational and recreational physical activity and Mediterranean diet | 15-year follow-up | Intervention reduces mortality in PCa patients (due to lowering serum insulin levels, IGF and inflammation). |
| Wang et al. [39] | non-metastatic prostate cancer (n = 10,864) | recreational physical activity, e.g., walking, bicycling, aerobics, dancing, jogging, tennis | MET-h/week < 3.5, 3.5–8.75, 8.75, ≤17.5, >17.5 | 37% lower risk of PCSM among men with lower-risk tumours (Gleason score 2–7, T1–T2, p = 0.02), 31% lower risk of PCSM (>17.5 vs. 3.5 ≤ 8.75 MET-h/week)—no difference observed by tumour risk category |
| Dieli-Conwright C.M. et al. [40] | overweight and obese survivors of breast cancer (n = 100) | aerobic, resistance exercise | 3 times per week for 16 weeks | Improved levels of insulin, IGF-1, leptin, adiponectin, BMI, skeletal mass index. At 3-month follow-up, all variables remained improved. |
| Tubiana-Mathieu N. et al. [42] | breast cancer (n = 138) | CPET, 6MWT- 6-min walk test | 6 min | 6MWT allows for the calculation of the required exercise intensity |
| Watson G.A. et al. [43] | Colorectal cancer (n = 832, n = 573) breast, colon cancer (multiple systematic reviews) | aerobic and resistance training | 150 min of moderate intensity aerobic exercise in 3–5 sessions per week; resistance training—at least 2 days per week for 6–12 weeks | PA: reduces mortality and risk of recurrence in cancer survivors; improves QoL, allows maintaining a healthy weight, decreases fatigue. |
| Rogers L.Q. et al. [44] | breast cancer (n = 222) | BEAT Cancer—physical activity behaviour change intervention | PA recommendations from American Cancer Society, 12 supervised exercise sessions for the first 6 weeks, then unsupervised home-based exercises, >150 min/week of moderate to vigorous PA, 3- and 6-month follow-up | BEAT Cancer was superior to usual care for improvement in sleep quality (p < 0.01) |
4. Discussion
5. Limitations
6. Conclusions
Author Contributions
Funding
Data Availability Statement
Conflicts of Interest
References
- Sung, H.; Ferlay, J.; Siegel, R.L.; Laversanne, M.; Soerjomataram, I.; Jemal, A.; Bray, F. Global cancer statistics 2020: GLOBOCAN estimates of incidence and mortality worldwide for 36 cancers in 185 countries. CA Cancer J. Clin. 2021, 71, 209–249. [Google Scholar] [CrossRef] [PubMed]
- Carayol, M.; Ninot, G.; Senesse, P.; Bleuse, J.P.; Gourgou, S.; Sancho-Garnier, H.; Jacot, W. Short-and long-term impact of adapted physical activity and diet counseling during adjuvant breast cancer therapy: The “APAD1” randomized controlled trial. BMC Cancer 2019, 19, 737. [Google Scholar] [CrossRef] [PubMed]
- Singh, B.; Hayes, S.C.; Spence, R.R.; Steele, M.L.; Millet, G.Y.; Gergele, L. Exercise and colorectal cancer: A systematic review and meta-analysis of exercise safety, feasibility and effectiveness. Int. J. Behav. Nutr. 2020, 17, 122. [Google Scholar] [CrossRef] [PubMed]
- Avancini, A.; Sartori, G.; Gkountakos, A.; Casali, M.; Trestini, I.; Tregnago, D.; Pilotto, S. Physical Activity and Exercise in Lung Cancer Care: Will Promises Be Fulfilled? Oncologist 2020, 25, e555–e569. [Google Scholar] [CrossRef]
