Simple Summary
Cannabinoids have been shown to suppress tumour cell proliferation, tumour invasion, metastasis, angiogenesis, chemoresistance and epithelial-mesenchymal transition and to induce tumour cell apoptosis, autophagy and immune response. This review focuses on the current status of investigations on the impact of inhibitors of endocannabinoid-degrading enzymes on tumour growth and spread in preclinical oncology research.
Abstract
Despite the long history of cannabinoid use for medicinal and ritual purposes, an endogenous system of cannabinoid-controlled receptors, as well as their ligands and the enzymes that synthesise and degrade them, was only discovered in the 1990s. Since then, the endocannabinoid system has attracted widespread scientific interest regarding new pharmacological targets in cancer treatment among other reasons. Meanwhile, extensive preclinical studies have shown that cannabinoids have an inhibitory effect on tumour cell proliferation, tumour invasion, metastasis, angiogenesis, chemoresistance and epithelial-mesenchymal transition (EMT) and induce tumour cell apoptosis and autophagy as well as immune response. Appropriate cannabinoid compounds could moreover be useful for cancer patients as potential combination partners with other chemotherapeutic agents to increase their efficacy while reducing unwanted side effects. In addition to the direct activation of cannabinoid receptors through the exogenous application of corresponding agonists, another strategy is to activate these receptors by increasing the endocannabinoid levels at the corresponding pathological hotspots. Indeed, a number of studies accordingly showed an inhibitory effect of blockers of the endocannabinoid-degrading enzymes fatty acid amide hydrolase (FAAH) and monoacylglycerol lipase (MAGL) on tumour development and spread. This review summarises the relevant preclinical studies with FAAH and MAGL inhibitors compared to studies with cannabinoids and provides an overview of the regulation of the endocannabinoid system in cancer.
1. Introduction
Individual components of the endocannabinoid system have been intensively studied in recent decades and evaluated as potential targets of pharmacological interventions in systemic tumour therapy. N-arachidonoylethanolamine (anandamide, AEA) and 2-arachidonoylglycerol (2-AG) were the first lipids discovered as endogenously synthesised agonists at cannabinoid receptors [1,2]. Other endogenously formed cannabinoid compounds are 2-arachidonoylglycerol ether (noladin ether, 2-AGE) [3], N-arachidonoyldopamine (NADA) [4] and O-arachidonoylethanolamine (virodhamine) [5]. Cannabinoid-triggered receptors include the pertussis toxin-sensitive, Gi/o protein-coupled cannabinoid receptors CB1 and CB2 [6,7]. The phytocannabinoid Δ9-tetrahydrocannabinol (THC), the main psychoactive constituent of Cannabis sativa L., has the properties of a partial agonist at the CB1 receptor and a full agonist at the CB2 receptor with Ki values in the nanomolar range, while the non-psychoactive phytocannabinoid cannabidiol (CBD) has a much weaker affinity for cannabinoid receptors, with Ki values about 1000-fold higher and inverse agonist effects at the CB1 and CB2 receptors [8]. Other receptors modulated by cannabinoid compounds include transient receptor potential family cation channels, such as transient receptor potential vanilloid 1 (TRPV1) activated by AEA [9] and CBD [10]. Among G protein-coupled receptors (GPCRs), GPR55, a rhodopsin-like class A GPCR [11], has been described to be antagonised by CBD and activated by various cannabinoids, such as CP 55,940, virodhamine and AEA, as well as by the endocannabinoid-like substance palmitoylethanolamide (PEA) [12]. In further studies, endocannabinoids such as AEA or structurally similar N-acylethanolamines, also known as endocannabinoid-like compounds such as PEA, oleoylethanolamide (OEA), stearoylethanolamide (SEA) and linoleoylethanolamide (LEA), have been described as activators of the peroxisome proliferator-activated receptor (PPAR)-α [13]. Endocannabinoid-like compounds utilise the biosynthetic and degradative enzymes of endocannabinoids but do not trigger activation of the cannabinoid receptors (for review see [14]).
The enzyme N-acyl phosphatidylethanolamine phospholipase D (NAPE-PLD) synthesises AEA and other N-acylethanolamines from membrane phospholipids. AEA is also produced via alternative biosynthetic pathways. On the other hand, 2-AG is generated by phospholipase C or by diacylglycerol lipase (DAGL) α and β (for review see [15]). AEA is catabolised by serine hydrolase fatty acid amide hydrolase (FAAH) [16], with FAAH-1 having much greater hydrolytic activity towards AEA than the FAAH-2 form, which is not expressed by rodents. The degradation of 2-AG is predominantly mediated by monoacylglycerol lipase (MAGL) or by α/β-hydrolase domain containing 6 (ABHD6) and 12 (ABHD12) [17]. Increased MAGL activity in tumour tissue is also the source of free fatty acids, which promote the growth and spread of tumours in the body through the formation of oncogenic lipids [18]. In addition, AEA and 2-AG were found to be oxidised by cyclooxygenase-2 (COX-2), leading to the corresponding prostaglandin (PG) ethanolamides and glycerol esters, respectively (for review see [14]).
In recent years, an increasing number of compounds designed to inhibit FAAH and MAGL have been tested for their preclinical potential, including antitumour activity as described later. The rationale here is to increase systemic and local endocannabinoid concentrations. Thus, shortly after the discovery of AEA degradation by FAAH [16], the same group introduced the synthetic fatty acid compound arachidonoyl trifluoromethyl ketone, which shows inhibitory properties towards FAAH with simultaneous binding affinity to the CB1 receptor [19]. However, this inhibitor still exhibited potent inhibitory action toward cytosolic phospholipase A2 (cPLA2) [20]. One of the first FAAH inhibitors that did not exhibit inhibition of cPLA2 and/or binding affinity to the CB1 cannabinoid receptor was N-arachidonoyl-serotonin (AA-5HT) [21]. This was later followed by URB597, a more potent FAAH inhibitor that even increased AEA levels in the brain, thereby exhibiting benzodiazepine-like properties [22].
A MAGL inhibitor tested in 2005 is the N-biphenyl carbamate URB602, which showed selectivity towards MAGL compared to FAAH inhibitor URB597 in an early attempt to establish MAGL inhibitors [23]. However, a study investigating the specificity of URB602 in measuring the inhibition of AEA hydrolysis in brain membranes found an IC50 value of 17 µM, while the IC50 value for MAGL inhibition was found to be 25 µM [24]. On the other hand, JZL184, a piperidine carbamate, proved to be a highly potent and selective MAGL inhibitor, irreversibly inhibiting the enzyme by carbamoylation of the active site catalytic serine nucleophile (Ser122). JZL184 shows IC50 values of 2 nM on murine and human MAGL and 25 nM on rat MAGL [25]. Later, MAGL inhibitors with lower cross-reactivity were synthesised, such as the N-hydroxysuccinimidyl carbamate MJN110, which showed an IC50 of 2.1 nM at MAGL in murine brain homogenates with concomitant lack of inhibition on AEA hydrolysis up to a concentration of 50 µM [26]. The O-aryl, O-hexafluoroisopropyl carbamate JW651 was found to be another selective inhibitor of MAGL, with an IC50 of 38 nM, showing less cross-reactivity with other brain serine hydrolases, such as ABHD6 (IC50 at 10.38 µM) and FAAH (IC50 > 100 µM), and inducing a 5- to 10-fold increase in brain 2-AG concentration in mice after doses of 5 to 40 mg/kg [27]. Currently, the hexafluoroisopropylcarbamate-based irreversible MAGL inhibitor ABX-1431 [28], developed in 2018, is being tested in clinical trials for its effect in, among others, neuralgias, neuropathies, motor tic disorders or Tourette’s syndrome [29]. Although appropriate clinical tests of MAGL inhibitors for efficacy and safety in relation to use as systemic cancer therapy are pending, the preclinical data presented below provide hope that such cannabinoid compounds could decisively expand the armamentarium for the treatment of tumour diseases.
It should also be mentioned that it was recently found in the distantly related roundworm C. elegans that JZL184 causes a prolongation of the lifespan despite the absence of a MAGL ortholog [30]. The cause of the effect of JZL184 was identified as an inhibition of the enzyme FAAH-4, which in this organism fulfils the MAGL-equivalent function of degrading endocannabinoid-related monoacylglycerides.
The chemical structures of the most important FAAH and MAGL inhibitors are shown together with the biochemical degradation reactions of AEA and 2-AG via FAAH and MAGL in Figure 1.
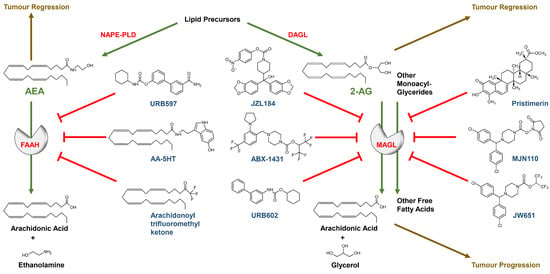
Figure 1.
Chemical structures of selected FAAH and MAGL inhibitors, endocannabinoids and their degradation products. Green arrows: enzymatic activity; red blocking arrows: blockade of enzymatic activity; brown arrows/letters: functional influence on tumour development; red letters: enzymes; green letters: FAAH and MAGL substrates; blue letters: inhibitors of enzymes; black letters: starting compound of biosynthesis and metabolic end products of endocannabinoid degradation. For illustration purposes, only the most important enzymes are listed.
2. Regulation of the Endocannabinoid System in Different Tumour Entities in Context with the Clinical Outcome of Cancer Patients
2.1. Regulation of Endocannabinoids in the Tumour Process
Despite many studies describing cannabinoid compounds and cannabinoid-activated receptors as mediators of anticarcinogenic effects, several investigations have reported increased activity of the endocannabinoid system in cancer tissue compared to healthy tissue and an associated unfavourable patient prognosis. However, particularly regarding the concentrations of endocannabinoids in the tumour, it is apparent that these regulations do not form a consensus of the overall profile.
Significantly elevated serum 2-AG levels have been detected, for example, in late-stage diffuse large B-cell lymphomas [31] and in biopsies from patients with endometrial carcinomas compared to healthy controls [32]. Interestingly, male patients with diffuse large B-cell lymphoma with a body mass index (BMI) ≥ 25 showed increased 2-AG levels compared to patients with a BMI < 25, while such effects were not detected in female patients [31]. Another study demonstrated increased AEA levels with almost unchanged 2-AG in colorectal carcinoma [33], whereas conversely a downregulation of AEA was observed in brain cancer tissue [34,35]. As can be seen from Table 1, endocannabinoid regulation does not correlate uniformly with tissue malignancy, the data on this are partly contradictory, and endocannabinoid levels are not yet reliable markers for tumour diseases according to current knowledge. Moreover, there are no robust studies to date linking endocannabinoid regulation to patient survival.

Table 1.
Regulation of endocannabinoids, cannabinoid receptors, FAAH, MAGL and NAPE-PLD in tumour tissues (in a few labelled cases serum/plasma) from cancer patients.
2.2. Regulation of Cannabinoid Receptors in the Tumour Process
Regarding the regulation of cannabinoid receptors in malignant tissues, similar to the regulation of endocannabinoids, studies have also shown partly contradictory results. Thereby, the majority of studies point to an upregulation of the CB2 receptor in tumour tissues. This is the case, for example, in non-small cell lung cancer (NSCLC) [75] (Table 1), and is consistent with other publications that have also found an association between CB2 receptor upregulation and cancer development and poorer survival, such as in squamous cell carcinoma of the head and neck [68], renal cell carcinoma [95] and HER2-positive breast cancer [47]. In addition, recent studies have reported that increased expression of the human epidermal growth factor 2 (HER2)-CB2 receptor heteromer in breast tumours is associated with lower disease-free survival in patients [48] and that lower CB2 levels in tumour-associated macrophages from colorectal cancer patients are associated with longer survival [57]. On the other hand, longer survival was found to be associated with higher expression of CB2 in hepatocellular carcinoma [69], lung cancer [76] and mobile tongue squamous cell carcinoma [83].
Analyses of CB1 receptor expression in cancer cells or tissues have yielded more pronounced conflicting results. An early study reported that the CB1 receptor in astrocytoma tissue had no dynamics associated with disease severity, while CB2 receptor expression in grade IV astrocytomas was higher than in lower grade tumours [37]. Another investigation concluded an association between high expression of the CB1 receptor in malignant tissues of patients with pancreatic cancer and poor patient prognosis, whereas CB2 receptor immunoreactivity did not correlate with survival [86]. Similar results have been reported for patients with prostate cancer, in whom high CB1 expression was associated with shorter survival [89]. In addition, high expression of CB1 correlated with metastasis to lymph nodes and distant organs as well as poor prognosis in esophageal squamous cell carcinoma [64]. The authors found no association with other factors such as age, sex, histologic differentiation and pathologic stage and thus concluded that immunohistochemical detection of CB1 could serve as a useful diagnostic marker for predicting metastases in lymph nodes and distant organs. In colorectal cancer patients undergoing surgical resection, low CB1 receptor expression was found more frequently in stage IV than in stage I/II or III [54]. Interestingly, stage IV patients with high CB1 expression in the tumours had significantly worse overall survival than patients of this stage with low CB1 expression, with no corresponding differences found in patients at other stages. In contrast, cannabinoid receptor regulation in lung cancer tissues was associated with increased survival in patients with high expression of CB1 [76]. A recent study investigating a possible function of cannabinoid receptors in the development and progression of metastases in patients with colorectal cancer found significant downregulation of CB1 at the mRNA and protein levels in cancer tissue compared to healthy mucosa. Interestingly, CB1 was also downregulated in patients with metastases in both normal mucosa and tumour tissue compared to patients without metastases, which is a possible indication that the development of metastases may be related to a reduction in CB1 receptor-dependent signalling pathways [58].
More detailed studies have shown how complex the regulation of cannabinoid receptors in tumour tissue actually is and that a simplified use of these receptor regulations as tumour markers is only possible to a limited extent. In some cancers, cannabinoid receptor regulations appear to even depend on specific molecular subsets of the cancers, such as the increased CB1 receptor expression in TP53-mutated Sonic Hedgehog (SHH) disease compared to other molecular categories within the medulloblastoma group [63]. In this study, none of the four molecular groups of medulloblastoma (wingless [WNT], SHH, group 3 and group 4) exhibited an association with differences in CB2 expression [63]. A publication focusing on gender differences in the expression of cannabinoid receptors in patients with mobile squamous cell carcinoma of the tongue reported that CB2 and simultaneous CB1/CB2 inductions were significantly more frequent in female patients than in male patients [83]. In the latter study, increased expression of CB1 in mobile squamous cell carcinomas of the tongue was associated with improved survival. A further investigation regarding the regulation of cannabinoid receptors in malignant tissue was also able to show a significant increase in CB1 receptors in early hepatocarcinomas [69]. Here, the Kaplan-Meyer curves revealed significant disease-free survival with high CB1 expression and, conversely, a significant association between low CB1 expression and recurrence of hepatocarcinomas. Thus, although there is a tendency for upregulation of cannabinoid receptors in cancer tissue (Table 1), this tendency cannot be consistently associated with unfavourable patient outcomes. Table 2 provides an overview of the regulation of cannabinoid receptors, FAAH and MAGL and the respective association with patient survival.

Table 2.
