Radiotherapy with Intensity-Modulated (IMRT) Techniques in the Treatment of Anal Carcinoma (RAINSTORM): A Multicenter Study on Behalf of AIRO (Italian Association of Radiotherapy and Clinical Oncology) Gastrointestinal Study Group
Simple Summary
Abstract
1. Introduction
2. Results
2.1. Patients and Treatment Characteristics
2.2. Treatment Compliance and Toxicity
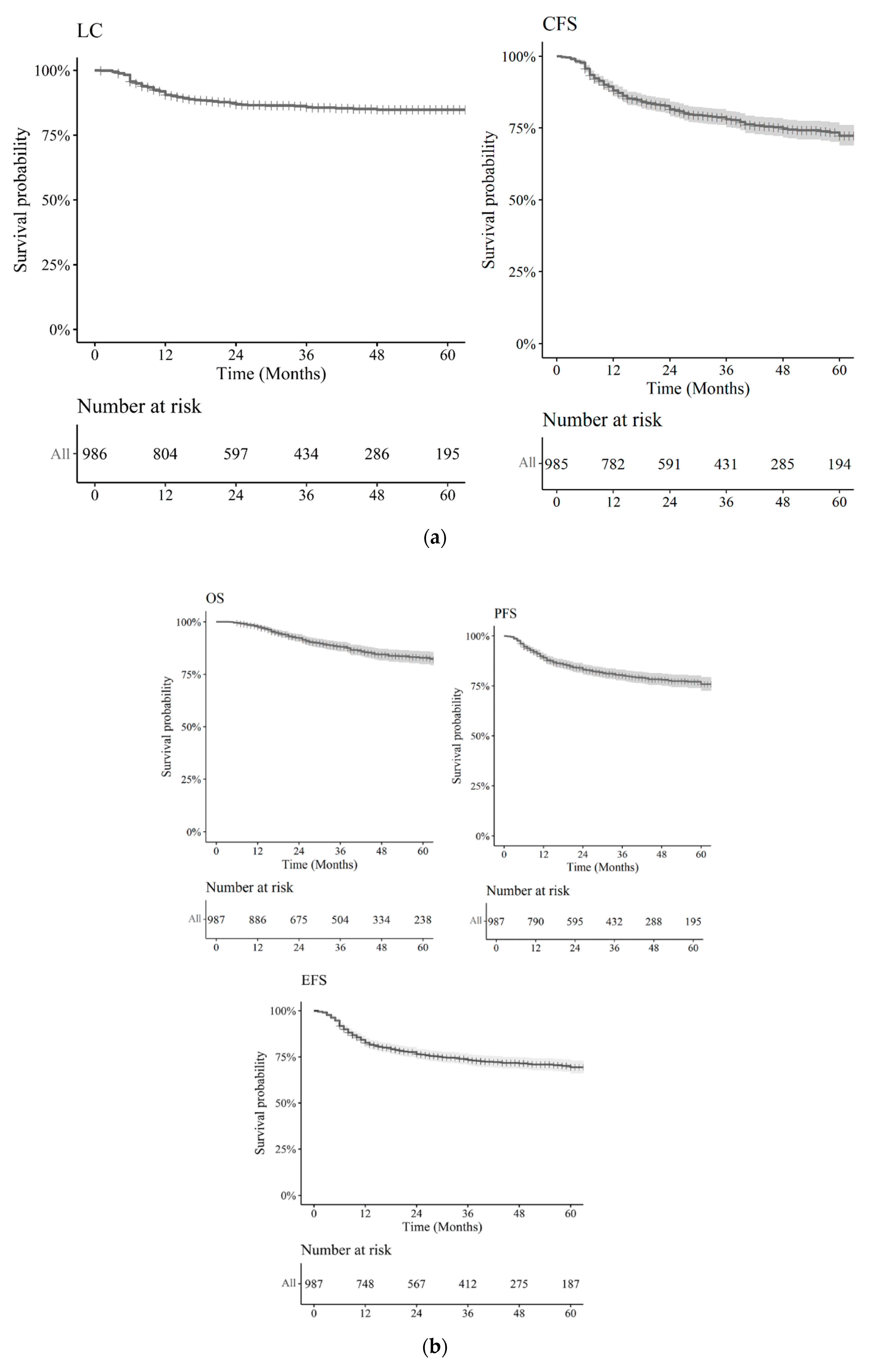
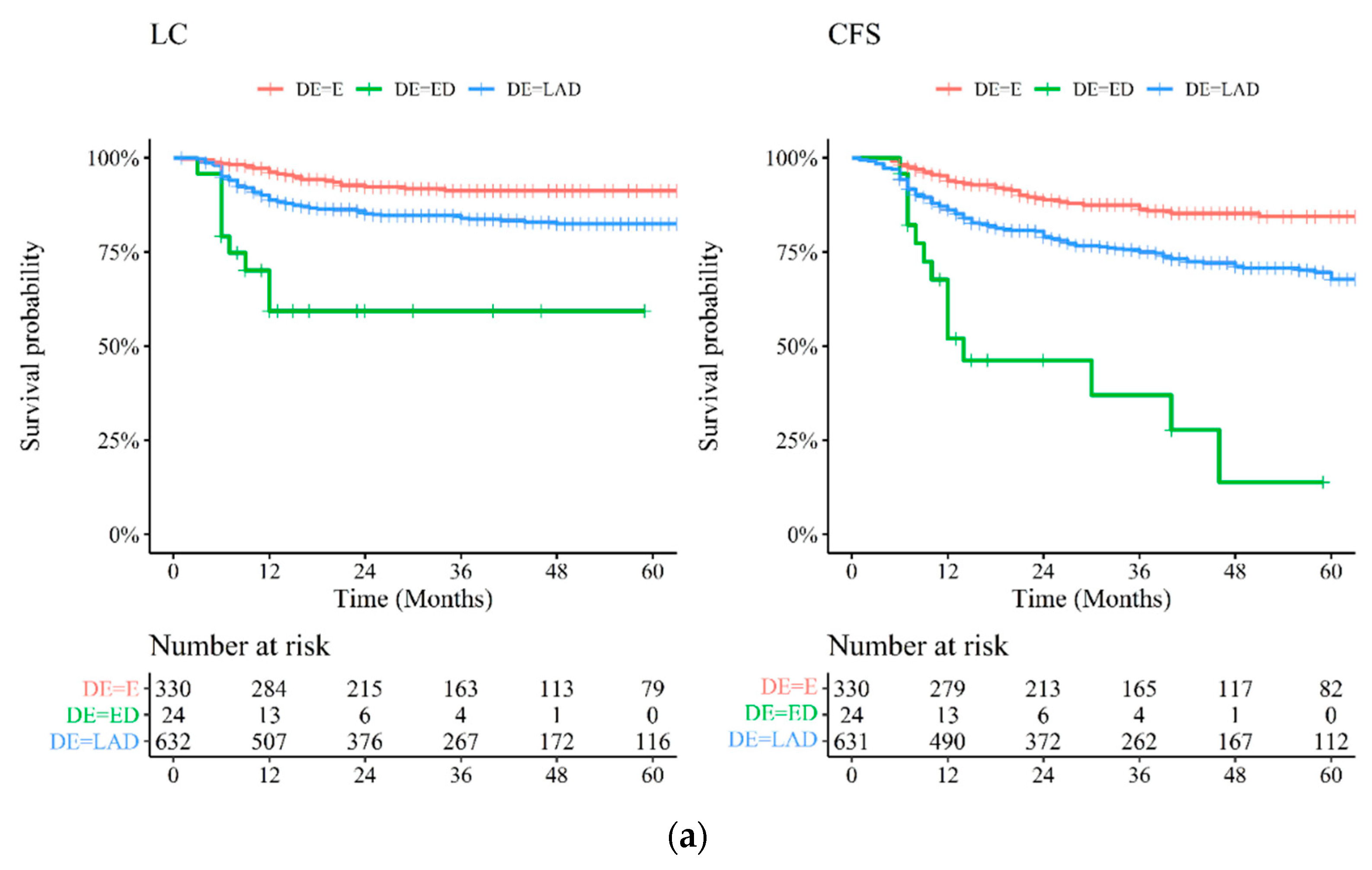
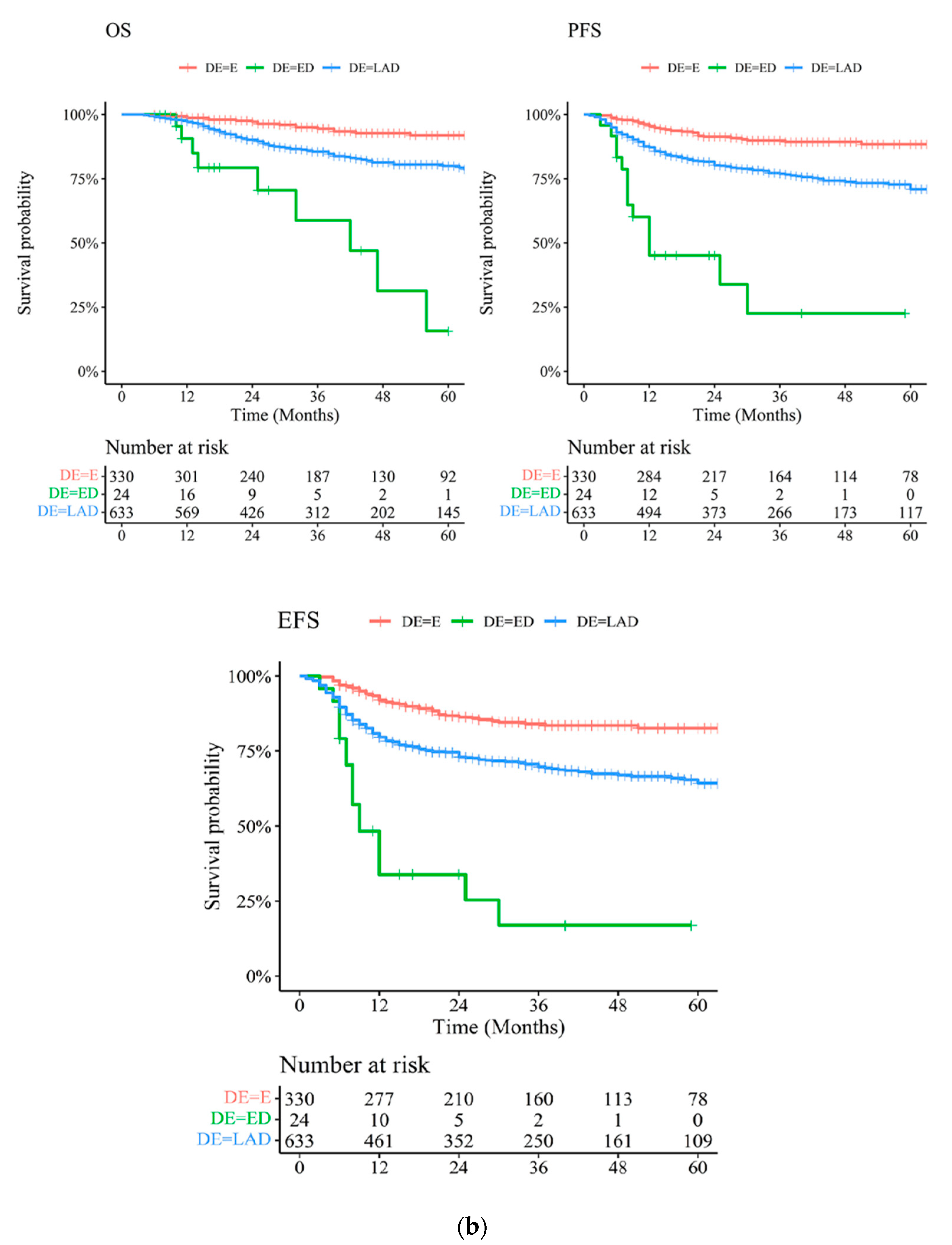
2.3. Treatment Response and Clinical Outcomes
2.4. Univariate and Multivariate Analyses
3. Discussion
4. Materials and Methods
4.1. Treatment Characteristics
4.2. Statistical Analysis
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Nigro, N.D.; Seydel, H.G.; Considine, B.; Vaitkevicius, V.K.; Leichman, L.; Kinzie, J.J. Combined preoperative radiation and chemo-therapy for squamous cell carcinoma of the anal canal. Cancer 1983, 51, 1826–1829. [Google Scholar] [CrossRef]
- Cummings, B.; Keane, T.; O’Sullivan, B.; Wong, C.; Catton, C. Epidermoid anal cancer: Treatment by radiation alone or by radiation and 5-fluorouracil with and without mitomycin C. Int. J. Radiat. Oncol. 1991, 21, 1115–1125. [Google Scholar] [CrossRef]
- Arnott, S.J.; Cunningham, D.; Gallagher, J.; Gray, R.; Hardcastle, J.; Houghton, J.; James, R.D.; Lennon, T.A.; Meadows, H.M.; Mossman, J.; et al. Epidermoid anal cancer: Results from the UKCCCR randomised trial of radiotherapy alone versus radiotherapy, 5-fluorouracil, and mitomycin. UKCCCR Anal Cancer Trial Working Party. UK Co-ordinating Committee on Cancer Research. Lancet 1996, 348, 1049–1054. [Google Scholar]
- Flam, M.; John, M.; Pajak, T.F.; Petrelli, N.; Myerson, R.; Doggett, S.; Quivey, J.; Rotman, M.; Kerman, H.; Coia, L.; et al. Role of mitomycin in combination with fluorouracil and radiotherapy, and of salvage chemoradiation in the definitive nonsurgical treatment of epidermoid carcinoma of the anal canal: Results of a phase III randomized intergroup study. J. Clin. Oncol. 1996, 14, 2527–2539. [Google Scholar] [CrossRef] [PubMed]
