A Cohort Study of Free Light Chain Ratio in Combination with Serum Protein Electrophoresis as a First-Line Test in General Practice
Abstract
:Simple Summary
Abstract
1. Introduction
2. Materials and Methods
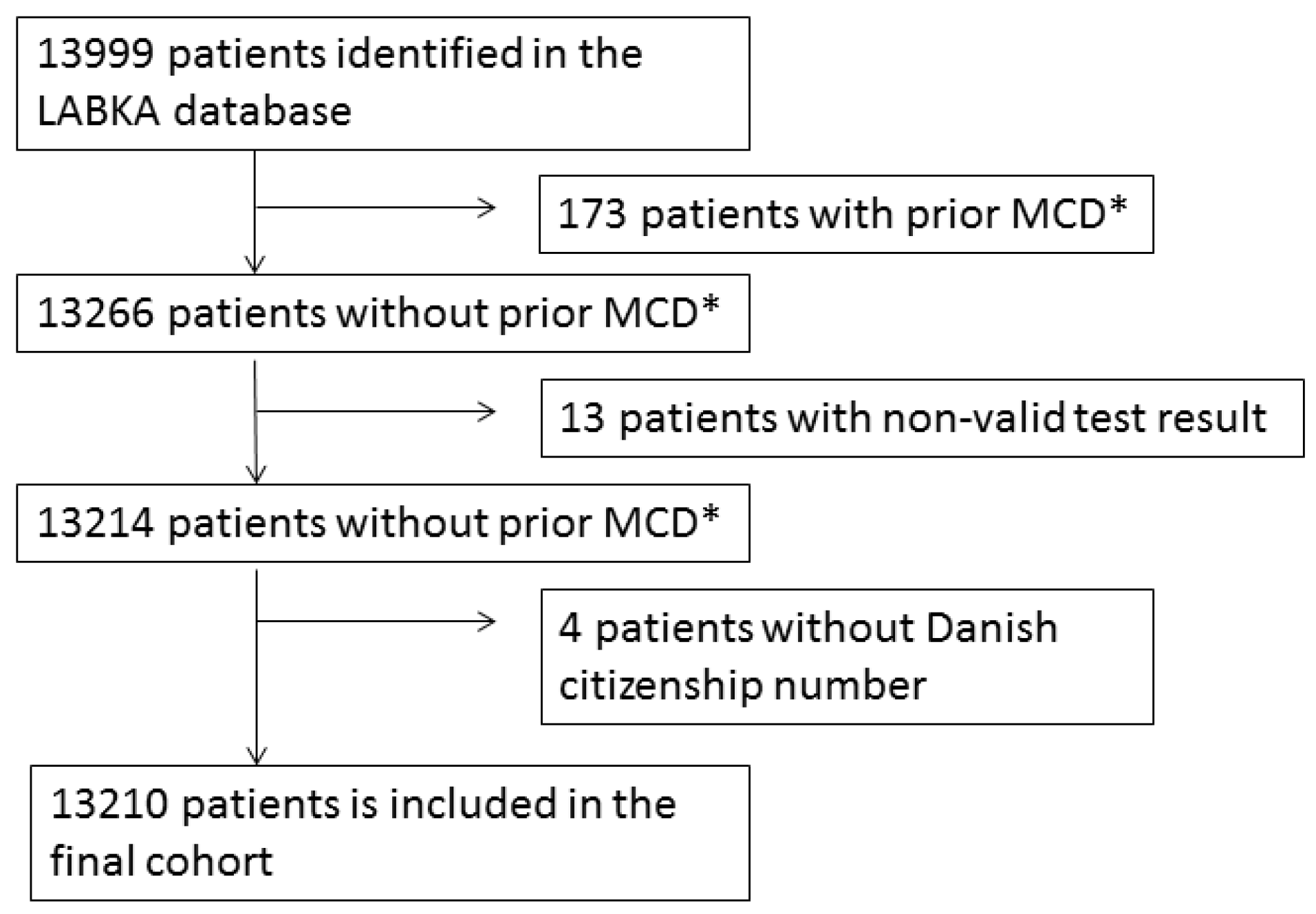
2.1. Patients
2.2. Laboratory Methods
2.3. Statistics
3. Results
3.1. Patients
3.2. sFLC Ratio Test Results
3.3. Diagnostic Value of Individual and Combined Tests
3.4. Clinical Cut-Offs
3.5. Comorbidity
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Ingeman, M.L.; Christensen, M.B.; Bro, F.; Knudsen, S.T.; Vedsted, P. The Danish cancer pathway for patients with serious non-specific symptoms and signs of cancer—A cross-sectional study of patient characteristics and cancer probability. BMC Cancer 2015, 15, 421. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Danckert, B.; Ferlay, J.; Engholm, G.; Hansen, H.L.; Johannesen, T.B.; Khan, S.; Køtlum, J.E.; Ólafsdóttir, E.; Schmidt, L.K.H.; Virtanen, A.; et al. NORDCAN: Cancer Incidence, Mortality, Prevalende and Survival in the Nordic Countries, Version 8.2 (26 March 2019). 2019. Available online: https://www-dep.iarc.fr/NORDCAN/DK/frame.asp (accessed on 8 October 2020).
- Hill, P.G.; Forsyth, J.M.; Rai, B.; Mayne, S. Serum free light chains: An alternative to the urine Bence Jones proteins screening test for monoclonal gammopathies. Clin. Chem. 2006, 52, 1743–1748. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Holding, S.; Spradbery, D.; Hoole, R.; Wilmot, R.; Shields, M.L.; Levoguer, A.M.; Doré, P.C. Use of serum free light chain analysis and urine protein electrophoresis for detection of monoclonal gammopathies. Clin. Chem. Lab. Med. 2011, 49, 83–88. [Google Scholar] [CrossRef] [PubMed]
- McTaggart, M.P.; Lindsay, J.; Kearney, E.M. Replacing urine protein electrophoresis with serum free light chain analysis as a first-line test for detecting plasma cell disorders offers increased diagnostic accuracy and potential health benefit to patients. Am. J. Clin. Pathol. 2013, 140, 890–897. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Rajkumar, S.V.; Dimopoulos, M.A.; Palumbo, A.; Blade, J.; Merlini, G.; Mateos, M.-V.; Kumar, S.; Hillengass, J.; Kastritis, E.; Richardson, P.; et al. International Myeloma Working Group updated criteria for the diagnosis of multiple myeloma. Lancet Oncol. 2014, 15, e538–e548. [Google Scholar] [CrossRef]
