Correlation of Clinical Parameters with Intracranial Outcome in Non-Small Cell Lung Cancer Patients with Brain Metastases Treated with Pd-1/Pd-L1 Inhibitors as Monotherapy
Abstract
Simple Summary
Abstract
1. Introduction
2. Results
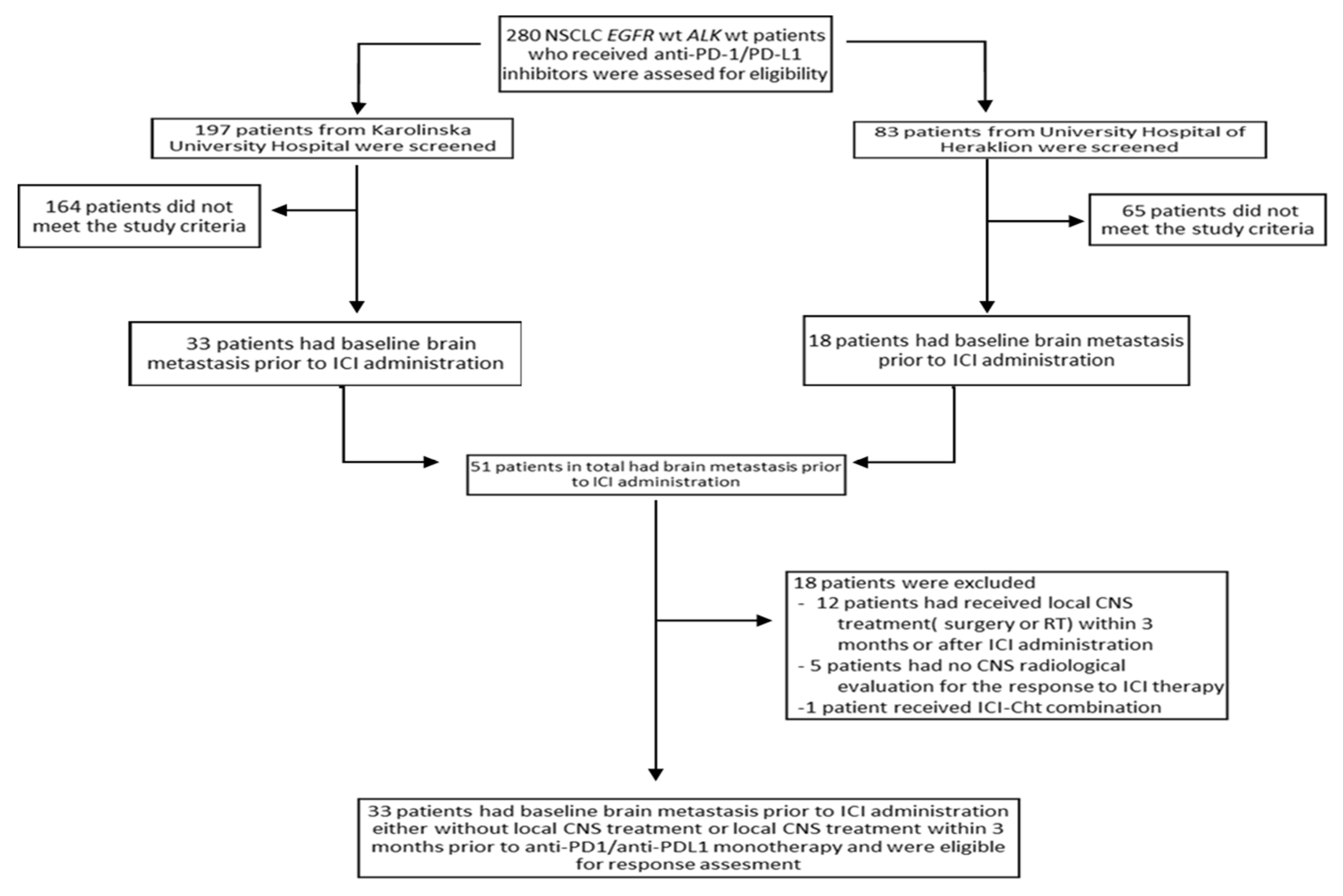
2.1. Patient Characteristics
2.2. Results from Response Assessment
2.3. Creation of Different Predictive Groups Based on I-O CNS Score
3. Discussion
4. Methods
4.1. Patient Eligibility
4.2. Study Design
4.3. Response Assessment
4.4. Statistical Analysis
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Barnholtz-Sloan, J.S.; Sloan, A.E.; Davis, F.G.; Vigneau, F.D.; Lai, P.; Sawaya, R.E. Incidence Proportions of Brain Metastases in Patients Diagnosed (1973 to 2001) in the Metropolitan Detroit Cancer Surveillance System. J. Clin. Oncol. 2004, 22, 2865–2872. [Google Scholar] [CrossRef] [PubMed]
- Villano, J.L.; Durbin, E.B.; Normandeau, C.; Thakkar, J.P.; Moirangthem, V.; Davis, F.G. Incidence of Brain Metastasis at Initial Presentation of Lung Cancer. Neuro Oncol. 2015, 17, 122–128. [Google Scholar] [CrossRef]
- Schouten, L.J.; Rutten, J.; Huveneers, H.A.M.; Twijnstra, A. Incidence of Brain Metastases in a Cohort of Patients with Carcinoma of the Breast, Colon, Kidney, and Lung and Melanoma. Cancer 2002, 94, 2698–2705. [Google Scholar] [CrossRef] [PubMed]
- Sperduto, P.W.; Yang, T.J.; Beal, K.; Pan, H.; Brown, P.D.; Bangdiwala, A.; Shanley, R.; Yeh, N.; Gaspar, L.E.; Braunstein, S.; et al. Estimating Survival in Patients With Lung Cancer and Brain Metastases: An Update of the Graded Prognostic Assessment for Lung Cancer Using Molecular Markers (Lung-MolGPA). JAMA Oncol. 2017, 3, 827–831. [Google Scholar] [CrossRef] [PubMed]
- Nieder, C.; Bremnes, R.M.; Andratschke, N.H. Prognostic Scores in Patients with Brain Metastases from Non-Small Cell Lung Cancer. J. Thorac. Oncol. 2009, 4, 1337–1341. [Google Scholar] [CrossRef]