- Bartelink, H.; Roelofsen, F.; Eschwege, F.; Rougier, P.; Bosset, J.F.; Gonzalez, D.G.; Peiffert, D.; Van Glabbeke, M.; Pierart, M. Concomitant radiotherapy and chemotherapy is superior to radiotherapy alone in the treatment of locally advanced anal cancer: Results of a phase III randomized trial of the European Organization for Research and Treatment of Cancer Radiotherapy and Gastrointestinal Cooperative Groups. J. Clin. Oncol. 1997, 15, 2040–2049. [Google Scholar] [CrossRef]
- Ajani, J.A.; Winter, K.A.; Gunderson, L.L.; Pedersen, J.; Benson, A.B.; Thomas, C.R.; Mayer, R.J.; Haddock, M.G.; Rich, T.A.; Willett, C. Fluorouracil, Mitomycin, and Radiotherapy vs Fluorouracil, Cisplatin, and Radiotherapy for Carcinoma of the Anal Canal: A randomized controlled trial. JAMA 2008, 299, 1914–1921. [Google Scholar] [CrossRef]
- Peiffert, D.; Tournier-Rangeard, L.; Gérard, J.-P.; Lemanski, C.; François, E.; Giovannini, M.; Cvitkovic, F.; Mirabel, X.; Bouché, O.; Luporsi, E.; et al. Induction Chemotherapy and Dose Intensification of the Radiation Boost in Locally Advanced Anal Canal Carcinoma: Final Analysis of the Randomized UNICANCER ACCORD 03 Trial. J. Clin. Oncol. 2012, 30, 1941–1948. [Google Scholar] [CrossRef]
- James, R.D.; Glynne-Jones, R.; Meadows, H.M.; Cunningham, D.; Myint, A.S.; Saunders, M.P.; Maughan, T.; McDonald, A.; Essapen, S.; Leslie, M.; et al. Mitomycin or cisplatin chemoradiation with or without maintenance chemotherapy for treatment of squamous-cell carcinoma of the anus (ACT II): A randomised, phase 3, open-label, 2 × 2 factorial trial. Lancet Oncol. 2013, 14, 516–524. [Google Scholar] [CrossRef]
- Gunderson, L.L.; Winter, K.A.; Ajani, J.A.; Pedersen, J.E.; Moughan, J.; Benson, A.B., 3rd; Thomas, C.R., Jr.; Mayer, R.J.; Haddock, M.G.; Rich, T.A.; et al. Long-Term Update of US GI Intergroup RTOG 98-11 Phase III Trial for Anal Carcinoma: Survival, Relapse, and Colostomy Failure with Concurrent Chemoradiation Involving Fluorouracil/Mitomycin Versus Fluorouracil/Cisplatin. J. Clin. Oncol. 2012, 30, 4344–4351. [Google Scholar] [CrossRef]
- Ben-Josef, E.; Moughan, J.; Ajani, J.A.; Flam, M.; Gunderson, L.; Pollock, J.; Myerson, R.; Anne, R.; Rosenthal, S.A.; Willett, C. Impact of Overall Treatment Time on Survival and Local Control in Patients with Anal Cancer: A Pooled Data Analysis of Radiation Therapy Oncology Group Trials 87-04 and 98-11. J. Clin. Oncol. 2010, 28, 5061–5066. [Google Scholar] [CrossRef]
