Lower Survival and Increased Circulating Suppressor Cells in Patients with Relapsed/Refractory Diffuse Large B-Cell Lymphoma with Deficit of Vitamin D Levels Using R-GDP Plus Lenalidomide (R2-GDP): Results from the R2-GDP-GOTEL Trial
Abstract
Simple Summary
Abstract
1. Introduction
2. Results
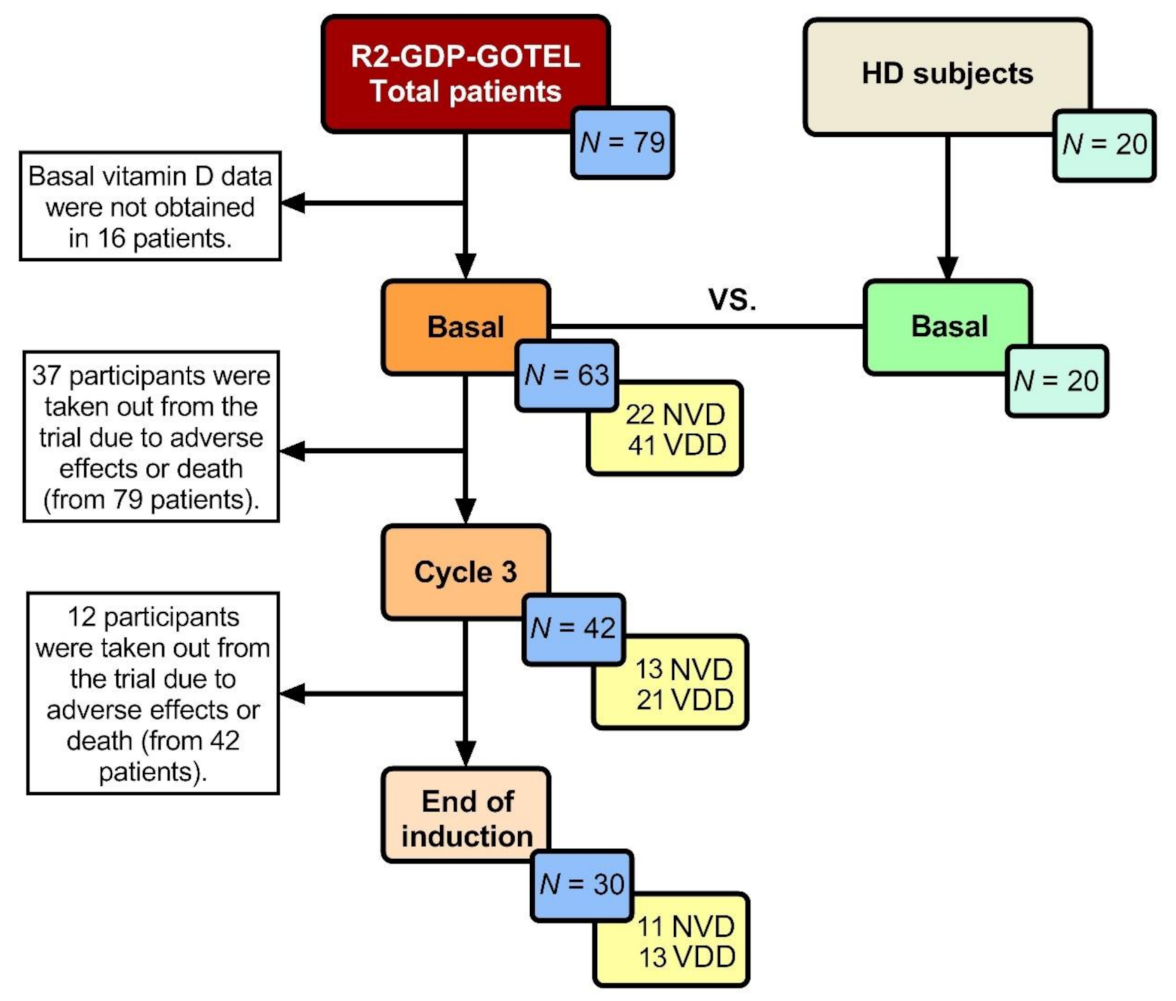
2.1. Clinical Characteristics of R/R DLBCL Patients
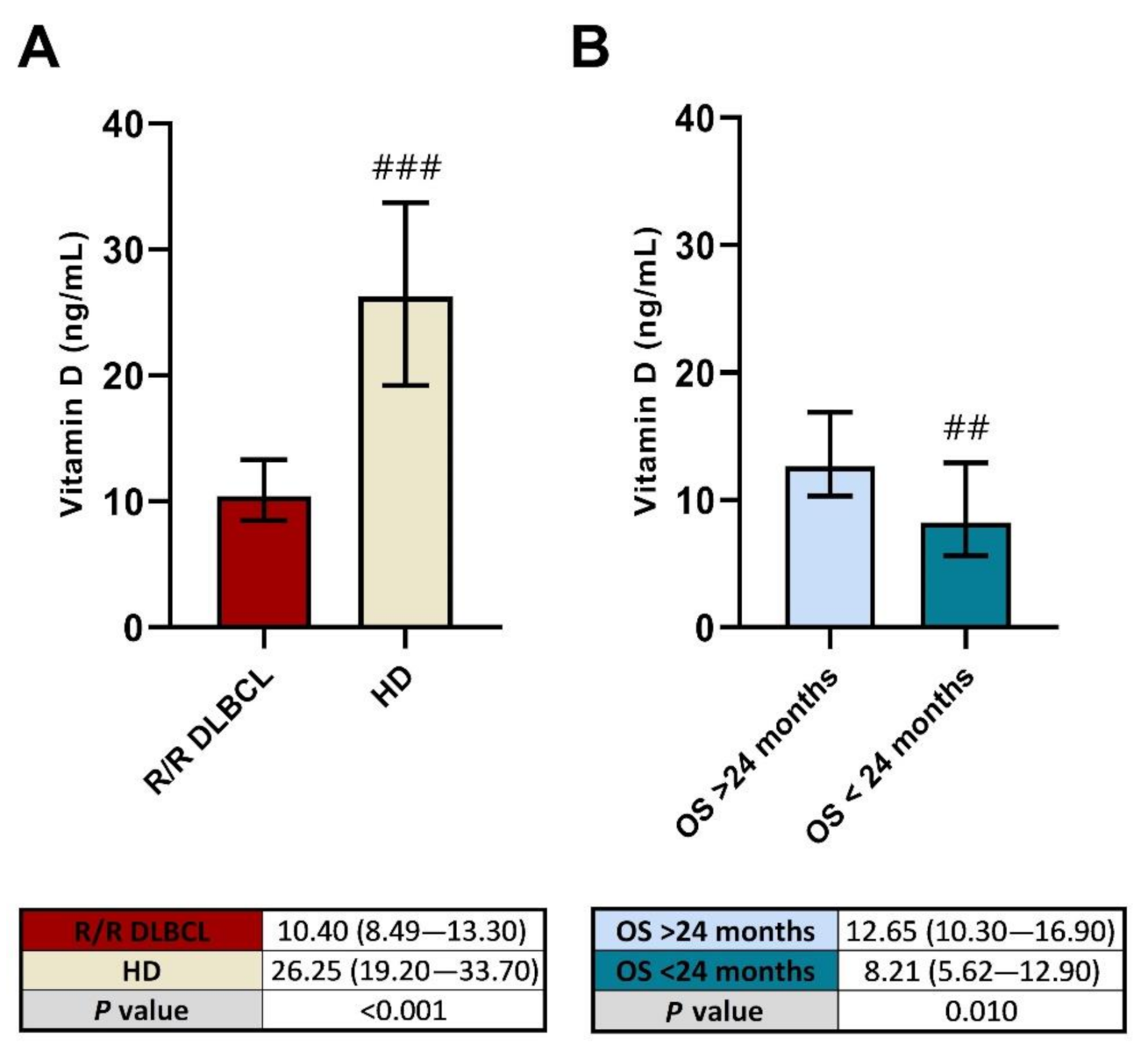
2.2. Low Vitamin D Levels before R2-GDP May Predict a Poorer OS in R/R DLBCL
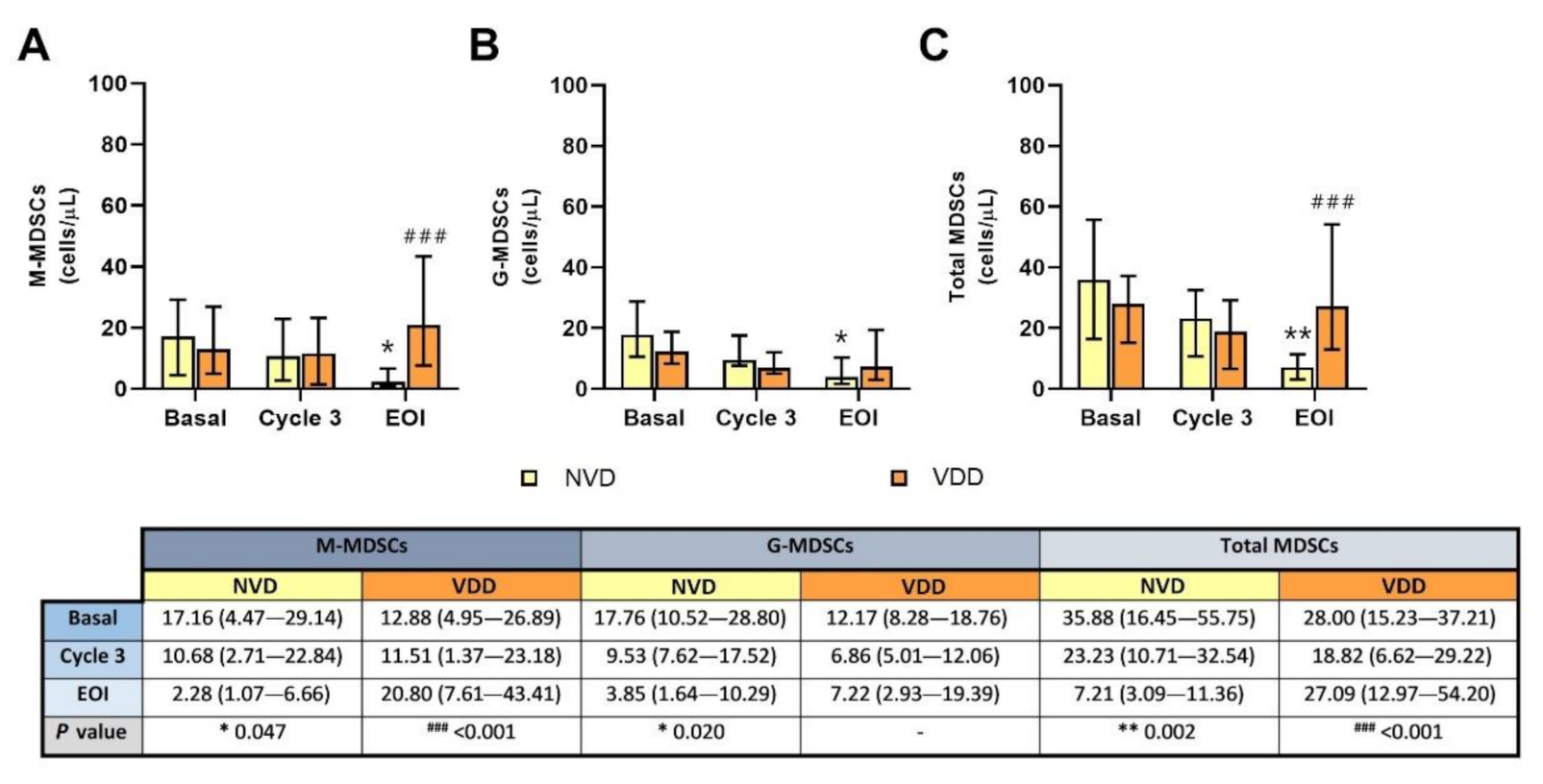
2.3. Circulating MDSCs Decreased in R/R DLBCL Patients with Better Vitamin D Levels after R2-GDP
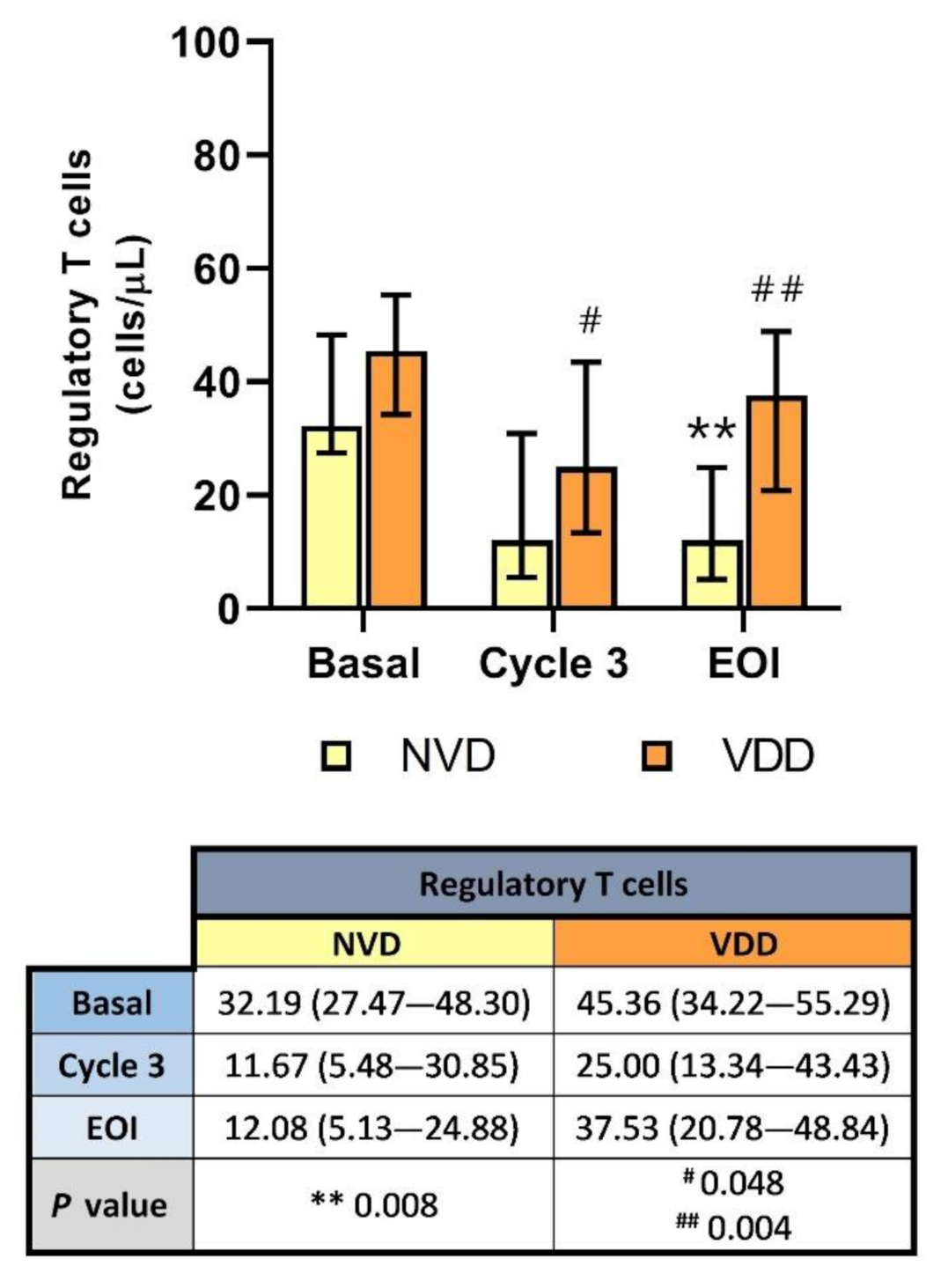
2.4. Peripheral Blood Treg Levels Were Significantly Reduced with R2-GDP in NVD Patients
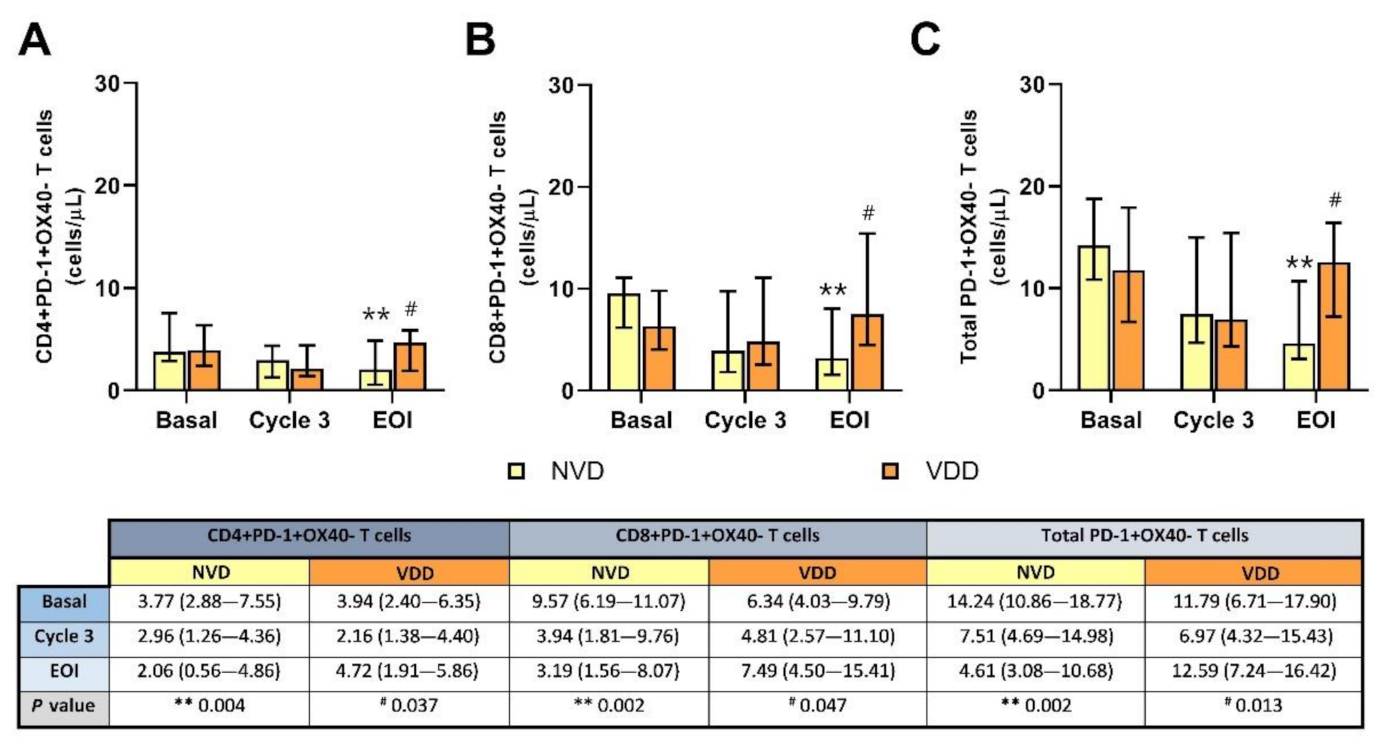
2.5. Inhibited PD-1+OX40− T Cells Were Notably Reduced after Treatment in Patients with Better Vitamin D Concentration
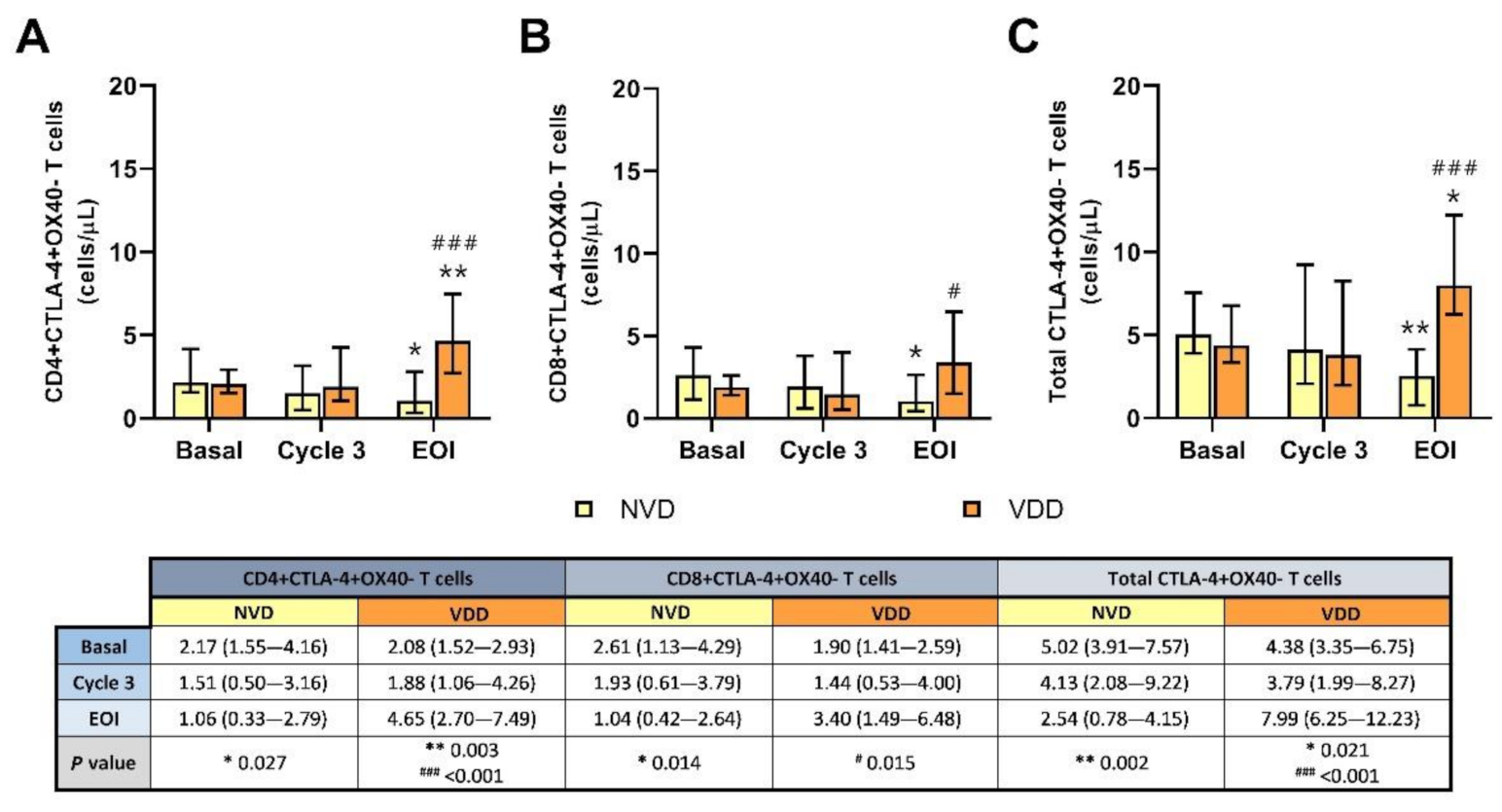
2.6. Exhausted CTLA-4+OX40- T Cells Were Significantly Depleted in NVD Patients Using R2-GDP Schedule
3. Discussion
4. Materials and Methods
4.1. Patients
4.2. Flow Cytometry Analysis in Whole Blood Samples
4.3. Monoclonal Antibodies
4.4. Vitamin D Analysis in Serum
4.5. Data Analysis
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Li, S.; Young, K.H.; Medeiros, L. Diffuse large B-cell lymphoma. Pathology 2018, 50, 74–87. [Google Scholar] [CrossRef]
- Gisselbrecht, C.; Van den Neste, E. How I manage patients with relapsed/refractory diffuse large B cell lymphoma. Br. J. Haematol. 2018, 182, 633–643. [Google Scholar] [CrossRef] [PubMed]
- Solimando, A.G.; Annese, T.; Tamma, R.; Ingravallo, G.; Maiorano, E.; Vacca, A.; Specchia, G.; Ribatti, D. New insights into diffusse large B-cell lymphoma pathobiology. Cancers 2020, 12, 1869. [Google Scholar] [CrossRef]
- Borel, P.; Caillaud, D.; Cano, N.J. Vitamin D Bioavailability: State of the Art. Crit. Rev. Food. Sci. 2015, 55, 1193–1205. [Google Scholar] [CrossRef]
- Bikle, D.; Christakos, S. New aspects of vitamin D metabolism and action—Addressing the skin as source and target. Nat. Rev. Endocrinol. 2020, 16, 234–252. [Google Scholar] [CrossRef] [PubMed]
- Dou, R.; Ng, K.; Giovannucci, E.L.; Manson, J.E.; Qian, Z.R.; Ogino, S. Vitamin D and colorectal cancer: Molecular, epidemiological, and clinical evidence. Br. J. Nutr. 2016, 115, 1643–1660. [Google Scholar] [CrossRef]
