Allogeneic Stem Cell Transplantation in Mantle Cell Lymphoma in the Era of New Drugs and CAR-T Cell Therapy
Abstract
Simple Summary
Abstract
1. Introduction
2. Materials and Methods
3. Outcomes with allo-SCT
4. Which Is the Best Time for allo-SCT?
Key Points
5. Allo-SCT as an Option for “Very High Risk” Patients
- primarily chemorefractory subjects, after a reinduction therapy;
- “very high risk” cases [35].
Key Points
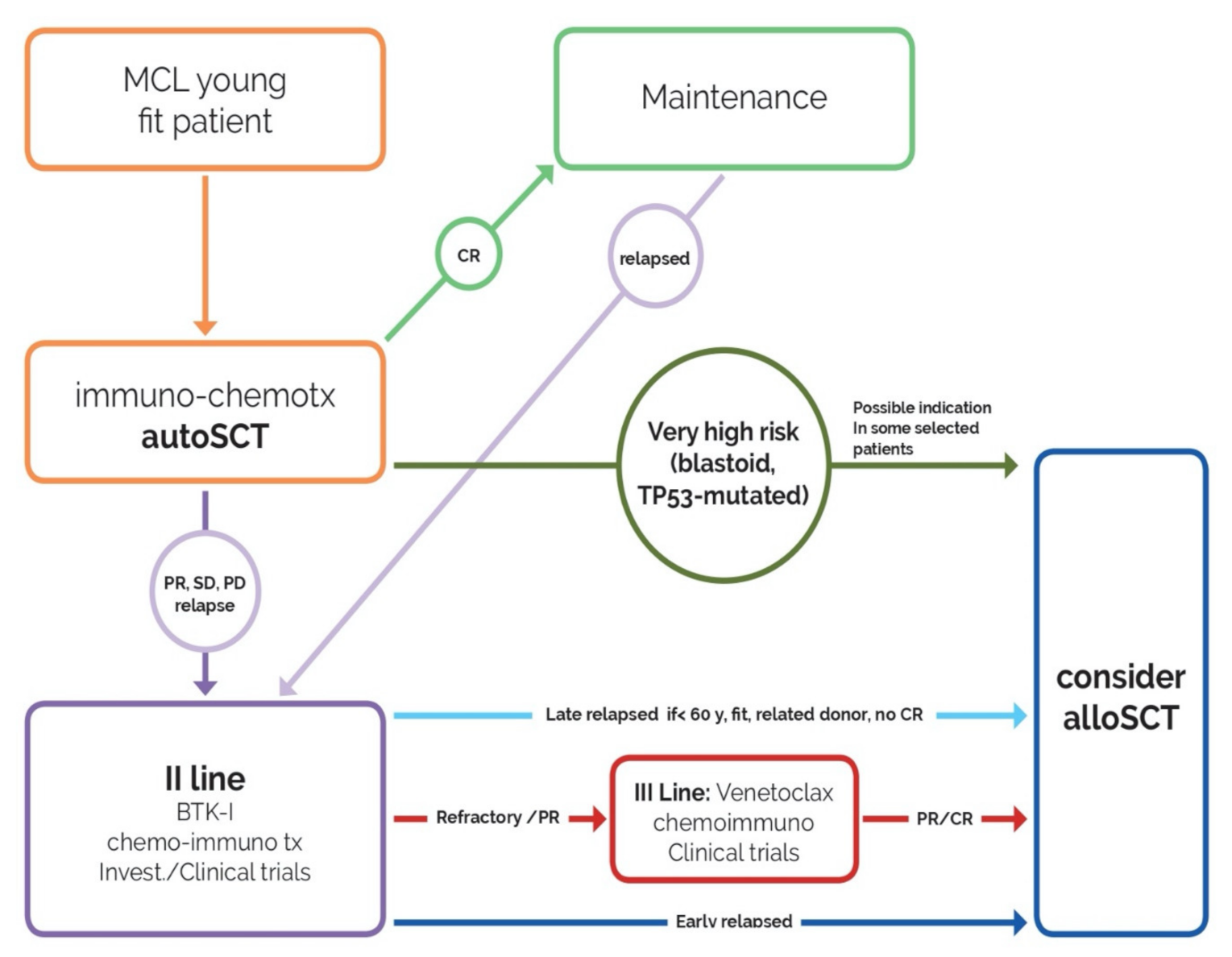
- after intensive immuno-chemotherapy (orange box), young and fit patients who achieve complete response (CR) proceed with maintenance (green arrow, green box).
- In case of partial response (PR), no response (SD) or relapse/progression (PD), patients receive re-induction therapy (BTK inhibitors, chemo-immunotherapy, investigational drug) (violet arrow, violet box).
- All patients relapsed before 24 months from the end of the first-line therapy who achieve a CR proceed immediately to allo-SCT (dark blue arrow, blue box).
- Subjects who relapse later than 24 months proceed to allo-SCT at the first signs of failure of second-line therapy (light blue arrow, blue box) if <60 years, with matched (related) donor.
- Patients who do not respond to second-line treatment, receive a third-line therapy (venetoclax, investigational drugs, chemo-immunotherapy) and proceed to allo-SCT at reaching of PR/CR (red arrow, red box, blue box).
- For cases at very high risk (blastoid variant, TP53-mutated), allo-SCT could be considered as first-line consolidation in some selected patients and discussed with each patient.
6. Chemosensitivity and the Role of allo-SCT
7. Bridging Strategies
7.1. Targeted Therapies
7.2. Chemo-Immunotherapy
7.3. Key Points
8. Conditioning Regimens
Key Points
9. Donor Selection and Stem-Cell Source
Key Points
10. Graft Versus Lymphoma
Key Points
11. Maintenance
12. Pre-Emptive Treatment
13. Post allo-SCT Salvage Treatment
Key Points
- No systematic available data on post allo-SCT maintenance in MCL;
- ESMO Clinical Practice Guidelines for MCL do not recommend MRD guided treatment in MCL, with the exception of the setting of DLI treatment post allo-SCT;
- salvage treatment: better considering DLI than 2 allo-SCT;
- salvage treatment: case reports of activity and safety of ibrutinib;
- a few prospective ongoing clinical trials (Table 2).
14. Positioning CAR-T in the allo-SCT Era, and Viceversa
- CAR-T KTE-X19 treatment is feasible and effective also in patients with active disease; allo-SCT is a curative option only in patients achieving a second response with salvage regimens.
- The follow-up for CAR-T KTE-X19 is short compared to allo-SCT; moreover, even if the experience of aggressive B-cell lymphomas showed that the PFS and OS rates were maintained at longer follow-up periods in patients treated with CAR-T cells, this has to be proven in MCL, a disease characterized by a continuous pattern of relapse even after many years of ongoing complete remission.
- The results obtained with CAR-T KTE-X19 were observed in a population with a poor prognosis, and all of the patients were pre-treated with iBTK.
