RTK Inhibitors in Melanoma: From Bench to Bedside
Abstract
Simple Summary
Abstract
1. Introduction
2. RTK Activation and Alterations in Melanoma
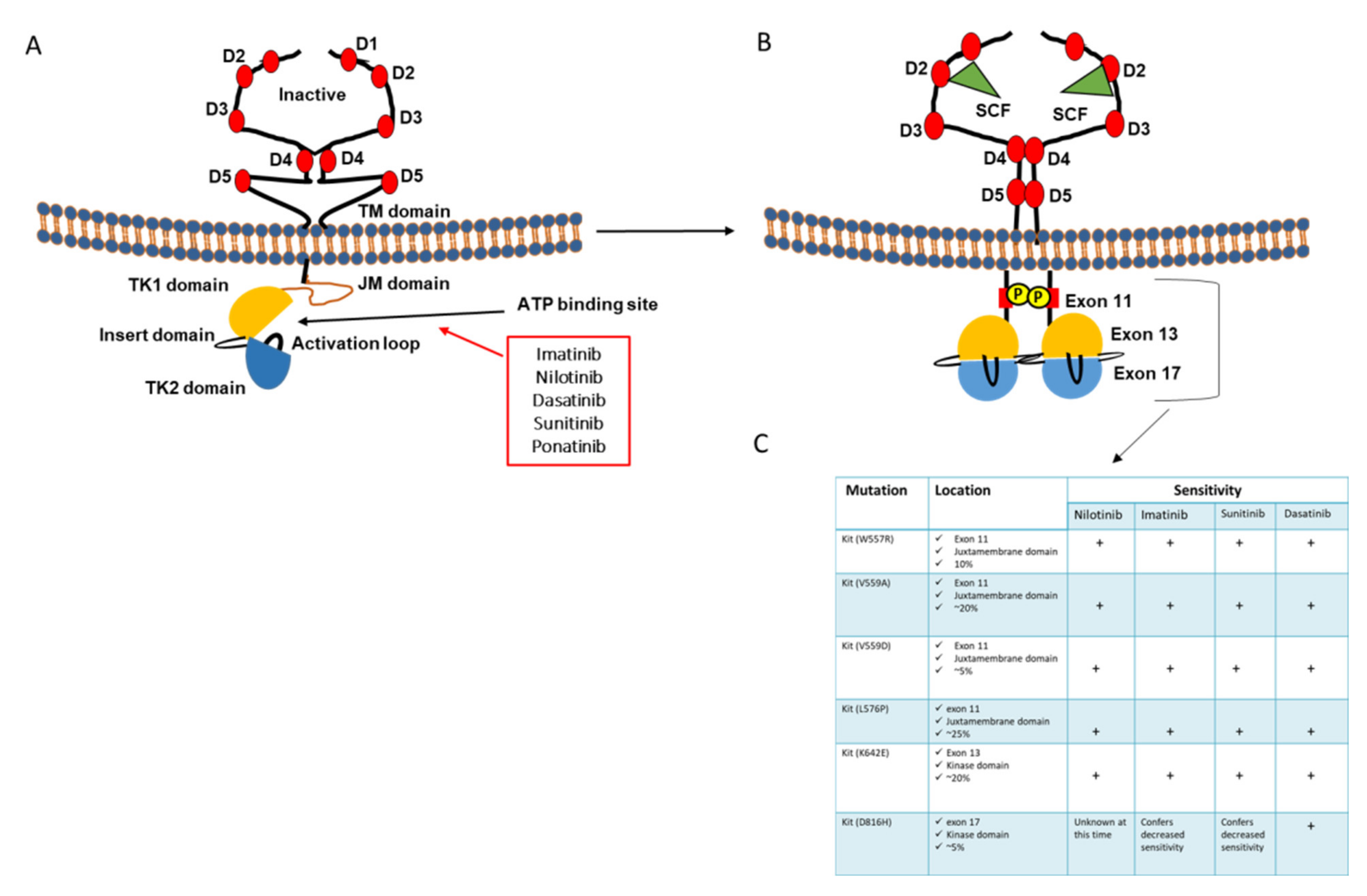
2.1. c-KIT (CD117)
2.1.1. Imatinib (GLEEVEC®)
2.1.2. Nilotinib (TASIGNA®)
2.1.3. Dasatinib (Sprycel®)
2.1.4. Sunitinib (SUTENT®)
2.1.5. Pexidartinib (TURALIO™)
2.1.6. Ponatinib (Iclusig®)
2.1.7. Sorafenib-Nexavar®
2.1.8. c-KIT Inhibitors and Future Perspectives
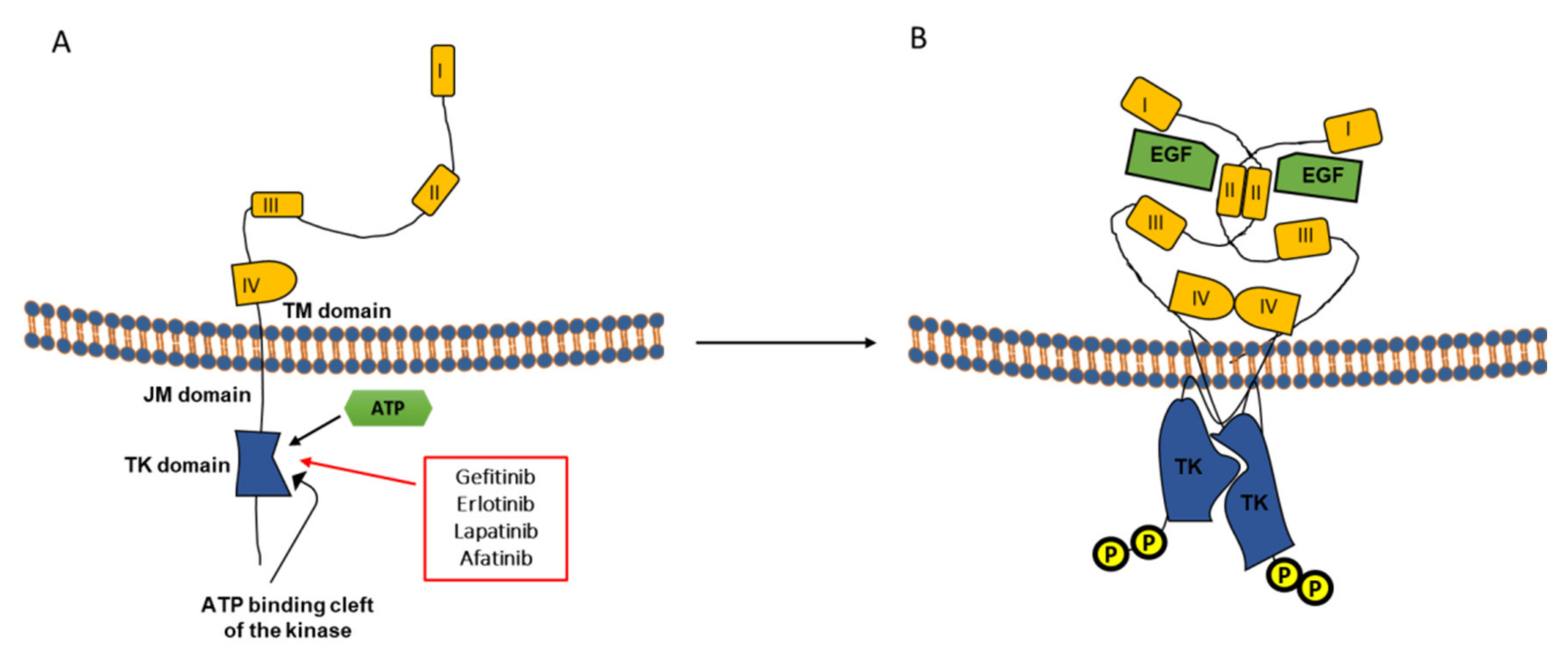
2.2. EGFR
2.2.1. Gefitinib (Iressa®)
2.2.2. Erlotinib (TARCEVA®)
2.2.3. Lapatinib (Tyverb®)
Second Generation of EGFR TK Inhibitors
2.2.4. Afatinib (Giotrif®)
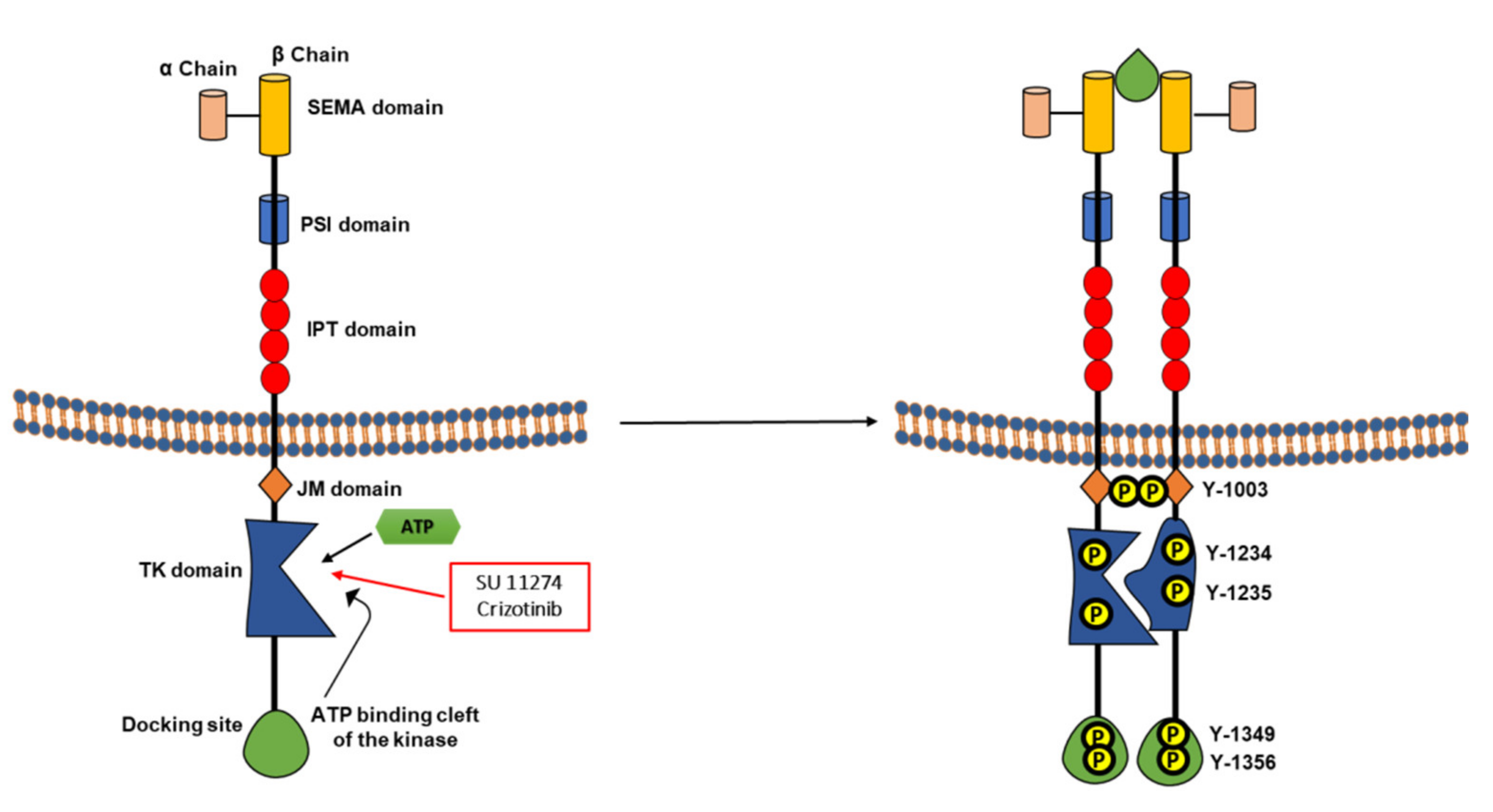
2.3. HGFR (MET Receptor)
2.3.1. SU 11274
2.3.2. Crizotinib (XALKORI®)
2.3.3. Tivantinib
2.3.4. PHA-665752
2.3.5. Quercetin
2.4. VEGFR
2.4.1. SU5416, Sugen (Semaxanib™)
2.4.2. Axitinib (INLYTA®)
Dual MET/HGF and VEGF/VEGFR Targeting
2.4.3. Cabozantinib (COMETRIQ®)
2.4.4. Foretinib (Exelixis, GlaxoSmithKline) (XL-880)
2.4.5. E7050
2.5. Other RTKs
2.5.1. IGF1R
2.5.2. FGFR
3. RTK Inhibitors as Immune Modulators
4. RTK Inhibitors and Check-Point Inhibitors Combination
5. Conclusions
Funding
Conflicts of Interest
References
- Hedger, G.; Sansom, M.S.P.; Koldsø, H. The Juxtamembrane Regions of Human Receptor Tyrosine Kinases Exhibit Conserved Interaction Sites with Anionic Lipids. Sci. Rep. 2015, 5, 9198. [Google Scholar] [CrossRef]
- Lemmon, M.A.; Schlessinger, J. Cell Signaling by Receptor Tyrosine Kinases. Cell 2010, 141, 1117–1134. [Google Scholar] [CrossRef] [PubMed]
- Wintheiser, G.A.; Silberstein, P. Physiology, Tyrosine Kinase Receptors. In StatPearls; StatPearls Publishing: Treasure Island, FL, USA, 2019. [Google Scholar]
- Du, Z.; Lovly, C.M. Mechanisms of Receptor Tyrosine Kinase Activation in Cancer. Mol. Cancer 2018, 17, 58. [Google Scholar] [CrossRef] [PubMed]
- Braeuer, R.R.; Watson, I.R.; Wu, C.-J.; Mobley, A.K.; Kamiya, T.; Shoshan, E.; Bar-Eli, M. Why Is Melanoma so Metastatic? Pigment Cell Melanoma Res. 2014, 27, 19–36. [Google Scholar] [CrossRef] [PubMed]
- McArthur, G.A.; Ribas, A. Targeting Oncogenic Drivers and the Immune System in Melanoma. J. Clin. Oncol. 2013, 31, 499–506. [Google Scholar] [CrossRef]
- Akbani, R.; Kadir, C.; Aksoy, B.A.; Albert, M.; Ally, A.; Amin, S.B.; Arachchi, H.; Arora, A.J.; Auman, T.; Ayala, B.; et al. Genomic Classification of Cutaneous Melanoma. Cell 2015, 161, 1681–1696. [Google Scholar] [CrossRef]
- Cheng, L.; Lopez-Beltran, A.; Massari, F.; MacLennan, G.T.; Montironi, R. Molecular Testing for BRAF Mutations to Inform Melanoma Treatment Decisions: A Move toward Precision Medicine. Mod. Pathol. 2018, 31, 24–38. [Google Scholar] [CrossRef]
- Ribas, A.; Lawrence, D.; Atkinson, V.; Agarwal, S.; Miller, W.H.; Carlino, M.S.; Fisher, R.; Long, G.V.; Hodi, F.S.; Tsoi, J.; et al. Combined BRAF and MEK Inhibition with PD-1 Blockade Immunotherapy in BRAF-Mutant Melanoma. Nat. Med. 2019, 25, 936–940. [Google Scholar] [CrossRef]
- Aasen, S.N.; Parajuli, H.; Hoang, T.; Feng, Z.; Stokke, K.; Wang, J.; Roy, K.; Bjerkvig, R.; Knappskog, S.; Thorsen, F. Effective Treatment of Metastatic Melanoma by Combining MAPK and PI3K Signaling Pathway Inhibitors. Int. J. Mol. Sci. 2019, 20, 4235. [Google Scholar] [CrossRef]
- Muñoz-Couselo, E.; Adelantado, E.Z.; Ortiz, C.; García, J.S.; Perez-Garcia, J. NRAS-Mutant Melanoma: Current Challenges and Future Prospect. OncoTargets Ther. 2017, 10, 3941–3947. [Google Scholar] [CrossRef]
- O’Bryan, J.P. Pharmacological Targeting of RAS: Recent Success with Direct Inhibitors. Pharmacol. Res. 2019, 139, 503–511. [Google Scholar] [CrossRef]
- McKean, M.; Oba, J.; Ma, J.; Roth, K.G.; Wang, W.-L.; Macedo, M.P.; Carapeto, F.C.L.; Haydu, L.E.; Siroy, A.E.; Vo, P.; et al. Tyrosine Kinase Inhibitor and Immune Checkpoint Inhibitor Responses in KIT-Mutant Metastatic Melanoma. J. Investig. Dermatol. 2019, 139, 728–731. [Google Scholar] [CrossRef]
- Newell, F.; Kong, Y.; Wilmott, J.S.; Johansson, P.A.; Ferguson, P.M.; Cui, C.; Li, Z.; Kazakoff, S.H.; Burke, H.; Dodds, T.J.; et al. Whole-Genome Landscape of Mucosal Melanoma Reveals Diverse Drivers and Therapeutic Targets. Nat. Commun. 2019, 10. [Google Scholar] [CrossRef]
- D’Angelo, S.P.; Larkin, J.; Sosman, J.A.; Lebbé, C.; Brady, B.; Neyns, B.; Schmidt, H.; Hassel, J.C.; Hodi, F.S.; Lorigan, P.; et al. Efficacy and Safety of Nivolumab Alone or in Combination with Ipilimumab in Patients With Mucosal Melanoma: A Pooled Analysis. J. Clin. Oncol. 2017, 35, 226–235. [Google Scholar] [CrossRef] [PubMed]
- Hintzsche, J.D.; Gorden, N.T.; Amato, C.M.; Kim, J.; Wuensch, K.E.; Robinson, S.E.; Applegate, A.J.; Couts, K.L.; Medina, T.M.; Wells, K.R.; et al. Whole-Exome Sequencing Identifies Recurrent SF3B1 R625 Mutation and Comutation of NF1 and KIT in Mucosal Melanoma. Melanoma Res. 2017, 27, 189–199. [Google Scholar] [CrossRef] [PubMed]
- Lyu, J.; Song, Z.; Chen, J.; Shepard, M.J.; Song, H.; Ren, G.; Li, Z.; Guo, W.; Zhuang, Z.; Shi, Y. Whole-Exome Sequencing of Oral Mucosal Melanoma Reveals Mutational Profile and Therapeutic Targets. J. Pathol. 2018, 244, 358–366. [Google Scholar] [CrossRef] [PubMed]
- Hayward, N.K.; Wilmott, J.S.; Waddell, N.; Johansson, P.A.; Field, M.A.; Nones, K.; Patch, A.-M.; Kakavand, H.; Alexandrov, L.B.; Burke, H.; et al. Whole-Genome Landscapes of Major Melanoma Subtypes. Nature 2017, 545, 175–180. [Google Scholar] [CrossRef] [PubMed]
- Tworkoski, K.; Singhal, G.; Szpakowski, S.; Zito, C.I.; Bacchiocchi, A.; Muthusamy, V.; Bosenberg, M.; Krauthammer, M.; Halaban, R.; Stern, D.F. Phospho-Proteomic Screen Identifies Potential Therapeutic Targets in Melanoma. Mol. Cancer Res. 2011, 9, 801–812. [Google Scholar] [CrossRef] [PubMed]
- Sun, C.; Wang, L.; Huang, S.; Heynen, G.J.J.E.; Prahallad, A.; Robert, C.; Haanen, J.; Blank, C.; Wesseling, J.; Willems, S.M.; et al. Reversible and Adaptive Resistance to BRAF(V600E) Inhibition in Melanoma. Nature 2014, 508, 118–122. [Google Scholar] [CrossRef] [PubMed]
- Li, F.Z.; Dhillon, A.S.; Anderson, R.L.; McArthur, G.; Ferrao, P.T. Phenotype Switching in Melanoma: Implications for Progression and Therapy. Front. Oncol. 2015, 5. [Google Scholar] [CrossRef] [PubMed]
- Albanesi, C.; Geremia, R.; Giorgio, M.; Dolci, S.; Sette, C.; Rossi, P. A Cell- and Developmental Stage-Specific Promoter Drives the Expression of a Truncated c-Kit Protein during Mouse Spermatid Elongation. Development 1996, 122, 1291–1302. [Google Scholar] [PubMed]
- Roskoski, R. Structure and Regulation of Kit Protein-Tyrosine Kinase—The Stem Cell Factor Receptor. Biochem. Biophys. Res. Commun. 2005, 338, 1307–1315. [Google Scholar] [CrossRef]
- Roskoski, R. Signaling by Kit Protein-Tyrosine Kinase—The Stem Cell Factor Receptor. Biochem. Biophys. Res. Commun. 2005, 337, 1–13. [Google Scholar] [CrossRef] [PubMed]
- Kitamura, Y.; Hirotab, S. Kit as a Human Oncogenic Tyrosine Kinase. Cell. Mol. Life Sci. 2004, 61, 2924–2931. [Google Scholar] [CrossRef] [PubMed]