- Langer, C.J.; Mehta, M.P. Current Management of Brain Metastases, with a Focus on Systemic Options. J. Clin. Oncol. 2005, 23, 6207–6219. [Google Scholar] [CrossRef]
- Moro-Sibilot, D.; Smit, E.; de Castro Carpeño, J.; Lesniewski-Kmak, K.; Aerts, J.G.; Villatoro, R.; Kraaij, K.; Nacerddine, K.; Dyachkova, Y.; Smith, K.T.; et al. Non-Small Cell Lung Cancer Patients with Brain Metastases Treated with First-Line Platinum-Doublet Chemotherapy: Analysis from the European FRAME Study. Lung Cancer 2015, 90, 427–432. [Google Scholar] [CrossRef]
- Schuette, W. Treatment of Brain Metastases from Lung Cancer: Chemotherapy. Lung Cancer 2004, 45 (Suppl. 2), S253–S257. [Google Scholar] [CrossRef]
- Doroshow, D.B.; Sanmamed, M.F.; Hastings, K.; Politi, K.; Rimm, D.L.; Chen, L.; Melero, I.; Schalper, K.A.; Herbst, R.S. Immunotherapy in Non-Small Cell Lung Cancer: Facts and Hopes. Clin. Cancer Res. 2019, 25, 4592–4602. [Google Scholar] [CrossRef]
- Berghmans, T.; Durieux, V.; Hendriks, L.E.L.; Dingemans, A.-M. Immunotherapy: From Advanced NSCLC to Early Stages, an Evolving Concept. Front. Med. 2020, 7. [Google Scholar] [CrossRef]
- Lim, S.M.; Hong, M.H.; Kim, H.R. Immunotherapy for Non-Small Cell Lung Cancer: Current Landscape and Future Perspectives. Immune Netw. 2020, 20. [Google Scholar] [CrossRef]
- Borghaei, H.; Paz-Ares, L.; Horn, L.; Spigel, D.R.; Steins, M.; Ready, N.E.; Chow, L.Q.; Vokes, E.E.; Felip, E.; Holgado, E.; et al. Nivolumab versus Docetaxel in Advanced Nonsquamous Non-Small-Cell Lung Cancer. N. Engl. J. Med. 2015, 373, 1627–1639. [Google Scholar] [CrossRef] [PubMed]
- Brahmer, J.; Reckamp, K.L.; Baas, P.; Crinò, L.; Eberhardt, W.E.E.; Poddubskaya, E.; Antonia, S.; Pluzanski, A.; Vokes, E.E.; Holgado, E.; et al. Nivolumab versus Docetaxel in Advanced Squamous-Cell Non–Small-Cell Lung Cancer. N. Engl. J. Med. 2015, 373, 123–135. [Google Scholar] [CrossRef]