- At the time of relapse after CAR-T KTE-X19, patients could perform a salvage treatment and could receive an allo-SCT as consolidation. On the other hand, previous allo-SCT might impact on the feasibility of a subsequent treatment with CAR-T KTE-X19. The use of allo-SCT as consolidation after the obtainment of a CR with CAR-T KTE-X19 is purely investigational.
- The NRM ranged between 10 to 24% with transplant strategies; NRM related to CAR-T KTE-X19 is 3%.
- Regarding feasibility, transplant is based on the availability of a donor (easier with the introduction of haploidentical donor). The rate of failure in the manufacture of CAR-T KTE-X19 cells was 4%. However, the relevant economic impact of CAR-T KTE-X19 cells should be considered, as well as its accessibility, which is still limited.
Key Points
15. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
Abbreviations
| aGVHD | acute Graft versus Host Disease |
| ALLO-SCT | allogeneic stem cell transplant |
| ASCT | autologous stem cell transplant |
| ATG | Anti Thymocyte Globuline |
| BCR | B-cell receptor |
| BR | Bendamustine—Rituximab |
| BSBMT | British Society of Blood and Marrow Transplantation |
| BTK | Bruton’s Tyrosine Kinase |
| CAR T | Chimeric Antigen Receptor T cell therapies |
| cGVHD | chronic Graft versus Host Disease |
| CIBMTR | Center for International Blood and Marrow Transplant Research |
| CLL | Chronic Lymphocytic Leukemia |
| c-MIPI | combined MIPI |
| CMV | cytomegalovirus |
| CR | complete response |
| CRS | Cytokine release syndrome |
| DLBCL | diffuse large B cell lymphoma |
| DLI | donor-lymphocyte infusion |
| DOR | duration of response |
| DSA | donor-specific antibodies |
| EBMT | European Society for Bone Marrow & Transplantation |
| EFS | event free survival |
| ESMO | European Society for Medical Oncology |
| FDG-PET | fluorodeoxyglucose—Positron Emission Tomography |
| FIL | Fondazione Italiana Linfomi |
| GVL | graft versus lymphoma |
| HCT | hematopoietic cell transplant |
| HCT-CI | hematopoietic cell transplantation comorbidity Index |
| HD ARA-C | High dose Cytarabine |
| HLA | Human Leukocyte Antigen |
| IBMTR | International Bone Marrow Transplant Registry |
| iBTK | BTK inhibitors |
| MCL | Mantle cell lymphoma |
| MIPI | Mantle cell International Prognostic index |
| miRNA | microRNA |
| MRD | minimal residual disease |
| MTX | methotrexate |
| MUD | matched unrelated donor |
| NHL | Non-Hodgkin Lymphoma |
| NMA | nonmyeloablative conditioning regimen |
| NRM | non-relapse mortality |
| ORR | overall response rate |
| OS | overall survival |
| PFS | progression free survival |
| POD | progression of disease |
| PR | partial response |
| R-BAC | Rituximab + Bendamustine + Cytarabine |
| R- CHOP | Rituximab + Cyclophosphamide, Doxorubicin, Vincristine, Prednisone |
| R-DHAP | Rituximab -Dexamethasone—high dose Cytarabine—Cisplatin |
| R- Hyper CVAD | Rituximab + Cyclophosphamide, Vincristine, Doxorubicin (Adriamycin), and Dexamethasone |
| RIC-ALLO SCT | reduced intensity conditioning—allogeneic stem cell transplant |
| RIC | reduced intensity conditioning |
| R/R | relapsed/refractory |
| TBI | total body irradiation |
| TRM | transplant related mortality |
| URD | unrelated donor |
| VR- CAP | bortezomib + rituximab + cyclophosphamide + doxorubicin and prednisone |
References
- Dreyling, M. European Mantle Cell Lymphoma Network Mantle cell lymphoma: Biology, clinical presentation, and therapeutic approaches. Am. Soc. Clin. Oncol. Educ. Book 2014, 34, 191–198. [Google Scholar] [CrossRef] [PubMed]
- Nordström, L.; Sernbo, S.; Eden, P.; Grønbæk, K.; Kolstad, A.; Räty, R.; Karjalainen, M.L.; Geisler, C.; Ralfkiær, E.; Sundström, C.; et al. SOX11 and TP53 add prognostic information to MIPI in a homogenously treated cohort of mantle cell lymphoma—A Nordic Lymphoma Group study. Br. J. Haematol. 2014, 166, 98–108. [Google Scholar] [CrossRef]
- Hoster, E.; Rosenwald, A.; Berger, F.; Bernd, H.W.; Hartmann, S.; Loddenkemper, C.; Barth, T.F.E.; Brousse, N.; Pileri, S.; Rymkiewicz, G.; et al. Prognostic value of Ki-67 index, cytology, and growth pattern in mantle-cell lymphoma: Results from randomized trials of the european mantle cell lymphoma network. J. Clin. Oncol. 2016, 34, 1386–1394. [Google Scholar] [CrossRef] [PubMed]
- Hoster, E.; Dreyling, M.; Klapper, W.; Gisselbrecht, C.; Van Hoof, A.; Kluin-Nelemans, H.C.; Pfreundschuh, M.; Reiser, M.; Metzner, B.; Einsele, H.; et al. A new prognostic index (MIPI) for patients with advanced-stage mantle cell lymphoma. Blood 2008, 111, 558–565. [Google Scholar] [CrossRef]
- Hermine, O.; Hoster, E.; Walewski, J.; Bosly, A.; Stilgenbauer, S.; Thieblemont, C.; Szymczyk, M.; Bouabdallah, R.; Kneba, M.; Hallek, M.; et al. Addition of high-dose cytarabine to immunochemotherapy before autologous stem-cell transplantation in patients aged 65 years or younger with mantle cell lymphoma (MCL Younger): A randomised, open-label, phase 3 trial of the European Mantle Cell Lymphoma N. Lancet 2016, 388, 565–575. [Google Scholar] [CrossRef]
- Le Gouill, S.; Thieblemont, C.; Oberic, L.; Moreau, A.; Bouabdallah, K.; Dartigeas, C.; Damaj, G.; Gastinne, T.; Ribrag, V.; Feugier, P.; et al. Rituximab after Autologous Stem-Cell Transplantation in Mantle-Cell Lymphoma. N. Engl. J. Med. 2017, 377, 1250–1260. [Google Scholar] [CrossRef]
- Ladetto, M.; Cortelazzo, S.; Ferrero, S.; Evangelista, A.; Mian, M.; Tavarozzi, R.; Zanni, M.; Cavallo, F.; Rocco, A. Di; Stefoni, V.; et al. Lenalidomide maintenance after autologous stem cell transplantation in mantle cell lymphoma (MCL): Results of a “Fondazione Italiana Linfomi” (FIL) multicentre randomised phase III trial. Lancet Haematol. 2021, 8, e34–e44. [Google Scholar] [CrossRef]
