BRCA 1/2 Germline Mutation Predicts the Treatment Response of FOLFIRINOX with Pancreatic Ductal Adenocarcinoma in Korean Patients
Abstract
:Simple Summary
Abstract
1. Introduction
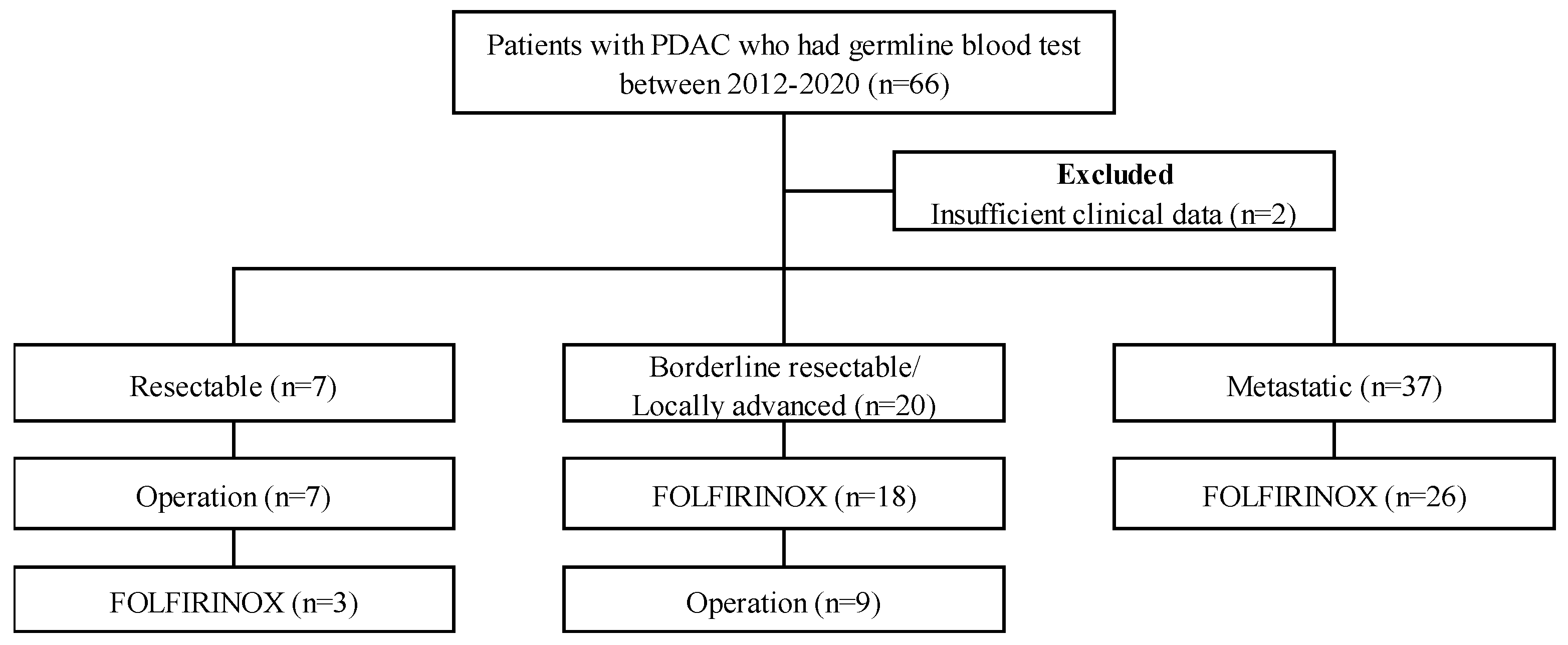
2. Materials and Methods
2.1. Study Population
2.2. Variables
2.3. DNA Extraction and Sequencing
2.4. Statistical Analysis
3. Results
3.1. Patients’ Characteristics and BRCA 1/2 Gene Mutations
3.2. FOLFIRINOX Treatment and Overall Response Rate
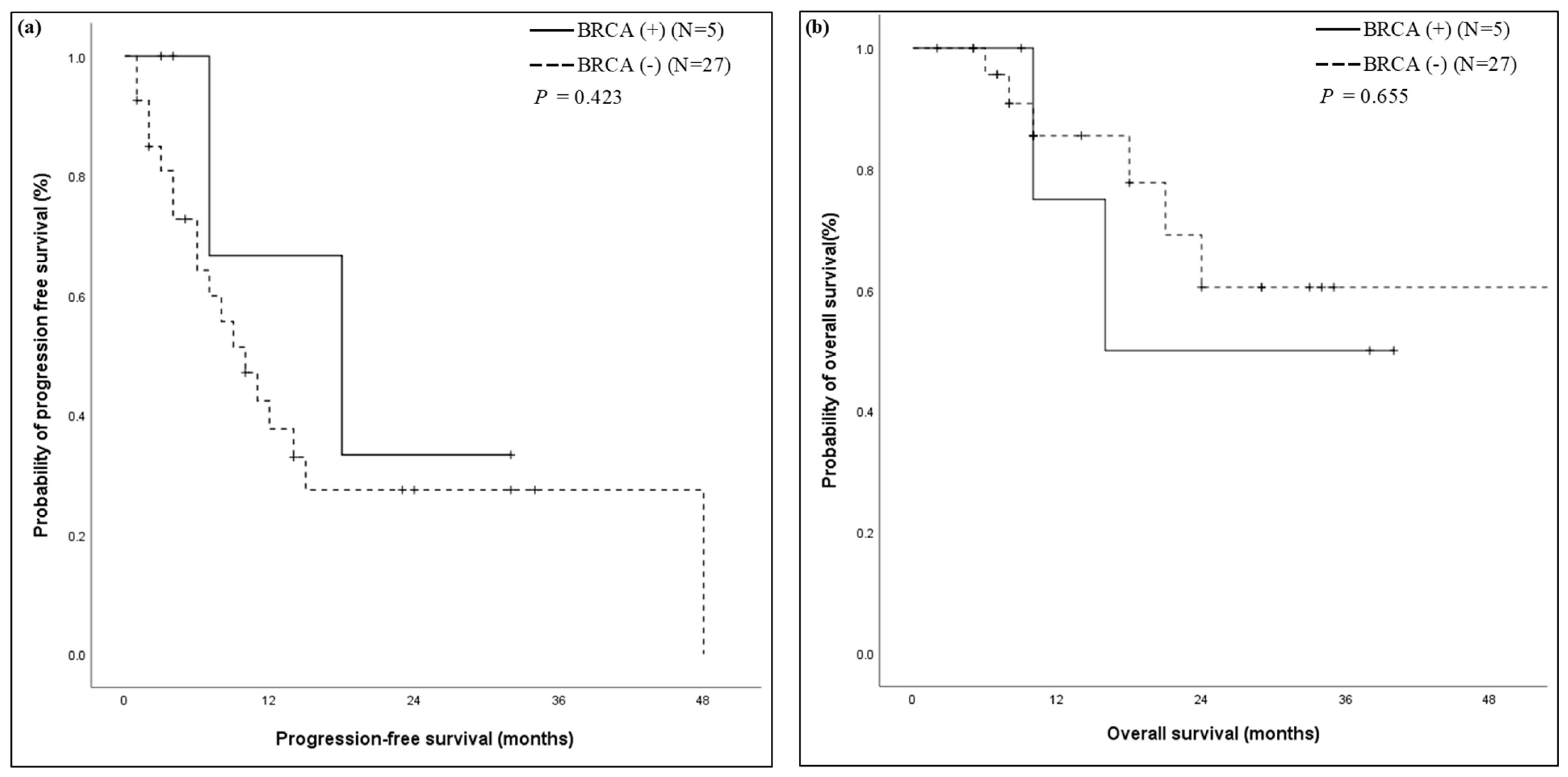
3.3. Progression-Free Survival and Overall Survival
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Rahib, L.; Smith, B.D.; Aizenberg, R.; Rosenzweig, A.B.; Fleshman, J.M.; Matrisian, L.M. Projecting Cancer Incidence and Deaths to 2030: The Unexpected Burden of Thyroid, Liver, and Pancreas Cancers in the United States. Cancer Res. 2014, 74, 2913–2921. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Tempero, M.A.; Malafa, M.P.; Al-Hawary, M.; Behrman, S.W.; Benson, A.B.; Cardin, D.B.; Chiorean, E.G.; Chung, V.; Czito, B.; Del Chiaro, M.; et al. Pancreatic Adenocarcinoma, Version 2.2021, NCCN Clinical Practice Guidelines in Oncology. J. Natl. Compr. Cancer Netw. 2021, 19, 439–457. [Google Scholar] [CrossRef] [PubMed]
- Zhu, H.; Wei, M.; Xu, J.; Hua, J.; Liang, C.; Meng, Q.; Zhang, Y.; Liu, J.; Zhang, B.; Yu, X.; et al. PARP inhibitors in pancreatic cancer: Molecular mechanisms and clinical applications. Mol. Cancer 2020, 19, 49. [Google Scholar] [CrossRef]
- Iqbal, J.; Ragone, A.; Lubinski, J.; Lynch, H.T.; Moller, P.; Ghadirian, P.; Foulkes, W.D.; Armel, S.; Eisen, A.; Neuhausen, S.L.; et al. The incidence of pancreatic cancer in BRCA1 and BRCA2 mutation carriers. Br. J. Cancer 2012, 107, 2005–2009. [Google Scholar] [CrossRef] [PubMed]
- Holter, S.; Borgida, A.; Dodd, A.; Grant, R.; Semotiuk, K.; Hedley, D.; Dhani, N.; Narod, S.; Akbari, M.; Moore, M.; et al. Germline BRCA Mutations in a Large Clinic-Based Cohort of Patients with Pancreatic Adenocarcinoma. J. Clin. Oncol. 2015, 33, 3124–3129. [Google Scholar] [CrossRef] [PubMed]