- Campbell, K.L.; Winters-Stone, K.; Wiskemann, J.; May, A.M.; Schwartz, A.L.; Courneya, K.S.; Schmitz, K.H. Exercise guidelines for cancer survivors: Consensus statement from international multidisciplinary roundtable. Med. Sci. Sports Exerc. 2019, 51, 2375. [Google Scholar] [CrossRef]
- Bull, F.C.; Al-Ansari, S.S.; Biddle, S.; Borodulin, K.; Buman, M.P.; Cardon, G.; Carty, C.; Chaput, J.P.; Chastin, S.; Chou, R.; et al. World Health Organization 2020 guidelines on physical activity and sedentary behaviour. Br. J. Sports Med. 2020, 54, 1451–1462. [Google Scholar] [CrossRef]
- Liska, T.M.; Kolen, A.M. The role of physical activity in cancer survivors’ quality of life. Health Qual. Life Outcomes 2020, 18, 197. [Google Scholar] [CrossRef]
- An, K.; Morielli, A.R.; Kang, D.; Friedenreich, C.M.; McKenzie, D.C.; Gelmon, K.; Courneya, K.S. Effects of exercise dose and type during breast cancer chemotherapy on longer-term patient-reported outcomes and health-related fitness: A randomized controlled trial. Int.J. Cancer Res. 2020, 146, 150–160. [Google Scholar] [CrossRef]
- Cannioto, R.A.; Hutson, A.; Dighe, S.; McCann, W.; McCann, S.E.; Zirpoli, G.R.; Ambrosone, C.B. Physical activity before, during and after chemotherapy for high-risk breast cancer: Relationships with survival. J. Natl. Cancer Inst. 2021, 113, 54–63. [Google Scholar] [CrossRef]
- Parker, N.H.; Arlinghaus, K.R.; Johnston, C.A. Integrating Physical Activity into Clinical Cancer Care. Am. J. Lifestyle Med. 2018, 12, 220–223. [Google Scholar] [CrossRef]
- Patel, A.V.; Friedenreich, C.M.; Moore, S.C.; Hayes, S.C.; Silver, J.K.; Campbell, K.L.; Winters-Stone, K.; Gerber, L.H.; George, S.M.; Fulton, J.E.; et al. American College of Sports Medicine Roundtable Report on Physical Activity, Sedentary Behavior, and Cancer Prevention and Control. Med. Sci. Sports Exerc. 2019, 51, 2391–2402. [Google Scholar] [CrossRef] [PubMed]
- National Cancer Institute Cancer Trends Progress Report. Available online: https://progressreport.cancer.gov/after/physical_activity (accessed on 23 July 2022).
- Avancini, A.; Pala, V.; Trestini, I.; Tregnago, D.; Mariani, L.; Sieri, S.; Krogh, V.; Boresta, M.; Milella, M.; Pilotto, S.; et al. Exercise Levels and Preferences in Cancer Patients: A Cross-Sectional Study. Int. J. Environ. Health Res. 2020, 17, 5351. [Google Scholar] [CrossRef]
- Yellen, S.B.; Cella, D.F.; Webster, K.; Blendowski, C.; Kaplan, E. Measuring fatigue and other anemia-related symptoms with the Functional Assessment of Cancer Therapy (FACT) measurement system. J. Pain Symptom Manag. 1997, 13, 63–74. [Google Scholar] [CrossRef]
- Cella, D.F.; Bonomi, A.E.; Leslie, W.T. Quality of life and nutritional well-being: Measurementand relationship. Oncology 1993, 7, 105–111. [Google Scholar]
- Cella, D.F.; Jacobsen, P.B.; Orav, E.J.; Holland, J.C.; Silberfarb, P.M.; Rafla, S. A brief POMS measure of distress for cancer patients. J. Chronic. Dis. 1987, 40, 939–942. [Google Scholar] [CrossRef]
- Nielsen, A.M.; Welch, W.A.; Gavin, K.L.; Cottrell, A.M.; Solk, P.; Torre, E.A.; Phillips, S.M. Preferences for mHealth physical activity interventions during chemotherapy for breast cancer: A qualitative evaluation. Supportive Care Cancer 2020, 28, 1919–1928. [Google Scholar] [CrossRef]