- Chihara, D.; Cheah, C.Y.; Westin, J.R.; Fayad, L.E.; Rodriguez, M.A.; Hagemeister, F.B.; Pro, B.; Mclaughlin, P.; Younes, A.; Samaniego, F.; et al. Rituximab plus hyper-CVAD alternating with MTX/Ara-C in patients with newly diagnosed mantle cell lymphoma: 15-year follow-up of a phase II study from the MD Anderson Cancer Center. Br. J. Haematol. 2016, 172, 80–88. [Google Scholar] [CrossRef]
- Massaro, F.; Stepanishyna, Y.; Manni, M.; Luminari, S.; Galimberti, S.; Marcheselli, L.; Visco, C.; Tecchio, C.; Stelitano, C.; Angrilli, F.; et al. Long-term results of the MCL01 phase II trial of rituximab plus HyperCVAD alternating with high-dose cytarabine and methotrexate for the initial treatment of patients with mantle cell lymphoma. Br. J. Haematol. 2020. [Google Scholar] [CrossRef]
- Kluin-Nelemans, H.C.; Hoster, E.; Hermine, O.; Walewski, J.; Trneny, M.; Geisler, C.H.; Stilgenbauer, S.; Thieblemont, C.; Vehling-Kaiser, U.; Doorduijn, J.K.; et al. Treatment of older patients with Mantle-Cell Lymphoma. N. Engl. J. Med. 2012, 367, 520–531. [Google Scholar] [CrossRef]
- Robak, T.; Huang, H.; Jin, J.; Zhu, J.; Liu, T.; Samoilova, O.; Pylypenko, H.; Verhoef, G.; Siritanaratkul, N.; Osmanov, E.; et al. Bortezomib-Based Therapy for Newly Diagnosed Mantle-Cell Lymphoma. N. Engl. J. Med. 2015, 372, 944–953. [Google Scholar] [CrossRef] [PubMed]
- Rummel, M.; Kaiser, U.; Balser, C.; Stauch, M.; Brugger, W.; Welslau, M.; Niederle, N.; Losem, C.; Boeck, H.-P.; Weidmann, E.; et al. Bendamustine plus rituximab versus fludarabine plus rituximab for patients with relapsed indolent and mantle-cell lymphomas: A multicentre, randomised, open-label, non-inferiority phase 3 trial. Lancet Oncol. 2016, 17, 57–66. [Google Scholar] [CrossRef]
- Flinn, I.W.; Van Der Jagt, R.; Kahl, B.; Wood, P.; Hawkins, T.; MacDonald, D.; Simpson, D.; Kolibaba, K.; Issa, S.; Chang, J.; et al. First-line treatment of patients with indolent non-hodgkin lymphoma or mantle-cell lymphoma with bendamustine plus rituximab versus R-CHOP or R-CVP: Results of the BRIGHT 5-year follow-up study. J. Clin. Oncol. 2019, 37, 984–991. [Google Scholar] [CrossRef]
- Visco, C.; Chiappella, A.; Nassi, L.; Patti, C.; Ferrero, S.; Barbero, D.; Evangelista, A.; Spina, M.; Molinari, A.; Rigacci, L.; et al. Rituximab, bendamustine, and low-dose cytarabine as induction therapy in elderly patients with mantle cell lymphoma: A multicentre, phase 2 trial from Fondazione Italiana Linfomi. Lancet Haematol. 2017, 4, e15–e23. [Google Scholar] [CrossRef]
- Leux, C.; Maynadié, M.; Troussard, X.; Cabrera, Q.; Herry, A.; Le Guyader-Peyrou, S.; Le Gouill, S.; Monnereau, A. Mantle cell lymphoma epidemiology: A population-based study in France. Ann. Hematol. 2014, 93, 1327–1333. [Google Scholar] [CrossRef]
- Sandoval-Sus, J.D.; Faramand, R.; Chavez, J.; Puri, S.; Parra, P.; Sokol, L.; Kharfan-Dabaja, M.A.; Shah, B.; Ayala, E. Allogeneic hematopoietic cell transplantation is potentially curative in mantle cell lymphoma: Results from a single institution study. Leuk. Lymphoma 2019, 60, 309–316. [Google Scholar] [CrossRef]
- Robinson, S.P.; Boumendil, A.; Finel, H.; Peggs, K.S.; Chevallier, P.; Sierra, J.; Finke, J.; Poiré, X.; Maillard, N.; Milpied, N.; et al. Long-term outcome analysis of reduced-intensity allogeneic stem cell transplantation in patients with mantle cell lymphoma: A retrospective study from the EBMT Lymphoma Working Party. Bone Marrow Transplant. 2018, 53, 617–624. [Google Scholar] [CrossRef]
- Vaughn, J.E.; Sorror, M.L.; Storer, B.E.; Chauncey, T.R.; Pulsipher, M.A.; Maziarz, R.T.; Maris, M.B.; Hari, P.; Laport, G.G.; Franke, G.N.; et al. Long-term sustained disease control in patients with mantle cell lymphoma with or without active disease after treatment with allogeneic hematopoietic cell transplantation after nonmyeloablative conditioning. Cancer 2015, 121, 3709–3716. [Google Scholar] [CrossRef]
- Le Gouill, S.; Kröger, N.; Dhedin, N.; Nagler, A.; Bouabdallah, K.; Yakoub-Agha, I.; Kanouni, T.; Bulabois, C.E.; Tournilhac, O.; Buzyn, A.; et al. Reduced-intensity conditioning allogeneic stem cell transplantation for relapsed/refractory mantle cell lymphoma: A multicenter experience. Ann. Oncol. 2012, 23, 2695–2703. [Google Scholar] [CrossRef]
- Cook, G.; Smith, G.M.; Kirkland, K.; Lee, J.; Pearce, R.; Thomson, K.; Morris, E.; Orchard, K.; Rule, S.; Russell, N.; et al. Outcome following reduced-intensity allogeneic stem cell transplantation (RIC AlloSCT) for relapsed and refractory mantle cell lymphoma (MCL): A study of the British society for blood and marrow transplantation. Biol. Blood Marrow Transplant. 2010, 16, 1419–1427. [Google Scholar] [CrossRef][Green Version]
- Dreyling, M.; Campo, E.; Hermine, O.; Jerkeman, M.; Le Gouill, S.; Rule, S.; Shpilberg, O.; Walewski, J.; Ladetto, M. Newly diagnosed and relapsed mantle cell lymphoma: ESMO Clinical Practice Guidelines for diagnosis, treatment and follow-up. Ann. Oncol. 2017, 28, iv62–iv71. [Google Scholar] [CrossRef]
- Okamoto, M.; Kusumoto, S. JSH practical guidelines for hematological malignancies, 2018: II. Lymphoma-4. Mantle cell lymphoma (MCL). Int. J. Hematol. 2020, 111, 5–15. [Google Scholar] [CrossRef]
- McKay, P.; Leach, M.; Jackson, B.; Robinson, S.; Rule, S. Guideline for the management of mantle cell lymphoma. Br. J. Haematol. 2018, 182, 46–62. [Google Scholar] [CrossRef]
- Dietrich, S. Mantle Cell Lymphoma. In The EBMT Handbook: Hematopoietic Stem Cell Transplantation and Cellular Therapies; Carreras, E., Dufour, C., Mohty, M., Kröger, N., Eds.; Springer Open: Cham, Switzerland, 2019; ISBN 9783030022778. [Google Scholar]
- National Comprehensive Cancer Network. Comprehensive Cancer Network Guideline—B Cell Lymphomas. Available online: www.nccn.org (accessed on 22 October 2020).