- Hellmann, M.D.; Paz-Ares, L.; Bernabe Caro, R.; Zurawski, B.; Kim, S.-W.; Carcereny Costa, E.; Park, K.; Alexandru, A.; Lupinacci, L.; de la Mora Jimenez, E.; et al. Nivolumab plus Ipilimumab in Advanced Non–Small-Cell Lung Cancer. N. Engl. J. Med. 2019, 381, 2020–2031. [Google Scholar] [CrossRef]
- Gandhi, L.; Rodríguez-Abreu, D.; Gadgeel, S.; Esteban, E.; Felip, E.; De Angelis, F.; Domine, M.; Clingan, P.; Hochmair, M.J.; Powell, S.F.; et al. Pembrolizumab plus Chemotherapy in Metastatic Non-Small-Cell Lung Cancer. N. Engl. J. Med. 2018, 378, 2078–2092. [Google Scholar] [CrossRef] [PubMed]
- Herbst, R.S.; Baas, P.; Kim, D.-W.; Felip, E.; Pérez-Gracia, J.L.; Han, J.-Y.; Molina, J.; Kim, J.-H.; Arvis, C.D.; Ahn, M.-J.; et al. Pembrolizumab versus Docetaxel for Previously Treated, PD-L1-Positive, Advanced Non-Small-Cell Lung Cancer (KEYNOTE-010): A Randomised Controlled Trial. Lancet 2016, 387, 1540–1550. [Google Scholar] [CrossRef]
- Paz-Ares, L.; Luft, A.; Vicente, D.; Tafreshi, A.; Gümüş, M.; Mazières, J.; Hermes, B.; Çay Şenler, F.; Csőszi, T.; Fülöp, A.; et al. Pembrolizumab plus Chemotherapy for Squamous Non–Small-Cell Lung Cancer. N. Engl. J. Med. 2018, 379, 2040–2051. [Google Scholar] [CrossRef] [PubMed]
- Reck, M.; Rodríguez-Abreu, D.; Robinson, A.G.; Hui, R.; Csőszi, T.; Fülöp, A.; Gottfried, M.; Peled, N.; Tafreshi, A.; Cuffe, S.; et al. Pembrolizumab versus Chemotherapy for PD-L1–Positive Non–Small-Cell Lung Cancer. N. Engl. J. Med. 2016, 375, 1823–1833. [Google Scholar] [CrossRef]
- Rittmeyer, A.; Barlesi, F.; Waterkamp, D.; Park, K.; Ciardiello, F.; von Pawel, J.; Gadgeel, S.M.; Hida, T.; Kowalski, D.M.; Dols, M.C.; et al. Atezolizumab versus Docetaxel in Patients with Previously Treated Non-Small-Cell Lung Cancer (OAK): A Phase 3, Open-Label, Multicentre Randomised Controlled Trial. Lancet 2017, 389, 255–265. [Google Scholar] [CrossRef]