- Heinrich, M.C.; Blanke, C.D.; Druker, B.J.; Corless, C.L. Inhibition of KIT Tyrosine Kinase Activity: A Novel Molecular Approach to the Treatment of KIT-Positive Malignancies. J. Clin. Oncol. 2002, 20, 1692–1703. [Google Scholar] [CrossRef] [PubMed]
- Shen, H.; Tang, Y.; Yang, S.; Qian, B.; Song, H.; Shi, S.; Xu, W. Analysis of CD117 expression on leukemia cells. Zhonghua Xue Ye Xue Za Zhi 2003, 24, 228–230. [Google Scholar]
- Duensing, A.; Medeiros, F.; McConarty, B.; Joseph, N.E.; Panigrahy, D.; Singer, S.; Fletcher, C.D.M.; Demetri, G.D.; Fletcher, J.A. Mechanisms of Oncogenic KIT Signal Transduction in Primary Gastrointestinal Stromal Tumors (GISTs). Oncogene 2004, 23, 3999–4006. [Google Scholar] [CrossRef]
- Tzen, C.-Y.; Wu, Y.-H.; Tzen, C.-Y. Characterization of KIT Mutation in Melanoma. Dermatol. Sin. 2014, 32, 7–12. [Google Scholar] [CrossRef]
- Conca, E.; Negri, T.; Gronchi, A.; Fumagalli, E.; Tamborini, E.; Pavan, G.M.; Fermeglia, M.; Pierotti, M.A.; Pricl, S.; Pilotti, S. Activate and Resist: L576P-KIT in GIST. Mol. Cancer Ther. 2009, 8, 2491–2495. [Google Scholar] [CrossRef][Green Version]
- Carvajal, R.D.; Antonescu, C.R.; Wolchok, J.D.; Chapman, P.B.; Roman, R.-A.; Teitcher, J.; Panageas, K.S.; Busam, K.J.; Chmielowski, B.; Lutzky, J.; et al. KIT as a Therapeutic Target in Metastatic Melanoma. JAMA 2011, 305, 2327–2334. [Google Scholar] [CrossRef]
- Hodi, F.S.; Corless, C.L.; Giobbie-Hurder, A.; Fletcher, J.A.; Zhu, M.; Marino-Enriquez, A.; Friedlander, P.; Gonzalez, R.; Weber, J.S.; Gajewski, T.F.; et al. Imatinib for Melanomas Harboring Mutationally Activated or Amplified KIT Arising on Mucosal, Acral, and Chronically Sun-Damaged Skin. J. Clin. Oncol. 2013, 31, 3182–3190. [Google Scholar] [CrossRef] [PubMed]
- Chris, H.; Takimoto, M.D. Principles of Oncologic Pharmacotherapy. Available online: https://www.cancernetwork.com/articles/principles-oncologic-pharmacotherapy (accessed on 25 March 2020).
- Potti, A.; Hille, R.C.; Koch, M. Immunohistochemical Determination of HER-2/Neu Overexpression in Malignant Melanoma Reveals No Prognostic Value, While c-Kit (CD117) Overexpression Exhibits Potential Therapeutic Implications. J. Carcinog. 2003, 2, 8. [Google Scholar] [CrossRef] [PubMed]
- Druker, B.J.; Talpaz, M.; Resta, D.J.; Peng, B.; Buchdunger, E.; Ford, J.M.; Lydon, N.B.; Kantarjian, H.; Capdeville, R.; Ohno-Jones, S.; et al. Efficacy and Safety of a Specific Inhibitor of the BCR-ABL Tyrosine Kinase in Chronic Myeloid Leukemia. N. Engl. J. Med. 2001, 344, 1031–1037. [Google Scholar] [CrossRef] [PubMed]
- Van Oosterom, A.T.; Judson, I.; Verweij, J.; Stroobants, S.; Donato di Paola, E.; Dimitrijevic, S.; Martens, M.; Webb, A.; Sciot, R.; Van Glabbeke, M.; et al. Safety and Efficacy of Imatinib (STI571) in Metastatic Gastrointestinal Stromal Tumours: A Phase I Study. Lancet 2001, 358, 1421–1423. [Google Scholar] [CrossRef]
- McGary, E.C.; Onn, A.; Mills, L.; Heimberger, A.; Eton, O.; Thomas, G.W.; Shtivelband, M.; Bar-Eli, M. Imatinib Mesylate Inhibits Platelet-Derived Growth Factor Receptor Phosphorylation of Melanoma Cells but Does Not Affect Tumorigenicity in Vivo. J. Investig. Dermatol. 2004, 122, 400–405. [Google Scholar] [CrossRef]
- Redondo, P.; Lloret, P.; Andreu, E.J.; Inoges, S. Imatinib Mesylate in Cutaneous Melanoma. J. Investig. Dermatol. 2004, 123, 1208–1209. [Google Scholar] [CrossRef] [PubMed]
- Ugurel, S.; Hildenbrand, R.; Zimpfer, A.; Rosée, P.L.; Paschka, P.; Sucker, A.; Keikavoussi, P.; Becker, J.C.; Rittgen, W.; Hochhaus, A.; et al. Lack of Clinical Efficacy of Imatinib in Metastatic Melanoma. Br. J. Cancer 2005, 92, 1398–1405. [Google Scholar] [CrossRef] [PubMed]
- Kantarjian, H.; Sawyers, C.; Hochhaus, A.; Guilhot, F.; Schiffer, C.; Gambacorti-Passerini, C.; Niederwieser, D.; Resta, D.; Capdeville, R.; Zoellner, U.; et al. Hematologic and Cytogenetic Responses to Imatinib Mesylate in Chronic Myelogenous Leukemia. N. Engl. J. Med. 2002, 346, 645–652. [Google Scholar] [CrossRef]
- Wyman, K.; Atkins, M.B.; Prieto, V.; Eton, O.; McDermott, D.F.; Hubbard, F.; Byrnes, C.; Sanders, K.; Sosman, J.A. Multicenter Phase II Trial of High-Dose Imatinib Mesylate in Metastatic Melanoma: Significant Toxicity with No Clinical Efficacy. Cancer 2006, 106, 2005–2011. [Google Scholar] [CrossRef] [PubMed]
- Kim, K.B.; Eton, O.; Davis, D.W.; Frazier, M.L.; McConkey, D.J.; Diwan, A.H.; Papadopoulos, N.E.; Bedikian, A.Y.; Camacho, L.H.; Ross, M.I.; et al. Phase II Trial of Imatinib Mesylate in Patients with Metastatic Melanoma. Br. J. Cancer 2008, 99, 734–740. [Google Scholar] [CrossRef]
- Curtin, J.A.; Busam, K.; Pinkel, D.; Bastian, B.C. Somatic Activation of KIT in Distinct Subtypes of Melanoma. J. Clin. Oncol. 2006, 24, 4340–4346. [Google Scholar] [CrossRef]
- Jiang, X.; Zhou, J.; Yuen, N.K.; Corless, C.L.; Heinrich, M.C.; Fletcher, J.A.; Demetri, G.D.; Widlund, H.R.; Fisher, D.E.; Hodi, F.S. Imatinib Targeting of KIT-Mutant Oncoprotein in Melanoma. Clin. Cancer Res. 2008, 14, 7726–7732. [Google Scholar] [CrossRef] [PubMed]
- Guo, J.; Si, L.; Kong, Y.; Flaherty, K.T.; Xu, X.; Zhu, Y.; Corless, C.L.; Li, L.; Li, H.; Sheng, X.; et al. Phase II, Open-Label, Single-Arm Trial of Imatinib Mesylate in Patients with Metastatic Melanoma Harboring c-Kit Mutation or Amplification. J. Clin. Oncol. 2011, 29, 2904–2909. [Google Scholar] [CrossRef]
- Rapisuwon, S.; Parks, K.; Al-Refaie, W.; Atkins, M.B. Novel Somatic KIT Exon 8 Mutation with Dramatic Response to Imatinib in a Patient with Mucosal Melanoma: A Case Report. Melanoma Res. 2014, 24, 509–511. [Google Scholar] [CrossRef]
- Kim, K.B.; Alrwas, A. Treatment of KIT-Mutated Metastatic Mucosal Melanoma. Chin. Clin. Oncol. 2014, 3, 35. [Google Scholar] [CrossRef] [PubMed]
- Flaherty, K.T.; Hamilton, B.K.; Rosen, M.A.; Amaravadi, R.K.; Schuchter, L.M.; Gallagher, M.; Chen, H.; Sehgal, C.; O’Dwyer, P.J. Phase I/II Trial of Imatinib and Bevacizumab in Patients with Advanced Melanoma and Other Advanced Cancers. Oncologist 2015, 20, 952–959. [Google Scholar] [CrossRef] [PubMed]
- Wang, H.; Cheng, F.; Cuenca, A.; Horna, P.; Zheng, Z.; Bhalla, K.; Sotomayor, E.M. Imatinib Mesylate (STI-571) Enhances Antigen-Presenting Cell Function and Overcomes Tumor-Induced CD4+ T-Cell Tolerance. Blood 2005, 105, 1135–1143. [Google Scholar] [CrossRef]
- Reilley, M.J.; Bailey, A.; Subbiah, V.; Janku, F.; Naing, A.; Falchook, G.; Karp, D.; Piha-Paul, S.; Tsimberidou, A.; Fu, S.; et al. Phase I Clinical Trial of Combination Imatinib and Ipilimumab in Patients with Advanced Malignancies. J. Immunother. Cancer 2017, 5, 35. [Google Scholar] [CrossRef] [PubMed]
- Fecher, L.A.; Nathanson, K.; Flaherty, K.T.; Amaravadi, R.; Giles, L.; McGettigan, S.; Carberry, M.; Elder, D.; Schuchter, L. Phase I/II Trial of Imatinib and Temozolomide in Advanced Unresectable Melanoma. JCO 2008, 26, 9059. [Google Scholar] [CrossRef]
- Pembrolizumab and Imatinib in Patients With Locally Advanced/Metastatic Melanoma With c-KIT Mutation/Amplification-Full Text View-ClinicalTrials.Gov. Available online: https://clinicaltrials.gov/ct2/show/NCT02812693 (accessed on 21 April 2020).