- Dreyling, M.; Aurer, I.; Cortelazzo, S.; Hermine, O.; Hess, G.; Jerkeman, M.; Le Gouill, S.; Ribrag, V.; Trněný, M.; Visco, C.; et al. Treatment for patients with relapsed/refractory mantle cell lymphoma: European-based recommendations. Leuk. Lymphoma 2018, 59, 1814–1828. [Google Scholar] [CrossRef]
- Visco, C.; Tisi, M.C.; Evangelista, A.; Di Rocco, A.; Zoellner, A.-K.; Zilioli, V.R.; Hohaus, S.; Sciarra, R.; Re, A.; Tecchio, C.; et al. Time to progression of mantle cell lymphoma after high-dose cytarabine-based regimens defines patients risk for death. Br. J. Haematol. 2019, 185, 940–944. [Google Scholar] [CrossRef]
- Visco, C.; Di Rocco, A.; Evangelista, A.; Quaglia, F.M.; Tisi, M.C.; Morello, L.; Zilioli, V.R.; Rusconi, C.; Hohaus, S.; Sciarra, R.; et al. Outcomes in first relapsed-refractory younger patients with mantle cell lymphoma: Results from the MANTLE-FIRST study. Leukemia 2020. [Google Scholar] [CrossRef]
- Dietrich, S.; Boumendil, A.; Finel, H.; Avivi, I.; Volin, L.; Cornelissen, J.; Jarosinska, R.J.; Schmid, C.; Finke, J.; Stevens, W.B.C.; et al. Outcome and prognostic factors in patients with mantle-cell lymphoma relapsing after autologous stem-cell transplantation: A retrospective study of the European Group for Blood and Marrow Transplantation (EBMT). Ann. Oncol. 2014, 25, 1053–1058. [Google Scholar] [CrossRef]
- Krüger, W.H.; Hirt, C.; Basara, N.; Sayer, H.G.; Behre, G.; Fischer, T.; Grobe, N.; Maschmeyer, G.; Niederwieser, D.; Dölken, G. Allogeneic stem cell transplantation for mantle cell lymphoma-final report from the prospective trials of the East German Study Group Haematology/Oncology (OSHO). Ann. Hematol. 2014, 93, 1587–1597. [Google Scholar] [CrossRef]
- Robinson, S.; Dreger, P.; Caballero, D.; Corradini, P.; Geisler, C.; Ghielmini, M.; Le Gouill, S.; Kimby, E.; Rule, S.; Vitolo, U.; et al. The EBMT/EMCL consensus project on the role of autologous and allogeneic stem cell transplantation in mantle cell lymphoma. Leukemia 2015, 29, 464–473. [Google Scholar] [CrossRef]
- Rule, S.; Cook, G.; Russell, N.H.; Hunter, A.; Robinson, S.; Morley, N.; Sureda, A.; Patrick, P.; Clifton-Hadley, L.; Adedayo, T.; et al. Allogeneic stem cell transplantation as part of front line therapy for Mantle cell lymphoma. Br. J. Haematol. 2019, 184, 999–1005. [Google Scholar] [CrossRef]
- Fenske, T.S.; Zhang, M.-J.; Carreras, J.; Ayala, E.; Burns, L.J.; Cashen, A.; Costa, L.J.; Freytes, C.O.; Gale, R.P.; Hamadani, M.; et al. Autologous or reduced-intensity conditioning allogeneic hematopoietic cell transplantation for chemotherapy-sensitive mantle-cell lymphoma: Analysis of transplantation timing and modality. J. Clin. Oncol. 2014, 32, 273–281. [Google Scholar] [CrossRef]
- Tessoulin, B.; Ceballos, P.; Chevallier, P.; Blaise, D.; Tournilhac, O.; Gauthier, J.; Maillard, N.; Tabrizi, R.; Choquet, S.; Carras, S.; et al. Allogeneic stem cell transplantation for patients with mantle cell lymphoma who failed autologous stem cell transplantation: A national survey of the SFGM-TC. Bone Marrow Transplant. 2016, 51, 1184–1190. [Google Scholar] [CrossRef]
- Cohen, J.B.; Burns, L.J.; Bachanova, V. Role of allogeneic stem cell transplantation in mantle cell lymphoma. Eur. J. Haematol. 2015, 94, 290–297. [Google Scholar] [CrossRef]
- Husby, S.; Ralfkiaer, U.; Garde, C.; Zandi, R.; Ek, S.; Kolstad, A.; Jerkeman, M.; Laurell, A.; Räty, R.; Pedersen, L.B.; et al. miR-18b overexpression identifies mantle cell lymphoma patients with poor outcome and improves the MIPI-B prognosticator. Blood 2015, 125, 2669–2677. [Google Scholar] [CrossRef][Green Version]
- Holte, H.; Beiske, K.; Boyle, M.; Trøen, G.; Blaker, Y.N.; Myklebust, J.; Kvaløy, S.; Rosenwald, A.; Lingjaerde, O.C.; Rimsza, L.M.; et al. The MCL35 gene expression proliferation assay predicts high-risk MCL patients in a Norwegian cohort of younger patients given intensive first line therapy. Br. J. Haematol. 2018, 183, 225–234. [Google Scholar] [CrossRef]
- Bomben, R.; Ferrero, S.; D’Agaro, T.; Dal Bo, M.; Re, A.; Evangelista, A.; Carella, A.M.; Zamò, A.; Vitolo, U.; Omedè, P.; et al. A B-cell receptor-related gene signature predicts survival in mantle cell lymphoma: Results from the Fondazione Italiana Linfomi MCL-0208 trial. Haematologica 2018, 103, 849–856. [Google Scholar] [CrossRef]
- Streich, L.; Sukhanova, M.; Lu, X.; Chen, Y.-H.; Venkataraman, G.; Mathews, S.; Zhang, S.; Kelemen, K.; Segal, J.; Gao, J.; et al. Aggressive morphologic variants of mantle cell lymphoma characterized with high genomic instability showing frequent chromothripsis, CDKN2A/B loss, and TP53 mutations: A multi-institutional study. Genes Chromosom. Cancer 2020, 59, 484–494. [Google Scholar] [CrossRef]
- Sakhdari, A.; Ok, C.Y.; Patel, K.P.; Kanagal-Shamanna, R.; Yin, C.C.; Zuo, Z.; Hu, S.; Routbort, M.J.; Luthra, R.; Medeiros, L.J.; et al. TP53 mutations are common in mantle cell lymphoma, including the indolent leukemic non-nodal variant. Ann. Diagn. Pathol. 2019, 41, 38–42. [Google Scholar] [CrossRef]