- Socinski, M.A.; Jotte, R.M.; Cappuzzo, F.; Orlandi, F.; Stroyakovskiy, D.; Nogami, N.; Rodríguez-Abreu, D.; Moro-Sibilot, D.; Thomas, C.A.; Barlesi, F.; et al. Atezolizumab for First-Line Treatment of Metastatic Nonsquamous NSCLC. N. Engl. J. Med. 2018, 378, 2288–2301. [Google Scholar] [CrossRef]
- Paz-Ares, L.; Ciuleanu, T.-E.; Cobo, M.; Schenker, M.; Zurawski, B.; Menezes, J.; Richardet, E.; Bennouna, J.; Felip, E.; Juan-Vidal, O.; et al. First-Line Nivolumab plus Ipilimumab Combined with Two Cycles of Chemotherapy in Patients with Non-Small-Cell Lung Cancer (CheckMate 9LA): An International, Randomised, Open-Label, Phase 3 Trial. Lancet Oncol. 2021, 22, 198–211. [Google Scholar] [CrossRef]
- Herbst, R.S.; Giaccone, G.; de Marinis, F.; Reinmuth, N.; Vergnenegre, A.; Barrios, C.H.; Morise, M.; Felip, E.; Andric, Z.; Geater, S.; et al. Atezolizumab for First-Line Treatment of PD-L1-Selected Patients with NSCLC. N. Engl. J. Med. 2020, 383, 1328–1339. [Google Scholar] [CrossRef]
- Goldberg, S.B.; Schalper, K.A.; Gettinger, S.N.; Mahajan, A.; Herbst, R.S.; Chiang, A.C.; Lilenbaum, R.; Wilson, F.H.; Omay, S.B.; Yu, J.B.; et al. Pembrolizumab for Management of Patients with NSCLC and Brain Metastases: Long-Term Results and Biomarker Analysis from a Non-Randomised, Open-Label, Phase 2 Trial. Lancet Oncol. 2020, 21, 655–663. [Google Scholar] [CrossRef]
- Hendriks, L.E.L.; Henon, C.; Auclin, E.; Mezquita, L.; Ferrara, R.; Audigier-Valette, C.; Mazieres, J.; Lefebvre, C.; Rabeau, A.; Le Moulec, S.; et al. Outcome of Patients with Non-Small Cell Lung Cancer and Brain Metastases Treated with Checkpoint Inhibitors. J. Thorac. Oncol. 2019, 14, 1244–1254. [Google Scholar] [CrossRef]