- Abdou, Y.; Kapoor, A.; Hamad, L.; Ernstoff, M.S. Combination of Pembrolizumab and Imatinib in a Patient with Double KIT Mutant Melanoma. Medicine 2019, 98. [Google Scholar] [CrossRef]
- Wei, X.; Mao, L.; Chi, Z.; Sheng, X.; Cui, C.; Kong, Y.; Dai, J.; Wang, X.; Li, S.; Tang, B.; et al. Efficacy Evaluation of Imatinib for the Treatment of Melanoma: Evidence from a Retrospective Study. Oncol. Res. 2019, 27, 495–501. [Google Scholar] [CrossRef]
- Todd, J.R.; Becker, T.M.; Kefford, R.F.; Rizos, H. Secondary C-Kit Mutations Confer Acquired Resistance to RTK Inhibitors in c-Kit Mutant Melanoma Cells. Pigment Cell Melanoma Res. 2013, 26, 518–526. [Google Scholar] [CrossRef] [PubMed]
- Delyon, J.; Chevret, S.; Jouary, T.; Dalac, S.; Dalle, S.; Guillot, B.; Arnault, J.-P.; Avril, M.-F.; Bedane, C.; Bens, G.; et al. STAT3 Mediates Nilotinib Response in KIT-Altered Melanoma: A Phase II Multicenter Trial of the French Skin Cancer Network. J. Investig. Dermatol. 2018, 138, 58–67. [Google Scholar] [CrossRef] [PubMed]
- Eustace, A.J.; Crown, J.; Clynes, M.; O’Donovan, N. Preclinical Evaluation of Dasatinib, a Potent Src Kinase Inhibitor, in Melanoma Cell Lines. J. Transl. Med. 2008, 6, 53. [Google Scholar] [CrossRef] [PubMed]
- Buettner, R.; Mesa, T.; Vultur, A.; Lee, F.; Jove, R. Inhibition of Src Family Kinases with Dasatinib Blocks Migration and Invasion of Human Melanoma Cells. Mol. Cancer Res. 2008, 6, 1766–1774. [Google Scholar] [CrossRef]
- Woodman, S.E.; Trent, J.C.; Stemke-Hale, K.; Lazar, A.J.; Pricl, S.; Pavan, G.M.; Fermeglia, M.; Gopal, Y.N.V.; Yang, D.; Podoloff, D.A.; et al. Activity of Dasatinib Against L576P KIT Mutant Melanoma: Molecular, Cellular and Clinical Correlates. Mol. Cancer Ther. 2009, 8, 2079–2085. [Google Scholar] [CrossRef] [PubMed]
- Homsi, J.; Cubitt, C.L.; Zhang, S.; Munster, P.N.; Yu, H.; Sullivan, D.M.; Jove, R.; Messina, J.L.; Daud, A.I. Src Activation in Melanoma and Src Inhibitors as Therapeutic Agents in Melanoma. Melanoma Res. 2009, 19, 167–175. [Google Scholar] [CrossRef]
- Wu, J.; Liao, X.; Yu, B.; Su, B. Dasatinib Inhibits Primary Melanoma Cell Proliferation through Morphology-Dependent Disruption of Src-ERK Signaling. Oncol. Lett. 2013, 5, 527–532. [Google Scholar] [CrossRef]
- Eustace, A.J.; Kennedy, S.; Larkin, A.-M.; Mahgoub, T.; Tryfonopoulos, D.; O’Driscoll, L.; Clynes, M.; Crown, J.; O’Donovan, N. Predictive Biomarkers for Dasatinib Treatment in Melanoma. Oncoscience 2014, 1, 158–166. [Google Scholar] [CrossRef]
- Karwaciak, I.; Sałkowska, A.; Karaś, K.; Sobalska-Kwapis, M.; Walczak-Drzewiecka, A.; Pułaski, Ł.; Strapagiel, D.; Dastych, J.; Ratajewski, M. SIRT2 Contributes to the Resistance of Melanoma Cells to the Multikinase Inhibitor Dasatinib. Cancers 2019, 11. [Google Scholar] [CrossRef]
- Gaustad, J.-V.; Simonsen, T.G.; Leinaas, M.N.; Rofstad, E.K. Sunitinib Treatment Does Not Improve Blood Supply but Induces Hypoxia in Human Melanoma Xenografts. BMC Cancer 2012, 12, 388. [Google Scholar] [CrossRef] [PubMed]
- Gaustad, J.-V.; Pozdniakova, V.; Hompland, T.; Simonsen, T.G.; Rofstad, E.K. Magnetic Resonance Imaging Identifies Early Effects of Sunitinib Treatment in Human Melanoma Xenografts. J. Exp. Clin. Cancer Res. 2013, 32, 93. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Han, Y.; Gu, Z.; Wu, J.; Huang, X.; Zhou, R.; Shi, C.; Tao, W.; Wang, L.; Wang, Y.; Zhou, G.; et al. Repurposing Ponatinib as a Potent Agent against KIT Mutant Melanomas. Theranostics 2019, 9, 1952–1964. [Google Scholar] [CrossRef] [PubMed]
- Cullinane, C.; Natoli, A.; Hui, Y.; Conus, N.; Jackson, S.; Brüggen, J.; Manley, P.W.; McArthur, G.A. Preclinical Evaluation of Nilotinib Efficacy in an Imatinib-Resistant KIT-Driven Tumor Model. Mol. Cancer Ther. 2010, 9, 1461–1468. [Google Scholar] [CrossRef]
- Cho, J.H.; Kim, K.M.; Kwon, M.; Kim, J.H.; Lee, J. Nilotinib in Patients with Metastatic Melanoma Harboring KIT Gene Aberration. Investig. New Drugs 2012, 30, 2008–2014. [Google Scholar] [CrossRef] [PubMed]
- Carvajal, R.D.; Lawrence, D.P.; Weber, J.S.; Gajewski, T.F.; Gonzalez, R.; Lutzky, J.; O’Day, S.J.; Hamid, O.; Wolchok, J.D.; Chapman, P.B.; et al. Phase II Study of Nilotinib in Melanoma Harboring KIT Alterations Following Progression to Prior KIT Inhibition. Clin. Cancer Res. 2015, 21, 2289–2296. [Google Scholar] [CrossRef]
- Lee, S.J.; Kim, T.M.; Kim, Y.J.; Jang, K.-T.; Lee, H.J.; Lee, S.N.; Ahn, M.S.; Hwang, I.G.; Lee, S.; Lee, M.-H.; et al. Phase II Trial of Nilotinib in Patients with Metastatic Malignant Melanoma Harboring KIT Gene Aberration: A Multicenter Trial of Korean Cancer Study Group (UN10-06). Oncologist 2015, 20, 1312–1319. [Google Scholar] [CrossRef]
- Guo, J.; Carvajal, R.D.; Dummer, R.; Hauschild, A.; Daud, A.; Bastian, B.C.; Markovic, S.N.; Queirolo, P.; Arance, A.; Berking, C.; et al. Efficacy and Safety of Nilotinib in Patients with KIT-Mutated Metastatic or Inoperable Melanoma: Final Results from the Global, Single-Arm, Phase II TEAM Trial. Ann. Oncol. 2017, 28, 1380–1387. [Google Scholar] [CrossRef]
- Successful Treatment with Imatinib after Nilotinib and Ipilimumab in a C-Kit-Mutated Advanced Melanoma Patient: A Case Report.-PubMed-NCBI. Available online: https://www.ncbi.nlm.nih.gov/pubmed/28410286 (accessed on 15 January 2020).
- Dasatinib. LiverTox: Clinical and Research Information on Drug-Induced Liver Injury; National Institute of Diabetes and Digestive and Kidney Diseases: Bethesda, MD, USA, 2012. [Google Scholar]
- Algazi, A.P.; Weber, J.S.; Andrews, S.C.; Urbas, P.; Munster, P.N.; DeConti, R.C.; Hwang, J.; Sondak, V.K.; Messina, J.L.; McCalmont, T.; et al. Phase I Clinical Trial of the Src Inhibitor Dasatinib with Dacarbazine in Metastatic Melanoma. Br. J. Cancer 2012, 106, 85–91. [Google Scholar] [CrossRef]
- Kluger, H.M.; Dudek, A.Z.; McCann, C.; Ritacco, J.; Southard, N.; Jilaveanu, L.B.; Molinaro, A.; Sznol, M. A Phase II Trial of Dasatinib in Advanced Melanoma. Cancer 2011, 117, 2202–2208. [Google Scholar] [CrossRef]
- Kalinsky, K.; Lee, S.; Rubin, K.; Lawrence, D.P.; Iafrarte, A.J.; Borger, D.R.; Margolin, K.A.; Leitao, M.M.; Tarhini, A.A.; Koon, H.B.; et al. A Phase II Trial of Dasatinib in Patients with Locally Advanced or Stage IV Mucosal, Acral and Vulvovaginal Melanoma: A Trial of the ECOG-ACRIN Cancer Research Group (E2607). Cancer 2017, 123, 2688–2697. [Google Scholar] [CrossRef]
- Skoko, J.; Rožanc, J.; Charles, E.M.; Alexopoulos, L.G.; Rehm, M. Post-Treatment de-Phosphorylation of P53 Correlates with Dasatinib Responsiveness in Malignant Melanoma. BMC Cell Biol. 2018, 19. [Google Scholar] [CrossRef] [PubMed]
- Imbulgoda, A.; Heng, D.Y.C.; Kollmannsberger, C. Sunitinib in the Treatment of Advanced Solid Tumors. Recent Results Cancer Res. 2014, 201, 165–184. [Google Scholar] [CrossRef] [PubMed]
- Demetri, G.D.; van Oosterom, A.T.; Garrett, C.R.; Blackstein, M.E.; Shah, M.H.; Verweij, J.; McArthur, G.; Judson, I.R.; Heinrich, M.C.; Morgan, J.A.; et al. Efficacy and Safety of Sunitinib in Patients with Advanced Gastrointestinal Stromal Tumour after Failure of Imatinib: A Randomised Controlled Trial. Lancet 2006, 368, 1329–1338. [Google Scholar] [CrossRef]
- Motzer, R.J.; Hutson, T.E.; Tomczak, P.; Michaelson, M.D.; Bukowski, R.M.; Oudard, S.; Negrier, S.; Szczylik, C.; Pili, R.; Bjarnason, G.A.; et al. Overall Survival and Updated Results for Sunitinib Compared with Interferon Alfa in Patients with Metastatic Renal Cell Carcinoma. J. Clin. Oncol. 2009, 27, 3584–3590. [Google Scholar] [CrossRef]
- Minor, D.R.; Kashani-Sabet, M.; Garrido, M.; O’Day, S.J.; Hamid, O.; Bastian, B.C. Sunitinib Therapy for Melanoma Patients with KIT Mutations. Clin. Cancer Res. 2012, 18, 1457–1463. [Google Scholar] [CrossRef] [PubMed]
- Decoster, L.; Vande Broek, I.; Neyns, B.; Majois, F.; Baurain, J.F.; Rottey, S.; Rorive, A.; Anckaert, E.; De Mey, J.; De Brakeleer, S.; et al. Biomarker Analysis in a Phase II Study of Sunitinib in Patients with Advanced Melanoma. Anticancer Res. 2015, 35, 6893–6899. [Google Scholar] [PubMed]
- Buchbinder, E.I.; Sosman, J.A.; Lawrence, D.P.; McDermott, D.F.; Ramaiya, N.H.; Van den Abbeele, A.D.; Linette, G.P.; Giobbie-Hurder, A.; Hodi, F.S. Phase 2 Study of Sunitinib in Patients with Metastatic Mucosal or Acral Melanoma. Cancer 2015, 121, 4007–4015. [Google Scholar] [CrossRef]
- Lamb, Y.N. Pexidartinib: First Approval. Drugs 2019, 79, 1805–1812. [Google Scholar] [CrossRef]
- A Study of PLX3397 in Patients with Unresectable or Metastatic KIT-Mutated Melanoma-Full Text View-ClinicalTrials.Gov. Available online: https://clinicaltrials.gov/ct2/show/NCT02975700 (accessed on 16 April 2020).
- Tan, F.H.; Putoczki, T.L.; Stylli, S.S.; Luwor, R.B. Ponatinib: A Novel Multi-Tyrosine Kinase Inhibitor against Human Malignancies. OncoTargets Ther. 2019, 12, 635–645. [Google Scholar] [CrossRef]
- Eisen, T.; Ahmad, T.; Flaherty, K.T.; Gore, M.; Kaye, S.; Marais, R.; Gibbens, I.; Hackett, S.; James, M.; Schuchter, L.M.; et al. Sorafenib in Advanced Melanoma: A Phase II Randomised Discontinuation Trial Analysis. Br. J. Cancer 2006, 95, 581–586. [Google Scholar] [CrossRef]
- Huse, M.; Kuriyan, J. The Conformational Plasticity of Protein Kinases. Cell 2002, 109, 275–282. [Google Scholar] [CrossRef]
- Vajpai, N.; Strauss, A.; Fendrich, G.; Cowan-Jacob, S.W.; Manley, P.W.; Grzesiek, S.; Jahnke, W. Solution Conformations and Dynamics of ABL Kinase-Inhibitor Complexes Determined by NMR Substantiate the Different Binding Modes of Imatinib/Nilotinib and Dasatinib. J. Biol. Chem. 2008, 283, 18292–18302. [Google Scholar] [CrossRef] [PubMed]
- Schlessinger, J. Ligand-Induced, Receptor-Mediated Dimerization and Activation of EGF Receptor. Cell 2002, 110, 669–672. [Google Scholar] [CrossRef]
- Wells, A. EGF Receptor. Int. J. Biochem. Cell Biol. 1999, 31, 637–643. [Google Scholar] [CrossRef]
- Macdonald-Obermann, J.L.; Pike, L.J. Different Epidermal Growth Factor (EGF) Receptor Ligands Show Distinct Kinetics and Biased or Partial Agonism for Homodimer and Heterodimer Formation. J. Biol. Chem. 2014, 289, 26178–26188. [Google Scholar] [CrossRef]
- Wilson, K.J.; Mill, C.; Lambert, S.; Buchman, J.; Wilson, T.R.; Hernandez-Gordillo, V.; Gallo, R.M.; Ades, L.M.C.; Settleman, J.; Riese, D.J. EGFR Ligands Exhibit Functional Differences in Models of Paracrine and Autocrine Signaling. Growth Factors 2012, 30, 107–116. [Google Scholar] [CrossRef]
- Landau, M.; Fleishman, S.J.; Ben-Tal, N. A Putative Mechanism for Downregulation of the Catalytic Activity of the EGF Receptor via Direct Contact between Its Kinase and C-Terminal Domains. Structure 2004, 12, 2265–2275. [Google Scholar] [CrossRef]
- Zhang, X.; Gureasko, J.; Shen, K.; Cole, P.A.; Kuriyan, J. An Allosteric Mechanism for Activation of the Kinase Domain of Epidermal Growth Factor Receptor. Cell 2006, 125, 1137–1149. [Google Scholar] [CrossRef]
- Jura, N.; Endres, N.F.; Engel, K.; Deindl, S.; Das, R.; Lamers, M.H.; Wemmer, D.E.; Zhang, X.; Kuriyan, J. Mechanism for Activation of the EGF Receptor Catalytic Domain by the Juxtamembrane Segment. Cell 2009, 137, 1293. [Google Scholar] [CrossRef]
- Yarden, Y.; Sliwkowski, M.X. Untangling the ErbB Signalling Network. Nat. Rev. Mol. Cell Biol. 2001, 2, 127–137. [Google Scholar] [CrossRef]
- Fleishman, S.J.; Schlessinger, J.; Ben-Tal, N. A Putative Molecular-Activation Switch in the Transmembrane Domain of ErbB2. Proc. Natl. Acad. Sci. USA 2002, 99, 15937–15940. [Google Scholar] [CrossRef]
- Moriki, T.; Maruyama, H.; Maruyama, I.N. Activation of Preformed EGF Receptor Dimers by Ligand-Induced Rotation of the Transmembrane Domain. J. Mol. Biol. 2001, 311, 1011–1026. [Google Scholar] [CrossRef]
- Burgess, A.W.; Cho, H.-S.; Eigenbrot, C.; Ferguson, K.M.; Garrett, T.P.J.; Leahy, D.J.; Lemmon, M.A.; Sliwkowski, M.X.; Ward, C.W.; Yokoyama, S. An Open-and-Shut Case? Recent Insights into the Activation of EGF/ErbB Receptors. Mol. Cell 2003, 12, 541–552. [Google Scholar] [CrossRef]
- Blume-Jensen, P.; Hunter, T. Oncogenic Kinase Signalling. Nature 2001, 411, 355–365. [Google Scholar] [CrossRef]
- Siwak, D.R.; Carey, M.; Hennessy, B.T.; Nguyen, C.T.; McGahren Murray, M.J.; Nolden, L.; Mills, G.B. Targeting the Epidermal Growth Factor Receptor in Epithelial Ovarian Cancer: Current Knowledge and Future Challenges. J. Oncol. 2010, 2010. [Google Scholar] [CrossRef]
- Veloso, E.S.; Gonçalves, I.N.N.; Silveira, T.L.; Oliveira, F.S.; Vieira, D.S.; Cassali, G.D.; Del Puerto, H.L.; Ferreira, E. Diverse Roles of Epidermal Growth Factors Receptors in Oral and Cutaneous Canine Melanomas. BMC Vet. Res. 2020, 16, 24. [Google Scholar] [CrossRef] [PubMed]
- Koprowski, H.; Herlyn, M.; Balaban, G.; Parmiter, A.; Ross, A.; Nowell, P. Expression of the Receptor for Epidermal Growth Factor Correlates with Increased Dosage of Chromosome 7 in Malignant Melanoma. Somat. Cell Mol. Genet. 1985, 11, 297–302. [Google Scholar] [CrossRef] [PubMed]
- De Wit, P.E.; Moretti, S.; Koenders, P.G.; Weterman, M.A.; van Muijen, G.N.; Gianotti, B.; Ruiter, D.J. Increasing Epidermal Growth Factor Receptor Expression in Human Melanocytic Tumor Progression. J. Investig. Dermatol. 1992, 99, 168–173. [Google Scholar] [CrossRef] [PubMed]
- Huang, T.S.; Rauth, S.; Das Gupta, T.K. Overexpression of EGF Receptor Is Associated with Spontaneous Metastases of a Human Melanoma Cell Line in Nude Mice. Anticancer Res. 1996, 16, 3557–3563. [Google Scholar] [PubMed]
- Mattei, S.; Colombo, M.P.; Melani, C.; Silvani, A.; Parmiani, G.; Herlyn, M. Expression of Cytokine/Growth Factors and Their Receptors in Human Melanoma and Melanocytes. Int. J. Cancer 1994, 56, 853–857. [Google Scholar] [CrossRef] [PubMed]
- Simiczyjew, A.; Pietraszek-Gremplewicz, K.; Dratkiewicz, E.; Podgórska, M.; Matkowski, R.; Ziętek, M.; Nowak, D. Combination of Selected MET and EGFR Inhibitors Decreases Melanoma Cells’ Invasive Abilities. Front. Pharmacol. 2019, 10. [Google Scholar] [CrossRef] [PubMed]
- Pearlman, R.L.; de Oca, M.K.M.; Pal, H.C.; Afaq, F. Potential Therapeutic Targets of Epithelial-Mesenchymal Transition in Melanoma. Cancer Lett. 2017, 391, 125–140. [Google Scholar] [CrossRef]
- Girotti, M.R.; Pedersen, M.; Sanchez-Laorden, B.; Viros, A.; Turajlic, S.; Niculescu-Duvaz, D.; Zambon, A.; Sinclair, J.; Hayes, A.; Gore, M.; et al. Inhibiting EGF Receptor or SRC Family Kinase Signaling Overcomes BRAF Inhibitor Resistance in Melanoma. Cancer Discov. 2013, 3, 158–167. [Google Scholar] [CrossRef]
- Luebker, S.A.; Koepsell, S.A. Diverse Mechanisms of BRAF Inhibitor Resistance in Melanoma Identified in Clinical and Preclinical Studies. Front. Oncol. 2019, 9. [Google Scholar] [CrossRef]
- EGFR Inhibitors—An Overview|ScienceDirect Topics. Available online: https://www.sciencedirect.com/topics/medicine-and-dentistry/egfr-inhibitors (accessed on 29 April 2020).
