Should Lymph Nodes Be Retrieved in Patients with Intrahepatic Cholangiocarcinoma? A Collaborative Korea–Japan Study
Abstract
Simple Summary
Abstract
1. Introduction
2. Results
2.1. General Characteristics of Patients and Primary Tumors
2.2. Distribution of Metastatic Lymph Nodes in Patients with Resected IHCC
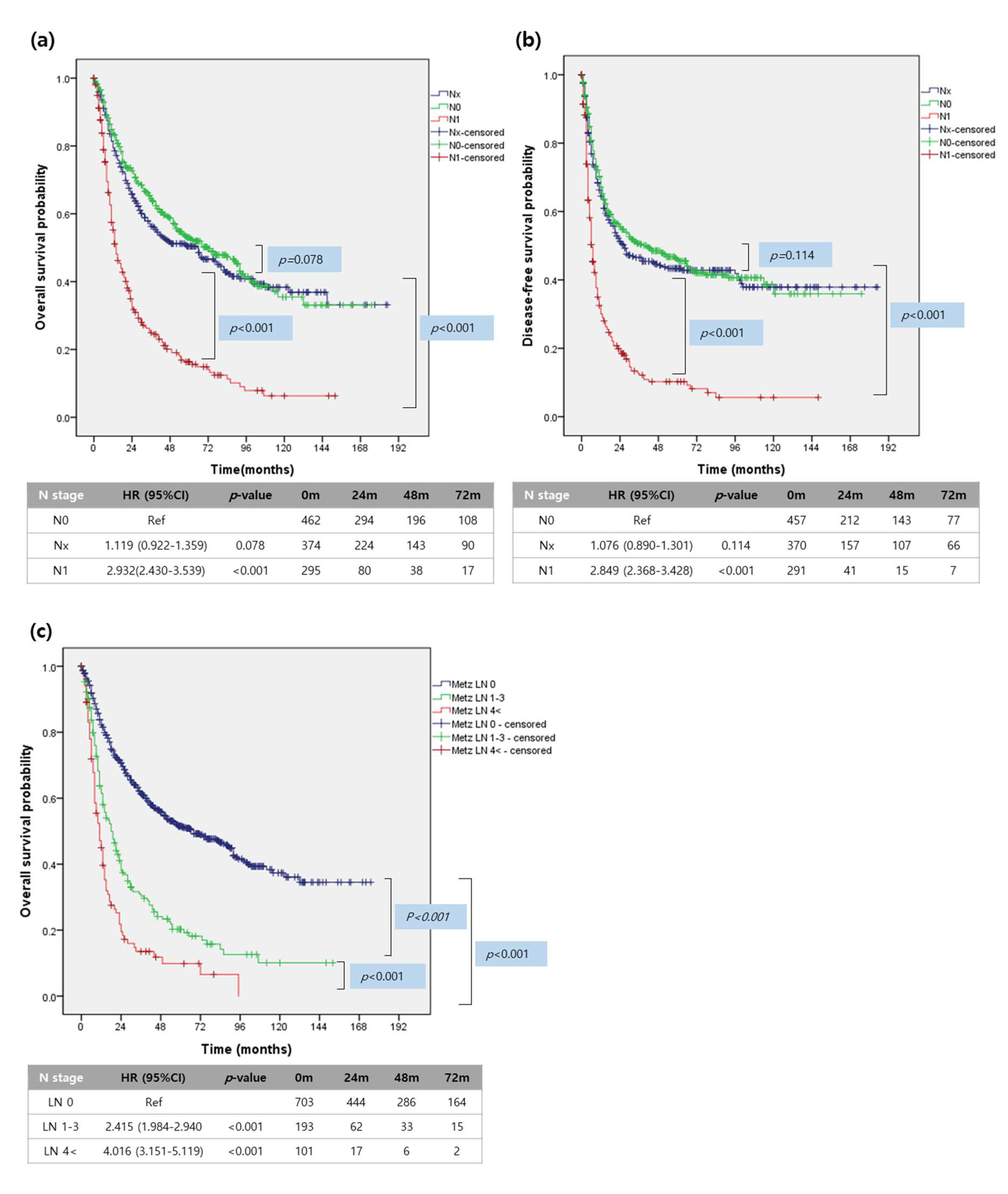
2.3. Oncologic Impact of Metastatic Lymph Nodes in Patients with Resected IHCC
2.4. Oncologic Impact of Number of Retrieved Lymph Nodes on N0, and N1 Patients with Resected IHCC
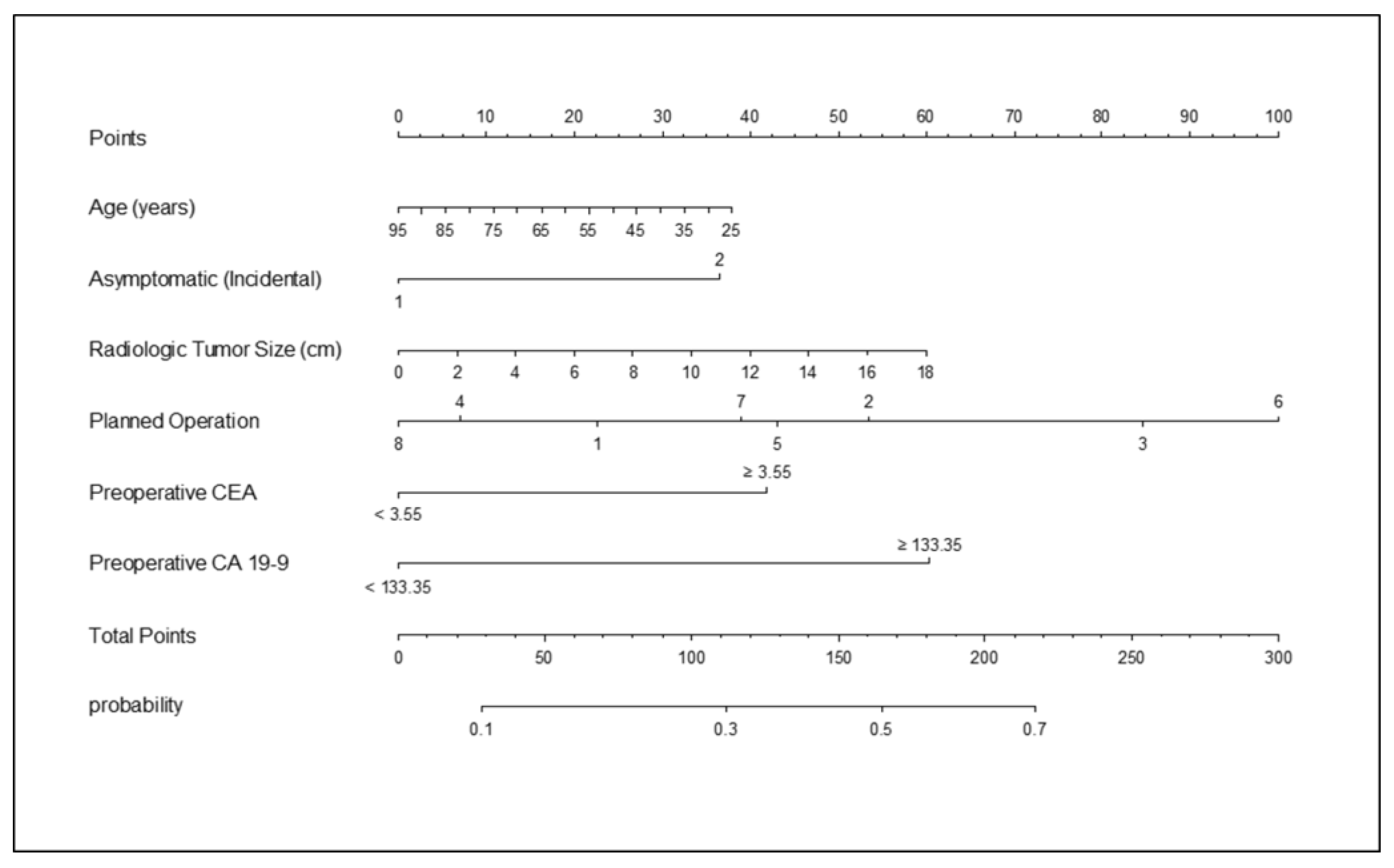
2.5. Can Lymph Node Metastasis Be Preoperatively Predicted in Patients with Resected IHCC? Developing a Surgeon-Oriented Nomogram to Predict Lymph Node Metastasis
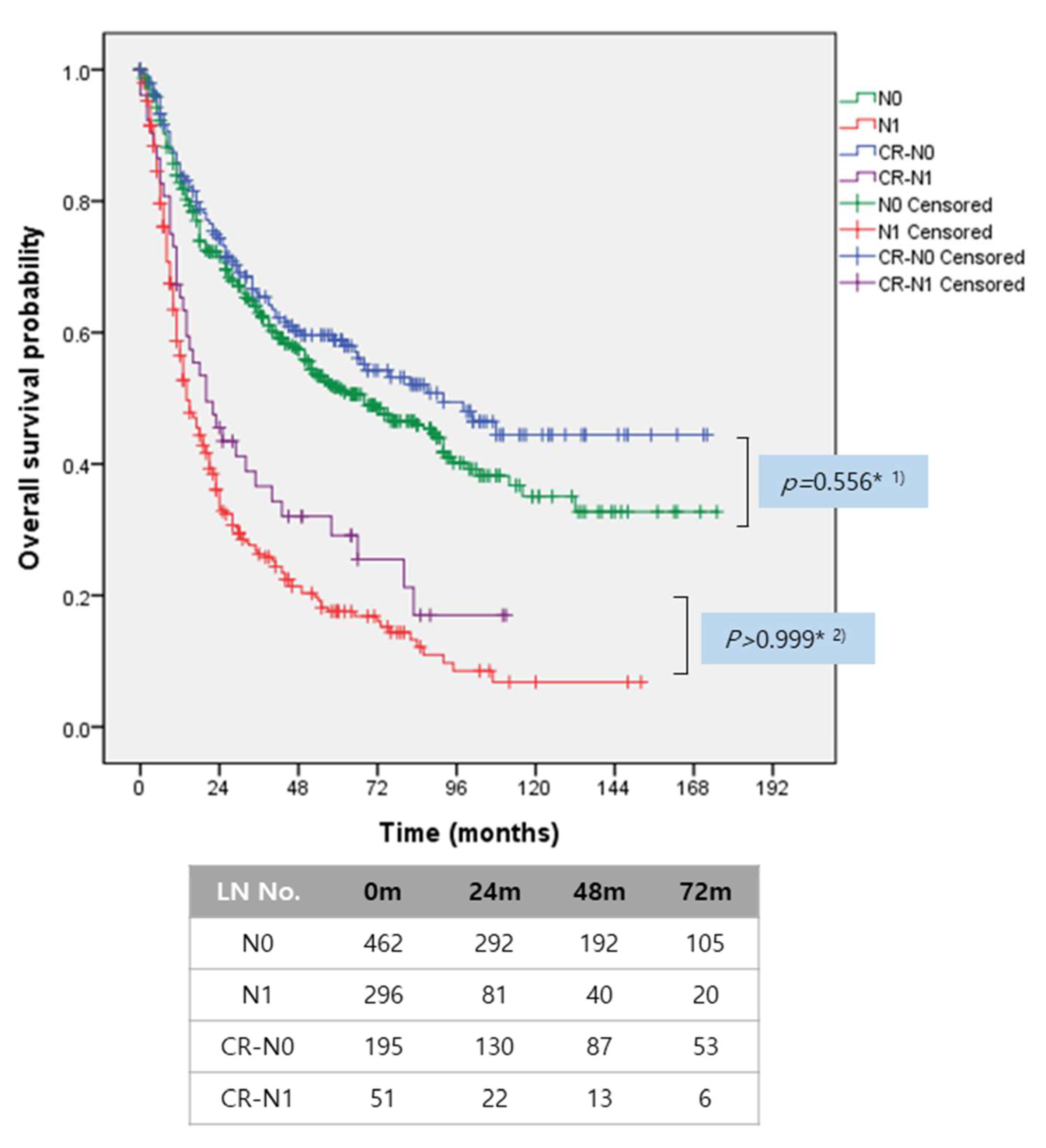
2.6. Indirect External Validation of Nomogram in Nx Patients with Resected IHCC
2.7. Proposed Surgical Strategy in Lymph Node Management for IHCC
3. Discussion
4. Materials and Methods
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Aljiffry, M.; Abdulelah, A.; Walsh, M.; Peltekian, K.; Alwayn, I.; Molinari, M. Evidence-based approach to cholangiocarcinoma: A systematic review of the current literature. J. Am. Coll. Surg. 2009, 208, 134–147. [Google Scholar] [CrossRef] [PubMed]
- Rahib, L.; Smith, B.D.; Aizenberg, R.; Rosenzweig, A.B.; Fleshman, J.M.; Matrisian, L.M. Projecting cancer incidence and deaths to 2030: The unexpected burden of thyroid, liver, and pancreas cancers in the United States. Cancer Res. 2014, 74, 2913–2921. [Google Scholar] [CrossRef] [PubMed]
- Massarweh, N.N.; El-Serag, H.B. Epidemiology of Hepatocellular Carcinoma and Intrahepatic Cholangiocarcinoma. Cancer Control 2017, 24. [Google Scholar] [CrossRef] [PubMed]