- John, E.M.; Hopper, J.L.; Beck, J.C.; Knight, J.A.; Neuhausen, S.L.; Senie, R.T.; Ziogas, A.; Andrulis, I.L.; Anton-Culver, H.; Boyd, N.; et al. The Breast Cancer Family Registry: An infrastructure for cooperative multinational, interdisciplinary and translational studies of the genetic epidemiology of breast cancer. Breast Cancer Res. 2004, 6, R375. [Google Scholar] [CrossRef] [Green Version]
- Hahn, S.A.; Greenhalf, B.; Ellis, I.; Sina-Frey, M.; Rieder, H.; Korte, B.; Gerdes, B.; Kress, R.; Ziegler, A.; Raeburn, J.A.; et al. BRCA2 germline mutations in familial pancreatic carcinoma. J. Natl. Cancer Inst. 2003, 95, 214–221. [Google Scholar] [CrossRef] [PubMed]
- Golan, T.; Hammel, P.; Reni, M.; Van Cutsem, E.; Macarulla, T.; Hall, M.J.; Park, J.O.; Hochhauser, D.; Arnold, D.; Oh, D.Y.; et al. Maintenance Olaparib for Germline BRCA-Mutated Metastatic Pancreatic Cancer. N. Engl. J. Med. 2019, 381, 317–327. [Google Scholar] [CrossRef]
- Navarro, E.B.; Lopez, E.V.; Quijano, Y.; Caruso, R.; Ferri, V.; Durand, H.; Cabrera, I.F.; Reques, E.D.; Ielpo, B.; Glagolieva, A.Y.; et al. Impact of BRCA 1/2 gene mutations on survival of patients with pancreatic cancer: A case-series analysis. Ann. Hepato-Biliary-Pancreat. Surg. 2019, 23, 200–205. [Google Scholar] [CrossRef] [Green Version]
- Turner, N.; Tutt, A.; Ashworth, A. Hallmarks of ‘BRCAness’ in sporadic cancers. Nat. Rev. Cancer 2004, 4, 814–819. [Google Scholar] [CrossRef]
- Husain, A.; He, G.; Venkatraman, E.S.; Spriggs, D.R. BRCA1 up-regulation is associated with repair-mediated resistance to cis-diamminedichloroplatinum(II). Cancer Res. 1998, 58, 1120–1123. [Google Scholar] [PubMed]
- Wattenberg, M.M.; Asch, D.; Yu, S.; O’Dwyer, P.J.; Domchek, S.M.; Nathanson, K.L.; Rosen, M.A.; Beatty, G.L.; Siegelman, E.S.; Reiss, K.A. Platinum response characteristics of patients with pancreatic ductal adenocarcinoma and a germline BRCA1, BRCA2 or PALB2 mutation. Br. J. Cancer 2020, 122, 333–339. [Google Scholar] [CrossRef]