- Penne, K.; Bohlin, C.; Schneider, S.; Allen, D. Gefitinib (Iressa, ZD1839) and Tyrosine Kinase Inhibitors: The Wave of the Future in Cancer Therapy. Cancer Nurs. 2005, 28, 481–486. [Google Scholar] [CrossRef] [PubMed]
- Djerf, E.A.; Trinks, C.; Abdiu, A.; Thunell, L.K.; Hallbeck, A.-L.; Walz, T.M. ErbB Receptor Tyrosine Kinases Contribute to Proliferation of Malignant Melanoma Cells: Inhibition by Gefitinib (ZD1839). Melanoma Res. 2009, 19, 156–166. [Google Scholar] [CrossRef]
- Patel, S.P.; Kim, K.B.; Papadopoulos, N.E.; Hwu, W.-J.; Hwu, P.; Prieto, V.G.; Bar-Eli, M.; Zigler, M.; Dobroff, A.; Bronstein, Y.; et al. A Phase II Study of Gefitinib in Patients with Metastatic Melanoma. Melanoma Res. 2011, 21, 357–363. [Google Scholar] [CrossRef] [PubMed]
- Wan, X.; Zhu, Y.; Zhang, L.; Hou, W. Gefitinib Inhibits Malignant Melanoma Cells through the VEGF/AKT Signaling Pathway. Mol. Med. Rep. 2018, 17, 7351–7355. [Google Scholar] [CrossRef]
- Kenessey, I.; Kramer, Z.; István, L.; Cserepes, M.T.; Garay, T.; Hegedűs, B.; Dobos, J.; Tímár, J.; Tóvári, J. Inhibition of Epidermal Growth Factor Receptor Improves Antitumor Efficacy of Vemurafenib in BRAF-Mutant Human Melanoma in Preclinical Model. Melanoma Res. 2018, 28, 536–546. [Google Scholar] [CrossRef]
- Schicher, N.; Paulitschke, V.; Swoboda, A.; Kunstfeld, R.; Loewe, R.; Pilarski, P.; Pehamberger, H.; Hoeller, C. Erlotinib and Bevacizumab Have Synergistic Activity against Melanoma. Clin. Cancer Res. 2009, 15, 3495–3502. [Google Scholar] [CrossRef]
- Deng, W.-G.; Kwon, J.; Ekmekcioglu, S.; Poindexter, N.J.; Grimm, E.A. IL-24 Gene Transfer Sensitizes Melanoma Cells to Erlotinib through Modulation of the Apaf-1 and Akt Signaling Pathways. Melanoma Res. 2011, 21, 44–56. [Google Scholar] [CrossRef]
- Dratkiewicz, E.; Pietraszek-Gremplewicz, K.; Simiczyjew, A.; Mazur, A.J.; Nowak, D. Gefitinib or Lapatinib with Foretinib Synergistically Induce a Cytotoxic Effect in Melanoma Cell Lines. Oncotarget 2018, 9, 18254–18268. [Google Scholar] [CrossRef]
- Dratkiewicz, E.; Simiczyjew, A.; Pietraszek-Gremplewicz, K.; Mazurkiewicz, J.; Nowak, D. Characterization of Melanoma Cell Lines Resistant to Vemurafenib and Evaluation of Their Responsiveness to EGFR- and MET-Inhibitor Treatment. Int. J. Mol. Sci. 2019, 21, 113. [Google Scholar] [CrossRef]
- Held, M.A.; Langdon, C.G.; Platt, J.T.; Graham-Steed, T.; Liu, Z.; Chakraborty, A.; Bacchiocchi, A.; Koo, A.; Bosenberg, M.W.; Stern, D.F. Genotype-Selective Combination Therapies for Melanoma Identified by High Throughput Drug Screening. Cancer Discov. 2013, 3, 52–67. [Google Scholar] [CrossRef]
- Das, I.; Wilhelm, M.; Höiom, V.; Franco Marquez, R.; Costa Svedman, F.; Hansson, J.; Tuominen, R.; Egyhàzi Brage, S. Combining ERBB Family and MET Inhibitors Is an Effective Therapeutic Strategy in Cutaneous Malignant Melanoma Independent of BRAF/NRAS Mutation Status. Cell Death Dis. 2019, 10. [Google Scholar] [CrossRef]
- Mudigonda, T.V.; Wyman, K.; Spigel, D.R.; Dahlman, K.B.; Greco, F.A.; Puzanov, I.; Kelley, M.C.; Hainsworth, J.D.; Sosman, J.A.; Johnson, D.B. A Phase II Trial of Erlotinib and Bevacizumab for Patients with Metastatic Melanoma. Pigment Cell Melanoma Res. 2016, 29, 101–103. [Google Scholar] [CrossRef] [PubMed]
- Erlotinib-an Overview ScienceDirect Topics. Available online: https://www.sciencedirect.com/topics/medicine-and-dentistry/erlotinib (accessed on 30 April 2020).
- Cohen, M.H.; Johnson, J.R.; Chen, Y.-F.; Sridhara, R.; Pazdur, R. FDA Drug Approval Summary: Erlotinib (Tarceva) Tablets. Oncologist 2005, 10, 461–466. [Google Scholar] [CrossRef] [PubMed]
- Loss of Novel Mda-7 Splice Variant (Mda-7s) Expression Is Associated with Metastatic Melanoma.-PubMed-NCBI. Available online: https://www.ncbi.nlm.nih.gov/pubmed/15304100 (accessed on 1 May 2020).
- Ekmekcioglu, S.; Ellerhorst, J.A.; Mumm, J.B.; Zheng, M.; Broemeling, L.; Prieto, V.G.; Stewart, A.L.; Mhashilkar, A.M.; Chada, S.; Grimm, E.A. Negative Association of Melanoma Differentiation-Associated Gene (Mda-7) and Inducible Nitric Oxide Synthase (INOS) in Human Melanoma: MDA-7 Regulates INOS Expression in Melanoma Cells. Mol. Cancer Ther. 2003, 2, 9–17. [Google Scholar] [PubMed]
- Chada, S.; Mhashilkar, A.M.; Ramesh, R.; Mumm, J.B.; Sutton, R.B.; Bocangel, D.; Zheng, M.; Grimm, E.A.; Ekmekcioglu, S. Bystander Activity of Ad-Mda7: Human MDA-7 Protein Kills Melanoma Cells via an IL-20 Receptor-Dependent but STAT3-Independent Mechanism. Mol. Ther. 2004, 10, 1085–1095. [Google Scholar] [CrossRef] [PubMed]
- Lebedeva, I.V.; Su, Z.-Z.; Sarkar, D.; Kitada, S.; Dent, P.; Waxman, S.; Reed, J.C.; Fisher, P.B. Melanoma Differentiation Associated Gene-7, Mda-7/Interleukin-24, Induces Apoptosis in Prostate Cancer Cells by Promoting Mitochondrial Dysfunction and Inducing Reactive Oxygen Species. Cancer Res. 2003, 63, 8138–8144. [Google Scholar]
- Ekmekcioglu, S.; Mumm, J.B.; Udtha, M.; Chada, S.; Grimm, E.A. Killing of Human Melanoma Cells Induced by Activation of Class I Interferon-Regulated Signaling Pathways via MDA-7/IL-24. Cytokine 2008, 43, 34–44. [Google Scholar] [CrossRef][Green Version]
- Bocangel, D.; Zheng, M.; Mhashilkar, A.; Liu, Y.; Ramesh, R.; Hunt, K.; Chada, S. Combinatorial Synergy Induced by Adenoviral-Mediated Mda-7 and Herceptin in Her-2 þ Breast Cancer Cells. Cancer Gene Ther. 2006, 13, 958–968. [Google Scholar] [CrossRef][Green Version]
- Emdad, L.; Lebedeva, I.V.; Su, Z.-Z.; Gupta, P.; Sarkar, D.; Settleman, J.; Fisher, P.B. Combinatorial Treatment of Non-Small-Cell Lung Cancers with Gefitinib and Ad.Mda-7 Enhances Apoptosis-Induction and Reverses Resistance to a Single Therapy. J. Cell. Physiol. 2007, 210, 549–559. [Google Scholar] [CrossRef] [PubMed]
- Wood, E.R.; Truesdale, A.T.; McDonald, O.B.; Yuan, D.; Hassell, A.; Dickerson, S.H.; Ellis, B.; Pennisi, C.; Horne, E.; Lackey, K.; et al. A Unique Structure for Epidermal Growth Factor Receptor Bound to GW572016 (Lapatinib): Relationships among Protein Conformation, Inhibitor off-Rate, and Receptor Activity in Tumor Cells. Cancer Res. 2004, 64, 6652–6659. [Google Scholar] [CrossRef] [PubMed]
- Manzano, J.L.; Layos, L.; Bugés, C.; de Los Llanos Gil, M.; Vila, L.; Martínez-Balibrea, E.; Martínez-Cardús, A. Resistant Mechanisms to BRAF Inhibitors in Melanoma. Ann. Transl. Med. 2016, 4, 237. [Google Scholar] [CrossRef] [PubMed]
- Yu, H.A.; Riely, G.J. Second Generation Epidermal Growth Factor Receptor Tyrosine Kinase Inhibitors in Lung Cancers. J. Natl. Compr. Cancer Netw. 2013, 11, 161–169. [Google Scholar] [CrossRef] [PubMed]
- Landi, L.; Cappuzzo, F. Irreversible EGFR-TKIs: Dreaming Perfection. Transl. Lung Cancer Res. 2013, 2, 40–49. [Google Scholar] [CrossRef] [PubMed]
- Dungo, R.T.; Keating, G.M. Afatinib: First Global Approval. Drugs 2013, 73, 1503–1515. [Google Scholar] [CrossRef]
- Cecchi, F.; Rabe, D.C.; Bottaro, D.P. The Hepatocyte Growth Factor Receptor: Structure, Function and Pharmacological Targeting in Cancer. Curr. Signal Transduct. Ther. 2011, 6, 146–151. [Google Scholar] [CrossRef] [PubMed]
- Bottaro, D.P.; Rubin, J.S.; Faletto, D.L.; Chan, A.M.; Kmiecik, T.E.; Vande Woude, G.F.; Aaronson, S.A. Identification of the Hepatocyte Growth Factor Receptor as the C-Met Proto-Oncogene Product. Science 1991, 251, 802–804. [Google Scholar] [CrossRef]
- Ronsin, C.; Muscatelli, F.; Mattei, M.G.; Breathnach, R. A Novel Putative Receptor Protein Tyrosine Kinase of the Met Family. Oncogene 1993, 8, 1195–1202. [Google Scholar]
- Medico, E.; Mongiovi, A.M.; Huff, J.; Jelinek, M.A.; Follenzi, A.; Gaudino, G.; Parsons, J.T.; Comoglio, P.M. The Tyrosine Kinase Receptors Ron and Sea Control “Scattering” and Morphogenesis of Liver Progenitor Cells in Vitro. Mol. Biol. Cell 1996, 7, 495–504. [Google Scholar] [CrossRef] [PubMed]
- Jung, K.H.; Park, B.H.; Hong, S.-S. Progress in Cancer Therapy Targeting C-Met Signaling Pathway. Arch. Pharm. Res. 2012, 35, 595–604. [Google Scholar] [CrossRef]
- Kozlov, G.; Perreault, A.; Schrag, J.D.; Park, M.; Cygler, M.; Gehring, K.; Ekiel, I. Insights into Function of PSI Domains from Structure of the Met Receptor PSI Domain. Biochem. Biophys. Res. Commun. 2004, 321, 234–240. [Google Scholar] [CrossRef] [PubMed]
- Gherardi, E.; Youles, M.E.; Miguel, R.N.; Blundell, T.L.; Iamele, L.; Gough, J.; Bandyopadhyay, A.; Hartmann, G.; Butler, P.J.G. Functional Map and Domain Structure of MET, the Product of the c-Met Protooncogene and Receptor for Hepatocyte Growth Factor/Scatter Factor. Proc. Natl. Acad. Sci. USA 2003, 100, 12039–12044. [Google Scholar] [CrossRef] [PubMed]
- A High Affinity Hepatocyte Growth Factor-Binding Site in the Immunoglobulin-like Region of Met. Available online: https://www.ncbi.nlm.nih.gov/pmc/articles/PMC2475716/ (accessed on 4 May 2020).