- Parker, N.H.; Ngo-Huang, A.; Lee, R.E.; O’Connor, D.P.; Basen-Engquist, K.M.; Petzel, M.Q.; Katz, M.H. Physical activity and exercise during preoperative pancreatic cancer treatment. Supportive Care Cancer 2019, 27, 2275–2284. [Google Scholar] [CrossRef]
- Servadio, M.; Cottone, F.; Sommer, K.; Oerlemans, S.; van de Poll-Franse, L.; Efficace, F. Physical activity and health-related quality of life in multiple myeloma survivors: The PROFILES registry. BMJ. Supportive Palliat. Care 2020, 10, 35. [Google Scholar] [CrossRef]
- Nicol, J.L.; Woodrow, C.; Burton, N.W.; Mollee, P.; Nicol, A.J.; Hill, M.M.; Skinner, T.L. Physical activity in people with multiple myeloma: Associated factors and exercise program preferences. J. Clin. Med. 2020, 9, 3277. [Google Scholar] [CrossRef]
- Chen, J.J.; Wu, P.T.; Middlekauff, H.R. Aerobic exercise in anthracycline-induced cardiotoxicity: A systematic review of current evidence and future directions. Am. J. Physiol. Heart Circ. Physiol. 2017, 312, H213–H222. [Google Scholar] [CrossRef]
- Dun, L.; Xian-Yi, W.; Xiao-Ying, J. Effects of Moderate-To-Vigorous Physical Activity on Cancer-Related Fatigue in Patients with Colorectal Cancer: A Systematic Review and Meta-Analysis. Arch. Med. Res. 2020, 51, 173–179. [Google Scholar] [CrossRef] [PubMed]
- Dieli-Conwright, C.M.; Courneya, K.S.; Demark-Wahnefried, W.; Sami, N.; Lee, K.; Sweeney, F.C.; Mortimer, J.E. Aerobic and resistance exercise improves physical fitness, bone health, and quality of life in overweight and obese breast cancer survivors: A randomized controlled trial. Breast Cancer Res. 2018, 20, 124. [Google Scholar] [CrossRef] [PubMed]
- Cheung, A.T.; Li, W.H.C.; Ho, L.L.K.; Ho, K.Y.; Chan, G.C.F.; Chung, J.O.K. Physical activity for pediatric cancer survivors: A systematic review of randomized controlled trials. J. Cancer Surviv. 2021, 15, 876–889. [Google Scholar] [CrossRef] [PubMed]
- Howell, C.R.; Krull, K.R.; Partin, R.E.; Kadan-Lottick, N.S.; Robison, L.L.; Hudson, M.M. Randomized web-based physical activity intervention in adolescent survivors of childhood cancer. Pediatr. Blood Cancer. 2018, 65, e27216. [Google Scholar] [CrossRef] [PubMed]
- Li, W.H.C.; Ho, K.Y.; Lam, K.K.W.; Lam, H.S.; Chui, S.Y.; Chan, G.C.F. Adventure-based training to promote physical activity and reduce fatigue among childhood cancer survivors: A randomized controlled trial. Int. J. Nurs. Stud. 2018, 83, 65–74. [Google Scholar] [CrossRef]
- Jones, T.L.; Sandler, C.X.; Spence, R.R.; Hayes, S.C. Physical activity and exercise in women with ovarian cancer: A systematic review. Gynecol. Oncol. 2020, 158, 803–811. [Google Scholar] [CrossRef]
- Groeneveldt, L.; Mein, G.; Garrod, R.; Jewell, A.P.; Someren, K.V.; Stephens, R.; D’Sa, S.P.; Yong, K.L. A mixed exercise training programme is feasible and safe and may improve quality of life and muscle strength in multiple myeloma survivors. BMC Cancer 2013, 13, 31. [Google Scholar] [CrossRef]
- Jones, L.W.; Courneya, K.S.; Vallance, J.K.; Ladha, A.B.; Mant, M.J.; Belch, A.R.; Stewart, D.A.; Reiman, T. Association between exercise and quality of life in multiple myeloma cancer survivors. Supportive Care Cancer 2004, 12, 780–788. [Google Scholar] [CrossRef]