- Bridgewater, J.; Galle, P.R.; Khan, S.A.; Llovet, J.M.; Park, J.W.; Patel, T.; Pawlik, T.M.; Gores, G.J. Guidelines for the diagnosis and management of intrahepatic cholangiocarcinoma. J. Hepatol. 2014, 60, 1268–1289. [Google Scholar] [CrossRef]
- Liver Cancer Study Group of Japan. General rules for the clinical and pathological study of primary liver cancer. Jpn. J. Surg. 1997, 19, 98–129. [Google Scholar]
- Amin, M.B. AJCC Cancer Staging Manual, 8th ed.; Springer: Chicago, IL, USA, 2016. [Google Scholar]
- Uenishi, T.; Yamamoto, T.; Takemura, S.; Kubo, S. Surgical treatment for intrahepatic cholangiocarcinoma. Clin. J. Gastroenterol. 2014, 7, 87–93. [Google Scholar] [CrossRef][Green Version]
- Amini, N.; Ejaz, A.; Spolverato, G.; Maithel, S.K.; Kim, Y.; Pawlik, T.M. Management of lymph nodes during resection of hepatocellular carcinoma and intrahepatic cholangiocarcinoma: A systematic review. J. Gastrointest. Surg. 2014, 18, 2136–2148. [Google Scholar] [CrossRef] [PubMed]
- Hyder, O.; Marques, H.; Pulitano, C.; Marsh, J.W.; Alexandrescu, S.; Bauer, T.W.; Gamblin, T.C.; Sotiropoulos, G.C.; Paul, A.; Barroso, E.; et al. A nomogram to predict long-term survival after resection for intrahepatic cholangiocarcinoma: An Eastern and Western experience. JAMA Surg. 2014, 149, 432–438. [Google Scholar] [CrossRef]
- Adachi, T.; Eguchi, S. Lymph node dissection for intrahepatic cholangiocarcinoma: A critical review of the literature to date. J. Hepatobiliary Pancreat. Sci. 2014, 21, 162–168. [Google Scholar] [CrossRef]
- Guro, H.; Kim, J.W.; Choi, Y.; Cho, J.Y.; Yoon, Y.S.; Han, H.S. Multidisciplinary management of intrahepatic cholangiocarcinoma: Current approaches. Surg. Oncol. 2017, 26, 146–152. [Google Scholar] [CrossRef]