- Aukema, S.M.; Hoster, E.; Rosenwald, A.; Canoni, D.; Delfau-Larue, M.-H.; Rymkiewicz, G.; Thorns, C.; Hartmann, S.; Kluin-Nelemans, H.; Hermine, O.; et al. Expression of TP53 is associated with the outcome of MCL independent of MIPI and Ki-67 in trials of the European MCL Network. Blood 2018, 131, 417–420. [Google Scholar] [CrossRef]
- Ferrero, S.; Zaccaria, G.M.; Grimaldi, D.; Moia, R.; Genuardi, E.; Ghislieri, M.; Favini, C.; Rocco, A. Di; Re, A.; Stefoni, V.; et al. Focus on Patients with TP53 Disruption in the Fondazione Italiana Linfomi (FIL) MCL0208 Trial: Uniform Poor Outcome, Regardless of Baseline Predictors, MRD Status and Lenalidomide Maintenance. EHA Library 2020, 293649, EP1160. [Google Scholar]
- Ferrero, S.; Rossi, D.; Rinaldi, A.; Bruscaggin, A.; Spina, V.; Eskelund, C.W.; Evangelista, A.; Moia, R.; Kwee, I.; Dahl, C.; et al. KMT2D mutations and TP53 disruptions are poor prognostic biomarkers in mantle cell lymphoma receiving high-dose therapy: A FIL study. Haematologica 2020, 105, 1604–1612. [Google Scholar] [CrossRef] [PubMed]
- Lin, R.J.; Ho, C.; Hilden, P.D.; Barker, J.N.; Giralt, S.A.; Hamlin, P.A.; Jakubowski, A.A.; Castro-Malaspina, H.R.; Robinson, K.S.; Papadopoulos, E.B.; et al. Allogeneic haematopoietic cell transplantation impacts on outcomes of mantle cell lymphoma with TP53 alterations. Br. J. Haematol. 2019, 184, 1006–1010. [Google Scholar] [CrossRef] [PubMed]
- Magnusson, E.; Cao, Q.; Linden, M.A.; Frolich, J.; Anand, V.; Burns, L.J.; Bachanova, V. Hematopoietic cell transplantation for mantle cell lymphoma: Predictive value of pretransplant positron emission tomography/computed tomography and bone marrow evaluations for outcomes. Clin. Lymphoma. Myeloma Leuk. 2014, 14, 114–121. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Hamadani, M.; Saber, W.; Ahn, K.W.; Carreras, J.; Cairo, M.S.; Fenske, T.S.; Gale, R.P.; Gibson, J.; Hale, G.A.; Hari, P.N.; et al. Allogeneic Hematopoietic Cell Transplantation for Chemotherapy-Unresponsive Mantle Cell Lymphoma: A Cohort Analysis from the Center for International Blood and Marrow Transplant Research. Biol. Blood Marrow Transplant. 2013, 19, 625–631. [Google Scholar] [CrossRef]
- Dreger, P.; Michallet, M.; Bosman, P.; Dietrich, S.; Sobh, M.; Boumendil, A.; Nagler, A.; Scheid, C.; Cornelissen, J.; Niederwieser, D.; et al. Ibrutinib for bridging to allogeneic hematopoietic cell transplantation in patients with chronic lymphocytic leukemia or mantle cell lymphoma: A study by the EBMT Chronic Malignancies and Lymphoma Working Parties. Bone Marrow Transplant. 2019, 54, 44–52. [Google Scholar] [CrossRef]
- Bachanova, V.; Burns, L.J.; Ahn, K.W.; Laport, G.G.; Akpek, G.; Kharfan-Dabaja, M.A.; Nishihori, T.; Agura, E.; Armand, P.; Jaglowski, S.M.; et al. Impact of Pretransplantation 18F-fluorodeoxy Glucose-Positron Emission Tomography Status on Outcomes after Allogeneic Hematopoietic Cell Transplantation for Non-Hodgkin Lymphoma. Biol. Blood Marrow Transpl. 2015, 21, 1605–1611. [Google Scholar] [CrossRef]
- Rule, S.; Jurczak, W.; Jerkeman, M.; Rusconi, C.; Trneny, M.; Offner, F.; Caballero, D.; Joao, C.; Witzens-Harig, M.; Hess, G.; et al. Ibrutinib versus temsirolimus: 3-year follow-up of patients with previously treated mantle cell lymphoma from the phase 3, international, randomized, open-label RAY study. Leukemia 2018, 32, 1799–1803. [Google Scholar] [CrossRef]
- Song, Y.; Zhou, K.; Zou, D.; Zhou, J.; Hu, J.; Yang, H.; Zhang, H.; Ji, J.; Xu, W.; Jin, J.; et al. Treatment of patients with relapsed or refractory Mantle-Cell Lymphoma with zanubrutinib, a selective inhibitor of Bruton’s Tyrosine Kinase. Clin. Cancer Res. 2020, 26, 4216–4224. [Google Scholar] [CrossRef]
- Witzig, T.E.; Inwards, D. Acalabrutinib for mantle cell lymphoma. Blood 2019, 133, 2570–2574. [Google Scholar] [CrossRef]
- Eyre, T.A.; Walter, H.S.; Iyengar, S.; Follows, G.; Cross, M.; Fox, C.P.; Hodson, A.; Coats, J.; Narat, S.; Morley, N.; et al. Efficacy of venetoclax monotherapy in patients with relapsed, refractory mantle cell lymphoma after Bruton tyrosine kinase inhibitor therapy. Haematologica 2019, 104, e68–e71. [Google Scholar] [CrossRef]
- Tam, C.S.; Anderson, M.A.; Pott, C.; Agarwal, R.; Handunnetti, S.; Hicks, R.J.; Burbury, K.; Turner, G.; Di Iulio, J.; Bressel, M.; et al. Ibrutinib plus Venetoclax for the treatment of Mantle-Cell Lymphoma. N. Engl. J. Med. 2018, 378, 1211–1223. [Google Scholar] [CrossRef] [PubMed]
- Rummel, M.J.; Al-Batran, S.E.; Kim, S.Z.; Welslau, M.; Hecker, R.; Kofahl-Krause, D.; Josten, K.M.; Dürk, H.; Rost, A.; Neise, M.; et al. Bendamustine plus rituximab is effective and has a favorable toxicity profile in the treatment of mantle cell and low-grade non-Hodgkin’s lymphoma. J. Clin. Oncol. 2005, 23, 3383–3389. [Google Scholar] [CrossRef] [PubMed]