- Glynne-Jones, R.; Sebag-Montefiore, D.; Adams, R.; McDonald, A.; Gollins, S.; James, R.; Northover, J.M.; Meadows, H.M.; Jitlal, M. “Mind the Gap”—The Impact of Variations in the Duration of the Treatment Gap and Overall Treatment Time in the First UK Anal Cancer Trial (ACT I). Int. J. Radiat. Oncol. Biol. Phys. 2011, 81, 1488–1494. [Google Scholar] [CrossRef]
- Han, K.; Cummings, B.J.; Dawson, L.A.; Ringash, J.; Krzyzanowska, M.K.; Moore, M.J.; Chen, E.X.; Easson, A.M.; Kassam, Z.; Cho, C.; et al. Prospective Evaluation of Acute Toxicity and Quality of Life After IMRT and Concurrent Chemotherapy for Anal Canal and Perianal Cancer. Int. J. Radiat. Oncol. Biol. Phys. 2014, 90, 587–594. [Google Scholar] [CrossRef]
- Mitchell, M.P.; Abboud, M.; Eng, C.; Beddar, A.S.; Krishnan, S.; Delclos, M.E.; Crane, C.H.; Das, P. Intensity-modulated Radiation Therapy with Concurrent Chemotherapy for Anal Cancer: Outcomes and toxicity. Am. J. Clin. Oncol. 2014, 37, 461–466. [Google Scholar] [CrossRef]
- Call, J.A.; Prendergast, B.M.; Jensen, L.G.; Ord, C.B.; Goodman, K.A.; Jacob, R.; Mell, L.K.; Thomas, C.R.; Jabbour, S.K.; Miller, R.C. Intensity-modulated Radiation Therapy for Anal Cancer: Results from a multi-institutional retrospective cohort study: Results from a multi-institutional retrospective cohort study. Am. J. Clin. Oncol. 2016, 39, 8–12. [Google Scholar] [CrossRef]
- Kachnic, L.A.; Winter, K.; Willett, C.G.; Myerson, R.J.; Goodyear, M.D.; Willins, J.; Esthappan, J.; Haddock, M.G.; Rotman, M.; Parikh, P.J.; et al. RTOG 0529: A Phase 2 Evaluation of Dose-Painted Intensity Modulated Radiation Therapy in Combination With 5-Fluorouracil and Mitomycin-C for the Reduction of Acute Morbidity in Carcinoma of the Anal Canal. Int. J. Radiat. Oncol. Biol. Phys. 2013, 86, 27–33. [Google Scholar] [CrossRef]
- Yates, A.; Carroll, S.; Kneebone, A.; Tse, R.; Horvath, L.; Byrne, C.; Solomon, M.; Hruby, G. Implementing Intensity-modulated Radiotherapy with Simultaneous Integrated Boost for Anal Cancer: 3 Year Outcomes at Two Sydney Institutions. Clin. Oncol. (R. Coll. Radiol.) 2015, 27, 700–707. [Google Scholar] [CrossRef]