- Golan, T.; Barenboim, A.; Lahat, G.; Nachmany, I.; Goykhman, Y.; Shacham-Shmueli, E.; Halpern, N.; Brazowski, E.; Geva, R.; Wolf, I.; et al. Increased Rate of Complete Pathologic Response after Neoadjuvant FOLFIRINOX for BRCA Mutation Carriers with Borderline Resectable Pancreatic Cancer. Ann. Surg. Oncol. 2020, 27, 3963–3970. [Google Scholar] [CrossRef] [PubMed]
- Shimmura, H.; Kuramochi, H.; Jibiki, N.; Katagiri, S.; Nishino, T.; Araida, T. Dramatic response of FOLFIRINOX regimen in a collision pancreatic adenocarcinoma patient with a germline BRCA2 mutation: A case report. Jpn. J. Clin. Oncol. 2019, 49, 1049–1054. [Google Scholar] [CrossRef]
- Rebelatto, T.F.; Falavigna, M.; Pozzari, M.; Spada, F.; Cella, C.A.; Laffi, A.; Pellicori, S.; Fazio, N. Should platinum-based chemotherapy be preferred for germline BReast CAncer genes (BRCA) 1 and 2-mutated pancreatic ductal adenocarcinoma (PDAC) patients? A systematic review and meta-analysis. Cancer Treat. Rev. 2019, 80, 101895. [Google Scholar] [CrossRef] [PubMed]
- Sehdev, A.; Gbolahan, O.; Hancock, B.A.; Stanley, M.; Shahda, S.; Wan, J.; Wu, H.H.; Radovich, M.; O’Neil, B.H. Germline and Somatic DNA Damage Repair Gene Mutations and Overall Survival in Metastatic Pancreatic Adenocarcinoma Patients Treated with FOLFIRINOX. Clin. Cancer Res. 2018, 24, 6204–6211. [Google Scholar] [CrossRef] [Green Version]
- Kim, B.; Won, D.; Jang, M.; Kim, H.; Choi, J.R.; Kim, T.I.; Lee, S.T. Next-generation sequencing with comprehensive bioinformatics analysis facilitates somatic mosaic APC gene mutation detection in patients with familial adenomatous polyposis. BMC Med. Genom. 2019, 12, 103. [Google Scholar] [CrossRef] [Green Version]
- Kim, H.; Choi, D.H. Distribution of BRCA1 and BRCA2 Mutations in Asian Patients with Breast Cancer. J. Breast Cancer 2013, 16, 357–365. [Google Scholar] [CrossRef] [Green Version]
- Golan, T.; Kanji, Z.S.; Epelbaum, R.; Devaud, N.; Dagan, E.; Holter, S.; Aderka, D.; Paluch-Shimon, S.; Kaufman, B.; Gershoni-Baruch, R.; et al. Overall survival and clinical characteristics of pancreatic cancer in BRCA mutation carriers. Br. J. Cancer 2014, 111, 1132–1138. [Google Scholar] [CrossRef]
