Home-Based Aerobic and Resistance Exercise Interventions in Cancer Patients and Survivors: A Systematic Review
Simple Summary
Abstract
1. Introduction
2. Materials and Methods
2.1. Search Strategy
2.2. Data Extraction and Quality Assessment
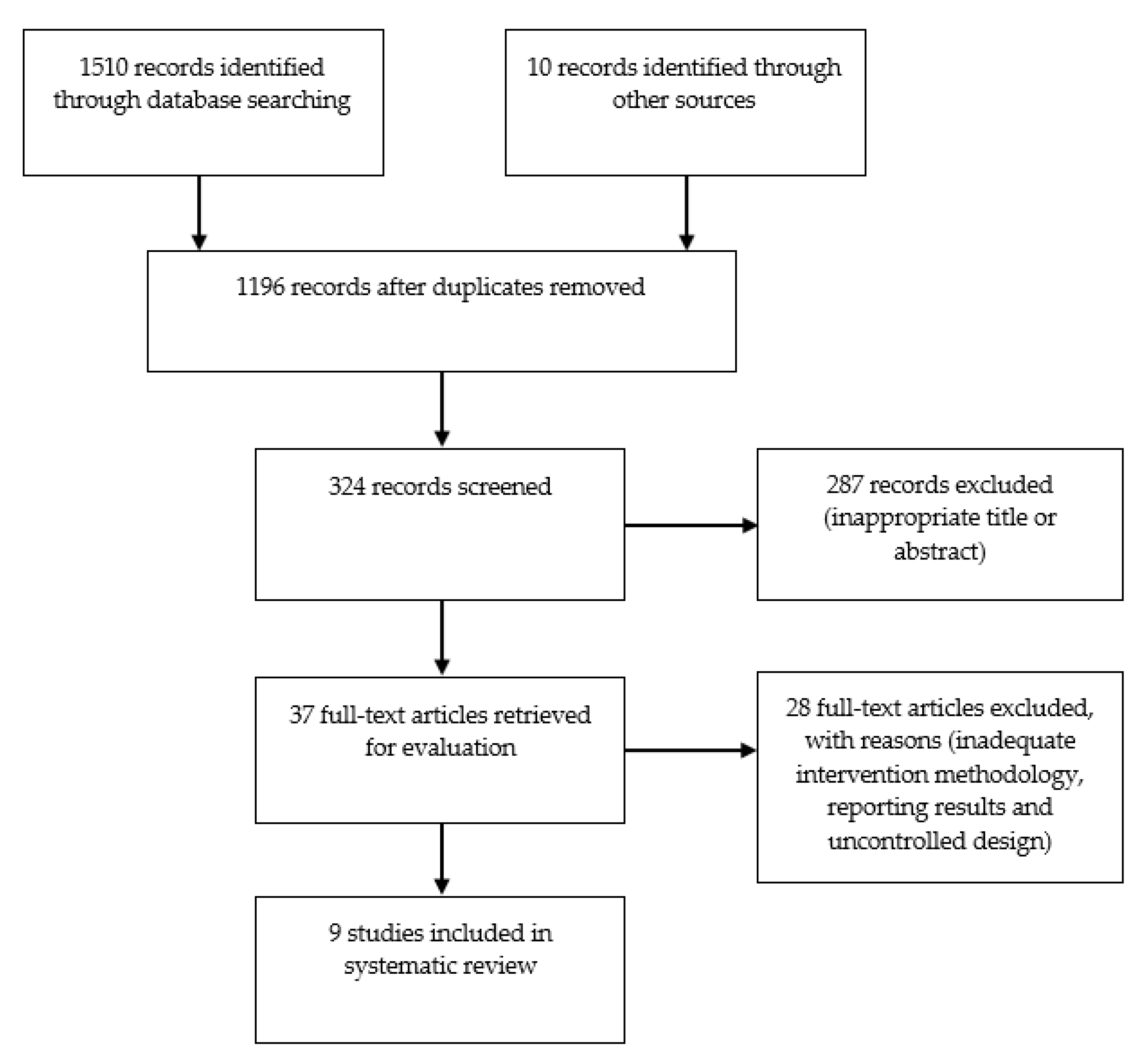
3. Results
3.1. Studies Included
3.2. Sample Size and Recruitment
3.3. Participants
3.4. Control Groups
3.5. Interventions
3.6. Adherence and Compliance
3.7. Safety and Adverse Events
3.8. Methodological Evaluation and Study Quality Results
3.9. Outcomes
3.9.1. CRF
3.9.2. Physical Activity (PA)
3.9.3. Fatigue and HRQOL
3.9.4. Other Secondary Outcomes
4. Discussion
4.1. Limitations
4.2. Future Directions
- define the outcomes and evaluate the effect relevant to the phase of cancer rehabilitation,
- establish a timeframe to define the phase of cancer rehabilitation,
- determine relevant measurements of outcomes for different cancer populations, and
- conduct further pilot studies in understudied cancer populations to ensure the feasibility of interventions and data analysis for future research, e.g., sample size determination.