- Mitra, D.; Hong, T.S.; Horick, N.; Rose, B.; Drapek, L.N.; Blaszkowsky, L.S.; Allen, J.N.; Kwak, E.L.; Murphy, J.E.; Clark, J.W.; et al. Long-term outcomes and toxicities of a large cohort of anal cancer patients treated with dose-painted IMRT per RTOG 0529. Adv. Radiat. Oncol. 2017, 2, 110–117. [Google Scholar] [CrossRef]
- Arcadipane, F.; Franco, P.; Ceccarelli, M.; Furfaro, G.; Rondi, N.; Trino, E.; Martini, S.; Iorio, G.C.; Mistrangelo, M.; Cassoni, P.; et al. Image-guided IMRT with simultaneous integrated boost as per RTOG 0529 for the treatment of anal cancer. Asia-Pac. J. Clin. Oncol. 2018, 14, 217–223. [Google Scholar] [CrossRef]
- Franco, P.; Mistrangelo, M.; Arcadipane, F.; Munoz, F.; Sciacero, P.; Spadi, R.; Migliaccio, F.; Angelini, V.; Bombaci, S.; Rondi, N.; et al. Intensity-Modulated Radiation Therapy with Simultaneous Integrated Boost Combined with Concurrent Chemotherapy for the Treatment of Anal Cancer Patients: 4-Year Results of a Consecutive Case Series. Cancer Investig. 2015, 33, 259–266. [Google Scholar] [CrossRef]
- Franco, P.; Arcadipane, F.; Ragona, R.; Mistrangelo, M.; Cassoni, P.; Rondi, N.; Morino, M.; Racca, P.; Ricardi, U. Locally Advanced (T3-T4 or N+) Anal Cancer Treated with Simultaneous Integrated Boost Radiotherapy and Concurrent Chemotherapy. Anticancer Res. 2016, 36, 2027–2032. [Google Scholar]
- De Bari, B.; Lestrade, L.; Franzetti-Pellanda, A.; Jumeau, R.; Biggiogero, M.; Kountouri, M.; Matzinger, O.; Miralbell, R.; Bourhis, J.; Ozsahin, M.; et al. Modern intensity-modulated radiotherapy with image guidance allows low toxicity rates and good local control in chemoradiotherapy for anal cancer patients. J. Cancer Res. Clin. Oncol. 2018, 144, 781–789. [Google Scholar] [CrossRef]
- Dell’Acqua, V.; Surgo, A.; Arculeo, S.; Zerella, M.A.; Bagnardi, V.; Frassoni, S.; Zampino, M.G.; Ravenda, P.S.; Rotundo, M.S.; Kraja, F.; et al. Intensity-modulated radiotherapy (IMRT) in the treatment of squamous cell anal canal cancer: Acute and early-late toxicity, outcome, and efficacy. Int. J. Color. Dis. 2020, 35, 685–694. [Google Scholar] [CrossRef]