- Brożyna, A.A.; Hoffman, R.M.; Slominski, A.T. Relevance of Vitamin D in Melanoma Development, Progression and Therapy. Anticancer Res. 2020, 40, 473–489. [Google Scholar] [CrossRef] [PubMed]
- Wang, W.; Hu, W.; Xue, S.; Chen, Q.; Jiang, Y.; Zhang, H.; Zuo, W. Vitamin D and Lung Cancer; Association, Prevention, and Treatment. Nutr. Cancer 2020, 11, 1–13. [Google Scholar] [CrossRef]
- Iqbal, S.; Naseem, J. Pancreatic cancer control: Is vitamin D the answer? Eur. J. Cancer. Prev. 2016, 25, 188–195. [Google Scholar] [CrossRef]
- Welsh, J. Vitamin D and breast cancer: Past and present. J. Steroid. Biochem. Mol. Biol. 2018, 177, 15–20. [Google Scholar] [CrossRef] [PubMed]
- Xu, Y.; Shao, X.; Yao, Y.; Xu, L.; Chang, L.; Jiang, Z.; Lin, Z. Positive association between circulating 25-hydroxivitamin D levels and prostate cancer risk: New findings from an updated meta-analysis. J. Cancer. Res. Clin. Oncol. 2014, 140, 1465–1477. [Google Scholar] [CrossRef] [PubMed]
- Xu, D.M.; Liang, J.H.; Wang, L.; Zhu, H.Y.; Xia, Y.; Fan, L.; Li, J.-Y.; Xu, W. 25-Hydroxy vitamin D deficiency predicts inferior prognosis in mantle cell lymphoma. J. Cancer. Res. Clin. Oncol. 2020, 146, 1003–1009. [Google Scholar] [CrossRef]
- Kelly, J.L.; Salles, G.; Goldman, B.; Fisher, R.I.; Brice, P.; Press, O.; Casasnovas, O.; Maloney, D.G.; Soubeyran, P.; Rimsza, L.; et al. Low serum vitamin D levels are associated with inferior survival in Follicular Lymphoma: A prospective evaluation in SWOG and LYSA Studies. J. Clin. Oncol. 2015, 33, 1482–1490. [Google Scholar] [CrossRef]
- Graklanov, V.; Popov, V. Vitamin D levels in patients with non-Hodgkin lymphoma/diffuse large B-cell lymphoma, chronic lymphocytic leukemia and multiple myeloma. J. Int. Med. Res. 2020, 48, 300060520943421. [Google Scholar] [CrossRef] [PubMed]
- Chen, P.; Cao, Y.; Duan, X.; Li, J.; Zhao, W.; Wang, H. Bioavailable 25(OH)D level is associated with clinical outcomes of patients with diffuse large B-cell lymphoma: An exploratory study. Clin. Nutr. 2021, 40, 157–165. [Google Scholar] [CrossRef]
- Salles, G.; Barret, M.; Foà, R.; Maurer, J.; O’Brien, S.; Valente, N.; Wenger, M.; Maloney, D.G. Rituximab in B-cell hematologic malignancies: A review of 20 years of clinical experience. Adv. Ther. 2017, 34, 2232–2273. [Google Scholar] [CrossRef] [PubMed]