- Jang, J.Y.; Park, T.; Lee, S.; Kim, Y.; Lee, S.Y.; Kim, S.W.; Kim, S.C.; Song, K.B.; Yamamoto, M.; Hatori, T.; et al. Proposed Nomogram Predicting the Individual Risk of Malignancy in the Patients with Branch Duct Type Intraductal Papillary Mucinous Neoplasms of the Pancreas. Ann. Surg. 2017, 266, 1062–1068. [Google Scholar] [CrossRef]
- Kim, D.S.; Kim, B.W.; Hatano, E.; Hwang, S.; Hasegawa, K.; Kudo, A.; Ariizumi, S.; Kaibori, M.; Fukumoto, T.; Baba, H.; et al. Surgical Outcomes of Hepatocellular Carcinoma with Bile Duct Tumor Thrombus: A Korea-Japan Multicenter Study. Ann. Surg. 2018. [Google Scholar] [CrossRef]
- Kaibori, M.; Kon, M.; Kitawaki, T.; Kawaura, T.; Hasegawa, K.; Kokudo, N.; Ariizumi, S.; Beppu, T.; Ishizu, H.; Kubo, S.; et al. Comparison of anatomic and non-anatomic hepatic resection for hepatocellular carcinoma. J. Hepatobiliary Pancreat. Sci. 2017, 24, 616–626. [Google Scholar] [CrossRef] [PubMed]
- Dunnett, C.W. A Multiple Comparison Procedure for Comparing Several Treatments with a Control. J. Am. Stat. Assoc. 1955, 50, 1096–1121. [Google Scholar] [CrossRef]
- Kimura, K.; Amano, R.; Nakata, B.; Yamazoe, S.; Hirata, K.; Murata, A.; Miura, K.; Nishio, K.; Hirakawa, T.; Ohira, M.; et al. Clinical and pathological features of five-year survivors after pancreatectomy for pancreatic adenocarcinoma. World J. Surg. Oncol. 2014, 12, 360. [Google Scholar] [CrossRef] [PubMed]
- Picozzi, V.J.; Oh, S.Y.; Edwards, A.; Mandelson, M.T.; Dorer, R.; Rocha, F.G.; Alseidi, A.; Biehl, T.; Traverso, L.W.; Helton, W.S.; et al. Five-Year Actual Overall Survival in Resected Pancreatic Cancer: A Contemporary Single-Institution Experience from a Multidisciplinary Perspective. Ann. Surg. Oncol. 2017, 24, 1722–1730. [Google Scholar] [CrossRef]