- specified eligibility criteria (clearly stated and fulfilled eligibility criteria),
- unbiased randomization with description (a description of the randomization method used to allocate patients into study groups should be provided),
- allocation concealment,
- blinding of the assessor (for at least one primary outcome), and
- intention-to-treat analysis.
- specifications of the time range between cancer treatment completion and study enrolment,
- participation rate,
- limitations on recruitment flow and exercise intervention implementation,
- adherence to exercise intervention and compliance with the exercise prescription,
- adverse events (any serious medical events, deaths, and hospitalizations),
- activity monitoring in control groups,
- exercise volume and energy expenditure (exercise session and intervention duration, session frequency, and exercise intensity and modality),
- between-group statistical comparisons, and
- findings from all analyses conducted including effect sizes.
5. Conclusions
Author Contributions
Funding
Data Availability Statement
Conflicts of Interest
Appendix A
| Study | Study Quality (Max. 5) | Study Reporting (Max. 10) | Total Score (Max. 15) |
|---|---|---|---|
| McNeil (2019) Canada [18] | 2 | 6 | 8 |
| Alibhai (2019) Canada [19] | 5 | 7 | 12 |
| Gehring (2018) Netherlands [20] | 5 | 6 | 11 |
| Lahart (2017) UK [21] | 4 | 4 | 8 |
| Hvid (2016) Denmark [22] | 3 | 7 | 10 |
| Cornette (2016) France [23] | 2 | 9 | 11 |
| van Waart (2015) Netherlands [24] | 3 | 6 | 9 |
| Husebo (2014) Norway [25] | 4 | 7 | 11 |
| Pinto (2013) USA [26] | 3 | 8 | 11 |
| Mean | 3.4 | 6.6 | 10.1 |
| Range | 2–5 | 4–7 | 8–12 |
References
- Sung, H.; Ferlay, J.; Siegel, R.L.; Laversanne, M.; Soerjomataram, I.; Jemal, A.; Bray, F. Global Cancer Statistics 2020: GLOBOCAN Estimates of Incidence and Mortality Worldwide for 36 Cancers in 185 Countries. CA Cancer J. Clin. 2020, 1–41. [Google Scholar] [CrossRef]
- Shapiro, C.L. Cancer survivorship. N. Engl. J. Med. 2018, 379, 2438–2450. [Google Scholar] [CrossRef]
- Bower, J.E. Cancer-related fatigue—Mechanisms, risk factors, and treatments. Nat. Rev. Clin. Oncol. 2014, 11, 597–609. [Google Scholar] [CrossRef] [PubMed]
- Vejpongsa, P.; Yeh, E.T. Prevention of anthracycline-induced cardiotoxicity: Challenges and opportunities. J. Am. Coll. Cardiol. 2014, 64, 938–945. [Google Scholar] [CrossRef]
- Awad, S.; Tan, B.H.; Cui, H.; Bhalla, A.; Fearon, K.C.H.; Parsons, S.L.; Catton, J.A.; Lobo, D.N. Marked changes in body composition following neoadjuvant chemotherapy for oesophagogastric cancer. Clin. Nutr. 2012, 31, 74–77. [Google Scholar] [CrossRef] [PubMed]
- Curigliano, G.; Mayer, E.L.; Burstein, H.J.; Winer, E.P.; Goldhirsch, A. Cardiac toxicity from systemic cancer therapy: A comprehensive review. Prog. Cardiovasc. Dis. 2010, 53, 94–104. [Google Scholar] [CrossRef]
- Phillips, J.L.; Currow, D.C. Cancer as a chronic disease. Collegian 2010, 17, 47–50. [Google Scholar] [CrossRef] [PubMed]