- Shallwani, S.; Dalzell, M.-A.; Sateren, W.; O’Brien, S. Exercise compliance among patients with multiple myeloma undergoing chemotherapy: A retrospective study. Supportive Care Cancer 2015, 23, 3081–3088. [Google Scholar] [CrossRef]
- Galvao, D.A.; Taaffe, D.R.; Spry, N.; Cormie, P.; Joseph, D.; Chambers, S.K.; Newton, R.U. Exercise Preserves Physical Function in Prostate Cancer Patients with Bone Metastases. Med. Sci. Sports Exerc. 2018, 50, 393–399. [Google Scholar] [CrossRef]
- Cataldi, S.; Amato, A.; Messina, G.; Iovane, A.; Greco, G.; Guarini, A.; Patrizia, P.; Fischetti, F. Effects of combined exercise on psychological and physiological variables in cancer patients: A pilot study. Acta Med. Mediterr. 2020, 36, 1105–1113. [Google Scholar] [CrossRef]
- McTiernan, A.; Friedenreich, C.M.; Katzmarzyk, P.T.; Powell, K.E.; Macko, R.; Buchner, D.; Piercy, K.L. Physical activity in cancer prevention and survival: A systematic review. Med. Sci. Sports Exerc. 2019, 51, 1252. [Google Scholar] [CrossRef]
- Palesh, O.; Kamen, C.; Sharp, S.; Golden, A.; Neri, E.; Spiegel, D.; Koopman, C. Physical Activity and Survival in Women with Advanced Breast Cancer. Cancer Nurs. 2018, 41, E31. [Google Scholar] [CrossRef] [PubMed]
- Di Maso, M.; Augustin, L.S.A.; Toffolutti, F.; Stocco, C.; Dal Maso, L.; Jenkins, D.J.A.; Fleshner, N.E.; Serraino, D.; Polesel, J. Adherence to Mediterranean Diet, Physical Activity and Survival after Prostate Cancer Diagnosis. Nutrients 2021, 13, 243. [Google Scholar] [CrossRef] [PubMed]
- Friedenreich, C.M.; Neilson, H.K.; Farris, M.S.; Courneya, K.S. Physical activity and cancer outcomes: A precision medicine approach. Clin. Cancer Res. 2016, 22, 4766–4775. [Google Scholar] [CrossRef]
- Barnard, R.J.; Ngo, T.H.; Leung, P.S.; Aronson, W.J.; Golding, L.A. A low-fat diet and/or strenuous exercise alters the IGF axis in vivo and reduces prostate tumour cell growth in vitro. Prostate 2003, 56, 201–206. [Google Scholar] [CrossRef]
- Barnard, R.J.; Kobayashi, N.; Aronson, W.J. Effect of diet and exercise intervention on the growth of prostate epithelial cells. Prostate Cancer Prostatic Dis. 2008, 11, 362–366. [Google Scholar] [CrossRef]
- Wang, Y.; Jacobs, E.J.; Gapstur, S.M.; Maliniak, M.L.; Gansler, T.; McCullough, M.L.; Patel, A.V. Recreational Physical Activity in Relation to Prostate Cancer–specific Mortality Among Men with Nonmetastatic Prostate Cancer. Eur. Urol. 2017, 72, 931–939. [Google Scholar] [CrossRef]
- Dieli-Conwright, C.M.; Courneya, K.S.; Demark-Wahnefried, W.; Sami, N.; Lee, K.; Buchanan, T.A.; Mortimer, J.E. Effects of aerobic and resistance exercise on metabolic syndrome, sarcopenic obesity, and circulating biomarkers in overweight or obese survivors of breast cancer: A randomized controlled trial. J. Clin. Oncol. 2018, 36, 875. [Google Scholar] [CrossRef]
- Ainsworth, B.E.; Haskell, W.L.; Herrmann, S.D. Compendium of Physical Activities: A second update ofcodes and MET values. Med. Sci. Sports Exerc. 2011, 43, 1575–1581. [Google Scholar] [CrossRef]
- Tubiana-Mathieu, N.; Cornette, T.; Mandigout, S.; Leobon, S.; Vincent, F.; Venat, L.; Deluche, E. Can the Six-Minute Walk Test Be Used to Individualize Physical Activity Intensity in Patients with Breast Cancer? Cancers 2021, 13, 5851. [Google Scholar] [CrossRef] [PubMed]