- Skribek, M.; Rounis, K.; Makrakis, D.; Agelaki, S.; Mavroudis, D.; De Petris, L.; Ekman, S.; Tsakonas, G. Outcome of Patients with NSCLC and Brain Metastases Treated with Immune Checkpoint Inhibitors in a “Real-Life” Setting. Cancers 2020, 12, 3707. [Google Scholar] [CrossRef] [PubMed]
- Kuremsky, J.G.; Urbanic, J.J.; Petty, W.J.; Lovato, J.F.; Bourland, J.D.; Tatter, S.B.; Ellis, T.L.; McMullen, K.P.; Shaw, E.G.; Chan, M.D. Tumor Histology Predicts Patterns of Failure and Survival in Patients with Brain Metastases from Lung Cancer Treated with Gamma Knife Radiosurgery. Neurosurgery 2013, 73, 641–647, discussion 647. [Google Scholar] [CrossRef]
- Burnette, B.C.; Liang, H.; Lee, Y.; Chlewicki, L.; Khodarev, N.N.; Weichselbaum, R.R.; Fu, Y.-X.; Auh, S.L. The Efficacy of Radiotherapy Relies upon Induction of Type i Interferon-Dependent Innate and Adaptive Immunity. Cancer Res. 2011, 71, 2488–2496. [Google Scholar] [CrossRef]
- Han, C.; Liu, Z.; Zhang, Y.; Shen, A.; Dong, C.; Zhang, A.; Moore, C.; Ren, Z.; Lu, C.; Cao, X.; et al. Tumor Cells Suppress Radiation-Induced Immunity by Hijacking Caspase 9 Signaling. Nat. Immunol. 2020, 21, 546–554. [Google Scholar] [CrossRef]
- Grassberger, C.; Ellsworth, S.G.; Wilks, M.Q.; Keane, F.K.; Loeffler, J.S. Assessing the Interactions between Radiotherapy and Antitumour Immunity. Nat. Rev. Clin. Oncol. 2019, 16, 729–745. [Google Scholar] [CrossRef]
- Eisenhauer, E.A.; Therasse, P.; Bogaerts, J.; Schwartz, L.H.; Sargent, D.; Ford, R.; Dancey, J.; Arbuck, S.; Gwyther, S.; Mooney, M.; et al. New Response Evaluation Criteria in Solid Tumours: Revised RECIST Guideline (Version 1.1). Eur. J. Cancer 2009, 45, 228–247. [Google Scholar] [CrossRef] [PubMed]

| Patient Characteristics | Total (n = 33) | 95% Confidence Intervals | |
|---|---|---|---|
| Age (years) | Median | 69 | |
| Range | 40–84 | ||
| Gender (n%) | Male | 16 (48.5%) | |
| Female | 17 (51.5%) | ||
| ECOG performance status (n%) | 0–1 | 25 (75.8%) | |