- Schmitz, K.H.; Campbell, A.M.; Stuiver, M.M.; Pinto, B.M.; Schwartz, A.L.; Morris, G.S.; Ligibel, J.A.; Cheville, A.; Galvão, D.A.; Alfano, C.M. Exercise is medicine in oncology: Engaging clinicians to help patients move through cancer. CA. Cancer J. Clin. 2019, 69, 468–484. [Google Scholar] [CrossRef] [PubMed]
- Spence, R.R.; Heesch, K.C.; Brown, W.J. Exercise and cancer rehabilitation: A systematic review. Cancer Treat. Rev. 2010, 36, 185–194. [Google Scholar] [CrossRef] [PubMed]
- Schmitz, K.H.; Holtzman, J.; Courneya, K.S.; Mâsse, L.C.; Duval, S.; Kane, R. Controlled physical activity trials in cancer survivors: A systematic review and meta-analysis. Cancer Epidemiol. Biomark. Prev. 2005, 14, 1588–1595. [Google Scholar] [CrossRef]
- Kruijsen-Jaarsma, M.; Revesz, D.; Bierings, M.B.; Buffart, L.M.; Takken, T. Effects of exercise on immune function in patients with cancer: A systematic review. Exerc. Imunol. Rev. 2013, 19, 120–143. [Google Scholar]
- Stubblefield, M.D. The underutilization of rehabilitation to treat physical impairments in breast cancer survivors. PM R 2017, 9, 317–323. [Google Scholar] [CrossRef]
- IJsbrandy, C.; Hermens, R.P.M.; Boerboom, L.W.M.; Gerritsen, W.R.; van Harten, W.H.; Ottevanger, P.B. Implementing physical activity programs for patients with cancer in current practice: Patients’ experienced barriers and facilitators. J. Cancer Surviv. 2019, 13, 703–712. [Google Scholar] [CrossRef]
- Rossi, A.; Garber, C.E.; Ortiz, M.; Shankar, V.; Goldberg, G.L.; Nevadunsky, N.S. Feasibility of a physical activity intervention for obese, socioculturally diverse endometrial cancer survivors. Gynecol. Oncol. 2016, 142, 304–310. [Google Scholar] [CrossRef] [PubMed]
- Stefani, L.; Klika, R.; Mascherini, G.; Mazzoni, F.; Lunghi, A.; Petri, C.; Petreni, P.; Di Costano, F.; Maffulli, N.; Galanti, G. Effects of a home-based exercise rehabilitation program for cancer survivors. J. Sports Med. Phys. Fitness 2019, 59, 846–852. [Google Scholar] [CrossRef]
- Scherrenberg, M.; Wilhelm, M.; Hansen, D.; Völler, H.; Cornelissen, V.; Frederix, I.; Kemps, H.; Dendale, P. The future is now: A call for action for cardiac telerehabilitation in the COVID-19 pandemic from the secondary prevention and rehabilitation section of the European Association of Preventive Cardiology. Eur. J. Prev. Cardiol. 2020, 27, 1–21. [Google Scholar] [CrossRef]
- Smart, N.A.; Waldron, M.; Ismail, H.; Giallauria, F.; Vigorito, C.; Cornelissen, V.; Dieberg, G. Validation of a new tool for the assessment of study quality and reporting in exercise training studies. Int. J. Evid. Based Healthc. 2015, 13, 9–18. [Google Scholar] [CrossRef] [PubMed]
- McNeil, J.; Brenner, D.R.; Stone, C.R.; O’Reilly, R.; Ruan, Y.; Vallance, J.K.; Courneya, K.S.; Thorpe, K.E.; Klein, D.J.; Friedenreich, C.M. Activity Tracker to Prescribe Various Exercise Intensities in Breast Cancer Survivors. Med. Sci. Sports Exerc. 2019, 51, 930–940. [Google Scholar] [CrossRef] [PubMed]
- Alibhai, S.M.H.; Santa Mina, D.; Ritvo, P.; Tomlinson, G.; Sabiston, C.; Krahn, M.; Durbano, S.; Matthew, A.; Warde, P.; O’Neill, M.; et al. A phase II randomized controlled trial of three exercise delivery methods in men with prostate cancer on androgen deprivation therapy. BMC Cancer 2019, 19, 2. [Google Scholar] [CrossRef]