- Witzig, T.E.; Nowakowski, G.S.; Habermann, T.M.; Goy, A.; Hernandez-Ilizaliturri, F.J.; Chiapella, A.; Vitolo, U.; Fowler, N.; Czuczman, M.S. A comprehensive review of lenalidomide therapy for B-cell non-Hodgkin lymphoma. Ann. Oncol. 2015, 26, 1667–1677. [Google Scholar] [CrossRef]
- Hohaus, S.; Tisi, M.C.; Bellesi, S.; Maiolo, E.; Alma, E.; Tartaglia, G.; Corrente, F.; Cuccaro, A.; D’Alo, F.; Basile, U.; et al. Vitamin D deficiency and supplementation in patients with aggressive B-cell lymphomas treated with immunotherapy. Cancer. Med. 2018, 7, 270–281. [Google Scholar] [CrossRef]
- Brosseau, C.; Dousset, C.; Touzeau, C.; Maïga, S.; Moreau, P.; Amiot, M.; Le Gouill, S.; Pellat-Deceunynck, C. Combination of lenalidomide with vitamin D3 induces apoptosis in mantle cell lymphoma via demethylation of BIK. Cell. Death. Dis. 2014, 5, e1389. [Google Scholar] [CrossRef][Green Version]
- Neumann, F.; Acker, F.; Schormann, C.; Pfreundschuh, M.; Bittenbring, J.T. Determination of optimum vitamin D3 levels for NK cell-mediated rituximab- and obinutuzumab-dependent cellular cytotoxicity. Cancer Immunol. Immunother. 2018, 67, 1709–1718. [Google Scholar] [CrossRef]
- Flamann, C.; Busch, L.; Mackensen, A.; Bruns, H. Combination of lenalidomide and vitamin D enhances MOR202-mediated cytotoxicity of macrophages: It takes three to tango. Oncotarget 2019, 10, 10–12. [Google Scholar] [CrossRef]
- Tomuleasa, C.; Iluta, S.; Pasca, S.; Roman, A.; Piciu, D.; Jitaru, C.; Teodorescu, P.; Rus, I.; Bojan, A.; Dima, D.; et al. Complete metabolic remission in an 84-year old patient with relapsed/refractory diffuse large B-cell lymphoma following combination immunotherapy with lenalidomide plus rituximab. Int. J. Hematol. 2020, 112, 597–598. [Google Scholar] [CrossRef]
- Zhang, Y.; Wang, X.; Liu, Y.; Sun, C.; Shi, W.; Huang, H. Lenalidomide combined with R-GDP in a patient with refractory CD5-positive diffuse large B-cell lymphoma: A promising response and review. Cancer Biol. Ther 2018, 19, 549–553. [Google Scholar] [CrossRef] [PubMed]
- De la Cruz-Merino, L.; Martín García-Sancho, A.; Nogales-Fernández, E.; Carnicero-González, F.; Ríos-Herranz, E.; De la Cruz-Vicente, F.; Rodríguez, G.; Nicolás, C.; Martínez-Banaclocha, N.; Guma, J.; et al. Lenalidomide plus R-GDP (R2-GDP) in relapsed/refractory diffuse large B-cell lymphoma. Preliminary results of the R2-GDP-GOTEL trial. Hematol. Oncol. 2019, 37 (Suppl. S2), 258–259. [Google Scholar] [CrossRef]