| 2 | 8 (24.2%) | ||
| Smoking status (n%) | Former or active smoker | 29 (87.9%) | |
| Never smoker | 4 (12.1%) | ||
| Histology (n%) | Adenocarcinoma | 29 (87.9%) | |
| Non-adenocarcinoma | 4 (12.1%) | ||
| Line of treatment of ICIs administration (n%) | 1st line | 5 (15.2%) | |
| 2nd or subsequent line | 28 (84.8%) | ||
| PD-L1 expression in extracranial lesions (n%) | <1% | 2 (6.1%) | |
| 1% ≤ PDL1 < 50% | 10 (30.3%) | ||
| PDL1 ≥ 50% | 14 (42.4%) | ||
| Missing data | 7 (21.2%) | ||
| CNS metastases at diagnosis | Yes | 24 (72.7%) | |
| No | 9 (27.3%) | ||
| Neurological symptoms attributed to CNS metastases | Yes | 18 (54.5%) | |
| No | 15 (45.5%) | ||
| Radiological evaluation method of CNS metastases | MRI | 23 (69.7%) | |
| CT | 10 (30.3%) | ||
| Size of largest CNS metastasis (mm) | Median | 17 | |
| Range | 4–50 | ||
| Number of CNS metastases | Median | 2 | |
| Range | 1–50 | ||
| >3 CNS metastases | Yes | 12 (36.4%) | |
| No | 21 (63.6%) | ||
| Bone metastases (n%) | Yes | 12 (36.4%) | |
| No | 21 (63.6%) | ||
| Liver metastases (n%) | Yes | 10 (30.3%) | |
| No | 23 (69.7%) | ||
| Disease burden (n%) | Number of organs with metastatic disease >2 | 13 (39.4%) | |
| Number of organs with metastatic disease ≤2 | 20 (60.6%) | ||
| Steroid administration > 10 mg for > 10 days (n%) | Yes | 16 (48.5%) | |
| No | 17 (51.5%) | ||
| CNS response to previous systemic treatment | PRi or SDi | 7 (21.2%) | |
| PDi | 21 (63.6%) | ||
| No previous treatment | 4 (12.2%) | ||
| Missing data | 1 (3%) | ||
| Active CNS metastases | Yes | 25 (75.8%) | |
| No | 8 (24.2%) | ||
| Previous surgery for CNS metastases | Yes | 4 (12.1%) | |
| No | 29 (87.9%) | ||
| Previous radiation treatment for CNS metastases | WBRT | 9 (27.3%) | |
| SRS | 9 (27.3%) | ||
| No RT | 15 (45.5%) | ||
| Intracranial response to ICIs* | CRi | 1 (3%) | |
| PRi | 7 (21.2%) | ||