Relationship between expression levels of cannabinoid receptors, FAAH and MAGL and patient survival.
2.3. Regulation of Endocannabinoid-Synthesising and -Degrading Enzymes in the Tumour Process
A similar ambivalent expression pattern is currently assumed for endocannabinoid-synthesising and -degrading enzymes in malignant tissue. In one study, for example, NAPE-PLD, FAAH and MAGL were found to be downregulated in gliomas, while the expression of DAGL remained almost unchanged [34]. MAGL expression has been found to be downregulated in cancer tissue in some studies [32,34,46,49,57,74,86] but upregulated in others [18,43,53,62,71,72,73,78,82,84,92] (see Table 1).
In one study, low FAAH and MAGL immunoreactivities in cancer cells were correlated with shorter patient survival [86]. Accordingly, in this work, median survival with low FAAH levels in cancer cells was 10 months, whereas patients with moderate to strong FAAH immunoreactivity showed a median survival of 19.1 months. After resection of pancreatic cancer, patients with high MAGL levels had a median survival of 8 months, while moderate to strong MAGL staining was associated with a median survival of 21.8 months. However, this result contrasts with a report showing that MAGL worsened the prognosis of patients with hepatocellular carcinoma [71], where conversely low MAGL levels were associated with longer survival. In a further investigation on hepatocellular carcinoma, it was reported that the clinical prognosis for the group with high MAGL expression was significantly worse than for the group with low MAGL expression, in terms of overall survival times and recurrence rates [72]. In a study with 412 prostate cancer patients, higher FAAH levels were also associated with disease severity in a subgroup with intermediate but not high CB1 receptor levels [91]. In the latter work, higher expression of tumour epithelial FAAH was associated with a poor disease-specific survival for patients at the end stages of the disease. Another paper that focused specifically on MAGL regulation in human tumour-associated macrophages described that MAGL expression in macrophages from fresh carcinoma tissue was significantly lower than in adjacent normal tissue in colorectal cancer patients [57]. Remarkably, higher MAGL levels in tumour-associated macrophages from colorectal cancer patients were associated with better survival here, again suggesting that endocannabinoid system regulations should be interpreted with caution in terms of functional involvement in carcinogenic processes.
Furthermore, individual studies described opposite regulations of the same parameter, such as the regulation of MAGL in lung tumour tissue, which was upregulated once with patients exhibiting high MAGL expression associated with worse outcomes [78] and downregulated in malignant tissue in another study [46,74]. One reason for such discrepancies could be different patient cohorts. For example, while in Zhang et al. [78] only about 53% of the 156 patients were older than ≥60 years, the patient population studied by Liu et al. [74] was older (79% of the 34 patients ≥60 years). Finally, there are also studies that did not find an association of overall survival with FAAH expression in breast cancer patients [45] or MAGL expression in lung cancer patients’ overall survival [77]. The latter investigation, however, found the overall survival gradually reduced with increasing ABHD6 levels.
Upregulation of NAPE-PLD and FAAH mRNA has been noted in colorectal cancer tissue [33]. Elevated levels of NAPE-PLD, associated with increased AEA concentrations, have also been reported in human hepatocellular carcinoma compared to adjacent healthy tissue [70]. However, in a study with biopsies of hepatocellular carcinomas, no significant differences in NAPE-PLD mRNA were detected between tumours and non-cancerous controls [73]. The latter investigation also reported elevated 2-AG levels as a result of strong overexpression of DAGL α with less pronounced MAGL induction in hepatocellular cancer tissue; downregulation of AEA was found to be thereby dependent on higher expression levels of FAAH. In contrast to these observations, a decrease in FAAH protein expression and an increase in NAPE-PLD in cancerous versus non-cancerous endometrial tissue has recently been reported [61], resulting in increased AEA and PEA levels in cancerous endometrial tissue and in plasma from patients with endometrial carcinoma compared to volunteers with an atrophic endometrium [60].
Thus, the quantitative ratio of endocannabinoid-synthesising and -degrading enzymes to each other is crucial for the effect on endocannabinoid levels and thus tumour progression. As with endocannabinoids and cannabinoid receptors, the relationship between tumour progression and the expression of enzymes of the endocannabinoid system is complex and requires further investigation.
3. Systemic Effects of Cannabinoid Compounds on Different Levels of Carcinogenesis
The first studies and descriptions of tumour-regressive effects of cannabinoids date back to a 1975 publication [100], which reported that Δ8-tetrahydrocannabinol, THC and cannabinol suppressed tumour growth in a murine Lewis lung adenocarcinoma model, leading to a life prolongation of the experimental animals. However, relevant studies that made cannabinoid receptors a research topic as targets for systemic cancer therapy were only published from the late 1990s onwards [101].
There are now several publications showing that cannabinoid agents mediate antitumour effects via the activation of CB1, CB2 and TRPV1 or independently of these receptors (for review see [102]). In addition to directly influencing downstream signalling pathways induced via cannabinoid-triggered receptors through the application of exogenous agonists, a second strategy is to activate these receptors by increasing endocannabinoid levels at the corresponding pathological hotspots through the inhibition of endocannabinoid-degrading enzymes (for review see [14]). In this context, it is hypothesised that the endocannabinoid system, as part of an endogenous tumour defence mechanism, can counteract the growth of neoplasms and thus selective inhibitors of endocannabinoid degradation could support this tumour defence system. In addition to blocking FAAH, inhibition of MAGL as a potential antitumour strategy has also attracted the attention of scientists in recent years, as it not only induces anticarcinogenic effects by activating cannabinoid receptors via 2-AG, but also reduces a number of tumour-promoting fatty acids (Figure 1) that would contribute to tumour growth via various mechanisms when MAGL is overactive (for review see [14,102]).
In the following, the effects of cannabinoids and subsequently of inhibitors of endocannabinoid degradation on the key events of tumour development and spread are described respectively. In the case of the cannabinoids presented for reasons of comparison, only selected studies are mentioned.
3.1. Cancer Cell Proliferation and Viability
3.1.1. Effect of Cannabinoids on Cancer Cell Proliferation and Viability
Publications on growth-inhibiting effects of cannabinoids have accumulated over the last two decades. Studies from the beginning of the millennium focused on the mechanisms leading to cannabinoid-induced apoptosis and cell cycle arrest of cancer cells. Later, some extensive studies showed a connection between cannabinoid-induced autophagy and subsequent apoptosis.
A seminal paper on this topic was published in 2000, showing that THC and the synthetic cannabinoid WIN 55,212-2 induced growth inhibition of glioma xenografts in Wistar rats and in Rag2-/- mice [103]. The crucial mechanism identified was a sustained accumulation of ceramide as a second messenger leading to cell death of glioma cells, which was later confirmed for R(+)-methanandamide (Met-AEA)-induced apoptosis of neuroglioma cells [104,105]. Meanwhile growth inhibitory effects of various cannabinoid compounds have been reported in numerous studies. In this context, protein kinase B (Akt) inhibition has been repeatedly described as a key event in the growth inhibitory effects of cannabinoids on various types of tumour cells, as shown for instance for CB1 receptor-dependent WIN 55,212-2-induced cell cycle arrest in melanoma cells [106], CB1 receptor dependent HU210- and THC-induced apoptosis of rhabdomyosarcoma cells [97] or CB2 receptor-dependent THC- and JWH-133-induced apoptosis of breast cancer cells [107]. In this context, a number of cell cycle regulators have been found to be affected by cannabinoids [42,108].
In addition, recent studies have shown that the induction of autophagy is associated with these apoptotic effects and is involved in the toxicity of cannabinoid compounds to cancer cells. This was first demonstrated by Salazar et al. [109], who reported that THC induces autophagy in glioma cells via CB1 receptor- and ceramide-dependent phosphorylation of eukaryotic translation initiation factor 2α (eIF2α) and subsequent endoplasmic reticulum stress, induction of the stress-associated transcriptional coactivator p8 and upregulation of tribbles pseudokinase 3 (TRB3). These regulations eventually lead to autophagy via inhibition of Akt and mTORC1 (mammalian target of rapamycin complex 1). In this publication, autophagy represents a mechanism that ultimately leads to apoptosis and cell death of tumour cells. In another study with human hepatocellular carcinoma cells, it was shown that autophagy induction occurs through two different mechanisms, both of which also lead to tumour cell death. Here, THC and the CB2 agonist JWH-015 also induced TRB3 upregulation and subsequent inhibition of the Akt/mTORC1 axis, but a second pathway involved cannabinoid-induced activation of Ca2+/calmodulin-activated kinase kinase β (CaCMKKβ) with a causally linked downstream stimulation of adenosine monophosphate-activated kinase (AMPK), which ultimately caused autophagy [110]. The same authors later further demonstrated upregulation of PPARγ as a link between autophagy and apoptosis in hepatocellular carcinoma cells treated with THC or the selective CB2 agonist JWH-015 [111]. Further evidence for autophagy inductions was found for THC in melanoma cells [112] and for CBD in various cancer entities, such as glioma tumour cells [113], glioma stem-like cells [114], breast cancer [115], lymphoblastic leukaemia [116] and head and neck squamous cell carcinoma cells [117].
3.1.2. Effect of FAAH and MAGL Inhibition on Cancer Cell Proliferation and Viability
Consistent with the concept that an increase in endogenous endocannabinoid levels also counteracts cancer processes in the body, inhibitors of endocannabinoid-metabolising enzymes revealed an antiproliferative effect on cancer cells. One of the early pioneering studies in this field showed that arachidonoyl trifluoromethyl ketone, an inhibitor of the major AEA-degrading enzyme FAAH, enhanced the antiproliferative effect of exogenously added AEA on breast cancer cell lines [101]. Subsequently, several publications confirmed the anticancer effect of inhibitors of FAAH. For example, AA-5HT, another FAAH inhibitor, was described to cause growth inhibition of thyroid cancer xenografts [118] and to reduce tumour development in an in vivo model of colon carcinogenesis [119]. The inhibition of tumour cell growth by FAAH inhibition was later also shown in melanoma cells. Thus, a study demonstrated concentration-dependent apoptosis of melanoma cells after incubation with AEA, which was enhanced by inhibition of FAAH and attenuated by blockade of COX-2 or lipoxygenase (LOX), suggesting involvement of eicosanoid metabolism in this endocannabinoid effect [120]. In another study, antiproliferative effects on melanoma cells were described for PEA and 2-AG in addition to AEA [121]. This work focused primarily on the antiproliferative effect of PEA and its enhancement due to FAAH inhibition, which was finally also confirmed in vivo using a murine xenograft model [121]. In another study, FAAH inhibition in combination with 2-methyl-2′-fluoro-anandamide (Met-F-AEA) was shown to lead to cell cycle arrest in the G0/G1 phase and apoptosis in lung cancer cells, mediated by a reduction in epidermal growth factor receptor (EGFR) activation and downstream signalling pathways [122]. In addition to the aforementioned tumour regressive findings on AA-5HT, which caused a reduction in tumour growth alone without the addition of exogenous FAAH substrates [118,119], there are also other in vivo studies with FAAH inhibitors in which such effects were not observed. For example, no reduction in tumour development was demonstrated when URB597 alone was administered every third day for three weeks at a dose of 1 mg/kg in a lung tumour model [122] or daily for six days at a dose of 10 mg/kg in a melanoma model [121]. Similarly, using a lung tumour xenograft model, it was shown that both URB597 and AA-5HT did not significantly affect tumour growth when the compounds were administered alone at a dose of 10 mg/kg every 72 h for 28 days [123].
In recent years in particular, data have accumulated supporting the inhibition of MAGL as a promising concept for inhibiting cancer progression. On the one hand, inhibition of the key 2-AG-degrading enzyme MAGL by JZL184 exhibited positive effects on the quality of life of experimental animals in preclinical studies, such as reduced cachexia in a mouse model for bone cancer [92] and in lithium chloride-induced vomiting in tree shrews [124]. In addition, inhibition of MAGL has also been found to induce systemic anti-cancer effects, either by affecting the endocannabinoid system or by modulating endogenous lipids. In a first of these studies with breast, ovarian and melanoma cancer cells, a decrease in free fatty acids due to MAGL inhibition was considered to be the main cause of the anti-cancer potential of MAGL inhibition [18]. Consistent with this hypothesis, aggressive melanoma cells in which MAGL was knocked down by small hairpin (sh) RNA showed decreased tumour growth, which was completely counter regulated in mice fed a high-fat diet, associated with increased free fatty acid content in the tumours. Knockdown of MAGL or treatment with the MAGL inhibitor JZL184 also caused inhibition of prostate cancer xenograft growth [125]. Here, the authors found partial reversal of tumour growth defects observed with MAGL shRNA-transfected PC3 cells by treatment of mice with a high-fat diet or a CB1 receptor antagonist, and fully reversal by pre-treatment with both regimes. The importance of lipid modulation by MAGL inhibition was further underscored by a recent study showing that JZL184-mediated blockade of oleic acid-stimulated proliferation of glioblastoma cells occurs via modulation of triglyceride metabolism and not via changes in cannabinoid receptor signalling pathways [126]. A tumour-regressive effect has also been reported for JZL184 in a murine xenograft model with MAGL-overexpressing hepatocellular cancer cells [71]. The growth inhibitory effect of MAGL inhibition by knockdown or treatment with JZL184 was further corroborated in a colon cancer cell xenograft model and associated with downregulation of cyclin D1 and B-cell lymphoma 2 (Bcl-2) [53]. Other studies have confirmed these findings and additionally found an increase in Bcl-2-associated X protein (Bax) in response to treatment with JZL184 in colorectal cancer cells [127]. Knockdown of MAGL by transfection with a shRNA likewise inhibited lung cancer cell proliferation via inhibition of cyclin B1 and cyclin D1 expression [78]. A reduction in the expression of cyclin D1 and Bcl-2, which was associated with a reduction in cell cycle progression after treatment with JZL184 and by small interfering (si) RNA-mediated MAGL inhibition, was finally also demonstrated in endometrial cancer cells [62]. However, not all studies could support these positive effects of MAGL knockdown on cancer progression. Accordingly, a recent publication found that inhibition of MAGL enhances rather than impairs cancer progression in mice [74]. The authors obtained their results in MAGL knockout mice, which showed a higher incidence of neoplasia in multiple organs with splenomegaly and particularly promoting effects on lung cancer progression. MAGL deficiency was associated with increased EGFR expression and phosphorylation, as well as increased activation of p42/44 mitogen-activated protein kinase and Akt, and induction of COX-2 and tumour necrosis factor α.
The in vitro evidence on the effects of MAGL inhibition on tumour cell viability is ambiguous. In a recent study, JZL184 at concentrations between 0.01 and 10 µM was shown to neither significantly reduce viability and proliferation of lung cancer cells under both serum-containing and serum-free conditions [128]. The same work also demonstrated that JZL184 did not significantly alter the colony forming properties of lung tumour cells in this concentration range. On the other hand, MAGL siRNA led to a slight but significant decrease in viability [128]. These data are in line with observations in melanoma, ovarian and breast cancer cells, where knockdown of MAGL via transfection of shRNA but not pharmacological inhibition of MAGL with JZL184 decreased cellular viability, implying that long-term inhibition of MAGL may be necessary for this effect [18]. Another study found that a 3-day treatment of prostate cancer cells with JZL184 resulted in an increase in cell density under basal conditions but a decrease in epidermal growth factor (EGF)-induced proliferation [129]. No effect of JZL184 was observed on the viability of neuroblastoma cells after an incubation of 3 days [130]. Here, the authors observed a moderate toxic effect on neuroblastoma cells only induced by the MAGL inhibitors methyl arachidonyl fluorophosphonate (MAFP) and CAY1049 or in the presence of 2-AG. On the other hand, cytotoxic effects of JZL184 have been observed in endometrial carcinoma [62], colorectal cancer [53] and hepatocellular carcinoma cells [71]. Accordingly, the effect of MAGL inhibitors seems to depend on the particular conditions and cell line as well as on the duration of action and the concentration used.