- Kong-Beltran, M.; Stamos, J.; Wickramasinghe, D. The Sema Domain of Met Is Necessary for Receptor Dimerization and Activation. Cancer Cell 2004, 6, 75–84. [Google Scholar] [CrossRef]
- Stamos, J.; Lazarus, R.A.; Yao, X.; Kirchhofer, D.; Wiesmann, C. Crystal Structure of the HGF β-Chain in Complex with the Sema Domain of the Met Receptor. EMBO J. 2004, 23, 2325–2335. [Google Scholar] [CrossRef]
- Corso, S.; Comoglio, P.M.; Giordano, S. Cancer Therapy: Can the Challenge Be MET? Trends Mol. Med. 2005, 11, 284–292. [Google Scholar] [CrossRef]
- Zhang, Y.-W.; Vande Woude, G.F. HGF/SF-Met Signaling in the Control of Branching Morphogenesis and Invasion. J. Cell. Biochem. 2003, 88, 408–417. [Google Scholar] [CrossRef]
- Organ, S.L.; Tsao, M.-S. An Overview of the C-MET Signaling Pathway. Ther. Adv. Med. Oncol. 2011, 3, S7–S19. [Google Scholar] [CrossRef] [PubMed]
- Ma, P.C.; Kijima, T.; Maulik, G.; Fox, E.A.; Sattler, M.; Griffin, J.D.; Johnson, B.E.; Salgia, R. C-MET Mutational Analysis in Small Cell Lung Cancer: Novel Juxtamembrane Domain Mutations Regulating Cytoskeletal Functions. Cancer Res. 2003, 63, 6272–6281. [Google Scholar] [PubMed]
- Schmidt, L.; Duh, F.M.; Chen, F.; Kishida, T.; Glenn, G.; Choyke, P.; Scherer, S.W.; Zhuang, Z.; Lubensky, I.; Dean, M.; et al. Germline and Somatic Mutations in the Tyrosine Kinase Domain of the MET Proto-Oncogene in Papillary Renal Carcinomas. Nat. Genet. 1997, 16, 68–73. [Google Scholar] [CrossRef]
- Parikh, R.A.; Wang, P.; Beumer, J.H.; Chu, E.; Appleman, L.J. The Potential Roles of Hepatocyte Growth Factor (HGF)-MET Pathway Inhibitors in Cancer Treatment. OncoTargets Ther. 2014, 7, 969–983. [Google Scholar] [CrossRef]
- Caenepeel, S.; Cooke, K.; Wadsworth, S.; Huang, G.; Robert, L.; Moreno, B.H.; Parisi, G.; Cajulis, E.; Kendall, R.; Beltran, P.; et al. MAPK Pathway Inhibition Induces MET and GAB1 Levels, Priming BRAF Mutant Melanoma for Rescue by Hepatocyte Growth Factor. Oncotarget 2017, 8, 17795–17809. [Google Scholar] [CrossRef] [PubMed]
- Al-U’datt, D.G.F.; Al-Husein, B.A.A.; Qasaimeh, G.R. A Mini-Review of c-Met as a Potential Therapeutic Target in Melanoma. Biomed. Pharmacother. 2017, 88, 194–202. [Google Scholar] [CrossRef]
- Zaidi, M.R.; Fisher, D.E.; Rizos, H. Biology of Melanocytes and Primary Melanoma. In Cutaneous Melanoma; Balch, C.M., Atkins, M.B., Garbe, C., Gershenwald, J.E., Halpern, A.C., Kirkwood, J.M., McArthur, G.A., Thompson, J.F., Sober, A.J., Eds.; Springer International Publishing: Cham, Switzerland, 2020; pp. 3–40. ISBN 978-3-030-05070-2. [Google Scholar]
- Cruz, J.; Reis-Filho, J.S.; Silva, P.; Lopes, J.M. Expression of C-Met Tyrosine Kinase Receptor Is Biologically and Prognostically Relevant for Primary Cutaneous Malignant Melanomas. Oncology 2003, 65, 72–82. [Google Scholar] [CrossRef]
- Moore, S.R.; Persons, D.L.; Sosman, J.A.; Bobadilla, D.; Bedell, V.; Smith, D.D.; Wolman, S.R.; Tuthill, R.J.; Moon, J.; Sondak, V.K.; et al. Detection of Copy Number Alterations in Metastatic Melanoma by a DNA Fluorescence in Situ Hybridization Probe Panel and Array Comparative Genomic Hybridization: A Southwest Oncology Group Study (S9431). Clin. Cancer Res. 2008, 14, 2927–2935. [Google Scholar] [CrossRef]
- Li, G.; Schaider, H.; Satyamoorthy, K.; Hanakawa, Y.; Hashimoto, K.; Herlyn, M. Downregulation of E-Cadherin and Desmoglein 1 by Autocrine Hepatocyte Growth Factor during Melanoma Development. Oncogene 2001, 20, 8125–8135. [Google Scholar] [CrossRef]
- Beuret, L.; Flori, E.; Denoyelle, C.; Bille, K.; Busca, R.; Picardo, M.; Bertolotto, C.; Ballotti, R. Up-Regulation of MET Expression by Alpha-Melanocyte-Stimulating Hormone and MITF Allows Hepatocyte Growth Factor to Protect Melanocytes and Melanoma Cells from Apoptosis. J. Biol. Chem. 2007, 282, 14140–14147. [Google Scholar] [CrossRef]
- Chattopadhyay, C.; Ellerhorst, J.A.; Ekmekcioglu, S.; Greene, V.R.; Davies, M.A.; Grimm, E.A. Association of Activated C-Met with NRAS-Mutated Human Melanomas. Int. J. Cancer 2012, 131, E56–E65. [Google Scholar] [CrossRef] [PubMed]
- Straussman, R.; Morikawa, T.; Shee, K.; Barzily-Rokni, M.; Qian, Z.R.; Du, J.; Davis, A.; Mongare, M.M.; Gould, J.; Frederick, D.T.; et al. Tumour Micro-Environment Elicits Innate Resistance to RAF Inhibitors through HGF Secretion. Nature 2012, 487, 500–504. [Google Scholar] [CrossRef] [PubMed]
- Wilson, T.R.; Fridlyand, J.; Yan, Y.; Penuel, E.; Burton, L.; Chan, E.; Peng, J.; Lin, E.; Wang, Y.; Sosman, J.; et al. Widespread Potential for Growth-Factor-Driven Resistance to Anticancer Kinase Inhibitors. Nature 2012, 487, 505–509. [Google Scholar] [CrossRef]
- Comoglio, P.M.; Giordano, S.; Trusolino, L. Drug Development of MET Inhibitors: Targeting Oncogene Addiction and Expedience. Nat. Rev. Drug Discov. 2008, 7, 504–516. [Google Scholar] [CrossRef]
- Berthou, S.; Aebersold, D.M.; Schmidt, L.S.; Stroka, D.; Heigl, C.; Streit, B.; Stalder, D.; Gruber, G.; Liang, C.; Howlett, A.R.; et al. The Met Kinase Inhibitor SU11274 Exhibits a Selective Inhibition Pattern toward Different Receptor Mutated Variants. Oncogene 2004, 23, 5387–5393. [Google Scholar] [CrossRef] [PubMed]
- Puri, N.; Ahmed, S.; Janamanchi, V.; Tretiakova, M.; Zumba, O.; Krausz, T.; Jagadeeswaran, R.; Salgia, R. C-Met Is a Potentially New Therapeutic Target for Treatment of Human Melanoma. Clin. Cancer Res. 2007, 13, 2246–2253. [Google Scholar] [CrossRef] [PubMed]
- Kenessey, I.; Keszthelyi, M.; Krámer, Z.; Berta, J.; Adám, A.; Dobos, J.; Mildner, M.; Flachner, B.; Cseh, S.; Barna, G.; et al. Inhibition of C-Met with the Specific Small Molecule Tyrosine Kinase Inhibitor SU11274 Decreases Growth and Metastasis Formation of Experimental Human Melanoma. Curr. Cancer Drug Targets 2010, 10, 332–342. [Google Scholar] [CrossRef] [PubMed]
- Kucerova, L.; Demkova, L.; Skolekova, S.; Bohovic, R.; Matuskova, M. Tyrosine Kinase Inhibitor SU11274 Increased Tumorigenicity and Enriched for Melanoma-Initiating Cells by Bioenergetic Modulation. BMC Cancer 2016, 16. [Google Scholar] [CrossRef] [PubMed]
- Etnyre, D.; Stone, A.L.; Fong, J.T.; Jacobs, R.J.; Uppada, S.B.; Botting, G.M.; Rajanna, S.; Moravec, D.N.; Shambannagari, M.R.; Crees, Z.; et al. Targeting C-Met in Melanoma. Cancer Biol. Ther. 2014, 15, 1129–1141. [Google Scholar] [CrossRef] [PubMed]
- Kumar, S.R.; Gajagowni, S.; Bryan, J.N.; Bodenhausen, H.M. Molecular Targets for Tivantinib (ARQ 197) and Vasculogenic Mimicry in Human Melanoma Cells. Eur. J. Pharmacol. 2019, 853, 316–324. [Google Scholar] [CrossRef]
- Cao, H.-H.; Cheng, C.-Y.; Su, T.; Fu, X.-Q.; Guo, H.; Li, T.; Tse, A.K.-W.; Kwan, H.-Y.; Yu, H.; Yu, Z.-L. Quercetin Inhibits HGF/c-Met Signaling and HGF-Stimulated Melanoma Cell Migration and Invasion. Mol. Cancer 2015, 14. [Google Scholar] [CrossRef]
- Parsons, B.M.; Meier, D.R.; Gurda, G.T.; Lofgren, K.A.; Kenny, P.A. Exceptional Response to Crizotinib in an MET-Amplified Triple-Negative Breast Tumor. JCO Precis. Oncol. 2017, 1, 1–6. [Google Scholar] [CrossRef]
- Crizotinib, a c-Met Inhibitor, Prevents Metastasis in a Metastatic Uveal Melanoma Model-PubMed-NCBI. Available online: https://www.ncbi.nlm.nih.gov/pubmed/24140933 (accessed on 6 May 2020).
- Adjei, A.A.; Schwartz, B.; Garmey, E. Early Clinical Development of ARQ 197, a Selective, Non-ATP-Competitive Inhibitor Targeting MET Tyrosine Kinase for the Treatment of Advanced Cancers. Oncologist 2011, 16, 788–799. [Google Scholar] [CrossRef]
- Jeay, S.; Munshi, N.; Hill, J.; Moussa, M.; Ashwell, M.; Leggett, D.; Li, C. ARQ 197, a Highly Selective Small Molecule Inhibitor of c-Met, with Selective Antitumor Properties in a Broad Spectrum of Human Cancer Cells. Cancer Res. 2007, 67, 2369. [Google Scholar]
- Jahangiri, A.; De Lay, M.; Miller, L.M.; Carbonell, W.S.; Hu, Y.-L.; Lu, K.; Tom, M.W.; Paquette, J.; Tokuyasu, T.A.; Tsao, S.; et al. Gene Expression Profile Identifies Tyrosine Kinase C-Met as a Targetable Mediator of Antiangiogenic Therapy Resistance. Clin. Cancer Res. 2013, 19, 1773–1783. [Google Scholar] [CrossRef] [PubMed]
- Puzanov, I.; Sosman, J.; Santoro, A.; Saif, M.W.; Goff, L.; Dy, G.K.; Zucali, P.; Means-Powell, J.A.; Ma, W.W.; Simonelli, M.; et al. Phase 1 Trial of Tivantinib in Combination with Sorafenib in Adult Patients with Advanced Solid Tumors. Investig. New Drugs 2015, 33, 159–168. [Google Scholar] [CrossRef]
- Cao, H.-H.; Tse, A.K.-W.; Kwan, H.-Y.; Yu, H.; Cheng, C.-Y.; Su, T.; Fong, W.-F.; Yu, Z.-L. Quercetin Exerts Anti-Melanoma Activities and Inhibits STAT3 Signaling. Biochem. Pharmacol. 2014, 87, 424–434. [Google Scholar] [CrossRef]
- Labbé, D.; Provençal, M.; Lamy, S.; Boivin, D.; Gingras, D.; Béliveau, R. The Flavonols Quercetin, Kaempferol, and Myricetin Inhibit Hepatocyte Growth Factor-Induced Medulloblastoma Cell Migration. J. Nutr. 2009, 139, 646–652. [Google Scholar] [CrossRef] [PubMed]
- Daud, A.; Kluger, H.M.; Kurzrock, R.; Schimmoller, F.; Weitzman, A.L.; Samuel, T.A.; Moussa, A.H.; Gordon, M.S.; Shapiro, G.I. Phase II Randomised Discontinuation Trial of the MET/VEGF Receptor Inhibitor Cabozantinib in Metastatic Melanoma. Br. J. Cancer 2017, 116, 432–440. [Google Scholar] [CrossRef]
- Wheeler, D.L.; Yarden, Y. Receptor Tyrosine Kinases: Family and Subfamilies; Springer: Berlin, Germany, 2015; ISBN 978-3-319-11888-8. [Google Scholar]
- Claesson-Welsh, L. Vascular Growth Factors and Angiogenesis; Springer Science & Business Media: Berlin, Germany, 2012; ISBN 978-3-642-59953-8. [Google Scholar]
- Shibuya, M.; Claesson-Welsh, L. Signal Transduction by VEGF Receptors in Regulation of Angiogenesis and Lymphangiogenesis. Exp. Cell Res. 2006, 312, 549–560. [Google Scholar] [CrossRef] [PubMed]
- Bruns, A.F.; Herbert, S.P.; Odell, A.F.; Jopling, H.M.; Hooper, N.M.; Zachary, I.C.; Walker, J.H.; Ponnambalam, S. Ligand-Stimulated VEGFR2 Signaling Is Regulated by Co-Ordinated Trafficking and Proteolysis. Traffic 2010, 11, 161–174. [Google Scholar] [CrossRef]
- Davis-Smyth, T.; Chen, H.; Park, J.; Presta, L.G.; Ferrara, N. The Second Immunoglobulin-like Domain of the VEGF Tyrosine Kinase Receptor Flt-1 Determines Ligand Binding and May Initiate a Signal Transduction Cascade. EMBO J. 1996, 15, 4919–4927. [Google Scholar] [CrossRef]
- Sawano, A.; Takahashi, T.; Yamaguchi, S.; Aonuma, M.; Shibuya, M. Flt-1 but Not KDR/Flk-1 Tyrosine Kinase Is a Receptor for Placenta Growth Factor, Which Is Related to Vascular Endothelial Growth Factor. Cell Growth Differ. 1996, 7, 213–221. [Google Scholar]
- Rahimi, N. VEGFR-1 and VEGFR-2: Two Non-Identical Twins with a Unique Physiognomy. Front. Biosci. 2006, 11, 818–829. [Google Scholar] [CrossRef] [PubMed]
- Autiero, M.; Waltenberger, J.; Communi, D.; Kranz, A.; Moons, L.; Lambrechts, D.; Kroll, J.; Plaisance, S.; De Mol, M.; Bono, F.; et al. Role of PlGF in the Intra- and Intermolecular Cross Talk between the VEGF Receptors Flt1 and Flk1. Nat. Med. 2003, 9, 936–943. [Google Scholar] [CrossRef] [PubMed]
- Pajusola, K.; Aprelikova, O.; Pelicci, G.; Weich, H.; Claesson-Welsh, L.; Alitalo, K. Signalling Properties of FLT4, a Proteolytically Processed Receptor Tyrosine Kinase Related to Two VEGF Receptors. Oncogene 1994, 9, 3545–3555. [Google Scholar] [PubMed]
- Shinkai, A.; Ito, M.; Anazawa, H.; Yamaguchi, S.; Shitara, K.; Shibuya, M. Mapping of the Sites Involved in Ligand Association and Dissociation at the Extracellular Domain of the Kinase Insert Domain-Containing Receptor for Vascular Endothelial Growth Factor. J. Biol. Chem. 1998, 273, 31283–31288. [Google Scholar] [CrossRef] [PubMed]
- Tanaka, K.; Yamaguchi, S.; Sawano, A.; Shibuya, M. Characterization of the Extracellular Domain in Vascular Endothelial Growth Factor Receptor-1 (Flt-1 Tyrosine Kinase). Jpn. J. Cancer Res. 1997, 88, 867–876. [Google Scholar] [CrossRef]
- Ruch, C.; Skiniotis, G.; Steinmetz, M.O.; Walz, T.; Ballmer-Hofer, K. Structure of a VEGF-VEGF Receptor Complex Determined by Electron Microscopy. Nat. Struct. Mol. Biol. 2007, 14, 249–250. [Google Scholar] [CrossRef] [PubMed]
- Holmes, K.; Roberts, O.L.; Thomas, A.M.; Cross, M.J. Vascular Endothelial Growth Factor Receptor-2: Structure, Function, Intracellular Signalling and Therapeutic Inhibition. Cell. Signal. 2007, 19, 2003–2012. [Google Scholar] [CrossRef] [PubMed]
- Manni, S. Structural and Functional Characterization of the Intracellular Kinase Domain of Vascular Endothelial Growth Factor Receptor-2. Ph.D. Thesis, University of Basel, Basel, Switzerland, 21 May 2013. [Google Scholar]
- Shibuya, M. Vascular Endothelial Growth Factor (VEGF) and Its Receptor (VEGFR) Signaling in Angiogenesis. Genes Cancer 2011, 2, 1097–1105. [Google Scholar] [CrossRef]
- Rajabi, P.; Neshat, A.; Mokhtari, M.; Rajabi, M.A.; Eftekhari, M.; Tavakoli, P. The Role of VEGF in Melanoma Progression. J. Res. Med. Sci. 2012, 17, 534–539. [Google Scholar] [PubMed]
- Sini, P.; Baffert, F.; Wood, J.; Hynes, N.E. Role of VEGFR Signaling in Tumor Cells. Cancer Res. 2006, 66, 659. [Google Scholar]
- Simonetti, O.; Lucarini, G.; Brancorsini, D.; Nita, P.; Bernardini, M.L.; Biagini, G.; Offidani, A. Immunohistochemical Expression of Vascular Endothelial Growth Factor, Matrix Metalloproteinase 2, and Matrix Metalloproteinase 9 in Cutaneous Melanocytic Lesions. Cancer 2002, 95, 1963–1970. [Google Scholar] [CrossRef] [PubMed]
- Potti, A.; Moazzam, N.; Tendulkar, K.; Javed, N.A.; Koch, M.; Kargas, S. Immunohistochemical Determination of Vascular Endothelial Growth Factor (VEGF) Overexpression in Malignant Melanoma. Anticancer Res. 2003, 23, 4023–4026. [Google Scholar] [PubMed]
- Graells, J.; Vinyals, A.; Figueras, A.; Llorens, A.; Moreno, A.; Marcoval, J.; Gonzalez, F.J.; Fabra, A. Overproduction of VEGF Concomitantly Expressed with Its Receptors Promotes Growth and Survival of Melanoma Cells through MAPK and PI3K Signaling. J. Investig. Dermatol. 2004, 123, 1151–1161. [Google Scholar] [CrossRef] [PubMed]
- Goydos, J.S.; Gorski, D.H. Vascular Endothelial Growth Factor C MRNA Expression Correlates with Stage of Progression in Patients with Melanoma. Clin. Cancer Res. 2003, 9, 5962–5967. [Google Scholar]
- Ugurel, S.; Rappl, G.; Tilgen, W.; Reinhold, U. Increased Serum Concentration of Angiogenic Factors in Malignant Melanoma Patients Correlates with Tumor Progression and Survival. J. Clin. Oncol. 2001, 19, 577–583. [Google Scholar] [CrossRef]
- Atzori, M.G.; Ceci, C.; Ruffini, F.; Trapani, M.; Barbaccia, M.L.; Tentori, L.; D’Atri, S.; Lacal, P.M.; Graziani, G. Role of VEGFR-1 in Melanoma Acquired Resistance to the BRAF Inhibitor Vemurafenib. J. Cell Mol. Med. 2020, 24, 465–475. [Google Scholar] [CrossRef]
- Jain, R.K. Normalizing Tumor Vasculature with Anti-Angiogenic Therapy: A New Paradigm for Combination Therapy. Nat. Med. 2001, 7, 987–989. [Google Scholar] [CrossRef]
- Mendel, D.B.; Schreck, R.E.; West, D.C.; Li, G.; Strawn, L.M.; Tanciongco, S.S.; Vasile, S.; Shawver, L.K.; Cherrington, J.M. The Angiogenesis Inhibitor SU5416 Has Long-Lasting Effects on Vascular Endothelial Growth Factor Receptor Phosphorylation and Function. Clin. Cancer Res. 2000, 6, 4848–4858. [Google Scholar] [PubMed]
- Mendel, D.B.; Laird, A.D.; Smolich, B.D.; Blake, R.A.; Liang, C.; Hannah, A.L.; Shaheen, R.M.; Ellis, L.M.; Weitman, S.; Shawver, L.K.; et al. Development of SU5416, a Selective Small Molecule Inhibitor of VEGF Receptor Tyrosine Kinase Activity, as an Anti-Angiogenesis Agent. Anticancer Drug Des. 2000, 15, 29–41. [Google Scholar]
- Kuenen, B.C.; Tabernero, J.; Baselga, J.; Cavalli, F.; Pfanner, E.; Conte, P.F.; Seeber, S.; Madhusudan, S.; Deplanque, G.; Huisman, H.; et al. Efficacy and Toxicity of the Angiogenesis Inhibitor SU5416 as a Single Agent in Patients with Advanced Renal Cell Carcinoma, Melanoma, and Soft Tissue Sarcoma. Clin. Cancer Res. 2003, 9, 1648–1655. [Google Scholar]
- Mita, M.M.; Rowinsky, E.K.; Forero, L.; Eckhart, S.G.; Izbicka, E.; Weiss, G.R.; Beeram, M.; Mita, A.C.; de Bono, J.S.; Tolcher, A.W.; et al. A Phase II, Pharmacokinetic, and Biologic Study of Semaxanib and Thalidomide in Patients with Metastatic Melanoma. Cancer Chemother. Pharmacol. 2007, 59, 165–174. [Google Scholar] [CrossRef]
- Hu-Lowe, D.D.; Zou, H.Y.; Grazzini, M.L.; Hallin, M.E.; Wickman, G.R.; Amundson, K.; Chen, J.H.; Rewolinski, D.A.; Yamazaki, S.; Wu, E.Y.; et al. Nonclinical Antiangiogenesis and Antitumor Activities of Axitinib (AG-013736), an Oral, Potent, and Selective Inhibitor of Vascular Endothelial Growth Factor Receptor Tyrosine Kinases 1, 2, 3. Clin. Cancer Res. 2008, 14, 7272–7283. [Google Scholar] [CrossRef]
- Fruehauf, J.; Lutzky, J.; McDermott, D.; Brown, C.K.; Meric, J.-B.; Rosbrook, B.; Shalinsky, D.R.; Liau, K.F.; Niethammer, A.G.; Kim, S.; et al. Multicenter, Phase II Study of Axitinib, a Selective Second-Generation Inhibitor of Vascular Endothelial Growth Factor Receptors 1, 2, and 3, in Patients with Metastatic Melanoma. Clin. Cancer Res. 2011, 17, 7462–7469. [Google Scholar] [CrossRef]
- Bose, A.; Lowe, D.B.; Rao, A.; Storkus, W.J. Combined Vaccine + Axitinib Therapy Yields Superior Anti-Tumor Efficacy in a Murine Melanoma Model. Melanoma Res. 2012, 22, 236–243. [Google Scholar] [CrossRef]
- Algazi, A.P.; Cha, E.; Ortiz-Urda, S.M.; McCalmont, T.; Bastian, B.C.; Hwang, J.; Pampaloni, M.H.; Behr, S.; Chong, K.; Cortez, B.; et al. The Combination of Axitinib Followed by Paclitaxel/Carboplatin Yields Extended Survival in Advanced BRAF Wild-Type Melanoma: Results of a Clinical/Correlative Prospective Phase II Clinical Trial. Br. J. Cancer 2015, 112, 1326–1331. [Google Scholar] [CrossRef]
- Study of the Anti-Angiogenesis Agent Axitinib in Patients with Stage III Malignant Melanoma-Full Text View-ClinicalTrials.Gov. Available online: https://clinicaltrials.gov/ct2/show/NCT01321437 (accessed on 18 May 2020).