| SDi | 8 (24.2%) | ||
| PDi | 17 (51.5%) | ||
| Intracranial response to ICIs in patients with active CNS metastases (n = 25) | CRi | 1 (4%) | |
| PRi | 5 (20%) | ||
| SDi | 2 (8%) | ||
| PDi | 17 (68%) | ||
| I-O CNS score | 0.5–1 | 12 (36.4%) | |
| 1.5–2 | 21 (63.6%) | ||
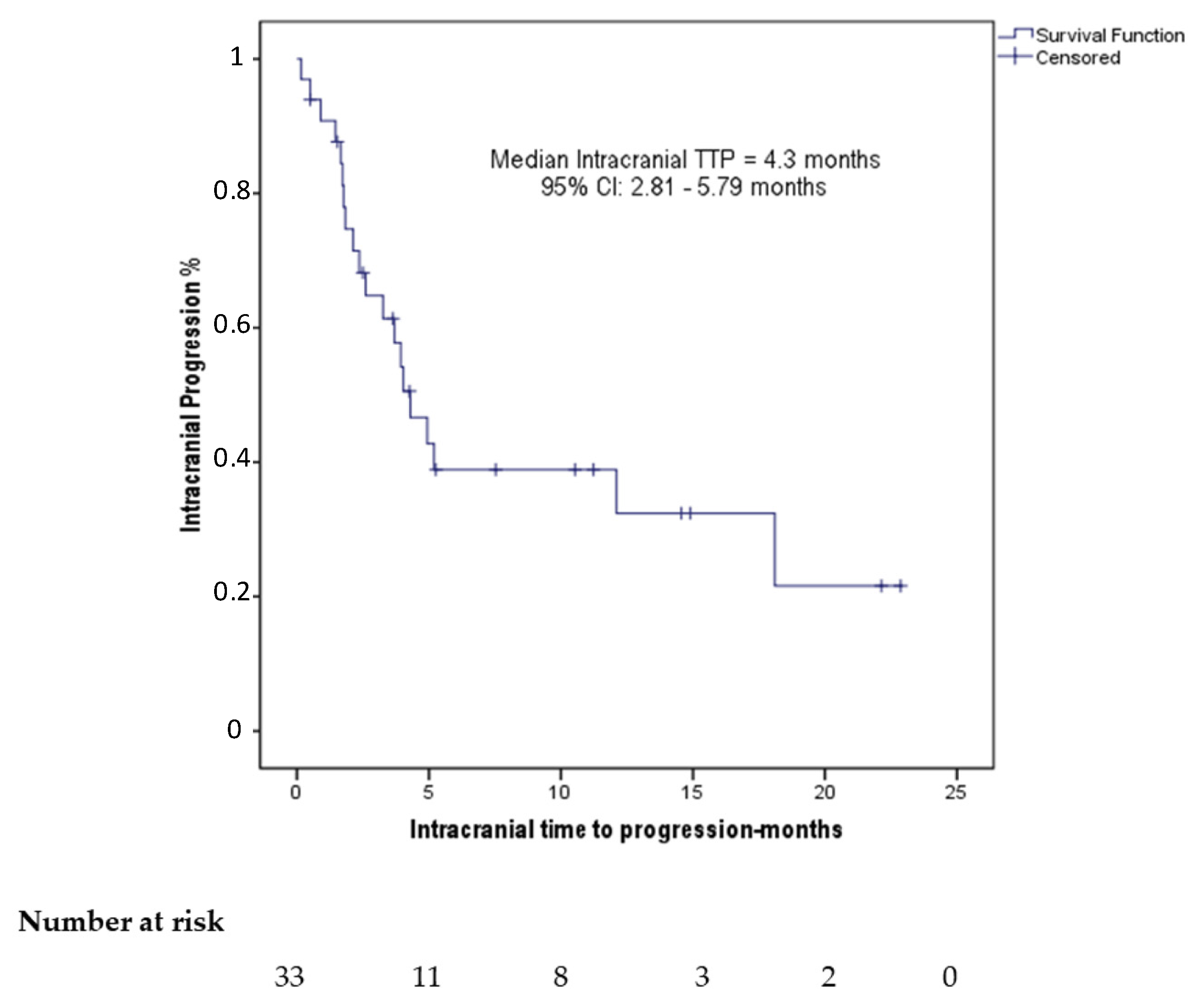
| Duration of intracranial response (months) | Median | 7.53 | 0–18.45 |
| Range | 0.5–22.47 | ||
| Intracranial progression (n%) | Yes | 28 (84.8%) | |
| No | 5 (15.2%) | ||
| Death (n%) | Yes | 25 (75.8%) | |
| No | 8 (24.2%) | ||
| Intracranial TTP (months) | Median | 4.3 | 2.81–5.79 |
| Range | 0.17–22.87 | ||
| OS (months) | Median | 6.77 | 2.34–11.19 |
| Range | 0.5–30.3 | ||
| Follow-up (months) | Median | 6.67 | 2.31–11.19 |
| Range | 0.13–22.13 | ||
| Univariate | ROC Analysis | ||
|---|---|---|---|
| Variable | OR (95% CI) | p-Value | AUC (95% CI) |
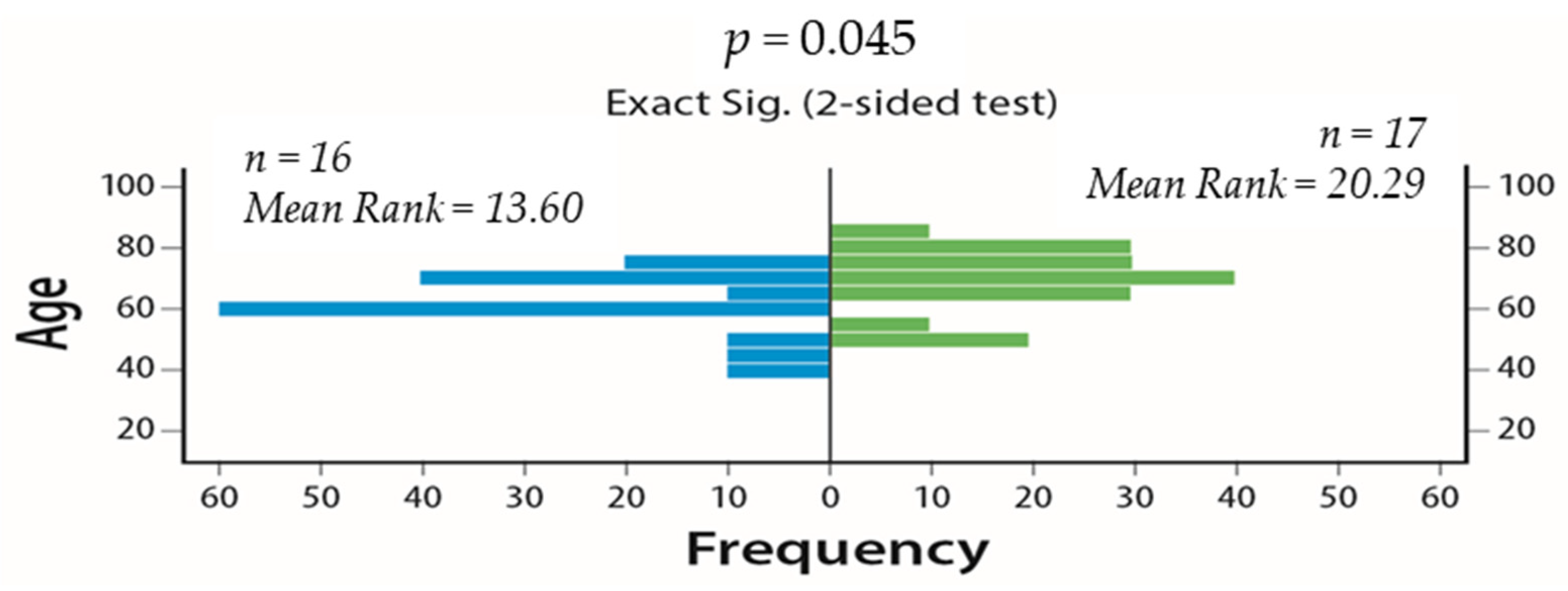
| Age ≥ 70 years old | 6.19 (1.27–30.170) | 0.024 | 0.700 (0.518–0.883) |
| Female gender | 1.84 (0.461–7.312) | 0.388 | 0.575 (0.378–0.773) |
| Performance status 2 | 3.82 (0.641–22.744) | 0.141 | 0.614 (0.420–0.808) |
| >2 organs with metastatic disease | 1.95 (0.471–8.130) | 0.356 | 0.579 (0.382–0.776) |
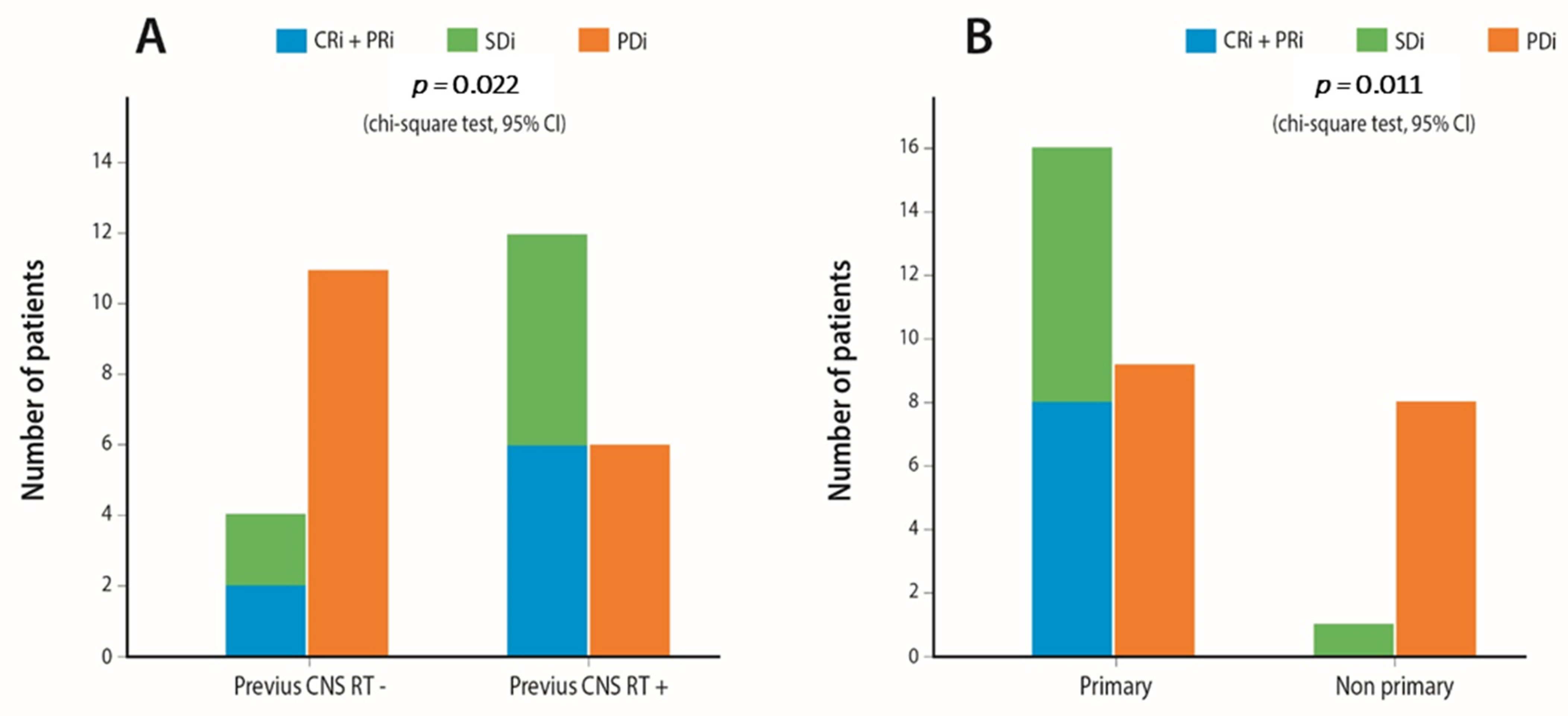
| Non-primary CNS metastases | 13.33 (1.424–124.880) | 0.023 | 0.704 (0.523–0.885) |
| >3 CNS metastases | 0.91 (0.220–3.758) | 0.895 | 0.511 (0.311–0.711) |
| Largest CNS metastases >3 cm | 0.52 (0.120–2.248) | 0.381 | 0.577 (0.373–0.781) |
| Symptoms attributed to CNS metastases | 0.53 (0.133–2.141) | 0.375 | 0.577 (0.380–0.775) |
| Previous RT for CNS metastases | 5.49 (1.210–25.000) | 0.027 | 0.699 (0.515–0.882) |
| 2nd or subsequent line of treatment | 0.22 (0.021–2.191) | 0.195 | 0.586 (0.390–0.783) |
| Steroid administration >10 mg for ≥10 days | 2.38 (0.588–9.646) | 0.224 | 0.607 (0.411–0.802) |