- Drayson, M.; Tang, L.X.; Drew, R.; Mead, G.P.; Carr-Smith, H.; Bradwell, A. Serum free light-chain measurements for identifying and monitoring patients with nonsecretory multiple myeloma. Blood 2001, 97, 2900–2902. [Google Scholar] [CrossRef] [Green Version]
- Lachmann, H.J.; Gallimore, R.; Gillmore, J.D.; Carr-Smith, H.D.; Bradwell, A.R.; Pepys, M.B.; Hawkins, P.N. Outcome in systemic AL amyloidosis in relation to changes in concentration of circulating free im-munoglobulin light chains following chemotherapy. Br. J. Haematol. 2003, 122, 78–84. [Google Scholar] [CrossRef]
- Gertz, M.A.; Abraham, R.S.; Clark, R.J.; Katzmann, J.A.; Bradwell, A.; Kyle, R.A. Quantitative analysis of serum free light chains: A new marker for the diagnostic evaluation of primary systemic amyloidosis. Am. J. Clin. Pathol. 2003, 119, 274–278. [Google Scholar] [CrossRef]
- Brockhurst, I.; Harris, K.P.G.; Chapman, C.S. Diagnosis and monitoring a case of light-chain deposition disease in the kidney using a new, sensitive immunoassay. Nephrol. Dial. Transplant. 2005, 20, 1251–1253. [Google Scholar] [CrossRef] [Green Version]
- Katzmann, J.A.; Clark, R.J.; Abraham, R.S.; Bryant, S.; Lymp, J.F.; Bradwell, A.R.; Kyle, R.A. Serum reference intervals and diagnostic ranges for free kappa and free lambda immunoglobulin light chains: Relative sensitivity for detection of monoclonal light chains. Clin. Chem. 2002, 48, 1437–1444. [Google Scholar] [CrossRef] [Green Version]
- Katzmann, J.A.; Kyle, R.A.; Benson, J.; Larson, D.R.; Snyder, M.R.; Lust, J.A.; Rajkumar, S.V.; Dispenzieri, A. Screening panels for detection of monoclonal gammopathies. Clin. Chem. 2009, 55, 1517–1522. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Bakshi, N.A.; Gulbranson, R.; Garstka, D.; Bradwell, A.R.; Keren, D.F. Serum free light chain (FLC) measurement can aid capillary zone electrophoresis in detecting subtle FLC-producing M proteins. Am. J. Clin. Pathol. 2005, 124, 214–218. [Google Scholar] [CrossRef] [PubMed]
- Augustson, B.M.; Begum, G.; Dunn, J.A.; Barth, N.J.; Davies, F.; Morgan, G.; Behrens, J.; Smith, A.; Child, J.A.; Drayson, M.T. Early mortality after diagnosis of multiple myeloma: Analysis of patients entered onto the United Kingdom Medical Research Council trials between 1980 and 2002—Medical Research Council Adult Leukaemia Working Party. J. Clin. Oncol. 2005, 23, 9219–9226. [Google Scholar] [CrossRef] [PubMed]