- De la Cruz-Merino, L.; Martín García-Sancho, A.; Nogales-Fernández, E.; Carnicero-González, F.; Ríos-Herranz, E.; De la Cruz-Vicente, F.; Rodríguez, G.; Nicolás, C.; Martínez-Banaclocha, N.; Guma, J.; et al. Lenalidomide plus R-GDP (R2-GDP) in relapsed/refractory diffuse large B-cell lymphoma: Final results of the R2-GDP-GOTEL trial. J. Clin. Oncol. 2020, 38 (Suppl. S15), 8019. [Google Scholar] [CrossRef]
- Jiménez-Cortegana, C.; Palazón Carrión, N.; Martin García-Sancho, A.; Nogales-Fernández, E.; Carnicero-Gonzalez, F.; Ríos-Herranz, E.; De la Cruz-Vicente, F.; Rodríguez-García, G.; Fernández-Álvarez, R.; Rueda-Domínguez, A.; et al. Circulating myeloid-derived suppressor cells and regulatory T cells as immunological biomarkers in refractory/relapsed diffuse large B-cell lymphoma: Translational results from the R2-GDP-GOTEL trial. J. Immunother. Cancer 2021, 9, e002323. [Google Scholar] [CrossRef]
- Aranow, C. Vitamin D and the immune system. J. Investig. Med. 2011, 59, 881–886. [Google Scholar] [CrossRef] [PubMed]
- Beake, F.; Takiishi, T.; Korf, H.; Gysemans, C.; Mathieu, C. Vitamin D: Modulator of the immune system. Curr. Opin. Pharmacol. 2010, 10, 482–496. [Google Scholar] [CrossRef] [PubMed]
- Fleet, J.C.; Burcham, G.N.; Calvert, R.D.; Elzey, B.D.; Ratliff, T.L. 1a, 25 Dihydroxyvitamin D (1,25(OH)2 D) inhibits the T cell suppressive functions of myeloid derived suppressor cells (MDSC). J. Steroid. Biochem. Mol. Biol. 2020, 198, 105557. [Google Scholar] [CrossRef] [PubMed]
- Toor, S.M.; Syed-Khaja, A.S.; El Salhat, H.; Faour, I.; Kanbar, J.; Quadri, A.A.; Albashir, M.; Elkord, E. Myeloid cells in circulation and tumor microenvironment of breast cancer patients. Cancer. Immunol. Immunother. 2017, 66, 753–764. [Google Scholar] [CrossRef] [PubMed]
- Galanina, N.; Kline, J.; Bishop, M.R. Emerging role of checkpoint blockade therapy in lymphoma. Ther. Adv. Hematol. 2017, 8, 81–90. [Google Scholar] [CrossRef] [PubMed]
- Jeffery, L.E.; Wood, A.M.; Qureshi, O.S.; Hou, T.Z.; Gardner, D.; Briggs, Z.; Kaur, S.; Raza, K.; Sansom, D.M. Availability of 25-hydroxyvitamin D(3) to APCs controls the balance between regulatory and inflammatory T cell responses. J. Immunol. 2012, 189, 5155–5164. [Google Scholar] [CrossRef]
- Unger, W.W.J.; Laban, S.; Kleijwegt, F.S.; van der Slik, A.R.; Roep, B.O. Induction of Treg by monocyte-derived DC modulated by vitamin D3 or dexamethasone: Differential role for PD-L1. Eur. J. Immunol. 2009, 39, 3147–3159. [Google Scholar] [CrossRef] [PubMed]