- Tram, E.; Savas, S.; Ozcelik, H. Missense variants of uncertain significance (VUS) altering the phosphorylation patterns of BRCA1 and BRCA2. PLoS ONE 2013, 8, e62468. [Google Scholar] [CrossRef] [Green Version]
- Hu, H.; Zhu, Y.; Pu, N.; Burkhart, R.A.; Burns, W.; Laheru, D.; Zheng, L.; He, J.; Goggins, M.G.; Yu, J. Association of Germline Variants in Human DNA Damage Repair Genes and Response to Adjuvant Chemotherapy in Resected Pancreatic Ductal Adenocarcinoma. J. Am. Coll. Surg. 2020, 231, 527–535.e514. [Google Scholar] [CrossRef]
- Jimenez-Sainz, J.; Jensen, R.B. Imprecise Medicine: BRCA2 Variants of Uncertain Significance (VUS), the Challenges and Benefits to Integrate a Functional Assay Workflow with Clinical Decision Rules. Genes 2021, 12, 780. [Google Scholar] [CrossRef]
- Lowery, M.A.; Kelsen, D.P.; Stadler, Z.K.; Yu, K.H.; Janjigian, Y.Y.; Ludwig, E.; D’Adamo, D.R.; Salo-Mullen, E.; Robson, M.E.; Allen, P.J.; et al. An emerging entity: Pancreatic adenocarcinoma associated with a known BRCA mutation: Clinical descriptors, treatment implications, and future directions. Oncologist 2011, 16, 1397–1402. [Google Scholar] [CrossRef] [Green Version]
- Golan, T.; Sella, T.; O’Reilly, E.M.; Katz, M.H.; Epelbaum, R.; Kelsen, D.P.; Borgida, A.; Maynard, H.; Kindler, H.; Friedmen, E.; et al. Overall survival and clinical characteristics of BRCA mutation carriers with stage I/II pancreatic cancer. Br. J. Cancer 2017, 116, 697–702. [Google Scholar] [CrossRef] [Green Version]
- Gostimir, M.; Bennett, S.; Moyana, T.; Sekhon, H.; Martel, G. Complete pathological response following neoadjuvant FOLFIRINOX in borderline resectable pancreatic cancer—A case report and review. BMC Cancer 2016, 16, 786. [Google Scholar] [CrossRef] [Green Version]
- Vyas, O.; Leung, K.; Ledbetter, L.; Kaley, K.; Rodriguez, T.; Garcon, M.C.; Saif, M.W. Clinical outcomes in pancreatic adenocarcinoma associated with BRCA-2 mutation. Anticancer Drugs 2015, 26, 224–226. [Google Scholar] [CrossRef] [PubMed]