- Abadie, J.M.; Bankson, D.D. Assessment of serum free light chain assays for plasma cell disorder screening in a Veterans Affairs population. Ann. Clin. Lab. Sci. 2006, 36, 157–162. [Google Scholar] [PubMed]
- Piehler, A.P.; Gulbrandsen, N.; Kierulf, P.; Urdal, P. Quantitation of serum free light chains in combination with protein electrophoresis and clinical infor-mation for diagnosing multiple myeloma in a general hospital population. Clin. Chem. 2008, 54, 1823–1830. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Vermeersch, P.; Van Hoovels, L.; Delforge, M.; Mariën, G.; Bossuyt, X. Diagnostic performance of serum free light chain measurement in patients suspected of a monoclonal B-cell disorder. Br. J. Haematol. 2008, 143, 496–502. [Google Scholar] [CrossRef]
- Bradwell, A.R.; Carr-Smith, H.D.; Mead, G.P.; Harvey, T.C.; Drayson, M. Serum test for assessment of patients with Bence Jones myeloma. Lancet 2003, 361, 489–491. [Google Scholar] [CrossRef]
- Starfield, B. Primary care. Participants or gatekeepers? Diabetes Care 1994, 17, 12–17. [Google Scholar]
- Jensen, H.; Tørring, M.L.; Olesen, F.; Overgaard, J.; Vedsted, P. Cancer suspicion in general practice, urgent referral and time to diagnosis: A population-based GP survey and registry study. BMC Cancer 2014, 14, 636. [Google Scholar] [CrossRef] [Green Version]
- Bossuyt, P.M.; Reitsma, J.B.; E Bruns, D.; A Gatsonis, C.; Glasziou, P.; Irwig, L.; Lijmer, J.G.; Moher, D.; Rennie, D.; De Vet, H.C.W.; et al. STARD 2015: An updated list of essential items for reporting diagnostic accuracy studies. BMJ 2015, 351, h5527. [Google Scholar] [CrossRef] [Green Version]
- Hutchison, C.A.; Harding, S.; Hewins, P.; Mead, G.P.; Townsend, J.; Bradwell, A.R.; Cockwell, P. Quantitative assessment of serum and urinary polyclonal free light chains in patients with chronic kidney disease. Clin. J. Am. Soc. Nephrol. 2008, 3, 1684–1690. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Hutchison, C.A.; Plant, T.; Drayson, M.; Cockwell, P.; Kountouri, M.; Basnayake, K.; Harding, S.; Bradwell, A.R.; Mead, G. Serum free light chain measurement aids the diagnosis of myeloma in patients with severe renal failure. BMC Nephrol. 2008, 9, 11–18. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Weiss, B.M.; Abadie, J.; Verma, P.; Howard, R.S.; Kuehl, W.M. A monoclonal gammopathy precedes multiple myeloma in most patients. Blood 2009, 113, 5418–5422. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Weiss, B.M.; Hebreo, J.; Cordaro, D.V.; Roschewski, M.J.; Baker, T.P.; Abbott, K.C.; Olson, S.W. Increased serum free light chains precede the presentation of immunoglobulin light chain amyloidosis. J. Clin. Oncol. 2014, 32, 2699–2704. [Google Scholar] [CrossRef] [Green Version]
- Heaney, J.L.J.; Richter, A.; Bowcock, S.; Pratt, G.; Child, J.A.; Jackson, G.; Morgan, G.; Turesson, I.; Drayson, M.T. Excluding myeloma diagnosis using revised thresholds for serum free light chain ratios and M-protein levels. Haematologica 2020, 105, e169–e171. [Google Scholar] [CrossRef] [PubMed]
- van de Donk, N.W.; Palumbo, A.; Johnsen, H.E.; Engelhardt, M.; Gay, F.; Gregersen, H.; Hajek, R.; Kleber, M.; Ludwig, H.; Morgan, G.; et al. The clinical relevance and management of monoclonal gammopathy of undetermined significance and related disorders: Recommendations from the European Myeloma Network. Haematologica 2014, 99, 984–996. [Google Scholar] [CrossRef] [Green Version]
- Schmidt, M.; Pedersen, L.; Sørensen, H.T. The Danish Civil Registration System as a tool in epidemiology. Eur. J. Epidemiol. 2014, 29, 541–549. [Google Scholar] [CrossRef]
- Gottenberg, J.E.; Aucouturier, F.; Goetz, J.; Sordet, C.; Jahn, I.; Busson, M.; Cayuela, J.M.; Sibilia, J.; Mariette, X. Serum immunoglobulin free light chain assessment in rheumatoid arthritis and primary Sjogren’s syndrome. Ann. Rheum. Dis. 2007, 66, 23–27. [Google Scholar] [CrossRef] [Green Version]
- Teng, M.; Pirrie, S.; Ward, D.; Assi, L.K.; Hughes, R.G.; Stocken, D.D.; Johnson, P.J. Diagnostic and mechanistic implications of serum free light chains, albumin and alpha-fetoprotein in hepatocellular carcinoma. Br. J. Cancer 2014, 110, 2277–2282. [Google Scholar] [CrossRef] [Green Version]
- Yang, Y.; Chen, L.; Jia, Y.; Liu, Y.; Wen, L.; Liang, Y.; An, Y.; Chen, S.; Su, Y.; Li, Z. Monoclonal gammopathy in rheumatic diseases. Clin. Rheumatol. 2018, 37, 1751–1762. [Google Scholar] [CrossRef]
- Galvani, L.; Flanagan, J.; Sargazi, M.; Neithercut, W.D. Validation of serum free light chain reference ranges in primary care. Ann. Clin. Biochem. Int. J. Lab. Med. 2016, 53, 399–404. [Google Scholar] [CrossRef] [PubMed] [Green Version]

| Patients Characteristics | Total | Patients without MCD | Patients with MCD |