- Gehring, K.; Kloek, C.J.; Aaronson, N.K.; Janssen, K.W.; Jones, L.W.; Sitskoorn, M.M.; Stuiver, M.M. Feasibility of a home-based exercise intervention with remote guidance for patients with stable grade II and III gliomas: A pilot randomized controlled trial. Clin. Rehabil. 2018, 32, 352–366. [Google Scholar] [CrossRef]
- Møller, T.; Andersen, C.; Lillelund, C.; Bloomquist, K.; Christensen, K.B.; Ejlertsen, B.; Tuxen, M.; Oturai, P.; Breitenstein, U.; Kolind, C.; et al. Physical deterioration and adaptive recovery in physically inactive breast cancer patients during adjuvant chemotherapy: A randomised controlled trial. Sci. Rep. 2020, 10, 9710. [Google Scholar] [CrossRef]
- Hvid, T.; Lindegaard, B.; Winding, K.; Iversen, P.; Brasso, K.; Solomon, T.P.; Pedersen, B.K.; Hojman, P. Effect of a 2-year home-based endurance training intervention on physiological function and PSA doubling time in prostate cancer patients. Cancer Causes Control 2016, 27, 165–174. [Google Scholar] [CrossRef]
- Cornette, T.; Vincent, F.; Mandigout, S.; Antonini, M.T.; Leobon, S.; Labrunie, A.; Venat, L.; Lavau-Denes, S.; Tubiana-Mathieu, N. Effects of home-based exercise training on VO2 in breast cancer patients under adjuvant or neoadjuvant chemotherapy (SAPA): A randomized controlled trial. Eur. J. Phys. Rehabil. Med. 2016, 52, 223–232. [Google Scholar]
- Van Waart, H.; Stuiver, M.M.; van Harten, W.H.; Geleijn, E.; Kieffer, J.M.; Buffart, L.M.; de Maaker-Berkhof, M.; Boven, E.; Schrama, J.; Geenen, M.M.; et al. Effect of Low-Intensity Physical Activity and Moderate to High-Intensity Physical Exercise During Adjuvant Chemotherapy on Physical Fitness, Fatigue, and Chemotherapy Completion Rates: Results of the PACES Randomized Clinical Trial. J. Clin. Oncol. 2015, 33, 1918–1927. [Google Scholar] [CrossRef]
- Husebø, A.M.; Dyrstad, S.M.; Mjaaland, I.; Søreide, J.A.; Bru, E. Effects of scheduled exercise on cancer-related fatigue in women with early breast cancer. Sci. World J. 2014, 15, 1–9. [Google Scholar] [CrossRef]
- Pinto, B.M.; Papandonatos, G.D.; Goldstein, M.G.; Marcus, B.H.; Farrell, N. Home-based physical activity intervention for colorectal cancer survivors. Psychooncology 2013, 22, 54–64. [Google Scholar] [CrossRef] [PubMed]
- Stevinson, C.; Lawlor, D.A.; Fox, K.R. Exercise interventions for cancer patients: Systematic review of controlled trials. Cancer Causes Control 2004, 15, 1035–1056. [Google Scholar] [CrossRef] [PubMed]
- Maltser, S.; Cristian, A.; Silver, J.K.; Morris, G.S.; Stout, N.L. A Focused Review of Safety Considerations in Cancer Rehabilitation. PM & R 2017, 9, 415–428. [Google Scholar] [CrossRef]
- Kampshoff, C.S.; Jansen, F.; van Mechelen, W.; May, A.M.; Brug, J.; Chinapaw, M.J.; Buffart, L.M. Determinants of exercise adherence and maintenance among cancer survivors: A systematic review. Int. J. Behav. Nutr. Phys. Act. 2014, 11, 80. [Google Scholar] [CrossRef] [PubMed]
- Courneya, K.S.; Segal, R.J.; Gelmon, K.; Reid, R.D.; Mackey, J.R.; Friedenreich, C.M.; Proulx, C.; Lane, K.; Ladha, A.B.; Vallance, J.K.; et al. Predictors of supervised exercise adherence during breast cancer chemotherapy. Med. Sci. Sports Exerc. 2008, 40, 1180–1187. [Google Scholar] [CrossRef] [PubMed]