- Glynne-Jones, R.; Nilsson, P.; Aschele, C.; Goh, V.; Peiffert, D.; Cervantes, A.; Arnold, D. Anal cancer: ESMO-ESSO-ESTRO clinical practice guidelines for diagnosis, treatment and follow-up. Eur. J. Surg. Oncol. (EJSO) 2014, 40, 1165–1176. [Google Scholar] [CrossRef] [PubMed]
- Myerson, R.J.; Garofalo, M.C.; El Naqa, I.; Abrams, R.A.; Apte, A.; Bosch, W.R.; Das, P.; Gunderson, L.L.; Hong, T.S.; Kim, J.J.; et al. Elective Clinical Target Volumes for Conformal Therapy in Anorectal Cancer: A Radiation Therapy Oncology Group Consensus Panel Contouring Atlas. Int. J. Radiat. Oncol. 2009, 74, 824–830. [Google Scholar] [CrossRef]
- Ng, M.; Leong, T.; Chander, S.; Chu, J.; Kneebone, A.; Carroll, S.; Wiltshire, K.; Ngan, S.; Kachnic, L. Australasian Gastrointestinal Trials Group (AGITG) Contouring Atlas and Planning Guidelines for Intensity-Modulated Radiotherapy in Anal Cancer. Int. J. Radiat. Oncol. 2012, 83, 1455–1462. [Google Scholar] [CrossRef] [PubMed]
- Muirhead, R.; Partridge, M.; Hawkins, M.A. A tumor control probability model for anal squamous cell carcinoma. Radiother. Oncol. 2015, 116, 192–196. [Google Scholar] [CrossRef] [PubMed]
- John, M.; Pajak, T.; Flam, M.; Hoffman, J.; Markoe, A.; Wolkov, H.; Paris, K. Dose escalation in chemoradiation for anal cancer: Preliminary results of RTOG 92-08. Cancer J. Sci. Am. 1996, 2, 205–211. [Google Scholar]
- Arcadipane, F.; Silvetti, P.; Olivero, F.; Gastino, A.; De Luca, V.; Mistrangelo, M.; Cassoni, P.; Racca, P.; Gallio, E.; Lesca, A.; et al. Bone Marrow-Sparing IMRT in Anal Cancer Patients Undergoing Concurrent Chemo-Radiation: Results of the First Phase of a Prospective Phase II Trial. Cancers 2020, 12, 3306. [Google Scholar] [CrossRef] [PubMed]
- Kim, J.J.; Tannock, I.F. Repopulation of cancer cells during therapy: An important cause of treatment failure. Nat. Rev. Cancer 2005, 5, 516–525. [Google Scholar] [CrossRef] [PubMed]
- Muirhead, R.; Adams, R.A.; Gilbert, D.C.; Harrison, M.; Glynne-Jones, R.; Sebag-Montefiore, D.; Hawkins, M. National Guidance for IMRT in Anal Cancer. 2016. Available online: http://analimrtguidance.co.uk/National-Guidance-IMRT-Anal-Cancer-V4-Jan17.pdf (accessed on 3 October 2018).