- Winter, J.M.; Cameron, J.L.; Campbell, K.A.; Arnold, M.A.; Chang, D.C.; Coleman, J.; Hodgin, M.B.; Sauter, P.K.; Hruban, R.H.; Riall, T.S.; et al. 1423 pancreaticoduodenectomies for pancreatic cancer: A single-institution experience. J. Gastrointest. Surg. 2006, 10, 1199–1210. [Google Scholar] [CrossRef] [PubMed]
- Ferrone, C.R.; Pieretti-Vanmarcke, R.; Bloom, J.P.; Zheng, H.; Szymonifka, J.; Wargo, J.A.; Thayer, S.P.; Lauwers, G.Y.; Deshpande, V.; Mino-Kenudson, M.; et al. Pancreatic ductal adenocarcinoma: Long-term survival does not equal cure. Surgery 2012, 152, S43–S49. [Google Scholar] [CrossRef]
- Birnbaum, D.J.; Vigano, L.; Russolillo, N.; Langella, S.; Ferrero, A.; Capussotti, L. Lymph node metastases in patients undergoing surgery for a gallbladder cancer. Extension of the lymph node dissection and prognostic value of the lymph node ratio. Ann. Surg. Oncol. 2015, 22, 811–818. [Google Scholar] [CrossRef]
- Liu, G.J.; Li, X.H.; Chen, Y.X.; Sun, H.D.; Zhao, G.M.; Hu, S.Y. Radical lymph node dissection and assessment: Impact on gallbladder cancer prognosis. World J. Gastroenterol. 2013, 19, 5150–5158. [Google Scholar] [CrossRef]
- Guglielmi, A.; Ruzzenente, A.; Campagnaro, T.; Valdegamberi, A.; Bagante, F.; Bertuzzo, F.; Conci, S.; Iacono, C. Patterns and prognostic significance of lymph node dissection for surgical treatment of perihilar and intrahepatic cholangiocarcinoma. J. Gastrointest. Surg. 2013, 17, 1917–1928. [Google Scholar] [CrossRef] [PubMed]