- Visco, C.; Finotto, S.; Zambello, R.; Paolini, R.; Menin, A.; Zanotti, R.; Zaja, F.; Semenzato, G.; Pizzolo, G.; D’Amore, E.S.G.; et al. Combination of rituximab, bendamustine, and cytarabine for patients with mantle-cell non-Hodgkin lymphoma ineligible for intensive regimens or autologous transplantation. J. Clin. Oncol. 2013, 31, 1442–1449. [Google Scholar] [CrossRef]
- McCulloch, R.; Visco, C.; Eyre, T.A.; Frewin, R.; Phillips, N.; Tucker, D.L.; Quaglia, F.M.; McMillan, A.; Lambert, J.; Crosbie, N.; et al. Efficacy of R-BAC in relapsed, refractory mantle cell lymphoma post BTK inhibitor therapy. Br. J. Haematol. 2020, 189, 684–688. [Google Scholar] [CrossRef]
- Chakraverty, R.; Mackinnon, S. Allogeneic Transplantation for Lymphoma. J. Clin. Oncol. 2011, 29, 1855–1863. [Google Scholar] [CrossRef]
- Khouri, I.F.; Champlin, R.E. Non-myeloablative allogeneic stem cell transplantation for non-Hodgkin lymphoma. Cancer J. 2012, 18, 457–462. [Google Scholar] [CrossRef]
- Appelbaum, F.R. Allogeneic hematopoietic cell transplantation for acute myeloid leukemia when a matched related donor is not available. Hematol. Am. Soc. Hematol. Educ. Progr. 2008, 412–417. [Google Scholar] [CrossRef]
- Gragert, L.; Eapen, M.; Williams, E.; Freeman, J.; Spellman, S.; Baitty, R.; Hartzman, R.; Rizzo, J.D.; Horowitz, M.; Confer, D.; et al. HLA match likelihoods for hematopoietic stem-cell grafts in the U.S. registry. N. Engl. J. Med. 2014, 371, 339–348. [Google Scholar] [CrossRef]
- Ballen, K.K.; Gluckman, E.; Broxmeyer, H.E. Umbilical cord blood transplantation: The first 25 years and beyond. Blood 2013, 122, 491–498. [Google Scholar] [CrossRef]
- Kanate, A.S.; Mussetti, A.; Kharfan-Dabaja, M.A.; Ahn, K.W.; Digilio, A.; Beitinjaneh, A.; Chhabra, S.; Fenske, T.S.; Freytes, C.; Gale, R.P.; et al. Reduced-intensity transplantation for lymphomas using haploidentical related donors vs. HLA-matched unrelated donors. Blood 2016, 127, 938–947. [Google Scholar] [CrossRef]
- Lu, D.P.; Dong, L.; Wu, T.; Huang, X.J.; Zhang, M.J.; Han, W.; Chen, H.; Liu, D.H.; Gao, Z.Y.; Chen, Y.H.; et al. Conditioning including antithymocyte globulin followed by unmanipulated HLA-mismatched/haploidentical blood and marrow transplantation can achieve comparable outcomes with HLA-identical sibling transplantation. Blood 2006, 107, 3065–3073. [Google Scholar] [CrossRef] [PubMed]
- Wang, Y.; Liu, D.-H.; Liu, K.-Y.; Xu, L.-P.; Zhang, X.-H.; Han, W.; Chen, H.; Chen, Y.-H.; Wang, F.-R.; Wang, J.-Z.; et al. Long-term follow-up of haploidentical hematopoietic stem cell transplantation without in vitro T cell depletion for the treatment of leukemia: Nine years of experience at a single center. Cancer 2013, 119, 978–985. [Google Scholar] [CrossRef] [PubMed]
- Ghosh, N.; Karmali, R.; Rocha, V.; Ahn, K.W.; DiGilio, A.; Hari, P.N.; Bachanova, V.; Bacher, U.; Dahi, P.; De Lima, M.; et al. Reduced-intensity transplantation for lymphomas using haploidentical related donors versus HLA-matched sibling donors: A center for international blood and marrow transplant research analysis. J. Clin. Oncol. 2016, 34, 3141–3149. [Google Scholar] [CrossRef]
- Bazarbachi, A.; Boumendil, A.; Finel, H.; Castagna, L.; Dominietto, A.; Blaise, D.; Diez-Martin, J.L.; Tischer, J.; Gülbas, Z.; Wallet, H.L.; et al. Influence of donor type, stem cell source and conditioning on outcomes after haploidentical transplant for lymphoma—a LWP-EBMT study. Br. J. Haematol. 2020, 188, 745–756. [Google Scholar] [CrossRef]
- Fatobene, G.; Rocha, V.; St. Martin, A.; Hamadani, M.; Robinson, S.; Bashey, A.; Boumendil, A.; Brunstein, C.; Castagna, L.; Dominietto, A.; et al. Nonmyeloablative alternative donor transplantation for Hodgkin and non-Hodgkin lymphoma: From the LWP-EBMT, Eurocord, and CIBMTR. J. Clin. Oncol. 2020, 38, 1518–1526. [Google Scholar] [CrossRef]
- Jones, R.J.; Ambinder, R.F.; Piantadosi, S.; Santos, G.W. Evidence of a graft-versus-lymphoma effect associated with allogeneic bone marrow transplantation. Blood 1991, 77, 649–653. [Google Scholar] [CrossRef]
- Ratanatharathorn, V.; Uberti, J.; Karanes, C.; Abella, E.; Lum, L.G.; Momin, F.; Cummings, G.; Sensenbrenner, L.L. Prospective comparative trial of autologous versus allogeneic bone marrow transplantation in patients with non-Hodgkin’s lymphoma. Blood 1994, 84, 1050–1055. [Google Scholar] [CrossRef]
- Schimmer, A.D.; Jamal, S.; Messner, H.; Keating, A.; Meharchand, J.; Huebsch, L.; Walker, I.; Benger, A.; Gluck, S.; Smith, A. Allogeneic or autologous bone marrow transplantation (BMT) for non-Hodgkin’s lymphoma (NHL): Results of a provincial strategy. Bone Marrow Transplant. 2000, 26, 859–864. [Google Scholar] [CrossRef]
- Van Besien, K.; Loberiza, F.R.; Bajorunaite, R.; Armitage, J.O.; Bashey, A.; Burns, L.J.; Freytes, C.O.; Gibson, J.; Horowitz, M.M.; Inwards, D.J.; et al. Comparison of autologous and allogeneic hematopoietic stem cell transplantation for follicular lymphoma. Blood 2003, 102, 3521–3529. [Google Scholar] [CrossRef]