- Shakir, R.; Adams, R.; Cooper, R.; Downing, A.; Geh, I.; Gilbert, D.; Jacobs, C.; Jones, C.; Lorimer, C.; Namelo, W.C.; et al. Patterns and Predictors of Relapse Following Radical Chemoradiation Therapy Delivered Using Intensity Modulated Radiation Therapy With a Simultaneous Integrated Boost in Anal Squamous Cell Carcinoma. Int. J. Radiat. Oncol. 2020, 106, 329–339. [Google Scholar] [CrossRef]
- Sekhar, H.; Zwahlen, M.; Trelle, S.; Malcomson, L.; Kochhar, R.; Saunders, M.P.; Sperrin, M.; van Herk, M.; Sebag-Montefiore, D.; Egger, M.; et al. Nodal stage migration and prognosis in anal cancer: A systematic review, meta-regression, and simulation study. Lancet Oncol. 2017, 18, 1348–1359. [Google Scholar] [CrossRef]
- Cancer Therapy Evaluation Program (CTEP) of the National Cancer Institute (NCI). Common Terminology Criteria for Adverse Events (CTCAE); Version 4.0; v4.02; US Department of Health and Human Services, National Institutes of Health, National Cancer Institute, Bethesda: Rockville, MD, USA, 2009. [Google Scholar]
- Eisenhauer, E.A.; Therasse, P.; Bogaerts, J.; Schwartz, L.H.; Sargent, D.; Ford, R.; Dancey, J.; Arbuck, S.; Gwyther, S.; Mooney, M.; et al. New response evaluation criteria in solid tumours: Revised RECIST guideline (version 1.1). Eur. J. Cancer 2009, 45, 228–247. [Google Scholar] [CrossRef]
- Glynne-Jones, R.; Adams, R.; Lopes, A.; Meadows, H. Clinical endpoints in trials of chemoradiation for patients with anal cancer. Lancet Oncol. 2017, 18, e218–e227. [Google Scholar] [CrossRef]
- Fish, R.; Sanders, C.; Ryan, N.; Van Der Veer, S.; Renehan, A.G.; Williamson, P.R. Systematic review of outcome measures following chemoradiotherapy for the treatment of anal cancer (CORMAC). Colorectal Dis. 2018, 20, 371–382. [Google Scholar] [CrossRef] [PubMed]
| Patient and Tumor Characteristics | n = 987 | % | |
|---|---|---|---|
| Age = median 68.5 years (range: 54.00–80.00) | |||
| Gender | Male | 281 | 28.4 |
| Female | 706 | 71.5 | |
| ECOG Performance Status | 0 | 775 | 78.5 |
| 1 | 197 | 19.9 | |
| 2 | 15 | 1.5 | |
| HPV | Negative | 210 | 21.3 |
| Positive | 229 | 23.2 | |
| NR | 548 | 55.5 | |
| HIV | Negative | 747 | 75.7 |
| Positive | 90 | 9.1 | |
| NR | 150 | 15.2 | |
| Tumor site | Anal Canal | 879 | 89.1 |
| Anal Margin | 108 | 10.9 | |
| Histology | Squamous | 880 | 89.1 |
| Basalioid | 84 | 8.5 | |
| Other | 23 | 2.3 | |
| Grading | G1 | 68 | 6.9 |
| G2 | 328 | 33.2 | |
| G3 | 233 | 23.6 | |
| NR | 358 | 36.3 | |
| TNM Stage | T1-T2, N0 | 330 | 33.4 |
| T3-T4, N0 | 106 | 10.7 | |
| Any T, N+ | 551 | 55.8 | |
| Disease extension | Early stage | 330 | 33.4 |
| LAD | 633 | 64.1 | |
| ED | 24 | 2.4 | |
| Staging | N = 987 (%) | |||
|---|---|---|---|---|
| T | N | M | ||
| Single Imaging modality | CT | 143 (14.5) | 186 (18.8) | 402 (40.7) |
| MRI | 541 (54.818) | 321 (32.5) | 12 (1.2) | |
| ERUS | 126 (12.8) | 24 (2.4) | 14 (1.4) | |
| 18FDG-PET | 99 (10.0) | 375 (38) | 467 (47.3) | |
| Multiple Imaging modalities * | 78 (7.9) | 79 (8.2) | 81 (8.2) | |
| Missing data | 0 (0.0) | 2 (0.02) | 11 (1.1) | |
| Treatment Details | n = 987 | % | |
|---|---|---|---|
| Median Total Dose 55 Gy (range: 45–75) | |||
| IMRT modalities | static or dynamic IMRT | 302 | 30.6 |
| VMAT | 470 | 47.6 | |
| HT | 215 | 21.8 | |
| Boost modalities | Sequential | 252 | 25.5 |
| SIB | 568 | 57.5 | |
| SIB+ Sequential | 122 | 12.4 | |
| SIB+ Sequential BRT | 45 | 4.6 | |
| Concomitant chemotherapy | MMC + 5-FU | 634 | 64.2 |
| MMC + Capecitabine | 145 | 14.7 | |
| CDDP + 5-FU | 21 | 2.1 | |