Among the compounds known to affect MAGL activity, the plant triterpenoid quinone methide pristimerin has attracted attention in recent years as a reversible MAGL inhibitor [131]. Inhibition of cancer growth by pristimerin treatments was demonstrated in a murine lung cancer xenograft model [132]. In addition, pristimerin was shown to induce apoptosis, autophagy and cell cycle arrest in human breast cancer cells [133]. However, an effect on the endocannabinoid system as the basis of the anticancer properties of pristimerin was not investigated in the aforementioned works.
In general, there are no studies on the influence of MAGL inhibition on the induction of autophagy in tumour cells. However, it should be noted that the above studies overwhelmingly support the hypothesis that endocannabinoid-degrading enzymes, especially MAGL, are key parameters in the endogenous control of cancer growth. This makes these enzymes promising targets for innovative pharmacotherapeutic options that could benefit cancer patients.
3.2. Cancer Cell Invasion and Metastasis
3.2.1. Effect of Cannabinoids on Cancer Cell Invasion and Metastasis
Many investigations have addressed anti-invasive and antimetastatic effects of various cannabinoid compounds, as summarised elsewhere [102]. Early studies on this topic showed that 2-AG exerts a CB1-dependent anti-invasive effect on prostate cancer cells [134]. Later, anti-invasive effects were confirmed for AEA in glioma [135] and lung cancer cells [123] as well as for the endocannabinoid derivatives Met-F-AEA in human breast cancer cells [136] and for Met-AEA in cervical and lung cancer cells [137]. Both endocannabinoid derivatives caused anti-invasive effects via CB1 receptor activation. In the case of Met-AEA, the CB2 receptor and TRPV1 additionally contributed to the inhibition of invasion in the latter study, with THC exerting an invasion-inhibiting effect via CB1 and CB2 in the same system [137]. Thereby, both cannabinoids mediated the inhibition of invasion via an induction of the tissue inhibitor of metalloproteinase-1 (TIMP-1), which could later also be confirmed for CBD in cervical and lung cancer cells [138]. Another study found a causal relationship in lung cancer cells between THC-, CBD- and Met-AEA-induced invasion inhibition and the induction of the expression of intercellular adhesion molecule-1 (ICAM-1), which acts as an upstream regulator of TIMP-1 [139]. In the same work, ICAM-1 was also shown to mediate the antimetastatic effect of CBD in athymic nude mice. Further proteolytic regulations involved in anti-invasive cannabinoid action include downregulation of matrix metalloproteinase (MMP)-2 by THC in glioma cells [140], downregulation of MMP-2 and -9 in hepatocellular carcinoma cells treated with the CB2 receptor agonist CB65 and the CB1 receptor agonist arachidonyl-2′-chloroethylamide (ACEA) [141] and downregulation of plasminogen activator inhibitor-1 (PAI-1) as underlying mechanism of CBD-induced inhibition of lung cancer cell invasion [142]. Moreover, reduced MMP-2 release from cancer-associated fibroblasts after treatment with WIN 55-212.2 led to reduced invasion of prostate cancer cells, suggesting that cannabinoids inhibit invasion not only by direct action on tumour cells [143].
Besides the regulation of proteolytic enzymes as the underlying mechanism of the anti-invasive effect of cannabinoids, other studies showed that the anti-invasive mechanism of CBD also relies on downregulation of the inhibitor of basic helix-loop-helix transcription factors, Id-1, in breast cancer [144] and brain tumour cells [145]. The antimetastatic effect on breast cancer cells demonstrated for CBD in a mouse model [146] was shown in a later work to be reversible by ectopic expression of Id-1 in breast cancer cells [147], so that the antimetastatic action of CBD can also be directly linked to Id-1 downregulation.
3.2.2. Effect of FAAH and MAGL Inhibition on Cancer Cell Invasion and Metastasis
Some recent publications have revealed that regulation of the endocannabinoid system through inhibition of endocannabinoid-degrading enzymes has a profound effect on tumour cell invasion and metastasis. This involves some of the mechanisms that have also been demonstrated for the synthetic and phytocannabinoids. For example, CB2- and TRPV1-mediated induction of TIMP-1 was confirmed as the mechanism of anti-invasive action for the FAAH inhibitors AA-5HT and URB597, as well as for FAAH siRNA in human lung cancer cells [123]. In the latter study, beyond the in vitro results, administration of AA-5HT and URB597 and the FAAH substrates AEA, 2-AG, OEA and PEA to athymic nude mice was reported to have an inhibitory effect on metastatic infiltration of the lung by previously intravenously injected lung carcinoma cells.
In addition, there are other anti-invasive mechanisms that have been shown to be specific to the inhibition of endocannabinoid degradation. In this context, the role of hydrolysis of monoacylglycerols and thus the function of MAGL in cancer metastasis has been intensively researched. In a pioneering comprehensive investigation [18], shRNA-mediated silencing of MAGL was shown to inhibit migration and invasion of the melanoma cell line C8161 and the ovarian cancer cell line SKOV3, with the migration reduced by shRNA being reversed in both cell lines by treatment with fatty acids. In a follow-up study with prostate cancer cells, inhibition of cancer cell migration after MAGL inhibition by JZL184 or MAGL shRNA was partially reversed by the addition of fatty acids or CB1 receptor antagonists and completely reversed by the combination of both substances [125]. Therefore, activation of the CB1 receptor by the increase in 2-AG and a decrease in tumour-promoting free fatty acids, both induced by MAGL inhibition, appear to produce these effects on prostate cancer cells. Other work with hepatocellular carcinoma cells showed increased invasion when the cells overexpressed MAGL by knock-in and, conversely, decreased invasion when MAGL was knocked out or inhibited by JZL184 [71]. Therefore, the authors considered an increase in PGE2 and lysophosphatidic acid as promoters for increased proliferation and invasion as the mechanism for this effect. In another study, using an shRNA approach, it was found that inhibition of MAGL expression was associated with reductions in lung cancer cell invasiveness, lung cancer xenograft growth and lung cancer metastasis [78]. In this context, the authors described a downregulation of MMP-14 that mediated a reduction in invasiveness triggered by MAGL knockdown. Another recent study addressing the role of MMP-dependent proteolytic action in the context of the anti-invasive properties of MAGL inhibition revealed CB1 receptor-dependent anti-invasive and antimetastatic effects of JZL184 on human lung cancer cells [128], with anti-invasion mediated by increased TIMP-1 expression. In the latter study, a TIMP-1-dependent anti-invasive effect was confirmed for the MAGL substrate 2-AG. In addition, invasion inhibitory effects associated with increased TIMP-1 expression were observed for the MAGL inhibitors JW651 and MJN110 or when cells were transfected with MAGL siRNA. Finally, anti-invasive effects of JZL184 have also been shown for colorectal cancer [53] and hepatocellular carcinoma cells [71].
In further work, different immunodeficient mouse models were used to investigate the effects of JZL184 on bone cancer metastasis by intracardiac injection of osteotropic human prostate and breast cancer cells and by paratibial injection of human osteosarcoma cells [92]. The latter system was complemented by a murine osteosarcoma cell line as syngeneic model. Here, JZL184 inhibited skeletal tumour growth in all applied systems reduced bone metastasis of breast and prostate cancer cells, and inhibited the spread of osteosarcoma cells to the lung. These results are consistent with another work reporting that knockdown of MAGL reduced invasion of highly metastatic nasopharyngeal carcinoma cells and decreased the proportion of popliteal lymph node metastases in vivo [84]. The latter study also described that MAGL expression and metastatic potential of nasopharyngeal carcinoma cells were associated with a decrease in epithelial-mesenchymal transition (EMT) markers. Thus, MAGL-overexpressing cells showed increased vimentin and snail levels and decreased E-cadherin levels, which was reversed by knockdown of MAGL [84]. Inhibition of migration by JZL184 was also associated with inhibition of EMT, i.e., downregulation of vimentin and Snail and upregulation of E-cadherin in another study with colon cancer cells [127]. Consistent with the notion of MAGL as a proinvasive parameter, further investigation revealed that MAGL-overexpressing hepatocellular carcinoma cells exhibited higher invasiveness, which was causally linked to increased expression of nuclear factor-κB p65, which regulates downstream markers of EMT [72].
3.3. Tumour Angiogenesis
3.3.1. Effects of Cannabinoid Compounds on Tumour Angiogenesis
In several publications, a reduction of neovascularisation of xenograft tumours in mice treated with cannabinoids has been demonstrated, sometimes only as an additional finding, as described elsewhere (for review see [102]). In terms of possible mechanisms, early studies indicated that cannabinoids downregulate VEGF (vascular endothelial growth factor) and angiopoietin-2 (Ang-2) in mouse skin [148] and glioma tumours [149], with additional inhibition of MMP-2 formation described in glioma tumours. Later, the same group found modulation of several hypoxia-related angiogenesis markers by the CB2 receptor agonist JWH-133 in mouse gliomas, including hypoxia-inducible factor-1α, connective tissue growth factor, midkine, Id-3, heme oxygenase-1 and, in addition to Ang-2, its receptor tyrosine kinase, with immunoglobulin-like and EGF-like domains 1 (Tie-1) [150].
Other studies focused on the interaction of cancer cells and their associated microenvironment. For example, an investigation with conditioned media of CBD-, THC-, Met-AEA- and JWH-133-treated lung cancer cells showed an inhibition of tube and sprout formation as well as endothelial cell migration [151]. Here, the authors showed that the increased release of TIMP-1 by cannabinoids from lung tumour cells mediates inhibition of the angiogenic properties of endothelial cells in addition to the anti-invasive effect described above, suggesting a dual anticancer mechanism of cannabinoid-induced TIMP-1 expression. Another report addressing the interplay between the immune system and tumour cells showed that ACEA and JWH-133 inhibited lipopolysaccharide (LPS)-induced VEGF-A release from human neutrophils, which was associated with the decrease in the corresponding LPS-induced angiogenic capacities of bovine aortic endothelial cells [152].
3.3.2. Effects of FAAH and MAGL Inhibition on Tumour Angiogenesis
Regarding the effects of endocannabinoids, it was found that the conditioned medium of AEA-treated breast cancer cells inhibited the proliferation of endothelial cells, which was attributed to the downregulation of factors involved in angiogenesis, such as leptin, thrombopoietin and VEGF [153]. Interestingly, there are no data on the effects of FAAH inhibition on the interaction between cancer cells and endothelial cells, as has been reported for other cannabinoids. In the case of MAGL, one study found that the MAGL inhibitor URB602 reduced the growth of colon carcinoma xenografts, which was associated with reduced VEGF and fibroblast growth factor (FGF)-2 expression [154]. In the same work, URB602 reduced colon carcinogenesis in a murine model of azoxymethane-induced preneoplastic lesions, polyps and tumours in the colon, with the authors suggesting this effect to be dependent on the anti-angiogenic properties of URB602. Thus, MAGL inhibition suppressed FGF-2-induced proliferation and migration of endothelial cells. Overall, these results suggest a previously neglected effect of MAGL inhibition on cancer development and progression that should receive increased attention in future studies.
Finally, it was reported that the natural MAGL inhibitor pristimerin reduced sonic hedgehog (Shh)-induced angiogenesis by inhibiting VEGF receptor phosphorylation in endothelial cells [132]. However, a contribution of MAGL inhibition to this anti-angiogenic effect was not investigated here.
3.4. Tumour-Immune Interactions
3.4.1. Effect of Cannabinoid Compounds on Tumour-Immune Interactions
An impact of cannabinoids on the immune system and thus on its components that influence tumour growth has been discussed for a long time. Indeed, cannabinoids greatly affect immune cells, as recently reported using single-cell transcriptome mapping, where THC altered a variety of genes involved in immune response, cytokine production, cell proliferation and apoptosis of immune cells [155]. A further recent study performed on pancreatic cancer cells indicated an inhibitory effect of THC and CBD on the expression of programmed death ligand 1 (PD-L1), a key target of immune checkpoint blockade, through an upstream reduction in the expression and phosphorylation of activated p21 kinase 1 (PAK1) [156]. Finally, another recent paper demonstrated an inhibitory effect of the CB1 agonist ACEA and the CB2 agonist JWH-133 on VEGF release from neutrophils [152]. Further exemplary studies have shown a stronger tumour-regressive effect of the synthetic cannabinoid WIN 55,212-2 in immunocompetent compared to immunodeficient mice [106], a tumour-regressive effect of THC on melanomas associated with reduced infiltration of macrophages and neutrophils [157], and increased cancer cell lysis by lymphokine-activated killer cells after treatment of lung tumour cells with CBD, THC and Met-AEA [158].
However, there are also results showing that THC accelerates tumour implant growth by inhibiting antitumour immunity via a CB2 receptor-mediated, cytokine-dependent pathway in Lewis lung carcinoma and alveolar carcinoma models of immunocompetent mice [159]. In another study using breast cancer cells expressing low levels of cannabinoid receptors, THC was found to increase tumour growth and metastasis due to inhibition of the specific antitumour immune response in vivo. Accordingly, THC induced the profile of Th2 polarisation, resulting in an increase in Th2-associated cytokines and a downregulation of Th1-related cytokines [160]. Finally, another investigation showed that the CB2 receptor-activating effect of cannabinoids attenuates its own antitumour effect by promoting M2 polarisation of microglia/tumour-associated macrophages (TAMs), causing a proangiogenic effect of M2 microglia [161].
3.4.2. Effect of FAAH and MAGL Inhibition on Tumour-Immune Interactions
Accumulating data suggest that the endocannabinoid system acts as an important regulator of tumour immune defence through multiple mechanisms. For example, the involvement of the endocannabinoid system in the immune response of tumours was recently highlighted by the finding that the FAAH inhibitor URB597 may enhance the immune surveillance of human hepatocellular carcinoma cells [162]. Specifically, URB597 via induction of TIMP-3 expression was found to inhibit proteolytic shedding of the proteins major histocompatibility complex class I polypeptide-related sequence A (MICA) and B (MICB) on the surface of cancer cells, thereby ensuring binding of these proteins to the natural killer group 2D (NKG2D) receptor on the surface of cytolytic lymphocytes and subsequent killing of tumour cells.