- Zhang, X.F.; Chakedis, J.; Bagante, F.; Chen, Q.; Beal, E.W.; Lv, Y.; Weiss, M.; Popescu, I.; Marques, H.P.; Aldrighetti, L.; et al. Trends in use of lymphadenectomy in surgery with curative intent for intrahepatic cholangiocarcinoma. Br. J. Surg. 2018, 105, 857–866. [Google Scholar] [CrossRef] [PubMed]
- Lendoire, J.C.; Gil, L.; Imventarza, O. Intrahepatic cholangiocarcinoma surgery: The impact of lymphadenectomy. Chin. Clin. Oncol. 2018. [Google Scholar] [CrossRef] [PubMed]
- Bagante, F.; Gani, F.; Spolverato, G.; Xu, L.; Alexandrescu, S.; Marques, H.P.; Lamelas, J.; Aldrighetti, L.; Gamblin, T.C.; Maithel, S.K.; et al. Intrahepatic Cholangiocarcinoma: Prognosis of Patients Who Did Not Undergo Lymphadenectomy. J. Am. Coll. Surg. 2015, 221, 1031–1040. [Google Scholar] [CrossRef]
- Doussot, A.; Groot-Koerkamp, B.; Wiggers, J.K.; Chou, J.; Gonen, M.; DeMatteo, R.P.; Allen, P.J.; Kingham, T.P.; D’Angelica, M.I.; Jarnagin, W.R. Outcomes after Resection of Intrahepatic Cholangiocarcinoma: External Validation and Comparison of Prognostic Models. J. Am. Coll. Surg. 2015, 221, 452–461. [Google Scholar] [CrossRef] [PubMed]
- Mavros, M.N.; Economopoulos, K.P.; Alexiou, V.G.; Pawlik, T.M. Treatment and Prognosis for Patients with Intrahepatic Cholangiocarcinoma: Systematic Review and Meta-analysis. JAMA Surg. 2014, 149, 565–574. [Google Scholar] [CrossRef]
- Meng, Z.W.; Lin, X.Q.; Zhu, J.H.; Han, S.H.; Chen, Y.L. A nomogram to predict lymph node metastasis before resection in intrahepatic cholangiocarcinoma. J. Surg. Res. 2018, 226, 56–63. [Google Scholar] [CrossRef]
- Liang, W.; Xu, L.; Yang, P.; Zhang, L.; Wan, D.; Huang, Q.; Niu, T.; Chen, F. Novel Nomogram for Preoperative Prediction of Early Recurrence in Intrahepatic Cholangiocarcinoma. Front. Oncol. 2018, 8, 360. [Google Scholar] [CrossRef]