- Kampshoff, C.S.; van Mechelen, W.; Schep, G.; Nijziel, M.R.; Witlox, L.; Bosman, L.; Chinapaw, M.J.; Brug, J.; Buffart, L.M. Participation in and adherence to physical exercise after completion of primary cancer treatment. Int. J. Behav. Nutr. Phys. Act. 2016, 13, 100. [Google Scholar] [CrossRef] [PubMed]
- Scott, J.M.; Zabor, E.C.; Schwitzer, E.; Koelwyn, G.J.; Adams, S.C.; Nilsen, T.S.; Moskowitz, C.S.; Matsoukas, K.; Iyengar, N.M.; Dang, C.T.; et al. Efficacy of Exercise Therapy on Cardiorespiratory Fitness in Patients with Cancer: A Systematic Review and Meta-Analysis. J. Clin. Oncol. 2018, 36, 2297–2305. [Google Scholar] [CrossRef]
- Gilchrist, S.C.; Barac, A.; Ades, P.A.; Alfano, C.M.; Franklin, B.A.; Jones, L.W.; La Gerche, A.; Ligibel, J.A.; Lopez, G.; Madan, K. American Heart Association Exercise, Cardiac Rehabilitation, and Secondary Prevention Committee of the Council on Clinical Cardiology; Council on Cardiovascular and Stroke Nursing; and Council on Peripheral Vascular Disease. Cardio-Oncology Rehabilitation to Manage Cardiovascular Outcomes in Cancer Patients and Survivors: A Scientific Statement from the American Heart Association. Circulation 2019, 139, 997–1012. [Google Scholar] [CrossRef]
- Brown, J.C.; Troxel, A.B.; Ky, B.; Damjanov, N.; Zemel, B.S.; Rickels, M.R.; Rhim, A.D.; Rustgi, A.K.; Courneya, K.S.; Schmitz, K.H. Dose-response Effects of Aerobic Exercise Among Colon Cancer Survivors: A Randomized Phase II Trial. Clin. Colorectal. Cancer. 2018, 17, 32–40. [Google Scholar] [CrossRef] [PubMed]
- Cormie, P.; Zopf, E.M.; Zhang, X.; Schmitz, K.H. The Impact of Exercise on Cancer Mortality, Recurrence, and Treatment-Related Adverse Effects. Epidemiol. Rev. 2017, 39, 71–92. [Google Scholar] [CrossRef] [PubMed]
- Schneider, C.M.; Hsieh, C.C.; Sprod, L.K.; Carter, S.D.; Hayward, R. Effects of supervised exercise training on cardiopulmonary function and fatigue in breast cancer survivors during and after treatment. Cancer 2007, 110, 918–925. [Google Scholar] [CrossRef] [PubMed]
- Brown, J.C.; Huedo-Medina, T.B.; Pescatello., L.S.; Pescatello, S.M.; Ferrer, R.A.; Johnson, B.T. Efficacy of exercise interventions in modulating cancer-related fatigue among adult cancer survivors: A meta-analysis. Cancer Epidemiol. Biomark. Prev. 2011, 20, 123–133. [Google Scholar] [CrossRef]
- Irwin, M.L. Physical activity interventions for cancer survivors. Br. J. Sports Med. 2009, 43, 32–38. [Google Scholar] [CrossRef] [PubMed]
- Maginador, G.; Lixandrão, M.E.; Bortolozo, H.I.; Vechin, F.C.; Sarian, L.O.; Derchain, S.; Telles, G.D.; Zopf, E.; Ugrinowitsch, C.; Conceição, M.S. Aerobic Exercise-Induced Changes in Cardiorespiratory Fitness in Breast Cancer Patients Receiving Chemotherapy: A Systematic Review and Meta-Analysis. Cancers 2020, 12, 2240. [Google Scholar] [CrossRef]
- Van Waart, H.; Stuiver, M.M.; van Harten, W.H.; Sonke, G.S.; Aaronson, N.K. Design of the Physical exercise during Adjuvant Chemotherapy Effectiveness Study (PACES): A randomized controlled trial to evaluate effectiveness and cost-effectiveness of physical exercise in improving physical fitness and reducing fatigue. BMC Cancer 2010, 10, 673. [Google Scholar] [CrossRef]