Likewise, MAGL has become the focus of tumour-immune interactions in recent work. Using inoculated and genetic cancer models, one study demonstrated that MAGL is oppositely regulated in cancer tissues and tumour-associated macrophages (TAMs) [57]. In this context, the authors reported an interaction between the CB2 receptor and Toll-like receptor 4 (TLR4) in the membrane of TAMs. Thus, upregulation of MAGL in macrophages and associated decreased levels of 2-AG led to unloading of TLR4 signalling via an interaction with the CB2 receptor. At the same time, overexpression of MAGL resulted in increased levels of free fatty acids, leading to activation of TLR4 signalling and M1 polarisation of TAMs. Lipid accumulation in TAMs thereby promotes CB2/TLR4-dependent macrophage activation, inhibition of CD8+ T cells and thus tumour progression. On the other hand, decreased MAGL activity in macrophages in the tumour microenvironment resulted in activation of CB2 receptor binding and inactivation of TLR4 via increased 2-AG concentration. The inhibition of MAGL activity in TAMs accordingly led to growth and metastasis of cancer cells in the tumour microenvironment. Consistently, in a mouse model with MAGL-overexpressing myeloid cells, cancer growth in experimental colorectal carcinomas was slower than in the wild type [57].
Another comprehensive study on this topic also addressed the role of MAGL in the interplay between cancer and immune cells and found that arsenite resistance protein 2 (ARS2) transcriptionally upregulates MAGL and thereby regulates the self-renewal and tumourigenicity of glioma stem cells via increased synthesis of PGE2 [163]. Consistent with the in vitro findings here, MAGL-specific shRNA-expressing lentivirally infected glioblastoma stem cells orthotopically injected into nude mice showed less pronounced xenograft growth and the corresponding mice survived significantly longer than the mice receiving cells infected with the shRNA control construct. Thus, JZL184 increased the survival of the xenografted mice in association with a downregulated M2-like signature of TAMs. Figure 2 summarises the highlights of the descriptions. Finally, it is important to note that in one study, administration of JZL184 had no effect on tumour burden in bone and spleen of immunocompetent female C57BL/6KalWRij mice that had received an injection of the murine multiple myeloma cell line 5TGM1 via the tail vein [164].
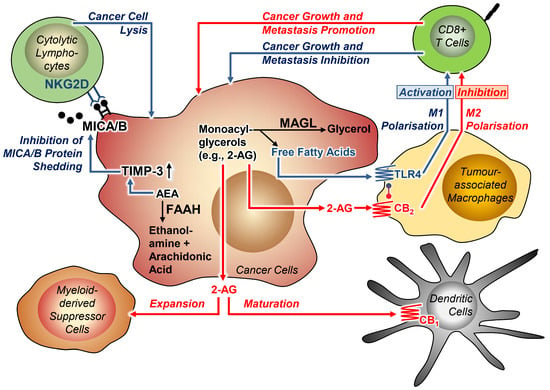
Figure 2.
Effects of MAGL and FAAH on tumour-immune interactions. Blue arrows indicate benign effects, red arrows indicate effects that cause cancer progression. Lines with circles at the ends indicate receptor interaction. All abbreviations are explained in the text.
4. Conclusions and Perspectives
Many of the preclinical results listed here give hope for cannabinoids as a possible additional approach for pharmacotherapeutic interventions in cancer. According to studies in past years, FAAH and MAGL inhibitors also show preclinical tumour-regressive effects. Recent investigations also highlight that MAGL inhibitors could be promising anticancer drugs in terms of their antimetastatic properties [78,92,128]. In addition, a dual antitumour mechanism of MAGL inhibition may prove beneficial, consisting of inhibiting the degradation of antitumour 2-AG while also preventing the MAGL-dependent formation of free fatty acids that promote tumour growth and spread. Regarding the translational aspect, where cannabinoid compounds would certainly be combined with other chemotherapeutic agents, there are a number of preclinical reports indicating that cannabinoids, especially CBD and THC, enhance the chemotherapeutic effects of established cytostatic treatments in cancer (for review see [165]). However, there are currently no data on potential synergies between FAAH or MAGL inhibitors and classical chemotherapies or immunotherapies, particularly related to systemic anticancer effects. Considering that cannabinoids are already used clinically to treat various chemotherapy-induced side effects, such as nausea and vomiting (for review see [166]) as well as pain [167], this could additionally lead to benefits for cancer patients. An analgesic effect has also been reported for FAAH and MAGL inhibitors as well as for dual inhibitors of both enzymes [168]. Furthermore, a recent study has shown that the MAGL inhibitor MJN110 can completely reverse paclitaxel-induced allodynia in a mouse model of chemotherapy-induced neuropathy [169]. Accordingly, combination studies with classical chemotherapeutic agents should also be conducted for these blockers of endocannabinoid degradation, looking at a possible synergistic effect on tumour growth and spread as well as a possible reduction of chemotherapy-typical side effects.
From the studies described above, on the regulation of the endocannabinoid system in cancer, a tendency toward upregulation of CB2 and CB1 receptors in tumour tissue can be inferred. However, these parameters do not represent reliable superior tumour markers. This is mainly due to such observations it always being necessary to distinguish exactly which cells (tumour tissue or immune cells), which degree of development of carcinogenesis and which molecular tumour subtype are present in order to derive a prognostic property from the respective parameters. Similarly, no clear indications of suitability as tumour markers can currently be derived for FAAH and MAGL.
While cannabinoids (THC, nabilone (synthetic THC analogue), nabiximols (standardised extract of Cannabis sativa L. with an approximate 1:1 ratio of THC and CBD), CBD) are already used therapeutically, several FAAH inhibitors and one MAGL inhibitor are still in phase II testing (for review see [170]). In the case of the MAGL inhibitor ABX-1431 investigated for use in Tourette’s syndrome, no major adverse events were described. However, preclinical studies indicate that MAGL inhibitors require a carefully designed dosing schedule to prevent CB1 receptor-dependent side effects. Here, animals treated with the dual FAAH and MAGL inhibitor, JZL195 or the MAGL inhibitor JZL184, showed hyperreflexia in the bar test, known as “popcorning”, and hypomotility in an open field test [168]. However, JZL184, like the FAAH inhibitor PF-3845, did not cause marked catalepsy in animals. The hypomotility typical of THC was also ruled out for FAAH inhibitors in another study [171]. On the other hand, THC-like psychoactive effects, such as catalepsy and THC-like drug discrimination reactions, were induced with dual inhibition of FAAH and MAGL [168]. Furthermore, the disaster of the clinical trial with the FAAH inhibitor BIA 10-2474 [172] clearly showed the importance of consistent preclinical selectivity testing to exclude off-target activities. Other cannabinoid-typical side effects on the cardiovascular system [173] as well as the increased risk of liver fibrosis associated with CB1 receptor activation [174] should also be critically evaluated.
However, the greatest challenge lies in the translational leap from the thus far successful preclinical oncological testing of cannabinoids or inhibitors of endocannabinoid degradation into the clinic. Current studies are limited to a few investigations, such as a pilot study from 2006 that showed that intracranially administered THC was safe in glioblastoma patients [175]. Another exploratory, randomised, placebo-controlled phase 1b clinical trial in patients with recurrent glioblastoma multiforme included 21 patients, 12 of whom were randomised to THC and CBD in addition to dose-intensive temozolomide, with a 1-year survival 83% for nabiximols- and 44% for placebo-treated patients [176].
There are a number of clinical trials registered with the United States National Library of Medicine that address the effects of cannabinoids in cancer patients beyond palliative effects, such as pain relief. These studies include an open-label, single-group phase I/Ib intervention study with 18 participants on the safety of CBD in patients with biochemically recurrent prostate cancer [177], monitoring adverse events leading to dose-limiting toxicity of CBD treatment, as well as prostate specific antigen (PSA) and testosterone level determination as an indication of biochemical response. Another clinical trial is a randomised, open-label, interventional phase III study with 160 participants on the effect of combination therapy in patients with early breast cancer using mifepristone, tamoxifen, retinoic acid and CBD as a selective CYP2D6 inhibitor to enhance the effect of tamoxifen [178]. Here, the expression of cytokeratin 5 is measured to identify a therapy-resistant subpopulation of breast cancer cells that show quantitatively increased cancer stem cell characteristics. Other parameters to be investigated include pathological complete response and concentrations of estradiol, progesterone, retinoic acid and tamoxifen in breast tissue and plasma, as well as tumour markers in serum compared to baseline. Another registered, yet to be recruiting clinical trial aims to determine the maximum tolerated dose and the type and number of adverse events occurring during treatment with a combination of THC and CBD [179]. The secondary outcome to be determined in this study is the antitumour activity of THC/CBD treatment in combination with temozolomide and radiotherapy, as well as overall survival and progression-free survival.
In summary, clinical studies need to clarify which combinations of chemotherapeutic agents with cannabinoids or, in perspective, inhibitors of endocannabinoid degradation are useful for cancer patients. To provide this development with the opportunity to succeed, uncertified products derived from marijuana should not continue to be marketed under the pretext of curing cancer [180]; this would be the second step before the first, which could jeopardise the potential use of cannabinoids in tumour diseases.
Author Contributions
Conceptualisation, R.R. and B.H.; writing—original draft preparation, R.R., F.W. and B.H.; writing—review and editing, R.R., F.W. and B.H. All authors have read and agreed to the published version of the manuscript.
Funding
This review received no special funding.
Acknowledgments
The corresponding author’s preclinical experimental work on the effects of cannabinoids and chemotherapeutic agents is currently funded by the German Research Foundation (DFG, HI 813/9-1), the joint research project ONKOTHER-H (supported by the European Social Fund, reference: ESF/14-BM-A55-0002/18, and the Ministry of Education, Science and Culture of Mecklenburg-Vorpommern, Germany) and by the joint research projects RESPONSE FV18 (reference: 03ZZ0928A) and RESPONSE TV3 (reference: 03ZZ0933A).
Conflicts of Interest
The authors declare no conflict of interest.
References
- Devane:, W.A.; Hanus, L.; Breuer, A.; Pertwee, R.G.; Stevenson, L.A.; Griffin, G.; Gibson, D.; Mandelbaum, A.; Etinger, A.; Mechoulam, R. Isolation and structure of a brain constituent that binds to the cannabinoid receptor. Science 1992, 258, 1946–1949. [Google Scholar] [CrossRef]
- Mechoulam, R.; Ben-Shabat, S.; Hanus, L.; Ligumsky, M.; Kaminski, N.E.; Schatz, A.R.; Gopher, A.; Almog, S.; Martin, B.R.; Compton, D.R.; et al. Identification of an endogenous 2-monoglyceride, present in canine gut, that binds to cannabinoid receptors. Biochem. Pharmacol. 1995, 50, 83–90. [Google Scholar] [CrossRef]
- Hanus, L.; Abu-Lafi, S.; Fride, E.; Breuer, A.; Vogel, Z.; Shalev, D.E.; Kustanovich, I.; Mechoulam, R. 2-arachidonyl glyceryl ether, an endogenous agonist of the cannabinoid CB1 receptor. Proc. Natl. Acad. Sci. USA 2001, 98, 3662–3665. [Google Scholar] [CrossRef] [PubMed]
- Bisogno, T.; Melck, D.; Bobrov, M.Y.; Gretskaya, N.M.; Bezuglov, V.V.; De Petrocellis, L.; Di Marzo, V. N-acyl-dopamines: Novel synthetic CB1 cannabinoid-receptor ligands and inhibitors of anandamide inactivation with cannabimimetic activity in vitro and in vivo. Biochem. J. 2000, 351, 817–824. [Google Scholar] [CrossRef] [PubMed]
- Porter, A.C.; Sauer, J.M.; Knierman, M.D.; Becker, G.W.; Berna, M.J.; Bao, J.; Nomikos, G.G.; Carter, P.; Bymaster, F.P.; Leese, A.B.; et al. Characterization of a novel endocannabinoid, virodhamine, with antagonist activity at the CB1 receptor. J. Pharmacol. Exp. Ther. 2002, 301, 1020–1024. [Google Scholar] [CrossRef] [PubMed]
- Matsuda, L.A.; Lolait, S.J.; Brownstein, M.J.; Young, A.C.; Bonner, T. Structure of a cannabinoid receptor and functional expression of the cloned cDNA. Nature 1990, 346, 561–564. [Google Scholar] [CrossRef]
- Munro, S.; Thomas, K.L.; Abu-Shaar, M. Molecular characterization of a peripheral receptor for cannabinoids. Nature 1993, 365, 61–65. [Google Scholar] [CrossRef]
- Pertwee, R.G. The diverse CB1 and CB2 receptor pharmacology of three plant cannabinoids: Δ9-tetrahydrocannabinol, cannabidiol and Δ9-tetrahydrocannabivarin. Br. J. Pharmacol. 2008, 153, 199–215. [Google Scholar] [CrossRef]
- Zygmunt, P.M.; Petersson, J.; Andersson, D.A.; Chuang, H.; Sørgård, M.; Di Marzo, V.; Julius, D.; Högestätt, E.D. Vanilloid receptors on sensory nerves mediate the vasodilator action of anandamide. Nature 1999, 400, 452–457. [Google Scholar] [CrossRef] [PubMed]
- Bisogno, T.; Hanus, L.; De Petrocellis, L.; Tchilibon, S.; Ponde, D.E.; Brandi, I.; Moriello, A.S.; Davis, J.B.; Mechoulam, R.; Di Marzo, V. Molecular targets for cannabidiol and its synthetic analogues: Effect on vanilloid VR1 receptors and on the cellular uptake and enzymatic hydrolysis of anandamide. Br. J. Pharmacol. 2001, 134, 845–852. [Google Scholar] [CrossRef] [PubMed]
- Sawzdargo, M.; Nguyen, T.; Lee, D.K.; Lynch, K.R.; Cheng, R.; Heng, H.H.; George, S.R.; O’Dowd, B.F. Identification and cloning of three novel human G protein-coupled receptor genes GPR52, ΨGPR53 and GPR55: GPR55 is extensively expressed in human brain. Brain Res. Mol. Brain Res. 1999, 64, 193–198. [Google Scholar] [CrossRef]
- Ryberg, E.; Larsson, N.; Sjögren, S.; Hjorth, S.; Hermansson, N.O.; Leonova, J.; Elebring, T.; Nilsson, K.; Drmota, T.; Greasley, P.J. The orphan receptor GPR55 is a novel cannabinoid receptor. Br. J. Pharmacol. 2007, 152, 1092–1101. [Google Scholar] [CrossRef] [PubMed]
- Artmann, A.; Petersen, G.; Hellgren, L.I.; Boberg, J.; Skonberg, C.; Nellemann, C.; Hansen, S.H.; Hansen, H.S. Influence of dietary fatty acids on endocannabinoid and N-acylethanolamine levels in rat brain, liver and small intestine. Biochim. Biophys. Acta. 2008, 1781, 200–212. [Google Scholar] [CrossRef] [PubMed]
- Schwarz, R.; Ramer, R.; Hinz, B. Targeting the endocannabinoid system as a potential anticancer approach. Drug Metab. Rev. 2018, 50, 26–53. [Google Scholar] [CrossRef] [PubMed]
- Di Marzo, V. The endocannabinoid system: Its general strategy of action, tools for its pharmacological manipulation and potential therapeutic exploitation. Pharmacol. Res. 2009, 60, 77–84. [Google Scholar] [CrossRef]
- Deutsch, D.G.; Chin, S.A. Enzymatic synthesis and degradation of anandamide, a cannabinoid receptor agonist. Biochem. Pharmacol. 1993, 46, 791–796. [Google Scholar] [CrossRef]
- Blankman, J.L.; Simon, G.M.; Cravatt, B.F. A comprehensive profile of brain enzymes that hydrolyze the endocannabinoid 2-arachidonoylglycerol. Chem. Biol. 2007, 14, 1347–1356. [Google Scholar] [CrossRef] [PubMed]
- Nomura, D.K.; Long, J.Z.; Niessen, S.; Hoover, H.S.; Ng, S.W.; Cravatt, B.F. Monoacylglycerol lipase regulates a fatty acid network that promotes cancer pathogenesis. Cell 2010, 140, 49–61. [Google Scholar] [CrossRef] [PubMed]
- Koutek, B.; Prestwich, G.D.; Howlett, A.C.; Chin, S.A.; Salehani, D.; Akhavan, N.; Deutsch, D.G. Inhibitors of arachidonoyl ethanolamide hydrolysis. J. Biol. Chem. 1994, 269, 22937–22940. [Google Scholar] [CrossRef]
- Street, I.P.; Lin, H.K.; Laliberté, F.; Ghomashchi, F.; Wang, Z.; Perrier, H.; Tremblay, N.M.; Huang, Z.; Weech, P.K.; Gelb, M.H. Slow- and tight-binding inhibitors of the 85-kDa human phospholipase A2. Biochemistry 1993, 32, 5935–5940. [Google Scholar] [CrossRef] [PubMed]
- Bisogno, T.; Melck, D.; De Petrocellis, L.; Bobrov, M.Y.; Gretskaya, N.M.; Bezuglov, V.V.; Sitachitta, N.; Gerwick, W.H.; Di Marzo, V. Arachidonoylserotonin and other novel inhibitors of fatty acid amide hydrolase. Biochem. Biophys. Res. Commun. 1998, 248, 515–522. [Google Scholar] [CrossRef] [PubMed]
- Kathuria, S.; Gaetani, S.; Fegley, D.; Valiño, F.; Duranti, A.; Tontini, A.; Mor, M.; Tarzia, G.; La Rana, G.; Calignano, A.; et al. Modulation of anxiety through blockade of anandamide hydrolysis. Nat. Med. 2003, 9, 76–81. [Google Scholar] [CrossRef]
- Hohmann, A.G.; Suplita, R.L.; Bolton, N.M.; Neely, M.H.; Fegley, D.; Mangieri, R.; Krey, J.F.; Walker, J.M.; Holmes, P.V.; Crystal, J.D.; et al. An endocannabinoid mechanism for stress-induced analgesia. Nature 2005, 435, 1108–1112. [Google Scholar] [CrossRef] [PubMed]
- Vandevoorde, S.; Jonsson, K.O.; Labar, G.; Persson, E.; Lambert, D.M.; Fowler, C.J. Lack of selectivity of URB602 for 2-oleoylglycerol compared to anandamide hydrolysis in vitro. Br. J. Pharmacol. 2007, 150, 186–191. [Google Scholar] [CrossRef] [PubMed]
- Long, J.Z.; Nomura, D.K.; Cravatt, B.F. Characterization of monoacylglycerol lipase inhibition reveals differences in central and peripheral endocannabinoid metabolism. Chem. Biol. 2009, 16, 744–753. [Google Scholar] [CrossRef] [PubMed]
- Niphakis, M.J.; Cognetta, A.B., 3rd; Chang, J.W.; Buczynski, M.W.; Parsons, L.H.; Byrne, F.; Burston, J.J.; Chapman, V.; Cravatt, B.F. Evaluation of NHS carbamates as a potent and selective class of endocannabinoid hydrolase inhibitors. ACS Chem. Neurosci. 2013, 4, 1322–1332. [Google Scholar] [CrossRef]
- Chang, J.W.; Cognetta, A.B., 3rd; Niphakis, M.J.; Cravatt, B.F. Proteome-wide reactivity profiling identifies diverse carbamate chemotypes tuned for serine hydrolase inhibition. ACS Chem. Biol. 2013, 8, 1590–1599. [Google Scholar] [CrossRef] [PubMed]
- Cisar, J.S.; Weber, O.D.; Clapper, J.R.; Blankman, J.L.; Henry, C.L.; Simon, G.M.; Alexander, J.P.; Jones, T.K.; Ezekowitz, R.A.B.; O’Neill, G.P.; et al. Identification of ABX-1431, a Selective Inhibitor of Monoacylglycerol Lipase and Clinical Candidate for Treatment of Neurological Disorders. J. Med. Chem. 2018, 61, 9062–9084. [Google Scholar] [CrossRef] [PubMed]
- U.S. National Library of Medicine. ClinicalTrials.gov. Available online: https://www.clinicaltrials.gov/ct2/results?recrs=&cond=&term=ABX-1431&cntry=&state=&city=&dist= (accessed on 20 October 2021).