| CDDP + Capecitabine | 67 | 6.8 | |
| MMC | 2 | 0.2 | |
| 5-FU | 8 | 0.8 | |
| CDDP | 3 | 0.3 | |
| Capecitabine | 29 | 2.9 | |
| Other | 21 | 2.1 | |
| NR | 4 | 0.4 | |
| None | 53 | 5.4 | |
| n = 987 (%) | ||||||
|---|---|---|---|---|---|---|
| Grade | G0 | G1 | G2 | G3 | G4 | G3 and G4 |
| ACUTE TOXICITY | ||||||
| Skin | 107 (10.8) | 136 (13.8) | 491 (49.7) | 251 (25.4) | 2 (0.2) | 253 (25.6) |
| Gastrointestinal | 232 (23.5) | 371 (37.6) | 319 (32.3) | 61 (6.2) | 4 (0.4) | 65 (6.6) |
| Urogenital | 552 (55.9) | 334 (3.8) | 96 (9.7) | 5 (0.5) | 0 (0.00) | 5 (0.50) |
| Hematologic | 510 (51.7) | 265 (26.8) | 126 (12.8) | 70 (7.1) | 16 (1.6) | 86 (8.7) |
| LATE TOXICITY | ||||||
| Skin | 792 (80.2) | 183 (18.5) | 10 (1.0) | 2 (0.2) | 0 (0.0) | 2 (0.2) |
| Subcutaneous tissue | 841 (85.2) | 130 (13.2) | 13 (1.3) | 2 (0.2) | 1 (0.1) | 3 (0.3) |
| Gastrointestinal | 699 (70.8) | 205 (20.8) | 65 (6.6) | 17 (1.7) | 1 (0.1) | 18 (1.8) |
| Urogenital | 905 (91.7) | 65 (6.6) | 13 (1.3) | 3 (0.3) | 1 (0.1) | 4 (0.4) |
| Variable | LC | CFS | OS | PFS | EFS | ||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| HR | 95% CI | p-Value | HR | 95% CI | p-Value | HR | 95% CI | p-Value | HR | 95% CI | p-Value | HR | 95% CI | p-Value | |
| Gender (ref. Male) | |||||||||||||||
| Female | 0.77 | (0.54–1.09) | 0.160 | 0.73 | (0.54–0.96) | 0.026 | 0.65 | (0.45–0.94) | 0.022 | 0.74 | (0.54–0.98) | 0.050 | 0.75 | (0.58–0.97) | 0.031 |
| Age (ref. < 68.5 year) | |||||||||||||||
| ≥68.5 year | 0.88 | (0.62–1.25) | 0.491 | 1.07 | (0.82–1.40) | 0.604 | 1.33 | (0.93–1.89) | 0.111 | 1.33 | (1.00–1.78) | 0.050 | 1.15 | (0.90–1.47) | 0.276 |
| ECOG PS (ref. 0) | |||||||||||||||
| 1 | 1.45 | (0.96–2.16) | 0.071 | 1.43 | (1.04–1.96) | 0.025 | 1.59 | (1.06–2.38) | 0.025 | 1.55 | (1.12–2.16) | 0.009 | 1.47 | (1.10–1.95) | 0.008 |
| 2 | 1.60 | (0.39 2.16) | 0.511 | 2.50 | (1.02–6.09) | 0.044 | 4.23 | (1.55–11.54) | 0.005 | 2.31 | (0.85–6.26) | 0.098 | 2.02 | (0.83–4.91) | 0.121 |
| HIV (ref. no) | |||||||||||||||
| Yes | 1.29 | (0.73–2.26) | 0.381 | 1.39 | (0.92–2.08) | 0.117 | 2.06 | (1.28–3.28) | 0.003 | 1.67 | (1.10–2.52) | 0.015 | 1.35 | (0.93–1.98) | 0.118 |
| HPV (ref. No) | |||||||||||||||
| Yes | 1.11 | (0.66–1.87) | 0.689 | 1.01 | (0.65–1.54) | 0.976 | 1.34 | (0.74–2.42) | 0.327 | 0.91 | (0.57–1.43) | 0.671 | 0.90 | (0.61–1.32) | 0.595 |
| Histology (ref. Squamous) | |||||||||||||||
| Basaloid | 0.66 | (0.32–1.35) | 0.255 | 0.73 | (0.43–1.23) | 0.242 | 0.74 | (0.37–1.45) | 0.379 | 0.75 | (0.43–1.32) | 0.320 | 0.76 | (0.47–1.22) | 0.256 |
| Histological Grade (ref. 1) | |||||||||||||||
| 2 | 2.60 | (0.80–8.42) | 0.111 | 0.97 | (0.55–1.68) | 0.904 | 1.03 | (0.50–2.10) | 0.941 | 1.07 | (0.57–1.98) | 0.837 | 1.07 | (0.63–1.83) | 0.800 |
| 3 | 3.83 | (1.18–12.42) | 0.025 | 0.97 | (0.54–1.72) | 0.917 | 1.14 | (0.55–2.39) | 0.720 | 1.32 | (0.70–2.47) | 0.391 | 1.17 | (0.67–2.02) | 0.582 |
| Stage (ref. cT1-2N0) | |||||||||||||||
| cT3-4N0 | 1.76 | (0.90–3.44) | 0.098 | 2.39 | (1.49–3.82) | <0.001 | 3.44 | (1.84–6.39) | <0.001 | 2.69 | (1.59–4.54) | <0.001 | 2.43 | (1.57–3.76) | <0.001 |
| Any T N+ | 2.37 | (1.52–3.68) | <0.001 | 2.26 | (1.61–3.18) | <0.001 | 2.90 | (1.78–4.72) | <0.001 | 2.76 | (1.88–4.07) | <0.001 | 2.36 | (1.72–3.24) | <0.001 |
| Disease extension (ref. Early) | |||||||||||||||
| ED | 7.59 | (3.53–16.31) | <0.001 | 7.90 | (4.30–14.50) | <0.001 | 12.50 | (5.66–27.60) | <0.001 | 11.27 | (5.98–21.22) | <0.001 | 8.44 | (4.85–14.69) | <0.001 |