|---|---|---|---|
| Total, n | 13,210 | 12,916 | 294 |
| Age | |||
| Years, Median (range) | 64 (1–106) | 64 (1–106) | 70 (39–97) |
| Sex | |||
| Female, n (%) | 7961 (60) | 7841 (61) | 120 (41) |
| Male, n (%) | 5249 (40) | 5075 (39) | 174 (59) |
| Creatinine, µmol/L, Median (range) | 71 (40–280) | 71 (41–189) | 76 (40–280) |
| Number of patients for each analysis, n (%) | |||
| sFLC κ:λ | 3742 (28) | 3595 (28) | 144 (49) |
| SPE | 12,950 (98) | 12,658 (98) | 292 (99) |
| UPE | 3373 (26) | 3237 (25) | 136 (46) |
| Combinations of analyses, n (%) | |||
| SPE | 6717 (51) | 6619 (51) | 98 (33) |
| SPE and UPE | 2649 (20) | 2598 (20) | 51 (17) |
| UPE | 102 (<1) | 101 (1) | 1(<1) |
| SPE and FLC ratio | 2964 (22) | 2905 (22) | 59 (20) |
| UPE and FLC ratio | 2 (<1) | 2 (<1) | 0 |
| FLC ratio | 156 (<1) | 155 (1) | 1 (<1) |
| SPE, UPE and FLC ratio | 620 (5) | 536 (4) | 84 (29) |
| Diagnostic Tests | Total (N) | MCD (N) | Sensitivity | Specificity | PPV | NPV | False Positive N (%) | False Negative N (%) |
|---|---|---|---|---|---|---|---|---|
| Individual tests | ||||||||
| SPE | 12,950 | 292 | 89.7 (85.7–93.0) | 95.9 (95.6–96.3) | 33.7 (30.4–37.2) | 99.8 (99.6–99.8) | 515 (4) | 30 (<1) |
| UPE | 3373 | 136 | 50.7 (42.0–59.4) | 92.3 (91.3–93.2) | 21.6 (17.2–26.6) | 97.8 (97.2–98.3) | 250 (7) | 67 (2) |
| sFLC κ:λ (0.26–1.65) | 3742 | 144 | 71.5 (63.4–78.7) | 83.7 (82.5–84.9) | 14.9 (12.4–17.8) | 98.7 (98.2–99.0) | 586 (16) | 41 (1) |
| sFLC κ:λ (0.26–4.32) | 3742 | 144 | 51.4 (42.9–59.8) | 97.8 (97.3–98.3) | 48.4 (40.2–56.6) | 98.0 (97.5–98.5) | 79 (2) | 70 (2) |
| sFLC κ:λ (0.26–7.0) | 3742 | 144 | 43.8 (35.5–52.3) | 98.4 (98.0–98.8) | 52.9 (43.6–62.2) | 97.8 (97.2–98.2) | 56 (1) | 81 (2) |
| sFLC κ:λ (0.1–10.0) | 3742 | 144 | 34.7 (27.0–43.1) | 99.4 (99.0–99.6) | 68.5 (56.6–78.9) | 97.4 (96.9–97.9) | 23 (1) | 93 (2) |
| Combination of test * SPE and UPE a | 3269 | 135 | 95.6 (90.6–98.4) | 88.4 (87.2–89.5) | 26.2 (22.3–30.3) | 99.8 (99.5–99.9) | 364 (11) | 6 (<1) |
| SPE and sFLC κ:λ (0.26–1.65) b | 3584 | 143 | 95.1 (90.2–98.0) | 80.3 (79.0–81.6) | 16.7 (14.2–19.5) | 99.7 (99.5–99.9) | 677 (19) | 7 (<1) |
| SPE a | 3269 | 135 | 87.4 (80.6–92.5) | 95.0 (94.2–95.7) | 42.9 (37.0–49.0) | 99.4 (99.1–99.7) | 157 (5) | 17 (3) |
| SPE b | 3584 | 143 | 86.0 (79.2–91.2) | 95.1 (94.3–95.8) | 42.1 (36.4–48.0) | 99.4 (99.1–99.6) | 169 (5) | 20 (1) |