- Chen, A.L.; Lum, K.M.; Lara-Gonzalez, P.; Ogasawara, D.; Cognetta, A.B., 3rd; To, A.; Parsons, W.H.; Simon, G.M.; Desai, A.; Petrascheck, M.; et al. Pharmacological convergence reveals a lipid pathway that regulates C. elegans lifespan. Nat. Chem. Biol. 2019, 15, 453–462. [Google Scholar] [CrossRef] [PubMed]
- Zhang, J.; Medina-Cleghorn, D.; Bernal-Mizrachi, L.; Bracci, P.M.; Hubbard, A.; Conde, L.; Riby, J.; Nomura, D.K.; Skibola, C.F. The potential relevance of the endocannabinoid, 2-arachidonoylglycerol, in diffuse large B-cell lymphoma. Oncoscience 2016, 3, 31–41. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Guida, M.; Ligresti, A.; De Filippis, D.; D’Amico, A.; Petrosino, S.; Cipriano, M.; Bifulco, G.; Simonetti, S.; Orlando, P.; Insabato, L.; et al. The levels of the endocannabinoid receptor CB2 and its ligand 2-arachidonoylglycerol are elevated in endometrial carcinoma. Endocrinology 2010, 151, 921–928. [Google Scholar] [CrossRef] [PubMed]
- Chen, L.; Chen, H.; Li, Y.; Li, L.; Qiu, Y.; Ren, J. Endocannabinoid and ceramide levels are altered in patients with colorectal cancer. Oncol. Rep. 2015, 34, 447–454. [Google Scholar] [CrossRef] [PubMed]
- Wu, X.; Han, L.; Zhang, X.; Li, L.; Jiang, C.; Qiu, Y.; Huang, R.; Xie, B.; Lin, Z.; Ren, J.; et al. Alteration of endocannabinoid system in human gliomas. J. Neurochem. 2012, 120, 842–849. [Google Scholar] [CrossRef]
- Maccarrone, M.; Attinà, M.; Cartoni, A.; Bari, M.; Finazzi-Agrò, A. Gas chromatography-mass spectrometry analysis of endogenous cannabinoids in healthy and tumoral human brain and human cells in culture. J. Neurochem. 2001, 76, 594–601. [Google Scholar] [CrossRef]
- Alberich Jordà, M.; Rayman, N.; Tas, M.; Verbakel, S.E.; Battista, N.; van Lom, K.; Löwenberg, B.; Maccarrone, M.; Delwel, R. The peripheral cannabinoid receptor Cb2, frequently expressed on AML blasts, either induces a neutrophilic differentiation block or confers abnormal migration properties in a ligand-dependent manner. Blood 2004, 104, 526–534. [Google Scholar] [CrossRef] [PubMed]
- Sánchez, C.; de Ceballos, M.L.; Gomez del Pulgar, T.; Rueda, D.; Corbacho, C.; Velasco, G.; Galve-Roperh, I.; Huffman, J.W.; Ramón y Cajal, S.; Guzmán, M. Inhibition of glioma growth in vivo by selective activation of the CB2 cannabinoid receptor. Cancer Res. 2001, 61, 5784–5789. [Google Scholar]
- Held-Feindt, J.; Dörner, L.; Sahan, G.; Mehdorn, H.M.; Mentlein, R. Cannabinoid receptors in human astroglial tumors. J. Neurochem. 2006, 98, 886–893. [Google Scholar] [CrossRef]
- De Jesús, M.L.; Hostalot, C.; Garibi, J.M.; Sallés, J.; Meana, J.J.; Callado, L.F. Opposite changes in cannabinoid CB1 and CB2 receptor expression in human gliomas. Neurochem. Int. 2010, 56, 829–833. [Google Scholar] [CrossRef]
- Sredni, S.T.; Huang, C.C.; Suzuki, M.; Pundy, T.; Chou, P.; Tomita, T. Spontaneous involution of pediatric low-grade gliomas: High expression of cannabinoid receptor 1 (CNR1) at the time of diagnosis may indicate involvement of the endocannabinoid system. Childs Nerv. Syst. 2016, 32, 2061–2067. [Google Scholar] [CrossRef]
- Ellert-Miklaszewska, A.; Ciechomska, I.A.; Kaminska, B. Synthetic Cannabinoids Induce Autophagy and Mitochondrial Apoptotic Pathways in Human Glioblastoma Cells Independently of Deficiency in TP53 or PTEN Tumor Suppressors. Cancers 2021, 13, 419. [Google Scholar] [CrossRef]
- Caffarel, M.M.; Sarrió, D.; Palacios, J.; Guzmán, M.; Sánchez, C. Δ9-tetrahydrocannabinol inhibits cell cycle progression in human breast cancer cells through Cdc2 regulation. Cancer Res. 2006, 66, 6615–6621. [Google Scholar] [CrossRef] [PubMed]
- Gjerstorff, M.F.; Benoit, V.M.; Laenkholm, A.V.; Nielsen, O.; Johansen, L.E.; Ditzel, H.J. Identification of genes with altered expression in medullary breast cancer vs. ductal breast cancer and normal breast epithelia. Int. J. Oncol. 2006, 28, 1327–1335. [Google Scholar] [CrossRef] [PubMed]
- Nasser, M.W.; Qamri, Z.; Deol, Y.S.; Smith, D.; Shilo, K.; Zou, X.; Ganju, R.K. Crosstalk between chemokine receptor CXCR4 and cannabinoid receptor CB2 in modulating breast cancer growth and invasion. PLoS ONE 2011, 6, e23901. [Google Scholar] [CrossRef]
- Shubbar, E.; Helou, K.; Kovács, A.; Nemes, S.; Hajizadeh, S.; Enerbäck, C.; Einbeigi, Z. High levels of γ-glutamyl hydrolase (GGH) are associated with poor prognosis and unfavorable clinical outcomes in invasive breast cancer. BMC Cancer 2013, 13, 47. [Google Scholar] [CrossRef]
- Sun, H.; Jiang, L.; Luo, X.; Jin, W.; He, Q.; An, J.; Lui, K.; Shi, J.; Rong, R.; Su, W.; et al. Potential tumor-suppressive role of monoglyceride lipase in human colorectal cancer. Oncogene 2013, 32, 234–241. [Google Scholar] [CrossRef] [PubMed]
- Pérez-Gómez, E.; Andradas, C.; Blasco-Benito, S.; Caffarel, M.M.; García-Taboada, E.; Villa-Morales, M.; Moreno, E.; Hamann, S.; Martín-Villar, E.; Flores, J.M.; et al. Role of cannabinoid receptor CB2 in HER2 pro-oncogenic signaling in breast cancer. J. Natl. Cancer Inst. 2015, 107, djv077. [Google Scholar] [CrossRef]
- Blasco-Benito, S.; Moreno, E.; Seijo-Vila, M.; Tundidor, I.; Andradas, C.; Caffarel, M.M.; Caro-Villalobos, M.; Urigüen, L.; Diez-Alarcia, R.; Moreno-Bueno, G.; et al. Therapeutic targeting of HER2-CB2R heteromers in HER2-positive breast cancer. Proc. Natl. Acad. Sci. USA 2019, 116, 3863–3872, Erratum in 2019, 116, 6505. [Google Scholar] [CrossRef]
- Yoneten, K.K.; Kasap, M.; Akpinar, G.; Gunes, A.; Gurel, B.; Utkan, N.Z. Comparative Proteome Analysis of Breast Cancer Tissues Highlights the Importance of Glycerol-3-phosphate Dehydrogenase 1 and Monoacylglycerol Lipase in Breast Cancer Metabolism. Cancer Genom. Proteom. 2019, 16, 377–397. [Google Scholar] [CrossRef]
- Ligresti, A.; Bisogno, T.; Matias, I.; De Petrocellis, L.; Cascio, M.G.; Cosenza, V.; D’argenio, G.; Scaglione, G.; Bifulco, M.; Sorrentini, I.; et al. Possible endocannabinoid control of colorectal cancer growth. Gastroenterology 2003, 125, 677–687. [Google Scholar] [CrossRef]
- Cianchi, F.; Papucci, L.; Schiavone, N.; Lulli, M.; Magnelli, L.; Vinci, M.C.; Messerini, L.; Manera, C.; Ronconi, E.; Romagnani, P.; et al. Cannabinoid receptor activation induces apoptosis through tumor necrosis factor α-mediated ceramide de novo synthesis in colon cancer cells. Clin. Cancer Res. 2008, 14, 7691–7700. [Google Scholar] [CrossRef]
- Wang, D.; Wang, H.; Ning, W.; Backlund, M.G.; Dey, S.K.; DuBois, R.N. Loss of cannabinoid receptor 1 accelerates intestinal tumor growth. Cancer Res. 2008, 68, 6468–6476. [Google Scholar] [CrossRef]
- Ye, L.; Zhang, B.; Seviour, E.G.; Tao, K.X.; Liu, X.H.; Ling, Y.; Chen, J.Y.; Wang, G.B. Monoacylglycerol lipase (MAGL) knockdown inhibits tumor cells growth in colorectal cancer. Cancer Lett. 2011, 307, 6–17. [Google Scholar] [CrossRef]
- Jung, C.K.; Kang, W.K.; Park, J.M.; Ahn, H.J.; Kim, S.W.; Taek Oh, S.; Choi, K.Y. Expression of the cannabinoid type I receptor and prognosis following surgery in colorectal cancer. Oncol. Lett. 2013, 5, 870–876. [Google Scholar] [CrossRef]
- Martínez-Martínez, E.; Gómez, I.; Martín, P.; Sánchez, A.; Román, L.; Tejerina, E.; Bonilla, F.; Merino, A.G.; de Herreros, A.G.; Provencio, M.; et al. Cannabinoids receptor type 2, CB2, expression correlates with human colon cancer progression and predicts patient survival. Oncoscience 2015, 2, 131–141. [Google Scholar] [CrossRef] [PubMed]
- Hasenoehrl, C.; Feuersinger, D.; Sturm, E.M.; Bärnthaler, T.; Heitzer, E.; Graf, R.; Grill, M.; Pichler, M.; Beck, S.; Butcher, L.; et al. G protein-coupled receptor GPR55 promotes colorectal cancer and has opposing effects to cannabinoid receptor 1. Int. J. Cancer 2018, 142, 121–132. [Google Scholar] [CrossRef]
- Xiang, W.; Shi, R.; Kang, X.; Zhang, X.; Chen, P.; Zhang, L.; Hou, A.; Wang, R.; Zhao, Y.; Zhao, K.; et al. Monoacylglycerol lipase regulates cannabinoid receptor 2-dependent macrophage activation and cancer progression. Nat. Commun. 2018, 9, 2574. [Google Scholar] [CrossRef] [PubMed]
- Tutino, V.; Caruso, M.G.; De Nunzio, V.; Lorusso, D.; Veronese, N.; Gigante, I.; Notarnicola, M.; Giannelli, G. Down-Regulation of Cannabinoid Type 1 (CB1) Receptor and its Downstream Signaling Pathways in Metastatic Colorectal Cancer. Cancers 2019, 11, 708. [Google Scholar] [CrossRef]
- Zhang, Y.; Zheng, W.; Shen, K.; Shen, W. ∆9-tetrahydrocannabinol inhibits epithelial-mesenchymal transition and metastasis by targeting matrix metalloproteinase-9 in endometrial cancer. Oncol. Lett. 2018, 15, 8527–8535. [Google Scholar] [CrossRef]
- Ayakannu, T.; Taylor, A.H.; Marczylo, T.H.; Maccarrone, M.; Konje, J.C. Identification of Novel Predictive Biomarkers for Endometrial Malignancies: N-Acylethanolamines. Front. Oncol. 2019, 9, 430. [Google Scholar] [CrossRef]
- Ayakannu, T.; Taylor, A.H.; Bari, M.; Mastrangelo, N.; Maccarrone, M.; Konje, J.C. Expression and Function of the Endocannabinoid Modulating Enzymes Fatty Acid Amide Hydrolase and N-Acylphosphatidylethanolamine-Specific Phospholipase D in Endometrial Carcinoma. Front. Oncol. 2019, 9, 1363. [Google Scholar] [CrossRef]
- Li, X.; Gao, S.; Li, W.; Liu, Z.; Shi, Z.; Qiu, C.; Jiang, J. Effect of monoacylglycerol lipase on the tumor growth in endometrial cancer. J. Obstet. Gynaecol. Res. 2019, 45, 2043–2054. [Google Scholar] [CrossRef]
- Andradas, C.; Byrne, J.; Kuchibhotla, M.; Ancliffe, M.; Jones, A.C.; Carline, B.; Hii, H.; Truong, A.; Storer, L.C.D.; Ritzmann, T.A.; et al. Assessment of Cannabidiol and Δ9-Tetrahydrocannabiol in Mouse Models of Medulloblastoma and Ependymoma. Cancers 2021, 13, 330. [Google Scholar] [CrossRef] [PubMed]
- Hijiya, N.; Shibata, T.; Daa, T.; Hamanaka, R.; Uchida, T.; Matsuura, K.; Tsukamoto, Y.; Nakada, C.; Iha, H.; Inomata, M.; et al. Overexpression of cannabinoid receptor 1 in esophageal squamous cell carcinoma is correlated with metastasis to lymph nodes and distant organs, and poor prognosis. Pathol. Int. 2017, 67, 83–90. [Google Scholar] [CrossRef] [PubMed]
- Xian, X.; Tang, L.; Wu, C.; Huang, L. mir-23b-3p and mir-130a-5p affect cell growth, migration and invasion by targeting CB1R via the Wnt/β-catenin signaling pathway in gastric carcinoma. Onco Targets Ther. 2018, 11, 7503–7512. [Google Scholar] [CrossRef] [PubMed]
- Petersen, G.; Moesgaard, B.; Schmid, P.C.; Schmid, H.H.; Broholm, H.; Kosteljanetz, M.; Hansen, H.S. Endocannabinoid metabolism in human glioblastomas and meningiomas compared to human non-tumour brain tissue. J. Neurochem. 2005, 93, 299–309. [Google Scholar] [CrossRef]
- Schley, M.; Ständer, S.; Kerner, J.; Vajkoczy, P.; Schüpfer, G.; Dusch, M.; Schmelz, M.; Konrad, C. Predominant CB2 receptor expression in endothelial cells of glioblastoma in humans. Brain Res. Bull. 2009, 79, 333–337. [Google Scholar] [CrossRef]