- Bierman, P.J.; Sweetenham, J.W.; Loberiza, F.R.; Taghipour, G.; Lazarus, H.M.; Rizzo, J.D.; Schmitz, N.; van Besien, K.; Vose, J.M.; Horowitz, M.; et al. Syngeneic Hematopoietic Stem-Cell Transplantation for Non-Hodgkin’s Lymphoma: A Comparison With Allogeneic and Autologous Transplantation—The Lymphoma Working Committee of the International Bone Marrow Transplant Registry and the European Group for Blood. J. Clin. Oncol. 2003, 21, 3744–3753. [Google Scholar] [CrossRef]
- Van Besien, K.W.; De Lima, M.; Giralt, S.A.; Moore, D.F.; Khouri, I.F.; Rondón, G.; Mehra, R.; Andersson, B.S.; Dyer, C.; Cleary, K.; et al. Management of lymphoma recurrence after allogeneic transplantation: The relevance of graft-versus-lymphoma effect. Bone Marrow Transplant. 1997, 19, 977–982. [Google Scholar] [CrossRef][Green Version]
- Mandigers, C.M.P.W.; Verdonck, L.F.; Meijerink, J.P.P.; Dekker, A.W.; Schattenberg, A.V.M.B.; Raemaekers, J.M.M. Graft-versus-lymphoma effect of donor lymphocyte infusion in indolent lymphomas relapsed after allogeneic stem cell transplantation. Bone Marrow Transplant. 2003, 32, 1159–1163. [Google Scholar] [CrossRef][Green Version]
- Khouri, I.F.; Champlin, R.E. Nonmyeloablative stem cell transplantation for lymphoma. Semin. Oncol. 2004, 31, 22–26. [Google Scholar] [CrossRef]
- Butcher, B.W.; Collins, R.H. The graft-versus-lymphoma effect: Clinical review and future opportunities. Bone Marrow Transplant. 2005, 36, 1–17. [Google Scholar] [CrossRef]
- Cavattoni, I.; Zabelina, T.; Ayuk, F.; Wolschke, C.; Bacher, U.; Zander, A.; Kröger, N. Pilot study of rituximab plus donor-lymphocyte infusion to prevent or treat relapse in B-cell lymphoma after allogeneic stem cell transplantation. Leuk. Lymphoma 2010, 51, 146–148. [Google Scholar] [CrossRef]
- Andersen, N.S.; Pedersen, L.B.; Laurell, A.; Elonen, E.; Kolstad, A.; Boesen, A.M.; Pedersen, L.M.; Lauritzsen, G.F.; Ekanger, R.; Nilsson-Ehle, H.; et al. Pre-Emptive treatment with rituximab of molecular relapse after autologous stem cell transplantation in mantle cell lymphoma. J. Clin. Oncol. 2009, 27, 4365–4370. [Google Scholar] [CrossRef]
- Ferrero, S.; Monitillo, L.; Mantoan, B.; Barbero, D.; Genuardi, E.; Barbiero, S.; Bernocco, E.; Caracciolo, D.; Ruella, M.; Drandi, D.; et al. Rituximab-based pre-emptive treatment of molecular relapse in follicular and mantle cell lymphoma. Ann. Hematol. 2013, 92, 1503–1511. [Google Scholar] [CrossRef]
- Horstmann, K.; Boumendil, A.; Finke, J.; Finel, H.; Kanfer, E.; Milone, G.; Russell, N.; Bacigalupo, A.; Chalandon, Y.; Diez-Martin, J.L.; et al. Second allo-SCT in patients with lymphoma relapse after a first allogeneic transplantation. A retrospective study of the EBMT Lymphoma Working Party. Bone Marrow Transplant. 2015, 50, 790–794. [Google Scholar] [CrossRef][Green Version]
- Wudhikarn, K.; Brunstein, C.G.; Bachanova, V.; Burns, L.J.; Cao, Q.; Weisdorf, D.J. Relapse of lymphoma after allogeneic hematopoietic cell transplantation: Management strategies and outcome. Biol. Blood Marrow Transpl. 2011, 17, 1497–1504. [Google Scholar] [CrossRef]
- Rich, J.D.; Clark, S.M.; Fedoriw, Y.; Jewells, V.; Wood, W.; Dittus, C. Complete remission with ibrutinib after allogeneic stem cell transplant for central nervous system relapse of mantle cell lymphoma: A case report and literature review. Clin. Case Rep. 2019, 7, 1957–1961. [Google Scholar] [CrossRef]
- Khouri, I.F.; Jabbour, E.; Saliba, R.M.; Ledesma, C.; Burger, J.A.; O’Brien, S.; Gulbis, A.M. Ibrutinib in Patients (Pts) with Mantle Cell Lymphoma (MCL) and Chronic Lymphocytic Leukemia (CLL) with Relapsed Disease Post Allogeneic Stem Cell Transplantation (alloSCT). Blood 2015, 126, 4345. [Google Scholar] [CrossRef]
- Michallet, M.; Dreger, P.; Sobh, M.; Hoek, J.; Boumendil, A.; Muller, L.; Corradini, P.; Bethge, W.; Russo, D.; Durakovic, N.; et al. Salvage use of ibrutinib after allogeneic hematopoietic stem cell transplantation (allo-HSCT) for B cell malignancies: A study of the French Cooperative Group for CLL, the French Soceity for Blood and Marrow Transplantation (SFGM-TC), and the European Soc. Blood 2016, 128, 4659. [Google Scholar] [CrossRef]
- Michallet, M.; Dreger, P.; Sobh, M.; Koster, L.; Hoek, J.; Boumendil, A.; Scheid, C.; Fox, C.P.; Wulf, G.; Krüger, W.; et al. Ibrutinib as a salvage therapy after allogeneic HCT for chronic lymphocytic leukemia. Bone Marrow Transplant. 2020, 55, 884–890. [Google Scholar] [CrossRef]
- Kochenderfer, J.N.; Dudley, M.E.; Carpenter, R.O.; Kassim, S.H.; Rose, J.J.; Telford, W.G.; Hakim, F.T.; Halverson, D.C.; Fowler, D.H.; Hardy, N.M.; et al. Donor-derived CD19-targeted T cells cause regression of malignancy persisting after allogeneic hematopoietic stem cell transplantation. Blood 2013, 122, 4129–4139. [Google Scholar] [CrossRef]
- Wang, M.; Munoz, J.; Goy, A.; Locke, F.L.; Jacobson, C.A.; Hill, B.T.; Timmerman, J.M.; Holmes, H.; Jaglowski, S.; Flinn, I.W.; et al. KTE-X19 CAR T-Cell Therapy in Relapsed or Refractory Mantle-Cell Lymphoma. N. Engl. J. Med. 2020, 382, 1331–1342. [Google Scholar] [CrossRef]