- Zhao, Q.; Rashid, A.; Gong, Y.; Katz, M.H.; Lee, J.E.; Wolf, R.; Balachandran, A.; Varadhachary, G.R.; Pisters, P.W.; Wang, H.; et al. Pathologic complete response to neoadjuvant therapy in patients with pancreatic ductal adenocarcinoma is associated with a better prognosis. Ann. Diagn. Pathol. 2012, 16, 29–37. [Google Scholar] [CrossRef] [Green Version]
- Sonnenblick, A.; Kadouri, L.; Appelbaum, L.; Peretz, T.; Sagi, M.; Goldberg, Y.; Hubert, A. Complete remission, in BRCA2 mutation carrier with metastatic pancreatic adenocarcinoma, treated with cisplatin based therapy. Cancer Biol. 2011, 12, 165–168. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Muckenhuber, A.; Berger, A.K.; Schlitter, A.M.; Steiger, K.; Konukiewitz, B.; Trumpp, A.; Eils, R.; Werner, J.; Friess, H.; Esposito, I.; et al. Pancreatic Ductal Adenocarcinoma Subtyping Using the Biomarkers Hepatocyte Nuclear Factor-1A and Cytokeratin-81 Correlates with Outcome and Treatment Response. Clin. Cancer Res. 2018, 24, 351–359. [Google Scholar] [CrossRef] [Green Version]
- van der Sijde, F.; Vietsch, E.E.; Mustafa, D.A.M.; Besselink, M.G.; Groot Koerkamp, B.; van Eijck, C.H.J. Circulating Biomarkers for Prediction of Objective Response to Chemotherapy in Pancreatic Cancer Patients. Cancers 2019, 11, 93. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Capello, M.; Lee, M.; Wang, H.; Babel, I.; Katz, M.H.; Fleming, J.B.; Maitra, A.; Wang, H.; Tian, W.; Taguchi, A.; et al. Carboxylesterase 2 as a Determinant of Response to Irinotecan and Neoadjuvant FOLFIRINOX Therapy in Pancreatic Ductal Adenocarcinoma. J. Natl. Cancer Inst. 2015, 107, djv132. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Hohla, F.; Hopfinger, G.; Romeder, F.; Rinnerthaler, G.; Bezan, A.; Stättner, S.; Hauser-Kronberger, C.; Ulmer, H.; Greil, R. Female gender may predict response to FOLFIRINOX in patients with unresectable pancreatic cancer: A single institution retrospective review. Int. J. Oncol. 2014, 44, 319–326. [Google Scholar] [CrossRef] [PubMed]