- Watson, G.A.; Leonard, G.D. Prescribing exercise for cancer survivors: Time for physicians to become more proactive. Ir. Med. J. 2020, 113, 25. [Google Scholar]
- Rogers, L.Q.; Courneya, K.S.; Oster, R.A.; Anton, P.M.; Robbs, R.S.; Forero, A.; Mcauley, E. Physical Activity and Sleep Quality in Breast Cancer Survivors. Med. Sci. Sports Exerc. 2009, 49, 2009–2015. [Google Scholar] [CrossRef] [PubMed]
- Kent, E.E.; Ambs, A.; Mitchell, S.A.; Clauser, S.B.; Smith, A.W.; Hays, R.D. Health-related quality of life in older adult survivors of selected cancers: Data from the SEER-MHOS linkage. Cancer 2015, 121, 758–765. [Google Scholar] [CrossRef] [PubMed]
- Cataldi, S.; Greco, G.; Mauro, M.; Fischetti, F. Effect of exercise on cancer-related fatigue: A systematic review. J. Hum. Sport. 2020, 16, 476–492. [Google Scholar] [CrossRef]
- Johnsen, A.T.; Tholstrup, D.; Petersen, M.A.; Pedersen, L.; Groenvold, M. Health related quality of life in a nationally representative sample of haematological patients. Eur. J. Haematol. 2009, 83, 139–148. [Google Scholar] [CrossRef] [PubMed]
- Shapiro, C.L. Cancer Survivorship. N. Engl J. Med. 2018, 379, 2438–2450. [Google Scholar] [CrossRef]
- Hojman, P.; Gehl, J.; Christensen, J.F.; Pedersen, B.K. Molecular Mechanisms Linking Exercise to Cancer Prevention and Treatment. Cell Metab. 2018, 27, 10–21. [Google Scholar] [CrossRef]
- Song, M.; Chan, A.T. The Potential Role of Exercise and Nutrition in Harnessing the Immune System to Improve Colorectal Cancer Survival. Gastroenterology 2018, 155, 596–600. [Google Scholar] [CrossRef]
- Derakhshani, A.; Hashemzadeh, S.; Asadzadeh, Z.; Shadbad, M.A.; Rasibonab, F.; Safarpour, H.; Baradaran, B. Cytotoxic T-lymphocyte antigen-4 in colorectal cancer: Another therapeutic side of capecitabine. Cancers 2021, 13, 2414. [Google Scholar] [CrossRef]
- Gustafson, M.P.; Wheatley-Guy, C.M.; Rosenthal, A.C.; Gastineau, D.A.; Katsanis, E.; Johnson, B.D.; Simpson, R.J. Exercise and the immune system: Taking steps to improve responses to cancer immunotherapy. J. Immunother. Cancer 2021, 9, e001872. [Google Scholar] [CrossRef] [PubMed]
- Toohey, K. Exercise in Cancer Care. Semin. Oncol. Nurs. 2020, 36, 51069. [Google Scholar] [CrossRef] [PubMed]
- Hayes, S.C.; Newton, R.U.; Spence, R.R.; Galvão, D.A. The Exercise and Sports Science Australia position statement: Exercise medicine in cancer management. J. Sci. Med. Sport 2019, 22, 1175–1199. [Google Scholar] [CrossRef]
- Romero, S.A.D.; Brown, J.C.; Bauml, J.M.; Hay, J.L.; Li, Q.S.; Cohen, R.B.; Mao, J.J. Barriers to physical activity: A study of academic and community cancer survivors with pain. J. Cancer Surviv. 2018, 12, 744–752. [Google Scholar] [CrossRef]
- Denlinger, C.S.; Sanft, T.; Baker, K.S.; Broderick, G.; Demark-Wahnefried, W.; Friedman, D.L.; Freedman-Cass, D.A. Survivorship, version 2.2018, NCCN clinical practice guidelines in oncology. J. Natl. Compr. Cancer Netw. 2018, 16, 1216–1247. [Google Scholar] [CrossRef] [PubMed]
- Newton, R.U.; Taaffe, D.R.; Galvao, D.A. Clinical Oncology Society of Australia position statement on exercise in cancer care. Med. J. Aust. 2019, 210, 54. [Google Scholar] [CrossRef]