- Guo, J. A Monocentric, Single-Arm, Open, Phase II Clinical Study on the Efficacy and Safety of Toripalimab Combined with Axitinib as Neoadjuvant Therapy in Patients with Localized Mucosal Melanoma. Available online: clinicaltrials.gov (accessed on 18 May 2020).
- Varker, K.A.; Biber, J.E.; Kefauver, C.; Jensen, R.; Lehman, A.; Young, D.; Wu, H.; Lesinski, G.B.; Kendra, K.; Chen, H.X.; et al. A Randomized Phase 2 Trial of Bevacizumab with or without Daily Low-Dose Interferon Alfa-2b in Metastatic Malignant Melanoma. Ann. Surg. Oncol. 2007, 14, 2367–2376. [Google Scholar] [CrossRef]
- Grignol, V.P.; Olencki, T.; Relekar, K.; Taylor, C.; Kibler, A.; Kefauver, C.; Wei, L.; Walker, M.J.; Chen, H.X.; Kendra, K.; et al. A Phase II Trial of Bevacizumab and High Dose Interferon Alpha-2B in Metastatic Melanoma. J. Immunother. 2011, 34, 509–515. [Google Scholar] [CrossRef] [PubMed]
- Kim, K.B.; Sosman, J.A.; Fruehauf, J.P.; Linette, G.P.; Markovic, S.N.; McDermott, D.F.; Weber, J.S.; Nguyen, H.; Cheverton, P.; Chen, D.; et al. BEAM: A Randomized Phase II Study Evaluating the Activity of Bevacizumab in Combination with Carboplatin Plus Paclitaxel in Patients with Previously Untreated Advanced Melanoma. J. Clin. Oncol. 2012, 30, 34–41. [Google Scholar] [CrossRef]
- Corrie, P.G.; Marshall, A.; Nathan, P.D.; Lorigan, P.; Gore, M.; Tahir, S.; Faust, G.; Kelly, C.G.; Marples, M.; Danson, S.J.; et al. Adjuvant Bevacizumab for Melanoma Patients at High Risk of Recurrence: Survival Analysis of the AVAST-M Trial. Ann. Oncol. 2018, 29, 1843–1852. [Google Scholar] [CrossRef] [PubMed]
- Simonsen, T.G.; Gaustad, J.-V.; Rofstad, E.K. Bevacizumab Treatment of Meningeal Melanoma Metastases. J. Transl. Med. 2020, 18, 13. [Google Scholar] [CrossRef] [PubMed]
- Yakes, F.M.; Chen, J.; Tan, J.; Yamaguchi, K.; Shi, Y.; Yu, P.; Qian, F.; Chu, F.; Bentzien, F.; Cancilla, B.; et al. Cabozantinib (XL184), a Novel MET and VEGFR2 Inhibitor, Simultaneously Suppresses Metastasis, Angiogenesis, and Tumor Growth. Mol. Cancer Ther. 2011, 10, 2298–2308. [Google Scholar] [CrossRef]
- Grüllich, C. Cabozantinib: Multi-Kinase Inhibitor of MET, AXL, RET, and VEGFR2. Recent Results Cancer Res. 2018, 211, 67–75. [Google Scholar] [CrossRef]
- Kurzrock, R.; Sherman, S.I.; Ball, D.W.; Forastiere, A.A.; Cohen, R.B.; Mehra, R.; Pfister, D.G.; Cohen, E.E.W.; Janisch, L.; Nauling, F.; et al. Activity of XL184 (Cabozantinib), an Oral Tyrosine Kinase Inhibitor, in Patients with Medullary Thyroid Cancer. J. Clin. Oncol. 2011, 29, 2660–2666. [Google Scholar] [CrossRef]
- Vergani, E.; Vallacchi, V.; Frigerio, S.; Deho, P.; Mondellini, P.; Perego, P.; Cassinelli, G.; Lanzi, C.; Testi, M.A.; Rivoltini, L.; et al. Identification of MET and SRC Activation in Melanoma Cell Lines Showing Primary Resistance to PLX4032. Neoplasia 2011, 13, 1132–1142. [Google Scholar] [CrossRef]
- Cabozantinib and Pembrolizumab for Advanced Metastatic Melanoma-Full Text View-ClinicalTrials.Gov. Available online: https://clinicaltrials.gov/ct2/show/NCT03957551 (accessed on 4 September 2020).
- Nivolumab/Ipilimumab Plus Cabozantinib in Patients with Unresectable Advanced Melanoma-Full Text View-ClinicalTrials.Gov. Available online: https://clinicaltrials.gov/ct2/show/NCT04091750 (accessed on 4 September 2020).
- Dufies, M.; Jacquel, A.; Robert, G.; Cluzeau, T.; Puissant, A.; Fenouille, N.; Legros, L.; Raynaud, S.; Cassuto, J.-P.; Luciano, F.; et al. Mechanism of Action of the Multikinase Inhibitor Foretinib. Cell Cycle 2011, 10, 4138–4148. [Google Scholar] [CrossRef] [PubMed]
- Qian, F.; Engst, S.; Yamaguchi, K.; Yu, P.; Won, K.-A.; Mock, L.; Lou, T.; Tan, J.; Li, C.; Tam, D.; et al. Inhibition of Tumor Cell Growth, Invasion, and Metastasis by EXEL-2880 (XL880, GSK1363089), a Novel Inhibitor of HGF and VEGF Receptor Tyrosine Kinases. Cancer Res. 2009, 69, 8009–8016. [Google Scholar] [CrossRef]
- Wang, W.; Li, Q.; Takeuchi, S.; Yamada, T.; Koizumi, H.; Nakamura, T.; Matsumoto, K.; Mukaida, N.; Nishioka, Y.; Sone, S.; et al. Met Kinase Inhibitor E7050 Reverses Three Different Mechanisms of Hepatocyte Growth Factor-Induced Tyrosine Kinase Inhibitor Resistance in EGFR Mutant Lung Cancer. Clin. Cancer Res. 2012, 18, 1663–1671. [Google Scholar] [CrossRef]
- E7050 in Combination With E7080 in Subjects With Advanced Solid Tumors (Dose Escalation) and in Subjects With Recurrent Glioblastoma or Unresectable Stage III or Stage IV Melanoma After Prior Systemic Therapy (Expansion Cohort and Phase 2)-No Study Results Posted-ClinicalTrials.Gov. Available online: https://clinicaltrials.gov/ct2/show/results/NCT01433991 (accessed on 8 May 2020).
- Adams, T.E.; Epa, V.C.; Garrett, T.P.; Ward, C.W. Structure and Function of the Type 1 Insulin-like Growth Factor Receptor. Cell. Mol. Life Sci. 2000, 57, 1050–1093. [Google Scholar] [CrossRef]
- Li, J.; Choi, E.; Yu, H.; Bai, X.-C. Structural Basis of the Activation of Type 1 Insulin-like Growth Factor Receptor. Nat. Commun. 2019, 10, 4567. [Google Scholar] [CrossRef] [PubMed]
- Kanter-Lewensohn, L.; Dricu, A.; Girnita, L.; Wejde, J.; Larsson, O. Expression of Insulin-like Growth Factor-1 Receptor (IGF-1R) and P27Kip1 in Melanocytic Tumors: A Potential Regulatory Role of IGF-1 Pathway in Distribution of P27Kip1 between Different Cyclins. Growth Factors 2000, 17, 193–202. [Google Scholar] [CrossRef] [PubMed]
- Constitutive Expression of Multiple Growth Factor Genes by Melanoma Cells but Not Normal Melanocytes-PubMed. Available online: https://pubmed.ncbi.nlm.nih.gov/2056188/ (accessed on 9 March 2021).
- Hilmi, C.; Larribere, L.; Giuliano, S.; Bille, K.; Ortonne, J.-P.; Ballotti, R.; Bertolotto, C. IGF1 Promotes Resistance to Apoptosis in Melanoma Cells through an Increased Expression of BCL2, BCL-X(L), and Survivin. J. Investig. Dermatol. 2008, 128, 1499–1505. [Google Scholar] [CrossRef]
- Wang, J.; Sinnberg, T.; Niessner, H.; Dölker, R.; Sauer, B.; Kempf, W.E.; Meier, F.; Leslie, N.; Schittek, B. PTEN Regulates IGF-1R-Mediated Therapy Resistance in Melanoma. Pigment Cell Melanoma Res. 2015, 28, 572–589. [Google Scholar] [CrossRef]
- Schlessinger, J. Receptor Tyrosine Kinases: Legacy of the First Two Decades. Cold Spring Harb. Perspect. Biol. 2014, 6. [Google Scholar] [CrossRef]
- Ornitz, D.M.; Itoh, N. The Fibroblast Growth Factor Signaling Pathway. Wiley Interdiscip. Rev. Dev. Biol. 2015, 4, 215–266. [Google Scholar] [CrossRef]
- Freier, K.; Schwaenen, C.; Sticht, C.; Flechtenmacher, C.; Mühling, J.; Hofele, C.; Radlwimmer, B.; Lichter, P.; Joos, S. Recurrent FGFR1 Amplification and High FGFR1 Protein Expression in Oral Squamous Cell Carcinoma (OSCC). Oral Oncol. 2007, 43, 60–66. [Google Scholar] [CrossRef]
- Wang, Y.; Becker, D. Antisense Targeting of Basic Fibroblast Growth Factor and Fibroblast Growth Factor Receptor-1 in Human Melanomas Blocks Intratumoral Angiogenesis and Tumor Growth. Nat. Med. 1997, 3, 887–893. [Google Scholar] [CrossRef]
- Li, L.; Zhang, S.; Li, H.; Chou, H. FGFR3 Promotes the Growth and Malignancy of Melanoma by Influencing EMT and the Phosphorylation of ERK, AKT, and EGFR. BMC Cancer 2019, 19, 963. [Google Scholar] [CrossRef]
- Czyz, M. Fibroblast Growth Factor Receptor Signaling in Skin Cancers. Cells 2019, 8. [Google Scholar] [CrossRef]
- Pfizer The LOGIC 2 Trial A Phase II, Multi-Center, Open-Label Study of Sequential LGX818/MEK162 Combination Followed by a Rational Combination with Targeted Agents After Progression, to Overcome Resistance in Adult Patients with Locally Advanced or Metastatic BRAF V600 Melanoma. Available online: clinicaltrials.gov (accessed on 18 May 2020).