- Conroy, T.; Desseigne, F.; Ychou, M.; Bouche, O.; Guimbaud, R.; Becouarn, Y.; Adenis, A.; Raoul, J.L.; Gourgou-Bourgade, S.; de la Fouchardiere, C.; et al. FOLFIRINOX versus gemcitabine for metastatic pancreatic cancer. N. Engl. J. Med. 2011, 364, 1817–1825. [Google Scholar] [CrossRef] [PubMed] [Green Version]
| Variables | Total (n = 64) | BRCA Mutation (+) | BRCA Mutation (–) | p Value | ||
|---|---|---|---|---|---|---|
| (n = 9) | (n = 55) | |||||
| Age at diagnosis (year) | 57.5 (48.0–66.8) | 50.0 (47.0–60.0) | 59.0 (48.0–71.0) | 0.122 | ||
| Male | 24 (37.5%) | 2 (22.2%) | 22 (40.0%) | 0.464 | ||
| History of prior malignancy, n (%) | ||||||
| Yes | 19 (29.7%) | 6 (66.7%) | 13 (23.6%) | 0.016 | ||
| Breast | 6 (9.4%) | 4 (44.4%) | 2 (3.64%) | 0.014 | ||
| Family history of any malignancy, n (%) | ||||||
| Yes | 29 (45.3%) | 6 (66.7%) | 23 (41.8%) | 0.279 | ||
| Pancreas | 14 (21.9%) | 2 (22.2%) | 12 (21.8%) | 1.000 | ||
| Breast | 3 (4.7%) | 1 (11.1%) | 2 (3.6%) | 1.000 | ||
| Tobacco use (%) | ||||||
| Yes (past or current) | 16 (36.4%) | 3 (33.3%) | 19 (34.5%) | 1.000 | ||
| BMI (kg/m2) | ||||||
| ≥25.0 | 9 (14.1%) | 1 (11.1%) | 8 (14.5%) | 1.000 | ||
| Diabetes Mellitus | 18 (28.1%) | 1 (11.1%) | 17 (30.9%) | 0.425 | ||
| Hypertension | 21 (32.8%) | 2 (22.2%) | 19 (34.5%) | 0.706 | ||
| CA 19-9 (U/mL) | ||||||
| Elevated (>34.0U/mL) | 51 (79.7%) | 8 (88.9%) | 43 (78.2%) | 0.672 | ||
| Pathology | ||||||
| Well-differentiated | 2 (3.1%) | 0 (0.0%) | 2 (3.6%) | |||
| Moderately differentiated | 24 (37.5%) | 7 (77.8%) | 17 (30.9%) | |||
| Poorly differentiated | 7 (10.9%) | 2 (22.2%) | 5 (9.1%) | |||
| Clinical T stage | ||||||
| T1/2 | 25 (39.1%) | 3 (33.3%) | 22 (40.0%) | 1.000 | ||
| T3/4 | 39 (60.9%) | 6 (66.7%) | 33 (60.0%) | |||
| Clinical n stage | ||||||
| N0 | 26 (40.6%) | 5 (55.6%) | 21 (38.2%) | 0.467 | ||
| Location of primary tumor | ||||||
| Head | 32 (50.0%) | 4 (44.4%) | 28 (50.9%) | 1.000 | ||
| Metastasis site | ||||||
| Liver | 22 (34.4%) | 6 (66.7%) | 16 (29.1%) | 0.053 | ||