| LAD | 2.12 | (1.36–3.29) | 0.001 | 2.14 | (1.52–2.99) | <0.001 | 2.78 | (1.71–4.50) | <0.001 | 2.55 | (1.74–3.74) | <0.001 | 2.22 | (1.62–3.03) | <0.001 |
| Variable | LC | CFS | OS | PFS | EFS | ||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| HR | 95% CI | p-Value | HR | 95% CI | p-Value | HR | 95% CI | p-Value | HR | 95% CI | p-Value | HR | 95% CI | p-Value | |
| OTT (Ref. <45) | |||||||||||||||
| ≥45 | 1.13 | (0.80–1.61) | 0.478 | 1.22 | (0.93–1.60) | 0.140 | 1.23 | (0.86–1.75) | 0.243 | 1.33 | (1.00–1.77) | 0.050 | 1.31 | (1.03–1.68) | 0.030 |
| Total dose 54 Gy (ref. ≤ 54 Gy) | |||||||||||||||
| >54 Gy | 0.81 | (0.57–1.15) | 0.238 | 0.90 | (0.69–1.18) | 0.463 | 0.85 | (0.60–1.22) | 0.383 | 1.03 | (0.77–1.39) | 0.821 | 0.98 | (0.76–1.26) | 0.882 |
| Total dose 55 Gy (ref. ≤ 55 Gy) | |||||||||||||||
| >55 Gy | 0.81 | (0.57–1.15) | 0.238 | 0.90 | (0.69–1.18) | 0.463 | 0.85 | (0.60–1.22) | 0.383 | 1.03 | (0.77–1.39) | 0.821 | 1.02 | (0.79–1.30) | 0.904 |
| Dose/Fraction HR PTV (ref. 1.8–2 Gy) | |||||||||||||||
| >2 Gy) | 0.79 | (0.56–1.12) | 0.187 | 0.89 | (0.67–1.17) | 0.385 | 0.88 | (0.61–1.25) | 0.471 | 0,93 | (0.69–1.25) | 0.647 | 0.91 | (0.71–1.17) | 0.452 |
| Dose/Fraction LR PTV (ref. 1.8–2 Gy) | |||||||||||||||
| <1.8 Gy | 0.96 | (0.67–1.39) | 0.835 | 0.98 | (0.74–1.31) | 0.908 | 0.74 | (0.52–1.06) | 0.102 | 0.78 | (0.58–1.04) | 0.090 | 1.01 | (0.67–1.51) | 0.972 |
| SIB (ref. No) | |||||||||||||||
| Yes | 0.92 | (0.64–1.30) | 0.639 | 0.92 | (0.70–1.20) | 0.527 | 0.94 | (0.65–1.33) | 0.713 | 0.93 | (0.69–1.24) | 0.616 | 0.89 | (0.69–1.13) | 0.334 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Caravatta, L.; Mantello, G.; Valvo, F.; Franco, P.; Gasparini, L.; Rosa, C.; Slim, N.; Manfrida, S.; De Felice, F.; Gerardi, M.A.; et al. Radiotherapy with Intensity-Modulated (IMRT) Techniques in the Treatment of Anal Carcinoma (RAINSTORM): A Multicenter Study on Behalf of AIRO (Italian Association of Radiotherapy and Clinical Oncology) Gastrointestinal Study Group. Cancers 2021, 13, 1902. https://doi.org/10.3390/cancers13081902
Caravatta L, Mantello G, Valvo F, Franco P, Gasparini L, Rosa C, Slim N, Manfrida S, De Felice F, Gerardi MA, et al. Radiotherapy with Intensity-Modulated (IMRT) Techniques in the Treatment of Anal Carcinoma (RAINSTORM): A Multicenter Study on Behalf of AIRO (Italian Association of Radiotherapy and Clinical Oncology) Gastrointestinal Study Group. Cancers. 2021; 13(8):1902. https://doi.org/10.3390/cancers13081902
Chicago/Turabian StyleCaravatta, Luciana, Giovanna Mantello, Francesca Valvo, Pierfrancesco Franco, Lucrezia Gasparini, Consuelo Rosa, Najla Slim, Stefania Manfrida, Francesca De Felice, Marianna A. Gerardi, and et al. 2021. "Radiotherapy with Intensity-Modulated (IMRT) Techniques in the Treatment of Anal Carcinoma (RAINSTORM): A Multicenter Study on Behalf of AIRO (Italian Association of Radiotherapy and Clinical Oncology) Gastrointestinal Study Group" Cancers 13, no. 8: 1902. https://doi.org/10.3390/cancers13081902
APA StyleCaravatta, L., Mantello, G., Valvo, F., Franco, P., Gasparini, L., Rosa, C., Slim, N., Manfrida, S., De Felice, F., Gerardi, M. A., Vagge, S., Krengli, M., Palazzari, E., Osti, M. F., Gonnelli, A., Catalano, G., Pittoni, P., Ivaldi, G. B., Galardi, A., ... Genovesi, D. (2021). Radiotherapy with Intensity-Modulated (IMRT) Techniques in the Treatment of Anal Carcinoma (RAINSTORM): A Multicenter Study on Behalf of AIRO (Italian Association of Radiotherapy and Clinical Oncology) Gastrointestinal Study Group. Cancers, 13(8), 1902. https://doi.org/10.3390/cancers13081902