- Klein Nulent, T.J.W.; Van Diest, P.J.; van der Groep, P.; Leusink, F.K.; Kruitwagen, C.L.; Koole, R.; Van Cann, E.M. Cannabinoid receptor-2 immunoreactivity is associated with survival in squamous cell carcinoma of the head and neck. Br. J. Oral Maxillofac. Surg. 2013, 51, 604–609. [Google Scholar] [CrossRef] [PubMed]
- Xu, X.; Liu, Y.; Huang, S.; Liu, G.; Xie, C.; Zhou, J.; Fan, W.; Li, Q.; Wang, Q.; Zhong, D.; et al. Overexpression of cannabinoid receptors CB1 and CB2 correlates with improved prognosis of patients with hepatocellular carcinoma. Cancer Genet. Cytogenet. 2006, 171, 31–38. [Google Scholar] [CrossRef] [PubMed]
- Mukhopadhyay, B.; Schuebel, K.; Mukhopadhyay, P.; Cinar, R.; Godlewski, G.; Xiong, K.; Mackie, K.; Lizak, M.; Yuan, Q.; Goldman, D.; et al. Cannabinoid receptor 1 promotes hepatocellular carcinoma initiation and progression through multiple mechanisms. Hepatology 2015, 61, 1615–1626. [Google Scholar] [CrossRef] [PubMed]
- Zhang, J.; Liu, Z.; Lian, Z.; Liao, R.; Chen, Y.; Qin, Y.; Wang, J.; Jiang, Q.; Wang, X.; Gong, J. Monoacylglycerol Lipase: A Novel Potential Therapeutic Target and Prognostic Indicator for Hepatocellular Carcinoma. Sci. Rep. 2016, 6, 35784. [Google Scholar] [CrossRef] [PubMed]
- Zhu, W.; Zhao, Y.; Zhou, J.; Wang, X.; Pan, Q.; Zhang, N.; Wang, L.; Wang, M.; Zhan, D.; Liu, Z.; et al. Monoacylglycerol lipase promotes progression of hepatocellular carcinoma via NF-κB-mediated epithelial-mesenchymal transition. J. Hematol. Oncol. 2016, 9, 127. [Google Scholar] [CrossRef] [PubMed]
- Yang, J.; Tian, Y.; Zheng, R.; Li, L.; Qiu, F. Endocannabinoid system and the expression of endogenous ceramides in human hepatocellular carcinoma. Oncol. Lett. 2019, 18, 1530–1538. [Google Scholar] [CrossRef] [PubMed]
- Liu, R.; Wang, X.; Curtiss, C.; Landas, S.; Rong, R.; Sheikh, M.S.; Huang, Y. Monoglyceride lipase gene knockout in mice leads to increased incidence of lung adenocarcinoma. Cell Death Dis. 2018, 9, 36. [Google Scholar] [CrossRef] [PubMed]
- Xu, S.; Ma, H.; Bo, Y.; Shao, M. The oncogenic role of CB2 in the progression of non-small-cell lung cancer. Biomed. Pharmacother. 2019, 117, 109080. [Google Scholar] [CrossRef] [PubMed]
- Milian, L.; Mata, M.; Alcacer, J.; Oliver, M.; Sancho-Tello, M.; Martín de Llano, J.J.; Camps, C.; Galbis, J.; Carretero, J.; Carda, C. Cannabinoid receptor expression in non-small cell lung cancer. Effectiveness of tetrahydrocannabinol and cannabidiol inhibiting cell proliferation and epithelial-mesenchymal transition in vitro. PLoS ONE 2020, 15, e0228909. [Google Scholar] [CrossRef] [PubMed]
- Tang, Z.; Xie, H.; Heier, C.; Huang, J.; Zheng, Q.; Eichmann, T.O.; Schoiswohl, G.; Ni, J.; Zechner, R.; Ni, S.; et al. Enhanced monoacylglycerol lipolysis by ABHD6 promotes NSCLC pathogenesis. eBioMedicine 2020, 53, 102696. [Google Scholar] [CrossRef] [PubMed]
- Zhang, H.; Guo, W.; Zhang, F.; Li, R.; Zhou, Y.; Shao, F.; Feng, X.; Tan, F.; Wang, J.; Gao, S.; et al. Monoacylglycerol Lipase Knockdown Inhibits Cell Proliferation and Metastasis in Lung Adenocarcinoma. Front. Oncol. 2020, 10, 559568. [Google Scholar] [CrossRef] [PubMed]
- Islam, T.C.; Asplund, A.C.; Lindvall, J.M.; Nygren, L.; Liden, J.; Kimby, E.; Christensson, B.; Smith, C.I.; Sander, B. High level of cannabinoid receptor 1, absence of regulator of G protein signalling 13 and differential expression of Cyclin D1 in mantle cell lymphoma. Leukemia 2003, 17, 1880–1890. [Google Scholar] [CrossRef]
- Wasik, A.M.; Nygren, L.; Almestrand, S.; Zong, F.; Flygare, J.; Wennerholm, S.B.; Saft, L.; Andersson, P.; Kimby, E.; Wahlin, B.E.; et al. Perturbations of the endocannabinoid system in mantle cell lymphoma: Correlations to clinical and pathological features. Oncoscience 2014, 1, 550–557. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Zhao, Z.; Yang, J.; Zhao, H.; Fang, X.; Li, H. Cannabinoid receptor 2 is upregulated in melanoma. J. Cancer Res. Ther. 2012, 8, 549–554. [Google Scholar] [CrossRef]
- Baba, Y.; Funakoshi, T.; Mori, M.; Emoto, K.; Masugi, Y.; Ekmekcioglu, S.; Amagai, M.; Tanese, K. Expression of monoacylglycerol lipase as a marker of tumour invasion and progression in malignant melanoma. J. Eur. Acad. Dermatol. Venereol. 2017, 31, 2038–2045. [Google Scholar] [CrossRef] [PubMed]
- Theocharis, S.; Giaginis, C.; Alexandrou, P.; Rodriguez, J.; Tasoulas, J.; Danas, E.; Patsouris, E.; Klijanienko, J. Evaluation of cannabinoid CB1 and CB2 receptors expression in mobile tongue squamous cell carcinoma: Associations with clinicopathological parameters and patients’ survival. Tumour Biol. 2016, 37, 3647–3656. [Google Scholar] [CrossRef]
- Hu, W.R.; Lian, Y.F.; Peng, L.X.; Lei, J.J.; Deng, C.C.; Xu, M.; Feng, Q.S.; Chen, L.Z.; Bei, J.X.; Zeng, Y.X. Monoacylglycerol lipase promotes metastases in nasopharyngeal carcinoma. Int. J. Clin. Exp. Pathol. 2014, 7, 3704–3713. [Google Scholar] [PubMed]
- Messalli, E.M.; Grauso, F.; Luise, R.; Angelini, A.; Rossiello, R. Cannabinoid receptor type 1 immunoreactivity and disease severity in human epithelial ovarian tumors. Am. J. Obstet. Gynecol. 2014, 211, 234.e1–234.e6. [Google Scholar] [CrossRef] [PubMed]
- Michalski, C.W.; Oti, F.E.; Erkan, M.; Sauliunaite, D.; Bergmann, F.; Pacher, P.; Batkai, S.; Müller, M.W.; Giese, N.A.; Friess, H.; et al. Cannabinoids in pancreatic cancer: Correlation with survival and pain. Int. J. Cancer 2008, 122, 742–750. [Google Scholar] [CrossRef] [PubMed]
- Pagotto, U.; Marsicano, G.; Fezza, F.; Theodoropoulou, M.; Grübler, Y.; Stalla, J.; Arzberger, T.; Milone, A.; Losa, M.; Di Marzo, V.; et al. Normal human pituitary gland and pituitary adenomas express cannabinoid receptor type 1 and synthesize endogenous cannabinoids: First evidence for a direct role of cannabinoids on hormone modulation at the human pituitary level. J. Clin. Endocrinol. Metab. 2001, 86, 2687–2696. [Google Scholar] [PubMed]
- Endsley, M.P.; Thill, R.; Choudhry, I.; Williams, C.L.; Kajdacsy-Balla, A.; Campbell, W.B.; Nithipatikom, K. Expression and function of fatty acid amide hydrolase in prostate cancer. Int. J. Cancer 2008, 123, 1318–1326. [Google Scholar] [CrossRef] [PubMed]
- Chung, S.C.; Hammarsten, P.; Josefsson, A.; Stattin, P.; Granfors, T.; Egevad, L.; Mancini, G.; Lutz, B.; Bergh, A.; Fowler, C.J. A high cannabinoid CB1 receptor immunoreactivity is associated with disease severity and outcome in prostate cancer. Eur. J. Cancer 2009, 45, 174–182. [Google Scholar] [CrossRef] [PubMed]
- Czifra, G.; Varga, A.; Nyeste, K.; Marincsák, R.; Tóth, B.I.; Kovács, I.; Kovács, L.; Bíró, T. Increased expressions of cannabinoid receptor-1 and transient receptor potential vanilloid-1 in human prostate carcinoma. J. Cancer Res. Clin. Oncol. 2009, 135, 507–514. [Google Scholar] [CrossRef] [PubMed]
- Thors, L.; Bergh, A.; Persson, E.; Hammarsten, P.; Stattin, P.; Egevad, L.; Granfors, T.; Fowler, C.J. Fatty acid amide hydrolase in prostate cancer: Association with disease severity and outcome, CB1 receptor expression and regulation by IL-4. PLoS ONE 2010, 5, e12275. [Google Scholar] [CrossRef] [PubMed]
- Marino, S.; de Ridder, D.; Bishop, R.T.; Renema, N.; Ponzetti, M.; Sophocleous, A.; Capulli, M.; Aljeffery, A.; Carrasco, G.; Gens, M.D.; et al. Paradoxical effects of JZL184, an inhibitor of monoacylglycerol lipase, on bone remodelling in healthy and cancer-bearing mice. eBioMedicine 2019, 44, 452–466. [Google Scholar] [CrossRef]
- Larrinaga, G.; Sanz, B.; Pérez, I.; Blanco, L.; Cándenas, M.L.; Pinto, F.M.; Gil, J.; López, J.I. Cannabinoid CB₁ receptor is downregulated in clear cell renal cell carcinoma. J. Histochem. Cytochem. 2010, 58, 1129–1134. [Google Scholar] [CrossRef]
- Larrinaga, G.; Sanz, B.; Blanco, L.; Perez, I.; Candenas, M.L.; Pinto, F.M.; Irazusta, A.; Gil, J.; López, J.I. Cannabinoid CB1 receptor is expressed in chromophobe renal cell carcinoma and renal oncocytoma. Clin. Biochem. 2013, 46, 638–641. [Google Scholar] [CrossRef]
- Wang, J.; Xu, Y.; Zhu, L.; Zou, Y.; Kong, W.; Dong, B.; Huang, J.; Chen, Y.; Xue, W.; Huang, Y.; et al. Cannabinoid receptor 2 as a novel target for promotion of renal cell carcinoma prognosis and progression. J. Cancer Res. Clin. Oncol. 2018, 144, 39–52. [Google Scholar] [CrossRef]
- Mergler, S.; Cheng, Y.; Skosyrski, S.; Garreis, F.; Pietrzak, P.; Kociok, N.; Dwarakanath, A.; Reinach, P.S.; Kakkassery, V. Altered calcium regulation by thermosensitive transient receptor potential channels in etoposide-resistant WERI-Rb1 retinoblastoma cells. Exp. Eye Res. 2012, 94, 157–173. [Google Scholar] [CrossRef] [PubMed]
- Oesch, S.; Walter, D.; Wachtel, M.; Pretre, K.; Salazar, M.; Guzmán, M.; Velasco, G.; Schäfer, B.W. Cannabinoid receptor 1 is a potential drug target for treatment of translocation-positive rhabdomyosarcoma. Mol. Cancer Ther. 2009, 8, 1838–1845. [Google Scholar] [CrossRef] [PubMed]
- Lakiotaki, E.; Giaginis, C.; Tolia, M.; Alexandrou, P.; Delladetsima, I.; Giannopoulou, I.; Kyrgias, G.; Patsouris, E.; Theocharis, S. Clinical Significance of Cannabinoid Receptors CB1 and CB2 Expression in Human Malignant and Benign Thyroid Lesions. Biomed. Res. Int. 2015, 2015, 839403. [Google Scholar] [CrossRef]
- Sailler, S.; Schmitz, K.; Jäger, E.; Ferreiros, N.; Wicker, S.; Zschiebsch, K.; Pickert, G.; Geisslinger, G.; Walter, C.; Tegeder, I.; et al. Regulation of circulating endocannabinoids associated with cancer and metastases in mice and humans. Oncoscience 2014, 1, 272–282. [Google Scholar] [CrossRef]
- Munson, A.E.; Harris, L.S.; Friedman, M.A.; Dewey, W.L.; Carchman, R.A. Antineoplastic activity of cannabinoids. J. Natl. Cancer Inst. 1975, 55, 597–602. [Google Scholar] [CrossRef] [PubMed]
- De Petrocellis, L.; Melck, D.; Palmisano, A.; Bisogno, T.; Laezza, C.; Bifulco, M.; Di Marzo, V. The endogenous cannabinoid anandamide inhibits human breast cancer cell proliferation. Proc. Natl. Acad. Sci. USA 1998, 95, 8375–8380. [Google Scholar] [CrossRef]
- Ramer, R.; Hinz, B. Antitumorigenic targets of cannabinoids—Current status and implications. Expert Opin. Ther. Targets 2016, 20, 1219–1235. [Google Scholar] [CrossRef]
- Galve-Roperh, I.; Sánchez, C.; Cortés, M.L.; Gómez del Pulgar, T.; Izquierdo, M.; Guzmán, M. Anti-tumoral action of cannabinoids: Involvement of sustained ceramide accumulation and extracellular signal-regulated kinase activation. Nat. Med. 2000, 6, 313–319. [Google Scholar] [CrossRef]