- Bendix, M.; Greisen, S.; Dige, A.; Hvas, C.L.; Bak, N.; Jorgensen, S.P.; Dahlerup, J.F.; Deleuran, B.; Agnholt, J. Vitamin D increases programmed death receptor-1 expression in Crohn’s disease. Oncotarget 2017, 8, 24177–24186. [Google Scholar] [CrossRef]
- Pincikova, T.; Paquin-Proulx, D.; Sandberg, J.K.; Flodstrom-Tullberg, M.; Hjelte, L. Vitamin D treatment modulates immune activation in cystic fibrosis. Clin. Exp. Immunol. 2017, 189, 359–371. [Google Scholar] [CrossRef] [PubMed]
- Apperly, F.L. The relationship of solar radiation to cancer mortality in North America. Cancer Res. 1941, 1, 191–195. [Google Scholar]
- Garland, C.F.; Garland, F.C. Do sunlight and vitamin D reduce the likelihood of colon cancer? Int. J. Epidemiol. 1980, 9, 227–231. [Google Scholar] [CrossRef]
- Grant, W.B.; Mohr, S.B. Ecological studies of ultraviolet B, vitamin D and cancer since 2000. Ann. Epidemiol. 2009, 19, 446–454. [Google Scholar] [CrossRef] [PubMed]
- Mondul, A.M.; Weinstein, S.J.; Layne, T.M.; Albanes, D. Vitamin D and cancer risk and mortality: State of the science, gaps, and challenges. Epidemiol. Rev. 2017, 39, 28–48. [Google Scholar] [CrossRef]
- Giammanco, M.; Di Majo, D.; La Guardia, M.; Aiello, S.; Crescimannno, M.; Flandina, C.; Tumminello, F.M.; Leto, G. Vitamin D in cancer chemoprevention. Pharm. Biol. 2015, 53, 1399–1434. [Google Scholar] [CrossRef]
- Ma, Y.; Trump, D.L.; Johnson, C.S. Vitamin D in combination cancer treatment. J. Cancer 2010, 1, 101–107. [Google Scholar] [CrossRef]
- Jeon, S.-M.; Shin, E.-A. Exploring vitamin D metabolism and function in cancer. Exp. Mol. Med. 2018, 50, 20. [Google Scholar] [CrossRef] [PubMed]
- Bittengring, J.T.; Neumann, F.; Altmann, B.; Achenbach, J.; Reichrath, J.; Ziepert, M.; Geisel, J.; Regitz, E.; Held, G.; Pfreundschuh, M. Vitamin D deficiency impairs rituximab-mediated cellular cytotoxicity and outcome of patients with diffuse large B-cell lymphoma treated with but not without rituximab. J. Clin. Oncol. 2014, 32, 3242–3248. [Google Scholar] [CrossRef] [PubMed]
- Wang, W.-T.; Liang, J.-H.; Wang, L.; Zhu, H.-Y.; Xia, Y.; Fan, L.; Li, J.-Y.; Xu, W. The prognostic value of 25-hydroxy vitamin D deficiency and its interaction with c-Myc expression in diffuse large B cell lymphoma. Ann. Hematol. 2020, 99, 2377–2384. [Google Scholar] [CrossRef] [PubMed]