- Shimada, M.; Yamashita, Y.; Aishima, S.; Shirabe, K.; Takenaka, K.; Sugimachi, K. Value of lymph node dissection during resection of intrahepatic cholangiocarcinoma. Br. J. Surg. 2001, 88, 1463–1466. [Google Scholar] [CrossRef]
- Schisterman, E.F.; Perkins, N.J.; Liu, A.; Bondell, H. Optimal cut-point and its corresponding Youden Index to discriminate individuals using pooled blood samples. Epidemiology 2005, 16, 73–81. [Google Scholar] [CrossRef]
- Hosmer, D.W., Jr.; Lemeshow, S.; Sturdivant, R.X. Applied Logistic Regression, 3rd ed.; Wiley: Hoboken, NJ, USA, 2013. [Google Scholar]

| Patients (N = 1138) | |
|---|---|
| Sex | |
| Male | 758 (66.6%) |
| Female | 380 (33.4%) |
| Age (years) | 63.4 ± 9.8 |
| Previous symptoms | |
| Yes | 535 (47.0%) |
| No | 603 (53.0%) |
| American Sosciety of Anesthesiologists (ASA) score | |
| 1 | 250 (24.6%) |
| 2 | 662 (65.2%) |
| 3 | 98 (9.6%) |
| 4 | 1 (0.1%) |
| 5 | 1 (0.1%) |
| Karnofsky scale | |
| 50 | 1 (0.1%) |
| 60 | 5 (0.5%) |
| 70 | 25 (2.6%) |
| 80 | 142 (14.8%) |
| 90 | 518 (54.1%) |
| 100 | 267 (27.9%) |
| Tumor location side | |
| Right liver | 558 (50.4%) |
| Left liver | 549 (49.6%) |
| Operation name | |
| Lt. lateral sectionectomy | 75 (6.6%) |
| Lt hemihepatectomy | 325 (28.6%) |
| Lt extended hepatectomy | 126 (11.1%) |
| Rt hemihepatectomy | 295 (25.9%) |
| Rt extended hepatectomy | 79 (6.9%) |
| Trisectionectomy | 31 (2.7%) |
| Bisegmentectomy | 65 (5.7%) |
| Segmentectomy | 142 (12.5%) |
| Lymph node retrieval | |
| No | 413 (36.3%) |
| Yes | 725 (63.7%) |
| Serum Carcinoembryonic antigen (CEA) (ng/mL) | 21.5 ± 111.3 |
| Serum 19-9 (U/mL) | 2273.9 ± 10,816.4 |
| Tumor Side | Right-Sided (N = 112) | Left-Sided (N = 181) | p |
|---|---|---|---|