- Angadi, S.S.; Mookadam, F.; Lee, C.D.; Tucker, W.J.; Haykowsky, M.J.; Gaesser, G.A. High-intensity interval training vs. moderate-intensity continuous exercise training in heart failure with preserved ejection fraction: A pilot study. J. Appl. Physiol. 2015, 119, 753–758. [Google Scholar] [CrossRef]
- Papathanasiou, J.V.; Petrov, I.; Tokmakova, M.P.; Dimitrova, D.D.; Spasov, L.; Dzhafer, N.S.; Tsekoura, D.; Dionyssiotis, Y.; Ferreira, A.S.; Lopes, A.J.; et al. Group-based cardiac rehabilitation interventions. A challenge for physical and rehabilitation medicine physicians: A randomized controlled trial. Eur. J. Phys. Rehabil. Med. 2020, 56, 479–488. [Google Scholar] [CrossRef]
- Cardinale, D.; Iacopo, F.; Cipolla, C.M. Cardiotoxicity of Anthracyclines. Front. Cardiovasc. Med. 2020, 7, 26. [Google Scholar] [CrossRef]
- Mercurio, V.; Pirozzi, F.; Lazzarinim, E.; Marone, G.; Rizzo, P.; Agnetti, G.; Tocchetti, C.G.; Ghigo, A.; Ameri, P. Models of Heart Failure Based on the Cardiotoxicity of Anticancer Drugs. J. Card. Fail. 2016, 22, 449–458. [Google Scholar] [CrossRef] [PubMed]
- Gielen, S.; Laughlin, M.H.; O’Conner, C.; Duncker, D.J. Exercise training in patients with heart disease: Review of beneficial effects and clinical recommendations. Prog. Cardiovasc. Dis. 2015, 57, 347–355. [Google Scholar] [CrossRef] [PubMed]
- Peçanha, T.; Goessler, K.F.; Roschel, H.; Gualano, B. Social isolation during the COVID-19 pandemic can increase physical inactivity and the global burden of cardiovascular disease. Am. J. Physiol. Heart Circ. Physiol. 2020, 318, 1441–1446. [Google Scholar] [CrossRef] [PubMed]
- Besnier, F.; Gayda, M.; Nigam, A.; Juneau, M.; Bherer, L. Cardiac Rehabilitation During Quarantine in COVID-19 Pandemic: Challenges for Center-Based Programs. Arch. Phys. Med. Rehabil. 2020, 101, 1835–1838. [Google Scholar] [CrossRef] [PubMed]
- Peretti, A.; Amenta, F.; Tayebati, S.K.; Nittari, G.; Mahdi, S.S. Telerehabilitation: Review of the State-of-the-Art and Areas of Application. JMIR Rehabil. Assist. Technol. 2017, 4, e7. [Google Scholar] [CrossRef]
- Aragaki, D.; Luo, J.; Weiner, E.; Zhang, G.; Darvish, B. Cardiopulmonary Telerehabilitation. Phys. Med. Rehabil. Clin. N. Am. 2021, in press. [Google Scholar] [CrossRef] [PubMed]
- Etiwy, M.; Akhrass, Z.; Gillinov, L.; Alashi, A.; Wang, R.; Blackburn, G.; Gillinov, S.M.; Phelan, D.; Gillinov, A.M.; Houghtaling, P.L.; et al. Accuracy of wearable heart rate monitors in cardiac rehabilitation. Cardiovasc. Diagn. Ther. 2019, 9, 262–271. [Google Scholar] [CrossRef]
- Gilgen-Ammann, R.; Schweizer, T.; Wyss, T. RR interval signal quality of a heart rate monitor and an ECG Holter at rest and during exercise. Eur. J. Appl. Physiol. 2019, 119, 1525–1532. [Google Scholar] [CrossRef] [PubMed]
- Liu, N.; Huang, R.; Baldacchino, T.; Sud, A.; Sud, K.; Khadra, M.; Kim, J. Telehealth for Noncritical Patients with Chronic Diseases During the COVID-19 Pandemic. J. Med. Internet Res. 2020, 22, e19493. [Google Scholar] [CrossRef] [PubMed]