- Ramer, R.; Weinzierl, U.; Schwind, B.; Brune, K.; Hinz, B. Ceramide is involved in R(+)-methanandamide-induced cyclooxygenase-2 expression in human neuroglioma cells. Mol. Pharmacol. 2003, 64, 1189–1198. [Google Scholar] [CrossRef]
- Hinz, B.; Ramer, R.; Eichele, K.; Weinzierl, U.; Brune, K. Up-regulation of cyclooxygenase-2 expression is involved in R(+)-methanandamide-induced apoptotic death of human neuroglioma cells. Mol. Pharmacol. 2004, 66, 1643–1651. [Google Scholar] [CrossRef] [PubMed]
- Blázquez, C.; Carracedo, A.; Barrado, L.; Real, P.J.; Fernández-Luna, J.L.; Velasco, G.; Malumbres, M.; Guzmán, M. Cannabinoid receptors as novel targets for the treatment of melanoma. FASEB J. 2006, 20, 2633–2635. [Google Scholar] [CrossRef] [PubMed]
- Caffarel, M.M.; Andradas, C.; Mira, E.; Pérez-Gómez, E.; Cerutti, C.; Moreno-Bueno, G.; Flores, J.M.; García-Real, I.; Palacios, J.; Mañes, S.; et al. Cannabinoids reduce ErbB2-driven breast cancer progression through Akt inhibition. Mol. Cancer 2010, 9, 196. [Google Scholar] [CrossRef] [PubMed]
- Laezza, C.; Pisanti, S.; Crescenzi, E.; Bifulco, M. Anandamide inhibits Cdk2 and activates Chk1 leading to cell cycle arrest in human breast cancer cells. FEBS Lett. 2006, 580, 6076–6082. [Google Scholar] [CrossRef]
- Salazar, M.; Carracedo, A.; Salanueva, I.J.; Hernández-Tiedra, S.; Lorente, M.; Egia, A.; Vázquez, P.; Blázquez, C.; Torres, S.; García, S.; et al. Cannabinoid action induces autophagy-mediated cell death through stimulation of ER stress in human glioma cells. J. Clin. Investig. 2009, 119, 1359–1372. [Google Scholar] [CrossRef] [PubMed]
- Vara, D.; Salazar, M.; Olea-Herrero, N.; Guzmán, M.; Velasco, G.; Díaz-Laviada, I. Anti-tumoural action of cannabinoids on hepatocellular carcinoma, role of AMPK-dependent activation of autophagy. Cell Death Differ. 2011, 18, 1099–1111. [Google Scholar] [CrossRef]
- Vara, D.; Morell, C.; Rodríguez-Henche, N.; Diaz-Laviada, I. Involvement of PPARγ in the antitumoural action of cannabinoids on hepatocellular carcinoma. Cell Death Dis. 2013, 4, e618. [Google Scholar] [CrossRef]
- Armstrong, J.L.; Hill, D.S.; McKee, C.S.; Hernandez-Tiedra, S.; Lorente, M.; Lopez-Valero, I.; Eleni Anagnostou, M.; Babatunde, F.; Corazzari, M.; Redfern, C.P.F.; et al. Exploiting cannabinoid-induced cytotoxic autophagy to drive melanoma cell death. J. Investig. Dermatol. 2015, 135, 1629–1637. [Google Scholar] [CrossRef]
- Ivanov, V.N.; Grabham, P.W.; Wu, C.C.; Hei, T.K. Inhibition of autophagic flux differently modulates cannabidiol-induced death in 2D and 3D glioblastoma cell cultures. Sci. Rep. 2020, 10, 2687. [Google Scholar] [CrossRef]
- Nabissi, M.; Morelli, M.B.; Amantini, C.; Liberati, S.; Santoni, M.; Ricci-Vitiani, L.; Pallini, R.; Santoni, G. Cannabidiol stimulates Aml-1a-dependent glial differentiation and inhibits glioma stem-like cells proliferation by inducing autophagy in a TRPV2-dependent manner. Int. J. Cancer 2015, 137, 1855–1869. [Google Scholar] [CrossRef]
- Shrivastava, A.; Kuzontkoski, P.M.; Groopman, J.E.; Prasad, A. Cannabidiol induces programmed cell death in breast cancer cells by coordinating the cross-talk between apoptosis and autophagy. Mol. Cancer Ther. 2011, 10, 1161–1172. [Google Scholar] [CrossRef]
- Olivas-Aguirre, M.; Torres-López, L.; Valle-Reyes, J.S.; Hernández-Cruz, A.; Pottosin, I.; Dobrovinskaya, O. Cannabidiol directly targets mitochondria and disturbs calcium homeostasis in acute lymphoblastic leukemia. Cell Death Dis. 2019, 10, 779. [Google Scholar] [CrossRef]
- Go, Y.Y.; Kim, S.R.; Kim, D.Y.; Chae, S.W.; Song, J.J. Cannabidiol enhances cytotoxicity of anti-cancer drugs in human head and neck squamous cell carcinoma. Sci. Rep. 2020, 10, 20622. [Google Scholar] [CrossRef]
- Bifulco, M.; Laezza, C.; Valenti, M.; Ligresti, A.; Portella, G.; Di Marzo, V. A new strategy to block tumor growth by inhibiting endocannabinoid inactivation. FASEB J. 2004, 18, 1606–1608. [Google Scholar] [CrossRef]
- Izzo, A.A.; Aviello, G.; Petrosino, S.; Orlando, P.; Marsicano, G.; Lutz, B.; Borrelli, F.; Capasso, R.; Nigam, S.; Capasso, F.; et al. Increased endocannabinoid levels reduce the development of precancerous lesions in the mouse colon. J. Mol. Med. 2008, 86, 89–98. [Google Scholar] [CrossRef] [PubMed]
- Adinolfi, B.; Romanini, A.; Vanni, A.; Martinotti, E.; Chicca, A.; Fogli, S.; Nieri, P. Anticancer activity of anandamide in human cutaneous melanoma cells. Eur. J. Pharmacol. 2013, 718, 154–159. [Google Scholar] [CrossRef] [PubMed]
- Hamtiaux, L.; Masquelier, J.; Muccioli, G.G.; Bouzin, C.; Feron, O.; Gallez, B.; Lambert, D.M. The association of N-palmitoylethanolamine with the FAAH inhibitor URB597 impairs melanoma growth through a supra-additive action. BMC Cancer 2012, 12, 92. [Google Scholar] [CrossRef] [PubMed]
- Ravi, J.; Sneh, A.; Shilo, K.; Nasser, M.W.; Ganju, R.K. FAAH inhibition enhances anandamide mediated anti-tumorigenic effects in non-small cell lung cancer by downregulating the EGF/EGFR pathway. Oncotarget 2014, 5, 2475–2486. [Google Scholar] [CrossRef]
- Winkler, K.; Ramer, R.; Dithmer, S.; Ivanov, I.; Merkord, J.; Hinz, B. Fatty acid amide hydrolase inhibitors confer anti-invasive and antimetastatic effects on lung cancer cells. Oncotarget 2016, 7, 15047–15064. [Google Scholar] [CrossRef]
- Sticht, M.A.; Long, J.Z.; Rock, E.M.; Limebeer, C.L.; Mechoulam, R.; Cravatt, B.F.; Parker, L.A. Inhibition of monoacylglycerol lipase attenuates vomiting in Suncus murinus and 2-arachidonoyl glycerol attenuates nausea in rats. Br. J. Pharmacol. 2012, 165, 2425–2435. [Google Scholar] [CrossRef]
- Nomura, D.K.; Lombardi, D.P.; Chang, J.W.; Niessen, S.; Ward, A.M.; Long, J.Z.; Hoover, H.H.; Cravatt, B.F. Monoacylglycerol lipase exerts dual control over endocannabinoid and fatty acid pathways to support prostate cancer. Chem. Biol. 2011, 18, 846–856. [Google Scholar] [CrossRef] [PubMed]
- Taïb, B.; Aboussalah, A.M.; Moniruzzaman, M.; Chen, S.; Haughey, N.J.; Kim, S.F.; Ahima, R.S. Lipid accumulation and oxidation in glioblastoma multiforme. Sci. Rep. 2019, 9, 19593. [Google Scholar] [CrossRef] [PubMed]
- Ma, M.; Bai, J.; Ling, Y.; Chang, W.; Xie, G.; Li, R.; Wang, G.; Tao, K. Monoacylglycerol lipase inhibitor JZL184 regulates apoptosis and migration of colorectal cancer cells. Mol. Med. Rep. 2016, 13, 2850–2856. [Google Scholar] [CrossRef]
- Prüser, J.L.; Ramer, R.; Wittig, F.; Ivanov, I.; Merkord, J.; Hinz, B. The Monoacylglycerol Lipase Inhibitor JZL184 Inhibits Lung Cancer Cell Invasion and Metastasis via the CB1 Cannabinoid Receptor. Mol. Cancer Ther. 2021, 20, 787–802. [Google Scholar] [CrossRef]
- Cipriano, M.; Gouveia-Figueira, S.; Persson, E.; Nording, M.; Fowler, C.J. The influence of monoacylglycerol lipase inhibition upon the expression of epidermal growth factor receptor in human PC-3 prostate cancer cells. BMC Res. Notes 2014, 7, 441. [Google Scholar] [CrossRef] [PubMed]
- Matuszak, N.; Hamtiaux, L.; Baldeyroux, B.; Muccioli, G.G.; Poupaert, J.H.; Lansiaux, A.; Lambert, D.M. Dual inhibition of MAGL and type II topoisomerase by N-phenylmaleimides as a potential strategy to reduce neuroblastoma cell growth. Eur. J. Pharm. Sci. 2012, 45, 263–271. [Google Scholar] [CrossRef] [PubMed]
- King, A.R.; Dotsey, E.Y.; Lodola, A.; Jung, K.M.; Ghomian, A.; Qiu, Y.; Fu, J.; Mor, M.; Piomelli, D. Discovery of potent and reversible monoacylglycerol lipase inhibitors. Chem. Biol. 2009, 16, 1045–1052. [Google Scholar] [CrossRef]
- Lei, X.; Zhong, Y.; Huang, L.; Li, S.; Fu, J.; Zhang, L.; Zhang, Y.; Deng, Q.; Yu, X. Identification of a novel tumor angiogenesis inhibitor targeting Shh/Gli1 signaling pathway in Non-small cell lung cancer. Cell Death Dis. 2020, 11, 232. [Google Scholar] [CrossRef]
- Zhao, Q.; Liu, Y.; Zhong, J.; Bi, Y.; Liu, Y.; Ren, Z.; Li, X.; Jia, J.; Yu, M.; Yu, X. Pristimerin induces apoptosis and autophagy via activation of ROS/ASK1/JNK pathway in human breast cancer in vitro and in vivo. Cell Death Dis. 2019, 5, 125. [Google Scholar] [CrossRef] [PubMed]
- Nithipatikom, K.; Endsley, M.P.; Isbell, M.A.; Falck, J.R.; Iwamoto, Y.; Hillard, C.J.; Campbell, W.B. 2-arachidonoylglycerol: A novel inhibitor of androgen-independent prostate cancer cell invasion. Cancer Res. 2004, 64, 8826–8830. [Google Scholar] [CrossRef] [PubMed]
- Ma, C.; Wu, T.T.; Jiang, P.C.; Li, Z.Q.; Chen, X.J.; Fu, K.; Wang, W.; Gong, R. Anti-carcinogenic activity of anandamide on human glioma in vitro and in vivo. Mol. Med. Rep. 2016, 13, 1558–1562. [Google Scholar] [CrossRef] [PubMed]
- Grimaldi, C.; Pisanti, S.; Laezza, C.; Malfitano, A.M.; Santoro, A.; Vitale, M.; Caruso, M.G.; Notarnicola, M.; Iacuzzo, I.; Portella, G.; et al. Anandamide inhibits adhesion and migration of breast cancer cells. Exp. Cell Res. 2006, 312, 363–373. [Google Scholar] [CrossRef]
- Ramer, R.; Hinz, B. Inhibition of cancer cell invasion by cannabinoids via increased expression of tissue inhibitor of matrix metalloproteinases-1. J. Natl. Cancer Inst. 2008, 100, 59–69. [Google Scholar] [CrossRef] [PubMed]
- Ramer, R.; Merkord, J.; Rohde, H.; Hinz, B. Cannabidiol inhibits cancer cell invasion via upregulation of tissue inhibitor of matrix metalloproteinases-1. Biochem. Pharmacol. 2010, 79, 955–966. [Google Scholar] [CrossRef]
- Ramer, R.; Bublitz, K.; Freimuth, N.; Merkord, J.; Rohde, H.; Haustein, M.; Borchert, P.; Schmuhl, E.; Linnebacher, M.; Hinz, B. Cannabidiol inhibits lung cancer cell invasion and metastasis via intercellular adhesion molecule-1. FASEB J. 2012, 26, 1535–1548. [Google Scholar] [CrossRef] [PubMed]
- Blázquez, C.; Salazar, M.; Carracedo, A.; Lorente, M.; Egia, A.; González-Feria, L.; Haro, A.; Velasco, G.; Guzmán, M. Cannabinoids inhibit glioma cell invasion by down-regulating matrix metalloproteinase-2 expression. Cancer Res. 2008, 68, 1945–1952. [Google Scholar] [CrossRef]
- Pourkhalili, N.; Ghahremani, M.H.; Farsandaj, N.; Tavajohi, S.; Majdzadeh, M.; Parsa, M.; Lavasani, N.J.; Ostad, S.N. Evaluation of anti-invasion effect of cannabinoids on human hepatocarcinoma cells. Toxicol. Mech. Methods 2013, 23, 120–126. [Google Scholar] [CrossRef] [PubMed]
- Ramer, R.; Rohde, A.; Merkord, J.; Rohde, H.; Hinz, B. Decrease of plasminogen activator inhibitor-1 may contribute to the anti-invasive action of cannabidiol on human lung cancer cells. Pharm. Res. 2010, 27, 2162–2174. [Google Scholar] [CrossRef] [PubMed]
- Pietrovito, L.; Iozzo, M.; Bacci, M.; Giannoni, E.; Chiarugi, P. Treatment with Cannabinoids as a Promising Approach for Impairing Fibroblast Activation and Prostate Cancer Progression. Int. J. Mol. Sci. 2020, 21, 787. [Google Scholar] [CrossRef]