| No. 7 Lymph node (LN) | 0.013 | ||
| No | 109 (97.3%) | 162 (89.5%) | |
| Metastasis | 3 (2.7%) | 19 (10.5%) | |
| No. 12 LN | 0.065 | ||
| No | 47 (42.0%) | 96 (53.0%) | |
| Metastasis | 65 (58.0%) | 85 (47.0%) | |
| No. 13 LN | 0.024 | ||
| No | 93 (83.0%) | 166 (91.7%) | |
| Metastasis | 19 (17.0%) | 15 (8.3%) | |
| Perigastric LN | 0.052 | ||
| No | 109 (97.3%) | 166 (91.7%) | |
| Metastasis | 3 (2.7%) | 15 (8.3%) |
| Variable | Univariate | Multivariate | ||
|---|---|---|---|---|
| OR (95% CI) | p-Value | OR (95% CI) | p-Value | |
| Sex (Male/Female) | 1.08 (0.798–1.463) | 0.6164 | ||
| Age, years | 0.989 (0.974–1.004) | 0.1401 | 0.991 (0.974–1.009) | 0.3427 |
| Chief complaint (no/yes) | 1.81 (1.344–2.438) | 0.0001 | 1.803 (1.245–2.612) | 0.0018 |
| ASA | 0.727 (0.505–1.046) | 0.0854 | ||
| 0.873 (0.479–1.591) | 0.6575 | |||
| Karnofsky score | 0.995 (0.975–1.016) | 0.657 | ||
| Radiologic tumor size, cm | 1.092 (1.033–1.156) | 0.0021 | 1.055 (0.982–1.134) | 0.1415 |
| Gross type | 0.917 (0.748–1.124) | 0.4023 | ||
| Tumor location (Right/Left) | 1.546 (1.148–2.082) | 0.0042 | ||
| Number of the tumor | 1.303 (0.985–1.723) | 0.064 | ||
| Left hemihepatectomy | 1.122 (0.555–2.268) | 0.7495 | 1.646 (0.685–3.952) | 0.2649 |
| Left extended hemihepatectomy | 1.608 (0.755–3.425) | 0.2185 | 2.713 (1.079–6.825) | 0.0339 |
| Right hemihepatectomy | 0.651 (0.315–1.343) | 0.2448 | 0.777 (0.319–1.896) | 0.5799 |
| Right extended hemihepatectomy | 1.004 (0.44–2.288) | 0.9934 | 1.39 (0.515–3.752) | 0.5156 |
| Trisectionentectomy | 2.514 (0.804–7.862) | 0.1129 | 3.488 (0.925–13.15) | 0.0651 |
| Bisegmentectomy | 0.933 (0.365–2.384) | 0.8854 | 1.301 (0.411–4.117) | 0.6539 |