| Study | Treatment | Cancer | Study | n | Sex | Age | Duration (Week) | Exercise Program | Intensity | Frequency | Monitoring/Feedback | Outcomes Primary/Secondary |
|---|---|---|---|---|---|---|---|---|---|---|---|---|
| McNeil (2019) [18] | Post CT, RT or surgery | BC | RCT (1:1:1) | 45 | W | 58 | 12 | Aerobic exercise; low-intensity group: 300 min/week, high-intensity group: 150 min/week | Low-intensity group: 40–59% HRR; high-intensity group: 60–80% HRR | NS | HR monitor; exercise diary; phone call or e-mail 1/3 weeks | PA/CRF, body composition—anthropometry |
| Alibhai (2019) [19] | During ADT | PC | RCT (1:1:1) | 59 | M | 70 | 24 | Aerobic exercise + resistance and flexibility exercises; 150 min/week | 60–70% HRR, 3–6 RPE (10 on Borg scale) | 4–5/week | HR monitor; phone call 1/week | Feasibility/CRF, fatigue, HRQOL, strength, satisfaction, cost-effectivity |
| Gehring (2018) [20] | Post CT, RT or surgery | Gliom | RCT (2:1) | 34 | M, W | 48 | 24 | Aerobic exercise | 60–85% HRmax | 3/week | HR monitor; training log online; e-mail 1/week | CRF/PA, body composition—anthropometry, satisfaction |
| Lahart (2017) [21] | Post ADT or surgery | BC | RCT (1:1) | 80 | W | 52 | 24 | Aerobic exercise; 30 min/session | NS | Gradually 3–7/week | PA diary recommended; phone call 1/4 weeks | CRF/PA, body composition—anthropometry |
| Hvid (2016) [22] | Post radical prostatectomy | PC | RCT (2:1) | 25 | M | 70 | 96 | Aerobic exercise; 45 min/session | 60–65% VO2max | 3/week | HR monitor; training log; in person control visit 1/4 weeks | NS/CRF, body composition—anthropometry, glucose |
| Cornette (2016) [23] | During ADT | BC | RCT (1:1) | 44 | W | 51 | 27 | Aerobic exercise, 30–50 min/session + resistance exercise; | 1-VT | 3/week | HR monitor; exercise diary; phone call 1/week | CRF/PA, fatigue, HRQOL, strength |
| Van Waart (2015) [24] | During ADT | BC + Colon | RCT (1:1:1) | 230 | W | 51 | NS | Aerobic exercise, self-managed PA ≥ 30 min/session | 12–14 RPE (6–20 Borg scale) | 5/week | Activity diary; in-person control visit 1/CT cycle | CRF, fatigue, strength/PA, HRQOL |
| Husebo (2014) [25] | During ADT | BC | RCT (1:1) | 67 | W | 52 | 12 | Aerobic exercise, 30 min/session + resistance exercise | At least 2 level of intensity out of 4 | 7/week | Exercise diary; phone call 1 /2 week | Fatigue, PA/CRF |
| Pinto (2013) [26] | Postsurgery, CT, or RT | CRC | RCT (1:1) | 46 | M, W | 57 | 12 | Aerobic exercise gradually to 30 min/session | 64–76% HRmax | Gradually 2–5/week | Accelerometer; home PA log; 1 phone call/week | PA/CRF, fatigue, HRQOL |
| Study | Randomization Process | Unbiased Randomization | Blinding of Assessors | Intention-to-Treat Analysis | Adverse Events | Adherence with Exercise Protocol |
|---|---|---|---|---|---|---|
| McNeil (2019) [18] | NR | NR | NR | Yes | NR | 111% of prescribed PA in higher-intensity group (mean 166 min/week), 309% of prescribed PA in lower-intensity group (mean 928 min/week) |