| Peritoneum | 13 (20.3%) | 0 (0.0%) | 13 (23.6%) | 0.185 | ||
| Distant LN | 9 (14.1%) | 1 (11.1%) | 8 (14.5%) | 1.000 | ||
| Number of metastasis site | ||||||
| 0 site | 33 (51.6%) | 3 (33.3%) | 30 (54.5%) | 0.296 | ||
| 1 or more sites | 31 (48.4%) | 6 (66.7%) | 25 (45.5%) |
| Variables | Total (n = 43) | BRCA Mutation (+) | BRCA Mutation (–) | p Value | ||
|---|---|---|---|---|---|---|
| (n = 7) | (n = 36) | |||||
| Age at diagnosis (year) | 51.0 (46.0–65.0) | 49.0 (46.0–56.0) | 51.5 (44.5–65.8) | 0.508 | ||
| Male | 18 (41.9%) | 2 (28.6%) | 16 (44.4%) | 0.680 | ||
| History of prior malignancy, n (%) | ||||||
| Yes | 10 (23.3%) | 4 (57.1%) | 6 (16.7%) | 0.040 | ||
| Breast | 5 (11.6%) | 3 (42.9%) | 2 (5.6%) | 0.024 | ||
| Family history of any malignancy, n (%) | ||||||
| Yes | 22 (51.2%) | 6 (85.7%) | 16 (44.4%) | 0.095 | ||
| Pancreas | 10 (23.3%) | 2 (28.6%) | 8 (22.2%) | 0.656 | ||
| Breast | 3 (7.0%) | 1 (14.3%) | 2 (5.6%) | 0.421 | ||
| Tobacco use (%) | ||||||
| Yes (past or current) | 15 (34.9%) | 3 (42.9%) | 12 (33.3%) | 0.680 | ||
| BMI (kg/m2) | ||||||
| ≥25.0 | 9 (20.9%) | 1 (14.3%) | 5 (13.9%) | 1.000 | ||
| Diabetes Mellitus | 9 (20.9%) | 0 (0.0%) | 9 (25.0%) | 0.314 | ||
| Hypertension | 11 (25.6%) | 2 (28.6%) | 9 (25.0%) | 1.000 | ||
| CA 19-9 (U/mL) | ||||||
| Elevated (>34.0 U/mL) | 36 (83.7%) | 6 (85.7%) | 30 (83.3%) | 1.000 | ||
| Pathology | ||||||
| Well-differentiated | 1 (2.3%) | 0 (0.0%) | 1 (2.8%) | |||
| Moderately differentiated | 16 (37.2%) | 5 (71.4%) | 11 (30.6%) | |||
| Poorly differentiated | 5 (11.6%) | 2 (28.6%) | 3 (8.3%) | |||
| Clinical T stage | ||||||
| T1/2 | 16 (37.2%) | 2 (28.6%) | 14 (38.9%) | 0.695 | ||
| T3/4 | 27 (62.8%) | 5 (71.4%) | 22 (61.1%) | |||
| Clinical n stage | ||||||
| N0 | 16 (37.2%) | 3 (42.9%) | 13 (36.1%) | 1.000 | ||
| Location of primary tumor | ||||||
| Head | 22 (51.2%) | 3 (42.9%) | 19 (52.8%) | 0.698 | ||
| Metastasis site | ||||||
| Liver | 17 (39.5%) | 5 (71.4%) | 12 (33.3%) | 0.093 | ||
| Peritoneum | 8 (18.6%) | 0 (0.0%) | 8 (22.2%) | 0.315 | ||