- McAllister, S.D.; Christian, R.T.; Horowitz, M.P.; Garcia, A.; Desprez, P.Y. Cannabidiol as a novel inhibitor of Id-1 gene expression in aggressive breast cancer cells. Mol. Cancer Ther. 2007, 6, 2921–2927. [Google Scholar] [CrossRef] [PubMed]
- Soroceanu, L.; Murase, R.; Limbad, C.; Singer, E.; Allison, J.; Adrados, I.; Kawamura, R.; Pakdel, A.; Fukuyo, Y.; Nguyen, D.; et al. Id-1 is a key transcriptional regulator of glioblastoma aggressiveness and a novel therapeutic target. Cancer Res. 2013, 73, 1559–1569. [Google Scholar] [CrossRef] [PubMed]
- McAllister, S.D.; Murase, R.; Christian, R.T.; Lau, D.; Zielinski, A.J.; Allison, J.; Almanza, C.; Pakdel, A.; Lee, J.; Limbad, C.; et al. Pathways mediating the effects of cannabidiol on the reduction of breast cancer cell proliferation invasion and metastasis. Breast Cancer Res. Treat. 2011, 129, 37–47, Erratum in 2012, 133, 401–404. [Google Scholar] [CrossRef] [PubMed]
- Murase, R.; Kawamura, R.; Singer, E.; Pakdel, A.; Sarma, P.; Judkins, J.; Elwakeel, E.; Dayal, S.; Martinez-Martinez, E.; Amere, M.; et al. Targeting multiple cannabinoid anti-tumour pathways with a resorcinol derivative leads to inhibition of advanced stages of breast cancer. Br. J. Pharmacol. 2014, 171, 4464–4477. [Google Scholar] [CrossRef]
- Casanova, M.L.; Blázquez, C.; -Palacio, J.; Villanueva, C.; Fernández-Aceñero, M.J.; Huffman, J.W. Inhibition of skin tumour growth and angiogenesis in vivo by activation of cannabinoid receptors. J. Clin. Investig. 2003, 111, 43–50. [Google Scholar] [CrossRef]
- Blázquez, C.; Casanova, M.L.; Planas, A.; Gómez Del Pulgar, T.; Villanuev, C.; Fernández-Aceñero, M.J.; Aragonés, J.; Huffman, J.W.; Jorcano, J.L.; Guzmán, M. Inhibition of tumor angiogenesis by cannabinoids. FASEB J. 2003, 17, 529–531. [Google Scholar] [CrossRef]
- Blázquez, C.; González-Feria, L.; Alvarez, L.; Haro, A.; Casanova, M.L.; Guzmán, M. Cannabinoids inhibit the vascular endothelial growth factor pathway in gliomas. Cancer Res. 2004, 64, 5617–5623. [Google Scholar] [CrossRef] [PubMed]
- Ramer, R.; Fischer, S.; Haustein, M.; Manda, K.; Hinz, B. Cannabinoids inhibit angiogenic capacities of endothelial cells via release of tissue inhibitor of matrix metalloproteinases-1 from lung cancer cells. Biochem. Pharmacol. 2014, 91, 202–216. [Google Scholar] [CrossRef] [PubMed]
- Braile, M.; Cristinziano, L.; Marcella, S.; Varricchi, G.; Marone, G.; Modestino, L.; Ferrara, A.L.; De Ciuceis, A.; Scala, S.; Galdiero, M.R.; et al. LPS-mediated neutrophil VEGF-A release is modulated by cannabinoid receptor activation. J. Leukoc. Biol. 2021, 109, 621–631. [Google Scholar] [CrossRef] [PubMed]
- Picardi, P.; Ciaglia, E.; Proto, M.; Pisanti, S. Anandamide inhibits breast tumor-induced angiogenesis. Transl. Med. UniSa. 2014, 10, 8–12. [Google Scholar] [PubMed]
- Pagano, E.; Borrelli, F.; Orlando, P.; Romano, B.; Monti, M.; Morbidelli, L.; Aviello, G.; Imperatore, R.; Capasso, R.; Piscitelli, F.; et al. Pharmacological inhibition of MAGL attenuates experimental colon carcinogenesis. Pharmacol. Res. 2017, 119, 227–236. [Google Scholar] [CrossRef]
- Hu, Y.; Ranganathan, M.; Shu, C.; Liang, X.; Ganesh, S.; Osafo-Addo, A.; Yan, C.; Zhang, X.; Aouizerat, B.E.; Krystal, J.H.; et al. Single-cell Transcriptome Mapping Identifies Common and Cell-type Specific Genes Affected by Acute Delta9-tetrahydrocannabinol in Humans. Sci. Rep. 2020, 10, 3450. [Google Scholar] [CrossRef] [PubMed]
- Yang, Y.; Huynh, N.; Dumesny, C.; Wang, K.; He, H.; Nikfarjam, M. Cannabinoids Inhibited Pancreatic Cancer via P-21 Activated Kinase 1 Mediated Pathway. Int. J. Mol. Sci. 2020, 21, 8035. [Google Scholar] [CrossRef] [PubMed]
- Glodde, N.; Jakobs, M.; Bald, T.; Tüting, T.; Gaffal, E. Differential role of cannabinoids in the pathogenesis of skin cancer. Life Sci. 2015, 138, 35–40. [Google Scholar] [CrossRef]
- Haustein, M.; Ramer, R.; Linnebacher, M.; Manda, K.; Hinz, B. Cannabinoids increase lung cancer cell lysis by lymphokine-activated killer cells via upregulation of ICAM-1. Biochem. Pharmacol. 2014, 92, 312–325. [Google Scholar] [CrossRef] [PubMed]
- Zhu, L.X.; Sharma, S.; Stolina, M.; Gardner, B.; Roth, M.D.; Tashkin, D.P.; Dubinett, S.M. Δ-9-tetrahydrocannabinol inhibits antitumor immunity by a CB2 receptor-mediated, cytokine-dependent pathway. J. Immunol. 2000, 165, 373–380. [Google Scholar] [CrossRef]
- McKallip, R.J.; Nagarkatti, M.; Nagarkatti, P.S. Δ-9-tetrahydrocannabinol enhances breast cancer growth and metastasis by suppression of the antitumor immune response. J. Immunol. 2005, 174, 3281–3289. [Google Scholar] [CrossRef] [PubMed]
- Lei, X.; Chen, X.; Quan, Y.; Tao, Y.; Li, J. Targeting CYP2J2 to Enhance the Anti-Glioma Efficacy of Cannabinoid Receptor 2 Stimulation by Inhibiting the Pro-Angiogenesis Function of M2 Microglia. Front. Oncol. 2020, 10, 574277. [Google Scholar] [CrossRef]
- Sekiba, K.; Otsuka, M.; Seimiya, T.; Tanaka, E.; Funato, K.; Miyakawa, Y.; Koike, K. The fatty-acid amide hydrolase inhibitor URB597 inhibits MICA/B shedding. Sci. Rep. 2020, 10, 15556. [Google Scholar] [CrossRef]
- Yin, J.; Kim, S.S.; Choi, E.; Oh, Y.T.; Lin, W.; Kim, T.H.; Sa, J.K.; Hong, J.H.; Park, S.H.; Kwon, H.J.; et al. ARS2/MAGL signaling in glioblastoma stem cells promotes self-renewal and M2-like polarization of tumor-associated macrophages. Nat. Commun. 2020, 11, 2978. [Google Scholar] [CrossRef]
- Marino, S.; Carrasco, G.; Li, B.; Shah, K.M.; Lath, D.L.; Sophocleous, A.; Lawson, M.A.; Idris, A.I. JZL184, A Monoacylglycerol Lipase Inhibitor, Induces Bone Loss in a Multiple Myeloma Model of Immunocompetent Mice. Calcif. Tissue Int. 2020, 107, 72–85. [Google Scholar] [CrossRef]
- Hinz, B.; Ramer, R. Anti-tumour actions of cannabinoids. Br. J. Pharmacol. 2019, 176, 1384–1394. [Google Scholar] [CrossRef] [PubMed]
- Tramèr, M.R.; Carroll, D.; Campbell, F.A.; Reynolds, D.J.; Moore, R.A.; McQuay, H.J. Cannabinoids for control of chemotherapy induced nausea and vomiting: Quantitative systematic review. BMJ 2001, 323, 16–21. [Google Scholar] [CrossRef]
- Khasabova, I.A.; Khasabov, S.; Paz, J.; Harding-Rose, C.; Simone, D.A.; Seybold, V.S. Cannabinoid type-1 receptor reduces pain and neurotoxicity produced by chemotherapy. J. Neurosci. 2012, 32, 7091–7101. [Google Scholar] [CrossRef]
- Long, J.Z.; Nomura, D.K.; Vann, R.E.; Walentiny, D.M.; Booker, L.; Jin, X.; Burston, J.J.; Sim-Selley, L.J.; Lichtman, A.H.; Wiley, J.L.; et al. Dual blockade of FAAH and MAGL identifies behavioral processes regulated by endocannabinoid crosstalk in vivo. Proc. Natl. Acad. Sci. USA 2009, 106, 20270–20275. [Google Scholar] [CrossRef]
- Curry, Z.A.; Wilkerson, J.L.; Bagdas, D.; Kyte, S.L.; Patel, N.; Donvito, G.; Mustafa, M.A.; Poklis, J.L.; Niphakis, M.J.; Hsu, K.L.; et al. Monoacylglycerol Lipase Inhibitors Reverse Paclitaxel-Induced Nociceptive Behavior and Proinflammatory Markers in a Mouse Model of Chemotherapy-Induced Neuropathy. J. Pharmacol. Exp. Ther. 2018, 366, 169–183. [Google Scholar] [CrossRef] [PubMed]
- van Egmond, N.; Straub, V.M.; van der Stelt, M. Targeting Endocannabinoid Signaling: FAAH and MAG Lipase Inhibitors. Annu. Rev. Pharmacol. Toxicol. 2021, 61, 441–463. [Google Scholar] [CrossRef] [PubMed]
- Schlosburg, J.E.; Boger, D.L.; Cravatt, B.F.; Lichtman, A.H. Endocannabinoid modulation of scratching response in an acute allergenic model: A new prospective neural therapeutic target for pruritus. J. Pharmacol. Exp. Ther. 2009, 329, 314–323. [Google Scholar] [CrossRef] [PubMed]
- van Esbroeck, A.C.M.; Janssen, A.P.A.; Cognetta, A.B., 3rd; Ogasawara, D.; Shpak, G.; van der Kroeg, M.; Kantae, V.; Baggelaar, M.P.; de Vrij, F.M.S.; Deng, H.; et al. Activity-based protein profiling reveals off-target proteins of the FAAH inhibitor BIA 10-2474. Science 2017, 356, 1084–1087. [Google Scholar] [CrossRef] [PubMed]
- Page, R.L., 2nd; Allen, L.A.; Kloner, R.A.; Carriker, C.R.; Martel, C.; Morris, A.A.; Piano, M.R.; Rana, J.S.; Saucedo, J.F.; American Heart Association Clinical Pharmacology Committee and Heart Failure and Transplantation Committee of the Council on Clinical Cardiology; et al. Medical Marijuana, Recreational Cannabis, and Cardiovascular Health: A Scientific Statement From the American Heart Association. Circulation 2020, 142, e131–e152. [Google Scholar] [CrossRef] [PubMed]
- Teixeira-Clerc, F.; Julien, B.; Grenard, P.; Tran Van Nhieu, J.; Deveaux, V.; Li, L.; Serriere-Lanneau, V.; Ledent, C.; Mallat, A.; Lotersztajn, S. CB1 cannabinoid receptor antagonism: A new strategy for the treatment of liver fibrosis. Nat. Med. 2006, 12, 671–676. [Google Scholar] [CrossRef] [PubMed]
- Guzmán, M.; Duarte, M.J.; Blazquez, C.; Ravina, J.; Rosa, M.C.; Galve-Roperh, I.; Sánchez, C.; Velasco, G.; González-Feria, L. A pilot study of Δ9-tetrahydrocannabinol in patients with recurrent glioblastoma multiforme. Br. J. Cancer 2006, 95, 197–203. [Google Scholar] [CrossRef]
- Twelves, C.; Sabel, M.; Checketts, D.; Miller, S.; Tayo, B.; Jove, M.; Brazil, L.; Short, S.C. GWCA1208 study group. A phase 1b randomised, placebo-controlled trial of nabiximols cannabinoid oromucosal spray with temozolomide in patients with recurrent glioblastoma. Br. J. Cancer 2021, 124, 1379–1387. [Google Scholar] [CrossRef] [PubMed]
- U.S. National Library of Medicine. ClinicalTrials.gov: A Phase I/Ib Study on the Safety of Epidiolex in Patients with Prostate Cancer with Rising PSA after Localized Therapy with Either Surgery or Radiation. Identifier: NCT04428203. Available online: https://clinicaltrials.gov/ct2/show/NCT04428203?term=CBD&cond=cancer&draw=2&rank=2 (accessed on 20 October 2021).
- U.S. National Library of Medicine. ClinicalTrials.gov: Why Antiprogestrone (Mifepristone) and Cyp 26 Inhibitor Must Be Combined with Tamoxifen or (Tamoxifen and Retinoic Acid) for Treating Early Breast Cancer. Identifier: NCT05016349. Available online: https://clinicaltrials.gov/ct2/show/NCT05016349?term=CBD&cond=cancer&draw=1&rank=5 (accessed on 20 October 2021).
- U.S. National Library of Medicine. ClinicalTrials.gov: Phase Ib, Open-Label, Multicenter, Intrapatient Dose-Escalation Clinical Trial to Assess the Safety Profile of the TN-TC11G (THC+CBD) Combination with Temozolomide and Radiotherapy in Patients with Newly-Diagnosed Glioblastoma. Identifier: NCT03529448. Available online: https://clinicaltrials.gov/ct2/show/NCT03529448?term=CBD&cond=cancer&draw=3&rank=42 (accessed on 20 October 2021).
- U.S. Food and Drug Administration. FDA News Release. Available online: https://www.fda.gov/news-events/press-announcements/fda-warns-companies-marketing-unproven-products-derived-marijuana-claim-treat-or-cure-cancer (accessed on 20 October 2021).
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).