| Alibhai (2019) [19] | Stratified by duration of prior ADT use | Yes | Yes | No 14 dropouts | Two adverse events grade 2 (primarily musculoskeletal), no events grade 3 or higher | 31% (self-reported questionnaire) or 50% (accelerometry) of participants achieved 150 min/week of prescribed PA |
| Gehring (2018) [20] | Stratified by age, education, tumor grade, disease duration, relative VO2 classification, and performance on the letter digit substitution task | NR | Yes | Yes | No adverse events (one aggravation of pre-existing osteoarthritis-related knee pain at 6th month of HB) | Participants completed 79% of prescribed sessions (mean 2.4 sessions/week) |
| Lahart (2017) [21] | Stratification based on ADT | Yes | No | Yes | NR | NR |
| Hvid (2016) [22] | Simple adaptive randomization procedure | NR | No | No 6 dropouts | NR | Participants completed 88% of prescribed sessions |
| Cornette (2016) [23] | Without stratification | NR | NR | Yes | No | Reported only in 14/20 participants, participants completed 88% of prescribed sessions (109% aerobic exercise, 46% resistance exercise) |
| van Waart (2015) [24] | Stratified by age, primary diagnosis, treating hospital, and ADT use | NR | NR | Yes | NR | 55% of participants followed prescribed daily PA levels at least 75% of HB program |
| Husebo (2014) [25] | NR | Yes | NR | No 7 dropouts | 1 knee discomfort, 1 syncope | 17% of participants achieved 210 min/week of prescribed PA (58% met the general recommendations of 150 min/week) |
| Pinto (2013) [26] | Stratified by age, cancer type, and sex | NR | Yes | No 3 dropouts | NR | 65% of participants achieved 150 min/week of prescribed PA |
| Benefits | Limitations |
|---|---|
| Higher protection from infection | Lack of exercise supervision |
| Independence in exercise planning | Less face-to-face contact |
| Less time and/or travel barriers | Lack of social interaction |
| Integration into daily PA | ICT literacy |
| Combination with tele-monitoring and/or counselling | Exercise data integration into medical records |
| Higher privacy | Lack of legal clarity and data protection |
| Lower costs | Lack of published guidelines |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Batalik, L.; Winnige, P.; Dosbaba, F.; Vlazna, D.; Janikova, A. Home-Based Aerobic and Resistance Exercise Interventions in Cancer Patients and Survivors: A Systematic Review. Cancers 2021, 13, 1915. https://doi.org/10.3390/cancers13081915
Batalik L, Winnige P, Dosbaba F, Vlazna D, Janikova A. Home-Based Aerobic and Resistance Exercise Interventions in Cancer Patients and Survivors: A Systematic Review. Cancers. 2021; 13(8):1915. https://doi.org/10.3390/cancers13081915
Chicago/Turabian StyleBatalik, Ladislav, Petr Winnige, Filip Dosbaba, Daniela Vlazna, and Andrea Janikova. 2021. "Home-Based Aerobic and Resistance Exercise Interventions in Cancer Patients and Survivors: A Systematic Review" Cancers 13, no. 8: 1915. https://doi.org/10.3390/cancers13081915
APA StyleBatalik, L., Winnige, P., Dosbaba, F., Vlazna, D., & Janikova, A. (2021). Home-Based Aerobic and Resistance Exercise Interventions in Cancer Patients and Survivors: A Systematic Review. Cancers, 13(8), 1915. https://doi.org/10.3390/cancers13081915