| Segmentectomy | 0.304 (0.121–0.766) | 0.0116 | 0.694 (0.233–2.066) | 0.5119 |
| Preoperative CEA | 2.113 (1.537–2.903) | <0.0001 | 1.966 (1.352–2.857) | 0.0004 |
| Preoperative CA 19-9 | 3.389 (2.475–4.643) | <0.0001 | 2.648 (1.837–3.819) | <0.0001 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Kang, C.M.; Suh, K.-S.; Yi, N.-J.; Hong, T.H.; Park, S.J.; Ahn, K.S.; Hayashi, H.; Choi, S.B.; Jeong, C.-Y.; Takahara, T.; et al. Should Lymph Nodes Be Retrieved in Patients with Intrahepatic Cholangiocarcinoma? A Collaborative Korea–Japan Study. Cancers 2021, 13, 445. https://doi.org/10.3390/cancers13030445
Kang CM, Suh K-S, Yi N-J, Hong TH, Park SJ, Ahn KS, Hayashi H, Choi SB, Jeong C-Y, Takahara T, et al. Should Lymph Nodes Be Retrieved in Patients with Intrahepatic Cholangiocarcinoma? A Collaborative Korea–Japan Study. Cancers. 2021; 13(3):445. https://doi.org/10.3390/cancers13030445
Chicago/Turabian StyleKang, Chang Moo, Kyung-Suk Suh, Nam-Joon Yi, Tae Ho Hong, Sang Jae Park, Keun Soo Ahn, Hiroki Hayashi, Sae Byeol Choi, Chi-Young Jeong, Takeshi Takahara, and et al. 2021. "Should Lymph Nodes Be Retrieved in Patients with Intrahepatic Cholangiocarcinoma? A Collaborative Korea–Japan Study" Cancers 13, no. 3: 445. https://doi.org/10.3390/cancers13030445
APA StyleKang, C. M., Suh, K.-S., Yi, N.-J., Hong, T. H., Park, S. J., Ahn, K. S., Hayashi, H., Choi, S. B., Jeong, C.-Y., Takahara, T., Shiozaki, S., Roh, Y. H., Yu, H. C., Fukumoto, T., Matsuyama, R., Naoki, U., Hashida, K., Seo, H. I., Okabayashi, T., ... Choi, D. W. (2021). Should Lymph Nodes Be Retrieved in Patients with Intrahepatic Cholangiocarcinoma? A Collaborative Korea–Japan Study. Cancers, 13(3), 445. https://doi.org/10.3390/cancers13030445