- Gu, B.; Shi, B.-Y.; Zhang, X.; Zhou, S.-Y.; Chu, J.-H.; Wu, X.-J.; Fu, C.-C.; Qiu, H.-Y.; Han, Y.; Chen, S.-N.; et al. Allogeneic haematopoietic stem cell transplantation improves outcome of adults with relapsed/refractory Philadelphia chromosome-positive acute lymphoblastic leukemia entering remission following CD19 chimeric antigen receptor T cells. Bone Marrow Transplant. 2020, 56, 91–100. [Google Scholar] [CrossRef]
- Zhao, H.; Wei, J.; Wei, G.; Luo, Y.; Shi, J.; Cui, Q.; Zhao, M.; Liang, A.; Zhang, Q.; Yang, J.; et al. Pre-transplant MRD negativity predicts favorable outcomes of CAR-T therapy followed by haploidentical HSCT for relapsed/refractory acute lymphoblastic leukemia: A multi-center retrospective study. J. Hematol. Oncol. 2020, 13, 42. [Google Scholar] [CrossRef]
- Zhang, M.; Huang, H. How to Combine the Two Landmark Treatment Methods—Allogeneic Hematopoietic Stem Cell Transplantation and Chimeric Antigen Receptor T Cell Therapy Together to Cure High-Risk B Cell Acute Lymphoblastic Leukemia? Front. Immunol. 2020, 11, 3300. [Google Scholar] [CrossRef]
- Barnes, D.W.H.; Corp, M.J.; Loutit, J.F.; Neal, F.E. Treatment of murine leukaemia with X rays and homologous bone marrow; preliminary communication. Br. Med. J. 1956, 2, 626–627. [Google Scholar] [CrossRef]
| Study | Timing | N° Patients | ORR/CR | Median FU | 2-yr NRM | 5-yr PFS | 5-yr OS | Main Toxicities |
|---|---|---|---|---|---|---|---|---|
| Le Gouill et al. | Salvage | 70 | 95%/89% | 24mo | 32% | ≈25% | ≈25% | GVHD |
| Krüger et al. | First line | 24 | 86%/76% | 2.8yr | ≈67% | ≈73% | Stomatitis, infection, GVHD | |
| Salvage | 15 | 91%/83% | 2.8yr | ≈67% | ≈73% | |||
| Rule et al. | First line | 25 | 92%/60% | 60.5mo | 13% | 56% | 76% | Infection, mucositis, GVHD |
| Fenske et al. | First response | 50 | 48mo | ≈30% | 55% | 62% | ||
| Salvage | 88 | 37mo | ≈25% | 24% | 31% | |||
| Tessoulin et al. | Salvage | 106 | 97%/86% | 45mo | ≈30% | ≈35% | ≈55% | GVHD, infection |
| Treatment Drugs | Trial Phase | Recruitment Status | Timing Administration | Malignancies Included | Clinical Trials Identifier |
|---|---|---|---|---|---|
| Idelalisib vs. Placebo | I | Recruiting | Maintenance post allo-HSCT | MCL, CLL, FL, DLBCL, B cell-tumors | NCT03151057 |
| Anti-CD19-CAR-transduced T cell | I | Recruiting | Salvage post allo-HSCT | NHL, HL, B cell-tumors | NCT01087294 |
| Ibrutinib | II | Ongoing, but not currently recruiting | Salvage post allo-HSCT | MCL, CLL, FL, HL, B cell-tumors | NCT02869633 |
| Ibrutinib and Selinexor | I | Recruiting | Salvage post allo-HSCT * | MCL, DLBCL, CLL, SLL, PLL | NCT02303392 |
| Study | Approach | N° Patients | Median Age (Range) | N° Prior Lines (Range) | N° Refractory (%) | N° Prior Rituximab (Range) | N° Prior iBTK (Range) | ORR/CR | Median FU | 1-yr NRM | 1-yr PFS | 1-yr OS |
|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Hamadani et al. | MAC | 74 | 54 (27–69) | 3 (2–5) | 37 (50) | 11 (52) | 0 | ? | 35 mo | 43% | 31% | 33% |
| Hamadani et al. | RIC/non -myeloablative | 128 | 59 (42–75) | 4 (1–5) | 71 (55) | 52 (80) | 0 | ? | 43 mo | 38% | 38% | 46% |
| Cook et al. | RIC | 70 | 52.2 (34.7–68.8) | 2 (1–6) | 12 (17%) | 40 (64%) | 0 | 51/48% | 37 mo | 18% | ≈50% | ≈75% |
| Le Gouill et al. | RIC | 70 | 56 (33–67.5) | 2 (1–5) | 15 (21%) | ? | 0 | 94%/89% of eval. pts | 24 mo | ≈20% | ? | ≈60% |
| Wang et al. | CAR-T | 68 | 65 (38–79) | 3 (1–5) | 27 (40) | 68 (100) | 68 (100) | 85%/59% | 12.3 mo | 3% | 61% | 83% |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Marangon, M.; Visco, C.; Barbui, A.M.; Chiappella, A.; Fabbri, A.; Ferrero, S.; Galimberti, S.; Luminari, S.; Musuraca, G.; Re, A.; et al. Allogeneic Stem Cell Transplantation in Mantle Cell Lymphoma in the Era of New Drugs and CAR-T Cell Therapy. Cancers 2021, 13, 291. https://doi.org/10.3390/cancers13020291
Marangon M, Visco C, Barbui AM, Chiappella A, Fabbri A, Ferrero S, Galimberti S, Luminari S, Musuraca G, Re A, et al. Allogeneic Stem Cell Transplantation in Mantle Cell Lymphoma in the Era of New Drugs and CAR-T Cell Therapy. Cancers. 2021; 13(2):291. https://doi.org/10.3390/cancers13020291
Chicago/Turabian StyleMarangon, Miriam, Carlo Visco, Anna Maria Barbui, Annalisa Chiappella, Alberto Fabbri, Simone Ferrero, Sara Galimberti, Stefano Luminari, Gerardo Musuraca, Alessandro Re, and et al. 2021. "Allogeneic Stem Cell Transplantation in Mantle Cell Lymphoma in the Era of New Drugs and CAR-T Cell Therapy" Cancers 13, no. 2: 291. https://doi.org/10.3390/cancers13020291
APA StyleMarangon, M., Visco, C., Barbui, A. M., Chiappella, A., Fabbri, A., Ferrero, S., Galimberti, S., Luminari, S., Musuraca, G., Re, A., Zilioli, V. R., & Ladetto, M. (2021). Allogeneic Stem Cell Transplantation in Mantle Cell Lymphoma in the Era of New Drugs and CAR-T Cell Therapy. Cancers, 13(2), 291. https://doi.org/10.3390/cancers13020291