- Prietl, B.; Treiber, G.; Pieber, T.R.; Amrein, K. Vitamin D and immune function. Nutrients 2013, 5, 2502–2521. [Google Scholar] [CrossRef]
- Di Rosa, M.; Malaquarnera, M.; Nicoletti, F.; Malaguarnera, L. Vitamin D3: A helpful immuno-modulator. Immunology 2011, 134, 123–139. [Google Scholar] [CrossRef] [PubMed]
- Gabrilovich, D.I. Myeloid-Derived Suppressor Cells. Cancer. Immunol. Res. 2017, 5, 3–8. [Google Scholar] [CrossRef] [PubMed]
- Wolf, D.; Sopper, S.; Pircher, A.; Gastl, G.; Wolf, A.M. Treg(s) in Cancer: Friends or Foe? J. Cell. Physiol. 2015, 230, 2598–2605. [Google Scholar] [CrossRef]
- Dermani, F.K.; Samadi, P.; Rahmani, G.; Kohlan, A.K.; Najafi, R. PD-1/PD-L1 immune checkpoint: Potential target for cancer therapy. J. Cell. Physiol. 2019, 234, 1313–1325. [Google Scholar] [CrossRef]
- Rowshanravan, B.; Halliday, N.; Sansom, D.M. CTLA-4: A moving target in immunotherapy. Blood 2018, 131, 58–67. [Google Scholar] [CrossRef]
- Görgün, G.T.; Samur, M.K.; Cowens, K.B.; Paula, S.; Bianchi, G.; Anderson, J.E.; White, R.E.; Singh, A.; Ohguchi, H.; Suzuki, R.; et al. Lenalidomide enhances immune checkpoint blockade induced immune response in multiple myeloma. Clin. Cancer. Res. 2015, 21, 4607–4618. [Google Scholar] [CrossRef] [PubMed]
- Lv, M.; Wang, K.; Huang, X.-J. Myeloid-derived suppressor cells in hematological malignancies: Friends or foes. J. Hematol. Oncol. 2019, 12, 105. [Google Scholar] [CrossRef] [PubMed]
- Ma, Y.-H.; Zhou, Y.-I.; Yue, C.-Y.; Zhang, G.-H.; Deng, L.; Xie, G.-H.; Xu, W.-P.; Shen, L.-S. Vitamin D deficiency contributes to the reduction and impaired function of CD45RA+ naïve regulatory T cell in chronic heart failure. J. Immunol. Res. 2015, 2015, 547697. [Google Scholar] [CrossRef]
- Gorman, S.; Geldenhuys, S.; Judge, M.; Weeden, C.E.; Waithman, J.; Hart, P.H. Dietary vitamin D increases percentages and function of regulatory T cells in the skin-draining lymph nodes and suppresses dermal inflammation. J. Immunol. Res. 2016, 2016, 1426503. [Google Scholar] [CrossRef] [PubMed]
- Ghoryani, M.; Sahebari, M.; Mahmoudi, M.; Abdollahi, N.; Reihani, H.; Taghizadeh-Rabe, S.Z.; Tabasi, N. Immunomodulatory vitamin D effects on regulatory T-cells and cytokines in an in vitro study on patients with systemic lupus erythematosus. Food. Agric. Immunol. 2016, 27, 377–387. [Google Scholar] [CrossRef][Green Version]