- Schmitz, K.H.; Courneya, K.S.; Matthews, C.; Demark-Wahnefried, W.; Galvao, D.A.; Pinto, B.M.; Irwin, M.L.; Wolin, K.Y.; Segal, R.J.; Lucia, A.; et al. American college of sports medicine roundtable on exercise guidelines for cancer survivors. Med. Sci. Sports Exerc. 2010, 42, 1409–1426. [Google Scholar] [CrossRef] [PubMed]
- Chasan-Taber, L.; Erickson, J.B.; McBride, J.W. Reproducibility of a self- administered lifetime physical activity questionnaire among female college alumnae. Am. J. Epidemiol. 2002, 155, 282–289. [Google Scholar] [CrossRef] [PubMed]
- Clinton, S.K.; Giovannucci, E.L.; Hursting, S.D. The World Cancer Research Fund/American Institute for Cancer Research Third Expert Report on Diet, Nutrition, Physical Activity, and Cancer: Impact and Future Directions. J. Nutr. 2019, 150, 663–671. [Google Scholar] [CrossRef]
- Ośmiałowska, E.; Misiąg, W.; Chabowski, M.; Jankowska-Polańska, B. Coping strategies, pain, and quality of life in patients with breast cancer. J. Clin. Med. 2021, 10, 4469. [Google Scholar] [CrossRef]
- Eng, L.; Pringle, D.; Su, J.; Shen, X.; Mahler, M.; Niu, C.; Jones, J.M. Patterns, perceptions, and perceived barriers to physical activity in adult cancer survivors. Supportive Care Cancer 2018, 26, 3755–3763. [Google Scholar] [CrossRef] [PubMed]
- Cho, D.; Park, C.L. Barriers to physical activity and healthy diet among breast cancer survivors: A multilevel perspective. Eur J. Cancer Care 2017, 27, e12772. [Google Scholar] [CrossRef] [PubMed]
- Min, J.; Yoo, S.; Kim, M.-J.; Yang, E.; Hwang, S.; Kang, M.; Jeon, J.Y. Exercise participation, barriers, and preferences in Korean prostate cancer survivors. Ethn. Health 2021, 26, 1130–1142. [Google Scholar] [CrossRef] [PubMed]
- Jiang, C.; Deng, L.; Karr, M.A.; Wen, Y.; Wang, Q.; Perimbeti, S.; Shapiro, C.L.; Han, X. Chronic comorbid conditions among adult cancer survivors in the United States: Results from the National Health Interview Survey, 2002–2018. Cancer 2022, 128, 828–838. [Google Scholar] [CrossRef] [PubMed]
- Greenlee, H.; Shi, Z.; Sardo Molmenti, C.L.; Rundle, A.; Wen, Y. Trends in Obesity Prevalence in Adults with a History of Cancer: Results from the US National Health Interview Survey, 1997 to 2014. J. Clin. Oncol. 2016, 34, 3133–3140. [Google Scholar] [CrossRef]
- Spence, R.R.; Sandler, C.X.; Newton, R.U.; Galvão, D.A.; Hayes, S.C. Physical Activity and Exercise Guidelines for People with Cancer: Why Are They Needed, Who Should Use Them, and When? Semin. Oncol. Nurs. 2020, 36, 151075. [Google Scholar] [CrossRef]

Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Misiąg, W.; Piszczyk, A.; Szymańska-Chabowska, A.; Chabowski, M. Physical Activity and Cancer Care—A Review. Cancers 2022, 14, 4154. https://doi.org/10.3390/cancers14174154
Misiąg W, Piszczyk A, Szymańska-Chabowska A, Chabowski M. Physical Activity and Cancer Care—A Review. Cancers. 2022; 14(17):4154. https://doi.org/10.3390/cancers14174154
Chicago/Turabian StyleMisiąg, Weronika, Anna Piszczyk, Anna Szymańska-Chabowska, and Mariusz Chabowski. 2022. "Physical Activity and Cancer Care—A Review" Cancers 14, no. 17: 4154. https://doi.org/10.3390/cancers14174154
APA StyleMisiąg, W., Piszczyk, A., Szymańska-Chabowska, A., & Chabowski, M. (2022). Physical Activity and Cancer Care—A Review. Cancers, 14(17), 4154. https://doi.org/10.3390/cancers14174154