- Zitvogel, L.; Rusakiewicz, S.; Routy, B.; Ayyoub, M.; Kroemer, G. Immunological Off-Target Effects of Imatinib. Nat. Rev. Clin. Oncol. 2016, 13, 431–446. [Google Scholar] [CrossRef] [PubMed]
- Finke, J.H.; Rini, B.; Ireland, J.; Rayman, P.; Richmond, A.; Golshayan, A.; Wood, L.; Elson, P.; Garcia, J.; Dreicer, R.; et al. Sunitinib Reverses Type-1 Immune Suppression and Decreases T-Regulatory Cells in Renal Cell Carcinoma Patients. Clin. Cancer Res. 2008, 14, 6674–6682. [Google Scholar] [CrossRef] [PubMed]
- De Lavallade, H.; Khoder, A.; Hart, M.; Sarvaria, A.; Sekine, T.; Alsuliman, A.; Mielke, S.; Bazeos, A.; Stringaris, K.; Ali, S.; et al. Tyrosine Kinase Inhibitors Impair B-Cell Immune Responses in CML through off-Target Inhibition of Kinases Important for Cell Signaling. Blood 2013, 122, 227–238. [Google Scholar] [CrossRef] [PubMed]
- Dörfel, D.; Lechner, C.J.; Joas, S.; Funk, T.; Gutknecht, M.; Salih, J.; Geiger, J.; Kropp, K.N.; Maurer, S.; Müller, M.R.; et al. The BCR-ABL Inhibitor Nilotinib Influences Phenotype and Function of Monocyte-Derived Human Dendritic Cells. Cancer Immunol. Immunother. 2018, 67, 775–783. [Google Scholar] [CrossRef]
- MacDonald, F.; Zaiss, D.M.W. The Immune System’s Contribution to the Clinical Efficacy of EGFR Antagonist Treatment. Front. Pharmacol. 2017, 8. [Google Scholar] [CrossRef]
- Pollack, B.P. EGFR Inhibitors, MHC Expression and Immune Responses: Can EGFR Inhibitors Be Used as Immune Response Modifiers? Oncoimmunology 2012, 1, 71–74. [Google Scholar] [CrossRef] [PubMed]
- Faivre, S.; Delbaldo, C.; Vera, K.; Robert, C.; Lozahic, S.; Lassau, N.; Bello, C.; Deprimo, S.; Brega, N.; Massimini, G.; et al. Safety, Pharmacokinetic, and Antitumor Activity of SU11248, a Novel Oral Multitarget Tyrosine Kinase Inhibitor, in Patients with Cancer. J. Clin. Oncol. 2006, 24, 25–35. [Google Scholar] [CrossRef]
- Van Cruijsen, H.; Van Der Veldt, A.A.; Vroling, L.; Oosterhoff, D.; Broxterman, H.J.; Scheper, R.J.; Giaccone, G.; Haanen, J.B.; Eertwegh, A.J.V.D.; Boven, E.; et al. Sunitinib-Induced Myeloid Lineage Redistribution in Renal Cell Cancer Patients: CD1c+ Dendritic Cell Frequency Predicts Progression-Free Survival. Clin. Cancer Res. 2008, 14, 5884–5892. [Google Scholar] [CrossRef]
- Papaccio, F.; Della Corte, C.M.; Viscardi, G.; Di Liello, R.; Esposito, G.; Sparano, F.; Ciardiello, F.; Morgillo, F. HGF/MET and the Immune System: Relevance for Cancer Immunotherapy. Int. J. Mol. Sci. 2018, 19. [Google Scholar] [CrossRef]
- Glodde, N.; Bald, T.; van den Boorn-Konijnenberg, D.; Nakamura, K.; O’Donnell, J.S.; Szczepanski, S.; Brandes, M.; Eickhoff, S.; Das, I.; Shridhar, N.; et al. Reactive Neutrophil Responses Dependent on the Receptor Tyrosine Kinase C-MET Limit Cancer Immunotherapy. Immunity 2017, 47, 789–802.e9. [Google Scholar] [CrossRef] [PubMed]
- Im, J.S.; Herrmann, A.C.; Bernatchez, C.; Haymaker, C.; Molldrem, J.J.; Hong, W.K.; Perez-Soler, R. Immune-Modulation by Epidermal Growth Factor Receptor Inhibitors: Implication on Anti-Tumor Immunity in Lung Cancer. PLoS ONE 2016, 11. [Google Scholar] [CrossRef] [PubMed]
- Farsaci, B.; Higgins, J.P.; Hodge, J.W. Consequence of Dose Scheduling of Sunitinib on Host Immune Response Elements and Vaccine Combination Therapy. Int. J. Cancer 2012, 130, 1948–1959. [Google Scholar] [CrossRef] [PubMed]
- Motzer, R.J.; Escudier, B.; McDermott, D.F.; George, S.; Hammers, H.J.; Srinivas, S.; Tykodi, S.S.; Sosman, J.A.; Procopio, G.; Plimack, E.R.; et al. Nivolumab versus Everolimus in Advanced Renal Cell Carcinoma. N. Engl. J. Med. 2015, 373, 1803–1813. [Google Scholar] [CrossRef] [PubMed]

| RTKi | Cell Lines Used | Results | Reference |
|---|---|---|---|
| Imatinib | A375SM (from pooled lung metastases by A375 cells (V600EBRAF), intravenously injected to nude mice MeWo cells (WTBRAF, WTNRAS) injected into nude mice | -Imatinib did not affect A375SM and MeWo growth in vivo but inhibits PDGFR-α and PDGFR-β phosphorylation in A375SM xenografts. | McGary, E.C., et al. 2004 [37] |
| Imatinib | B16F10 murine melanoma cells in C57B16 mice | -Imatinib inhibits B16F10 melanoma cell proliferation and growth in a mouse model. | Redondo, P., et al. 2004 [38] |
| Imatinib | M6 (V559Ac-KIT), M40 (WTc-KIT), GIST 882(K642Ec-KIT) | -Imatinib inhibits cell proliferation, MAPK, P3K/AKT, STAT pathways, favors G1 arrest, enhances apoptosis, and reduces cyclin D1, in M6 and GIST 882 cells. -Imatinib reduces BCL-2, MCL-1, ML-IAP, and survivin in both M6 and GIST882 cells. | Jiang, X., et al. 2008 [44] |
| Imatinib/ nilotinib | SKMel28 (V600EBRAF), M230 (L576Pc-KIT), IMR_A829P (imatinib-resistant clone) NR T670I (nilotinib-resistant clone) | -M230 but not SKMel28 melanoma cells are sensitive to imatinib and nilotinib. -Drug-resistant clones exhibit secondary c-KIT mutations and retain c-KIT activation even in the presence of inhibitors. -IMR_A829P cells retains a strong apoptotic response to nilotinib and dasatinib. -NR_T670I cells undergo significant apoptosis in response to sunitinib. | Todd, J.R., et al. 2013 [55] |
| Nilotinib | M230 (L576Pc-KIT) | -Nilotinib inhibits cell proliferation and reduces STAT3 signaling. | Delyon, J., et al. 2018 [56] |
| Dasatinib | Lox-IMVI (V600EBRAF), Malme-3M (V600EBRAF), Sk-Mel-5 (V600EBRAF), Sk-Mel-28 (V600EBRAF) and HT144 (V600EBRAF) | -Dasatinib inhibits growth of Lox-IMVI, Malme-3M, HT144, cell migration and invasion in HT144 and Sk-Mel-28. -Dasatinib increases apoptosis in Lox-IMVI, Malme-3M, favors G1 arrest in Lox-IMVI and HT144 and enhances response to temozolomide in HT144, Malme-3M and Lox-IMVI. | Eustace, A.J., et al. 2008 [57] |
| Dasatinib | MeWo (WTBRAF, WTNRAS), SK-Mel-5 (V600EBRAF), SK-Mel-28 (V600EBRAF), A375 (V600EBRAF), A2058 (V600EBRAF), G361(V599EBRAF), 1205-Lu (V600EBRAF) 451-Lu cells (V600EBRAF) | -Dasatinib inhibits migration and invasion in 1205-Lu and A2058 cell lines. -Dasatinib decreases MMP9, inhibits EphA2 kinase activity and blocks SFK in A2058 cells. | Buettner, R., et al. 2008 [58] |
| Dasatinib | WM3211 (L576Pc-KIT), A375 (V600EBRAF), MeWo (WTBRAF, WTNRAS) | -L576P mutation induces structural changes in KIT that reduce imatinib affinity. -Dasatinib alone reduces cell viability of the L576P mutant cell line. | Woodman, S.E., et al. 2009 [59] |
| Dasatinib | A375(V600EBRAF), Sk-Mel-5 (V600EBRAF), Sk-Mel-28 (V600EBRAF) | -Dasatinib inhibits growth of melanoma cell lines and synergized with cisplatin | Homsi, J., et al., 2009 [60] |
| Dasatinib | Mel-p (primary melanoma), A375 (V600EBRAF, Q61KNRAS) | -Dasatinib leads to growth inhibition of Mel-p. -Dasatinib induces cell differentiation, remodels the actin cytoskeleton, and inhibits nuclear translocation of ERK1/2 | Wu, J., et al. 2013 [61] |
| Dasatinib | Lox-IMVI (V600EBRAF), Malme-3M (V600EBRAF), M14(V600EBRAF), Sk-Mel-5 (V600EBRAF), Sk-Mel-28 (V600EBRAF) | -Lox-IMV, WM-115 and HT144 cells showed sensitivity to dasatinib. -Malme-3M, WM266-4, M14, Sk-Mel-28 and Sk-Mel-5 cells were resistant to dasatinib. -High protein expression of ANXA1, CAV-1 or EphA2 in the sensitive melanoma cells that predicts sensitivity to dasatinib | Eustace, A.J., et al. 2014 [62] |
| Dasatinib | MDA-MB-435S, A375(V600EBRAF, Q61KNRAS), WM853 (V600EBRAF) | -SIRT2 silencing renders melanoma cells more sensitive to dasatinib. -SIRT2 loss enhances dasatinib effect on cell migration inhibition and cell cycle arrest. | Karwaciak, I., et al. 2019 [63] |
| Sunitinib | A-07 and R-18 human melanoma cells transfected with GFP in female BALB/c nu/nu mice | -Sunitinib increases hypoxia, vessel segment length, and median vessel diameter, does not affect blood supply time (BST), vascular basement membrane, vessel tortuosity and pericyte-coverage but reduces vessel density. -Prolonged exposure, reduces tumor growth. | Gaustad, J.-V., et al. 2012 [64] |
| Sunitinib | Amelanotic human melanoma A-07 in female BALB/c-nu/nu mice | -Sunitinib treatment does not affect tumor growth but increases microvascular density (MVD), hypoxia, necrosis, and ADC, but reduces K trans. | Gaustad, J.-V., et al. 2013 [65] |
| Ponatinib | KITWT, KITV560D, KITK642E, KITD816V PDX mice | -Ponatinib reduces cell viability, KIT, AKT, ERK phosphorylation, cell proliferation, tumor growth and induces apoptosis in the KIT mutant PDX in vitro and in vivo with high affinity to KITD816V. | Han, Y., et al. 2019 [66] |
| RTKi | Phase/Year Published or Presented | Population | N | Dose | Survival | Response | Adverse Reactions |
|---|---|---|---|---|---|---|---|
| Imatinib | Phase II, Ugurel, S., et al. 2005 [39] | Median age of 54.2 years (range, 38.9–72.0) years | 18 | 800 mg/day | Median OS and PFS = 3.9 and 1.9 months, respectively | No objective responses | Severe (AE)s of grade 3 and 4: Exanthema, Constipation, intestinal perforation, arterial thromboembolism, suicide attempt |
| Imatinib | Phase II, Wyman, K., et al. 2006 [41] | Median age of 59 years (range, 37–82) years | 26 | 800 mg/day | Median OS and PFS = 6.5 and two months respectively | No objective responses | Grade 3 and grade 4 toxicity: Gastrointestinal toxicities, nausea and emesis |
| Imatinib | Phase II, Kim, K.B., et al. 2008 [42] | Median age of 58 years (range, 33–83) years | 21 | 400 mg (twice daily) | Median OS and PFS = 7.5 and 1.4 months respectively | 4 SD and 1 PR | Common toxicity of grade 3 or 4: Fatigue and oedema |
| Imatinib | Phase II, Carvajal, R.D., et al. 2011 [31] | Median age of 71 (range, 49–88) years | 28 | 400 mg (twice daily) | Median OS and PFS = 10.7 and 2.8 months, respectively | 2 CRs, 2 PRs, 2 transient PRs, and 5 SD | |
| Imatinib | Phase II trial, Guo, J., et al. 2011 [45] | Median age of 57 (range, 27–76) years | 43 | 400 mg/d | Median OS and PFS = 15 and 9 months, respectively. | 10 PRs, 13 SD and 18 showed tumor regression | Common AEs: edema, fatigue, anorexia, nausea, neutropenia, elevated AST ALT |
| Imatinib | Phase II trial, Hodi, F.S., et al. 2013 [32] | Median age of 65 (range, 42–84) years | 25 | 400 mg/day | Median OS and TTP = 12.5 and 3.7 months, respectively | 7 patients achieved CRs or PRs | The common reported (AE)s: nausea, fatigue, anemia, hyperglycemia, and vomiting |
| Imatinib + Bevacizumab (Bevax) | Phase I/II trial, Flaherty, K., et al. 2015 [48] | Median age of 63 (range, 49–86) years | 23 | Bevax 10 mg/kg + imatinib 400 or 600 or twice 800 mg | The median PFS = 7.7 weeks | A PR was observed in 1 patient and 7 showed SD | Common toxicities: fatigue, nausea, vomiting, edema, proteinuria, and anemia, but were not commonly severe |
| Imatinib + Ipilimumab (IPI) | Phase I Reilley, M.J., et al. 2017 [50] | Median age of 55 years, showing KIT positive tumour | 7 | 400 mg imatinib (one or twice daily) +IPI 1 mg/kg/3 mg/kg) on day 1 of each 21 day/cycle | One partial response observed in one KIT -mutated melanoma patient | The common (AE)s were fatigue, nausea, vomiting, anorexia, anemia, edema, diarrhea, rash, shortness of breath, constipation, neuropathy, thrombocytopenia, and infection | |
| Imatinib | Retrospective study, Wei, X., et al. 2019 [54] | Median age of 54 (range, 11–80) with c-KIT alterations | 78 | 400 mg/day | Median OS and PFS = 13.1 and 4.2 months respectively | 2 patients were alive without disease progression | The common (AE)s were: edema, rash, fatigue, anorexia, nausea, and neutropenia. Vomiting, psychiatric symptoms, and elevated ALT or AST, in a fraction of patients |
| RTKi | Phase/Year Published or Presented | Population | N | Dose | Survival | Response | Adverse Reactions |
|---|---|---|---|---|---|---|---|
| Nilotinib | Phase II, Cho, J.H., et al. 2012 [68] | Median age 51 (range, 37–68) | 11 | 400 mg twice daily | Median OS and PFS = 7.7 and 2.5 months, respectively | 2 PRs and 5 SD | Common AEs: alopecia, skin rash and headache |
| Nilotinib | Phase II, Carvajal, R.D., et al. 2015 [69] | Median age of 67 years (range, 38–85 years) in 2 cohorts: (A) refractory/shows resistance to a prior KIT inhibitor; (B) patients with brain metastases | 19 | 400 mg twice daily | Median OS = 9.1 months and TTP = 3.3 months | In Cohort A, 2 patients achieved PRs and none observed in cohort B | Toxicity rates and patterns were similar for Cohorts A and B. The Common AEs: fatigue, low-grade musculoskeletal, gastrointestinal discomfort. |
| Nilotinib | Phase II, Lee, S.J., et al. 2015 [70] | Median age of 56 (range, 28–81) years | 42 | 400 mg twice daily | Median OS and PFS = 74 and 34 weeks, respectively | 1 CR and 6 PRs | Most common AEs: anemia, skin rash, liver enzyme elevation, jaundice, anorexia, fatigue, and nausea. |
| Nilotinib | Phase II, Guo, J., et al. 2017 [71] | Median age 65.5 (range, 20–87) years | 42 | 400 mg twice daily | Median OS and PFS = 18 and 4.2 months, respectively | 3 PRs | Rash, increased blood bilirubin, nausea, decreased appetite fatigue |
| Nilotinib | Phase II, Delyon, J., et al. 2018 [56] | Median age 70 (range, 62–76) years | 25 | 400 mg twice daily | Median OS and PFS = 13.2 and 6 months, respectively | 1 CR and 4 PRs | The most common AEs: fatigue, rash, increased AST/ALT or cholestasis, and nausea. Three patients had drug with drawal because of grade 3 AEs |
| RTKi | Phase/ Year Published or Presented | Population | N | Dose | Survival | Response | Adverse Reactions |
|---|---|---|---|---|---|---|---|
| Dasatinib +Dacarbazine | Phase I,Algazi, A.P., et al. 2011 [74] | Median age 62.3 | 50 | dasatinib 70 mg dacarbazine 800 mg·m−2 | Median OS and PFS = 40.6 and 13.4 weeks respectively | Two patients showed PRs | The most common grade 3 and 4 (AE)s were: haematological, neutropenia, anaemia, and thrombocytopenia |
| Dasatinib | Phase II, Kluger, H.M., et al. 2011 [75] | Median age of 64 (range, 37–84) years | 39 |
100 mg PO BID or 70 mg PO BID | Median OS and PFS = 55 and 8 weeks respectively | Two patients showed PRs | The most common (AE)s were: fatigue, dyspnea,pleural effusion, nausea, anorexia and diarrhea |
| Dasatinib | Phase II,Kalinsky, K., et al. 2017 [76] | Median age of 69 (range, 41–87) years | 30 |
70 mg orally twice daily | Median OS and PFS = 7.5 and 2.1 months respectively | 4 of 22 evaluable patients had PRs | The most common (AE)s: fatigue, dyspnea, nausea, anemia, pleural effusion, neutropenia, vomiting, anorexia, hypoxia, hypertension lymphopenia, myocardial infarction |
| RTKi | Phase/ Year Published or Presented | Population | N | Dose | Survival | Response | Adverse Reactions |
|---|---|---|---|---|---|---|---|
| Sunitinib | Phase 2, Minor, D.R., et al. 2012 [81] | Median age 75 (range, 39–92) years | 12 | 50 mg/d, dose modifications sequentially to 37.5 and 25 mg/d for grade III or IV toxicities. | Median survival = 6 months (patients with KIT mutations); PFS = 15 months | 1 CR, 2 PRs in the 4 KIT patients and 1 PR in 6 patients with KIT amplification | The frequently observed toxicities: nausea or vomiting, skin and subcutaneous disorders, hematologic toxicity, fatigue, and hypertension |