| UPE a | 3269 | 135 | 50.4 (41.6–59.1) | 92.2 (91.2–93.1) | 21.8 (17.3–26.8) | 97.7 (97.1–98.2) | 244 (7) | 67 (2) |
| sFLC κ:λ (0.26–1.65) b | 3584 | 143 | 71.3 (63.2–78.6) | 83.3 (82.1–84.6) | 15.1 (12.5–18.0) | 98.6 (98.1–99.0) | 573 (16) | 41 (7) |
| Alternative reference ranges for sFLC κ:λ SPE + sFLC κ:λ (0.26–4.32) b | 3584 | 143 | 93.0 (87.5–96.6) | 93.6 (92.7–94.4) | 37.7 (32.6–43.0) | 99.7 (99.4–99.9) | 229 (6) | 10 (<1) |
| FLC- κ:λ b | 3584 | 143 | 51.0 (42.6–59.5) | 97.8 (97.2–98.2) | 48.7 (40.4–57.0) | 98.0 (97.4–98.4) | 77 (2) | 70 (2) |
| SPE + sFLC κ:λ (0.26–7.00) b | 3584 | 143 | 92.3 (86.7–96.1) | 94.0 (93.2–94.8) | 39.2 (33.9–44.6) | 99.7 (99.4–99.8) | 205 (6) | 11 (<1) |
| sFLC κ:λ b | 3584 | 143 | 43.4 (35.1–51.9) | 98.4 (98.0–98.8) | 53.4 (44.0–62.8) | 97.7 (97.1–98.1) | 54 (2) | 81 (2) |
| SPE + sFLC κ:λ (0.1–10) b | 3584 | 143 | 92.3 (86.7–96.1) | 94.8 (94.0–95.5) | 42.3 (36.8–48.0) | 99.7 (99.4–99.8) | 180 (5) | 11 (<1) |
| sFLC κ:λ b | 3584 | 143 | 34.3 (26.5–47.7) | 99.3 (99.0–99.6) | 68.1 (56.0–78.6) | 97.3 (96.7–97.8) | 23 (<1) | 94 (3) |
| Patient | Diagnosis | κ | λ | sFLC κ:λ | UPE | Days from Sample until Diagnosis |
|---|---|---|---|---|---|---|
| 1 | MM | 1728 | 1 | 1728 | N/A | 2 |
| 2 | MM | 13 | 386 | 0.03 | pos | 150 |
| 3 | Amyloidosis | 681 | 11 | 61.9 | N/A | 35 |
| 4 | Amyloidosis * | 64 | 859 | 0.07 | pos | 47 |
| 5 | WM * | 33 | 6 | 5.48 | pos | 65 |
| 6 | MGUS | 27 | 15 | 1.8 ** | neg | 34 |
| 7 | MGUS | 1350 | 6 | 225 | pos | 351 |
| 8 | MGUS | 559 | 18 | 31.1 | pos | 9 |
| 9 | MGUS* | 155 | 36 | 4.31 ** | pos | 890 |
| 10 | MGUS | 490 | 39 | 12.6 | pos | 17 |
| 11 | MGUS | 187 | 7 | 26.7 | pos | 12 |
| 12 | MGUS* | 22 | 13 | 1.69 ** | pos | 20 |
| 13 | MGUS | 13 | 287 | 0.05 | pos | 2 |
| Comorbidity | Comorbidity Prior to Blood Test | Comorbidity 3 Months after Blood Test | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| n = 3742 | MCD | Non-MCD | MCD | Non-MCD | ||||||||||
| Total | sFLC Abnomal | sFLC Normal | p-Value * | sFLC Abnormal | sFLC Normal | p-Value * | Total | sFLC Abnormal | sFLC Normal | p-Value * | sFLC Abnormal | sFLC Normal | p-Value * | |
| Myocardial infarction | 152 | 4 | 0 | NA | 24 | 124 | 0.98 | 166 | 4 | 0 | NA | 25 | 137 | 0.76 |
| Congestive heart failure | 398 | 9 | 2 | NA | 62 | 325 | 0.88 | 438 | 10 | 3 | NA | 72 | 353 | 0.70 |