- Fisher, S.A.; Rahimzadeh, M.; Brierley, C.; Gration, B.; Doree, C.; Kimber, C.E.; Plaza-Cajide, A.; Lamikanra, A.A.; Roberts, D.J. The role of vitamin D in increasing circulating T regulatory cell numbers and modulating T regulatory cell phenotypes in patients with inflammatory disease or in healthy volunteers: A systematic review. PLoS ONE 2019, 14, e0222313. [Google Scholar] [CrossRef] [PubMed]
- Khoo, A.-L.; Joosten, I.; Michels, M.; Woestenenk, R.; Preijers, F.; He, X.-H.; Netea, M.G.; van der Ven, A.J.A.M.; Koenen, H.J.P.M. 1,25-dihydroxyvitamin D3 inhibits proliferation but not the suppressive function of regulatory T cells in the absence of antigen-presenting cells. Immunology 2011, 134, 459–468. [Google Scholar] [CrossRef] [PubMed]
- Ohue, Y.; Nishikawa, H. Regulatory T (Treg) cells in cancer: Can Treg cells be a new therapeutic target? Cancer Sci. 2019, 110, 2080–2089. [Google Scholar] [CrossRef]
- Karkeni, E.; Morin, S.O.; Tayeh, B.B.; Goubard, A.; Josselin, E.; Castellano, R.; Fauriat, C.; Guittard, G.; Olive, D.; Nunès, J.A. Vitamin D controls tumor growth and CD8+ T cell infiltration in breast cancer. Front. Immunol. 2019, 10, 1307. [Google Scholar] [CrossRef]
- Dimitrov, V.; Bouttier, M.; Boukhaled, G.; Salehi-Tabar, R.; Avramescu, R.G.; Memari, B.; Hasaj, B.; Lukacs, G.L.; Krawczyk, C.M.; White, J.H. Hormonal vitamin D up-regulates tissue-specific PD-L1 and PD-L2 surface glycoprotein expression in humans but not mice. J. Biol. Chem. 2017, 292, 20657–20668. [Google Scholar] [CrossRef]
- Jeffery, L.E.; Burke, F.; Mura, M.; Zheng, Y.; Qureshi, O.S.; Hewison, M.; Walker, L.S.K.; Lammas, D.A.; Raza, K.; Sansom, D.M. 1,25-Dihidroxyvitamin D3 and IL-2 combine to inhibit T cell production of inflammatory cytokines and promote development of regulatory T cells expressing CTLA-4 and FOXP3. J. Immunol. 2009, 183, 5458–5467. [Google Scholar] [CrossRef] [PubMed]
- Jeffery, L.E.; Qureshi, O.S.; Gardner, D.; Hou, T.Z.; Briggs, Z.; Soskic, B.; Baker, J.; Raza, K.; Sansom, D.M. Vitamin D antagonises the suppressive effect of inflammatory cytokines on CTLA-4 expression and regulatory function. PLoS ONE 2015, 10, e0131539. [Google Scholar] [CrossRef]
- Donati, B.; Ferrari, A.; Ruffini, A.; Manzotti, G.; Fragliasso, V.; Merli, F.; Zanelli, M.; Valli, R.; Luminari, S.; Ciarrochi, A. Gene expression profile unveils diverse biological effect of serum vitamin D in Hodgkin’s and diffuse large B-cell lymphoma. Hematol. Oncol. 2021, 39, 205–214. [Google Scholar] [CrossRef] [PubMed]