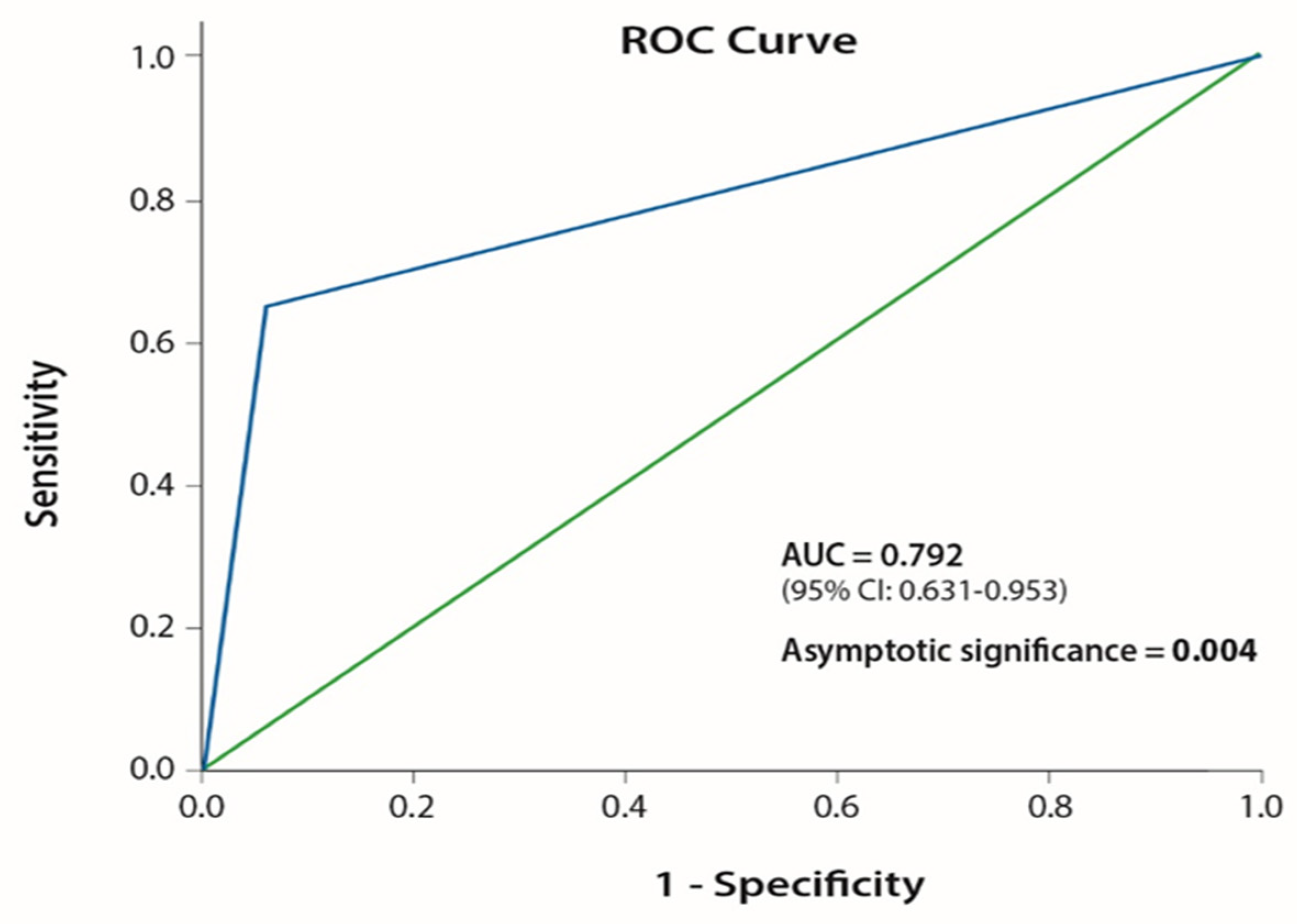
| I-O CNS score 0–1 | 27.50 (2.883–262.34) | 0.004 | 0.792 (0.631–0.953) |
| Parameter | Point Allocation | |
|---|---|---|
| Age (years) | <70 | 0.5 |
| ≥70 | 0 | |
| CNS metastases at presentation | Yes | 1 |
| No | 0 | |
| Previous CNS RT | Yes | 0.5 |
| No | 0 | |
| Predictive Groups | I-O CNS Score |
|---|---|
| Group A (good) | 1.5–2 |
| Group B (poor) | 0–1 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Rounis, K.; Skribek, M.; Makrakis, D.; De Petris, L.; Agelaki, S.; Ekman, S.; Tsakonas, G. Correlation of Clinical Parameters with Intracranial Outcome in Non-Small Cell Lung Cancer Patients with Brain Metastases Treated with Pd-1/Pd-L1 Inhibitors as Monotherapy. Cancers 2021, 13, 1562. https://doi.org/10.3390/cancers13071562
Rounis K, Skribek M, Makrakis D, De Petris L, Agelaki S, Ekman S, Tsakonas G. Correlation of Clinical Parameters with Intracranial Outcome in Non-Small Cell Lung Cancer Patients with Brain Metastases Treated with Pd-1/Pd-L1 Inhibitors as Monotherapy. Cancers. 2021; 13(7):1562. https://doi.org/10.3390/cancers13071562
Chicago/Turabian StyleRounis, Konstantinos, Marcus Skribek, Dimitrios Makrakis, Luigi De Petris, Sofia Agelaki, Simon Ekman, and Georgios Tsakonas. 2021. "Correlation of Clinical Parameters with Intracranial Outcome in Non-Small Cell Lung Cancer Patients with Brain Metastases Treated with Pd-1/Pd-L1 Inhibitors as Monotherapy" Cancers 13, no. 7: 1562. https://doi.org/10.3390/cancers13071562
APA StyleRounis, K., Skribek, M., Makrakis, D., De Petris, L., Agelaki, S., Ekman, S., & Tsakonas, G. (2021). Correlation of Clinical Parameters with Intracranial Outcome in Non-Small Cell Lung Cancer Patients with Brain Metastases Treated with Pd-1/Pd-L1 Inhibitors as Monotherapy. Cancers, 13(7), 1562. https://doi.org/10.3390/cancers13071562