| Distant LN | 5 (11.6%) | 1 (14.3%) | 4 (11.1%) | 1.000 | ||
| Number of metastasis site | ||||||
| 0 site | 19 (44.2%) | 2 (28.6%) | 17 (47.2%) | 0.243 | ||
| 1 or more sites | 24 (55.8%) | 5 (71.4%) | 19 (52.8%) |
| Outcome | BRCA Mutation (+) | BRCA Mutation (-) | p Value | |
|---|---|---|---|---|
| (n = 7) | (n = 36) | |||
| Overall response rate, n (%) | 5 (71.4%) | 5 (13.9%) | 0.004 | |
| Complete response | 0 | 0 | ||
| Partial response | 5 (71.4%) | 5 (13.9%) | ||
| Stable disease | 2 (28.6%) | 26 (72.2%) | ||
| Progressive disease | 0 | 5 (13.9%) | ||
| Line of FOLFIRINOX therapy | ||||
| First | 6 (85.7%) | 34 (94.4%) | 0.421 | |
| Second | 1 (14.3%) | 2 (5.6%) |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Park, J.H.; Jo, J.H.; Jang, S.I.; Chung, M.J.; Park, J.Y.; Bang, S.; Park, S.W.; Song, S.Y.; Lee, H.S.; Cho, J.H. BRCA 1/2 Germline Mutation Predicts the Treatment Response of FOLFIRINOX with Pancreatic Ductal Adenocarcinoma in Korean Patients. Cancers 2022, 14, 236. https://doi.org/10.3390/cancers14010236
Park JH, Jo JH, Jang SI, Chung MJ, Park JY, Bang S, Park SW, Song SY, Lee HS, Cho JH. BRCA 1/2 Germline Mutation Predicts the Treatment Response of FOLFIRINOX with Pancreatic Ductal Adenocarcinoma in Korean Patients. Cancers. 2022; 14(1):236. https://doi.org/10.3390/cancers14010236
Chicago/Turabian StylePark, Ji Hoon, Jung Hyun Jo, Sung Ill Jang, Moon Jae Chung, Jeong Youp Park, Seungmin Bang, Seung Woo Park, Si Young Song, Hee Seung Lee, and Jae Hee Cho. 2022. "BRCA 1/2 Germline Mutation Predicts the Treatment Response of FOLFIRINOX with Pancreatic Ductal Adenocarcinoma in Korean Patients" Cancers 14, no. 1: 236. https://doi.org/10.3390/cancers14010236
APA StylePark, J. H., Jo, J. H., Jang, S. I., Chung, M. J., Park, J. Y., Bang, S., Park, S. W., Song, S. Y., Lee, H. S., & Cho, J. H. (2022). BRCA 1/2 Germline Mutation Predicts the Treatment Response of FOLFIRINOX with Pancreatic Ductal Adenocarcinoma in Korean Patients. Cancers, 14(1), 236. https://doi.org/10.3390/cancers14010236