- Martens, P.-J.; Gysemans, C.; Verstuyf, A.; Mathieu, C. Vitamin D’s effect on immune function. Nutrients 2020, 12, 1248. [Google Scholar] [CrossRef] [PubMed]
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Jiménez-Cortegana, C.; Sánchez-Martínez, P.M.; Palazón-Carrión, N.; Nogales-Fernández, E.; Henao-Carrasco, F.; Martín García-Sancho, A.; Rueda, A.; Provencio, M.; de la Cruz-Merino, L.; Sánchez-Margalet, V. Lower Survival and Increased Circulating Suppressor Cells in Patients with Relapsed/Refractory Diffuse Large B-Cell Lymphoma with Deficit of Vitamin D Levels Using R-GDP Plus Lenalidomide (R2-GDP): Results from the R2-GDP-GOTEL Trial. Cancers 2021, 13, 4622. https://doi.org/10.3390/cancers13184622
Jiménez-Cortegana C, Sánchez-Martínez PM, Palazón-Carrión N, Nogales-Fernández E, Henao-Carrasco F, Martín García-Sancho A, Rueda A, Provencio M, de la Cruz-Merino L, Sánchez-Margalet V. Lower Survival and Increased Circulating Suppressor Cells in Patients with Relapsed/Refractory Diffuse Large B-Cell Lymphoma with Deficit of Vitamin D Levels Using R-GDP Plus Lenalidomide (R2-GDP): Results from the R2-GDP-GOTEL Trial. Cancers. 2021; 13(18):4622. https://doi.org/10.3390/cancers13184622
Chicago/Turabian StyleJiménez-Cortegana, Carlos, Pilar M. Sánchez-Martínez, Natalia Palazón-Carrión, Esteban Nogales-Fernández, Fernando Henao-Carrasco, Alejandro Martín García-Sancho, Antonio Rueda, Mariano Provencio, Luis de la Cruz-Merino, and Víctor Sánchez-Margalet. 2021. "Lower Survival and Increased Circulating Suppressor Cells in Patients with Relapsed/Refractory Diffuse Large B-Cell Lymphoma with Deficit of Vitamin D Levels Using R-GDP Plus Lenalidomide (R2-GDP): Results from the R2-GDP-GOTEL Trial" Cancers 13, no. 18: 4622. https://doi.org/10.3390/cancers13184622
APA StyleJiménez-Cortegana, C., Sánchez-Martínez, P. M., Palazón-Carrión, N., Nogales-Fernández, E., Henao-Carrasco, F., Martín García-Sancho, A., Rueda, A., Provencio, M., de la Cruz-Merino, L., & Sánchez-Margalet, V. (2021). Lower Survival and Increased Circulating Suppressor Cells in Patients with Relapsed/Refractory Diffuse Large B-Cell Lymphoma with Deficit of Vitamin D Levels Using R-GDP Plus Lenalidomide (R2-GDP): Results from the R2-GDP-GOTEL Trial. Cancers, 13(18), 4622. https://doi.org/10.3390/cancers13184622