| Peripheral vascular | 386 | 5 | 4 | NA | 56 | 321 | 0.43 | 437 | 7 | 5 | 0.32 | 63 | 362 | 0.38 |
| Cerebrovascular | 205 | 5 | 0 | NA | 34 | 166 | 0.78 | 232 | 5 | 0 | NA | 38 | 189 | 0.85 |
| CPD | 290 | 7 | 2 | NA | 45 | 236 | 0.90 | 344 | 7 | 2 | NA | 49 | 286 | 0.39 |
| Diabetes | 210 | 5 | 0 | NA | 42 | 163 | 0.09 | 251 | 5 | 0 | NA | 50 | 196 | 0.08 |
| Diabetes with end-organ damage | 68 | 2 | 0 | NA | 12 | 54 | 0.84 | 79 | 2 | 0 | NA | 14 | 63 | 0.65 |
| Renal disease | 82 | 0 | 2 | NA | 25 | 55 | <0.001 | 118 | 2 | 2 | NA | 32 | 82 | 0.001 |
| Mild liver disease | 15 | 1 | 0 | NA | 2 | 12 | NA | 24 | 1 | 0 | NA | 2 | 21 | NA |
| Ulcer disease | 51 | 2 | 1 | NA | 7 | 41 | 1.00 | 76 | 3 | 1 | NA | 8 | 64 | 0.26 |
| Connective tissue disease | 50 | 1 | 0 | NA | 11 | 48 | 0.24 | 67 | 8 | 1 | NA | 14 | 52 | 0.27 |
| Any tumour | 204 | 5 | 1 | NA | 53 | 145 | <0.001 | 396 | 8 | 1 | NA | 77 | 310 | 0.04 |
| Leukaemia | 2 | 0 | 0 | 1 | 1 | NA | 10 | 0 | 0 | 4 | 6 | NA | ||
| Lymphoma | 0 | 4 | 1 | 0 | NA | 0 | 3 | NA | ||||||
| Metastatic solid tumor | 57 | 2 | 1 | NA | 14 | 40 | 0.05 | 907 | 17 | 8 | 0.64 | 138 | 744 | 0.55 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Sandfeld-Paulsen, B.; Aggerholm-Pedersen, N.; Samson, M.H.; Møller, H.J. A Cohort Study of Free Light Chain Ratio in Combination with Serum Protein Electrophoresis as a First-Line Test in General Practice. Cancers 2022, 14, 2930. https://doi.org/10.3390/cancers14122930
Sandfeld-Paulsen B, Aggerholm-Pedersen N, Samson MH, Møller HJ. A Cohort Study of Free Light Chain Ratio in Combination with Serum Protein Electrophoresis as a First-Line Test in General Practice. Cancers. 2022; 14(12):2930. https://doi.org/10.3390/cancers14122930
Chicago/Turabian StyleSandfeld-Paulsen, Birgitte, Ninna Aggerholm-Pedersen, Mie Hessellund Samson, and Holger Jon Møller. 2022. "A Cohort Study of Free Light Chain Ratio in Combination with Serum Protein Electrophoresis as a First-Line Test in General Practice" Cancers 14, no. 12: 2930. https://doi.org/10.3390/cancers14122930
APA StyleSandfeld-Paulsen, B., Aggerholm-Pedersen, N., Samson, M. H., & Møller, H. J. (2022). A Cohort Study of Free Light Chain Ratio in Combination with Serum Protein Electrophoresis as a First-Line Test in General Practice. Cancers, 14(12), 2930. https://doi.org/10.3390/cancers14122930