| Sunitinib | Phase II study, Decoster, L., et al. 2015 [82] | Median age of 55 years | 39 | 50 mg/d for 4 weeks, followed by 2 weeks off, | The median OS and PFS = 4.3 and 1.3 months, respectively | PRs were observed in 4 patients | The most grade 3 or 4 (AE)s were asthenia, thrombocytopenia, neutropenia, and anorexia |
| Sunitinib | Phase 2 trial, Bucbinder, E.I., et al. 2015 [83] | Median age of 63 (range 38–86) years | 52 | 50 mg daily or 37.5 mg daily for 4 weeks of a 6-week cycle | The median OS and PFS = 7.7 and 3.1 months, respectively | 4 patients showed PRs | The most common (AE)s were fatigue, leukopenia, thrombocytopenia, nausea, neutropenia, and diarrhea |
| RTKi | Cell Lines Used | Results | Literature |
|---|---|---|---|
| Gefitinib | RaH3 RaH5 (both WTBRAF WTNRAS) | -Gefitinib exhibits a dose dependent inhibition of growth, without effect on apoptosis, favors cell arrest in G1, increased expression of p27KIP1 and reduces phosphorylation of ErbB1, ErbB2, ErbB3, ERK1/2, and AKT. | Djerf, E.A., et al. 2011 [114] |
| Gefitinib | A375 (V600EBRAF) | -Gefitinib suppressed cell proliferation, mRNA, and protein expression of VEGF and AKT, invasion and induced apoptosis. | Wan, X., et al. 2018 [116] |
| Gefitinib | A2058 (V600EBRAF), HT168-M1(V600EBRAF), HT199(V600EBRAF), WM983B (V600EBRAF), M24met (61RNRAS), MEWO (WTBRAF, WTNRAS) A431 (squamous carcinoma cells) | -Gefitinib inhibited the activity of EGFR in HT168-M1 and WM983B. -Synergistic inhibitory effect of vemurafenib with gefitinib in BRAF mutant melanoma cells. -Gefitinib reduced cell migration in just melanoma cells expressing mutant BRAF, and inhibited in-vivo liver colonization of WM983B and HT168-M1 xenografts. | Kenessey, I., et al. 2018 [117] |
| Erlotinib + Bevacizumab | 518A2 (V600EBRAF), 607B (activated Ras), Sk-Mel-28 (V600EBRAF), A375 (V600EBRAF), Mel-Juso (Q61L NRAS), M24met (61R NRAS), 6F (isolated from an ovaric metastasis) | -Erlotinib reduces transmigration in 518A2, M24met and SK-Mel-28 cells and increase the antiangiogenic effect of bevacizumab. -Erlotinib inhibited MEK/AKT pathways. -Erlotinib in combination with bevacizumab reduces sprout length in HUVECs conditioned with 518A2 and in cells conditioned with M24met medium. -Erlotinib and bevacizumab reduce tumor volume and proliferation, enhance apoptosis, and reduce lymph node diameter and lung metastasis in mice injected with 518A2. | Schicher, N., et al. 2009 [118] |
| Erlotinib + Ad-IL-24 | WM35(V600EBRAF), WM793 (V600EBRAF), A375(V600EBRAF), MeWo (WTBRAF, WTNRAS) (metastatic), Skmel-2 (Q61RNRAS), SB2 (advanced-stage), Mel-2 (metastatic, passage 6) Mel-3(metastatic, passage3), A431 (squamous carcinoma) | -Erlotinib decreased the cell viability of WM35, WM793, A375, and MeWo cells. -Co-treatment of melanoma cells with Ad-IL-24 and erlotinib decreases cell growth and enhanced apoptosis through Apaf-1 and AKT signaling pathways. | Deng, W.G., et al. 2011 [119] |
| Lapatinib or Gefitinib + Foretinib | A375(V600EBRAF), Hs294T(WTBRAF) WM9(V600EBRAF) | -Lapatinib/gefitinib shows slight effect alone but in combination with foretinib decreased melanoma cell viability in A375 and Hs294T cells. The WM9 cell was the most resistant to treatment. -Foretinib alone/with EGFR inhibitors reduced proliferation of A375, induced higher apoptosis in Hs294T than A375, and reduced pAkt and pErk levels. -Lapatinib/gefitinib does not affect cell morphology or actin cytoskeleton organization, but foretinib alone or in combination changed nuclei morphology -Foretinib alone or its combination with lapatinib/gefitinib induced G2/M cycle arrest in A375, Hs294T and WW9 cells. | Dratkiewicz, E., et al. 2018 [120] |
| Lapatinib or Gefitinib + Foretinib | A375(V600EBRAF), Hs294T (WTBRAF) WM9 (V600EBRAF) | -This combination is effective in WM9 and Hs294T, while in A375 cells, the effect was similar to foretinib alone in terms of reduction of cell migration. -This combination reduces invasion in A375 and WM9 cells but is less evident in Hs294T cells. -Foretinib alone or in combination decreases proteolytic activity. | Simiczyjew, A., et al. 2019 [108] |
| Lapatinib + Foretinib | A375 (V600EBRAF)/A375 RL and WM9 (V600EBRAF)/WM9 RL | -This combination or foretinib alone inhibit viability and migration in both resistant cells, especially in WM9 RL. | Dratkiewicz, E., et al. 2020 [121] |
| Gefitinib or Afatinib/BIBW2992 + MK-2206/GSK692094 | vemurafenib-resistant YUKSI cells | -Afatinib/BIBW2992 paired with MK-2206/GSK692094 and reduced growth. | Held, M.A., et al. 2013 [122] |
| Afatinib + crizotinib | A375 (V600EBRAF), SkMel24(V600EBRAF), SkMel28 (V600EBRAF), A375PR1& A375VR4 were PLX4720 or vemurafenib-resistant sublines derived from A375, SkMel2 (Q61RNRAS), ESTDAB102 (Q61RNRAS), ESTDAB105 (WTBRAF/NRAS), ESTDAB138 (WT BRAF/NRAS), ESTDAB140 (WTBRAF/NRAS) ESTDAB149 (Q61RNRAS) | -This combination decreased cell proliferation, colony formation, invasion, promoted cell death in distinct melanoma cells, and decreased tumor growth rate. | Das, I., et al. 2019 [123] |
| RTKi | Phase/Year Published or Presented | Population | N | Dose | Survival | Response | Adverse Reactions |
|---|---|---|---|---|---|---|---|
| Gefitinib | Phase II, Patel, S.P., et al. 2011 [115] | Median age of 62.5 years, (range, 19–90) years | 52 | 250 mg/day | Median OS and PFS = 9.7 and 1.4 months, respectively | Two PRs and 13 showed SD | Well tolerated drug, and fatigue was the only grade 3 adverse event. |
| Erlotinib+ Bevacizumab | Phase II trial,Mudigonda, T.V., et al. 2016 [124] | Median age of 60 years, (range, 18–65) years | 28 | 150 mg/day of erlotinib and 10 mg/kg of bevacizumab | Median OS and PFS = 6.7 and 2 months, respectively | Two PRs and 11 showed SD | Rare grade 3–4 events: fatigue and dysarthria. |
| RTKi | Cell Lines Used | Results | Literature |
|---|---|---|---|
| SU11274 | MM-AN, MU, PM-WK, MM-RU, MM-MC, MM-LH, and RPM-EP |
-SU11274 inhibits proliferation and induces cell death in all melanoma cells expressing MET. -SU11274 induces a differentiated phenotype in MM-RU, MU, and MM-MC. -SU11274 decreases ROS and inhibits tyrosine phosphorylation of c-Met in MU melanoma cells. | Puri, N., et al. 2007 [167] |
| SU11274 | HT168(V600EBRAF), HT168-M1(V600EBRAF), HT199 (V600EBRAF), WM35 (V600EBRAF), WM983A (V600EBRAF), WM983B (V600EBRAF), M24met (61RNRAS), HT168-M1 human melanoma cells in SCID-mice |
-SU11274 inhibited Met phosphorylation in HT168-M1 cells. -SU11274 inhibited cells proliferation in HT168-M1, HT199, WM983B, and M24met. -SU11274 favor apoptosis and inhibit migration of HT168- M1 cells. -SU11274 inhibited intrasplenic growth and liver colonization of HT168-M1 xenograft. | Kenessey, I., et al. 2010 [168] |
| SU11274 | M14 (V600EBRAF), M4Beu, A375 (V600EBRAF), EGFP-A375 and Rel3 (hyper metastatic variant of A375); Untreated or SU11274-treated Rel3 injected into the flank of immunodeficient mice | -SU11274 increased phosphorylation of c-Met on Tyr1349 inA375 and Rel3 cells. -SU11274 inhibits cell proliferation in all melanoma cells, changes cell morphology, mediates bioenergetic alterations, increases pluripotent stem cell proteins, phosphokinase proteome profile, tumor initiation in Rel3, and mediates in vivo tumorigenicity. | Kucerova, L., et al. 2016 [169] |
| SU11274 | MU-P (V600EBRAF), RU-P, EP-P, WK-P (explant culture); MU-R and RU-R (SU11274 resistant cell lines) |
-Treatment with SU11274 favors seven-fold reduction in tumor size of xenografts from RU-P melanoma cells. -SU11274 in combination with everolimus and XAV939 overcome resistance associated to c-Met inhibitor in MU-R and RU-R cells. | Etnyre, D., et al. 2014 [170] |
| Tivatinib | C8161 (G464EBRAF), WM793 (V600EBRAF), WM293 (V600EBRAF), UACC, WM278 (V600EBRAF) |
-Tivatinib inhibits cell viability and induces apoptosis and cytotoxicity in the tested melanoma cells. -Tivatinib increases vinculin in C8161 and UACC cells, RhoC in C8161 cells and reduces zyxin and FN1 mRNA in C8161 and UACC cells. -Tivatinib decreases VM formation in C8161 and WM793 cells. | Kumar, S.R., et al. 2019 [171] |
| PHA-665752 | A375 (V600EBRAF), MeWo (WTBRAFWTNRAS),SK-Mel-2 (Q61RNRAS), SB2 cells (NRAS mutants), WM852 (Q61RNRAS), 451Lu (V600EBRAF), WM1361A (Q61RNRAS), WM35 (V600EBRAF) WM793 (V600EBRAF) |
-PHA-66752 favors dose dependent inhibition of MET phosphorylation in all cells and completes Akt phosphorylation inhibition in NRAS mutant cells. -IC50 of PHA-66752 was lower for NRAS mutant cells. -PHA-66752 reduces migration and induces G0/G1 cell cycle arrest and apoptosis more dramatically in NRAS mutant cells. | Chattopadhyay, C., et al. 2012 [162] |
| Quercetin |
A375 (V600EBRAF), A2058 (V600EBRAF), SK-Mel-2 (Q61RNRAS) MeWo (WTBRAFWTNRAS) |
-Quercetin inhibited migration, invasion, MET activation, and downstream molecules in A375 and A2058 cells. -Quercetin reduced c-Met levels through FAS inhibition in A375, A2058, MeWo, and skmel-2 cells | Cao, H., et al. 2015 [172] |
| RTKi | Phase/Year Published or Presented | Population | N | Dose | Survival | Response | Adverse Reactions |
|---|---|---|---|---|---|---|---|
| Tivantinib + Sorafenib | Phase I, Puzanov, I., et al. 2015 [178] | Median age of 65.1 years | 19 | tivantinib 360 mg BID/sorafenib 200 mg BID) or tivantinib 360 mg BID/sorafenib 400 mg BID | Median PFS = 4.9 months | 1 CR, 4 patients achieved PRs | The most common AEs included: rash, diarrhea, anorexia, fatigue, alopecia, palmar plantar erythrodysaesthesia syndrome, and weight reduction |
| Cabozantinib | Phase II trial, Daud, A., et al. 2017 [181] | Median age of 65 (range, 30–90) years | 77 | 100 mg daily | Median OS and PFS = 9.4 and 3.8 month, respectively | 5 PRs | The most common AEs were grade 3/4: fatigue, hypertension, and abdominal pain |
| RTKi | Phase/Year Published or Presented | Study Population | N | Dose | Survival | Response | Adverse Reactions |
|---|---|---|---|---|---|---|---|
| SU5416, Semaxanib | Phase II, Kuenen, B.C., et al. 2003 [208] | Median age of 53.5 (range, 23–71) years | 20 | 145 mg·m−2, twice weekly | Median OS and PFS = 107.5 and 41 days, respectively. | No Response | Main (AEs): headache, phlebitis, nausea, vomiting, anorexia, diarrhea, and asthenia |
| SU5416 + Thalidomide (THAL) | Phase II, Mita, M.M., et al. 2007 [209] | Median age of 58 (range, 43–71) | 12 | Semaxanib (145 mg·m−2, twice/week + THAL starting 200 mg/d | Median survival = 7.3 months | One CR and one PR | The common toxicities: deep venous, thrombosis, headache, and lower extremity edema |
| Axitinib | Phase II, Fruehauf, J. et al. 2011 [211] | Median age 65 (range, 30–86) | 32 | 5 mg/twice a day | Median OS and PFS = 6.6 and 3.9 months respectively | One CR and five PRs | The most common (AE)s included fatigue, hypertension, hoarseness, diarrhea |
| Axitinib + carboplation+ paclitaxel | Phase II, Algazi, A.P., et al. 2015 [213] | Median age 65.4 years | 36 | Axitinib 5 mg PO b.i.d. + carboplatin (AUC = 5) with paclitaxel (175 mg·m−2) | Median OS and PFS = 14 and 8.7 months, respectively | 8 patients achieved PRS | The most common (AE)s: hypertension, neutropenia, and anaemia |
| Bevacizumab (Bevax) + low-dose interferon alfa-2b | Phase 2, Varker, K.A., et al. 2007 [216] | Median age 57.5 (range, 28–83) years | 32 | Bevax (15mg/kg every 2 weeks) + low-dose IFN-α2b (1MU·m−2 daily) | Median OS and PFS = 10 and 3 months respectively | One PR | The most (AE)s were of grade 1/2: fatigue, anorexia, myalgia, headache, nausea, vomiting |
| Bevacizumab (Bevax) + high dose interferon alfa-2b | Phase II, Grignol, V.P., et al. 2011 [217] | Median age 58.4 (range, 31–79) years | 25 | Bevax 15 mg/kg + 5MU/m IFN-α thrice weekly | Median OS and PFS = 17 and 4.8 months respectively | 6 PRs, and 5 SD | The common (AE)s were fatigue, anorexia, nausea/vomiting, fever/chills, anemia |
| Bevacizumab (Bevax) + paclitaxel + carboplatin | Phase II, Kim, K.B., et al.2012 [218] | Median age 60 (range, 27–85) years | 143 | Bevax (15 mg/kg), carboplatin (area under the curve, 5) plus paclitaxel (175 mg·m−2) | Median OS and PFS = 12.3 and 5.6 months, respectively | 3 CRs and 33 PRs | The most common (AE)s: neutropenia, peripheral neuropathy, febrile neutropenia, arterial thromboembolic events, and hypertension |
| Bevacizumab | Phase III Corrie, P.G., et al. 2018 [219] | Median age 56 years (18–88 years) | 671 | 7.5 mg/kg | Median DFI = 63 months | Adjuvant bevacizumab improved DFI but did not improve OS | Common grade 3 or 4 adverse events was hypertension |
| RTKi | Cell Lines Used | Results | Literature |
|---|---|---|---|
| Axitinib | M24met (61RNRAS), A375 (V600EBRAF) A2058 (V600EBRAF) | -Axitinib inhibited VEGFR-2 phosphorylation and partially ERK1/2 signal in angiogenic vessels of xenograft tumors of M24 met. -Axitinib in combination with bevacizumab inhibited lymph node metastasis and prolonged survival. | Hu-Lowe, D.D., et al. 2008 [210] |
| Axitinib + OVA peptide-based vaccination | MO5 (B16.OVA) | -Axitinib enhances vaccine effect to prevent melanoma growth and to favor T cell infiltration and activity. -This combination prevents accumulation of MDSC and Treg suppressor cells and promotes type-1 LT cells function in vivo. | Bose, A., et al. 2012 [212] |
| Bevacizumab | A-07 and D-12 meningeal human melanoma cells inoculated in the intracranial region of BALB/c nu/nu mice | -Bevacizumab inhibits angiogenesis and, increases cerebral invasion and genes related angiogenesis expression of A-07 tumors | Simonsen, T.G., et al. 2020 [220] |
| RTKi | Cell Lines Used | Results | Literature |
|---|---|---|---|
| Cabozantinib | Murine B16F10 cells | -Cabozantinib inhibits cell tubule formation, migration, and invasion | Yakes F et al. 2011 [221] |
| Foretinib | Murine B16F10 cells |
-Foretinib inhibits migration, invasion, and anchorage dependent growth. -Foretinib inhibits phosphorylation of Met, Flk-1/KDR, and reduces tumor burden injected in mice. | Qian F et al. 2009 [228] |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Sabbah, M.; Najem, A.; Krayem, M.; Awada, A.; Journe, F.; Ghanem, G.E. RTK Inhibitors in Melanoma: From Bench to Bedside. Cancers 2021, 13, 1685. https://doi.org/10.3390/cancers13071685
Sabbah M, Najem A, Krayem M, Awada A, Journe F, Ghanem GE. RTK Inhibitors in Melanoma: From Bench to Bedside. Cancers. 2021; 13(7):1685. https://doi.org/10.3390/cancers13071685
Chicago/Turabian StyleSabbah, Malak, Ahmad Najem, Mohammad Krayem, Ahmad Awada, Fabrice Journe, and Ghanem E. Ghanem. 2021. "RTK Inhibitors in Melanoma: From Bench to Bedside" Cancers 13, no. 7: 1685. https://doi.org/10.3390/cancers13071685
APA StyleSabbah, M., Najem, A., Krayem, M., Awada, A., Journe, F., & Ghanem, G. E. (2021). RTK Inhibitors in Melanoma: From Bench to Bedside. Cancers, 13(7), 1685. https://doi.org/10.3390/cancers13071685







